Recent Advances in Supercritical CO2 Extraction of Pigments, Lipids and Bioactive Compounds from Microalgae
Abstract
1. Introduction
2. Microalgal Products
3. Pretreatment Methods
3.1. Classification of Methods
- Mechanical pretreatment
- Non mechanical pretreatment
- Chemical
- Enzymatic [138]
3.1.1. Mechanical
3.1.2. Chemical
- Acids
- Solvents (organic, ionic liquids, etc.)
- Salts (e.g., osmotic shock with NaCl)
- Nanoparticles
- Surfactants [138]
3.1.3. Enzymatic
3.2. Pretreatment of Microalgae
3.2.1. Arthrospira
3.2.2. Chlorella
3.2.3. Haematococcus
3.2.4. Nannochloropsis
3.2.5. Scenedesmus
3.2.6. Other Cultures
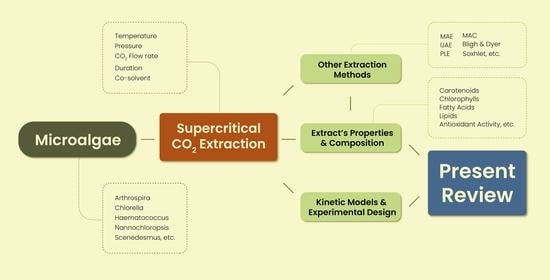
4. Supercritical CO2 Extraction
4.1. Principles and Process
4.2. Extraction of Bioactive Compounds
4.2.1. Arthrospira
4.2.2. Chlorella
4.2.3. Haematococcus
4.2.4. Nannochloropsis
4.2.5. Scenedesmus
4.2.6. Other Cultures
4.3. Extraction of Lipids and Fatty Acids
4.3.1. Arthrospira
4.3.2. Chlorella
4.3.3. Nannochloropsis
4.3.4. Scenedesmus
4.3.5. Other Cultures
4.4. Kinetic Models
4.4.1. Broken Plus Intact Cell Model
4.4.2. Other Models
5. Other Extraction Methods
5.1. Maceration
5.2. Soxhlet
5.3. Bligh and Dyer and Folch
5.4. Ultrasound Assisted Extraction
5.5. Microwave Assisted Extraction
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AA | Arachidic acid |
| ACE | Acetone |
| AM | Antimicrobial activity |
| AO | Antioxidant activity (IC50) |
| ASE | Accelerated Solvent Extractor |
| AST | Astaxanthin |
| B-D | Bligh and Dyer |
| BICM | Broken Plus Intact Cell Model |
| CAN | Canthaxanthin |
| CCD | Central Composite Design |
| CCRD | Central Composite Rotatable Design |
| CHF | Chloroform |
| CHL-a | Chlorophyll a |
| CHL-b | Chlorophyll b |
| CHL-c | Chlorophyll c |
| Comp. | Composition |
| Co-Solv | Co-solvent |
| CRY | Cryptoxanthin |
| DCM | Dichloromethane |
| DHA | Docosahexaenoic Acid |
| DMF | Dimethylformamide |
| DMC | Dimethyl Carbonate |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| EPA | Eicosapentaenoic Acid |
| EtA | Ethyl Acetate |
| EtOH | Ethanol |
| Extr | Extract |
| FA | Fatty Acids |
| FAME | Fatty Acid Methyl Esters |
| GAE | Gallic Acid Equivalent |
| GXL | Gas Expanded Liquid |
| GLA | γ-Linolenic Acid |
| GR | Ground |
| IC50/EC50 | 50% Inhibition |
| LA | Linolic Acid |
| LAA | Lauric acid |
| LDF | Linear Driving Force Model |
| LNA | Linolenic Acid |
| LUT | Lutein |
| LY | Lyophilized |
| MA | Myristic acid |
| MAC | Maceration |
| MAE | Microwave Assisted Extraction |
| MBC | Minimum Bactericidal Concentration |
| MeOH | Methanol |
| MFC | Minimal Fungicidal Concentration |
| MUFA | Monounsaturated Fatty Acids |
| MW | Microwave |
| MYX | Myxoxanthophyll |
| NEO | Neoxanthin |
| OA | Oleic Acid |
| P | Pressure |
| PA | Palmitic Acid |
| PHY | Phycocyanine |
| PLA | Palmitoleic Acid |
| PLE | Pressurized Liquid Extraction |
| PUFA | Polyunsaturated Fatty Acids |
| Rec | Recovery |
| RSM | Response Surface Methodology |
| SEP | Separator |
| SFA | Saturated Fatty Acids |
| SFE | Supercritical Fluid Extraction |
| STA | Stearic Acid |
| STP | Standard Temperature and Pressure |
| SX | Soxhlet |
| t | Time/Duration |
| T | Temperature |
| T.CAR | Total Carotenoids |
| T.CHL | Total Chlorophyll |
| TE | Trolox Equivalent |
| TFA | Total Fatty Acids |
| TOC | Tocopherol |
| TPC | Total Phenolic Content |
| UAE | Ultrasound Assisted Extraction |
| VAU | Vaucheriaxanthin |
| VIO | Violaxanthin |
| Y | Yield |
| ZEA | Zeaxanthin |
| β-CAR | β-Carotene |
References
- Picot-Allain, C.; Mahomoodally, M.F.; Ak, G.; Zengin, G. Conventional versus green extraction techniques—A comparative perspective. Curr. Opin. Food Sci. 2021, 40, 144–156. [Google Scholar] [CrossRef]
- Lorenzen, J.; Igl, N.; Tippelt, M.; Stege, A.; Qoura, F.; Sohling, U.; Bruck, T. Extraction of microalgae derived lipids with supercritical carbon dioxide in an industrial relevant pilot plant. Bioprocess. Biosyst. Eng. 2017, 40, 911–918. [Google Scholar] [CrossRef]
- Silva, S.C.; Ferreira, I.C.F.R.; Dias, M.M.; Barreiro, M.F. Microalgae-Derived Pigments: A 10-Year Bibliometric Review and Industry and Market Trend Analysis. Molecules 2020, 25, 3406. [Google Scholar] [CrossRef]
- Daneshvar, E.; Sik Ok, Y.; Tavakoli, S.; Sarkar, B.; Shaheen, S.M.; Hong, H.; Luo, Y.; Rinklebe, J.; Song, H.; Bhatnagar, A. Insights into upstream processing of microalgae: A review. Bioresour. Technol. 2021, 329, 124870. [Google Scholar] [CrossRef]
- Vale, M.A.; Ferreira, A.; Pires, J.C.M.; Gonçalves, A.L. Chapter 17—CO2 capture using microalgae. In Advances in Carbon Capture; Rahimpour, M.R., Farsi, M., Makarem, M.A., Eds.; Woodhead Publishing: Sawston, UK, 2020; pp. 381–405. [Google Scholar]
- Borowitzka, M.A. High-value products from microalgae—Their development and commercialisation. J. Appl. Phycol. 2013, 25, 743–756. [Google Scholar] [CrossRef]
- Borowiak, D.; Krzywonos, M. Bioenergy, Biofuels, Lipids and Pigments—Research Trends in the Use of Microalgae Grown in Photobioreactors. Energies 2022, 15, 5357. [Google Scholar] [CrossRef]
- Gong, M.; Bassi, A. Carotenoids from microalgae: A review of recent developments. Biotechnol. Adv. 2016, 34, 1396–1412. [Google Scholar] [CrossRef]
- Yen, H.-W.; Yang, S.-C.; Chen, C.-H.; Jesisca; Chang, J.-S. Supercritical fluid extraction of valuable compounds from microalgal biomass. Bioresour. Technol. 2015, 184, 291–296. [Google Scholar] [CrossRef]
- Chemat, F.; Vian, M.A.; Cravotto, G. Green Extraction of Natural Products: Concept and Principles. Int. J. Mol. Sci. 2012, 13, 8615–8627. [Google Scholar] [CrossRef]
- Mandal, S.C.; Mandal, V.; Das, A.K. Chapter 6—Classification of Extraction Methods. In Essentials of Botanical Extraction; Mandal, S.C., Mandal, V., Das, A.K., Eds.; Academic Press: Boston, MA, USA, 2015; pp. 83–136. [Google Scholar]
- Leonelli, C.; Veronesi, P.; Cravotto, G. Microwave-Assisted Extraction: An Introduction to Dielectric Heating. In Microwave-assisted Extraction for Bioactive Compounds: Theory and Practice; Chemat, F., Cravotto, G., Eds.; Springer US: Boston, MA, USA, 2013; pp. 1–14. [Google Scholar]
- Al-Nimer, M.; Wahbee, Z. Ultraviolet light assisted extraction of flavonoids and allantoin from aqueous and alcoholic extracts of Symphytum officinale. J. Intercult. Ethnopharmacol. 2017, 6, 280–283. [Google Scholar] [CrossRef]
- Hitchen, S.M.; Dean, J.R. Properties of Supercritical Fluids. In Applications of Supercritical Fluids in Industrial Analysis; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1993; pp. 1–11. [Google Scholar]
- Ghasemi, E.; Raofie, F.; Najafi, N.M. Application of response surface methodology and central composite design for the optimisation of supercritical fluid extraction of essential oils from Myrtus communis L. leaves. Food Chem. 2011, 126, 1449–1453. [Google Scholar] [CrossRef]
- Uwineza, P.A.; Waśkiewicz, A. Recent Advances in Supercritical Fluid Extraction of Natural Bioactive Compounds from Natural Plant Materials. Molecules 2020, 25, 3847. [Google Scholar] [CrossRef]
- Cardoso, L.C.; Serrano, C.M.; Rodríguez, M.R.; de la Ossa, E.J.M.; Lubián, L.M. Extraction of Carotenoids and Fatty Acids from Microalgae using Supercritical Technology. Am. J. Anal. Chem. 2012, 03, 877–883. [Google Scholar] [CrossRef]
- Cassanelli, M.; Prosapio, V.; Norton, I.; Mills, T. Design of a Cost-Reduced Flexible Plant for Supercritical Fluid-Assisted Applications. Chem. Eng. Technol. 2018, 41, 1368–1377. [Google Scholar] [CrossRef]
- Tabernero, A.; Martín del Valle, E.M.; Galan, M.A. Microalgae Technology: A Patent Survey. Int. J. Chem. React. Eng. 2013, 11, 733–763. [Google Scholar] [CrossRef]
- WIPO. W.I.P.O. Patent Landscape Report on Microalgae-Related Technologies; WIPO: Geneva, Switzerland, 2016. [Google Scholar]
- Ambati, R.R.; Gogisetty, D.; Aswathanarayana, R.G.; Ravi, S.; Bikkina, P.N.; Bo, L.; Yuepeng, S. Industrial potential of carotenoid pigments from microalgae: Current trends and future prospects. Crit. Rev. Food Sci. Nutr. 2019, 59, 1880–1902. [Google Scholar] [CrossRef]
- Maltsev, Y.; Maltseva, K. Fatty acids of microalgae: Diversity and applications. Rev. Environ. Sci. Bio/Technol. 2021, 20, 515–547. [Google Scholar] [CrossRef]
- Lang, I.; Hodac, L.; Friedl, T.; Feussner, I. Fatty acid profiles and their distribution patterns in microalgae: A comprehensive analysis of more than 2000 strains from the SAG culture collection. BMC Plant Biol. 2011, 11, 124. [Google Scholar] [CrossRef]
- Mendes, R.L.; Nobre, B.P.; Cardoso, M.T.; Pereira, A.P.; Palavra, A.F. Supercritical carbon dioxide extraction of compounds with pharmaceutical importance from microalgae. Inorg. Chim. Acta 2003, 356, 328–334. [Google Scholar] [CrossRef]
- Mendes, R.L.; Reis, A.D.; Pereira, A.P.; Cardoso, M.T.; Palavra, A.F.; Coelho, J.P. Supercritical CO2 extraction of γ-linolenic acid (GLA) from the cyanobacterium Arthrospira (Spirulina) maxima: Experiments and modeling. Chem. Eng. J. 2005, 105, 147–151. [Google Scholar] [CrossRef]
- Mendes, R.L.; Reis, A.D.; Palavra, A.F. Supercritical CO2 extraction of γ-linolenic acid and other lipids from Arthrospira (Spirulina) maxima: Comparison with organic solvent extraction. Food Chem. 2006, 99, 57–63. [Google Scholar] [CrossRef]
- Canela, A.P.R.F.; Rosa, P.T.V.; Marques, M.O.M.; Meireles, M.A.A. Supercritical Fluid Extraction of Fatty Acids and Carotenoids from the Microalgae Spirulina maxima. Ind. Eng. Chem. Res. 2002, 41, 3012–3018. [Google Scholar] [CrossRef]
- Valderrama, J.O.; Perrut, M.; Majewski, W. Extraction of Astaxantine and Phycocyanine from Microalgae with Supercritical Carbon Dioxide. J. Chem. Eng. Data 2003, 48, 827–830. [Google Scholar] [CrossRef]
- Careri, M.; Furlattini, L.; Mangia, A.; Musci, M.; Anklam, E.; Theobald, A.; von Holst, C. Supercritical fluid extraction for liquid chromatographic determination of carotenoids in Spirulina Pacifica algae: A chemometric approach. J. Chromatogr. A 2001, 912, 61–71. [Google Scholar] [CrossRef]
- Esquivel-Hernandez, D.A.; Lopez, V.H.; Rodriguez-Rodriguez, J.; Aleman-Nava, G.S.; Cuellar-Bermudez, S.P.; Rostro-Alanis, M.; Parra-Saldivar, R. Supercritical Carbon Dioxide and Microwave-Assisted Extraction of Functional Lipophilic Compounds from Arthrospira platensis. Int. J. Mol. Sci. 2016, 17, 658. [Google Scholar] [CrossRef]
- Golmakani, M.-T.; Mendiola, J.A.; Rezaei, K.; Ibáñez, E. Expanded ethanol with CO2 and pressurized ethyl lactate to obtain fractions enriched in γ-Linolenic Acid from Arthrospira platensis (Spirulina). J. Supercrit. Fluids 2012, 62, 109–115. [Google Scholar] [CrossRef]
- Sajilata, M.G.; Singhal, R.S.; Kamat, M.Y. Supercritical CO2 extraction of γ-linolenic acid (GLA) from Spirulina platensis ARM 740 using response surface methodology. J. Food Eng. 2008, 84, 321–326. [Google Scholar] [CrossRef]
- Qiuhui, H. Supercritical Carbon Dioxide Extraction of Spirulina platensis Component and Removing the Stench. J. Agric. Food Chem. 1999, 47, 2705–2706. [Google Scholar] [CrossRef]
- Mendiola, J.A.; Marín, F.R.; Hernández, S.F.; Arredondo, B.O.; Señoráns, F.J.; Ibañez, E.; Reglero, G. Characterization via liquid chromatography coupled to diode array detector and tandem mass spectrometry of supercritical fluid antioxidant extracts of Spirulina platensis microalga. J. Sep. Sci. 2005, 28, 1031–1038. [Google Scholar] [CrossRef]
- Wang, L.; Pan, B.; Sheng, J.; Xu, J.; Hu, Q. Antioxidant activity of Spirulina platensis extracts by supercritical carbon dioxide extraction. Food Chem. 2007, 105, 36–41. [Google Scholar] [CrossRef]
- Mendiola, J.A.; Jaime, L.; Santoyo, S.; Reglero, G.; Cifuentes, A.; Ibañez, E.; Señoráns, F.J. Screening of functional compounds in supercritical fluid extracts from Spirulina platensis. Food Chem. 2007, 102, 1357–1367. [Google Scholar] [CrossRef]
- Tong, Y.; Gao, L.; Xiao, G.; Pan, X. Supercritical CO2 Extraction of Chlorophyll a from Spirulina platensis with a Static Modifier. Chem. Eng. Technol. 2011, 34, 241–248. [Google Scholar] [CrossRef]
- Mouahid, A.; Crampon, C.; Toudji, S.-A.A.; Badens, E. Supercritical CO2 extraction of neutral lipids from microalgae: Experiments and modelling. J. Supercrit. Fluids 2013, 77, 7–16. [Google Scholar] [CrossRef]
- Andrich, G.; Zinnai, A.; Nesti, U.; Venturi, F. Supercritical fluid extraction of oil from microalga Spirulina (arthrospira) platensis. Acta Aliment. 2006, 35, 195–203. [Google Scholar] [CrossRef]
- Palavra, A.M.F.; Coelho, J.P.; Barroso, J.G.; Rauter, A.P.; Fareleira, J.M.N.A.; Mainar, A.; Urieta, J.S.; Nobre, B.P.; Gouveia, L.; Mendes, R.L.; et al. Supercritical carbon dioxide extraction of bioactive compounds from microalgae and volatile oils from aromatic plants. J. Supercrit. Fluids 2011, 60, 21–27. [Google Scholar] [CrossRef]
- Santana, A.; Jesus, S.; Larrayoz, M.A.; Filho, R.M. Supercritical Carbon Dioxide Extraction of Algal Lipids for the Biodiesel Production. Procedia Eng. 2012, 42, 1755–1761. [Google Scholar] [CrossRef]
- Mendiola, J.A.; Torres, C.F.; Toré, A.; Martín-Álvarez, P.J.; Santoyo, S.; Arredondo, B.O.; Señoráns, F.J.; Cifuentes, A.; Ibáñez, E. Use of supercritical CO2 to obtain extracts with antimicrobial activity from Chaetoceros muelleri microalga. A correlation with their lipidic content. Eur. Food Res. Technol. 2006, 224, 505–510. [Google Scholar] [CrossRef]
- Chen, Y.H.; Walker, T.H. Fed-batch fermentation and supercritical fluid extraction of heterotrophic microalgal Chlorella protothecoides lipids. Bioresour. Technol. 2012, 114, 512–517. [Google Scholar] [CrossRef]
- Solana, M.; Rizza, C.S.; Bertucco, A. Exploiting microalgae as a source of essential fatty acids by supercritical fluid extraction of lipids: Comparison between Scenedesmus obliquus, Chlorella protothecoides and Nannochloropsis salina. J. Supercrit. Fluids 2014, 92, 311–318. [Google Scholar] [CrossRef]
- Viguera, M.; Marti, A.; Masca, F.; Prieto, C.; Calvo, L. The process parameters and solid conditions that affect the supercritical CO2 extraction of the lipids produced by microalgae. J. Supercrit. Fluids 2016, 113, 16–22. [Google Scholar] [CrossRef]
- Hu, Q.; Pan, B.; Xu, J.; Sheng, J.; Shi, Y. Effects of supercritical carbon dioxide extraction conditions on yields and antioxidant activity of Chlorella pyrenoidosa extracts. J. Food Eng. 2007, 80, 997–1001. [Google Scholar] [CrossRef]
- Wu, Z.; Wu, S.; Shi, X. Supercritical Fluid Extraction and Determination of Lutein in Heterotrophically Cultivated Chlorella Pyrenoidosa. J. Food Process Eng. 2007, 30, 174–185. [Google Scholar] [CrossRef]
- Alhattab, M.; Kermanshahi-pour, A.; Su-Ling Brooks, M. Dispersed air flotation of Chlorella saccharophila and subsequent extraction of lipids—Effect of supercritical CO2 extraction parameters and surfactant pretreatment. Biomass Bioenergy 2019, 127, 105297. [Google Scholar] [CrossRef]
- Morcelli, A.; Cassel, E.; Vargas, R.; Rech, R.; Marcílio, N. Supercritical fluid (CO2+ethanol) extraction of chlorophylls and carotenoids from Chlorella sorokiniana: COSMO-SAC assisted prediction of properties and experimental approach. J. CO2 Util. 2021, 51, 101649. [Google Scholar] [CrossRef]
- Abrahamsson, V.; Jumaah, F.; Turner, C. Continuous multicomponent quantification during supercritical fluid extraction applied to microalgae using in-line UV/Vis absorption spectroscopy and on-line evaporative light scattering detection. J. Supercrit. Fluids 2018, 131, 157–165. [Google Scholar] [CrossRef]
- Char, J.-M.; Wang, J.-K.; Chow, T.-J.; Chien, Q.-C. Biodiesel Production from Microalgae through Supercritical Carbon Dioxide Extraction. J. Jpn. Inst. Energy 2011, 90, 369–373. [Google Scholar] [CrossRef]
- Safi, C.; Camy, S.; Frances, C.; Varela, M.M.; Badia, E.C.; Pontalier, P.-Y.; Vaca-Garcia, C. Extraction of lipids and pigments of Chlorella vulgaris by supercritical carbon dioxide: Influence of bead milling on extraction performance. J. Appl. Phycol. 2013, 26, 1711–1718. [Google Scholar] [CrossRef]
- Zhou, D.; Qiao, B.; Li, G.; Xue, S.; Yin, J. Continuous production of biodiesel from microalgae by extraction coupling with transesterification under supercritical conditions. Bioresour. Technol. 2017, 238, 609–615. [Google Scholar] [CrossRef]
- Tai, D.C.; Hai, D.T.T.; Vinh, N.H.; Phung, L.T.K. Extraction fatty acid as a source to produce biofuel in microalgae Chlorella sp. and Spirulina sp. using supercritical carbon dioxide. AIP Conf. Proc. 2016, 1737, 060004. [Google Scholar]
- Bahadar, A.; Khan, M.; Asim, M.; Jalwana, K. Supercritical Fluid Extraction of Microalgae (Chlorella vulagaris) Biomass. In Handbook of Marine Microalgae: Biotechnology Advances; Elsevier: Amsterdam, The Netherlands, 2015. [Google Scholar]
- Bahadar, A.; Khan, M.B.; Willmann, J.C. Accelerated production and analysis of biofuel derived from photobioreactor engineered microalgae using super critical fluid extraction. Energy Sources Part A Recovery Util. Environ. Eff. 2016, 38, 1132–1139. [Google Scholar] [CrossRef]
- Dejoye, C.; Vian, M.A.; Lumia, G.; Bouscarle, C.; Charton, F.; Chemat, F. Combined extraction processes of lipid from Chlorella vulgaris microalgae: Microwave prior to supercritical carbon dioxide extraction. Int. J. Mol. Sci. 2011, 12, 9332–9341. [Google Scholar] [CrossRef]
- Gouveia, L.; Nobre, B.P.; Marcelo, F.M.; Mrejen, S.; Cardoso, M.T.; Palavra, A.F.; Mendes, R.L. Functional food oil coloured by pigments extracted from microalgae with supercritical CO2. Food Chem. 2007, 101, 717–723. [Google Scholar] [CrossRef]
- Kitada, K.; Machmudah, S.; Sasaki, M.; Goto, M.; Nakashima, Y.; Kumamoto, S.; Hasegawa, T. Supercritical CO2 extraction of pigment components with pharmaceutical importance from Chlorella vulgaris. J. Chem. Technol. Biotechnol. 2009, 84, 657–661. [Google Scholar] [CrossRef]
- Moradi-kheibari, N.; Ahmadzadeh, H. Supercritical carbon dioxide extraction and analysis of lipids from Chlorella vulgaris using gas chromatography. J. Iran. Chem. Soc. 2017, 14, 2427–2436. [Google Scholar] [CrossRef]
- Mendes, R.L.; Fernandes, H.L.; Coelho, J.; Reis, E.C.; Cabral, J.M.; Novais, J.M.; Palavra, A.F. Supercritical CO2 extraction of carotenoids and other lipids from Chlorella vulgaris. Food Chem. 1995, 53, 99–103. [Google Scholar] [CrossRef]
- Mendes, R.L.; Coelho, J.P.; Fernandes, H.L.; Marrucho, I.J.; Cabral, J.M.S.; Novais, J.M.; Palavra, A.F. Applications of supercritical CO2 extraction to microalgae and plants. J. Chem. Technol. Biotechnol. 1995, 62, 53–59. [Google Scholar] [CrossRef]
- Obeid, S.; Beaufils, N.; Camy, S.; Takache, H.; Ismail, A.; Pontalier, P.-Y. Supercritical carbon dioxide extraction and fractionation of lipids from freeze-dried microalgae Nannochloropsis oculata and Chlorella vulgaris. Algal Res. 2018, 34, 49–56. [Google Scholar] [CrossRef]
- Ruen-ngam, D.; Shotipruk, A.; Pavasant, P.; Machmudah, S.; Goto, M. Selective Extraction of Lutein from Alcohol Treated Chlorella vulgaris by Supercritical CO2. Chem. Eng. Technol. 2012, 35, 255–260. [Google Scholar] [CrossRef]
- Wang, H.-M.; Pan, J.-L.; Chen, C.-Y.; Chiu, C.-C.; Yang, M.-H.; Chang, H.-W.; Chang, J.-S. Identification of anti-lung cancer extract from Chlorella vulgaris C-C by antioxidant property using supercritical carbon dioxide extraction. Process Biochem. 2010, 45, 1865–1872. [Google Scholar] [CrossRef]
- Georgiopoulou, I.; Tzima, S.; Louli, V.; Magoulas, K. Supercritical CO2 Extraction of High-Added Value Compounds from Chlorella vulgaris: Experimental Design, Modelling and Optimization. Molecules 2022, 27, 5884. [Google Scholar] [CrossRef]
- khorramdashti Mohammad, S.; Giri Mohammad, S.; Majidian, N. Extraction lipids from chlorella vulgaris by supercritical CO2 for biodiesel production. S. Afr. J. Chem. Eng. 2021, 38, 121–131. [Google Scholar] [CrossRef]
- Halim, R.; Gladman, B.; Danquah, M.K.; Webley, P.A. Oil extraction from microalgae for biodiesel production. Bioresour. Technol. 2011, 102, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Ota, M.; Watanabe, H.; Kato, Y.; Watanabe, M.; Sato, Y.; Smith, R.L., Jr.; Inomata, H. Carotenoid production from Chlorococcum littorale in photoautotrophic cultures with downstream supercritical fluid processing. J. Sep. Sci. 2009, 32, 2327–2335. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.T.; Cheng, C.H.; Wu, Y.H.; Lu, W.C.; Lin, Y.H.; Lee, H.T. Continuous lipid extraction of microalgae using high-pressure carbon dioxide. Bioresour. Technol. 2013, 146, 23–26. [Google Scholar] [CrossRef]
- Couto, R.M.; Simões, P.C.; Reis, A.; Da Silva, T.L.; Martins, V.H.; Sánchez-Vicente, Y. Supercritical fluid extraction of lipids from the heterotrophic microalga Crypthecodinium cohnii. Eng. Life Sci. 2010, 158–164. [Google Scholar] [CrossRef]
- Jaime, L.; Mendiola, J.A.; Ibáñez, E.; Martin-Álvarez, P.J.; Cifuentes, A.; Reglero, G.; Señoráns, F.J. β-Carotene Isomer Composition of Sub- and Supercritical Carbon Dioxide Extracts. Antioxidant Activity Measurement. J. Agric. Food Chem. 2007, 55, 10585–10590. [Google Scholar] [CrossRef]
- Mendiola, J.A.; Santoyo, S.; Cifuentes, A.; Reglero, G.; IbÁÑEz, E.; SeÑOrÁNs, F.J. Antimicrobial Activity of Sub- and Supercritical CO2 Extracts of the Green Alga Dunaliella salina. J. Food Prot. 2008, 71, 2138–2143. [Google Scholar] [CrossRef]
- Macias-Sanchez, M.D.; Mantell, C.; Rodriguez, M.; Martinez de la Ossa, E.; Lubian, L.M.; Montero, O. Comparison of supercritical fluid and ultrasound-assisted extraction of carotenoids and chlorophyll a from Dunaliella salina. Talanta 2009, 77, 948–952. [Google Scholar] [CrossRef]
- Macías-Sánchez, M.D.; Serrano, C.M.; Rodríguez, M.R.; Martínez de la Ossa, E. Kinetics of the supercritical fluid extraction of carotenoids from microalgae with CO2 and ethanol as cosolvent. Chem. Eng. J. 2009, 150, 104–113. [Google Scholar] [CrossRef]
- Macias-Sanchez, M.D.; Mantell Serrano, C.; Rodriguez, M.R.; Martinez de la Ossa, E.; Lubian, L.M.; Montero, O. Extraction of carotenoids and chlorophyll from microalgae with supercritical carbon dioxide and ethanol as cosolvent. J. Sep. Sci. 2008, 31, 1352–1362. [Google Scholar] [CrossRef]
- Pour Hosseini, S.R.; Tavakoli, O.; Sarrafzadeh, M.H. Experimental optimization of SC-CO2 extraction of carotenoids from Dunaliella salina. J. Supercrit. Fluids 2017, 121, 89–95. [Google Scholar] [CrossRef]
- Molino, A.; Larocca, V.; Di Sanzo, G.; Martino, M.; Casella, P.; Marino, T.; Karatza, D.; Musmarra, D. Extraction of Bioactive Compounds Using Supercritical Carbon Dioxide. Molecules 2019, 24, 782. [Google Scholar] [CrossRef] [PubMed]
- Tirado, D.F.; Calvo, L. The Hansen theory to choose the best cosolvent for supercritical CO2 extraction of β-carotene from Dunaliella salina. J. Supercrit. Fluids 2019, 145, 211–218. [Google Scholar] [CrossRef]
- Yothipitak, W.; Goto, M.; Shotipruk, A. Experiments and Statistical Analysis of Supercritical Carbon Dioxide Extraction. Chiang Mai J. Sci. 2008, 35, 109–115. [Google Scholar]
- Thana, P.; Machmudah, S.; Goto, M.; Sasaki, M.; Pavasant, P.; Shotipruk, A. Response surface methodology to supercritical carbon dioxide extraction of astaxanthin from Haematococcus pluvialis. Bioresour. Technol. 2008, 99, 3110–3115. [Google Scholar] [CrossRef]
- Bustamante, A.; Roberts, P.J.; Aravena, R.I.; Valle, J.M.d. Supercritical extraction of astaxanthin from H. pluvialis using ethanol-modified CO2. Experiments and modeling. In Proceedings of the 11th International Conference of Eng Food, Athens, Greece, 22–26 May 2011. [Google Scholar]
- Pan, J.-L.; Wang, H.-M.; Chen, C.-Y.; Chang, J.-S. Extraction of astaxanthin from Haematococcus pluvialis by supercritical carbon dioxide fluid with ethanol modifier. Eng. Life Sci. 2012, 12, 638–647. [Google Scholar] [CrossRef]
- Aravena, R.I.; del Valle, J.M. Effect of microalgae preconditioning on supercritical CO2 extraction of astaxanthin from Haematococcus pluvialis. In Proceedings of the 10th International Symposium of Supercritical Fluids, San Francisco, CA, USA, 13–16 May 2012. [Google Scholar]
- Kwan, T.A.; Kwan, S.E.; Peccia, J.; Zimmerman, J.B. Selectively biorefining astaxanthin and triacylglycerol co-products from microalgae with supercritical carbon dioxide extraction. Bioresour. Technol. 2018, 269, 81–88. [Google Scholar] [CrossRef]
- Nobre, B.; Marcelo, F.; Passos, R.; Beirão, L.; Palavra, A.; Gouveia, L.; Mendes, R. Supercritical carbon dioxide extraction of astaxanthin and other carotenoids from the microalga Haematococcus pluvialis. Eur. Food Res. Technol. 2006, 223, 787–790. [Google Scholar] [CrossRef]
- Machmudah, S.; Shotipruk, A.; Goto, M.; Sasaki, M.; Hirose, T. Extraction of Astaxanthin from Haematococcus pluvialis Using Supercritical CO2 and Ethanol as Entrainer. Ind. Eng. Chem. Res. 2006, 45, 3652–3657. [Google Scholar] [CrossRef]
- Krichnavaruk, S.; Shotipruk, A.; Goto, M.; Pavasant, P. Supercritical carbon dioxide extraction of astaxanthin from Haematococcus pluvialis with vegetable oils as co-solvent. Bioresour. Technol. 2008, 99, 5556–5560. [Google Scholar] [CrossRef]
- Wang, L.; Yang, B.; Yan, B.; Yao, X. Supercritical fluid extraction of astaxanthin from Haematococcus pluvialis and its antioxidant potential in sunflower oil. Innov. Food Sci. Emerg. Technol. 2012, 13, 120–127. [Google Scholar] [CrossRef]
- Reyes, F.A.; Mendiola, J.A.; Ibañez, E.; del Valle, J.M. Astaxanthin extraction from Haematococcus pluvialis using CO2-expanded ethanol. J. Supercrit. Fluids 2014, 92, 75–83. [Google Scholar] [CrossRef]
- Sanzo, G.D.; Mehariya, S.; Martino, M.; Larocca, V.; Casella, P.; Chianese, S.; Musmarra, D.; Balducchi, R.; Molino, A. Supercritical Carbon Dioxide Extraction of Astaxanthin, Lutein, and Fatty Acids from Haematococcus pluvialis Microalgae. Mar. Drugs 2018, 16, 334. [Google Scholar] [CrossRef] [PubMed]
- Molino, A.; Mehariya, S.; Iovine, A.; Larocca, V.; Di Sanzo, G.; Martino, M.; Casella, P.; Chianese, S.; Musmarra, D. Extraction of Astaxanthin and Lutein from Microalga Haematococcus pluvialis in the Red Phase Using CO(2) Supercritical Fluid Extraction Technology with Ethanol as Co-Solvent. Mar. Drugs 2018, 16, 432. [Google Scholar] [CrossRef]
- Hernández, D.; Solana, M.; Riaño, B.; García-González, M.C.; Bertucco, A. Biofuels from microalgae: Lipid extraction and methane production from the residual biomass in a biorefinery approach. Bioresour. Technol. 2014, 170, 370–378. [Google Scholar] [CrossRef]
- Gilbert-López, B.; Mendiola, J.A.; Fontecha, J.; van den Broek, L.A.M.; Sijtsma, L.; Cifuentes, A.; Herrero, M.; Ibáñez, E. Downstream processing of Isochrysis galbana: A step towards microalgal biorefinery. Green Chem. 2015, 17, 4599–4609. [Google Scholar] [CrossRef]
- Fujii, K. Process integration of supercritical carbon dioxide extraction and acid treatment for astaxanthin extraction from a vegetative microalga. Food Bioprod. Process. 2012, 90, 762–766. [Google Scholar] [CrossRef]
- Macías-Sánchez, M.D.; Mantell, C.; Rodríguez, M.; Martínez de la Ossa, E.; Lubián, L.M.; Montero, O. Supercritical fluid extraction of carotenoids and chlorophyll a from Nannochloropsis gaditana. J. Food Eng. 2005, 66, 245–251. [Google Scholar] [CrossRef]
- Sánchez-Camargo, A.d.P.; Pleite, N.; Mendiola, J.A.; Cifuentes, A.; Herrero, M.; Gilbert-López, B.; Ibáñez, E. Development of green extraction processes for Nannochloropsis gaditana biomass valorization. Electrophoresis 2018, 39, 1875–1883. [Google Scholar] [CrossRef]
- Molino, A.; Martino, M.; Larocca, V.; Di Sanzo, G.; Spagnoletta, A.; Marino, T.; Karatza, D.; Iovine, A.; Mehariya, S.; Musmarra, D. Eicosapentaenoic Acid Extraction from Nannochloropsis gaditana using Carbon Dioxide at Supercritical Conditions. Mar. Drugs 2019, 17, 132. [Google Scholar] [CrossRef]
- Bjornsson, W.J.; MacDougall, K.M.; Melanson, J.E.; O’Leary, S.J.B.; McGinn, P.J. Pilot-scale supercritical carbon dioxide extractions for the recovery of triacylglycerols from microalgae: A practical tool for algal biofuels research. J. Appl. Phycol. 2011, 24, 547–555. [Google Scholar] [CrossRef]
- Tibbetts, S.M.; Bjornsson, W.J.; McGinn, P.J. Biochemical composition and amino acid profiles of Nannochloropsis granulata algal biomass before and after supercritical fluid CO2 extraction at two processing temperatures. Anim. Feed Sci. Technol. 2015, 204, 62–71. [Google Scholar] [CrossRef]
- Liau, B.-C.; Shen, C.-T.; Liang, F.-P.; Hong, S.-E.; Hsu, S.-L.; Jong, T.-T.; Chang, C.-M.J. Supercritical fluids extraction and anti-solvent purification of carotenoids from microalgae and associated bioactivity. J. Supercrit. Fluids 2010, 55, 169–175. [Google Scholar] [CrossRef]
- Crampon, C.; Mouahid, A.; Toudji, S.-A.A.; Lépine, O.; Badens, E. Influence of pretreatment on supercritical CO2 extraction from Nannochloropsis oculata. J. Supercrit. Fluids 2013, 79, 337–344. [Google Scholar] [CrossRef]
- Bong, S.C.; Loh, S. A study of fatty acid composition and tocopherol content of lipid extracted from marine microalgae, Nannochloropsis oculata and Tetraselmis suecica, using solvent extraction and supercritical fluid extraction. Int. Food Res. J. 2013, 20, 721–729. [Google Scholar]
- Andrich, G.; Nesti, U.; Venturi, F.; Zinnai, A.; Fiorentini, R. Supercritical fluid extraction of bioactive lipids from the microalga Nannochloropsis sp. Eur. J. Lipid Sci. Technol. 2005, 107, 381–386. [Google Scholar] [CrossRef]
- Nobre, B.P.; Villalobos, F.; Barragan, B.E.; Oliveira, A.C.; Batista, A.P.; Marques, P.A.; Mendes, R.L.; Sovova, H.; Palavra, A.F.; Gouveia, L. A biorefinery from Nannochloropsis sp. microalga—Extraction of oils and pigments. Production of biohydrogen from the leftover biomass. Bioresour. Technol. 2013, 135, 128–136. [Google Scholar] [CrossRef]
- Leone, G.P.; Balducchi, R.; Mehariya, S.; Martino, M.; Larocca, V.; Di Sanzo, G.; Iovine, A.; Casella, P.; Marino, T.; Karatza, D.; et al. Selective Extraction of ω-3 Fatty Acids from Nannochloropsis sp. using Supercritical CO2 Extraction. Molecules 2019, 24, 2406. [Google Scholar] [CrossRef]
- Polak, J.T.; Balaban, M.; Peplow, A.; Phlips, A.J. Supercritical Carbon Dioxide Extraction of Lipids from Algae. In Supercritical Fluid Science and Technology; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 1989; Volume 406, pp. 449–467. [Google Scholar]
- Cheng, C.H.; Du, T.B.; Pi, H.C.; Jang, S.M.; Lin, Y.H.; Lee, H.T. Comparative study of lipid extraction from microalgae by organic solvent and supercritical CO2. Bioresour. Technol. 2011, 102, 10151–10153. [Google Scholar] [CrossRef]
- Tommasi, E.; Cravotto, G.; Galletti, P.; Grillo, G.; Mazzotti, M.; Sacchetti, G.; Samorì, C.; Tabasso, S.; Tacchini, M.; Tagliavini, E. Enhanced and Selective Lipid Extraction from the Microalga P. tricornutum by Dimethyl Carbonate and Supercritical CO2 using Deep Eutectic Solvents and Microwaves as Pretreatment. ACS Sustain. Chem. Eng. 2017, 5, 8316–8322. [Google Scholar] [CrossRef]
- Chatterjee, D.; Bhattacharjee, P. Supercritical carbon dioxide extraction of antioxidant rich fraction from Phormidium valderianum: Optimization of experimental process parameters. Algal Res. 2014, 3, 49–54. [Google Scholar] [CrossRef]
- Macías-Sánchez, M.D.; Fernandez-Sevilla, J.M.; Fernández, F.A.; García, M.C.; Grima, E. Supercritical fluid extraction of carotenoids from Scenedesmus almeriensis. Food Chem. 2010, 123, 928–935. [Google Scholar] [CrossRef]
- Mehariya, S.; Iovine, A.; Di Sanzo, G.; Larocca, V.; Martino, M.; Leone, G.P.; Casella, P.; Karatza, D.; Marino, T.; Musmarra, D.; et al. Supercritical Fluid Extraction of Lutein from Scenedesmus almeriensis. Molecules 2019, 24, 1324. [Google Scholar] [CrossRef] [PubMed]
- Soh, L.; Zimmerman, J. Biodiesel production: The potential of algal lipids extracted with supercritical carbon dioxide. Green Chem. 2011, 13, 1422–1429. [Google Scholar] [CrossRef]
- Gilbert-López, B.; Mendiola, J.A.; van den Broek, L.A.M.; Houweling-Tan, B.; Sijtsma, L.; Cifuentes, A.; Herrero, M.; Ibáñez, E. Green compressed fluid technologies for downstream processing of Scenedesmus obliquus in a biorefinery approach. Algal Res. 2017, 24, 111–121. [Google Scholar] [CrossRef]
- Choi, K.J.; Nakhost, Z.; Krukonis, V.J.; Karel, M. Supercritical fluid extraction and characterization of lipids from algae Scenedesmus obliquus. Food Biotechnol. 1987, 1, 263–281. [Google Scholar] [CrossRef]
- Guedes, A.C.; Gião, M.S.; Matias, A.A.; Nunes, A.V.M.; Pintado, M.E.; Duarte, C.M.M.; Malcata, F.X. Supercritical fluid extraction of carotenoids and chlorophylls a, b and c, from a wild strain of Scenedesmus obliquus for use in food processing. J. Food Eng. 2013, 116, 478–482. [Google Scholar] [CrossRef]
- Klejdus, B.; Lojkova, L.; Plaza, M.; Snoblova, M.; Sterbova, D. Hyphenated technique for the extraction and determination of isoflavones in algae: Ultrasound-assisted supercritical fluid extraction followed by fast chromatography with tandem mass spectrometry. J. Chromatogr. A 2010, 1217, 7956–7965. [Google Scholar] [CrossRef]
- Yen, H.-W.; Chiang, W.-C.; Sun, C.-H. Supercritical fluid extraction of lutein from Scenedesmus cultured in an autotrophical photobioreactor. J. Taiwan Inst. Chem. Eng. 2012, 43, 53–57. [Google Scholar] [CrossRef]
- Abrahamsson, V.; Rodriguez-Meizoso, I.; Turner, C. Determination of carotenoids in microalgae using supercritical fluid extraction and chromatography. J. Chromatogr. A 2012, 1250, 63–68. [Google Scholar] [CrossRef]
- Taher, H.; Al-Zuhair, S.; Al-Marzouqi, A.H.; Haik, Y.; Farid, M.; Tariq, S. Supercritical carbon dioxide extraction of microalgae lipid: Process optimization and laboratory scale-up. J. Supercrit. Fluids 2014, 86, 57–66. [Google Scholar] [CrossRef]
- Shomal, R.; Hisham, H.; Mlhem, A.; Hassan, R.; Al-Zuhair, S. Simultaneous extraction–reaction process for biodiesel production from microalgae. Energy Rep. 2019, 5, 37–40. [Google Scholar] [CrossRef]
- Montero, O.; Macías-Sánchez, M.D.; Lama, C.M.; Lubián, L.M.; Mantell, C.; Rodríguez, M.; de la Ossa, E.M. Supercritical CO2 extraction of beta-carotene from a marine strain of the cyanobacterium Synechococcus species. J. Agric. Food Chem. 2005, 53, 9701–9707. [Google Scholar] [CrossRef] [PubMed]
- Macías-Sánchez, M.D.; Mantell, C.; Rodríguez, M.; Martínez de la Ossa, E.; Lubián, L.M.; Montero, O. Supercritical fluid extraction of carotenoids and chlorophyll a from Synechococcus sp. J. Supercrit. Fluids 2007, 39, 323–329. [Google Scholar] [CrossRef]
- Grierson, S.; Strezov, V.; Bray, S.; Mummacari, R.; Danh, L.T.; Foster, N. Assessment of Bio-oil Extraction from Tetraselmis chui Microalgae Comparing Supercritical CO2, Solvent Extraction, and Thermal Processing. Energy Fuels 2012, 26, 248–255. [Google Scholar] [CrossRef]
- Li, Y.; Ghasemi Naghdi, F.; Garg, S.; Adarme-Vega, T.C.; Thurecht, K.J.; Ghafor, W.A.; Tannock, S.; Schenk, P.M. A comparative study: The impact of different lipid extraction methods on current microalgal lipid research. Microb. Cell Factories 2014, 13, 14. [Google Scholar] [CrossRef]
- Safafar, H.; van Wagenen, J.; Moller, P.; Jacobsen, C. Carotenoids, Phenolic Compounds and Tocopherols Contribute to the Antioxidative Properties of Some Microalgae Species Grown on Industrial Wastewater. Mar. Drugs 2015, 13, 7339–7356. [Google Scholar] [CrossRef]
- Hosikian, A.; Lim, S.; Halim, R.; Danquah, M.K. Chlorophyll Extraction from Microalgae: A Review on the Process Engineering Aspects. Int. J. Chem. Eng. 2010, 2010, 391632. [Google Scholar] [CrossRef]
- Fu, W.; Nelson, D.R.; Yi, Z.; Xu, M.; Khraiwesh, B.; Jijakli, K.; Chaiboonchoe, A.; Alzahmi, A.; Al-Khairy, D.; Brynjolfsson, S.; et al. Chapter 6 - Bioactive Compounds from Microalgae: Current Development and Prospects. In Studies in Natural Products Chemistry; Attaur, R., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; Volume 54, pp. 199–225. [Google Scholar]
- Balasubramaniam, V.; Gunasegavan, R.D.; Mustar, S.; Lee, J.C.; Mohd Noh, M.F. Isolation of Industrial Important Bioactive Compounds from Microalgae. Molecules 2021, 26, 943. [Google Scholar] [CrossRef]
- Novoveská, L.; Ross, M.E.; Stanley, M.S.; Pradelles, R.; Wasiolek, V.; Sassi, J.F. Microalgal Carotenoids: A Review of Production, Current Markets, Regulations, and Future Direction. Mar. Drugs 2019, 17, 640. [Google Scholar] [CrossRef]
- Guedes, A.C.; Amaro, H.M.; Malcata, F.X. Microalgae as Sources of Carotenoids. Mar. Drugs 2011, 9, 625–644. [Google Scholar] [CrossRef] [PubMed]
- Smaoui, S.; Barkallah, M.; Ben Hlima, H.; Fendri, I.; Mousavi Khaneghah, A.; Michaud, P.; Abdelkafi, S. Microalgae Xanthophylls: From Biosynthesis Pathway and Production Techniques to Encapsulation Development. Foods 2021, 10, 2835. [Google Scholar] [CrossRef] [PubMed]
- Henríquez, V.; Escobar, C.; Galarza, J.; Gimpel, J. Carotenoids in microalgae. Carotenoids Nat. 2016, 79, 219–237. [Google Scholar]
- da Silva, J.C.; Lombardi, A.T. Chlorophylls in Microalgae: Occurrence, Distribution, and Biosynthesis. In Pigments from Microalgae Handbook; Jacob-Lopes, E., Queiroz, M.I., Zepka, L.Q., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–18. [Google Scholar]
- da Silva Ferreira, V.; Sant’Anna, C. Impact of culture conditions on the chlorophyll content of microalgae for biotechnological applications. World J. Microbiol. Biotechnol. 2016, 33, 20. [Google Scholar] [CrossRef] [PubMed]
- Mimouni, V.; Couzinet-Mossion, A.; Ulmann, L.; Wielgosz-Collin, G. Chapter 5—Lipids from Microalgae. In Microalgae in Health and Disease Prevention; Levine, I.A., Fleurence, J., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 109–131. [Google Scholar]
- de Carvalho, J.C.; Magalhaes, A.I., Jr.; de Melo Pereira, G.V.; Medeiros, A.B.P.; Sydney, E.B.; Rodrigues, C.; Aulestia, D.T.M.; de Souza Vandenberghe, L.P.; Soccol, V.T.; Soccol, C.R. Microalgal biomass pretreatment for integrated processing into biofuels, food, and feed. Bioresour. Technol. 2020, 300, 122719. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Cho, J.M.; Chang, Y.K.; Oh, Y.K. Cell disruption and lipid extraction for microalgal biorefineries: A review. Bioresour. Technol. 2017, 244, 1317–1328. [Google Scholar] [CrossRef]
- Bernaerts, T.M.M.; Gheysen, L.; Foubert, I.; Hendrickx, M.E.; Van Loey, A.M. The potential of microalgae and their biopolymers as structuring ingredients in food: A review. Biotechnol. Adv. 2019, 37, 107419. [Google Scholar] [CrossRef]
- Lee, A.K.; Lewis, D.M.; Ashman, P.J. Disruption of microalgal cells for the extraction of lipids for biofuels: Processes and specific energy requirements. Biomass Bioenergy 2012, 46, 89–101. [Google Scholar] [CrossRef]
- Postma, P.R.; Suarez-Garcia, E.; Safi, C.; Yonathan, K.; Olivieri, G.; Barbosa, M.J.; Wijffels, R.H.; Eppink, M.H.M. Energy efficient bead milling of microalgae: Effect of bead size on disintegration and release of proteins and carbohydrates. Bioresour. Technol. 2017, 224, 670–679. [Google Scholar] [CrossRef]
- Suarez Garcia, E.; Lo, C.; Eppink, M.H.M.; Wijffels, R.H.; van den Berg, C. Understanding mild cell disintegration of microalgae in bead mills for the release of biomolecules. Chem. Eng. Sci. 2019, 203, 380–390. [Google Scholar] [CrossRef]
- Greenly, J.M.; Tester, J.W. Ultrasonic cavitation for disruption of microalgae. Bioresour. Technol. 2015, 184, 276–279. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Muppaneni, T.; Sun, Y.; Reddy, H.K.; Fu, J.; Lu, X.; Deng, S. Microwave-assisted extraction of lipids from microalgae using an ionic liquid solvent [BMIM][HSO4]. Fuel 2016, 178, 49–55. [Google Scholar] [CrossRef]
- Passos, F.; Uggetti, E.; Carrère, H.; Ferrer, I. Algal Biomass. In Pretreatment of Biomass; Routledge: London, UK, 2015; pp. 195–226. [Google Scholar]
- D’Hondt, E.; Martín-Juárez, J.; Bolado, S.; Kasperoviciene, J.; Koreiviene, J.; Sulcius, S.; Elst, K.; Bastiaens, L. 6—Cell disruption technologies. In Microalgae-Based Biofuels and Bioproducts; Gonzalez-Fernandez, C., Muñoz, R., Eds.; Woodhead Publishing: Sawston, UK, 2017; pp. 133–154. [Google Scholar]
- Yoo, G.; Park, M.S.; Yang, J.-W. Chemical Pretreatment of Algal Biomass. In Pretreatment of Biomass; Routledge: London, UK, 2015; pp. 227–258. [Google Scholar]
- Dular, M.; Griessler-Bulc, T.; Gutierrez-Aguirre, I.; Heath, E.; Kosjek, T.; Krivograd Klemencic, A.; Oder, M.; Petkovsek, M.; Racki, N.; Ravnikar, M.; et al. Use of hydrodynamic cavitation in (waste) water treatment. Ultrason. Sonochem. 2016, 29, 577–588. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Liu, J.; Ji, L.; Wang, G.; Zhao, S.; Yoon, J.Y.; Chen, S. A review on hydrodynamic cavitation disinfection: The current state of knowledge. Sci. Total Environ. 2020, 737, 139606. [Google Scholar] [CrossRef] [PubMed]
- Lam, G.P.t.; Postma, P.R.; Fernandes, D.A.; Timmermans, R.A.H.; Vermuë, M.H.; Barbosa, M.J.; Eppink, M.H.M.; Wijffels, R.H.; Olivieri, G. Pulsed Electric Field for protein release of the microalgae Chlorella vulgaris and Neochloris oleoabundans. Algal Res. 2017, 24, 181–187. [Google Scholar] [CrossRef]
- Zbinden, M.D.; Sturm, B.S.; Nord, R.D.; Carey, W.J.; Moore, D.; Shinogle, H.; Stagg-Williams, S.M. Pulsed electric field (PEF) as an intensification pretreatment for greener solvent lipid extraction from microalgae. Biotechnol. Bioeng. 2013, 110, 1605–1615. [Google Scholar] [CrossRef]
- Mishra, V.; Dubey, A.; Prajapti, S.K. Algal Biomass Pretreatment for Improved Biofuel Production. In Algal Biofuels; Springer: Cham, Switzerland, 2017; pp. 259–280. [Google Scholar]
- Lorente, E.; Haponska, M.; Clavero, E.; Torras, C.; Salvado, J. Microalgae fractionation using steam explosion, dynamic and tangential cross-flow membrane filtration. Bioresour. Technol. 2017, 237, 3–10. [Google Scholar] [CrossRef]
- Aarthy, A.; Kumari, S.; Turkar, P.; Subramanian, S. An insight on algal cell disruption for biodiesel production. Asian J. Pharm. Clin. Res. 2018, 11, 21–26. [Google Scholar] [CrossRef]
- Velazquez-Lucio, J.; Rodríguez-Jasso, R.M.; Colla, L.M.; Sáenz-Galindo, A.; Cervantes-Cisneros, D.E.; Aguilar, C.N.; Fernandes, B.D.; Ruiz, H.A. Microalgal biomass pretreatment for bioethanol production: A review. Biofuel Res. J. 2018, 5, 780–791. [Google Scholar] [CrossRef]
- Khoo, K.S.; Lee, S.Y.; Ooi, C.W.; Fu, X.; Miao, X.; Ling, T.C.; Show, P.L. Recent advances in biorefinery of astaxanthin from Haematococcus pluvialis. Bioresour. Technol. 2019, 288, 121606. [Google Scholar] [CrossRef]
- Scholz, M.J.; Weiss, T.L.; Jinkerson, R.E.; Jing, J.; Roth, R.; Goodenough, U.; Posewitz, M.C.; Gerken, H.G. Ultrastructure and Composition of the Nannochloropsis gaditana Cell Wall. Eukaryot. Cell 2014, 13, 1450–1464. [Google Scholar] [CrossRef] [PubMed]
- Mularczyk, M.; Michalak, I.; Marycz, K. Astaxanthin and other Nutrients from Haematococcus pluvialis—Multifunctional Applications. Mar. Drugs 2020, 18, 459. [Google Scholar] [CrossRef] [PubMed]
- Burczyk, J.; Szkawran, H.; Zontek, I.; Czygan, F.-C. Carotenoids in the Outer Cell-Wall Layer of Scenedesmus (Chlorophyceae). Planta 1981, 247–250. [Google Scholar] [CrossRef]
- Kopcak, U.; Mohamed, R.S. Caffeine solubility in supercritical carbon dioxide/co-solvent mixtures. J. Supercrit. Fluids 2005, 34, 209–214. [Google Scholar] [CrossRef]
- Sovová, H. Mathematical model for supercritical fluid extraction of natural products and extraction curve evaluation. J. Supercrit. Fluids 2005, 33, 35–52. [Google Scholar] [CrossRef]
- Oliveira, E.L.G.; Silvestre, A.J.D.; Silva, C.M. Review of kinetic models for supercritical fluid extraction. Chem. Eng. Res. Des. 2011, 89, 1104–1117. [Google Scholar] [CrossRef]
- Sovová, H. Rate of the vegetable oil extraction with supercritical CO2—I. Modelling of extraction curves. Chem. Eng. Sci. 1994, 49, 409–414. [Google Scholar] [CrossRef]
- Huang, Z.; Shi, X.-h.; Jiang, W.-j. Theoretical models for supercritical fluid extraction. J. Chromatogr. A 2012, 1250, 2–26. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Iverson, S.J.; Lang, S.L.C.; Cooper, M.H. Comparison of the bligh and dyer and folch methods for total lipid determination in a broad range of marine tissue. Lipids 2001, 36, 1283–1287. [Google Scholar] [CrossRef] [PubMed]
- Araujo, G.S.; Matos, L.J.B.L.; Fernandes, J.O.; Cartaxo, S.J.M.; Gonçalves, L.R.B.; Fernandes, F.A.N.; Farias, W.R.L. Extraction of lipids from microalgae by ultrasound application: Prospection of the optimal extraction method. Ultrason. Sonochemistry 2013, 20, 95–98. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, B.; Christen, P. Recent extraction techniques for natural products: Microwave-assisted extraction and pressurised solvent extraction. Phytochem. Anal. 2002, 13, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Veggi, P.C.; Martinez, J.; Meireles, M.A.A. Fundamentals of Microwave Extraction. In Microwave-assisted Extraction for Bioactive Compounds: Theory and Practice; Chemat, F., Cravotto, G., Eds.; Springer US: Boston, MA, USA, 2013; pp. 15–52. [Google Scholar]
- Mandal, V.; Mohan, Y.; Hemalatha, S. Microwave Assisted Extraction—An Innovative and Promising Extraction Tool for Medicinal Plant Research. Pharmacogn. Rev. 2006, 1, 7–18. [Google Scholar]

| Algae | Pretreatment 1 | Carotenoids 1 | Chlorophylls 1 | Other Bioactive 1 | Lipids 1 | Fatty Acids 1 | TPC AO AM 1,2 | Kinetic Model 3 | Exp. Design 3 | Other Methods 3 | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Arthrospira maxima | ✓ | ✓ | [24] | ||||||||
| ✓ | ✓ | ✓ | ✓ | [25,26] | |||||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [27] | ||||||
| ✓ | ✓ | [28] | |||||||||
| Arthrospira pacifica | ✓ | ✓ | ✓ | [29] | |||||||
| Arthrospira platensis | ✓ | ✓ | ✓ | ✓ | ✓ | [30] | |||||
| ✓ | ✓ | ✓ | [31] | ||||||||
| ✓ | ✓ | ✓ | ✓ | [32] | |||||||
| ✓ | ✓ | [33] | |||||||||
| ✓ | ✓ | ✓ | AO | [34] | |||||||
| ✓ | ✓ | ✓ | ✓ | [35] | |||||||
| ✓ | AO AM | [36] | |||||||||
| ✓ | ✓ | [37] | |||||||||
| ✓ | ✓ | [38] | |||||||||
| ✓ | ✓ | ✓ | ✓ | [39] | |||||||
| Botryococcus braunii | ✓ | ✓ | [24,40] | ||||||||
| ✓ | ✓ | [41] | |||||||||
| Chaetoceros muelleri | AM | [42] | |||||||||
| Chlorella protothecoides | ✓ | ✓ | [43] | ||||||||
| ✓ | ✓ | ✓ | [44] | ||||||||
| ✓ | [45] | ||||||||||
| Chlorella pyrenoidosa | AO | ✓ | [46] | ||||||||
| ‘✓ | ✓ | [47] | |||||||||
| Chlorella saccharophila | ✓ | ✓ | [48] | ||||||||
| Chlorella sorokiniana | ✓ | ✓ | ✓ | ✓ | ✓ | [49] | |||||
| Chlorella sp. | ✓ | ✓ | ✓ | [50] | |||||||
| ✓ | [51] | ||||||||||
| ✓ | ✓ | ✓ | ✓ | [52] | |||||||
| ✓ | [53] | ||||||||||
| ✓ | [54] | ||||||||||
| Chlorella vulgaris | ✓ | ✓ | ✓ | [55] | |||||||
| ✓ | [56] | ||||||||||
| ✓ | ✓ | ✓ | [57] | ||||||||
| ✓ | ✓ | ✓ | [58] | ||||||||
| ✓ | ✓ | ✓ | [59] | ||||||||
| ✓ | ✓ | [60] | |||||||||
| ✓ | ✓ | [24] | |||||||||
| ✓ | ✓ | ✓ | [61] | ||||||||
| ✓ | ✓ | [62] | |||||||||
| ✓ | ✓ | [38] | |||||||||
| ✓ | ✓ | ✓ | [63] | ||||||||
| ✓ | ✓ | ✓ | [40] | ||||||||
| ✓ | ✓ | [64] | |||||||||
| ✓ | TPC | ✓ | [65] | ||||||||
| ✓ | ✓ | AO TPC | ✓ | ✓ | ✓ | [66] | |||||
| ✓ | [67] | ||||||||||
| Chlorococcum sp. | ✓ | ✓ | ✓ | ✓ | [68] | ||||||
| Chlorococcum littorale | ✓ | ✓ | ✓ | [69] | |||||||
| Commercial DHA algae | ✓ | ✓ | [70] | ||||||||
| Crypthecodinium cohnii | ✓ | ✓ | ✓ | [71] | |||||||
| Cylindrotheca. closterium | ✓ | ✓ | [38] | ||||||||
| Dunaliella salina | ✓ | ✓ | AO | ✓ | [72] | ||||||
| ✓ | ✓ | [73] | |||||||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [74] | ||||||
| ✓ | ✓ | [75] | |||||||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [76] | ||||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [77] | ||||||
| ✓ | ✓ | ✓ | [78] | ||||||||
| ✓ | [79] | ||||||||||
| Haematococcus pluvialis | ✓ | ✓ | [80] | ||||||||
| ✓ | ✓ | AO | ✓ | ✓ | [81] | ||||||
| ✓ | ✓ | ✓ | ✓ | [82] | |||||||
| ✓ | ✓ | ✓ | ✓ | [83] | |||||||
| ✓ | ✓ | ✓ | [84] | ||||||||
| ✓ | ✓ | ✓ | ✓ | [85] | |||||||
| ✓ | ✓ | ✓ | [28] | ||||||||
| ✓ | ✓ | ✓ | [86] | ||||||||
| ✓ | ✓ | ✓ | [87] | ||||||||
| ✓ | ✓ | ✓ | [88] | ||||||||
| ✓ | ✓ | [89] | |||||||||
| ✓ | AO | ✓ | ✓ | [90] | |||||||
| ✓ | ✓ | ✓ | [91] | ||||||||
| ✓ | ✓ | [92] | |||||||||
| Isochrysis sp. | ✓ | ✓ | ✓ | ✓ | ✓ | [93] | |||||
| Isochrysis galbana | ✓ | ✓ | ✓ | ✓ | ✓ | [94] | |||||
| Monoraphidium sp. | ✓ | ✓ | ✓ | ✓ | [95] | ||||||
| Nannochloropsis gaditana | ✓ | ✓ | ✓ | ✓ | ✓ | [96] | |||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [76] | ||||||
| ✓ | ✓ | ✓ | [75] | ||||||||
| ✓ | ✓ | AO | ✓ | ✓ | ✓ | [93] | |||||
| ✓ | ✓ | ✓ | ✓ | [97] | |||||||
| ✓ | ✓ | AO | [98] | ||||||||
| Nannochloropsis granulata | ✓ | ✓ | ✓ | [99] | |||||||
| ✓ | ✓ | ✓ | [100] | ||||||||
| Nannochloropsis oculata | ✓ | ✓ | AO | ✓ | [101] | ||||||
| ✓ | ✓ | ✓ | ✓ | [102] | |||||||
| ✓ | ✓ | ✓ | ✓ | [103] | |||||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [38] | ||||||
| Nannochloropsis salina | ✓ | [44] | |||||||||
| Nannochloropsis sp. | ✓ | ✓ | ✓ | ✓ | [104] | ||||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [105] | ||||||
| ✓ | ✓ | ✓ | ✓ | [106] | |||||||
| Ochromonas danica | ✓ | ✓ | [107] | ||||||||
| Pavlova sp. | ✓ | ✓ | ✓ | [108] | |||||||
| Phaeodactylum tricornutum | ✓ | ✓ | ✓ | ✓ | [109] | ||||||
| Phormidium valderianum | ✓ | ✓ | AO TPC | ✓ | ✓ | [110] | |||||
| Scenedesmus almeriansis | ✓ | ✓ | ✓ | ✓ | [111] | ||||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [93] | ||||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [112] | ||||||
| Scenedesmus dimorphus | ✓ | ✓ | ✓ | [113] | |||||||
| Scenedesmus obliquus | ✓ | ✓ | ✓ | ✓ | ✓ | [44] | |||||
| ✓ | ✓ | ✓ | ✓ | [2] | |||||||
| ✓ | ✓ | ✓ | ✓ | ✓ | [114] | ||||||
| ✓ | ✓ | [115] | |||||||||
| ✓ | ✓ | ✓ | ✓ | [116] | |||||||
| Scenedesmus obtusiusculus | ✓ | ✓ | ✓ | [2] | |||||||
| Scenedesmus sp. | ✓ | [117] | |||||||||
| ✓ | ✓ | ✓ | [118] | ||||||||
| ✓ | ✓ | ✓ | [119] | ||||||||
| ✓ | ✓ | ✓ | ✓ | [120] | |||||||
| ✓ | ✓ | ✓ | [121] | ||||||||
| Skeletonema costatum | ✓ | [107] | |||||||||
| Synechococcus sp. | ✓ | ✓ | ✓ | ✓ | [122] | ||||||
| ✓ | ✓ | ✓ | ✓ | [76] | |||||||
| ✓ | ✓ | ✓ | ✓ | [123] | |||||||
| ✓ | ✓ | [75] | |||||||||
| ✓ | ✓ | ✓ | ✓ | [17] | |||||||
| Tetraselmis chui | ✓ | ✓ | [124] | ||||||||
| Tetraselmis sp. | ✓ | ✓ | ✓ | ✓ | [93] | ||||||
| ✓ | ✓ | ✓ | [125] |
| Algae | Pretreatment | Parametric Investigation | Optimal Conditions | Ext. Yield/Recovery | Carotenoids | Chlorophylls | Other Pigments | Extract Properties | Lipids | Fatty Acids | Ref. |
|---|---|---|---|---|---|---|---|---|---|---|---|
| A. maxima | GR | T (20–70 °C), P (15–18 MPa), CO2 Flow (3.33 × 10−5 kg/s), t (660 min) | T (30 °C), P (18 MPa), CO2 Flow (3.33 × 10−5 kg/s) t (660 min) | 2.27 mg T.CAR/0.8 kg/cm3 bed | 23.64 mg/0.8 kg/cm3 bed FA content | [27] | |||||
| Crushed by cutting mills | T (60 °C), P (30 MPa), Co-solv (EtOH 0–10% w/w) | T (60 °C), P (30 MPa), Co-solv (EtOH 10% w/w) | 2.97% | 3% PHY >97% AST Rec | [28] | ||||||
| T (50–60 °C), P (25–35 MPa), Co-solv (EtOH 0–10% v/v) | T (60 °C), P (35 MPa), Co-solv (EtOH 10% v/v) | 0.44% GLA | [24] | ||||||||
| LY and GR | T (50–60 °C), P (25–35 MPa), CO2 Flow (2 g/min), t (390 min), Co-solv (EtOH 0–10% v/v) | T (60 °C), P (35 MPa), CO2 Flow (2 g/min), t (390 min), Co-solv (EtOH 10% v/v) | 0.44% GLA | [25,26] | |||||||
| A. pacifica | T (40–80 °C), P (15–35 MPa), CO2 Flow (2 mL/min), t (40–100 min), Co-solv (EtOH 5–15% v/v) | T (60–80 °C), P (35 MPa), CO2 Flow (2 mL/min), t (100 min), Co-solv (EtOH 15% v/v) | 48 mg/100 g ZEA, 7.5 mg/100 g β-CRY 118 mg/100 g β-CAR | [29] | |||||||
A. platensis | LY and milled | T (45–60 °C), P (15–45 MPa), CO2 Flow (0.015 kg/h), t (50 min), Co-solv (EtOH 26.70–53.22% v/v) | T (60 °C), P (45 MPa), CO2 Flow (0.015 kg/h), t (50 min), Co-solv (EtOH 53.22% v/v) | 4.07% | 283 μg/g T.CAR | 5.01 μg/g TOC | 34.76 mg/g FA | [30] | |||
| T (32–48 °C), P (20–40 MPa), t (120–240 min), Co-solv (EtOH) | T (48 °C), P (20 MPa), t (240 min), Co-solv (EtOH) | 10.26 g/kg | 77.8 g/kg β-CAR 113.2 g/kg Vitamin A | 85.1 g/kg Flavonoids 3.4 g/kg α-TOC | 35.32% PA, 21.66% LNA, 20.58% LOA | [35] | |||||
| Air-dried | T (55 °C), P (8–36 MPa), CO2 Flow (3 L/h), Co-solv (EtOH 10% mol) | T (55 °C), P (22 MPa), CO2 Flow (3 L/h), Co-solv (EtOH 10% mol) | 0.63% (SEP 1) 2.46% (SEP 2) | 178.2 ppm ZEA (SEP 1) 109.3 ppm ZEA (SEP 2) 19.8 ppm MYX fucoside (SEP 1) 52.9 ppm MYX fucoside (SEP 2) 55.0 ppm β-CAR (SEP 2) | CHL-a 480.1 ppm (SEP 1) 55.0 ppm (SEP 2) | AO 66.6 μg/mL EC50 (SEP 1) 73.5 μg/mL EC50 (SEP 2) | [34] | ||||
| T (40–80 °C), P (10–30 MPa), t (30–90 min), Co-solv (EtOH 10–50% v/v) | T (40 °C), P (30 MPa), t (90 min), Co-solv (EtOH 50% v/v) | 6.7% w/w | 24.7% GLA Rec | [31] | |||||||
| T (33.18–66.82 °C), P (23.2–56.8 MPa), CO2 Flow (0.24–0.9 kg/h), t (0–120 min soaking and 30–180 min extraction) Co-solv (MeOH, ACE, EtA 0–10 mL, Aq.EtOH (20–80%) 5–28.4 mL) | T (53.4 °C), P (48.7 MPa), CO2 Flow (0.6 kg/h), t (60 min soaking and 120 min extraction) Co-solv (Aq.EtOH (40%) 21.2 mL) | 6.84 mg/g CHL-a | [37] | ||||||||
| LY | T (40 °C), P (31.6–48.4 MPa), CO2 Flow (0.7 L/min), t (26.4–94 min), Co-solv (EtOH 9.64–16.36 mL) | T (40 °C), P (40 MPa), CO2 Flow (0.7 L/min), t (60 min), Co-solv (EtOH 13.7 mL) | 102% GLA Rec | [32] | |||||||
| Air-flow dried | T (60 °C), P (40 MPa), CO2 Flow (0.35 kg/h) | T (60 °C), P (40 MPa), CO2 Flow (0.35 kg/h) | 10.98% | [38] | |||||||
| LY and GR | T (40–55 °C), P (25–70 MPa), CO2 Flow (10 kg/h), t (90–240 min) | T (55 °C), P (70 MPa), CO2 Flow (10 kg/h), t (90 min) | 7.79% Lipid | 37–41% Total FA | [39] | ||||||
| T (27–83 °C), P (7.8–36.1 MPa), t (75 min), Co-solv (EtOH 0–10% v/v) | T (55 °C), P (22–32 MPa), t (75 min), Co-solv (wihout) or T (75 °C), P (32 MPa), t (75 min) Co-solv (EtOH 10% v/v) | AO (EC50) 66.7 μg/mL (SEP 1) 36.1 μg/mL (SEP 2) OR 20.0 μg/mL (SEP 1), 129.4 μg/mL (SEP 2) MBC 10–30 mg/mL E. coli, 10–25 mg/mL S.aureus, 10–15 mg/mL C.albicans, >35 mg/mL A.niger | 44.4%(SEP 1), 36.6% (SEP 2) PA, 30.6%(SEP 1), 25%(SEP 2) OA | [36] | |||||||
| T (40 °C), P (30–40 MPa), CO2 Flow (24 kg/h), t (120–240 min) | T (40 °C), P (35 MPa), CO2 Flow (24 kg/h), t (240 min) | 7.2% lipid | Composition 16.91% OA, 36.51% LA., 9.16% α-LNA., 19.68% GLA. | [33] | |||||||
| Botryococcus braunii | T (40 °C), P (12.5–30 MPa) | T (40 °C), P (30 MPa) | ~72 g/kg Hydrocarbons | [24,40] | |||||||
| T (50–80 °C), P (20–25 MPa), t (10–150 min) | T (50 °C), P (25 MPa) | ~10.5% | ~18% FA | [41] | |||||||
| Chaetoceros muelleri | T (40–80 °C), P (20–40 MPa), t (60 min), Co-solv (EtOH 0.2 mL) | T (40 °C), P (40 MPa), t (60 min), Co-solv (EtOH 0.2 mL) | 3.9% | MBC 12 mg/mL E. coli 12 mg/mL S. aureus 7 mg/mL C. albicans | [42] | ||||||
| Chlorella protothecoides | T (50 °C), P (35 MPa), CO2 Flow (0.0439 kg/h), t (180 min) | T (50 °C), P (35 MPa), CO2 Flow (0.0439 kg/h), t (180 min) | 0.23 g/gbiom lipid 75% Rec | [43] | |||||||
| Oven dried, GR and sieved | T (60 °C), P (30 MPa), CO2 Flow (30 g/h), t (90 min), Co-solv (EtOH 5%) | T (60 °C), P (30 MPa), CO2 Flow (30 g/h), t (90 min), Co-solv (EtOH 5%) | 10% Lipid | Composition 25.68% SFA 13.1% MUFA 61.77% PUFA 15.13% Ω-3 23.63% Ω-6 | [44] | ||||||
| Oven dried, milled, MW, sonication, autoclave | T (35–70 °C), P (15–30 MPa), CO2 Flow (3–7 g/min) | T (70 °C), P (30 MPa), CO2 Flow (3 g/min) | 21% | [45] | |||||||
| C. pyrenoidosa | LY, superfine pulverized | T (40–60 °C), P (20–30 MPa), CO2 Flow (20 kg/h), t (2–8 h), Co-solv (EtOH 0–70%) | T (50 °C), P (25 MPa), CO2 Flow (20 kg/h), t (4 h), Co-solv (EtOH 50%) | 87% LUT Rec | [47] | ||||||
| T (32–55 °C), P (25–40 MPa), CO2 Flow (15–30 kg/h), t (1.5–180 min), Co-solv (EtOH 0–1.5 mL/gbiom) | T (32 °C), P (40 MPa), CO2 Flow (20 kg/h), t (180 min), Co-solv (EtOH 1 mL/gbiom) | 7.78% | AO 42.03% Inhibition | [46] | |||||||
| C. saccharophila | T (42–73 °C), P (24.1–41.4 MPa), t (30–90 min) | T (73 °C), P (24.1 MPa), t (86 min) | 20.4% T-FAME Comp. | [48] | |||||||
| C. sorokiniana | High-pressure cell disruption | T (40–60 °C), P (10–30 MPa), t (180 min), Co-solv (EtOH 0–10%) | T (50 °C), P (20 MPa), t (180 min), Co-solv (EtOH 5%) | 35.03 mg/g | 0.526 mg/g (18.8% Rec) LUT 0.056 mg/g (26.2% Rec) VIO 0.051 mg/g (16.8% Rec) ZEA 0.557 mg/g (73.7% Rec) Carotene | 4.60 mg/g (36.2% Rec) CHL-a 3.92 mg/g (82.3% Rec) CHL-b | [49] | ||||
| Chlorella sp. | T (40–60 °C) P (15–30 MPa), CO2 Flow (15 g/min), t (180 min), Co-solv (Hexane/MeOH 1–3 v/v) | T (40 °C), P (30 MPa), CO2 Flow (15 g/min), t (180 min), Co-solv (Hexane/MeOH 2 v/v) | 47.2% | [51] | |||||||
| T (60 °C), P (20 MPa), CO2 Flow (0.5–2 L/min), t (240 min), Co-solv (Hexane 0.4 mL/min) | T (60 °C), P (20 MPa), CO2 Flow (0.5 L/min), t (240 min), Co-solv (Hexane 0.4 mL/min) | 63.78% Lipid Y | [53] | ||||||||
| T (40–60 °C), P (20–30 MPa), CO2 Flow (6.7–20 g/min) | T (60 °C), P (30 MPa), | 2.2% | 79.53% Lipid Y | [54] | |||||||
| T (40–50 °C), P (15–30 MPa), CO2 Flow (0.5–4 g/min), Co-solv (EtOH 0–30%) | T (40 °C), P (30 MPa), CO2 Flow (1.88 g/min), Co-solv (EtOH ~30%) | 160–222 μg/g T.CAR | 830–1050 μg/g CHL-a | 360–400 μg/g Ergosterol | [50] | ||||||
| LY and bead milled | T (60 °C), P (20–30 MPa), CO2 Flow (30 g/h), t (180 min), Co-solv (EtOH 0–5%) | T (60 °C), P (30 MPa), CO2 Flow (30 g/h), t (180 min), Co-solv (EtOH 5% w/v) | 5 mg/g (26.2% Rec) T.CAR | 9 mg/g T. CHL | [52] | ||||||
| C. vulgaris | T (60–80 °C), P (20–50 MPa), CO2 Flow (2.5 mL/min), t (3–6 h), Co-solv (EtOH or ACE 7.5% v/v) | T (60 °C), P (30 MPa), CO2 Flow (2.5 mL/min), t (6 h), Co-solv (EtOH 7.5% v/v) | 3 mg/g LUT 0.06 mg/g Carotene | 7 mg/g CHL-a 3 mg/g CHL-b | [59] | ||||||
| LY, 3 degrees of crushing | T (40–55 °C), P (20–35 MPa) | T (55 °C), P (35 MPa) | 40% T.CAR Rec | [24] | |||||||
| 3 degrees of crushing | T (40 °C), P (12.5–30 MPa), CO2 Flow (0.04 kg/h) | T (40 °C), P (30 MPa), CO2 Flow (0.04 kg/h) | >70% T.CAR Rec | [40] | |||||||
| MW | T (40–70 °C), P (20–28 MPa), CO2 Flow (10 kg/h), t (9 h) | T (70 °C), P (28 MPa), CO2 Flow (10 kg/h), t (9 h) | 4.86% | 26.598 mg/100 mgoil PA 27.296 mg/100 mgoil OA 10.403 mg/100 mgoil LNA 16.163 mg/100 mgoil a-LNA | [57] | ||||||
| Air dried | T (45 °C), P (45 MPa), CO2 Flow (25 g/min) | T (45 °C), P (45 MPa), CO2 Flow (25 g/min) | ~14 % | [38] | |||||||
| LY, crushed | T (40–55 °C), P (15–35 MPa), CO2 Flow (0.4 dm3/min), t (125–480) | T (55 °C), P (35 MPa), CO2 Flow (0.4 dm3/min), t (330 min) | 5% T.CAR | [61] | |||||||
| T (40–60 °C), P (11–25 MPa), CO2 Flow (20–40 g/min) | T (60 °C), P (25 MPa), CO2 Flow (40 g/min) | 3.37% | 21.14 mg/gextr T.CAR 10.00 mg/gextr Sel. CAR | 35.55 mg/gextr T.CHL | AO 44.35 mgextr/mgDPPH TPC 18.29 mgGA/gextr | [66] | |||||
| LY | T (50 °C), P (31 MPa), CO2 Flow (6 NL/min), t (20 min), Co-solv (Aq. EtOH (50%) 50 mL) | T (50 °C), P (31 MPa), CO2 Flow (6 NL/min), t (20 min), Co-solv (Aq. EtOH (50%) 50 mL) | 8.71% | TPC 13.40 mg GAE/gextr | [65] | ||||||
| T (40–60 °C), P (27.6–48.3 MPa), CO2 Flow (1–3 g/min), t (1–180 min) | T (60 °C), P (48.3 MPa), CO2 Flow (3 g/min), t (180 min) | 17.7% | [55] | ||||||||
| T (40–80 °C), P (27.6–62.1 MPa), t (180 min) | T (80 °C), P (62.1 MPa), t (180 min) | 19% >99% Rec | [56] | ||||||||
| Crushed (3 degrees) | T (40 °C), P (30 MPa), CO2 Flow (0.34–0.6 L/min), Co-solv (EtOH or oil) | T (40 °C), P (30 MPa), CO2 Flow (0.34 L/min), Co-solv (oil) | [58] | ||||||||
| LY, crushed | T (40–55 °C), P (20–35 MPa), CO2 Flow (0.4 dm3/min), t (125–480 min) | T (55 °C), P (35 MPa), CO2 Flow (0.4 dm3/min), t (330 min) | 0.05% | 54.26 mg/g Total Lipid Y | [62] | ||||||
| T (40–80 °C), P (20–37 MPa), CO2 Flow (100–200 g/min), t (60 min), Co-solv (Hexane/EtOH (1:1) 4–12 w/w biomass) | T (40 °C), P (37 MPa) | Composition 30.05% PA 30.22% STA 3.24% LAA 4.82% MA 3.01% AA 2.54% PLA 3.38% OA 1.63% LNA 1.71% DHA 2.98% EPA | [67] | ||||||||
| LY | T (50 °C), P (25 MPa), CO2 Flow (0.5 kg/h), t (210–230 min) Co-solv (EtOH 0–10% v/v) | T (50 °C), P (25 MPa), CO2 Flow (0.5 kg/h), t (230 min), Co-solv (EtOH 10% v/v) | ~40% | 97% Rec Neutral Lipid ~25% Rec Glycolipid ~35% Rec Phospholipid | [63] | ||||||
| Spray-dried, eluent pretreated | T (40–80 °C), P (20–40 MPa), CO2 Flow (3 mL/min), t (100 min), Co-solv (EtOH 0.3–0.5 mL/min) | T (40 °C), P (40 MPa), CO2 Flow (3 mL/min), t (100 min), Co-solv (EtOH 0.4 mL/min) | ~1.8% | 52.9% LUT Rec | [64] | ||||||
| Chlorococcum littorale | LY | T (60 °C), P (30 MPa), CO2 Flow (0.36 dm3/min), t (80 min), Co-solv (EtOH 0–10% mol) | ~0.2 mg/mg Rec | ~89% T.CAR | ~48% T.CHL | [69] | |||||
| Chlorococcum sp. | Dried, GR or wet biomass | T (60–80 °C), P (30 MPa), CO2 Flow (400 mL/min), t (80 min) | 7.1% Lipid | 1.4% FAME | [68] | ||||||
| Commercial DHA algae | Lyophilized or high-pressure ruptured | T (30–60 °C), P (10.5–30 MPa), CO2 Flow (20 mL/min), t (90–2700 min), Co-solv (EtOH, EtA, 1-Propanol 30:1–10:1) | T (30 °C), P (30 MPa), CO2 Flow (20 mL/min), t (2700 min), Co-solv (1-Propanol 30:1) | 90.56% | [70] | ||||||
| Crypthecodinium cohnii | LY | T (40–50 °C), P (20–30 MPa), CO2 Flow (0.6 kg/h), t (180 min) | 8.6% Lipid | 72% DHA Composition | [71] | ||||||
| Cylindrotheca closterium | Air-dried or LY | T (60 °C), P (40 MPa), CO2 Flow (0.41 kg/h) | 12.73% | [38] | |||||||
| Dunaliella salina | LY, homogenized | T (40–60 °C), P (10–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min), Co-solv (EtOH 0–5% mol) | T (60 °C), P (40 MPa), CO2 Flow (4.5 mmol/min) t (180 min), Co-solv (EtOH 5% mol) | 1.2% | [75] | ||||||
| LY | T (9.8–45.2 °C), P (18.5–44.2 MPa), t (100 min) | T (9.8 °C), P (31.4 MPa), t (100 min) | MBC 3.1 mg/mL E. coli, 3.9 mg/mL S. aureus MFC 8.3 mg/mL C. albicans, 30 mg/mL A. niger | [73] | |||||||
| LY | T (9.8–45.2 °C), P (18.5–44.2 MPa), t (100 min) | T (27.5 °C), P (44.2 MPa), t (100 min) | 6.58% | 7.199 mg T.CAR/100 mgextr, 3.751 mg β-CAR/100 mgextr | AO 0.452 mmol TE/gextr | [72] | |||||
| LY | T (40–60 °C), P (10–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min) | T (60 °C), P (40 MPa), CO2 Flow (4.5 mmol/min), t (180 min) OR T (60 °C), P (50 MPa), CO2 Flow (4.5 mmol/min), t (180 min) | 12.17 μg/mg or 9.3 μg/mg T.CAR | 0.227 μg/mg or 0.376 μg/mg T.CHL | [74] | ||||||
| LY | T (40–60 °C), P (10–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min), Co-solv (EtOH 5% mol) | T (60 °C), P (40 MPa), CO2 Flow (4.5 mmol/min), t (180 min), Co-solv (EtOH 5% mol) | 9.629 μg/mg T.CAR | 0.700 μg/mg T.CHL | [76] | ||||||
| Spray-dried | T (30–60 °C), P (10–50 MPa), CO2 Flow (3 L/min), t (90 min) | T (55 °C), P (40 MPa), CO2 Flow (3 L/min), t (90 min) | 115.44 μg/g T.CAR | 32.68 μg/g T.CHL | [77] | ||||||
| GR (in different conditions) | T (50–75 °C), P (10–55 MPa), CO2 Flow (7.24–14.48 g/min), t (30–110 min) | T (65 °C), P (14 MPa), CO2 Flow (14.48 g/min), t (110 min) OR T (75 °C), P (55 MPa), CO2 Flow (14.48 g/min), t (110 min) | 25.48% β-CAR Rec | 7.91 mg/g OR 8.47 mg/g Lipids | 95.88% OR 97.07% FAME Rec | [78] | |||||
| T (35–55 °C), P (20–30 MPa), t (180 min), Co-solv (EtOH/MeOH 0–5% w/w) | T (45 °C), P (20 MPa), t (180 min), Co-solv (EtOH 5% w/w) | 25 g/kg T.CAR | [79] | ||||||||
| Haematococcus pluvialis | T (40–80 °C), P (30–50 MPa), t (60–240 min) | T (90 °C), P (64.0 MPa), t (174 min) | 22.66 mg/g AST | [92] | |||||||
| Dried | T (40–80 °C), P (30–50 MPa), CO2 Flow (3 mL/min), t (60–240 min) | T (80 °C), P (50 MPa), CO2 Flow (3 mL/min), t (60 min) | 22.844 mg/g (83.05% Rec) OR 11.780 mg/g AST | AO (IC50) 2.37 mg/L OR 1.77 mg/L | [81] | ||||||
| Disrupted | T (40–70 °C), P (30–55 MPa), t (300 min), Co-solv (EtOH 0–8% v/v) | T (40 °C), P (55 MPa), t (300 min), Co-solv (EtOH 4.5% v/v) | 84% AST Rec | [82] | |||||||
| LY | T (30–80 °C), P (6.9–34.5 MPa), CO2 Flow (2–12 ΝL/min) t (20–100 min), Co-solv (EtOH/H2O 19.5–78 mL 0–99.5% v/v) | T (50 °C), P (31 MPa), CO2 Flow (6 ΝL/min) t (20 min), Co-solv (EtOH/H2O 9.23 mL/g 99.5% v/v) | 10.92 mg/L (73.9% Rec) AST | [83] | |||||||
| Dried | T (35–75 °C), P (30–50 MPa), CO2 Flow (10 L/h), t (210 min), Co-solv (EtOH 0.5–3.5 mL/g) | T (65 °C), P (43.5 MPa), CO2 Flow (10 L/h), t (210 min), Co-solv (EtOH 2.3 mL/g) | 87.42% AST | [89] | |||||||
| LY | T (45 °C), P (11.7–48.3 MPa), CO2 Flow (2.7 mL/min) t (240 min) | T (45 °C), P (48.3 MPa), CO2 Flow (2.7 mL/min), t (240 min) | 84.8% AST Rec | 85.3% Total TAG Rec | [85] | ||||||
| Crushed and/or GR | T (60 °C), P (30 MPa), Co-solv (EtOH 0–9.4% w/w) | T (60 °C), P (30 MPa), Co-solv (EtOH 9.4% w/w) | ~1.6% AST ~3% PHY | [28] | |||||||
| LY, crushed (3 degrees) | T (40–60 °C), P (20–30 MPa), Co-solv (EtOH 0–10%) | T (60 °C), P (30 MPa), Co-solv (EtOH 10%) | ~59–92% T.CAR Rec, ~76% β-CAR Rec, ~90% AST Rec | [86] | |||||||
| Dried | T (40–80 °C), P (20–55 MPa), CO2 Flow (2–4 mL/min), t (240 min), Co-solv (EtOH 0–7.5% v/v) | T (70 °C), P (40 MPa), CO2 Flow (3 mL/min), t (240 min), Co-solv (EtOH 5% v/v) | 80.6% AST Rec | [87] | |||||||
| Dried | T (50–80 °C), P (30–50 MPa), CO2 Flow (2–4 mL/min), t (300 min), Co-solv (EtOH/Soy bean oil/Olive oil 0–12% v/v) | T (70 °C), P (40 MPa), CO2 Flow (3 mL/min), t (300 min), Co-solv (Olive oil 10% v/v) | 51% AST | [88] | |||||||
| Disrupted, powdered or homogenized with water | T (40–70 °C), P (35–75 MPa), CO2 Flow (10 g/min) t (270–600 min) | T (70 °C), P (55 MPa), CO2 Flow (10 g/min) t (270 min) for powdered OR T (70 °C), P (45 MPa), CO2 Flow (10 g/min) t (600 min) for homogenized | 61% OR 54% AST Rec | [84] | |||||||
| T (40–70 °C), P (20–35 MPa), CO2 Flow (0.06 g/min), t (120 min), Co-solv (EtOH 0–13% w/w) | T (55 °C), P (20 MPa), CO2 Flow (0.06 g/min), t (120 min), Co-solv (EtOH 13% w/w) | 282.5 mg/g | 53.48 mg/g (82.3% Rec) AST | AO 0.243 mM TE/g | [90] | ||||||
| Ball-milled, HPR (H. Red Phase) | T (50–80 °C), P (10–55 MPa), CO2 Flow (3.62–14.48 g/min), t (20–120 min) | T (50 or 65 °C), P (55 MPa), CO2 Flow (3.62 g/min), t (120 min) | 237.4 mg/g | 19.72 mg/g (98.6% Rec) AST 4.03 mg/g (52.3% Rec) LUT | 21.41 mg/g Y, 93.2% Rec | [91] | |||||
| HPR (H. Red Phase) | T (50–80 °C), P (10–55 MPa), CO2 Flow (3.62 g/min), t (20–80 min), Co-solv (EtOH 0–1 mL/min) | T (65 °C), P (55 MPa), CO2 Flow (3.62 g/min), t (80 min), Co-solv (EtOH 1 mL/min) | 280.78 mg/g | 18.5 mg/g (~92% Rec) AST 7.15 mg/g (~93% Rec) LUT | [92] | ||||||
| Isochrysis galbana | LY | T (40–60 °C), P (10–30 MPa), CO2 Flow (5 L/min), t (60 min) | T (50 °C), P (30 MPa), CO2 Flow (5 L/min), t (60 min) | 5% | 16.2 mg/g T.CAR | 4.5 mg/g T.CHL | [94] | ||||
| Isochrysis sp. | LY and/or MW | T (45 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (120 min) Co-solv (EtOH 5%) | 15.5% | 9.3% Lipid Y | 61.9% Free FA Conversion | [93] | |||||
| Monoraphidium sp. | LY | T (30–60 °C), P (20 MPa), t (15–60 min), Co-solv (EtOH 0–20 mL) | T (60 °C), P (20 MPa), t (60 min), Co-solv (EtOH 20 mL) | 2.46 mg/g (101% Rec) AST | 29.5 mg/g (103% Rec) T. CHL | [95] | |||||
| Nannochloropsis gaditana | LY, homogenized | T (40–60 °C), P (10–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min) | T (60 °C), P (40 MPa), CO2 Flow (4.5 mmol/min), t (180 min) OR T (60 °C), P (20 MPa), CO2 Flow (4.5 mmol/min), t (180 min) | 0.343 μg/mg OR 0.125 μg/mg T.CAR | 2.238 μg/mg OR 0.090 μg/mg CHL-a | [96] | |||||
| LY | T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min) Co-solv (EtOH 5% mol) | T (60 °C), P (50 MPa), CO2 Flow (4.5 mmol/min), t (180 min) Co-solv (EtOH 5% mol) | 2.893 μg/mg T.CAR | 0.369 μg/mg CHL-a | [76] | ||||||
| LY, ASE | T (50–65 °C), P (25–55 MPa), CO2 Flow (7.24–14.48 g/min), t (100 min) | T (65 °C), P (25 MPa), CO2 Flow (7.24 - 14.48 g/min), t (100 min) | 77.68 mg/g | 34.15 mg/g Lipid Y | ~7.5 mg/g SFAs, ~8 mg/g MUFAs, ~10.5 mg/g PUFAs ~11.50 mg/g EPA | [98] | |||||
| LY and/or MW | T (45 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (120 min) Co-solv (EtOH 5%) | 12.9% | 7.9% Lipid Y | 61.2% Free FA Conversion | [93] | ||||||
| LY | T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min) Co-solv (EtOH 0–5% mol) | T (60 °C), P (50 MPa), CO2 Flow (4.5 mmol/min) t (180 min) Co-solv (EtOH 5% mol) | ~0.33% T.CAR | [75] | |||||||
| LY, High-pressure homogenized | T (55 °C), P (40 MPa), CO2 Flow (10 L/min), t (270 min) | 11.48% | 0.18 mg/g (8.3% Rec) VIO | [97] | |||||||
| N. granulata | LY, milled | T (50–90 °C), P (35–55 MPa), CO2 Flow (100 g/min), t (180–270 min) | T (70 °C), P (35 MPa), CO2 Flow (100 g/min), t (270 min) | 28.45 mg/g ash free biomass | 18.23 mg/g FAME | [99] | |||||
| LY | T (70–90 °C), P (35 MPa), CO2 Flow (100 g/min), t (270 min) | T (70 °C), P (35 MPa), CO2 Flow (100 g/min), t (270 min) | 165.9 g/kg Carbohydrates, 363.9 g/kg Sum of amino acids, 21.9 g/kg Non-protein | 256.3 g/kg Crude Lipid | [100] | ||||||
| N. oculata | LY, GR | T (50 °C), P (25–35 MPa), CO2 Flow (20 mL/min), Co-solv (EtOH, DCM, Toluene, n-Hexane) | T (50 °C), P (35 MPa), CO2 Flow (20 mL/min), Co-solv (EtOH) | 13.7 mg/gextr (63.2% Rec) ZEA | AO 1.612 mg/mL sample EC50, 0.313 mmol TE/g sample | [101] | |||||
| LY, homogenized | T (40–80 °C), P (20.7–62.1 MPa), CO2 Flow (24 mL/min), t (240 min) | T (40 °C), P (20.7 MPa), CO2 Flow (24 mL/min), t (240 min) | 47.30 mg/g | 10.36 mg/g Total TOC | Composition 35% T. SFA 45.31% T.MUFA 19.69% T.PUFA | [103] | |||||
| LY or air dried, crushed or GR | T (60 °C), P (30–85 MPa), CO2 Flow (0.5–100 kg/h), t (270 min) | T (60 °C), P (40 MPa), CO2 Flow (0.5 kg/h), t (270 min) | ~15% | Composition 93.82% Triglycerides 1.80% Sterol | 2.62% Free FA Comp. | [102] | |||||
| LY or air dried | T (60 °C), P (40 MPa), CO2 Flow (0.4–0.5 kg/h), t (120 min) | T (60 °C), P (40 MPa), CO2 Flow (0.5 kg/h), t (120 min) | ~12% | 1.76% Pigments Comp. | Composition 93.82% Triglycerides 1.80% Sterol | 2.62% Free FA Comp. | [38] | ||||
| N. gaditana | LY | T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min) Co-solv (EtOH 0–5% mol) | T (60 °C), P (50 MPa), CO2 Flow (4.5 mmol/min) t (180 min) Co-solv (EtOH 5% mol) | ~0.33% T.CAR | [75] | ||||||
| LY, High-pressure homogenized | T (55 °C), P (40 MPa), CO2 Flow (10 L/min), t (270 min) | 11.48% | 0.18 mg/g (8.3% Rec) VIO | [97] | |||||||
| N. granulata | LY, milled | T (50–90 °C), P (35–55 MPa), CO2 Flow (100 g/min), t (180–270 min) | T (70 °C), P (35 MPa), CO2 Flow (100 g/min), t (270 min) | 28.45 mg/g ash free biomass | 18.23 mg/g FAME | [99] | |||||
| LY | T (70–90 °C), P (35 MPa), CO2 Flow (100 g/min), t (270 min) | T (70 °C), P (35 MPa), CO2 Flow (100 g/min), t (270 min) | 165.9 g/kg Carbohydrates, 363.9 g/kg Sum of amino acids, 21.9 g/kg Non-protein | 256.3 g/kg Crude Lipid | [100] | ||||||
| N. oculata | LY, GR | T (50 °C), P (25–35 MPa), CO2 Flow (20 mL/min), Co-solv (EtOH, DCM, Toluene, n-Hexane) | T (50 °C), P (35 MPa), CO2 Flow (20 mL/min), Co-solv (EtOH) | 13.7 mg/gextr (63.2% Rec) ZEA | AO 1.612 mg/mL sample EC50, 0.313 mmol TE/g sample | [101] | |||||
| LY, homogenized | T (40–80 °C), P (20.7–62.1 MPa), CO2 Flow (24 mL/min), t (240 min) | T (40 °C), P (20.7 MPa), CO2 Flow (24 mL/min), t (240 min) | 47.30 mg/g | 10.36 mg/g Total TOC | Composition 35% T. SFA 45.31% T.MUFA 19.69% T.PUFA | [103] | |||||
| LY or air dried, crushed or GR | T (60 °C), P (30–85 MPa), CO2 Flow (0.5–100 kg/h), t (270 min) | T (60 °C), P (40 MPa), CO2 Flow (0.5 kg/h), t (270 min) | ~15% | Composition 93.82% Triglycerides 1.80% Sterol | 2.62% Free FA Comp. | [102] | |||||
| LY or air dried | T (60 °C), P (40 MPa), CO2 Flow (0.4–0.5 kg/h), t (120 min) | T (60 °C), P (40 MPa), CO2 Flow (0.5 kg/h), t (120 min) | ~12% | 1.76% Pigments Comp. | Composition 93.82% Triglycerides 1.80% Sterol | 2.62% Free FA Comp. | [38] | ||||
| N. salina | T (60 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (90 min), Co-solv (EtOH 5%) | ~30% | [44] | ||||||||
| Nannochloropsis sp. | LY, GR | T (40–55 °C), P (40–70 MPa), CO2 Flow (10 kg/h), t (360 min) | T (55 °C), P (40 MPa), CO2 Flow (10 kg/h), t (360 min) | ~257 mg/g Lipid | Composition 25.3% SFA 20.1% Monoenoic 54.6% PUFA 44% n-3 PUFAs | [104] | |||||
| Dried, milled | T (40–60 °C), P (12.5–30 MPa), CO2 Flow (0.35–0.62 g/min), t (60–105 min), Co-solv (EtOH 0–20% w/w) | T (40 °C), P (30 MPa), CO2 Flow (0.62 g/min), Co-solv (EtOH 20% w/w) | Composition: 13.71% AST 22.35% LUT, 13.20% VIO/NEO, 34.30% VAU, 4.71% CAN, 5.06% β-CAR | ~1 mg/g Pigment Rec | 45% Lipid Y | [105] | |||||
| Bead milled | T (50–75 °C), P (10–55 MPa), CO2 Flow (7.24–14.48 g/min), t (100 min) | T (75 °C), P (55 MPa), CO2 Flow (14.48 g/min), t (100 min) OR T (50 °C), P (40 MPa), CO2 Flow (14.48 g/min), t (100 min) | 94.28 mg/g OR 58.26 mg/g | 18.39 mg/g OR 10.37 mg/g Lipid Y | 5.69 mg/g (15.59% Rec) EPA OR 0.12 mg/g (79.63% Rec) DHA | [106] | |||||
| Ochromonas danica | LY | T (40 °C), P (17.2–31 MPa), t (~240 min) | T (40 °C), P (17.2 MPa), t (~240 min) | 234.2 mg/g Lipid Y | [107] | ||||||
| Pavlova sp. | Bead milled | T (45 °C), P (30.6 MPa), t (360 min) | 17.9% | 15.7% (98.7% Rec) FAME | [108] | ||||||
| Phaeodactylum tricornutum | MW with DES | T (45 °C), P (30.6 MPa), CO2 Flow (2.5 L/min), t (360 min) | 7.1% Lipid Y | 7.0% TFA Y, 1.0% EPA Y, 2.0% PUFA Y | [109] | ||||||
| Phormidium valderianum | T (35.86–64.14 °C), P (13.79–56.21 MPa), CO2 Flow (2 L/min), t (90 min) | T (50 °C), P (50 MPa), CO2 Flow (2 L/min), t (90 min) | 3.96 mg/g | 13.43 μg β-CAR eq. /g T.CAR | 1.41 mg/g Anatoxin-a | 2596.57 μg BHT eq./g Reducing Power, 5.29 mM FeSO4 eq./g FRAP value, 0.38 mg/mL IC50 TPC 94.87 μg GAE/g | [110] | ||||
| Scenedesmus almeriansis | LY, milled, and/or bead milled with alumina A | T (32–60 °C), P (20–60 MPa), CO2 Flow (1 g/min), t (300 min) | T (60 °C), P (40 MPa), CO2 Flow (1 g/min), t (300 min) | 0.0466 mg/g LUT 1.50 mg/g β-CAR | [111] | ||||||
| LY and matrix solid-phase dispersion | T (50–65 °C), P (25–55 MPa), CO2 Flow (7.24–14.48 g/min), t (120 min) | T (65 °C), P (55 MPa), CO2 Flow (14.48 g/min), t (120 min) | 8.74 mg/g | 2.97 mg/g (17% Rec) LUT | 3.42 mg/g Lipid Y | 15% FA Rec | [112] | ||||
| LY and/or MW | T (45 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (90 min), Co-solv (EtOH 5% v/v) | 13.2% | 10.1% Lipid Y | 76.5% Free FA Conversion | [93] | ||||||
| S. dimorphus | LY and/or MW, sonicated and bead milled | T (50–100 °C), P (16.6–50 MPa), t (60 min) | T (100 °C), P (41.4 MPa), t (60 min) | 98.8% FAME Rec | [113] | ||||||
| S. obliquus | LY and/or high-pressure homogenized | T (40–60 °C), P (10–40 MPa), CO2 Flow (7 L/min), t (120 min) | T (50 °C), P (36 MPa), CO2 Flow (7 L/min), t (120 min) | 0.97% | 35.85 mg/gextr T.CAR | 11.03 mg/gextr T.CHL | [114] | ||||
| LY | T (20–200 °C), P (7–80 MPa), t (540 min) | T (20 °C), P (120 MPa), t (540 min) | 6.4% | 92% Lipid Rec | 59% PUFA Conc. | [2] | |||||
| Dried | T (45–65 °C), P (15–30 MPa), CO2 Flow (0.4 kg/h), t (30–90 min), Co-solv (EtOH 5% v/v) | T (60 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (30 min), Co-solv (EtOH 5% v/v) OR T (65 oC), P (30 MPa), CO2 Flow (0. kg/h), t (90 min), Co-solv (EtOH 5% v/v) | 24.67% | 18.15% Lipid Y | 73.57% Free FA Conv. 33.76% Ω-3, 23.63% Ω-6, 26.71% SFA, 22.00% MUFA, 51.28% PUFA | [44] | |||||
| LY, homogenized | T (40–60 °C), P (15–25 MPa), CO2 Flow (2–4.3 g/min), t (240 min), Co-solv (EtOH 0–9.5% v/v) | T (60 °C), P (25 MPa), CO2 Flow (2 g/min), t (240 min) Co-solv (EtOH 0% v/v) | 0.182 mg/g T.CAR | 0.016 mg/g CHL-a, 0.016 mg/g CHL-b, 0.011 mg/g CHL-c | [116] | ||||||
| LY, protein concentrate | T (40 °C), P (37.9 MPa), CO2 Flow (3 sL/min), Co-solv (EtOH 0–15% v/v) | T (40 °C), P (37.9 MPa), CO2 Flow (3 sL/min), Co-solv (EtOH 15% v/v) | Composition 12.48% Lipid 67.89% Neutral Lipids 22.52% Glycolipids 9.59% Phospholipids | [115] | |||||||
| S. obtusiusculus | LY | T (20 °C), P (12 MPa), t (540 min) | 6.4% | 42.52% FA Y | [2] | ||||||
| Scenedesmus sp. | LY, GR | T (35–65 °C), P (20–50 MPa), CO2 Flow (1.38–4.02 g/min) | T (53 °C), P (50 MPa), CO2 Flow (1.9 g/min) | 7.06% | 7.41% Lipid Y | [120] | |||||
| LY | T (35–50 °C), P (40 MPa), t (120–360 min), Co-solv (MeOH) | T (35 °C), P (40 MPa) t (360 min), Co-solv (MeOH) | 19.32% Lipid Y | [121] | |||||||
| LY | T (35–80 °C), P (20–40 MPa), CO2 Flow (750–800 mL/min), t (60 min), Co-solv (MeOH, EtOH, Propanol, Butanol, ACE 0–40% mol) | T (70 °C), P (40 MPa), CO2 Flow (750–800 mL/min) t (60 min), Co-solv (EtOH 30% mol) | 2.210 mg/g (76.7% Rec) LUT | [118] | |||||||
| LY, GR | T (60 °C), P (30 MPa), CO2 Flow (2 mL/min), t (60 min), Co-solv (EtOH 0–10% mol) | T (60 °C), P (30 MPa), CO2 Flow (2 mL/min), t (60 min), Co-solv (EtOH 10% mol) | 72.9 μg/g AST 436.1 μg/g LUT 59.9 μg/g β-CAR 670.8 μg/g NEO 89.6 μg/g ZEA | [119] | |||||||
| T (40 °C), P (35 MPa), CO2 Flow (800 mL/min), t (60 min), Co-solv (MeOH/Water 90:10 v/v, 0.3 mL) | 0.96 ng/g Daidzin, 4.91 ng/g Genistin, 9.14 ng/g Ononin, 10.6 ng/g Daidzein, 3.82 ng/g Sissotrin, 6.11 ng/g Genistein 5.92 ng/g Formononetin, 6.8 ng/g Biochanin A | [117] | |||||||||
| Skeletonema costatum | LY | T (40 °C), P (17.2–31 MPa) t (240 min) | T (40 °C), P (24 MPa) t (240 min) | ~65 mg/g | [107] | ||||||
| Synechococcus sp. | LY and homogenized | T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min), Co-solv (EtOH 0–5% mol) | [75] | ||||||||
| T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min) | T (50 °C), P (30 MPa), CO2 Flow (4.5 mmol/min) t (180 min) | 1.511 μg/mg T.CAR | 0.078 μg/mg T.CHL | [123] | |||||||
| LY | T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min), Co-solv (EtOH 5% mol) | T (50 °C), P (30 MPa), CO2 Flow (4.5 mmol/min), t (180 min), Co-solv (EtOH 5% mol) | 1.86 μg/mg T.CAR | 0.286 μg/mg T.CHL | [76] | ||||||
| LY | T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min) t (240 min), Co-solv (EtOH 15% mol) | CO2 Flow (4.5 mmol/min) t (240 min), Co-solv (EtOH 15% mol) T (50 °C), P (35.8 MPa), OR T (59 °C), P (45.4 MPa), OR T (60 °C), P (50.0 MPa), | 71.6% β-CAR max. Rec 90.3% β-CRY max. Rec 36.4% ZEA max. Rec | [122] | |||||||
| LY and homogenized | T (40–60 °C), P (20–40 MPa), CO2 Flow (0.8 g/min), t (180 min), Co-solv (EtOH 0–5% mol) | T (40 °C), P (40 MPa), CO2 Flow (0.8 g/min), t (180 min), Co-solv (EtOH 5% mol) | 20.35 mg/gextr β-CAR 25.96 mg/gextr ZEA | 193.75 mg/gextr PA, 5.3 mg/gextr PLA 71.96 mg/gextr STA, 4.13 mg/gextr OA, 94.66 mg/gextr LNA, 2.95 mg/gextr GLA | [17] | ||||||
| T. chui | Dried and GR | T (40–60 °C), P (18–25 MPa), CO2 Flow (2 mL/min), t (60–90 min), Co-solv (EtOH, MeOH) | T (40 °C), P (18 MPa), CO2 Flow (2 mL/min), t (60–90 min), Co-solv (MeOH) | 4.3% | [124] | ||||||
| Tetraselmis sp. | LY and/or MW | T (45 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (90 min), Co-solv (EtOH 5%) | 14.8% | 11.1% Lipid Y | [93] | ||||||
| LY | T (40 °C), P (15 MPa), CO2 Flow (5 mL/min), t (30 min), Co-solv (EtOH 5%) | 10.88% Lipid Y | [125] |
| Algae | Parametric Investigation | Ext. Yield/ Recovery | Kinetic Model | Experimental Design | Other Extraction Methods | Results | Ref. |
|---|---|---|---|---|---|---|---|
| A. maxima | T (50–60 °C), P (25–35 MPa), Co-solv (EtOH 0–10% v/v) | B-D | Total Lipids Determination | [24] | |||
| Hexane MAC ( T = 25 °C, t = 2 h, stirring = 100 rpm) | 2.6% wt lipid/biomass 0.01% wt GLA/biomass | ||||||
| EtOH MAC ( T = 25 °C, t = 2 h, stirring = 100 rpm) | 5.7% wt lipid/biomass 0.68% wt GLA/biomass | ||||||
| ACE MAC ( T = 25 °C, t = 2 h, stirring = 100 rpm) | 4.7% wt lipid/biomass 0.63% wt GLA/biomass | ||||||
| T (50–60 °C), P (25–35 MPa), CO2 Flow (2 g/min), t (390 min), Co-solv (EtOH 0–10% v/v) | internal mass transfer | Lepage and Roy | 1.23% wt GLA/biomass | [25,26] | |||
| B-D | 7.8% wt lipid/biomass 0.98% wt GLA/biomass | ||||||
| Hexane MAC ( T = 25 °C, t = 2 h, stirring = 100 rpm) | 2.6% wt lipid/biomass 0.01% wt GLA/biomass | ||||||
| EtOH MAC ( T = 25 °C, t = 2 h, stirring = 100 rpm) | 5.7% wt lipid/biomass 0.68% wt GLA/biomass | ||||||
| ACE MAC ( T = 25 °C, t = 2 h, stirring = 100 rpm) | 4.7% wt lipid/biomass 0.63% wt GLA/biomass | ||||||
| T (20–70 °C), P (15–18 MPa), CO2 Flow (3.33 × 10–5 kg/s), t (660 min) | Goto et al.-LDF | Two-level factorial design | [27] | ||||
| A. pacifica | T (40–80 °C), P (15–35 MPa), CO2 Flow (2 mL/min), t (40–100 min), Co-solv (EtOH 5–15% v/v) | Two-level factorial design | Tetrahydrofuran/MeOH MAC | 50 mg/100 g ZEA, 8 mg/100 g β-CRY, 120 mg/100 g β-CAR | [29] | ||
| A. platensis | T (45–60 °C), P (15–45 MPa), CO2Flow (0.015 kg/h), t (50 min), Co-solv (EtOH 26.70–53.22% v/v) | 4.07% | Two-level factorial design | MAE with MeOH/EtA/light petroleum (1:1:1 v/v/v) (T = 50 °C, W = 40 W) | 2.03% Y, 2.46 μg/g TOCs, 629 μg/g T.CAR, 15.88 mg/g FAs | [30] | |
| T (32–48 °C), P (20–40 MPa), t (120–240 min), Co-solv (EtOH) | 10.26 g/kg | RSM, Box-Behnken design | [35] | ||||
| T (33.18–66.82 °C), P (23.2–56.8 MPa), CO2 Flow (0.24–0.9 kg/h), t (0–120 min soaking and 30–180 min Extr.) Co-solv (MeOH, ACE, EtA 0–10 mL, Aq.EtOH (20–80%) 5–28.4 mL) | RSM, CCD | [37] | |||||
| T (40–80 °C), P (10–30 MPa), t (30–90 min), Co-solv (EtOH 10–50% v/v) | 6.7% w/w | Taguchi’s orthogonal array | PLE (T = 60–180 °C, P = 3.4–20.7 MPa, t = 5–15 min, ethyl lactate 0–100% v/v) | 20.7% Y, 68.3% GLA Rec (in optimal conditions) | [31] | ||
| T (40 °C), P (31.6–48.4 MPa), CO2 Flow (0.7 L/min), t (26.4–94 min), Co-solv (EtOH 9.64–16.36 mL) | RSM, CCD | B-D (UAE) | for GLA Rec | [32] | |||
| MeOH/acetyl chloride MAC (T = 80 °C, 1 h) | for GLA Rec | ||||||
| T (60 °C), P (40 MPa), CO2 Flow (0.35 kg/h) | 10.98% | Sovová | [38] | ||||
| T (40–55 °C), P (25–70 MPa), CO2 Flow (10 kg/h), t (90–240 min) | 7.79% Lipid | Andrich et al. | Hexane MAC (t = 8 h) | 7.77% Lipid Y | [39] | ||
| B. braunii | T (40 °C), P (12.5–30 MPa) | Hexane MAC | ~76 g/kg Hydrocarbons | [24,40] | |||
| T (50–80 °C), P (20–25 MPa), t (10–150 min) | ~10.5% | B-D | 18.2% FA Y | [41] | |||
| C. protothecoides | T (50 °C), P (35 MPa), CO2 Flow (0.0439 kg/h), t (180 min) | 0.23 g/gbiom lipid 75% Rec | Goto et al. | SX (n-Hexane, t = 24 h) | 0.32 g/g Lipid Y | [43] | |
| T (60 °C), P (30 MPa), CO2 Flow (30 g/h), t (90 min), Co-solv (EtOH 5%) | 10% Lipid | Sovová & Semiemperical solubility models | [44] | ||||
| C. pyrenoidosa | T (32–55 °C), P (25–40 MPa), CO2 Flow (15–30 kg/h), t (1.5–180 min), Co-solv (EtOH 0–1.5 mL/gbiom) | 7.78% | Orthogonal design (L1645) | [46] | |||
| C. saccharophila | T (42–73 °C), P (24.1–41.4 MPa), t (30–90 min) | RSM, Box-Behnken design | [48] | ||||
| C. sorokiniana | T (40–60 °C), P (10–30 MPa), t (180 min), Co-solv (EtOH 0–10%) | 35.03 mg/g | RSM, CCD | EtA and MeOH MAC | 0.215 mg/g VIO Y 2.797 mg/g LUT Y 0.756 mg/g Carotene Y | [49] | |
| Chlorella sp. | T (40–60 °C) P (15–30 MPa), CO2 Flow (15 g/min), t (180 min), Co-solv (Hexane/MeOH 1–3 v/v) | 47.2% | RSM, Box-Behnken design | [51] | |||
| T (60 °C), P (20–30 MPa), CO2 Flow (30 g/h), t (180 min), Co-solv (EtOH 0–5%) | B-D | 15.2% Y | [52] | ||||
| C. vulgaris | T (60–80 °C), P (20–50 MPa), CO2 Flow (2.5 mL/min), t (3–6 h), Co-solv (EtOH or ACE 7.5% v/v) | SX (EtOH, t = 5 h) | 2 mg/g Extr LUT Y, 18 mg/g Extr CHL Y | [59] | |||
| T (40–55 °C), P (15–35 MPa), CO2 Flow (0.4 dm3/min), t (125–480) | B-D | 24.5% Lipid Y | [61] | ||||
| n-hexane MAC (t = 72 h) | 0.03% Y | ||||||
| ACE MAC (t = 72 h) | 0.04% Y | ||||||
| T (40 °C), P (12.5–30 MPa), CO2 Flow (0.04 kg/h) | ACE MAC | 0.43% T.CAR Y | [40] | ||||
| T (50 °C), P (31 MPa), CO2 Flow (6 NL/min), t (20 min), Co-solv (Aq. EtOH (50%) 50 mL) | 8.71% | UAE (0.5 g algae with 60 mL 50% aqueous EtOH, t = 15 h) | 9.73% Y, 0.46 mg GAE/g Extr, 0.86 mg quercetin/g Extr | [65] | |||
| T (40–60 °C), P (27.6–48.3 MPa), CO2 Flow (1–3 g/min), t (1–180 min) | 17.7% | BICM, LDF, shrinking core model, BICM + shrinking core model | RSM, CCD | SX (n-hexane, t = 14 h) | 18% Y | [55] | |
| T (40–80 °C), P (27.6–62.1 MPa), t (180 min) | 19% > 99% Rec | SX (n-hexane, t = 12 h) | 18% Y | [56] | |||
| T (40–70 °C), P (20–28 MPa), CO2 Flow (10 kg/h), t (9 h) | 4.86% | RSM, CCD | [57] | ||||
| T (40 °C), P (30 MPa), CO2 Flow (0.34–0.6 L/min) Co-solv (EtOH or oil) | Soybean oil MAC (T= ambient, t = 17 h or T= 100 °C, t = 30 min) | 0.438% or 0.306% Y, 100% or 70.9% Rec | [58] | ||||
| ACE MAC | 0.426% Y 100% Rec | ||||||
| T (45 °C), P (45 MPa), CO2 Flow (25 g/min) | ~14 % | Sovová | [38] | ||||
| T (50 °C), P (25 MPa), CO2 Flow (0.5 kg/h), t (210–230 min) Co-solv (EtOH 0–10% v/v) | ~40% | SX (CHF/MeOH 35:65 v/v, t = 18 h) | 0.244 g/g Total Lipid Y 26% Neutral Lipid Rec 59% Glycolipid Rec 15% Phospholipid Rec | [63] | |||
| Chlorococcum sp. | T (60–80 °C), P (30 MPa), CO2 Flow (400 mL/min), t (80 min) | 7.1% Lipid | Ozkal et al. | Hexane MAC (t = 7.5 h, T = ambient) | 1.5% Lipid Y | [68] | |
| Hexane and hexane/isopropanol (3:2) MAC (t = 7.5 h, T = ambient) | 1.0% Lipid Y | ||||||
| SX (Hexane, t = 7.5 h) | 3.2% Lipid Y | ||||||
| Commercial DHA algae | T (30–60 °C), P (10.5–30 MPa), CO2 Flow (20 mL/min), t (90–2700 min), Co-solv (EtOH, EtA, 1-Propanol 30:1–10:1) | 90.56% | UAE (0.9 g algae, 48 mL EtA + 24 mL MeOH, T = 80 °C, t = 3 h) | for total lipid determination | [70] | ||
| Crypthecodinium cohnii | T (40–50 °C), P (20–30 MPa), CO2 Flow (0.6 kg/h), t (180 min) | 8.6% Lipid | B-D | 19.9% Lipid Y | [71] | ||
| Cylindrotheca closterium | T (60 °C), P (40 MPa), CO2 Flow (0.41 kg/h) | 12.73% | Sovová | [38] | |||
| D. salina | T (40–60 °C), P (10–50 MPa), CO2 Flow(4.5 mmol/min), t (180 min), Co-solv (EtOH 0–5% mol) | 1.2% | Reverchon et al. | [75] | |||
| T (9.8–45.2 °C), P (18.5–44.2 MPa), t (100 min) | CCRD | [73] | |||||
| T (9.8–45.2 °C), P (18.5–44.2 MPa), t (100 min) | 6.58% | CCRD | [72] | ||||
| T (40–60 °C), P (10–50 MPa), CO2 Flow (4.5 mmol/min) t (180 min) | Multi-level factorial design | UAE (0.105 g algae in 5 mL DMF, t = 3 min) | 27.7 μg T.CAR/mg, 3.1 μg CHL/mg | [74] | |||
| UAE (0.105 g algae in 5 mL MeOH, t = 3 min) | 14.1 μg T.CAR/mg, 2.5 μg CHL/mg | ||||||
| T (40–60 °C), P (10–50 MPa), CO2 Flow (4.5 mmol/min) t (180 min), Co-solv (EtOH 5% mol) | Multi-level factorial design | UAE (t = 3 min, 5 mL MeOH, 0.025 g biomass) | 14.1 μg/mg T.CAR, 2.5 μg/mg Total CHL | [76] | |||
| UAE (t = 3 min, 5 mL DMF, 0.025 g biomass) | 27.7 μg/mg T.CAR, 3.1 μg/mg Total CHL | ||||||
| T (30–60 °C), P (10–50 MPa), CO2 Flow (3 L/min), t (90 min) | RSM | MeOH MAC (t = 8 h, 2 g biomass, 150 mL) | 245.74 μg/g T.CAR, 917.96 μg/g Total CHL | [77] | |||
| H. pluvialis | T (40–80 °C), P (30–50 MPa), t (60–240 min) | RSM, CCD | [92] | ||||
| T (40–80 °C), P (30–50 MPa), CO2 Flow (3 mL/min), t (60–240 min) | RSM, CCD | SX (ACE 250 mL, 0.5 g biomass, t = 6 h) | for total AST determination (27.46 mg/g) | [81] | |||
| T (40–70 °C), P (30–55 MPa), t (300 min), Co-solv (EtOH 0–8% v/v) | Sovová | Two-level factorial design | [82] | ||||
| T (30–80 °C), P (6.9–34.5 MPa), CO2 Flow (2–12 ΝL/min) t (20–100 min), Co-solv (EtOH/Water 19.5–78 mL 0–99.5% v/v) | Design with 7 factors | SX (DCM 200 mL, 1.0 g biomass, T = 45 °C) | for total AST determination | [83] | |||
| T (40–70 °C), P (35–75 MPa), CO2 Flow (10 g/min) t (270–600 min) | ACE MAC (multiple circles) | for total AST determination | [84] | ||||
| T (45 °C), P (11.7–48.3 MPa), CO2 Flow (2.7 mL/min) t (240 min) | B-D | for total TAG (366.3 mg for GR and 468.3 mg for homogenized biomass) | [85] | ||||
| ACE MAC | for total AST (41.4 mg for GR and 71.0 mg for homogenized biomass) | ||||||
| T (40–60 °C), P (20–30 MPa), Co-solv (EtOH 0–10%) | ACE MAC | for T.CAR determination (1.80% Y, 3.3% LUT, 2.2% CAN, 7.2% β-CAR, 75.0% Total AST) | [86] | ||||
| T (40–80 °C), P (20–55 MPa), CO2 Flow (2–4 mL/min), t (240 min), Co-solv(EtOH 0–7.5% v/v) | SX (DCM 200 mL, 6 g biomass, t = 6 h) | for total AST Rec (3.43% AST Y) | [87] | ||||
| T (50–80 °C), P (30–50 MPa), CO2 Flow (2–4 mL/min), t (300 min), Co-solv (EtOH/Soy bean oil/Olive oil 0–12% v/v) | SX (DCM 200 mL, 1 g biomass, t = 2 h) | for total AST Rec | [88] | ||||
| T (40–70 °C), P (20–35 MPa), CO2 Flow (0.06 g/min), t (120 min), Co-solv (EtOH 0–13% w/w) | 282.5 mg/g | RSM, Box-Behnken design | CO2 - Expanded EtOH (30–60 °C, EtOH 50–70% w/w, 7 MPa) | 333.1 mg/g Y, 62.57 mg/g AST Content, 124.2% w/w AST Rec, 0.233 mM TE/g | [90] | ||
| I. galbana | T (40–60 °C), P (10–30 MPa), CO2 Flow (5 L/min), t (60 min) | 5% | Factorial design | Reyes (ACE/BHT (99.9:0.01) 20 mL, t = 24 h, 200 mg biomass) | for total extr. compounds determination | [94] | |
| GXL ( T = 50 °C, P = 7 MPa, EtOH 15–75%) | as stage 2 - for enhanced CAR and CHL extr. | ||||||
| EtOH MAC ( T = 80 °C, P = 10 MPa) | as stage 3 - for mid- and highly-polar lipids, proteins and sugars extr. | ||||||
| Water MAC ( T = 80 °C, P = 10 MPa) | as stage 4 - for protein and sugars extr. | ||||||
| Isochrysis sp. | T (45 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (120 min) Co-solv (EtOH 5%) | 15.5% | Sovová | SX (MeOH/CHF (2:1), t = 18 h, T = 105 °C) | 23.1% Y, 31.2% Free FA Conversion, 7.2% Lipid Y | [93] | |
| Kochert (MeOH/CHF (2:1), t = 1 h, T = 45 °C) | 12.7% Y | ||||||
| Monoraphidium sp. | T (30–60 °C), P (20 MPa), t (15–60 min), Co-solv (EtOH 0–20 mL) | Bead beater method (BBM) (ACE/hexane (35:65) 500 μL, 30 mg biomass) | 2.44 mg/g AST, 100% AST Rec, 27.6 mg/g Total CHL, 100% Total CHL Rec | [95] | |||
| EtOH MAC (20 mL, 1 g biomass, t = 30 min) | 1.16 mg/g AST, 48% AST Rec, 16.1 mg/g Total CHL, 56% Total CHL Rec | ||||||
| N. gaditana | T (40–60 °C), P (10–50 MPa), CO2 Flow (4.5 mmol/min) t (180 min) | Multilevel factorial Design | UAE MeOH (5 mL, 0.2 g biomass, t = 10 min, T = 4 °C, t = 24 h) | 0.8 μg/mg T.CAR Y 18.5 μg/mg CHL-a Y | [96] | ||
| T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min)t (180 min) Co-solv (EtOH 5% mol) | Multilevel factorial design | UAE MeOH (5 mL, 0.2 g biomass, t = 10 min, T = 4 °C, t = 24 h) | 2.2 μg/mg T.CAR Y, 26.4 μg/mg T.CHL Y | [76] | |||
| UAE DMF (5 mL, 0.2 g biomass, t = 10 min, T = 4 °C, t = 24 h) | 6.9 μg/mg T.CAR Y 41.5 μg/mg T.CHL Y | ||||||
| T (45 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (120 min) Co-solv (EtOH 5%) | 12.9% | Sovová | SX (MeOH/CHF (2:1), t = 18 h, T = 105 °C) | 23.1% Y, 31.2% Free FA Conversion, 7.2% Lipid Y | [93] | ||
| Kochert (MeOH/CHF (2:1), t = 1 h, T = 45 °C) | 12.7% Y | ||||||
| T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min) t (180 min) Co-solv (EtOH 0–5% mol) | Reverchon et al. | [75] | |||||
| T (55 °C), P (40 MPa), CO2 Flow (10 L/min), t (270 min) | 11.48% | Sovová | PLE (water or EtOH/water (1:1) or EtOH, T = 40–170 °C, t = 20 min) | 37.71% Y, 9.04 mg/g Extr T.CAR Y, 69.14% Lipid Y, 59.85 mg GAE/g Extr, 0.8 mmol TE/g Extr (optimum conditions) | [97] | ||
| ACE MAC (t = 24 h) | for total VIO determination | ||||||
| N. granulata | T (50–90 °C), P (35–55 MPa), CO2 Flow (100 g/min), t (180–270 min) | 28.45 mg/g ash free biomass | SX (hexane, 0.5 g biomass, t = 1 h) | 57.34 mg/g Y, 17.35 mg/g FAME | [99] | ||
| N. oculata | T (50 °C), P (25–35 MPa), CO2 Flow (20 mL/min), Co-solv (EtOH, DCM, Toluene, n-Hexane) | SX (hexane 300 mL, 10 g biomass, t = 16 h) | 5.79% Y, 56.3% CAR Rec | [101] | |||
| SX (EtOH 300 mL, 10 g biomass, t = 16 h) | 40.90% Y, 70.3% CAR Rec | ||||||
| SX (DCM 300 mL, 10 g biomass, t = 16 h) | 9% Y, 100% CAR Rec | ||||||
| T (40–80 °C), P (20.7–62.1 MPa), CO2 Flow (24 mL/min), t (240 min) | 47.30 mg/g | Chen method (hexane 1 mL, 5 mg biomass) | for TOC Rec 4.722 mg/gextr, 163 mg/g Y | [103] | |||
| Cequier-Sanchez method (DCM/MeOH) | 665.33 mg/g Y, Composition 74.63 mg/g Total SFA 23.41 mg/g Total MUFA 1.96 mg/g Total PUFA | ||||||
| T (60 °C), P (30–85 MPa), CO2 Flow (0.5–100 kg/h), t (270 min) | ~15% | B-D | Composition 0.71% Free FA 72.13% Triglycerides 4.58% Sterol | [102] | |||
| T (60 °C), P (40 MPa), CO2 Flow (0.4–0.5 kg/h), t (120 min) | ~12% | Sovová | [38] | ||||
| N. salina | T (60 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (90 min), Co-solv (EtOH 5%) | ~30% | Sovová | [44] | |||
| Nannochloropsis sp. | T (40–55 °C), P (40–70 MPa), CO2 Flow (10 kg/h), t (360 min) | ~257 mg/g Lipid | SX (hexane, t = 6 h) | 237 mg/g Lipid Y, 25.6% SFA Comp., 21.9% Monoenoic Comp., 52.2% PUFA Comp., 42.6% n-3 PUFAs Comp. | [104] | ||
| T (50–75 °C), P (10–55 MPa), CO2 Flow(7.2–14.5 g/min) t (100 min) | 94.28 mg/g OR 58.26 mg/g | B-D | for total lipid determination | [106] | |||
| T (40–60 °C), P (12.5–30 MPa), CO2 Flow(0.35–0.62 g/min) t (60–105 min), Co-solv (EtOH 0–20% w/w) | B-D method (MeOH/CHF/H2 O (10:5:4 v/v/v), 150 mg biom., t = 24 h) | 25.3% Lipid Y | [105] | ||||
| SX (hexane, 1 g biom., t = 6 h) | 40.7% Lipid Y | ||||||
| SX (EtOH, 1 g biom., t = 6 h) | 50.6% Lipid Y | ||||||
| EtA MAC (19 mL, 1 g biom., t = 24 min, T = 65 °C) | for T.CAR determination | ||||||
| EtA or ACE MAC (2 mL, 5 g biom., t = 10 min, T =−22 °C) | for T.CAR determination | ||||||
| Pavlova sp. | T (45 °C), P (30.6 MPa), t (360 min) | 17.9% | UAE (10 mL water/24 mL MeOH/48 mL EtA, 10 g biom., t = 3 h) | 44.7% Y, 15.6% (98.1% Rec) FAME | [108] | ||
| SX (hexane 450 mL, 2 g biom., t = 15 h) | 13.5% Y, 7.2% (45.2% Rec) FAME | ||||||
| SX (hexane 450 mL, 2 g biomass, t = 100 h) | 18.5% Y, 9.8% (61.6% Rec) FAME | ||||||
| SX (hexane 450 mL, 2 g biom., t = 15 h, bead milled) | 15.3% Y, 9.3% (58.5% Rec) FAME | ||||||
| Phaeodactylum tricornutum | T (45 °C), P (30.6 MPa), CO2 Flow (2.5 L/min), t (360 min) | B-D method (3 mL MeOH/CHF 1:2 v/v, 100 mg biom., t = 2 h, T = 50 °C) | 31.3% Lipid Y, 11.1% TFA Y, 2.0% EPA Y, 4.4% PUFA Y | [109] | |||
| DMC MAC (3 mL, 100 mg biom., t = 2 h, T = 50 °C) | 11.3% Lipid Y, 4.5% TFA Y, 1.1% EPA Y, 2.6% PUFA Y | ||||||
| DMC MAC (3 mL, 100 mg biom., t = 2 h, T = 50 °C, DES pretreated) | 14.1% Lipid Y, 8.1% TFA Y, 1.6% EPA Y, 3.6% PUFA Y | ||||||
| DMC MAC (3 mL, 100 mg biom., t = 2 h, T = 50 °C, MW and DES pretreated for t = 30 min, T = 150 °C) | 9.2% Lipid Y, 3.9% TFA Y, 2.2% EPA Y, 4.4% PUFA Y | ||||||
| DMC MAC (3 mL, 100 mg biom., t = 2 h, T = 50 °C, MW and DES pretreated for t = 60 min, T = 100 °C) | 12.5% Lipid Y, 11.0% TFA Y, 2.2% EPA Y, 4.6% PUFA Y | ||||||
| Phormidium valderianum | T (35.86–64.14 °C), P (13.79–56.21 MPa), CO2 Flow (2 L/min), t (90 min) | 3.96 mg/g | CCRD | SX (hexane, 10 g biomass, t = 8 h) | 125.15 μg GAE/g TPC, 19.21 μg β-CAR eq./g T.CAR, 2451 μg BH equivalent/g Reducing power | [110] | |
| S. almeriansis | T (32–60 °C), P (20–60 MPa), CO2 Flow (1 g/min), t (300 min) | RSM | ACE MAC | 2.33 mg/g LUT, 3.07 mg/g β-CAR | [111] | ||
| T (50–65 °C), P (25–55 MPa), CO2 Flow(7.2–14.5 g/min)t (120 min) | 8.74 mg/g | B-D (3.75 mL MeOH/CHF 2:1, 120 mg biomass, t = 1 h) | for lipid determination | [112] | |||
| T (45 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (90 min), Co-solv (EtOH 5% v/v) | 13.2% | Sovová | Kochert Method (MeOH/CHF 1:2 v/v, t = 1 h, T = 45 °C) | 15.7% Y | [93] | ||
| SX (MeOH/CHF 2:1 v/v, t = 18 h) | 22.4% Y, 8.0% Lipid Y, 35.7% Free FA Conversion | ||||||
| S. dimorphus | T (50–100 °C), P (16.6–50 MPa), t (60 min) | B-D | for total lipid determination | [113] | |||
| S. obliquus | T (40–60 °C), P (10–40 MPa), CO2 Flow (7 L/min), t (120 min) | 0.97% | RSM | Axelsson-Gentili method (MeOH/CHF 1:2 v/v 8 mL, 25 mg biomass) | for total lipid determination | [114] | |
| ACE MAC (20 mL with BHT 0.1% w/v, t = 24 h, 200 mg biomass) | for total extr. compounds determination | ||||||
| PLE (EtOH 0–100%, T = 50–170 °C, P = 70 MPa) | 4.83–78.04% Y, 0.66–124.1 mg/gextr CHL, 6.3–49.41 mg GAE/gextr TPC 0.11–1.6 mmol TE/ gextr AO | ||||||
| T (45–65 °C), P (15–30 MPa), CO2 Flow (0.4 kg/h), t (30–90 min), Co-solv (EtOH 5% v/v ) | 24.67% | Sovová | SX (MeOH/CHF 2:1 v/v, t = 18 h) | 29.03% Y, 51.13% Free FA Conv., 14.84% Lipid Y, 27.38% SFA, 19.95% MUFA, 52.67% PUFA, 36.32% Ω-3, 11.20% Ω-6 | [44] | ||
| T (20–200 °C), P (7–80 MPa), t (540 min) | 6.4% | B-D (with hexane, t = 8 h) | for total lipid determination | [2] | |||
| T (40–60 °C), P (15–25 MPa), CO2 Flow (2–4.3 g/min), t (240 min), Co-solv (EtOH 0–9.5% v/v ) | ACE MAC (5 mL, t = 2 h) | 24.00 mg/g CHL-a, 19.04 mg/g CHL-b, 18.90 mg/g CHL-c, 17.78 mg/g T.CAR | [116] | ||||
| S. obtusiusculus | T (20 °C), P (12 MPa), t (540 min) | 6.4% | B-D (with hexane, t = 8 h) | for total lipid determination | [2] | ||
Scenedesmus sp. | T (35–80 °C), P (20–40 MPa), CO2 Flow (750–800 mL/min), t (60 min), Co-solv (MeOH, EtOH, Propanol, Butanol, ACE 0–40% mol) | MeOH MAC | 0.388 mg/g LUT Y | [118] | |||
| EtOH MAC | 0.345 mg/g LUT Y | ||||||
| Propanol MAC | 0.291 mg/g LUT Y | ||||||
| Butanol MAC | 0.269 mg/g LUT Y | ||||||
| ACE MAC | 0.3579 mg/g LUT Y | ||||||
| T (60 °C), P (30 MPa), CO2 Flow (2 mL/min), t (60 min), Co-solv (EtOH 0–10% mol) | UAE (hexane 40 mL,1 g biom.) | 4.00% lipid | [119] | ||||
| UAE (CHF/MeOH/H2O 1:1:0.9 v/v 40 mL, 1 g biomass) | 4.26% Lipid Y | ||||||
| UAE (n-hexane/iso-propanol 3:2 v/v 40 mL, 1 g biomass) | 4.62% Lipid Y | ||||||
| T (35–65 °C), P (20–50 MPa), CO2 Flow (1.38–4.02 g/min) | 7.06% | Multilevel Factorial Design | B-D (hexane 40 mL, 1 g biomass, UAE, t = 30 min) | 4.00% Lipid Y | [120] | ||
| B-D (CHF/MeOH/Water 1:1:0.9 v/v/v 40 mL, 1 g biomass, UAE, t = 30 min) | 4.26% Lipid Y | ||||||
| B-D (hexane/isopropanol 3:2 v/v 40 mL, 1 g biomass, UAE, t = 30 min) | 4.62% Lipid Y | ||||||
| Folch (Hexane, CHF/MeOH/Water 1:1:0.9 v/v/v, Hexane/Isopropanol 3:2 v/v, 40 mL, 1 g biomass, UAE, t = 30 min) | for total lipid determination | ||||||
| Hara & Radin (Hexane, CHF/MeOH/Water 1:1:0.9 v/v/v, Hexane/Isopropanol 3:2 v/v, 40 mL, 1 g biomass, UAE, t = 30 min) | for total lipid determination | ||||||
| SX (Hexane 75 mL, 1 g biomass, t = 12 h) | 2.61% Lipid Y | ||||||
| T (35–50 °C), P (40 MPa), t (120–360 min), Co-solv (MeOH) | Folch (MeOH/CHF 1:2 v/v ) | 5.8% Lipid Y | [121] | ||||
| Synechococcus sp. | T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min), Co-solv (EtOH 0–5% mol) | Reverchon et al. | [75] | ||||
| T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (240 min), Co-solv (EtOH 15% mol) | Multilevel Factorial Design | UAE DMF (1 mL, 2–5 mg) | 5.4 mg/g CHL, 0.48 mg/g MYX, 2.15 mg/g β-CAR, 0.12 mg/g β-CRY, 1.79 mg/g ZEA, 4.93 mg/g T.CAR | [122] | |||
| T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min), Co-solv (EtOH 5% mol) | UAE DMF (5 mL, 0.1 g) | 3.3 μg/mg Total T.CAR, 9.6 μg/mg Total CHL | [76] | ||||
| UAE MeOH (5 mL, 0.1 g) | 1.4 μg/mg T.CAR, 4.1 μg/mg Total CHL | ||||||
| T (40–60 °C), P (20–50 MPa), CO2 Flow (4.5 mmol/min), t (180 min) | Multilevel Factorial Design | UAE MeOH (5 mL, 0.1 g, t = 10 min) | 1.353 μg/mg T.CAR, 4.096 μg/mg Total CHL | [123] | |||
| T (40–60 °C), P (20–40 MPa), CO2 Flow (0.8 g/min), t (180 min), Co-solv (EtOH 0–5% mol) | UAE (DMF 5 mL, 0.105 g biomass, t = 3 min) | 42.53 mg/gextr β-CAR, 10.09 mg/gextr ZEA 59.38 mg/gextr PA, 8.89 mg/gextr PLA, 6.47 mg/gextr OA, 2.11 mg/gextr LOA, 0.23 mg/g Extr LNA | [17] | ||||
| T. chui | T (40–60 °C), P (18–25 MPa), CO2 Flow (2 mL/min), t (60–90 min), Co-solv (EtOH, MeOH) | 4.3% | ASE (DCM/MeOH 9:1, t = 60 min) | 14.6% Y | [124] | ||
| Tetraselmis sp. | T (45 °C), P (30 MPa), CO2 Flow (0.4 kg/h), t (90 min), Co-solv (EtOH 5%) | 14.8% | Sovová | SX (MeOH/CHF 2:1, t = 18 h, T = 105 °C) | 17.7% Y, 38.7% Free FA Conv., 7.0% Lipid Y | [93] | |
| Kochert (MeOH/CHF 2:1, t = 1 h, T = 45 °C) | 19.1% Y | ||||||
| T (40 °C), P (15 MPa), CO2 Flow (5 mL/min), t (30 min), Co-solv (EtOH 5%) | B-D (MeOH/CHF 2:1 v/v 5 mL, 200 mg biomass, t = 4 h) | 11.66% Lipid Y | [125] | ||||
| Cequier-Sanchez (MeOH/DCM 1:2 v/v 6–8 mL, 200 mg biomass, t = 2 h) | 15.05% Lipid Y | ||||||
| Schlechtriem (Propan-2-ol/Cyclohexane 1:1.25 v/v 9 mL, 200 mg biomass, UAE, t = 30 min) | 13.35% Lipid Y | ||||||
| Burja (3 mM KOH in 96% EtOH 15.2 mL, 200 mg biomass, UAE, t = 1 h) | 9.40% Lipid Y |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tzima, S.; Georgiopoulou, I.; Louli, V.; Magoulas, K. Recent Advances in Supercritical CO2 Extraction of Pigments, Lipids and Bioactive Compounds from Microalgae. Molecules 2023, 28, 1410. https://doi.org/10.3390/molecules28031410
Tzima S, Georgiopoulou I, Louli V, Magoulas K. Recent Advances in Supercritical CO2 Extraction of Pigments, Lipids and Bioactive Compounds from Microalgae. Molecules. 2023; 28(3):1410. https://doi.org/10.3390/molecules28031410
Chicago/Turabian StyleTzima, Soultana, Ioulia Georgiopoulou, Vasiliki Louli, and Kostis Magoulas. 2023. "Recent Advances in Supercritical CO2 Extraction of Pigments, Lipids and Bioactive Compounds from Microalgae" Molecules 28, no. 3: 1410. https://doi.org/10.3390/molecules28031410
APA StyleTzima, S., Georgiopoulou, I., Louli, V., & Magoulas, K. (2023). Recent Advances in Supercritical CO2 Extraction of Pigments, Lipids and Bioactive Compounds from Microalgae. Molecules, 28(3), 1410. https://doi.org/10.3390/molecules28031410