Advance in Detection Technique of Lean Meat Powder Residues in Meat Using SERS: A Review
Abstract
:1. Introduction
2. Physical and Chemical Properties of LMP
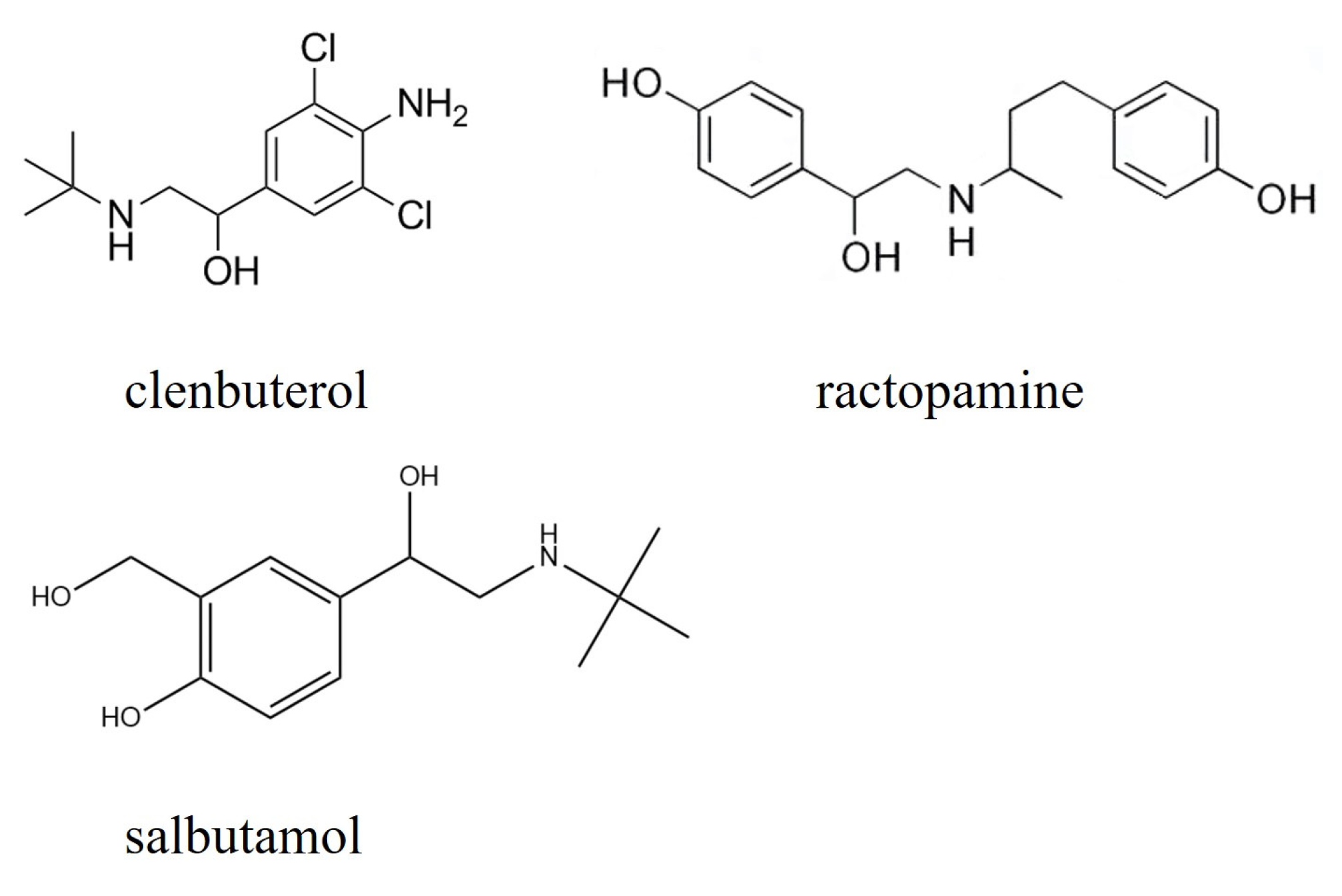
2.1. Types of LMP
2.2. Molecular Structure of LMP
2.3. Mechanisms of Action
3. Raman Spectroscopy and Surface-Enhanced Raman Scattering Techniques
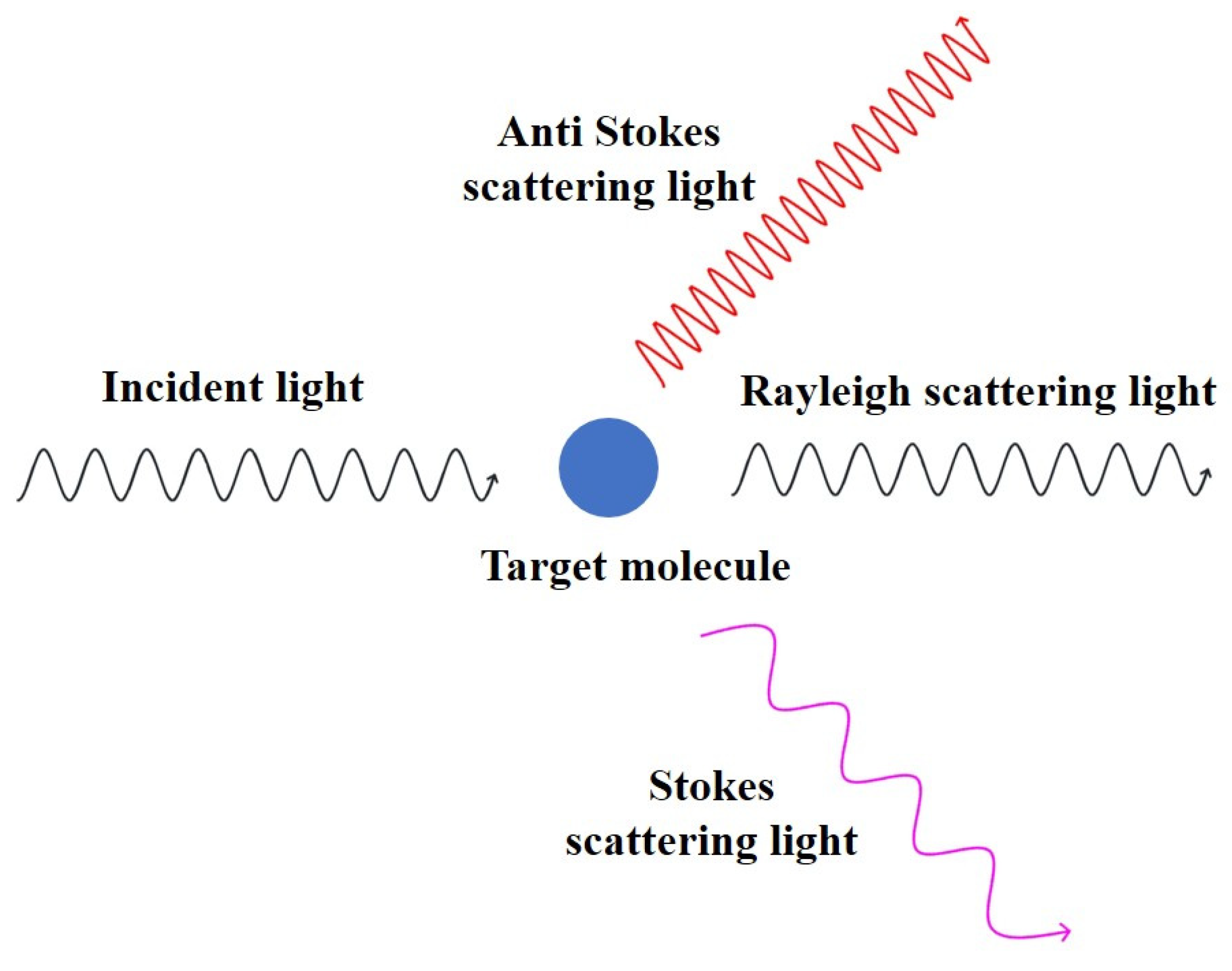
3.1. Raman Spectroscopy
3.2. Surface-Enhanced Raman Scattering Spectroscopy
3.2.1. Enhancement Mechanism of EM
3.2.2. Enhancement Mechanism of CM
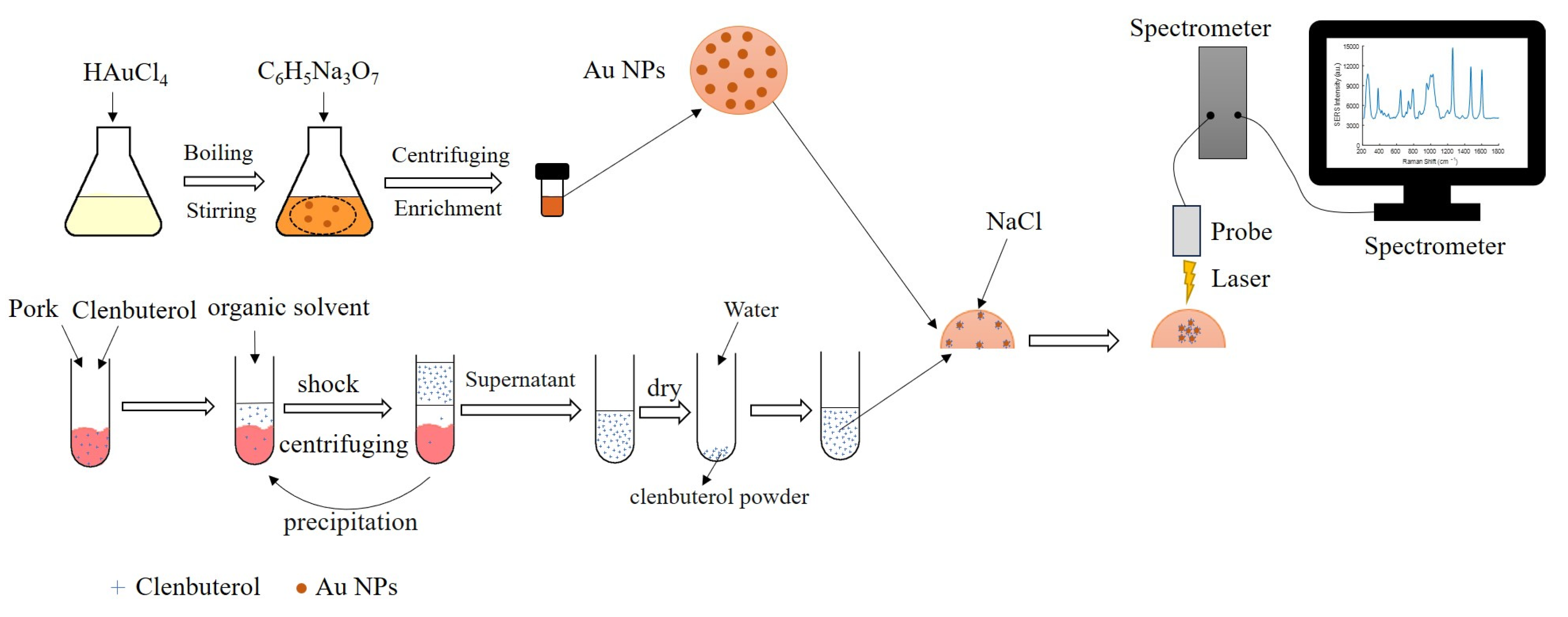
3.3. Procedure for the Detection of LMP Residues in Meats by SERS Technology
4. Applications for LMP Detection
4.1. Raman Spectroscopy without Enhancement
4.2. Application of SERS Spectroscopy for Detecting LMP Residues in Meat
4.2.1. Common Metal Nanoparticle Substrates
4.2.2. Metal Nanoparticles Bound to Adsorbent Materials
4.2.3. Metal Nanoparticles Bound to Chemical Substances
4.3. Application of SERS in Combination with Related Techniques for Detecting LMP in Meat
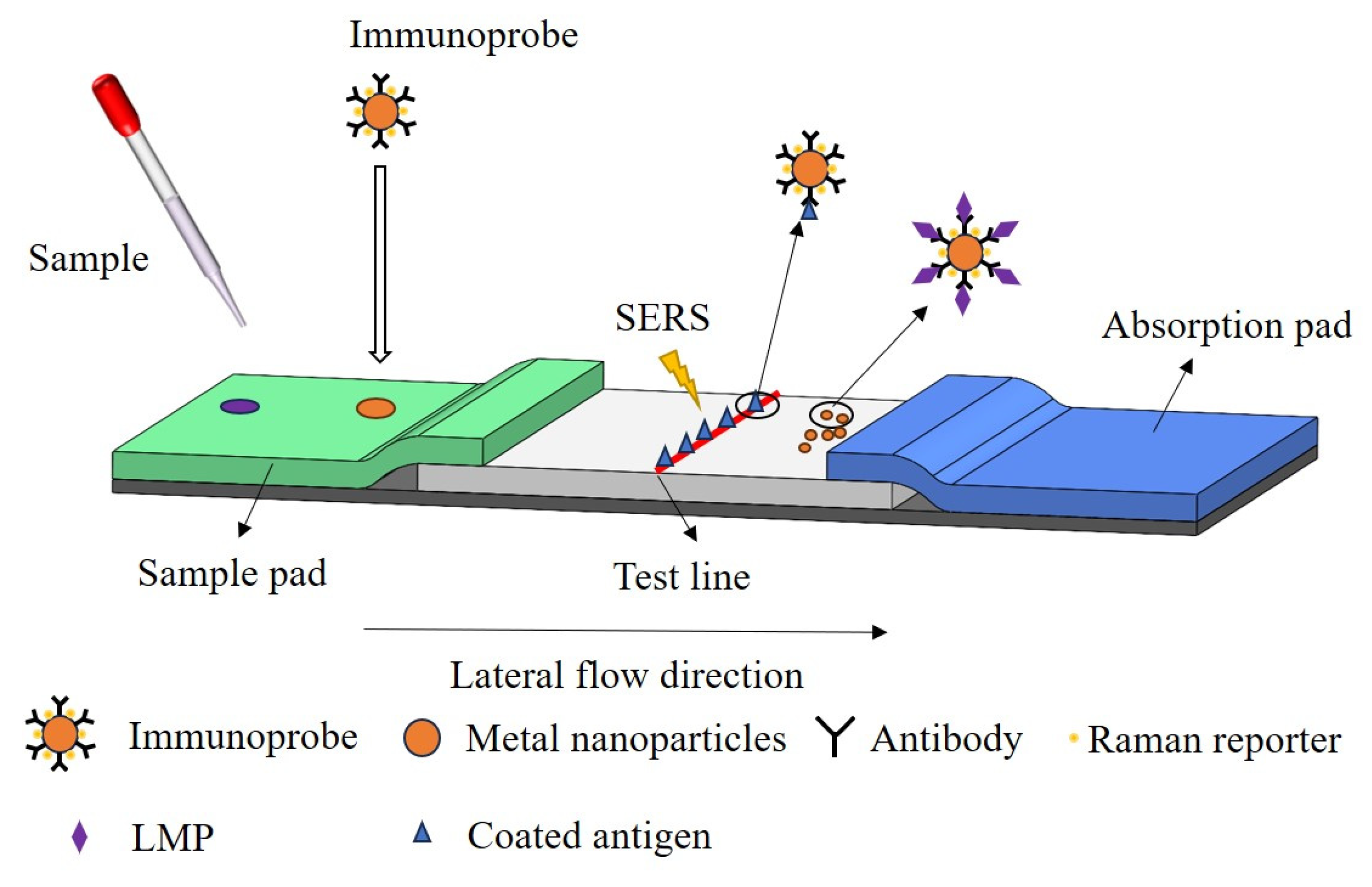
4.3.1. SERS Combined with Immunochromatography
4.3.2. SERS in Combination with Other Techniques
5. Conclusions and Future Trends
- (1)
- Development of novel enhancement substrates with larger SERS enhancement factors and stabilized SERS signals will greatly enhance the SERS signal.
- (2)
- Deposition of metal nanoparticles on a densely ordered solid substrate results in a more uniform “hot spot” produced by the metal nanoparticles.
- (3)
- Combining metal nanoparticles with physically or chemically adsorbent materials allows for better adsorption of target molecules on the surface of the nanoparticles, resulting in higher SERS intensities and stable signals.
- (4)
- Development of solid transparent substrates to reduce the extraction process of LMP residues in meat and to detect them directly on the meat, thus realizing non-destructive and rapid detection.
- (5)
- The combination of two or more nanoparticles for the simultaneous detection of target molecules can greatly enhance the SERS signal of target molecules.
- (6)
- More information on SERS spectra should be mined. Currently, it has been reported that the characteristic bands of SERS signals are mainly selected to establish a ULR, MLR, or PLSR model with the concentration of substances, and the modeling process is relatively simple. The information on the characteristic bands is underutilized, such as the shape of the characteristic bands, the area, etc. The prediction of residual concentrations of target molecules using neural network modeling will be the future direction.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhang, J.; Lu, J.; Zhang, Y.; Wang, Y. A LC-MS/MS method for determination of clenbuterol enantiomers in animal tissues and its application to the enantioselective distribution in Bama mini-pigs. J. Chromatogr. B 2023, 1226, 123790. [Google Scholar] [CrossRef]
- Piribauer, M.; Jiang, L.; Kostov, T.; Parr, M.; Steidel, S.; Bizjak, D.A.; Steinacker, J.M.; Diel, P. Combinatory in vitro effects of the β2-agonists salbutamol and formoterol in skeletal muscle cells. Toxicol. Lett. 2023, 378, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Gill, S.K.; Marriott, H.M.; Suvarna, S.K.; Peachell, P.T. Evaluation of the anti-inflammatory effects of β-adrenoceptor agonists on human lung macrophages. Eur. J. Pharmacol. 2016, 793, 49–55. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Piribauer, M.; Kostov, T.; Steidel, S.; Bizjak, D.; Steinacker, J.; Parr, M.; Diel, P. Testing anabolic activity, potency and mechanisms of action of the phyto-derived beta 2 agonist higenamine. Toxicol. Lett. 2023, 385, 21–28. [Google Scholar] [CrossRef]
- Lin, C.; Li, Y. Detection of clenbuterol in meat samples using a molecularly imprinted electrochemical sensor with MnFe2O4-CQDs composite material. Int. J. Electrochem. Sci. 2023, 18, 100178. [Google Scholar] [CrossRef]
- Abhikha Sherlin, V.; Stanley, M.M.; Wang, S.; Sriram, B.; Baby, J.N.; George, M. Nanoengineered disposable sensor fabricated with lanthanum stannate nanocrystallite for detecting animal feed additive: Ractopamine. Food Chem. 2023, 423, 136268. [Google Scholar] [CrossRef] [PubMed]
- Luan, W.; Ma, Y.; Li, X. Comparative analysis using status and the residue limits of “clenbuterol” domestic and foreign. China Food Saf. Mag. 2022, 19, 181–183. [Google Scholar] [CrossRef]
- Mohamed, R.A.; Elbialy, Z.I.; Abd El Latif, A.S.; Shukry, M.; Assar, D.H.; El Nokrashy, A.M.; Elsheshtawy, A.; Dawood, M.A.O.; Paray, B.A.; Doan, H.V.; et al. Dietary clenbuterol modifies the expression of genes involved in the regulation of lipid metabolism and growth in the liver, skeletal muscle, and adipose tissue of Nile tilapia (Oreochromis niloticus). Aquac. Rep. 2020, 17, 100319. [Google Scholar] [CrossRef]
- Zhao, Y.; Tian, L.; Zhang, X.; Sun, Z.; Shan, X.; Wu, Q.; Chen, R.; Lu, J. A novel molecularly imprinted polymer electrochemiluminescence sensor based on Fe2O3@Ru(bpy)32+ for determination of clenbuterol. Sens. Actuators B Chem. 2022, 350, 130822. [Google Scholar] [CrossRef]
- Shieh, J.; Yang, S. Statutory safety quarantine and its compensation of consumer’s long-term intake of food additives. J. Agric. Food Res. 2022, 7, 100264. [Google Scholar] [CrossRef]
- Chan, S.H.; Lee, W.; Asmawi, M.Z.; Tan, S.C. Chiral liquid chromatography–mass spectrometry (LC-MS/MS) method development for the detection of salbutamol in urine samples. J. Chromatogr. B 2016, 1025, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Wang, T.; Chen, S.; Wang, X.; Reynoso, L.C. Highly sensitive electrochemical sensor based on rGO/Fe3O4 composite as electrocatalyst for clenbuterol detection in doping analysis. Int. J. Electrochem. Sci. 2022, 17, 220128. [Google Scholar] [CrossRef]
- Li, F.; Zhou, J.; Wang, M.; Zhang, L.; Yang, M.; Deng, L. Production of a matrix certified reference material for measurement and risk monitoring of clenbuterol in mutton. Anal. Bioanal. Chem. 2023, 415, 1487–1496. [Google Scholar] [CrossRef]
- Zhong, Y.; Hou, C.; Gao, X.; Wang, M.; Yao, Y.; Chen, M.; Di, B.; Su, M. Application of wastewater-based epidemiology to estimate the usage of beta-agonists in 31 cities in China. Sci. Total Environ. 2023, 894, 164956. [Google Scholar] [CrossRef]
- Hsieh, Y.; Jung, W.; Lee, H. Novel vinylene-based covalent organic framework as a promising adsorbent for the rapid extraction of beta-agonists in meat samples. Anal. Chim. Acta 2023, 1272, 341492. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Agriculture of the People’s Republic of China. Regulation No. 1519; Substances Prohibited for Use in Feed and Drinking Water for Animals; Ministry of Agriculture of the People’s Republic of China: Beijing, China, 2011. Available online: http://www.moa.gov.cn/govpublic/XMYS/201101/t20110113_1806088.htm (accessed on 10 September 2023.).
- Balram, D.; Lian, K.; Sebastian, N.; Alharthi, S.S.; Al-Saidi, H.M.; Kumar, D. Nanomolar electrochemical detection of feed additive ractopamine in meat samples using spinel zinc ferrite decorated 3-dimensional graphene nanosheets to combat food fraud in livestock industries. Food Chem. 2023, 437, 137868. [Google Scholar] [CrossRef]
- Gressler, V.; Franzen, A.R.L.; de Lima, G.J.M.M.; Tavernari, F.C.; Dalla Costa, O.A.; Feddern, V. Development of a readily applied method to quantify ractopamine residue in meat and bone meal by QuEChERS-LC–MS/MS. J. Chromatogr. B 2016, 1015–1016, 192–200. [Google Scholar] [CrossRef]
- Niño, A.M.M.; Granja, R.H.M.M.; Wanschel, A.C.B.A.; Salerno, A.G. The challenges of ractopamine use in meat production for export to European Union and Russia. Food Control 2017, 72, 289–292. [Google Scholar] [CrossRef]
- Velasco-Bejarano, B.; Gómez-Tagle, A.; Noguez-Córdova, M.O.; Zambrano-Zaragoza, M.L.; Miranda-Molina, A.; Bautista, J.; Rodríguez, L.; Velasco-Carrillo, R. Determination of clenbuterol at trace levels in raw gelatin powder and jellies using ultra-high-performance liquid chromatography coupled to triple quadrupole mass spectrometry. Food Chem. 2022, 370, 131261. [Google Scholar] [CrossRef] [PubMed]
- Furusawa, N. An isocratic solvent-free mobile phase HPLC-PDA analysis of clenbuterol and ractopamine. Int. J. Chem. Anal. Sci. 2013, 4, 169–173. [Google Scholar] [CrossRef]
- Protti, M.; Sberna, P.M.; Sardella, R.; Vovk, T.; Mercolini, L.; Mandrioli, R. VAMS and StAGE as innovative tools for the enantioselective determination of clenbuterol in urine by LC-MS/MS. J. Pharmaceut. Biomed. 2021, 195, 113873. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, L.; Jin, Y.; Shuang, Y.; Wang, H. Preparation of a teicoplanin-bonded chiral stationary phase for simultaneous determination of clenbuterol and salbutamol enantiomers in meat by LC-MS/MS. Microchem. J. 2020, 157, 104925. [Google Scholar] [CrossRef]
- Li, G.; Fu, Y.; Han, X.; Li, X.; Li, C. Metabolomic investigation of porcine muscle and fatty tissue after Clenbuterol treatment using gas chromatography/mass spectrometry. J. Chromatogr. A 2016, 1456, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Piña-Olmos, S.; Dolores-Hernández, M.; Villaseñor, A.; Díaz-Torres, R.; Ramírez Bribiesca, E.; López-Arellano, R.; Ramírez-Noguera, P. Extracellular and intracellular zilpaterol and clenbuterol quantification in Hep G2 liver cells by UPLC-PDA and UPLC–MS/MS. J. Pharmaceut. Biomed. 2021, 195, 113817. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Zhu, C.; Liu, X.; Wang, Y.; Qu, F. Screening of Clenbuterol Hydrochloride Aptamers Based on Capillary Electrophoresis. Chin. J. Anal. Chem. 2018, 46, 1595–1603. [Google Scholar] [CrossRef]
- Mao, J.; Huang, L.; Fan, L.; Chen, F.; Lou, J.; Shan, X.; Yu, D.; Zhou, J. 60-Nt DNA Direct Detection without Pretreatment by Surface-Enhanced Raman Scattering with Polycationic Modified Ag Microcrystal Derived from AgCl Cube. Molecules 2021, 26, 6790. [Google Scholar] [CrossRef] [PubMed]
- Muniz-Miranda, M.; Muniz-Miranda, F.; Menziani, M.C.; Pedone, A. Can DFT calculations provide useful information for SERS applications. Molecules 2023, 28, 573. [Google Scholar] [CrossRef] [PubMed]
- Fatkhutdinova, L.I.; Babich, E.; Boldyrev, K.; Shipilovskikh, S.; Terterev, I.; Baranenko, D.; Redkov, A.; Timin, A.; Zyuzin, M.V.; Kivshar, Y.; et al. Rapid and sensitive detection of nucleoprotein SARS-CoV-2 virus: SERS vs ELISA. Photonics Nanostructures—Fundam. Appl. 2023, in press. [Google Scholar] [CrossRef]
- Postnikov, E.B.; Wasiak, M.; Bartoszek, M.; Polak, J.; Zyubin, A.; Lavrova, A.I.; Chora̧żewski, M. Accessing properties of molecular compounds involved in cellular metabolic processes with electron paramagnetic resonance, raman spectroscopy, and differential scanning calorimetry. Molecules 2023, 28, 6417. [Google Scholar] [CrossRef] [PubMed]
- Villa, N.S.; Picarelli, C.; Iacoe, F.; Zanchi, C.G.; Ossi, P.M.; Lucotti, A.; Tommasini, M. Investigating perampanel antiepileptic drug by DFT calculations and SERS with custom spinning cell. Molecules 2023, 28, 5968. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Liu, S.; Huang, L.; Jin, M.; Wang, J. Nanohybrid SERS substrates intended for food supply chain safety. Coordin. Chem. Rev. 2023, 494, 215349. [Google Scholar] [CrossRef]
- Albini, B.; Galinetto, P.; Schiavi, S.; Giulotto, E. Food Safety Issues in the Oltrepò Pavese Area: A SERS Sensing Perspective. Sensors 2023, 23, 9015. [Google Scholar] [CrossRef]
- Dikmen, Z.; Dikmen, G.; Bütün, V. Fluorophore-assisted green fabrication of flexible and cost-effective Ag nanoparticles decorated PVA nanofibers for SERS based trace detection. J. Photochem. Photobiol. A Chem. 2023, 445, 115074. [Google Scholar] [CrossRef]
- Yao, G.; Liu, C.; Elsherbiny, S.M.; Huang, Q. Chiral recognition of D/L-Ribose by visual and SERS assessments. Molecules 2023, 28, 6480. [Google Scholar] [CrossRef] [PubMed]
- Dina, N.E.; Tahir, M.A.; Bajwa, S.Z.; Amin, I.; Valev, V.K.; Zhang, L. SERS-based antibiotic susceptibility testing: Towards point-of-care clinical diagnosis. Biosens. Bioelectron. 2023, 219, 114843. [Google Scholar] [CrossRef]
- Shan, J.; Ren, T.; Li, X.; Jin, M.; Wang, X. Study of microplastics as sorbents for rapid detection of multiple antibiotics in water based on SERS technology. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 284, 121779. [Google Scholar] [CrossRef]
- Barbillon, G.; Cheap-Charpentier, H. Advances in Surface-Enhanced Raman scattering sensors of pollutants in water treatment. Nanomaterials 2023, 13, 2417. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Xu, D.; Dong, X.; Huang, Q. A review: Research progress of SERS-based sensors for agricultural applications. Trends Food Sci. Technol. 2022, 128, 90–101. [Google Scholar] [CrossRef]
- Jigyasa; Rajput, J.K. Nanosensors: A smart remedy for early detection of clenbuterol contamination in food. Food Chem. 2023, 426, 136569. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Tang, X.; Xiong, R.; Li, L.; Du, X.; He, L. Simultaneous determination of fourteen β2-agonist enantiomers in food animal muscles by liquid chromatography coupled with tandem mass spectrometry. J. Chromatogr. B 2022, 1193, 123169. [Google Scholar] [CrossRef] [PubMed]
- Jing, H.; Ouyang, H.; Li, W.; Long, Y. Molten salt synthesis of BCNO nanosheets for the electrochemical detection of clenbuterol. Microchem. J. 2022, 178, 107359. [Google Scholar] [CrossRef]
- Ho, J.; Huo, T.; Lin, L.; Tsai, T. Pharmacokinetics of ractopamine and its organ distribution in rats. J. Agric. Food Chem. 2014, 62, 9273–9278. [Google Scholar] [CrossRef] [PubMed]
- Itoh, T.; Procházka, M.; Dong, Z.; Ji, W.; Yamamoto, Y.S.; Zhang, Y.; Ozaki, Y. Toward a new era of SERS and TERS at the nanometer scale: From fundamentals to innovative applications. Chem. Rev. 2023, 123, 1552–1634. [Google Scholar] [CrossRef] [PubMed]
- Shan, J.; Shi, L.; Li, Y.; Yin, X.; Wang, S.; Liu, S.; Sun, J.; Zhang, D.; Ji, Y.; Wang, J. SERS-based immunoassay for amplified detection of food hazards: Recent advances and future trends. Trends Food Sci. Technol. 2023, 140, 104149. [Google Scholar] [CrossRef]
- Ott, A.K.; Ferrari, A.C. Raman spectroscopy of graphene and related materials. In Encyclopedia of Condensed Matter Physics, 2nd ed.; Chakraborty, T., Ed.; Academic Press: Oxford, UK, 2024; pp. 233–247. [Google Scholar]
- Qin, J.; Kim, M.S.; Chao, K.; Dhakal, S.; Cho, B.; Lohumi, S.; Mo, C.; Peng, Y.; Huang, M. Advances in Raman spectroscopy and imaging techniques for quality and safety inspection of horticultural products. Postharvest Biol. Tec. 2019, 149, 101–117. [Google Scholar] [CrossRef]
- Das, R.S.; Agrawal, Y.K. Raman spectroscopy: Recent advancements, techniques and applications. Vib. Spectrosc. 2011, 57, 163–176. [Google Scholar] [CrossRef]
- Tanwar, S.; Paidi, S.K.; Prasad, R.; Pandey, R.; Barman, I. Advancing Raman spectroscopy from research to clinic: Translational potential and challenges. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2021, 260, 119957. [Google Scholar] [CrossRef]
- Bērziņš, K.; Fraser-Miller, S.J.; Gordon, K.C. Recent advances in low-frequency Raman spectroscopy for pharmaceutical applications. Int. J. Pharmaceut. 2021, 592, 120034. [Google Scholar] [CrossRef]
- Fleischmann, M.; Hendra, P.J.; Mcquillan, A.J. Raman spectra of pyridzne adsorbed at a silver electrode. Chem. Phys. Lett. 1974, 26, 163–166. [Google Scholar] [CrossRef]
- Jeanmaire, D.L.; Van Duyhe, R.P. Surface Raman spectroelectrochemistry: Part I. Heterocyclic, aromatic, and aliphatic amines adsorbed on the anodized silver electrode. J. Electroanal. Chem. Interfacial Electrochem. 1977, 84, 1–20. [Google Scholar] [CrossRef]
- Bi, S.; Shao, D.; Yuan, Y.; Zhao, R.; Li, X. Sensitive surface-enhanced Raman spectroscopy (SERS) determination of nitrofurazone by β-cyclodextrin-protected AuNPs/γ-Al2O3 nanoparticles. Food Chem. 2022, 370, 131059. [Google Scholar] [CrossRef]
- Panneerselvam, R.; Xiao, L.; Waites, K.B.; Atkinson, T.P.; Dluhy, R.A. A rapid and simple chemical method for the preparation of Ag colloids for surface-enhanced Raman spectroscopy using the Ag mirror reaction. Vib. Spectrosc. 2018, 98, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Song, L.; Li, J.; Li, H.; Chang, Y.; Dai, S.; Xu, R.; Dou, M.; Li, Q.; Lv, G.; Zheng, T. Highly sensitive SERS detection for Aflatoxin B1 and Ochratoxin a based on aptamer-functionalized photonic crystal microsphere array. Sens. Actuators B Chem. 2022, 364, 131778. [Google Scholar] [CrossRef]
- Jiang, Y.; Sun, D.; Pu, H.; Wei, Q. Surface enhanced Raman spectroscopy (SERS): A novel reliable technique for rapid detection of common harmful chemical residues. Trends Food Sci. Technol. 2018, 75, 10–22. [Google Scholar] [CrossRef]
- Kaur, B.; Kumar, S.; Kaushik, B.K. Recent advancements in optical biosensors for cancer detection. Biosens. Bioelectron. 2022, 197, 113805. [Google Scholar] [CrossRef]
- Stuart, D.D.; Ebel, C.P.; Cheng, Q. Biosensing empowered by molecular identification: Advances in surface plasmon resonance techniques coupled with mass spectrometry and Raman spectroscopy. Sens. Actuators Rep. 2022, 4, 100129. [Google Scholar] [CrossRef]
- Mikac, L.; Rigó, I.; škrabić, M.; Ivanda, M.; Veres, M. Comparison of glyphosate detection by Surface-Enhanced raman spectroscopy using gold and silver nanoparticles at different laser excitations. Molecules 2022, 27, 5767. [Google Scholar] [CrossRef] [PubMed]
- Issatayeva, A.; Farnesi, E.; Cialla-May, D.; Schmitt, M.; Rizzi, F.M.A.; Milanese, D.; Selleri, S.; Cucinotta, A. SERS-based methods for the detection of genomic biomarkers of cancer. Talanta 2023, 267, 125198. [Google Scholar] [CrossRef] [PubMed]
- Shah, K.C.; Shah, M.B.; Solanki, S.J.; Makwana, V.D.; Sureja, D.K.; Gajjar, A.K.; Bodiwala, K.B.; Dhameliya, T.M. Recent advancements and applications of Raman spectroscopy in pharmaceutical analysis. J. Mol. Struct. 2023, 1278, 134914. [Google Scholar] [CrossRef]
- Pu, H.; Fang, T.; Wu, Z.; Sun, D. Advancements in recyclable photocatalytic semiconductor substrates for SERS detection in food safety applications. Trends Food Sci. Technol. 2023, 138, 697–707. [Google Scholar] [CrossRef]
- Estefany, C.; Sun, Z.; Hong, Z.; Du, J. Raman spectroscopy for profiling physical and chemical properties of atmospheric aerosol particles: A review. Ecotoxicol. Environ. Saf. 2023, 249, 114405. [Google Scholar] [CrossRef]
- Du, X.; Liu, D.; An, K.; Jiang, S.; Wei, Z.; Wang, S.; Ip, W.F.; Pan, H. Advances in oxide semiconductors for surface enhanced Raman scattering. Appl. Mater. Today 2022, 29, 101563. [Google Scholar] [CrossRef]
- Wu, Z.; Pu, H.; Sun, D. Fingerprinting and tagging detection of mycotoxins in agri-food products by surface-enhanced Raman spectroscopy: Principles and recent applications. Trends Food Sci. Technol. 2021, 110, 393–404. [Google Scholar] [CrossRef]
- Paudel, A.; Raijada, D.; Rantanen, J. Raman spectroscopy in pharmaceutical product design. Adv. Drug Deliv. Rev. 2015, 89, 3–20. [Google Scholar] [CrossRef]
- Meza-Marquez, O.G.; Gallardo-Velazquez, T.; Dorantes-Alvarez, L.; Osorio-Revilla, G.; de la Rosa, A.J. FT-MIR and Raman spectroscopy coupled to multivariate analysis for the detection of clenbuterol in murine model. Analyst 2011, 136, 3355–3365. [Google Scholar] [CrossRef]
- Pham, T.B.N.; Bui, T.T.T.; Tran, V.Q.; Dang, V.Q.; Hoang, L.N.; Tran, C.K. Surface-enhanced Raman scattering (SERS) performance on salbutamol detection of colloidal multi-shaped silver nanoparticles. Appl. Nanosci. 2020, 10, 703–714. [Google Scholar] [CrossRef]
- Izquierdo-Lorenzo, I.; Sanchez-Cortes, S.; Garcia-Ramos, J.V. Adsorption of Beta-Adrenergic Agonists Used in Sport Doping on Metal Nanoparticles: A Detection Study Based on Surface-Enhanced Raman Scattering. Langmuir 2010, 26, 14663–14670. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.H.; Yuan, H.C.; Peng, Y.J.; Hong, Q.; Liu, M.H. Detection of ractopamine and clenbuterol hydrochloride residues in pork using surface enhanced Raman spectroscopy. J. Appl. Spectrosc. 2017, 84, 76–81. [Google Scholar] [CrossRef]
- Zhai, F.; Huang, Y.; Wang, X.; Lai, K. Surface-Enhanced Raman spectroscopy for rapid determination of β-Agonists in swine urine. Chin. J. Anal. Chem. 2012, 40, 718–723. [Google Scholar] [CrossRef]
- Zhai, F.; Huang, Y.; Li, C.; Wang, X.; Lai, K. Rapid determination of ractopamine in swine urine using Surface-Enhanced Raman spectroscopy. J. Agric. Food Chem. 2011, 59, 10023–10027. [Google Scholar] [CrossRef] [PubMed]
- Feng, D.; Xu, S.; Wen, B.; Kathiresan, M.; Zhang, Y.; Wang, A.; Zhang, F.; Jin, S.; Li, J. Ultrasensitive and rapid detection for multiple doping in saliva and urine using surface-enhanced Raman spectroscopy. Spectrosc. Lett. 2023, 56, 249–262. [Google Scholar] [CrossRef]
- Guo, Q.; Peng, Y.; Chao, K. Raman enhancement effect of different silver nanoparticles on salbutamol. Heliyon 2022, 8, e9576. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Peng, Y.; Chao, K.; Zhuang, Q.; Chen, Y. Raman enhancement effects of gold nanoparticles with different particle sizes on clenbuterol and ractopamine. Vib. Spectrosc. 2022, 123, 103444. [Google Scholar] [CrossRef]
- Guo, Q.; Peng, Y.; Zhao, X.; Chen, Y. Rapid detection of clenbuterol residues in pork using enhanced Raman spectroscopy. Biosensors 2022, 12, 859. [Google Scholar] [CrossRef]
- Guo, Q.; Peng, Y.; Chao, K.; Qin, J.; Chen, Y.; Yin, T. A determination method for clenbuterol residue in pork based on optimal particle size gold colloid using SERS. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 302, 123097. [Google Scholar] [CrossRef] [PubMed]
- Yan, B.; Sun, K.; Chao, K.; Alharbi, N.S.; Li, J.; Huang, Q. Fabrication of a novel transparent SERS substrate comprised of ag-nanoparticle arrays and its application in rapid detection of ractopamine on meat. Food Anal. Methods 2018, 11, 2329–2335. [Google Scholar] [CrossRef]
- Sun, Y.; Chen, H.; Ma, P.; Li, J.; Zhang, Z.; Shi, H.; Zhang, X. In situ synthesis of graphene oxide/gold nanocomposites as ultrasensitive surface-enhanced Raman scattering substrates for clenbuterol detection. Anal. Bioanal. Chem. 2020, 412, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Su, X.; Han, C.; Wang, S.; Wang, P.; Zhang, S.; Xie, J. Ultrasensitive detection of salbutamol in animal urine by immunomagnetic bead treatment coupling with surface-enhanced Raman spectroscopy. Sens. Actuators B Chem. 2018, 255, 2329–2338. [Google Scholar] [CrossRef]
- Cheng, J.; Su, X.; Wang, S.; Zhao, Y. Highly sensitive detection of clenbuterol in animal urine using immunomagnetic bead treatment and Surface-Enhanced raman spectroscopy. Sci. Rep. 2016, 6, 32637. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Wang, S.; Zhang, S.; Wang, P.; Xie, J.; Han, C.; Su, X. Rapid and sensitive determination of clenbuterol residues in animal urine by surface-enhanced Raman spectroscopy. Sens. Actuators B Chem. 2019, 279, 7–14. [Google Scholar] [CrossRef]
- Gu, X.; Tian, S.; Chen, Y.; Wang, Y.; Gu, D.; Guo, E.; Liu, Y.; Li, J.; Deng, A. A SERS-based competitive immunoassay using highly ordered gold cavity arrays as the substrate for simultaneous detection of β-adrenergic agonists. Sens. Actuators B Chem. 2021, 345, 130230. [Google Scholar] [CrossRef]
- Yu, S.; Liu, Z.; Zhang, J.; Li, H.; Xu, N.; Yuan, X.; Wu, Y. An azo-coupling reaction-based surface enhanced resonance Raman scattering approach for ultrasensitive detection of salbutamol. RSC Adv. 2018, 8, 5536–5541. [Google Scholar] [CrossRef] [PubMed]
- Yao, D.; Li, C.; Wen, G.; Liang, A.; Jiang, Z. A highly sensitive and accurate SERS/RRS dual-spectroscopic immunosensor for clenbuterol based on nitrogen/silver-codoped carbon dots catalytic amplification. Talanta 2020, 209, 120529. [Google Scholar] [CrossRef]
- Duan, N.; Qi, S.; Guo, Y.; Xu, W.; Wu, S.; Wang, Z. Fe3O4@Au@Ag nanoparticles as surface-enhanced Raman spectroscopy substrates for sensitive detection of clenbuterol hydrochloride in pork with the use of aptamer binding. LWT 2020, 134, 110017. [Google Scholar] [CrossRef]
- Wei, C.; Zong, Y.; Guo, Q.; Xu, M.; Yuan, Y.; Yao, J. Magnetic separation of clenbuterol based on competitive immunoassay and evaluation by surface-enhanced Raman spectroscopy. RSC Adv. 2017, 7, 3388–3397. [Google Scholar] [CrossRef]
- Wei, C.; Zhang, C.; Xu, M.; Yuan, Y.; Yao, J. Liquid magnetic competitive immunoassay of clenbuterol based on SERS. J. Raman Spectrosc. 2017, 48, 1307–1317. [Google Scholar] [CrossRef]
- Liang, J.; Liu, H.; Huang, C.; Yao, C.; Fu, Q.; Li, X.; Cao, D.; Luo, Z.; Tang, Y. Aggregated silver nano-particles based Surface-Enhanced Raman scattering Enzyme-Linked immunosorbent assay for ultrasensitive detection of protein biomarkers and small molecules. Anal. Chem. 2015, 87, 5790–5796. [Google Scholar] [CrossRef]
- Li, M.; Yang, H.; Li, S.; Zhao, K.; Li, J.; Jiang, D.; Sun, L.; Deng, A. Ultrasensitive and quantitative detection of a new β-Agonist phenylethanolamine a by a novel immunochromatographic assay based on Surface-Enhanced Raman scattering (SERS). J. Agric. Food Chem. 2014, 62, 10896–10902. [Google Scholar] [CrossRef]
- Xie, Y.; Chang, H.; Zhao, K.; Li, J.; Yang, H.; Mei, L.; Xu, S.; Deng, A. A novel immunochromatographic assay (ICA) based on surface-enhanced Raman scattering for the sensitive and quantitative determination of clenbuterol. Anal. Methods 2015, 7, 513–552. [Google Scholar] [CrossRef]
- Fu, X.; Chu, Y.; Zhao, K.; Li, J.; Deng, A. Ultrasensitive detection of the β-adrenergic agonist brombuterol by a SERS-based lateral flow immunochromatographic assay using flower-like gold-silver core-shell nanoparticles. Microchim. Acta 2017, 184, 1711–1719. [Google Scholar] [CrossRef]
- Su, L.; Hu, H.; Tian, Y.; Jia, C.; Wang, L.; Zhang, H.; Wang, J.; Zhang, D. Highly sensitive Colorimetric/Surface-Enhanced Raman spectroscopy immunoassay relying on a metallic core–shell Au/Au nanostar with clenbuterol as a target analyte. Anal. Chem. 2021, 93, 8362–8369. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Hu, Y.; Liu, J. Simultaneous detection of clenbuterol and ractopamine based on multiplexed competitive surface enhanced Raman scattering (SERS) immunoassay. New J. Chem. 2017, 41, 10407–10414. [Google Scholar] [CrossRef]
- Wu, T.; Li, J.; Zheng, S.; Yu, Q.; Qi, K.; Shao, Y.; Wang, C.; Tu, J.; Xiao, R. Magnetic Nanotag-Based Colorimetric/SERS Dual-Readout immunochromatography for ultrasensitive detection of clenbuterol hydrochloride and ractopamine in food samples. Biosensors 2022, 12, 709. [Google Scholar] [CrossRef] [PubMed]
- Tu, J.; Wu, T.; Yu, Q.; Li, J.; Zheng, S.; Qi, K.; Sun, G.; Xiao, R.; Wang, C. Introduction of multilayered magnetic core–dual shell SERS tags into lateral flow immunoassay: A highly stable and sensitive method for the simultaneous detection of multiple veterinary drugs in complex samples. J. Hazard. Mater. 2023, 448, 130912. [Google Scholar] [CrossRef] [PubMed]
- Zheng, T.; Gao, Z.; Luo, Y.; Liu, X.; Zhao, W.; Lin, B. Manual-slide-engaged paper chip for parallel SERS-immunoassay measurement of clenbuterol from swine hair. Electrophoresis 2016, 37, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Dou, B.; Luo, Y.; Chen, X.; Shi, B.; Du, Y.; Gao, Z.; Zhao, W.; Lin, B. Direct measurement of beta-agonists in swine hair extract in multiplexed mode by surface-enhanced Raman spectroscopy and microfluidic paper. Electrophoresis 2015, 36, 485–487. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.; Hu, Y.; Gao, J.; Zhong, L. Highly sensitive detection of clenbuterol using competitive surface-enhanced Raman scattering immunoassay. Anal. Chim. Acta 2011, 697, 61–66. [Google Scholar] [CrossRef]
- Xiao, X.; Yan, K.; Xu, X.; Li, G. Rapid analysis of ractopamine in pig tissues by dummy-template imprinted solid-phase extraction coupling with surface-enhanced Raman spectroscopy. Talanta 2015, 138, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Wang, K.; Tian, S.; Shao, X.; Li, J.; Deng, A. A SERS/electrochemical dual-signal readout immunosensor using highly-ordered Au/Ag bimetallic cavity array as the substrate for simultaneous detection of three β-adrenergic agonists. Talanta 2023, 254, 124159. [Google Scholar] [CrossRef]
- Ali, H.R.H.; Edwards, H.G.M.; Kendrick, J.; Scowen, I.J. Vibrational spectroscopic study of terbutaline hemisulphate. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2009, 72, 715–719. [Google Scholar] [CrossRef]
- Ali, H.R.H.; Edwards, H.G.M.; Kendrick, J.; Scowen, I.J. Vibrational spectroscopic study of salbutamol hemisulphate. Drug Test. Anal. 2009, 1, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Jing, W.; Wang, J.; Kuipers, B.; Bi, W.; Chen, D.D.Y. Recent applications of graphene and graphene-based materials as sorbents in trace analysis. TrAC Trends Anal. Chem. 2021, 137, 116212. [Google Scholar] [CrossRef]
- Jiang, L.; Mehedi Hassan, M.; Ali, S.; Li, H.; Sheng, R.; Chen, Q. Evolving trends in SERS-based techniques for food quality and safety: A review. Tends Food Sci. Technol. 2021, 112, 225–240. [Google Scholar] [CrossRef]
| Method | Enhanced Substrate | Material | Target Molecule | Quantitative Range | LOD | References |
|---|---|---|---|---|---|---|
| SERS | - | Rat meat | Clenbuterol | 5–10,000 ppb | – | [67] |
| SERS | MAgNPs | Water | Salbutamol | 0–1000 ng/µL | 1.25 ng/μL | [68] |
| SERS | Au NPs | Water | Clenbuterol, salbutamol, terbutaline | 0–10−4 mol/L | 35, 765, 55 ng/mL | [69] |
| SERS | Au NPs | Water | Clenbuterol, ractopamine | 0.1–5 mg/L | 0.1 mg/L | [70] |
| SERS | Au NPs | Standard solution | Clenbuterol, ractopamine, salbutamol, | 1–20 mg/L | 0.002, 0.1, 0.002 mg/L | [71] |
| SERS | Gold-plated Klarite | Swine urine | Ractopamine, | 0.4–8 µg/mL | 0.4 μg/mL | [72] |
| SERS | Ag NPs | Saliva, urine | Clenbuterol | 2.5–1000 ng/mL | 25 ng/mL | [73] |
| SERS | Ag NPs | Standard solution | Salbutamol | 0.2−1 mg/L | 0.2 mg/L | [74] |
| SERS | Au NPs | Standard solution | Clenbuterol, ractopamine | 0.1–1 µg/mL | 18 ng/mL, 24 ng/mL | [75] |
| SERS | Au NPs | Pork | Clenbuterol | 1–10 µg/g | 1 µ g/g | [76] |
| SERS | Au NPs | Pork | Clenbuterol | 0.5–15 µg/mL | 42 ng/g | [77] |
| SERS | Ag NPs | Pork | Ractopamine | 10−8−10−4 M | 1.0 × 10−8 M | [78] |
| SERS | GO/AuNP | Standard solution | Clenbuterol | 5 × 10−8–10−6 mol/L | 3.34 × 10−8 mol/L | [79] |
| SERS | RoGO/AuNPs | Animal urine | Salbutamol | 1–20 ng/mL | 0.5 ng/mL | [80] |
| SERS | Au NPs | Swine urine | Clenbuterol | 0.5–20 ng/mL | 0.5 ng/mL | [81] |
| SERS | roGO/AuNPs | Animal urine | Clenbuterol | 1–20 ng/mL | 0.5 ng/mL | [82] |
| SERS | GCA-Ab | Pork, pig liver, urine | Salbutamol, trombuterol | 0.005–100, 0.003–200 ng/mL | 1.0, 2.0 pg/mL | [83] |
| SERS | Fe3O4@Ag | Pork | Salbutamol | 10−11–10−6 M | 1.0 × 10−10 M | [84] |
| SERS | Au NPs | Standard solution | Clenbuterol | 0.0033–0.067 ng/mL | 0.68 pg mL | [85] |
| SERS-IA | Fe3O4@Au@Ag | Pork | Clenbuterol | 0.01–10 ng/mL | 3 pg/mL | [86] |
| SERS-IA | Fe3O4@Au-Ab | Standard solution | Clenbuterol, salbutamol | - | 17 fg/mL | [87] |
| SERS-IA | Fe3O4@Au-Ab | Standard solution | Clenbuterol | 1–100 ng/mL | 0.22 fg/mL | [88] |
| SERS-Elisa | Ag NPs | Swine urine | Ractopamine | - | 10−6 ng/mL | [89] |
| SERS-IAC | AuMBA @Ag-Ab | Swine urine | Phenylethanolamine A | 0–100 ng/mL | 0.32 pg/mL | [90] |
| SERS-ICA | AuMBA @Ag–Ab | Swine urine | Clenbuterol | 0–10 ng/mL | 0.24 pg/mL | [91] |
| SERS-LFIA | AuNFsMBA@Ag-Ab | Pork, swine urine | Brombuterol | 0–100 ng/mL | 0.5 pg/mL | [92] |
| SERS-LFIA | Au@Au nanostar | Standard solution | Clenbuterol | 0.05–1 ng/mL | 0.05 ng/mL | [93] |
| SERS- | Ab–BPAuDP–Ab | Standard solution | Clenbuterol, ractopamine | 1–1000 pg/mL | 1 pg/mL | [94] |
| SERS-IAC | Fe3O4@Au-Ab | Pork, beef, and lamb | Clenbuterol, ractopamine | 0–3 ng/mL | 7.8 pg/mL 3.5 pg/mL | [95] |
| SERS-LFA | Fe3O4@Au@Ag | Pork, lake water | Ractopamine, clenbuterol | - | 2.5, 6.2 pg/mL | [96] |
| SERS | AuMBA-Ab | Pig hair | Clenbuterol | 0.1–100 pg/mL | 0.1 pg/mL | [97] |
| SERS | Ag NPs | Pig hair | clenbuterol, ractopamine, salbutamol | - | 20, 20, 30 ng/mL | [98] |
| SERS-IA | Au NPs-Ab | Swine urine | Clenbuterol | 0.1–100 pg/mL | 0.1 pg/mL | [99] |
| SERS | Au NPs | Pork, pig liver | Ractopamine | 20.0–200.0 μg/L | 3.1 μg/L | [100] |
| SERS | Au/Ag NPs | Pig liver | Salbutamol, ractopamine, phenylethanolamine A | 0.002–200 0.001–200 0.005–100 ng/mL | 0.8, 0.4, 1.3 pg/mL | [101] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, Q.; Peng, Y.; Qin, J.; Chao, K.; Zhao, X.; Yin, T. Advance in Detection Technique of Lean Meat Powder Residues in Meat Using SERS: A Review. Molecules 2023, 28, 7504. https://doi.org/10.3390/molecules28227504
Guo Q, Peng Y, Qin J, Chao K, Zhao X, Yin T. Advance in Detection Technique of Lean Meat Powder Residues in Meat Using SERS: A Review. Molecules. 2023; 28(22):7504. https://doi.org/10.3390/molecules28227504
Chicago/Turabian StyleGuo, Qinghui, Yankun Peng, Jianwei Qin, Kuanglin Chao, Xinlong Zhao, and Tianzhen Yin. 2023. "Advance in Detection Technique of Lean Meat Powder Residues in Meat Using SERS: A Review" Molecules 28, no. 22: 7504. https://doi.org/10.3390/molecules28227504
APA StyleGuo, Q., Peng, Y., Qin, J., Chao, K., Zhao, X., & Yin, T. (2023). Advance in Detection Technique of Lean Meat Powder Residues in Meat Using SERS: A Review. Molecules, 28(22), 7504. https://doi.org/10.3390/molecules28227504







