Investigating Perampanel Antiepileptic Drug by DFT Calculations and SERS with Custom Spinning Cell
Abstract
1. Introduction
2. Results and Discussion
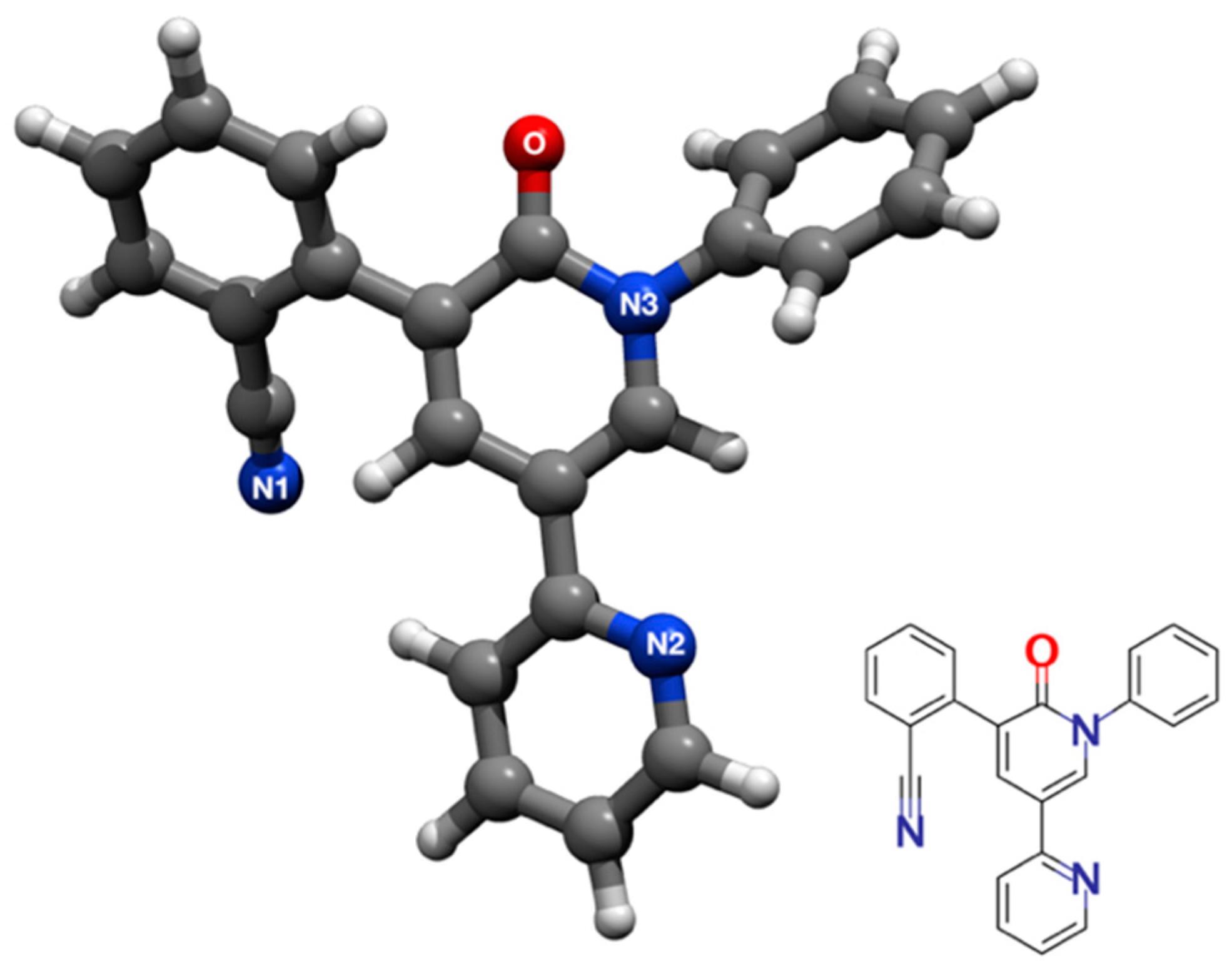
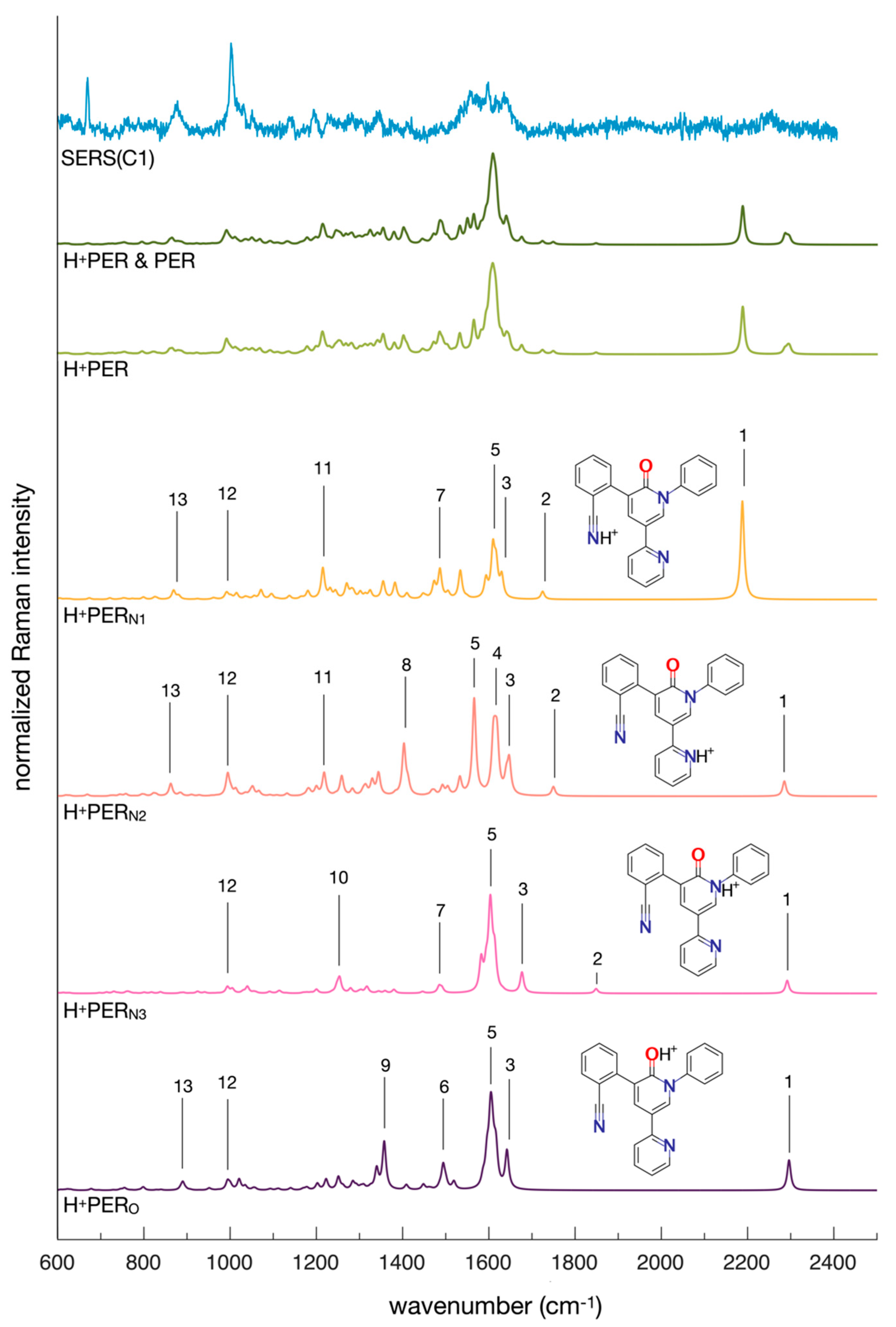
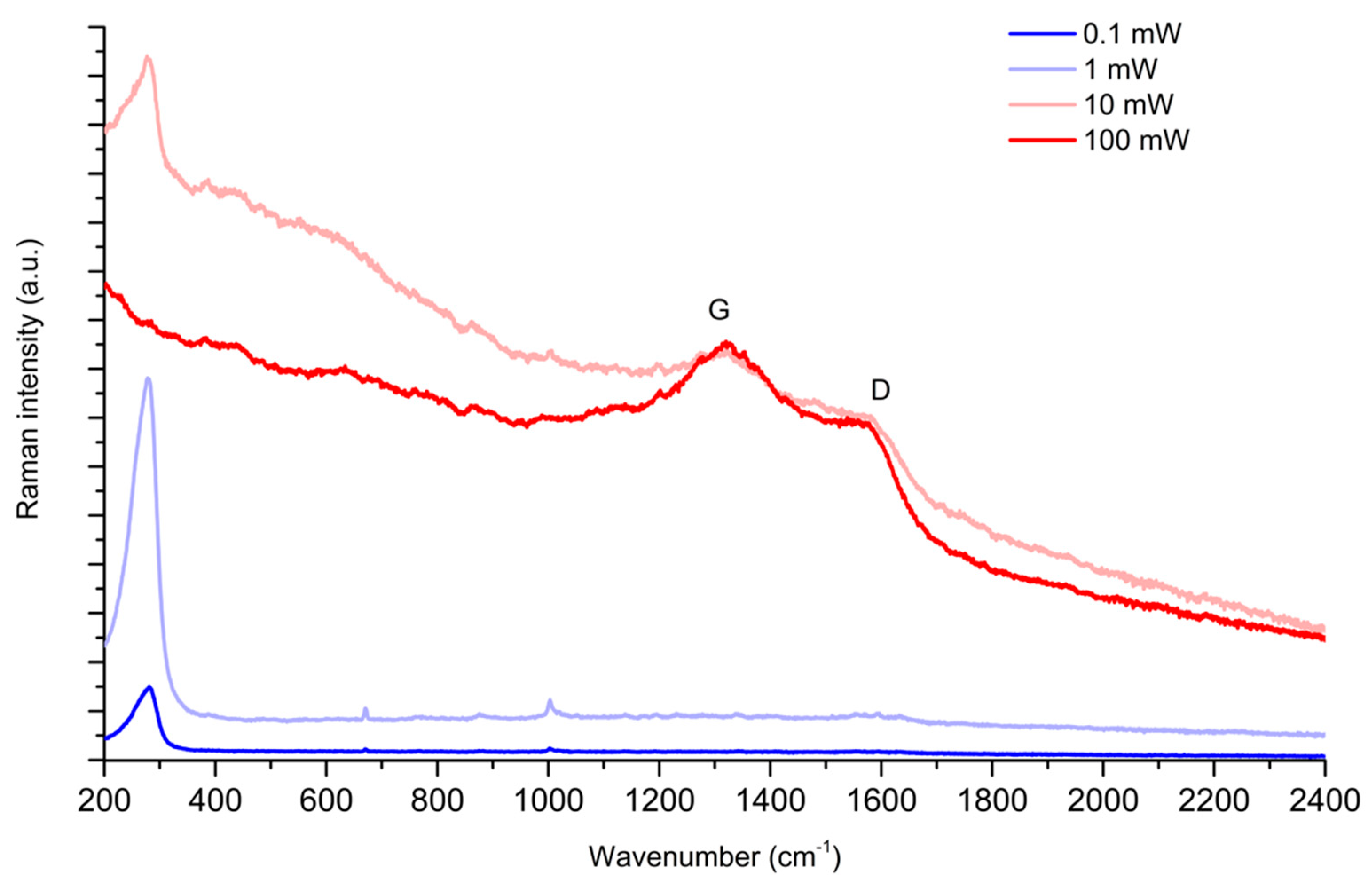
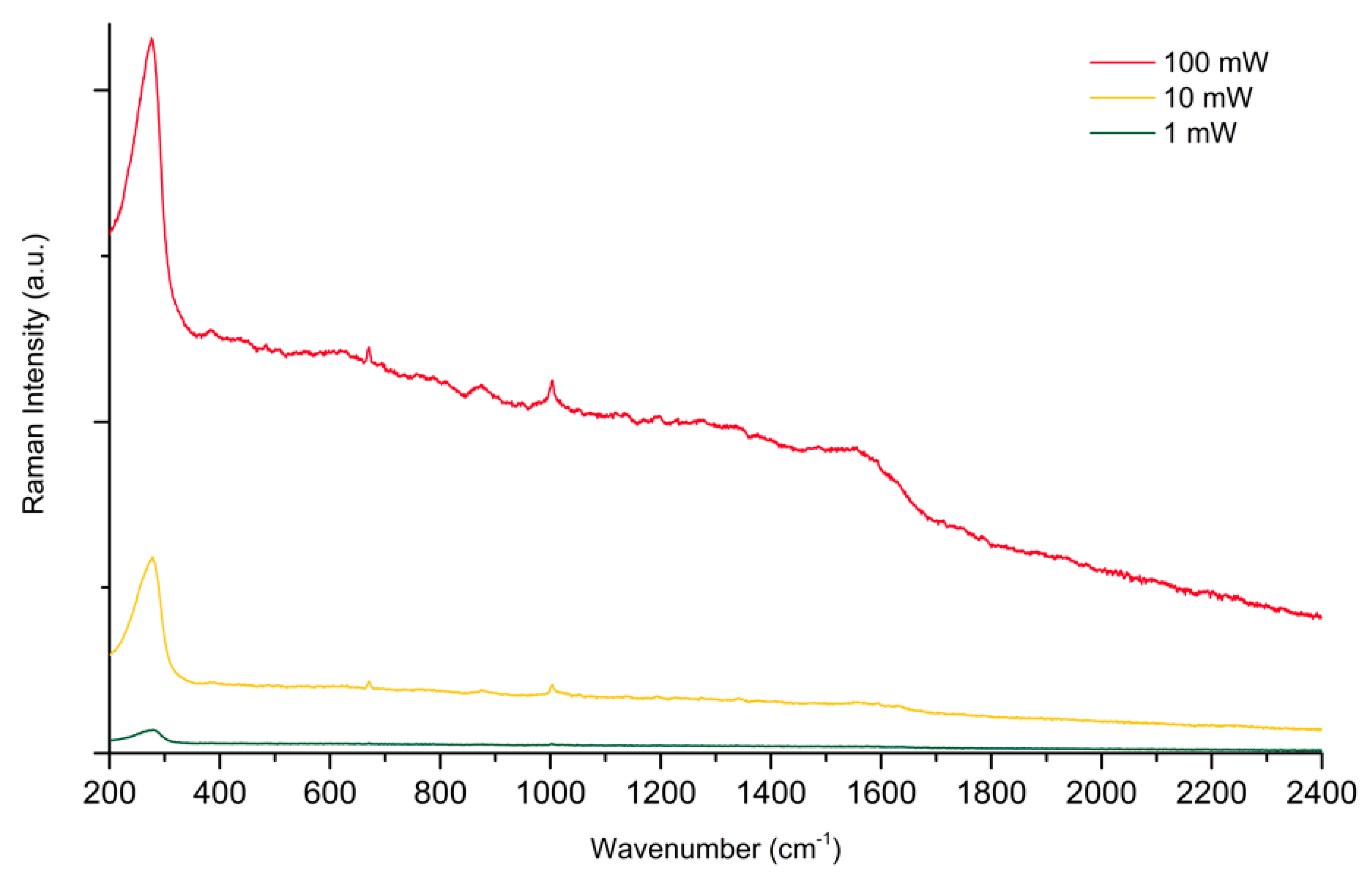
2.1. The Protonation of PER and Its Effects on SERS
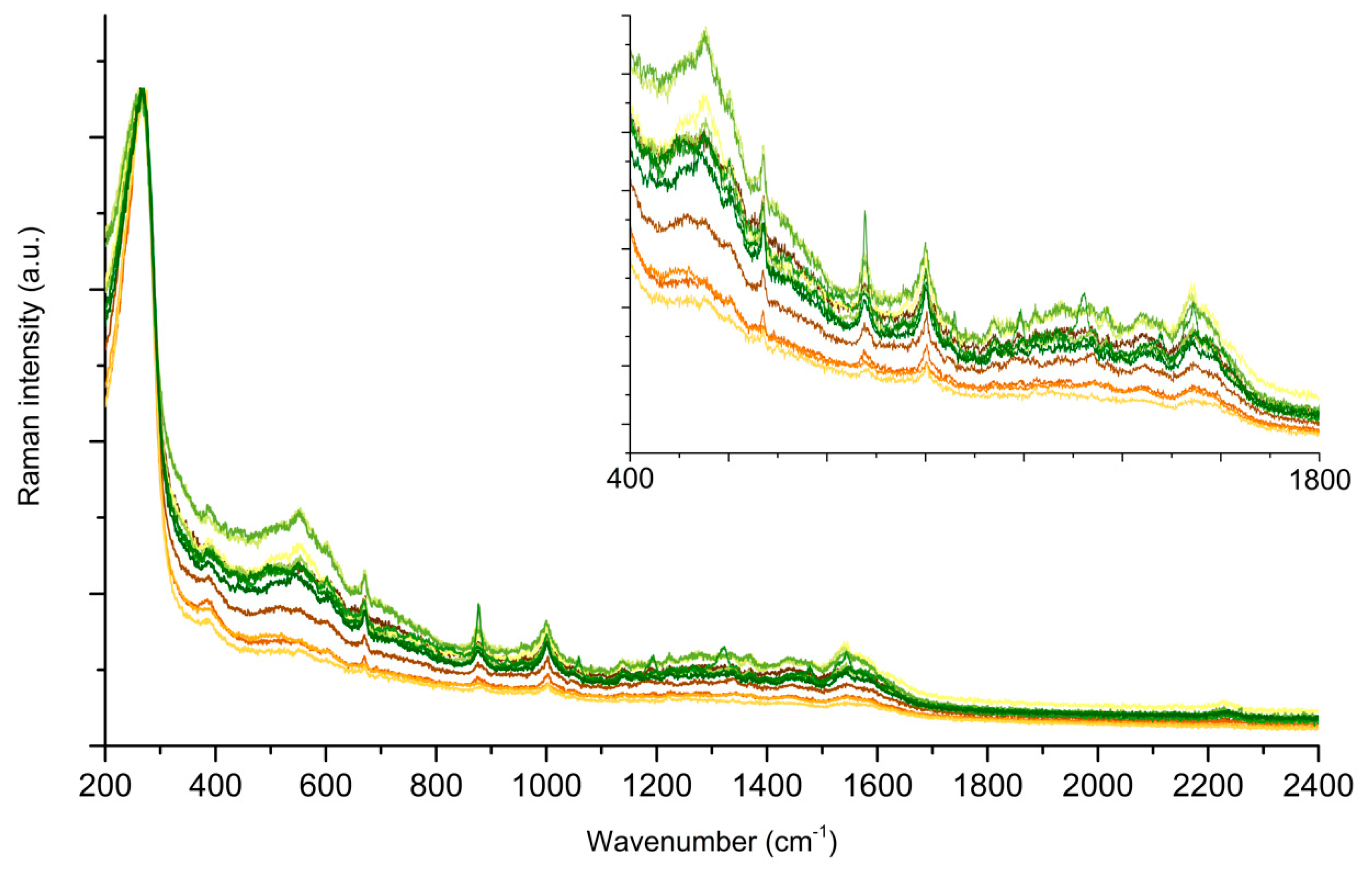
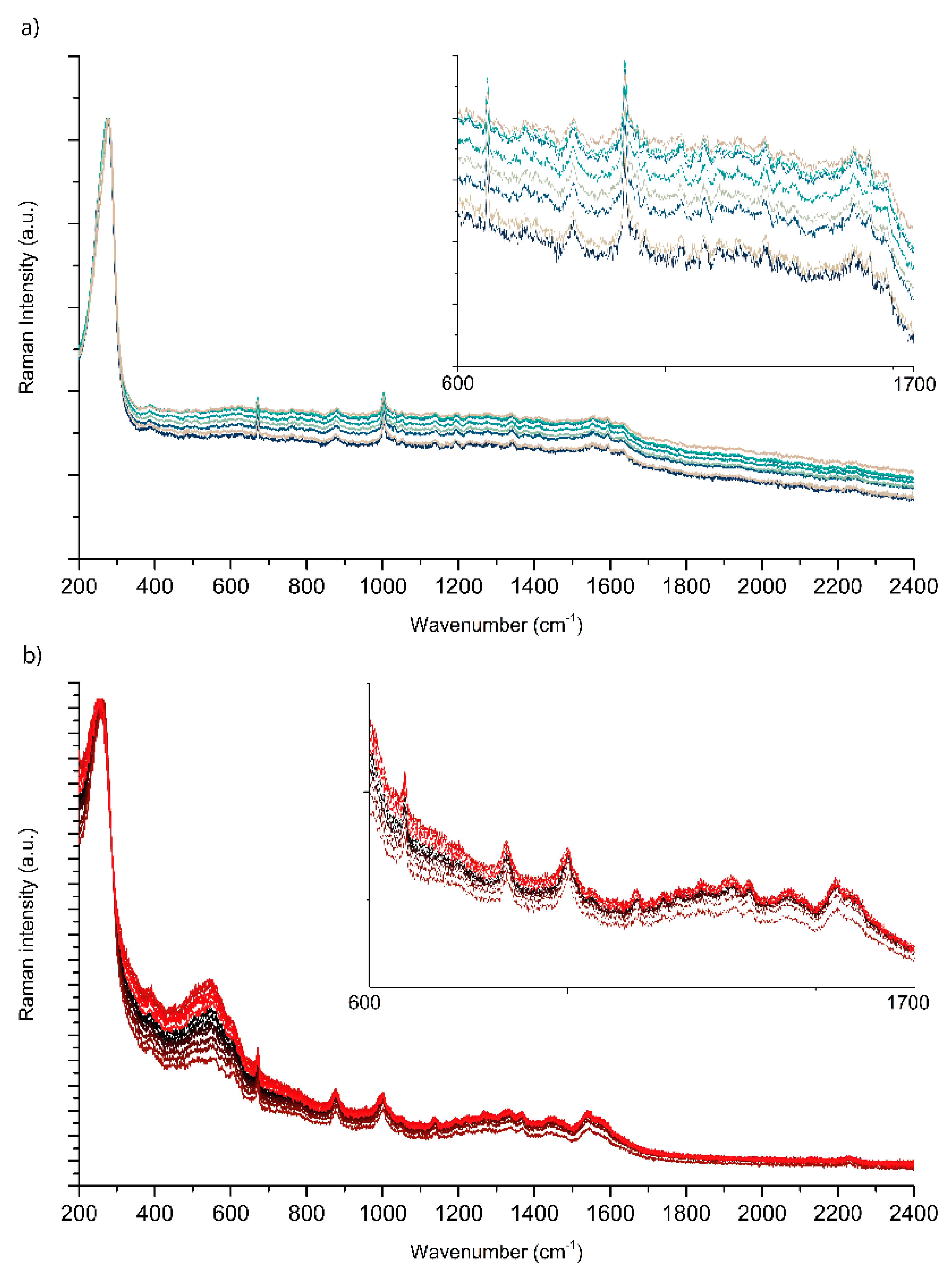
2.2. Testing the Spinning Cell on SERS of PER
3. Materials and Methods
3.1. Chemicals
3.2. Sample Preparation
- (a)
- PER was first dissolved in methanol to obtain a reference solution at 10−3 M concentration. Then, following ref. [15], this reference solution (0.3 mL) was added to an aqueous mixture of HCl and H2SO4 in a 1:9 molar ratio (2.7 mL) to reach a final pH of 2 at a 10−4 M concentration of PER. This allowed us to achieve the protonation of PER without an excess of Cl- that was shown to be detrimental to SERS (see Section 2.1). This relatively high concentration for SERS experiments (10−4 M) was selected in such a way as to monitor the evolution of the signal as a function of the tested experimental conditions in the most effective way.
- (b)
- The Au nanoparticles (NPs) were prepared as colloidal suspensions obtained by a modified Turkevich method [32], reducing HAuCl4∙3H2O with trisodium citrate (TSC). First, in a conical flask, HAuCl4∙3H2O was dissolved in 180 mL deionized water reaching a concentration of 10−3 M, which is about four times higher than the original recipe [32]. Separately, a solution of 1% wt. TSC in deionized water (9.6 mL) was prepared. The two solutions were then heated until boiling, and subsequently, they were mixed in the conical flask. The boiling mixture was kept under magnetic stirring during the formation of the gold NPs for about 60 min. The magnetic stirring was maintained during the cooling of the produced colloidal suspension. Before further use, the colloid was let naturally settle in the conical flask for about one week. To produce the SERS substrates, we withdrew the colloid from the bottom of the flask, where it was more concentrated. A representative SEM image of the gold colloid cast on a Si wafer is shown in Figure 9. The image was acquired in the NanoLab (Energy Dept., Politecnico di Milano) by a Zeiss Supra 40 field-emission scanning electron microscope (FE-SEM), operating in a high vacuum and equipped with the GEMINI column.
- (c)
- We produced SERS-active films (from here on simply denoted as SERS pads) by drop-casting controlled volumes of the Au colloid (20 μL) and letting them dry fully.
3.3. Raman Setup
3.4. Spinning Cell
3.5. Density Functional Theory Calculations
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Fornasaro, S.; Alsamad, F.; Baia, M.; de Carvalho, L.A.E.B.; Beleites, C.; Byrne, H.J.; Chiadò, A.; Chis, M.; Chisanga, M.; Daniel, A.; et al. Surface Enhanced Raman Spectroscopy for Quantitative Analysis: Results of a Large-Scale European Multi-Instrument Interlaboratory Study. Anal. Chem. 2020, 92, 4053–4064. [Google Scholar] [CrossRef]
- Alessandri, I.; Lombardi, J.R. Editorial: Surface Enhanced Raman Scattering: New Theoretical Approaches, Materials and Strategies. Front. Chem. 2020, 8, 63. [Google Scholar] [CrossRef]
- Bruni, S.; Guglielmi, V.; Pozzi, F. Historical organic dyes: A surface-enhanced Raman scattering (SERS) spectral database on Ag Lee-Meisel colloids aggregated by NaClO4. J. Raman Spectrosc. 2011, 42, 1267–1281. [Google Scholar] [CrossRef]
- Innocenti, S.; Ricci, M.; Balbas, D.Q.; Fontana, R.; Striova, J.; Becucci, M. Surface-enhanced Raman spectroscopy for madder lake detection in painting layers. Eur. Phys. J. Plus 2023, 138, 381. [Google Scholar] [CrossRef]
- Berganza, L.B.; Litti, L.; Meneghetti, M.; Lanceros-Méndez, S.; Reguera, J. Enhancement of Magnetic Surface-Enhanced Raman Scattering Detection by Tailoring Fe3O4@Au Nanorod Shell Thickness and Its Application in the On-site Detection of Antibiotics in Water. ACS Omega 2022, 7, 45493–45503. [Google Scholar] [CrossRef] [PubMed]
- Weber, V.; Brigo, L.; Brusatin, G.; Mattei, G.; Pedron, D.; Pilot, R.; Signorini, R. Hybrid Sol-Gel Surface-Enhanced Raman Sensor for Xylene Detection in Solution. Sensors 2021, 21, 7912. [Google Scholar] [CrossRef] [PubMed]
- Droghetti, E.; Nicoletti, F.P.; Guandalini, L.; Bartolucci, G.; Smulevich, G. SERS detection of benzophenones on viologen functionalized Ag nanoparticles: Application to breakfast cereals. J. Raman Spectrosc. 2013, 44, 1428–1434. [Google Scholar] [CrossRef]
- Chen, C.; Li, Y.; Kerman, S.; Neutens, P.; Willems, K.; Cornelissen, S.; Lagae, L.; Stakenborg, T.; Van Dorpe, P. High spatial resolution nanoslit SERS for single-molecule nucleobase sensing. Nat. Commun. 2018, 9, 1733. [Google Scholar] [CrossRef]
- Mao, P.; Liu, C.; Favraud, G.; Chen, Q.; Han, M.; Fratalocchi, A.; Zhang, S. Broadband single molecule SERS detection designed by warped optical spaces. Nat. Commun. 2018, 9, 5428. [Google Scholar] [CrossRef] [PubMed]
- Nie, S.; Emory, S.R. Probing Single Molecules and Single Nanoparticles by Surface-Enhanced Raman Scattering. Science 1997, 275, 1102–1106. [Google Scholar] [CrossRef]
- Kneipp, K.; Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Single Molecule Detection Using Surface-Enhanced Raman Scattering (SERS). Phys. Rev. Lett. 1997, 78, 1667–1670. [Google Scholar] [CrossRef]
- Darby, B.L.; Etchegoin, P.G.; Le Ru, E.C. Single-molecule surface-enhanced Raman spectroscopy with nanowatt excitation. Phys. Chem. Chem. Phys. 2014, 16, 23895–23899. [Google Scholar] [CrossRef]
- Pilot, R.; Signorini, R.; Durante, C.; Orian, L.; Bhamidipati, M.; Fabris, L. A Review on Surface-Enhanced Raman Scattering. Biosensors 2019, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Vo-Dinh, T.; Liu, Y.; Fales, A.M.; Ngo, H.; Wang, H.; Register, J.K.; Yuan, H.; Norton, S.J.; Griffin, G.D. SERS Nanosensors and Nanoreporters: Golden Opportunities in Biomedical Applications. WIREs Nanomed. Nanobiotechnol. 2015, 7, 17–33. [Google Scholar] [CrossRef] [PubMed]
- Tommasini, M.; Zanchi, C.; Lucotti, A.; Bombelli, A.; Villa, N.S.; Casazza, M.; Ciusani, E.; de Grazia, U.; Santoro, M.; Fazio, E.; et al. Laser-Synthesized SERS Substrates as Sensors toward Therapeutic Drug Monitoring. Nanomaterials 2019, 9, 677. [Google Scholar] [CrossRef] [PubMed]
- Buclin, T.; Thoma, Y.; Widmer, N.; André, P.; Guidi, M.; Csajka, C.; Decosterd, L. The Steps to Therapeutic Drug Monitoring: A Structured Approach Illustrated With Imatinib. Front. Pharmacol. 2020, 11, 177. [Google Scholar] [CrossRef]
- Kang, J.-S.; Lee, M.-H. Overview of Therapeutic Drug Monitoring. Korean J. Intern. Med. 2009, 24, 1–10. [Google Scholar] [CrossRef]
- Krasowski, M.D.; McMillin, G.A. Advances in anti-epileptic drug testing. Clin. Chim. Acta 2014, 436, 224–236. [Google Scholar] [CrossRef]
- Goldsmith, D.; Minassian, B.A. Efficacy and tolerability of perampanel in ten patients with Lafora disease. Epilepsy Behav. 2016, 62, 132–135. [Google Scholar] [CrossRef]
- Frampton, J.E. Perampanel: A Review in Drug-Resistant Epilepsy. Drugs 2015, 75, 1657–1668. [Google Scholar] [CrossRef]
- Satlin, A.; Kramer, L.D.; Laurenza, A. Development of perampanel in epilepsy. Acta Neurol. Scand. 2013, 127, 3–8. [Google Scholar] [CrossRef]
- Shushan, B. A review of clinical diagnostic applications of liquid chromatography–tandem mass spectrometry. Mass Spectrom. Rev. 2010, 29, 930–944. [Google Scholar] [CrossRef]
- Urban, P.L. Quantitative mass spectrometry: An overview. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2016, 374, 20150382. [Google Scholar] [CrossRef]
- Sahu, P.K.; Ramisetti, N.R.; Cecchi, T.; Swain, S.; Patro, C.S.; Panda, J. An overview of experimental designs in HPLC method development and validation. J. Pharm. Biomed. Anal. 2018, 147, 590–611. [Google Scholar] [CrossRef]
- Fornasaro, S.; Bonifacio, A.; Marangon, E.; Buzzo, M.; Toffoli, G.; Rindzevicius, T.; Schmidt, M.S.; Sergo, V. Label-Free Quantification of Anticancer Drug Imatinib in Human Plasma with Surface Enhanced Raman Spectroscopy. Anal. Chem. 2018, 90, 12670–12677. [Google Scholar] [CrossRef]
- Liu, C.; Weber, S.; Peng, R.; Wu, L.; Zhang, W.-S.; Luppa, P.B.; Popp, J.; Cialla-May, D. Toward SERS-based therapeutic drug monitoring in clinical settings: Recent developments and trends. TrAC Trends Anal. Chem. 2023, 164, 117094. [Google Scholar] [CrossRef]
- Panikar, S.S.; Cialla-May, D.; De la Rosa, E.; Salas, P.; Popp, J. Towards translation of surface-enhanced Raman spectroscopy (SERS) to clinical practice: Progress and trends. TrAC Trends Anal. Chem. 2021, 134, 116122. [Google Scholar] [CrossRef]
- Jaworska, A.; Fornasaro, S.; Sergo, V.; Bonifacio, A. Potential of Surface Enhanced Raman Spectroscopy (SERS) in Therapeutic Drug Monitoring (TDM). A Critical Review. Biosensors 2016, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, C.; MacMillan, D.; McCardle, C.; Smith, W.E. Quantitative Analysis of Mitoxantrone by Surface-Enhanced Resonance Raman Scattering. Anal. Chem. 2002, 74, 3160–3167. [Google Scholar] [CrossRef] [PubMed]
- Zanchi, C.; Lucotti, A.; Tommasini, M.; Trusso, S.; de Grazia, U.; Ciusani, E.; Ossi, P.M. Laser tailored nanoparticle arrays to detect molecules at dilute concentration. Appl. Surf. Sci. 2017, 396, 1866–1874. [Google Scholar] [CrossRef]
- Agarwal, N.; Ossi, P.; Trusso, S. Driving electromagnetic field enhancements in tailored gold surface nanostructures: Optical properties and macroscale simulations. Appl. Surf. Sci. 2019, 466, 19–27. [Google Scholar] [CrossRef]
- Turkevich, J.; Stevenson, P.C.; Hillier, J. A study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss. Faraday Soc. 1951, 11, 55–75. [Google Scholar] [CrossRef]
- Lee, P.C.; Meisel, D. Adsorption and surface-enhanced Raman of dyes on silver and gold sols. J. Phys. Chem. 1982, 86, 3391–3395. [Google Scholar] [CrossRef]
- Ye, Z.; Li, C.; Xu, Y.; Bell, S.E.J. Exploiting the chemical differences between Ag and Au colloids allows dramatically improved SERS detection of “non-adsorbing” molecules. Analyst 2019, 144, 448–453. [Google Scholar] [CrossRef]
- Kruszewski, S.; Cyrankiewicz, M. Aggregated Silver Sols as SERS Substrates. Acta Phys. Pol. A 2012, 121, 1A. [Google Scholar] [CrossRef]
- Zhu, Q.; Li, H.; Lu, F.; Chai, Y.; Yuan, Y. A Widely Applicable Silver Sol for TLC Detection with Rich and Stable SERS Features. Nanoscale Res. Lett. 2016, 11, 220. [Google Scholar] [CrossRef] [PubMed]
- Bell, S.E.J.; Sirimuthu, N.M.S. Quantitative surface-enhanced Raman spectroscopy. Chem. Soc. Rev. 2008, 37, 1012–1024. [Google Scholar] [CrossRef] [PubMed]
- Fu, H.; Bao, H.; Zhang, H.; Zhao, Q.; Zhou, L.; Zhu, S.; Wei, Y.; Li, Y.; Cai, W. Quantitative Surface-Enhanced Raman Spectroscopy for Field Detections Based on Structurally Homogeneous Silver-Coated Silicon Nanocone Arrays. ACS Omega 2021, 6, 18928–18938. [Google Scholar] [CrossRef]
- Chen, N.; Yuan, Y.; Lu, P.; Wang, L.; Zhang, X.; Chen, H.; Ma, P. Detection of carbamazepine in saliva based on surface-enhanced Raman spectroscopy. Biomed. Opt. Express 2021, 12, 7673–7688. [Google Scholar] [CrossRef] [PubMed]
- Macrelli, A.; Villa, N.S.; Lucotti, A.; Dellasega, D.; Ossi, P.M.; Tommasini, M. Sensing the Anti-Epileptic Drug Perampanel with Paper-Based Spinning SERS Substrates. Molecules 2022, 27, 30. [Google Scholar] [CrossRef]
- Cañamares, M.V.; Mieites-Alonso, M.G.; Leona, M. Raman, SERS and DFT analysis of the natural red dyes of Japanese origin alkannin and shikonin. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 265, 120382. [Google Scholar] [CrossRef]
- Steen, J.D.; Volker, A.; Duijnstee, D.R.; Sardjan, A.S.; Browne, W.R. pH-Induced Changes in the SERS Spectrum of Thiophenol at Gold Electrodes during Cyclic Voltammetry. J. Phys. Chem. C 2022, 126, 7680–7687. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, F.; Yin, H.; Hong, M. Nonuniform Distribution of Capping Ligands Promoting Aggregation of Silver Nanoparticles for Use as a Substrate for SERS. Adv. Nanoparticles 2013, 2, 104–111. [Google Scholar] [CrossRef][Green Version]
- Gong, X.; Liao, X.; Li, Y.; Cao, H.; Zhao, Y.; Li, H.; Cassidy, D.P. Sensitive detection of polycyclic aromatic hydrocarbons with gold colloid coupled chloride ion SERS sensor. Analyst 2019, 144, 6698–6705. [Google Scholar] [CrossRef] [PubMed]
- Socrates, G. Infrared and Raman Characteristic Group Frequencies: Tables and Charts; John Wiley & Sons: Hoboken, NJ, USA, 2004. [Google Scholar]
- The MathWorks Inc. Bioinformatics Toolbox Version: 4.15 (R2020b); The MathWorks Inc.: Natick, MA, USA, 2022; Available online: https://www.mathworks.com (accessed on 25 April 2023).
- Keresztury, G. Raman Spectroscopy: Theory. In Handbook of Vibrational Spectroscopy; Chalmers, J.M., Griffiths, P.R., Eds.; Wiley: Hoboken, NJ, USA, 2006. [Google Scholar] [CrossRef]
- Hahm, E.; Cha, M.G.; Kang, E.J.; Pham, X.-H.; Lee, S.H.; Kim, H.-M.; Kim, D.-E.; Lee, Y.-S.; Jeong, D.-H.; Jun, B.-H. Multilayer Ag-Embedded Silica Nanostructure as a Surface-Enhanced Raman Scattering-Based Chemical Sensor with Dual-Function Internal Standards. ACS Appl. Mater. Interfaces 2018, 10, 40748–40755. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Ding, R. Quantitative SERS Detection of Trace Glutathione with Internal Reference Embedded Au-core/Ag-shell Nanoparticles. Nano Life 2016, 6, 1642003. [Google Scholar] [CrossRef]
- Sun, L.; Zhang, M.; Natarajan, V.; Yu, X.; Zhang, X.; Zhan, J. Au@Ag core–shell nanoparticles with a hidden internal reference promoted quantitative solid phase microextraction-surface enhanced Raman spectroscopy detection. RSC Adv. 2017, 7, 23866–23874. [Google Scholar] [CrossRef]
- Lin, S.; Lin, X.; Zhao, H.; Hasi, W.; Wang, L. Quantitative SERS measurements by self-assembled ultra-smooth Au nanosphere superlattice with embedded internal reference. Appl. Phys. A 2018, 124, 796. [Google Scholar] [CrossRef]
- Zhong, Z.; Subramanian, A.S.; Highfield, J.; Carpenter, K.; Gedanken, A. From Discrete Particles to Spherical Aggregates: A Simple Approach to the Self-Assembly of Au Colloids. Chem.—A Eur. J. 2005, 11, 1473–1478. [Google Scholar] [CrossRef]
- Airy, G.B. On the Diffraction of an Object-glass with Circular Aperture. In Transactions of the Cambridge Philosophical Society; Cambridge University Press: Cambridge, UK, 1835; Volume 5, pp. 283–291. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09, Revision D.01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Grimme, S.; Ehrlich, S.; Goerigk, L. Effect of the damping function in dispersion corrected density functional theory. J. Comput. Chem. 2011, 32, 1456–1465. [Google Scholar] [CrossRef]
- Becke, A.D.; Johnson, E.R. A density-functional model of the dispersion interaction. J. Chem. Phys. 2005, 123, 154101. [Google Scholar] [CrossRef] [PubMed]
- Pracht, P.; Bohle, F.; Grimme, S. Automated exploration of the low-energy chemical space with fast quantum chemical methods. Phys. Chem. Chem. Phys. 2020, 22, 7169–7192. [Google Scholar] [CrossRef] [PubMed]

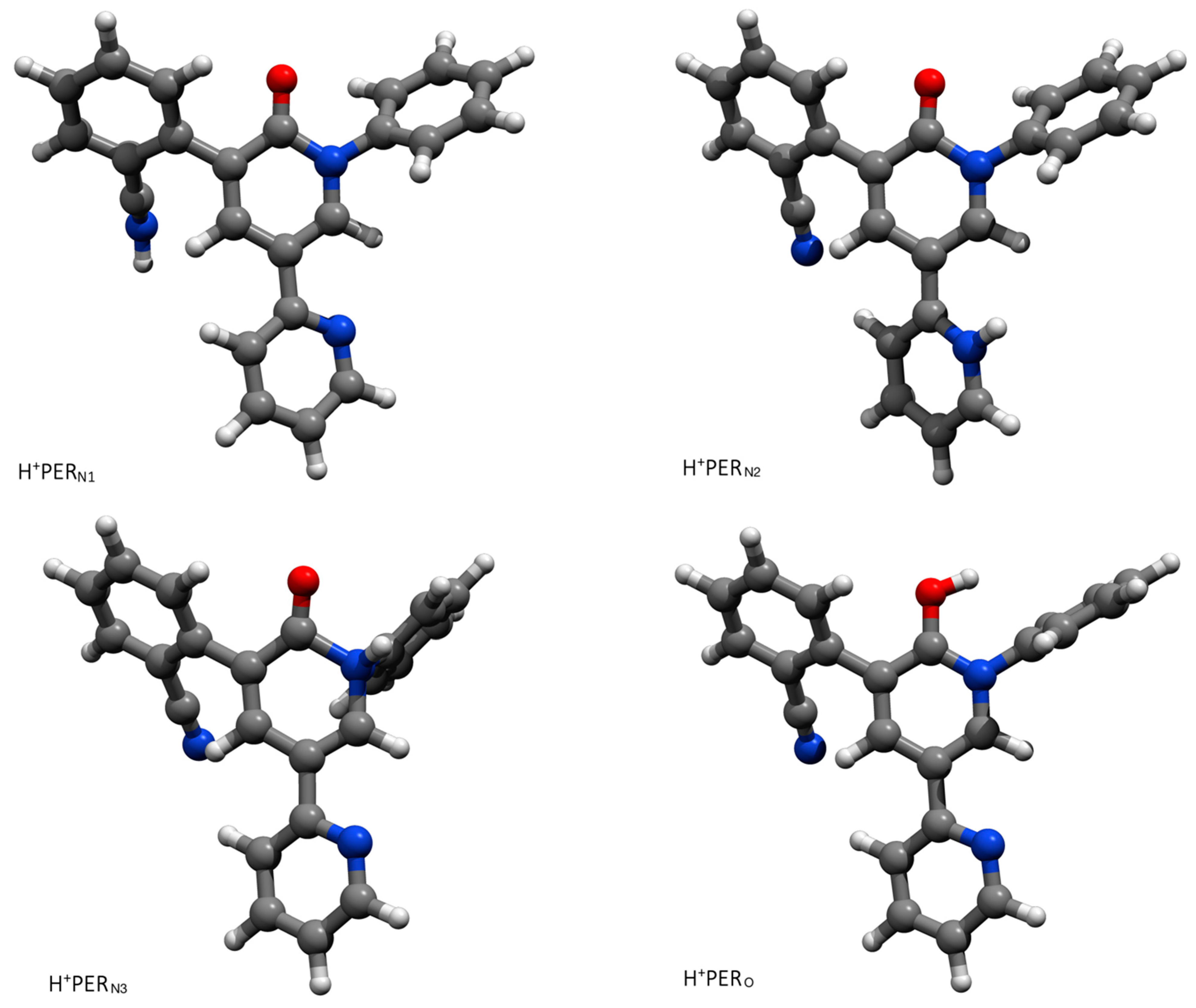


| Structure | ΔE (kcal/mol) | E (ha) |
|---|---|---|
| PER | // | −1.124985978599593 × 103 |
| H+PERN2 | −244.02 | −1.125372595588159 × 103 |
| H+PERO | −241.30 | −1.125368720495620 × 103 |
| H+PERN1 | −216.55 | −1.125330688734299 × 103 |
| H+PERN3 | −204.36 | −1.125306411724009 × 103 |
| NH3 | // | −56.55898800030168 |
| NH4+ | −218.86 | −56.90775684108217 |
| H+PERN1 | Wavenumber (cm−1) | Assignment |
|---|---|---|
| 1 | 2187 | C≡N stretching |
| 2 | 1724 | C=O stretching |
| 3, 5 | 1629, 1610 | in-plane ring deformation |
| 7, 11 | 1486, 1215 | C-N stretching |
| 12,13 | 993, 868 | ring deformation and out-of-plane C-H bending |
| H+PERN2 | ||
| 1 | 2287 | C≡N stretching |
| 2 | 1746 | C=O stretching |
| 3, 4, 5 | 1649, 1611, 1565 | in-plane ring deformation |
| 8, 11 | 1401, 1219 | C-N stretching |
| 12,13 | 992, 859 | ring deformation and out-of-plane C-H bending |
| H+PERN3 | ||
| 1 | 2292 | C≡N stretching |
| 2 | 1848 | C=O stretching |
| 3, 5 | 1677, 1603 | in-plane ring deformation |
| 7 | 1486 | C-N stretching |
| 10 | 1250 | in-plane ring deformation |
| 12 | 990 | in-plane ring deformation |
| H+PERO | ||
| 1 | 2296 | C≡N stretching |
| 3, 5, 6 | 1641, 1606, 1486 | in-plane ring deformation |
| 9 | 1251 | O-C-N stretching |
| 12 | 993 | ring deformation |
| 13 | 888 | ring deformation and out-of-plane C-H bending |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villa, N.S.; Picarelli, C.; Iacoe, F.; Zanchi, C.G.; Ossi, P.M.; Lucotti, A.; Tommasini, M. Investigating Perampanel Antiepileptic Drug by DFT Calculations and SERS with Custom Spinning Cell. Molecules 2023, 28, 5968. https://doi.org/10.3390/molecules28165968
Villa NS, Picarelli C, Iacoe F, Zanchi CG, Ossi PM, Lucotti A, Tommasini M. Investigating Perampanel Antiepileptic Drug by DFT Calculations and SERS with Custom Spinning Cell. Molecules. 2023; 28(16):5968. https://doi.org/10.3390/molecules28165968
Chicago/Turabian StyleVilla, Nicolò Simone, Chiara Picarelli, Federica Iacoe, Chiara Giuseppina Zanchi, Paolo M. Ossi, Andrea Lucotti, and Matteo Tommasini. 2023. "Investigating Perampanel Antiepileptic Drug by DFT Calculations and SERS with Custom Spinning Cell" Molecules 28, no. 16: 5968. https://doi.org/10.3390/molecules28165968
APA StyleVilla, N. S., Picarelli, C., Iacoe, F., Zanchi, C. G., Ossi, P. M., Lucotti, A., & Tommasini, M. (2023). Investigating Perampanel Antiepileptic Drug by DFT Calculations and SERS with Custom Spinning Cell. Molecules, 28(16), 5968. https://doi.org/10.3390/molecules28165968