Recent Progress in Distiller’s Grains: Chemical Compositions and Biological Activities
Abstract
1. Introduction
2. Research Progress on the Chemical Constituents of DGs
2.1. Volatile Constituents
2.2. Fatty Acids
2.3. Protein and Amino Acid Constituents
2.4. Bioactive Peptides
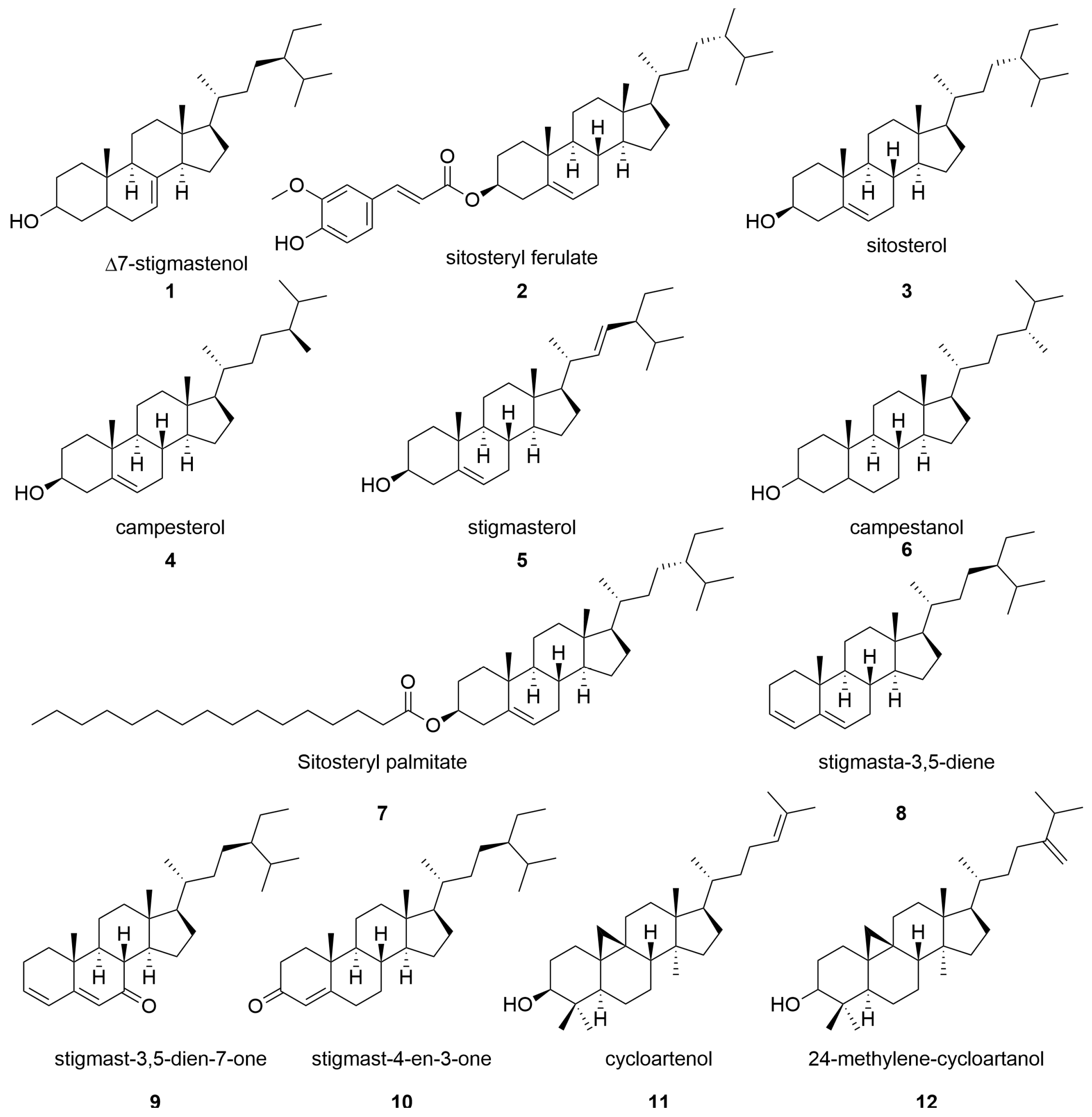
2.5. Phytosterols
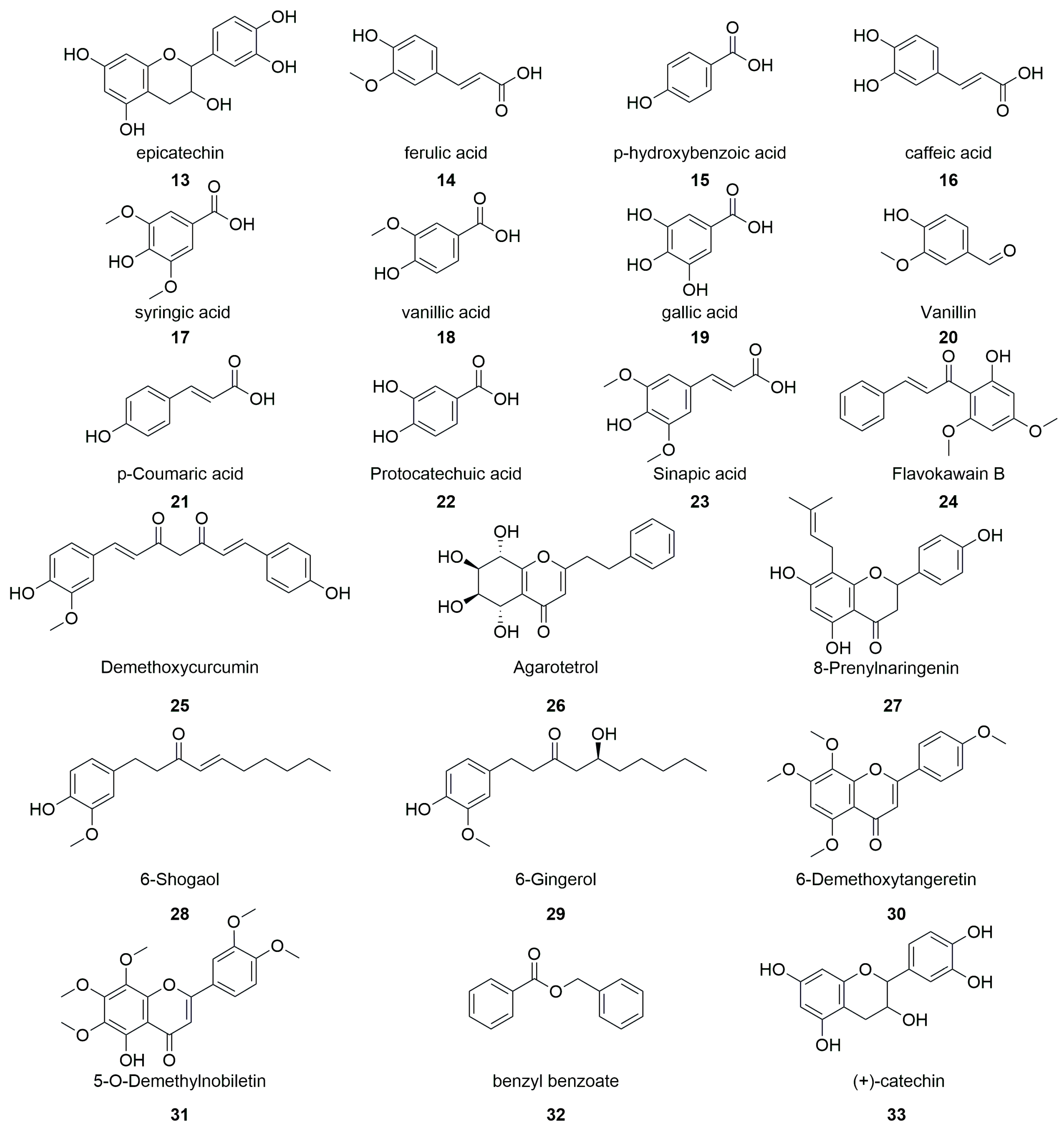
2.6. Polyphenols
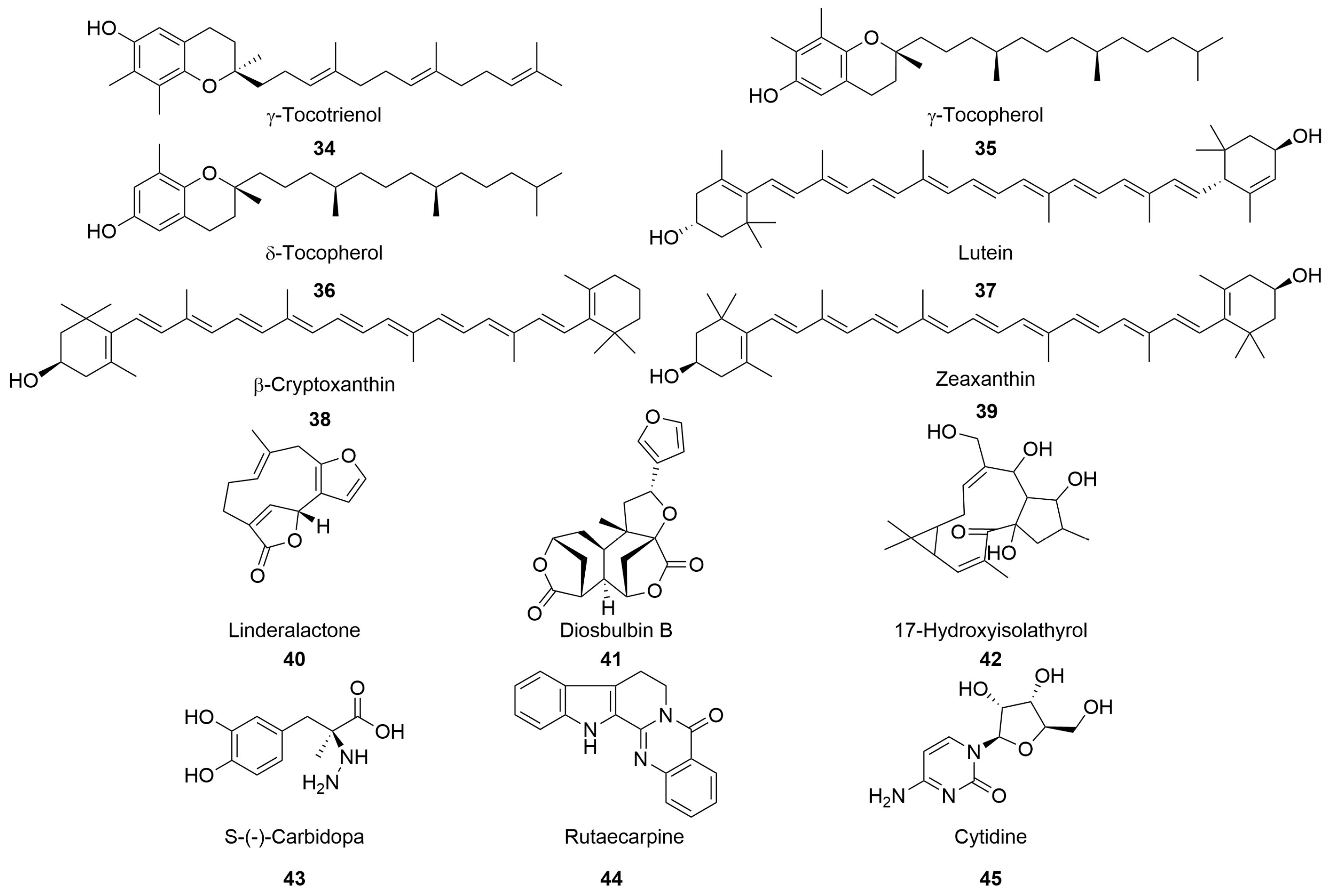
2.7. Other Constituents
3. Research Progress on the Biological Activity of DGs
3.1. Antioxidant Activity
3.2. Anti-Inflammatory Activity
| DG Type | Constituents | Test Methods | Inhibitory Concentration | Ref. |
|---|---|---|---|---|
| Baijiu DG | Bioactive peptide VNP | DPPH | 0.10 μmol TE/μmol | [25] |
| Bioactive peptide YGD | ABTS | 0.95 μmol TE/μmol | ||
| Baijiu DG | Polysaccharides | DPPH | IC50 of extruded sample: 9.8 mg/mL IC50 of unextruded sample: 11.12 mg/mL | [46] |
| ABTS | IC50 of extruded sample: 51.39 mg/mL IC50 of unextruded sample: 156.21 mg/mL | |||
| •OH radical scavenging | IC50 of extruded sample: 0.16 mg/mL IC50 of unextruded sample: 0.32 mg/mL | |||
| Rice wine lees | Polyphenols | DPPH | 86% at 1.4 mg/mL (soxhlet 95% ethanol extract) | [51] |
| Ferrous chelating ability | 91% at 1 mg/mL (soxhlet 95% ethanol extract) | |||
| Reducing ability | 1.877 OD at 1.25 mg/mL (soxhlet 95% ethanol extract) | |||
| BSG | Phenols | DPPH | 1.28 mg TE/g of 60% (v/v) acetone extract 0.85 mg TE/g of 60% (v/v) ethanol extract | [52] |
| BSG | Protein hydrolysate | SOD | 0.5 U/mg protein BSG against DNA damage in U937 cells under oxidative stress conditions | [53] |
| BSG | Phenolic acids | ABTS | 0.77 mg ascorbic acid/g BSG (60% ethanol extract) | [37] |
| ORAC | 24.80 mg TE/g BSG (80% ethanol extract) | |||
| Sorghum spent grains | Protein hydrolysate | ABTS | 15.49% at 0.5 mg protein/mL | [26] |
| DDGS | Phenols | DPPH | 1.49~6.53 μmol TE/g | [54] |
| Liquor DG | Phenols | DPPH | IC50 of 60% ethanol extract: 33.3 µg/mL | [55] |
| ABTS | IC50 of 60% ethanol extract: 22.1 µg/mL | |||
| Baijiu DG | Polyphenols | DPPH | IC50 of purified extract: 34.03 μg/mL IC50 of unpurified extract: 16.21 μg/mL | [36] |
| ABTS | IC50 of unpurified extract: 20.31 μg/mL IC50 of unpurified extract: 5.73 μg/mL | |||
| Baijiu DG | Flavonoids | DPPH | 62.3% at 79 mg/mL (methanol extract) | [38] |
| Baijiu DG | Phenols and anthocyanins | DPPH | Above 70% at 1 mg/mL (red yeast rice DG) Above 40% at 1 mg/mL (sorghum DG) | [56] |
| Dried DSG | Phenols | ABTS | 0.32 and 0.44 mg TE/g of aqueous extract 0.54~0.57 mg TE/g of ethanolic extract | [57] |
| Baijiu DG | Polyphenols | FRAP | 2.60~2.61 mg TE/mL of free polyphenols 0.19~0.22 mg TE/mL of bound polyphenols | [58] |
| ABTS | 7.11~7.38 mg TE/mL of free polyphenols 2.22~3.33 mg TE/mL of bound polyphenols | |||
| DPPH | 5.46~5.71 mg TE/mL of free polyphenols 0.79~1.85 mg TE/mL of bound polyphenols | |||
| Baijiu DG | Polyphenols | DPPH | 64.37% (70% acetone vinasse extract) | [59] |
| FRAP | 4.63 mM FE(II)/g of 70% acetone vinasse extract |
3.3. Antihypertensive Activity
3.4. Antihyperglycaemic Activity
3.5. Other Biological Activities
4. Conclusions and Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, Y.; Liu, S.; Huang, C.; Ge, X.; Xi, B.; Mao, J. Chinese Baijiu distiller’s grains resourcing: Current progress and future prospects. Resour. Conserv. Recycl. 2022, 176, 105900. [Google Scholar] [CrossRef]
- Buenavista, R.M.E.; Siliveru, K.; Zheng, Y. Utilization of Distiller’s dried grains with solubles: A review. J. Agric. Food Res. 2021, 5, 100195. [Google Scholar] [CrossRef]
- Qian, Y.; Zhang, L.; Sun, Y.; Tang, Y.; Li, D.; Zhang, H.; Yuan, S.; Li, J. Differentiation and classification of Chinese Luzhou-flavor liquors with different geographical origins based on fingerprint and chemometric analysis. J. Food Sci. 2021, 86, 1861–1877. [Google Scholar] [CrossRef]
- Liu, K. Chemical Composition of Distillers Grains, a Review. J. Agric. Food. Chem. 2011, 59, 1508–1526. [Google Scholar] [CrossRef] [PubMed]
- Jose Jara-Palacios, M. Wine Lees as a Source of Antioxidant Compounds. Antioxidants 2019, 8, 45. [Google Scholar] [CrossRef] [PubMed]
- Li, F.X.; Zhang, W.; Shi, W.; Yu, J.S.; Yue, Q.Q.; Yu, J.P.; Wang, X.Y. GC-MS analysis and application of sauce-flavor distilled grains extract. Chin. Brew. 2021, 40, 159–164. [Google Scholar]
- Zhang, S.X.; Jin, Q.; Zeng, Q.H.; Yu, J.S.; Yue, Q.Q.; Yu, J.P.; Wang, X.Y. Analysis of aromatic compounds in sauce-flavor Moutai lees by GC-MS. Chin. Brew. 2012, 31, 188–189. [Google Scholar]
- Wang, X.D.; Qiu, S.Y.; Gao, W.T.; Zhou, H.X.; Luo, X.Y.; Ban, S.D. Preliminary analysis of quality and volatile components of sauce-flavor distiller’s grains before and after fermentation. Chin. Brew. 2021, 40, 97–101. [Google Scholar]
- Lei, X.J.; Yang, K.Z.; Zhang, J.M.; Zhang, X.; Luo, Q.C.; Qiao, Z.W.; Zhao, D.; Zheng, D. Spatial distribution of aroma compounds in fermented grains of multi-grains strong-aroma Baijiu. Food. Ferment. Ind. 2020, 46, 48–54. [Google Scholar]
- Jiang, S.Y.; Song, H.; Chen, C.; Zheng, Y.Z. Analysis of Organic Acids and Flavoring Compositions Extracted from Distiller’s Grains. Food. Ind. Sci. 2019, 40, 206–211. [Google Scholar]
- Ye, X.; Leng, Y.W.; Chen, X.; Li, H.; Kong, X.Y. GC Analysis of leaching liquid of distiller’s grains. Liquor-Mak. Sci. Technol. 2011, 6, 100–102. [Google Scholar]
- Wang, X.; Zhou, J.; Ming, H.M.; Zhang, S.Y.; Li, D.L.; Luo, J.; Yu, F. The quality and aroma components of distiller’s grain in mechanized and manual brewing methods. Food. Ferment. Ind. 2020, 46, 210–216. [Google Scholar]
- Roth, M.; Meiringer, M.; Kollmannsberger, H.; Zarnkow, M.; Jekle, M.; Becker, T. Characterization of Key Aroma Compounds in Distiller’s Grains from Wheat as a Basis for Utilization in the Food Industry. J. Agric. Food Chem. 2014, 62, 10873–10880. [Google Scholar] [CrossRef]
- Gomez, M.E.; Igartuburu, J.M.; Pando, E.; Luis, F.R.; Mourente, G. Lipid composition of lees from sherry wine. J. Agric. Food. Chem. 2004, 52, 4791–4794. [Google Scholar] [CrossRef] [PubMed]
- Fărcaş, A.C.; Socaci, S.A.; Dulf, F.V.; Tofană, M.; Mudura, E.; Diaconeasa, Z. Volatile profile, fatty acids composition and total phenolics content of brewers’ spent grain by-product with potential use in the development of new functional foods. J. Cereal Sci. 2015, 64, 34–42. [Google Scholar] [CrossRef]
- Hwang, K.T.; Weller, C.L.; Cuppett, S.L.; Hanna, M.A. Policosanol Contents and Composition of Grain Sorghum Kernels and Dried Distillers Grains. Cereal Chem. 2004, 81, 345–349. [Google Scholar] [CrossRef]
- Wang, L.; Weller, C.L.; Schlegel, V.L.; Carr, T.P.; Cuppett, S.L. Comparison of supercritical CO2 and hexane extraction of lipids from sorghum distillers grains. Eur. J. Lipid Sci. Technol. 2010, 109, 567–574. [Google Scholar] [CrossRef]
- Larson, E.M.; Stock, R.A.; Klopfenstein, T.J.; Sindt, M.H.; Huffman, R.P. Feeding value of wet distillers byproducts for finishing ruminants. J. Anim. Sci. 1993, 71, 2228–2236. [Google Scholar] [CrossRef]
- Cromwell, G.L.; Herkelman, K.L.; Stahly, T.S. Physical, chemical, and nutritional characteristics of distillers dried grains with solubles for chicks and pigs. J. Anim. Sci. 1993, 71, 679–686. [Google Scholar] [CrossRef][Green Version]
- Spiehs, M.J.; Whitney, M.H.; Shurson, G.C. Nutrient database for distiller’s dried grains with solubles produced from new ethanol plants in Minnesota and South Dakota. J. Anim. Sci. 2002, 80, 2639–2645. [Google Scholar]
- Kim, Y.; Mosier, N.S.; Hendrickson, R.; Ezeji, T.; Blaschek, H.; Dien, B.; Cotta, M.; Dale, B.; Ladisch, M.R. Composition of corn dry-grind ethanol by-products: DDGS, wet cake, and thin stillage. Bioresour. Technol. 2008, 99, 5165–5176. [Google Scholar] [CrossRef] [PubMed]
- Böttger, C.; Südekum, K.H. Review: Protein value of distillers dried grains with solubles (DDGS) in animal nutrition as affected by the ethanol production process. Anim. Feed Sci. Technol. 2018, 244, 11–17. [Google Scholar] [CrossRef]
- Shen, Y.; Huang, W.Q.; Luo, H. Functions of Bioactive Peptides and Their Relationship with Liquor-Making. Wine Sci. Technol. 2022, 1, 91–96. [Google Scholar]
- Liu, X.G.; Chang, R.; Zhou, Z.L. Conversion of Baijiu distillers’ grains to functional peptides: Process optimization and antioxidant activity evaluation. J. Funct. Foods 2023, 108, 105722. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhao, D.; Sun, J.; Luo, X.; Li, H.; Sun, X.; Zheng, F. Analysis of antioxidant effect of two tripeptides isolated from fermented grains (Jiupei) and the antioxidative interaction with 4-methylguaiacol, 4-ethylguaiacol, and vanillin. Food Sci. Nutr. 2019, 7, 2391–2403. [Google Scholar] [CrossRef] [PubMed]
- Guadalupe Garzon, A.; Esteban Cian, R.; Estefania Aquino, M.; Rosa Drago, S. Isolation and identification of cholesterol esterase and pancreatic lipase inhibitory peptides from brewer’s spent grain by consecutive chromatography and mass spectrometry. Food Funct. 2020, 11, 4994–5003. [Google Scholar] [CrossRef] [PubMed]
- Wei, D.; Fan, W.L.; Xu, Y. Identification of water-soluble peptides in distilled spent grain and its angiotensin converting enzyme (ACE) inhibitory activity based on UPLC-Q-TOF-MS and proteomics analysis. Food Chem. 2021, 353, 129521. [Google Scholar] [CrossRef]
- Liao, Q.J.; An, M.Z.; Li, Y.H.; Zhou, H.L.; Wang, X.Q.; Wang, F.; Guo, Y. Study of Dipeptide and Cyclodipeptide in Yellow Water of Distillers Grains. Liquor-Mak. Sci. Technol. 2018, 292, 17–23. [Google Scholar]
- Garzón, A.G.; Veras, F.F.; Brandelli, A.; Drago, S.R. Purification, identification and in silico studies of antioxidant, antidiabetogenic and antibacterial peptides obtained from sorghum spent grain hydrolysate. Food Sci. Technol. 2021, 153, 112414. [Google Scholar] [CrossRef]
- Zhang, R.; Han, Y.; McClements, D.J.; Xu, D.; Chen, S. Production, Characterization, Delivery, and Cholesterol-Lowering Mechanism of Phytosterols: A Review. J. Agric. Food Chem. 2022, 70, 2483–2494. [Google Scholar] [CrossRef]
- Winkler, J.K.; Rennick, K.A.; Eller, F.J.; Vaughn, S.F. Phytosterol and Tocopherol Components in Extracts of Corn Distiller’s Dried Grain. J. Agric. Food Chem. 2007, 55, 6482–6486. [Google Scholar] [CrossRef] [PubMed]
- del Río, J.C.; Prinsen, P.; Gutiérrez, A. Chemical composition of lipids in brewer’s spent grain: A promising source of valuable phytochemicals. J. Cereal Sci. 2013, 58, 248–254. [Google Scholar] [CrossRef]
- Winkler-Moser, J.K.; Vaughn, S.F. Antioxidant Activity of Phytochemicals from Distillers Dried Grain Oil. J. Am. Oil Chem. Soc. 2009, 86, 1073–1082. [Google Scholar] [CrossRef]
- Leguizamón, C.; Weller, C.L.; Schlegel, V.L.; Carr, T.P. Plant Sterol and Policosanol Characterization of Hexane Extracts from Grain Sorghum, Corn and their DDGS. J. Am. Oil Chem. Soc. 2009, 86, 707–716. [Google Scholar] [CrossRef]
- Wang, G.L.; Song, J.M.; Xu, W.F.; Zhao, E.L. Effects of Pressure on Extraction Rate and Antioxidant Activity of Polyphenols from Ginger. Chin. Brew. 2020, 45, 63–65. [Google Scholar]
- Wang, X.; Wang, S.; Huang, S.; Zhang, L.; Ge, Z.; Sun, L.; Zong, W. Purification of Polyphenols from Distiller’s Grains by Macroporous Resin and Analysis of the Polyphenolic Components. Molecules 2019, 24, 1284. [Google Scholar] [CrossRef]
- Bonifacio-Lopes, T.; Vilas Boas, A.A.; Coscueta, E.R.; Costa, E.M.; Silva, S.; Campos, D.; Teixeira, J.A.; Pintado, M. Bioactive extracts from brewer’s spent grain. Food Funct. 2021, 12, 12162, Erratum in Food Funct. 2020, 11, 8963–8977. [Google Scholar]
- Yang, J.; Zhang, Z.; Ding, X.; Chen, X.; Yin, C.; Yang, E.; Sun, D.; Wang, W.; Guo, F. Multiple responses optimization of antioxidative components extracted from distiller’s grains using response surface methodology and identify their chemical compositions. J. Food Process. Preserv. 2021, 45, 3475–3484. [Google Scholar] [CrossRef]
- Wang, J.H.; Zhang, Z.H. Extraction and chemical composition of soluble dietary fiber from Beer’s spent grains. Food Sci. Technol. 2002, 11, 64–67. [Google Scholar]
- Maphosa, Y.; Jideani, V.A. Dietary fiber extraction for human nutrition—A review. Food Rev. Int. 2016, 32, 98–115. [Google Scholar] [CrossRef]
- Dahl, W.J.; Stewart, M.L. Position of the Academy of Nutrition and Dietetics: Health Implications of Dietary Fiber. J. Acad. Nutr. Diet. 2015, 115, 1861–1870. [Google Scholar] [CrossRef]
- Wang, X.Q.; Wang, W.; Peng, M.; Zhang, X.Z. Free radicals for cancer theranostics. Biomaterials 2021, 266, 120474. [Google Scholar] [CrossRef] [PubMed]
- Ziada, A.S.; Smith, M.S.R.; Cote, H.C.F. Updating the Free Radical Theory of Aging. Front. Cell Dev. Biol. 2020, 8, 575645. [Google Scholar] [CrossRef]
- Zaric, B.L.; Macvanin, M.T.; Isenovic, E.R. Free radicals: Relationship to Human Diseases and Potential Therapeutic applications. Int. J. Biochem. Cell Biol. 2023, 154, 106346. [Google Scholar] [CrossRef] [PubMed]
- Baenas, N.; García-Viguera, C.; Domínguez-Perles, R.; Medina, S. Winery By-Products as Sources of Bioactive Tryptophan, Serotonin, and Melatonin: Contributions to the Antioxidant Power. Foods 2023, 12, 1571. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, X.; Qin, H.; Huang, M.; Liu, S.; Chang, R.; Xi, B.; Mao, J.; Zhang, S. Obtaining non-digestible polysaccharides from distillers’ grains of Chinese baijiu after extrusion with enhanced antioxidation capability. Int. J. Biol. Macromol. 2023, 243, 124799. [Google Scholar] [CrossRef]
- Naziri, E.; Glisic, S.B.; Mantzouridou, F.T.; Tsimidou, M.Z.; Nedovic, V.; Bugarski, B. Advantages of supercritical fluid extraction for recovery of squalene from wine lees. J. Supercrit. Fluids. 2016, 107, 560–565. [Google Scholar] [CrossRef]
- Murakami, A.; Nakamura, Y.; Koshimizu, K.; Takahashi, D.; Matsumoto, K.; Hagihara, K.; Taniguchi, H.; Nomura, E.; Hosoda, A.; Tsuno, T. FA15, a hydrophobic derivative of ferulic acid, suppresses inflammatory responses and skin tumor promotion: Comparison with ferulic acid. Cancer Lett. 2002, 180, 121–129. [Google Scholar] [CrossRef]
- Peng, L.; Kong, X.; Wang, Z.; Ai-Lati, A.; Mao, J. Baijiu vinasse as a new source of bioactive peptides with antioxidant and anti-inflammatory activity. Food Chem. 2021, 339, 128159. [Google Scholar] [CrossRef]
- Crowley, D.; O’Callaghan, Y.; McCarthy, A.; Connolly, A.; Piggott, C.O.; FitzGerald, R.J.; O’Brien, N.M. Immunomodulatory potential of a brewers’ spent grain protein hydrolysate incorporated into low-fat milk following in vitro gastrointestinal digestion. Int. J. Food Sci. Nutr. 2015, 66, 672–676. [Google Scholar] [CrossRef]
- Wu, J.J.; Lin, J.C.; Wang, C.H.; Jong, T.T.; Yang, H.L.; Hsu, S.L.; Chang, C.M.J. Extraction of antioxidative compounds from wine lees using supercritical fluids and associated anti-tyrosinase activity. J. Supercrit. Fluids 2009, 50, 33–41. [Google Scholar] [CrossRef]
- Zuorro, A.; Iannone, A.; Lavecchia, R. Water-Organic Solvent Extraction of Phenolic Antioxidants from Brewers’ Spent Grain. Processes 2019, 7, 126. [Google Scholar] [CrossRef]
- McCarthy, A.L.; O’Callaghan, Y.C.; Connolly, A.; Piggott, C.O.; FitzGerald, R.J.; O’Brien, N.M. Brewers’ spent grain (BSG) protein hydrolysates decrease hydrogen peroxide (H2O2)-induced oxidative stress and concanavalin-A (con-A) stimulated IFN-γ production in cell culture. Food Funct. 2013, 4, 1709–1716. [Google Scholar] [CrossRef] [PubMed]
- Inglett, G.E.; Rose, D.J.; Stevenson, D.G.; Chen, D.; Biswas, A. Total Phenolics and Antioxidant Activity of Water and Ethanolic Extracts from Distillers Dried Grains with Solubles with or without Microwave Irradiation. Cereal Chem. 2009, 86, 661–664. [Google Scholar] [CrossRef]
- Wang, X.Y.; Ding, J.H.; Zhang, Y.Y. Ultrasonic-assisted extraction and antioxidant activity evaluation of polyphenols from liquor distiller’s grains. Food Sci. Technol. 2018, 43, 192–197. [Google Scholar]
- Lu, Q.; Luo, Q.; Li, J.; Wang, X.; Ban, C.; Qin, J.; Tian, Y.; Tian, X.; Chen, X. Evaluation of the Chemical Composition, Bioactive Substance, Gas Production, and Rumen Fermentation Parameters of Four Types of Distiller’s Grains. Molecules 2022, 27, 6134. [Google Scholar] [CrossRef]
- Cenkowski, S.; Sosa-Morales, M.E.; Flores-Alvarez, M.D.C. Protein Content and Antioxidant Activity of Distillers’ Spent Grain Dried at 150 °C with Superheated Steam and Hot Air. Drying Technol. 2012, 30, 1292–1296. [Google Scholar] [CrossRef]
- Zhou, D.; Zhong, J.; Huang, Y.; Cheng, Y. Effect of free and bound polyphenols from Rosa roxburghii Tratt distiller’s grains on moderating fecal microbiota. Food Chem. 2023, 19, 100747. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, H.; Zhang, D.; Liu, J.; Wang, J.; Wang, S.; Sun, B. Baijiu Vinasse Extract Scavenges Glyoxal and Inhibits the Formation of Nε-Carboxymethyllysine in Dairy Food. Molecules 2019, 24, 1526. [Google Scholar] [CrossRef]
- Cian, R.E.; Hernández-Chirlaque, C.; Gámez-Belmonte, R.; Drago, S.R.; Sánchez de Medina, F.; Martínez-Augustin, O. Molecular action mechanism of anti-inflammatory hydrolysates obtained from brewers’ spent grain. J. Sci. Food Agric. 2020, 100, 2880–2888. [Google Scholar] [CrossRef]
- Wu, J.; Liao, W.; Udenigwe, C.C. Revisiting the mechanisms of ACE inhibitory peptides from food proteins. Trends Food Sci. Technol. 2017, 69, 214–219. [Google Scholar] [CrossRef]
- Zhang, L.; Jiang, A.Y.; Yin, B.Z.; Sun, B.J.; Li, A.H. Isolation and evaluation of two angiotensin-I-converting enzyme inhibitory peptides from fermented grains (Jiupei) used in Chinese Baijiu production. RSC Adv. 2018, 8, 37451–37461. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Liu, G.; Qiao, Z.; Cao, Y.; Song, M. Novel Angiotensin-I Converting Enzyme Inhibitory Peptides Isolated From Rice Wine Lees: Purification, Characterization, and Structure-Activity Relationship. Front. Nutr. 2021, 8, 746113. [Google Scholar] [CrossRef] [PubMed]
- López-Fernández-Sobrino, R.; Soliz-Rueda, J.R.; vila-Román, J.; Arola-Arnal, A.; Suárez, M.; Muguerza, B.; Bravo, F.I. Blood Pressure-Lowering Effect of Wine Lees Phenolic Compounds Is Mediated by Endothelial-Derived Factors: Role of Sirtuin. Antioxidants 2021, 10, 1073. [Google Scholar] [CrossRef]
- Muguerza, B. ACE Inhibitory and Antihypertensive Activities of Wine Lees and Relationship among Bioactivity and Phenolic Profile. Nutrients 2021, 13, 679. [Google Scholar]
- López-Fernández-Sobrino, R.; Margalef, M.; Torres-Fuentes, C.; Ávila-Román, J.; Aragonès, G.; Muguerza, B.; Bravo, F.I. Enzyme-Assisted Extraction to Obtain Phenolic-Enriched Wine Lees with Enhanced Bioactivity in Hypertensive Rats. Antioxidants 2021, 10, 517. [Google Scholar] [CrossRef]
- Isabel Bravo, F.; Mas-Capdevila, A.; Lopez-Fernandez-Sobrino, R.; Torres-Fuentes, C.; Mulero, M.; Maria Alcaide-Hidalgo, J.; Muguerza, B. Identification of novel antihypertensive peptides from wine lees hydrolysate. Food Chem. 2022, 366, 130690. [Google Scholar] [CrossRef]
- Ribeiro-Oliveira, R.; Martins, Z.E.; Faria, M.A.; Sousa, J.B.; Ferreira, I.M.P.L.V.O.; Diniz, C. Protein Hydrolysates from Brewing By-Products as Natural Alternatives to ACE-Inhibitory Drugs for Hypertension Management. Life 2022, 12, 1554. [Google Scholar] [CrossRef]
- Garzon, A.G.; Cian, R.E.; Drago, S.R. Effects of agar-carrageenan wall materials and core-to-wall material ratio on physicochemical properties and in vitro bioaccessibility of microencapsulated bioactive peptides. Food Hydrocoll. 2023, 139, 108570. [Google Scholar] [CrossRef]
- Cermeno, M.; Connolly, A.; O’Keeffe, M.B.; Flynn, C.; Alashi, A.M.; Aluko, R.E.; FitzGerald, R.J. Identification of bioactive peptides from brewers’ spent grain and contribution of Leu/Ile to bioactive potency. J. Funct. Foods 2019, 60, 103455. [Google Scholar] [CrossRef]
- Nongonierma, A.B.; FitzGerald, R.J. Features of dipeptidyl peptidase IV (DPP-IV) inhibitory peptides from dietary proteins. J. Food Biochem. 2019, 43, e12451. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, T.; Uraji, M.; Fujita, A.; Kawakami, K. Anti-oxidation Activities of Rice-Derived Peptides and Their Inhibitory Effects on Dipeptidylpeptidase-IV. Int. J. Pept. Res. Ther. 2015, 21, 479–485. [Google Scholar] [CrossRef]
- Connolly, A.; Cermeno, M.; Crowley, D.; O’Callaghan, Y.; O’Brien, N.M.; FitzGerald, R.J. Characterisation of the in vitro bioactive properties of alkaline and enzyme extracted brewers’ spent grain protein hydrolysates. Food Res. Int. 2019, 121, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Connolly, A.; O’Keeffe, M.B.; Nongonierma, A.B.; Piggott, C.O.; FitzGerald, R.J. Isolation of peptides from a novel brewers spent grain protein isolate with potential to modulate glycaemic response. Int. J. Food Sci. Technol. 2017, 52, 146–153. [Google Scholar] [CrossRef]
- Kawakami, K.; Moritani, C.; Uraji, M.; Fujita, A.; Kawakami, K.; Hatanaka, T.; Suzaki, E.; Tsuboi, S. Sake lees hydrolysate protects against acetaminophen-induced hepatotoxicity via activation of the Nrf2 antioxidant pathway. J. Clin. Biochem. Nutr. 2017, 61, 203–209. [Google Scholar] [CrossRef]
- Jeon, H.J.; Noda, M.; Maruyama, M.; Matoba, Y.; Kumagai, T.; Sugiyama, M. Identification and kinetic study of tyrosinase inhibitors found in sake lees. J. Agric. Food Chem. 2006, 54, 9827–9833. [Google Scholar] [CrossRef]
- Tagkouli, D.; Tsiaka, T.; Kritsi, E.; Sokovic, M.; Sinanoglou, V.J.; Lantzouraki, D.Z.; Zoumpoulakis, P. Towards the Optimization of Microwave-Assisted Extraction and the Assessment of Chemical Profile, Antioxidant and Antimicrobial Activity of Wine Lees Extracts. Molecules 2022, 27, 2189. [Google Scholar] [CrossRef]
- Cian, R.E.; Garzon, A.G.; Martinez-Augustin, O.; Botto, C.C.; Drago, S.R. Antithrombotic Activity of Brewers’ Spent Grain Peptides and their Effects on Blood Coagulation Pathways. Plant Foods Hum. Nutr. 2018, 73, 241–246. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, R.; Yang, Y.; Li, Y.; Yang, M.; Kong, L.; Yang, F. Recent Progress in Distiller’s Grains: Chemical Compositions and Biological Activities. Molecules 2023, 28, 7492. https://doi.org/10.3390/molecules28227492
He R, Yang Y, Li Y, Yang M, Kong L, Yang F. Recent Progress in Distiller’s Grains: Chemical Compositions and Biological Activities. Molecules. 2023; 28(22):7492. https://doi.org/10.3390/molecules28227492
Chicago/Turabian StyleHe, Ran, Yubo Yang, Yongsu Li, Minghua Yang, Lingyi Kong, and Fan Yang. 2023. "Recent Progress in Distiller’s Grains: Chemical Compositions and Biological Activities" Molecules 28, no. 22: 7492. https://doi.org/10.3390/molecules28227492
APA StyleHe, R., Yang, Y., Li, Y., Yang, M., Kong, L., & Yang, F. (2023). Recent Progress in Distiller’s Grains: Chemical Compositions and Biological Activities. Molecules, 28(22), 7492. https://doi.org/10.3390/molecules28227492






