Caffeic Acid in Spent Coffee Grounds as a Dual Inhibitor for MMP-9 and DPP-4 Enzymes
Abstract
:1. Introduction
2. Results and Discussion
2.1. DPP-4 and MMP-9 Inhibitory Assay
2.2. Molecular Docking Simulations of Caffeic Acid Targeting DPP-4 and MMP-9
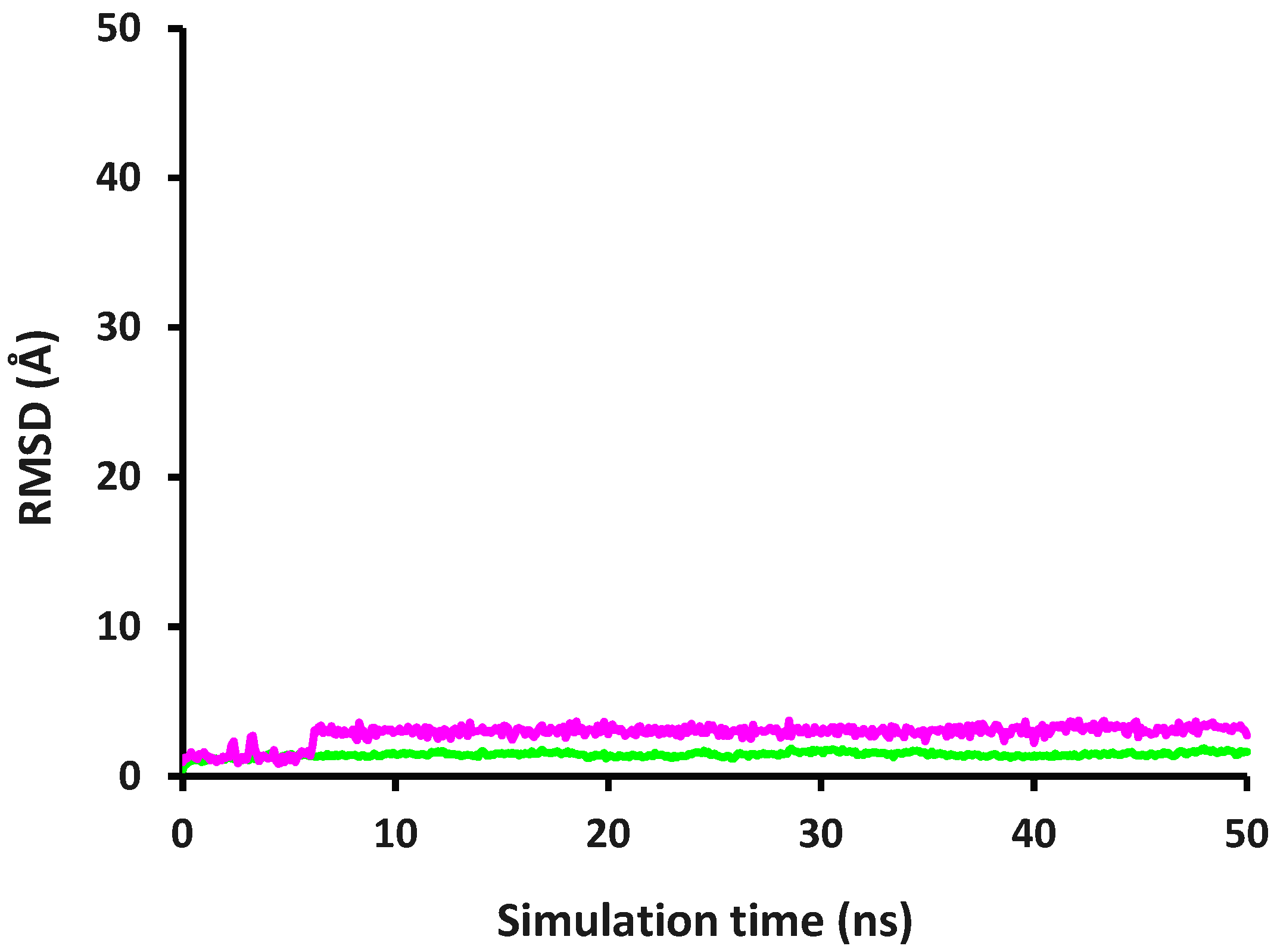
2.3. Molecular Dynamics and Interaction Hotspot Identifications of Caffeic Acid-DPP-4
2.4. Molecular Dynamics and Interaction Hotspot Identifications of Caffeic Acid-MMP-9
3. Materials and Methods
3.1. Chemicals
3.2. In Silico Instrumentations
3.3. Standard Caffeic Acid and Sample Preparation
3.4. DPP-4 Inhibitory Assay
3.5. MMP-9 Inhibitory Assay
3.6. Receptor–Ligand Preparation
3.7. Redocking of the Native Ligand
3.8. Molecular Docking of Caffeic Acid
3.9. Molecular Dynamics Simulations
3.10. Interaction Hotspot Identifications
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Westman, E.C. Type 2 Diabetes Mellitus: A Pathophysiologic Perspective. Front. Nutr. 2021, 8, 707371. [Google Scholar] [CrossRef] [PubMed]
- Donath, M.Y.; Ehses, J.A.; Maedler, K.; Schumann, D.M.; Ellingsgaard, H.; Eppler, E.; Reinecke, M. Mechanisms of β-Cell Death in Type 2 Diabetes. Diabetes 2005, 54, S108–S113. [Google Scholar] [CrossRef] [PubMed]
- Mariadoss, A.V.A.; Sivakumar, A.S.; Lee, C.H.; Kim, S.J. Diabetes Mellitus and Diabetic Foot Ulcer: Etiology, Biochemical and Molecular Based Treatment Strategies via Gene and Nanotherapy. Biomed. Pharmacother. 2022, 151, 113134. [Google Scholar] [CrossRef]
- Diabetes Facets and Figures. International Diabetes Federation. Available online: https://idf.org/about-diabetes/diabetes-facts-figures/ (accessed on 3 September 2023).
- Pang, B.; Zhou, Q.; Li, J.L.; Zhao, L.H.; Tong, X.L. Treatment of Refractory Diabetic Gastroparesis: Western Medicine and Traditional Chinese Medicine Therapies. World J. Gastroenterol. 2014, 20, 6504–6514. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Li, Y.; Dai, Y.; Peng, J. Natural Products for the Treatment of Type 2 Diabetes Mellitus: Pharmacology and Mechanisms. Pharmacol. Res. 2018, 130, 451–465. [Google Scholar] [CrossRef]
- Herman, A.; Herman, A.P. Herbal Products and Their Active Constituents for Diabetic Wound Healing—Preclinical and Clinical Studies: A Systematic Review. Pharmaceutics 2023, 15, 281. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Ma, Q.; Ye, L.; Piao, G. The Traditional Medicine and Modern Medicine from Natural Products. Molecules 2016, 21, 559. [Google Scholar] [CrossRef] [PubMed]
- Monteiro Espíndola, K.M.; Ferreira, R.G.; Mosquera Narvaez, L.E.; Rocha Silva Rosario, A.C.; Machado Da Silva, A.H.; Bispo Silva, A.G.; Oliveira Vieira, A.P.; Chagas Monteiro, M. Chemical and Pharmacological Aspects of Caffeic Acid and Its Activity in Hepatocarcinoma. Front. Oncol. 2019, 9, 467241. [Google Scholar] [CrossRef]
- Gani, M.R.; Istyastono, E.P. Determination of Caffeic Acid in Ethanolic Extract of Spent Coffee Grounds by High-Performance Liquid Chromatography with UV Detection. Indones. J. Chem. 2021, 21, 1281–1286. [Google Scholar] [CrossRef]
- Spagnol, C.M.; Assis, R.P.; Brunetti, I.L.; Isaac, V.L.B.; Salgado, H.R.N.; Corrêa, M.A. In Vitro Methods to Determine the Antioxidant Activity of Caffeic Acid. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2019, 219, 358–366. [Google Scholar] [CrossRef]
- Saivish, M.V.; Pacca, C.C.; da Costa, V.G.; de Lima Menezes, G.; da Silva, R.A.; Nebo, L.; da Silva, G.C.D.; de Aguiar Milhim, B.H.G.; da Silva Teixeira, I.; Henrique, T.; et al. Caffeic Acid Has Antiviral Activity against Ilhéus Virus In Vitro. Viruses 2023, 15, 494. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Li, S.; Liu, X.; An, H.; Kang, X.; Guo, S. Caffeic Acid, an Active Ingredient in Coffee, Combines with DOX for Multitarget Combination Therapy of Lung Cancer. J. Agric. Food Chem. 2022, 70, 8326–8337. [Google Scholar] [CrossRef] [PubMed]
- Alson, S.G.; Jansen, O.; Cieckiewicz, E.; Rakotoarimanana, H.; Rafatro, H.; Degotte, G.; Francotte, P.; Frederich, M. In-Vitro and in-Vivo Antimalarial Activity of Caffeic Acid and Some of Its Derivatives. J. Pharm. Pharmacol. 2018, 70, 1349–1356. [Google Scholar] [CrossRef] [PubMed]
- Oršolić, N.; Sirovina, D.; Odeh, D.; Gajski, G.; Balta, V.; Šver, L.; Jembrek, M.J. Efficacy of Caffeic Acid on Diabetes and Its Complications in the Mouse. Molecules 2021, 26, 3262. [Google Scholar] [CrossRef] [PubMed]
- Salau, V.F.; Erukainure, O.L.; Ijomone, O.M.; Islam, M.S. Caffeic Acid Regulates Glucose Homeostasis and Inhibits Purinergic and Cholinergic Activities While Abating Oxidative Stress and Dyslipidaemia in Fructose-Streptozotocin-Induced Diabetic Rats. J. Pharm. Pharmacol. 2022, 74, 973–984. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Shen, S.C. Caffeic Acid and Cinnamic Acid Ameliorate Glucose Metabolism via Modulating Glycogenesis and Gluconeogenesis in Insulin-Resistant Mouse Hepatocytes. J. Funct. Foods 2012, 4, 358–366. [Google Scholar] [CrossRef]
- Shaikh, S.; Lee, E.J.; Ahmad, K.; Ahmad, S.S.; Lim, J.H.; Choi, I. A Comprehensive Review and Perspective on Natural Sources as Dipeptidyl Peptidase-4 Inhibitors for Management of Diabetes. Pharmaceuticals 2021, 14, 591. [Google Scholar] [CrossRef]
- Hariono, M.; Yuliani, S.H.; Istyastono, E.P.; Riswanto, F.D.O.; Adhipandito, C.F. Matrix Metalloproteinase 9 (MMP9) in Wound Healing of Diabetic Foot Ulcer: Molecular Target and Structure-Based Drug Design. Wound Med. 2018, 22, 1–13. [Google Scholar] [CrossRef]
- Wang, L.; Li, X.; Zhang, S.; Lu, W.; Liao, S.; Liu, X.; Shan, L.; Shen, X.; Jiang, H.; Zhang, W.; et al. Natural Products as a Gold Mine for Selective Matrix Metalloproteinases Inhibitors. Bioorg. Med. Chem. 2012, 20, 4164–4171. [Google Scholar] [CrossRef]
- Chen, J.; Qin, S.; Liu, S.; Zhong, K.; Jing, Y.; Wu, X.; Peng, F.; Li, D.; Peng, C. Targeting Matrix Metalloproteases in Diabetic Wound Healing. Front. Immunol. 2023, 14, 1089001. [Google Scholar] [CrossRef]
- Fan, S.L.; Lin, J.A.; Chen, S.Y.; Lin, J.H.; Lin, H.T.; Chen, Y.Y.; Yen, G.C. Effects of Hsian-Tsao (Mesona procumbens Hemsl.) Extracts and Its Polysaccharides on the Promotion of Wound Healing under Diabetes-like Conditions. Food Funct. 2021, 12, 119–132. [Google Scholar] [CrossRef]
- Istyastono, E.P.; Riswanto, F.D.O. Molecular Dynamics Simulations of the Caffeic Acid Interactions to Dipeptidyl Peptidase Iv. Int. J. Appl. Pharm. 2022, 14, 274–278. [Google Scholar] [CrossRef]
- Istyastono, E.P.; Radifar, M.; Yuniarti, N.; Prasasty, V.D.; Mungkasi, S. PyPLIF HIPPOS: A Molecular Interaction Fingerprinting Tool for Docking Results of AutoDock Vina and PLANTS. J. Chem. Inf. Model. 2020, 60, 3697–3702. [Google Scholar] [CrossRef]
- Saberian, M.; Li, J.; Donnoli, A.; Bonderenko, E.; Oliva, P.; Gill, B.; Lockrey, S.; Siddique, R. Recycling of Spent Coffee Grounds in Construction Materials: A Review. J. Clean. Prod. 2021, 289, 125837. [Google Scholar] [CrossRef]
- Campos-Vega, R.; Loarca-Piña, G.; Vergara-Castañeda, H.A.; Dave Oomah, B. Spent Coffee Grounds: A Review on Current Research and Future Prospects. Trends Food Sci. Technol. 2015, 45, 24–36. [Google Scholar] [CrossRef]
- Franca, A.S.; Oliveira, L.S. Potential Uses of Spent Coffee Grounds in the Food Industry. Foods 2022, 11, 2064. [Google Scholar] [CrossRef] [PubMed]
- Biftu, T.; Sinha-Roy, R.; Chen, P.; Qian, X.; Feng, D.; Kuethe, J.T.; Scapin, G.; Gao, Y.D.; Yan, Y.; Krueger, D.; et al. Omarigliptin (MK-3102): A Novel Long-Acting DPP-4 Inhibitor for Once-Weekly Treatment of Type 2 Diabetes. J. Med. Chem. 2014, 57, 3205–3212. [Google Scholar] [CrossRef]
- Mourad, A.A.E.; Khodir, A.E.; Saber, S.; Mourad, M.A.E. Novel Potent and Selective DPP-4 Inhibitors: Design, Synthesis and Molecular Docking Study of Dihydropyrimidine Phthalimide Hybrids. Pharmaceuticals 2021, 14, 144. [Google Scholar] [CrossRef]
- Lin, Y.S.; Chen, C.R.; Wu, W.H.; Wen, C.L.; Chang, C.I.; Hou, W.C. Anti-α-Glucosidase and Anti-Dipeptidyl Peptidase-IV Activities of Extracts and Purified Compounds from Vitis Thunbergii Var. Taiwaniana. J. Agric. Food Chem. 2015, 63, 6393–6401. [Google Scholar] [CrossRef]
- Gupta, A.; Jacobson, G.A.; Burgess, J.R.; Jelinek, H.F.; Nichols, D.S.; Narkowicz, C.K.; Al-Aubaidy, H.A. Citrus Bioflavonoids Dipeptidyl Peptidase-4 Inhibition Compared with Gliptin Antidiabetic Medications. Biochem. Biophys. Res. Commun. 2018, 503, 21–25. [Google Scholar] [CrossRef]
- Majeed, M.; Majeed, S.; Mundkur, L.; Nagabhushanam, K.; Arumugam, S.; Beede, K.; Ali, F. Standardized Emblica Officinalis Fruit Extract Inhibited the Activities of α-Amylase, α-Glucosidase, and Dipeptidyl Peptidase-4 and Displayed Antioxidant Potential. J. Sci. Food Agric. 2020, 100, 509–516. [Google Scholar] [CrossRef] [PubMed]
- Laronha, H.; Carpinteiro, I.; Portugal, J.; Azul, A.; Polido, M.; Petrova, K.T.; Salema-Oom, M.; Caldeira, J. Challenges in Matrix Metalloproteinases Inhibition. Biomolecules 2020, 10, 717. [Google Scholar] [CrossRef] [PubMed]
- Crascì, L.; Basile, L.; Panico, A.; Puglia, C.; Bonina, F.P.; Basile, P.M.; Rizza, L.; Guccione, S. Correlating in Vitro Target-Oriented Screening and Docking: Inhibition of Matrix Metalloproteinases Activities by Flavonoids. Planta Med. 2017, 83, 901–911. [Google Scholar] [CrossRef] [PubMed]
- Saragusti, A.C.; Ortega, M.G.; Cabrera, J.L.; Estrin, D.A.; Marti, M.A.; Chiabrando, G.A. Inhibitory Effect of Quercetin on Matrix Metalloproteinase 9 Activity Molecular Mechanism and Structure-Activity Relationship of the Flavonoid-Enzyme Interaction. Eur. J. Pharmacol. 2010, 644, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Hariono, M.; Rollando, R.; Karamoy, J.; Hariyono, P.; Salin, N.; Wahab, H. Bioguided Fractionation of Local Plants against Matrix Metalloproteinase9 and Its Cytotoxicity against Breast Cell Models: In Silico and In Vitro Study. Molecules 2020, 25, 4691. [Google Scholar] [CrossRef]
- Chen, Y.C. Beware of Docking! Trends Pharmacol. Sci. 2015, 36, 78–95. [Google Scholar] [CrossRef]
- Liu, K.; Kokubo, H. Exploring the Stability of Ligand Binding Modes to Proteins by Molecular Dynamics Simulations: A Cross-Docking Study. J. Chem. Inf. Model. 2017, 57, 2514–2522. [Google Scholar] [CrossRef]
- Musoev, A.; Numonov, S.; You, Z.; Gao, H. Discovery of Novel DPP-IV Inhibitors as Potential Candidates for the Treatment of Type 2 Diabetes Mellitus Predicted by 3D QSAR Pharmacophore Models, Molecular Docking and de Novo Evolution. Molecules 2019, 24, 2870. [Google Scholar] [CrossRef]
- Tomovic, K.; Ilic, B.S.; Miljkovic, M.; Dimov, S.; Yancheva, D.; Kojic, M.; Mavrova, A.T.; Kocic, G.; Smelcerovic, A. Benzo[4,5]Thieno[2,3-d]Pyrimidine Phthalimide Derivative, One of the Rare Noncompetitive Inhibitors of Dipeptidyl Peptidase-4. Arch. Pharm. Chem. Life Sci. 2020, 353, 1900238. [Google Scholar] [CrossRef]
- Nongonierma, A.B.; Mooney, C.; Shields, D.C.; Fitzgerald, R.J. In Silico Approaches to Predict the Potential of Milk Protein-Derived Peptides as Dipeptidyl Peptidase IV (DPP-IV) Inhibitors. Peptides 2014, 57, 43–51. [Google Scholar] [CrossRef]
- Nongonierma, A.B.; Fitzgerald, R.J. Inhibition of Dipeptidyl Peptidase IV (DPP-IV) by Tryptophan Containing Dipeptides. Food Funct. 2013, 4, 1843–1849. [Google Scholar] [CrossRef] [PubMed]
- Malekipour, M.H.; Shirani, F.; Moradi, S.; Taherkhani, A. Cinnamic Acid Derivatives as Potential Matrix Metalloproteinase-9 Inhibitors: Molecular Docking and Dynamics Simulations. Genom. Inform. 2023, 21, e9. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Zhang, Q.; Zhang, C.; Yang, W.; Liu, H.; Lv, Z.; Liu, J.; Jiao, Z. Inhibition of Dipeptidyl Peptidase-4 by Flavonoids: Structure–Activity Relationship, Kinetics and Interaction Mechanism. Front. Nutr. 2022, 9, 892426. [Google Scholar] [CrossRef] [PubMed]
- Ende, C.; Gebhardt, R. Inhibition of Matrix Metalloproteinase-2 and -9 Activities by Selected Flavonoids. Planta Med. 2004, 70, 1006–1008. [Google Scholar] [CrossRef]
- Diallo, B.N.; Swart, T.; Hoppe, H.C.; Tastan Bishop, Ö.; Lobb, K. Potential Repurposing of Four FDA Approved Compounds with Antiplasmodial Activity Identified through Proteome Scale Computational Drug Discovery and in Vitro Assay. Sci. Rep. 2021, 11, 1413. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the Speed and Accuracy of Docking with a New Scoring Function, Efficient Optimization, and Multithreading. J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]

| Interacting Residue | Interaction Percentage |
|---|---|
| Arg61 | 0.80% |
| Trp62 | 1.80% |
| Pro159 | 2.40% |
| Pro218 | 1.60% |
| Lys463 | 47.50% |
| Tyr547 | 2.79% |
| Asn562 | 0.80% |
| Tyr631 | 0.60% |
| Tyr662 | 0.20% |
| Tyr666 | 1.40% |
| Interacting Residue | Interaction Type | Interaction Percentage |
|---|---|---|
| Tyr48 | Aromatic (Edge to face) | 0.20% |
| Ser59 | H-bond (Acceptor) | 0.20% |
| Arg61 | H-bond (Donor) | 3.59% |
| Arg61 | Ionic (Cation) | 4.39% |
| Trp62 | Aromatic (Edge to face) | 12.57% |
| Trp62 | Aromatic (Face to face) | 1.00% |
| Trp62 | H-bond (Donor) | 0.20% |
| Ser106 | H-bond (Donor) | 1.60% |
| Glu206 | H-bond (Acceptor) | 0.80% |
| Phe357 | Aromatic (Edge to face) | 0.60% |
| Phe357 | Aromatic (Face to face) | 2.00% |
| Ser460 | H-bond (Acceptor) | 0.40% |
| Lys463 | H-bond (Donor) | 6.59% |
| Lys463 | Ionic (Cation) | 5.59% |
| Glu464 | H-bond (Acceptor) | 1.60% |
| Arg471 | H-bond (Donor) | 2.20% |
| Arg471 | Ionic (Cation) | 2.79% |
| Ser473 | H-bond (Acceptor) | 0.20% |
| Tyr547 | Aromatic (Face to face) | 0.60% |
| Lys554 | H-bond (Donor) | 0.80% |
| Lys554 | Ionic (Cation) | 1.20% |
| Arg560 | H-bond (Donor) | 1.20% |
| Arg560 | Ionic (Cation) | 1.40% |
| Asn562 | H-bond (Donor) | 1.20% |
| Tyr666 | Aromatic (Edge to face) | 1.60% |
| Tyr666 | Aromatic (Face to face) | 0.40% |
| Interacting Residues | Interaction Type | Interaction Percentage |
|---|---|---|
| His226 | Aromatic (Edge-to-face) | 15.56% |
| Aromatic (Face-to-face) | 73.85% | |
| Hydrophobic | 90.01% | |
| H-bond (Donor) | 1.79% | |
| Pro246 | Hydrophobic | 0.79% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Istyastono, E.P.; Yuniarti, N.; Prasasty, V.D.; Mungkasi, S.; Waskitha, S.S.W.; Yanuar, M.R.S.; Riswanto, F.D.O. Caffeic Acid in Spent Coffee Grounds as a Dual Inhibitor for MMP-9 and DPP-4 Enzymes. Molecules 2023, 28, 7182. https://doi.org/10.3390/molecules28207182
Istyastono EP, Yuniarti N, Prasasty VD, Mungkasi S, Waskitha SSW, Yanuar MRS, Riswanto FDO. Caffeic Acid in Spent Coffee Grounds as a Dual Inhibitor for MMP-9 and DPP-4 Enzymes. Molecules. 2023; 28(20):7182. https://doi.org/10.3390/molecules28207182
Chicago/Turabian StyleIstyastono, Enade P., Nunung Yuniarti, Vivitri D. Prasasty, Sudi Mungkasi, Stephanus S. W. Waskitha, Michael R. S. Yanuar, and Florentinus D. O. Riswanto. 2023. "Caffeic Acid in Spent Coffee Grounds as a Dual Inhibitor for MMP-9 and DPP-4 Enzymes" Molecules 28, no. 20: 7182. https://doi.org/10.3390/molecules28207182
APA StyleIstyastono, E. P., Yuniarti, N., Prasasty, V. D., Mungkasi, S., Waskitha, S. S. W., Yanuar, M. R. S., & Riswanto, F. D. O. (2023). Caffeic Acid in Spent Coffee Grounds as a Dual Inhibitor for MMP-9 and DPP-4 Enzymes. Molecules, 28(20), 7182. https://doi.org/10.3390/molecules28207182








