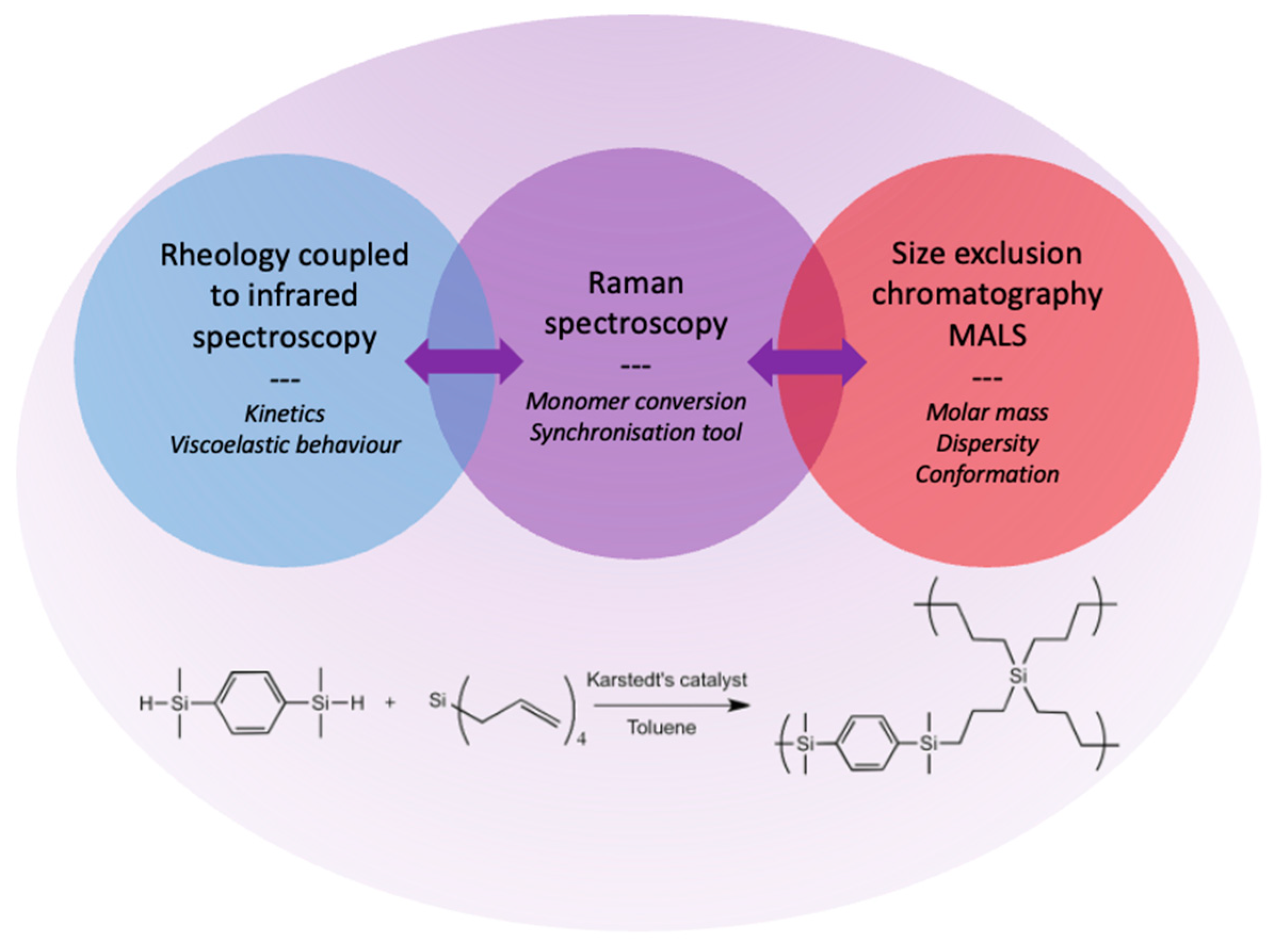
Contribution of Dynamic Rheology Coupled to FTIR and Raman Spectroscopies to the Real-Time Shaping Ability of a Hyperbranched Polycarbosilane
Abstract
:1. Introduction
2. Results and Discussion
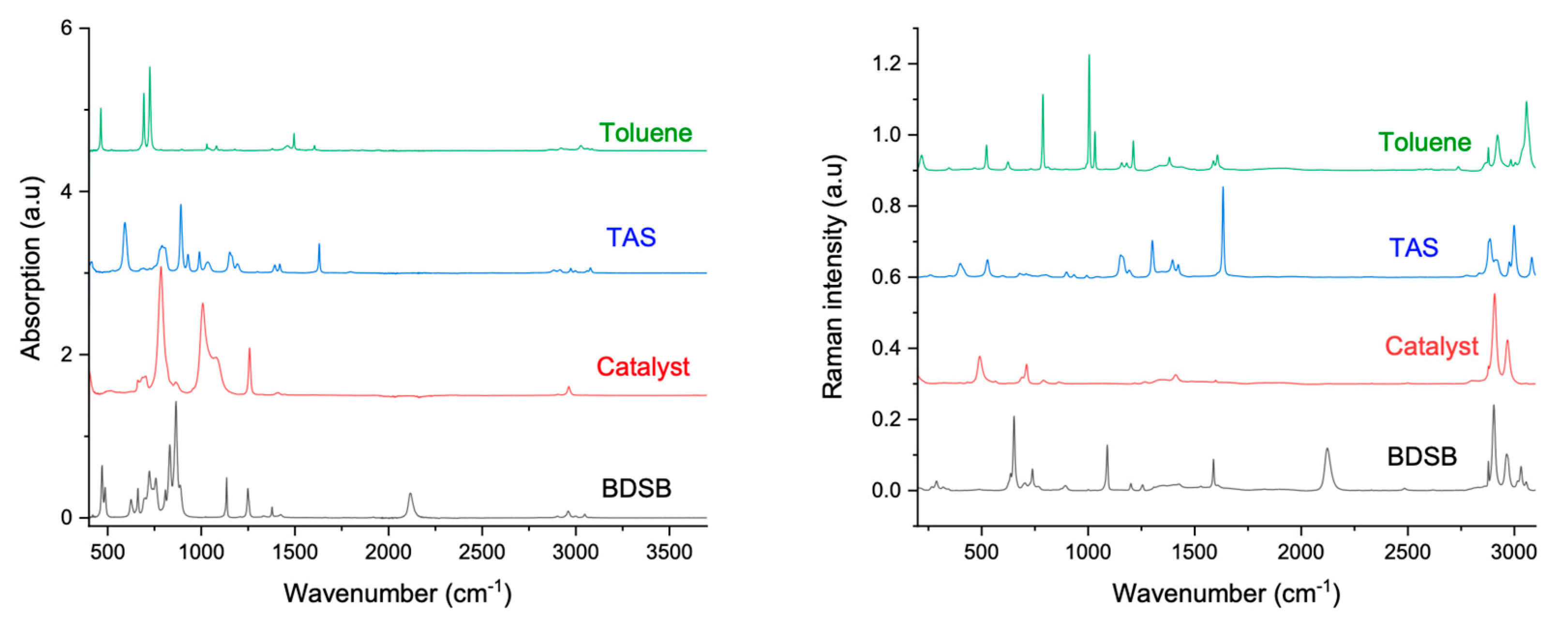
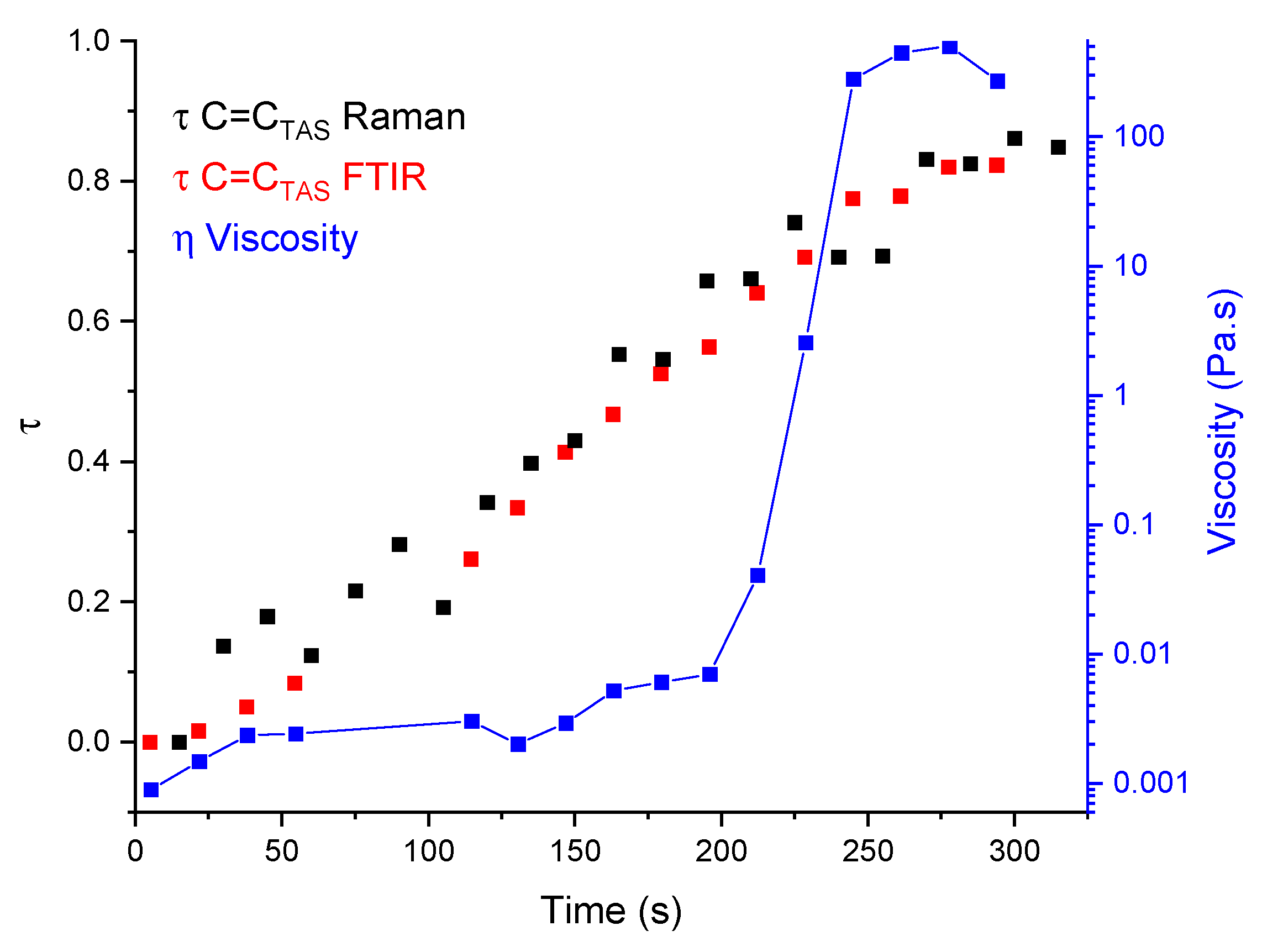
2.1. Rheology Coupled to Raman and Infrared Spectroscopies
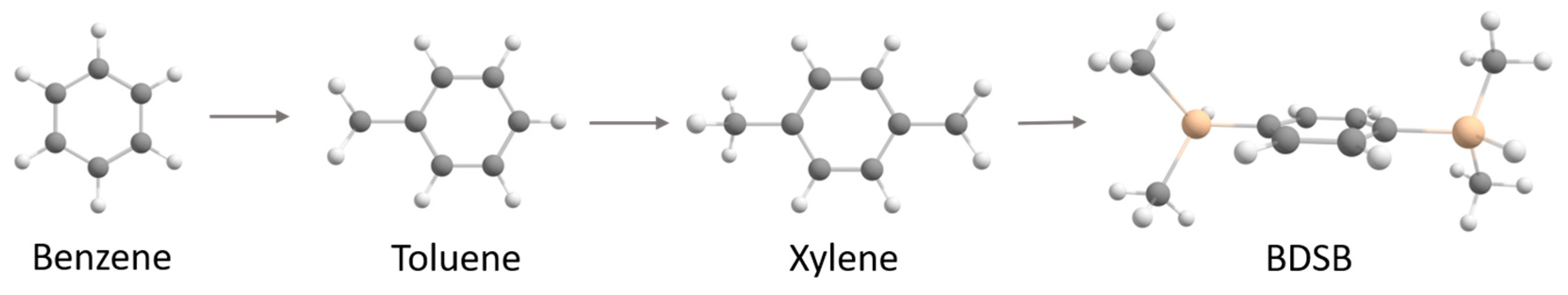
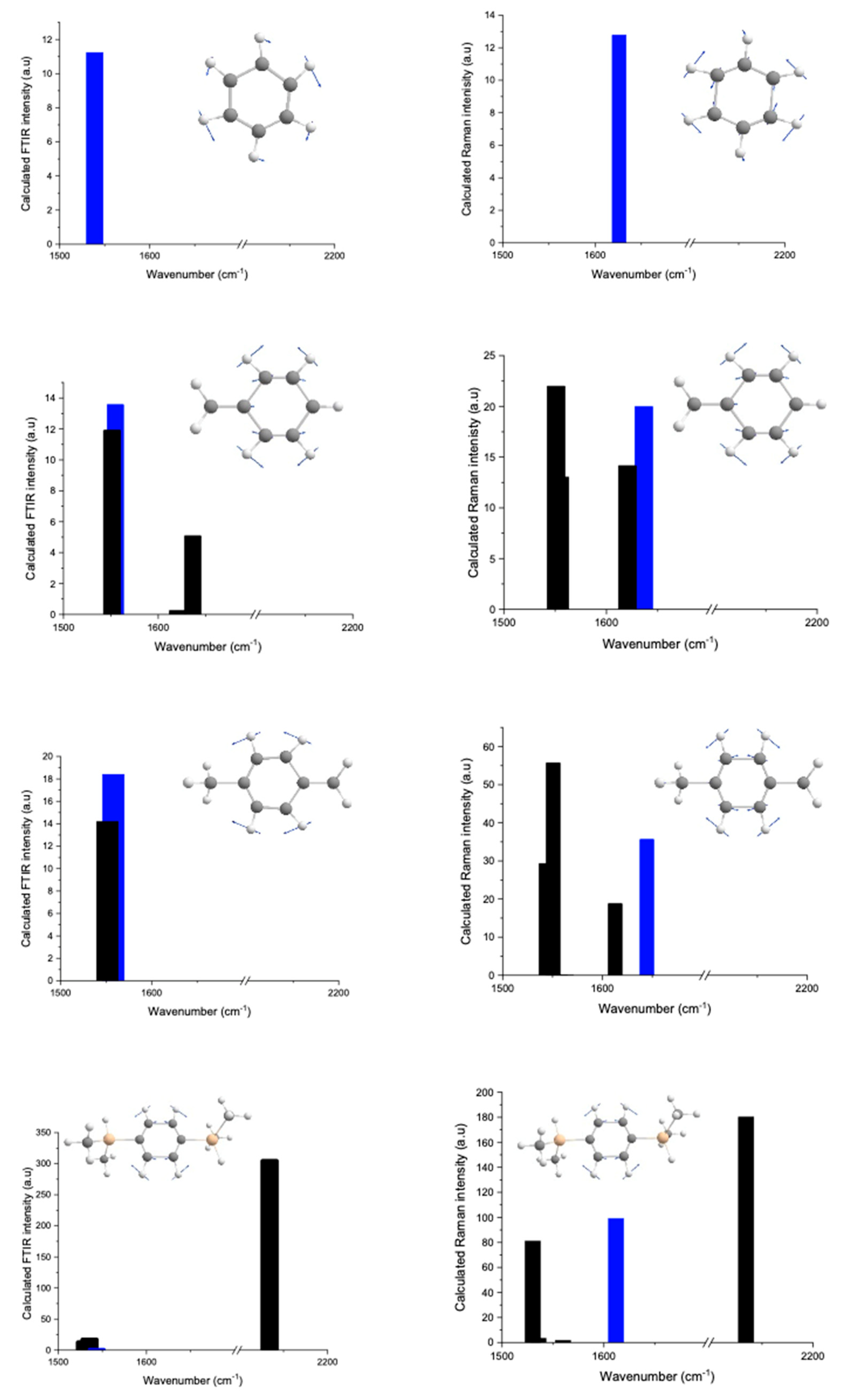
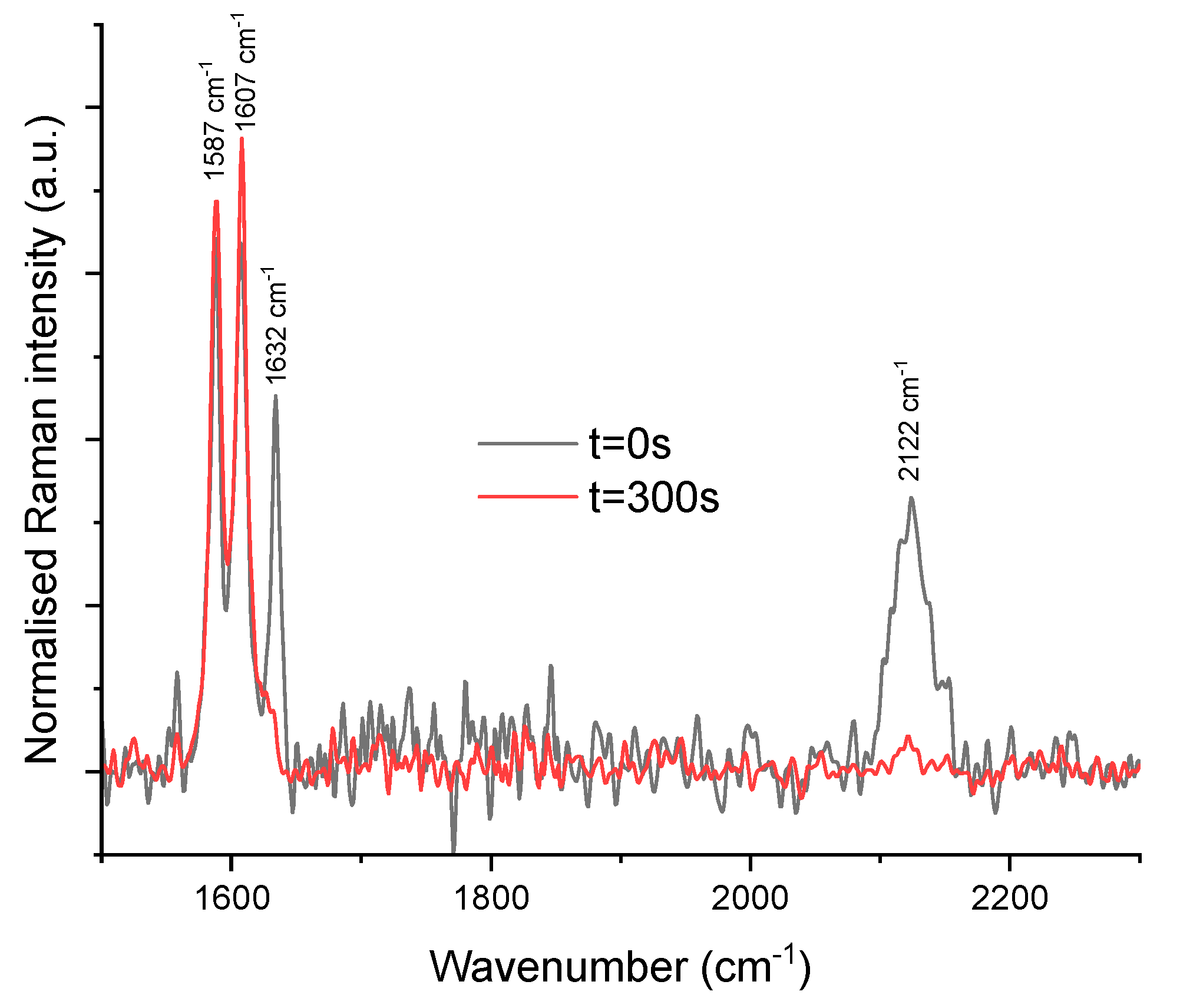
2.1.1. From Spectroscopic Signatures
2.1.2. To the Optimisation of a New Coupling Instrumentation
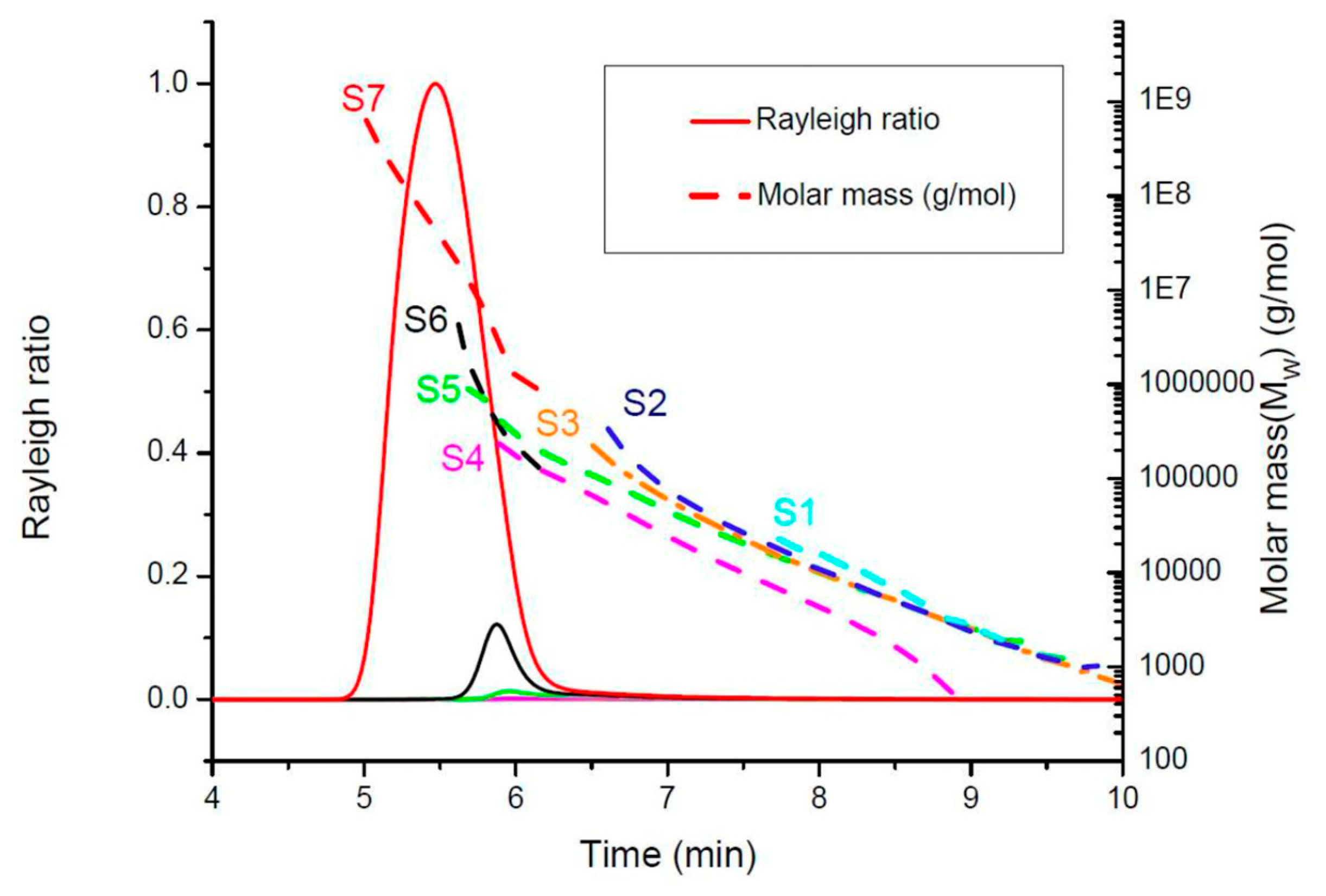
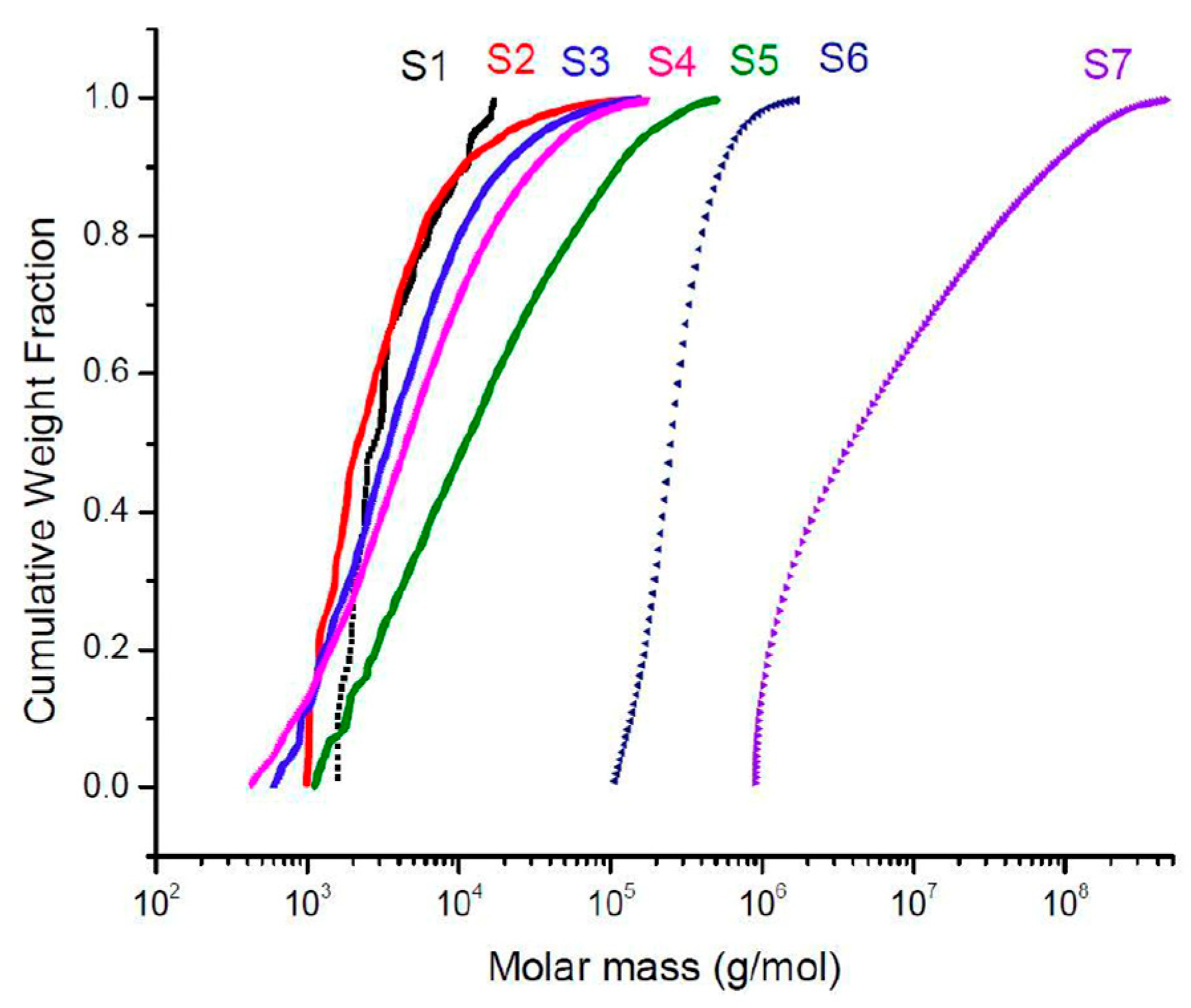
2.2. Kinetics of the Hyperbranched Polymerisation through SEC-MALS Analyses
2.3. Linking Polymer Structure to Rheology, towards a Shaping Ability
3. Materials and Methods
3.1. Materials
3.2. Characterisations
3.2.1. Rheometer Coupled to FTIR Spectroscopy
3.2.2. Raman Spectroscopy
3.2.3. Size-Exclusion Chromatography/Multiangle Light Scattering and Sampling
3.3. Synthesis
3.3.1. Classic Synthesis
3.3.2. In Situ Monitored Synthesis
3.3.3. Monomer Conversion Calculation
3.4. Computational Details
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bougoin, M.; Thevenot, F.; Dubois, J.; Fantozzi, G. Synthesis and thermomechanical properties of dense carbide ceramic composites produced from boron carbide and silicon. J. Less-Common. Metals 1987, 132, 209–228. [Google Scholar] [CrossRef]
- Balat, M.J.H. Determination of the active-to-passive transition in the oxidation of silicon carbide in standard and microwave-excited air. J. Eur. Ceram. Soc. 1996, 16, 55–62. [Google Scholar] [CrossRef]
- Li, H.; Zhang, L.; Cheng, L.; Wang, Y. Oxidation analysis of 2D C/ZrC–SiC composites with different coating structures in CH4 combustion gas environment. Ceram. Int. 2009, 35, 2277–2282. [Google Scholar] [CrossRef]
- Yajima, S.; Okamura, K.; Hayashi, J.; Omori, M. Synthesis of continuous SiC fibers with high tensile strength. J. Am. Ceram. Soc. 1976, 59, 324–327. [Google Scholar] [CrossRef]
- Colombo, P.; Mera, G.; Riedel, R.; Sorarù, G.D. Polymer-Derived Ceramics: 40 Years of Research and Innovation in Advanced Ceramics. J. Am. Ceram. Soc. 2010, 93, 1805–1837. [Google Scholar] [CrossRef]
- Bernardo, E.; Fiocco, L.; Parcianello, G.; Storti, E.; Colombo, P. Advanced Ceramics from Preceramic Polymers Modified at the Nano-Scale: A Review. Materials 2014, 7, 1927–1956. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Ishida, H.; Maia, J. Vinyl Ester-Clay Based Nanocomposites: A Complementary Study of the Vinyl Ester Polymerization by Hyphenated Rheo-FTIR and Separate Rheology /FTIR Measurements. Polym. Int. 2014, 63, 521–528. [Google Scholar] [CrossRef]
- Bose, R.K.; Kötteritzsch, J.; Garcia, S.J.; Hager, M.D.; Schubert, U.S.; Van der Zwaag, S. A rheological and spectroscopic study on the kinetics of self-healing in a single-component diels-alder copolymer and its underlying chemical reaction. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 1669–1675. [Google Scholar] [CrossRef]
- Auriemma, M.; Piscitelli, A.; Pasquino, R.; Cerruti, P.; Malinconico, M.; Grizzuti, N. Blending poly (3-hydroxybutyrate) with tannic acid: Influence of a polyphenolic natural additive on the rheological and thermal behavior. Eur. Polym. J. 2015, 63, 123–131. [Google Scholar] [CrossRef]
- Darsy, G.; Bouzat, F.; Muñoz-Hoyos, M.; Lucas, R.; Foucaud, S.; Coelho, C.; Babonneau, F.; Maître, A. Monitoring a polycycloaddition by the combination of dynamic rheology and FTIR spectroscopy. Polymer 2015, 79, 283–289. [Google Scholar] [CrossRef]
- Piriou, C.; Viers, L.; Lucas, R.; Bouzat, F.; Laadoua, H.; Champavier, Y.; Foucaud, S.; Coelho, C.; Babonneau, F. Rheological and thermal behaviours of a hyperbranched polycarbosilane. Appl. Organomet. Chem. 2018, e4443. [Google Scholar] [CrossRef]
- Kudelski, A. Analytical applications of Raman spectroscopy. Talanta 2008, 76, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Simsek, G.; Colomban, P.; Milande, V. Tentative differentiation between Iznik tiles and copies with Raman spectroscopy using both laboratory and portable instruments. J. Raman Spectrosc. 2010, 41, 529–536. [Google Scholar] [CrossRef]
- Quang, L.X.; Lim, C.; Seong, G.H.; Choo, J.; Do, K.J.; Yoo, S.Y. A portable surface-enhanced Raman scattering sensor integrated with a lab-on-a-chip for field analysis. Lab Chip 2008, 8, 2214–2219. [Google Scholar] [CrossRef] [PubMed]
- Chevrel, M.C.; Hoppe, S.; Falk, L.; Brun, N.; Chapron, D.; Bourson, P.; Durand, A. In situ monitoring of styrene polymerization using Raman spectroscopy. Multi-scale approach of homogeneous and heterogeneous polymerization processes. Ind. Eng. Chem. Res. 2012, 51, 16151–16156. [Google Scholar] [CrossRef]
- Brun, N.; Youssef, I.; Chevrel, M.C.; Chapron, D.; Schrauwen, C.; Hoppe, S.; Bourson, P.; Durand, A. In situ monitoring of styrene polymerization using Raman spectroscopy. Multi-scale approach of homogeneous and heterogeneous polymerization processes. J. Raman Spectrosc. 2013, 44, 909–915. [Google Scholar] [CrossRef]
- Brun, N.; Chevrel, M.C.; Falk, L.; Hoppe, S.; Durand, A.; Chapron, D.; Bourson, P. Contribution of Raman Spectroscopy to In Situ Monitoring of a High-Impact Polystyrene Process. Chem. Eng. Technol. 2014, 37, 275–282. [Google Scholar] [CrossRef]
- Kotula, A.P.; Meyer, M.W.; De Vito, F.; Plog, J.; Hight Walker, A.R.; Migler, K.B. The rheo-Raman microscope: Simultaneous chemical, conformational, mechanical, and microstructural measures of soft materials. Rev. Sci. Instrument. 2016, 87, 105105. [Google Scholar] [CrossRef]
- Podzimek, Š. A review of the analysis of branched polymers by SEC-MALS. Am. Lab. 2002, 34, 38. [Google Scholar]
- Podzimek, Š. Multi-Angle Light Scattering: An Efficient Tool Revealing Molecular Structure of Synthetic Polymers. Macromol. Symp. 2019, 384, 1800174. [Google Scholar] [CrossRef]
- Carnicer Cervera, V.; Alcazar Rodrigo, M.D.C.; Orts Tarí, M.J.; Sánchez-Vilches, E.; Moreno, R. Microfluidic rheology: A new approach to measure viscosity of ceramic suspensions at extremely high shear rates. Open Ceram. 2020, 5, 100052. [Google Scholar] [CrossRef]

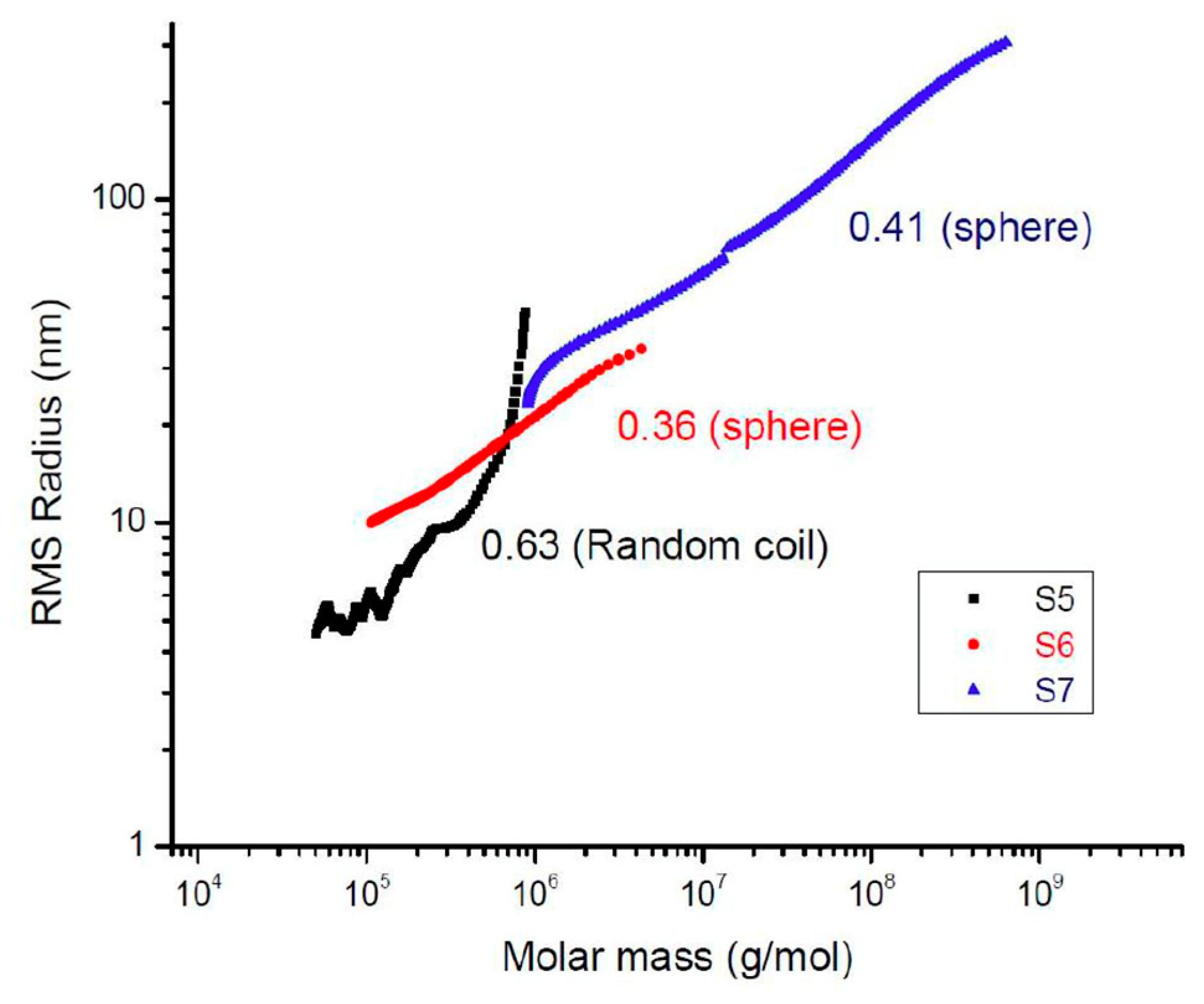
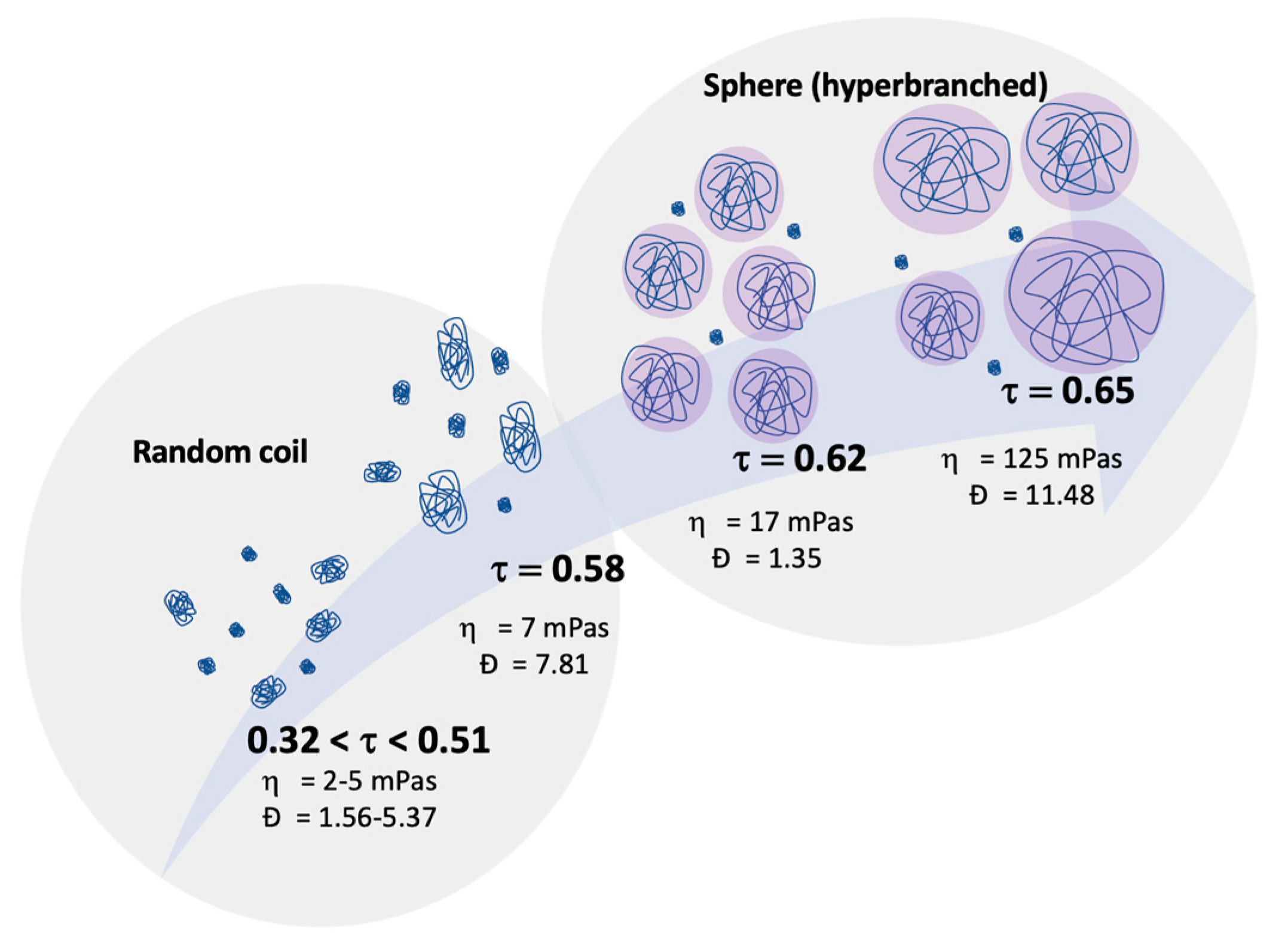

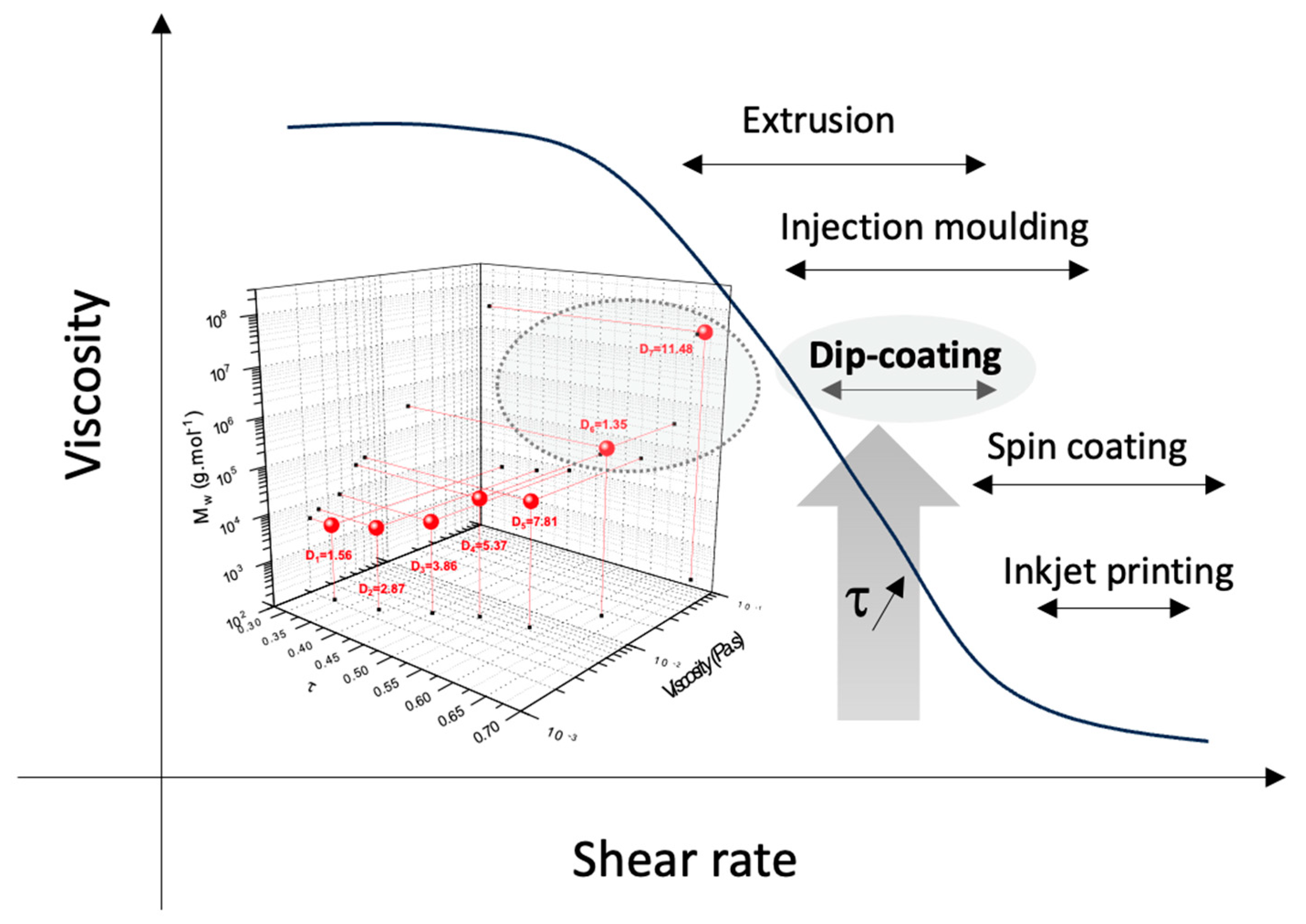
| Sample | /g·mol−1 | ƉM/- |
|---|---|---|
| S1 1 | 4386 | 1.56 |
| S2 1 | 5885 | 2.87 |
| S3 1 | 8984 | 3.86 |
| S4 1 | 31,080 | 5.37 |
| S5 1 | 40,230 | 7.81 |
| S6 1 | 320,200 | 1.35 |
| S7 2 | 28,200,000 | 11.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dhondoo, N.; Cornette, J.; Foucaud, S.; Colas, M.; Lucas-Roper, R. Contribution of Dynamic Rheology Coupled to FTIR and Raman Spectroscopies to the Real-Time Shaping Ability of a Hyperbranched Polycarbosilane. Molecules 2023, 28, 6476. https://doi.org/10.3390/molecules28186476
Dhondoo N, Cornette J, Foucaud S, Colas M, Lucas-Roper R. Contribution of Dynamic Rheology Coupled to FTIR and Raman Spectroscopies to the Real-Time Shaping Ability of a Hyperbranched Polycarbosilane. Molecules. 2023; 28(18):6476. https://doi.org/10.3390/molecules28186476
Chicago/Turabian StyleDhondoo, Nilesh, Julie Cornette, Sylvie Foucaud, Maggy Colas, and Romain Lucas-Roper. 2023. "Contribution of Dynamic Rheology Coupled to FTIR and Raman Spectroscopies to the Real-Time Shaping Ability of a Hyperbranched Polycarbosilane" Molecules 28, no. 18: 6476. https://doi.org/10.3390/molecules28186476
APA StyleDhondoo, N., Cornette, J., Foucaud, S., Colas, M., & Lucas-Roper, R. (2023). Contribution of Dynamic Rheology Coupled to FTIR and Raman Spectroscopies to the Real-Time Shaping Ability of a Hyperbranched Polycarbosilane. Molecules, 28(18), 6476. https://doi.org/10.3390/molecules28186476







