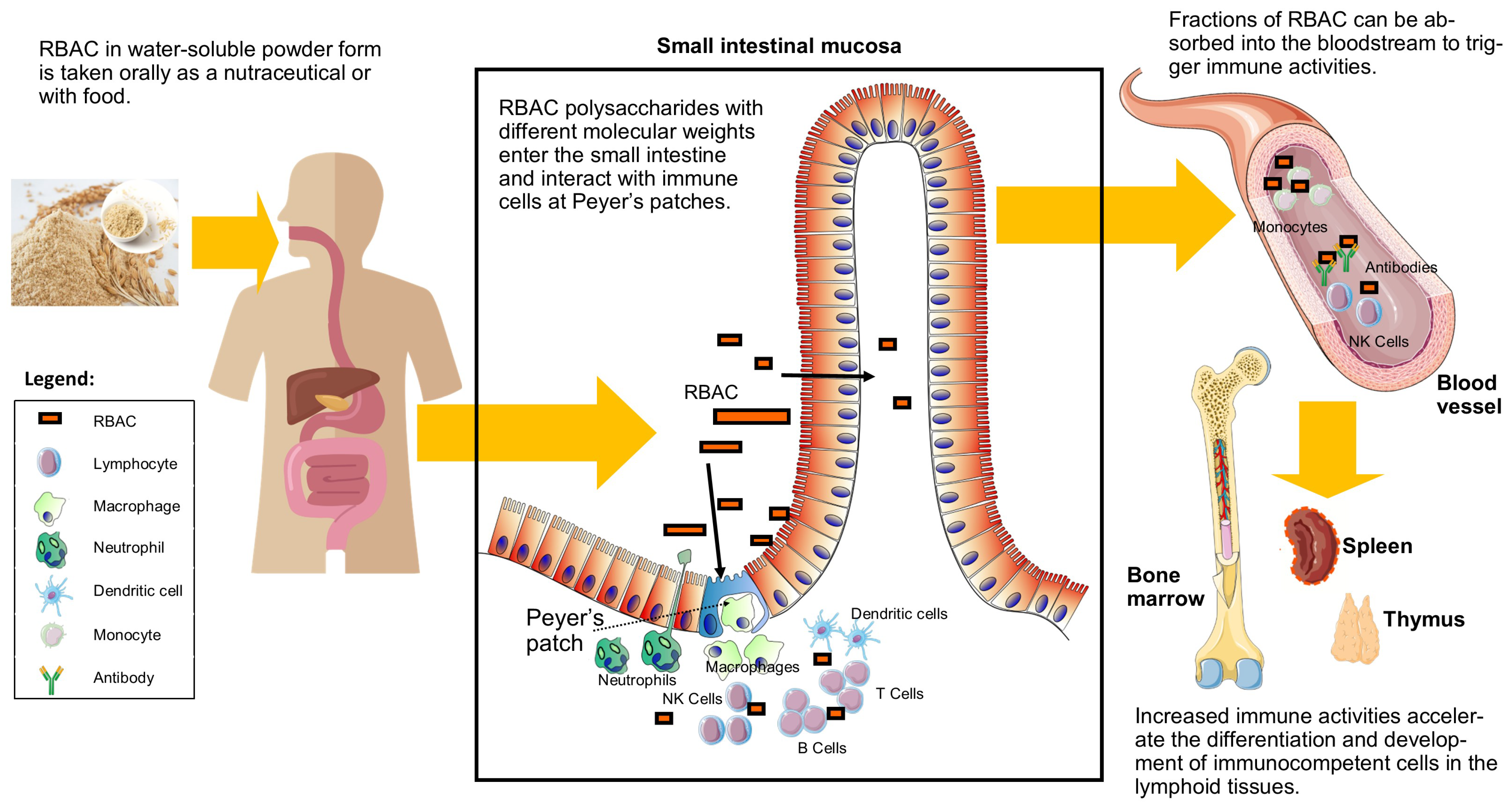
3.1. Chemical Composition
Only one article by Miura et al. [
22] attempted to identify the chemical structure of the active ingredient of RBAC responsible for its immunomodulatory activity. Through methanol fractional precipitation, the study discovered that several components of Biobran MGN-3 possess immunomodulatory activities. Among them are the 50% MeOH insoluble fraction (50 ppt) and subfractions II-4, II-5, and II-6 of 80% MeOH insoluble material eluted with 0.2 M NaCl which demonstrated macrophage-stimulating activity with increased nitric oxide (NO) formation while inducing interleukin (IL)-1
and tumour necrosis factor (TNF)-
production in vitro. Among these components, subfraction II-6 demonstrated the highest macrophage-stimulating activity and was found to have a molecular weight between 10,000–20,000 daltons.
Through methylation analysis based on the sugar linkage composition of subfraction II-6, Miura et al. [
22] proposed four possible polysaccharide structures in the active ingredient of Biobran MGN-3 to be: (1) arabinogalactan with a main chain of 1,4-
-galactan and side chains of arabinose, (2) arabinoxylan with a main chain of 1,4-
-xylan and side chains of arabinose, (3) arabinan with the main chain and side chains of arabinose, and (4)
-1.3:1,4-glucan. The possible structures are shown in
Figure 1. While Miura et al. [
22] suggested further study to determine whether these were intermixing structures or parts of a composite molecule, there was no follow-up research.
Natural sugar analysis with chromatography was performed on various RBAC products by different authors [
22,
23,
24,
25], with the results summarised in
Table 1. The subfraction II-6 of MeOH insoluble material of Biobran MGN-3 mentioned earlier has glucose (30.2%), galactose (23%), arabinose (22.2%), and xylose (13.7%) as the main components. As arabinoxylan comprises arabinose and xylose, the active ingredient of Biobran MGN-3 thus consists of complex polysaccharides with arabinoxylan as its primary structure (36%) while also containing galactan and glucan. However, the composition can vary drastically across different subfractions of Biobran MGN-3, with fraction IV in
Table 1 consisting mainly of glucose (89.2%) [
22].
RBEP (sample 1) from Erom was reported to have a much higher content of mannose (22.9%), similar in arabinose content (21.8%) but lower in xylose, glucose, and galactose contents compared to Biobran MGN-3 (Fr-II) [
25]. However, in a separate study by Choi et al. [
26], the primary sugar composition of RBEP was reported as xylose (22.25%) and glucose (11.71%), with only traces of galactose and arabinose detected (sample 2). Hence, the results from these two analyses were markedly different.
Furthermore, the two RBAC products from STR Biotech, namely BPP (a black rice bran derivative) and BPRBE, also appeared to have drastically different sugar compositions than Biobran MGN-3 and RBEP, as shown in
Table 1 [
23,
24]. It is unclear how different sugar compositions may affect the immunomodulating capacity of the products in the absence of any direct comparison. Regardless, these RBAC products (Biobran MGN-3, RBEP, BPP, BPRBE) were reported to have enhanced macrophage-stimulation activities, with details presented in a later section.
3.2. Antioxidant Capacity
Reactive oxygen species (ROS), such as superoxide radicals (O
2•−), hydrogen peroxide (H
2O
2), and hydroxyl radicals (•OH), are metabolic by-products of biological processes [
27]. Low levels of ROS can be beneficial to the immune system. Phagocytes, such as neutrophils and macrophages, synthesise and release ROS in respiratory bursts to facilitate phagocytosis and the killing of microbial [
28]. Excess production of ROS, however, can lead to oxidative stress that harm the host cells, damaging the mitochondria and deoxyribonucleic acid (DNA), thus inducing premature cell death or leading to neoplasm [
29]. Therefore, it is widely accepted that oxidative damage caused by ROS is the primary cause of aging, although the theory is yet to be substantiated by research evidence [
30]. An imbalance in ROS also plays a role in the dysfunction of immune cell activity, such as the adaptive immune responses by T and B cells [
31,
32], and excess ROS has been identified as a contributing factor to autoimmune disorders [
33,
34]. Therefore, maintaining ROS homeostasis is crucial for the immune system, health, and aging.
Antioxidants are stable molecules readily donating electrons to neutralise free radicals and hence counteract the effects of ROS [
35]. The endogenous antioxidant system, which consists of enzymatic, hydrophilic, and lipophilic antioxidants, eliminates excess ROS to protect the host against oxidative stress, thus maintaining homeostasis [
36]. The antioxidant activities of plant-based polysaccharides are known to improve markedly after chemical modification [
37]. Specifically, fermentation and treatment with enzymes enhance the antioxidant capacity in rice bran by removing bonds between bioactive and cell wall components [
38]. Therefore, RBAC, as a form of denatured hemicellulose of rice bran, may be a rich source of antioxidants needed for ROS balance.
Table 2 summarises the research on the antioxidant capacity of RBAC.
Tazawa et al. [
39] studied the free radical scavenging activities of Biobran MGN-3 using the compound and three of its fractions (L, M, S) extracted with fractional ethanol condensation. The study found high scavenging activities in Biobran MGN-3 and all its fractions. The scavenging rates were dose-dependent against O
2•− and •OH at 20, 2.0, and 0.2 mg/mL of the test samples. In particular, the S fraction of Biobran MGN-3 (<3000 molecules) was the most effective in the inhibition of •OH caused by O
2•− and ultraviolet irradiation [
39].
In another study, An [
40] evaluated the antioxidant capacity of RBEP compared to broccoli, a known high-antioxidant food, with two assays: the Oxygen Radical Antioxidant Capacity (ORAC, Cell Biolabs Inc., San Diego, CA, USA) and Total Antioxidant Status (TAS, Randox Labs. Ltd., Gortnagallon, Crumlin, UK). The study found the fat-soluble (lipophilic) sample of RBEP to have a higher antioxidant capacity than its water-soluble (hydrophilic) counterpart (446 vs. 326 μM Trolox equivalent/g). Both fat- and water-soluble RBEP had much higher ORAC values than lipophilic and hydrophilic extracts from raw broccoli, broccoli seed, and sprout. However, in the TAS assay, broccoli sprout extract had the highest measurement at 0.56 mmol/100 g, whereas RBEP ranked second with 0.4 mmol/100 g compared to 0.18 and 0.38 mmol/100 g of raw broccoli and broccoli seeds, respectively (An, 2011). Moreover, co-culturing L929 (mouse fibroblast) cell line with 1, 10, 100, and 500 μg/μL of RBEP for 72 h did not affect cell count. Hence, the study confirmed that RBEP was not cytotoxic and could be a functional food comparable to or even better than broccoli in antioxidant content.
Noaman et al. [
41] investigated the in vivo antioxidant effect of RBAC. Adult female Swiss albino mice (
n = 10) were injected intraperitoneally (i.p.) with 25 mg/kg body weight (BW) of Biobran MGN-3 six times a week over 25 days. These Biobran MGN-3-treated mice were found to have higher glutathione, an antioxidant, in the liver compared to untreated mice (
n = 10) by 24.79% (
p < 0.05) at the end of the study, even though the plasma glutathione level remained unchanged from the control values. Correspondingly Biobran MGN-3 appeared to significantly increase the production of glutathione peroxidase in the liver by 29.34% (
p < 0.05) over control but not in the blood. Other endogenous antioxidant enzymes in the liver and plasma, including glutathione-S-transferase, superoxide dismutase and catalase, were no different from the control values. The study also found significant increases (
p < 0.05) in the gene expression of glutathione peroxidase and catalase in Biobran MGN-3-treated mice compared to the control mice. So, Biobran MGN-3 is a potent antioxidant and could affect the production of endogenous antioxidants epigenetically, particularly glutathione and glutathione peroxidase to improve the host defence against ROS.
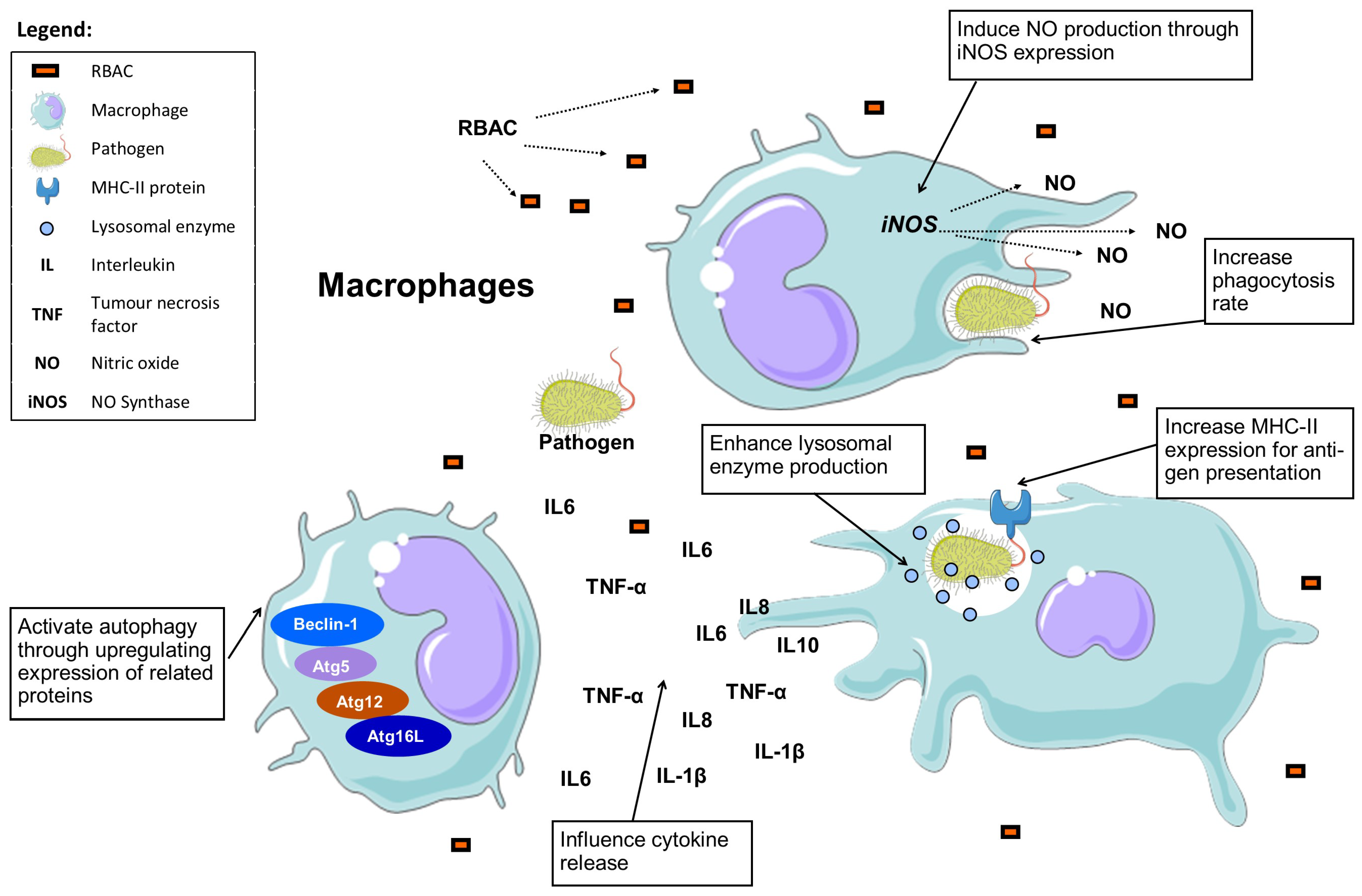
3.4. Macrophage Stimulation and Phagocytosis
Macrophages are derived from the circulating monocytes originating from the bone marrow. As a type of professional phagocytic cell, macrophages embed in tissues and engulf, digest, and eliminate invading microorganisms, foreign bodies, and damaged or mutated cells while initiating proinflammatory responses if necessary [
46,
47]. Hence, macrophages are an essential component of the innate defence mechanism of the host tissues. Additionally, macrophages play a role in antigen presentation, triggering adaptive immune responses from the cluster differentiation (CD) 8+ T lymphocytes during infection and inflammation [
48]. After pathogen elimination, macrophages actively assist in wound healing by dampening inflammation, clearing of cell debris, and coordinating tissue repair [
49]. Hence, there are two opposing phenotypes of macrophages: M1 and M2. M1 fights infections and may cause cellular destruction and tissue damage, whereas M2 activity fixes the damage and promotes tissue repair [
50]. Plant-based polysaccharides are known to trigger immune responses in M1 macrophages with increased NO production for pathogen killing and cytokine secretion for inflammatory process regulation [
51]. As such, the macrophage stimulation intensity is commonly used as an immunomodulating indicator of the strength of RBAC products or components [
22,
25].
Table 3 summarises the effects of RBAC on macrophages based on available in vitro and in vivo evidence.
Yu et al. [
25] compared the macrophage stimulation activity of modified rice bran from the submerged culture of nine fungi (
Grifola frondose, L. edodes, Cordyceps militaris, Clonorchis sinensis, Agaricus blazei, Flammulina velutipes, Auricularia auricula-judae, Schizophyllum commune, and
Coriolus versicolor). Peritoneal macrophages (P-M∅) were isolated from male ICR mice and cultured with 10 and 100 μg/mL of rice bran modified by enzymes of different mushrooms. All modified rice bran showed higher macrophage stimulation activities measured with acid phosphatase (a lysosomal enzyme with a direct role in microbe killing and antigen presentation) when cultured at 100 μg/mL compared to saline control. Notwithstanding, the
L. edodes product (RBEP) demonstrated the highest activity, followed by those produced with
G. frondose, S. commune, and
C. versicolor. Furthermore, RBEP’s macrophage stimulation level was similar to LPS at 10 μg/mL and higher at 100 μg/mL.
Biobran MGN-3 was shown to stimulate M1 macrophages to enhance the rate of attachment and phagocytosis of yeast (
Saccharomyces cerevisiae), increase the production of proinflammatory cytokines (TNF-
and IL-6), and raise NO release in a dose-dependent manner (1 < 10 < 100 μg/mL). Ghoneum and Matsuura [
54] demonstrated these effects in vitro by utilising three different macrophage cell types: human (U937), murine (RAW264.7), and P-M∅ freshly prepared from female C3H/HeN mice. Albeit, the responsiveness varied across cell lines. The macrophage stimulation activity of Biobran MGN-3 was also validated based on NO and cytokine production (TNF-
and IL-1
) by Miura et al. [
22] while investigating the chemical structure of active ingredients of Biobran MGN-3.
Similar observations were made by Chae et al. [
55] while comparing the immunomodulating effects of Biobran MGN-3 compared to polysaccharide-peptide (PSP) extracted from
C. versicolor. The phagocytic activities of the P-M∅ extracted from Balb/c mice orally fed with 1.5 mg/L of either Biobran MGN-3 (69%) or PSP (65%) against
Candida parapsilosis were significantly higher (
p < 0.05) than P-M∅ of control mice fed with saline only (47%). Furthermore, both Biobran MGN-3 and PSP were observed to increase NO production in the RAW264.7 cell line, but Biobran MGN-3 appeared to be less effective than PSP (1.0 NO
2-mM vs. >2.0 NO
2-mM at 1000 μg/mL).
Regarding in vitro cytokine production in RAW264.7 macrophage cells, Biobran MGN-3 (1000 μg/mL) induced higher levels of IL-6 and TNF-
production than control but less than the levels influenced by PSP and LPS and was found to increase the major histocompatibility complex (MHC) class II expression in RAW264.7 cells compared to control after two days of incubation but also at a level lower than PSP, LPS, and interferon (IFN)-
[
55].
Biobran MGN-3 also upregulated human phagocytic cells, including monocytes, the precursors of macrophages, and neutrophils, to enhance intracellular killings of microbes, as Ghoneum et al. [
56] reported. Treatment with Biobran MGN-3 (100 μg/mL) significantly improved (
p < 0.01) the phagocytosis of
Escherichia coli by monocytes (110%) and neutrophils (400%) in human peripheral blood lymphocytes (PBL) compared to control. Increased oxidative burst by monocytes and neutrophils with elevated H
2O
2 production in the presence of
E. coli was observed. Culturing human macrophage cells (U937) with Biobran MGN-3 (1, 10, 100, 1000 μg/mL) also dose-dependently increased TNF-
, IL-6, IL-8, and IL-10 production with significant differences (
p < 0.01) observed from 10 μg/mL onward, compared to control. However, Biobran MGN-3 was not bactericidal as no in vitro activity was observed against 31 strains of selected anaerobic and microaerobic bacteria [
56].
The stimulating effect of RBEP on macrophages was also confirmed by Kim et al. [
45] in vitro and in vivo by measuring the lysosomal enzyme level. P-M∅ were extracted from male ICR mice and cultured with 10 and 100 μg/mL of RBEP. Both doses elicited more than twice the control activity level, similar to the levels of LPS under the same dose. The effects were also replicated in vivo with C3H/HeN mice fed with RBEP (50 and 250 mg/kg BW) for 5 days showing a 1.41- and 1.44-fold increase in P-M∅ lysosomal enzyme activity compared to the control.
Another study also reported that EFR (Erom’s fermented rice bran) possessed macrophage stimulation activity through experiments with P-M∅ (C57/BL6 mice) and RAW264.7 cell line. Dose-dependent significant increases (
p < 0.05) in NO production were observed by Kang et al. [
53] when RAW264.7 cells were treated with 0.5, 1, 2, 4, and 8 μg/mL of EFR, compared to untreated control. The level of NO production in 8 μg/mL of EFR was comparable to that of 1 μg/mL of LPS. At the gene expression level, the mRNA expression of inducible nitric oxide synthase (iNOS), a key marker of M1 macrophage activation (Xue et al., 2018), was detected by reverse transcriptase (RT) polymerase chain reaction (PCR) to increase in a similar trend under different EFR treatment concentrations. RAW264.7 cells treated with EFR (0.5–8 μg/mL) also demonstrated a significant increase (
p < 0.05) in IL-1
, IL-10, IL-6, and TNF-
secretion dose-dependently when compared to control. The lysosomal enzyme activity of P-M∅ treated with EFR was significantly increased (
p < 0.05) to 134.9–142.2% at a concentration of 0.5–8 μg/mL relative to the control. Such levels are at par with the 135.8% increase demonstrated by LPS [
53].
Kim et al. [
52] investigated the macrophage stimulation abilities of three variants of RBAC from STR Biotech: CFP (a crude fermentation-polysaccharide CFP extracted from black rice bran cultured with
L. edodes), a secondary CFP product fermented with
Lactobacillus gasseri (CFP-L), and another secondary CFP extract from fermentation with baker’s yeast,
S. cerevisiae (CFP-S). P-M∅ extracted from Balb/c mice orally administered with 250 mg/kg BW/day of treatment for 4 weeks showed lysosomal enzyme activities of 104.60 ± 10.97% (CFP), 115.21 ± 18.94% (CFP-S), 97.99 ± 16.79% (CFP-L) relative to control. In the presence of LPS, however, macrophage activity significantly increased (
p < 0.05) for CFP (113.21 ± 13.10%) and CFP-S (138.78 ± 24.41%) but decreased for CFP-L (93.60 ± 15.34%). Hence, the macrophage stimulation effect of CFP was enhanced by a secondary fermentation with
S. cerevisiae but dampened by adding
L. gasseri.
Kim et al. [
24] found that polysaccharides extracted from a combined culture of black rice bran and
L. edodes (BPP from STR Biotech) could increase the lysosomal enzyme activity level of P-M∅ (BALB/c mice) in vitro by about 5.4-fold at 10 μg/mL (15% to 81%) and 2.5-fold (49% to 120%) at 100 μg/mL, compared to the polysaccharides of
L. edodes without black rice bran. The macrophage stimulation activity of BPP at 100 μg/mL was also higher than the level stimulated by 100 μg/mL of LPS. Although BPP was not bactericidal in vitro against
Salmonella typhimurium, RAW 264.7 macrophages pretreated with 1, 10, and 100 μg/mL BPP demonstrated 1.4-, 2.4-, and 3.5-fold increases in bacterial uptake rates against
S. typhimurium compared to untreated macrophages, respectively (Kim et al., 2014). The phagocytotic stimulatory effect of BPP was also tested in vivo. In female BALB/c mice inoculated with
S. typhimurium in the peritoneal cavity, pretreatment with 10 mg/kg BW of BPP orally or i.p. showed significantly reduced (
p < 0.05) bacteria growth compared to control.
Further experiments also revealed that BPP, at 100 μg/mL, could induce RAW 264.7 monocyte/macrophage cells to morphological change into the dendrite-like structure, reaching up to 3.6-, 10.3-, and 12.4-fold changes after 2, 4, and 8 h of incubation (Kim et al., 2014). It was observed that BPP-treated cells (RAW 264.7) that were subsequently infected with
S. typhimurium showed more significant intracellular bacteria presence (
p < 0.05) after 2 h compared to the control. In contrast, the bacterial count markedly decreased (
p < 0.05) relative to the control after 4 and 8 h. The results suggested an initially enhanced engulfment of bacteria by the BPP and a subsequent switch to intracellular bacteriolysis through activation of T helper type 1 (Th1) cell cytotoxic reaction initiated and directed by M1 macrophages over time [
57,
59].
BPRBE from STR Biotech was also shown to significantly increase (
p < 0.05) NO production of RAW 264.7 macrophage cells compared to non-bioprocessed rice bran extract and vehicle control [
23]. The NO formation levels were dose-dependent (1, 10, and 100 μg/mL), and at 10 μg/mL or greater doses, they were significantly higher than that obtained with LPS [
23]. Through RT-PCR and western blot analysis, Kim et al. [
23] confirmed mRNA expression level of the iNOS gene of
Salmonella-infected RAW 264.7 cells pretreated with BPRBE was about 1.9-fold higher than that in the infected control cells without treatment. The phagocytotic effects of BPRBE on
S. typhimurium-infected RAW 264.7 cells were also enhanced significantly (
p < 0.05) with 1.3-, 2.3-, and 3.4-fold rate increment compared to control with 1, 10 and 100 μg/mL BPRBE treatment, respectively.
Salmonella was known to downregulate the expression of the autophagy-related membrane proteins (Beclin-1, Atg5, Atg12, Atg16L) of infected cells to evade autophagic recognition. Through, Western blot analysis, BPRBE treatment was found to upregulate the expression of these autophagy-related proteins regardless of bacterial infection [
23]. Additionally, BPRBE treatment (100 μg/mL) also markedly induced (
p < 0.05) the production of IFN-
in the
Salmonella-infected macrophage cells via the interferon regulatory transcription factor 3 (IFR3) pathway [
23]. In short, RBAC possesses remarkable macrophage-stimulating effects based on the available in vivo and in vitro evidence, as depicted in
Figure 3.
3.5. Natural Killer Cell Activity
Natural killer cells (NKC), representing about 5–20% of circulating lymphocytes in humans, are large granular lymphocytes developed and matured in the bone marrow and secondary lymphoid tissues such as tonsils, spleen, and lymph nodes [
60]. As part of the innate immune system, NKC are naturally cytotoxic and can detect and destroy viral-infected or tumorigenic cells without priming by expressing a myriad of activating and inhibitory cell surface receptors [
61]. Hence, harnessing NKC’s killing power has been a strong interest in suppressing malignant neoplasm since its first discovery in the 1960s [
62]. The bioactivities of RBAC on NKC have been studied extensively in vitro and in vivo.
Table 4 summarises the effects of RBAC on NKC activity based on the available preclinical evidence.
In vitro experiments with the PBL from healthy donors (
n = 6) cultured with Biobran MGN-3 showed significant increases (
p < 0.001) in NKC cytotoxicity tested with chromium (
51Cr) release assay. The effect was dose-dependent (130% at 25 μg/mL vs. 150% at 100 μg/mL) [
63]. Biobran MGN-3 also increased IFN-
production significantly (
p < 0.001), showing 340, 390, and 580 pg/mL of IFN-
production at 25, 50, and 100 μg/mL, respectively, compared to <100 pg/mL in control. Quantification of total NKC, however, indicated no increase in the percentage of NKC populations in PBL cultured with Biobran MGN-3 of all concentrations as identified by the CD56+/CD3− and CD16+ markers [
63]. Hence, Biobran MGN-3 increased the cytotoxicity of existing NKC but not the cell count.
A follow-up study on the in vitro effects of Biobran MGN-3 on NKC by Ghoneum and Jewett [
64] found significant increases in NKC cytotoxicity in both PBL (
p < 0.001) and purified NKC (
p < 0.01) by Biobran MGN-3 (500 μg/mL). The NKC augmentation effect of Biobran MGN-3 was enhanced by co-culturing with IL-2 (500 U/mL) in PBL but not in purified NKC. Biobran MGN-3 (100 and 1000 μg/mL) was also shown to significantly increase (
p < 0.001) the secretion of TNF-
in PBL samples collected from 25 donors in a dose-dependent manner. However, there were variations across individuals with responses categorised into three groups at 1000 μg/mL: ≤20-fold (40%,
n = 10), 20 to 100-fold (24%,
n = 6), and >100-fold (36%,
n = 9). TNF-
secretion by PBL cultured with Biobran MGN-3 (1000 μg/mL) was also significantly enhanced (
p < 0.001) with the presence of IL-2. In contrast, TNF-
secretion in purified NKC treated with Biobran MGN-3 (1000 μg/mL) did not increase further with the addition of IL-2. These experiments showed that while Biobran MGN-3 worked synergistically with IL-2 to increase NKC cytotoxicity and TNF-
in PBL, Biobran MGN-3 alone could enhance the NKC cytotoxicity, as demonstrated by its effects on purified NKC [
64].
Production of IFN-
by PBL was tested with samples from 14 donors by Ghoneum and Jewett [
64]. Again, Biobran MGN-3 (100 and 1000 μg/mL) treatment significantly increased (
p < 0.03) IFN-
secretion with variation across individuals. Adding IL-2 resulted in a synergistic increase (
p < 0.03) in IFN-
production in all samples, and the effect was dose-dependent. Furthermore, a synergistic increase in IFN-
production (
p < 0.01) was also observed when adding IL-2 to Biobran MGN-3 treatment of purified NKC.
Ghoneum and Jewett [
64] further explored the mechanisms of NKC activation by Biobran MGN-3. CD69 cell surface receptor, CD54 (intercellular adhesion molecule-1, ICAM-1), and CD25 (IL-2 receptor alpha chain) expressions of NKC were all observed to be upregulated in PBL treated with Biobran MGN-3 comparable to those with IL-2. Upregulation of CD69, CD54, and CD25 was also observed in other lymphocytes after Biobran MGN-3 treatment but to a lower extent.
The enhancement of depleted NKC activity in aged C57BL/6 and C3H female mice (18 months old) by Biobran MGN-3 was explored by Ghoneum and Abedi [
65]. Biobran MGN-3 (0.1 mL with 10 mg/mL, i.p.) was reported to significantly increase the cellularity (404–470%,
p < 0.01) and NKC activity against YAC-1 target cells (4.8–6-fold increase,
p < 0.01) in the peritoneal cavity after 2 days with the levels remained elevated through to the 14th day. However, the treatment did not increase NKC activity in the spleen or the bone marrow, even though a significant increase in splenic cellularity was observed (145–192%,
p < 0.025). In contrast, oral feeding of Biobran MGN-3 at the same dose elicited a 200% increase (
p < 0.01) in splenic NKC activity in C57BL/6 mice 14 days after treatment but did not affect peritoneal NKC activity and cell count compared to the control. For comparison, the in vitro NKC activity of splenic lymphocytes isolated from aged C57BL/6 mice exhibited a 4-fold increase (
p < 0.01) over the control after culturing with Biobran MGN-3 at 25 and 100 μg/mL.
Furthermore, the peritoneal NKC of C57BL/6 mice (
n = 3) demonstrated a remarkable increase in BLT-esterase activity (
p < 0.01) and granularity after 5 days of i.p. treatment, which were alternative markers for increased cytotoxicity [
65]. The binding capacity of peritoneal NKC of mice treated with Biobran MGN-3 was also reported to have significantly higher percentage conjugates (26% vs. 13%,
p < 0.01), a 2-fold increase, compared to the control. Ghoneum and Abedi [
65] also found the presence of P-M∅ could substantially lower (
p < 0.025) the in vivo peritoneal NKC activity.
Badr El-Din et al. [
66] investigated the in vivo effect of Biobran MGN-3 on NKC activity in young female Swiss albino mice (2 months old) administered with 100 μg/mL/day of Biobran MGN-3 with intramuscular injection for 14 days. The splenic NKC activity was measured against the YAC-1 tumour target cells. The results showed a significant increase in NKC activity in the treated mice (27.1 [lytic units] LUs vs. 8.3 LUs,
p < 0.01) compared to the control. Furthermore, a two-fold increase (27.5% vs. 14%,
p < 0.01) in the NKC binding capacity in terms of the percentage of conjugate formation to target cells was also detected.
Likewise, the splenic NKC activity (against YAC-1 target cells) of male Lewis rats fed with 250 mg/day of Biobran MGN-3 as a dietary supplement after two weeks was also enhanced significantly (
p < 0.045) compared to rats fed with only a control diet, as reported by Giese et al. [
67]. Such an increase in activity was associated with elevations of IL-2 (
p < 0.05) and IFN-
(
p < 0.08) production in splenocytes. The effect of RBEP on the splenic NKC activity of healthy mice was investigated by Kim et al. (2007). Male ICR mice (4 weeks old) were fed orally with 50 mg/kg BW or 250 mg/kg BW of RBEP for 5 days. RBEP was shown to enhance the NKC activity against YAC-1 cells significantly, reporting 32.8 LUs (50 mg/kg,
p < 0.05) and 46.3 LUs (250 mg/kg,
p < 0.01) compared to the 22.8 LUs of the controls.
Pérez-Martínez et al. [
58] conducted in vitro cytotoxicity assays of NKC isolated from PBL of healthy volunteers against multiple cancer cell lines (K562, NB1691, Jurkat, A673, A-04, RD, RH30). Significant increases in NKC cytotoxicity against all tested cell lines (
p < 0.05) compared to resting NKC were reported after overnight stimulation with Biobran MGN-3 (100 μg/mL). The cytotoxicity measured in percentage lysis of treated NKC ranged from 34% to 80% in different cell lines compared to 13% to 69% of the resting NKC. Such levels were less than IL-5 (10 ng/mL) stimulated NKC. Nonetheless, Biobran MGN-3 (100 μg/mL) was found to have a synergistic effect in significantly enhancing the stimulatory effect (
p < 0.05) of low dose IL-2 (40 IU/ml) on NKC cytotoxicity level to that obtained with 1000 IU/mL of IL-2. Regarding phenotyping, NKC stimulated with Biobran MGN-3 showed increased expression of CD69 and CD25 by 3.1-fold and 3.2-fold over resting NKC (measured in mean fluorescence intensity), respectively. However, Biobran MGN-3 did not affect the expression of other receptors on NKC, including NKG2D (natural killer group 2D), DNAM (DNAX accessory molecule), NCRs (nuclear receptor coregulators) and TLRs (toll-like receptors), unlike IL-5, the positive control [
58]. The biological effects of RBAC on NKC are summarised in
Figure 4.
RBAC’s ability to enhance NKC activity in a human clinical trial was first reported by Ghoneum [
63]. Healthy adults (
n = 24) were divided into three groups and received Biobran MGN-3 in doses of 15, 30, and 45 mg/kg BW/day (
n = 8 in each group) for two months. The NKC activities in PBL were tested against K562 tumour cells at baseline and after 1 week, 1 month, 2 months, and 3 months. Biobran MGN-3 treatment at all concentrations demonstrated enhanced NKC cytotoxicity against K562 over time with significant differences over baseline achieved at 1 week (∼3.1-fold,
p < 0.001) for 30 and 45 mg/kg dose and after 1 month (2-fold) for 15 mg/kg dose. Peak response was recorded after 2 months (∼5-fold) for all doses, and the NKC activity returned to baseline after 1 month of discontinuing treatment. Furthermore, after 1 month of treatment at 45 mg/kg BW/day, the binding capacity of NKC to K562 tumour targets, measuring in the percentage of conjugate formations, increased significantly over baseline (38.5% vs. 9.4%,
p < 0.005). A similar trend of augmented NKC effect was also observed against the more resistant Raji tumour cells with 45 mg/kg dose [
63].
Two other human studies also evaluated RBAC’s ability to enhance NKC activity in healthy adults. Ali et al. [
69] recruited 20 adult volunteers (mean age = 33.6 ± 13.2) randomly assigned to take either 1 g/day or 3 g/day of Biobran MGN-3 for 60 days. Peripheral blood-derived mononuclear cells (PBMC) from these participants were obtained at baseline, 48 h, 1 week, 30 days and 60 days (5 time points) for evaluation. The results showed a significant effect for time (
p = 0.001) based on repeated measure analysis of variance (ANOVA) on the NKC activity against K562 cell lysis but not for the group and the interaction of group and time. Total NKC activity peaked at 1 week and was significantly higher than the values at other time points. Unlike the previous study by Ghoneum [
63], the NKC activity did not remain elevated throughout the treatment period but dropped back to baseline levels at 30 and 60 days.
Choi et al. [
26] investigated the effects of RBEP on the NKC activity compared to placebo in healthy participants in a double-blind, randomised controlled trial (RCT). The participants in this study (
n = 80) were randomly assigned to consume either 3 g/day of RBEP or placebo powder for 8 weeks. NKC activity against K562 cells was tested with the PBL of participants collected from baseline, 4 weeks, and 8 weeks. This study, however, did not find any significant effect of RBEP supplementation over placebo at all time points. Such a finding was consistent with what was reported by Ali et al. (2012) since any initial stimulating effects by RBAC would have subsided after 4 weeks, contrasting the findings of Ghoneum [
63]. Possible explanations could be individual differences, with some healthy participants more receptive to the NKC stimulation effect of RBAC or product differences. Hence, more research on how RBAC could affect the NKC of healthy adults is warranted.
Biobran MGN-3 could also restore NKC activity in individuals with weakened immunity due to exogenous sources. Ghoneum [
70] reported a study among 11 individuals who had workplace chemical exposure and received Biobran MGN-3 at 15 mg/kg BW/day for 4 months. The participants had low levels of NKC activity (10.2 ± 4.2 LUs) at baseline, but treatment with MGN-3 increased NKC activity 4- and 7-fold at 2 and 4 months, respectively.
Among older adults under institutional care (age 75–90), consuming Biobran MGN-3 (500 mg/day) for two weeks did not significantly affect the NKC compared to control according to a cross-over randomised RCT (
n = 32) by Tazawa et al. [
71]. However, a subgroup analysis by Maeda et al. [
72] of the same study showed that those with low NKC activity at baseline (
n = 12, ≤30%) had a higher increment in activity during Biobran MGN-3 treatment (34.1% increase) compared to the control (15.6% increase). However, the between-group difference was not statistically significant.
In another study with a younger cohort of older people, Elsaid et al. [
73] showed that consuming Biobran MGN-3 could enhance NKC activity after 1 month in a placebo-controlled RCT. The study recruited 12 participants over 56 years old and randomly assigned to take either 500 mg/day of Biobran MGN-3 or a placebo (
n = 6 in each group). Blood samples were analysed for NKC expressing CD107a, a functional marker for NKC activation, before and after the trial. The median CD107a expression in the Biobran MGN-3 significantly increased from 60.5% to 83.0% posttreatment (Wilcoxon,
p < 0.046), whereas no significant differences were detected in the placebo group. However, the percentage of NKC in blood did not differ between groups and over time [
73].
Such findings were also confirmed by Elsaid et al. [
74] in a study with randomly selected geriatric volunteers (age ≥ 56) from a larger placebo-RCT (
n = 80). Twelve participants who took 500 mg/day of Biobran MGN-3 or placebo (6 in each group) were tested for NKC CD107a expression before treatment and after study completion at 3 months. Mean NKC CD107a expression significantly increased (
p = 0.004) from 49.5 ± 10.4% to 75.2 ± 6.6% for the Biobran MGN-3 group compared to insignificant difference pre- and posttreatment values in the placebo group (45.3 ± 12 vs. 50.8 ± 19.5). Hence, the enhanced NKC activity stimulated by RBAC could be sustained for 3 months in the geriatric group.
Table 5 summarises RBAC’s effects on the NKC activity of healthy adults with evidence from human clinical trials.
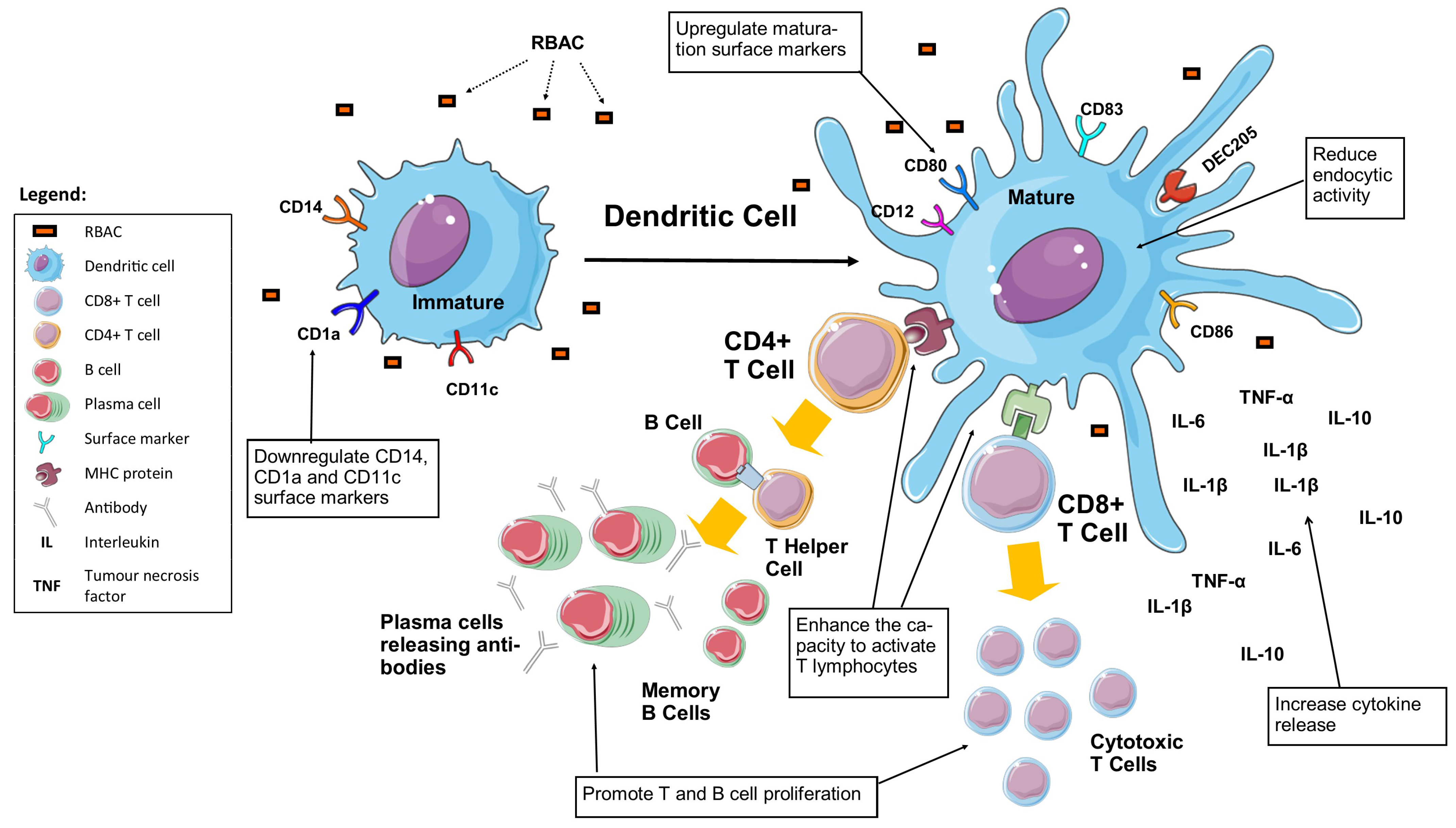
3.6. Dendritic Cell Maturation
Dendritic cells (DC) are considered professional antigen-presenting cells in the immune system, alongside macrophages and B cells [
75]. Like macrophages, DC sense and engulf invading pathogens [
76]. Instead of directly destroying an invading pathogen, DC communicate to T lymphocytes by antigen presentation to MHC complexes or other cell surface molecules, initiating long-lasting antigen-specific responses [
77]. Furthermore, DC also modify ongoing immune responses through the secretion of cytokines and growth factors while interacting with other immune cells, such as NKC [
78]. As such, DC are regarded as master regulators of the immune system, which play a critical role in bridging innate to adaptive immune responses [
77]. DC can be found in practically all tissues but remain in the ‘immature’ state until tissue homeostasis disturbances are triggered by PAMP, damage-associated molecular patterns, proinflammatory cytokines, or pathogens. Maturation involves significant changes in surface proteins, intracellular pathways and metabolic activity, together with the migration of DC from peripheral tissue to secondary lymphoid organs where T lymphocyte activation may occur [
78].
RBAC also possesses the capacity to upregulate the maturation of DC with evidence from research conducted on monocyte-derived DC, as summarised in
Table 6.
Cholujova et al. [
79] investigated the modulatory effects of RBAC on human DC differentiation and maturation using monocyte-derived DC in vitro. Immature DC (iDC) were derived from peripheral monocytes isolated from the buffy coat blood of healthy human donors. Cytokine maturation mix (CMM1) (TNF-
, IL-1
and IL-6) or CMM2 (LPS and IFN-
) was used to induce DC maturation co-cultured with different concentrations of BioBran MGN3 (0, 10, 100, 400 and 1000 μg/mL) to assess the maturation processes. The study found BioBran MGN3 down-regulated the expression of CD14 (a monocyte marker) and CD1a (an antigen-presenting molecule) on the surface of iDC while markedly increasing CD83 expression (a DC maturation marker) in a dose-dependent manner.
Similar observations were made in both matured DC samples (matDC1 and 2, matured with CMM1 and 2, respectively) with higher intensity of up- or down-regulation compared to the level seen in iDC using the same dose of BioBran MGN3 [
79]. Furthermore, the endocytic activity of iDC (evaluated by the uptake of FITC-conjugated dextran at 37 °C) also reduced from 73% to 27.7% (100 μg/mL), 17.7% (400 μg/mL), and 14.4% (1000 μg/mL) under different concentrations of BioBran MGN3, approaching the levels of mDC (14.9% for matDC1 and 5.6% for matDC2). BioBran MGN3 increased the surface density of costimulatory molecules CD80 and CD86 on iDC and further enhanced the CD80 and CD86 density on matDC1 and matDC2. Furthermore, the study also found iDC, matDC1, and matDC2 expressed higher levels of CD123 (IL-3 receptor
chain) and lower levels of CD11c cell surface antigens, the phenotype represented by CD123+/CD11c- plasmacytoid DC, in the presence of BioBran MGN3 [
79].
In parallel, the stimulatory effects of BioBran MGN3 were also validated by Ghoneum and Agrawal [
80] utilising the monocyte-derived DC isolated from PBMC of healthy donors. DC were treated for 24 h with BioBran MGN3 (0, 5, 10 and 20 μg/mL) or LPS (1 μg/mL) as a positive control. Activation of DC was determined by assessing the expression of costimulatory and maturation markers (CD40, CD80, CD83, CD86 and HLA-DR). Flow cytometry analysis showed a dose-dependent upregulation in CD83 and CD86 surface markers similar to the effects of LPS on DC. Moreover, the BioBran MGN3 treated DC also produced significantly higher levels (
p < 0.05) of IL-1
, IL-6, IL-10, TNF-
, IL-12p40, IL-12p70, and IL-2, compared to untreated DC. In comparison, LPS also significantly induced the production of these cytokines in DC, except IL-1
. In particular, BioBran MGN3 increased the production of IL-1
from DC in vitro in a dose-dependent manner [
80].
The BioBran MGN3-activated DC also demonstrated the capacity to boost T-cell proliferation in vitro [
80]. Co-culturing of DC pretreated with 10 μg/mL BioBran MGN3 with allogeneic CD4+ T cells increased the proliferation by 1.4-fold, a 73.6% increase compared to CD4+ T cells cultured with untreated DC. Such activation level is similar to that of DC activated with LPS. Furthermore, the study also detected a 1.3-fold increase in the expression of CD25 markers on the T lymphocytes co-cultured with BioBran MGN3 (10 μg/mL) induced DC compared to the control. In terms of cytokine production from T lymphocytes, significant increases (
p < 0.05) in IFN-
, IL-10, and IL-17 were reported in Biobran MGN-3 (10 μg/mL) primed DC-induced T lymphocytes compared to control [
80].
In a follow-up study, Ghoneum and Agrawal [
81] continued to explore the effects of BioBran MGN3-primed DC on cytotoxic CD8+ T lymphocytes. Again, monocyte-derived DC isolated from PBMC of healthy donors were incubated with BioBran MGN3 (20 and 40 μg/mL) for 24 h. DC treated with BioBran MGN3 showed signs of maturation with increased DEC-205 expression for antigen-presenting in a dose-dependent manner. Treatment of 20 μg/mL of BioBran MGN3 also significantly increased IL-29 production in DC (
p < 0.05) compared to the control. At the same time, increased secretion of IFN-
and IFN-
were also detected but did not achieve statistical significance. Culturing the activated DC (by 20 μg/mL BioBran MGN3) with purified, allogeneic CD8+T cells for 7 days resulted in significantly higher levels of granzyme B-positive CD8+ T cells (
p < 0.05) as compared to unstimulated DC treated CD8+ T cells. Granzyme-expressing CD8+ T cells possess an increased capacity to kill tumour cells. Experiments by Ghoneum and Agrawal [
81] further demonstrated that CD8+ T cells cultured with BioBran MGN3-activated DC had increased cytotoxicity against tumour cell targets significantly (
p < 0.05) compared to those treated with unstimulated DC.
Figure 5 is a graphical illustration of the maturation of DC under the influence of RBAC.
3.7. T and B Cell Proliferation
The T and B cells are the two main types of lymphocytes of the adaptive immune response, which are antigen-specific. A distinct T and B cell population recognises and responds to a specific antigenic epitope [
82]. Developed from stem cells in bone marrow while matured in the thymus, naïve T cells constantly migrate to secondary lymphoid organs, including lymph nodes, tonsils, spleen, and Peyer’s patches, where they can encounter antigen presented by the antigen-presenting cells such as DC and macrophages. Once activated, naïve T cells differentiate into effector cells with specialised phenotypes. Among them are cytotoxic CD8+ T cells, which can attack and destroy malignant or virus-infected cells via apoptosis induction, and CD4+ T cells are T helper cells that produce cytokines and stimulate B cells to differentiate into antibody-secreting plasma cells [
82]. B lymphocytes are also developed and differentiated from stem cells in bone marrow, but unlike their T counterpart, B cells continue to mature in the bone marrow. Mature B cells also migrate to secondary lymphoid organs and can transform into plasmocytes for producing specific antibodies after activation [
82].
As presented in the previous section, RBAC is an inducer of DC maturation to initiate the adaptive immune response of T and B cells. Ghoneum [
70] also observed in participants (
n = 11) with chemical exposure the increase of T and B cell mitogens by 130–150% compared to baseline after consuming Biobran MGN-3 (15 mg/kg/day) for 4 months (
Table 5). Several preclinical studies confirmed RBAC’s effects on enhancing T and B cell proliferation in the secondary lymphoid organs. A summary of the evidence on RBAC’s capacity to promote splenic T and B cell proliferation is shown in
Table 7.
Using rosette-forming cell assay, Bae et al. [
83] reported that the splenocytes of BALB/c mice fed with 1.5 mg/day of Biobran MGN-3 for 10 days could significantly induce rosette formation due to higher antibody production by 30% (
p < 0.005) compared to the control and the level was also higher than splenocytes of mice fed with PSP (1.5 mg/day). Similarly, the splenocyte plague formation was also highest in the Biobran MGN-3 group, with a 14% increase over the control group, compared to 11% higher than the control in the PSP group. Hence, these results showcase the in vivo capacity of Biobran MGN-3 in B cell activation and T cell proliferation [
55,
83].
Biobran MGN-3 also exhibited in vitro effects in inducing splenocyte proliferation in a dose-dependent manner (1, 10, 100, 1000 μg/mL), albeit at a much lower level than LPS (which proliferated B cells specifically), concanavalin A (Con A, a T cell proliferation inducer), and PSP. Biobran MGN-3 also induced IFN-
secretion like LPS in splenocytes [
83]. Giese et al. [
67] confirmed such observations in vivo with male Lewis rats fed with Biobran MGN3 (0.25 g/day) for 14 days. Splenocytes of Biobran MGN3-fed rats (
n = 12) showed significantly higher proliferative activity against the superantigen toxic shock syndrome toxin-1 compared to those from the control rats associated with a higher level of IFN-
secretion. In contrast, Sudo et al. (2001) reported no significant difference in the splenocyte count and lymphocyte subsets of BALB/c mice fed with Biobran MGN-3 (0, 0.25%, and 0.5%) after 12 h of restrain stress. Significant reductions of splenocyte counts (
p < 0.001) in all three groups compared to baseline were observed [
67]. Hence, Biobran MGN-3 did not prevent the decrease in immune cell count due to psychological stress.
Kim et al. [
45] also conducted a splenic lymphocyte proliferation assay with RBEP (10 and 100 μg/mL) and reported a 1.39- and 1.44-fold increase in cell proliferation levels in vitro relative to untreated control. However, the levels were lower compared to those produced with LPS and Con A. Similarly, Kang et al. [
53] demonstrated that EFR could significantly increase splenocyte proliferation by 1.33 to 1.74 times (
p < 0.05) compared to the control group at different concentrations in vitro (0.5 to 8 μg/mL). These results are consistent with the observation by Ghoneum and Abedi [
65] that Biobran MGN-3 (0.1 mL with 10 mg/mL, i.p.) significantly increased splenic cellularity (145–192%,
p < 0.025) in aged mice. Since the spleen cell population consists of all types of immune cells, further experiments with purified T and B cells by Chae et al. [
55] showed that Biobran MGN-3 had minimal in vitro effects on splenic T cell proliferation and only induced B cell proliferation at a very high concentration (1000 μg/mL). Hence, the observations confirmed that while Biobran MGN-3 could induce immune cell proliferation in the spleen, it only indirectly affects T and B cell proliferation, likely by enhancing DC and macrophages’ antigen-presenting capability, as shown in
Figure 5 earlier.
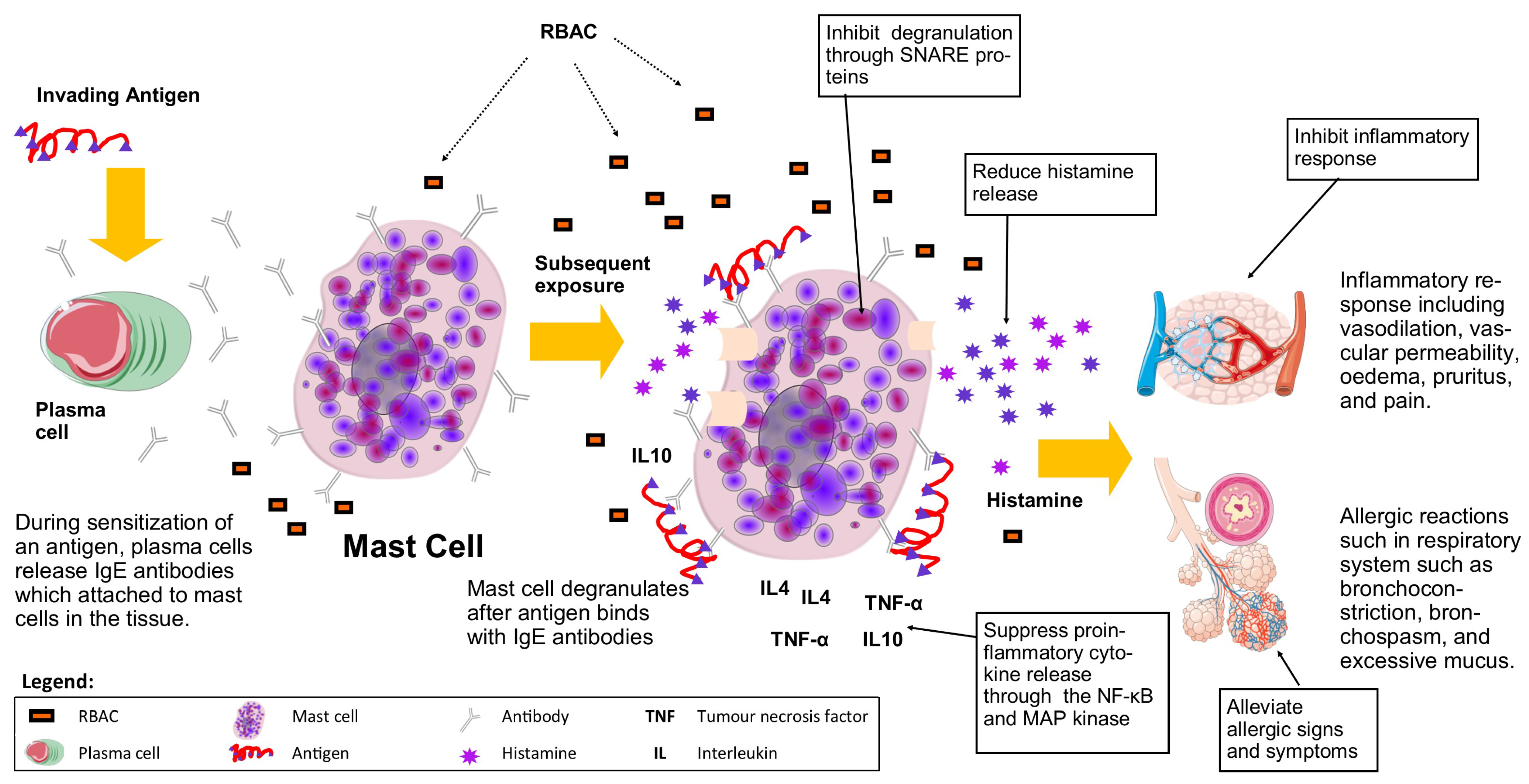
3.8. Mast Cells, Allergy, and Inflammation
The immune system is often considered a double-edged sword. While the system protects the host against infections and malignancies, an overreacting immunity could lead to detrimental consequences such as allergies, chronic inflammation, or autoimmune conditions. As RBAC has shown the capacity to stimulate different immune cells and enhance their activity, it is also essential to understand the role of RBAC during exaggerated immune responses in the host. Mast cells are immune cells widely distributed in host tissues and play a crucial role in the inflammatory response during infection and allergic reactions by releasing histamine and proinflammatory cytokines. Several studies were conducted to investigate the effects of RBAC on mast cells in various hypersensitivity scenarios, including anaphylaxis, asthma, and dermatitis, as shown in
Table 8.
Kambayashi and Endo [
84] studied the antiallergy effect of Biobran MGN-3 with a toluene diisocyanate (TDI)-induced asthmatic mice model. Female Balb/c mice sensitised with TDI were divided into four groups. Group A consumed Biobran MGN-3 (2 g/L dissolved in drinking water) one month before sensitisation and continued throughout the sensitisation and induction periods. Group B was administered Biobran MGN-3 (as prevention) one month before sensitisation and continued until the end of the sensitisation period. Group C was fed Biobran MGN-3 only during the TDI induction period (for symptom reduction). The last (D) was a control group that received no Biobran MGN-3. The effect of Biobran MGN-3 was evaluated by blood histamine concentration, eosinophil count in bronchoalveolar lavage fluid (BALF), TDI ear provocation test, and blood antibody titres (immunoglobulin [Ig] G1, IgG2a, and IgE) at the time of induction [
84].
The study found the lowest blood histamine concentration in group A seven minutes after TDI induction at 2.5 ± 0.53 ng/mL. The levels were 4.2 ± 0.75 ng/mL in group B and 4.3 ± 0.78 ng/mL in group C [
84]. All were significantly lower than the 6.4 ± 0.87 ng/mL of group D (
p < 0.05). Furthermore, a 10- to 100-fold decrease in sensitivity compared to control was observed in the treatment groups with TDI ear provocation test at 0.01–10% concentrations. Biobran MGN-3 administration also significantly lowered the BALF eosinophil counts of the mice compared to the control. However, no effects on IgG and IgE production were found, suggesting that Biobran MGN-3 was a suppressor of mast cell activity during hypersensitivity [
84].
In another study, Bae et al. [
83] utilised the passive cutaneous anaphylaxis (PCA) titre to evaluate the antiallergy effect of Biobran MGN-3 in a murine model. All mice (Balb/c mice,
n = 24) were sensitised with egg albumin as an allergen through subcutaneous injection before receiving Biobran MGN-3 or PSP (both at 1.5 mg/day p.o.) or saline (0.4 mL/day p.o.) for 14 days. PCA was carried out to induce an immediate dermal response caused by IgE and allergen reaction, showing as blue spots at the intradermal injection site. Biobran MGN-3-fed mice showed only 20 spots compared to 40 in the PSP group and 80 among the saline control mice (Bae et al., 2004). The plasma histamine content in the Biobran MGN-3-fed mice was also 25% lower than the control and PSP (6.5% lower) groups. Hence, in the presence of an allergen, Biobran MGN-3 downregulated the release of histamine from mast cells to reduce the inflammatory response [
83].
Hoshino et al. [
86] confirmed the in vitro effects of Biobran MGN-3 on mast cells. Using bone marrow-derived mast cells from BALB/c mice, the study reported that pretreatment with Biobran MGN-3 (0, 200, 500, 1000, 3000 μg/mL) for 30 min resulted in significant inhibition (
p < 0.01) of
-hexosaminidase release (a marker for mast cell degranulation) after antigen stimulation in a dose-dependent manner. Notably, secretions of TNF-
and IL-4 by mast cells also demonstrated a significant dose-dependent reduction (
p < 0.01) after antigen stimulation in mast cells pretreated with Biobran MGN-3. Follow-on investigations by Hoshino et al. [
86] also revealed that Biobran MGN-3 pretreatment did not affect the early events of mast cell signal transduction (IgE binding, Ca2+ mobilisation, and phosphorylation of Akt) but significantly inhibited (
p < 0.05) the distal event of membrane fusion between liposomes during degranulation activities mediated by soluble N-ethylmaleimide-sensitive factor attachment protein receptor-derived (SNARE) peptides. Moreover, suppression of the proinflammatory cytokine (TNF-
and IL-4) syntheses by Biobran MGN-3 pretreatment was shown to be through the nuclear factor kappa B (NF-
B) and mitogen-activated protein (MAP) kinase (extracellular signal-regulated kinase [ERK] and c-Jun N-terminal Kinase [JNK]) activation pathways [
86].
Similarly, Kim et al. [
85] confirmed that treatment with fermented SuperC3GHi bran (100 and 250 μg/mL) significantly reduced (
p < 0.05) the release of
-hexosaminidase from RBL-2H3 mast cell line stimulated with the allergen sensitiser Compound 48/80. The
-hexosaminidase activity of mast cells was decreased by 25% (100 μg/mL) and 40% (250 μg/mL) compared to the control with no treatment. Analysis of histamine secretion also showed a similar trend, with a statistically significant 18% reduction (
p < 0.05) recorded in mast cells treated with 100 μg/mL fermented SuperC3GHi bran and 43% with 250 μg/mL. Further experiments were carried out by Kim et al. [
85] to assess the anti-inflammatory effect of fermented SuperC3GHi bran in an ear-oedema mice mode. Arachidonic acid (3%) was applied to the ears of ICR mice to induce inflammation via increased production of prostaglandins. Topical applications of the fermented SuperC3GHi bran were shown to reduce the inflammatory response by 35.6% compared to the controls. Such inhibition rate was less effective than indomethacin (68.5% reduction), a nonsteroidal anti-inflammatory drug, used as a positive control [
85].
An [
40] explored the antiallergy effects of RBEP on dinitrochlorobenzene (DNCB)-induced contact hypersensitivity in NC/Nga mice. The mice (
n = 21) were divided equally into RBEP (250 mg/kg BW/day for 4 weeks), negative and positive control groups. Mice in RBEP and negative control groups were induced with dermatitis but not the positive controls. Significantly lower symptom scores (
p < 0.05) in the RBEP group compared to the negative control group were recorded throughout the study during weekly assessments of skin lesions. Even though rising IgE concentrations in plasma and splenocytes were observed in the RBEP group compared to the positive controls at the end of the experiment, the levels were significantly lower (50% less) than those in the negative controls (
p < 0.05). The RBEP group also recorded significantly lower IL-4 and IL-10 in both plasma and splenocytes compared to the negative controls (
p < 0.05). The splenocyte IFN-
and IFN-
/IL-4 ratio of the RBEP group remained normal compared to the positive controls. In comparison, the negative control group registered significantly lower (
p < 0.05) levels of IFN-
and IFN-
/IL-4 [
40]. DNCB is a known allergen that causes dermatitis by forming immune complex deposits (IgG1 conjugates) that activate the complement system, contributing to the development of hypersensitivity reactions [
87]. Hence, RBEP alleviated the allergic reaction to DNCB by preventing the cytokine production imbalance favouring Th2 responses (humoural immunity of B cells).
Figure 6 summarises the role of mast cells in an allergic reaction and the effects of RBAC on mast cells to alleviate the allergic signs and symptoms and inhibit inflammatory responses.
3.10. Effects on Circulating Cytokines
As discussed in the previous sections, many preclinical studies have demonstrated RBAC’s ability to modulate cytokine production for priming macrophages, augmenting NKC activity, inducing DC maturation, promoting T and B cell proliferation, and downregulating mast cells in allergic responses. Cytokines are potent immune modulators often produced in a cascade, with one cytokine stimulating the target cells to produce additional cytokines [
95]. Cytokines can be pro- or anti-inflammatory. Proinflammatory cytokines, the key ones being IL-1
, IL-6, and TNF-
, are involved in the upregulation of inflammatory reactions, resulting in pathological signs of pain, heat, redness, swelling, and loss of function [
95,
96]. Major anti-inflammatory cytokines are the IL-1 receptor antagonist, IL-4, IL-10, IL-11, and IL-13, and they attenuate inflammation to allow tissue repair and regeneration [
97]. Dysregulation of cytokine production can lead to an inflammatory state which increases the risk of chronic diseases, of which aging is an associative factor [
98,
99]. As such, circulating cytokine levels, especially proinflammatory cytokines, have been suggested as a potential biomarker for inflammatory diseases and aging [
100,
101,
102].
Table 9 shows a list of clinical studies evaluating RBAC’s effects on circulating cytokines in healthy adults.
Ali et al. [
69] investigated the effects of Biobran MGN-3 (1 and 3 g/day for 60 days) on plasma cytokines and growth factors in healthy individuals (
n = 20). A total of 12 cytokines and growth factors (IL-2, IL-4, IL-6, IL-8, IL-10, IL-1
, IL-1
, IFN-
, TNF-
, monocyte chemoattractant protein-1, VEGF, and epidermal growth factor [EGF]) were tested at baseline, 1 week, 30 days, and 60 days. Except for IL-4 and IL-8, the study found significant effects (
p < 0.05) for time but not between groups (1 g/day vs. 3 g/day) and group-by-time interaction. IFN-
, TNF-
, IL-1
, IL-1
, IL-8, and IL-10, and EGF peaked at 30 days and returned to the baseline level at 60 days. Hence, this study showed that the effects of Biobran MGN-3 in healthy adults were not purely proinflammatory nor anti-inflammatory but immunomodulatory over time [
69].
Choi et al. [
26] studied the serum cytokine level of healthy volunteers (
n = 80) randomly assigned to take either RBEP or placebo (3 g/day) for 8 weeks. IFN-
, TNF-
, IL-2, IL-4, IL-10, and IL-12 were tested at baseline, week 4 and week 8. The study showed that RBEP significantly increased serum IFN-
compared to placebo at week 8 (35.56 ± 17.66 vs. 27.04 ± 12.51 pg/mL,
p < 0.05), whereas no significant differences were detected for other cytokines. Moreover, there was no significant difference in reported adverse events among the participants in both RBEP and placebo groups. IFN-
responses are implicated in the host defence against intracellular pathogens, immune modulation, inflammatory processes associated with tissue damage and tumour immunosurveillance [
103]. The increased serum IFN-
, a proinflammatory cytokine, could indicate enhanced immunity against infections in healthy participants as IFN-
is produced predominantly by NKC during the innate immune response and by CD4+ and CD8+ T lymphocytes once adaptive immunity develops [
104]. Hence, IFN-
production was suggested as a supportive marker of NKC activity in certain malignancies [
105]. In this case, the increased IFN-
could indirectly indicate elevated NKC activity in the healthy participants in the short term, even though the same study did not detect a significant difference in NKC cytotoxic activity between RBAC and placebo group [
26].
Nevertheless, combining available evidence from both human studies [
26,
69], healthy individuals may likely experience fluctuations in circulating cytokine levels after taking RBAC due to increased immune activity. The effect may be short-term and may eventually stabilise. However, the immune cells have been primed with RBAC and thus could mount a more potent immune response should there be an actual infection based on the premise of trained immunity, a function of the innate defence mechanism [
106].
3.11. Effects among Older Adults
With RBAC’s immunomodulatory capacity, how RBAC could impact immunosenescence is another area of interest. The effects of Biobran MGN-3 on geriatric participants with reduced immune functions were investigated in three studies shown in
Table 10.
The field of psychoneuroimmunology strongly suggests a reciprocal and complex interaction between mind and body with perceived health-related quality of life (QoL) as a result of and directly affects cognitive functioning, the immune system and disease process [
107]. Hence, improving the immunity in aging adults was hypothesised to enhance the QoL. Elsaid et al. [
108] evaluated the effects of Biobran MGN-3 on the QoL of generally healthy older adults (≥56 years old). The study recruited 60 participants (40 males and 20 females) and randomly assigned them to either Biobran MGN-3 (250 mg/day) or placebo groups with a supplementation duration of 3 months. QoL was assessed with the validated SF-12v2 health questionnaire before and after the intervention. The study found that Biobran MGN-3 significantly improved the QoL of older adults after 3 months compared to baseline, whereas no significant differences in QoL scores were detected in the placebo group. Between-group comparisons showed that Biobran MGN-3 had significantly improved both the physical (
p < 0.037) and mental (
p < 0.012) component summary scores and the specific component scores of role physical (
p < 0.002), bodily pain (
p < 0.006), vitality (
p < 0.000), and social functioning (
p < 0.001). Hence, Elsaid et al. [
108] concluded that a low dose of Biobran MGN-3 (250 mg/day) over 3 months could improve the QoL of older adults.
Table 10.
The immunomodulating effects of RBAC on older adults with evidence from human clinical trials.
Table 10.
The immunomodulating effects of RBAC on older adults with evidence from human clinical trials.
| No | RBAC (dose) | Participants | Key Findings | Author (Year) |
|---|
| 1. | Biobran MGN-3 (500 mg/day for 2 weeks with cross-over) | Older adults (age 75–90, n = 36) | The mean total symptom score for CCS during the control period (0.1535 ± 0.38591) was significantly higher than that of the Biobran MGN-3 treatment period (0.0491 ± 0.09083, p = 0.0426). Biobran MGN-3 significantly prevented the decrease in T lymphocyte blastogenesis based on PHA mitogen test compared to the control. | Tazawa et al. (2003) [71] |
| 2. | Biobran MGN-3 (250 mg/day for 3 months) | Older adults (age ≥ 56, n = 60, 30 in each group) | Biobran MGN-3 significantly improved the QoL of older adults compared to baseline. Compared to placebo, Biobran MGN-3 significantly improved both the physical (p < 0.037) and mental (p < 0.012) component summary scores and the specific component scores of role physical (p < 0.002), bodily pain (p < 0.006), vitality (p < 0.000), and social functioning (p < 0.001). | Elsaid et al. (2020) [108] |
| 3. | Biobran MGN-3 (500 mg/day for 3 months) | Older adults (age ≥ 56, n = 80, 40 in each group) | The Biobran MGN-3 group had significantly lower ILI incidents than the placebo group (5% vs. 22.5%, p = 0.048). The risk of ILI infection in the Biobran MGN-3 group was estimated to be 18.2% (95% CI: 3.9–48.9), which was significantly lower (p < 0.05) than the 55.1% (95% CI: 43.4–66.3) for the placebo group. | Elsaid et al. (2021) [74] |
Another RCT by Elsaid et al. [
74] focused on the effects of Biobran MGN-3 on strengthening the innate immunity of older adults by assessing the incidents of influenza-like illnesses (ILI). Immunosenescence is known to amplify susceptibility to viral infections, such as influenza, even with vaccination. Older people also have a greater risk of more severe ILI, leading to severe respiratory symptoms, systemic complications, hospitalization, and death [
109,
110]. Hence, Biobran MGN-3, as an antioxidant and immunomodulator, could reduce the risk of contracting ILI while lowering the symptom severity in those infected.
Eighty generally healthy older adults (≥56 years old, male-to-female ratio: 1 to 1) enrolled in the study by Elsaid et al. [
74]. Participants were randomly allocated to take either 500 mg/day of Biobran MGN-3 or placebo powder for 3 months, with the incidence of ILI infection monitored during the trial. The study found significantly lower ILI incidents (
p = 0.048) in the Biobran MGN-3 group (5%, 2/40) compared to the placebo group (22.5%, 9/40). The risk of ILI infection in the Biobran MGN-3 group was estimated to be 18.2% (95% confidence interval [CI]: 3.9–48.9), which was significantly lower (
p < 0.05) than the 55.1% (95% CI: 43.4-66.3) for the placebo group. Hence, the data suggest that Biobran MGN-3 could have prophylactic effects against ILI among older adults [
74].
Similarly, an earlier study by Tazawa et al. [
71] among older adults (age 70–95) under institutional elderly care also confirmed the effects of Biobran MGN-3 in reducing the severity of common cold symptoms (CCS). In this cross-over study, participants were taking 500 mg/day Biobran MGN-3 (treatment) and a water-soluble unmodified rice bran powder of similar taste (control) for 2 weeks each in random order with 2-week washout in between (total 6 weeks). During the 42 days trial period, participants were monitored for CCS (fever, headache, malaise, chills, cough, sputum, running/stuffy nose, sore throat and chest pain) by care personnel at the facility who were blinded to their intervention allocation. Each symptom was scored based on a severity grade of 0 (no symptom) to 3 (severe) [
71].
Results from participants (
n = 36, mean age 84 ± 7.2) who completed the trial of Tazawa et al. [
71] showed that there was no significant difference in the number of participants for whom CCS was observed (
n = 15 each). The mean duration of CCS during the control period was double that of the treatment period (2.6 ± 5.94 vs. 1.2 ± 2.20 days) [
71]. However, the difference did not reach statistical significance (
p = 0.2721). Nonetheless, the mean total symptom score for CCS during the control period (0.1535 ± 0.38591) was significantly higher than that of the treatment period (0.0491 ± 0.09083,
p = 0.0426). Furthermore, a decrease in T lymphocyte blastogenesis by the phytohemagglutinin mitogens test was detected in both periods. However, the reduction during the Biobran MGN3 treatment period was significantly lower than in the control period (
p < 0.05) [
71]. As such, the results supported the hypothesis that Biobran MGN-3 could reduce the severity of CCS among older people by improving immunity.