Effects and Stress-Relieving Mechanisms of Dark Tea Polysaccharide in Human HaCaT Keratinocytes and SZ95 Sebocytes
Abstract
:1. Introduction
2. Results
2.1. Analysis of the DTP Molecular Weight Composition
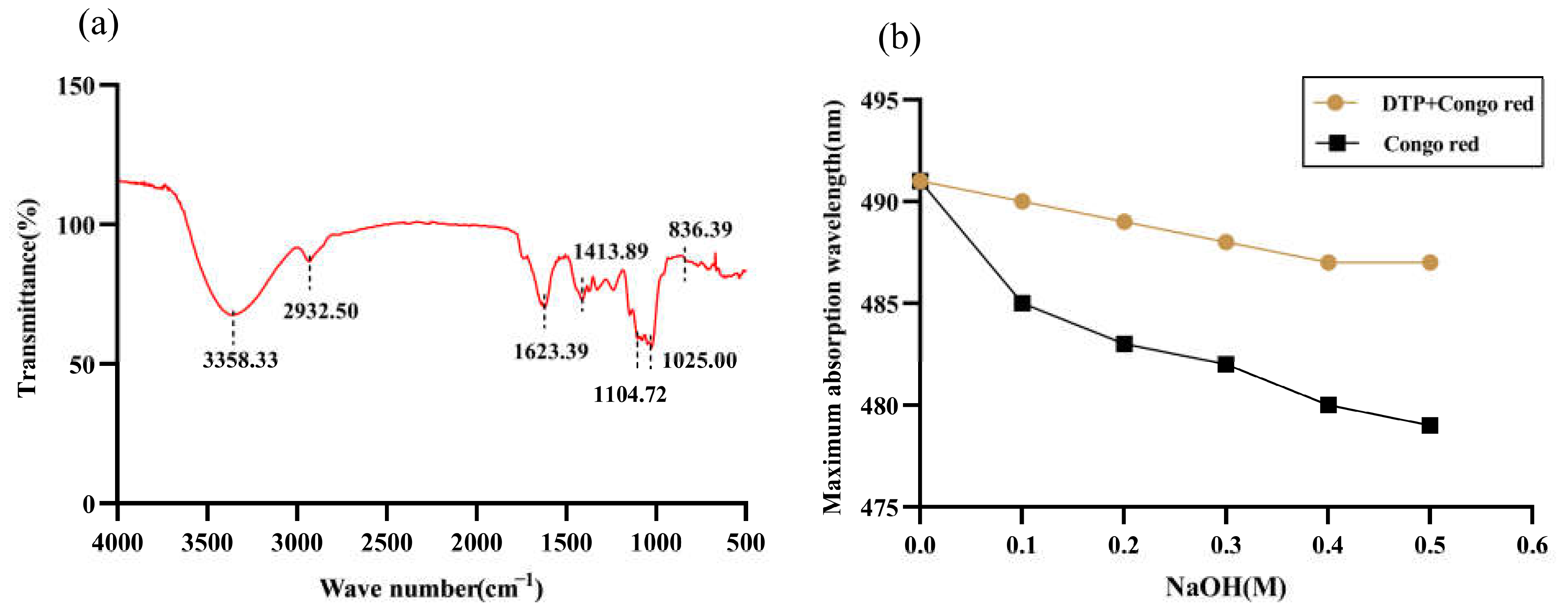
2.2. Analysis of the DTP Chemical Composition
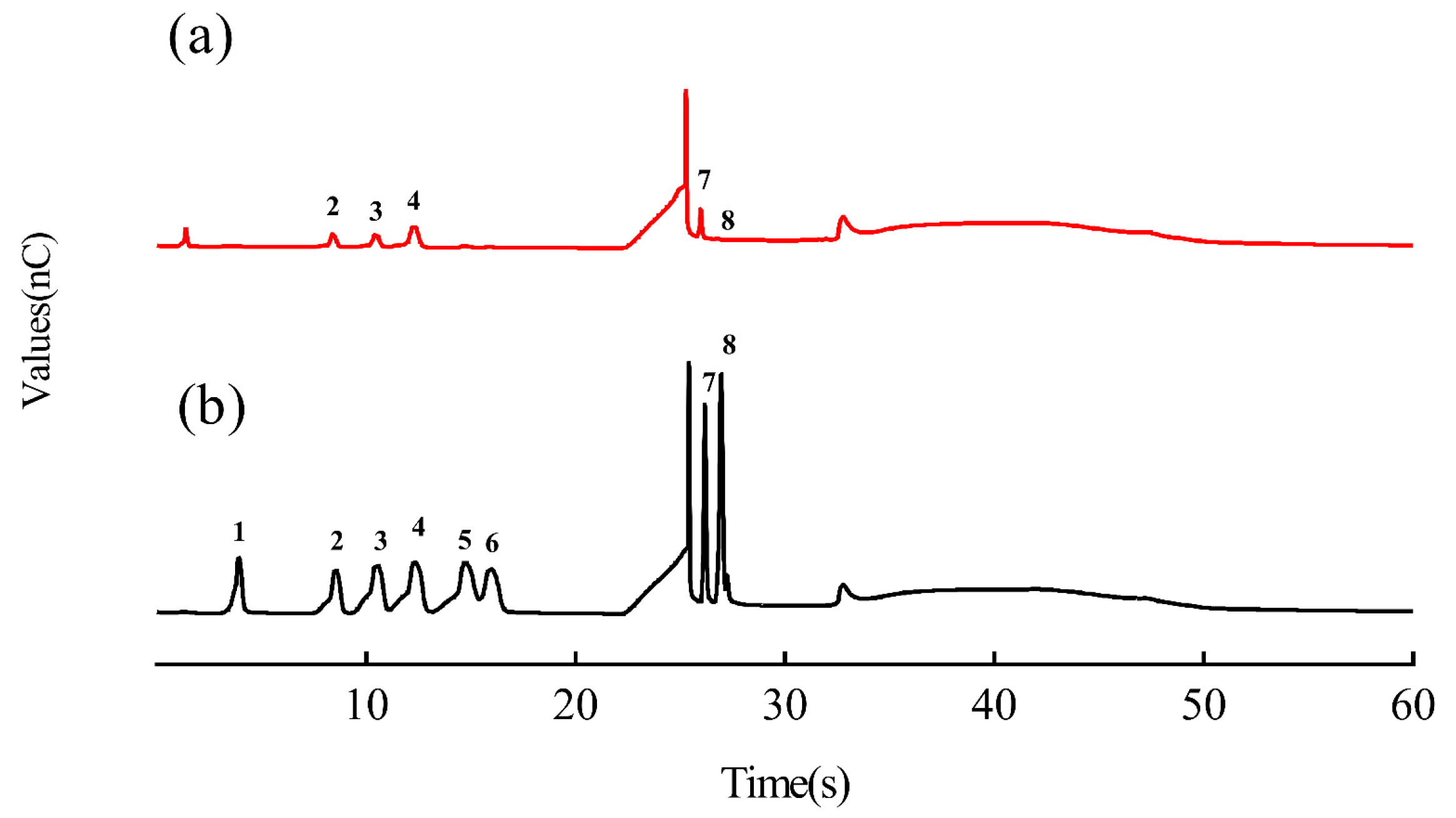
2.3. Analysis of the DTP Monosaccharide Composition
2.4. Analysis of DTP Structural Features
2.5. Effect of DTP on Viability of HaCaT Keratinocyte Cells
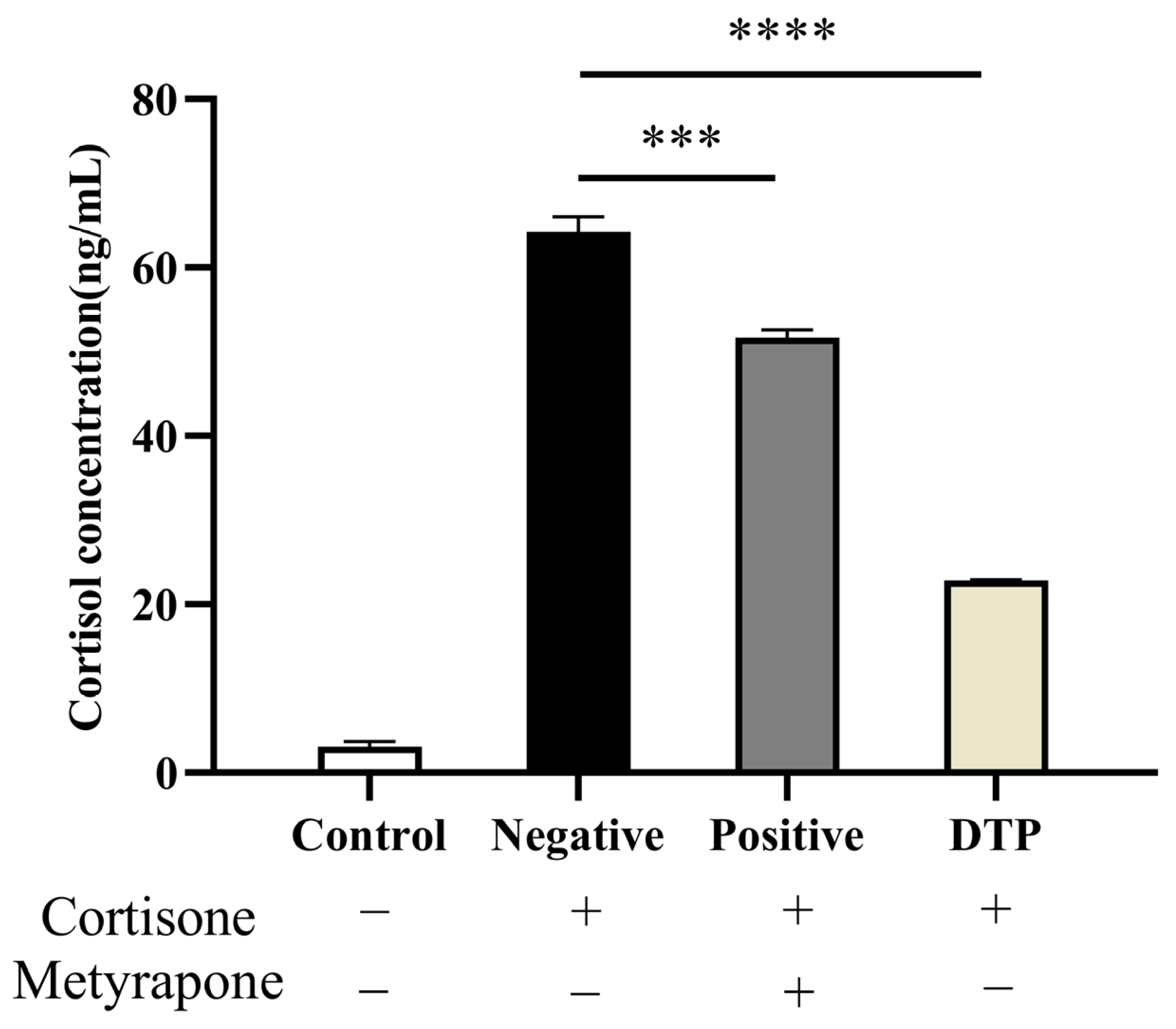
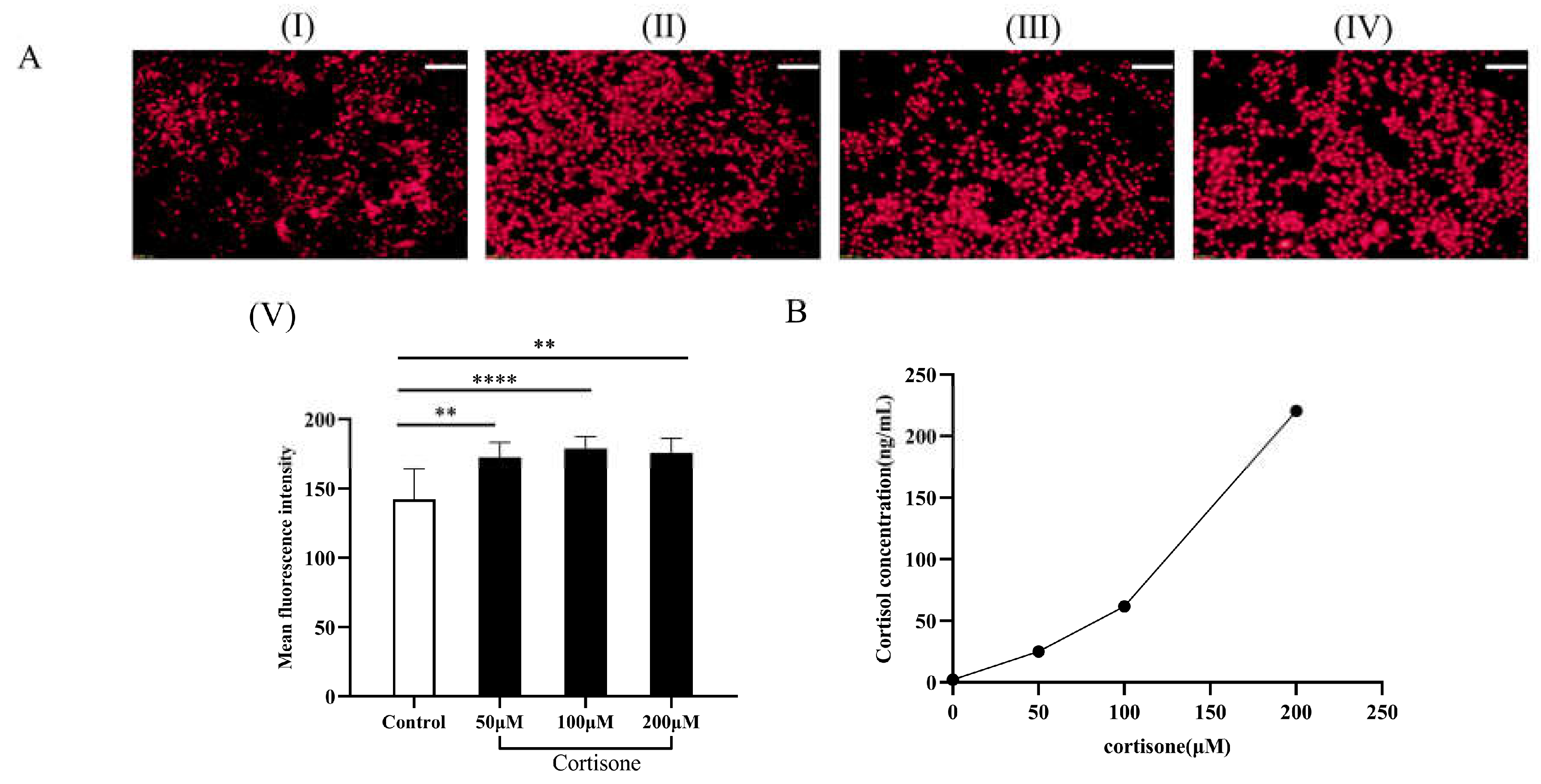
2.6. Cortisol Production in HaCaT Keratinocytes after DTP Treatment
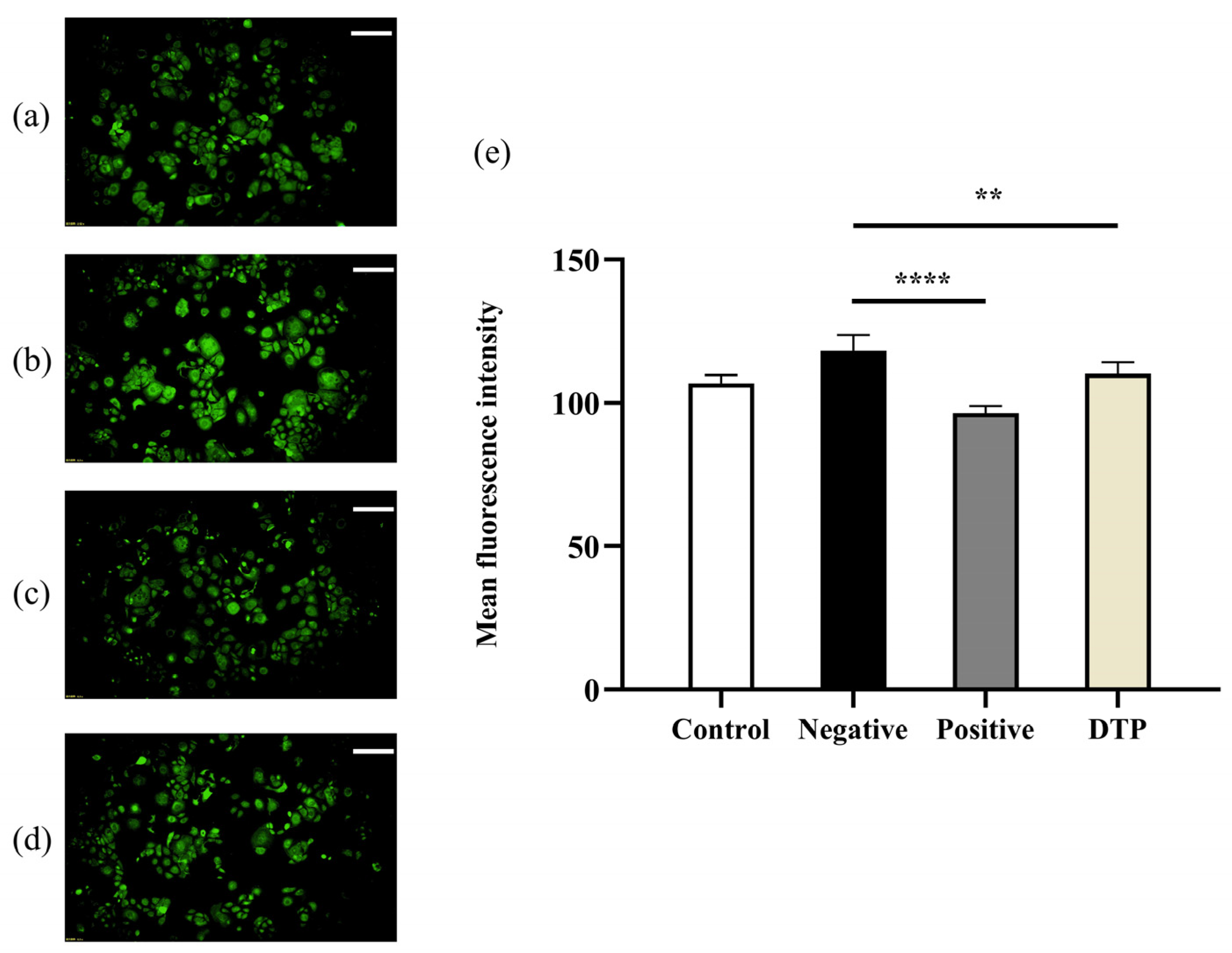
2.7. Effect of DTP on HSD11B1 Enzyme Expression
2.8. Effect of DTP on Viability of SZ95 Sebocytes
2.9. Cortisol Production and Lipid Secretion in SZ95 Sebocytes after Cortisone Treatment
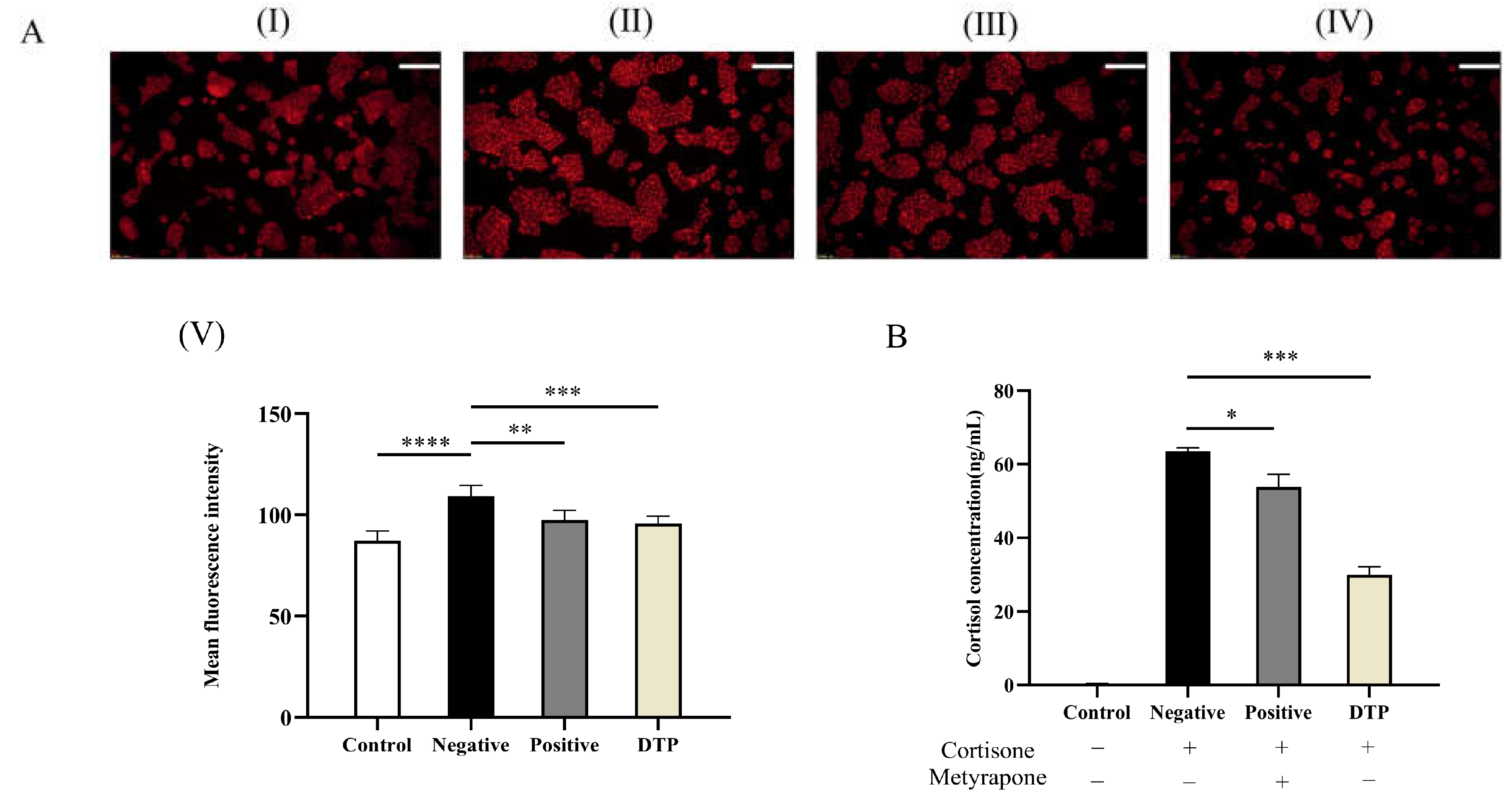
2.10. Effect of DTP on Lipid Secretion Induced by Cortisone
3. Discussion
4. Materials and Methods
4.1. DTP Preparation
4.2. Analysis of the Molecular Weight Composition in DTP Using Ultrafiltration
4.3. Analysis of the Chemical Composition in DTP Using UV-VIS
4.4. Analysis of the Monosaccharide Composition in DTP Using HPAEC-PAD
4.5. Infrared Spectral Analysis of DTP
4.6. Congo Red Experiment of DTP
4.7. Cell Culture
4.8. MTT Assay
4.9. ELISA
4.10. Experiment of Cortisol Inhibition in HaCaT Keratinocytes
4.11. Experiment on the Change of HSD11B1 Enzyme Expression in HaCaT Keratinocytes
4.12. Experiment on Cortisol Conversion/Lipid Secretion Assay Induced by Cortisone in Sebaceous Gland Cells
4.13. Experiment on Lipid Secretion Induced by Cortisone with DTP in Sebaceous Gland Cells
4.14. Image J Fluorescence Intensity Analysis
4.15. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Papadimitriou, A.; Priftis, K.N. Regulation of the hypothalamic-pituitary-adrenal axis. Neuroimmunomodulation 2009, 16, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Elenkov, I.J.; Chrousos, G.P. Stress system—Organization, physiology and immunoregulation. Neuroimmunomodulation 2006, 13, 257–267. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Zhang, T.; Xu, X.; Wan, S.; Wang, Y. Review of Skin Problems Caused by Stress. J. Clin. Med. Res. 2022, 3, 116–122. [Google Scholar] [CrossRef]
- Chen, Y.; Lyga, J. Brain-skin connection: Stress, inflammation and skin aging. Inflamm. Allergy Drug Targets 2014, 13, 177–190. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Hao, F. Research progression of independent HPA system in skin. Chin. J. Dermatovenereol. 2009, 23, 841–844. [Google Scholar]
- Youm, J.K.; Park, K.; Uchida, Y.; Chan, A.; Mauro, T.M.; Holleran, W.M.; Elias, P.M. Local blockade of glucocorticoid activation reverses stress- and glucocorticoid-induced delays in cutaneous wound healing. Wound Repair Regen. 2013, 21, 715–722. [Google Scholar] [CrossRef]
- Guichard, A.; Humbert, P.; Tissot, M.; Muret, P.; Courderot-Masuyer, C.; Viennet, C. Effects of topical corticosteroids on cell proliferation, cell cycle progression and apoptosis: In vitro comparison on HaCaT. Int. J. Pharm. 2015, 479, 422–429. [Google Scholar] [CrossRef]
- Terao, M.; Katayama, I. Local cortisol/corticosterone activation in skin physiology and pathology. J. Dermatol. Sci. 2016, 84, 11–16. [Google Scholar] [CrossRef]
- Evers, A.W.; Van, P.C.; Brouwer, S.D.; Jong, E.D.; Schalkwijk, J.; Verhoeven, E.W.; Kraaimaat, F.W.; Sweep, F.C. How stress gets under the skin: Cortisol and stress reactivity in psoriasis. Br. J. Dermatol. 2010, 163, 986–991. [Google Scholar] [CrossRef]
- Qin, L.; Shuo, C.; Yongdi, L.; Jianan, H.; Yu, L.; Lizheng, X.; Zhonghua, L. Biochemical Components Associated with Microbial Community Shift During the Pile-Fermentation of Primary Dark Tea. Front. Microbiol. 2018, 9, 1509. [Google Scholar]
- Liang, Z.; Zhang, Z.Z.; Zhou, Y.B.; Ling, T.J.; Wan, X.C. Chinese dark teas: Postfermentation, chemistry and biological activities. Food Res. Int. 2013, 53, 600–607. [Google Scholar]
- Zhou, P.; Liu, K.; Xiao, G.; You, P.; Wen, R. Adsorption-Desorption of Dark Tea Polysaccharides by Resin and Its Antioxidant Activity. J. Hunan City Univ. Nat. Sci. 2018, 26, 4162. [Google Scholar]
- Xu, J.; Wang, W.; Liang, X.; Li, P.; Du, X. Inhibitory Effect of the Theabrownin and Tea Polysaccharide Extracts of Dark Tea on Lipase. J. Phys. Conf. Ser. 2020, 1549, 032048. [Google Scholar] [CrossRef]
- Yang, X.; Huang, M.; Ma, W. Composition and antioxidant activity of polysaccharides of different dark tea. Sci. Technol. Food Ind. 2017, 38, 16–20. [Google Scholar]
- Zhu, J.; Zhou, H.; Zhang, J.; Li, F.; Wei, K.; Wei, X.; Wang, Y. Valorization of Polysaccharides Obtained from Dark Tea: Preparation, Physicochemical, Antioxidant, and Hypoglycemic Properties. Foods 2021, 205, 2276. [Google Scholar] [CrossRef] [PubMed]
- Xiang, G.; Sun, H.; Chen, Y.; Guo, H.; Liu, Y.; Li, Y.; Lu, C.; Wang, X. Antioxidant and hypoglycemic activity of tea polysaccharides with different degrees of fermentation. Int. J. Biol. Macromol. 2023, 228, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Song, L.B.; Huang, J.A.; Liu, Z.H.; Huang, H.; Wang, K.B. Study on the Activity of Dark Tea to Gastrointestinal Tumor. J. Tea Sci. 2009, 29, 191–195. [Google Scholar]
- Liu, M.; Gong, Z.; Liu, H.; Wang, J.; Wang, D.; Yang, Y.; Zhong, S. Structural characterization and anti-tumor activity in vitro of a water-soluble polysaccharide from dark brick tea. Int. J. Biol. Macromol. 2022, 205, 615–625. [Google Scholar] [CrossRef]
- Gong, Z.-P.; Ouyang, J.; Wu, X.-L.; Zhou, F.; Lu, D.-M.; Zhao, C.-J.; Liu, C.-F.; Zhu, W.; Zhang, J.-C.; Li, N.-X. Dark tea extracts: Chemical constituents and modulatory effect on gastrointestinal function. Biomed. Pharmacother. 2020, 130, 110514. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.Y.; Huang, Y.W.; Wang, X.J.; Shao, W.F. Research on Anti-fatigue Effect of Fermented Pu-erh Tea. J. Tea Sci. 2010, 30, 218–222. [Google Scholar]
- Yamaguchi, M.; Nishimiya, H. Noninvasive Evaluation of the Chronic Influence of Local Air Velocity from an Air Conditioner Using Salivary Cortisol and Skin Caspase-14 as Biomarkers of Psychosomatic and Environmental Stress. J. Int. Med. Res. 2012, 40, 1429–1437. [Google Scholar] [CrossRef] [PubMed]
- Cirillo, N.; Prime, S.S. Keratinocytes synthesize and activate cortisol. J. Cell Biochem. 2011, 112, 1499–1505. [Google Scholar] [CrossRef] [PubMed]
- Vukelic, S.; Stojadinovic, O.; Pastar, I.; Rabach, M.; Krzyzanowska, A.; Lebrun, E.; Davis, S.C.; Resnik, S.; Brem, H.; Tomic-Canic, M. Cortisol synthesis in epidermis is induced by IL-1 and tissue injury. J. Biol. Chem. 2011, 286, 10265–10275. [Google Scholar] [CrossRef] [PubMed]
- Tiganescu, A.; Walker, E.A.; Hardy, R.S.; Mayes, A.E.; Stewart, P.M. Localization, age- and site-dependent expression, and regulation of 11beta-hydroxysteroid dehydrogenase type 1 in skin. J. Investig. Dermatol. 2011, 131, 30–36. [Google Scholar] [CrossRef]
- Dodd, S.; Skvarc, D.R.; Dean, O.M.; Anderson, A.; Kotowicz, M.; Berk, M. Effect of Glucocorticoid and 11β-Hydroxysteroid-Dehydrogenase Type 1 (11β-HSD1) in Neurological and Psychiatric Disorders. Int. J. Neuropsychopharmacol. 2022, 25, 387–398. [Google Scholar] [CrossRef]
- Terao, M.; Tani, M.; Itoi, S.; Yoshimura, T.; Hamasaki, T.; Murota, H.; Katayama, I. 11beta-hydroxysteroid dehydrogenase 1 specific inhibitor increased dermal collagen content and promotes fibroblast proliferation. PLoS ONE 2014, 9, e93051. [Google Scholar] [CrossRef]
- Shi, V.Y.; Leo, M.; Hassoun, L.; Chahal, D.S.; Maibach, H.I.; Sivamani, R.K. Role of sebaceous glands in inflammatory dermatoses. J. Am. Acad. Dermatol. 2015, 73, 856–863. [Google Scholar] [CrossRef]
- Zouboulis, C.C.; Seltmann, H.; Hiroi, N.; Chen, W.; Young, M.; Oeff, M.; Scherbaum, W.A.; Orfanos, C.E.; McCann, S.M.; Bornstein, S.R. Corticotropin releasing hormone: An autocrine hormone that promotes lipogenesis in human sebocytes. Proc. Natl. Acad. Sci. USA 2002, 99, 7148–7153. [Google Scholar] [CrossRef]
- Zbytek, B.; Mysliwski, A.; Slominski, A.; Wortsman, J.; Wei, E.T.; Mysliwska, J. Corticotropin-releasing hormone affects cytokine production in human HaCaT keratinocytes. Life Sci. 2002, 70, 1013–1021. [Google Scholar] [CrossRef]
- Lei, X.; Wang, L.; Tang, M.; Huang, X.; Chen, P.; Hou, A. 14 Effects of sample extract methods on the production and antioxidant activity of Lepista sordida polysaccharides. J. Investig. Med. 2016, 64, A5. [Google Scholar]
- Qu, C.X.; Shen, S.D.; Wang, X.F.; Cui, Y.H.; Song, W.P. Method research of measuring soluble protein contents of plant rough extraction using Coomassie Brilliant Blue. J. Suzhou Univ. Nat. Sci. Ed. 2006, 22, 82–85. [Google Scholar]
- Qin, A.X.; Ji, S.J.; Gao, Z.; He, B.B.; Kang, X.H.; Jia, Y.N.; Chen, C.; Tong, C. Determination of oil-soluble tea polyphenols in oil by solid phase extraction and Folin-Ciocalteu colorimetry. J. Food Saf. Qual. 2014, 5, 1185–1190. [Google Scholar]
- Percival, E.; Ross, A.G. A colorimetric method for the Estimation of Alginic acid in seaweed Specimens. J. Chem. Technol. Biotechnol. 2010, 67, 420–421. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, R.; Li, Y.; Li, X.; Hileuskaya, K. Degradation of polysaccharides from Sargassum fusiforme using UV/H2O2 and its effects on structural characteristics. Carbohydr. Polym. 2019, 230, 115647. [Google Scholar] [CrossRef] [PubMed]
- Zouboulis, C.C.; Seltmann, H.; Neitzel, H.; Orfanos, C.E. Establishment and characterization of an immortalized human sebaceous gland cell line (SZ95). J. Investig. Dermatol. 1999, 113, 1011–1020. [Google Scholar] [CrossRef]


| Molecular Weight | Mass Ratio |
|---|---|
| <10 KD | 36.13% |
| 10–30 KD | 7.92% |
| 30–100 KD | 5.94% |
| >100 KD | 50.01% |
| No.1 | Components | Regression Equation | Composition (mg/g) |
|---|---|---|---|
| 1 | Total sugar | Y = 0.0036X + 0.0014 (R2 = 0.9991) | 509.13 |
| 2 | Total protein | Y = 0.0037X + 0.0384 (R2 = 0.9952) | 57.17 |
| 3 | Total polyphenols | Y = 0.0050X + 0.0102 (R2 = 0.9985) | 49.84 |
| 4 | Uronic acid | Y = 0.0104X +0.2597 (R2 = 0.9926) | 101.42 |
| No.1 | Components | Regression Equation | Range (μg/mL) | RT (min) |
|---|---|---|---|---|
| 1 | Fucose | Y = 1.3322X + 3.9023 (R2 = 0.9992) | 2.5–12.5 | 3.9 |
| 2 | Arabinose | Y = 1.5408X + 3.0415 (R2 = 0.9995) | 2.5–12.5 | 8.4 |
| 3 | Galactose | Y = 2.3057X + 5.0154 (R2 = 0.9998) | 2.5–12.5 | 10.5 |
| 4 | Glucose | Y = 2.8522X + 5.3423 (R2 = 0.9991) | 2.5–12.5 | 12.3 |
| 5 | Xylose | Y = 3.3568X + 4.6115 (R2 = 0.9998) | 2.5–12.5 | 14.9 |
| 6 | Mannose | Y = 2.3615X + 3.3531 (R2 = 0.9991) | 2.5–12.5 | 16.2 |
| 7 | Galacturonic acid | Y = 2.1277X − 1.7009 (R2 = 0.9998) | 1.25–12.5 | 25.9 |
| 8 | Glucuronic acid | Y = 2.9729X − 0.4388 (R2 = 0.9995) | 0–12.5 | 26.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, C.; Fu, J.; Cui, J.; Zhang, T.; Zouboulis, C.C.; Wang, J.; Yan, S. Effects and Stress-Relieving Mechanisms of Dark Tea Polysaccharide in Human HaCaT Keratinocytes and SZ95 Sebocytes. Molecules 2023, 28, 6128. https://doi.org/10.3390/molecules28166128
Gao C, Fu J, Cui J, Zhang T, Zouboulis CC, Wang J, Yan S. Effects and Stress-Relieving Mechanisms of Dark Tea Polysaccharide in Human HaCaT Keratinocytes and SZ95 Sebocytes. Molecules. 2023; 28(16):6128. https://doi.org/10.3390/molecules28166128
Chicago/Turabian StyleGao, Chang, Jiafeng Fu, Junyi Cui, Tingzhi Zhang, Christos C. Zouboulis, Jing Wang, and Shaowei Yan. 2023. "Effects and Stress-Relieving Mechanisms of Dark Tea Polysaccharide in Human HaCaT Keratinocytes and SZ95 Sebocytes" Molecules 28, no. 16: 6128. https://doi.org/10.3390/molecules28166128
APA StyleGao, C., Fu, J., Cui, J., Zhang, T., Zouboulis, C. C., Wang, J., & Yan, S. (2023). Effects and Stress-Relieving Mechanisms of Dark Tea Polysaccharide in Human HaCaT Keratinocytes and SZ95 Sebocytes. Molecules, 28(16), 6128. https://doi.org/10.3390/molecules28166128









