Effect of the Synthetic Parameters over ZnO in the CO2 Photoreduction
Abstract
1. Introduction
2. Result and Discussion
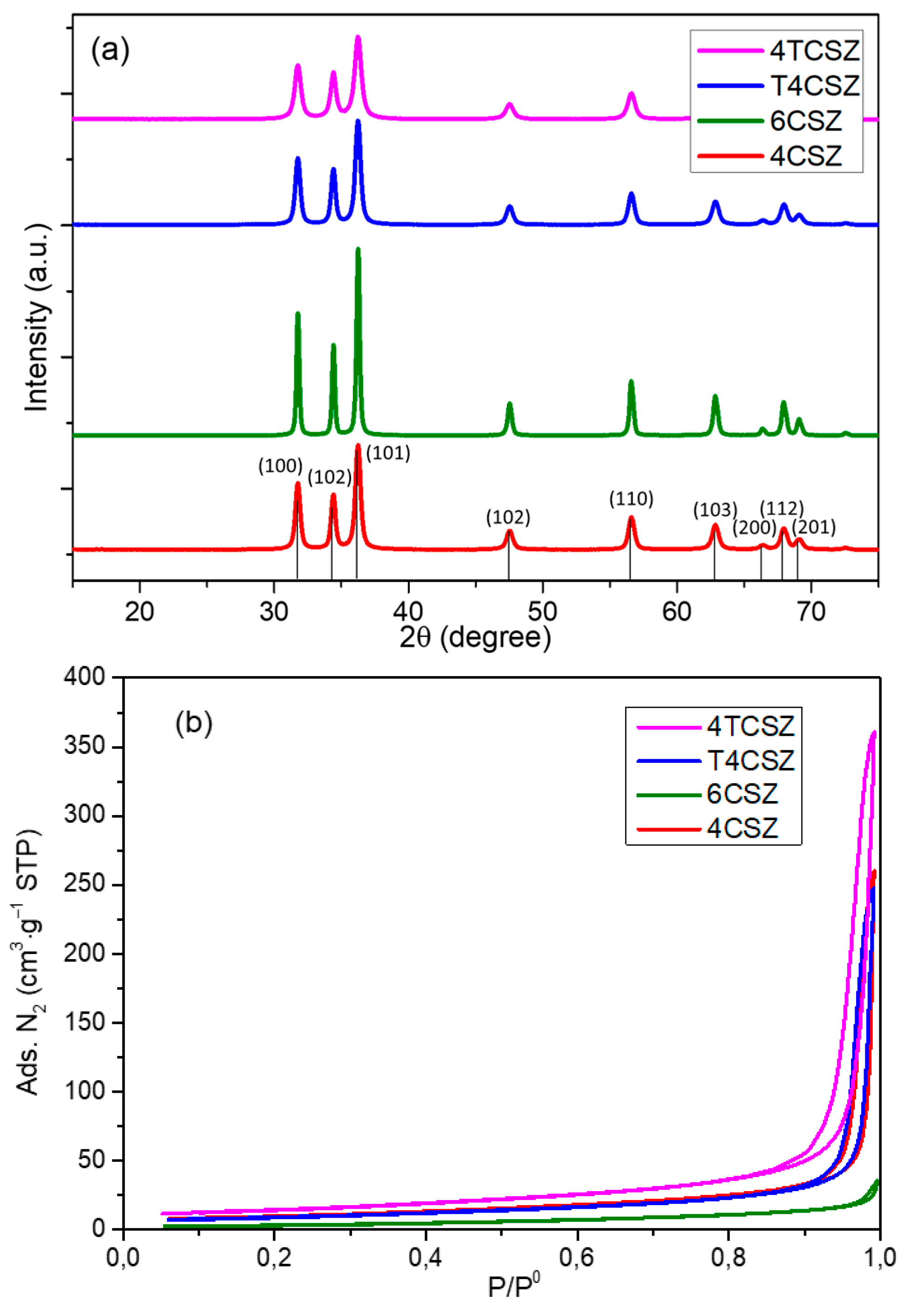
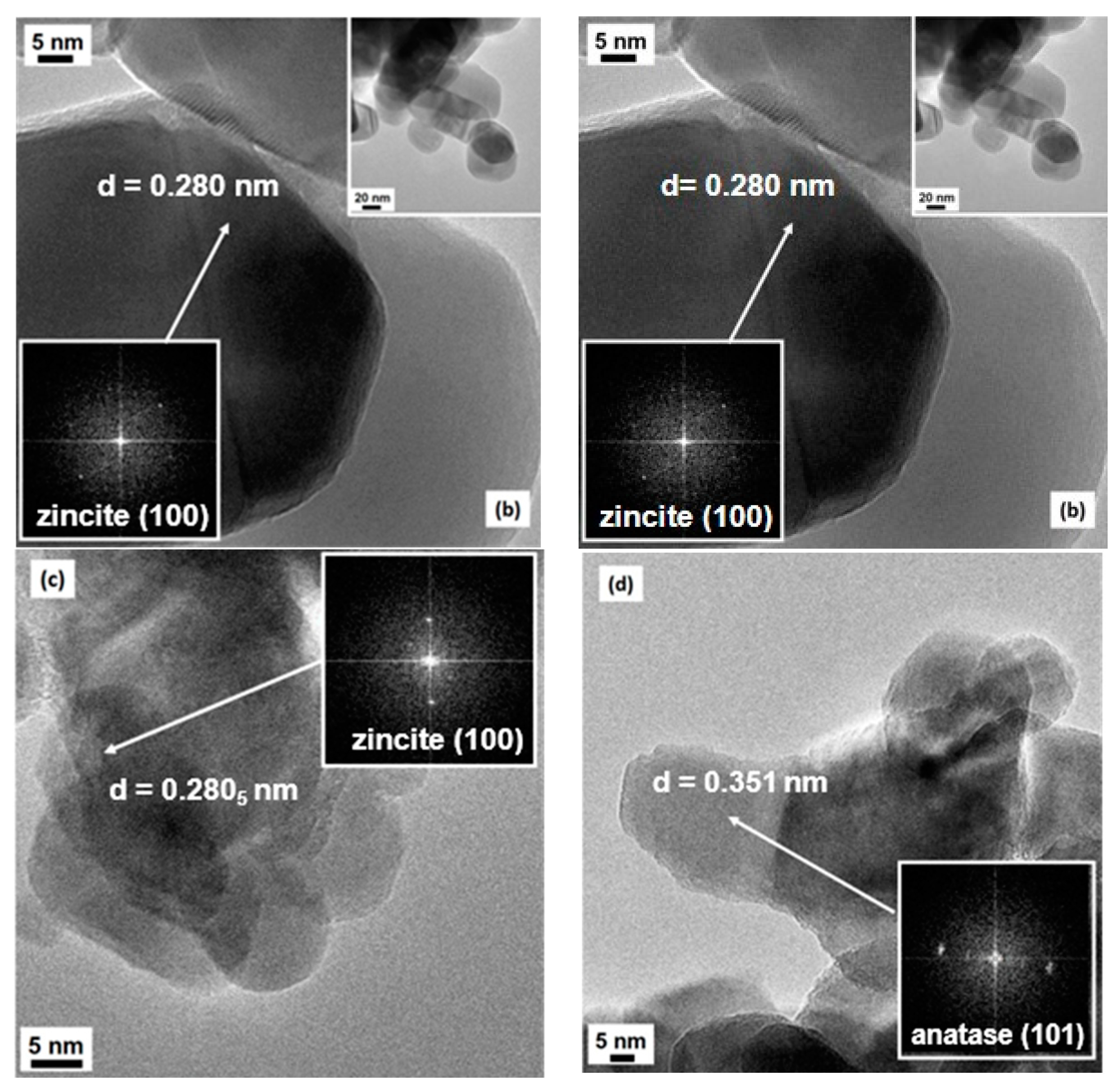
2.1. Structural and Morphological Characterization
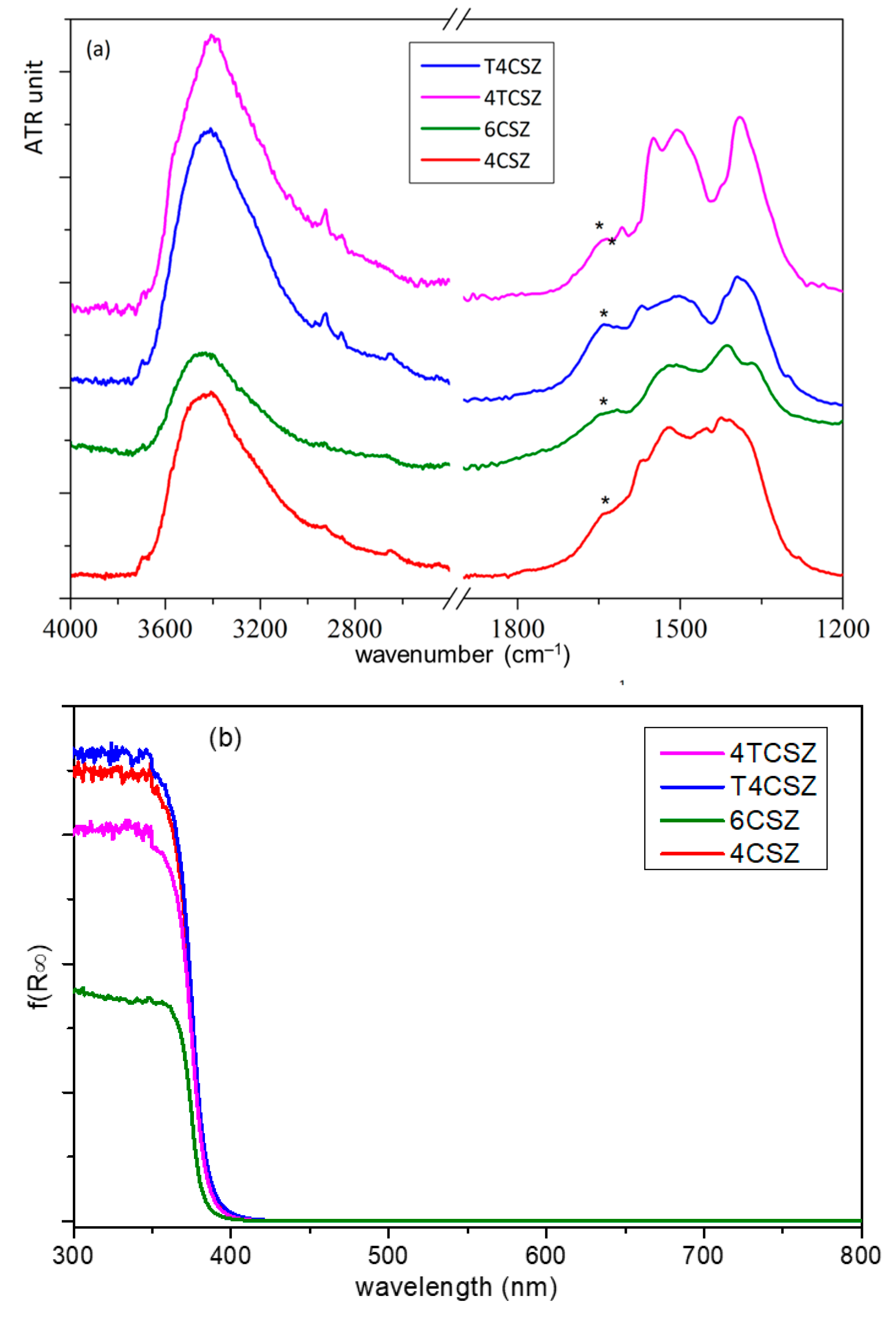
2.2. Surface and Optical Characterization
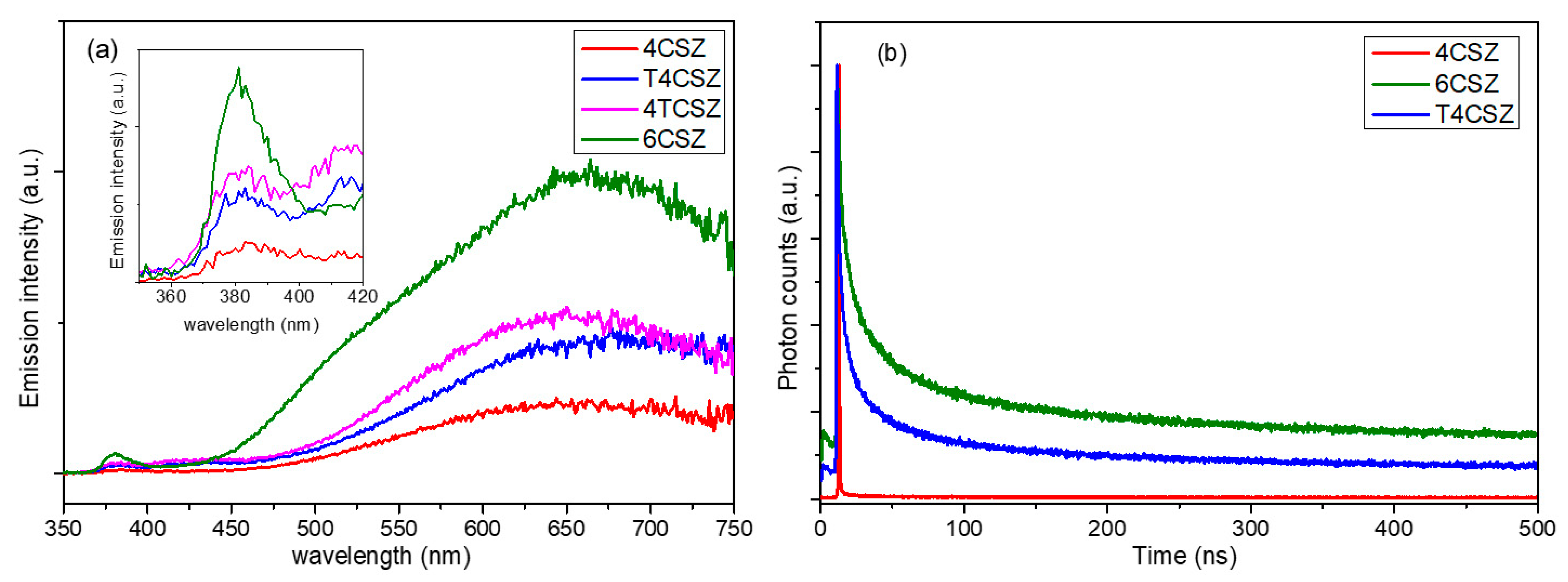
2.3. Photoluminescence Characterization
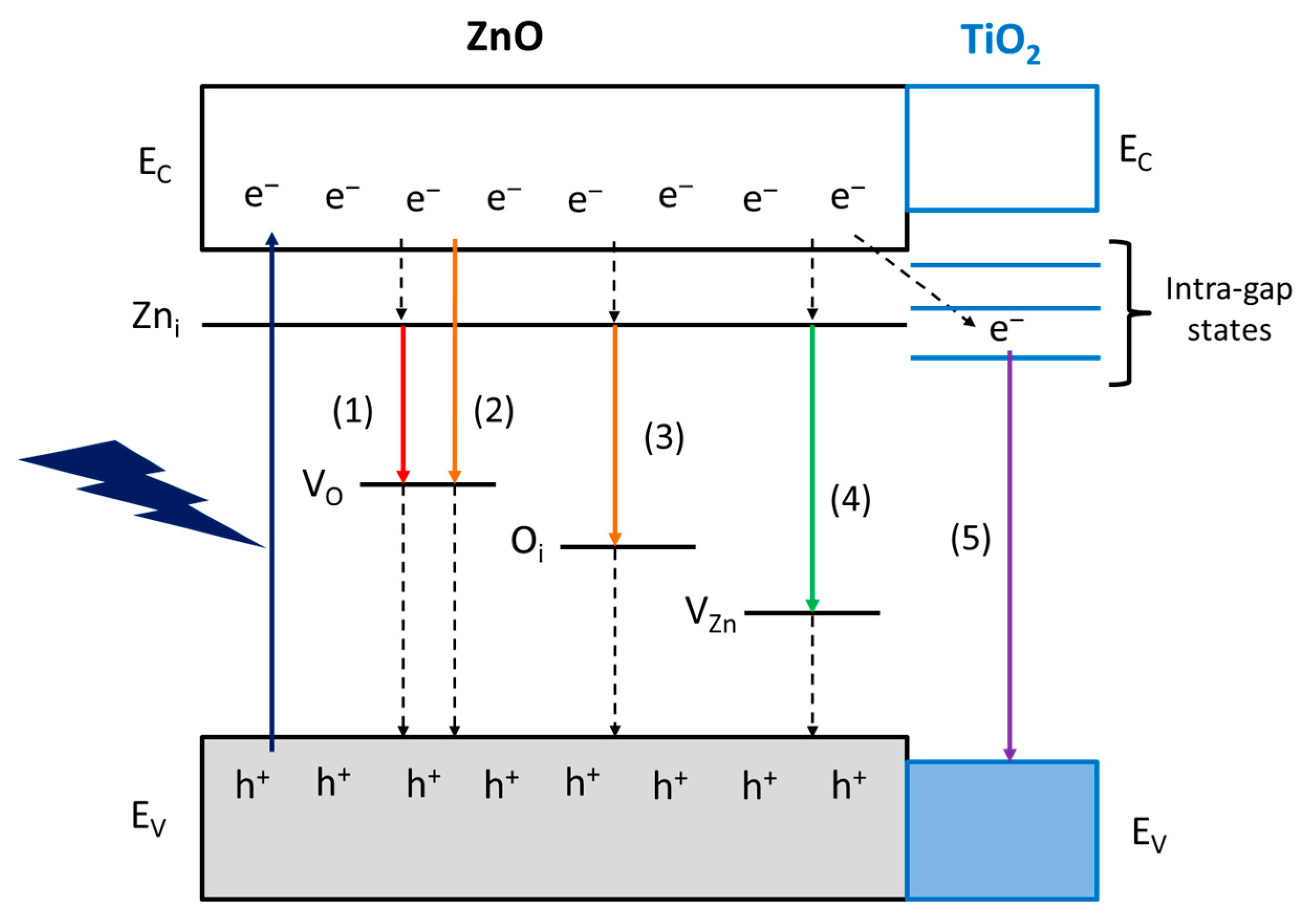
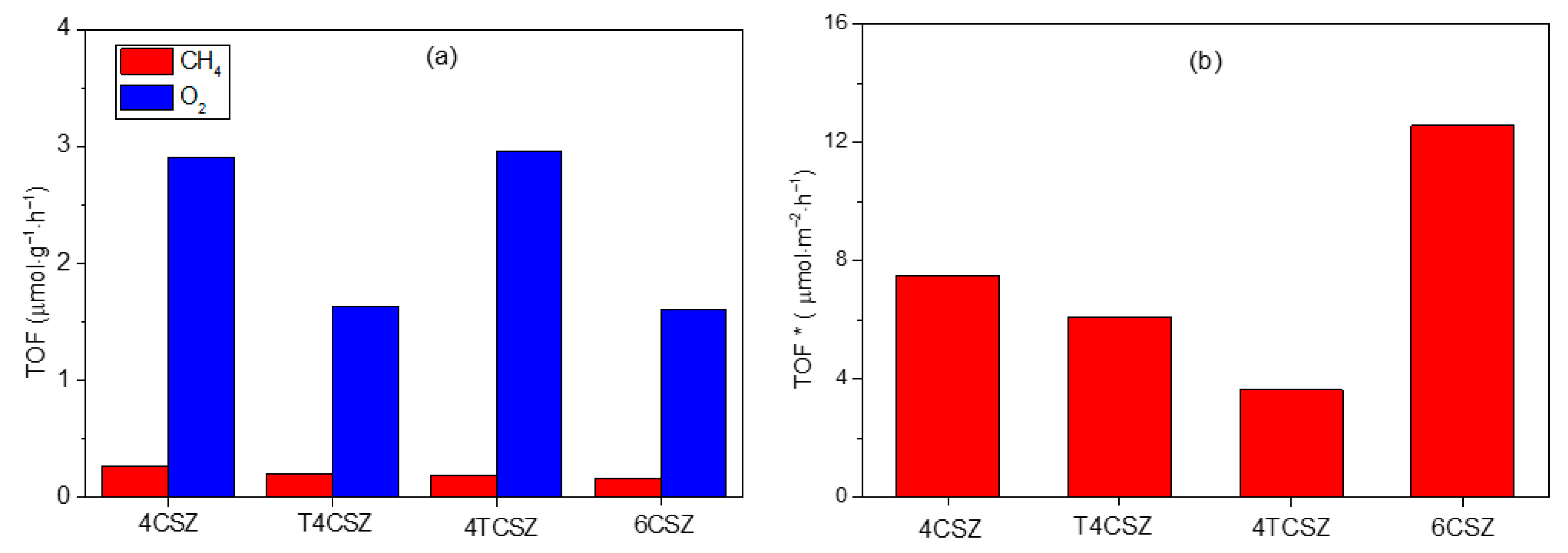
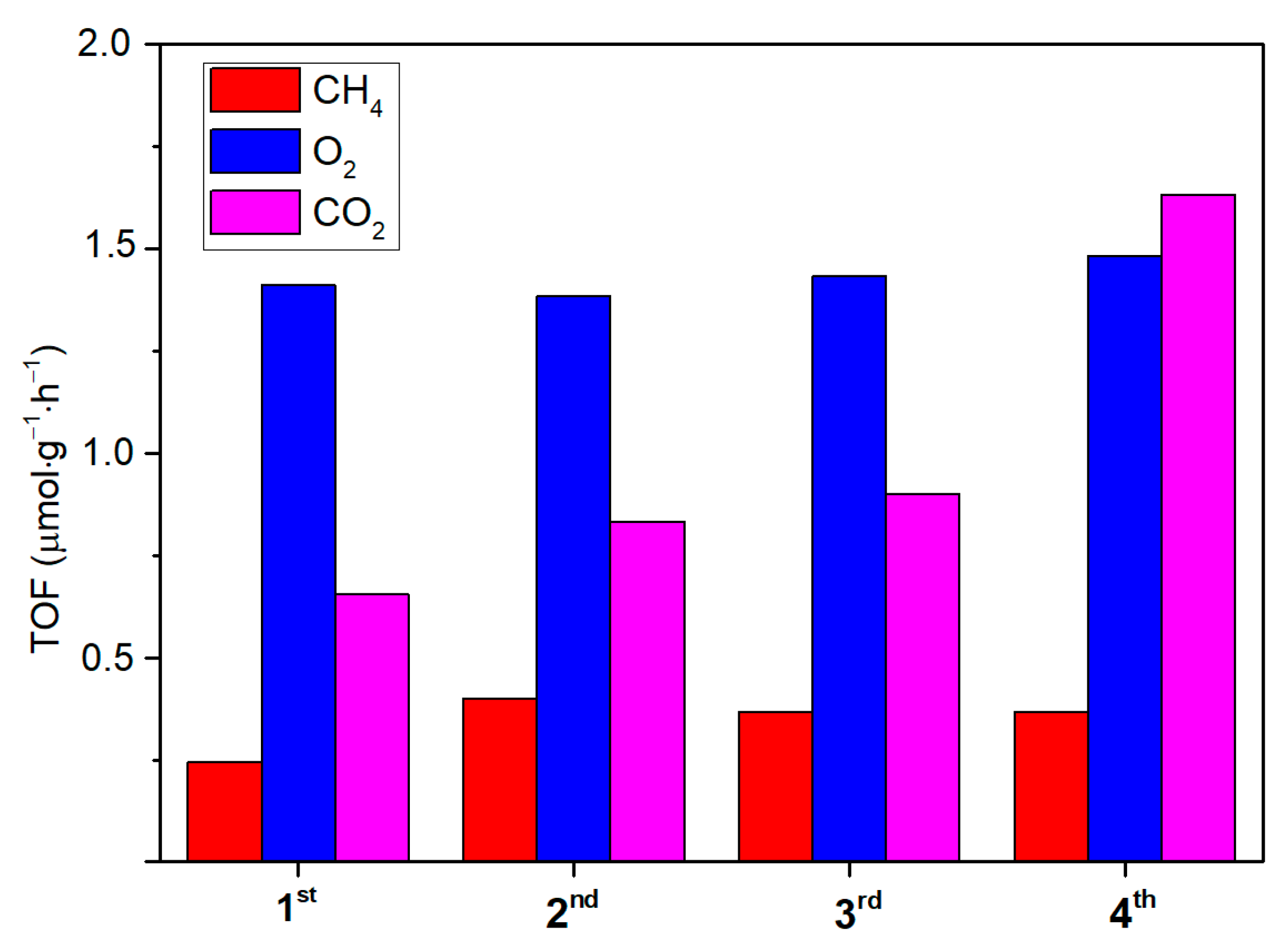
2.4. CO2 Photoreduction with H2O and Hybrid CO2 Capture–Photoconversion
3. Materials and Methods
3.1. Materials
3.2. ZnO-Based Materials Synthesis
3.3. Characterization
3.4. Photocatalytic Tests
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Janotti, A.; Van de Walle, C.G. Fundamentals of zinc oxide as a semiconductor. Rep. Prog. Phys. 2009, 72, 126501. [Google Scholar] [CrossRef]
- Ozgür, Ü.; Alivov, Y.I.; Liu, C.; Teke, A.; Reshchikov, M.A.; Doğan, S.; Avrutin, V.; Cho, S.-J.; Morkoç, H. A comprehensive review of ZnO materials and devices. J. Appl. Phys. 2005, 98, 041301. [Google Scholar] [CrossRef]
- Pearton, S.J.; Ren, F. ZnO-based materials for light emitting diodes. Curr. Opin. Chem. Eng. 2014, 3, 51–55. [Google Scholar] [CrossRef]
- Petti, L.; Münzenrieder, N.; Vogt, C.; Faber, H.; Büthe, L.; Cantarella, G.; Bottacchi, F.; Anthopoulos, T.D.; Tröster, G. Metal oxide semiconductor thin-film transistors for flexible electronics. Appl. Phys. Rev. 2016, 3, 021303. [Google Scholar] [CrossRef]
- Vittal, R.; Ho, K.-C. Zinc oxide based dye-sensitized solar cells: A review. Renew. Sustain. Energy Rev. 2017, 70, 920–935. [Google Scholar] [CrossRef]
- Khodja, A.A.; Sehili, T.; Pilichowski, J.-F.; Boule, P. Photocatalytic degradation of 2-phenylphenol on TiO2 and ZnO in aqueous suspensions. J. Photochem. Photobiol. A 2001, 141, 231–239. [Google Scholar] [CrossRef]
- Peng, T.-Y.; Lv, H.-J.; Zeng, P.; Zhang, X.-H. Preparation of ZnO Nanoparticles and Photocatalytic H2 Production Activity from Different Sacrificial Reagent Solutions. Chin. J. Chem. Phys. 2011, 24, 464–470. [Google Scholar] [CrossRef]
- Guo, Q.; Zhang, Q.; Wang, H.; Liu, Z.; Zhao, Z. Unraveling the role of surface property in the photoreduction performance of CO2 and H2O catalyzed by the modified ZnO. Mol. Catal. 2017, 436, 19–28. [Google Scholar] [CrossRef]
- Llorente, M.; Froehlich, J.; Dang, T.; Sathrum, A.; Kubiak, C.P. Photochemical and Photoelectrochemical Reduction of CO2. Annu. Rev. Phys. Chem. 2012, 63, 541–569. [Google Scholar]
- Centi, G.; Quadrelli, E.A.; Perathoner, S. Catalysis for CO2 conversion: A key technology for rapid introduction of renewable energy in the value chain of chemical industries. Energy Environ. Sci. 2013, 6, 1711–1731. [Google Scholar] [CrossRef]
- Xin, C.; Hu, M.; Wang, K.; Wang, X. Significant Enhancement of Photocatalytic Reduction of CO2 with H2O over ZnO by the Formation of Basic Zinc Carbonate. Langmuir 2017, 33, 6667–6676. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Hu, H.; Luo, G.; Zhu, S.; Guo, L.; Qu, P.; Shen, Q.; He, T. Crystal Facet-Dependent CO2 Photoreduction over Porous ZnO. ACS Appl. Mater. Interfaces 2020, 12, 56039–56048. [Google Scholar] [CrossRef] [PubMed]
- Boppella, R.; Anjaneyulu, K.; Basak, P.; Manorama, S.V. Facile Synthesis of Face Oriented ZnO Crystals: Tunable Polar Facets and Shape Induced Enhanced Photocatalytic Performance. J. Phys. Chem. C 2013, 117, 4597–4605. [Google Scholar] [CrossRef]
- Li, Y.; Xie, W.; Hu, X.; Shen, G.; Zhou, X.; Xiang, Y.; Zhao, X.; Fang, P. Comparison of Dye Photodegradation and its Coupling with Light-to-Electricity Conversion over TiO2 and ZnO. Langmuir 2010, 26, 591–597. [Google Scholar] [CrossRef]
- Thompson, W.A.; Olivo, A.; Zanardo, D.; Cruciani, G.; Signoretto, M.; Menegazzo, F.; Maroto-Valer, M.M. Systematic study of TiO2/ZnO mixed metal oxides for CO2 photoreduction. RSC Adv. 2019, 9, 21660–21666. [Google Scholar] [CrossRef]
- Zhang, X.; Qin, J.; Xue, Y.; Yu, P.; Zhang, B.; Wang, L.; Liu, R. Effect of aspect ratio and surface defects on the photocatalytic activity of ZnO nanorods. Sci. Rep. 2014, 4, 4596. [Google Scholar] [CrossRef]
- Liu, X.; Ye, L.; Liu, S.; Li, Y.; Ji, X. Photocatalytic Reduction of CO2 by ZnO Micro/nanomaterials with Different Morphologies and Ratios of {0001} Facets. Sci. Rep. 2016, 6, 38474. [Google Scholar] [CrossRef]
- Zhu, L.; Li, H.; Xia, P.; Liu, Z.; Xiong, D. Hierarchical ZnO Decorated with CeO2 Nanoparticles as the Direct Z-Scheme Heterojunction for Enhanced Photocatalytic Activity. ACS Appl. Mater. Interfaces 2018, 10, 39679–39687. [Google Scholar] [CrossRef]
- Li, J.; Wang, Z.; Chen, H.; Zhang, Q.; Hu, H.; Liu, L.; Ye, J.; Wang, D. A surface-alkalinized Ti3C2 MXene as an efficient cocatalyst for enhanced photocatalytic CO2 reduction over ZnO. Catal. Sci. Technol. 2021, 11, 4953–4961. [Google Scholar] [CrossRef]
- Kumar, S.; Saxena, S.K. A comparative study of CO2 sorption properties for different oxides. Mater. Renew. Sustain. Energy 2014, 3, 30. [Google Scholar] [CrossRef]
- Tan, L.-L.; Ong, W.-J.; Chai, S.-P.; Mohamed, A.R. Photocatalytic reduction of CO2 with H2O over graphene oxide-supported oxygen-rich TiO 2 hybrid photocatalyst under visible light irradiation: Process and kinetic studies. Chem. Eng. J. 2017, 308, 248–255. [Google Scholar] [CrossRef]
- Olivo, A.; Trevisan, V.; Ghedini, E.; Pinna, F.; Bianchi, C.; Naldoni, A.; Cruciani, G.; Signoretto, M. CO2 photoreduction with water: Catalyst and process investigation. J. CO2 Util. 2015, 12, 86–94. [Google Scholar] [CrossRef]
- Steingrube, A.; Voll, P. Selecting CO2 Sources for CO2 Utilization by Environmental-Merit-Order Curves. Environ. Sci. Technol. 2016, 50, 1093–1101. [Google Scholar]
- Liu, L.; Zhao, C.; Xu, J.; Li, Y. Integrated CO2 capture and photocatalytic conversion by a hybrid adsorbent/photocatalyst material. Appl. Catal. B 2015, 179, 489–499. [Google Scholar] [CrossRef]
- Zanardo, D.; Forghieri, G.; Tieuli, S.; Ghedini, E.; Menegazzo, F.; Di Michele, A.; Cruciani, G.; Signoretto, M. Effects of SiO2-based scaffolds in TiO2 photocatalyzed CO2 reduction. Catal. Today 2021, 387, 54–60. [Google Scholar] [CrossRef]
- Zhang, R.; Yin, P.-G.; Wang, N.; Guo, L. Photoluminescence and Raman scattering of ZnO nanorods. Solid State Sci. 2009, 11, 865–869. [Google Scholar] [CrossRef]
- Zhang, X.; Zheng, H.; Zhao, J.; Li, S.; Zhang, W. Structural and Optical Characteristics of Titanium-Doped Zinc Oxide Thin Films and Applications in Thin Film Transistors. J. Nanosci. Nanotechnol. 2017, 17, 4343–4347. [Google Scholar] [CrossRef]
- Leofanti, G.; Padovan, M.; Tozzola, G.; Venturelli, B. Surface area and pore texture of catalysts. Catal. Today 1998, 41, 207–219. [Google Scholar] [CrossRef]
- O1s Line, NIST XPS Database Website. Available online: https://srdata.nist.gov/xps/main_search_menu.aspx (accessed on 1 May 2023).
- Ballerini, G.; Ogle, K.; Barthés-Labrousse, M.-G. The acid–base properties of the surface of native zinc oxide layers: An XPS study of adsorption of 1,2-diaminoethane. Appl. Surf. Sci. 2007, 253, 6860–6867. [Google Scholar] [CrossRef]
- Fan, J.C.C.; Goodenough, J.B. X-ray photoemission spectroscopy studies of Sn-doped indium-oxide films. J. Appl. Phys. 1977, 48, 3524. [Google Scholar] [CrossRef]
- C 1s Line, NIST XPS Database Website. Available online: https://srdata.nist.gov/xps/main_search_menu.aspx (accessed on 1 May 2023).
- Ti 2p3/2 Line, NIST XPS Database Website. Available online: https://srdata.nist.gov/xps/main_search_menu.aspx (accessed on 1 May 2023).
- Wahyuono, R.A.; Hermann-Westendorf, F.; Dellith, A.; Schmidt, C.; Dellith, J.; Plentz, J.; Schulz, M.; Presselt, M.; Seyring, M.; Rettenmeyer, M.; et al. Effect of annealing on the sub-bandgap, defects and trapping states of ZnO nanostructures. Chem. Phys. 2017, 483–484, 112–121. [Google Scholar] [CrossRef]
- Shalish, I.; Temkin, H.; Narayanamurti, V. Size-dependent surface luminescence in ZnO nanowires. Phys. Rev. B 2004, 69, 245401. [Google Scholar] [CrossRef]
- Procházková, L.; Vaněcěk, V.; Čuba, V.; Pjatkan, R.; Martinez-Turtos, R.; Jakubec, I.; Buryi, M.; Omelkov, S.; Auffray, E.; Lecoq, P.; et al. Core–shell ZnO:Ga-SiO2 nanocrystals: Limiting particle agglomeration and increasing luminescence via surface defect passivation. RSC Adv. 2019, 9, 28946–28952. [Google Scholar] [CrossRef] [PubMed]
- Bandopadhyay, K.; Mitra, J. Zn interstitials and O vacancies responsible for n-type ZnO: What do the emission spectra reveal? RSC Adv. 2015, 5, 23540–23547. [Google Scholar] [CrossRef]
- Usui, H. Influence of Surfactant Micelles on Morphology and Photoluminescence of Zinc Oxide Nanorods Prepared by One-Step Chemical Synthesis in Aqueous Solution. J. Phys. Chem. C 2007, 111, 9060–9065. [Google Scholar] [CrossRef]
- Ahn, C.H.; Kim, Y.Y.; Kim, D.C.; Mohanta, S.K.; Cho, H.K. A comparative analysis of deep level emission in ZnO layers deposited by various methods. J. Appl. Phys. 2009, 105, 1–6. [Google Scholar] [CrossRef]
- Xu, Y.; Schoonen, M.A. The absolute energy positions of conduction and valence bands of selected semiconducting minerals. Am. Mineral. 2000, 85, 543–556. [Google Scholar] [CrossRef]
- Wu, Q.; Huang, F.; Zhao, M.; Xu, J.; Zhou, J.; Wang, Y. Ultra-small yellow defective TiO2 nanoparticles for co-catalyst free photocatalytic hydrogen production. Nano Energy 2016, 24, 63–71. [Google Scholar] [CrossRef]
- Naeem, M.; Qaseem, S.; Gul, I.H.; Maqsood, A. Study of active surface defects in Ti doped ZnO nanoparticles. J. Appl. Phys. 2010, 107, 124303. [Google Scholar] [CrossRef]
- Habisreutinger, S.N.; Schmidt-Mende, L.; Stolarczyk, J.K. Photocatalytic Reduction of CO2 on TiO2 and Other Semiconductors. Angew. Chem. Int. Ed. 2013, 52, 7372–7408. [Google Scholar] [CrossRef]
- Zanardo, D.; Ghedini, E.; Menegazzo, F.; Giordana, A.; Cerrato, G.; Di Michele, A.; Cruciani, G.; Signoretto, M. Traditional Venetian marmorino: Effect of zinc-based oxides on self-bleaching properties. J. Cult. Herit. 2021, 50, 171–178. [Google Scholar] [CrossRef]
- Brunauer, S.; Emmett, P.H.; Teller, E. Adsorption of gases in multimolecolar layers. J. Am. Chem. Soc. 1938, 60, 309–313. [Google Scholar] [CrossRef]
- Kubelka, P.; Munk, F. Ein Beitrag Zur Optik Der Farbanstriche. Z. Tech. Phys. 1931, 12, 593–601. [Google Scholar]
- Tauc, J.; Menth, A. States in the gap. J. Non-Cryst. Solids 1972, 8, 569–585. [Google Scholar] [CrossRef]
- Studenyak, I.; Kranjec, M.; Kurik, M. Urbach Rule in Solid State Physics. Int. J. Opt. Appl. 2014, 4, 76–83. [Google Scholar]
- Zn 2p3/2 Line, NIST XPS Database Website. Available online: https://srdata.nist.gov/xps/main_search_menu.aspx (accessed on 1 June 2023).


| Sample | Crystallite Size (nm) | SSA (m2·g−1) |
|---|---|---|
| 4CSZ | 18 | 35 |
| 6CSZ | 42 | 13 |
| T4CSZ | 17 | 33 |
| 4TCSZ | 14 | 51 |
| Sample | O at. % | Oa/Ob Ratio | C at. % | Cb at. % | O/Zn at. Ratio |
|---|---|---|---|---|---|
| 4CSZ | 41 | 2.3 | 7.5 | 1.7 | 0.79 |
| 6CSZ | 44 | 2.0 | 5.3 | 1.4 | 0.89 |
| T4CSZ | 40 | 2.2 | 4.8 | 1.2 | 0.76 |
| 4TCSZ | 40 | 2.7 | 4.6 | 1.2 | 0.76 |
| Sample | τ1 (ns) | τ2 (ns) | τ3 (ns) |
|---|---|---|---|
| 4CSZ | 0.2 | 5.2 | 93.7 |
| 6CSZ | 5.1 | 38.2 | 325.9 |
| T4CSZ | 2.9 | 27.3 | 264.7 |
| Sample | TiO2 Promotion | Annealing Temperature and Time (°C; h) |
|---|---|---|
| CSZ | / | / |
| 4CSZ | / | 400; 4 |
| 6CSZ | / | 600; 4 |
| T4CSZ | On annealed ZnO (4CSZ) (post-annealing addition) | 400; 1 |
| 4TCSZ | On unannealed Zn hydroxy carbonate (CSZ) (pre-annealing addition) | 400; 4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zanardo, D.; Forghieri, G.; Ghedini, E.; Menegazzo, F.; Giordana, A.; Cerrato, G.; Cattaruzza, E.; Di Michele, A.; Cruciani, G.; Signoretto, M. Effect of the Synthetic Parameters over ZnO in the CO2 Photoreduction. Molecules 2023, 28, 4798. https://doi.org/10.3390/molecules28124798
Zanardo D, Forghieri G, Ghedini E, Menegazzo F, Giordana A, Cerrato G, Cattaruzza E, Di Michele A, Cruciani G, Signoretto M. Effect of the Synthetic Parameters over ZnO in the CO2 Photoreduction. Molecules. 2023; 28(12):4798. https://doi.org/10.3390/molecules28124798
Chicago/Turabian StyleZanardo, Danny, Giulia Forghieri, Elena Ghedini, Federica Menegazzo, Alessia Giordana, Giuseppina Cerrato, Elti Cattaruzza, Alessandro Di Michele, Giuseppe Cruciani, and Michela Signoretto. 2023. "Effect of the Synthetic Parameters over ZnO in the CO2 Photoreduction" Molecules 28, no. 12: 4798. https://doi.org/10.3390/molecules28124798
APA StyleZanardo, D., Forghieri, G., Ghedini, E., Menegazzo, F., Giordana, A., Cerrato, G., Cattaruzza, E., Di Michele, A., Cruciani, G., & Signoretto, M. (2023). Effect of the Synthetic Parameters over ZnO in the CO2 Photoreduction. Molecules, 28(12), 4798. https://doi.org/10.3390/molecules28124798











