Terpenoids: Natural Compounds for Non-Alcoholic Fatty Liver Disease (NAFLD) Therapy
Abstract
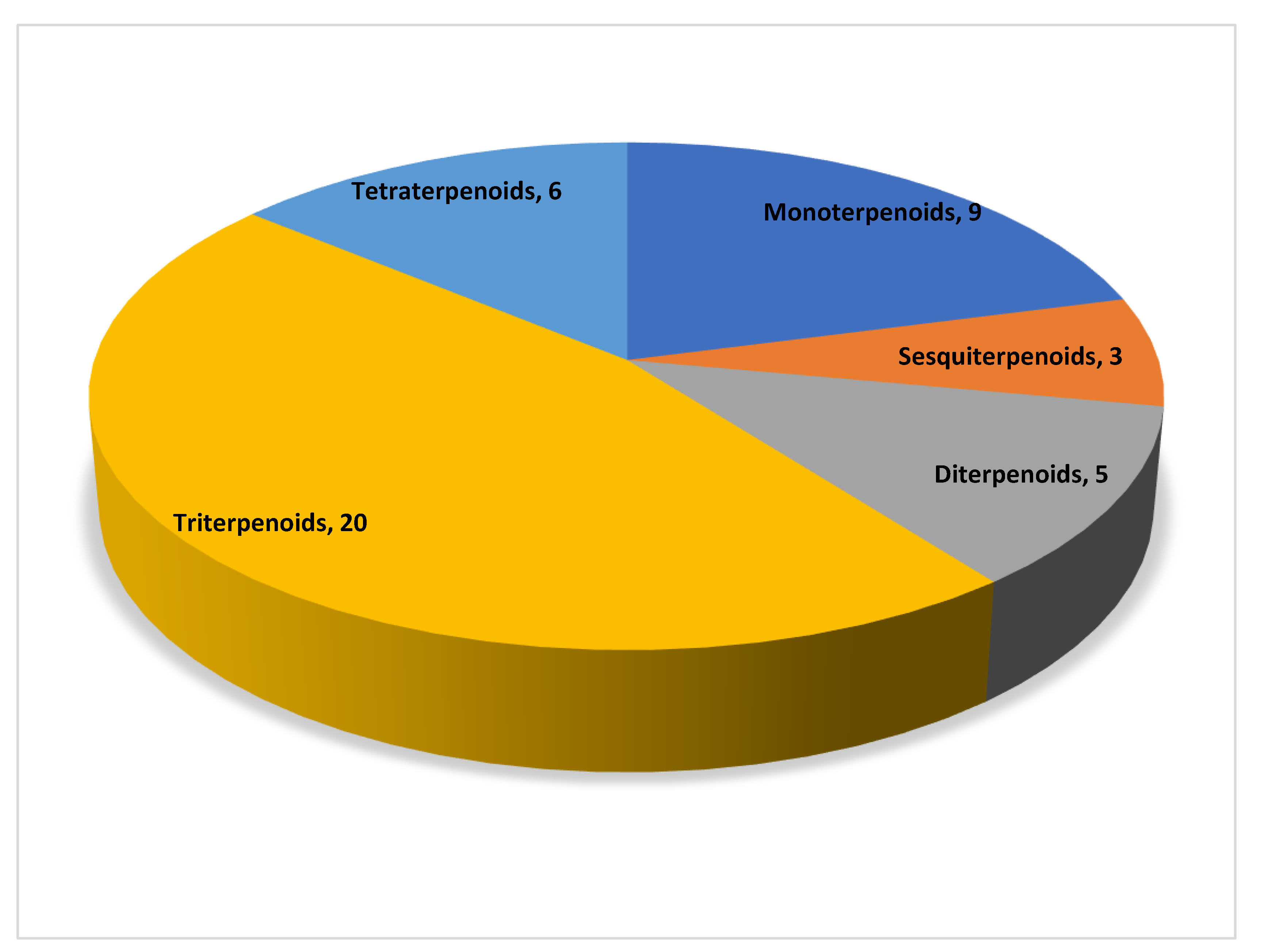
1. Introduction
2. Methods
3. Terpenoids and Their Mechanism of Action in the Treatment of Non-Alcoholic Fatty Liver Disease
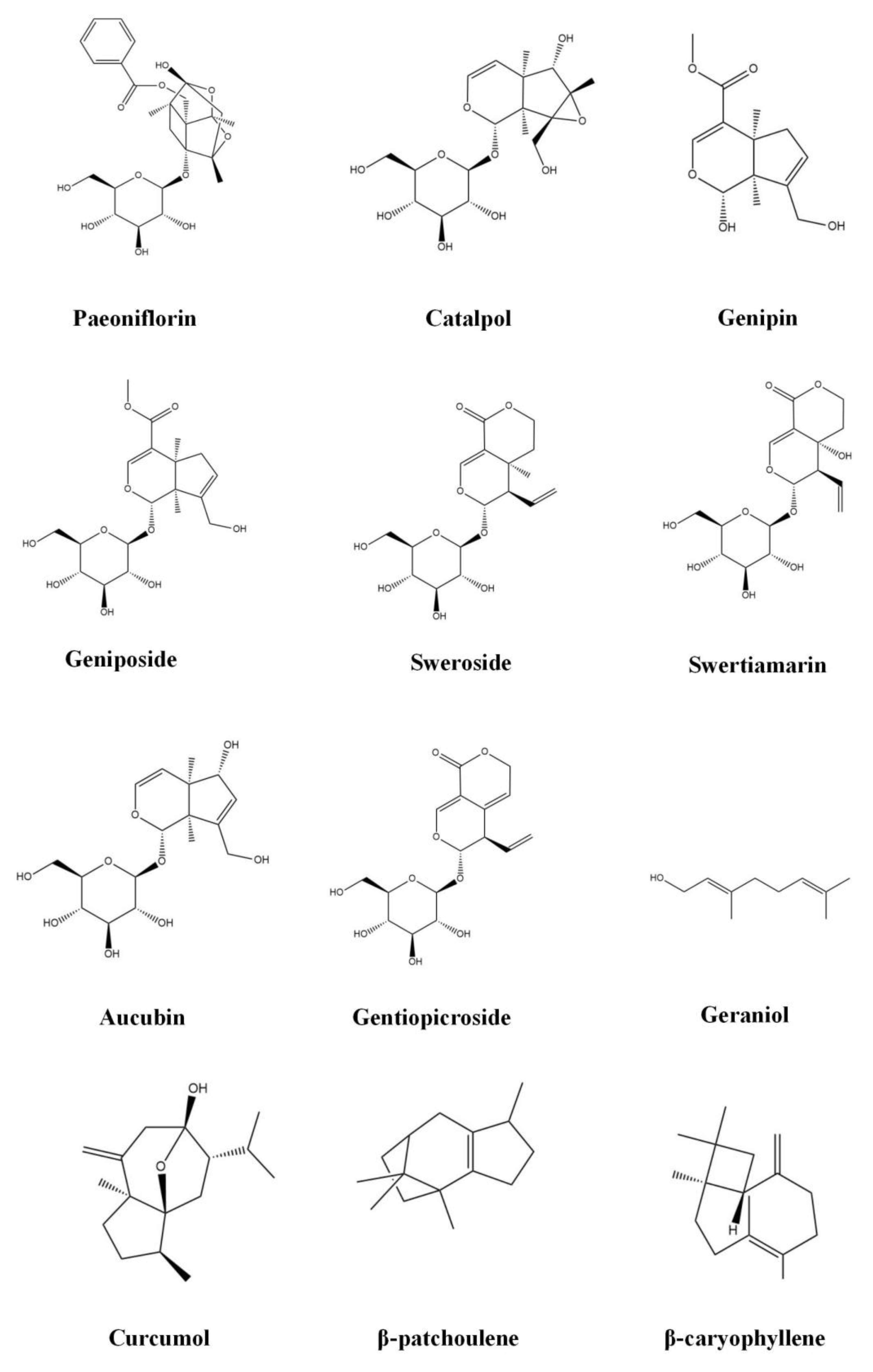
3.1. Monoterpenoids
3.1.1. Paeoniflorin
3.1.2. Catalpol
3.1.3. Geniposide
3.1.4. Genipin
3.1.5. Sweroside
3.1.6. Swertiamarin
3.1.7. Aucubin
3.1.8. Gentiopicroside
3.1.9. Geraniol
3.2. Sesquiterpenoids
3.2.1. Curcumol
3.2.2. β-Patchoulene
3.2.3. β-Caryophyllene
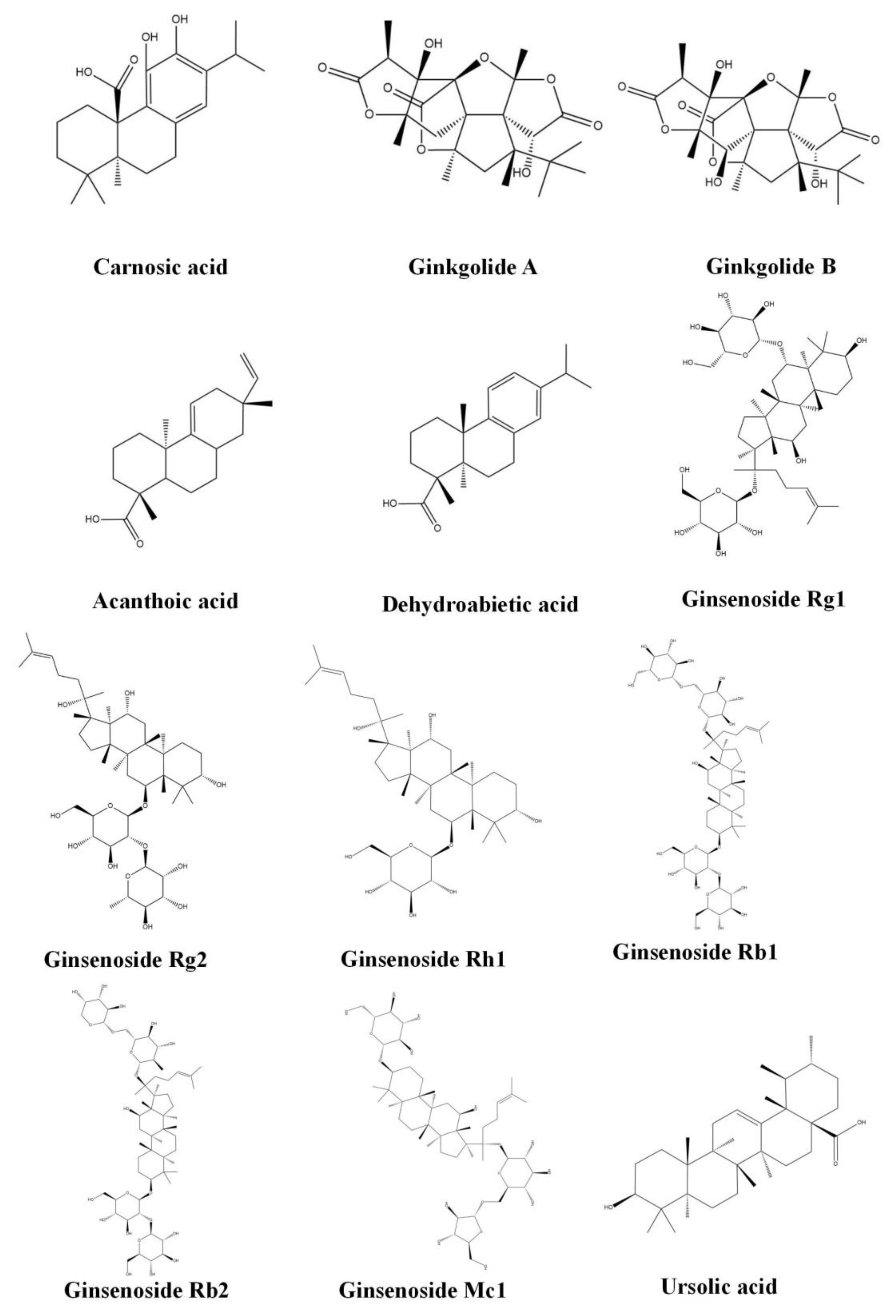
3.3. Diterpenoids
3.3.1. Carnosic Acid
3.3.2. Ginkgolide (A,B)
3.3.3. Acanthoic Acid
3.3.4. Dehydroabietic Acid
3.4. Triterpenoids
3.4.1. Ginsenoside (Rg1, Rg2, Rh1, Rb1, Rb2 and Mc1)
3.4.2. Ursolic Acid
3.4.3. Betulinic Acid
3.4.4. Glycyrrhizic Acid
3.4.5. Glycyrrhetinic Acid
3.4.6. Oleanolic Acid
3.4.7. Astragaloside IV
3.4.8. Mogroside V
3.4.9. Asiatic Acid
3.4.10. Corosolic Acid
3.4.11. Arjunolic Acid
3.4.12. Ganoderic Acid A
3.4.13. Ilexgenin A
3.4.14. Rotundic Acid
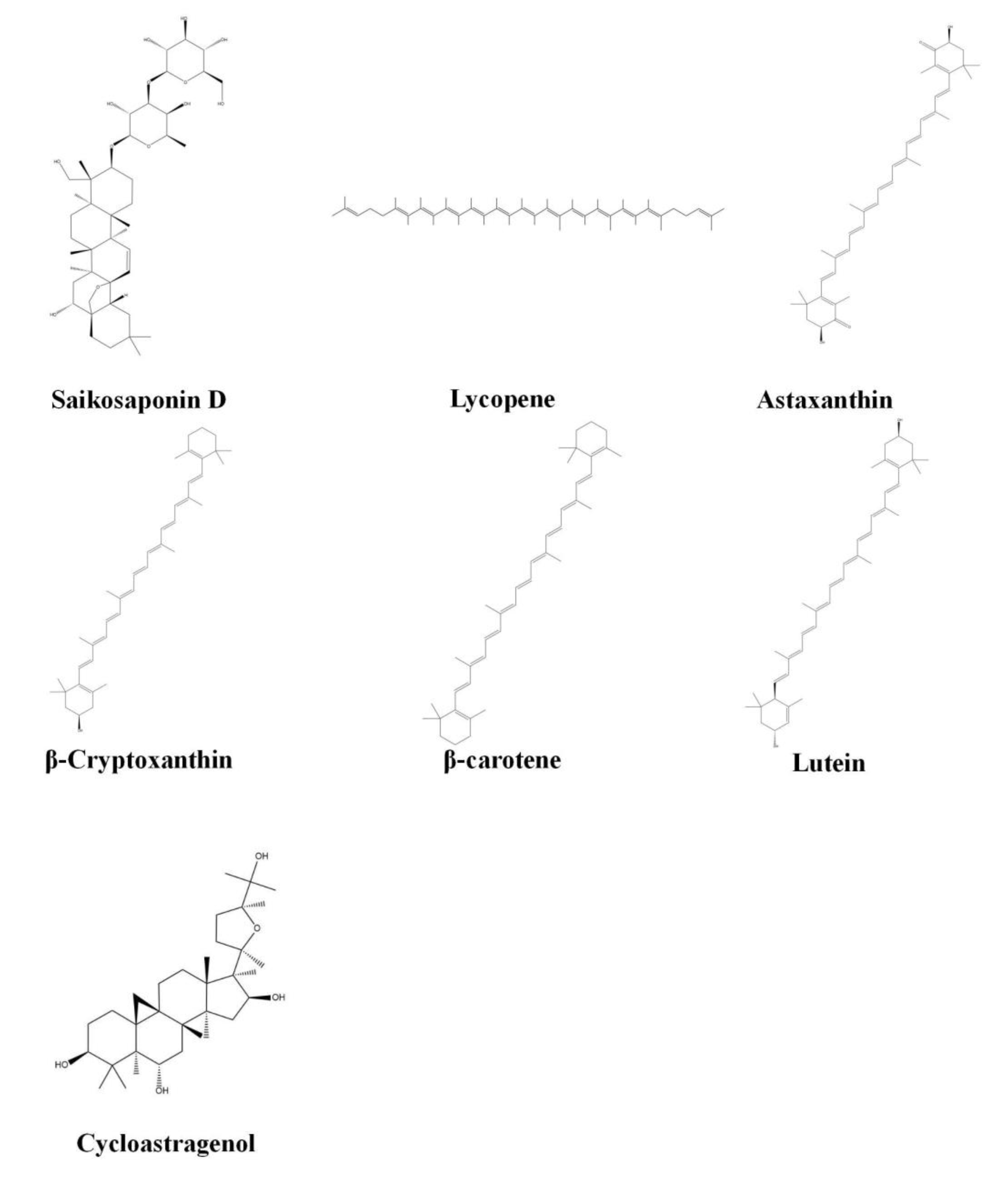
3.4.15. Saikosaponin D
3.5. Tetraterpenoids
3.5.1. Lycopene
3.5.2. Astaxanthin
3.5.3. β-Cryptoxanthin
3.5.4. β-Carotene
3.5.5. Lutein
3.5.6. Cycloastragenol
| Subclass | Compound | Molecular Formula | Weight (g/mol) | Sources |
|---|---|---|---|---|
| Monoterpenoid | Paeoniflorin | C23H28O11 | 480.5 | Paeonia albiflora Pallas |
| Iridoid | Catalpol | C15H22O10 | 362.3 | Rehmannia glutinosa (Gaertn.) Libosch. ex Fisch. and C. A. Mey. |
| Iridoid | Geniposide | C17H24O10 | 388.4 | Gardenia jasminoides J. Ellis |
| Iridoid | Genipin | C11H14O5 | 226.2 | Gardenia jasminoides J. Ellis |
| Iridoid | Sweroside | C16H22O9 | 358.3 | Swertia bimaculata (Siebold and Zucc.) Hook. f. and Thomson ex C. B. Clarke |
| Iridoid | Swertiamarin | C16H22O10 | 374.3 | Swertia bimaculata (Siebold and Zucc.) Hook. f. and Thomson ex C. B. Clarke |
| Iridoid | Aucubin | C15H22O9 | 346.3 | Eucommia ulmoides Oliv., Plantago asiatica L., Scrophularia ningpoensis Hemsl. |
| Iridoid | Gentiopicroside | C16H20O9 | 356.3 | Gentiana scabra Bunge |
| Monoterpenoid | Geraniol | C10H18O | 154.3 | Elsholtzia ciliata (Thunb.) Hyl., Murraya exotica L., Rosa rugosa Thunb. |
| Sesquiterpenoid | Curcumol | C15H24O2 | 236.4 | Curcuma phaeocaulis Valeton |
| Sesquiterpenoid | β-patchoulene | C15H24 | 204.4 | Pogostemon cablin (Blanco) Benth. |
| Sesquiterpenoid | β-caryophyllene | C15H24 | 204.4 | Rosmarinus officinalis L., Cinnamomum cassia (L.) D. Don, Ocimum basilicum L., Lavandula angustifolia Mill. |
| Diterpenoid | Carnosic acid | C20H28O4 | 332.4 | Rosmarinus officinalis Linnaeus, Salvia japonica Thunb. |
| Diterpenoid | Ginkgolide A | C20H24O9 | 408.4 | Ginkgo biloba L. |
| Diterpenoid | Ginkgolide B | C20H24O10 | 424.4 | Ginkgo biloba L. |
| Diterpenoid | Acanthoic acid | C20H30O2 | 302.5 | Eleutherococcus senticosus (Rupr. and Maxim.) Maxim. |
| Diterpenoid | Dehydroabietic acid | C20H28O2 | 300.4 | Pinus massoniana Lamb., Picea asperata Mast. |
| Triterpenoid | Ginsenoside Rg1 | C42H72O14 | 801 | Panax ginseng C. A. Mey. |
| Triterpenoid | Ginsenoside Rg2 | C42H72O13 | 785 | Panax ginseng C. A. Mey. |
| Triterpenoid | Ginsenoside Rh1 | C36H62O9 | 638.9 | Panax ginseng C. A. Mey. |
| Triterpenoid | Ginsenoside Rb1 | C54H92O23 | 1109.3 | Panax ginseng C. A. Mey. |
| Triterpenoid | Ginsenoside Rb2 | C53H90O22 | 1079.3 | Panax ginseng C. A. Mey. |
| Triterpenoid | Ginsenoside Mc1 | C47H80O17 | 917.1 | Panax ginseng C. A. Mey. |
| Triterpenoid | Ursolic acid | C30H48O3 | 456.7 | Gentiana scabra Bunge, Pseudocydonia sinensis (Thouin) C. K. Schneid., Eriobotrya japonica (Thunb.) Lindl., Pyrrosia lingua (Thunb.) Farw. and Plantago asiatica L. |
| Triterpenoid | Betulinic acid | C30H48O3 | 456.7 | Betula platyphylla Sukaczev |
| Triterpenoid | Glycyrrhizic acid | C42H62O16 | 822.9 | Glycyrrhiza uralensis Fisch. |
| Triterpenoid | Glycyrrhetinic acid | C30H46O4 | 470.7 | Glycyrrhiza uralensis Fisch. |
| Triterpenoid | Oleanolic acid | C30H48O3 | 456.7 | Ophiopogon japonicus (L. f.) Ker Gawl., Swertia leducii Franch., Ligustrum lucidum W. T. Aiton |
| Triterpenoid | Astragaloside IV | C41H68O14 | 785 | Astragalus membranaceus var. mongholicus (Bunge) P. K. Hsiao |
| Triterpenoid | Mogroside V | C60H102O29 | 1287.4 | Siraitia grosvenorii (Swingle) C. Jeffrey ex A. M. Lu and Zhi Y. Zhang |
| Triterpenoid | Asiatic acid | C30H48O5 | 488.7 | Centella asiatica (L.) Urb. |
| Triterpenoid | Corosolic acid | C30H48O4 | 472.7 | Eriobotrya japonica (Thunb.) Lindl., Actinidia chinensis Planch., Hippophae rhamnoides L. and Lagerstroemia speciosa (L.) Pers |
| Triterpenoid | Arjunolic acid | C30H48O5 | 488.7 | Cymbidium goeringii (Rchb. f.) Rchb. f., Chrysanthemum morifolium (Ramat.) Hemsl. |
| Triterpenoid | Ganoderic acid A | C30H44O7 | 516.7 | Ganoderma lucidum (Curtis) P. Karst. |
| Triterpenoid | Ilexgenin A | C30H46O6 | 502.7 | Quercus aliena Blume, Ilex brachyphylla (Hand.-Mazz.) S. Y. Hu |
| Triterpenoid | Rotundic acid | C30H48O5 | 488.7 | Ilex rotunda Thunb. |
| Triterpenoid | Saikosaponin D | C42H68O13 | 781 | Radix bupleuri |
| Tetraterpenoid | Lycopene | C40H56 | 536.9 | Solanum lycopersicum L., Citrullus lanatus (Thunb.) Matsum. and Nakai, Citrus aurantium (Lour.) Engl., Daucus carota var. sativa Hoffm. |
| Tetraterpenoid | Astaxanthin | C40H52O4 | 596.8 | Shrimp, fish, crab, algae, etc. |
| Tetraterpenoid | β-cryptoxanthin | C₄₀H₅₆O | 552.9 | Orange, papaya, egg yolk, butter, apples, etc. |
| Tetraterpenoid | β-carotene | C40H56 | 536.9 | Carrots, apricots, sweet potatoes, mature squashes, pumpkins, mangoes, etc. |
| Tetraterpenoid | Lutein | C40H56O2 | 568.9 | Spinach, kale, yellow carrots, etc. |
| Tetraterpenoid | Cycloastragenol | C30H50O5 | 490.7 | Astragalus membranaceus var. mongholicus (Bunge) P. K. Hsiao |
| Compound | Animal/Cell Model | Dosage (mg/kg/d; μM/24 h) | Target/Pathways/Mechanism | Effects | Reference |
|---|---|---|---|---|---|
| Paeoniflorin | HFD-induced NAFLD mice | 0.05% in diet | Activation of the CD36/AMPK signaling pathway | Reduced body weight, improved insulin resistance, anti-inflammatory, inhibition of lipid accumulation, attenuated hepatic adipose infiltration | [24] |
| HFD-induced NAFLD rats | 20 | Regulation of the IRS/Akt/GSK3β signaling pathway | Inhibition of lipid accumulation, improved insulin resistance, anti-oxidative stress, liver protection | [25] | |
| Fructose-induced metabolic syndrome rats | 10, 20, 40 | Activation of the AMPK signaling pathway | Inhibition of hepatic lipid accumulation, improved insulin resistance, inhibition of hepatic steatosis, inhibition of hepatic lipogenesis, promotion of fatty acid oxidation | [26] | |
| HFD-induced NAFLD rats | 20, 60, 100 | Inhibition of the ROCK/NF-κB signaling pathway | Anti-inflammatory, ameliorated hepatic steatosis, reduced lipids | [27] | |
| Catalpol | HFD-induced NAFLD mice; PA-induced HepG2 cells | 100, 200, 400; 100, 200, 400 | Activation of the AMPK signaling pathway | Ameliorated hepatic steatosis, reduced body weight, inhibition of lipid accumulation | [29] |
| HFD-induced NAFLD mice; PA-induced HepG2 cells | 100; 10 μg/mL/24h | / | Inhibition of autophagy, ameliorated hepatic steatosis, reduced liver weight, reduced liver fat | [31] | |
| (LDLr−/−)+ HFD-induced NAFLD mice | 100 | Regulation of the p66shc/cytochrome C signaling pathway | Attenuated liver injury, anti-oxidative stress, inhibition of hepatic steatosis, anti-apoptosis | [32] | |
| HFD-induced NAFLD mice | 100, 200, 400 | / | Ameliorated hepatic steatosis, reduced body weight, improvement of lipid metabolism disorders, inhibition of lipid accumulation, anti-inflammatory, anti-apoptosis | [33] | |
| Geniposide | (PA + OA)-induced HepG2 cells | 0, 65, 130, 260, 390, 520 μmol/L/24 h | Upregulation of the Nrf2/AMPK/mTOR signaling pathways | Inhibition of lipid accumulation, anti-oxidative stress, anti-inflammatory | [35] |
| HFD-induced NAFLD rats | 25, 50, 100 | Increased expression of PPARa gene | Ameliorated hepatic steatosis, anti-oxidative stress | [36] | |
| Genipin | HFD-induced NAFLD mice; (PA + OA)-induced cells primary hepatocytes of mice | 5, 20; 5, 20 | Regulation of the miR-142a-5p/SREBP-1c axis | Reduced body weight gain, increased locomotor activity, improved insulin resistance, alleviated hyperlipidemia, inhibition of lipid accumulation | [38] |
| HFD-induced NAFLD mice | 5, 20 | Suppressed UCP2 | Reversed liver damage, anti-pyroptosis | [39] | |
| Sweroside | HFD-induced NAFLD mice | 60, 120, 240 | Increases expression of PPARa gene | Reduced body weight, improved insulin resistance, inhibited hepatic steatosis, anti-inflammatory | [42] |
| MCD diet-induced NAFLD mice | 5, 30 | Suppressed activation of the NLRP3 inflammasome | Anti-inflammatory, inhibition of hepatic lipid accumulation, anti-fibrosis | [43] | |
| Swertiamarin | HFD-induced NAFLD mice; LPSO-induced murine monocytic cells | 10, 100; 1, 10, 50 | Suppressed activation of the p38 MAPK and NF-κB signaling pathways | Ameliorated hepatic steatosis, anti-inflammatory, reduced body weight, improved insulin resistance | [45] |
| Aucubin | Tyloxapol-induced NAFLD mice | 10, 20, 40 | Activation of the Nrf2/HO-1 and AMPK signaling pathways | Inhibition of lipid accumulation, anti-oxidative stress, anti-inflammatory | [47] |
| Gentiopicroside | Tyloxapol-induced NAFLD mice; (PA + OA)-induced HepG2 cells | 20, 40, 80; 0, 4, 20, 100, 200, 500 | Upregulation of the Nrf2 signaling pathway | Inhibition of hepatic lipid accumulation, anti-oxidative stress | [49] |
| Geraniol | MCD-induced NAFLD rats | 25, 100, 200 | / | Inhibition of hepatic lipid accumulation, anti-fibrosis, anti-apoptosis, anti-inflammatory, anti-oxidative stress | [50] |
| Curcumol | HFD-induced NAFLD mice | 15, 30, 60 | Regulation of the YAP/NCOA4 signaling pathway | Inhibition of hepatocyte senescence, suppressed ferritinophagy | [52] |
| HFD-induced NAFLD rats | 25, 50, 100 | Regulation of the TLR4, TAK1, and NF-κB/P65 signaling pathways | Anti-inflammatory, improved liver function, anti-fibrosis, anti-apoptosis | [53] | |
| β-patchoulene | HFD-induced NAFLD rats; (FFA + PA + OA)-induced HepG2 cells | 10, 20, 40; 40 | Activation of the AMPK signaling pathway | Inhibition of hepatic lipid accumulation, improved insulin resistance, ameliorated hepatic steatosis, inhibition of hepatic lipogenesis, promotion of fatty acid oxidation | [54] |
| HFD-induced NAFLD rats | 10, 20, 40 | Activation of the CD36/AMPK signaling pathway | Reduced body weight, reversed liver damage, ameliorated hepatic steatosis, anti-oxidative stress, anti-inflammatory | [55] | |
| β-caryophyllene | PA-induced HepG2 cells | 40 | Activation o the f AMPK signaling pathway | Inhibition of hepatic lipid accumulation | [56] |
| Carnosic acid | Obese leptin-deficient (ob/ob) mice | 0.05% in diet | / | Reduced body weight, inhibition of lipid accumulation, recovered glucose tolerance | [60] |
| Obese leptin-deficient (ob/ob) mice | 0.01, 0.02% in diet | / | Improved glucose tolerance, inhibition of lipid accumulation, reduced body weight | [61] | |
| HFD-induced NAFLD mice | 15 | Upregulation of MARCKS expression/impairment of the PI3K/AKT and NLRP3 inflammasome signaling pathways | Inhibition of lipid accumulation, anti-inflammatory, improved insulin resistance | [63] | |
| HFD-induced NAFLD mice, PA-induced human L0246 hepatic cell | 30, 60; 10 | Inhibition of the miR-34a/SIRT1/p66shc signaling pathway | Inhibition of lipid accumulation, anti-apoptosis | [64] | |
| Ginkgolide A | HFD-induced NAFLD mice; NEFA-induced HepG2 cell | 5; 0, 10, 50, 100 | Inhibition of lipid accumulation, induced cellular lipoapoptosis, anti-inflammatory, reduced body weight | [69] | |
| Ginkgolide B | HFD-induced NAFLD mice | 0.1 % in diet | Activation of pregnane X receptor | Reduced body weight, ameliorated hepatic steatosis | [70] |
| HFD-induced NAFLD mice; (PA + OA)-induced HepG2 cells | 20, 30; 4, 8, 16 | Increased Nrf2 expression | Anti-oxidative stress, reduced body weight, inhibition of lipid accumulation, anti-inflammatory | [71] | |
| Acanthoic acid | Modified Lieber–DeCarli diet-induced mice | 20, 40 | Via FXR–LXR axis | Inhibition of hepatic lipid accumulation, anti-fibrosis, regulation of fatty acid synthesis | [75] |
| Dehydroabietic acid | HFD-induced NAFLD mice; OA-induced HL7702 cells | 10, 20; 2.5, 5, 10 | Activation of the Keap1/Nrf2-ARE signaling pathway | Reduced blood lipid, inhibition of ferroptosis | [77] |
| Ginsenoside Rg1 | (PA + OA)-induced HepG2 cells | 25, 50 | Regulation of PPAR ɑ and PPAR γ expression | Inhibition of lipid accumulation, ameliorated hepatic steatosis | [80] |
| D-galactose-induced fatty liver disease mice | 40 | Upregulation of FOXO1 gene | Anti-inflammatory, inhibition of lipid accumulation | [81] | |
| HFD-induced NAFLD mice | 20, 40 | / | Anti-inflammatory, reduced body weight, alleviated ER stress | [82] | |
| HSD-induced NAFLD rats | 100 | Regulation of Atf3 and Acox2 gene | Reduced body weight, reduced blood lipid, alleviated hepatic steatosis | [83] | |
| Ginsenoside Rg2 | HFD-induced NAFLD mice | 2.5, 5, 10 | Regulation of the SIRT1 signaling pathways | Improvement of lipid and glucose disorders, anti-oxidative stress, anti-apoptosis, inhibition of lipid accumulation | [85] |
| Ginsenoside Rh1 | HFD-induced NAFLD rats | 3 | / | Anti-fibrotic | [86] |
| Ginsenoside Rb1 | HFD-induced NAFLD mice | 10 | Activation of PPAR-γ expression | Reduced body weight, improved glucose metabolism, inhibition of lipid accumulation, anti-apoptosis | [87] |
| Ginsenoside Rb2 | db/db mice, OA-induced HepG2 cells | 10; pretreated with 0.1, 1, 10, 50, 100 μmol/L/4h | Regulation of the SIRT1 and AMPK signaling pathways | Alleviated hepatic steatosis, improved glucose tolerance, regulation of hepatic autophagy, inhibition of lipid accumulation | [88] |
| Ginsenoside Mc1 | HFD-induced NAFLD mice | 10 | / | Alleviated ER stress, anti-apoptosis, improved insulin resistance, alleviated hepatic steatosis | [89] |
| Ursolic acid | HFD-induced NAFLD rats; human normally hepatic immortal cell line HL-7702 | 0.125, 0.25, 0.5% in diet; 0, 25, 50, 100 | Regulation of PPAR ɑ expression | Reduced body weight, alleviated hepatic steatosis, improved metabolic disorders, improved insulin resistance, anti-inflammatory, anti-oxidative stress | [91] |
| db/db mice (type 2 diabetic mouse model); PA-induced HepG2 cells | 0.14% in diets; 10–30 | / | Inhibition of lipid accumulation, alleviated ER stress, reduced liver weight and reduced liver injury, alleviated hepatic steatosis | [92] | |
| DMSO-induced human hepatocellular carcinoma cell | 5, 10 | Regulation of LXRα | Inhibition of lipid accumulation, alleviated hepatic steatosis, reduced blood lipids | [93] | |
| Fructose induced NAFLD newborn rats | 10 | / | Inhibition of lipid accumulation | [94] | |
| Betulinic acid | HFD-induced NAFLD rats | 0.1% in diet | Regulation of the PERK/EIF2α/ATF4/CHOP signaling pathway | Enhanced energy expenditure, modulation of bile acids, alleviated hepatic steatosis, anti-inflammatory, alleviated ER stress | [95] |
| HFD-induced NAFLD rats; insulin-resistant HepG2 cells | 5, 10; 10–40 | Regulation of the AMPK–mTOR–SREBP signaling pathway | Inhibition of lipid accumulation | [96] | |
| HFD-induced NAFLD mice; (PA + OA)-induced mice primary hepatocytes | 150; 10 | Inhibition of the YY1/FAS signaling pathway | Inhibition of lipid accumulation, alleviated fatty acid synthesis, anti-fibrosis, anti-inflammatory, inhibition of excessive lipogenesis | [97] | |
| Glycyrrhizic acid | MCD diet-induced NAFLD mice | 12.5, 25, 50 | / | Inhibition of lipid accumulation, anti-inflammatory, anti-fibrosis, improved lipid metabolism | [98] |
| HFD-induced NAFLD mice | 15, 30, 60 | / | Inhibition of lipid accumulation, reduced hepatic lipogenesis, reduced body weight, ameliorated hepatic steatosis, reduced serum glucose, improved glucose tolerance and insulin sensitivity | [99] | |
| MCD diet-induced NAFLD mice | 30, 50 | / | Inhibition of lipid accumulation, modulation of bile acids, anti-inflammatory, anti-fibrosis | [101] | |
| Glycyrrhetinic acid | HFD-induced NAFLD mice | 15, 30, 60 | / | Regulation of vitamin A metabolism, protection against hepatic injury | [103] |
| HFD-induced NAFLD mice | 60 | Suppression of HNF4α | Reduced blood glucose, improved insulin resistance | [104] | |
| Oleanolic acid | HFD-induced NAFLD rats | 25, 50, 100 | Inhibition of LXRs, activation of the AMPK pathways | Alleviated hepatic steatosis, anti-inflammatory, anti-oxidative stress, improved insulin resistance | [107] |
| HFHCD-induced NAFLD rats | 80 | / | Decreased blood lipids, anti-oxidative stress, reversed hepatic degeneration | [109] | |
| Astragaloside IV | (PA + OA)-induced HepG2 cells and primary murine hepatocytes | 50–200 | Activation of the AMPK signaling pathway | Inhibition of lipid accumulation, inhibition of lipogenesis, alleviated ER stress | [113] |
| High-concentration insulin or OA-induced HepG2 cells | 25.6, 51.2, 102.4 | Inhibition of protein tyrosine phosphatase 1B | Improved insulin resistance, inhibition of lipid accumulation | [114] | |
| Mogroside V | HFD-induced NAFLD mice | 400, 800 | Upregulation of pAMPK expression | Inhibition of lipid accumulation, anti-inflammatory, anti-oxidative stress | [116] |
| HFD-induced NAFLD mice; (PA + OA)-induced human LO2 cells | 25, 50, 100; 15, 30, 60,120 | Activation of the AMPK signaling pathway | Inhibition of lipid accumulation, ameliorated hepatic steatosis | [117] | |
| Asiatic acid | HFD-induced NAFLD rats | 4, 8 | Inhibition of the ERS signaling pathway | Inhibition of lipid accumulation, anti-inflammatory, anti-oxidative stress | [119] |
| Corosolic acid | HFD + CCl4-induced NAFLD mice; FFA + OA + PA-induced HepG2 cells | 10, 20; 5, 10, 20 | Regulation of the TGF-β1/Smad2, NF-κB, and AMPK signaling pathways | Inhibition of lipid accumulation, anti-inflammatory, anti-fibrosis | [121] |
| Arjunolic acid | HFD-induced NAFLD rats; (PA + OA)-induced HepG2 cells | 100, 200; 12.5, 50 | Upregulation of PPARγ expression | Inhibition of lipid accumulation, ameliorated hepatic steatosis, reduced blood lipids | [123] |
| Ganoderic acid A | HFD-induced NAFLD rats | 20, 40 | Activation of the AMPK signaling pathway | Inhibition of lipid accumulation, anti-inflammatory, reduced live weight | [125] |
| Ilexgenin A | HFD-induced NAFLD rats | 80 | / | Ameliorated hepatic steatosis, hypolipidemic, anti-inflammatory, enhanced effects of simvastatin | [126] |
| Rotundic acid | HFD-induced NAFLD rats; insulin-induced primary hepatocytes | 10, 30, 100; 6.25–200 | Downregulation of the SREBP-1c/SCD1 signaling pathway | Inhibition of lipid accumulation, improved dyslipidemia, protection against hepatic injury, anti-inflammatory, inhibition of excessive lipogenesis | [128] |
| Saikosaponin D | TAA-induced liver injury mice; HFD-induced NAFLD mice | 2 | / | Reduced blood lipids, anti-oxidative stress, anti-inflammatory | [130] |
| Lycopene | HFD-induced NAFLD mice | 100, 1000 | Upregulation of PPARa-inducible genes | Ameliorated hepatic steatosis | [135] |
| HFD-induced NAFLD mice | 0.004, 0.012% in diet | / | Inhibition of lipid accumulation, improved insulin resistance, anti-fibrosis, anti-inflammatory, anti-oxidative stress | [136] | |
| HFD-induced NAFLD rats | 5, 10, 20 | Downregulated expression of TNF-ɑ and CYP2E1 | Improved lipid profiles, reduced lipid peroxides, reduced blood lipids | [137] | |
| HFD-induced NAFLD mice | 0.05% in diet | microRNA-21-induced downregulation of fatty-acid-binding protein 7 | Ameliorated hepatic steatosis, inhibition of hepatic lipid accumulation | [138] | |
| HFD-induced NAFLD rats | 20 | / | Ameliorated hepatic steatosis, reduced liver weight, reduced blood lipids | [140] | |
| Astaxanthin | HFD-induced NAFLD mice | 0.02% in diet | Inhibition o the f eIF-2 signaling pathway | Inhibition of lipid accumulation, anti-inflammatory, anti-fibrosis, anti-oxidative stress | [147] |
| HFD-induced NAFLD mice | 0.003, 0.01, 0.03% in diet | / | Alleviated hepatic steatosis, anti-inflammatory, anti-oxidative stress | [148] | |
| Hepatic stellate cells from humans and mice | 0–200 | Inhibition of the TGFβ1–Smad3 signaling pathway | Anti-oxidative stress, anti-fibrosis | [149] | |
| HFD-induced NAFLD mice | 80 | / | Alleviated hepatic steatosis, anti-inflammatory, anti-oxidative stress | [150] | |
| HFD-induced NAFLD mice | 0.0067, 0.02% in diet | / | Inhibition of lipid accumulation, alleviated hepatic steatosis, improved glucose intolerance, improved insulin resistance, anti-inflammatory, anti-fibrosis | [151] | |
| HFD-induced NAFLD mice; human liver cell line | 10, 30, 60; 30, 60, 90 | Upregulating the FGF21/PGC-1α signaling pathway | Inhibition of lipid accumulation, anti-oxidative stress, anti-apoptosis, anti-inflammatory, anti-fibrosis, attenuated mitochondrial dysfunction | [152] | |
| β-cryptoxanthin | HRCD + DKO-induced NAFLD mice | 10 | Activation of the SIRT1/AMPK signaling pathway | Inhibition of lipid accumulation, alleviated hepatic steatosis, increased cholesterol efflux | [156] |
| HFD-induced NASH mice | 0.003% in diet | / | Anti-inflammatory, anti-oxidative stress, anti-fibrosis, alleviated hepatic steatosis, inhibition of lipid accumulation | [157] | |
| HFD-induced NASH mice | 0.003% in diet | / | Anti-inflammatory, anti-oxidative stress, anti-fibrosis, alleviated hepatic steatosis, inhibition of lipid accumulation, improved liver dysfunction | [158] | |
| β-carotene | HFD-induced NAFLD rats | 70 | / | Alleviated hepatic steatosis, anti-inflammatory | [163] |
| Lutein | HFD-induced NAFLD rats | 0, 12.5, 25, 50 | Activation of the SIRT1/PPAR-α signaling pathway | Reduced body weight, alleviated hepatic steatosis, improved insulin resistance | [165] |
| Cycloastragenol | HFD-induced NAFLD mice; FXR deletion HepG2 cells | 0.1% in diet; 25 | Regulation of the FXR signaling pathway | Alleviated hepatic steatosis, inhibition of lipid accumulation, reduced blood glucose, anti-oxidative stress | [167] |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
Abbreviations
| non-alcoholic fatty liver disease | NAFLD |
| non-alcoholic fatty liver | NAFL |
| non-alcoholic steatohepatitis | NASH |
| metabolic-associated fatty liver disease | MAFLD |
| hepatic fibrosis | HF |
| palmitate | PA |
| methionine-choline deficient | MCD |
| low-density lipoprotein receptor knockout | LDLr−/− |
| lipopolysaccharide | LPSO |
| non-esterified fatty acid | NEFA |
| Yin Yang 1 | YY1 |
| fatty acid synthase | FAS |
| endoplasmic reticulum | ER |
| free fatty acids | FFA |
| carbon tetrachloride | CCl4 |
| thioacetamide | TAA |
| high-fat + high-carbohydrate diet | HFHCD |
| uncoupling protein-2 | UCP2 |
| liver X receptors | LXRs |
| anti-oxidant redux elements | ARE |
| over the counter | OTC |
| Food and Drug Administration | FDA |
| high-refined carbohydrate diet | HRCD |
| double knockout | DKO |
| wild-type | WT |
| β-carotene oxygenase 1 | BCO1 |
| β-carotene oxygenase 2 | BCO2 |
| sirtuin 1 | SIRT 1 |
| farnesoid X receptor | FXR |
| liver X receptor | LXR |
| sterol regulatory element binding proteins-1c | SREBP-1c |
| steroyl-coA desaturase-1 | SCD-1 |
References
- Lazarus, J.V.; Mark, H.E.; Anstee, Q.M.; Arab, J.P.; Batterham, R.L.; Castera, L.; Cortez-Pinto, H.; Crespo, J.; Cusi, K.; Dirac, M.A.; et al. Advancing the global public health agenda for NAFLD: A consensus statement. Nat. Rev. Gastroenterol. Hepatol. 2022, 19, 60–78. [Google Scholar] [CrossRef] [PubMed]
- Powell, E.E.; Wong, V.W.; Rinella, M. Non-alcoholic fatty liver disease. Lancet 2021, 397, 2212–2224. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.H.; Lee, H.W.; Yoo, J.J.; Cho, Y.; Kim, S.U.; Lee, T.H.; Jang, B.K.; Kim, S.G.; Ahn, S.B.; Kim, H.; et al. KASL clinical practice guidelines: Management of nonalcoholic fatty liver disease. Clin. Mol. Hepatol. 2021, 27, 363–401. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, D.S.; An, T.H.; Park, H.J.; Kim, W.K.; Bae, K.H.; Oh, K.J. Metabolic Spectrum of Liver Failure in Type 2 Diabetes and Obesity: From NAFLD to NASH to HCC. Int. J. Mol. Sci. 2021, 22, 4495. [Google Scholar] [CrossRef] [PubMed]
- Eslam, M.; Sarin, S.K.; Wong, V.W.; Fan, J.G.; Kawaguchi, T.; Ahn, S.H.; Zheng, M.H.; Shiha, G.; Yilmaz, Y.; Gani, R.; et al. The Asian Pacific Association for the Study of the Liver clinical practice guidelines for the diagnosis and management of metabolic associated fatty liver disease. Hepatol. Int. 2020, 14, 889–919. [Google Scholar] [CrossRef]
- Rinella, M.E.; Tacke, F.; Sanyal, A.J.; Anstee, Q.M. Report on the AASLD/EASL Joint Workshop on Clinical Trial Endpoints in NAFLD. Hepatology 2019, 70, 1424–1436. [Google Scholar] [CrossRef]
- Marchisello, S.; Di Pino, A.; Scicali, R.; Urbano, F.; Piro, S.; Purrello, F.; Rabuazzo, A.M. Pathophysiological, Molecular and Therapeutic Issues of Nonalcoholic Fatty Liver Disease: An Overview. Int. J. Mol. Sci. 2019, 20, 1948. [Google Scholar] [CrossRef]
- Rhee, E.J. Nonalcoholic Fatty Liver Disease and Diabetes: An Epidemiological Perspective. Endocrinol. Metab. 2019, 34, 226–233. [Google Scholar] [CrossRef]
- Stahl, E.P.; Dhindsa, D.S.; Lee, S.K.; Sandesara, P.B.; Chalasani, N.P.; Sperling, L.S. Nonalcoholic Fatty Liver Disease and the Heart: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2019, 73, 948–963. [Google Scholar] [CrossRef]
- Kasper, P.; Martin, A.; Lang, S.; Kütting, F.; Goeser, T.; Demir, M.; Steffen, H.M. NAFLD and cardiovascular diseases: A clinical review. Clin. Res. Cardiol. 2021, 110, 921–937. [Google Scholar] [CrossRef]
- Hamaguchi, M.; Hashimoto, Y.; Obora, A.; Kojima, T.; Fukui, M. Non-alcoholic fatty liver disease with obesity as an independent predictor for incident gastric and colorectal cancer: A population-based longitudinal study. BMJ Open Gastroenterol. 2019, 6, e000295. [Google Scholar] [CrossRef] [PubMed]
- Fernando, D.H.; Forbes, J.M.; Angus, P.W.; Herath, C.B. Development and Progression of Non-Alcoholic Fatty Liver Disease: The Role of Advanced Glycation End Products. Int. J. Mol. Sci. 2019, 20, 5037. [Google Scholar] [CrossRef] [PubMed]
- Fougerat, A.; Montagner, A.; Loiseau, N.; Guillou, H.; Wahli, W. Peroxisome Proliferator-Activated Receptors and Their Novel Ligands as Candidates for the Treatment of Non-Alcoholic Fatty Liver Disease. Cells 2020, 9, 1638. [Google Scholar] [CrossRef] [PubMed]
- Delli Bovi, A.P.; Marciano, F.; Mandato, C.; Siano, M.A.; Savoia, M.; Vajro, P. Oxidative Stress in Non-alcoholic Fatty Liver Disease. An Updated Mini Review. Front. Med. 2021, 8, 595371. [Google Scholar] [CrossRef] [PubMed]
- Di Ciaula, A.; Passarella, S.; Shanmugam, H.; Noviello, M.; Bonfrate, L.; Wang, D.Q.; Portincasa, P. Nonalcoholic Fatty Liver Disease (NAFLD). Mitochondria as Players and Targets of Therapies? Int. J. Mol. Sci. 2021, 22, 5375. [Google Scholar] [CrossRef]
- Fujii, H.; Kawada, N.; Japan Study Group Of Nafld Jsg-Nafld. The Role of Insulin Resistance and Diabetes in Nonalcoholic Fatty Liver Disease. Int. J. Mol. Sci. 2020, 21, 3863. [Google Scholar] [CrossRef]
- Chopra, B.; Dhingra, A.K. Natural products: A lead for drug discovery and development. Phytother. Res. 2021, 35, 4660–4702. [Google Scholar] [CrossRef]
- Cheng, C.; Zhuo, S.; Zhang, B.; Zhao, X.; Liu, Y.; Liao, C.; Quan, J.; Li, Z.; Bode, A.M.; Cao, Y.; et al. Treatment implications of natural compounds targeting lipid metabolism in nonalcoholic fatty liver disease, obesity and cancer. Int. J. Biol. Sci. 2019, 15, 1654–1663. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Huang, M.; Lu, J.J.; Huang, M.Q.; Bao, J.L.; Chen, X.P.; Wang, Y.T. Terpenoids: Natural products for cancer therapy. Expert Opin. Investig. Drugs 2012, 21, 1801–1818. [Google Scholar] [CrossRef]
- Lai Shi Min, S.; Liew, S.Y.; Chear, N.J.Y.; Goh, B.H.; Tan, W.N.; Khaw, K.Y. Plant Terpenoids as the Promising Source of Cholinesterase Inhibitors for Anti-AD Therapy. Biology 2022, 11, 307. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhu, Y.; Lu, S.; Hu, C.; Zhong, W.; Chai, Y. Beneficial effects of paeoniflorin on osteoporosis induced by high-carbohydrate, high-fat diet-associated hyperlipidemia in vivo. Biochem. Biophys. Res. Commun. 2018, 498, 981–987. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liang, Y.; Liu, H.; Huang, Y.; Li, H.; Chen, B. Paeoniflorin attenuates gestational diabetes via Akt/mTOR pathway in a rat model. Food. Nutr. Res. 2020, 64. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, B.; Yu, B. Paeoniflorin Protects against Nonalcoholic Fatty Liver Disease Induced by a High-Fat Diet in Mice. Biol. Pharm. Bull. 2015, 38, 1005–1011. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.; Chu, L.; Liu, H.; Wang, W.; Li, J.; Yao, W.; Yi, J.; Gao, Y. Beneficial effects of paeoniflorin on non-alcoholic fatty liver disease induced by high-fat diet in rats. Sci. Rep. 2017, 7, 44819. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Qiao, J.Y.; Wang, B.Y.; Bai, M.; Shen, J.D.; Cheng, Y.X. Paeoniflorin Ameliorates Fructose-Induced Insulin Resistance and Hepatic Steatosis by Activating LKB1/AMPK and AKT Pathways. Nutrients 2018, 10, 1024. [Google Scholar] [CrossRef]
- Ma, Z.; Chu, L.; Liu, H.; Li, J.; Zhang, Y.; Liu, W.; Dai, J.; Yi, J.; Gao, Y. Paeoniflorin alleviates non-alcoholic steatohepatitis in rats: Involvement with the ROCK/NF-κB pathway. Int. Immunopharmacol. 2016, 38, 377–384. [Google Scholar] [CrossRef]
- Chen, D.; Guo, J.; Li, L. Catalpol promotes mitochondrial biogenesis in chondrocytes. Arch. Physiol. Biochem. 2022, 128, 802–808. [Google Scholar] [CrossRef]
- Tian, X.; Ru, Q.; Xiong, Q.; Wen, R.; Chen, Y. Catalpol Attenuates Hepatic Steatosis by Regulating Lipid Metabolism via AMP-Activated Protein Kinase Activation. Biomed. Res. Int. 2020, 2020, 6708061. [Google Scholar] [CrossRef]
- Herzig, S.; Shaw, R.J. AMPK: Guardian of metabolism and mitochondrial homeostasis. Nat. Rev. Mol. Cell Biol. 2018, 19, 121–135. [Google Scholar] [CrossRef]
- Ren, H.; Wang, D.; Zhang, L.; Kang, X.; Li, Y.; Zhou, X.; Yuan, G. Catalpol induces autophagy and attenuates liver steatosis in ob/ob and high-fat diet-induced obese mice. Aging 2019, 11, 9461–9477. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, C.; Lu, J.; Jin, Y.; Xu, C.; Meng, Q.; Liu, Q.; Dong, D.; Ma, X.; Liu, K.; et al. Targeting of miR-96-5p by catalpol ameliorates oxidative stress and hepatic steatosis in LDLr-/- mice via p66shc/cytochrome C cascade. Aging 2020, 12, 2049–2069. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Xiong, Q.; Chen, L.; Wen, J.R.; Ru, Q. Intervention of Catalpol on High-fat Diet Induced Nonalcoholic Fatty Liver Disease in Mice. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 2019, 41, 746–755. (In Chinese) [Google Scholar] [CrossRef]
- Wen, M.; Liu, Y.; Chen, R.; He, P.; Wu, F.; Li, R.; Lin, Y. Geniposide suppresses liver injury in a mouse model of DDC-induced sclerosing cholangitis. Phytother. Res. 2021, 35, 3799–3811. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.; Feng, H.; Cheng, J.; Li, Z.; Jin, M.; Zhao, L.; Wang, Q.; Qin, H.; Liu, G. Geniposide alleviates non-alcohol fatty liver disease via regulating Nrf2/AMPK/mTOR signalling pathways. J. Cell Mol. Med. 2020, 24, 5097–5108. [Google Scholar] [CrossRef]
- Ma, T.; Huang, C.; Zong, G.; Zha, D.; Meng, X.; Li, J.; Tang, W. Hepatoprotective effects of geniposide in a rat model of nonalcoholic steatohepatitis. J. Pharm. Pharmacol. 2011, 63, 587–593. [Google Scholar] [CrossRef]
- Fan, X.; Lin, L.; Cui, B.; Zhao, T.; Mao, L.; Song, Y.; Wang, X.; Feng, H.; Qingxiang, Y.; Zhang, J.; et al. Therapeutic potential of genipin in various acute liver injury, fulminant hepatitis, NAFLD and other non-cancer liver diseases: More friend than foe. Pharmacol. Res. 2020, 159, 104945. [Google Scholar] [CrossRef]
- Zhong, H.; Chen, K.; Feng, M.; Shao, W.; Wu, J.; Chen, K.; Liang, T.; Liu, C. Genipin alleviates high-fat diet-induced hyperlipidemia and hepatic lipid accumulation in mice via miR-142a-5p/SREBP-1c axis. FEBS J. 2018, 285, 501–517. [Google Scholar] [CrossRef]
- Zhong, H.; Liu, M.; Ji, Y.; Ma, M.; Chen, K.; Liang, T.; Liu, C. Genipin Reverses HFD-Induced Liver Damage and Inhibits UCP2-Mediated Pyroptosis in Mice. Cell Physiol. Biochem. 2018, 49, 1885–1897. [Google Scholar] [CrossRef]
- Wang, R.; Dong, Z.; Lan, X.; Liao, Z.; Chen, M. Sweroside Alleviated LPS-Induced Inflammation via SIRT1 Mediating NF-κB and FOXO1 Signaling Pathways in RAW264.7 Cells. Molecules 2019, 24, 872. [Google Scholar] [CrossRef]
- Mirza, A.Z.; Althagafi, I.I.; Shamshad, H. Role of PPAR receptor in different diseases and their ligands: Physiological importance and clinical implications. Eur. J. Med. Chem. 2019, 166, 502–513. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Shu, F.; Gong, J.; Ding, P.; Cheng, R.; Li, J.; Tong, R.; Ding, L.; Sun, H.; Huang, W.; et al. Sweroside ameliorates NAFLD in high-fat diet induced obese mice through the regulation of lipid metabolism and inflammatory response. J. Ethnopharmacol. 2020, 255, 112556. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Jang, J.H.; Kim, S.W.; Han, S.H.; Ma, K.H.; Jang, J.K.; Kang, H.C.; Cho, Y.Y.; Lee, H.S.; Lee, J.Y. Sweroside Prevents Non-Alcoholic Steatohepatitis by Suppressing Activation of the NLRP3 Inflammasome. Int. J. Mol. Sci. 2020, 21, 2790. [Google Scholar] [CrossRef] [PubMed]
- Jaishree, V.; Narsimha, S. Swertiamarin and quercetin combination ameliorates hyperglycemia, hyperlipidemia and oxidative stress in streptozotocin-induced type 2 diabetes mellitus in wistar rats. Biomed. Pharmacother. 2020, 130, 110561. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Li, D.; Zhu, Y.; Cai, S.; Liang, X.; Tang, Y.; Jin, S.; Ding, C. Swertiamarin supplementation prevents obesity-related chronic inflammation and insulin resistance in mice fed a high-fat diet. Adipocyte 2021, 10, 160–173. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Guo, F.; Ouyang, D. A review of the pharmacology and toxicology of aucubin. Fitoterapia 2020, 140, 104443. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.; Zhao, C.; Wang, Y.; Peng, Y.; Cheng, J.; Li, Z.; Wu, L.; Jin, M.; Feng, H. Aucubin inhibited lipid accumulation and oxidative stress via Nrf2/HO-1 and AMPK signalling pathways. J. Cell. Mol. Med. 2019, 23, 4063–4075. [Google Scholar] [CrossRef]
- Chang, Y.; Tian, Y.; Zhou, D.; Yang, L.; Liu, T.M.; Liu, Z.G.; Wang, S.W. Gentiopicroside ameliorates ethanol-induced gastritis via regulating MMP-10 and pERK1/2 signaling. Int. Immunopharmacol. 2021, 90, 107213. [Google Scholar] [CrossRef]
- Jin, M.; Feng, H.; Wang, Y.; Yan, S.; Shen, B.; Li, Z.; Qin, H.; Wang, Q.; Li, J.; Liu, G. Gentiopicroside Ameliorates Oxidative Stress and Lipid Accumulation through Nuclear Factor Erythroid 2-Related Factor 2 Activation. Oxid. Med. Cell Longev. 2020, 2020, 2940746. [Google Scholar] [CrossRef]
- Chen, J.; Fan, X.; Zhou, L.; Gao, X. Treatment with geraniol ameliorates methionine-choline-deficient diet-induced non-alcoholic steatohepatitis in rats. J. Gastroenterol. Hepatol. 2016, 31, 1357–1365. [Google Scholar] [CrossRef]
- Hashem, S.; Nisar, S.; Sageena, G.; Macha, M.A.; Yadav, S.K.; Krishnankutty, R.; Uddin, S.; Haris, M.; Bhat, A.A. Therapeutic Effects of Curcumol in Several Diseases; An Overview. Nutr. Cancer 2021, 73, 181–195. [Google Scholar] [CrossRef] [PubMed]
- Qi, X.; Song, A.; Ma, M.; Wang, P.; Zhang, X.; Lu, C.; Zhang, J.; Zheng, S.; Jin, H. Curcumol inhibits ferritinophagy to restrain hepatocyte senescence through YAP/NCOA4 in non-alcoholic fatty liver disease. Cell Prolif. 2021, 54, e13107. [Google Scholar] [CrossRef] [PubMed]
- Qi, S.Y.; Huang, H.; Li, Y.K.; Pei, L.G.; Zhang, W.J. Effects of curcumol on liver function and fibrosis in rats of nonalcoholic fatty liver disease and its mechanism. Zhongguo Ying Yong Sheng Li Xue Za Zhi 2021, 37, 611–615. (In Chinese) [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Luo, H.; Li, M.; Wu, J.; Wu, X.; Chen, L.; Gan, Y.; Guan, F.; Li, M.; Su, Z.; et al. β-patchoulene improves lipid metabolism to alleviate non-alcoholic fatty liver disease via activating AMPK signaling pathway. Biomed. Pharmacother. 2021, 134, 111104. [Google Scholar] [CrossRef]
- Luo, H.; Xu, N.; Wu, J.; Gan, Y.; Chen, L.; Guan, F.; Li, M.; Li, Y.; Chen, J.; Su, Z.; et al. β-patchoulene protects against non-alcoholic steatohepatitis via interrupting the vicious circle among oxidative stress, histanoxia and lipid accumulation in rats. Int. Immunopharmacol. 2021, 98, 107915. [Google Scholar] [CrossRef]
- Kamikubo, R.; Kai, K.; Tsuji-Naito, K.; Akagawa, M. β-Caryophyllene attenuates palmitate-induced lipid accumulation through AMPK signaling by activating CB2 receptor in human HepG2 hepatocytes. Mol. Nutr. Food. Res. 2016, 60, 2228–2242. [Google Scholar] [CrossRef]
- Zi, J.; Mafu, S.; Peters, R.J. To gibberellins and beyond! Surveying the evolution of (di)terpenoid metabolism. Annu. Rev. Plant. Biol. 2014, 65, 259–286. [Google Scholar] [CrossRef]
- Lou, T.Y.; Ma, B.B.; Liang, Y.Y.; Wang, C.X.; Liu, J.H.; Li, R.J.; Yu, S.Y.; Wang, Z.B. Analysis of carnosic acid metabolites in rats by UHPLC-Q-Exactive M.S. Zhongguo Zhong Yao Za Zhi 2020, 45, 3952–3960. (In Chinese) [Google Scholar] [CrossRef]
- Birtić, S.; Dussort, P.; Pierre, F.X.; Bily, A.C.; Roller, M. Carnosic acid. Phytochemistry 2015, 115, 9–19. [Google Scholar] [CrossRef]
- Wang, T.; Takikawa, Y.; Satoh, T.; Yoshioka, Y.; Kosaka, K.; Tatemichi, Y.; Suzuki, K. Carnosic acid prevents obesity and hepatic steatosis in ob/ob mice. Hepatol. Res. 2011, 41, 87–92. [Google Scholar] [CrossRef]
- Park, M.Y.; Sung, M.K. Carnosic acid attenuates obesity-induced glucose intolerance and hepatic fat accumulation by modulating genes of lipid metabolism in C57BL/6J-ob/ob mice. J. Sci. Food. Agric. 2015, 95, 828–835. [Google Scholar] [CrossRef] [PubMed]
- Dickmann, L.J.; VandenBrink, B.M.; Lin, Y.S. In vitro hepatotoxicity and cytochrome P450 induction and inhibition characteristics of carnosic acid, a dietary supplement with antiadipogenic properties. Drug. Metab. Dispos. 2012, 40, 1263–1267. [Google Scholar] [CrossRef] [PubMed]
- Song, H.M.; Li, X.; Liu, Y.Y.; Lu, W.P.; Cui, Z.H.; Zhou, L.; Yao, D.; Zhang, H.M. Carnosic acid protects mice from high-fat diet-induced NAFLD by regulating MARCKS. Int. J. Mol. Med. 2018, 42, 193–207. [Google Scholar] [CrossRef] [PubMed]
- Shan, W.; Gao, L.; Zeng, W.; Hu, Y.; Wang, G.; Li, M.; Zhou, J.; Ma, X.; Tian, X.; Yao, J. Activation of the SIRT1/p66shc antiapoptosis pathway via carnosic acid-induced inhibition of miR-34a protects rats against nonalcoholic fatty liver disease. Cell Death. Dis. 2015, 6, e1833. [Google Scholar] [CrossRef]
- Lombardi, R.; Fargion, S.; Fracanzani, A.L. Brain involvement in non-alcoholic fatty liver disease (NAFLD): A systematic review. Dig. Liver Dis. 2019, 51, 1214–1222. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Zhou, J.; Zhu, J.; Zhang, S.; Zhang, N.; Zhao, Y.; Ding, C.; Shi, X.; Yao, J. Carnosic acid protects non-alcoholic fatty liver-induced dopaminergic neuron injury in rats. Metab. Brain Dis. 2017, 32, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Du, J.F.; Li, P.; Lu, X. Advance in biosynthesis and metabolic regulation of ginkgolides. Zhongguo Zhong Yao Za Zhi 2021, 46, 3288–3297. (In Chinese) [Google Scholar] [CrossRef]
- Sarkar, C.; Quispe, C.; Jamaddar, S.; Hossain, R.; Ray, P.; Mondal, M.; Abdulwanis Mohamed, Z.; Sani Jaafaru, M.; Salehi, B.; Islam, M.T.; et al. Therapeutic promises of ginkgolide A: A literature-based review. Biomed. Pharmacother. 2020, 132, 110908. [Google Scholar] [CrossRef]
- Jeong, H.S.; Kim, K.H.; Lee, I.S.; Park, J.Y.; Kim, Y.; Kim, K.S.; Jang, H.J. Ginkgolide A ameliorates non-alcoholic fatty liver diseases on high fat diet mice. Biomed. Pharmacother. 2017, 88, 625–634. [Google Scholar] [CrossRef]
- Luo, L.; Yin, L.; Dongshan, W.; Zhao, Y.; Wang, Y.; Li, F.; Fang, J.; Chen, H.; Fan, S.; Huang, C. Ginkgolide B lowers body weight and ameliorates hepatic steatosis in high-fat diet-induced obese mice correlated with pregnane X receptor activation. RSC Advances 2017, 7, 37858–37866. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, J.; Gao, Q.; Shan, X.; Wang, J.; Lv, Z. Study on the attenuated effect of Ginkgolide B on ferroptosis in high fat diet induced nonalcoholic fatty liver disease. Toxicology 2020, 445, 152599. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Han, X.; Yao, Y.L.; Li, Y.M.; Zhang, J.; Shao, D.Y.; Hou, L.S.; Fan, Y.; Song, S.Z.; Lian, L.H.; et al. Acanthoic acid suppresses lipin1/2 via TLR4 and IRAK4 signalling pathways in EtOH- and lipopolysaccharide-induced hepatic lipogenesis. J. Pharm. Pharmacol. 2018, 70, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.L.; Jiang, Y.Z.; Jin, X.J.; Lian, L.H.; Piao, J.Y.; Wan, Y.; Jin, H.R.; Joon Lee, J.; Nan, J.X. Acanthoic acid, a diterpene in Acanthopanax koreanum, protects acetaminophen-induced hepatic toxicity in mice. Phytomedicine 2010, 17, 475–479. [Google Scholar] [CrossRef] [PubMed]
- Nan, J.X.; Jin, X.J.; Lian, L.H.; Cai, X.F.; Jiang, Y.Z.; Jin, H.R.; Lee, J.J. A diterpenoid acanthoic acid from Acanthopanax koreanum protects against D-galactosamine/lipopolysaccharide-induced fulminant hepatic failure in mice. Biol. Pharm. Bull. 2008, 31, 738–742. [Google Scholar] [CrossRef]
- Han, X.; Cui, Z.Y.; Song, J.; Piao, H.Q.; Lian, L.H.; Hou, L.S.; Wang, G.; Zheng, S.; Dong, X.X.; Nan, J.X.; et al. Acanthoic acid modulates lipogenesis in nonalcoholic fatty liver disease via FXR/LXRs-dependent manner. Chem. Biol. Interact. 2019, 311, 108794. [Google Scholar] [CrossRef]
- Kim, E.; Kang, Y.G.; Kim, Y.J.; Lee, T.R.; Yoo, B.C.; Jo, M.; Kim, J.H.; Kim, J.H.; Kim, D.; Cho, J.Y. Dehydroabietic Acid Suppresses Inflammatory Response Via Suppression of Src-, Syk-, and TAK1-Mediated Pathways. Int. J. Mol. Sci. 2019, 20, 1593. [Google Scholar] [CrossRef]
- Gao, G.; Xie, Z.; Li, E.W.; Yuan, Y.; Fu, Y.; Wang, P.; Zhang, X.; Qiao, Y.; Xu, J.; Hölscher, C.; et al. Dehydroabietic acid improves nonalcoholic fatty liver disease through activating the Keap1/Nrf2-ARE signaling pathway to reduce ferroptosis. J. Nat. Med. 2021, 75, 540–552. [Google Scholar] [CrossRef]
- Abboud, R.; Charcosset, C.; Greige-Gerges, H. Interaction of triterpenoids with human serum albumin: A review. Chem. Phys. Lipids 2017, 207, 260–270. [Google Scholar] [CrossRef]
- Hou, M.; Wang, R.; Zhao, S.; Wang, Z. Ginsenosides in Panax genus and their biosynthesis. Acta Pharm. Sin. B. 2021, 11, 1813–1834. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, S.; Li, J.; Zhao, J.; Xiao, Q.; Zhu, Y.; Zhang, J.; Huang, W. Effect and mechanism of ginsenoside Rg1-regulating hepatic steatosis in HepG2 cells induced by free fatty acid. Biosci. Biotechnol. Biochem. 2020, 84, 2228–2240. [Google Scholar] [CrossRef]
- Qi, R.; Jiang, R.; Xiao, H.; Wang, Z.; He, S.; Wang, L.; Wang, Y. Ginsenoside Rg1 protects against d-galactose induced fatty liver disease in a mouse model via FOXO1 transcriptional factor. Life Sci. 2020, 254, 117776. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Yang, C.; Zhang, S.; Li, J.; Xiao, Q.; Huang, W. Ginsenoside Rg1 Protects against Non-alcoholic Fatty Liver Disease by Ameliorating Lipid Peroxidation, Endoplasmic Reticulum Stress, and Inflammasome Activation. Biol. Pharm. Bull. 2018, 41, 1638–1644. [Google Scholar] [CrossRef] [PubMed]
- Gu, D.; Yi, H.; Jiang, K.; Fakhar, S.H.; Shi, J.; He, Y.; Liu, B.; Guo, Y.; Fan, X.; Li, S. Transcriptome analysis reveals the efficacy of ginsenoside-Rg1 in the treatment of nonalcoholic fatty liver disease. Life Sci. 2021, 267, 118986. [Google Scholar] [CrossRef]
- Wang, F.; Park, J.S.; Ma, Y.; Ma, H.; Lee, Y.J.; Lee, G.R.; Yoo, H.S.; Hong, J.T.; Roh, Y.S. Ginseng Saponin Enriched in Rh1 and Rg2 Ameliorates Nonalcoholic Fatty Liver Disease by Inhibiting Inflammasome Activation. Nutrients 2021, 13, 856. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.; Gao, W.; Wu, X.; Zheng, M.; Yu, Y.; Song, C.; Miao, W.; Yang, Z.; He, Y.; Liu, C.; et al. Ginsenoside Rg2 Ameliorates High-Fat Diet-Induced Metabolic Disease through SIRT1. J. Agric. Food. Chem. 2020, 68, 4215–4226. [Google Scholar] [CrossRef]
- Chen, X.J.; Liu, W.J.; Wen, M.L.; Liang, H.; Wu, S.M.; Zhu, Y.Z.; Zhao, J.Y.; Dong, X.Q.; Li, M.G.; Bian, L.; et al. Ameliorative effects of Compound K and ginsenoside Rh1 on non-alcoholic fatty liver disease in rats. Sci. Rep. 2017, 7, 41144. [Google Scholar] [CrossRef]
- Song, B.; Sun, Y.; Chu, Y.; Wang, J.; Zheng, H.; Liu, L.; Cai, W.; Zhang, H. Ginsenoside Rb1 Alleviated High-Fat-Diet-Induced Hepatocytic Apoptosis via Peroxisome Proliferator-Activated Receptor γ. Biomed. Res. Int. 2020, 2020, 2315230. [Google Scholar] [CrossRef]
- Huang, Q.; Wang, T.; Yang, L.; Wang, H.Y. Ginsenoside Rb2 Alleviates Hepatic Lipid Accumulation by Restoring Autophagy via Induction of Sirt1 and Activation of AMPK. Int. J. Mol. Sci. 2017, 18, 1063. [Google Scholar] [CrossRef]
- Roh, E.; Hwang, H.J.; Kim, J.W.; Hong, S.H.; Kim, J.A.; Lee, Y.B.; Choi, K.M.; Baik, S.H.; Yoo, H.J. Ginsenoside Mc1 improves liver steatosis and insulin resistance by attenuating ER stress. J. Ethnopharmacol. 2020, 259, 112927. [Google Scholar] [CrossRef]
- Jinhua, W. Ursolic acid: Pharmacokinetics process in vitro and in vivo, a mini review. Arch. Pharm. 2019, 352, e1800222. [Google Scholar] [CrossRef]
- Li, S.; Liao, X.; Meng, F.; Wang, Y.; Sun, Z.; Guo, F.; Li, X.; Meng, M.; Li, Y.; Sun, C. Therapeutic role of ursolic acid on ameliorating hepatic steatosis and improving metabolic disorders in high-fat diet-induced non-alcoholic fatty liver disease rats. PLoS ONE 2014, 9, e86724, Erratum in PLoS ONE 2014, 9, e92364. [Google Scholar] [CrossRef] [PubMed]
- Li, J.S.; Wang, W.J.; Sun, Y.; Zhang, Y.H.; Zheng, L. Ursolic acid inhibits the development of nonalcoholic fatty liver disease by attenuating endoplasmic reticulum stress. Food Funct. 2015, 6, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.N.; Wang, C.C.N.; Chang, H.Y.; Chu, F.Y.; Hsu, Y.A.; Cheng, W.K.; Ma, W.C.; Chen, C.J.; Wan, L.; Lim, Y.P. Ursolic Acid, a Novel Liver X Receptor α (LXRα) Antagonist Inhibiting Ligand-Induced Nonalcoholic Fatty Liver and Drug-Induced Lipogenesis. J. Agric. Food. Chem. 2018, 66, 11647–11662. [Google Scholar] [CrossRef] [PubMed]
- Mukonowenzou, N.C.; Dangarembizi, R.; Chivandi, E.; Nkomozepi, P.; Erlwanger, K.H. Administration of ursolic acid to new-born pups prevents dietary fructose-induced non-alcoholic fatty liver disease in Sprague Dawley rats. J. Dev. Orig. Health Dis. 2021, 12, 101–112. [Google Scholar] [CrossRef]
- Gu, M.; Zhao, P.; Zhang, S.; Fan, S.; Yang, L.; Tong, Q.; Ji, G.; Huang, C. Betulinic acid alleviates endoplasmic reticulum stress-mediated nonalcoholic fatty liver disease through activation of farnesoid X receptors in mice. Br. J. Pharmacol. 2019, 176, 847–863. [Google Scholar] [CrossRef]
- Quan, H.Y.; Kim, D.Y.; Kim, S.J.; Jo, H.K.; Kim, G.W.; Chung, S.H. Betulinic acid alleviates non-alcoholic fatty liver by inhibiting SREBP1 activity via the AMPK-mTOR-SREBP signaling pathway. Biochem. Pharmacol. 2013, 85, 1330–1340. [Google Scholar] [CrossRef]
- Mu, Q.; Wang, H.; Tong, L.; Fang, Q.; Xiang, M.; Han, L.; Jin, L.; Yang, J.; Qian, Z.; Ning, G.; et al. Betulinic acid improves nonalcoholic fatty liver disease through YY1/FAS signaling pathway. FASEB J. 2020, 34, 13033–13048. [Google Scholar] [CrossRef]
- Wang, C.; Duan, X.; Sun, X.; Liu, Z.; Sun, P.; Yang, X.; Sun, H.; Liu, K.; Meng, Q. Protective effects of glycyrrhizic acid from edible botanical glycyrrhiza glabra against non-alcoholic steatohepatitis in mice. Food Funct. 2016, 7, 3716–3723. [Google Scholar] [CrossRef]
- Sun, X.; Duan, X.; Wang, C.; Liu, Z.; Sun, P.; Huo, X.; Ma, X.; Sun, H.; Liu, K.; Meng, Q. Protective effects of glycyrrhizic acid against non-alcoholic fatty liver disease in mice. Eur. J. Pharmacol. 2017, 806, 75–82, Erratum in Eur. J. Pharmacol.2022, 928, 175106. [Google Scholar] [CrossRef]
- Gillard, J.; Clerbaux, L.A.; Nachit, M.; Sempoux, C.; Staels, B.; Bindels, L.B.; Tailleux, A.; Leclercq, I.A. Bile acids contribute to the development of non-alcoholic steatohepatitis in mice. JHEP Rep. 2021, 4, 100387. [Google Scholar] [CrossRef]
- Yan, T.; Wang, H.; Cao, L.; Wang, Q.; Takahashi, S.; Yagai, T.; Li, G.; Krausz, K.W.; Wang, G.; Gonzalez, F.J.; et al. Glycyrrhizin Alleviates Nonalcoholic Steatohepatitis via Modulating Bile Acids and Meta-Inflammation. Drug Metab. Dispos. 2018, 46, 1310–1319. [Google Scholar] [CrossRef] [PubMed]
- Chen, G. The link between Hepatic Vitamin A Metabolism and Nonalcoholic Fatty Liver Disease. Curr. Drug Targets 2015, 16, 1281–1292. [Google Scholar] [CrossRef]
- Shi, L.; Guo, S.; Zhang, S.; Gao, X.; Liu, A.; Wang, Q.; Zhang, T.; Zhang, Y.; Wen, A. Glycyrrhetinic acid attenuates disturbed vitamin a metabolism in non-alcoholic fatty liver disease through AKR1B10. Eur. J. Pharmacol. 2020, 883, 173167. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Zhang, M.; Liu, Q.; Xu, T.; Huang, T.; Yao, D.; Wong, C.W.; Liu, J.; Guan, M. 18β-Glycyrrhetinic acid acts through hepatocyte nuclear factor 4 alpha to modulate lipid and carbohydrate metabolism. Pharmacol. Res. 2020, 157, 104840. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Zhang, L.; Gurley, E.; Studer, E.; Shang, J.; Wang, T.; Wang, C.; Yan, M.; Jiang, Z.; Hylemon, P.B.; et al. Prevention of free fatty acid-induced hepatic lipotoxicity by 18beta-glycyrrhetinic acid through lysosomal and mitochondrial pathways. Hepatology 2008, 47, 1905–1915. [Google Scholar] [CrossRef] [PubMed]
- Pollier, J.; Goossens, A. Oleanolic acid. Phytochemistry 2012, 77, 10–15. [Google Scholar] [CrossRef]
- Xue, C.; Li, Y.; Lv, H.; Zhang, L.; Bi, C.; Dong, N.; Shan, A.; Wang, J. Oleanolic Acid Targets the Gut-Liver Axis to Alleviate Metabolic Disorders and Hepatic Steatosis. J. Agric. Food Chem. 2021, 69, 7884–7897. [Google Scholar] [CrossRef]
- Lin, Y.N.; Chang, H.Y.; Wang, C.C.N.; Chu, F.Y.; Shen, H.Y.; Chen, C.J.; Lim, Y.P. Oleanolic Acid Inhibits Liver X Receptor Alpha and Pregnane X Receptor to Attenuate Ligand-Induced Lipogenesis. J. Agric. Food Chem. 2018, 66, 10964–10976. [Google Scholar] [CrossRef]
- Gamede, M.; Mabuza, L.; Ngubane, P.; Khathi, A. Plant-derived oleanolic acid ameliorates markers associated with non-alcoholic fatty liver disease in a diet-induced pre-diabetes rat model. Diabetes Metab. Syndr. Obes. 2019, 12, 1953–1962. [Google Scholar] [CrossRef]
- Ou-Yang, Q.; Xuan, C.X.; Wang, X.; Luo, H.Q.; Liu, J.E.; Wang, L.L.; Li, T.T.; Chen, Y.P.; Liu, J. 3-Acetyl-oleanolic acid ameliorates non-alcoholic fatty liver disease in high fat diet-treated rats by activating AMPK-related pathways. Acta Pharmacol. Sin. 2018, 39, 1284–1293. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.; Shen, Q.; Wu, J.; Li, J.X. Oleanolic acid derivative HA-20 inhibits adipogenesis in a manner involving PPARγ-FABP4/aP2 pathway. J. Mol. Endocrinol. 2021, 66, 245–258. [Google Scholar] [CrossRef] [PubMed]
- You, L.Z.; Lin, Y.X.; Fang, Z.H.; Shen, G.M.; Zhao, J.D.; Wang, T.T. Research advances on astragaloside-IV in treatment of diabetes mellitus and its complications pharmacological effects. Zhongguo Zhong Yao Za Zhi 2017, 42, 4700–4706. (In Chinese) [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Zhou, D.L.; Wei, X.H.; Zhong, R.Y.; Xu, J.; Sun, L. Astragaloside I.V attenuates free fatty acid-induced ER stress and lipid accumulation in hepatocytes via AMPK activation. Acta Pharmacol. Sin. 2017, 38, 998–1008. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Wang, L.L.; Tang, W.J.; Tang, B. Astragaloside I.V inhibits protein tyrosine phosphatase 1B and improves insulin resistance in insulin-resistant HepG2 cells and triglyceride accumulation in oleic acid (OA)-treated HepG2 cells. J. Ethnopharmacol. 2021, 268, 113556. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Dai, L.; Liu, Y.; Dou, D.; Sun, Y.; Ma, L. Pharmacological activities of mogrosides. Future Med. Chem. 2018, 10, 845–850. [Google Scholar] [CrossRef]
- Zhang, X.; Song, Y.; Ding, Y.; Wang, W.; Liao, L.; Zhong, J.; Sun, P.; Lei, F.; Zhang, Y.; Xie, W. Effects of Mogrosides on High-Fat-Diet-Induced Obesity and Nonalcoholic Fatty Liver Disease in Mice. Molecules 2018, 23, 1894. [Google Scholar] [CrossRef]
- Li, L.; Zheng, W.; Wang, C.; Qi, J.; Li, H. Mogroside V Protects against Hepatic Steatosis in Mice on a High-Fat Diet and LO2 Cells Treated with Free Fatty Acids via AMPK Activation. Evid. Based Complement. Alternat. Med. 2020, 2020, 7826874. [Google Scholar] [CrossRef]
- Lv, J.; Sharma, A.; Zhang, T.; Wu, Y.; Ding, X. Pharmacological Review on Asiatic Acid and Its Derivatives: A Potential Compound. SLAS Technol. 2018, 23, 111–127. [Google Scholar] [CrossRef]
- Wang, D.; Lao, L.; Pang, X.; Qiao, Q.; Pang, L.; Feng, Z.; Bai, F.; Sun, X.; Lin, X.; Wei, J. Asiatic acid from Potentilla chinensis alleviates non-alcoholic fatty liver by regulating endoplasmic reticulum stress and lipid metabolism. Int. Immunopharmacol. 2018, 65, 256–267, Erratum in Int. Immunopharmacol.2020, 84, 106291. [Google Scholar] [CrossRef]
- Zhao, J.; Zhou, H.; An, Y.; Shen, K.; Yu, L. Biological effects of corosolic acid as an anti-inflammatory, anti-metabolic syndrome and anti-neoplasic natural compound. Oncol. Lett. 2021, 21, 84. [Google Scholar] [CrossRef]
- Liu, G.; Cui, Z.; Gao, X.; Liu, H.; Wang, L.; Gong, J.; Wang, A.; Zhang, J.; Ma, Q.; Huang, Y.; et al. Corosolic acid ameliorates non-alcoholic steatohepatitis induced by high-fat diet and carbon tetrachloride by regulating TGF-β1/Smad2, N.F-κB, and AMPK signaling pathways. Phytother. Res. 2021, 35, 5214–5226. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, J.; Sil, P.C. Arjunolic acid: A new multifunctional therapeutic promise of alternative medicine. Biochimie 2013, 95, 1098–1109. [Google Scholar] [CrossRef] [PubMed]
- Toppo, E.; Sylvester Darvin, S.; Esakkimuthu, S.; Buvanesvaragurunathan, K.; Ajeesh Krishna, T.P.; Antony Caesar, S.; Stalin, A.; Balakrishna, K.; Pandikumar, P.; Ignacimuthu, S.; et al. Curative effect of arjunolic acid from Terminalia arjuna in non-alcoholic fatty liver disease models. Biomed. Pharmacother. 2018, 107, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Xu, C.; Yang, R.; Zhang, G. Ganoderic Acid A Alleviates OVA-Induced Asthma in Mice. Inflammation 2021, 44, 1908–1915. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Shi, K.; Dong, J.; Jin, Z.; Wu, Y.; Cai, Y.; Lin, T.; Cai, Q.; Liu, L.; Zhang, Y. Ganoderic acid A attenuates high-fat-diet-induced liver injury in rats by regulating the lipid oxidation and liver inflammation. Arch. Pharm. Res. 2020, 43, 744–754. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.W.; Zhu, Y.C.; Zhang, L.; Li, P.; Yang, J.; Wen, X.D. Ilexgenin A enhances the effects of simvastatin on non-alcoholic fatty liver disease without changes in simvastatin pharmacokinetics. Chin. J. Nat. Med. 2018, 16, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Roy, G.; Guan, S.; Liu, H.; Zhang, L. Rotundic Acid Induces DNA Damage and Cell Death in Hepatocellular Carcinoma Through AKT/mTOR and MAPK Pathways. Front. Oncol. 2019, 9, 545. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.J.; Cao, S.T.; Wen, B.Y.; Han, X.; Li, Y.; Li, S.; Li, J.; Zhang, L. Rotundic acid ameliorates non-alcoholic steatohepatitis via SREBP-1c/ SCD1 signaling pathway and modulating gut microbiota. Int. Immunopharmacol. 2021, 99, 108065. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Li, J.; Sun, Z.B.; Sun, C.; Yu, Z.H.; Guo, X. Saikosaponin D inhibits proliferation of human osteosarcoma cells via the p53 signaling pathway. Exp. Ther. Med. 2019, 17, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Chang, G.R.; Lin, W.L.; Lin, T.C.; Liao, H.J.; Lu, Y.W. The Ameliorative Effects of Saikosaponin in Thioacetamide-Induced Liver Injury and Non-Alcoholic Fatty Liver Disease in Mice. Int. J. Mol. Sci. 2021, 22, 11383. [Google Scholar] [CrossRef]
- Avila, C. Terpenoids in Marine Heterobranch Molluscs. Mar. Drugs 2020, 18, 162. [Google Scholar] [CrossRef] [PubMed]
- Petyaev, I.M. Lycopene Deficiency in Ageing and Cardiovascular Disease. Oxid. Med. Cell Longev. 2016, 2016, 3218605. [Google Scholar] [CrossRef] [PubMed]
- Khan, U.M.; Sevindik, M.; Zarrabi, A.; Nami, M.; Ozdemir, B.; Kaplan, D.N.; Selamoglu, Z.; Hasan, M.; Kumar, M.; Alshehri, M.M.; et al. Lycopene: Food Sources, Biological Activities, and Human Health Benefits. Oxid. Med. Cell Longev. 2021, 2021, 2713511. [Google Scholar] [CrossRef]
- Tan, H.L.; Moran, N.E.; Cichon, M.J.; Riedl, K.M.; Schwartz, S.J.; Erdman, J.W., Jr.; Pearl, D.K.; Thomas-Ahner, J.M.; Clinton, S.K. β-Carotene-9’,10’-oxygenase status modulates the impact of dietary tomato and lycopene on hepatic nuclear receptor-, stress-, and metabolism-related gene expression in mice. J. Nutr. 2014, 144, 431–439, Erratum in J. Nutr.2014, 144, 1664. [Google Scholar] [CrossRef] [PubMed]
- Ip, B.C.; Liu, C.; Lichtenstein, A.H.; Von Lintig, J.; Wang, X.D. Lycopene and apo-10’-lycopenoic acid have differential mechanisms of protection against hepatic steatosis in β-carotene-9’,10’-oxygenase knockout male mice. J. Nutr. 2015, 145, 268–276. [Google Scholar] [CrossRef]
- Ni, Y.; Zhuge, F.; Nagashimada, M.; Nagata, N.; Xu, L.; Yamamoto, S.; Fuke, N.; Ushida, Y.; Suganuma, H.; Kaneko, S.; et al. Lycopene prevents the progression of lipotoxicity-induced nonalcoholic steatohepatitis by decreasing oxidative stress in mice. Free Radic. Biol. Med. 2020, 152, 571–582. [Google Scholar] [CrossRef]
- Jiang, W.; Guo, M.H.; Hai, X. Hepatoprotective and antioxidant effects of lycopene on non-alcoholic fatty liver disease in rat. World J. Gastroenterol. 2016, 22, 10180–10188. [Google Scholar] [CrossRef]
- Ahn, J.; Lee, H.; Jung, C.H.; Ha, T. Lycopene inhibits hepatic steatosis via microRNA-21-induced downregulation of fatty acid-binding protein 7 in mice fed a high-fat diet. Mol. Nutr. Food Res. 2012, 56, 1665–1674. [Google Scholar] [CrossRef]
- Chen, G.; Ni, Y.; Nagata, N.; Zhuge, F.; Xu, L.; Nagashimada, M.; Yamamoto, S.; Ushida, Y.; Fuke, N.; Suganuma, H.; et al. Lycopene Alleviates Obesity-Induced Inflammation and Insulin Resistance by Regulating M1/M2 Status of Macrophages. Mol. Nutr. Food Res. 2019, 63, e1900602. [Google Scholar] [CrossRef]
- Mustra Rakic, J.; Liu, C.; Veeramachaneni, S.; Wu, D.; Paul, L.; Ausman, L.M.; Wang, X.D. Dietary lycopene attenuates cigarette smoke-promoted nonalcoholic steatohepatitis by preventing suppression of antioxidant enzymes in ferrets. J. Nutr. Biochem. 2021, 91, 108596. [Google Scholar] [CrossRef]
- Li, Z.; Chen, J.; Zhang, D. Association between dietary carotenoid intakes and hypertension in adults: National Health and Nutrition Examination Survey 2007–2014. J. Hypertens. 2019, 37, 2371–2379. [Google Scholar] [CrossRef] [PubMed]
- Jha, P.; Kumari, S.; Jobby, R.; Desai, N.; Ali, A. Dietary Phytonutrients in the Prevention of Diabetes-related Complications. Curr. Diabetes Rev. 2020, 16, 657–673. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Fan, J.; Wang, J.; Li, Y.; Xiao, L.; Duan, D.; Wang, Q. Protective effect of lycopene on high-fat diet-induced cognitive impairment in rats. Neurosci. Lett. 2016, 627, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Liu, R.; Shen, Z.; Cai, G. Combination of luteolin and lycopene effectively protect against the “two-hit” in NAFLD through Sirt1/AMPK signal pathway. Life Sci. 2020, 256, 117990. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Tang, X.; Luo, Y.; Hu, X.; Ren, L. Accumulation and conversion of β-carotene and astaxanthin induced by abiotic stresses in Schizochytrium sp. Bioprocess Biosyst. Eng. 2022, 45, 911–920. [Google Scholar] [CrossRef]
- Li, J.; Guo, C.; Wu, J. Astaxanthin in Liver Health and Disease: A Potential Therapeutic Agent. Drug Des. Dev. Ther. 2020, 14, 2275–2285. [Google Scholar] [CrossRef]
- Kobori, M.; Takahashi, Y.; Sakurai, M.; Ni, Y.; Chen, G.; Nagashimada, M.; Kaneko, S.; Ota, T. Hepatic Transcriptome Profiles of Mice with Diet-Induced Nonalcoholic Steatohepatitis Treated with Astaxanthin and Vitamin E. Int. J. Mol. Sci. 2017, 18, 593. [Google Scholar] [CrossRef]
- Yang, Y.; Pham, T.X.; Wegner, C.J.; Kim, B.; Ku, C.S.; Park, Y.K.; Lee, J.Y. Astaxanthin lowers plasma TAG concentrations and increases hepatic antioxidant gene expression in diet-induced obesity mice. Br. J. Nutr. 2014, 112, 1797–1804. [Google Scholar] [CrossRef]
- Yang, Y.; Kim, B.; Park, Y.K.; Koo, S.I.; Lee, J.Y. Astaxanthin prevents TGFβ1-induced pro-fibrogenic gene expression by inhibiting Smad3 activation in hepatic stellate cells. Biochim. Biophys. Acta 2015, 1850, 178–185. [Google Scholar] [CrossRef]
- Yang, M.; Kimchi, E.T.; Staveley-O’Carroll, K.F.; Li, G. Astaxanthin Prevents Diet-Induced NASH Progression by Shaping Intrahepatic Immunity. Int. J. Mol. Sci. 2021, 22, 11037. [Google Scholar] [CrossRef]
- Ni, Y.; Nagashimada, M.; Zhuge, F.; Zhan, L.; Nagata, N.; Tsutsui, A.; Nakanuma, Y.; Kaneko, S.; Ota, T. Astaxanthin prevents and reverses diet-induced insulin resistance and steatohepatitis in mice: A comparison with vitamin E. Sci. Rep. 2015, 5, 17192. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Mo, W.; Feng, J.; Li, J.; Yu, Q.; Li, S.; Zhang, J.; Chen, K.; Ji, J.; Dai, W.; et al. Astaxanthin attenuates hepatic damage and mitochondrial dysfunction in non-alcoholic fatty liver disease by up-regulating the FGF21/PGC-1βpathway. Br. J. Pharmacol. 2020, 177, 3760–3777. [Google Scholar] [CrossRef] [PubMed]
- Nishino, A.; Maoka, T.; Yasui, H. Preventive Effects of β-Cryptoxanthin, a Potent Antioxidant and Provitamin A Carotenoid, on Lifestyle-Related Diseases-A Central Focus on Its Effects on Non-Alcoholic Fatty Liver Disease (NAFLD). Antioxidants 2021, 11, 43. [Google Scholar] [CrossRef] [PubMed]
- Haidari, F.; Hojhabrimanesh, A.; Helli, B.; Seyedian, S.S.; Ahmadi-Angali, K.; Abiri, B. A hypocaloric high-protein diet supplemented with β-cryptoxanthin improves non-alcoholic fatty liver disease: A randomized controlled trial. BMC Gastroenterol. 2020, 20, 349. [Google Scholar] [CrossRef] [PubMed]
- Haidari, F.; Hojhabrimanesh, A.; Helli, B.; Seyedian, S.S.; Ahmadi-Angali, K. An energy-restricted high-protein diet supplemented with β-cryptoxanthin alleviated oxidative stress and inflammation in nonalcoholic fatty liver disease: A randomized controlled trial. Nutr. Res. 2020, 73, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.; Liu, C.; Hu, K.Q.; Smith, D.E.; Wu, D.; Lamon-Fava, S.; Ausman, L.M.; Wang, X.D. Dietary β-Cryptoxanthin Inhibits High-Refined Carbohydrate Diet-Induced Fatty Liver via Differential Protective Mechanisms Depending on Carotenoid Cleavage Enzymes in Male Mice. J. Nutr. 2019, 149, 1553–1564. [Google Scholar] [CrossRef]
- Kobori, M.; Ni, Y.; Takahashi, Y.; Watanabe, N.; Sugiura, M.; Ogawa, K.; Nagashimada, M.; Kaneko, S.; Naito, S.; Ota, T. β-Cryptoxanthin alleviates diet-induced nonalcoholic steatohepatitis by suppressing inflammatory gene expression in mice. PLoS ONE 2014, 9, e98294. [Google Scholar] [CrossRef]
- Ni, Y.; Nagashimada, M.; Zhan, L.; Nagata, N.; Kobori, M.; Sugiura, M.; Ogawa, K.; Kaneko, S.; Ota, T. Prevention and reversal of lipotoxicity-induced hepatic insulin resistance and steatohepatitis in mice by an antioxidant carotenoid, β-cryptoxanthin. Endocrinology 2015, 156, 987–999. [Google Scholar] [CrossRef]
- Yilmaz, B.; Sahin, K.; Bilen, H.; Bahcecioglu, I.H.; Bilir, B.; Ashraf, S.; Halazun, K.J.; Kucuk, O. Carotenoids and non-alcoholic fatty liver disease. Hepatobiliary Surg. Nutr. 2015, 4, 161–171. [Google Scholar] [CrossRef]
- Wang, L.; Ding, C.; Zeng, F.; Zhu, H. Low Levels of Serum β-Carotene and β-Carotene/Retinol Ratio Are Associated with Histological Severity in Nonalcoholic Fatty Liver Disease Patients. Ann. Nutr. Metab. 2019, 74, 156–164. [Google Scholar] [CrossRef]
- Christensen, K.; Lawler, T.; Mares, J. Dietary Carotenoids and Non-Alcoholic Fatty Liver Disease among U.S Adults, NHANES 2003–2014. Nutrients 2019, 11, 1101. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Wang, C.; Liu, J.; Liu, Z.M.; Ling, W.H.; Chen, Y.M. Greater serum carotenoid levels associated with lower prevalence of nonalcoholic fatty liver disease in Chinese adults. Sci. Rep. 2015, 5, 12951. [Google Scholar] [CrossRef] [PubMed]
- Seif El-Din, S.H.; El-Lakkany, N.M.; El-Naggar, A.A.; Hammam, O.A.; Abd El-Latif, H.A.; Ain-Shoka, A.A.; Ebeid, F.A. Effects of rosuvastatin and/or β-carotene on non-alcoholic fatty liver in rats. Res. Pharm. Sci. 2015, 10, 275–287. [Google Scholar] [PubMed]
- Li, J.; Abdel-Aal, E.M. Dietary Lutein and Cognitive Function in Adults: A Meta-Analysis of Randomized Controlled Trials. Molecules 2021, 26, 5794. [Google Scholar] [CrossRef]
- Qiu, X.; Gao, D.H.; Xiang, X.; Xiong, Y.F.; Zhu, T.S.; Liu, L.G.; Sun, X.F.; Hao, L.P. Ameliorative effects of lutein on non-alcoholic fatty liver disease in rats. World J. Gastroenterol. 2015, 21, 8061–8072. [Google Scholar] [CrossRef]
- Chen, C.; Ni, Y.; Jiang, B.; Yan, S.; Xu, B.; Fan, B.; Huang, H.; Chen, G. Anti-aging derivatives of cycloastragenol produced by biotransformation. Nat. Prod. Res. 2021, 35, 2685–2690. [Google Scholar] [CrossRef]
- Gu, M.; Zhang, S.; Zhao, Y.; Huang, J.; Wang, Y.; Li, Y.; Fan, S.; Yang, L.; Ji, G.; Tong, Q.; et al. Cycloastragenol improves hepatic steatosis by activating farnesoid X receptor signalling. Pharmacol. Res. 2017, 121, 22–32. [Google Scholar] [CrossRef]
- Lin, C.; Wen, X.; Sun, H. Oleanolic acid derivatives for pharmaceutical use: A patent review. Expert Opin. Ther. Pat. 2016, 26, 643–655. [Google Scholar] [CrossRef]
- Zhang, B.Z.; Ding, F.; Tan, L.W. Clinical and experimental study on yi-gan-ning granule in treating chronic hepatitis B. Zhongguo Zhong Xi Yi Jie He Za Zhi 1993, 13, 597–599, 580. (In Chinese) [Google Scholar]
- Kandiah, N.; Ong, P.A.; Yuda, T.; Ng, L.L.; Mamun, K.; Merchant, R.A.; Chen, C.; Dominguez, J.; Marasigan, S.; Ampil, E.; et al. Treatment of dementia and mild cognitive impairment with or without cerebrovascular disease: Expert consensus on the use of Ginkgo biloba extract, EGb 761®. CNS Neurosci. Ther. 2019, 25, 288–298. [Google Scholar] [CrossRef]
- Patel, P.; Aknouk, M.; Dawson, A.; Aya, A.; Kanukuntla, A.; Kata, P.; De Dona, A. How Much Is Too Much? Exploring Pseudohyperaldosteronism in Glycyrrhizic Acid Toxicity From Chronic Licorice Root Consumption. Cureus 2021, 13, e16454. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.S.; Bril, F.; Cusi, K.; Newsome, P.N. Modulation of Insulin Resistance in Nonalcoholic Fatty Liver Disease. Hepatology 2019, 70, 711–724. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Lin, S.; Wan, B.; Velani, B.; Zhu, Y. Pyroptosis in Liver Disease: New Insights into Disease Mechanisms. Aging Dis. 2019, 10, 1094–1108. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Jin, Z.; Bandyopadhyay, G.; Wang, G.; Zhang, D.; Rocha, K.C.E.; Liu, X.; Zhao, H.; Kisseleva, T.; Brenner, D.A.; et al. Aberrant iron distribution via hepatocyte-stellate cell axis drives liver lipogenesis and fibrosis. Cell Metab. 2022, 34, 1201–1213.e5. [Google Scholar] [CrossRef] [PubMed]
- Carling, D. AMPK signalling in health and disease. Curr. Opin. Cell Biol. 2017, 45, 31–37. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Ru, X.; Wen, T. NRF2, a Transcription Factor for Stress Response and Beyond. Int. J. Mol. Sci. 2020, 21, 4777. [Google Scholar] [CrossRef]
- Ding, R.B.; Bao, J.; Deng, C.X. Emerging roles of SIRT1 in fatty liver diseases. Int. J. Biol. Sci. 2017, 13, 852–867. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, P.; Liu, Y. Terpenoids: Natural Compounds for Non-Alcoholic Fatty Liver Disease (NAFLD) Therapy. Molecules 2023, 28, 272. https://doi.org/10.3390/molecules28010272
Yao P, Liu Y. Terpenoids: Natural Compounds for Non-Alcoholic Fatty Liver Disease (NAFLD) Therapy. Molecules. 2023; 28(1):272. https://doi.org/10.3390/molecules28010272
Chicago/Turabian StyleYao, Pengyu, and Yajuan Liu. 2023. "Terpenoids: Natural Compounds for Non-Alcoholic Fatty Liver Disease (NAFLD) Therapy" Molecules 28, no. 1: 272. https://doi.org/10.3390/molecules28010272
APA StyleYao, P., & Liu, Y. (2023). Terpenoids: Natural Compounds for Non-Alcoholic Fatty Liver Disease (NAFLD) Therapy. Molecules, 28(1), 272. https://doi.org/10.3390/molecules28010272





