Therapeutic Potential of Flavonoids and Tannins in Management of Oral Infectious Diseases—A Review
Abstract
1. Introduction
1.1. Oral Cavity Pathogens
1.2. Natural Products in Oral Health
2. Results and Discussion
2.1. Flavonoids in Oral Health
2.2. Tannins in Oral Health
2.3. Bioaccessebility of Tannis and Flavonoids
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CEP | Chilean propolis |
| CHX | Chlorhexidine |
| COX | Cyclooxygenase |
| EC | Epicatechin |
| ECG | Epicatechin gallate |
| EGCG | Epigallocatechin gallate |
| EMA | European Medicines Agency |
| EPS | Exopolysaccharide |
| F-ATPase | F-Type ATPase |
| GTF | Glucosyltransferase |
| HPLC/MS | High-performance liquid chromatography/mass spectrometry |
| IC50 | Half maximal inhibitory concentration |
| IgG | Immunoglobulin G |
| IL | Interleukin |
| LOX | Lipoxygenase |
| MBC | Minimum bactericidal concentration |
| MBIC50 | Half maximum biofilm inhibition concentration |
| MIC | Minimum inhibitory concentration |
| NF-κB | Nuclear factor kappa B |
| PACs | Proanthocyanidins |
| PGE | Pomegranate peel glycolic extract |
| sHA | Saliva-coated hydroxyapatite |
| SrtA | Sortase A |
| TNF-α | Tumor necrosis factor alpha |
| VAS | Visual analogue scale |
References
- WHO. Oral Health. Available online: https://www.who.int/news-room/fact-sheets/detail/oral-health (accessed on 3 October 2022).
- Arweiler, N.B.; Netuschil, L. The Oral Microbiota. Microbiota Hum. Body 2016, 45–60. [Google Scholar] [CrossRef]
- Bennett, J.; Dolin, R.; Blaser, M.J. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases, 9th ed.; Elsevier: Amsterdam, The Netherlands, 2019. [Google Scholar]
- Peng, X.; Cheng, L.; You, Y.; Tang, C.; Ren, B.; Li, Y.; Xu, X.; Zhou, X. Oral Microbiota in Human Systematic Diseases. Int. J. Oral Sci. 2022, 14, 14. [Google Scholar] [CrossRef] [PubMed]
- Read, E.; Curtis, M.A.; Neves, J.F. Inflammatory Bowel Disease. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Cortelli, R.J.; Barbosa, M.D.S.; Westphal, M.A. Halitosis: A Review of Associated Factors and Therapeutic Approach. Braz. Oral Res. 2008, 22, 44–54. [Google Scholar] [CrossRef]
- Ogle, O.E. Odontogenic Infections. Dent. Clin. NA 2017, 61, 235–252. [Google Scholar] [CrossRef]
- Singh, U.; Tandon, T.; Sinha, D. Apical Periodontitis—Virulence Factors of Enterococcus Faecalis and Candida Albicans. Austin. J. Dent. 2020, 7, 1147. [Google Scholar]
- Patil, S.; Rao, S.R.; Majumdar, B.; Anil, S. Clinical Appearance of Oral Candida Infection and Therapeutic Strategies. Front. Microbiol. 2015, 6, 1–10. [Google Scholar] [CrossRef]
- Conrads, G.; About, I. Pathophysiology of Dental Caries. In Caries Excavation: Evolution of Treating Cavitated Carious Lesions; Schwendicke, F., Frencken, J., Eds.; Monogr Oral Sci: Basel, Karger, 2018; Volume 27, pp. 1–10. [Google Scholar] [CrossRef]
- Könönen, E.; Gursoy, M.; Gursoy, U.K. Periodontitis: A Multifaceted Disease of Tooth-Supporting Tissues. J. Clin. Med. 2019, 8, 1135. [Google Scholar] [CrossRef]
- Checchi, V.; Maravic, T.; Bellini, P.; Generali, L.; Consolo, U.; Breschi, L.; Mazzoni, A. The Role of Matrix Metalloproteinases in Periodontal Disease. Int. J. Environ. Res. Public Health 2020, 17, 4923. [Google Scholar] [CrossRef]
- Moghadam, T.E.; Yazdanian, M.; Tahmasebi, E.; Tebyanian, H.; Ranjbar, R.; Yazdanian, A.; Seifalian, A.; Tafazoli, A. Current Herbal Medicine as an Alternative Treatment in Dentistry: In Vitro, in Vivo and Clinical Studies. Eur. J. Pharmacol. 2020, 889, 173665. [Google Scholar] [CrossRef]
- Palombo, E.A. Traditional Medicinal Plant Extracts and Natural Products with Activity against Oral Bacteria: Potential Application in the Prevention and Treatment of Oral Diseases. Evid. Based Complement. Altern. Med. 2011, 2011, 15. [Google Scholar] [CrossRef] [PubMed]
- Al Alsheikh, M.H.; Sultan, I.; Kumar, V.; Rather, I.A.; Al-sheikh, H.; Jan, A.T.; Mohd, Q.; Haq, R. Plant-Based Phytochemicals as Possible Alternative to Antibiotics in Combating Bacterial Drug Resistance. Antibiotics 2020, 9, 480. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liu, Y.; Yang, X.; Li, C.; Song, Z.; Vernon, J. The Oral Microbiota: Community Composition, Influencing Factors, Pathogenesis, and Interventions. Front. Microbiol. 2022, 13, 895537. [Google Scholar] [CrossRef] [PubMed]
- Slobodníková, L.; Fialová, S.; Rendeková, K.; Kováč, J. Antibiofilm Activity of Plant Polyphenols. Molecules 2016, 21, 1717. [Google Scholar] [CrossRef] [PubMed]
- Flemming, J.; Meyer-Probst, C.T.; Speer, K.; Kölling-Speer, I.; Hannig, C.; Hannig, M. Preventive Applications of Polyphenols in Dentistry—A Review. Int. J. Mol. Sci. 2021, 22, 4892. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Mirza, M.A.; Naseef, P.P.; Kuruniyan, M.S. Exploring the Potential of Natural Product-Based Nanomedicine for Maintaining Oral Health. Molecules 2022, 27, 1725. [Google Scholar] [CrossRef] [PubMed]
- Nagy, M.; Mučaji, P.; Grančai, D. Pharmacognosy: Biologically Active Plant Metabolites and Their Sources, 2nd ed.; Osveta: Martin, Slovakia, 2017. [Google Scholar]
- Górniak, I.; Bartoszewski, R.; Króliczewski, J. Comprehensive Review of Antimicrobial Activities of Plant Flavonoids. Phytochem. Rev. 2019, 18, 241–272. [Google Scholar] [CrossRef]
- Wang, T.; Li, Q.; Bi, K. Bioactive Flavonoids in Medicinal Plants: Structure, Activity and Biological Fate. Asian J. Pharm. Sci. 2018, 13, 12–23. [Google Scholar] [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Journal of Nutritional Science. J. Nutr. Sci. 2016, 5, 1–15. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013, 2013, 16. [Google Scholar] [CrossRef]
- Fialova, S.; Rendekova, K.; Mucaji, P.; Slobodnikova, L. Plant Natural Agents: Polyphenols, Alkaloids and Essential Oils as Perspective Solution of Microbial Resistance. Curr. Org. Chem. 2017, 21, 1875–1884. [Google Scholar] [CrossRef]
- Fernández-Rojas, B.; Gutiérrez-Venegas, G. Flavonoids Exert Multiple Periodontic Benefits Including Anti-Inflammatory, Periodontal Ligament-Supporting, and Alveolar Bone-Preserving Effects. Life Sci. 2018, 209, 435–454. [Google Scholar] [CrossRef] [PubMed]
- Samy, R.P.; Gopalakrishnakone, P. Therapeutic Potential of Plants as Anti-Microbials for Drug Discovery. Evid. Based Complement. Altern. Med. 2010, 7, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Yuan, G.; Guan, Y.; Yi, H.; Lai, S.; Sun, Y.; Cao, S. Antibacterial Activity and Mechanism of Plant Flavonoids to Gram-Positive Bacteria Predicted from Their Lipophilicities. Sci. Rep. 2021, 11, 10471. [Google Scholar] [CrossRef] [PubMed]
- Bittner Fialová, S.; Rendeková, K.; Mučaji, P.; Nagy, M.; Slobodníková, L. Antibacterial Activity of Medicinal Plants and Their Constituents in the Context of Skin and Wound Infections, Considering European Legislation and Folk Medicine—A Review. Int. J. Mol. Sci. 2021, 22, 10746. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Yang, W.; Tang, F.; Chen, X.; Ren, L. Antibacterial Activities of Flavonoids: Structure-Activity Relationship and Mechanism. Curr. Med. Chem. 2014, 22, 132–149. [Google Scholar] [CrossRef]
- Duarte, S.; Gregoire, S.; Singh, A.P.; Vorsa, N.; Schaich, K.; Bowen, W.H.; Koo, H. Inhibitory Effects of Cranberry Polyphenols on Formation and Acidogenicity of Streptococcus Mutans Biofilms. FEMS Microbiol. Lett. 2006, 257, 50–56. [Google Scholar] [CrossRef]
- Cascioferro, S.; Totsika, M.; Schillaci, D. Microbial Pathogenesis Sortase A: An Ideal Target for Anti-Virulence Drug Development. Microb. Pathog. 2014, 77, 105–112. [Google Scholar] [CrossRef]
- Salmanli, M.; Tatar, G.; Tuzuner, T. Computer Methods and Programs in Biomedicine Investigation of the Antimicrobial Activities of Various Antimicrobial Agents on Streptococcus Mutans Sortase A through Computer-Aided Drug Design ( CADD ) Approaches. Comput. Methods Programs Biomed. 2021, 212, 106454. [Google Scholar] [CrossRef]
- Koo, H.; Rosalen, P.L.; Cury, J.A.; Park, Y.K.; Bowen, W.H. Effects of Compounds Found in Propolis on Streptococcus mutans Growth and on Glucosyltransferase Activity. Antimicrob. Agents Chemother. 2002, 46, 1302–1309. [Google Scholar] [CrossRef]
- Koo, H.; Hayacibara, M.F.; Schobel, B.D.; Cury, J.A.; Rosalen, P.L.; Park, Y.K.; Vacca-Smith, A.M.; Bowen, W.H. Inhibition of Streptococcus mutans Biofilm Accumulation and Polysaccharide Production by Apigenin and Tt-Farnesol. J. Antimicrob. Chemother. 2003, 52, 782–789. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Nikitkova, A.; Abdelsalam, H.; Li, J. Archives of Oral Biology Activity of Quercetin and Kaemferol against Streptococcus Mutans Bio Fi Lm. Arch. Oral Biol. 2019, 98, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Patra, J.K.; Kim, E.S.; Oh, K.; Kim, H.-J.; Kim, Y.; Baek, K.-H. Antibacterial Effect of Crude Extract and Metabolites of Phytolacca Americana on Pathogens Responsible for Periodontal Inflammatory Diseases and Dental Caries. BMC Complement. Altern. Med. 2014, 14, 343. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.-Y.; Kim, C.-K.; Ahn, C.-H.; Kim, H.; Shin, J.; Oh, K.-B. Flavonoid Glycosides Inhibit Sortase A and Sortase A-Mediated Aggregation of Streptococcus mutans, an Oral Bacterium Responsible for Human Dental Caries. J. Microbiol. Biotechnol. 2016, 26, 1566–1569. [Google Scholar] [CrossRef] [PubMed]
- Shu, Y.; Liu, Y.; Li, L.; Feng, J.; Lou, B.; Zhou, X.; Wu, H. Antibacterial Activity of Quercetin on Oral Infectious Pathogens. Afr. J. Microbiol. Res. 2011, 5, 5358–5361. [Google Scholar] [CrossRef]
- Cha, S.; Kim, G.; Cha, J. Synergistic Antimicrobial Activity of Apigenin against Oral Pathogens. Int. J. Eng. Res. Sci. 2016, 2, 27–37. [Google Scholar]
- André, C.B.; Rosalen, P.L.; de Galvão, L.C.C.; Fronza, B.M.; Ambrosano, G.M.B.; Ferracane, J.L.; Giannini, M. Modulation of Streptococcus Mutans Virulence by Dental Adhesives Containing Anti-Caries Agents. Dent. Mater. 2017, 33, 1084–1092. [Google Scholar] [CrossRef]
- Koo, H.; Schobel, B.; Scott-Anne, K.; Watson, G.; Bowen, W.H.; Cury, J.A.; Rosalen, P.L.; Park, Y.K. Apigenin and Tt-Farnesol with Fluoride Effects on S. mutans Biofilms and Dental Caries. J. Dent. Res. 2005, 84, 1016–1020. [Google Scholar] [CrossRef]
- Koo, H.; Seils, J.; Abranches, J.; Burne, R.A.; Bowen, W.H.; Quivey, R.G. Influence of Apigenin on Gtf Gene Expression in Streptococcus mutans UA159. Antimicrob. Agents Chemother. 2006, 50, 542–546. [Google Scholar] [CrossRef]
- Jeon, J.-G.; Klein, M.I.; Xiao, J.; Gregoire, S.; Rosalen, P.L.; Koo, H. Influences of Naturally Occurring Agents in Combination with Fluoride on Gene Expression and Structural Organization of Streptococcus mutans in Biofilms. BMC Microbiol. 2009, 9, 228. [Google Scholar] [CrossRef]
- Gregoire, S.; Singh, A.P.; Vorsa, N.; Koo, H. Influence of Cranberry Phenolics on Glucan Synthesis by Glucosyltransferases and Streptococcus Mutans Acidogenicity. J. Appl. Microbiol. 2007, 103, 1960–1968. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Venegas, G.; Gomez-Mora, J.A.; Meraz-Rodríguez, M.; Flores-Sanchez, M.; Ortiz-Miranda, L. Heliyon Effect of Fl Avonoids on Antimicrobial Activity of Microorganisms Present in Dental Plaque. Heliyon 2019, 5, e03013. [Google Scholar] [CrossRef] [PubMed]
- Huang, P.; Hu, P.; Yun, S. Morin Inhibits Sortase A and Subsequent Biofilm Formation in Streptococcus Mutans. Curr. Microbiol. 2014, 68, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Guan, X.; Zhou, Y.; Liang, X.; Xiao, J.; He, L.; Li, J. Effects of Compounds Found in Nidus Vespae on the Growth and Cariogenic Virulence Factors of Streptococcus Mutans. Microbiol. Res. 2012, 167, 61–68. [Google Scholar] [CrossRef] [PubMed]
- El Mihyaoui, A.; Esteves da Silva, J.C.G.; Charfi, S.; Candela Castillo, M.E.; Lamarti, A.; Arnao, M.B. Chamomile (Matricaria chamomilla L.): A Review of Ethnomedicinal Use, Phytochemistry and Pharmacological Uses. Life 2022, 12, 479. [Google Scholar] [CrossRef]
- Braga, A.S.; de Simas, L.L.M.; Pires, J.G.; Souza, B.M.; de Melo, F.P.S.R.; Saldanha, L.L.; Dokkedal, A.L.; Magalhães, A.C. Antibiofilm and Anti-Caries Effects of an Experimental Mouth Rinse Containing Matricaria chamomilla L. Extract under Microcosm Biofilm on Enamel. J. Dent. 2020, 99, 103415. [Google Scholar] [CrossRef]
- Andishe Tadbir, A.; Pourshahidi, S.; Ebrahimi, H.; Hajipour, Z.; Memarzade, M.R.; Shirazian, S. The Effect of Matricaria chamomilla (Chamomile) Extract in Orabase on Minor Aphthous Stomatitis, a Randomized Clinical Trial. J. Herb. Med. 2015, 5, 71–76. [Google Scholar] [CrossRef]
- Sychrová, A.; Škovranová, G.; Čulenová, M.; Bittner Fialová, S. Prenylated Flavonoids in Topical Infections and Wound Healing. Molecules 2022, 27, 4491. [Google Scholar] [CrossRef]
- Chen, L.; Cheng, X.; Shi, W.; Lu, Q.; Liang, V.; Heber, D.; Ma, L. Letters to the Editor Inhibition of Growth of Streptococcus mutans, Methicillin-Resistant Staphylococcus. J. Clin. Microbiol. 2005, 43, 3574–3575. [Google Scholar] [CrossRef]
- Badria, F.A.; Zidan, O.A. Natural Products for Dental Caries Prevention. J. Med. Food 2004, 7, 381–384. [Google Scholar] [CrossRef]
- Furiga, A.; Lonvaud-funel, A.; Badet, C. In Vitro Study of Antioxidant Capacity and Antibacterial Activity on Oral Anaerobes of a Grape Seed Extract. Food Chem. 2009, 113, 1037–1040. [Google Scholar] [CrossRef]
- Veloz, J.J.; Saavedra, N.; Lillo, A.; Alvear, M.; Barrientos, L.; Salazar, L.A. Antibiofilm Activity of Chilean Propolis on Streptococcus mutans Is Influenced by the Year of Collection. Biomed Res. Int. 2015, 2015, 291351. [Google Scholar] [CrossRef] [PubMed]
- Veloz, J.J.; Alvear, M.; Salazar, L.A. Antimicrobial and Antibiofilm Activity against Streptococcus mutans of Individual and Mixtures of the Main Polyphenolic Compounds Found in Chilean Propolis. Biomed Res. Int. 2019, 2019, 7602343. [Google Scholar] [CrossRef] [PubMed]
- Barrientos, L.; Herrera, C.L.; Montenegro, G.; Ortega, X.; Veloz, J.; Alvear, M.; Cuevas, A.; Saavedra, N.; Salazar, L.A. Chemical and Botanical Characterization of Chilean Propolis and Biological Activity on Cariogenic Bacteria Streptococcus mutans and Streptococcus sobrinus. Braz. J. Microbiol. 2013, 44, 577–585. [Google Scholar] [CrossRef]
- Nagy, M.; Mučaji, P.; Grančai, D. Pharmacognosy Biogenesis of Natural Substances, 1st ed.; Osveta: Martin, Slovakia, 2011. [Google Scholar]
- Engels, C.; Schieber, A.; Gänzle, M.G. Inhibitory Spectra and Modes of Antimicrobial Action of Gallotannins from Mango Kernels (Mangifera indica L.). Appl. Environ. Microbiol. 2011, 77, 2215–2223. [Google Scholar] [CrossRef]
- Farha, A.K.; Yang, Q.-Q.; Kim, G.; Li, H.-B.; Zhu, F.; Liu, H.-Y.; Gan, R.-Y.; Corke, H. Tannins as an Alternative to Antibiotics. Food Biosci. 2020, 38, 100751. [Google Scholar] [CrossRef]
- Fraga-Corral, M.; Otero, P.; Cassani, L.; Echave, J.; Garcia-Oliveira, P.; Carpena, M.; Chamorro, F.; Lourenço-Lopes, C.; Prieto, M.A.; Simal-Gandara, J. Traditional Applications of Tannin Rich Extracts Supported by Scientific Data: Chemical Composition, Bioavailability and Bioaccessibility. Foods 2021, 10, 251. [Google Scholar] [CrossRef]
- Rauf, A.; Imran, M.; Abu-Izneid, T.; Iahtisham-Ul-Haq; Patel, S.; Pan, X.; Naz, S.; Sanches Silva, A.; Saeed, F.; Rasul Suleria, H.A. Proanthocyanidins: A Comprehensive Review. Biomed. Pharmacother. 2019, 116, 108999. [Google Scholar] [CrossRef]
- Krenn, L.; Steitz, M.; Schlicht, C.; Kurth, H.; Gaedcke, F. Anthocyanin- and Proanthocyanidin-Rich Extracts of Berries in Food Supplements—Analysis with Problems. Die Pharm. Int. J. Pharm. Sci. 2007, 62, 803–812. [Google Scholar]
- Hellström, J.K.; Törrönen, A.R.; Mattila, P.H. Proanthocyanidins in Common Food Products of Plant Origin. J. Agric. Food Chem. 2009, 57, 7899–7906. [Google Scholar] [CrossRef]
- Patel, S. Rose Hip as an Underutilized Functional Food: Evidence-Based Review. Trends Food Sci. Technol. 2017, 63, 29–38. [Google Scholar] [CrossRef]
- González, O.A.; Escamilla, C.; Danaher, R.J.; Dai, J.; Ebersole, J.L.; Mumper, R.J.; Miller, C.S. Antibacterial Effects of Blackberry Extract Target Periodontopathogens. J. Periodontal Res. 2013, 48, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Riihinen, K.R.; Ou, Z.M.; Gödecke, T.; Lankin, D.C.; Pauli, G.F.; Wu, C.D. The Antibiofilm Activity of Lingonberry Flavonoids against Oral Pathogens Is a Case Connected to Residual Complexity. Fitoterapia 2014, 97, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Feng, G.; Klein, M.I.; Gregoire, S.; Singh, A.P.; Vorsa, N.; Koo, H. The Specific Degree-of-Polymerization of A-Type Proanthocyanidin Oligomers Impacts Streptococcus mutans Glucan-Mediated Adhesion and Transcriptome Responses within Biofilms. Biofouling 2013, 29, 629–640. [Google Scholar] [CrossRef]
- Philip, N.; Walsh, L.J. Cranberry Polyphenols: Natural Weapons against Dental Caries. Dent. J. 2019, 7, 20. [Google Scholar] [CrossRef]
- Koo, H.; Duarte, S.; Murata, R.M.; Scott-Anne, K.; Gregoire, S.; Watson, G.E.; Singh, A.P.; Vorsa, N. Influence of Cranberry Proanthocyanidins on Formation of Biofilms by Streptococcus mutans on Saliva-Coated Apatitic Surface and on Dental Caries Development in Vivo. Caries Res. 2010, 44, 116–126. [Google Scholar] [CrossRef]
- Yamanaka-Okada, A.; Sato, E.; Kouchi, T.; Kimizuka, R.; Kato, T.; Okuda, K. Inhibitory Effect of Cranberry Polyphenol on Cariogenic Bacteria. Bull. Tokyo Dent. Coll. 2008, 49, 107–112. [Google Scholar] [CrossRef]
- Kim, D.; Hwang, G.; Liu, Y.; Wang, Y.; Singh, A.P.; Vorsa, N.; Koo, H. Cranberry Flavonoids Modulate Cariogenic Properties of Mixed-Species Biofilm through Exopolysaccharides-Matrix Disruption. PLoS ONE 2016, 10, e0145844. [Google Scholar] [CrossRef]
- Puupponen-Pimiä, R.; Nohynek, L.; Alakomi, H.-L.; Oksman-Caldentey, K.-M. Bioactive Berry Compounds—Novel Tools against Human Pathogens. Appl. Microbiol. Biotechnol. 2005, 67, 8–18. [Google Scholar] [CrossRef]
- Satoh, Y.; Ishihara, K. Investigation of the Antimicrobial Activity of Bilberry (Vaccinium myrtillus L.) Extract against Periodontopathic Bacteria. J. Oral Biosci. 2020, 62, 169–174. [Google Scholar] [CrossRef]
- Dutreix, L.; Bernard, C.; Juin, C.; Imbert, C.; Girardot, M. Do Raspberry Extracts and Fractions Have Antifungal or Anti-Adherent Potential against Candida Spp.? Int. J. Antimicrob. Agents 2018, 52, 947–953. [Google Scholar] [CrossRef] [PubMed]
- Malheiros, J.; Simões, D.M.; Figueirinha, A.; Cotrim, M.D.; Fonseca, D.A. Agrimonia Eupatoria L.: An Integrative Perspective on Ethnomedicinal Use, Phenolic Composition and Pharmacological Activity. J. Ethnopharmacol. 2022, 296, 115498. [Google Scholar] [CrossRef] [PubMed]
- EMA. EMA. Agrimoniae Herba. Available online: https://www.ema.europa.eu/en/medicines/herbal/agrimoniae-herba. (accessed on 1 November 2022).
- Ham, Y.; Kim, T. Plant Extracts Inhibiting Biofilm Formation by Streptococcus mutans without Antibiotic Activity. J. Korean Wood Sci. Technol. 2018, 46, 692–702. [Google Scholar] [CrossRef]
- Melzig, M.F.; Böttger, S. Tormentillae Rhizoma—Review for an Underestimated European Herbal Drug. Planta Med. 2020, 86, 1050–1057. [Google Scholar] [CrossRef] [PubMed]
- Tomczyk, M.; Sosnowska, K.; Pleszczyńska, M.; Strawa, J.; Wiater, A.; Grochowski, D.M.; Tomczykowa, M.; Winnicka, K. Hydrogel Containing an Extract of Tormentillae Rhizoma for the Treatment of Biofilm-Related Oral Diseases. Nat. Prod. Commun. 2017, 12, 1934578X1701200328. [Google Scholar] [CrossRef]
- Cheesman, M.J.; Alcorn, S.; Verma, V.; Cock, I.E. Journal of Traditional and Complementary Medicine An Assessment of the Growth Inhibition pro Fi Les of Hamamelis virginiana L. Extracts against Streptococcus and Staphylococcus Spp. J. Tradit. Chinese Med. Sci. 2021, 11, 457–465. [Google Scholar] [CrossRef]
- Mouchrek Júnior, J.C.E.; de Araújo Castro Nunes, L.H.; Arruda, C.S.; Rizzi, C.; Mouchrek, A.Q.S.; Tavarez, R.; Tonetto, M.; Bandeca, M.C.; Filho, E.M.M. Effectiveness of Oral Antiseptics on Tooth Biofilm: A Study in Vivo. J. Contemp. Dent. Pract. 2015, 16, 674–678. [Google Scholar] [CrossRef]
- EMA. EMA. Quercus Cortex. Available online: https://www.ema.europa.eu/en/medicines/herbal/quercus-cortex (accessed on 2 November 2022).
- Kandra, L.; Gyémánt, G.; Zajácz, Á.; Batta, G. Inhibitory Effects of Tannin on Human Salivary α-Amylase. Biochem. Biophys. Res. Commun. 2004, 319, 1265–1271. [Google Scholar] [CrossRef]
- Basri, D.F.; Tan, L.S.; Shafiei, Z.; Zin, N.M. In Vitro Antibacterial Activity of Galls of Quercus Infectoria Olivier against Oral Pathogens. Evid. Based Complement. Altern. Med. 2012, 2012, 632796. [Google Scholar] [CrossRef]
- Limsuwan, S.; Subhadhirasakul, S.; Voravuthikunchai, S.P. Medicinal Plants with Significant Activity against Important Pathogenic Bacteria Medicinal Plants with Significant Activity against Important Pathogenic Bacteria. Pharm. Biol. 2009, 47, 683–689. [Google Scholar] [CrossRef]
- Khameneh, B.; Eskin, N.A.M.; Iranshahy, M.; Fazly Bazzaz, B.S. Phytochemicals: A Promising Weapon in the Arsenal against Antibiotic-Resistant Bacteria. Antibiotics 2021, 10, 1044. [Google Scholar] [CrossRef] [PubMed]
- Saremi, A.; Arora, R. The Cardiovascular Implications of Alcohol and Red Wine. Am. J. Ther. 2008, 15, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Monagas, M.; Bartolomé, B.; Gómez-Cordovés, C. Updated Knowledge About the Presence of Phenolic Compounds in Wine. Crit. Rev. Food Sci. Nutr. 2005, 45, 85–118. [Google Scholar] [CrossRef]
- Daglia, M.; Stauder, M.; Papetti, A.; Signoretto, C.; Giusto, G.; Canepari, P.; Pruzzo, C.; Gazzani, G. Isolation of Red Wine Components with Anti-Adhesion and Anti-Biofilm Activity against Streptococcus mutans. Food Chem. 2010, 119, 1182–1188. [Google Scholar] [CrossRef]
- Lee, J.-H.; Shim, J.S.; Chung, M.-S.; Lim, S.-T.; Kim, K.H. In Vitro Anti-Adhesive Activity of Green Tea Extract against Pathogen Adhesion. Phyther. Res. 2009, 23, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, T.; Tomofuji, T.; Endo, Y.; Irie, K.; Azuma, T.; Ekuni, D.; Tamaki, N.; Yamamoto, T.; Morita, M. Supplementation of Green Tea Catechins in Dentifrices Suppresses Gingival Oxidative Stress and Periodontal Inflammation. Arch. Oral Biol. 2011, 56, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Ferrazzano, G.F.; Roberto, L.; Amato, I.; Cantile, T.; Sangianantoni, G.; Ingenito, A. Antimicrobial Properties of Green Tea Extract Against Cariogenic Microflora: An In Vivo Study. J. Med. Food 2011, 14, 907–911. [Google Scholar] [CrossRef] [PubMed]
- Shahakbari, R.; Eshghpour, M.; Rajaei, A.; Rezaei, N.M.; Golfakhrabadi, P.; Nejat, A. Effectiveness of Green Tea Mouthwash in Comparison to Chlorhexidine Mouthwash in Patients with Acute Pericoronitis: A Randomized Clinical Trial. Int. J. Oral Maxillofac. Surg. 2014, 43, 1394–1398. [Google Scholar] [CrossRef]
- Chopra, A.; Thomas, B.S.; Sivaraman, K.; Prasad, H.K.; Kamath, S.U. Green Tea Intake as an Adjunct to Mechanical Periodontal Therapy for the Management of Mild to Moderate Chronic Periodontitis: A Randomized Controlled Clinical Trial. Oral Health Prev. Dent. 2016, 4, 293–303. [Google Scholar] [CrossRef]
- Asahi, Y.; Noiri, Y.; Miura, J.; Maezono, H.; Yamaguchi, M.; Yamamoto, R.; Azakami, H.; Hayashi, M.; Ebisu, S. Effects of the Tea Catechin Epigallocatechin Gallate on Porphyromonas Gingivalis Biofilms. J. Appl. Microbiol. 2014, 116, 1164–1171. [Google Scholar] [CrossRef]
- Sakanaka, S.; Aizawa, M.; Kim, M.; Yamamoto, T. Inhibitory Effects of Green Tea Polyphenols on Growth and Cellular Adherence of an Oral Bacterium, Porphyromonas Gingivalis. Biosci. Biotechnol. Biochem. 1996, 60, 745–749. [Google Scholar] [CrossRef] [PubMed]
- Hirasawa, M.; Takada, K.; Makimura, M.; Otake, S. Improvement of Periodontal Status by Green Tea Catechin Using a Local Delivery System: A Clinical Pilot Study. J. Periodontal Res. 2002, 37, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Zhou, X.D.; Wu, C.D. Tea Catechin Epigallocatechin Gallate Inhibits Streptococcus mutans Biofilm Formation by Suppressing Gtf Genes. Arch. Oral Biol. 2012, 57, 678–683. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, T.; Nakahara, A.; Minnatul, K.M.; Noiri, Y.; Ebisu, S.; Kato, A.; Azakami, H. The Inhibitory Effects of Catechins on Biofilm Formation by the Periodontopathogenic Bacterium, Eikenella corrodens. Biosci. Biotechnol. Biochem. 2010, 74, 2445–2450. [Google Scholar] [CrossRef] [PubMed]
- Kushiyama, M.; Shimazaki, Y.; Murakami, M.; Yamashita, Y. Relationship Between Intake of Green Tea and Periodontal Disease. J. Periodontol. 2009, 80, 372–377. [Google Scholar] [CrossRef]
- Shumi, W.; Hossain, M.A.; Park, D.-J.; Park, S. Inhibitory Effects of Green Tea Polyphenol Epigallocatechin Gallate (EGCG) on Exopolysaccharide Production by Streptococcus mutans under Microfluidic Conditions. BioChip J. 2014, 8, 179–186. [Google Scholar] [CrossRef]
- Cho, Y.-S.; Oh, J.J.; Oh, K.-H. Antimicrobial Activity and Biofilm Formation Inhibition of Green Tea Polyphenols on Human Teeth. Biotechnol. Bioprocess Eng. 2010, 15, 359–364. [Google Scholar] [CrossRef]
- Shimamura, T.; Zhao, W.-H.; Hu, Z.-Q. Mechanism of Action and Potential for Use of Tea Catechin as an Antiinfective Agent. Anti Infect. Agents Med. Chem. 2007, 6, 57–62. [Google Scholar] [CrossRef]
- Abdulbaqi, H.R.; Himratul-Aznita, W.H.; Baharuddin, N.A. Anti-Plaque Effect of a Synergistic Combination of Green Tea and Salvadora persica L. against Primary Colonizers of Dental Plaque. Arch. Oral Biol. 2016, 70, 117–124. [Google Scholar] [CrossRef]
- Kawarai, T.; Narisawa, N.; Yoneda, S.; Tsutsumi, Y.; Ishikawa, J.; Hoshino, Y.; Senpuku, H. Inhibition of Streptococcus Mutans Biofilm Formation Using Extracts from Assam Tea Compared to Green Tea. Arch. Oral Biol. 2016, 68, 73–82. [Google Scholar] [CrossRef]
- Vahid Dastjerdi, E.; Abdolazimi, Z.; Ghazanfarian, M.; Amdjadi, P.; Kamalinejad, M.; Mahboubi, A. Effect of Punica granatum L. Flower Water Extract on Five Common Oral Bacteria and Bacterial Biofilm Formation on Orthodontic Wire. Iran. J. Public Health 2014, 43, 1688–1694. [Google Scholar] [PubMed]
- Abdollahzadeh, S.; Mashouf, R.Y.; Mortazavi, H.; Moghaddam, M.H. Antibacterial and Antifungal Activities of Punica Granatum Peel Extracts Against Oral Pathogens. J. Dent. 2011, 8, 1–6. [Google Scholar]
- Rahmani, A.H.; Alsahli, M.A.; Almatroodi, S.A. Active Constituents of Pomegranates (Punica granatum) as Potential Candidates in the Management of Health through Modulation of Biological Activities. Pharmacogn. J. 2017, 9, 689–695. [Google Scholar] [CrossRef]
- Gomes, L.A.P.; Alves Figueiredo, L.M.; do Rosário Palma, A.L.; Corrêa Geraldo, B.M.; Isler Castro, K.C.; de Oliveira Fugisaki, L.R.; Jorge, A.O.C.; de Oliveira, L.D.; Junqueira, J.C. Punica granatum L. (Pomegranate) Extract: In Vivo Study of Antimicrobial Activity against Porphyromonas Gingivalis in Galleria Mellonella Model. Sci. World J. 2016, 2016, 8626987. [Google Scholar] [CrossRef]
- Widyarman, S.A.; Lay, S.H.; Wendhita, I.P.; Tjakra, E.E.; Murdono, F.I.; Binartha, C.T.O. Indonesian Mangosteen Fruit (Garcinia mangostana L.) Peel Extract Inhibits Streptococcus Mutans and Porphyromonas Gingivalis in Biofilms In Vitro. Contemp. Clin. Dent. 2019, 10, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Araghizadeh, A.; Kohanteb, J.; Fani, M.M. Inhibitory Activity of Green Tea (Camellia sinensis) Extract on Some Clinically Isolated Cariogenic and Periodontopathic Bacteria. Med. Princ. Pract. 2013, 22, 368–372. [Google Scholar] [CrossRef] [PubMed]
- Esawy, M.A.; Ragab, T.I.M.; Shalaby, A.S.G.; Basha, M.; Emam, M. Evaluated Bioactive Component Extracted from Punica Granatum Peel and Its Ag NPs Forms as Mouthwash against Dental Plaque. Biocatal. Agric. Biotechnol. 2019, 18, 101073. [Google Scholar] [CrossRef]
- Steinberg, D.; Feldman, M.; Ofek, I.; Weiss, E.I. Cranberry High Molecular Weight Constituents Promote Streptococcus sobrinus Desorption from Artificial Biofilm. Int. J. Antimicrob. Agents 2005, 25, 247–251. [Google Scholar] [CrossRef]
- Nagula, R.L.; Wairkar, S. Recent advances in topical delivery of flavonoids: A review. J. Control Release 2019, 296, 190–201. [Google Scholar] [CrossRef]
- Ayala-Fuentes, J.C.; Chavez-Santoscoy, R.A. Nanotechnology as a Key to Enhance the Benefits and Improve the Bioavailability of Flavonoids in the Food Industry. Foods 2021, 10, 2701. [Google Scholar] [CrossRef]
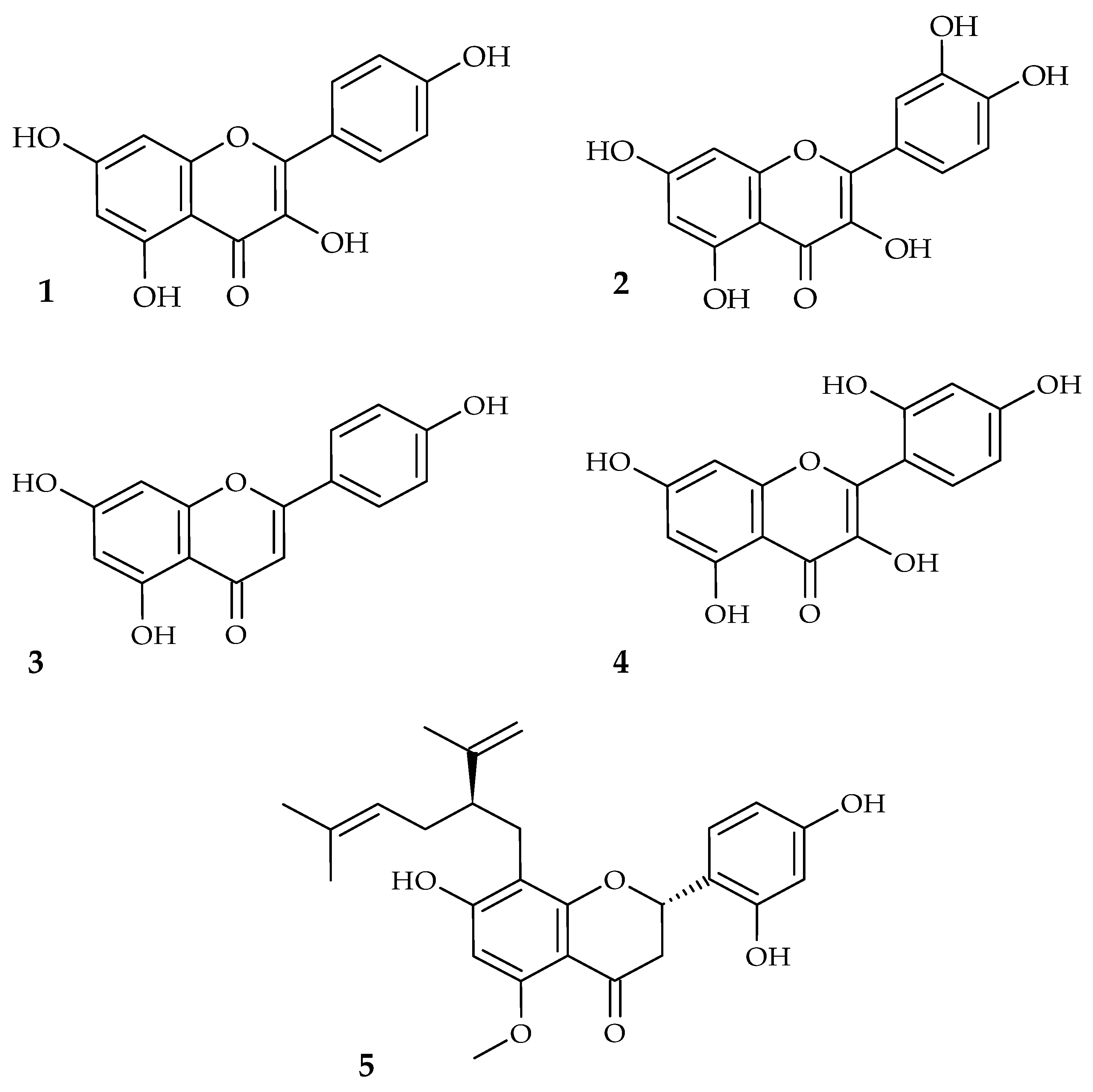
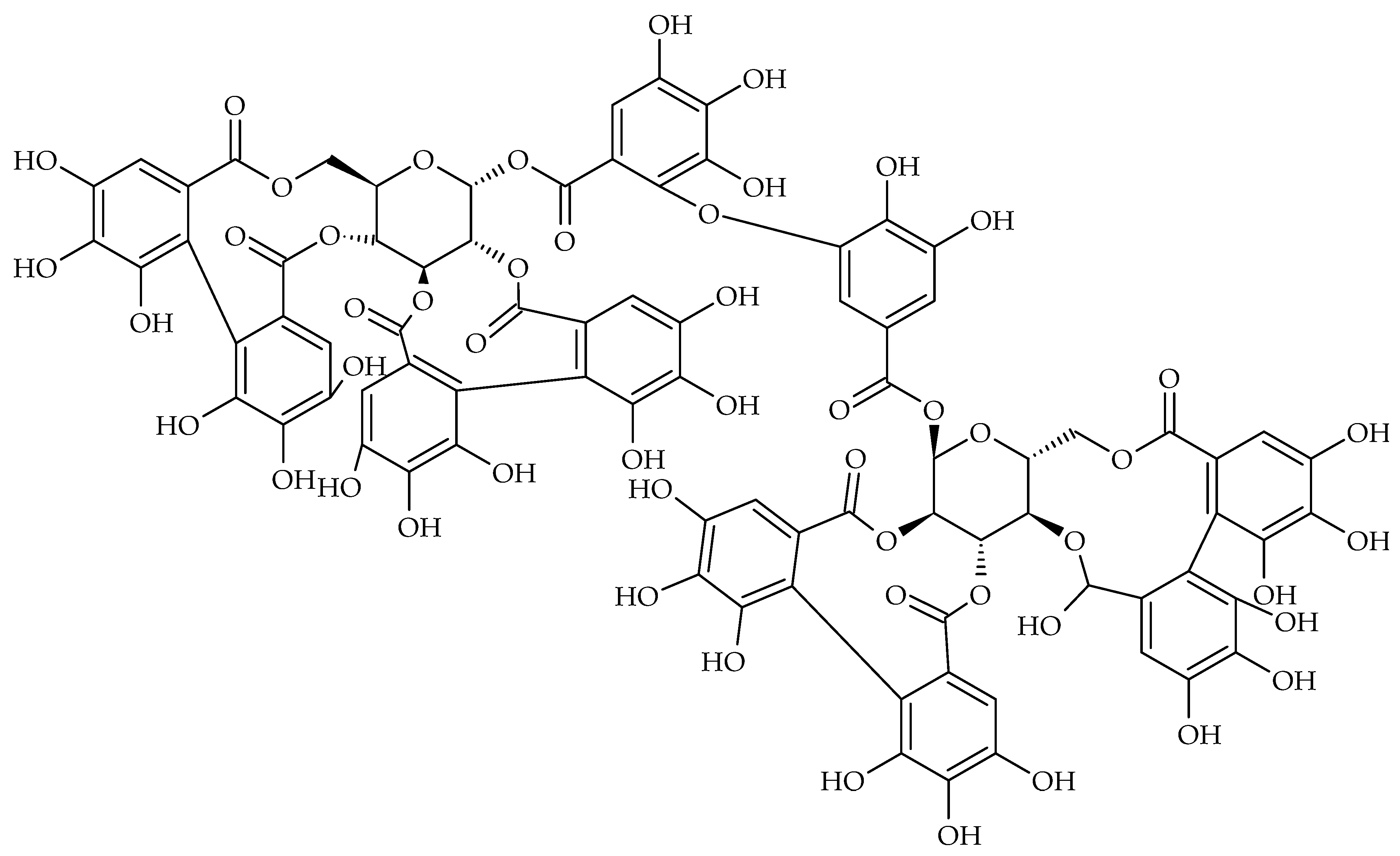
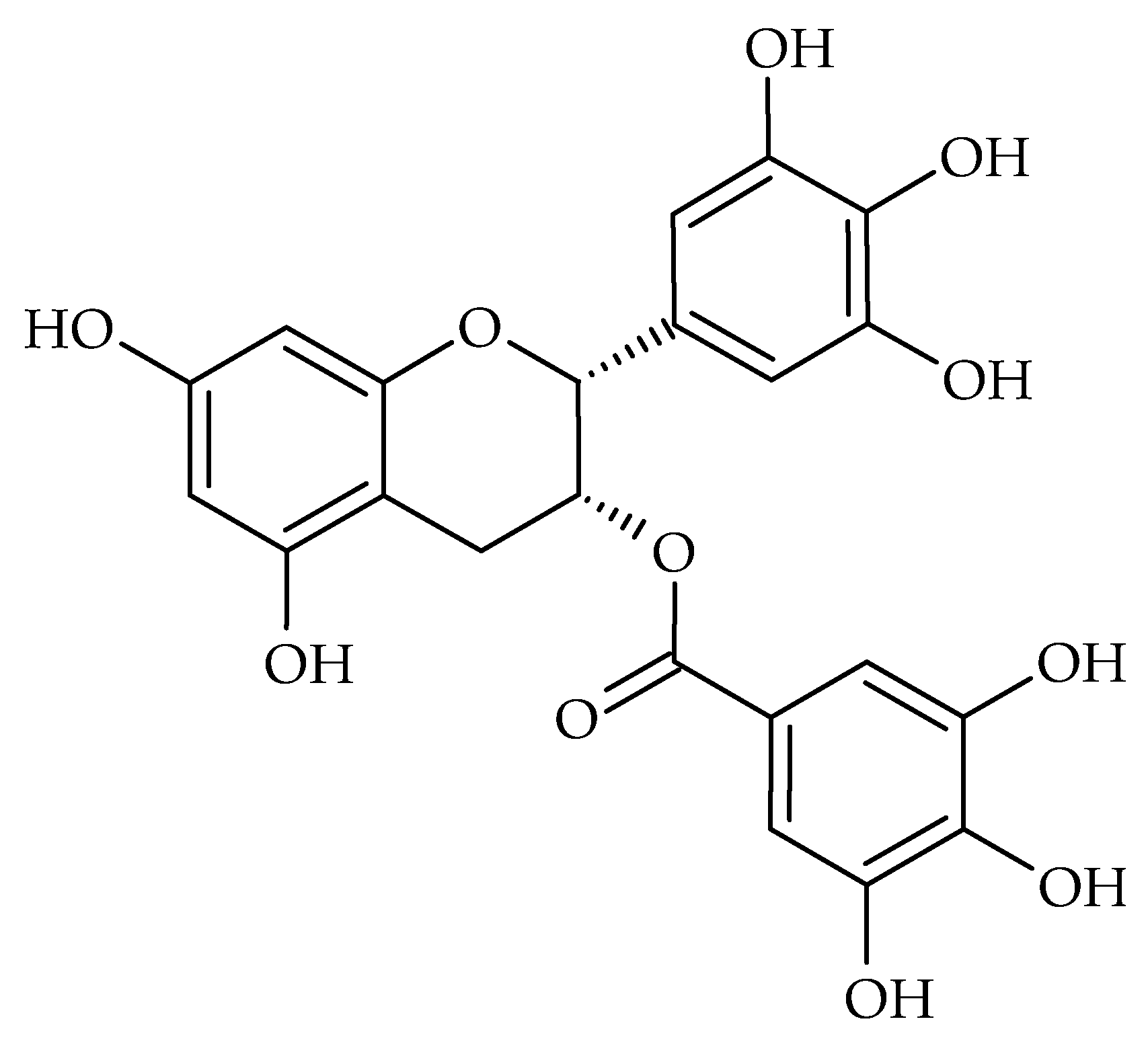
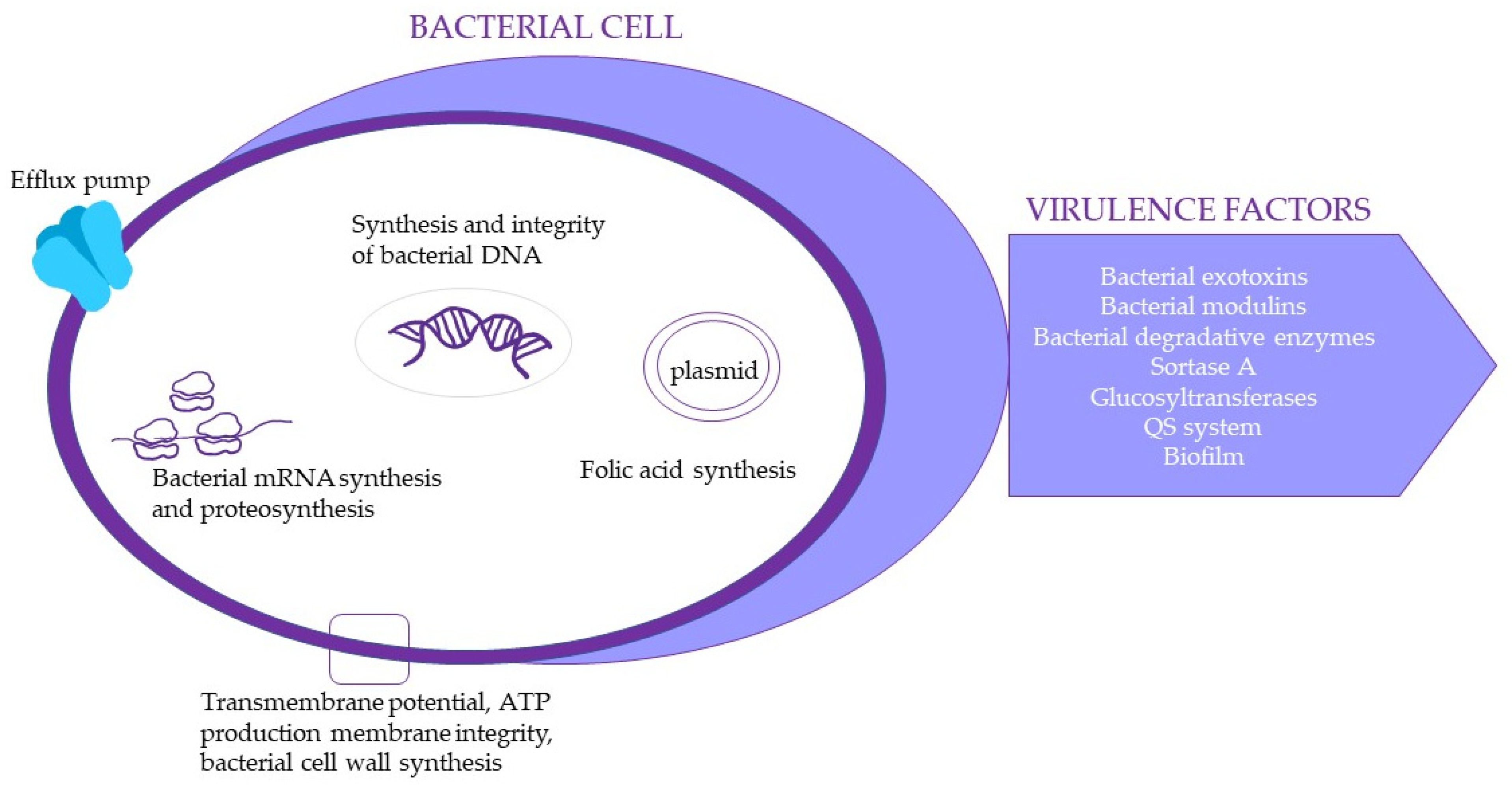
| Disease | Underlying Factors | Contributing Oral Microbial Communities | Complications |
|---|---|---|---|
| Dental caries | Xerostomia Sugars-rich diet Insufficient oral hygiene Genetic factors Immunodeficiency | Cariogenic supragingival dental plaque communities Cariogenic subgingival dental plaque communities (cervical and root caries) | Pulpitis, pulp necrosis Periapical abscess, periapical granuloma Dissemination and focal infections, dental sepsis Aesthetic defects and psychological impact |
| Chronic infection of the dental canal | Improper canal cleaning, shaping and irrigation Insufficient disinfection of the treated dental canal | Non-fastidious members of oral microbiota | Dental canal treatment failure |
| Periodontal disease | Bad oral hygiene, dental calculus Smoking Hormonal disturbances Genetic predisposition Stress Immunodeficiency | Dysbalanced subgingival dental plaque communities, esp. proteolytic anaerobic bacteria | Tooth loss Chronic low-level inflammation and systemic impact (cardiovascular diseases, Alzheimer’s disease, inflammatory bowel disease, complications during pregnancy) Dissemination and focal infections Halitosis, aesthetic defects, and psychological impact Cancrum oris, Vincent`s angina |
| Oral candidiasis | Impaired local and systemic defence mechanisms Xerostomia Dental prostheses Endocrine disorders (e.g., diabetes mellitus) Malnutrition Malignancies Damaged oral mucosa, underlying mucosal diseases Poor oral hygiene Altered or immature oral microbiota (antimicrobial therapy; neonates) Smoking | Candida spp. colonizing the oral cavity | Spread into the larynx, pharynx, or oesophagus Disseminated candidiasis |
| Disease | Pathogens | Important Virulence Factors |
|---|---|---|
| Dental caries | Streptococcus mutans, Streptococcus sobrinus, Bifidobacterium dentium, Scardovia wiggsiae, lactobacilli (Lactobacillus fermentum, L. rhamnosus, L. gasseri, L. salivarius, L. plantarum, L. casei-paracasei group) | Adhesivity, biofilm production (glucans production), acidogenicity—sugar metabolism (acid production), aciduric properties |
| Chronic infection of the dental canal | Enterococcus faecalis, Enterococcus faecium, Candida albicans, other Candida spp., coliforms Pseudomonas aeruginosa | Adhesivity, biofilm production, resistance to external factors, proteolytic and cytolytic enzymes, inflammatory potential, antimicrobial resistance, enhanced resistance to disinfectious agents |
| Periodontal disease | Aggregatibacter actinomycetemcomitans Porphyromonas gingivalis, Treponema denticola, Tannerella forsythia, Fusobacterium nucleatum | Adhesivity, biofilm production, proteolytic activity and other aggressins, invasion, inflammatory activity |
| Oral candidiasis | Candida albicans C. glabrata, C. guilliermondii, C. krusei, C. lusitaniae, C. parapsilosis, C. pseudotropicalis, C. stellatoidea, C. tropicalis | Adhesivity and biofilm production Proteolytic and lipolytic activity Invasivity Switching to filamentous forms |
| Flavonoids | Bacteria | Antibacterial/Antibiofilm Action | Reference |
|---|---|---|---|
| Quercetin Kaempferol | S. mutans | Increasing of the bacterial culture pH. Reduction of the total dry weight of the biofilm. Reduction of the cell viability. Reduction of the formation of insoluble and soluble glucans. Half maximum biofilm inhibition concentration (MBIC50 = 16 and 8 mg/mL, respectively), was comparable to chlorhexidine (CHX). Antibacterial activity in concentration 8 μg/mL. | [36,37] |
| Kaempferol | P. gingivalis | Antibacterial activity in concentration 8 μg/mL. | [37] |
| Rutin Quercetin-3′-O-methyl-3-O-α-L-rhamnopyranosyl-(1→6)-β-D-glucopyranoside Quercetin | S. mutans | Inhibition of sortase A (SrtA) with half maximum inhibition concentration (IC50) 134 μM, 186 μM and 2011 μM, respectively. | [38] |
| Quercetin | S. sobrinus, L. acidophilus, S. sanguis, A. actinomycetemocomitans and P. intermedia | Antibacterial activity in the concentration range from 1 to 4 mg/mL. | [39] |
| Apigenin | S. mutans, S. sanguinis, S. sobrinus, S. ratti, S. criceti, S. anginosus, S. gordonii, A. actinomycetemcomitans, F. nucleatum, P. intermedia, P. gingivalis | Antibacterial activity against cariogenic bacteria: minimum inhibitory concentration (MIC) 25–200 µg/mL, minimum bactericidal concentration (MBC) 100–800 µg/mL. Antibacterial activity against periodontopathogenic bacteria: MICs 100–200 µg/mL, MBCs 200–400 µg/mL. Synergistic effect in combination with antibiotics: 4-fold reduction of MICs of ampicillin or erythromycin and 4–8-fold reduction of MIC of gentamicin. | [40] |
| Apigenin | S. mutans | Reduction in the biofilm total biomass (dry weight), but without changes in bacterial viability. Inhibition of the production of extracellular glucans. Synergy: the combination with tt-farnesol and fluoride reduces the acidogenicity of biofilm. | [35,41,42] |
| Apigenin | S. sobrinus | Inhibition of glucosyltransferase (GTF) at the concentration of 1.33 mM, whether the enzyme was in solution (90–95% inhibition) or on saliva-coated hydroxyapatite (sHA) surface (35–58%). | [35] |
| Apigenin | different streptococci | Inhibition of various GTFs; the IC50 in solution were from 58 µM to 98 µM, for the surface absorbed enzymes the IC50 was higher (458 µM–1 mM). Modulation of the expression of genes that encode GTFs in S. mutans in a planktonic state or in biofilm (c = 0.1 mM to 1 mM). | [34,43] |
| Kaempferol Apigenin | S. mutans S. sobrinus | Inhibition of GTFs at the concentration of 500 µM:
| [34] |
| Pinocembrin | S. mutans | Growth inhibition; MIC ˃ 500 µM. | [34] |
| Pinocembrin | S. sobrinus | MIC = 250 µM, MBC = 500 µM. | [34] |
| Myricetin | S. mutans | Synergistic effect in combination with tt-farnesol and fluoride:
| [44] |
| Quercetin-3-arabinofuranoside Myricetin Procyanidin A2 | S. mutans S. anginosus | Inhibition of the surface-adsorbed glucosyltransferases B and C and F-ATPases at the concentration 500 µmol/L flavonoids. | [45] |
| Luteolin Morin Naringin Quercetin Rutin | A. naeslundii, A. viscosus, A. actinomycecomitans, E. faecalis, and L. casei | Growth inhibition. | [46] |
| Morin | S. mutans | SrtA inhibition (IC50 of 27.2 ± 2.6 μM). Reduction of the biofilm mass (in the concentration of 30 μM). | [47] |
| Bacteria | MIC | Mechanism | Reference |
|---|---|---|---|
| P.gingivalis | EGCG (500 μg/mL or 5 mg/mL) | At concentrations above the MIC, established biofilms were disrupted. At concentrations below the MIC, biofilm formation was inhibited. | [97] |
| P. gingivalis | MIC = 250–500 μg/mL | Green tea polyphenols, especially EGCG, completely inhibited the growth and adherence onto the buccal epithelial cells. | [98] |
| P. gingivalis Prevotella spp. | MIC of catechin = 1 mg/mL | Hydroxypropylcellulose strips containing green tea catechin as a slow-release topical delivery system were applied to the pockets of patients once a week for eight weeks. Green tea catechin showed a bactericidal effect in vitro with MIC of 1.0 mg/mL. | [99] |
| S. mutans | EGCG (7.8–31.25 μg/mL) | EGCG showed a dose-dependent inhibition. At sub-MIC concentration (15.6 μg/mL), it significantly suppressed the genes encoding GTFs. EGCG at a concentration of less than 78 μg/mL induced cellular aggregation of S. mutans. | [100] |
| Eikenella corrodens | EGCG (MIC50 = 0.1–0.25 mM) | Sub-MIC concentration inhibited biofilm formation. | [101] |
| Medicinal Plant | Extract/Fraction/Material | Microorganism | Activity | Reference(s) |
|---|---|---|---|---|
| Agrimonia eupatoria L. | methanol, water, 50% ethanol and 95% ethanol extracts | S. mutans | Antibiofilm | [79] |
| Assam tea (Camelia sinenssis var. assamica) | water extract | S. mutans | Antibiofilm | [107] |
| Chilean propolis | crude extract | S. mutans S. sobrinus, | Antibacterial Antibiofilm | [56,57,58] |
| Garcinia mangostana L. (mangosteen) | ethanol extracts | S. mutans P. gingivalis | Antibiofilm | [112] |
| Green tea (Camelia sinenssis) | water, water/ethyl acetate extract | Staphylococcus spp., Streptococcus spp., P. gingivalis, Prevotella spp. | Antimicrobial | [92,113] |
| Hamamelis virginiana L. | methanolic and water extracts | S. oralis | Antibacterial | [82] |
| Matricaria chamomilla L. | water extract | polymicrobial | Antibiofilm | [50] |
| Nidus vespae (honeycomb) | chloroform/methanol extract | S. mutans, S. sobrinus, S. sanguis, A. viscosus, A. naeslundii and L. rhamnosus | Antibacterial Antibiofilm | [31] |
| Potentilla erecta L. (rhizome) | methanol extract | S. mutans | Antibiofilm | [81] |
| propolis | isolates | S. mutans, S. sobrinus | Antibacterial Antibiofilm synergy | [34] |
| Punica granatum (peel) | crude extract methanol extract in nanoparticles water extract | Lysinibacillus cresolivorans L. boronitolerans S. mutans S. sanguinis, S. sobrinus, S. salivarius P. gingivalis | Antibacterial Biofilm inhibition | [108,109,111,114] |
| Quercus infectoria (galls) | methanol and acetone extracts | S. mutans, S. salivarius P. gingivalis F. nucleatum | Antibacterial | [86] |
| Red wine Italian | dealcoholized extract | S. mutans | Antibacterial In vitro, ex vivo biofilm inhibition | [91] |
| Rhus coriaria L. | water extract | S. sanguinis, S. sobrinus, S. salivarius, S. mutans | Antibacterial | [108] |
| Rubus idaeus (raspberry) | ethyl acetate extract | C. albicans C. glabrata C. parapsilosis | Antiadhesive | [76] |
| Salvadora persica L. (miswak) | Water | S. mitis S. sanguinis A. viscosus | Antimicrobial Synergistic anti-plaque | [106] |
| Sophora flavescens L. | water-ethanol extract | S. mutans | Antibacterial | [53] |
| Vaccinium oxycoccos L. or Vaccinium macrocarpon L. (cranberry) | flavonoid/proanthocyaidin fractions non-dialysable material derived from cranberry juice | S. mutans S. sorbinus | Antibacterial Antibiofilm Antiadhesive | [31,69,70,71,72,73,115] |
| Vaccinium vitis-idaea L | juice concentrate | F. nucleatum S. mutans | Antibacterial | [68] |
| Vitis vinifera L. (seeds) | extract | P. gingivalis F. nucleatum | Antibacterial Antibiofilm | [55] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kováč, J.; Slobodníková, L.; Trajčíková, E.; Rendeková, K.; Mučaji, P.; Sychrová, A.; Bittner Fialová, S. Therapeutic Potential of Flavonoids and Tannins in Management of Oral Infectious Diseases—A Review. Molecules 2023, 28, 158. https://doi.org/10.3390/molecules28010158
Kováč J, Slobodníková L, Trajčíková E, Rendeková K, Mučaji P, Sychrová A, Bittner Fialová S. Therapeutic Potential of Flavonoids and Tannins in Management of Oral Infectious Diseases—A Review. Molecules. 2023; 28(1):158. https://doi.org/10.3390/molecules28010158
Chicago/Turabian StyleKováč, Ján, Lívia Slobodníková, Eva Trajčíková, Katarína Rendeková, Pavel Mučaji, Alice Sychrová, and Silvia Bittner Fialová. 2023. "Therapeutic Potential of Flavonoids and Tannins in Management of Oral Infectious Diseases—A Review" Molecules 28, no. 1: 158. https://doi.org/10.3390/molecules28010158
APA StyleKováč, J., Slobodníková, L., Trajčíková, E., Rendeková, K., Mučaji, P., Sychrová, A., & Bittner Fialová, S. (2023). Therapeutic Potential of Flavonoids and Tannins in Management of Oral Infectious Diseases—A Review. Molecules, 28(1), 158. https://doi.org/10.3390/molecules28010158