Formulation and Testing of Antioxidant and Protective Effect of Hyalurosomes Loading Extract Rich in Rosmarinic Acid Biotechnologically Produced from Lavandula angustifolia Miller
Abstract
:1. Introduction
2. Results
2.1. Extract Bioproduction and Quantification
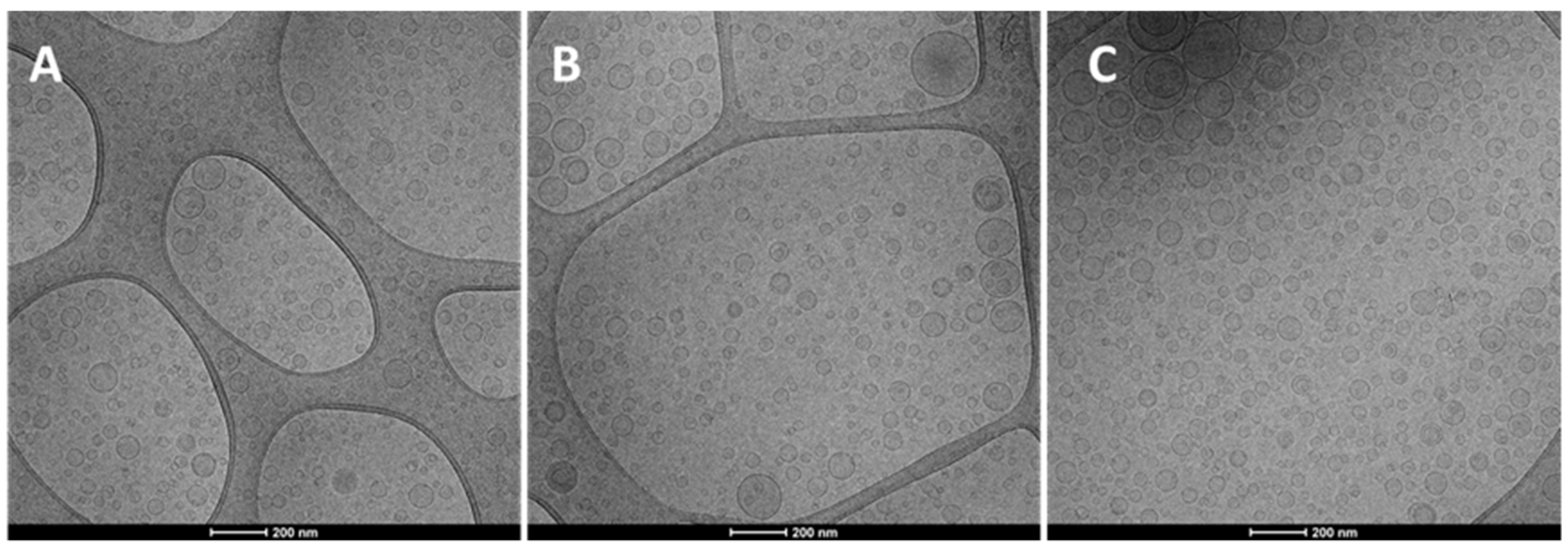
2.2. Vesicle Preparation and Characterization
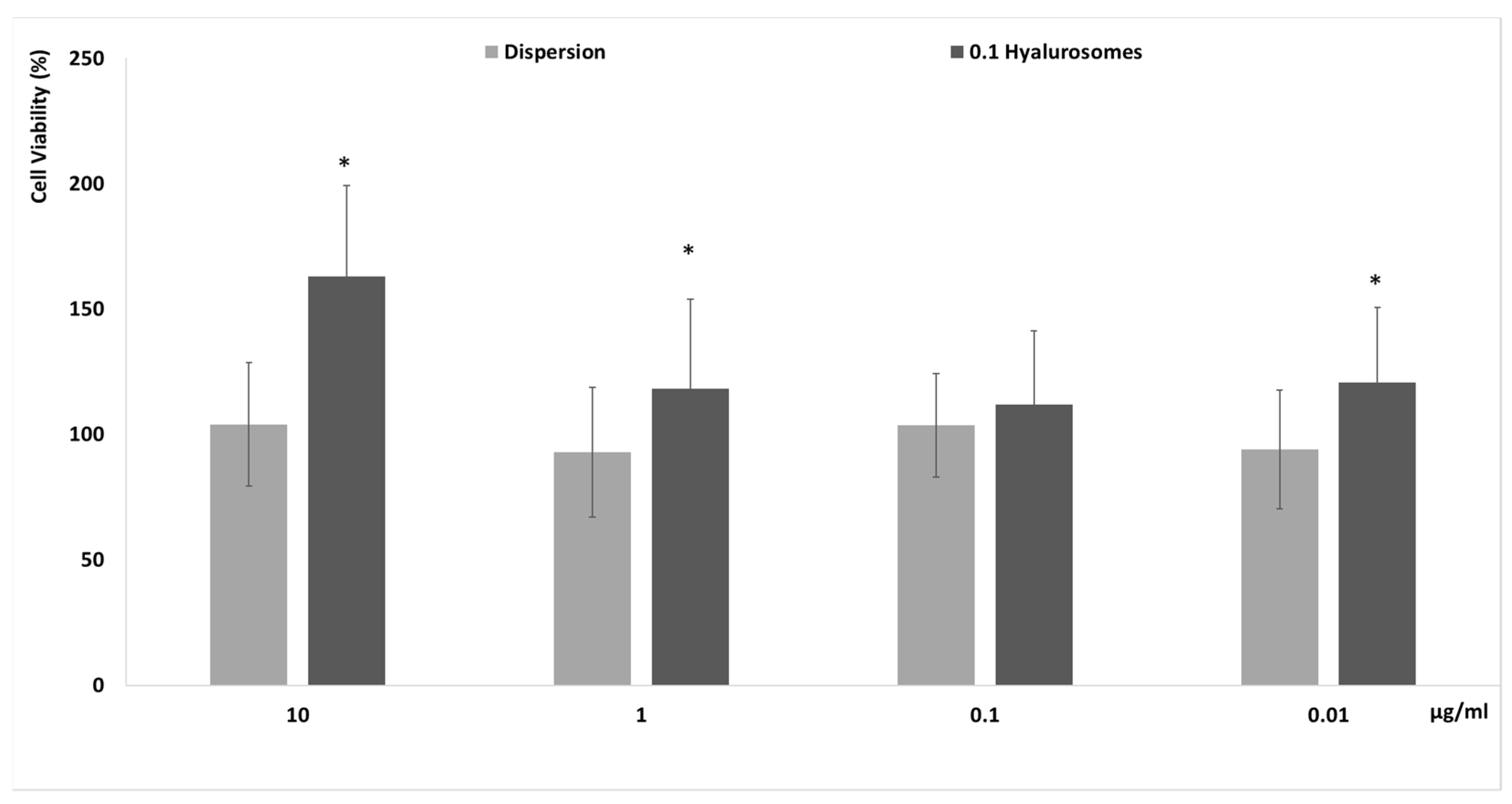
2.3. Biocompatibility of Formulations
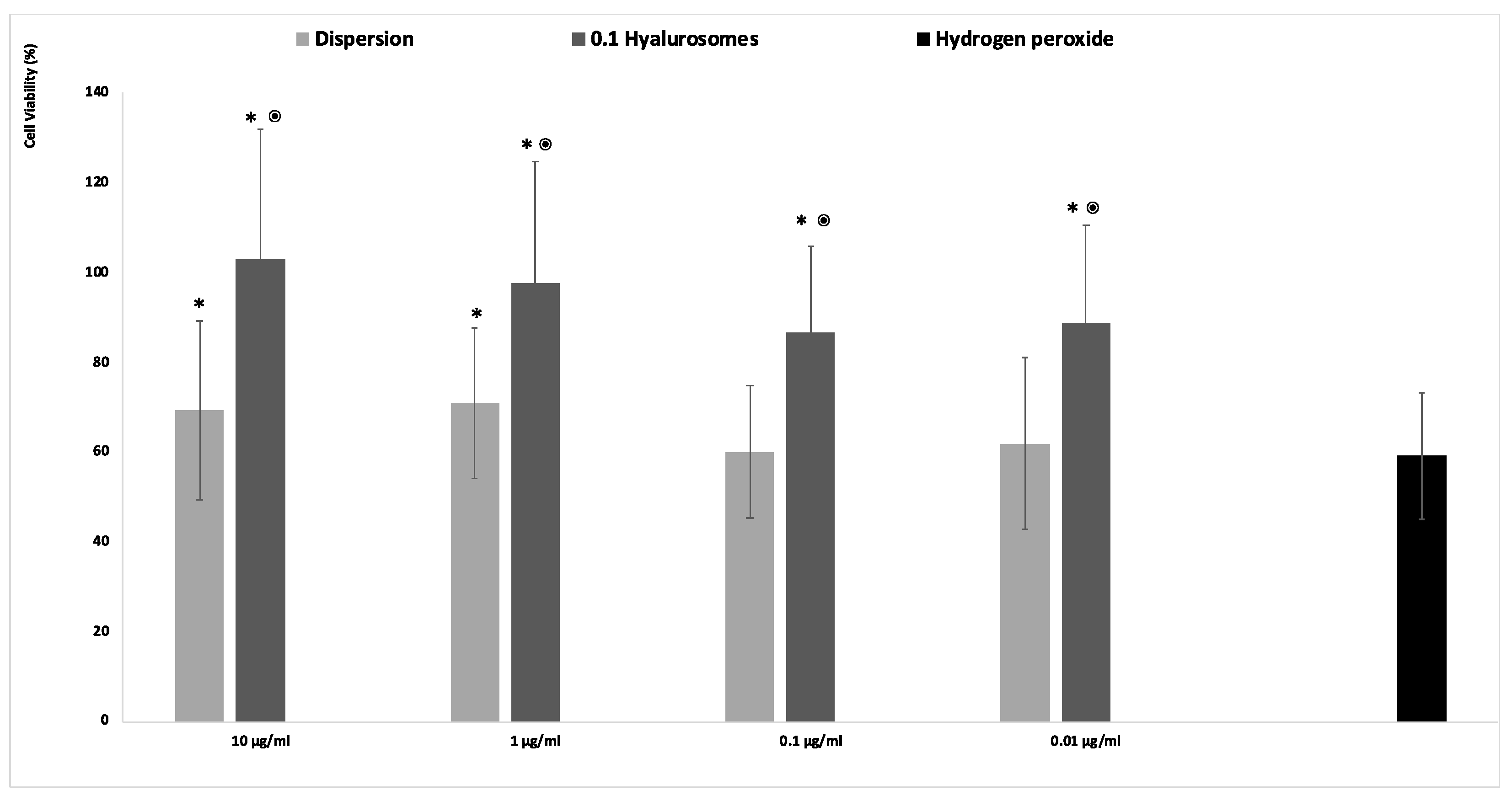
2.4. Protective Effect of the Formulations against Damages Induced by Hydrogen Peroxide
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Bioproduction of Extract
4.3. Ability of Extract to Scavenge Free Radicals
4.4. Ferric Reducing Antioxidant Assay
4.5. Vesicle Preparation
4.6. Vesicle Characterization
4.7. In Vitro Cytotoxicity of Formulations
4.8. In Vitro Protective Effect of Formulations against Oxidative Damage in Fibroblasts
4.9. Statistical Analysis of Data
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Demasi, S.; Caser, M.; Lonati, M.; Cioni, P.L.; Pistelli, L.; Najar, B.; Scariot, V. Latitude and Altitude Influence Secondary Metabolite Production in Peripheral Alpine Populations of the Mediterranean Species Lavandula Angustifolia Mill. Front. Plant Sci. 2018, 9, 983. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hajhashemi, V.; Ghannadi, A.; Sharif, B. Anti-Inflammatory and Analgesic Properties of the Leaf Extracts and Essential Oil of Lavandula Angustifolia Mill. J. Ethnopharmacol. 2003, 89, 67–71. [Google Scholar] [CrossRef]
- Héral, B.; Stierlin, É.; Fernandez, X.; Michel, T. Phytochemicals from the Genus Lavandula: A Review. Phytochem. Rev. 2021, 20, 751–771. [Google Scholar] [CrossRef]
- Alasalvar, H.; Yildirim, Z. Ultrasound-Assisted Extraction of Antioxidant Phenolic Compounds from Lavandula Angustifolia Flowers Using Natural Deep Eutectic Solvents: An Experimental Design Approach. Sustain. Chem. Pharm. 2021, 22, 100492. [Google Scholar] [CrossRef]
- Ivanova, D.; Gerova, D.; Chervenkov, T.; Yankova, T. Polyphenols and Antioxidant Capacity of Bulgarian Medicinal Plants. J. Ethnopharmacol. 2005, 96, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Yadikar, N.; Bobakulov, K.; Li, G.; Aisa, H.A. Seven New Phenolic Compounds from Lavandula Angustifolia. Phytochem. Lett. 2018, 23, 149–154. [Google Scholar] [CrossRef]
- Adaszyńska-Skwirzyńska, M.; Dzięcioł, M. Comparison of Phenolic Acids and Flavonoids Contents in Various Cultivars and Parts of Common Lavender (Lavandula Angustifolia) Derived from Poland. Nat. Prod. Res. 2017, 31, 2575–2580. [Google Scholar] [CrossRef]
- Koycheva, I.K.; Vasileva, L.V.; Amirova, K.M.; Marchev, A.S.; Balcheva-Sivenova, Z.P.; Georgiev, M.I. Biotechnologically Produced Lavandula Angustifolia Mill. Extract Rich in Rosmarinic Acid Resolves Psoriasis-Related Inflammation Through Janus Kinase/Signal Transducer and Activator of Transcription Signaling. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef]
- Kirakosyan, A.; Cseke, L.J.; Kaufman, P.B. The Use of Plant Cell Biotechnology for the Production of Phytochemicals. In Recent Advances in Plant Biotechnology; Springer: Boston, MA, USA, 2009. [Google Scholar]
- Wink, M.; Alfermann, A.W.; Franke, R.; Wetterauer, B.; Distl, M.; Windhövel, J.; Krohn, O.; Fuss, E.; Garden, H.; Mohagheghzadeh, A.; et al. Sustainable Bioproduction of Phytochemicals by Plant in Vitro Cultures: Anticancer Agents. Plant Genet. Resour. 2005, 3, 90–100. [Google Scholar] [CrossRef]
- Pant, B. Application of Plant Cell and Tissue Culture for the Production of Phytochemicals in Medicinal Plants. Adv. Exp. Med. Biol. 2014, 808, 25–39. [Google Scholar] [CrossRef]
- Lai, F.; Cucca, F.; Frau, R.; Corrias, F.; Schlich, M.; Caboni, P.; Fadda, A.M.; Bassareo, V. Systemic Administration of Orexin A Loaded Liposomes Potentiates Nucleus Accumbens Shell Dopamine Release by Sucrose Feeding. Front. Psychiatry 2018, 9, 640. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, M.J.; Billingsley, M.M.; Haley, R.M.; Wechsler, M.E.; Peppas, N.A.; Langer, R. Engineering Precision Nanoparticles for Drug Delivery. Nat. Rev. Drug Discov. 2021, 20, 101–124. [Google Scholar] [CrossRef] [PubMed]
- Allaw, M.; Manca, M.L.; Castangia, I.; Manconi, M. From Plants to Phospholipid Vesicles: A Comprehensive Review on the Incorporation of Phytochemicals into Phospholipid Vesicles Designed for Skin Applications with Special Focus on Scalability and in Vitro and in Vivo Efficacy. J. Drug Deliv. Sci. Technol. 2022, 67, 103049. [Google Scholar] [CrossRef]
- Georgiev, M.; Pavlov, A.; Ilieva, M. Selection of High Rosmarinic Acid Producing Lavandula Vera MM Cell Lines. Process Biochem. 2006, 41, 2068–2071. [Google Scholar] [CrossRef]
- Joardar, S.; Dewanjee, S.; Bhowmick, S.; Dua, T.K.; Das, S.; Saha, A.; de Feo, V. Rosmarinic Acid Attenuates Cadmium-Induced Nephrotoxicity via Inhibition of Oxidative Stress, Apoptosis, Inflammation and Fibrosis. Int. J. Mol. Sci. 2019, 20, 2027. [Google Scholar] [CrossRef] [Green Version]
- Soheili, M.; Salami, M. Lavandula Angustifolia Biological Characteristics: An in Vitro Study. J. Cell. Physiol. 2019, 234, 16424–16430. [Google Scholar] [CrossRef]
- Csepregi, K.; Neugart, S.; Schreiner, M.; Hideg, É. Comparative Evaluation of Total Antioxidant Capacities of Plant Polyphenols. Molecules 2016, 21, 208. [Google Scholar] [CrossRef] [Green Version]
- Castangia, I.; Manca, M.L.; Catalán-Latorre, A.; Maccioni, A.M.; Fadda, A.M.; Manconi, M. Phycocyanin-Encapsulating Hyalurosomes as Carrier for Skin Delivery and Protection from Oxidative Stress Damage. J. Mater. Sci. Mater. Med. 2016, 27, 1–10. [Google Scholar] [CrossRef]
- Castangia, I.; Caddeo, C.; Manca, M.L.; Casu, L.; Latorre, A.C.; Díez-Sales, O.; Ruiz-Saurí, A.; Bacchetta, G.; Fadda, A.M.; Manconi, M. Delivery of Liquorice Extract by Liposomes and Hyalurosomes to Protect the Skin against Oxidative Stress Injuries. Carbohydr. Polym. 2015, 134, 657–663. [Google Scholar] [CrossRef]
- Huycke, M.M.; Abrams, V.; Moore, D.R. Enterococcus Faecalis Produces Extracellular Superoxide and Hydrogen Peroxide That Damages Colonic Epithelial Cell DNA. Carcinogenesis 2002, 23, 529–536. [Google Scholar] [CrossRef]
- Manca, M.L.; Manconi, M.; Falchi, A.M.; Castangia, I.; Valenti, D.; Lampis, S.; Fadda, A.M. Close-Packed Vesicles for Diclofenac Skin Delivery and Fibroblast Targeting. Colloids Surf. B Biointerfaces 2013, 111, 609–617. [Google Scholar] [CrossRef] [PubMed]
- Wickett, R.R.; Visscher, M.O. Structure and Function of the Epidermal Barrier. Am. J. Infect. Control. 2006, 34, S98–S110. [Google Scholar] [CrossRef]
- Bickers, D.R.; Athar, M. Oxidative Stress in the Pathogenesis of Skin Disease. J. Investig. Dermatol. 2006, 126, 2565–2575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Portugal, M.; Barak, V.; Ginsburg, I.; Kohen, R. Interplay among Oxidants, Antioxidants, and Cytokines in Skin Disorders: Present Status and Future Considerations. Biomed. Pharmacother. 2007, 61, 412–422. [Google Scholar] [CrossRef]
- Black, H.S. ROS: A Step Closer to Elucidating Their Role in the Etiology of Light-Induced Skin Disorders. J. Investig. Dermatol. 2004, 122, xiii–xiv. [Google Scholar] [CrossRef] [Green Version]
- Schulze, C.; Wetzel, F.; Kueper, T.; Malsen, A.; Muhr, G.; Jaspers, S.; Blatt, T.; Wittern, K.P.; Wenck, H.; Käs, J.A. Stiffening of Human Skin Fibroblasts with Age. Biophys. J. 2010, 99, 2434–2442. [Google Scholar] [CrossRef] [Green Version]
- Godic, A.; Poljšak, B.; Adamic, M.; Dahmane, R. The Role of Antioxidants in Skin Cancer Prevention and Treatment. Oxidative Med. Cell. Longev. 2014, 2014, 1–6. [Google Scholar] [CrossRef]
- Santos-Sánchez, N.F.; Salas-Coronado, R.; Villanueva-Cañongo, C.; Hernández-Carlos, B. Antioxidant Compounds and Their Antioxidant Mechanism. Antioxidants 2019, 10, 1–29. [Google Scholar]
- Chen, W.P.; Jin, G.J.; Xiong, Y.; Hu, P.F.; Bao, J.P.; Wu, L.D. Rosmarinic Acid Down-Regulates NO and PGE2 Expression via MAPK Pathway in Rat Chondrocytes. J. Cell. Mol. Med. 2018, 22, 346–353. [Google Scholar] [CrossRef]
- Manca, M.L.; Castangia, I.; Zaru, M.; Nácher, A.; Valenti, D.; Fernàndez-Busquets, X.; Fadda, A.M.; Manconi, M. Development of Curcumin Loaded Sodium Hyaluronate Immobilized Vesicles (Hyalurosomes) and Their Potential on Skin Inflammation and Wound Restoring. Biomaterials 2015, 71, 100–109. [Google Scholar] [CrossRef]
- Bukhari, S.N.A.; Roswandi, N.L.; Waqas, M.; Habib, H.; Hussain, F.; Khan, S.; Sohail, M.; Ramli, N.A.; Thu, H.E.; Hussain, Z. Hyaluronic Acid, a Promising Skin Rejuvenating Biomedicine: A Review of Recent Updates and Pre-Clinical and Clinical Investigations on Cosmetic and Nutricosmetic Effects. Int. J. Biol. Macromol. 2018, 120, 1682–1695. [Google Scholar] [CrossRef] [PubMed]
- Abruzzo, A.; Cappadone, C.; Farruggia, G.; Luppi, B.; Bigucci, F.; Cerchiara, T. Glycyrrhetinic Acid Liposomes and Hyalurosomes on Spanish Broom, Flax, and Hemp Dressings to Heal Skin Wounds. Molecules 2020, 25, 2558. [Google Scholar] [CrossRef] [PubMed]
- Nicolai, M.; Pereira, P.; Vitor, R.F.; Reis, C.P.; Roberto, A.; Rijo, P. Antioxidant Activity and Rosmarinic Acid Content of Ultrasound-Assisted Ethanolic Extracts of Medicinal Plants. Meas. J. Int. Meas. Confed. 2016, 89, 328–332. [Google Scholar] [CrossRef]
- Kovatcheva-Apostolova, E.G.; Georgiev, M.I.; Ilieva, M.P.; Skibsted, L.H.; Rødtjer, A.; Andersen, M.L. Extracts of Plant Cell Cultures of Lavandula Vera and Rosa Damascena as Sources of Phenolic Antioxidants for Use in Foods. Eur. Food Res. Technol. 2008, 227, 1243–1249. [Google Scholar] [CrossRef]
- Borocci, S.; Bozzuto, G.; Bombelli, C.; Ceccacci, F.; Formisano, G.; Stringaro, A.; Molinari, A.; Mancini, G. How Stereochemistry of Lipid Components Can Affect Lipid Organization and the Route of Liposome Internalization into Cells. Nanoscale 2021, 13, 11976–11993. [Google Scholar] [CrossRef]
| Extract, mg/mL | P90G, mg/mL | Hyaluronan, mg/mL | Water, mL | |
|---|---|---|---|---|
| 0.05% hyalurosomes | 5 | 195 | 0.5 | 2 |
| 0.05% hyalurosomes | 10 | 195 | 0.5 | 2 |
| 0.05% hyalurosomes | 20 | 195 | 0.5 | 2 |
| 0.1% hyalurosomes | 5 | 195 | 1 | 2 |
| 0.1% hyalurosomes | 10 | 195 | 1 | 2 |
| 0.1% hyalurosomes | 20 | 195 | 1 | 2 |
| 0.2% hyalurosomes | 5 | 195 | 2 | 2 |
| 0.2% hyalurosomes | 10 | 195 | 2 | 2 |
| 0.2% hyalurosomes | 20 | 195 | 2 | 2 |
| Extract, mg/mL | MD, nm | PI | ZP, mV | EE, % | |
|---|---|---|---|---|---|
| 0.05% hyalurosomes | 5 | ° 123 ± 5 | 0.26 ± 0.01 | −21 ± 1 | 99 ± 1 |
| 0.05% hyalurosomes | 10 | # 133 ± 4 | 0.26 ± 0.01 | −24 ± 1 | 100 ± 1 |
| 0.05% hyalurosomes | 20 | # 135 ± 3 | 0.27 ± 0.02 | −27 ± 1 | 100 ± 1 |
| 0.1% hyalurosomes | 5 | * 142 ± 4 | 0.30 ± 0.03 | −21 ± 1 | 106 ± 3 |
| 0.1% hyalurosomes | 10 | * 141 ± 5 | 0.28 ± 0.01 | −24 ± 1 | 101 ± 1 |
| 0.1% hyalurosomes | 20 | *# 137 ± 20 | 0.30 ± 0.04 | −26 ± 1 | 101 ± 1 |
| 0.2% hyalurosomes | 5 | # 130 ± 2 | 0.26 ± 0.01 | −23 ± 1 | 100 ± 1 |
| 0.2% hyalurosomes | 10 | § 157 ± 4 | 0.28 ± 0.01 | −25 ± 1 | 100 ± 1 |
| 0.2% hyalurosomes | 20 | § 161 ± 12 | 0.32 ± 0.04 | −26 ± 1 | 100 ± 1 |
| Extract mg/mL | TE mg/mL of Sample | FRAP µmol Fe2+/mL of Sample | |
|---|---|---|---|
| 5-Dispersion | 5 | 1.72 ± 0.08 | 228.69 ± 22.40 |
| 10-Dispersion | 10 | 1.85 ± 0.06 | 368.02 ± 49.29 |
| 20-Dispersion | 20 | 1.76 ± 0.03 | 554.53 ± 35.80 |
| 0.05% hyalurosomes | 5 | 1.71 ± 0.04 | 200.97 ± 16.61 |
| 0.05% hyalurosomes | 10 | 1.81 ± 0.01 | 325.83 ± 3.95 |
| 0.05% hyalurosomes | 20 | 1.76 ± 0.02 | 531.63 ± 7.89 |
| 0.1% hyalurosomes | 5 | 1.54 ± 0.07 | 223.18 ± 13 |
| 0.1% hyalurosomes | 10 | 1.93 ± 0.01 | 362.68 ± 5.43 |
| 0.1% hyalurosomes | 20 | 1.80 ± 0.02 | 533.18 ± 1.30 |
| 0.2% hyalurosomes | 5 | 1.72 ± 0.02 | 249.19 ± 16.13 |
| 0.2% hyalurosomes | 10 | 1.80 ± 0.01 | 426.58 ± 7.89 |
| 0.2% hyalurosomes | 20 | 1.74 ± 0.01 | 600.18 ± 4.14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perra, M.; Fancello, L.; Castangia, I.; Allaw, M.; Escribano-Ferrer, E.; Peris, J.E.; Usach, I.; Manca, M.L.; Koycheva, I.K.; Georgiev, M.I.; et al. Formulation and Testing of Antioxidant and Protective Effect of Hyalurosomes Loading Extract Rich in Rosmarinic Acid Biotechnologically Produced from Lavandula angustifolia Miller. Molecules 2022, 27, 2423. https://doi.org/10.3390/molecules27082423
Perra M, Fancello L, Castangia I, Allaw M, Escribano-Ferrer E, Peris JE, Usach I, Manca ML, Koycheva IK, Georgiev MI, et al. Formulation and Testing of Antioxidant and Protective Effect of Hyalurosomes Loading Extract Rich in Rosmarinic Acid Biotechnologically Produced from Lavandula angustifolia Miller. Molecules. 2022; 27(8):2423. https://doi.org/10.3390/molecules27082423
Chicago/Turabian StylePerra, Matteo, Laura Fancello, Ines Castangia, Mohamad Allaw, Elvira Escribano-Ferrer, José Esteban Peris, Iris Usach, Maria Letizia Manca, Ivanka K. Koycheva, Milen I. Georgiev, and et al. 2022. "Formulation and Testing of Antioxidant and Protective Effect of Hyalurosomes Loading Extract Rich in Rosmarinic Acid Biotechnologically Produced from Lavandula angustifolia Miller" Molecules 27, no. 8: 2423. https://doi.org/10.3390/molecules27082423
APA StylePerra, M., Fancello, L., Castangia, I., Allaw, M., Escribano-Ferrer, E., Peris, J. E., Usach, I., Manca, M. L., Koycheva, I. K., Georgiev, M. I., & Manconi, M. (2022). Formulation and Testing of Antioxidant and Protective Effect of Hyalurosomes Loading Extract Rich in Rosmarinic Acid Biotechnologically Produced from Lavandula angustifolia Miller. Molecules, 27(8), 2423. https://doi.org/10.3390/molecules27082423










