Integration of Metal-Organic Frameworks with Bi-Nanoprobes as Dual-Emissive Ratiometric Sensors for Fast and Highly Sensitive Determination of Food Hazards
Abstract
:1. Introduction
2. Results and Discussion
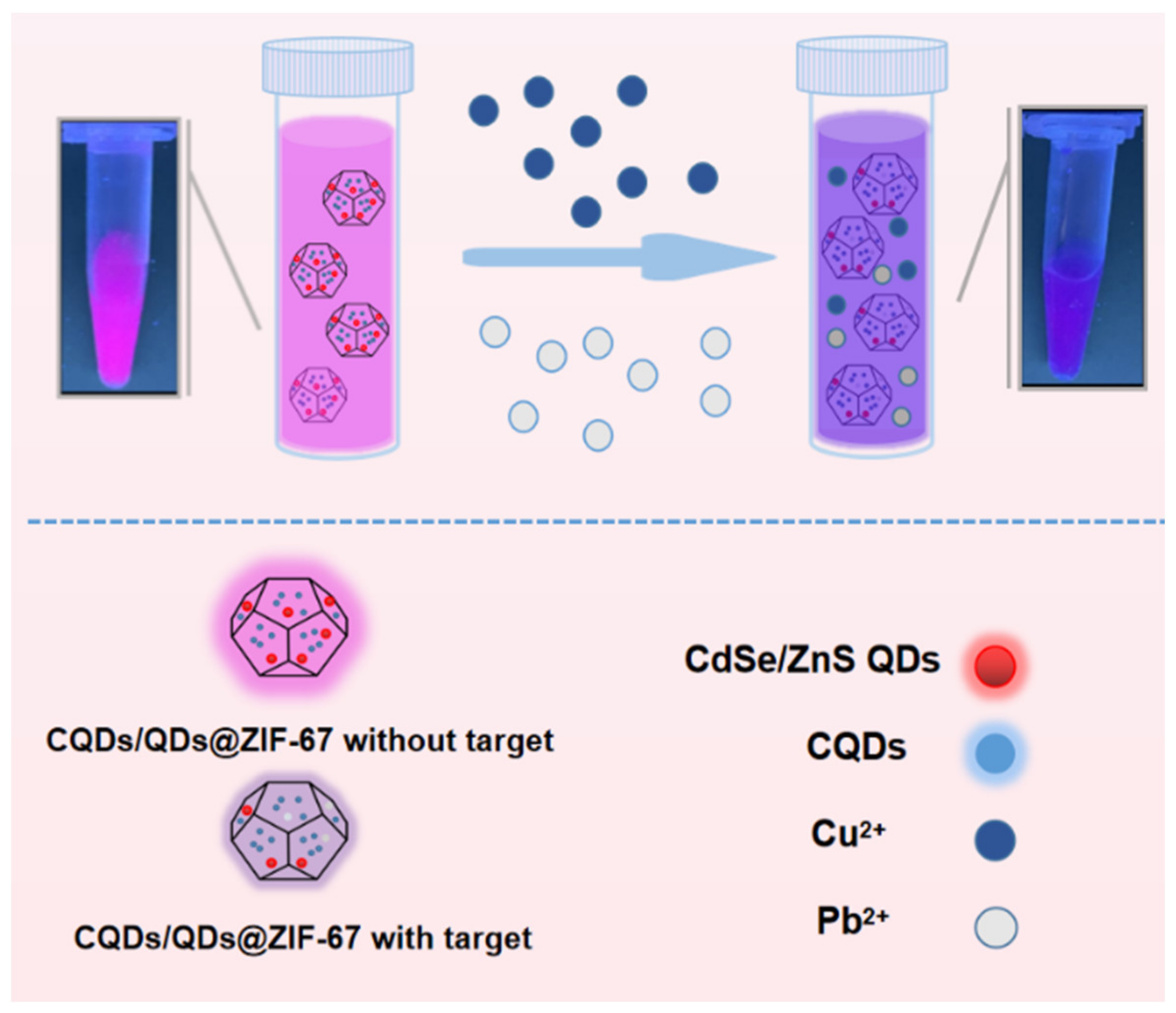
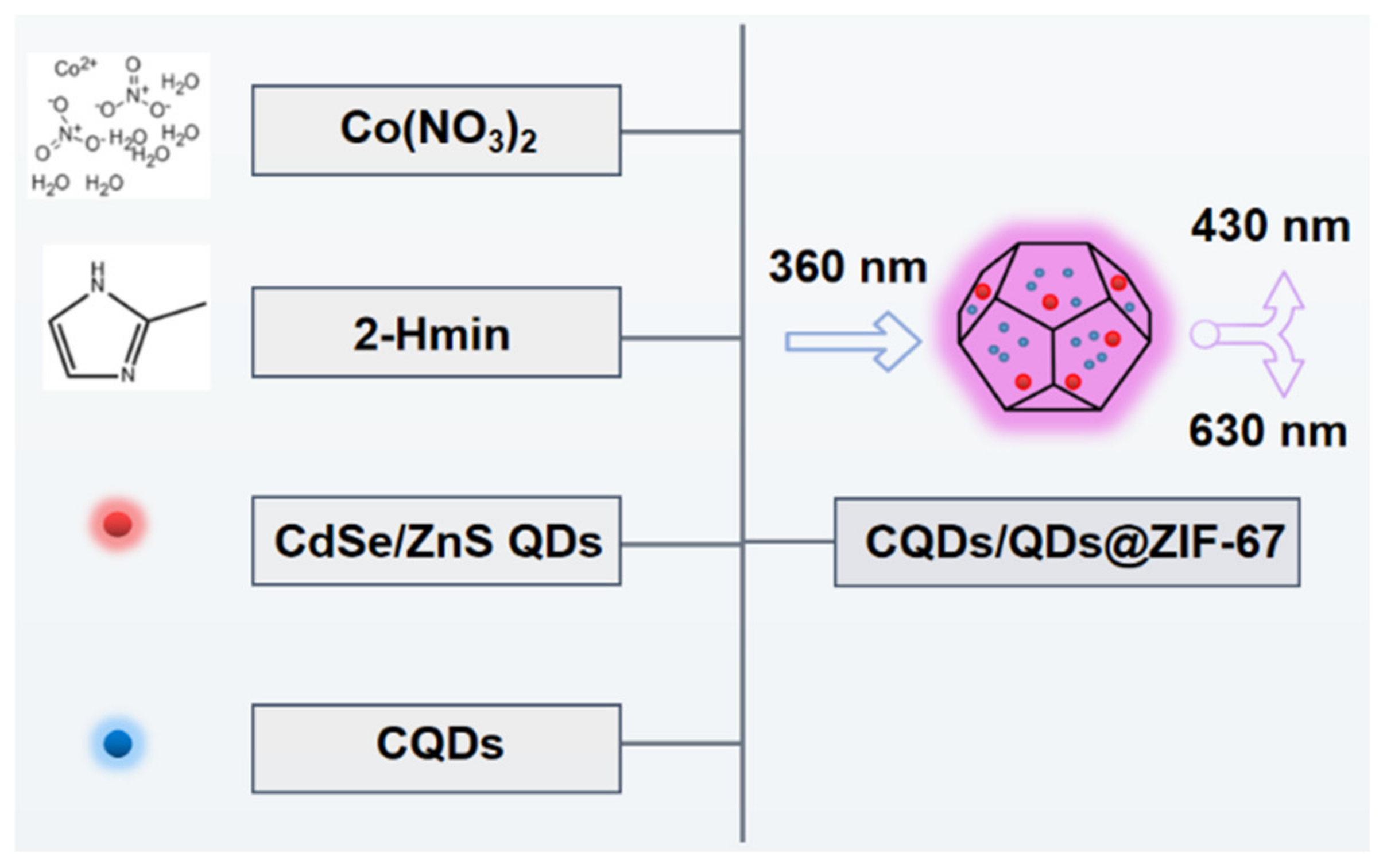
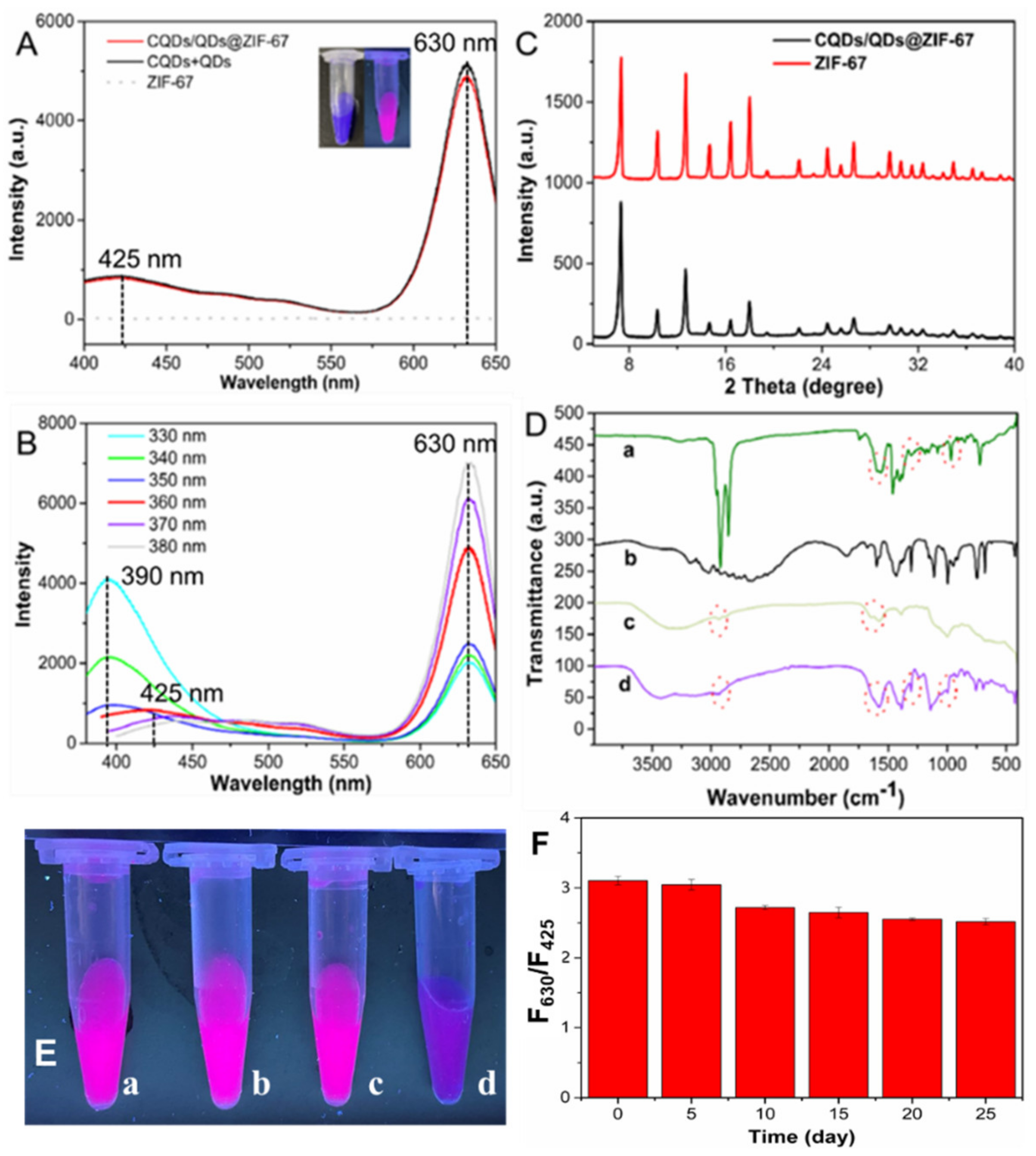
2.1. Preparation of the CQDs/QDs@ZIF-67 Composite
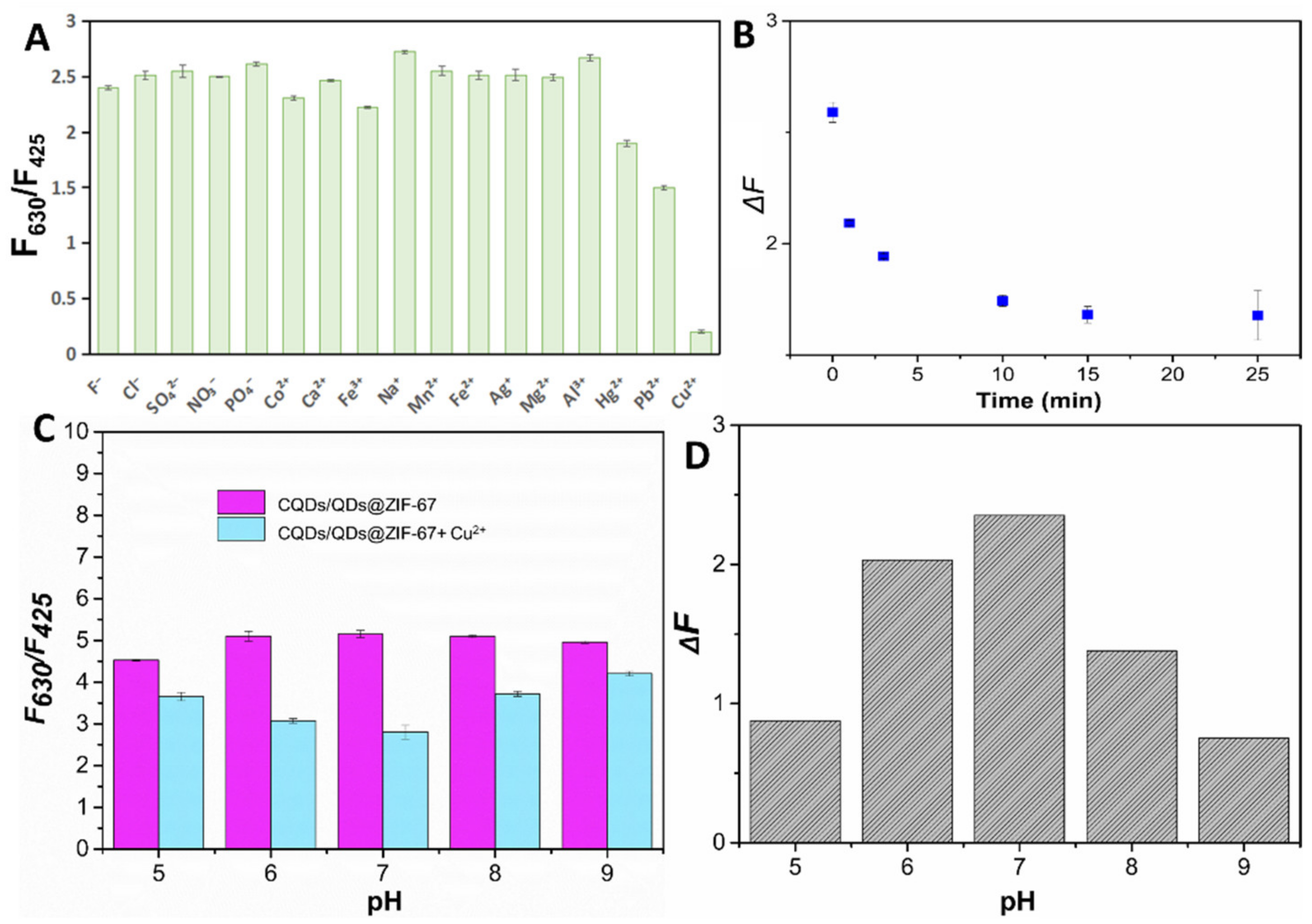
2.2. The Sensing Performance of CQDs/QDs@ZIF-67
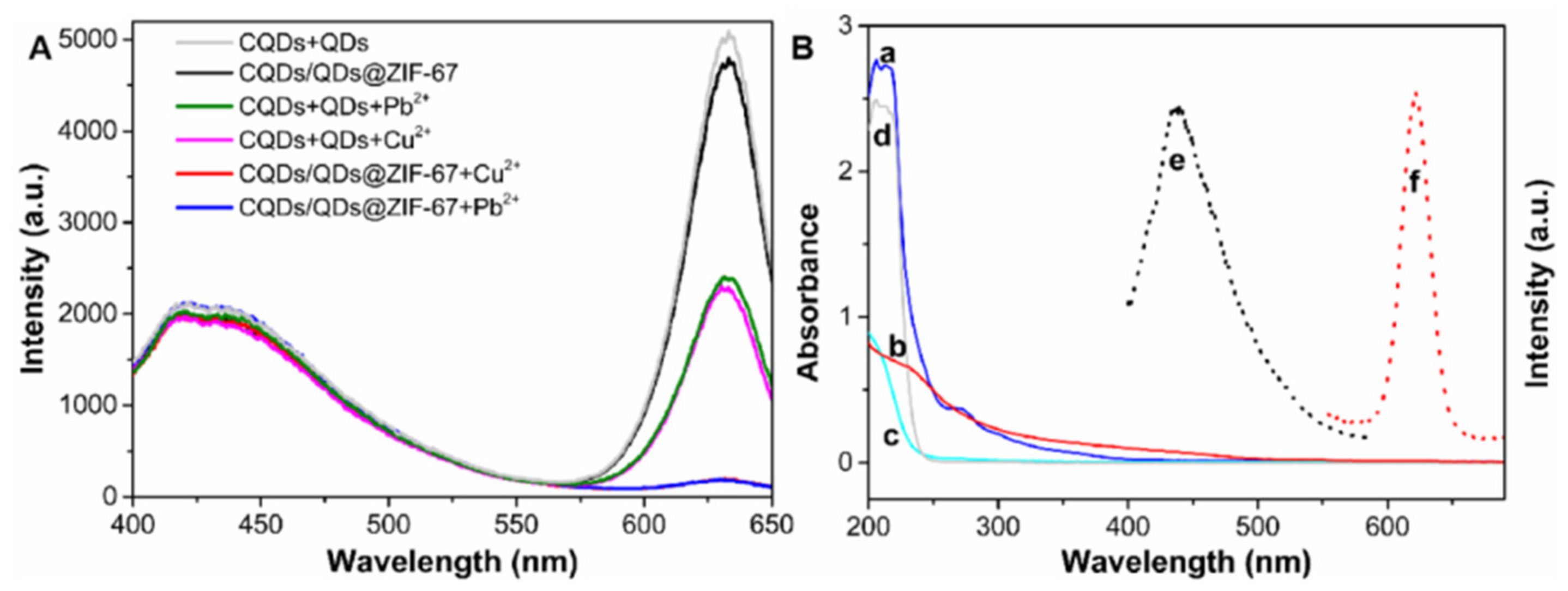
2.3. The Interaction between CQDs/QDs@ZIF-67 and Targets
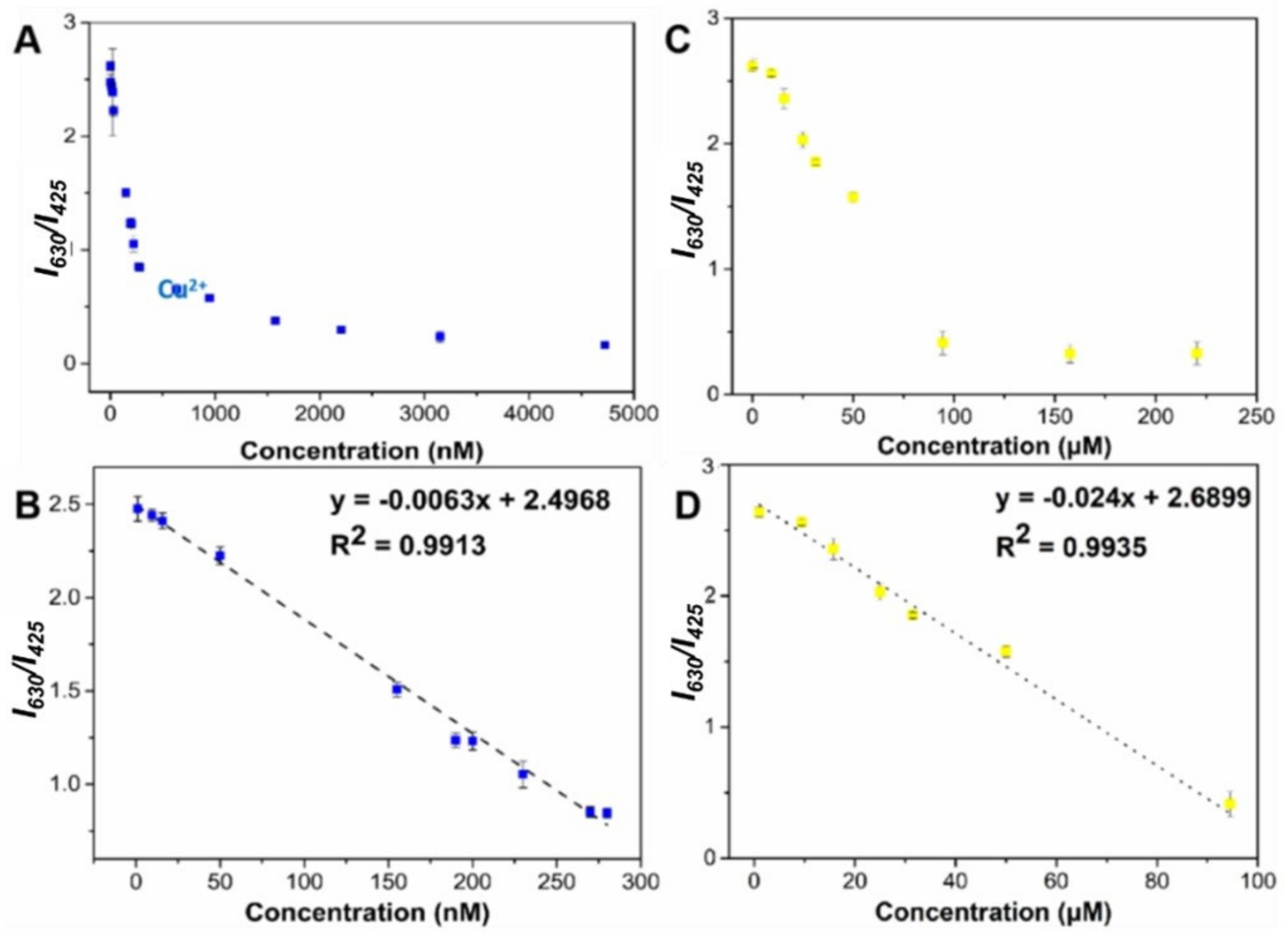
2.4. Method Establishment
2.5. Application to Practical Samples
3. Materials and Methods
3.1. Materials
3.2. Instrumentation
3.3. Preparation of Carbon Quantum Dots (CQDs)
3.4. Synthesis of the CQDs/QDs@ZIF-67 Composite
3.5. Determination of Cu2+ and Pb2+
3.6. Sample Preparation
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Lu, Y.; Song, S.; Wang, R.; Liu, Z.; Meng, J.; Sweetman, A.J.; Jenkins, A.; Ferrier, R.C.; Li, H.; Luo, W.; et al. Impacts of soil and water pollution on food safety and health risks in China. Environ. Int. 2015, 77, 5–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, P.; Pournara, A.; Kim, K.-H.; Bansal, V.; Rapti, S.; Manos, M.J. Metal-organic frameworks: Challenges and opportunities for ion-exchange/sorption applications. Prog. Mater. Sci. 2017, 86, 25–74. [Google Scholar] [CrossRef]
- Wu, Y.; Xu, M.; Chen, X.; Yang, S.; Wu, H.; Pan, J.; Xiong, X. CTAB-assisted synthesis of novel ultrathin MoSe2 nanosheets perpendicular to graphene for the adsorption and photodegradation of organic dyes under visible light. Nanoscale 2016, 8, 440–450. [Google Scholar] [CrossRef] [PubMed]
- Wei, M.-P.; Chai, H.; Cao, Y.-L.; Jia, D.-Z. Sulfonated graphene oxide as an adsorbent for removal of Pb2+ and methylene blue. J. Colloid. Interface Sci. 2018, 524, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Tan, P.; Hu, Y.; Bi, Q. Competitive adsorption of Cu2+, Cd2+ and Ni2+ from an aqueous solution on graphene oxide membranes. Colloid. Surf. A. 2016, 509, 56–64. [Google Scholar] [CrossRef]
- Reddad, Z.; Gerente, C.; Andres, Y.; Le Cloirec, P. Adsorption of Several Metal Ions onto a Low-Cost Biosorbent: Kinetic and Equilibrium Studies. Environ. Sci. Technol. 2002, 36, 2067–2073. [Google Scholar] [CrossRef]
- Stafiej, A.; Pyrzynska, K. Adsorption of heavy metal ions with carbon nanotubes. Sep. Purif. Technol. 2007, 58, 49–52. [Google Scholar] [CrossRef]
- Gao, C.; Zhu, H.; Zhang, M.; Tan, T.; Chen, J.; Qiu, H. A new highly Zn2+-selective and “off–on” fluorescent chemosensor based on the pyrene group. Anal. Methods 2015, 7, 8172–8176. [Google Scholar] [CrossRef]
- Rajasekhar, K.; Madhu, C.; Govindaraju, T. Natural Tripeptide-Based Inhibitor of Multifaceted Amyloid β Toxicity. ACS Chem. Neurosci. 2016, 7, 1300–1310. [Google Scholar] [CrossRef]
- Georgopoulos, P.G.; Roy, A.; Yonone-Lioy, M.J.; Opiekun, R.E.; Lioy, P.J. Environmental copper: Its dynamics and human exposure issues. J. Toxicol. Environ. Health B 2001, 4, 341–394. [Google Scholar] [CrossRef]
- Kim, Y.-R.; Kim, H.J.; Kim, J.S.; Kim, H. Rhodamine-Based “Turn-On” Fluorescent Chemodosimeter for Cu(II) on Ultrathin Platinum Films as Molecular Switches. Adv. Mater. 2008, 20, 4428–4432. [Google Scholar] [CrossRef]
- Zong, C.; Ai, K.; Zhang, G.; Li, H.; Lu, L. Dual-Emission Fluorescent Silica Nanoparticle-Based Probe for Ultrasensitive Detection of Cu2+. Anal. Chem. 2011, 83, 3126–3132. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.; Yang, Y.; Chen, X.; Hu, X.; Zhang, L.; Liu, L.; Zhao, Y.; Wu, H.-C. Visual Detection of Copper(II) Ions Based on an Anionic Polythiophene Derivative Using Click Chemistry. Anal. Chem. 2013, 85, 5650–5653. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Bai, R.; San Ly, Q. Selective removal of copper and lead ions by diethylenetriamine-functionalized adsorbent: Behaviors and mechanisms. Water Res. 2008, 42, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Parmar, P.; Daya, S. The Effect of Copper on (3H)-Tryptophan Metabolism in Organ Cultures of Rat Pineal Glands. Metab. Brain Dis. 2001, 16, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Awual, M.R.; Ismael, M.; Yaita, T.; El-Safty, S.A.; Shiwaku, H.; Okamoto, Y.; Suzuki, S. Trace copper(II) ions detection and removal from water using novel ligand modified composite adsorbent. Chem. Eng. J. 2013, 222, 67–76. [Google Scholar] [CrossRef]
- Zhao, H.; Xue, C.; Nan, T.; Tan, G.; Li, Z.; Li, Q.X.; Zhang, Q.; Wang, B. Detection of copper ions using microcantilever immunosensors and enzyme-linked immunosorbent assay. Anal. Chim. Acta 2010, 676, 81–86. [Google Scholar] [CrossRef]
- Li, X.; Wang, G.; Ding, X.; Chen, Y.; Gou, Y.; Lu, Y. A “turn-on” fluorescent sensor for detection of Pb2+ based on graphene oxide and G-quadruplex DNA. Phys. Chem. Chem. Phys. 2013, 15, 12800–12804. [Google Scholar] [CrossRef]
- Eddaoudi, M.; Moler, D.B.; Li, H.; Chen, B.; Reineke, T.M.; O’Keeffe, M.; Yaghi, O.M. Modular Chemistry: Secondary Building Units as a Basis for the Design of Highly Porous and Robust Metal-Organic Carboxylate Frameworks. Acc. Chem. Res. 2001, 34, 319–330. [Google Scholar] [CrossRef]
- Farha, O.K.; Hupp, J.T. Rational Design, Synthesis, Purification, and Activation of Metal-Organic Framework Materials. Acc. Chem. Res. 2010, 43, 1166–1175. [Google Scholar] [CrossRef] [Green Version]
- Kumar, P.; Deep, A.; Kim, K.-H. Metal organic frameworks for sensing applications. TrAC-Trends Anal. Chem. 2015, 73, 39–53. [Google Scholar] [CrossRef]
- Howarth, A.J.; Liu, Y.; Li, P.; Li, Z.; Wang, T.C.; Hupp, J.T.; Farha, O.K. Chemical, thermal and mechanical stabilities of metal-organic frameworks. Nat. Rev. Mater. 2016, 1, 15018. [Google Scholar] [CrossRef]
- Rubin, H.N.; Reynolds, M.M. Amino-Incorporated Tricarboxylate Metal-Organic Framework for the Sensitive Fluorescence Detection of Heavy Metal Ions with Insights into the Origin of Photoluminescence Response. Inorg. Chem. 2019, 58, 10671–10679. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Qi, Y.; Shen, Y.; Yuan, Y.; Zhang, L.; Zhang, C.; Sun, Y. A ratiometric electrochemical sensor for simultaneous detection of multiple heavy metal ions based on ferrocene-functionalized metal-organic framework. Sens. Actuators B Chem. 2020, 310, 127756. [Google Scholar] [CrossRef]
- Ghorbani, Y.A.; Ghoreishi, S.M.; Ghani, M. Derived N-doped carbon through core-shell structured metal-organic frameworks as a novel sorbent for dispersive solid phase extraction of Cr(III) and Pb(II) from water samples followed by quantitation through flame atomic absorption spectrometry. Microchem. J. 2020, 155, 104786. [Google Scholar] [CrossRef]
- Zhang, Z.; Lou, Y.; Guo, C.; Jia, Q.; Song, Y.; Tian, J.-Y.; Zhang, S.; Wang, M.; He, L.; Du, M. Metal-organic frameworks (MOFs) based chemosensors/biosensors for analysis of food contaminants. Trends Food Sci. Tech. 2021, 118, 569–588. [Google Scholar] [CrossRef]
- Yue, D.; Zhao, D.; Zhang, J.; Zhang, L.; Jiang, K.; Zhang, X.; Cui, Y.; Yang, Y.; Chen, B.; Qian, G. A luminescent cerium metal-organic framework for the turn-on sensing of ascorbic acid. Chem. Commun. 2017, 53, 11221–11224. [Google Scholar] [CrossRef]
- Xiao, J.; Liu, J.; Gao, X.; Ji, G.; Wang, D.; Liu, Z. A multi-chemosensor based on Zn-MOF: Ratio-dependent color transition detection of Hg (II) and highly sensitive sensor of Cr(VI). Sens. Actuators B Chem. 2018, 269, 164–172. [Google Scholar] [CrossRef]
- Bagheri, M.; Masoomi, M.Y.; Morsali, A.; Schoedel, A. Two Dimensional Host–Guest Metal-Organic Framework Sensor with High Selectivity and Sensitivity to Picric Acid. ACS Appl. Mater. Interfaces 2016, 8, 21472–21479. [Google Scholar] [CrossRef]
- Venna, S.R.; Carreon, M.A. Highly Permeable Zeolite Imidazolate Framework-8 Membranes for CO2/CH4 Separation. J. Am. Chem. Soc. 2010, 132, 76–78. [Google Scholar] [CrossRef]
- Phan, A.; Doonan, C.J.; Uribe-Romo, F.J.; Knobler, C.B.; O’Keeffe, M.; Yaghi, O.M. Synthesis, Structure, and Carbon Dioxide Capture Properties of Zeolitic Imidazolate Frameworks. Acc. Chem. Res. 2010, 43, 58–67. [Google Scholar] [CrossRef]
- Park, K.S.; Ni, Z.; Côté, A.P.; Choi, J.Y.; Huang, R.; Uribe-Romo, F.J.; Chae, H.K.; O’Keeffe, M.; Yaghi, O.M. Exceptional chemical and thermal stability of zeolitic imidazolate frameworks. Proc. Natl. Acad. Sci. USA 2006, 103, 10186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, K.-Y.A.; Chang, H.-A. Ultra-high adsorption capacity of zeolitic imidazole framework-67 (ZIF-67) for removal of malachite green from water. Chemosphere 2015, 139, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Hu, X.; Chen, T.; Zhang, S.; Zhou, M. Adsorptive removal of 1-naphthol from water with Zeolitic imidazolate framework-67. J. Phys. Chem. Solids 2017, 107, 50–54. [Google Scholar] [CrossRef]
- Zhou, K.; Mousavi, B.; Luo, Z.; Phatanasri, S.; Chaemchuen, S.; Verpoort, F. Characterization and properties of Zn/Co zeolitic imidazolate frameworks vs. ZIF-8 and ZIF-67. J. Mater. Chem. A 2017, 5, 952–957. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, Y.; Jiao, Y.; Gao, Q.; Yan, X.; Yang, Y. Facile construction of Fe@ zeolite imidazolate Framework-67 to selectively remove uranyl ions from aqueous solution. J. Taiwan Inst. Chem. E 2018, 91, 309–315. [Google Scholar] [CrossRef]
- Huang, Y.; Zeng, X.; Guo, L.; Lan, J.; Zhang, L.; Cao, D. Heavy metal ion removal of wastewater by zeolite-imidazolate frameworks. Sep. Purif. Technol. 2018, 194, 462–469. [Google Scholar] [CrossRef]
- Saliba, D.; Ammar, M.; Rammal, M.; Al-Ghoul, M.; Hmadeh, M. Crystal Growth of ZIF-8, ZIF-67, and Their Mixed-Metal Derivatives. J. Am. Chem. Soc. 2018, 140, 1812–1823. [Google Scholar] [CrossRef]
- Xu, L.; Fang, G.; Liu, J.; Pan, M.; Wang, R.; Wang, S. One-pot synthesis of nanoscale carbon dots-embedded metal-organic frameworks at room temperature for enhanced chemical sensing. J. Mater. Chem. A 2016, 4, 15880–15887. [Google Scholar] [CrossRef]
- Luan, W.; Yang, H.; Wan, Z.; Yuan, B.; Yu, X.; Tu, S.-T. Mercaptopropionic acid capped CdSe/ZnS quantum dots as fluorescence probe for lead(II). J. Nanopart. Res. 2012, 14, 762. [Google Scholar] [CrossRef]
- Qu, H.; Cao, L.; Su, G.; Liu, W.; Gao, R.; Xia, C.; Qin, J. Silica-coated ZnS quantum dots as fluorescent probes for the sensitive detection of Pb2+ ions. J. Nanopart. Res. 2014, 16, 2762. [Google Scholar] [CrossRef]
- Sung, T.-W.; Lo, Y.-L. Highly sensitive and selective sensor based on silica-coated CdSe/ZnS nanoparticles for Cu2+ ion detection. Sens. Actuators B Chem. 2012, 165, 119–125. [Google Scholar] [CrossRef]
- Callan, J.F.; Mulrooney, R.C. Luminescent detection of Cu(II) ions in aqueous solution using CdSe and CdSe-ZnS quantum dots functionalised with mercaptosuccinic acid. Phys. Status Solidi R 2009, 6, 920–923. [Google Scholar] [CrossRef]
- Wang, L.; Luo, D.; Qin, D.; Shan, D.; Lu, X. Cathodic electrochemiluminescence of a CdSe/ZnS QDs-modified glassy carbon electrode and its application in sensing of Pb2+. Anal. Methods 2015, 7, 1395–1400. [Google Scholar] [CrossRef]
- Bo, C.; Ping, Z. A new determining method of copper(II) ions at ng ml−1 levels based on quenching of the water-soluble nanocrystals fluorescence. Anal. Bioanal. Chem. 2005, 381, 986–992. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Xu, G.; Wei, F.; Cen, Y.; Ma, Y.; Song, Y.; Xu, X.; Shi, M.; Muhammad, S.; Hu, Q. A dual-emissive fluorescent sensor fabricated by encapsulating quantum dots and carbon dots into metal-organic frameworks for the ratiometric detection of Cu2+ in tap water. J. Mater. Chem. C 2017, 5, 8566–8571. [Google Scholar] [CrossRef]
- Shih, C.-I.; Chou, Y.-C.; Chen, H.-Y.; Chen, K.-H.; Wang, I.H.; Yeh, Y.-C. Colorimetric and Fluorometric Paper-Based Assay for Cu2+ Detection Based on Green Synthesis of 2-Aminoterephthalic Acid-Derived Pigments. ACS Appl. Bio Mater. 2020, 3, 2516–2521. [Google Scholar] [CrossRef]
- Wang, X.; Guo, X. Ultrasensitive Pb2+ detection based on fluorescence resonance energy transfer (FRET) between quantum dots and gold nanoparticles. Analyst 2009, 134, 1348–1354. [Google Scholar] [CrossRef] [Green Version]
- Shi, X.; Gu, W.; Zhang, C.; Zhao, L.; Peng, W.; Xian, Y. A label-free colorimetric sensor for Pb2+ detection based on the acceleration of gold leaching by graphene oxide. Dalton Trans. 2015, 44, 4623–4629. [Google Scholar] [CrossRef]
- Qiao, D.; Zhang, Z.; Wang, L.; Sheng, W.; Deng, Q.; Wang, S. In-situ preparation of molecularly imprinted fluorescent sensing test strips for on-site detection of tyramine in vinegar. Microchem. J. 2021, 160, 105638. [Google Scholar] [CrossRef]
- Asadi, F.; Azizi, S.N.; Chaichi, M.J. Green synthesis of fluorescent PEG-ZnS QDs encapsulated into Co-MOFs as an effective sensor for ultrasensitive detection of copper ions in tap water. Mat. Sci. Eng. C Mater. 2019, 105, 110058. [Google Scholar] [CrossRef]
- Qin, J.; Wang, S.; Wang, X. Visible-light reduction CO2 with dodecahedral zeolitic imidazolate framework ZIF-67 as an efficient co-catalyst. Appl. Catal. B Environ. 2017, 209, 476–482. [Google Scholar] [CrossRef]

| Material | Target | Linear Range (nM) | Method | LOD (nM) | Ref. |
|---|---|---|---|---|---|
| QDs/CDs@ZIF-8 | Cu2+ | 5–1.0 × 102 | Fluorescence | 1.53 | [46] |
| 2-AIPA–BX | Cu2+ | 2.5–50.0 × 104 | Fluorescence | 100 | [47] |
| CQDs/QDs@ZIF-67 | Cu2+ | 1.0–280.0 | Fluorescence | 0.51 | This work |
| Au NPs/CA-CdTe-QDs | Pb2+ | 1.06 × 103–2.2 × 104 | Fluorescence | 140 | [48] |
| AuNPs | Pb2+ | 1.0 × 102–2.0 × 104 | Colorimetry | 50 | [49] |
| CQDs/QDs@ZIF-67 | Pb2+ | 2.0 × 103–9.5 × 104 | Fluorescence | 15.7 | This work |
| Target | Sample | Initial (nM) | Added (nM) | Detected (nM) | Recovery (%) | RSD (%) |
|---|---|---|---|---|---|---|
| Cu2+ | Tap water | 0 | 20 | 19.2 | 96.1 | 3.5 |
| 2.0 × 102 | 1.96 × 102 | 97.8 | 3.0 | |||
| Cu2+ | Apple juice | 0 | 20 | 18.5 | 92.4 | 3.3 |
| 2.0 × 102 | 1.92 × 102 | 95.5 | 7.2 | |||
| Pb2+ | Tap water | 0 | 4 × 103 | 3.9 × 103 | 99.3 | 1.6 |
| 2.0 × 104 | 2.01 × 104 | 100.2 | 1.3 | |||
| Pb2+ | Apple juice | 0 | 4 × 103 | 3.88 × 103 | 97.1 | 2.2 |
| 2.0 × 104 | 1.95 × 104 | 97.5 | 1.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yao, C.-X.; Dong, L.; Yang, L.; Wang, J.; Li, S.-J.; Lv, H.; Ji, X.-M.; Liu, J.-M.; Wang, S. Integration of Metal-Organic Frameworks with Bi-Nanoprobes as Dual-Emissive Ratiometric Sensors for Fast and Highly Sensitive Determination of Food Hazards. Molecules 2022, 27, 2356. https://doi.org/10.3390/molecules27072356
Yao C-X, Dong L, Yang L, Wang J, Li S-J, Lv H, Ji X-M, Liu J-M, Wang S. Integration of Metal-Organic Frameworks with Bi-Nanoprobes as Dual-Emissive Ratiometric Sensors for Fast and Highly Sensitive Determination of Food Hazards. Molecules. 2022; 27(7):2356. https://doi.org/10.3390/molecules27072356
Chicago/Turabian StyleYao, Chi-Xuan, Lu Dong, Lu Yang, Jin Wang, Shi-Jie Li, Huan Lv, Xue-Meng Ji, Jing-Min Liu, and Shuo Wang. 2022. "Integration of Metal-Organic Frameworks with Bi-Nanoprobes as Dual-Emissive Ratiometric Sensors for Fast and Highly Sensitive Determination of Food Hazards" Molecules 27, no. 7: 2356. https://doi.org/10.3390/molecules27072356
APA StyleYao, C.-X., Dong, L., Yang, L., Wang, J., Li, S.-J., Lv, H., Ji, X.-M., Liu, J.-M., & Wang, S. (2022). Integration of Metal-Organic Frameworks with Bi-Nanoprobes as Dual-Emissive Ratiometric Sensors for Fast and Highly Sensitive Determination of Food Hazards. Molecules, 27(7), 2356. https://doi.org/10.3390/molecules27072356







