Plants Used in Mexican Traditional Medicine for the Management of Urolithiasis: A Review of Preclinical Evidence, Bioactive Compounds, and Molecular Mechanisms
Abstract
:1. Introduction
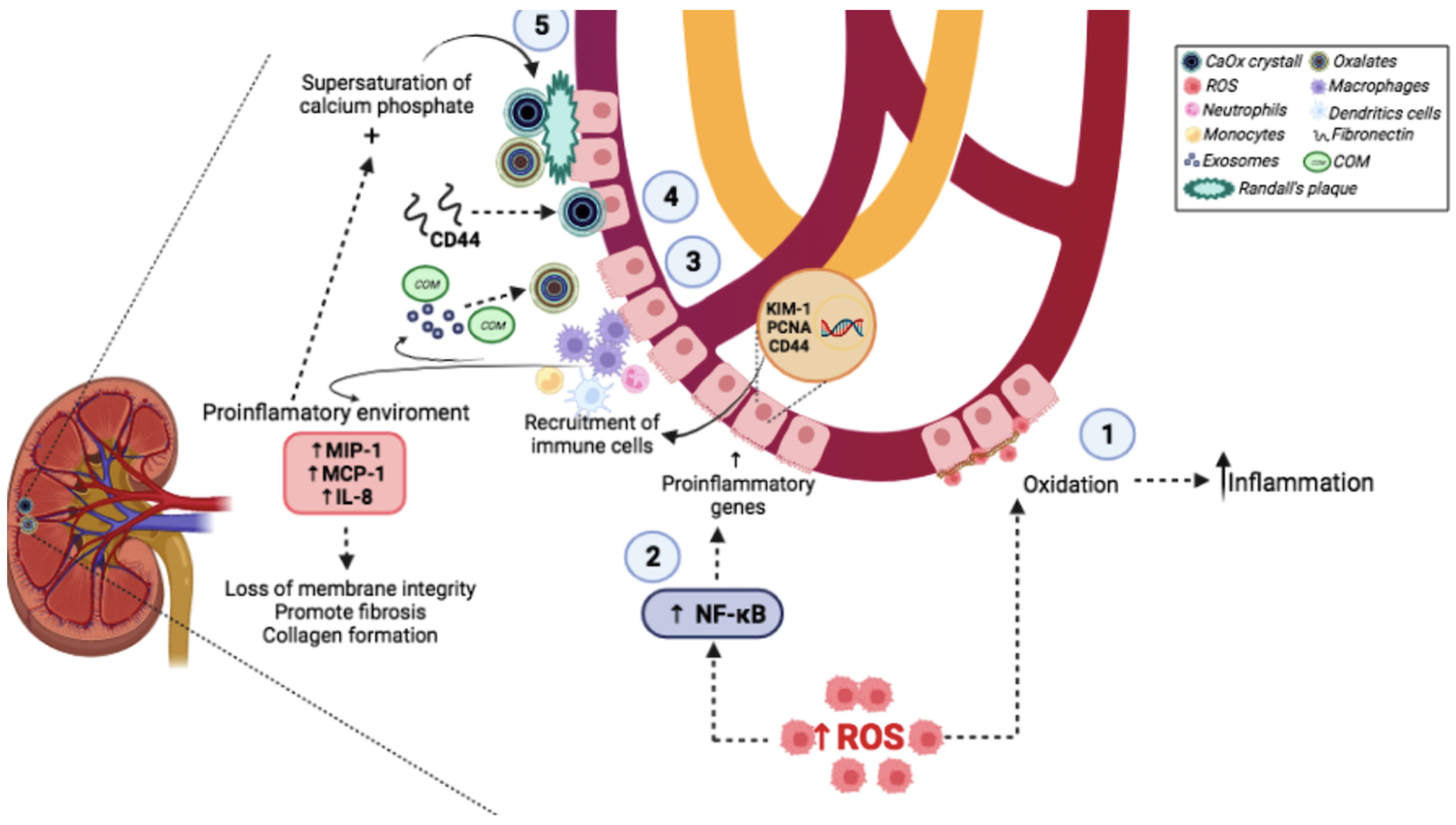
2. Molecular Mechanisms Involved in Urinary Stone Formation
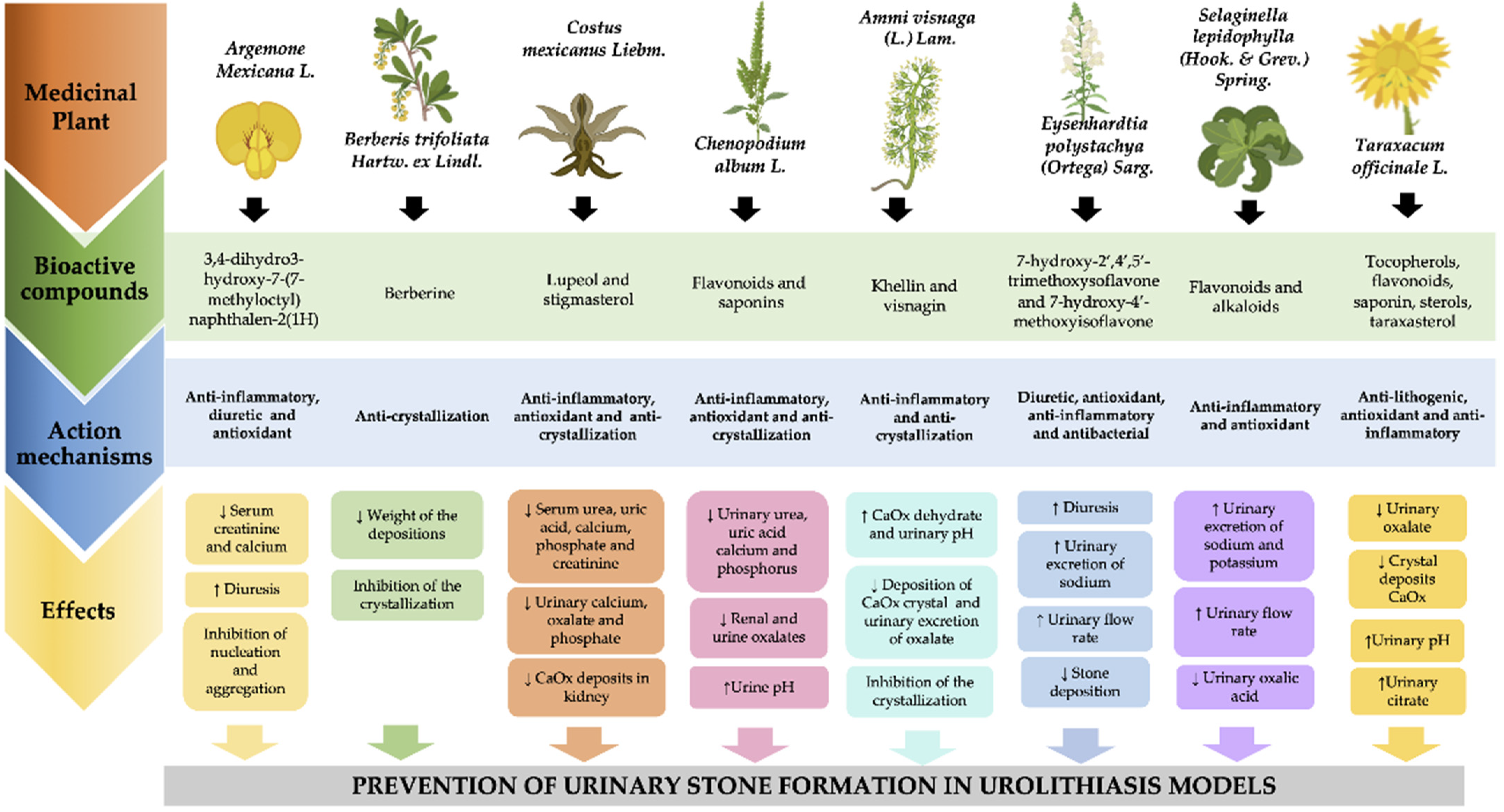
3. Plants Used in Mexican Traditional Medicine against Urolithiasis
3.1. Chicalote Amarillo (Argemone mexicana L.)
3.2. Palo Amarillo (Berberis trifoliata Hartw. ex Lindl.)
3.3. Planta de la Insulina (Costus Mexicanus Liebm)
3.4. Cenizo (Chenopodium album L.)
3.5. Visnaga (Ammi visnaga (L.) Lam.)
3.6. Palo Azul (Eysenhardtia polystachya (Ortega) Sarg.)
3.7. Doradilla (Selaginella Lepidophylla (Hook. & Grev.) Spring)
3.8. Diente de León (Taraxacum Officinale L.)
| Common Name | Binomial Nomenclature | Bioactive Compounds | Study Type | Study Design | Main Results | References |
|---|---|---|---|---|---|---|
| Chicalote amarillo | Argemone mexicana L. | 3,4-dihidro-3-hidroxi-7- (7-metiloctil) naftalen-2 (1 H) | In vitro | A mix of reaction to induce nucleation or aggregation where it was incubated with methanol leaf extract (100 mg/mL) | Inhibited nucleation and aggregation compared to standard cystone drug | [34] |
| In vivo | Wistar rats induced to UL by EG, and received following treatments for 13 days (n = 12 each group): Control group + water UL group+ water UL +cystone (750 mg/kg) UL+ methanolic extract (400 mg/kg) UL+ methanolic extract (750 mg/kg) | ↓↓ Serum creatinine and calcium in extract groups ↑ Diuresis * in all groups | [35] | |||
| Palo amarillo | Berberis trifoliata Hartw. ex Lindl. | Berberine | In vitro | Nucleation of CaOx crystals was induced by a mix of calcium chloride and sodium oxalate, and incubated with different concentration of B. trifoliata methanolic extract (100–1000 µg/mL) | Dose-dependent inhibition of crystallization | [37] |
| In vivo | Wistar rats induced to UL by zinc disc in bladder and received following treatments for 20 days (n = 6 each group): Control UL group UL + methanolic extract (50 mg/kg) UL + methanolic extract (100 mg/kg) UL + methanolic extract (150 mg/kg) | ↓↓ Weight of the depositions around the implants with all doses | [38] | |||
| Planta de la insulina | Costus mexicanus Liebm | Lupeol and stigmasterol | In vitro | A mix to induce nucleation and growth COM crystals was generated and incubated with water or different concentrations of plant aqueous extract (0.15–1%) | ↓ Mass of crystals and nucleation was delayed with a dose-dependent concentration | [41] |
| In vivo | Wistar rats induced to UL by EG and received following treatments (n = 4, each group). Control: UL group + water (1 g/kg) UL group + Aqueous extract of stem of C. igneus (100 mg/kg) UL group + ethanolic extract of stem of C. igneus (100 mg/kg) UL group + Lupeol (50 mg/kg) UL group + Lupeol (100 mg/kg) UL group + Stigmasterol (50 mg/kg) UL group + Stigmasterol (100 mg/kg) UL + gallium nitrate (50 mg/kg) | ↓ Serum levels of urea, uric acid, calcium, phosphate, and creatinine levels in lupeol and stigmasterol groups ↓ Urine levels of calcium, oxalate, creatinine, phosphate, and uric acid levels in all groups ↓ Calcium oxalate deposits in kidney in all groups | [42] | |||
| Cenizo | Chenopodium album L. | Flavonoids and saponins | In vitro | A mix to induce crystallization, nucleation and aggregation of CaOx crystals were generated and incubated with aqueous extract of the leaves (500 and 1000 μg/mL) | ↓↓↓ Size of crystals Inhibited nucleation and aggregation Inhibited COM growth * | [43] |
| In vivo | Adult Wistar rats model of UL induced by EG was used to administer CAME or CAAE from the leaves Chenopodium album (n = 6) during 28 days. Control group + water UL group + water UL group + Cystone (750 mg/kg) UL group + CAME (100 mg/kg) UL group + CAME (200 mg/kg) UL group + CAME (400 mg/kg) UL group + CAAE (100 mg/kg) UL group + CAAE (200 mg/kg) UL group + CAAE (400 mg/kg) | ↑ Urine volume with CAME and CAAE (200 and 400 mg/kg) ↑ Urine pH with CAME and CAAE (400 mg/kg) ↓↓↓ Urinary levels of urea, uric acid, calcium, and phosphorus and creatinine with CAME (200 and 400 mg/kg) and CAAE (100, 200 and 400 mg/kg) ↓ Plasma levels of creatinine and urea with treatment with CAME and CAAE (400 mg/kg) ↓ Urine oxalates level with CAME and CAAE (400 mg/kg) ↓↓↓ Renal oxalate level with CAME and CAAE (400 mg/kg) | [44] | |||
| Visnaga | Ammi visnaga (L.) Lam. | Khellin and visnagin | In vitro | CaOx-supersaturated human urine was used to evaluate the effect of aqueous extract from the whole plant (200 μL and 600 μL) and from the seeds (200 μL and 600 μL) of AVL | Inhibition of the crystallization (extract of seeds at 200 µL and 600 µL) In the presence of extract a full AVL or seed there is a great increase in the CaOx dihydrate * | [47] |
| In vitro | Cell lines (MDCK and LLC-PK1) were exposed to oxalate (300 µmol) and COM crystals (133 µg/cm2). Cells were incubated during 1, 3, 6, and 12 h as follows: Control + vehicle Control + Oxalate 300 µM Oxalate + KE (10, 50, 100, or 200 µg/mL) Control + COM COM + KE (10, 50, 100, or 200 µg/mL) | ↓↓↓ Cellular damage (% LDH release) in LLC-PK1 cells (50, 100 and 200 µg/mL KE) ↓↓↓ Cellular damage (% LDH release) in MDCK cells (100 and 200 µg/mL KE) | [49] | |||
| In vivo | Male Sprague-Dawley rats were induced to UL with EG. The animals were divided into the following experimental groups (n = 8 per group). The intervention included vehicle or KE. All treatments were administered orally for 14 days. Control group + vehicle UL group + vehicle UL group + KE (125 mg/kg) UL group + KE (250 mg/kg) UL group + KE (500 mg/kg) | ↓↓ Deposition of CaOx crystal in kidneys ↑↑↑ Urinary excretion of citrate in all doses of KE ↓↓Urinary excretion of oxalate (KE 250 and 500 mg/kg) ↑ Urinary pH in all doses of KE ↑ Urinary volume and urinary calcium (KE 500 mg/kg) | [48] | |||
| Palo Azul | Eysenhardtia polystachya (Ortega) Sarg. | 7-hydroxy-2′,4′,5′-trimethoxyisoflavone and 7-hydroxy-4′- methoxyisoflavone, | In vivo | Male Wistar rats induced UL by implantation of a zinc disc in bladder were divided into seven groups (n = 10, each group): Control group; Sham operated group and UL-induced group with different dosed of plant extract (25, 50, and 100 mg/kg) | ↓↓↓ Stone deposition in all treatment groups, dose dependent ↑ Diuretic activity in all treatments groups, dose dependent | [50] |
| In vivo | Female Wistar rats were divided into six treatments (n = 6). Control group received water (1 mL/kg) Furosemide-treated group (4 mg/kg) Groups treated with aqueous extract of Eysenhardtia polystachya (Ortega) Sarg at the doses of 125, 250, 500, and 750 mg/kg | ↑ Urinary flow rate (500 and 750 mg/kg) ↑ Urinary excretion of sodium (750 mg/kg) | [51] | |||
| Doradilla | Selaginella lepidophylla (Hook. & Grev.) Spring | Flavonoids and alkaloids | In vivo | Wistar female rats were induced to UL by administration of EG. After this period, the rats were divided into two groups (n = 6, each group): UL and UL + CE from Selaginella lepidophylla (Hook. & Grev.) Spring (50 mg/kg). | ↓↓↓ Urinary oxalic acid concentration compared to UL group ↑↑↑ Urinary flow rate ↑↑↑ Glomerular filtration rate in CE-treated compared with UL group ↑↑↑ Urinary excretions of sodium and potassium | [53] |
| In vivo | Female healthy Wistar rats were divided into six groups (n = 6, each group): Control group (water 1 mL/kg), furosemide group (4 mg/kg), group treated with aqueous extract from S. lepidophylla (200 mg/kg), and groups treated with different concentrations of alkaloid fraction from S. lepidophylla (10, 40, and 100 mg/kg). | ↑ Urinary excretion of sodium, potassium, and water in alkaloids fraction group | [55] | |||
| Diente de león | Taraxacum officinale L. | Tocopherols, phenols, flavonoids, saponins and sterols, including taraxasterol | In vitro | A model of CaOx crystallization in synthetic urine was generated by sodium oxalate. This was incubated with different concentrations of extract of Taraxacum officinale (1, 2, 4, and 8 mg/mL), taraxasterol (2.5, 5, 7.5, and 12.5 lg/mL), and PC (100, 150, 200, and 350 mg/mL) | ↓ Nucleation of crystals with extract and taraxasterol and extract in all doses, dose dependent ↓↓↓ Number of CaOx crystals in a dose-dependent manner with extract and taraxasterol ↓↓↓ Diameter of CaOx crystals with extract and taraxasterol | [57] |
| In vivo | A model of UL induced by ammonium chloride and EG in adult male Wistar albino rats. These were divided into the following groups (n = 6, each group): Control group, UL group, and UL with different concentrations of taraxasterol (2, 4, and 8 mg/kg), and UL with PC (2.5 g/kg). | Taraxasterol 2, 4, and 8 mg/kg and PC 2.5 g/kg ↓↓↓ Urinary oxalate levels. ↓↓ Score of inflammation in kidney Taraxasterol 2, 4, and 8 mg/kg ↓↓↓ Crystal deposits ↑↑ Urine pH ↑ Urinary citrate Taraxasterol 8 mg/kg: ↑↑ Superoxide dismutase and glutathione peroxidase in serum and kidney | [58] |
4. Molecular Mechanism of Bioactive Compounds Present in Medicinal Plants
4.1. Flavonoids
4.2. Phytosterols
4.3. Saponins
4.4. γ-Pyrones (Furanochromone Derivatives)
4.5. Alkaloids
4.6. Terpenoids
5. Materials and Methods
Literature Search
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aggarwal, K.P.; Narula, S.; Kakkar, M.; Tandon, C. Nephrolithiasis: Molecular mechanism of renal stone formation and the critical role played by modulators. BioMed Res. Int. 2013, 2013, 292953. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, J.K.; Teoh, J.Y.; Ng, C.F. Updates in endourological management of urolithiasis. Int. J. Urol. 2019, 26, 172–183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alelign, T.; Petros, B. Kidney Stone Disease: An Update on Current Concepts. Adv. Urol. 2018, 2018, 3068365. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, Y.; Zhang, J.; Deng, Q.; Liang, H. Recent advances on the mechanisms of kidney stone formation (Review). Int. J. Mol. Med. 2021, 48, 149. [Google Scholar] [CrossRef]
- Raheem, O.A.; Khandwala, Y.S.; Sur, R.L.; Ghani, K.R.; Denstedt, J.D. Burden of Urolithiasis: Trends in Prevalence, Treatments, and Costs. Eur. Urol. Focus 2017, 3, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Sorokin, I.; Mamoulakis, C.; Miyazawa, K.; Rodgers, A.; Talati, J.; Lotan, Y. Epidemiology of stone disease across the world. World J. Urol. 2017, 35, 1301–1320. [Google Scholar] [CrossRef] [PubMed]
- Medina-Escobedo, M.; Alcocer-Dzul, R.; López-López, J.; Villanueva-Jorge, S. Obesidad como factor de riesgo para alteraciones metabólicas en adultos con litiasis urinaria. Rev. Méd. Inst. Mex. Del Seguro Soc. 2015, 53, 692–697. [Google Scholar]
- Abeywickarama, B.; Ralapanawa, U.; Chandrajith, R. Geoenvironmental factors related to high incidence of human urinary calculi (kidney stones) in Central Highlands of Sri Lanka. Environ. Geochem. Health 2016, 38, 1203–1214. [Google Scholar] [CrossRef] [PubMed]
- Ziemba, J.B.; Matlaga, B.R. Epidemiology and economics of nephrolithiasis. Investig. Clin. Urol. 2017, 58, 299–306. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, P.M.; Taylor, E.N.; Gambaro, G.; Curhan, G.C. Dietary and Lifestyle Risk Factors Associated with Incident Kidney Stones in Men and Women. J. Urol. 2017, 198, 858–863. [Google Scholar] [CrossRef]
- Sorokin, I.; Pearle, M.S. Medical therapy for nephrolithiasis: State of the art. Asian J. Urol. 2018, 5, 243–255. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Hasan, M.M.; Alam Mahmood, Z. Antiurolithiatic plants: Formulations used in different countries and cultures. Pak. J. Pharm. Sci. 2016, 29, 2129–2139. [Google Scholar]
- Mutalib, L.Y. Comparison between phytotherapy and conventional drug therapy used in urolithiasis management in Hawler city, Kurdistan Region\Iraq. J. Pharmacogn. Phytochem. 2015, 4, 83–86. [Google Scholar]
- Bahmani, M.; Baharvand-Ahmadi, B.; Tajeddini, P.; Rafieian-Kopaei, M.; Naghdi, N. Identification of medicinal plants for the treatment of kidney and urinary stones. J. Ren. Inj. Prev. 2016, 5, 129–133. [Google Scholar] [CrossRef]
- Heinrich, M.; Ankli, A.; Frei, B.; Weimann, C.; Sticher, O. Medicinal plants in Mexico: Healers’ consensus and cultural importance. Soc. Sci. Med. 1998, 47, 1859–1871. [Google Scholar] [CrossRef]
- García-Alvarado, J.S.; Verde-Star, M.J.; Heredia, N.L. Traditional uses and scientific knowledge of medicinal plants from Mexico and Central America. J. Herbs Spices Med. Plants 2001, 8, 37–89. [Google Scholar] [CrossRef]
- Balaji, K.C.; Menon, M. Mechanism of stone formation. Urol. Clin. N. Am. 1997, 24, 1–11. [Google Scholar] [CrossRef]
- Khan, S.R. Reactive oxygen species, inflammation and calcium oxalate nephrolithiasis. Transl. Androl. Urol. 2014, 3, 256–276. [Google Scholar] [CrossRef] [PubMed]
- Mulay, S.R.; Linkermann, A.; Anders, H.J. Necroinflammation in Kidney Disease. J. Am. Soc. Nephrol. 2016, 27, 27–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wigner, P.; Grebowski, R.; Bijak, M.; Szemraj, J.; Saluk-Bijak, J. The Molecular Aspect of Nephrolithiasis Development. Cells 2021, 10, 1926. [Google Scholar] [CrossRef]
- Chaiyarit, S.; Thongboonkerd, V. Mitochondrial Dysfunction and Kidney Stone Disease. Front. Physiol. 2020, 11, 566506. [Google Scholar] [CrossRef]
- Birben, E.; Sahiner, U.M.; Sackesen, C.; Erzurum, S.; Kalayci, O. Oxidative stress and antioxidant defense. World Allergy Organ. J. 2012, 5, 9–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stowe, D.F.; Camara, A.K. Mitochondrial reactive oxygen species production in excitable cells: Modulators of mitochondrial and cell function. Antioxid Redox Signal 2009, 11, 1373–1414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Khan, S.R. Calcium oxalate crystal interaction with renal tubular epithelium, mechanism of crystal adhesion and its impact on stone development. Urol. Res. 1995, 23, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Wiessner, J.H.; Hasegawa, A.T.; Hung, L.Y.; Mandel, N.S. Oxalate-induced exposure of phosphatidylserine on the surface of renal epithelial cells in culture. J. Am. Soc. Nephrol. 1999, 10 (Suppl. S14), S441–S445. [Google Scholar]
- Okada, A.; Yasui, T.; Fujii, Y.; Niimi, K.; Hamamoto, S.; Hirose, M.; Kojima, Y.; Itoh, Y.; Tozawa, K.; Hayashi, Y. Renal macrophage migration and crystal phagocytosis via inflammatory-related gene expression during kidney stone formation and elimination in mice: Detection by association analysis of stone-related gene expression and microstructural observation. J. Bone Miner. Res. 2010, 25, 2701–2711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kusmartsev, S.; Dominguez-Gutierrez, P.R.; Canales, B.K.; Bird, V.G.; Vieweg, J.; Khan, S.R. Calcium Oxalate Stone Fragment and Crystal Phagocytosis by Human Macrophages. J. Urol. 2016, 195, 1143–1151. [Google Scholar] [CrossRef] [Green Version]
- Nikolic-Paterson, D.J.; Wang, S.; Lan, H.Y. Macrophages promote renal fibrosis through direct and indirect mechanisms. Kidney Int. Suppl. 2014, 4, 34–38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Singhto, N.; Thongboonkerd, V. Exosomes derived from calcium oxalate-exposed macrophages enhance IL-8 production from renal cells, neutrophil migration and crystal invasion through extracellular matrix. J. Proteom. 2018, 185, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.; Holmes, R.P.; Assimos, D.G.; Mitchell, T. Monocyte Mitochondrial Function in Calcium Oxalate Stone Formers. Urology 2016, 93, 224.e1–224.e6. [Google Scholar] [CrossRef] [Green Version]
- Patel, M.; Yarlagadda, V.; Adedoyin, O.; Saini, V.; Assimos, D.G.; Holmes, R.P.; Mitchell, T. Oxalate induces mitochondrial dysfunction and disrupts redox homeostasis in a human monocyte derived cell line. Redox Biol. 2018, 15, 207–215. [Google Scholar] [CrossRef]
- Dominguez-Gutierrez, P.R.; Kwenda, E.P.; Khan, S.R.; Canales, B.K. Immunotherapy for stone disease. Curr. Opin. Urol. 2020, 30, 183–189. [Google Scholar] [CrossRef]
- Khan, S.R.; Canales, B.K.; Dominguez-Gutierrez, P.R. Randall’s plaque and calcium oxalate stone formation: Role for immunity and inflammation. Nat. Rev. Nephrol. 2021, 17, 417–433. [Google Scholar] [CrossRef]
- Pedro, R.N.; Aslam, A.U.; Bello, J.O.; Bhatti, K.H.; Philipraj, J.; Sissoko, I.; Vasconcellos, G.S.; Trinchieri, A.; Buchholz, N. Nutrients, vitamins, probiotics and herbal products: An update of their role in urolithogenesis. Urolithiasis 2020, 48, 285–301. [Google Scholar] [CrossRef]
- Brahmachari, G.; Gorai, D.; Roy, R. Argemone mexicana: Chemical and pharmacological aspects. Rev. Bras. Farmacogn. 2013, 23, 559–567. [Google Scholar] [CrossRef] [Green Version]
- Chilivery, R.K.; Alagar, S.; Darsini, T.P. In Vitro Anti-Urolithiasis Potentials of Argemone mexicana L. Leaves. Curr. Clin. Pharmacol. 2016, 11, 286–290. [Google Scholar] [CrossRef]
- Ravi Kiran, C.; Rajkuberan, C.; Sangilimuthu, A.Y.; Hakkim, F.L.; Bakshi, H.; Manoharan, S.P. Intrinsic Evaluation of Antiurolithiatic Capacity of Argemone mexicana L. in Wistar Albino Rats. J. Herbs Spices Med. Plants 2021, 27, 289–304. [Google Scholar] [CrossRef]
- Brahmachari, G. Chemistry and Pharmacology of Naturally Occurring Bioactive Compounds; CRC Press: Boca Raton, FL, USA, 2013. [Google Scholar]
- Pérez Hernández, R.A.; Leos Rivas, C.; Oranday Cárdenas, A.; Hernández Luna, C.E.; Sánchez García, E.; Rivas Morales, C. Efecto in vitro en la inhibición del proceso de nucleación en litiasis renal, capacidad de captura de radicales libres, actividad antimicrobiana y tóxica del extracto metanólico de Berberis trifoliata. Rev. Mex. Cienc. Farm. 2015, 46, 70–76. [Google Scholar]
- Pérez-Hernández, R.A.; Treviño-Moreno, S.G.; Arévalo-Martínez, G.; Sánchez-García, E.; Leos-Rivas, C.; Rivas-Morales, C. Antiurolithic activity of Berberis trifoliate extract on induced urolithiasis in rats by zinc disc implantation. Afr. J. Tradit. Complementary Altern. Med. 2018, 15, 168–173. [Google Scholar] [CrossRef] [Green Version]
- Bashir, S.; Gilani, A.H.; Siddiqui, A.A.; Pervez, S.; Khan, S.R.; Sarfaraz, N.J.; Shah, A.J. Berberis vulgaris root bark extract prevents hyperoxaluria induced urolithiasis in rats. Phytother. Res. 2010, 24, 1250–1255. [Google Scholar] [CrossRef]
- Meléndez-Camargo, M.E.; Castillo-Nájera, R.; Silva-Torres, R.; Campos-Aldrete, M.E. Evaluation of the diuretic effect of the aqueous extract of Costus pictus D. Don in rat. In Proceedings of the Western Pharmacology Society; 2006; pp. 72–74. Available online: https://drive.google.com/file/d/1R02Y0x4wE0qJNRtiC6gvyUR4LTeQIUtL/view?usp=sharing (accessed on 1 February 2022).
- Kesavan, M.; Kaliaperumal, R.; Tamilmani, E.; Shanmugam, K. In vitro evaluation of calcium oxalate monohydrate crystals influenced by Costus igneus aqueous extract. Scand. J. Urol. Nephrol. 2012, 46, 290–297. [Google Scholar] [CrossRef]
- Manjula, K.; Rajendran, K.; Eevera, T.; Kumaran, S. Effect of Costus igneus stem extract on calcium oxalate urolithiasis in albino rats. Urol. Res. 2012, 40, 499–510. [Google Scholar] [CrossRef]
- Sharma, D.; Dey, Y.N.; Sikarwar, I.; Sijoria, R.; Wanjari, M.M.; Jadhav, A.D. In vitro study of aqueous leaf extract of Chenopodium album for inhibition of calcium oxalate and brushite crystallization. Egypt. J. Basic Appl. Sci. 2016, 3, 164–171. [Google Scholar] [CrossRef] [Green Version]
- Sikarwar, I.; Dey, Y.N.; Wanjari, M.M.; Sharma, A.; Gaidhani, S.N.; Jadhav, A.D. Chenopodium album Linn. leaves prevent ethylene glycol-induced urolithiasis in rats. J. Ethnopharmacol. 2017, 195, 275–282. [Google Scholar] [CrossRef]
- Khalil, N.; Bishr, M.; Desouky, S.; Salama, O. Ammi visnaga L., a potential medicinal plant: A review. Molecules 2020, 25, 301. [Google Scholar] [CrossRef] [Green Version]
- Khan, Z.A.; Assiri, A.M.; Al-Afghani, H.; Maghrabi, T. Inhibition of oxalate nephrolithiasis with Ammi visnaga (AI-Khillah). Int. Urol. Nephrol. 2001, 33, 605–608. [Google Scholar] [CrossRef]
- Charafi, S.; Kzaiber, F.; Hafid, A.; Berkani, M.; Oussama, A. Study of Ammi visnaga Lam. on oxalocalcic crystallization. Glob. J. Tradit. Med. Syst. 2012, 1, 7–12. [Google Scholar]
- Vanachayangkul, P.; Chow, N.; Khan, S.; Butterweck, V. Prevention of renal crystal deposition by an extract of Ammi visnaga L. and its constituents khellin and visnagin in hyperoxaluric rats. Urol. Res. 2011, 39, 189–195. [Google Scholar] [CrossRef] [Green Version]
- Vanachayangkul, P.; Byer, K.; Khan, S.; Butterweck, V. An aqueous extract of Ammi visnaga fruits and its constituents khellin and visnagin prevent cell damage caused by oxalate in renal epithelial cells. Phytomedicine 2010, 17, 653–658. [Google Scholar] [CrossRef] [Green Version]
- Perez, G.R.; Vargas, S.R.; Perez, G.S.; Zavala, S.M. Antiurolithiatic activity of Eysenhardtia polystachya aqueous extract on rats. Phytother. Res. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 1998, 12, 144–145. [Google Scholar]
- Pablo-Pérez, S.S.; Estévez-Carmona, M.M.; Meléndez-Camargo, M.E. Diuretic activity of the bark of Eysenhardtia polystachya. III. Bangladesh J. Pharmacol. 2016, 11, 212–217. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Campoy, A.; Garcia, E.; Muñiz-Ramirez, A. Phytochemical and Pharmacological Study of the Eysenhardtia Genus. Plants 2020, 9, 1124. [Google Scholar] [CrossRef]
- Mirian, E.-C.M.; Juanita, N.-M.; Christophe, B.O.; Estela, M.-C.M. Molecular mechanisms involved in the protective effect of the chloroform extract of Selaginella lepidophylla (Hook. et Grev.) Spring in a lithiasic rat model. Urolithiasis 2013, 41, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, M.I.; Benítez, W.V.; Colín, A.; Bye, R.; Ríos-Gómez, R.; Calzada, F. Evaluation of the diuretic activity in two Mexican medicinal species: Selaginella nothohybrida and Selaginella lepidophylla and its effects with ciclooxigenases inhibitors. J. Ethnopharmacol. 2015, 163, 167–172. [Google Scholar] [CrossRef] [PubMed]
- Melendez-Camargo, M.E.; Contreras-León, I.; Silva-Torres, R. Diuretic effect of alkaloids fraction extracted from Selaginella lepidophylla (Hook. et Grev.) Spring. Boletín Latinoam. Y Caribe Plantas Med. Y Aromáticas 2014, 13, 92–99. [Google Scholar]
- Kisiel, W.; Barszcz, B. Further sesquiterpenoids and phenolics from Taraxacum officinale. Fitoterapia 2000, 71, 269–273. [Google Scholar] [CrossRef]
- Yousefi Ghale-Salimi, M.; Eidi, M.; Ghaemi, N.; Khavari-Nejad, R.A. Inhibitory effects of taraxasterol and aqueous extract of Taraxacum officinale on calcium oxalate crystallization: In vitro study. Ren. Fail. 2018, 40, 298–305. [Google Scholar] [CrossRef] [Green Version]
- Yousefi Ghale-Salimi, M.; Eidi, M.; Ghaemi, N.; Khavari-Nejad, R.A. Antiurolithiatic effect of the taraxasterol on ethylene glycol induced kidney calculi in male rats. Urolithiasis 2018, 46, 419–428. [Google Scholar] [CrossRef]
- Monti, E.; Trinchieri, A.; Magri, V.; Cleves, A.; Perletti, G. Herbal medicines for urinary stone treatment. A systematic review. Arch. Ital. Urol. E Androl. 2016, 88, 38–46. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nirumand, M.C.; Hajialyani, M.; Rahimi, R.; Farzaei, M.H.; Zingue, S.; Nabavi, S.M.; Bishayee, A. Dietary plants for the prevention and management of kidney stones: Preclinical and clinical evidence and molecular mechanisms. Int. J. Mol. Sci. 2018, 19, 765. [Google Scholar] [CrossRef] [Green Version]
- Jucá, M.M.; Cysne Filho, F.M.S.; de Almeida, J.C.; Mesquita, D.d.S.; Barriga, J.R.d.M.; Dias, K.C.F.; Barbosa, T.M.; Vasconcelos, L.C.; Leal, L.K.A.M.; Ribeiro, J.E. Flavonoids: Biological activities and therapeutic potential. Nat. Prod. Res. 2020, 34, 692–705. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Xi, Y.; Jiang, W. Protective roles of flavonoids and flavonoid-rich plant extracts against urolithiasis: A review. Crit. Rev. Food Sci. Nutr. 2019, 59, 2125–2135. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Hasan, M.M.; Khan, H.; Mahmood, Z.A.; Patel, S. The mechanistic insight of polyphenols in calcium oxalate urolithiasis mitigation. Biomed. Pharm. 2018, 106, 1292–1299. [Google Scholar] [CrossRef]
- Ding, T.; Zhao, T.; Li, Y.; Liu, Z.; Ding, J.; Ji, B.; Wang, Y.; Guo, Z. Vitexin exerts protective effects against calcium oxalate crystal-induced kidney pyroptosis in vivo and in vitro. Phytomedicine 2021, 86, 153562. [Google Scholar] [CrossRef]
- Mandal, B.; Madan, S.; Ahmad, S.; Sharma, A.K.; Ansari, M.H.R. Antiurolithic efficacy of a phenolic rich ethyl acetate fraction of the aerial parts of Aerva lanata (Linn) Juss. ex Schult. in ethylene glycol induced urolithic rats. J. Pharm. Pharmacol. 2021, 73, 560–572. [Google Scholar] [CrossRef] [PubMed]
- Marhoume, F.Z.; Aboufatima, R.; Zaid, Y.; Limami, Y.; Duval, R.E.; Laadraoui, J.; Belbachir, A.; Chait, A.; Bagri, A. Antioxidant and Polyphenol-Rich Ethanolic Extract of Rubia tinctorum L. Prevents Urolithiasis in an Ethylene Glycol Experimental Model in Rats. Molecules 2021, 26, 1005. [Google Scholar] [CrossRef] [PubMed]
- Kangsamaksin, T.; Chaithongyot, S.; Wootthichairangsan, C.; Hanchaina, R.; Tangshewinsirikul, C.; Svasti, J. Lupeol and stigmasterol suppress tumor angiogenesis and inhibit cholangiocarcinoma growth in mice via downregulation of tumor necrosis factor-α. PLoS ONE 2017, 12, e0189628. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, P.; Patel, M.; Saralai, M.; Gandhi, T. Antiurolithiatic effects of Solanum xanthocarpum fruit extract on ethylene-glycol-induced nephrolithiasis in rats. J. Young Pharm. 2012, 4, 164–170. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, Y.; Su, R.; Zhang, W.; Yao, G.-L.; Chen, J. Antibacterial activity of tea saponin from Camellia oleifera shell by novel extraction method. Ind. Crops Prod. 2020, 153, 112604. [Google Scholar] [CrossRef]
- Peeters, L.; Foubert, K.; Breynaert, A.; Schreurs, G.; Verhulst, A.; Pieters, L.; Hermans, N. Effects of medicagenic acid metabolites, originating from biotransformation of an Herniaria hirsuta extract, on calcium oxalate crystallization in vitro. J. Ethnopharmacol. 2021, 285, 114860. [Google Scholar] [CrossRef]
- Chung, J.; Granja, I.; Taylor, M.G.; Mpourmpakis, G.; Asplin, J.R.; Rimer, J.D. Molecular modifiers reveal a mechanism of pathological crystal growth inhibition. Nature 2016, 536, 446–450. [Google Scholar] [CrossRef] [PubMed]
- Xueshibojie, L.; Duo, Y.; Tiejun, W. Taraxasterol inhibits cigarette smoke-induced lung inflammation by inhibiting reactive oxygen species-induced TLR4 trafficking to lipid rafts. Eur. J. Pharmacol. 2016, 789, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Sharifa, A.; Jamaludin, J.; Kiong, L.; Chia, L.; Osman, K. Anti-urolithiatic terpenoid compound from Plantago major Linn.(ekor anjing). Sains Malays. 2012, 41, 33–39. [Google Scholar]
- Nieber, K. The Impact of Coffee on Health. Planta Med. 2017, 83, 1256–1263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lobine, D.; Ahmed, S.; Aschner, M.; Khan, H.; Mirzaei, H.; Mahomoodally, M.F. Antiurolithiatic effects of pentacyclic triterpenes: The distance traveled from therapeutic aspects. Drug Dev. Res. 2020, 81, 671–684. [Google Scholar] [CrossRef] [PubMed]
- Guzmán, S.; Díaz, R.; González, M. Plantas Medicinales. La Realidad de una Tradición Ancestral; INIFAP: Celaya, México, 2020. [Google Scholar]
- Comisión Nacional Forestal. Plantas Medicinales de la Farmacia Viviente del CEFOFOR: Usos Terapéuticos y Dosificación, 1st ed.; SEMARNAT: Ciudad de México, México, 2020; pp. 48–49;58–59. [Google Scholar]
- Biblioteca Digital de la Medicina Tradicional Mexicana. Atlas de las Plantas de la Medicina Tradicional Mexicana. Ciudad de México, México. 2009. Available online: http://www.medicinatradicionalmexicana.unam.mx (accessed on 4 March 2022).
- Biodiversidad. Conocimiento de información especies y arboles. México. Available online: https://apps1.semarnat.gob.mx:8443/dgeia/informe_12/pdf/Cap4_biodiversidad.pdf (accessed on 4 March 2022).
- Salazar Rodríguez, M. Estudio Etnobotánico de Eysenhardtia Polystachya (Ortega) Sarg en una Comunidad del Municipio de Zempoala, Hidalgo y Evaluación del Efecto Diurético en Rata; Universidad Autónoma del Estado de Hidalgo: Pachuca, Hidalgo, México, 2007. [Google Scholar]
- Manzanilla Interián, A.M. Plantas Medicinales en Oxcutzcab, un Reencuentro Con Nuestras Raices Mayas; Universidad Autónoma Chapingo: Texcoco, Estado de México, México, 2009. [Google Scholar]
- Méndez, M.D.A.; Topacio, G.; Durán, R. Guía de la Colección de Plantas Medicinales del Banco de Germoplasma; Parque Cientifico Tecnológico de Yucatán: Mérida, Yucatán, México, 2016. [Google Scholar]
- Méndez, A.H.A.; López, M.; Morales, J. Herbolaria Oaxaqueña Para la Salud; Instituto Nacional de las Mujeres: Oaxaca, México, 2009.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sansores-España, D.; Pech-Aguilar, A.G.; Cua-Pech, K.G.; Medina-Vera, I.; Guevara-Cruz, M.; Gutiérrez-Solis, A.L.; Reyes-García, J.G.; Avila-Nava, A. Plants Used in Mexican Traditional Medicine for the Management of Urolithiasis: A Review of Preclinical Evidence, Bioactive Compounds, and Molecular Mechanisms. Molecules 2022, 27, 2008. https://doi.org/10.3390/molecules27062008
Sansores-España D, Pech-Aguilar AG, Cua-Pech KG, Medina-Vera I, Guevara-Cruz M, Gutiérrez-Solis AL, Reyes-García JG, Avila-Nava A. Plants Used in Mexican Traditional Medicine for the Management of Urolithiasis: A Review of Preclinical Evidence, Bioactive Compounds, and Molecular Mechanisms. Molecules. 2022; 27(6):2008. https://doi.org/10.3390/molecules27062008
Chicago/Turabian StyleSansores-España, Delia, Alfredo Geovanny Pech-Aguilar, Karol Guadalupe Cua-Pech, Isabel Medina-Vera, Martha Guevara-Cruz, Ana Ligia Gutiérrez-Solis, Juan G. Reyes-García, and Azalia Avila-Nava. 2022. "Plants Used in Mexican Traditional Medicine for the Management of Urolithiasis: A Review of Preclinical Evidence, Bioactive Compounds, and Molecular Mechanisms" Molecules 27, no. 6: 2008. https://doi.org/10.3390/molecules27062008
APA StyleSansores-España, D., Pech-Aguilar, A. G., Cua-Pech, K. G., Medina-Vera, I., Guevara-Cruz, M., Gutiérrez-Solis, A. L., Reyes-García, J. G., & Avila-Nava, A. (2022). Plants Used in Mexican Traditional Medicine for the Management of Urolithiasis: A Review of Preclinical Evidence, Bioactive Compounds, and Molecular Mechanisms. Molecules, 27(6), 2008. https://doi.org/10.3390/molecules27062008









