Characterization of the Phenolic Profile and Antioxidant Activity of Cathissa reverchonii (Lange) Speta
Abstract
:1. Introduction

2. Results and Discussion
2.1. Characterization of Phytochemicals in Methanolic Extracts
2.2. Quantification of Phenolic Compounds
2.3. Antioxidant Properties
2.4. Differences between Both Populations
3. Materials and Methods
3.1. Plant Materials
3.2. Extraction Techniques
3.3. Antioxidant Capacity
3.4. Analysis of Phenolic Compounds by HPLC-ESI-Q-TOF-MS
3.5. Quantification of Main Compounds by HPLC-DAD
3.6. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Stevens, P.F. Angiosperm Phylogeny Website. Version 14 July 2017. Available online: http://www.mobot.org/MOBOT/research/APweb/ (accessed on 4 February 2022).
- Engler, A. Liliaceae. In Die Natürlichen Pflanzenfamilien; Engler, A., Prantl, K., Eds.; W. Engelmann: Leipzig, Germany, 1888; Available online: https://bibdigital.rjb.csic.es/records/item/10928-die-naturlichen-pflanzenfamilien-ii-teil-5-abteilung (accessed on 4 February 2022).
- Zahariadi, C. Ornithogalum L. In Flora Europaea; Tutin, T.G., Heywood, V.H., Burges, N.A., Moore, D.M., Valentine, D.H., Walters, S.M., Webb, D.A., Eds.; Cambridge University Press: Cambridge, UK, 1980; Volume 5, pp. 35–40. [Google Scholar]
- Martínez Azorín, M.; Crespo, M.B.; Juan, A.; Fay, M.F. Molecular phylogenetics of subfamily Ornithogaloideae (Hyacinthaceae) based on nuclear and plastid DNA regions, including a new taxonomic arrangement. Ann. Bot. 2011, 107, 1–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tercero, A.; Benavente, A.; Rivas Rangel, A.; Lendínez Barriga, M.L.; Salazar, C. Ornithogalum reverchonii Lange ex Willk. In Atlas y Libro Rojo de la Flora Vascular Amenazada de España. Adenda 2017; Moreno Saiz, J.C., Iriondo Alegría, J.M., Martínez García, F., Martínez Rodríguez, J., Salazar Mendías, C., Eds.; Ministerio Para la Transición Ecológica-Sociedad Española de Biología de la Conservación de Plantas: Madrid, Spain, 2019; pp. 150–151. Available online: https://www.miteco.gob.es/es/biodiversidad/temas/inventarios-nacionales/inventario-especies-terrestres/inventario-nacional-de-biodiversidad/ieet_flora_vasc_adenda_2017_capitulos.aspx (accessed on 4 February 2022).
- Navarro Reyes, F.B. Ornithogalum L. In Flora Vascular de Andalucía Oriental, 2th ed.; Blanca, G., Cabezudo, B., Cueto, M., Morales Torres, C., Salazar, C., Eds.; Servicios de Publicaciones de las Universidades de Almería, Granada, Jaén and Málaga: Granada, Spain, 2011; pp. 153–156. Available online: https://bibdigital.rjb.csic.es/idurl/1/1526073 (accessed on 4 February 2022).
- Martínez Azorín, M.; Crespo, M.B.; Juan, A. Ornithogalum L. In Flora Iberica; Rico, E., Crespo, M.B., Quintanar, A., Herrero, A., Aedo, C., Eds.; Real Jardín Botánico: Madrid, Spain, 2013; pp. 188–207. [Google Scholar]
- European Union. Council Directive 92/43/EEC of 21 May 1992 on the Conservation of Natural Habitats and of Wild Fauna and Flora. OJ. 7 January 2013. 01992L0043, p. 1. Available online: https://eur-lex.europa.eu/legal-content/FR/TXT/PDF/?uri=CELEX:31992L0043&from=EN (accessed on 4 February 2022).
- Ministry of the Environment, and Rural and Marine Affairs. Royal Decree 139/2011 4 February, for the Development of the List of Wild Species in Regime of Special Protection and the Spanish Catalogue of Endangered Species. BOE Num. 46. 23 February 2011. Available online: https://www.boe.es/eli/es/rd/2011/02/04/139/con (accessed on 4 February 2022).
- Council of Andalusia. Decree 23/2012, of February 14, by Which Regulates the Conservation and Sustainable use of Wild Flora and Fauna and Their Habitats. BOJA Num. 60. 27 March 2012. Available online: https://www.juntadeandalucia.es/boja/2012/60/boletin.60.pdf (accessed on 4 February 2022).
- Speta, F. Die Echte und die Falsche Meerzwiebel: Charybdis Speta und Stellarioides Medicus (Hyacinthaceae), mit Neubeschreibungen und Neukombinationen im Anhang. Stapfia 2001, 75, 139–176. [Google Scholar]
- Zengin, G.; Uysal, S.; Ceylan, R.; Aktumsek, A. Phenolic constituent, antioxidative and tyrosinase inhibitory activity of Ornithogalum narbonense L. from Turkey: A phytochemical study. Ind. Crops Prod. 2015, 70, 1–6. [Google Scholar] [CrossRef]
- Renda, G.; Özel, A.; Turumtay, E.A.; Barut, B.; Korkmaz, B.; Çol Ayvaz, M.; Demir, A. Comparison of phenolic profiles and antioxidant activity of three Ornithogalum L. species. Turk. J. Biochem. 2018, 44, 299–306. [Google Scholar] [CrossRef]
- Temiz, M.A. Investigation of phenolic composition, antioxidant capacity, and antidiabetic effect of Ornithogalum lanceolatum L.: An in vitro study. Int. J. Second. Metabol. 2021, 8, 94–103. [Google Scholar] [CrossRef]
- Han, J.; Ye, M.; Qiao, X.; Xu, M.; Wang, B.-r.; Guo, D.-A. Characterization of phenolic compounds in the Chinese herbal drug Artemisia annua by liquid chromatography coupled to electrospray ionization mass spectrometry. J. Pharm. Biomed. Anal. 2008, 47, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Barreca, D.; Bellocco, E.; Caristi, C.; Leuzzi, U.; Gattuso, G. Kumquat (Fortunella japonica Swingle) juice: Flavonoid distribution and antioxidant properties. Food Res. Int. 2011, 44, 2190–2197. [Google Scholar] [CrossRef]
- Waridel, P.; Wolfender, J.-L.; Ndjoko, K.; Hobby, K.R.; Major, H.J.; Hostettmann, K. Evaluation of quadrupole time-of-flight tandem mass spectrometry and ion-trap multiple-stage mass spectrometry for the differentiation of C-glycosidic flavonoid isomers. J. Chromatogr. A 2001, 926, 29–41. [Google Scholar] [CrossRef]
- Geng, P.; Chen, P.; Lin, L.-Z.; Sun, J.; Harrington, P.; Harnly, J.M. Classification of structural characteristics facilitate identifying steroidal saponins in Alliums using ultra-high performance liquid chromatography high-resolution mass spectrometry. J. Food Comp. Anal. 2021, 102, 103994. [Google Scholar] [CrossRef]
- Jaramillo-Carmona, S.; Rodríguez-Arcos, R.; Jiménez-Araujo, A.; López, S.; Gil, J.; Moreno, R.; Guillén-Bejarano, R. Saponin profile of wild Asparagus species. J. Food Sci. 2017, 82, 638–646. [Google Scholar] [CrossRef] [Green Version]
- Van Hoyweghen, L.; De Bosscher, K.; Haegeman, G.; Deforce, D.; Heyerick, A. In vitro inhibition of the transcription factor NF-κB and cyclooxygenase by Bamboo extracts. Phytother. Res. 2014, 28, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Capanoglu, E.; Jassbi, A.R.; Miron, A. Advance on the Flavonoid C-glycosides and Health Benefits. Crit. Rev. Food Sci. Nutr. 2016, 56, S29–S45. [Google Scholar] [CrossRef] [PubMed]
- Angelino, D.; Berhow, M.; Ninfali, P.; Jeffery, E.H. Caecal absorption of vitexin-2-O-xyloside and its aglycone apigenin, in the rat. Food Funct. 2013, 4, 1339–1345. [Google Scholar] [CrossRef] [PubMed]
- Sorbo, S.; Basile, A.; Cobianchi, R. Antibacterial, antioxidant and allelopathic activities in bryophytes. Recent Res. Dev. Phytochem. 2004, 8, 69–82. [Google Scholar]
- Erel, S.B.; Karaalp, C.; Bedir, E.; Kaehlig, H.; Glasl, S.; Khan, S.; Krenn, L. Secondary metabolites of Centaurea calolepis and evaluation of cnicin for anti-inflammatory, antioxidant, and cytotoxic activities. Pharm. Biol. 2011, 49, 840–849. [Google Scholar] [CrossRef]
- El-Toumy, S.A.; Omara, E.A.; Nada, S.A.; Bermejo, J. Flavone C-glycosides from Montanoa bipinnatifida stems and evaluation of hepatoprotective activity of extract. J. Med. Plant Res. 2011, 5, 1291–1296. [Google Scholar]
- Osbourn, A.E. Molecules of interest, saponins in cereals. Phytochemistry 2003, 62, 1–4. [Google Scholar] [CrossRef]
- Sparg, S.G.; Light, M.E.; van Staden, J. Biological activities and distribution of plant saponins. J. Ethnopharmacol. 2004, 94, 219–243. [Google Scholar] [CrossRef]
- Howe, G.A.; Schilmiller, A.L. Oxylipin metabolism in response to stress. Curr. Opin. Plant Biol. 2002, 5, 230–236. [Google Scholar] [CrossRef]
- Özcan, M.M.; Doğu, S.; Uslu, N. Effect of species on total phenol, antioxidant activity and phenolic compounds of different wild onion bulbs. J. Food Meas. Charact. 2017, 12, 902–905. [Google Scholar] [CrossRef]
- Aşkın, D.; Şentürk, Ş.; Güler, E.M.; Doğan, A.; Omurtag, G.Z.; Ermanoğlu, M.; Taşkın, T. Biological activities of the Ornithogalum orthophyllum and its in silico ADMET profile. Tradit. Med. Res. 2021, 6, 56. [Google Scholar] [CrossRef]
- Llorent-Martínez, E.J.; Ortega-Vidal, J.; Ruiz-Riaguas, A.; Ortega-Barrales, P.; Fernández-de Córdova, M.L. Comparative study of the phytochemical and mineral composition of fresh and cooked broccolini. Food Res. Int. 2020, 129, 108798. [Google Scholar] [CrossRef] [PubMed]

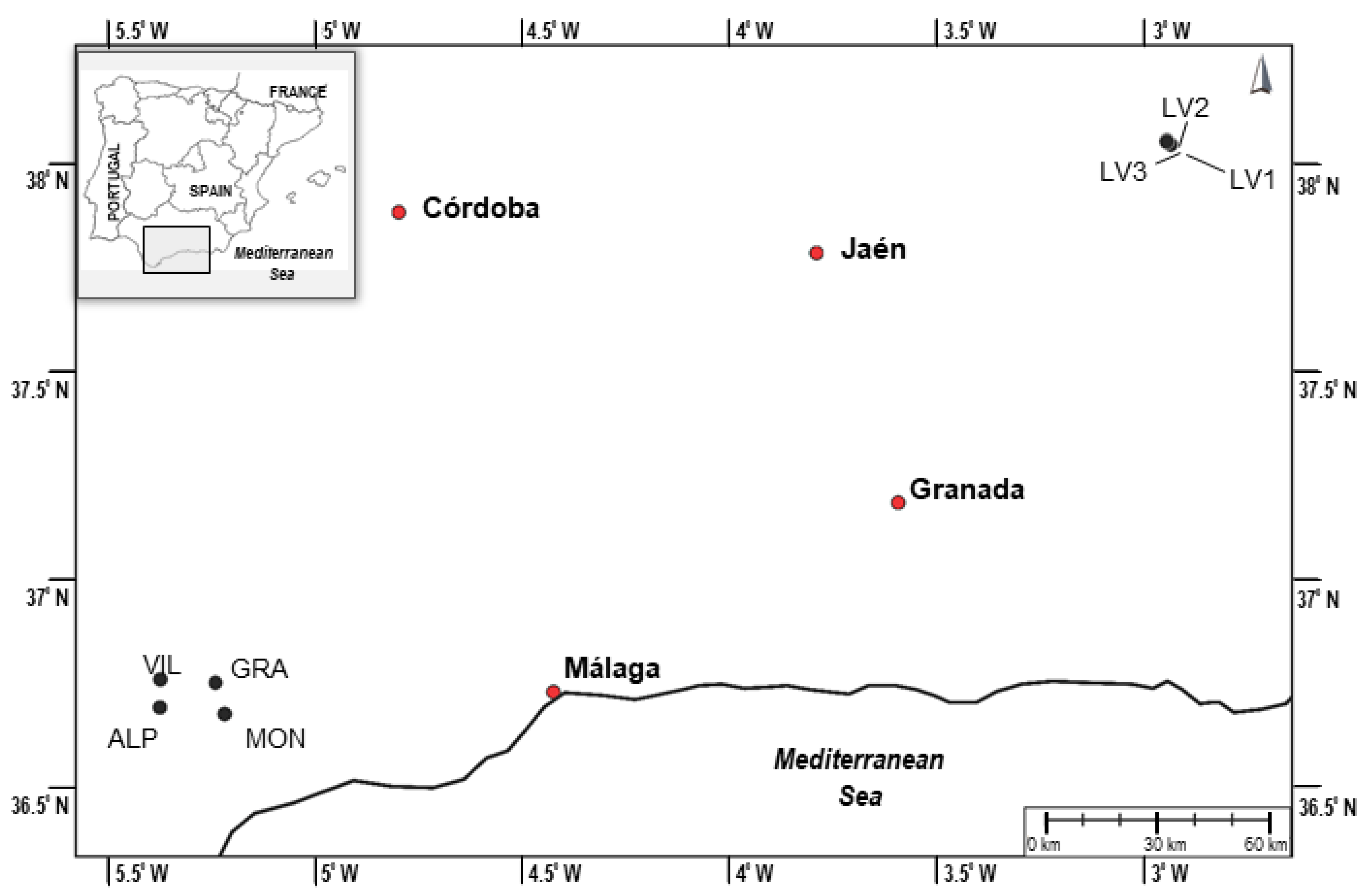
| Sample | Location | Geographical Coordinates | Altitude (m.a.s.l.) | Slope/ Aspect | Soil | Vegetation | T (°C) | RH (%) | P (MM) | SSM | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Population 1 | ALP | Puerto Encinas Borrachas (Alpandeire, Málaga) | 36°40′29.90″ N 5°12′58.10″ W | 970 | 60°/SE | Limestone rock | Rupicolous | 17.90 | 72.38 | 529 | 0.49 |
| GRA | Carretera Grazalema-Benamahoma (Grazalema, Cádiz) | 36°45′29.43″ N 5°22′16.68″ W | 893 | 90°/NE | Limestone rock | Rupicolous | 18.39 | 65.00 | 460 | 0.48 | |
| MON | Hundidero (Montejaque, Málaga) | 36°45′1.20″ N 5°14′18.10″ W | 812 | 70°/N | Limestone rock | Rupicolous | 17.24 | 65.69 | 456 | 0.49 | |
| VIL | Puerto de Las Viñas (Villaluenga del Rosario, Cádiz) | 36°41′25.62″ N 5°22′19.88″ W | 936 | 45°/E | Limestone rock | Rupicolous | 18.19 | 69.62 | 511 | 0.48 | |
| Population 2 | LV1 | Poyo Llano (Villacarrillo, Jaén) | 38° 2′35.02″ N 2°56′0.93″ W | 1295 | 5°/W | Limestone rock/Clay | Dry grassland | 16.49 | 57.75 | 361 | 0.45 |
| LV2 | Afluente Aguacebas Fuente del Tajo (Villacarrillo, Jaén) | 38° 3′0.02″ N 2°56′35.95″ W | 1100 | 0° | Clay | Wet grassland | 16.49 | 57.75 | 361 | 0.45 | |
| LV3 | Carril Senda de la Berrea (Villacarrillo, Jaén) | 38° 3′11.99″ N 2°56′34.30″ W | 1135 | 0° | Clay | Dry grassland | 16.49 | 57.75 | 361 | 0.45 |
| No. | tR (min) | Observed [M − H]− | Molecular Formula | Error (ppm) | Fragment Ions | Assigned Identification | ALP | GRA | MON | VIL | LV1 | LV2 | LV3 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1.76 | 341.1091 | C12H22O11 | −0.48 | 179.0556, 161.0470, 119.0346, 113.0239, 101.0241, 89.0246 | Disaccharide * | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 2 | 2.59 | 191.0200 | C6H8O7 | −1.22 | 173.0093, 129.0190, 111.0086, 87.0091 | Citric acid * | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 3 | 6.21 | 203.0826 | C11H12N2O2 | 0.24 | 186.0544, 159.0926, 142.0661, 116.0503, 74.0250 | Tryptophan * | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 4 | 9.51 | 625.1412 | C27H30O17 | −0.12 | 607.1285, 565.1206, 535.1089, 505.0986, 463.0870,415.0655, 385.0545 | Quercetin-di-C-glucoside | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 5 | 11.46 | 609.1465 | C27H30O16 | −0.51 | 591.1426, 519.1144, 489.1085, 399.0730, 369.0049 | Lucenin-2 (luteolin-6,8-di-C-glucoside) | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 6 | 16.99 | 447.0933 | C21H20O11 | −0.09 | 429.0844, 387.0596, 357.0618, 327.0519 | Isoorientin (luteolin-6-C-glucoside) | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 7 | 20.51 | 431.0985 | C21H20O10 | −0.09 | 341.0669, 311.0558, 283.0596 | Vitexin (apigenin-8-C-glucoside) | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 8 | 27.87 | 887.4646 | C44H72O18 | 0.09 | 741.4062, 609.3619, 447.3191 | Saponin-1 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | |
| 9 | 28.18 | 887.4643 | C44H72O18 | 0.4 | 741.4078, 609.3636, 447.3156 | Saponin-2 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 10 | 29.39 | 917.3518 | C37H54N14O10S2 | −0.22 | 838.3987, 458.1735 | Unknown | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 11 | 30.94 | 803.4426 | C40H68O16 | 1.01 | 757.4370, 611.3786, 449.3267 | Saponin-3 (formate adduct) | ✓ | ✓ | |||||
| 12 | 36.17 | 1065.5476 | C51H86O23 | 1.16 | 903.4958, 757.4346, 595.3835, 433.3329 | Saponin-4 | ✓ | ||||||
| 13 | 36.41 | 1065.546 | C51H86O23 | 2.31 | 903.4951, 757.4366, 595.3866, 433.3297 | Saponin-5 | ✓ | ✓ | |||||
| 14 | 37.23 | 903.496 | C45H76O18 | −0.05 | 757.4407, 595.3696, 427.5059 | Saponin-6 | ✓ | ||||||
| 15 | 38.76 | 327.2178 | C18H32O5 | −0.3 | 309.2053, 291.1955, 229.1442, 211.1334 | Trihydroxy-octadecadienoic acid | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| 16 | 39.05 | 695.3652 | C36H56O13 | −0.34 | 651.3744, 591.3528, 489.3220 | Unknown | ✓ | ||||||
| 17 | 40.26 | 329.2334 | C18H34O5 | −0.19 | 311.2233, 293.2109, 229.1442, 211.1337 | Trihydroxy-octadecenoic acid | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ |
| Nº | Assigned Identification | mg g−1 DE | ||||||
|---|---|---|---|---|---|---|---|---|
| ALP | GRA | MON | VIL | LV1 | LV2 | LV3 | ||
| Flavonoids | ||||||||
| 4 | Quercetin-di-C-glucoside | 1.8 ± 0.1 c | 2.3 ± 0.1 b | 1.8 ± 0.1 c | 2.0 ± 0.1 bc | 3.8 ± 0.3 a | 1.33 ± 0.09 d | 2.4 ± 0.2 b |
| 5 | Lucenin-2 | 5.1 ± 0.3 d | 5.8 ± 0.4 cd | 6.3 ± 0.4 bc | 6.5 ± 0.4 bc | 8.9 ± 0.5 a | 4.8 ± 0.3 d | 7.2 ± 0.4 b |
| 6 | Isoorientin | 0.71 ± 0.05 a | 0.41 ± 0.03 c | 0.60 ± 0.04 b | 0.55 ± 0.04 b | 0.18 ± 0.01 d | 0.11 ± 0.01 d | 0.10 ± 0.01 d |
| 7 | Vitexin | 1.03 ± 0.06 a | 0.47 ± 0.03 c | 0.68 ± 0.04 b | 0.62 ± 0.04 b | 0.18 ± 0.01 d | 0.12 ± 0.01 d | 0.10 ± 0.01 d |
| TIPC | 8.6 ± 0.3 c | 9.0 ± 0.4 bc | 9.4 ± 0.4 bc | 9.7 ± 0.4 bc | 13.1 ± 0.6 a | 6.4 ± 0.3 d | 9.8 ± 0.4 b | |
| Antioxidant Assay | ALP | GRA | MON | VIL | LV1 | LV2 | LV3 |
|---|---|---|---|---|---|---|---|
| ABTS radical-scavenging (g TE/100 g DE) | 2.0 ± 0.3 b | 2.4 ± 0.3 ab | 2.3 ± 0.1 ab | 2.2 ± 0.1 b | 2.9 ± 0.4 a | 1.9 ± 0.2 b | 1.9 ± 0.1 b |
| DPPH radical-scavenging (g TE/100 g DE) | 1.4 ± 0.2 bc | 1.7 ± 0.2 ab | 1.3 ± 0.2 bc | 1.2 ± 0.2 bc | 2.0 ± 0.3 a | 1.0 ± 0.1 c | 1.5 ± 0.2 abc |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Llorent-Martínez, E.J.; Gordo-Moreno, A.I.; Fernández-de Córdova, M.L.; Salazar-Mendías, C.; Tercero-Araque, A. Characterization of the Phenolic Profile and Antioxidant Activity of Cathissa reverchonii (Lange) Speta. Molecules 2022, 27, 1979. https://doi.org/10.3390/molecules27061979
Llorent-Martínez EJ, Gordo-Moreno AI, Fernández-de Córdova ML, Salazar-Mendías C, Tercero-Araque A. Characterization of the Phenolic Profile and Antioxidant Activity of Cathissa reverchonii (Lange) Speta. Molecules. 2022; 27(6):1979. https://doi.org/10.3390/molecules27061979
Chicago/Turabian StyleLlorent-Martínez, Eulogio J., Ana I. Gordo-Moreno, María Luisa Fernández-de Córdova, Carlos Salazar-Mendías, and Amanda Tercero-Araque. 2022. "Characterization of the Phenolic Profile and Antioxidant Activity of Cathissa reverchonii (Lange) Speta" Molecules 27, no. 6: 1979. https://doi.org/10.3390/molecules27061979
APA StyleLlorent-Martínez, E. J., Gordo-Moreno, A. I., Fernández-de Córdova, M. L., Salazar-Mendías, C., & Tercero-Araque, A. (2022). Characterization of the Phenolic Profile and Antioxidant Activity of Cathissa reverchonii (Lange) Speta. Molecules, 27(6), 1979. https://doi.org/10.3390/molecules27061979









