Abstract
The solvent extraction, complexing ability, and basicity of tetradentate N-donor 2,9-bis(5,5,8,8-tetramethyl-5,6,7,8-tetrahydro-1,2,4-benzotriazin-3-yl)-1,10-phenanthroline (CyMe4-BT- Phen) and its derivatives functionalized by Br, hydroxyphenyl, nitryl were discussed and compared. It was demonstrated that four BTPhen ligands are able to selectively extract Am(lll) over Eu(lll). It was notable that the distribution ratio of 5-nitryl-CyMe4-BTPhen for Eu(lll) was suppressed under 0.02, which was much lower compared to DEu(lll) = 1 by CyMe4-BTPhen. The analysis of the effect of the substituent on the affinity to lanthanides was conducted by UV/vis and fluorescence spectroscopic titration. The stability constants of various ligands with Eu(lll) were obtained by fitting titration curve. Additionally, the basicity of various ligands was determined to be 3.1 ± 0.1, 2.3 ± 0.2, 0.9 ± 0.2, 0.5 ± 0.1 by NMR in the media of CD3OD with the addition of DClO4. The basicity of ligands follows the order of L1 > L2 > L3 > L4, indicating the tendency of protonation decreases with the electron-withdrawing ability increase.
1. Introduction
In the reprocessing of spent fuel, minor actinides such as Americium, Neptunium, and Curium dominate the long-term radiotoxicity of high-level liquid waste (HLLW) after the PUREX reprocessing of uranium and plutonium. It is essential to shorten the lifetime of these radionuclides and minimize the volumes of waste for storage and isolation away from biosphere. One strategy currently being pursued is Partitioning and Transmutation (P&T) [1,2,3,4,5], whereby the radioactive nuclides are first separated from non-radioactive lanthanides by the Selective ActiNides EXtraction (SANEX) process and then converted into short-lived or stable elements by neutron-induced fission. This separation is indispensable since lanthanides with higher neutron capture cross sections will absorb neutrons in preference to actinides, resulting in an inefficient utilization of neutrons [6,7]. The separation of radioactive actinides from lanthanides is therefore a primary challenge in the process of nuclear fuel reprocessing for efficient transmutation. However, the similarities in chemical properties such as ionic radius and coordination models between two groups of f-block elements make the separation hard to achieve [8,9]. It is acknowledged that there is a greater availability of valence orbitals in the actinides leading to more covalent bonding natures in the actinide–ligand than those of lanthanides [10,11].
In recent years, many ligands have been developed and investigated for their extraction performance, coordination models, resistance to hydrolysis, and radiolysis [12,13,14,15]. It was found that ligands containing N-donor atoms or S-donor atoms could discriminate trivalent actinides from trivalent lanthanides. Due to second waste generated by α, β, and γ radiolysis during the use of ligands containing S-donor atoms, the development of N-donor ligands has been the priority. Among these N-donor ligands, 2,6-bis(5,6-dialkyl-1,2,4-triazin-3-yl) pyridines (BTPs) [16,17,18] and 6,6′-bis(5,6-dialkyl-1,2,4-triazin-3-yl)-2,2′-bipyridines (BTBPs) [19,20,21,22], have been extensively studied and considered the promising N-donor ligands for the continuous separation of actinides and lanthanides since they are able to directly extract actinides from nitric acid solution. It has been demonstrated in SANEX that BTBP is suitable for the separation of actinides and lanthanides at laboratory scale. Unfortunately, the kinetics of extraction using CyMe4-BTBP was found to be rather slow necessitating the addition of a phase-transfer agents such as TODGA (N,N,N’,N’-tetra-octyl-diglycolamide) or DMDOHEMA (N,N’-dimethyl-N,N’-dioctyl-2-hexylethoxymalonamide) to improve the extraction kinetics [20,23]. This disadvantage led to the development of CyMe4-BTPhen (L1 as depicted in Figure 1) [24]. In this molecule, the bipyridine moiety is replaced by a phenanthroline moiety thus prescribing the cis-conformation that is less favored in the BTBP ligand but essential for metal ion coordination. The reason for the improved kinetics may be attributed to a large dipole moment in the phenanthroline framework, making it able to coordinate with water via hydrogen bonds at the interface during its extraction [24]. The conformational rigidity and juxtaposition of N-donor atoms endows L1 optimum extraction thermodynamics and kinetics. Unfortunately, the affinity of L1 for Am is so high that it leads to difficult stripping of Am(lll) (HNO3 = 0.1 mol/L, DAm = 17, (Ligand) = 10 mmol/L in n-octanol). Based on that, ligands with Br and 4-hydroxyphenyl substitution at the 5 position (5-bromine-2,9-bis(5,5,8,8-tetramethyl-5,6,7,8-tetrahydro-1,2,4-benzotriazin-3-yl)-1,10-phenanthroline L2 (Figure 1) and 5-(4-hydroxyphenyl)-2,9-bis(5,5,8,8-tetramethyl-5,6,7,8-tetrahydro-1,2,4-benzotriazin-3-yl)-1,10-phenanthroline L3 (Figure 1) were synthesized and investigated for their extraction ability of actinides and lanthanides in this paper [25]. In order to gain further insight to elucidate reasons for the differences between these BTPhen ligands including a novel substituted structure L4 (Figure 1), the comparison of solvent extraction under same conditions, complexing ability, and protonation inclination were carried out in our work.

Figure 1.
Structures of four Bis-Triazine Phenanthrolines.
2. Results and Discussion
2.1. Solvent Extraction Study
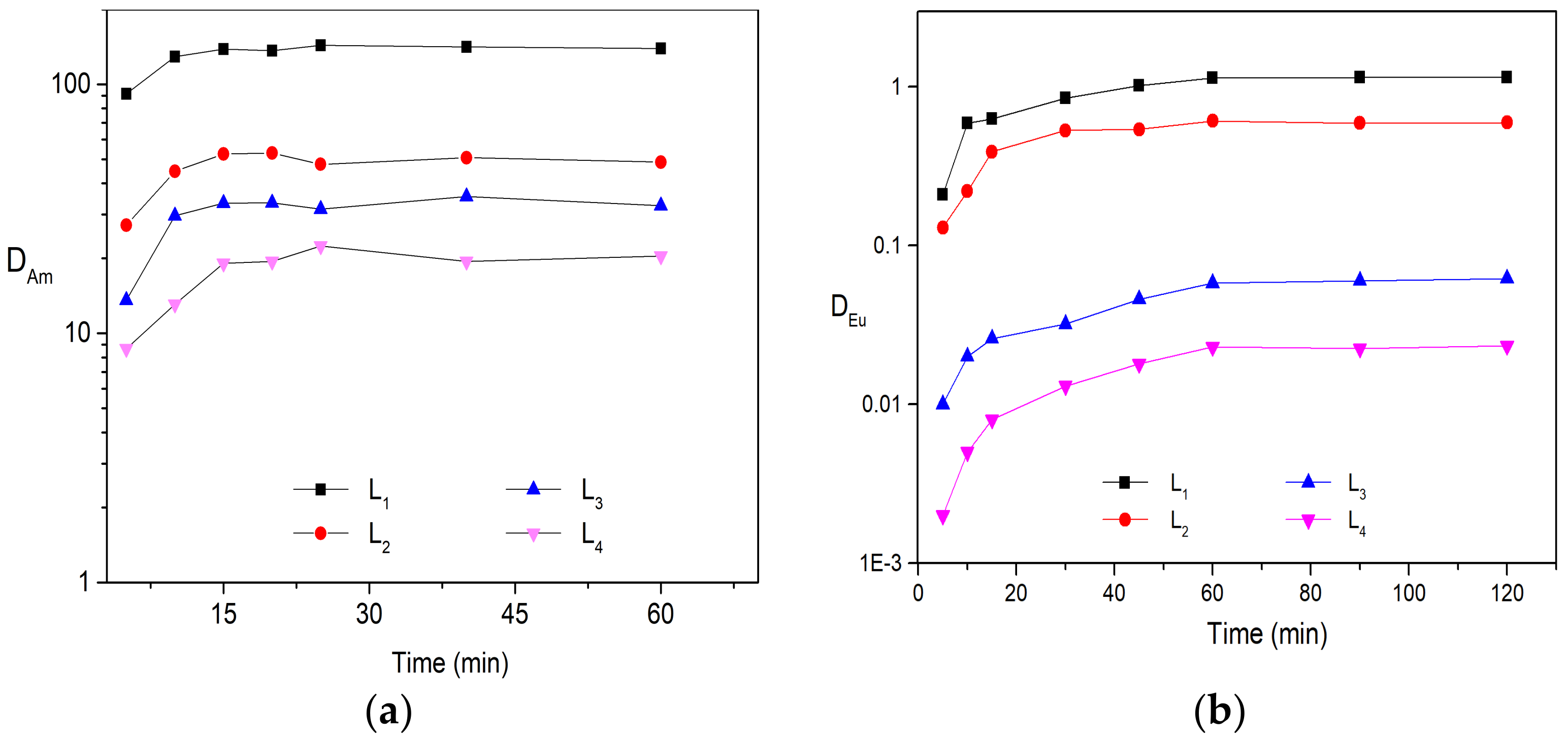
Liquid–liquid solvent extraction was carried out to determine the distribution ratio of Am (lll) and Eu(lll) by four substituted BTPhen ligands in the media of HNO3. The organic phases were prepared by dissolving four ligands into n-octanol and were contacted with nitric acid solution containing 241Am and inactive Eu, respectively. Various solvents (such as kerosene, 3-Nitrobenzotrifluoride, n-octanol, nitrobenzene, and dichloromethane) were tested for ligands; it turned out that n-octanol was the best selection for BTPhen ligands though L4 exhibited poor solubility in it. Based on this conclusion, 5 mmol/L extractant solution was employed for all ligands in order to compare the extraction performance under the same concentration. The dependence of distribution ratio on extraction balance time for Am (lll) and Eu(lll) was confirmed in advance to obtain the equilibrium time. The results indicate that the types of substitution have no influence on the dynamic extraction of Bis-Triazine Phenanthroline toward Am(lll) and Eu(lll). BTPhen ligand L1–L4 almost simultaneously reach extraction equilibrium for Eu(lll) after 60 min vibration and 15 min for Am(lll) as shown from Figure 2.
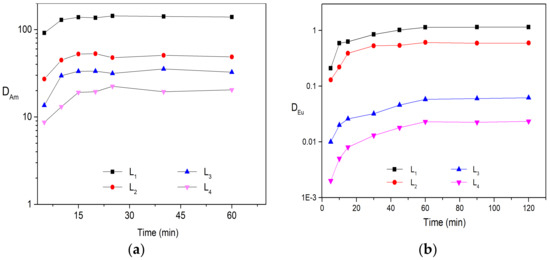
Figure 2.
Extraction results of Am and Eu: (a) Description of dependence of Am(lll) distribution ratios on balance time. Organic phase: 5 mmol/L BTPhen/n-octanol; Aqueous phase: trace amount of 241Am in 1 mol/L HNO3 solution, Contact time, 5–60 min; (b) Description of dependence of Eu(lll) distribution ratios on balance time. Organic phase: 5 mmol/L BTPhen/n-octanol; Aqueous phase: 0.1 mmol/L Eu (NO3)3 in 1 mol/L HNO3 solution, Contact time, 5–120 min (Temperature, 298 ± 0.1 K).
According to the extraction behaviors of four Bis-Triazine Phenanthroline ligands toward Am (lll) and Eu (lll), the distribution ratio value of Am by ligand L1 and L2 is considerably high even only 5 mmol/L ligand was employed in the organic phase. In addition, the affinity of ligands to Am is significantly stronger than Eu, SFAm/Eu for ligands L1, L2, L3, and L4 are 130, 83, 550, and 870, respectively, which is roughly in agreement with the results in the previous reports [24,25]. Due to excellent separation performance, extremely fast extraction rates, and strong extraction ability of ligand L1 for minor actinide (Am as representation) and lanthanides, it has been considered the promising candidate to treat HLLW in the SANEX process. However, the distribution ratio for Am even at low acidic conditions (DAm = 17, HNO3 = 0.1mol/L, (ligand) = 0.01 mol/L) is still very high leading to difficult stripping from extracted organic phase for subsequent step.
It has been reported that the extraction ability of Bis-Triazine Phenanthroline mainly originated from two phenanthroline-N atom coordinating with most lanthanide ions [26]. The average of metal–ligand bond length acquired from crystal structures such as Eu-Nphen = 2.515 is shorter than Eu-Ntriazinyl = 2.541, implying that there is a greater interaction between lanthanides ions and phenanthroline N-donor atoms than triazinyl N-donor atoms [26].
Quantum chemical calculation of complexes of various BTPhen ligands withAm/Eu also showed the similar results [27]. Thus, it might be effective to weaken the electronic density of phenanthroline skeleton via inductive effect exerted by substituted group Br, hydroxyphenyl, nitryl. It is observed that the order of affinity to metal ion from extraction results inversely follows the electron-withdrawing ability of substitution on phenanthroline: L1 > L2 > L3 > L4.
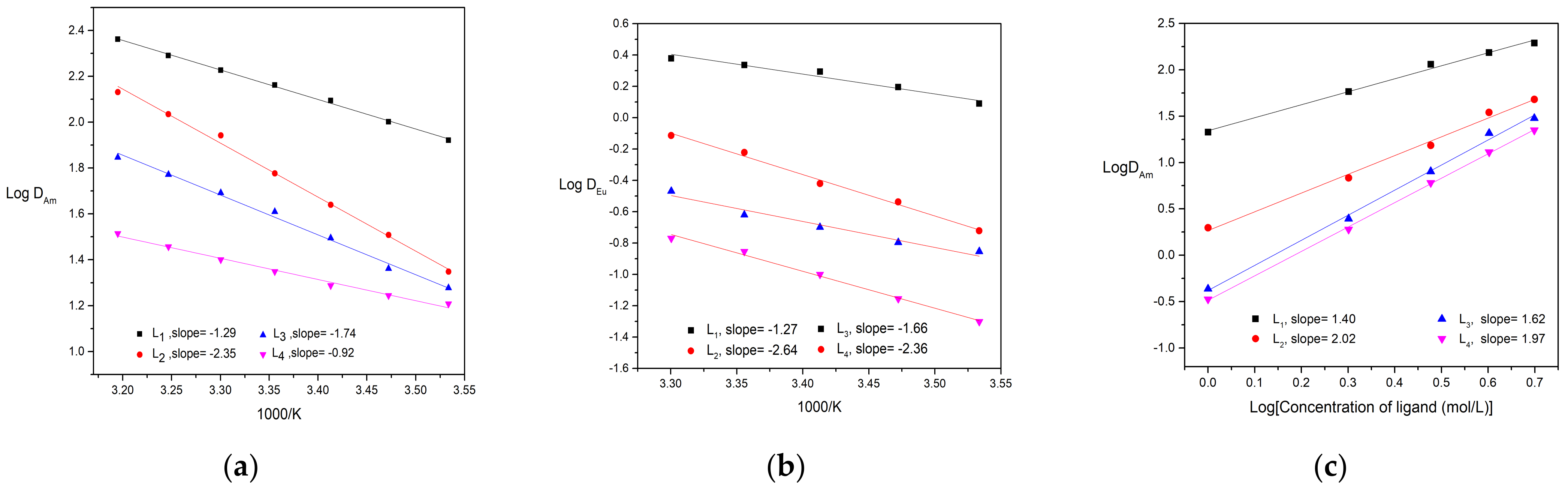
The dependence of distribution ratio of Am(lll) and Eu(lll) for ligands on aqueous nitric concentration was similar to CyMe4-BTPhen reported in the previous work [24]. As the initial concentration of HNO3 in the aqueous phase increases, the distribution ratio toward metal ion goes up at first and then slightly declines at 2 mol/L HNO3. The increase of distribution ratio at first might be accounted for the growing number of present, which is in favor of formation of complexes. With continuing increase in nitric acid, the proportion of protonated ligands increases, resulting in available ligands decreases. The influence of substituent on the protonation inclination of BTPhen ligands will be investigated in the following section. The extraction performance of Bis-Triazine Phenanthroline ligands for Am(lll) and Eu(lll) as a function of extraction temperature are presented in Figure 3. It turns out that the extraction process of Am(lll) and Eu(lll) by four ligands belong to endothermic reaction. To probe the extraction mechanism, the influence of ligand concentration on the distribution ratio of Am(lll) was carried out and depicted in Figure 3. The plots of Log DAm versus Log (L) of four types of BTPhen were obtained and gave four straight lines. Slopes analysis was employed to determine the number of ligands involved in the extracted complexes. The slopes of Ligand L1 and ligand L3 were around 2, indicating the type of 1:2 metal/ligand complexes during extraction by ligand L1 and L3 was formed. The crystal structure of [Eu(L1)NO3]2+ was obtained also verified our results [24]. Nevertheless, slopes of ligand L2 and L4, which differ from ligand L1 and L3, were between 1 and 2. Since crystal structure of maximum metal:ligand (1:2) of ligand 1 was formed, metal:ligand (2:3 or others) of homogeneous L2 and L4 was impossible due to the steric effect of many ligands. We considered that the formation of both 1:1 and 1:2 metal/ligand complexes was extracted into the organic phase during the extraction by ligand L2 and L4.
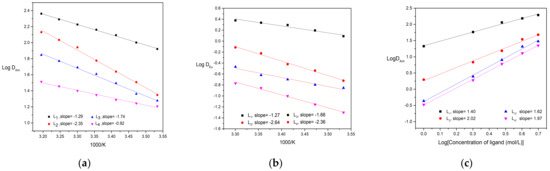
Figure 3.
Extraction results of Am and Eu–dependence of Am(lll) and Eu(lll) distribution ratios on extraction temperature and ligand concentration: (a) Description of dependence of Am(lll) on extraction temperature. Organic phase: 5 mmol/L BTPhen/n–octanol; Aqueous phase: trace amount of 241Am in 1 mol/L HNO3 solution (Temperature, 298 ± 0.1 K; Contact time, 20 min); (b) Description of dependence of Eu(lll) distribution ratios on extraction temperature. Organic phase: 5 mmol/L BTPhen/n–octanol; Aqueous phase: 0.1 mmol/L Eu(NO3)3 in 1 mol/L HNO3 solution. (Temperature, 298 ± 0.1 K; Contact time, 60 min); (c): Description of influence of ligand concentration on the extraction of Am(lll). Organic phase: 1–5 mmol/L BTPhen/n–octanol; Aqueous phase: trace amount of 241Am in 1 mol/L HNO3 solution (Temperature, 298 ± 0.1 K; Contact time, 20 min).
2.2. pKa Determination by NMR Titration
Since solvent extraction of reprocessing is accomplished in the media of HNO3 solution (2~3 mol/L), the basicity of ligands that determines the extent to which H+ ions compete with metal ions for ligands, is a very important property. Typically, the N-heterocyclics are readily prone to be protonated in highly acidic conditions. For example, Terpy(2,2′:6′,2′’-terpyridine) possesses the ability to extract Am from 0.1 mol/L HNO3 but fails to accomplish the extraction at 2 mol/L HNO3 [28,29]. This could be ascribed to highly nitric acid conditions caused by protonated Terpy, which is predominant, in which protonated ligands are transferred into the aqueous phase where their concentration in the organic phase is negligible in comparison to that of disprotonated species. However, the structures of easily protonated bilateral triazine and phenanthroline rings enable BTPhen ligands to extract metal ions from HNO3 solution. This phenomenon is mainly because the basicity of BTPhen ligands was strongly decreased by the conjugation effect between triazine and phenanthroline. So, it is meaningful to further investigate the effect of substitution on the basicity of BTPhen ligands.
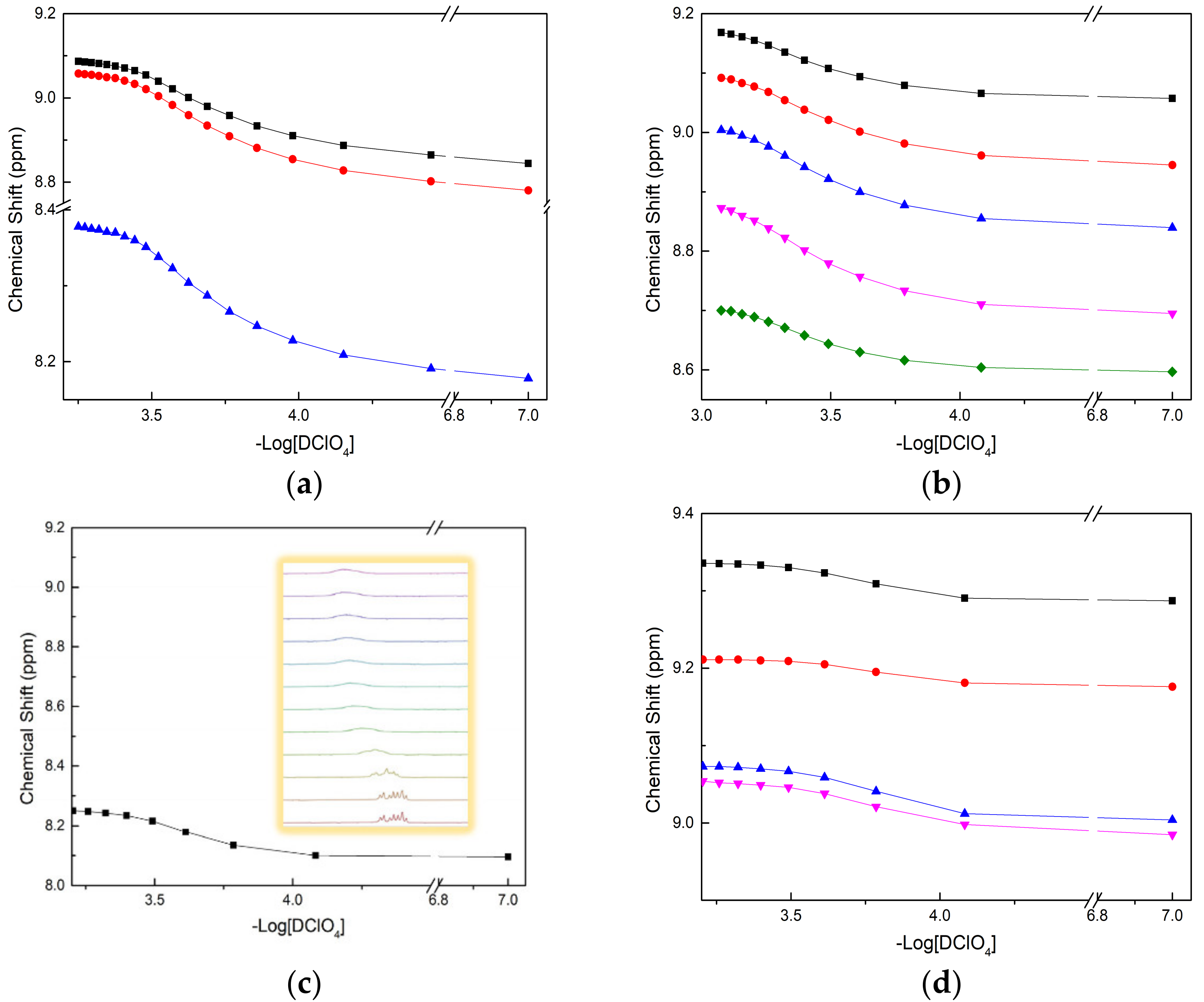
The protonation constants of four BTPhen ligands were determined by recording the chemical shift in H1 with the titration of DClO4 [30,31]. The titration was not suspended until the chemical shift in H1 remain unchanged. Different amounts of acid were added into each solution to simulate one step in a titration, in which each solution represented a point in a typical titration. The calculated protonation constants of four BTPhen ligands are presented in Table 1. As we can see from Table 1, the original large dipole moment of phenanthroline makes it able to coordinate with H+ even with the effect of withdrawing electrons by biliteral triazine [32]. The protonation process caused the alteration of the chemical environment of H3, H4, H5, H6, H7, and H8 at the corresponding carbon C3, C4, C5, C6, C7, and C8. The chemical shift results of H5 or H6 on phenanthroline versus -Log [DClO4] are presented in Figure 4. Due to the symmetric property of L1, there are only three types of peak signal whose chemical shifts were visible in the spectra. As for L2, L3, and L4, chemical environment change caused by different substituted groups result in five types of peak signal appeared in the map. The reason for only one plot of chemical shifts presented in Figure 4c was that the shift signal of hydrogen on phenanthroline developed into unimodal from multimodal, which it is unable to identify. The peculiar phenomenon of peak signal of H in L4, which was weak at first, and then vanished, makes it hard to distinguish.

Table 1.
Protonation constant of N-heterocyclics.
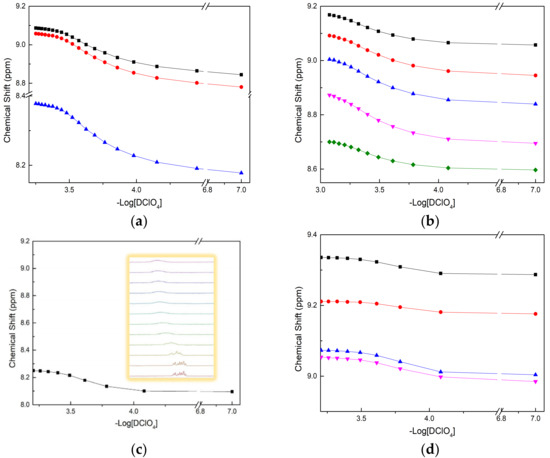
Figure 4.
Plot of chemical shift (δ, ppm) as a function of pD for the H1 signals with addition of DClO4 in CD3OD: (a) Description of chemical shift in L1 (■—H at C3(or 8), ●—H at C4(or 7), ▲—H at C5 (or 6)); (b) Description of chemical shift in L2 (■—H at C8, ●—H at C3, ▲—H at C7, ▼—H at C4, ◆—H at C6); (c) Description of chemical shift in L3 (■—H at C6); (d) Description of chemical shift in L4 (■—H at C8, ●—H at C3, ▲—H at C7, ▼—H at C4).
2.3. UV–vis Titration
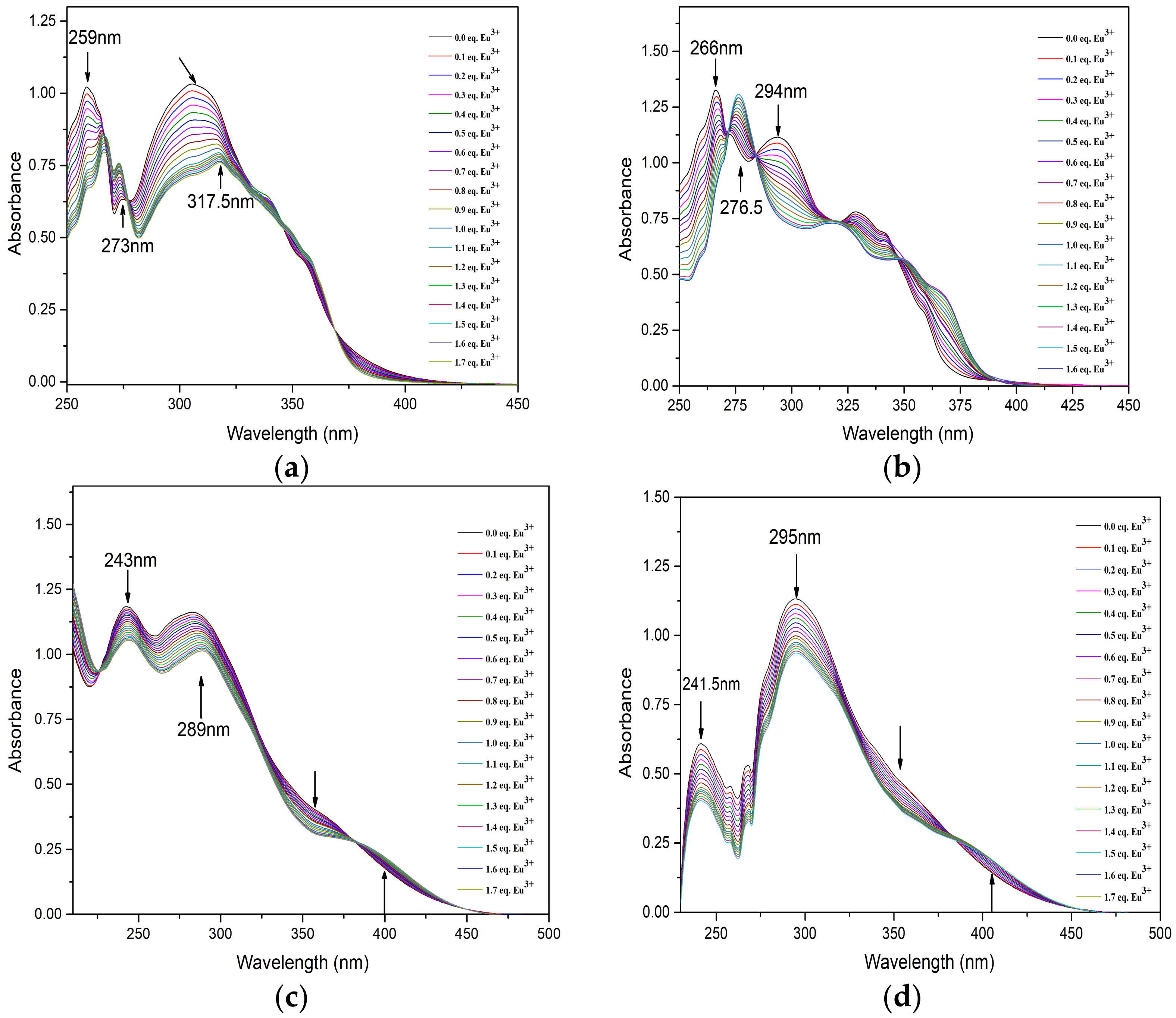
The complexation behavior of BTPhen ligands with lanthanides in methanol was investigated by UV–vis titration. The alteration of absorbance intensity of solution as a function of Eu(lll) concentration is shown in Figure 5. The absorption spectra of four ligands during the Ultra Visible region of 200–300 nm are dominated by the charge transfer of π–π* transition from the aromatic nature of the BTPhen structure. As Eu(lll) concentration increased in the L1 solution, the absorbance of ligands at 259 nm sharply decreased. The new peak at 265 nm and 273 nm emerged with the addition of up to 0.5 equivalent of Eu3+ corresponding to the M–L complex as presented in Figure 5a.
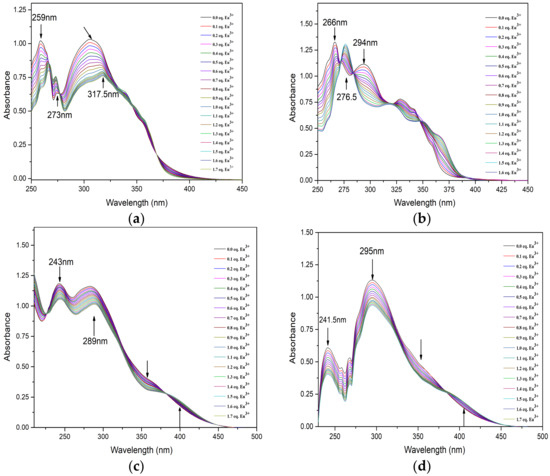
Figure 5.
UV–visible absorption spectroscopic titrations of ligands ((a) for L1, (b) for L2, (c) for L3, (d) for L4) complexing with Eu3+ in CH3OH solution (T = 298 ± 0.1 K, I = 0.01 M Et4NNO3, initial condition (L1~L4) = 2 × 10−5 mol/L, Vini = 2.00 mL, titrant condition [Eu(NO3)3] = 2 × 10−4 mol/L).
The increase in the absorption intensity for the new peak with the addition of Eu(lll) was also observed. The absorption at 265 nm shifted to 266 with the addition of Eu(lll) solution. It also could be seen that the peak at 317.5 nm apparently begun to form after one equivalent Eu3+ solution was added in the ligand solution. This indicates the 1:2 Eu3+/L1 complexes were formed. As for L2, sharp peak of spectrum at 266 nm and wide peak at 294 nm was obviously decreased as shown from Figure 5b. The absorption intensity at 276.5 nm was gradually enhanced and red-shifted upon the addition of Eu3+. This alteration on the behavior of spectra was analogous to the titration of L1. Similar results have been observed in the titration of L3 and L4, but more persuasive changes happened in the region from 325 to 425 nm. Moreover, two or more isosbestic points on every titration curve were generated, which confirms that more than one type of complex was formed upon the addition of Eu(lll). The stability constants of the 2:1 species of BTPhen ligands with Eu(lll) were determined to be 14.2 ± 0.5, 11.5 ± 0.3, 8.3 ± 0. 6, and 7.1 ± 0.4, respectively. It was found that the stability constants of Logβ12 the 2:1 complex formed between BTPhen ligands and trivalent europium ions increased from L4, L3, L2, to L1. This trend in the complexation strength agrees well the extraction properties of BTPhen for Eu as displayed in Figure 2a. It is reasonable to conclude that withdrawing substitution functionalized on the phenanthroline framework significantly decreased the complex ability of BTPhen ligands for trivalent lanthanides.
2.4. Fluorescence Spectroscopy
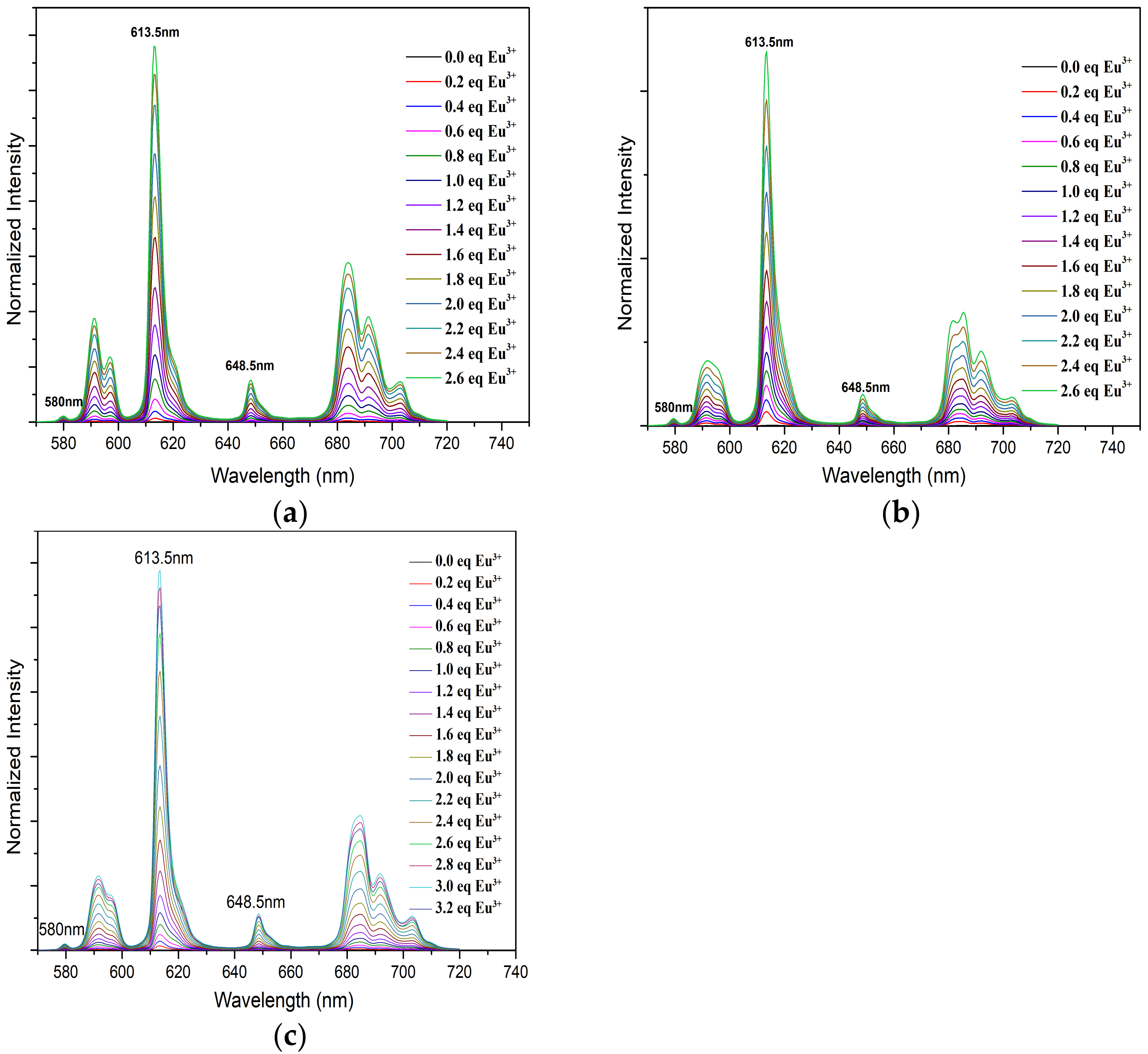
The fluorescence spectra of Eu(III) with CyMe4-BTPhen (a), 5-Br-CyMe4-BTPhen (b), and 5-(nitryl)-CyMe4-BTPhen (c) were illustrated in Figure 6. It is necessary to mention that there is no intensity increase in full spectra with the addition 5-(4-hydroxyphenyl)- CyMe4-BTPhen solution. The rigid skeleton and conjugation effect of phenanthroline should have enhanced the fluorescence intensity of Eu(lll) solution as observed in the titration of L1, L2, and L4. Nevertheless, the intensity of the spectra was nearly stationary while its complexation ability with Eu(lll) was stronger than L4 according to the investigation by UV/Vis. The reason for this “paradoxical phenomenon” in 5-(4-hydroxyphenyl)-CyMe4-BTPhen is still not fully understood and remains unsolved. The titration by Ligand L1, L2, and L4 exhibited remarkable enhancement in intensity of emissions across the addition of the corresponding ligand solution. A strongest intensity increase compared to the original Eu(lll) solution at 613.5 nm was observed resulting from 5D0→7F2 transitions [35]. The change in the remaining emission peaks in typical fluorescence spectra Eu (lll), which is at 580 nm, 594 nm, 648.5 nm, and 702 nm corresponding to 5D0→7F0, 5D0→7F1, 5D0→7F3, and 5D0→7F4 is relatively implicit [36,37]. The intensity plateau was reached for L1 and L2 after 2.6 μL equivalent ligands was added. As for L4, the spectra intensity did not stop growing until 2.4 μL of equivalent ligands was added. The stability constants of Eu(lll) with ligand 1, ligand 2, and ligand 4 by fitting spectra at 613.5 nm were determined to be 12.6 ± 0.4, 12.1 ± 0.8, and 8.6 ± 0.2, respectively. The calculated value of L1 with Eu(lll) was consistent with the previous report as shown from Table 2 [26]. The decrease in complexation ability of BTPhen ligands (except L3) with Eu(lll) indicates the affinity of ligands to lanthanides as the electron-withdrawing ability grows. This tendency was the manifestation of extraction results of BTPhen ligands toward Eu(lll).
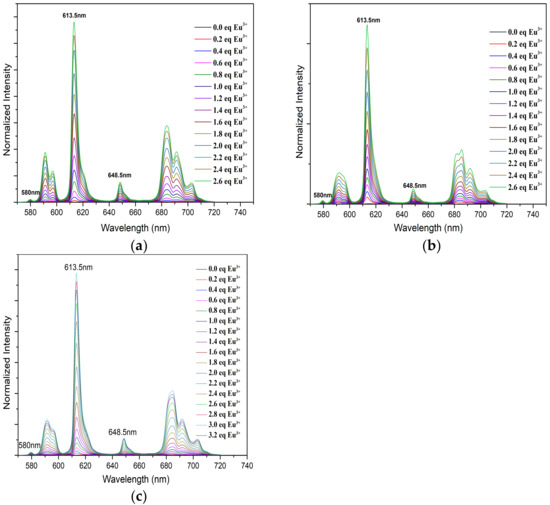
Figure 6.
Normalized fluorescence emission spectra of Eu(III) in methanol with increasing BTPhen concentration. (Eu(lll))ini = 1 × 10−5 mol/L; Vini = 2 mL; (L1, L2, L4) = 1 × 10−3 mol/L; 4 μL of ligand solution was added at one time. (a) Description of emission spectra of Eu (lll) with L1; (b) Description of emission spectra of Eu(lll) with L2; (c) Description of emission spectra of Eu(lll) with L4.

Table 2.
Conditional stability constants of BTPhen ligands with Eu(lll).
3. Materials and Methods
3.1. Syntheses
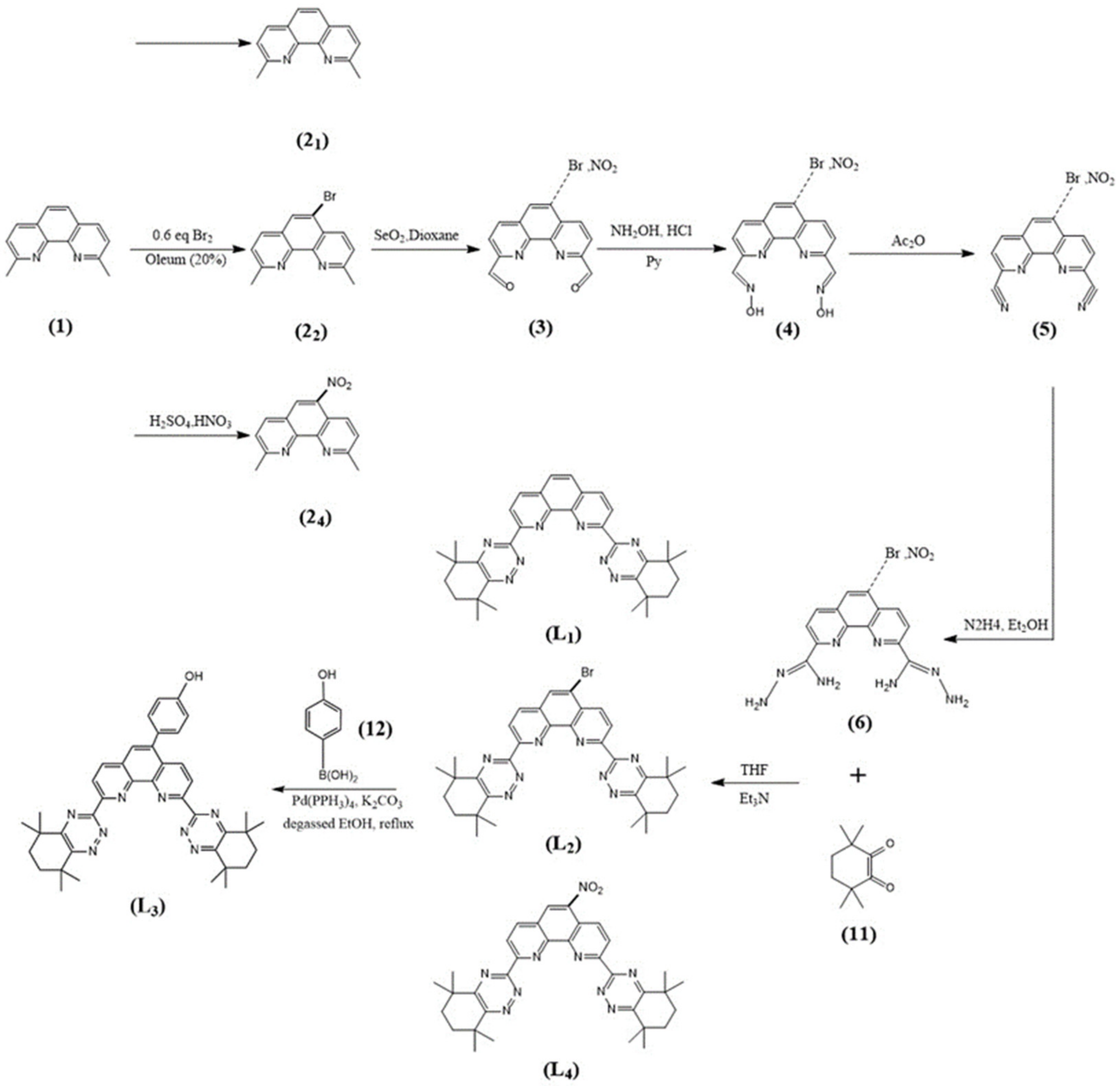
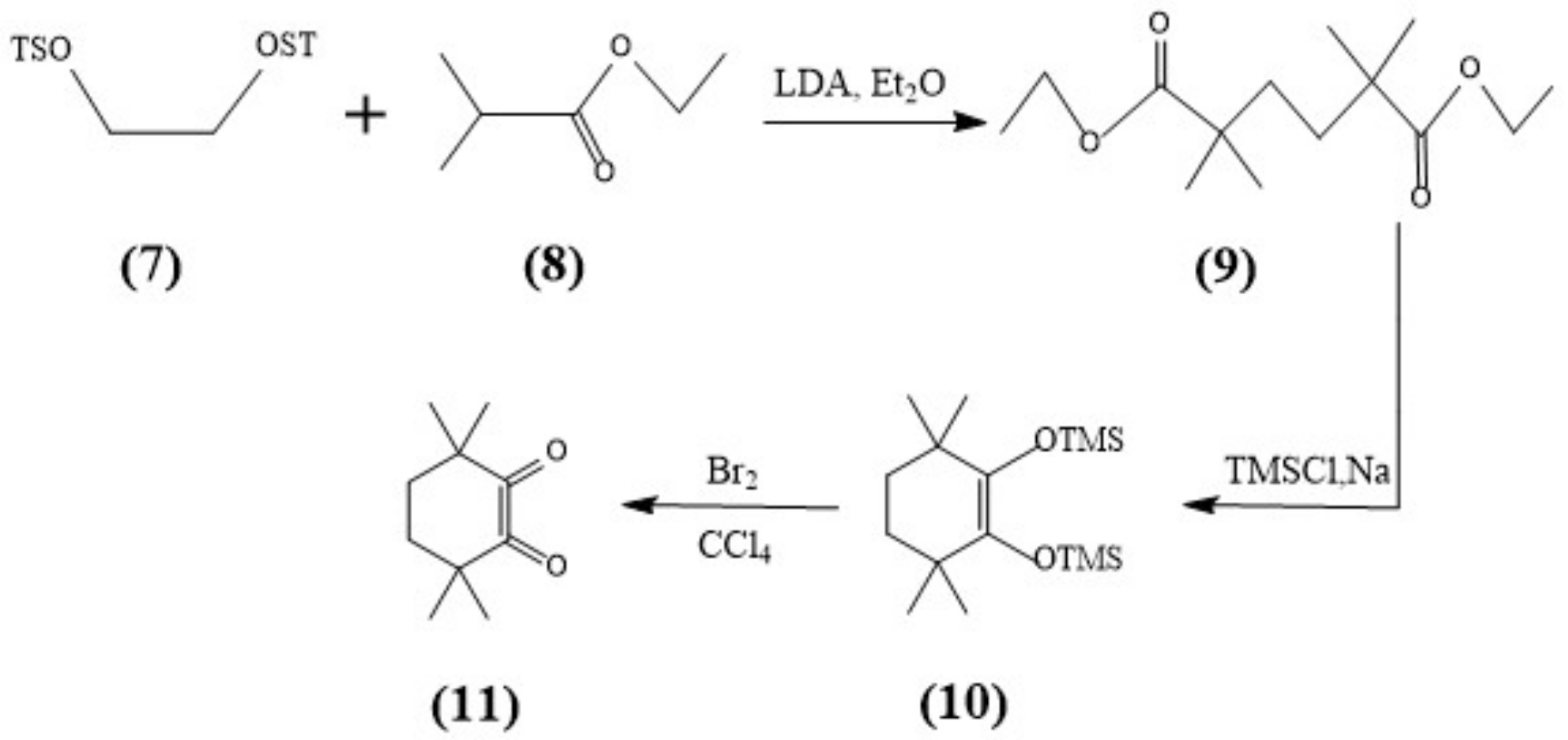
The syntheses of CyMe4-BTPhen, 5-Br-CyMe4-BTPhen, and 5-(4-hydroxyphenyl)-CyMe4-BTPhen were achieved according to the route described by Lewis [24] and Afsar [25]. The difference in synthesis between 5-nitryl-CyMe4-BTPhen and CyMe4-BTPhen is 2,9-dimethyl-1,10-phenanthroline needs to be nitrified in the media of sulfuric acid preceding the synthesis of dialdehyde. The schemes of synthesis route of ligands were depicted in Figure 7 and Figure 8.
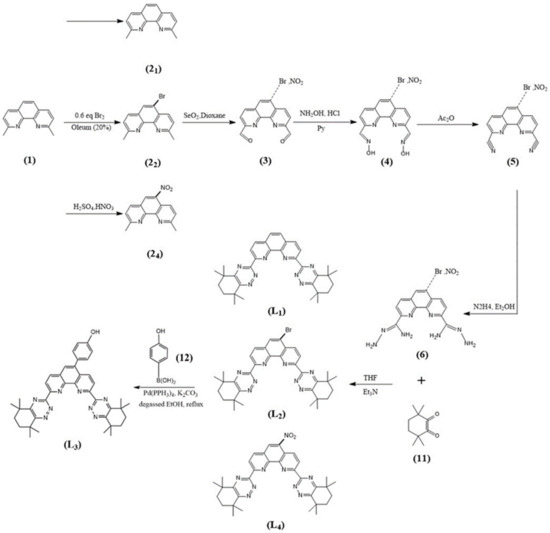
Figure 7.
Syntheses of the BTPhen Ligand L1, L2, L3, and L4.
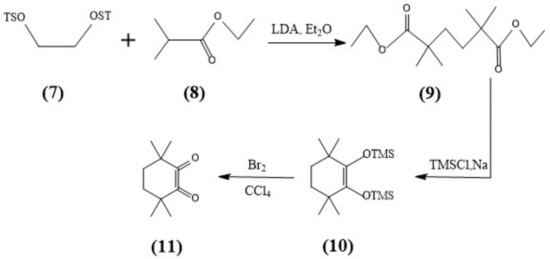
Figure 8.
Synthesis of the Diketone (11).
CyMe4-BTPhen: 1H NMR (400 MHz, CDCl3, ppm) δ 8.92 (d, J = 8.3 Hz, 2H), 8.61 (d, J = 8.4 Hz, 2H), 8.03 (s, 2H), 2.85 (s, 8H), 1.90 (s, 8H), 1.56 (d, J = 12.7 Hz, 24H). FT-IR (KBr, ν/cm-): 2864.48, 2932.80, 2960.50, 1363.05, 1385.681, 1151.89, 1249.23, 1640.78, 706.25, 1456.41, 1589.47, 1511.26, 840.33, 859.64, 869.89 MS(m/z):559.3298(M+H+), 581.3123(M+ Na+).
5-Br-CyMe4-BTPhen:1H NMR (400 MHz, CDCl3, ppm) δ 8.95 (d, J = 8.6 Hz, 1H), 8.89 (dd, J = 11.3, 8.4 Hz, 2H), 8.40 (d, J = 8.4 Hz, 1H), 8.29 (s, 1H), 2.62–2.26 (m, 8H), 1.90 (d, J = 2.7 Hz, 8H), 1.55 (dd, J = 11.9, 3.0 Hz, 24H).FT-IR (KBr, ν/cm-1): 2864.57, 2928.94, 2961.82, 1388.91, 1363.07, 1245.99, 1143.34, 705.44, 1641.98, 1514.61, 1602.92, 1602.9, 904.66, 866.63, 830.61, 632.65 MS(m/z):637.2409(M+H+),659.2231(M+Na+).
5-nitryl-CyMe4-BTPhen. 1H NMR (400 MHz, CDCl3, ppm) δ 9.24 (d, J = 8.8 Hz, 1H), 8.98 (dd, J = 21.1, 8.6 Hz, 2H), 8.81 (s, 1H), 8.64 (d, J = 8.3 Hz, 1H), 2.48 (s, 7H), 1.91 (s, 8H), 1.60–1.53 (m, 24H). FT-IR (KBr, ν/cm-1): 2863.24, 2929.24, 2960.67, 1363.72, 1384.77, 1123.05, 1247.46, 1508.88, 1630.01, 704.28cm-1, 1489.12, 1589.07, 843.80, 832.72, 771.54 MS(m/z): 604.3143 (M+H+), 626.2973 (M+ Na+).
5-(4-hydroxyphenyl)-CyMe4-BTPhen. 1H NMR (400 MHz, CDCl3, ppm): δ 8.89-8.90 (s,1H), δ 8.78-8,79(s,1H), δ 8.42-8,44 (s,1H), δ 8.30-8,31(s,1H), δ 7.68(s,1H), δ 6.64-6.66(d,2H), δ 6.48-6.49 (d,2H), δ 1.95 (m,8H), δ1.60-1.63(m,24H). FT-IR (KBr, ν/cm-1): 2864.42, 2928.68, 2960.25, 1368.76, 1388.07, 1144.54, 1244.18, 1612.78, 709.24, 1520.01, 1455.07, 1552.62, 839.13, 859.64, 869.89. MS(m/z): 651.3559(M+H+), 673.3379(M+ Na+).
3.2. Solvent Extraction
All the reagents supplied by Aladdin were used as received without further purification unless otherwise stated. Equal volumes (1 mL) of organic phase and aqueous phase were stirred for the extraction of Am(lll) and Eu(lll). The extraction of Am from HNO3 solution was performed at 298 ± 0.1 K. Nitric acid solution spiked with 241Am tracer amount was mixed with 0.5 mmol/L BTPhen ligands dissolved in n-octanol by Vortex oscillator for desired time (max. 120 min and min. 5 min). Temperature controlled extraction was carried out in the water bath magnetic stirrer with magneton in the plastic vials to maintain constant temperature during extraction. As for extraction of Eu(lll), it was conducted in the nonradioactive condition. The mixture for both Am and Eu extraction was centrifuged for 5 min to separate phases. The activity of 241Am and amount of Eu were separately determined by Tricarb 2910 tr liquid scintillator (Perkin Elmer company) and inductively coupled plasma atomic emission spectrometer ICP-AES. Distribution ratio of Am is determined by the ratio between the radioactivity counts of organic phase and aqueous phase after extraction. Distribution ratio of Eu(lll) is defined by the concentration of the Eu3+ in the organic phase divided by that in the aqueous phase. The separation factor (SFAm/Eu) was calculated by the ratio of distribution ratios between Am(lll) and Eu(lll).
3.3. pKa Determination
The protonation behavior of Bis-Triazine Phenanthrolines in the acid was investigated by NMR titration. Chemical shift was recorded by adding 0.1 mol/L DClO4 into 2 mmol/L ligand/ CD3OD solution. In the NMR measurements (ADVANCEⅢ, Bruker BioSpin GmbH), the magnetic field was stabilized by locking it to the D signal of the solvent. The sample temperatures were regulated to be 295 ± 0.1 K during all acquisitions. The titration was completed with addition of 90 μL L1, 65 μL L2, 60 μL L3, and 55 μL L4 with 5 μL at each time. The binding constants were determined using the hypNMR2008 software [39].
3.4. UV–vis Spectrometric Titration
The UV–vis spectroscopic titrations were conducted at 298 ± 0.1 K in the medium of methanol by Lambda950, Perkin Elmer. The stock solution was prepared by dissolving proper amounts of BTPhen ligands into methanol at concentration of 1.0 × 10−3 mol/L. Then ligand solution was diluted into 1.0 × 10−5 mol/L for use, and 2.0 mL BTPhen solution of 1.0 × 10−5 mol/L was added into quarts cell of 1 cm path length, then the initial spectrum was recorded. The each titration was performed by adding 1.0 × 10−4 mol/L Eu(NO3)3·6 H2O in methanol into ligand solution. Before the measurement, the mixture solution was vigorously shaken for 1 min to assure the complexation equilibrium. Stability constants were calculated by HypSpec program in the spectra of wavelength from 200 to 400 nm [40,41].
3.5. Fluorescence Spectroscopy
The fluorescence spectra of four BTPhen ligands with Eu (lll) were investigated in the cuvette cell of 1 cm path length at 298 ± 0.1 K by HORIBA Scientific. Ligand solution was prepared by dissolving 5.58 mg, 6.36 mg, 6.50 mg, and 6.03 mg into 10 mL methanol resulting in initial concentrations of 1.0 × 10−3 mol/L, respectively. Eu(NO3)3 stock solution was diluted into 1.0 × 10−5 mol/L for use. It has been reported that the complexation of CyMe4-BTPhen with f block elements exhibit slow kinetics, so the batch experiments were carried out with equilibrium of 32 h in Extraction Oscillator (200 rpm) at 298 ± 1 K [38]. The spectrum was recorded in the range of 570–720 nm with an interval of 0.2 nm with an excitation wavelength at 394 nm. The bandwidth of excitation and emission was set to be 5 nm and 2 nm. The stability constants of the complexes were determined by a nonlinear least-squares regression method using the HyperSpec program [40,41]. It is worthy to mention that emission spectra are recorded perpendicular to the laser beam with a time delay of 1 µs after excitation. This delay allows discrimination of the short-lived fluorescence of the organic ligand without compromising the analysis of the Eu(lll) complexes since the fluorescence lifetime of these species is considerably longer.
4. Conclusions
The four tetradentate N-donor ligands CyMe4-BTPhen, 5-Br-CyMe4-BTPhen, 5-(4-hydroxyphenyl)-CyMe4-BTPhen, and 5-nitryl-CyMe4-BTPhen were synthesized for extraction of actinides and lanthanides. The former three ligands exhibited good performance for selectively extract Am(lll) over Eu(lll) from HNO3 solution and their distribution ratio for metal ion was in agreement with previous reports. 5-nitryl-CyMe4-BTPhen was first developed by us to suppress the extraction of Eu(lll) into an organic phase for efficient separation from Am(lll). The separation factor of Am(lll) and Eu(lll) was enhanced to 870 (HNO3 = 1M, (ligand) = 5 mmol/L). The basicity of four BTPhen ligands was investigated by NMR titration in the media of CD3OD. Their protonation constants were determined to be 3.1 ± 0.1, 2.3 ± 0.2, 0.9 ± 0.2, and 0.5 ± 0.1, respectively. The tendency of affinity to H+ of these ligands follows the order of L1 > L2 > L3 > L4, which is inverse to the strength of the withdrawing electron substituent: nitryl (L4) > hydroxyphenyl (L3) > bromine (L2) > no substitution (L1). In addition, the stability constants of four BTPhen ligands with Eu(III) were obtained by UV–vis and fluorescence investigation. The results demonstrate that the affinity of these ligands to lanthanides gradually diminishes with the inductive effect by a substituent, which is in accordance with the solvent extraction results. Even though the novel ligands L4 shows the best selective extraction performance of actinides over lanthanides from a highly acidic solution, its solubility in hydrocarbon still needs to be improved. The results of this paper are of important reference in new phenanthroline-derived ligand design for industrial processing of used nuclear fuel. Further effort needs to be made in the tuning solubility of hydrocarbon solvents and available stripping (back-extraction) by dilute acid.
Author Contributions
Conceptualization, M.-M.L. and X.-J.L.; methodology, M.-M.L. and Q.Y.; formal analysis, Y.-K.L.; J.N. and S.-M.Y.; M.-M.L.; X.-J.L.; resources, S.-T.X.; Y.-G.O. and G.-A.Y.; data curation, F.-Y.L.; S.-T.X.; Y.-Y.Y.; writing—original draft preparation, M.-M.L.; writing—M.-M.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of the compounds are not available from the authors upon request.
References
- OECD—Nuclear Energy Agency (NEA). Status and Assessment Report on Actinide and Fission Product Partitioning and Transmutation; NEA: Paris, France, 1999. [Google Scholar]
- Magill, J.; Berthou, V.; Haas, D.; Galy, J.; Schenkel, R.; Wiese, H.W.; Heusener, G.; Tommasi, J.; Youinou, G. Impact limits of partitioning and transmutation scenarios on the radiotoxicity of actinides in radioactive waste. Nucl. Energy 2003, 42, 263–277. [Google Scholar] [CrossRef]
- Salvatores, M.; Palmiotti, G. Radioactive waste partitioning and transmutation within advanced fuel cycles: Achievements and challenges. Prog. Part. Nucl. Phys. 2011, 66, 144–166. [Google Scholar] [CrossRef]
- OECD—Nuclear Energy Agency (NEA). Potential Benefits and Impacts of Advanced Nuclear Fuel Cycles with Actinide Partitioning and Transmutation; Report NEA No. 6894; NEA: Paris, France, 2011. [Google Scholar]
- Sood, D.D.; Patil, S.K. Chemistry of Nuclear Fuel Reprocessing: Current Status. Radioanal. Nucl. Chem. 1996, 203, 547–573. [Google Scholar] [CrossRef]
- Lumetta, G.J.; Gelis, A.V.; Vandegrift, G.F. Review: Solvent Systems Combining Neutral and Acidic Extractants for Separating Trivalent Lanthanides from the Transuranic Elements. Solvent Extr. Ion Exch. 2010, 28, 287–312. [Google Scholar] [CrossRef]
- Nash, K.L. A Review of the Basic Chemistry and Recent Developments in Trivalent f-Elements Separations. Solvent Extr. Ion Exch. 1993, 11, 729–768. [Google Scholar] [CrossRef]
- Cotton, S. Comprehensive Coordination Chemistry II; McCleverty, J.A., Meyer, T.J., Eds.; Elsevier: Oxford, UK, 2004; Volume 3, pp. 93–188. [Google Scholar]
- Katz, J.J.; Morss, L.R.; Edelstein, N.M.; Fuger, J. (Eds.) The Chemistry of the Actinide and Transactinide Elements; Springer: Dordrecht, The Netherlands, 2006; Volume 1, pp. 1–17. [Google Scholar]
- Afsar, A.; Laventine, D.M.; Harwood, L.M.; Hudson, M.J.; Geist, A. Utilizing electronic effects in the modulation of BTPhen ligands with respect to the partitioning of minor actinides from lanthanides. Chem. Commun. 2013, 49, 8534. [Google Scholar] [CrossRef]
- Lewis, F.W.; Harwood, L.M.; Hudson, M.J.; Drew, M.G.B.; Sypula, M.; Modolo, G.; Whittaker, D.; Sharrad, C.A.; Videva, V.; Hubscher-Bruder, V.; et al. Complexation of lanthanides, actinides and transition metal cations with a 6-(1,2,4-triazin-3-yl)-2,2′:6′,2″-terpyridine ligand: Implications for actinide(lll)/lanthanide(lll) partitioning. Dalton Trans. 2012, 41, 9209–9219. [Google Scholar] [CrossRef] [Green Version]
- Panak, P.J.; Geist, A. Complexation and Extraction of Trivalent Actinides and Lanthanides by Triazinylpyridine N-Donor Ligands. Chem. Rev. 2013, 113, 1199–1236. [Google Scholar] [CrossRef]
- Leoncini, A.; Huskens, J.; Verboom, W. Ligands for f-element extraction used in the nuclear fuel cycle. Chem. Soc. Rev. 2017, 46, 7229–7273. [Google Scholar] [CrossRef]
- Xiao, C.L.; Wang, C.Z.; Chai, Z.F.; Shi, W.Q. Excellent Selectivity for Actinides with a Tetradentate 2,9-Diamide-1,10-Phenanthroline Ligand in Highly Acidic Solution: A Hard–Soft Donor Combined Strategy. Inorg. Chem. 2014, 53, 1712–1720. [Google Scholar] [CrossRef]
- Kong, X.H.; Wu, Q.Y.; Lan, J.H. Theoretical Insights into Preorganized Pyridylpyrazole-Based Ligands toward the Separation of Am(III)/Eu(III). Inorg. Chem. 2018, 57, 14810–14820. [Google Scholar] [CrossRef] [PubMed]
- Trumm, S.; Geist, A.; Panak, P.J.; Fanghanel, T. An Improved Hydrolytically-Stable Bis-Triazinyl-Pyridine (BTP) for Selective Actinide Extraction. Solvent Extr. Ion Exch. 2011, 29, 213–229. [Google Scholar] [CrossRef]
- Kolarik, Z.; Mullich, U.; Gassner, F. Selective Extraction of Am(lll) over Eu(lll) by 2,6-ditriazolyl and 2,6-ditriazintlpridinies1. Solvent Extr. Ion Exch. 1999, 17, 23–32. [Google Scholar] [CrossRef]
- Kolarik, Z.; Mullich, U.; Gassner, F. Extraction of Am(lll) and Eu(lll) Nitrates by 2-6-di-(5,6-dipropyl-1,2,4-triazine-3-yl) pyridines1. Solvent Extr. Ion Exch. 1999, 17, 1155–1170. [Google Scholar] [CrossRef]
- Drew, M.G.B.; Foreman, M.R.S.J.; Hudson, M.J.; Madic, C. 6,6′-bis-(5,6-diethyl-[1,2,4]triazin-3-yl)-2,2′-bipyridyl the first example of a new class of quadridentate heterocyclic extraction reagents for the separation of americium(lll) and europium(lll). Inorg. Chem. Commun. 2005, 8, 239–241. [Google Scholar] [CrossRef]
- Geist, A.; Hill, C.; Modolo, G.; Foreman, M.R.S.; Weigl, M.; Gompper, K.; Hudson, M.J.; Madic, C. 6,6′-Bis(5,5,8,8-tetramethyl-5,6,7,8-tetrahydro-benzo[1,2,4]triazin-3-yl) [2,2′]bipyridine, an Effective Extracting Agent for the Separation of Americium(lll) and Curium(lll) from the Lanthanides. Solvent Extr. Ion Exch. 2006, 24, 463–483. [Google Scholar] [CrossRef]
- Foreman, M.R.S.; Hudson, M.J.; Drew, M.G.B. Complexes formed between the quadridentate, heterocyclic molecules 6,6′-bis-(5,6-dialkyl-1,2,4-triazin-3-yl)-2,2′-bipyridine (BTBP) and lanthanides(lll): Implications for the partitioning of actinides(lll) and lanthanides(lll). Dalton Trans. 2006, 1645–1653. [Google Scholar] [CrossRef]
- Foreman, M.R.S.; Hudson, M.J.; Geist, A.; Madic, C.; Weigl, M. An Investigation into the Extraction of Americium(lll), Lanthanides and D-Block Metals by 6,6′-Bis-(5,6-dipentyl-[1,2,4]triazin-3-yl)-[2,2′]bipyridinyl (C5-BTBP). Solvent Extr. Ion Exch. 2005, 23, 645–662. [Google Scholar] [CrossRef]
- Magnusson, D.; Christiansen, B.; Foreman, M.R.S.; Geist, A.; Glatz, J.P.; Malmbeck, R.; Modolo, G.; Serrano-Purroy, D.; Sorel, C. Demonstration of a SANEX Process in Centrifugal Contactors using the CyMe4-BTBP Molecule on a Genuine Fuel Solution. Solvent Extr. Ion Exch. 2009, 27, 97. [Google Scholar] [CrossRef]
- Lewis, F.W.; Harwood, L.M.; Hudson, M.J. Highly Efficient Separation of Actinides from Lanthanides by a Phenanthroline-Derived Bis-triazine Ligand. J. Am. Chem. Soc. 2011, 133, 13093–13102. [Google Scholar] [CrossRef] [Green Version]
- Afsar, A.; Harwood, L.M.; Westwood, J. Effective separation of the actinides Am(lll) and Cm(Ⅲ) by electronic modulation of bis-(1,2,4-triazin-3-yl) phenanthrolines. Chem. Commun. 2015, 51, 5860–5863. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, D.M.; Griffiths, T.L.; Helliwell, M. Lanthanide Speciation in Potential SANEX and GANEX Actinide/Lanthanide Separations Using Tetra-N-Donor Extractants. Inorg. Chem. 2013, 52, 3429–3444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ebenezer, C.; Solomon, R.V. Tailoring the selectivity of phenanthroline derivatives for the partitioning of trivalent Am/Eu ions—A relativistic DFT study. Inorg. Chem. Front. 2021, 8, 3012–3024. [Google Scholar] [CrossRef]
- Morgan, S.G.; Burstall, F.H. Dehydrogenation of pyridine by anhydrous ferric chloride. J. Chem. Soc. 1932, 20–30. [Google Scholar] [CrossRef]
- Morgan, S.G.; Burstall, F.H. Researches on residual affinity and coordination. Part XXXVII. Complex metallic salts containing 2:6-di-2′-pyridyl-pyridine (2:2′:2″-tripyridyl). J. Chem. Soc. 1937, 1649–1655. [Google Scholar] [CrossRef]
- Ackerman, J.J.H.; Soto, G.E.; Spees, W.M.; Zhu, Z.; Evelhoch, J.L. The NMR chemical shift pH measurement revisited: Analysis of error and modeling of a pH dependent reference. Magn. Reson. Med. 1996, 36, 674–683. [Google Scholar] [CrossRef]
- Grycova, L.; Dommisse, R.; Pieters, L.; Marek, R. NMR determination of pKa values of indoloquinoline alkaloids. Magn. Reson. Chem. 2009, 47, 977–981. [Google Scholar] [CrossRef]
- Xu, L.; Pu, N.; Ye, G. Unraveling the complexation mechanism of actinide(lll) and lanthanide(Ⅲ) with a new tetradentate phenanthroline-derived phosphonate ligand. Inorg. Chem. Front. 2020, 7, 1726–1740. [Google Scholar] [CrossRef]
- Kolarik, Z. Complexation and Separation of Lanthanides(lll) and Actinides(lll) by Heterocyclic N-Donors in Solutions. Chem. Rev. 2008, 108, 4208–4252. [Google Scholar] [CrossRef]
- Martin, R.B.; Lissfelt, J.A. The 2,2′,2″-Tripyridine System. J. Am. Chem. Soc. 1956, 78, 938–940. [Google Scholar] [CrossRef]
- Kou, F.; Yang, S.; Qian, H.; Zhang, L.; Beavers, C.M.; Teat, S.J.; Tian, G. A fluorescence study on the complexation of Sm(III), Eu(lll) and Tb(lll) with tetraalkyldiglycolamides (TRDGA) in aqueous solution, in solid state, and in solvent extraction. Dalton Trans. 2016, 45, 18484–18493. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Fang, M.; Wang, X. Sorption Speciation of Lanthanides/Actinides on Minerals by TRLFS, EXAFS and DFT Studies: A Review. Molecules 2010, 15, 8431–8468. [Google Scholar] [CrossRef] [PubMed]
- Stumpf, T.; Bauer, A.; Coppin, F.; Fanghanel, T.; Kim, J.I. Inner-sphere, outer-sphere and ternary surface complexes: A TRLFS study of the sorption process of Eu (lll) onto smectite and kaolinite. Radiochim. Acta 2002, 90, 345–349. [Google Scholar] [CrossRef]
- Bremer, A.; Whittaker, D.M.; Sharrad, C.A. Complexation of Cm(lll) and Eu(lll) with CyMe4-BTPhen and CyMe4-BTBP studied by time resolved laser fluorescence spectroscopy. Dalton Trans. 2014, 43, 2684–2694. [Google Scholar] [CrossRef]
- Frassineti, C.; Ghelli, S.; Gans, P.; Sabatini, A.; Moruzzi, M.S.; Vacca, A. Nuclear magnetic resonance as a tool for determining protonation constants of natural polyprotic bases in solution. Anal Biochem. 1995, 231, 374–382. [Google Scholar] [CrossRef]
- Gans, P.; Sabatini, A.; Vacca, A. Investigation of equilibria in solution. Determination of equilibrium constants with the HYPERQUAD suite of programs. Talanta 1996, 43, 1739–1753. [Google Scholar]
- Alderighi, L.; Gans, P.; Ienco, A.; Peters, D.; Sabatini, A.; Vacca, A. Hyperquad simulation and speciation (HySS): A utility program for the investigation of equilibria involving soluble and partially soluble species. Coord. Chem. Rev. 1999, 184, 311–318. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).