Optimization of Extraction Conditions and Characterization of Volatile Organic Compounds of Eugenia klotzschiana O. Berg Fruit Pulp
Abstract
:1. Introduction
2. Results and Discussion
2.1. Evaluation of the Effect of Independent Variables on the Extraction of VOCs
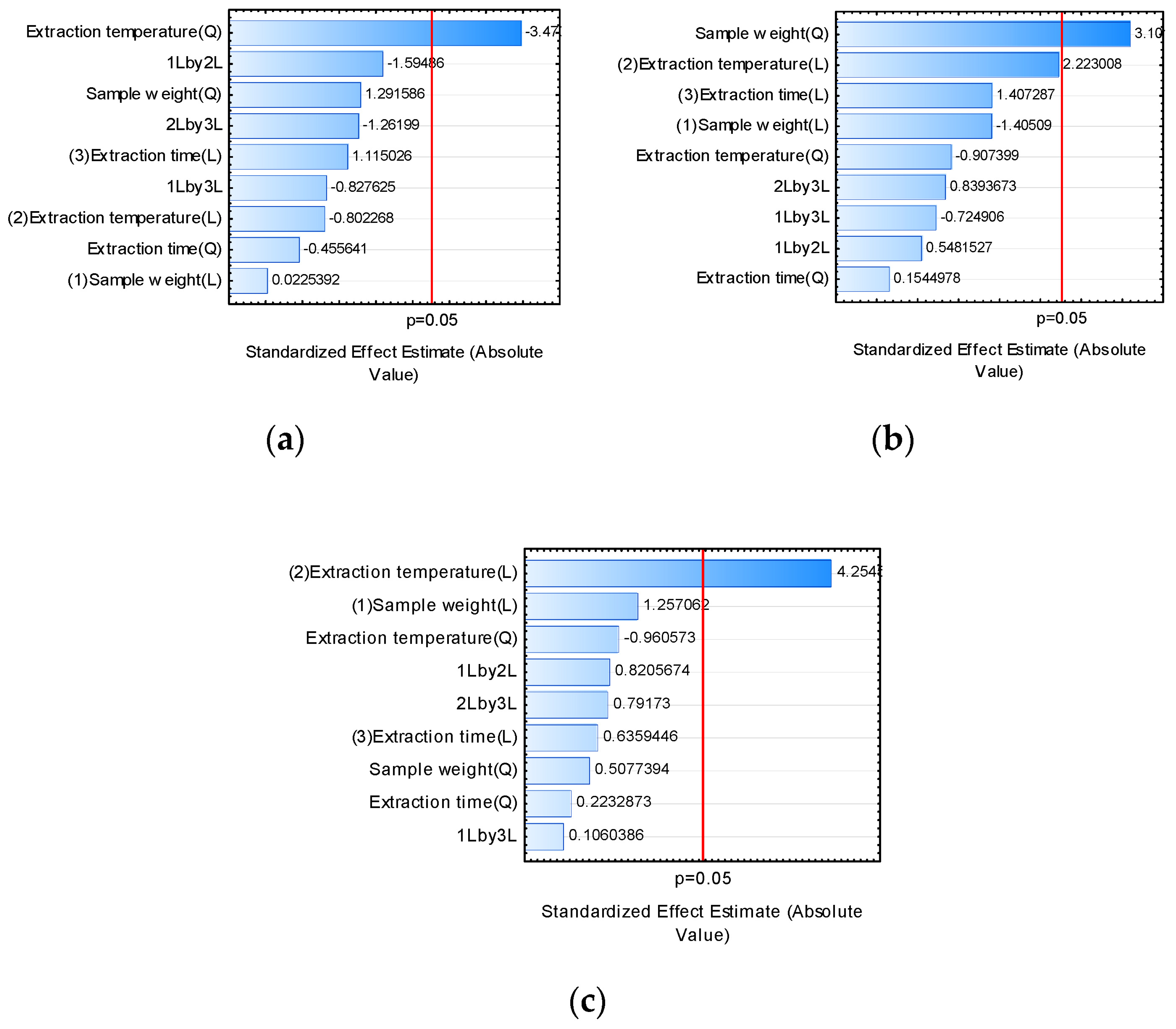
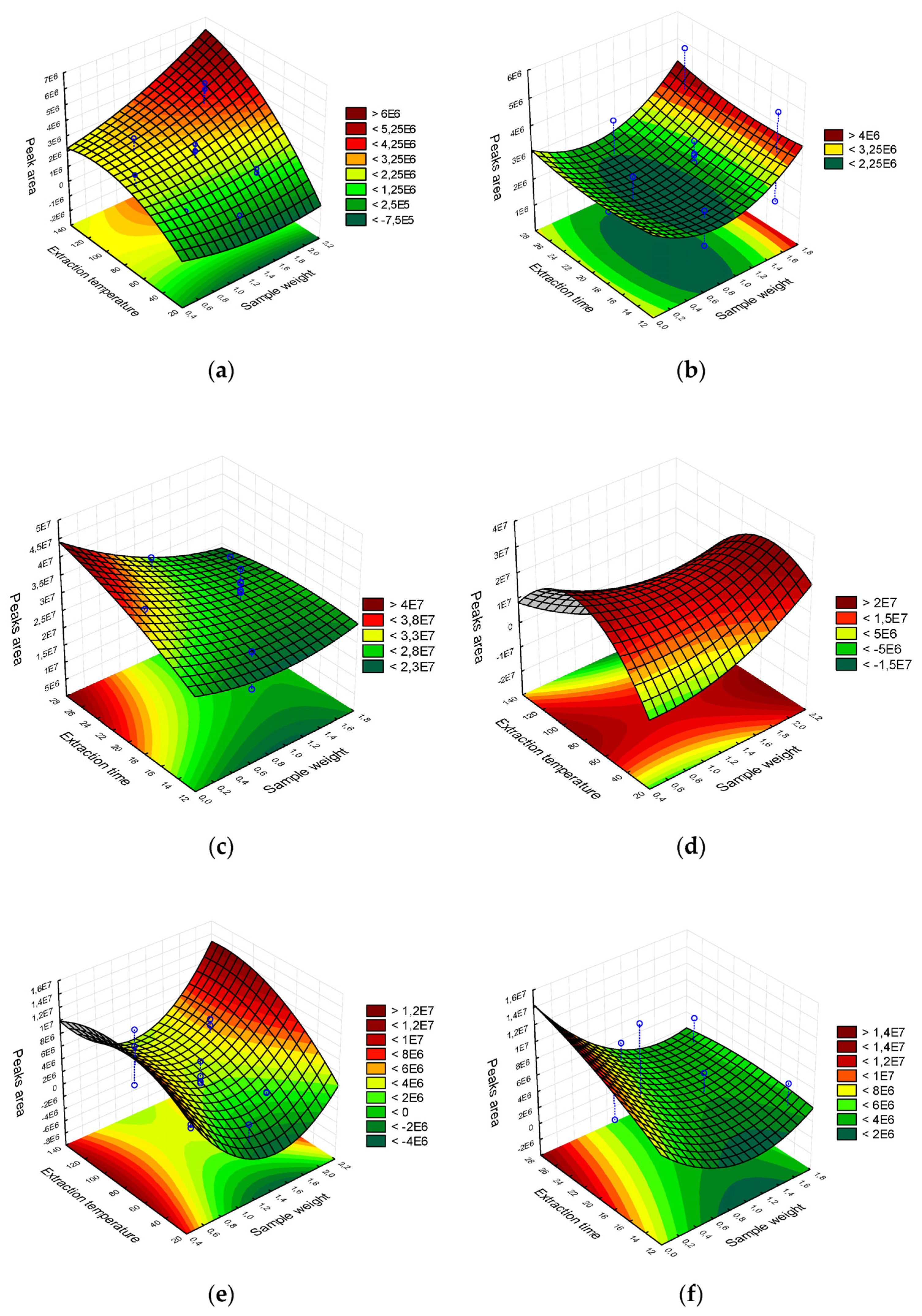
2.1.1. Extraction Temperature
2.1.2. Extraction Time
2.1.3. Sample Weight
3. Materials and Methods
3.1. Sample Acquisition
3.2. Experimental Design
3.3. Headspace Solid-Phase Microextraction (HS-SPME)
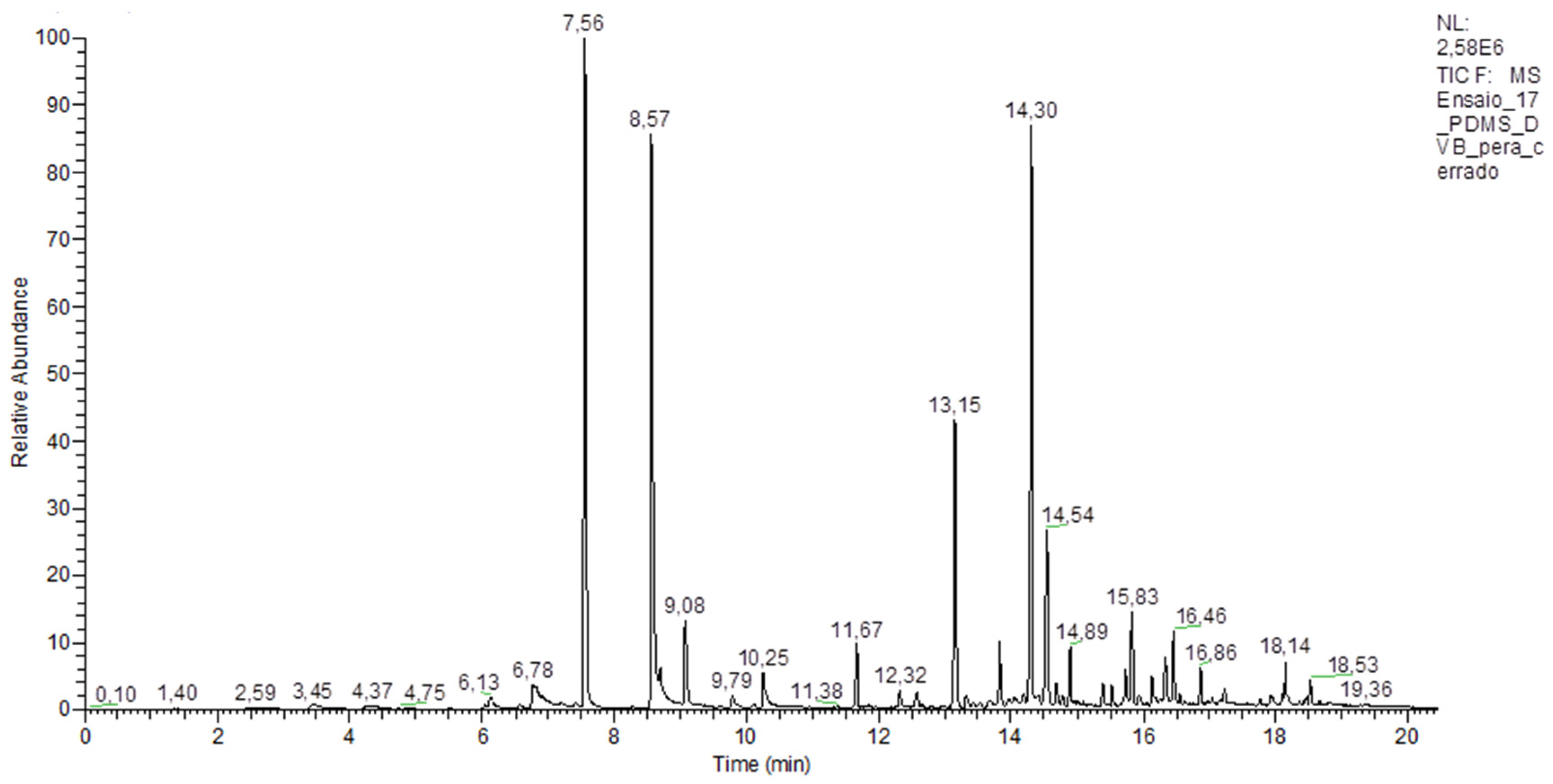
3.4. Gas Chromatography Coupled with Mass Spectrometry Analysis and Identification of Volatile Compounds
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Appendix A
| N. | Name | Formula | CAS | Reference | PA | PDMS/ DVB | CAR/PDMS/DVB |
|---|---|---|---|---|---|---|---|
| Monoterpenes | |||||||
| 01 | (±)-α-pinene | C10H16 | 80-56-8 | [10] | X | ||
| 02 | p-mentha-1,8-dien-7-ol | C10H14O | 536-59-4 | [37] | X | ||
| 03 | (2S, 5S)-2-methyl-5-propan-2-ylbicyclo [3.1.0] hexan-2-ol | C10H18O | ------------ | X | |||
| 04 | myrtenol | C10H16O | 515-00-4 | [30] | X | ||
| 05 | p-mentha-3,8-diene | C10H16 | 586-67-4 | [38] | X | ||
| 06 | (±)-camphene | C10H16 | 79-92-5 | [29] | X | ||
| 07 | α-fenchene | C10H16 | 471-84-1 | [10] | X | X | |
| 08 | linalyl acetate | C12H20O2 | 115-95-7 | [31] | X | ||
| 09 | ocimene | C10H16 | 29714-87-2 | [31] | X | X | |
| 10 | (−)-lavandulol | C10H18O | 498-16-8 | [31] | X | ||
| 11 | sabinene hydrate trans acetate | C12H20O2 | ------------ | ------------ | X | ||
| 12 | isogeraniol | C10H18O | 5944-20-7 | [30] | X | ||
| Sesquiterpenes | |||||||
| 13 | δ-elemene | C15H24 | 20307-84-0 | [10] | X | X | |
| 14 | isoledene | C15H24 | 95910-36-4 | [21] | X | ||
| 15 | β-guaiene | C15H24 | 88-84-6 | [39] | X | ||
| 16 | α-humulene | C15H24 | ------------ | [29] | X | X | |
| 17 | (+)-calarene | C15H24 | 17334-55-3 | [40] | X | X | |
| 18 | cadinene | C15H24 | 29350-73-0 | [21] | X | ||
| 19 | viridiflorene | C15H24 | 21747-46-6 | [10] | X | ||
| 20 | α-guaiene | C15H24 | 3691-12-1 | [10] | X | X | |
| 21 | γ-himachalene | C15H24 | 53111-25-4 | [10] | X | ||
| 22 | (−)-γ-muurolene | C15H24 | 24268-39-1 | [10] | X | X | |
| 23 | acoradiene | C15H24 | 24048-44-0 | [41] | X | X | |
| 24 | (+)-cyclosativene | C15H24 | 22469-52-9 | [21] | X | X | |
| 25 | (−)-caryophyllene | C15H24 | 87-44-5 | [21] | X | ||
| 26 | cadina-3,9-diene | C15H24 | 523-47-7 | [42] | X | ||
| 27 | globulol | C15H26O | 489-41-8 | [10] | X | X | |
| 28 | patchoulene | C15H24 | ----------- | [10] | X | ||
| 29 | α-caryophyllene alcohol | C15H26O | 4586-22-5 | [43] | X | ||
| 30 | (−)-guaiol | C15H26O | 489-86-1 | [10] | X | ||
| 31 | cedryl acetate | C17H28O2 | 77-54-3 | [44] | X | ||
| 32 | 2-naphthalenol,2,3,4,4alpha,5,6, 7-octahydro-1,4alpha-dimethyl-7-(2-hydroxi-1-methylethyl) | C15H26O2 | ----------- | ----------- | X | ||
| 33 | spiro [4.5]decan-7-one, 1,8-dimethyl-8,9-epoxi-4-isopropyl | C15H24O2 | 61050-91-7 | [20] | X | ||
| Other | |||||||
| 34 | N-(2-hydroxipropyl) ethylenediamine | C15H14N2O | 123-84-2 | ----------- | X | ||
| 35 | ethyl hexanoate | C8H16O2 | 123-66-0 | [45] | X | ||
| 36 | benzyl alcohol | C7H8O | 100-51-6 | [45] | X | ||
| 37 | benzyl acetate | C9H10O2 | 140-11-4 | [46] | X | X | X |
| 38 | (E)-3,7-dimetlnon-6-enal | C11H20O | ----------- | ----------- | X | ||
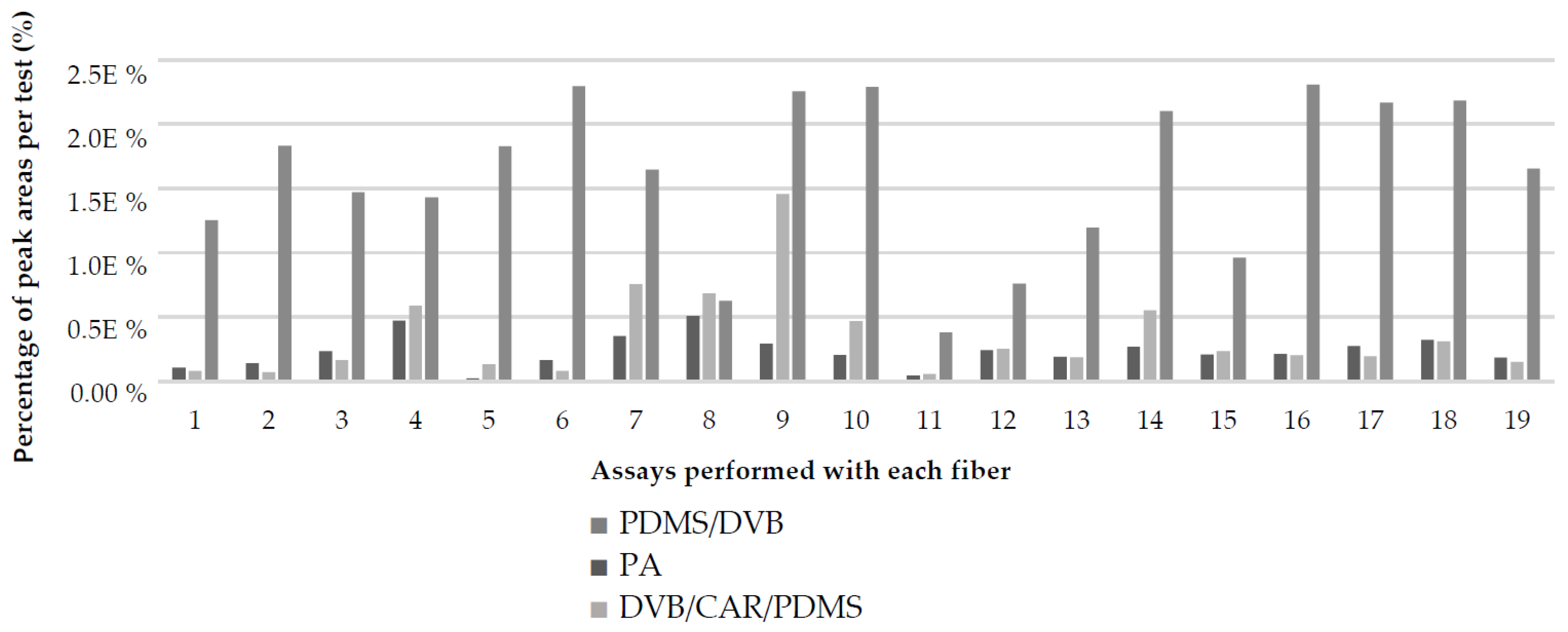
| Total compounds identified by each fiber | 18 | 23 | 08 | ||||
References
- Junqueira, N.T.V.; Junqueira, K.P.; Pereira, A.; Pereira, E.; Braga, M.; Conceição, L.D.; Faleiro, F. Frutíferas nativas do cerrado: O extrativismo e a busca da domesticação. In Proceedings of the Embrapa Cerrados-Artigo em Anais de Congresso (ALICE), Congresso Brasileiro de Fruticultura, Bento Gonçalves, Brazil, 22–26 October 2012. [Google Scholar]
- Sano, E.E.; Bettiol, G.M.; Martins, E.D.S.; Couto Júnior, A.F.; Vasconcelos, V.; Bolfe, E.L.; Victoria, D.D.C. Características gerais da paisagem do Cerrado. In Embrapa Informática Agropecuária-Capítulo em livro científico (ALICE); Embrapa: Brasília, Brazil, 2020. [Google Scholar]
- Vieira, R.F.; Agostini-Costa, T.; Silva, D.B.; Ferreira, F.R.; Sano, S.M. Frutas Nativas da Região Centro-Oeste do Brasil; Embrapa Recursos Genéticos e Biotecnologia: Brasília, Brasil, 2006. [Google Scholar]
- Arruda, H.S.; Pereira, G.A.; de Morais, D.R.; Eberlin, M.N.; Pastore, G.M. Determination of free, esterified, glycosylated and insoluble-bound phenolics composition in the edible part of araticum fruit (Annona crassiflora Mart.) and its by-products by HPLC-ESI-MS/MS. Food Chem. 2018, 245, 738–749. [Google Scholar] [CrossRef] [PubMed]
- Ramos, A.L.C.C.; Mendes, D.D.; Silva, M.R.; Augusti, R.; Melo, J.O.F.; de Araújo, R.L.B.; Lacerda, I.C.A. Chemical profile of Eugenia brasiliensis (Grumixama) pulp by PS/MS paper spray and SPME-GC/MS solid-phase microextraction. Res. Soc. Dev. 2020, 9, e318974008. [Google Scholar] [CrossRef]
- Teixeira, L.D.L.; Bertoldi, F.C.; Lajolo, F.M.; Hassimotto, N.M.A. Identification of ellagitannins and flavonoids from Eugenia brasilienses Lam. (Grumixama) by HPLC-ESI-MS/MS. J. Agric. Food Chem. 2015, 63, 5417–5427. [Google Scholar] [CrossRef]
- García, Y.M.; Rufini, J.; Campos, M.P.; Guedes, M.N.; Augusti, R.; Melo, J.O. SPME fiber evaluation for volatile organic compounds extraction from acerola. J. Braz. Chem. Soc. 2019, 30, 247–255. [Google Scholar] [CrossRef]
- Silva, M.R.; De Souza, A.G.; De Araújo, R.L.B.; Lacerda, I.C.A.; Augusti, R.; Melo, J.O.F.; Mendonça, H.D.O.P. Análise metabolômica de cagaitas utilizando a espectrometria de massas com ionização por paper spray. Avanços Ciência E Tecnol. Alimentos. Científica 2020, 1, 25–41. [Google Scholar] [CrossRef]
- García, Y.M.; Ramos, A.L.C.C.; de Paula, A.C.C.F.F.; do Nascimento, M.H.; Augusti, R.; de Araújo, R.L.B.; Melo, J.O.F. Chemical Physical Characterization and Profile of Fruit Volatile Compounds from Different Accesses of Myrciaria floribunda (H. West Ex Wild.) O. Berg through Polyacrylate Fiber. Molecules 2021, 26, 5281. [Google Scholar] [CrossRef]
- García, Y.M.; Ramos, A.L.C.C.; de Oliveira Júnior, A.H.; de Paula, A.C.C.F.F.; de Melo, A.C.; Andrino, M.A.; Melo, J.O.F. Physicochemical Characterization and Paper Spray Mass Spectrometry Analysis of Myrciaria Floribunda (H. West ex Willd.) O. Berg Accessions. Molecules 2021, 26, 7206. [Google Scholar] [CrossRef]
- Bess, J.; Araújo, S.S.; Grellmann, B.P.; Cruz, D.C.; Alamino, M.; Melo, J.O.F.; Reina, L.D.C.B. Biometric and physical-chemical analysis of pequis collected in the state of Mato Grosso. Sci. Electron. Arch. 2020, 13, 24–30. [Google Scholar] [CrossRef]
- Santos, B.O.; Augusti, R.; Melo, J.O.F.; Takahashi, J.A.; de Araújo, R.L.B. Optimization of extraction conditions of volatile compounds from pequi peel (Caryocar brasiliense Camb.) using HS-SPME. Res. Soc. Dev. 2020, 9, e919974893. [Google Scholar] [CrossRef]
- Ramos, S.A.; Silva, M.R.; Jacobino, A.R.; Damasceno, I.A.N.; Rodrigues, S.M.; Carlos, G.A.; Capobiango, M. Caracterização físico-química, microbiológica e da atividade antioxidante de farinhas de casca e amêndoa de manga (Mangifera indica) e sua aplicação em brownie. Res. Soc. Dev. 2021, 10, e22310212436. [Google Scholar] [CrossRef]
- Figueiredo, Y.G.; Bueno, F.C.; Júnior, A.H.D.O.; Mazzinghy, A.C.D.C.; Mendonça, H.D.O.P.; de Oliveira, A.F.; De Melo, A.C.; Reina, L.D.C.B.; Augusti, R.; Melo, J.O.F. Profile of the volatile organic compounds of pink pepper and black pepper. Sci. Electron. Arch. 2021, 14. [Google Scholar] [CrossRef]
- Silva, M.R.; Freitas, L.G.; Mendonça, H.D.O.; Souza, A.G.; Pereira, H.V.; Augusti, R.; Melo, J.O.F.; Araújo, R.L. determination of chemical profile of Eugenia dysenterica ice cream using PS-MS and HS-SPME/GC-MS. Química Nova 2021, 44, 129–136. [Google Scholar] [CrossRef]
- Carneiro, N.S. Caracterização Química e Zvidade Antioxidante da Polpa e Óleo Essencial da Pera do Cerrado (Eugenia klotzschiana Berg.). 2016. Available online: https://repositorio.ifgoiano.edu.br/handle/prefix/42 (accessed on 20 October 2020).
- Carneiro, N.S.; Alves, J.M.; Alves, C.C.F.; Esperandim, V.R.; Miranda, M.L.D. Essential oil of flowers from Eugenia klotzschiana (myrtaceae): Chemical composition and in vitro trypanocidal and cytotoxic activities. Rev. Virtual Quim. 2017, 9, 1381–1392. [Google Scholar] [CrossRef]
- Mariano, A.P.X.; Ramos, A.L.C.C.; Augusti, R.; Araújo, R.L.B.; Melo, J.O.F. Analysis of the chemical profile of cerrado pear fixed compounds by mass spectrometry with paper spray and volatile ionization by SPME-HS CG-MS. Res. Soc. Dev. 2020, 9, e949998219. [Google Scholar] [CrossRef]
- da Rocha, R.F.J.; da Silva Araújo, Í.M.; de Freitas, S.M.; dos Santos Garruti, D. Optimization of headspace solid phase micro-extraction of volatile compounds from papaya fruit assisted by GC–olfactometry. J. Food Sci. Technol. 2017, 54, 4042–4050. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Hernández, E.; Buzón-Durán, L.; Andrés-Juan, C.; Lorenzo-Vidal, B.; Martín-Gil, J.; Martín-Ramos, P. Physicochemical characterization of Crithmum maritimum L. and Daucus carota subsp. gummifer (Syme) Hook. fil. and their antimicrobial activity against apple tree and grapevine phytopathogens. Agronomy 2021, 11, 886. [Google Scholar] [CrossRef]
- Wei, C.; Ma, Z.; Liu, Y.; Qiao, J.; Sun, G. Effect of boron on fruit quality in pineapple. In AIP Conference Proceedings; AIP Publishing LLC: Melville, NY, USA, 2018. [Google Scholar]
- Biajoli, A.F.P. Fibras para SPME (microextração em fase solida) Recobertas com Novos Ormosils Sol-gel. (Doctoral Dissertation, Universidade Estadual de Campinas (UNICAMP). Instituto de Química). 2008. Available online: http://repositorio.unicamp.br/jspui/handle/REPOSIP/250195 (accessed on 20 July 2021).
- Pawliszyn, J. Theory of solid-phase microextraction. In Handbook of Solid Phase Microextraction; Elsevier: Amsterdam, The Netherlands, 2012; pp. 13–59. [Google Scholar] [CrossRef]
- Sigma, S. SPME for GC Analysis: Getting Started with Solid Phase Microextraction; Merck: Darmstadt, Germany, 2018. [Google Scholar]
- Canuto, K.; Garruti DD, S.; Magalhaes, H. Microextração em Fase Sólida: Métodos Analíticos Práticos Para Extração de Compostos Voláteis de Frutas. Embrapa Agroindústria Tropical-Comunicado Técnico (INFOTECA-E). 2011. Available online: https://ainfo.cnptia.embrapa.br/digital/bitstream/item/42119/1/COT11003.pdf (accessed on 20 October 2020).
- Silva, M.R.; Bueno, G.H.; Araújo, R.L.; Lacerda, I.C.; Freitas, L.G.; Morais, H.A.; Augusti, R.; Melo, J.O. Evaluation of the influence of extraction conditions on the isolation and identification of volatile compounds from cagaita (Eugenia dysenterica) using HS-SPME/GC-MS. J. Braz. Chem. Soc. 2019, 30, 379–387. [Google Scholar] [CrossRef]
- García, Y.M.; Lemos, E.E.P.D.; Augusti, R.; Melo, J.O.F. Optimization of extraction and identification of volatile compounds from Myrciaria Floribunda. Rev. Ciência Agronômica 2021, 52, 1–8. [Google Scholar] [CrossRef]
- Hui, Y.H.; Chen, F.; Nollet, L.M.; Guiné, R.P.; Martín-Belloso, O.; Mínguez-Mosquera, M.I.; Stanfield, P. (Eds.) Handbook of Fruit and Vegetable Flavors; John Wiley and Sons: Hoboken, NJ, USA, 2010. [Google Scholar]
- Gasiński, A.; Kawa-Rygielska, J.; Szumny, A.; Czubaszek, A.; Gąsior, J.; Pietrzak, W. Volatile compounds content, physicochemical parameters, and antioxidant activity of beers with addition of mango fruit (Mangifera Indica). Molecules 2020, 25, 3033. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, W.; Yu, W.; Zhao, L.; Song, S.; Xu, W.; Zhang, C.; Ma, C.; Wang, L.; Wang, S. Study on the volatile composition of table grapes of three aroma types. LWT 2019, 115, 108450. [Google Scholar] [CrossRef]
- Xiao, Z.; Li, Q.; Niu, Y.; Zhou, X.; Liu, J.; Xu, Y.; Xu, Z. Odor-active compounds of different lavender essential oils and their correlation with sensory attributes. Ind. Crops Prod. 2017, 108, 748–755. [Google Scholar] [CrossRef]
- Freitas, T.P.; Taver, I.B.; Spricigo, P.C.; do Amaral, L.B.; Purgatto, E.; Jacomino, A.P. Volatile Compounds and Physicochemical Quality of Four Jabuticabas (Plinia sp.). Molecules 2020, 25, 4543. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhu, Y.; He, Z.; Zhang, T.; Xiao, Z.; Xu, R.; He, J. Plantanone D, a new rare methyl-flavonoid from the flowers of Hosta plantaginea with anti-inflammatory and antioxidant activities. Nat. Prod. Res. 2021, 35, 4331–4337. [Google Scholar] [CrossRef] [PubMed]
- Pawliszyn, J. Solid Phase Microextraction: Theory and Practice; John Wiley & Sons: Hoboken, NJ, USA, 1997. [Google Scholar]
- Krokou, A.; Kokkinofta, R.; Stylianou, M.; Agapiou, A. Decoding carob flavor aroma using HS–SPME–GC–MS and chemometrics. Eur. Food Res. Technol. 2020, 246, 1419–1428. [Google Scholar] [CrossRef]
- Kataoka, H.; Lord, H.L.; Pawliszyn, J. Applications of solid-phase microextraction in food analysis. J. Chromatogr. A 2000, 880, 35–62. [Google Scholar] [CrossRef]
- Garcia, C.V.; Stevenson, R.J.; Atkinson, R.G.; Winz, R.A.; Quek, S.Y. Changes in the bound aroma profiles of ‘Hayward’and ‘Hort16A’kiwifruit (Actinidia spp.) during ripening and GC-olfactometry analysis. Food Chem. 2013, 137, 45–54. [Google Scholar] [CrossRef]
- Do Nascimento, A.F. Atividade do Óleo Essencial de Frutos de Schinus Terebinthifolius Raddi (Anacardiaceae) em Tetranychus urticae Koch (Acari: Tetranychidae) e Rhyzopherta dominica Fabricius (Coleoptera: Bostrichidae). 2012. Available online: http://www.tede2.ufrpe.br:8080/tede2/handle/tede2/5951 (accessed on 20 January 2021).
- Rondán Sanabria, G.G.; Cabezas Garcia, A.J.; Oliveira Lima, A.W.; Brousett-Minaya, M.A.; Narain, N. HS-SPME-GC-MS detection of volatile compounds in Myrciaria jabuticaba Fruit. Sci. Agropecu. 2018, 9, 319–327. [Google Scholar] [CrossRef] [Green Version]
- Núñez-Carmona, E.; Abbatangelo, M.; Zottele, I.; Piccoli, P.; Tamanini, A.; Comini, E.; Sberveglieri, G.; Sberveglieri, V. Nanomaterial gas sensors for online monitoring system of fruit jams. Foods 2019, 8, 632. [Google Scholar] [CrossRef] [Green Version]
- Asadi, K.; Abbasi-Maleki, S.; Hashjin, G.S. Antidepressant-like effect of Cuminum cyminum essential oil on the forced swim and tail suspension tests in male mice. J. Shahrekord Univ. Med. Sci. 2020, 22, 167–172. [Google Scholar] [CrossRef]
- Boachon, B.; Burdloff, Y.; Ruan, J.-X.; Rojo, R.; Junker, R.R.; Vincent, B.; Nicolè, F.; Bringel, F.; Lesot, A.; Henry, L.; et al. A promiscuous CYP706A3 reduces terpene volatile emission from Arabidopsis flowers, affecting florivores and the floral microbiome. Plant Cell 2019, 31, 2947–2972. [Google Scholar] [CrossRef] [Green Version]
- Thongkham, E.; Aiemsaard, J.; Kaenjampa, P. Antioxidant and Antimicrobial Properties of Ethanolic Extract of Asam Gelugor Fruit (Garcinia atroviridis). Burapha Sci. J. 2021, 26, 1293–1307. [Google Scholar]
- Mei, L.; Shi, L.; Song, X.; Liu, S.; Cheng, Q.; Zhu, K.; Zhuge, R. Characterization of Carboxymethyl Cellulose Films Incorporated with Chinese Fir Essential Oil and Their Application to Quality Improvement of Shine Muscat Grape. Coatings 2021, 11, 97. [Google Scholar] [CrossRef]
- Fang, H.; Chen, J.; Tian, Y.; Liu, Y.; Li, H.; Cheng, H. Chemometrics characterization of volatile changes in processed bayberry juice versus intact fruit during storage by headspace solid-phase micro-extraction combined with GC–MS. J. Food Processing Preserv. 2020, 44, e14444. [Google Scholar] [CrossRef]
- Cano-Lamadrid, M.; Galindo, A.; Collado-González, J.; Rodríguez, P.; Cruz, Z.N.; Legua, P.; Burló, F.; Morales, D.; Carbonell-Barrachina, Á.A.; Hernández, F. Influence of deficit irrigation and crop load on the yield and fruit quality in Wonderful and Mollar de Elche pomegranates. J. Sci. Food Agric. 2018, 98, 3098–3108. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mariano, A.P.X.; Ramos, A.L.C.C.; de Oliveira Júnior, A.H.; García, Y.M.; de Paula, A.C.C.F.F.; Silva, M.R.; Augusti, R.; de Araújo, R.L.B.; Melo, J.O.F. Optimization of Extraction Conditions and Characterization of Volatile Organic Compounds of Eugenia klotzschiana O. Berg Fruit Pulp. Molecules 2022, 27, 935. https://doi.org/10.3390/molecules27030935
Mariano APX, Ramos ALCC, de Oliveira Júnior AH, García YM, de Paula ACCFF, Silva MR, Augusti R, de Araújo RLB, Melo JOF. Optimization of Extraction Conditions and Characterization of Volatile Organic Compounds of Eugenia klotzschiana O. Berg Fruit Pulp. Molecules. 2022; 27(3):935. https://doi.org/10.3390/molecules27030935
Chicago/Turabian StyleMariano, Ana P. X., Ana L. C. C. Ramos, Afonso H. de Oliveira Júnior, Yesenia M. García, Ana C. C. F. F. de Paula, Mauro R. Silva, Rodinei Augusti, Raquel L. B. de Araújo, and Júlio O. F. Melo. 2022. "Optimization of Extraction Conditions and Characterization of Volatile Organic Compounds of Eugenia klotzschiana O. Berg Fruit Pulp" Molecules 27, no. 3: 935. https://doi.org/10.3390/molecules27030935
APA StyleMariano, A. P. X., Ramos, A. L. C. C., de Oliveira Júnior, A. H., García, Y. M., de Paula, A. C. C. F. F., Silva, M. R., Augusti, R., de Araújo, R. L. B., & Melo, J. O. F. (2022). Optimization of Extraction Conditions and Characterization of Volatile Organic Compounds of Eugenia klotzschiana O. Berg Fruit Pulp. Molecules, 27(3), 935. https://doi.org/10.3390/molecules27030935






