Risk Assessment of Passive Smoking Based on Analysis of Hair Nicotine and Cotinine as Exposure Biomarkers by In-Tube Solid-Phase Microextraction Coupled On-Line to LC-MS/MS
Abstract
1. Introduction
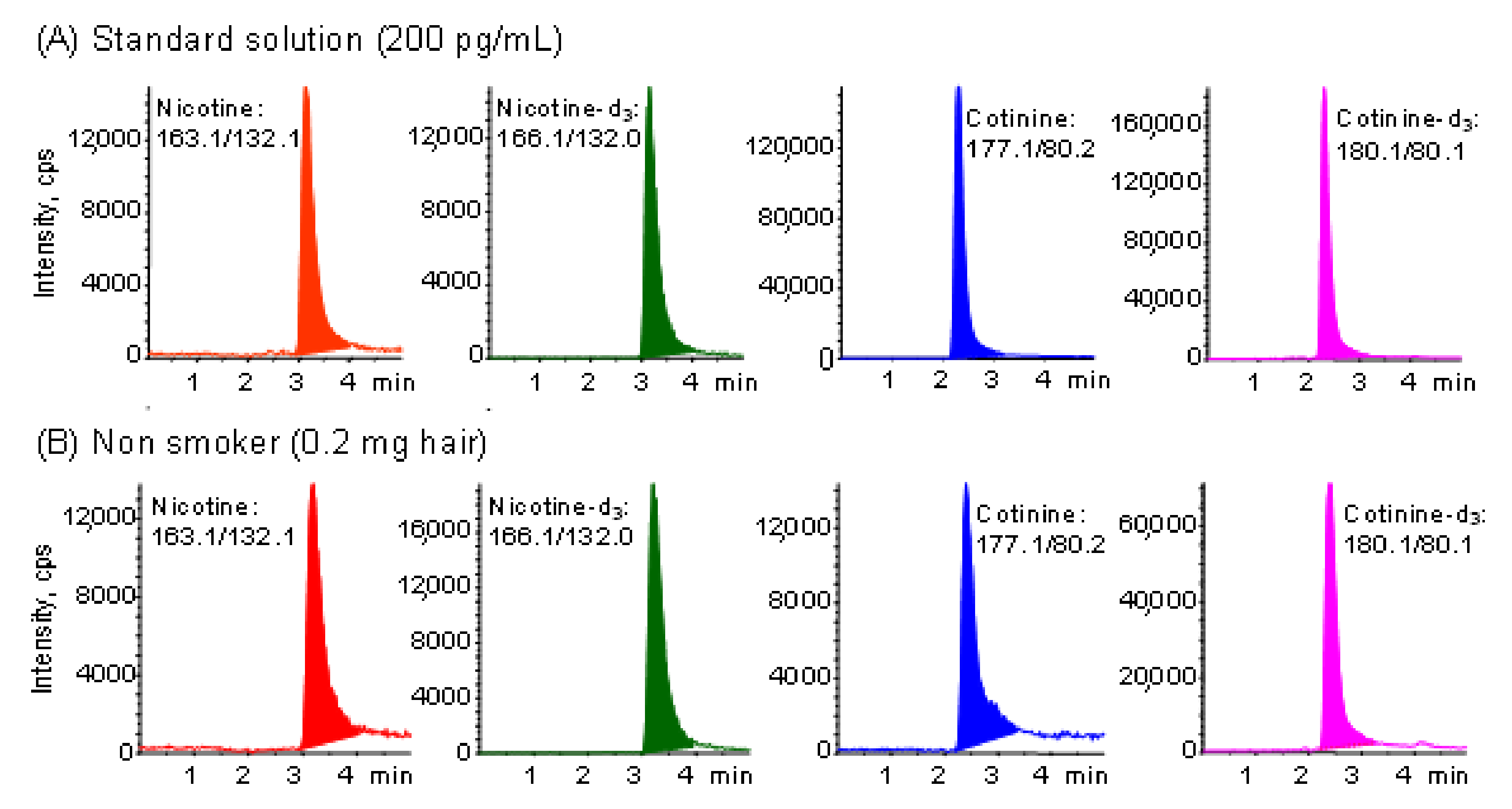
2. Results
3. Discussion
4. Materials and Methods
4.1. Reagents and Standard Solutions
4.2. On-Line Automated Analysis System and Analytical Conditions
4.3. Preparation and Analysis of Hair Samples and Lifestyle Questionnaires
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Hu, Q.; Hou, H. (Eds.) Tobacco Smoke Exposure Biomarkers; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2015. [Google Scholar]
- Talhout, R.; Schulz, T.; Florek, E.; van Benthem, J.; Wester, P.; Opperhuizen, A. Hazardous compounds in tobacco smoke. Int. J. Environ. Res. Public Health 2011, 8, 613–628. [Google Scholar] [CrossRef] [PubMed]
- Mattes, W.; Yang, X.; Orr, M.S.; Richter, P.; Mendrick, D.L. Biomarkers of tobacco smoke exposure. Adv. Clin. Chem. 2014, 67, 1–45. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Tobacco Fact Sheet. 2020. Available online: https://www.who.int/news-room/fact-sheets/detail/tobacco (accessed on 1 October 2021).
- Öberg, M.; Jaakkola, M.S.; Woodward, A.; Peruga, A.; Prüss-Ustün, A. Worldwide burden of disease from exposure to second-hand smoke: A retrospective analysis of data from 192 countries. Lancet 2011, 377, 139–146. [Google Scholar] [CrossRef]
- Torres, S.; Merino, C.; Paton, B.; Correig, X.; Ramírez, N. Biomarkers of exposure to secondhand and thirdhand tobacco smoke: Recent advances and future perspectives. Int. J. Environ. Res. Public Health 2018, 15, 2693. [Google Scholar] [CrossRef]
- Hori, M.; Tanaka, H.; Wakai, K.; Sasazuki, S.; Katanoda, K. Secondhand smoke exposure and risk of lung cancer in Japan: A systematic review and meta-analysis of epidemiologic studies. Jap. J. Clin. Oncol. 2016, 46, 942–951. [Google Scholar] [CrossRef]
- Kurahashi, N.; Inoue, M.; Liu, Y.; Iwasaki, M.; Sasazuki, S.; Sobue, T.; Tsugane, S. Passive smoking and lung cancer in Japanese non-smoking women: A prospective study. Int. J. Cancer 2008, 122, 653–657. [Google Scholar] [CrossRef]
- Ni, X.; Xu, N.; Wang, Q. Meta-analysis and systematic review in environmental tobacco smoke risk of female lung cancer by research type. Int. J. Environ. Res. Public Health 2018, 15, 1348. [Google Scholar] [CrossRef]
- Cao, S.; Yang, C.; Gan, Y.; Lu, Z. The health effects of passive smoking: An overview of systematic reviews based on observational epidemiological evidence. PLoS ONE 2015, 10, e0139907. [Google Scholar] [CrossRef]
- Macacu, A.; Autier, P.; Boniol, M.; Boyle, P. Active and passive smoking and risk of breast cancer: A meta-analysis. Breast Cancer Res. Treat. 2015, 154, 213–224. [Google Scholar] [CrossRef]
- Lee, P.N.; Hamling, J.S. Environmental tobacco smoke exposure and risk of breast cancer in nonsmoking women. An updated review and meta-analysis. Inhal. Toxicol. 2016, 28, 431–454. [Google Scholar] [CrossRef]
- Lee, P.N.; Thornton, A.J.; Hamling, J.S. Epidemiological evidence on environmental tobacco smoke and cancers other than lung or breast. Regul. Toxicol. Pharmacol. 2016, 80, 134–163. [Google Scholar] [CrossRef]
- Sheng, L.; Tu, J.W.; Tian, J.H.; Chen, H.J.; Pan, C.L.; Zhou, R.Z. A meta-analysis of the relationship between environmental tobacco smoke and lung cancer risk of nonsmoker in China. Medicine 2018, 97, e11389. [Google Scholar] [CrossRef]
- De Groot, P.M.; Wu, C.C.; Carter, B.W.; Munden, R.F. The epidemiology of lung cancer. Transl. Lung Cancer Res. 2018, 7, 220–233. [Google Scholar] [CrossRef]
- Law, M.R.; Wald, N.J. Environmental tobacco smoke and ischemic heart disease. Prog. Cardiovasc. Dis. 2003, 46, 31–38. [Google Scholar] [CrossRef]
- Ahijevych, K.; Wewers, M.E. Passive smoking and vascular disease. Cardiovasc. Nurs. 2003, 18, 69–74. [Google Scholar] [CrossRef]
- Dunbar, A.; Gotsis, W.; Frishman, W. Second-hand tobacco smoke and cardiovascular disease risk: An epidemiological review. Cardiol. Rev. 2013, 21, 94–100. [Google Scholar] [CrossRef]
- Messner, B.; Bernhard, D. Smoking and cardiovascular disease: Mechanisms of endothelial dysfunction and early atherogenesis. Arterioscler Thromb. Vasc. Biol. 2014, 34, 509–515. [Google Scholar] [CrossRef]
- DiGiacomo, S.I.; Jazayeri, M.A.; Barua, R.S.; Ambrose, J.A. Environmental tobacco smoke and cardiovascular disease. Int. J. Environ. Res. Public Health 2018, 16, 96. [Google Scholar] [CrossRef]
- Pistilli, M.; Howard, V.J.; Safford, M.M.; Lee, B.K.; Lovasi, G.S.; Cushman, M.; Malek, A.M.; McClure, L.A. Association of secondhand tobacco smoke exposure during childhood on adult cardiovascular disease risk among never-smokers. Ann. Epidemiol. 2019, 32, 28–34. [Google Scholar] [CrossRef]
- Khoramdad, M.; Vahedian-Azimi, A.; Karimi, L.; Rahimi-Bashar, F.; Amini, H.; Sahebkar, A. Association between passive smoking and cardiovascular disease: A systematic review and meta-analysis. IUBMB Life 2020, 72, 677–686. [Google Scholar] [CrossRef]
- Akpa, O.M.; Okekunle, A.P.; Asowata, J.O.; Adedokun, B. Passive smoking exposure and the risk of hypertension among non-smoking adults: The 2015-2016 NHANES data. Clin. Hypertens. 2021, 27, 1–10. [Google Scholar] [CrossRef]
- Hayashino, Y.; Fukuhara, S.; Okamura, T.; Yamato, H.; Tanaka, H.; Tanaka, T.; Kadowaki, T.; Ueshima, H. A prospective study of passive smoking and risk of diabetes in a cohort of workers. Diabetes Care 2008, 31, 732–734. [Google Scholar] [CrossRef]
- Wei, X.; Meng, E.; Yu, S. A meta-analysis of passive smoking and risk of developing type 2 diabetes mellitus. Diabetes Res. Clin. Prac. 2015, 107, 9–14. [Google Scholar] [CrossRef]
- Weitzman, M.; Cook, S.R.; Auinger, P.; Florin, T.A.; Daniels, S.; Nguyen, M.; Winickoff, J.P. Tobacco smoke exposure is associated with the metabolic syndrome in adolescents. Circulation 2005, 112, 862–869. [Google Scholar] [CrossRef]
- Hamer, M.; Stamatakis, E.; Batty, G.D. Objectively assessed secondhand smoke exposure and mental health in adults: Cross-sectional and prospective evidence from the Scottish Health Survey. Arch. Gen. Psychiatry 2010, 67, 850–855. [Google Scholar] [CrossRef]
- Chen, R. Association of environmental tobacco smoke with dementia and Alzheimer’s disease among never smokers. Alzheimers Dement. 2012, 8, 590–595. [Google Scholar] [CrossRef]
- Bamoya, J.; Navas-Acien, A. Protecting the world from secondhand tobacco smoke exposure: Where do we stand and where do we go from here? Nicotine Tob. Res. 2013, 15, 789–804. [Google Scholar] [CrossRef]
- Al-Sayed, E.M.; Ibrahim, K.S. Second-hand tobacco smoke and children. Toxicol. Ind. Health 2014, 30, 635–644. [Google Scholar] [CrossRef]
- Leonardi-Bee, J.; Smyth, A.; Britton, J.; Coleman, T. Environmental tobacco smoke and fetal health: Systematic review and meta-analysis. Arch. Dis. Child Fetal Neonatal. Ed. 2008, 93, F351–F361. [Google Scholar] [CrossRef]
- Abraham, M.; Alramadhan, S.; Iniguez, C.; Duijts, L.; Jaddoe, V.W.; den Dekker, H.T.; Crozier, S.; Godfrey, K.M.; Hindmarsh, P.; Vik, T.; et al. A systematic review of maternal smoking during pregnancy and fetal measurements with meta-analysis. PLoS ONE 2017, 12, e0170946. [Google Scholar] [CrossRef]
- Anderson, H.R.; Cook, D.G. Passive smoking and sudden infant death syndrome: Review of the epidemiological evidence. Thorax 1997, 52, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- Adgent, M.A. Environmental tobacco smoke and sudden infant death syndrome: A review. Birth Defects Res. B Dev. Reprod. Toxicol. 2006, 77, 69–85. [Google Scholar] [CrossRef] [PubMed]
- Gould, G.S.; Havard, A.; Lim, L.L.; Kumar, R. Exposure to tobacco, environmental tobacco smoke and nicotine in pregnancy: A pragmatic overview of reviews of maternal and child outcomes, effectiveness of interventions and barriers and facilitators to quitting. Int. J. Environ. Res. Public Health 2020, 17, 2034. [Google Scholar] [CrossRef] [PubMed]
- Braun, M.; Klingelhöfer, D.M.; Oremek, G.M.; Quarcoo, D.; David, A.; Groneberg, D.A. Influence of second-hand smoke and prenatal tobacco smoke exposure on biomarkers, genetics and physiological processes in children-an overview in research insights of the last few years. Int. J. Environ. Res. Public Health 2020, 17, 3212. [Google Scholar] [CrossRef] [PubMed]
- Avila-Tang, E.; Al-Delaimy, W.K.; Ashley, D.L.; Benowitz, N.; Bernert, J.T.; Kim, S.; Samet, J.M.; Hecht, S.S. Assessing secondhand smoke using biological markers. Tob. Control. 2013, 22, 164–171. [Google Scholar] [CrossRef]
- Boumba, V.A.; Ziavrou, K.S.; Vougiouklakis, T. Hair as a biological indicator of drug use, drug abuse or chronic expo-sure to environmental toxicants. Int. J. Toxicol. 2006, 25, 143–163. [Google Scholar] [CrossRef]
- Schramm, K.W. Hair-biomonitoring of organic pollutants. Chemosphere 2008, 72, 1103–1111. [Google Scholar] [CrossRef]
- Al-Delaimy, W.K. Hair as a biomarker for exposure to tobacco smoke. Tob. Control. 2002, 11, 176–182. [Google Scholar] [CrossRef]
- Vogliardi, S.; Tucci, M.; Stocchero, G.; Ferrara, S.D.; Favretto, D. Sample preparation methods for determination of drugs of abuse in hair samples: A review. Anal. Chim. Acta 2015, 857, 1–27. [Google Scholar] [CrossRef]
- Pérez-Ortuño, R.; Martínez-Sánchez, J.M.; Fu, M.; Fernández, E.; Pascual, J.A. Evaluation of tobacco specific nitrosamines exposure by quantification of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) in human hair of non-smokers. Sci. Rep. 2016, 6, 25043. [Google Scholar] [CrossRef]
- Benowitz, N.L.; Hukkanen, J.; Jacob, P. Nicotine chemistry, metabolism, kinetics and biomarkers. Handb. Exp. Pharmacol. 2009, 192, 29–60. [Google Scholar] [CrossRef]
- Kataoka, H.; Inoue, R.; Yagi, K.; Saito, K. Determination of nicotine, cotinine, and related alkaloids in human urine and saliva by automated in-tube solid-phase microextraction coupled with liquid chromatography-mass spectrometry. J. Pharm. Biomed. Anal. 2009, 49, 108–114. [Google Scholar] [CrossRef]
- Inukai, T.; Kaji, S.; Kataoka, H. Analysis of nicotine and cotinine in hair by on-line in-tube solid-phase microextraction coupled with liquid chromatography-tandem mass spectrometry as biomarkers of exposure to tobacco smoke. J. Pharm. Biomed. Anal. 2018, 156, 272–277. [Google Scholar] [CrossRef]
- Ishizaki, A.; Kataoka, H. In-tube solid-phase microextraction coupled to liquid chromatography-tandem mass spectrometry for the determination of tobacco-specific nitrosamines in hair samples. Molecules 2021, 26, 2056. [Google Scholar] [CrossRef]
- Kataoka, H. In-tube solid-phase microextraction: Current trends and future perspectives. J. Chromatogr. A 2021, 1636, 461787. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Ishizaki, A.; Kataoka, H. Biomonitoring method for the determination of polycyclic aromatic hydrocarbons in hair by online in-tube solid-phase microextraction coupled with high performance liquid chromatography and fluorescence detection. J. Chromatogr. B 2015, 1000, 187–191. [Google Scholar] [CrossRef]
- Kataoka, H.; Ehara, K.; Yasuhara, R.; Saito, K. Simultaneous determination of testosterone, cortisol and dehydroepiandrosterone in saliva by stable isotope dilution on-line in-tube solid-phase microextraction coupled with liquid chromatography-tandem mass spectrometry. Anal. Bioanal. Chem. 2013, 405, 331–340. [Google Scholar] [CrossRef]
- Balcerek, M.; Matławska, I. Preventive role of curcumin in lung cancer. Prz. Lek. 2005, 62, 1180–1181. [Google Scholar]
| Question/Answer | n | Content in Hair/Mean (Min.~Max.) | |
|---|---|---|---|
| Nicotine (ng mg−1) | Cotinine (pg mg−1) | ||
| 1. Sex | |||
| Male | 30 | 1.63 (0.20~7.72) | 14.4 (4.8~43.9) |
| Female | 80 | 1.29 (0.07~4.99) | 12.2 (2.7~99.1) |
| P 1 (Male/female) | 0.120 | 0.234 | |
| 2. Age | |||
| 29 years old and under | 79 | 1.39 (0.07~5.40) | 12.1 (2.7~99.1) |
| 30 years old and over | 31 | 1.37 (0.17~7.72) | 14.6 (4.3~59.3) |
| P (Younger/older) | 0.477 | 0.198 | |
| 3. Daily sleeping time | |||
| Less than 6 h | 30 | 1.19 (0.07~3.89) | 10.8 (4.1~59.3) |
| More than 6 h | 80 | 1.45 (0.10~37.72) | 13.6 (2.7~99.1) |
| P (Shorter/longer) | 0.188 | 0.171 | |
| 4. Stress awareness | |||
| Sometimes 2 | 68 | 1.39 (0.07~7.72) | 13.8 (2.7~99.1) |
| Frequently 2 | 42 | 1.38 (0.17~4.99) | 11.2 (4.4~59.3) |
| P (Sometimes/frequently) | 0.483 | 0.169 | |
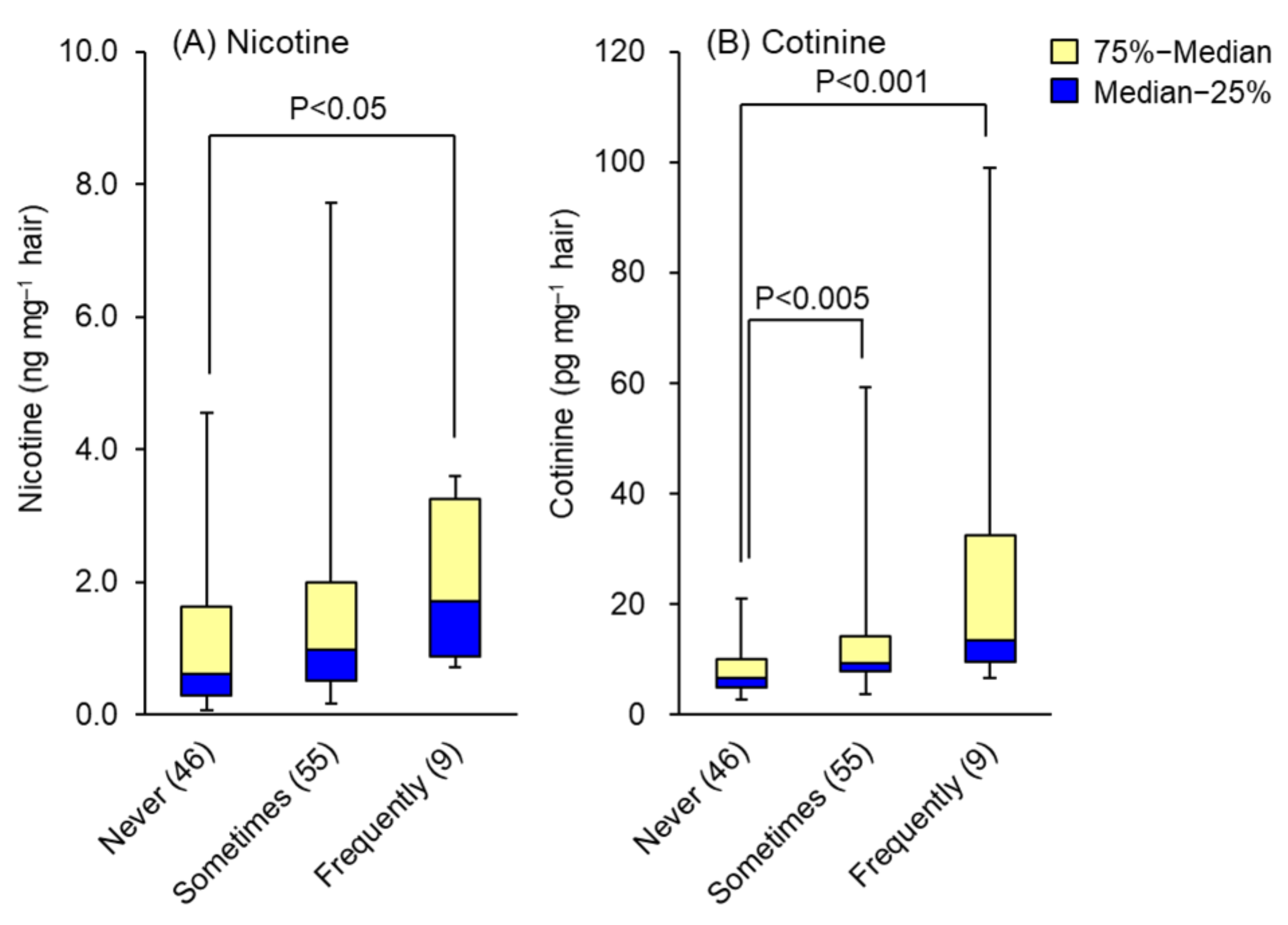
| 5. Passive smoking awareness | |||
| Never | 46 | 1.11 (0.07~4.55) | 8.1 (2.7~21.0) |
| Sometimes | 55 | 1.52 (0.16~7.72) | 13.8 (3.8~59.3) |
| Frequently | 9 | 1.94 (0.72~3.60) | 31.0 (6.6~99.1) |
| P (Never/sometimes) | 0.064 | 0.001 | |
| P (Never/frequently) | 0.023 | 0.00001 | |
| 6. Exposure to other smoke | |||
| Never | 55 | 1.35 (0.12~5.40) | 14.0 (2.7~99.1) |
| Sometimes~Always | 55 | 1.42 (0.07~7.72) | 11.6 (3.8~47.0) |
| P (Never/yes) | 0.397 | 0.177 | |
| 7. Frequency of tea drinking | |||
| Sometimes/Frequently | 46 | 1.49 (0.17~7.72) | 12.4 (4.0~59.3) |
| Always | 64 | 1.30 (0.07~5.40) | 13.1 (2.7~99.1) |
| P (Less/more) | 0.241 | 0.409 | |
| 8. Fat and fatty food intake | |||
| Sometimes | 8 | 1.82 (0.37~7.72) | 12.7 (5.1~35.2) |
| Frequently | 81 | 1.27 (0.07~4.99) | 11.8 (2.7~99.1) |
| Always | 21 | 1.64 (0.20~5.40) | 16.9 (3.8~47.0) |
| P (Frequently/always) | 0.405 | 0.227 | |
| 9. Vegetable intake | |||
| Sometimes | 31 | 1.27 (0.10~4.54) | 9.7 (2.7~32.5) |
| Frequently | 64 | 1.36 (0.07~7.72) | 15.1 (4.0~99.1) |
| Always | 15 | 1.71 (0.20~4.99) | 9.4 (3.8~22.0) |
| P (Sometimes/always) | 0.135 | 0.450 | |
| 10. Consumption of vegetables | |||
| Raw | 55 | 1.29 (0.07~7.72) | 12.0 (2.7~99.1) |
| Boiled | 21 | 1.30 (0.12~3.60) | 13.0 (4.3~59.3) |
| Pan-fried | 34 | 1.57 (0.10~5.42) | 14.0 (3.8~67.0) |
| P (Raw/pan-fried) | 0.232 | 0.390 | |
| P (Boiled/pan-fried) | 0.192 | 0.249 | |
| 11. Spice use | |||
| Sometimes | 36 | 1.54 (0.12~5.40) | 12.7 (4.0~59.3) |
| Frequently | 58 | 1.41 (0.10~7.72) | 13.7 (2.7~99.1) |
| Always | 16 | 0.93 (0.07~3.48) | 9.7 (4.9~31.7) |
| P (Sometimes/always) | 0.055 | 0.169 | |
| 12. Meat intake | |||
| Sometimes | 14 | 1.20 (0.28~4.99) | 10.5 (5.1~31.7) |
| Frequently | 67 | 1.47 (0.07~7.72) | 13.5 (2.7~99.1) |
| Always | 29 | 1.28 (0.12~3.60) | 12.2 (4.8~67.0) |
| P (Sometimes/always) | 0.414 | 0.311 | |
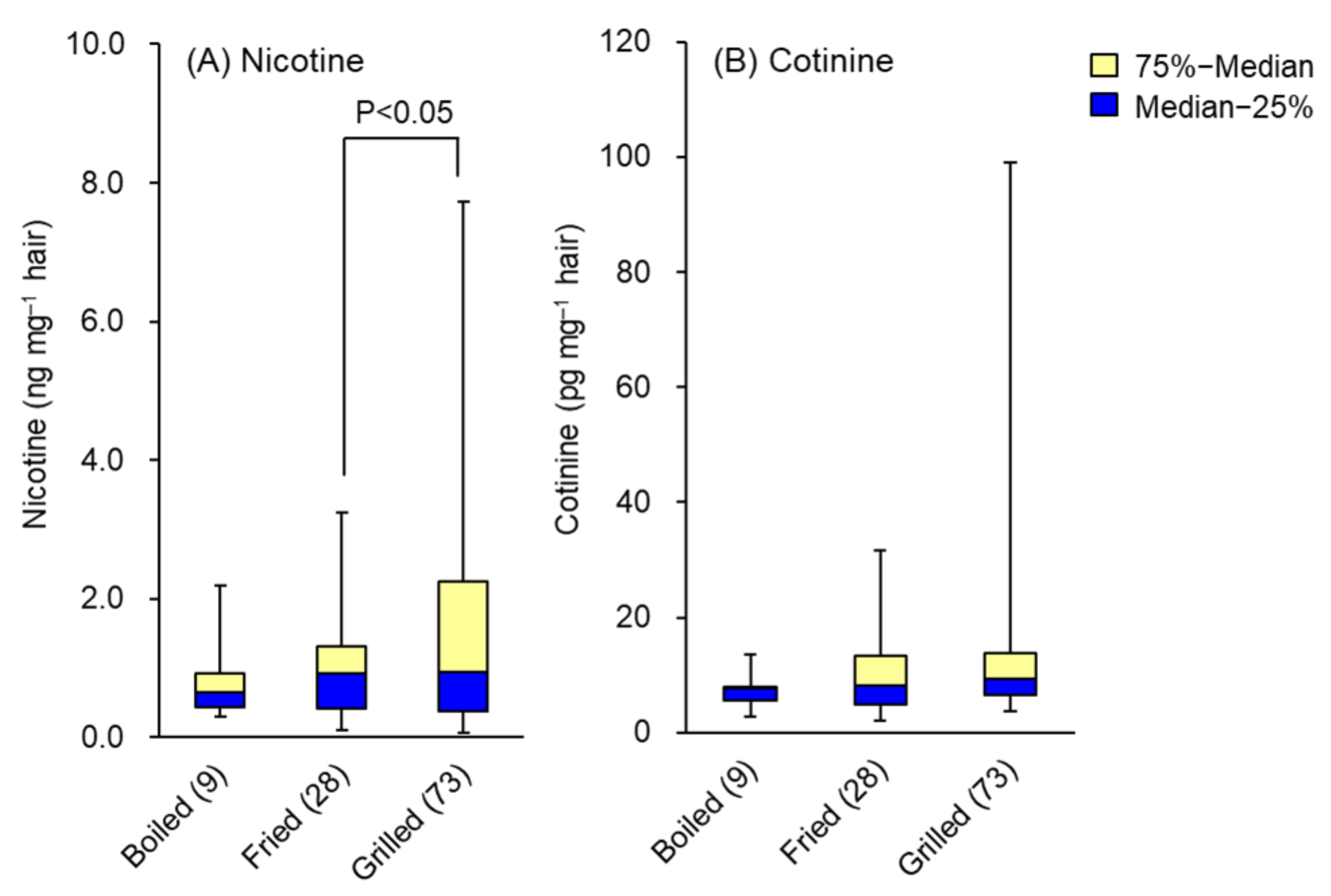
| 13. Consumption of meat | |||
| Boiled | 9 | 0.81 (0.30~2.19) | 7.6 (5.1~13.7) |
| Pan-fried/Deep-fried | 28 | 1.02 (0.10~3.25) | 9.6 (2.7~31.7) |
| Grilled | 73 | 1.59 (0.07~7.72) | 19.1 (4.0~99.1) |
| P (Boiled/grilled) | 0.071 | 0.096 | |
| P (Fried/grilled) | 0.034 | 0.053 | |
| 14. Seafood intake | |||
| Sometimes | 48 | 1.40 (0.12~5.40) | 12.0 (2.7~47.0) |
| Frequently | 54 | 1.37 (0.07~7.72) | 14.1 (3.8~99.1) |
| Always | 8 | 1.24 (0.17~3.44) | 8.9 (5.6~1.45) |
| P (Sometimes/always) | 0.478 | 0.205 | |
| 15. Consumption of seafood | |||
| Raw | 21 | 1.12 (0.12~4.99) | 17.9 (4.4~99.1) |
| Boiled | 33 | 1.38 (0.29~5.40) | 12.6 (4.0~43.9) |
| Grilled | 56 | 1.48 (0.07~7.72) | 11.0 (2.7~67.0) |
| P (Raw/grilled) | 0.155 | 0.036 | |
| P (Boiled/grilled) | 0.369 | 0.228 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kataoka, H.; Kaji, S.; Moai, M. Risk Assessment of Passive Smoking Based on Analysis of Hair Nicotine and Cotinine as Exposure Biomarkers by In-Tube Solid-Phase Microextraction Coupled On-Line to LC-MS/MS. Molecules 2021, 26, 7356. https://doi.org/10.3390/molecules26237356
Kataoka H, Kaji S, Moai M. Risk Assessment of Passive Smoking Based on Analysis of Hair Nicotine and Cotinine as Exposure Biomarkers by In-Tube Solid-Phase Microextraction Coupled On-Line to LC-MS/MS. Molecules. 2021; 26(23):7356. https://doi.org/10.3390/molecules26237356
Chicago/Turabian StyleKataoka, Hiroyuki, Sanae Kaji, and Maki Moai. 2021. "Risk Assessment of Passive Smoking Based on Analysis of Hair Nicotine and Cotinine as Exposure Biomarkers by In-Tube Solid-Phase Microextraction Coupled On-Line to LC-MS/MS" Molecules 26, no. 23: 7356. https://doi.org/10.3390/molecules26237356
APA StyleKataoka, H., Kaji, S., & Moai, M. (2021). Risk Assessment of Passive Smoking Based on Analysis of Hair Nicotine and Cotinine as Exposure Biomarkers by In-Tube Solid-Phase Microextraction Coupled On-Line to LC-MS/MS. Molecules, 26(23), 7356. https://doi.org/10.3390/molecules26237356







