A Green Method of Extracting and Recovering Flavonoids from Acanthopanax senticosus Using Deep Eutectic Solvents
Abstract
:1. Introduction
2. Results
2.1. Determination of Total Flavonoids Content
2.2. Physical and Chemical Properties of DESs
2.3. Selection of DESs
2.4. Fourier-Transform Infrared Spectroscopy (FT-IR) Analysis of Gly-La DES
2.5. Selection of the Most Suitable Ultrasonic Power Level
2.6. Determination of the Optimal HBA-HBD Ratio
2.7. Selection of the Most Suitable Water Content
2.8. Determination of the Optimal Solid-Liquid Ratio
2.9. Selection of the Suitable Extraction Temperature
2.10. Selection of the Optimal Extraction Time
2.11. RSM Optimization Test Data and Analysis
2.11.1. RSM Optimization Test
2.11.2. Analysis of the Response Surface Plot
2.11.3. Model Verification and Method Comparison
2.12. SEM Observation of AS Powder Microstructure
2.13. Recovery of Total Flavonoids and Reusability of DES
2.13.1. Screening for the Resin
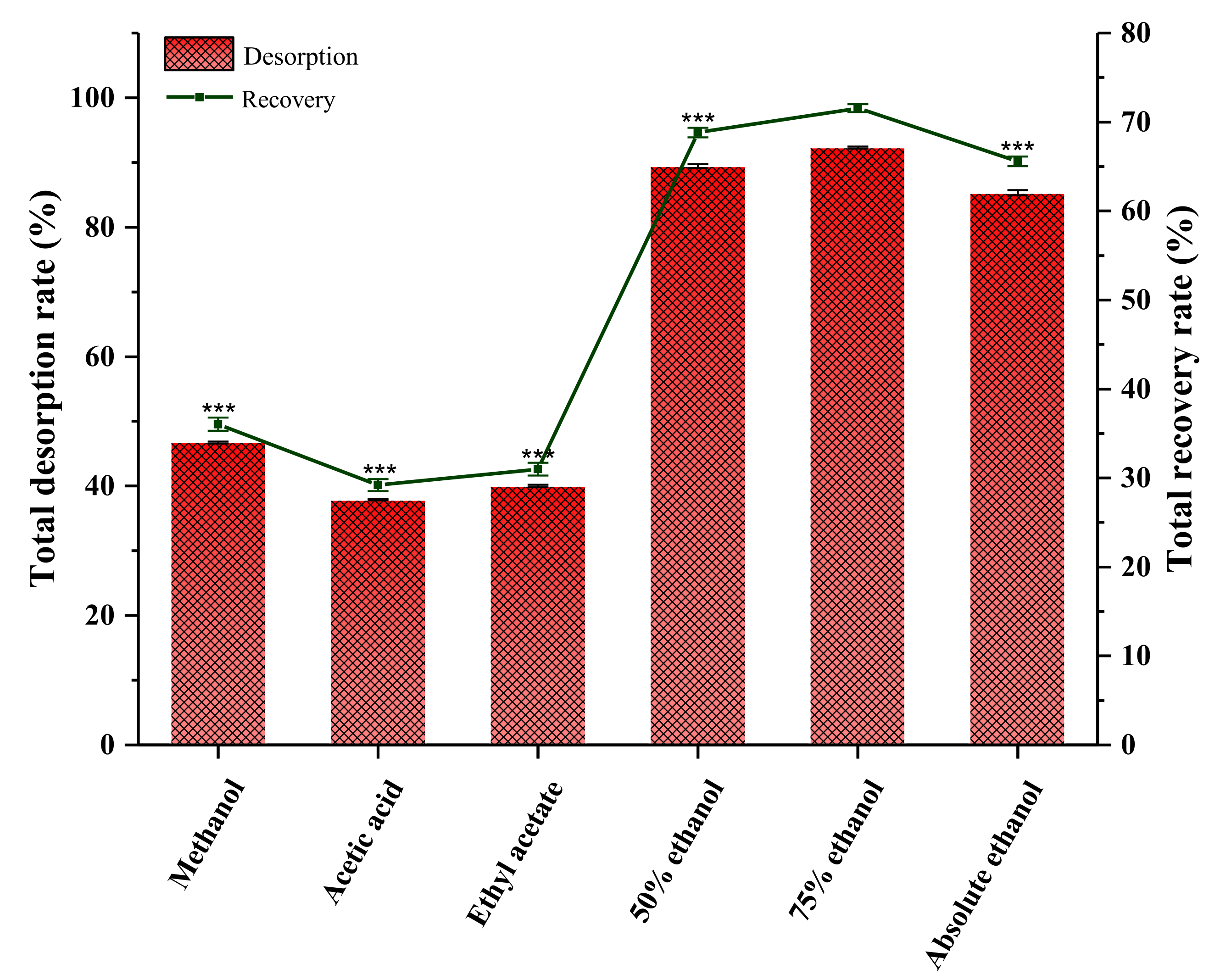
2.13.2. Screening Eluent Solvent
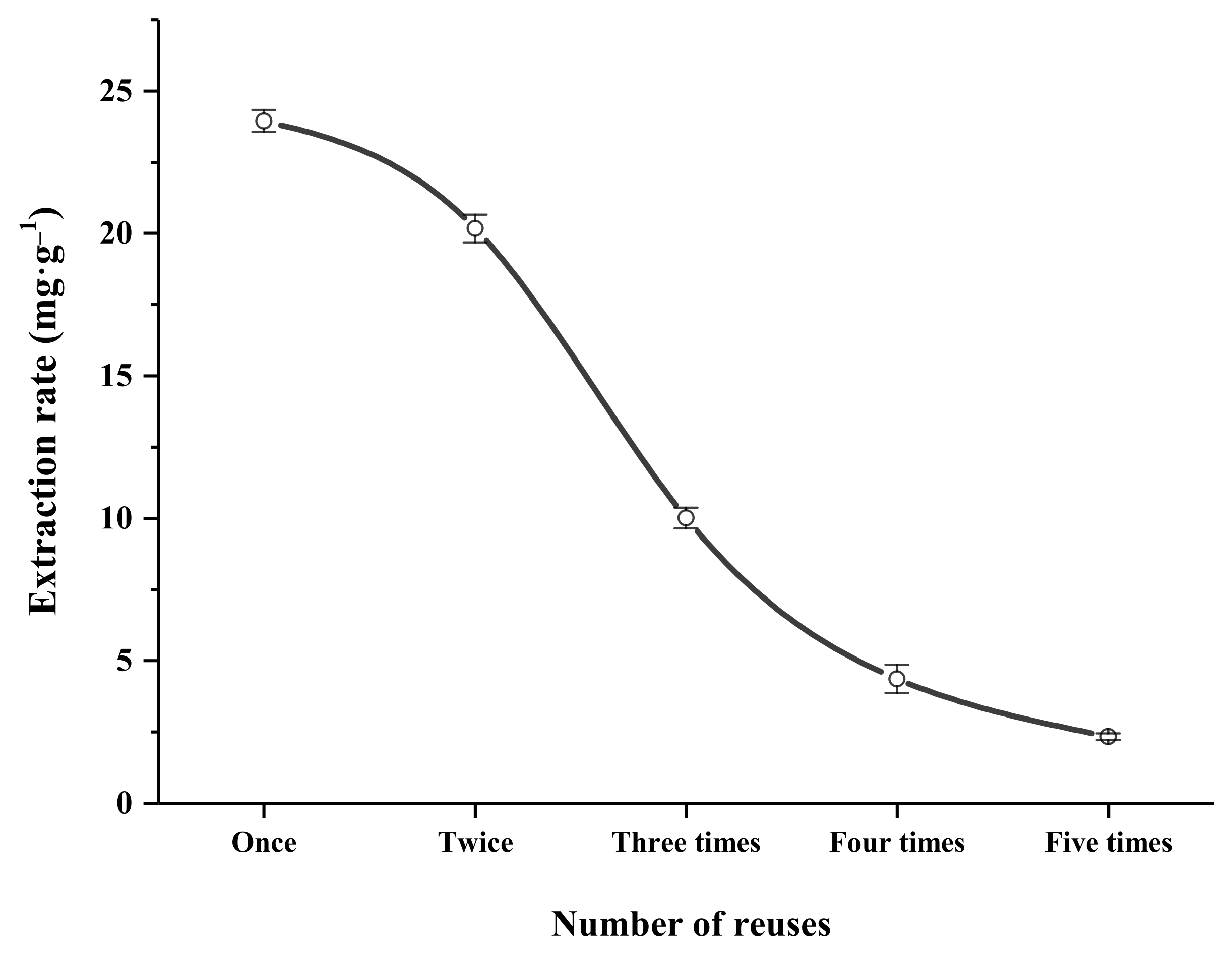
2.13.3. Reusability of DES
3. Materials and Methods
3.1. Reagents and Instruments
3.2. Experimental Methods
3.2.1. Preparation of DESs
3.2.2. Determination of pH and Viscosity of DES
3.2.3. Determination of Total Flavonoids Content
3.2.4. Extraction Process
3.2.5. Single Factor Effects
3.2.6. Optimization Extraction Method with RSM
3.2.7. Fourier-Infrared Spectrophotometry
3.2.8. SEM Observation of AS Powder Microstructure
3.2.9. Total Flavonoid Recovery Using Macroporous Resin and Reusability of DES
3.3. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Meng, Q.; Pan, J.; Liu, Y.; Chen, L.; Ren, Y. Anti-tumour effects of polysaccharide extracted from Acanthopanax�senticosus and cell-mediated immunity. Exp. Ther. Med. 2017, 15, 1694–1701. [Google Scholar] [CrossRef] [PubMed]
- Qun, P.; Jianyuan, C.; Hanying, D.; Chao, W. Determination of Kaurenoic Acid in Acanthopanax trifoliatus by Ultra-High Performance Liquid Chromatography Coupled with Tandem Mass Spectrometry (UHPLC-MS/MS). Sci. Rep. 2020, 10, 1–8. [Google Scholar]
- Ailing, J.; Yuhang, Z.; Han, G.; Zhen, Z.; Yanfei, Z.; Zhe, W.; Jinming, Z.; Bin, D.; Zhidong, Q.; Chaomei, F. A review of Acanthopanax senticosus (Rupr and Maxim.) harms: From ethnopharmacological use to modern application. J. Ethnopharmacol. 2021, 268, 113586. [Google Scholar]
- YunQiang, Z.; YunLu, Z.; ZiKui, L. Effects of Acanthopanax senticosus supplementation on innate immunity and changes of related immune factors in healthy mice. Innate Immun. 2020, 27, 1753425920955200. [Google Scholar] [CrossRef]
- Hongxia, L.; Jun, Q.; Changyou, S.; Pao, X. Transcriptome profiling reveal Acanthopanax senticosus improves growth performance, immunity and antioxidant capacity by regulating lipid metabolism in GIFT (Oreochromis niloticus). Comp. Biochem. Physiol.—Part D Genom. Proteom. 2021, 37, 100784. [Google Scholar]
- Haohao, W.; Chen, C.; Ruijuan, L.; Xu, W.; Yan, Z.; Zhaowei, Y.; Enbo, C.; Hongyan, Z. Potential Myocardial Protection of 3,4-seco-Lupane Triterpenoids from Acanthopanax sessiliflorus Leaves. Chem. Biodivers. 2020, 18, e2000830. [Google Scholar]
- Li, Q.; Luo, J.; Liu, X.; Kwon, D.; Kang, O. Eleutheroside K isolated from Acanthopanax henryi (Oliv.) Harms suppresses methicillin resistance of Staphylococcus aureus. Lett. Appl. Microbiol. 2020, 72, 669–676. [Google Scholar] [CrossRef]
- Chunyun, W.; Hongbo, G.; Enbo, C.; Lianxue, Z.; Xiaoman, Z.; Shengbo, Z.; Nian, S.; Yan, Z. Protective effects of Acanthopanax senticosus—Ligustrum lucidum combination on bone marrow suppression induced by chemotherapy in mice. Biomed. Pharmacother. 2019, 109, 2062–2069. [Google Scholar]
- Płotka-Wasylka, J.; Rutkowska, M.; Owczarek, K.; Tobiszewski, M.; Namiesnik, J. Extraction with environmentally friendly solvents. TrAC Trends Anal. Chem. 2017, 91, 12–25. [Google Scholar] [CrossRef]
- Ivanović, M.; Alañón, M.E.; Arráez-Román, D.; Segura-Carretero, A. Enhanced and green extraction of bioactive compounds from Lippia citriodora by tailor-made natural deep eutectic solvents. Food Res. Int. 2018, 111, 67–76. [Google Scholar] [CrossRef]
- Katarzyna, T.; Marcin, K.; Edward, R. The Application of Supercritical Fluid Extraction in Phenolic Compounds Isolation from Natural Plant Materials. Molecules 2018, 23, 2625. [Google Scholar]
- Zhou, Y.; Wu, D.; Cai, P.; Cheng, G.; Huang, C.; Pan, Y. Special Effect of Ionic Liquids on the Extraction of Flavonoid Glycosides from Chrysanthemum morifolium Ramat by Microwave Assistance. Molecules 2015, 20, 7683–7699. [Google Scholar] [CrossRef]
- Flieger, J.; Flieger, M. Ionic Liquids Toxicity—Benefits and Threats. Int. J. Mol. Sci. 2020, 21, 6267. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Munro, H.L.; Rasheed, R.K.; Tambyrajah, V. Preparation of novel, moisture-stable, Lewis-acidic ionic liquids containing quaternary ammonium salts with functional side chains. Chem. Commun. 2001, 19, 2010–2011. [Google Scholar] [CrossRef] [Green Version]
- Zhuang, B.; Dou, L.-L.; Li, P.; Liu, E.-H. Deep eutectic solvents as green media for extraction of flavonoid glycosides and aglycones from Platycladi Cacumen. J. Pharm. Biomed. Anal. 2017, 134, 214–219. [Google Scholar] [CrossRef]
- Lamei, N.; Ezoddin, M.; Abdi, K. Air assisted emulsification liquid-liquid microextraction based on deep eutectic solvent for preconcentration of methadone in water and biological samples. Talanta 2017, 165, 176–181. [Google Scholar] [CrossRef]
- Sheldon, R.A.; Woodley, J.M. Role of Biocatalysis in Sustainable Chemistry. Chem. Rev. 2018, 118, 801–838. [Google Scholar] [CrossRef]
- Xizhe, F.; Di, W.; Tarun, B.; Jing, X.; Yanqun, X.; Li, L.; Ligen, Z.; Lixia, Z.; Zisheng, L. Natural deep eutectic solvent enhanced pulse-ultrasonication assisted extraction as a multi-stability protective and efficient green strategy to extract anthocyanin from blueberry pomace. LWT 2021, 144, 111220. [Google Scholar]
- Yan, H.; Jiehong, Y.; Yu, Z.; Li, Y.; Yu, H.; Haitong, W.; Chang, L. Screening, Optimization, and Bioavailability Research of Natural Deep Eutectic Solvent Extracts from Radix Pueraria. Molecules 2021, 26, 729. [Google Scholar]
- Jianxiong, Y.; Zhenbao, Z.; Jianhua, Y.; Yang, L.; Bingcan, C.; Jiajia, R. Structure and functionality of oat protein extracted by choline chloride-dihydric alcohol deep eutectic solvent and its water binary mixtures. Food Hydrocoll. 2021, 112, 106330. [Google Scholar]
- da Silva, D.T.; Rodrigues, R.F.; Machado, N.M.; Maurer, L.H.; Ferreira, L.F.; Somacal, S.; da Leite, V.M.; da Rocha, R.M.I.d.U.M.; Marcia, V.; Eliseu, R.; et al. Natural deep eutectic solvent (NADES)-based blueberry extracts protect against ethanol-induced gastric ulcer in rats. Food Res. Int. 2020, 138, 109718. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Feng, F.; Jiang, J.; Qiao, Y.; Wu, T.; Voglmeir, J.; Chen, Z.-G. Green and efficient extraction of rutin from tartary buckwheat hull by using natural deep eutectic solvents. Food Chem. 2017, 221, 1400–1405. [Google Scholar] [CrossRef] [PubMed]
- Jeong, K.M.; Ko, J.; Zhao, J.; Jin, Y.; Yoo, D.E.; Han, S.Y.; Lee, J. Multi-functioning deep eutectic solvents as extraction and storage media for bioactive natural products that are readily applicable to cosmetic products. J. Clean. Prod. 2017, 151, 87–95. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, M.; Tan, T.; Yan, A.; Guo, L.; Jiang, K.; Tan, C.; Wan, Y. Deep eutectic solvents used as extraction solvent for the determination of flavonoids from Camellia oleifera flowers by high-performance liquid chromatography. Phytochem. Anal. 2018, 29, 639–648. [Google Scholar] [CrossRef] [PubMed]
- Su, E.; Yang, M.; Cao, J.; Lu, C.; Wang, J.; Cao, F. Deep eutectic solvents as green media for efficient extraction of terpene trilactones from Ginkgo biloba leaves. J. Liq. Chromatogr. Relat. Technol. 2017, 40, 385–391. [Google Scholar] [CrossRef]
- Zhao, B.-Y.; Xu, P.; Yang, F.-X.; Wu, H.; Zong, M.-H.; Lou, W.-Y. Biocompatible Deep Eutectic Solvents Based on Choline Chloride: Characterization and Application to the Extraction of Rutin from Sophora japonica. ACS Sustain. Chem. Eng. 2015, 3, 2746–2755. [Google Scholar] [CrossRef]
- Duan, L.; Dou, L.-L.; Guo, L.; Li, P.; Liu, E.-H. Comprehensive Evaluation of Deep Eutectic Solvents in Extraction of Bioactive Natural Products. ACS Sustain. Chem. Eng. 2016, 4, 2405–2411. [Google Scholar] [CrossRef]
- Wang, H.; Ma, X.; Cheng, Q.; Wang, L.; Zhang, L. Deep Eutectic Solvent-Based Ultrahigh Pressure Extraction of Baicalin from Scutellaria baicalensis Georgi. Molecules 2018, 23, 3233. [Google Scholar] [CrossRef] [Green Version]
- Qin, H.; Hu, X.; Wang, J.; Cheng, H.; Chen, L.; Qi, Z. Overview of acidic deep eutectic solvents on synthesis, properties and applications. Green Energy Environ. 2020, 5, 8–21. [Google Scholar] [CrossRef]
- Kumar, S.; Pandey, A.K. Chemistry and Biological Activities of Flavonoids: An Overview. Sci. World J. 2013, 2013, 162750. [Google Scholar] [CrossRef] [Green Version]
- Hao, C.; Chen, L.; Dong, H.; Xing, W.; Xue, F.; Cheng, Y. Extraction of Flavonoids from Scutellariae Radix using Ultrasound-Assisted Deep Eutectic Solvents and Evaluation of Their Anti-Inflammatory Activities. ACS Omega 2020, 5, 23140–23147. [Google Scholar] [CrossRef]
- Nia, N.N.; Hadjmohammadi, M.R. The application of three-phase solvent bar microextraction based on a deep eutectic solvent coupled with high-performance liquid chromatography for the determination of flavonoids from vegetable and fruit juice samples. Anal. Methods 2019, 11, 5134–5141. [Google Scholar] [CrossRef]
- Bubalo, M.C.; Ćurko, N.; Tomašević, M.; Ganić, K.K.; Redovniković, I.R. Green extraction of grape skin phenolics by using deep eutectic solvents. Food Chem. 2016, 200, 159–166. [Google Scholar] [CrossRef]
- Lu, L.; JuZhao, L.; Meng, L.; Wei, W.; YuYan, H.; Thomas, E.; HuiMei, W.; YuJie, F. Efficient extraction and preparative separation of four main isoflavonoids from Dalbergia odorifera T. Chen leaves by deep eutectic solvents-based negative pressure cavitation extraction followed by macroporous resin column chromatography. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2016, 40-48, 1033–1034. [Google Scholar]
- Mjalli, F.S.; Naser, J. Viscosity model for choline chloride-based deep eutectic solvents. Asia-Pac. J. Chem. Eng. 2015, 10, 273–281. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, Q.; Zhu, T. Comparison of hydrophilic and hydrophobic deep eutectic solvents for pretreatment determination of sulfonamides from aqueous environments. Anal. Methods 2019, 11, 5901–5909. [Google Scholar] [CrossRef]
- Aljerf, L.; Nadra, R. Developed greener method based on MW implementation in manufacturing CNFs. Int. J. Nanomanuf. 2019, 15, 269–289. [Google Scholar] [CrossRef]
- Socas-Rodríguez, B.; Torres-Cornejo, M.; Álvarez-Rivera, G.; Mendiola, J. Deep Eutectic Solvents for the Extraction of Bioactive Compounds from Natural Sources and Agricultural By-Products. Appl. Sci. 2021, 11, 4897. [Google Scholar] [CrossRef]
- Smith, E.L.; Abbott, A.P.; Ryder, K.S. Deep eutectic solvents (DESs) and their applications. Chem. Rev. 2014, 114, 11060–11082. [Google Scholar] [CrossRef] [Green Version]
- Kalhor, P.; Ghandi, K. Deep Eutectic Solvents for Pretreatment, Extraction, and Catalysis of Biomass and Food Waste. Molecules 2019, 24, 4012. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Lang, J.; Lan, P.; Yang, H.; Lu, J.; Wang, Z. Study on the Dissolution Mechanism of Cellulose by ChCl-Based Deep Eutectic Solvents. Materials 2020, 13, 278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mulia, K.; Fauzia, F.; Krisanti, E.A. Polyalcohols as Hydrogen-Bonding Donors in Choline Chloride-Based Deep Eutectic Solvents for Extraction of Xanthones from the Pericarp of Garcinia mangostana L. Molecules 2019, 24, 636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbott, A.P.; Capper, G.; Gray, S. Design of improved deep eutectic solvents using hole theory. Chemphyschem A Eur. J. Chem. Phys. Phys. Chem. 2006, 7, 803–806. [Google Scholar] [CrossRef] [PubMed]
- Patil, S.S.; Pathak, A.; Rathod, V.K. Optimization and kinetic study of ultrasound assisted deep eutectic solvent based extraction: A greener route for extraction of curcuminoids from Curcuma longa. Ultrason. Sonochem. 2021, 70, 105267. [Google Scholar] [CrossRef]
- Rozas, S.; Benito, C.; Alcalde, R.; Atilhan, M.; Aparicio, S. Insights on the water effect on deep eutectic solvents properties and structuring: The archetypical case of choline chloride + ethylene glycol. J. Mol. Liq. 2021, 344, 117717. [Google Scholar] [CrossRef]
- Dai, Y.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Tailoring properties of natural deep eutectic solvents with water to facilitate their applications. Food Chem. 2015, 187, 14–19. [Google Scholar] [CrossRef]
- Xu, M.; Ran, L.; Chen, N.; Fan, X.; Ren, D.; Yi, L. Polarity-dependent extraction of flavonoids from citrus peel waste using a tailor-made deep eutectic solvent. Food Chem. 2019, 297, 124970. [Google Scholar] [CrossRef]
- Gutiérrez, M.C.; Ferrer, M.L.; Mateo, C.R.; del Monte, F. Freeze-Drying of Aqueous Solutions of Deep Eutectic Solvents: A Suitable Approach to Deep Eutectic Suspensions of Self-Assembled Structures. Langmuir 2009, 25, 5509–5515. [Google Scholar] [CrossRef]
- Ghaedi, H.; Ayoub, M.; Sufian, S.; Shariff, A.M.; Lal, B. The study on temperature dependence of viscosity and surface tension of several Phosphonium-based deep eutectic solvents. J. Mol. Liq. 2017, 241, 500–510. [Google Scholar] [CrossRef]
- Abbott, A.P.; Harris, R.C.; Ryder, K.S. Application of Hole Theory to Define Ionic Liquids by their Transport Properties. J. Phys. Chem. B 2007, 111, 4910–4913. [Google Scholar] [CrossRef]
- Onydinma, U.P.; Aljerf, L.; Obike, A.; Onah, O.E.; Caleb, N.J. Evaluation of Physicochemical characteristics and health risk of polycyclic aromatic hydrocarbons in borehole waters around automobile workshops in Southeastern Nigeria. Groundw. Sustain. Dev. 2021, 14, 100615. [Google Scholar] [CrossRef]
- Li, Y.; Pan, Z.; Wang, B.; Yu, W.; Song, S.; Feng, H.; Zhao, W.; Zhang, J. Ultrasound-assisted extraction of bioactive alkaloids from Phellodendri amurensis cortex using deep eutectic solvent aqueous solutions. New J. Chem. 2020, 44, 9172–9178. [Google Scholar] [CrossRef]
- Mansur, A.R.; Song, N.-E.; Jang, H.W.; Lim, T.-G.; Yoo, M.; Nam, T.G. Optimizing the ultrasound-assisted deep eutectic solvent extraction of flavonoids in common buckwheat sprouts. Food Chem. 2019, 293, 438–445. [Google Scholar] [CrossRef]
- Nam, M.W.; Zhao, J.; Lee, M.S.; Jeong, J.H.; Lee, J. Enhanced extraction of bioactive natural products using tailor-made deep eutectic solvents: Application to flavonoid extraction from Flos sophorae. Green Chem. 2015, 17, 1718–1727. [Google Scholar] [CrossRef]
- Na, G.; Peng, Z.Y.; Kun, L.H.; Ping, K.; Ming, L.Z.; Jie, F.Y. Effective extraction and recovery of linarin from Chrysanthemum indicum L. flower using deep eutectic solvents. Microchem. J. 2020, 159, 105586. [Google Scholar]
- Dong, Y.; Zhao, M.; Sun-Waterhouse, D.; Zhuang, M.; Chen, H.; Feng, M.; Lin, L. Absorption and desorption behaviour of the flavonoids from Glycyrrhiza glabra L. leaf on macroporous adsorption resins. Food Chem. 2015, 168, 538–545. [Google Scholar] [CrossRef]
- Hou, M.; Hu, W.; Xiu, Z.; Shi, Y.; Hao, K.; Cao, D.; Guan, Y.; Yin, H. Efficient enrichment of total flavonoids from Pteris ensiformis Burm. extracts by macroporous adsorption resins and in vitro evaluation of antioxidant and antiproliferative activities. J. Chromatogr. B 2020, 1138, 121960. [Google Scholar] [CrossRef]
- Zeng, J.; Shang, X.; Zhang, P.; Wang, H.; Gu, Y.; Tan, J.-N. Combined Use of Deep Eutectic Solvents, Macroporous Resins, and Preparative Liquid Chromatography for the Isolation and Purification of Flavonoids and 20-Hydroxyecdysone from Chenopodium quinoa Willd. Biomolecules 2019, 9, 776. [Google Scholar] [CrossRef] [Green Version]
- Tang, B.; Park, H.E.; Row, K.H. Simultaneous Extraction of Flavonoids from Chamaecyparis obtusa Using Deep Eutectic Solvents as Additives of Conventional Extractions Solvents. J. Chromatogr. Sci. 2014, 53, 836–840. [Google Scholar] [CrossRef] [Green Version]
- Feng, S.; Luo, Z.; Zhang, Y.; Zhong, Z.; Lu, B. Phytochemical contents and antioxidant capacities of different parts of two sugarcane (Saccharum officinarum L.) cultivars. Food Chem. 2014, 151, 452–458. [Google Scholar] [CrossRef]
- Zhu, Y.; Yang, L.; Zhang, C.; Tian, Y.; Zhang, F.; Li, X. Structural and functional analyses of three purified polysaccharides isolated from Chinese Huaishan-yams. Int. J. Biol. Macromol. 2018, 120, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Bao, J.; Cai, Y.; Sun, M.; Wang, G.; Corke, H. Anthocyanins, Flavonols, and Free Radical Scavenging Activity of Chinese Bayberry (Myrica rubra) Extracts and Their Color Properties and Stability. J. Agric. Food Chem. 2005, 53, 2327–2332. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Zhao, M.; Lin, L. Adsorption and desorption characteristics of adlay bran free phenolics on macroporous resins. Food Chem. 2016, 194, 900–907. [Google Scholar] [CrossRef] [PubMed]
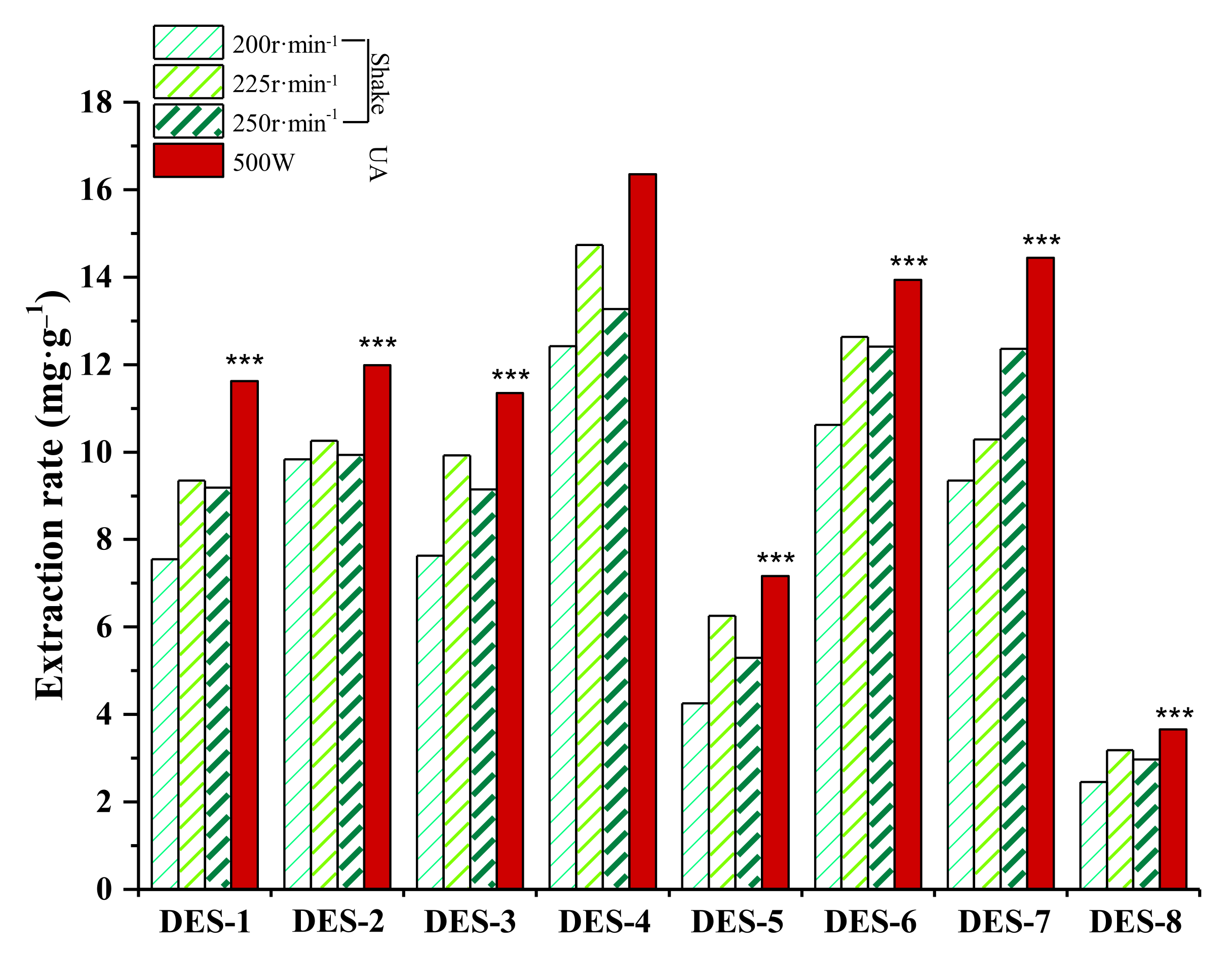
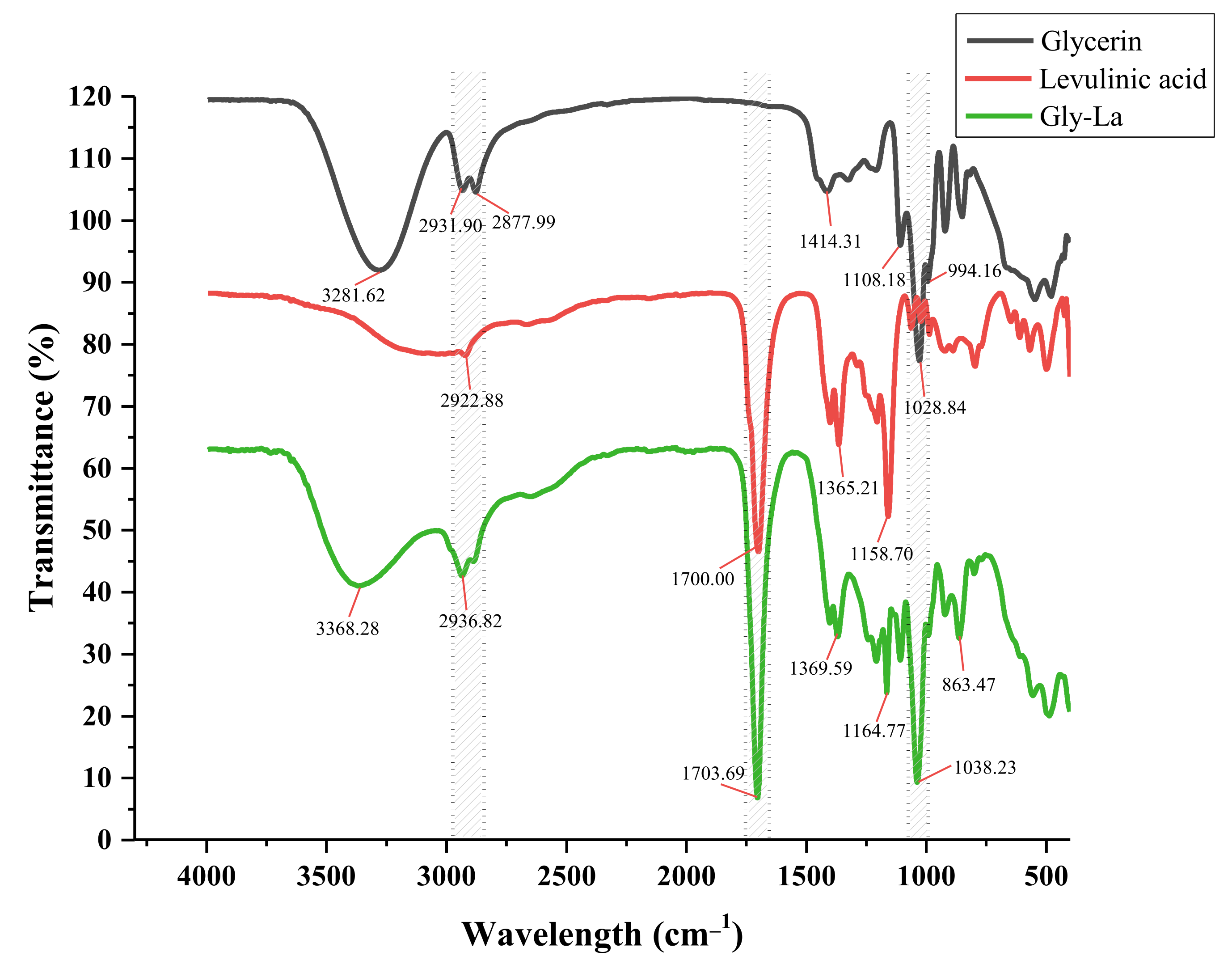
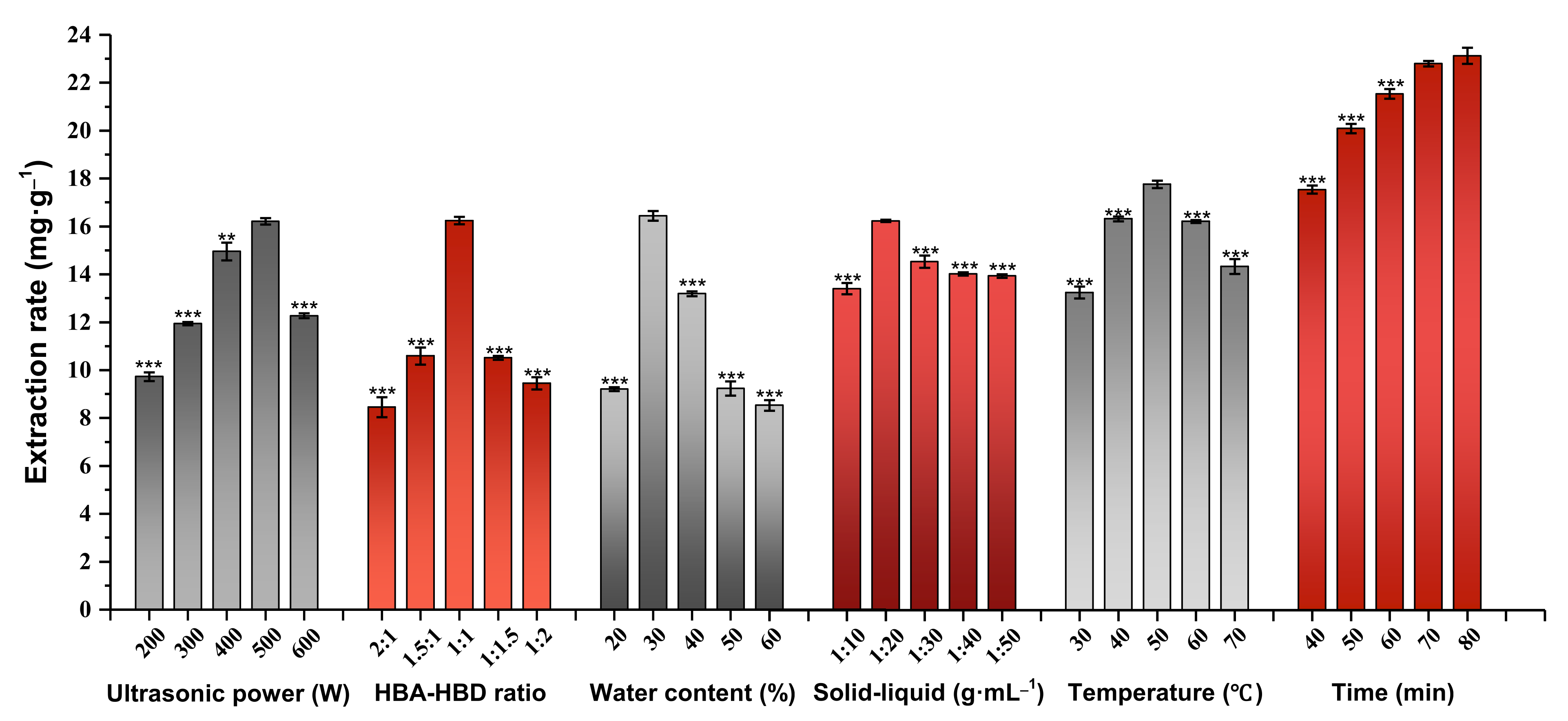
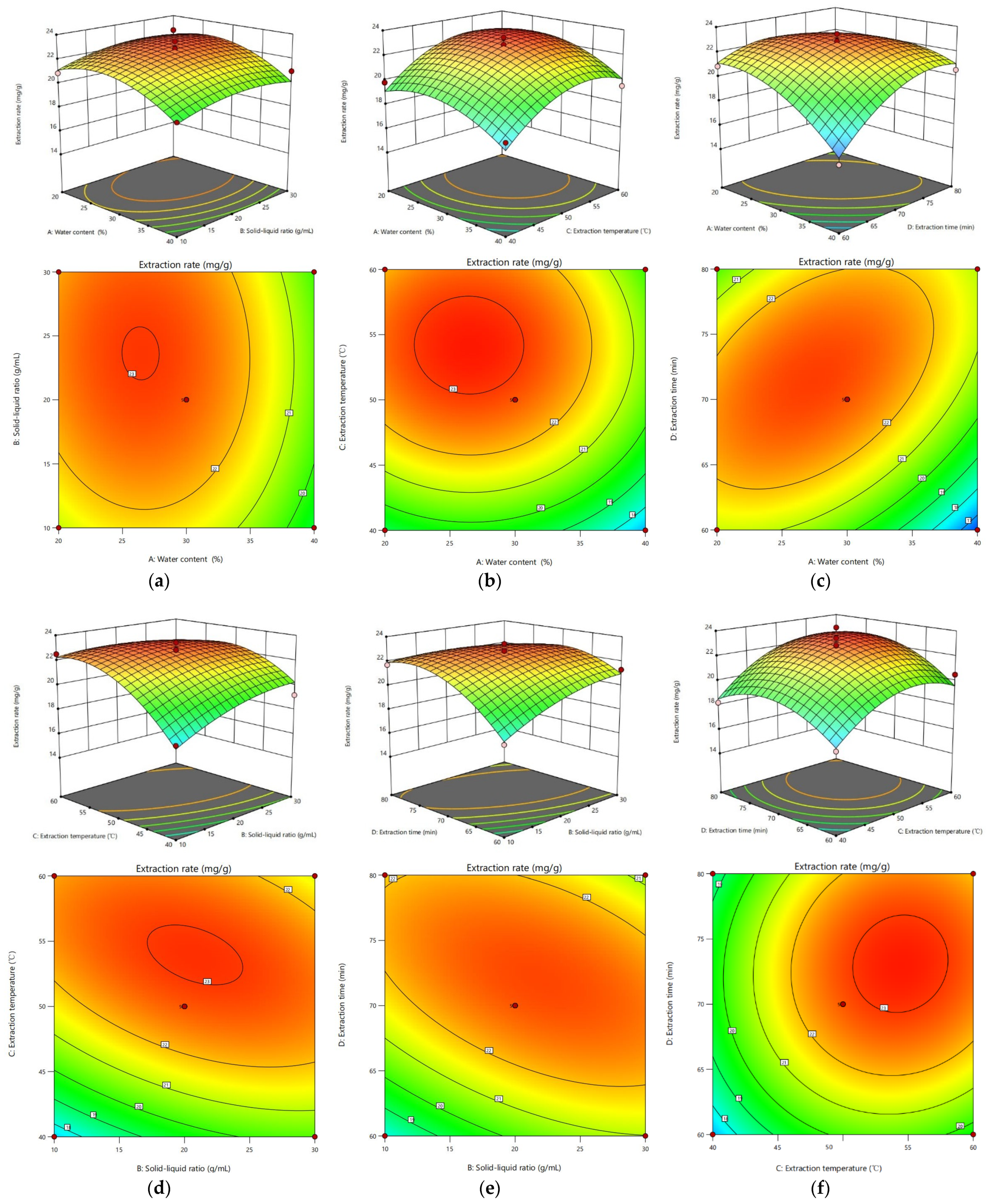

| Serial Number | HBA | HBD | DESs | ||
|---|---|---|---|---|---|
| Molar Ratio | pH | Viscosity/mPa∙s | |||
| DES-1 | ChCl | Eg | 1:2 | 4.40 | 26.00 119.00 47.00 12.00 21.00 45.39 9.65 64.01 |
| DES-2 | ChCl | La | 1:2 | 1.40 | |
| DES-3 | ChCl | Buta | 1:3 | 3.74 | |
| DES-4 | La | Gly | 1:1 | 1.85 | |
| DES-5 | LA | Gly | 1:1 | 1.86 | |
| DES-6 | Ma | Gly | 1:1 | 1.03 | |
| DES-7 | La | Buta | 2:1 | 2.25 | |
| DES-8 | Ca | Gly | 1:2 | 1.06 | |
| Run | A Water Content (%) | B Solid-Liquid Ratio (g/mL) | C Extraction Temperature (°C) | D Extraction Time (min) | Extraction Rate (mg/g) |
|---|---|---|---|---|---|
| 1 | 0 | 0 | −1 | −1 | 16.953 |
| 2 | −1 | 0 | −1 | 0 | 19.815 |
| 3 | 1 | 1 | 0 | 0 | 20.963 |
| 4 | 0 | −1 | 1 | 0 | 22.500 |
| 5 | −1 | 0 | 1 | 0 | 21.647 |
| 6 | −1 | −1 | 0 | 0 | 20.85 |
| 7 | 0 | 0 | 1 | −1 | 20.502 |
| 8 | 0 | 0 | 0 | 0 | 22.089 |
| 9 | 0 | 0 | 0 | 0 | 22.667 |
| 10 | 1 | 0 | −1 | 0 | 17.754 |
| 11 | 1 | 0 | 0 | 1 | 20.502 |
| 12 | 0 | 1 | −1 | 0 | 19.202 |
| 13 | 0 | −1 | 0 | −1 | 17.700 |
| 14 | −1 | 0 | 0 | 1 | 19.929 |
| 15 | 0 | −1 | 0 | 1 | 21.700 |
| 16 | 1 | 0 | 0 | −1 | 15.863 |
| 17 | 0 | 1 | 0 | −1 | 21.317 |
| 18 | 1 | 0 | 1 | 0 | 19.472 |
| 19 | 0 | 0 | −1 | 1 | 18.212 |
| 20 | 0 | −1 | −1 | 0 | 17.694 |
| 21 | 0 | 0 | 0 | 0 | 22.844 |
| 22 | 0 | 0 | 0 | 0 | 22.817 |
| 23 | 0 | 0 | 0 | 0 | 23.418 |
| 24 | 0 | 1 | 1 | 0 | 20.301 |
| 25 | −1 | 1 | 0 | 0 | 22.68 |
| 26 | −1 | 0 | 0 | −1 | 20.846 |
| 27 | 0 | 0 | 1 | 1 | 22.843 |
| 28 | 0 | 1 | 0 | 1 | 21.135 |
| 29 | 1 | −1 | 0 | 0 | 19.412 |
| Source | Sum of Squares | df | Mean Square | F-Value | p-Value | Significance |
|---|---|---|---|---|---|---|
| Model | 104.04 | 14 | 7.43 | 12.4 | <0.0001 | significant |
| A | 11.61 | 1 | 11.61 | 19.36 | 0.0006 | ** |
| B | 2.75 | 1 | 2.75 | 4.58 | 0.0504 | |
| C | 25.92 | 1 | 25.92 | 43.24 | <0.0001 | *** |
| D | 10.34 | 1 | 10.34 | 17.25 | 0.001 | ** |
| AB | 0.0195 | 1 | 0.0195 | 0.0325 | 0.8596 | |
| AC | 0.0032 | 1 | 0.0032 | 0.0054 | 0.9424 | |
| AD | 7.72 | 1 | 7.72 | 12.88 | 0.003 | ** |
| BC | 3.44 | 1 | 3.44 | 5.73 | 0.0312 | * |
| BD | 4.37 | 1 | 4.37 | 7.29 | 0.0172 | * |
| CD | 0.2927 | 1 | 0.2927 | 0.4883 | 0.4961 | |
| A2 | 12.86 | 1 | 12.86 | 21.46 | 0.0004 | ** |
| B2 | 3.12 | 1 | 3.12 | 5.2 | 0.0388 | * |
| C2 | 20.16 | 1 | 20.16 | 33.63 | <0.0001 | *** |
| D2 | 18.46 | 1 | 18.46 | 30.8 | <0.0001 | *** |
| Residual | 8.39 | 14 | 0.5994 | |||
| Lack of Fit | 7.49 | 10 | 0.749 | 3.32 | 0.1293 | not significant |
| Pure Error | 0.9019 | 4 | 0.2255 | |||
| Cor Total | 112.43 | 28 | ||||
| R-Squared | 0.9254 | |||||
| Adj R-Squared | 0.8507 | |||||
| Pred R-Squared | 0.6038 | |||||
| Adeq Precision | 11.4871 |
| Resin Types | Polarity | Specific Surface Area (m2·g −1) | Average Pore Diameter/A° | Total Adsorption Rate/% | Total Desorption Rate/% | Recovery Rate/% |
|---|---|---|---|---|---|---|
| D101 | Non-polar | 500–550 | 90–100 | 66.46 ± 0.389 bc | 83.77 ± 0.237 c | 55.76 ± 0.222 cd |
| AB-8 | Weakly polar | 480–520 | 130–140 | 77.10 ± 0.254 a | 89.38 ± 0.143 a | 77.10 ± 0.254 a |
| S-8 | Polar | 100–120 | 280–300 | 67.90 ± 0.111 b | 82.25 ± 0.150 d | 55.85 ± 0.181 c |
| HPD-600 | Polar | 550–600 | 80 | 66.13 ± 0.085 cd | 85.47 ± 0.168 b | 56.54 ± 0.066 b |
| Levels | −1 | 0 | 1 |
|---|---|---|---|
| Water content (%) | 20 | 30 | 40 |
| Solid-liquid ratio (g·mL−1) | 10 | 20 | 30 |
| Extraction temperature (°C) | 40 | 50 | 60 |
| Extraction time (min) | 60 | 70 | 80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Su, J.; Chu, X.; Wang, X. A Green Method of Extracting and Recovering Flavonoids from Acanthopanax senticosus Using Deep Eutectic Solvents. Molecules 2022, 27, 923. https://doi.org/10.3390/molecules27030923
Zhang X, Su J, Chu X, Wang X. A Green Method of Extracting and Recovering Flavonoids from Acanthopanax senticosus Using Deep Eutectic Solvents. Molecules. 2022; 27(3):923. https://doi.org/10.3390/molecules27030923
Chicago/Turabian StyleZhang, Xinyu, Jianqing Su, Xiuling Chu, and Xiaoya Wang. 2022. "A Green Method of Extracting and Recovering Flavonoids from Acanthopanax senticosus Using Deep Eutectic Solvents" Molecules 27, no. 3: 923. https://doi.org/10.3390/molecules27030923
APA StyleZhang, X., Su, J., Chu, X., & Wang, X. (2022). A Green Method of Extracting and Recovering Flavonoids from Acanthopanax senticosus Using Deep Eutectic Solvents. Molecules, 27(3), 923. https://doi.org/10.3390/molecules27030923






