Natural Dyeing of Modified Cotton Fabric with Cochineal Dye
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Arora, J.; Agarwal, P.; Gupta, G. Rainbow of Natural Dyes on Textiles Using Plants Extracts: Sustainable and Eco-Friendly Processes. Green Sustain. Chem. 2017, 7, 35–47. [Google Scholar] [CrossRef] [Green Version]
- Zarkogianni, M.; Mikropoulou, E.; Varella, E.; Tsatsaroni, E. Colour and fastness of natural dyes: Revival of traditional dyeing techniques. Color. Technol. 2010, 127, 18–27. [Google Scholar] [CrossRef]
- Deveoglu, O. A Review on Cochineal (Dactylopius Coccus Costa) Dye. Res. J. Recent Sci. 2020, 9, 37–43. [Google Scholar]
- Rehman, F.; Sanbhal, N.; Naveed, T.; Farooq, A.; Wang, Y.; Wei, W. Antibacterial performance of Tencel fabric dyed with pomegranate peel extracted via ultrasonic method. Cellulose 2018, 25, 4251–4260. [Google Scholar] [CrossRef]
- Maxia, A.; Meli, F.; Gaviano, C.; Picciau, R.; De Martis, B.; Kasture, S.; Kasture, V. Dye plants: Natural resources from traditional botanical knowledge of Sardinia Island, Italy. Indian J. Tradit. Knowl. 2013, 12, 651–656. [Google Scholar]
- Saakshy; Sharma, A.K.; Jain, R.K. Chapter in Biotechnology for Environmental Management and Resource Recovery. In Application of Natural Dyes: An Emerging Environment-Friendly Solution to Handmade Paper Industry; Kuhad, R.C., Singh, A., Eds.; Springer: New Delhi, India; Berlin/Heidelberg, Germany; New York, NY, USA; Dordrecht, The Netherlands; London, UK, 2013; pp. 279–288. [Google Scholar]
- Parac-Osterman, Đ.; Karaman, B. Osnove Teorije Bojenja Tekstila (Eng. Basics of Textile Dyeing Theory); University of Zagreb Faculty of Textile Technology: Zagreb, Croatia, 2013. [Google Scholar]
- Sutlović, A. Study of Natural Dyestuff—Contribution to Human Ecology. Ph.D. Thesis, University of Zagreb Faculty of Textile Technology, Zagreb, Croatia, 2008. [Google Scholar]
- Sutlović, A.; Parac-Osterman, Đ.; Đurašević, V. Croatian Traditional Herbal Dyes for Textile Dyeing. TEDI 2011, 1, 65–69. [Google Scholar]
- Ansari, A.A.; Thakur, B.D. Extraction, characterization and application of a natural dye: The eco-friendly textile colorant. Colourage 2000, 47, 15–20. [Google Scholar]
- Buttler Greenfield, A. A Perfect Red, Empire, Espionage and the Quest for the Colour of Desire; Harper Perennial: New York, NY, USA, 2005; pp. 103–117. [Google Scholar]
- Brenko, A.; Randić, M. Exhibition “The Power of Colour” (Moć boje); Etnografski Muzej: Zagreb, Croatia, 2009; pp. 7–153. [Google Scholar]
- Borges, M.E.; Tejera, R.L.; Diaz, L.; Esparza, P.; Ibanez, E. Natural dyes extraction from cochineal (Dactylopius coccus). New extraction methods. Food Chem. 2012, 132, 1855–1860. [Google Scholar] [CrossRef]
- Campana, M.G.; Robles Garcia, N.M.; Tuross, N. America’s red gold: Multiple lineages of cultivated cochineal in Mexico. Ecol. Evol. 2015, 5, 607. [Google Scholar] [CrossRef]
- Canamares, M.V.; Garcia-Ramos, J.V.; Domingo, C.; Sanchez-Cortes, S. Surface-Enhanced Raman Scattering Study of the Anthraquinone Red Pigment Carminic Acid. Vib. Spectrosc. 2006, 40, 161–167. [Google Scholar] [CrossRef]
- Dapson, R.W. The History, Chemistry and Modes of Action of Carmine and Related Dyes. Biotech. Histochem. 2007, 82, 173–187. [Google Scholar] [CrossRef] [PubMed]
- Allevi, P.; Anastasia, M.; Bingham, S.; Ciuffreda, P.; Fiecchi, A.; Cighetti, G.; Muir, M.; Tyman, J. Synthesis of Carminic Acid, the Colourant Principle of Cochineal. J. Chem. Soc. Perkin Trans. 1 1998, 575–582. [Google Scholar] [CrossRef]
- Naturex Focuses on Clean & Clear Labels at IFT18 with Launch of Plant-Based Alternative to Edta. Available online: https://www.naturex.com/Media2/Press-releases/Naturex-focuses-on-clean-clear-labels-at-IFT18-with-launch-of-plant-based-alternative-to-EDTA (accessed on 27 December 2021).
- Sutlović, A.; Brlek, I.; Ljubić, V.; Glogar, M.I. Optimization of Dyeing Process of Cotton Fabric with Cochineal Dye. Fibers Polym. 2020, 21, 555–563. [Google Scholar] [CrossRef]
- Prikhodko, S.V.; Rambaldi, D.C.; King, A.; Burr, E.; Muros, V.; Kakoulli, I. New advancements in SERS dye detection using infrared SEM and Raman spectromicroscopy (µRS). J. Raman Spectrosc. 2015, 46, 632–635. [Google Scholar] [CrossRef]
- Stathopoulou, K.; Valianou, L.; Skaltsounis, A.-L.; Karapanagiotis, I.; Magiatis, P. Structure elucidation and chromatographic identification of anthraquinone components of cochineal (Dactylopius coccus) detected in historical objects. Anal. Chim. Acta 2013, 804, 264–272. [Google Scholar] [CrossRef]
- Hebeish, A.; Elnagar, K.; Shaaban, M.F. Innovative Approach for Effecting Improved Mordant Dyeing of Cotton Textiles. Egypt. J. Chem. 2015, 58, 415–430. [Google Scholar]
- Arroyo-Figueroa, G.; Ruiz-Aguilar, G.M.L.; Cuevas-Rodriguez, G.; Gonzalez-Sanchez, G. Cotton fabric dyeing with cochineal extract: Influence of mordant concentration. Color. Technol. 2011, 127, 39–46. [Google Scholar] [CrossRef]
- Valipour, P.; Ekrami, E.; Shams-Nateri, A. Colorimetric Properties of Wool Dyed with Cochineal: Effect of Dye-Bath pH. Prog. Color Colorants Coat. 2014, 7, 129–138. [Google Scholar]
- Lokhande, H.T.; Dorugade, V.A.; Sandeep, R.N. Applicaton of Natural Dyes on Polyester. Am. Dyest. Report. 1998, 87, 40–50. [Google Scholar]
- Samanta, A.K.; Singhee, D.; Sethia, M. Application of single and mixture of selected natural dyes on cotton fabric: A scientific approach. Colourage 2003, 50, 29–42. [Google Scholar]
- Angelini, L.G.; Bertoli, A.; Rolandelli, S.; Pistelli, L. Agronomic potential of Reseda luteola L. as new crop for natural dyes in textiles production. Ind. Crops Prod. 2003, 17, 199–207. [Google Scholar] [CrossRef]
- Bechtold, T.; Mahmud-Ali, A.; Mussak, R.A.M. Natural dyes for textile dyeing: A comparison of methods to assess the quality of Canadian golden rod plant material. Dye. Pigment. 2007, 75, 287–293. [Google Scholar] [CrossRef]
- Bechtold, T.; Mahmud-Ali, A.; Mussak, R.A.M. Reuse of ash-tree (Fraxinus excelsior L.) bark as natural dyes for textile dyeing: Process conditions and process stability. Color. Technol. 2007, 123, 271–279. [Google Scholar] [CrossRef]
- Micheal, M.N.; Tera, F.M.; Aboelanwar, S.A. Colour measurements and colourant estimation of natural red dyes on natural fabrics using differnt mordants. Colourage 2003, 50, 31–42. [Google Scholar]
- Brückner, U.; Struckmeier, S.; Dittrich, J.H.; Reumann, R.D. Zur Echtheit von Färbungen mit ausgewählten Naturfarbstoffen auf Synthesefasergeweben. Texilveredlung 1997, 32, 112–116. [Google Scholar]
- Sutlović, A.; Glogar, M.I.; Tarbuk, A. Cochineal Colored Cotton as UV Shield: UV Protective Properties of Cotton Material Dyed with Cochineal Dyestuff. In Scientific Notes of the Color Society of Russia; Griber, Y.A., Schindler, V.M., Eds.; Smolensk State University Press: Smolensk, Russia, 2020; pp. 71–78. [Google Scholar]
- Haddar, W.; Ticha, M.B.; Guesmi, A.; Khoffi, F.; Durand, B. A novel approach for a natural dyeing process of cotton fabric with Hibiscus mutabilis (Gulzuba): Process development and optimization using statistical analysis. J. Clean. Prod. 2014, 68, 114–120. [Google Scholar] [CrossRef]
- Haddar, W.; Elksibi, I.; Meksi, N.; Mhenni, M.F. Valorization of the leaves of fennel (Foeniculum vulgare) as natural dyes fixed on modified cotton: A dyeing process optimization based on a response surface methodology. Ind. Crops Prod. 2014, 52, 588–596. [Google Scholar] [CrossRef]
- Ticha, M.B.; Haddar, W.; Meksi, N.; Guesmi, A.; Mhenni, M.F. Improving dyeability of modified cotton fabrics by the natural aqueous extract from red cabbage using ultrasonic energy. Carbohydr. Polym. 2016, 154, 287–295. [Google Scholar] [CrossRef]
- Peran, J.; Ercegović Ražić, S.; Sutlović, A.; Ivanković, T.; Glogar, M.I. Oxygen Plasma Pre-Treatment Improves Dyeing and Antimicrobial Properties of Wool Fabric Dyed with Natural Extract from Pomegranate Peel. Color. Technol. 2020, 136, 177–187. [Google Scholar]
- Haji, A. Plasma activation and chitosan attachment on cotton and wool for improvement of dyeability and fastness properties. Pigment Resin Technol. 2020, 49, 483–489. [Google Scholar] [CrossRef]
- Haji, A. Improved natural dyeing of cotton by plasma treatment and chitosan coating; optimization by response surface methodology. Cellul. Chem. Technol. 2017, 51, 975–982. [Google Scholar]
- Gulzar, T.; Adeel, S.; Hanif, I.; Rehman, F.; Hanif, R.; Zuber, M.; Akhtar, N. Eco-friendly dyeing of gamma ray induced cotton using natural quercetin extracted from Acacia bark (A. Nilotica). J. Nat. Fibers 2015, 12, 494–504. [Google Scholar] [CrossRef]
- Tarbuk, A.; Sutlović, A.; Grancarić, A.M.; Kopanska, A.; Trela, N.; Draczyński, Z. The Modified Cotton Dyed with Juglans Regia L. without Mordant. In Book of Proceedings of the 8th International Textile, Clothing & Design Conference—Magic World of Textiles; Dragčević, Z., Hursa Šajatović, A., Vujasinović, E., Eds.; University of Zagreb Faculty of Textile Technology: Zagreb, Croatia, 2016; pp. 212–217. [Google Scholar]
- Tarbuk, A.; Grancarić, A.M.; Leskovac, M. Novel cotton cellulose by cationisation during the mercerisation process—Part 1: Chemical and morphological changes. Cellulose 2014, 21, 2167–2179. [Google Scholar] [CrossRef]
- Tarbuk, A.; Grancarić, A.M.; Leskovac, M. Novel cotton cellulose by cationisation during the mercerization—Part 2: Interface phenomena. Cellulose 2014, 21, 2089–2099. [Google Scholar] [CrossRef]
- Rupin, M.; Veatue, J.; Balland, B. Utilization of reactive epoxy-ammonium quaternaries on cellulose treatment for dyeing with direct and reactive dyes. Textilveredlung 1970, 5, 829–838. [Google Scholar]
- Lewis, D.M.; McIlroy, K.A. The Chemical Modification of Cellulosic fibers to Enhance Dyeability. Rev. Prog. Color 1997, 27, 5–17. [Google Scholar]
- Hauser, P.J.; Tabba, A.H. Improving the Environmental and Economic Aspects of Dyeing Cotton. Color. Technol. 2001, 117, 282–288. [Google Scholar] [CrossRef]
- Draper, S.L.; Beck, K.R.; Smith, C.B.; Hauser, P. Characterization of the Dyeing Behavior of Cationic Cotton with Acid Dyes. AATCC Rev. 2003, 3, 51–55. [Google Scholar]
- Sutlović, A.; Glogar, M.I.; Čorak, I.; Tarbuk, A. Trichromatic Vat Dyeing of Cationized Cotton. Materials 2021, 14, 5731. [Google Scholar] [CrossRef]
- Aktek, T.; Millat, A.K.M.M. Salt free dyeing of cotton fiber—A critical review. Int. J. Text. Sci. 2017, 6, 21–33. [Google Scholar]
- Correia, J.; Rainert, K.T.; Oliveira, F.R.; Valle, R.C.S.C.; Valle, J.A.B. Cationization of cotton fiber: An integrated view of cationic agents, processes variables, properties, market and future prospects. Cellulose 2020, 27, 8527–8550. [Google Scholar] [CrossRef]
- Choudhury, A.K.R. Coloration of Cationized Cellulosic Fibers—A Review. AATCC J. Res. 2014, 1, 11–19. [Google Scholar] [CrossRef]
- Tarbuk, A. Interface Phenomena of Cationized Cotton. Ph.D. thesis, University of Zagreb Faculty of Textile Technology, Zagreb, Croatia, 2009. [Google Scholar]
- Tarbuk, A.; Grancarić, A.M. Chapter 6 in Cellulose and Cellulose Derivatives: Synthesis, Modification and Applications, Part I: Cellulose Synthesis and Modification. In Interface Phenomena of Cotton Cationized in Mercerization; Mondal, I.H., Ed.; Nova Science Publishers: New York, NY, USA, 2015; pp. 103–126. [Google Scholar]
- Grancarić, A.M.; Tarbuk, A.; Dekanić, T. Elektropozitivan pamuk (eng. Electropositive Cotton). Tekstil 2004, 53, 47–51. [Google Scholar]
- Soljačić, I.; Žerdik, M. Cotton mercerization. Tekstil 1968, 17, 495–518. [Google Scholar]
- Dinand, E.; Vignon, M.; Chanzy, H.; Heux, L. Mercerization of Primary Wall Cellulose and its Implication for the Conversion of Cellulose I to Cellulose II. Cellulose 2002, 9, 7–18. [Google Scholar] [CrossRef]
- Stana-Kleinschek, K.; Strand, S.; Ribitsch, V. Surface Characterization and Adsorption Abilities of Cellulose Fibers. Polym. Eng. Sci. 1999, 39, 1412–1424. [Google Scholar] [CrossRef]
- Grancarić, A.M.; Tarbuk, A.; Pušić, T. Electrokinetic Properties of Textile Fabrics. Color. Technol. 2005, 121, 221–227. [Google Scholar] [CrossRef]
- Parac-Osterman, Đ. Osnove o Boji i Sustavi Vrjednovanja, (Eng. Color Basics and Evaluation Systems); University of Zagreb Faculty of Textile Technology: Zagreb, Croatia, 2007. [Google Scholar]
- Lewis, D.M. Wool Dyeing; Society of Dyers and Colourists: Bradford, UK, 1992. [Google Scholar]
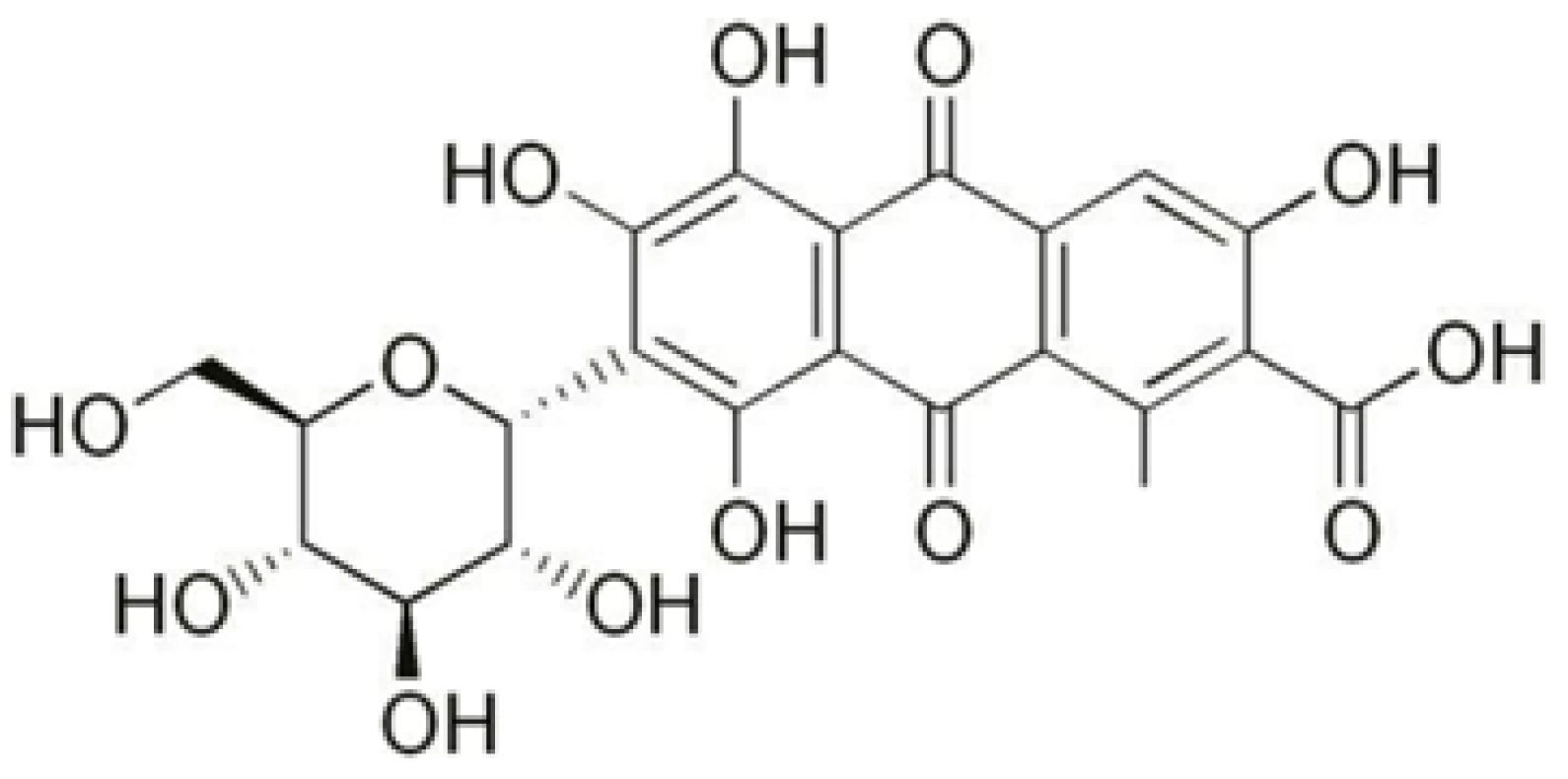

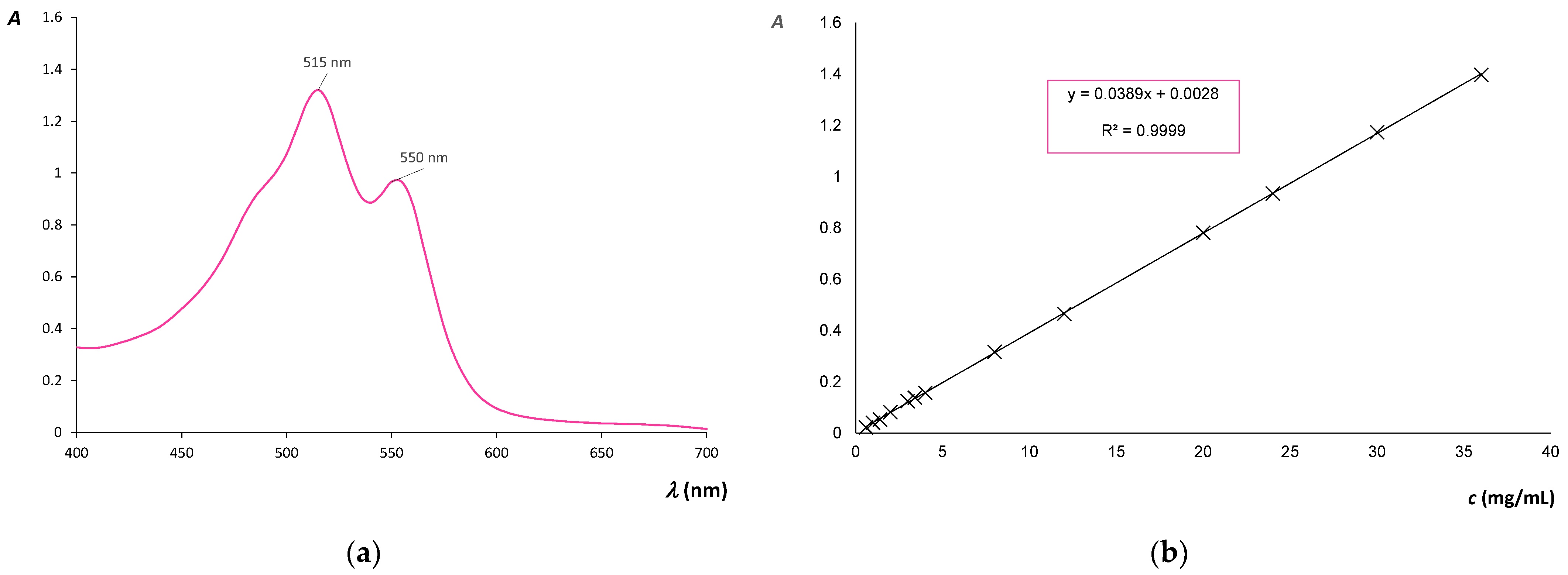

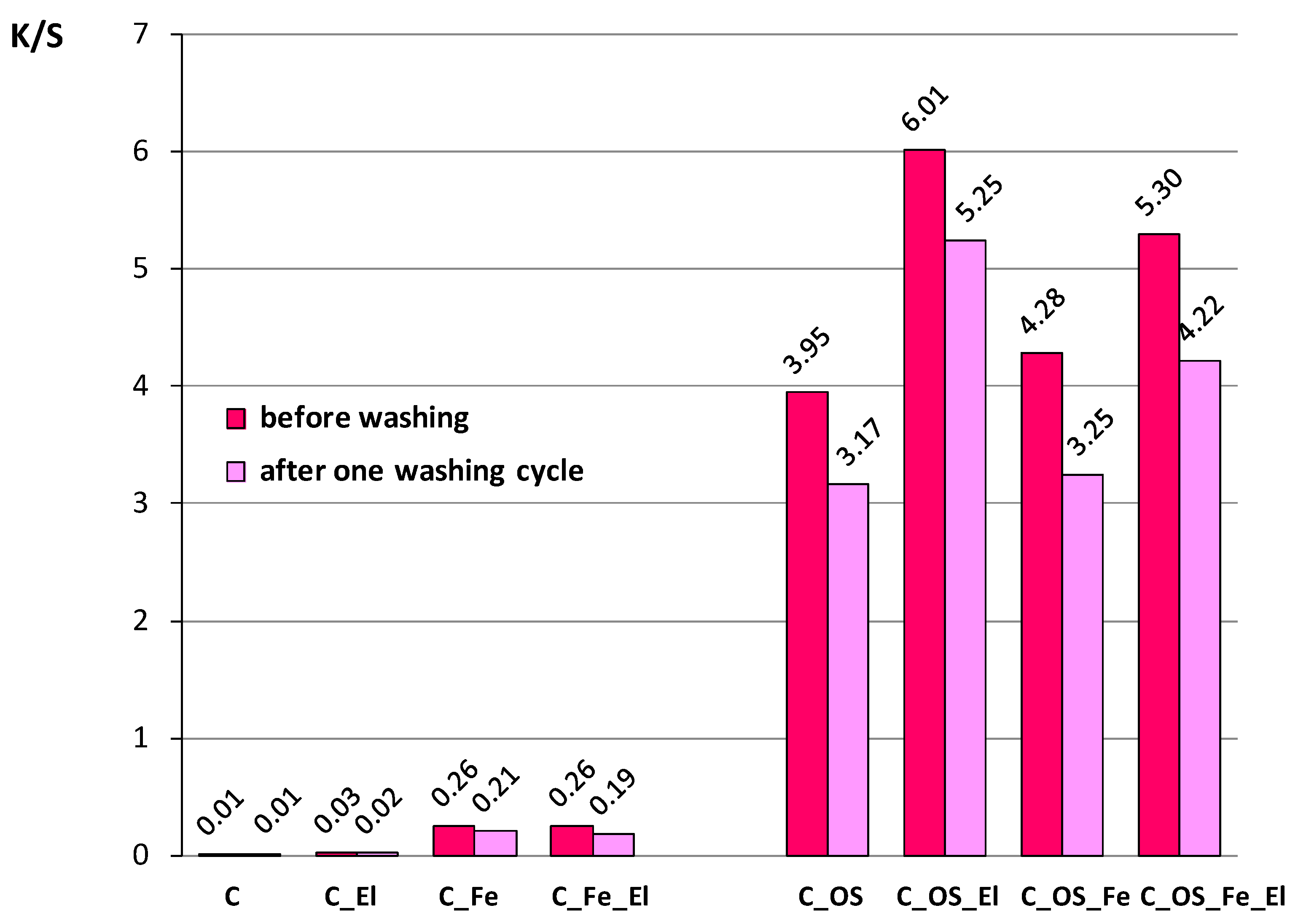

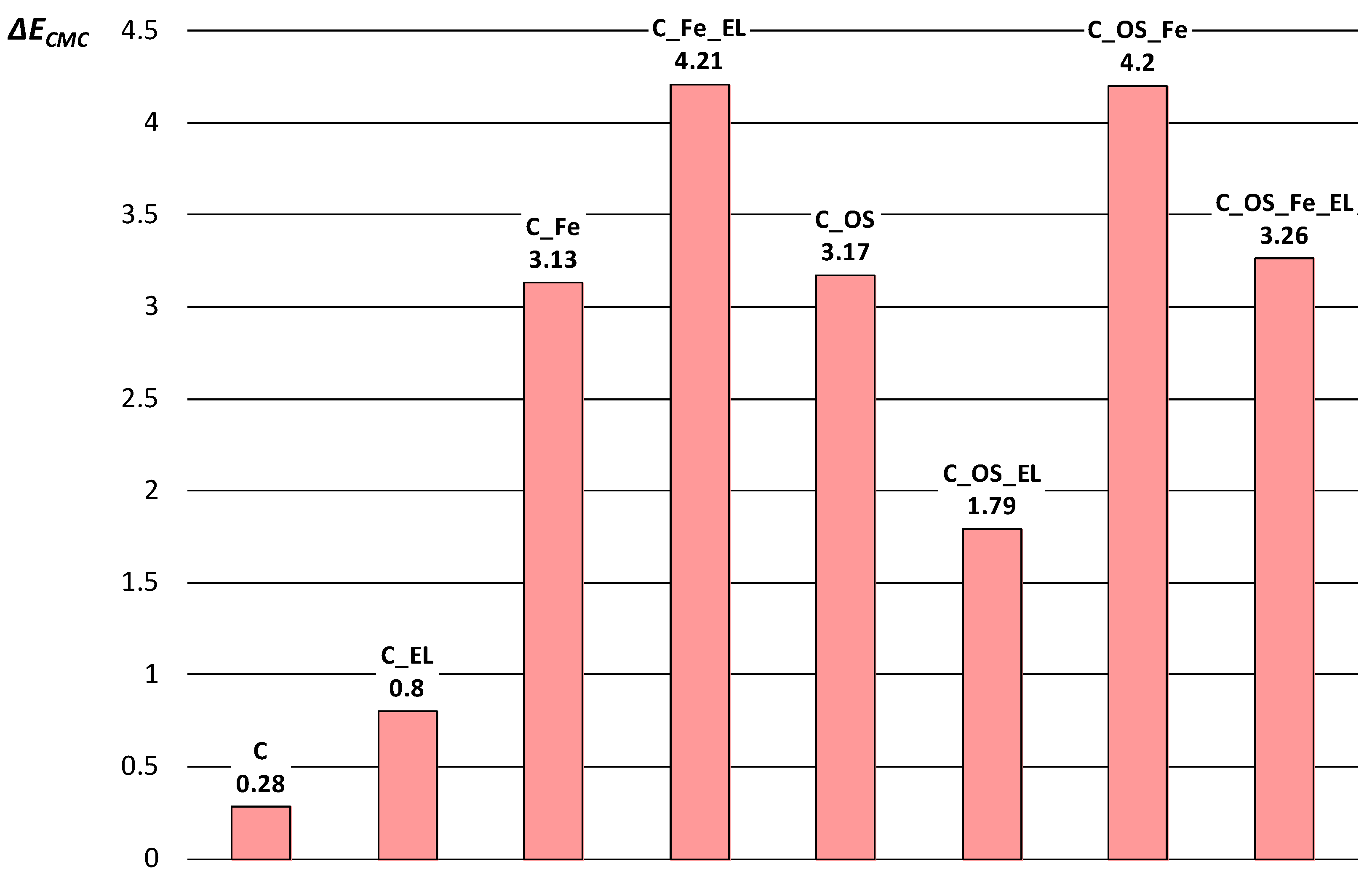
| Label | Description of Cotton Fabric Treatment |
|---|---|
| B_ | Bleached cotton fabric |
| _OS_ | Cationized cotton with Rewin OS |
| _Fe_ | Pre-mordanting using iron(II) sulfate heptahydrate (FeSO4·7H2O) |
| C_ | Cotton dyed with cochineal |
| …_EL | The electrolyte added in the dyeing bath |
| …_W | One washing cycle after dyeing |
| Sample | ZP at pH 3.5/mV | ZP at pH 6.5/mV | ZP at pH 8.5/mV | IEP | WCIE |
|---|---|---|---|---|---|
| B | −15.0 | −29.1 | −31.3 | 2.4 | 84.01 |
| B_Fe | −5.0 | −30.0 | −32.1 | 3.2 | −31.57 |
| B_OS | 2.2 | −14.4 | −15.0 | 3.8 | 77.81 |
| B_OS_Fe | 7.3 | −17.5 | −22.5 | 4.4 | −3.49 |
| Sample | L* | a* | b* | C* | h° |
|---|---|---|---|---|---|
| C | 94.20 | 0.80 | 2.04 | 2.19 | 68.48 |
| C_EL | 92.02 | 2.85 | 0.73 | 2.94 | 14.42 |
| C_Fe | 79.02 | 6.90 | 13.78 | 15.41 | 63.39 |
| C_Fe_EL | 78.80 | 6.77 | 11.18 | 13.07 | 58.81 |
| C_OS | 49.17 | 36.44 | −5.54 | 36.86 | 351.35 |
| C_OS_EL | 43.73 | 38.36 | −5.45 | 38.75 | 351.91 |
| C_OS_Fe | 42.28 | 18.51 | −7.48 | 19.96 | 338.00 |
| C_OS_Fe_EL | 39.77 | 20.68 | −8.20 | 22.24 | 338.38 |
| Sample | L* | a* | b* | C* | h° |
|---|---|---|---|---|---|
| C_W | 94.32 | 0.56 | 1.96 | 2.04 | 73.95 |
| C_EL_W | 92.50 | 2.20 | 0.75 | 2.33 | 18.85 |
| C_Fe_W | 81.29 | 6.44 | 15.88 | 17.14 | 67.93 |
| C_Fe_EL_W | 81.96 | 5.89 | 13.83 | 15.03 | 66.94 |
| C_OS_W | 51.93 | 34.89 | −5.46 | 35.32 | 351.11 |
| C_OS_EL_W | 45.26 | 37.44 | −5.50 | 37.84 | 351.65 |
| C_OS_Fe_W | 46.33 | 18.02 | −6.48 | 19.15 | 340.23 |
| C_OS_Fe_EL_W | 42.97 | 20.47 | −7.56 | 21.82 | 339.73 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Čorak, I.; Brlek, I.; Sutlović, A.; Tarbuk, A. Natural Dyeing of Modified Cotton Fabric with Cochineal Dye. Molecules 2022, 27, 1100. https://doi.org/10.3390/molecules27031100
Čorak I, Brlek I, Sutlović A, Tarbuk A. Natural Dyeing of Modified Cotton Fabric with Cochineal Dye. Molecules. 2022; 27(3):1100. https://doi.org/10.3390/molecules27031100
Chicago/Turabian StyleČorak, Ivana, Iva Brlek, Ana Sutlović, and Anita Tarbuk. 2022. "Natural Dyeing of Modified Cotton Fabric with Cochineal Dye" Molecules 27, no. 3: 1100. https://doi.org/10.3390/molecules27031100
APA StyleČorak, I., Brlek, I., Sutlović, A., & Tarbuk, A. (2022). Natural Dyeing of Modified Cotton Fabric with Cochineal Dye. Molecules, 27(3), 1100. https://doi.org/10.3390/molecules27031100








