An Investigation into the PVA:MC:NH4Cl-Based Proton-Conducting Polymer-Blend Electrolytes for Electrochemical Double Layer Capacitor (EDLC) Device Application: The FTIR, Circuit Design and Electrochemical Studies
Abstract
:1. Introduction
2. Results and Discussion
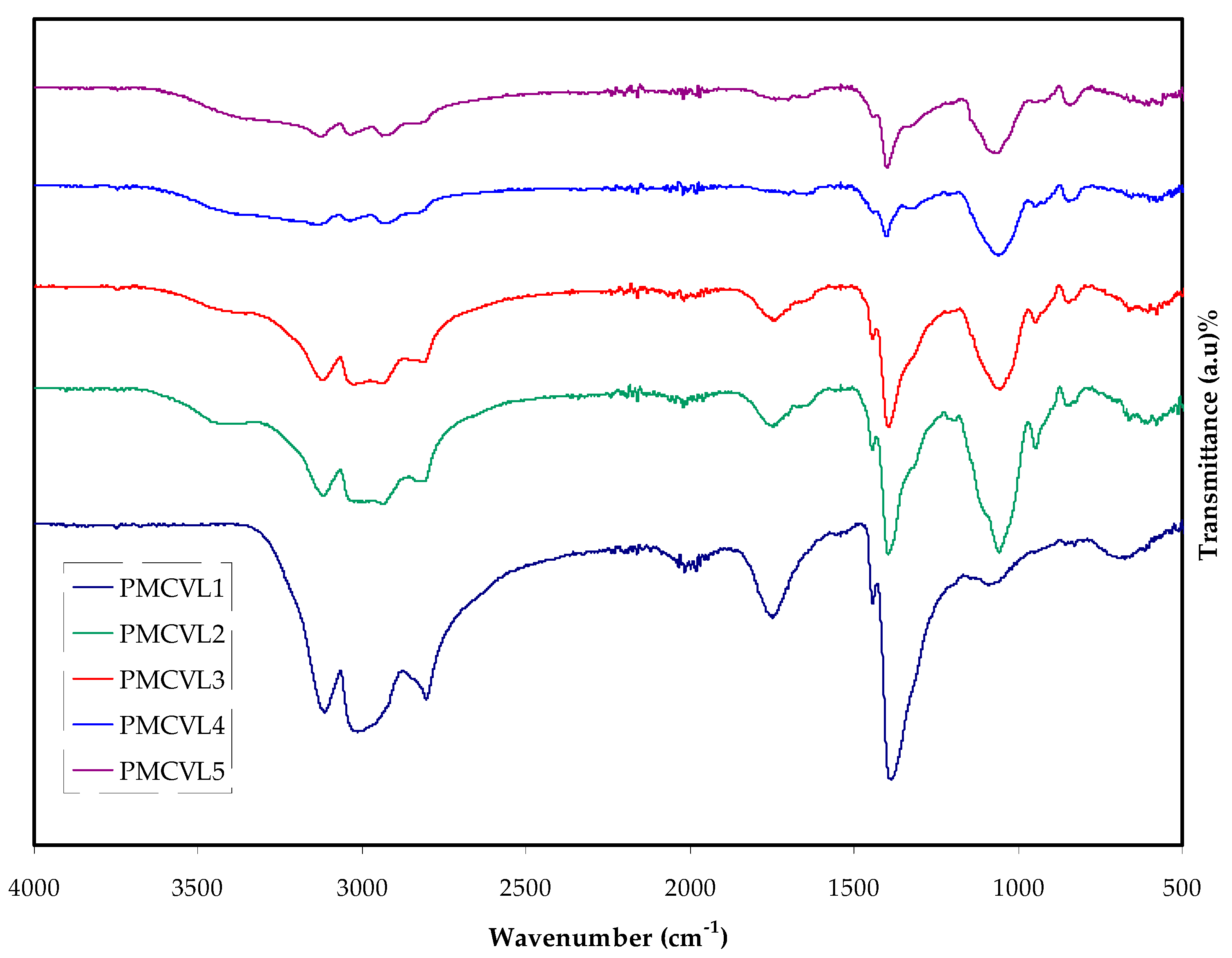
2.1. FTIR Study
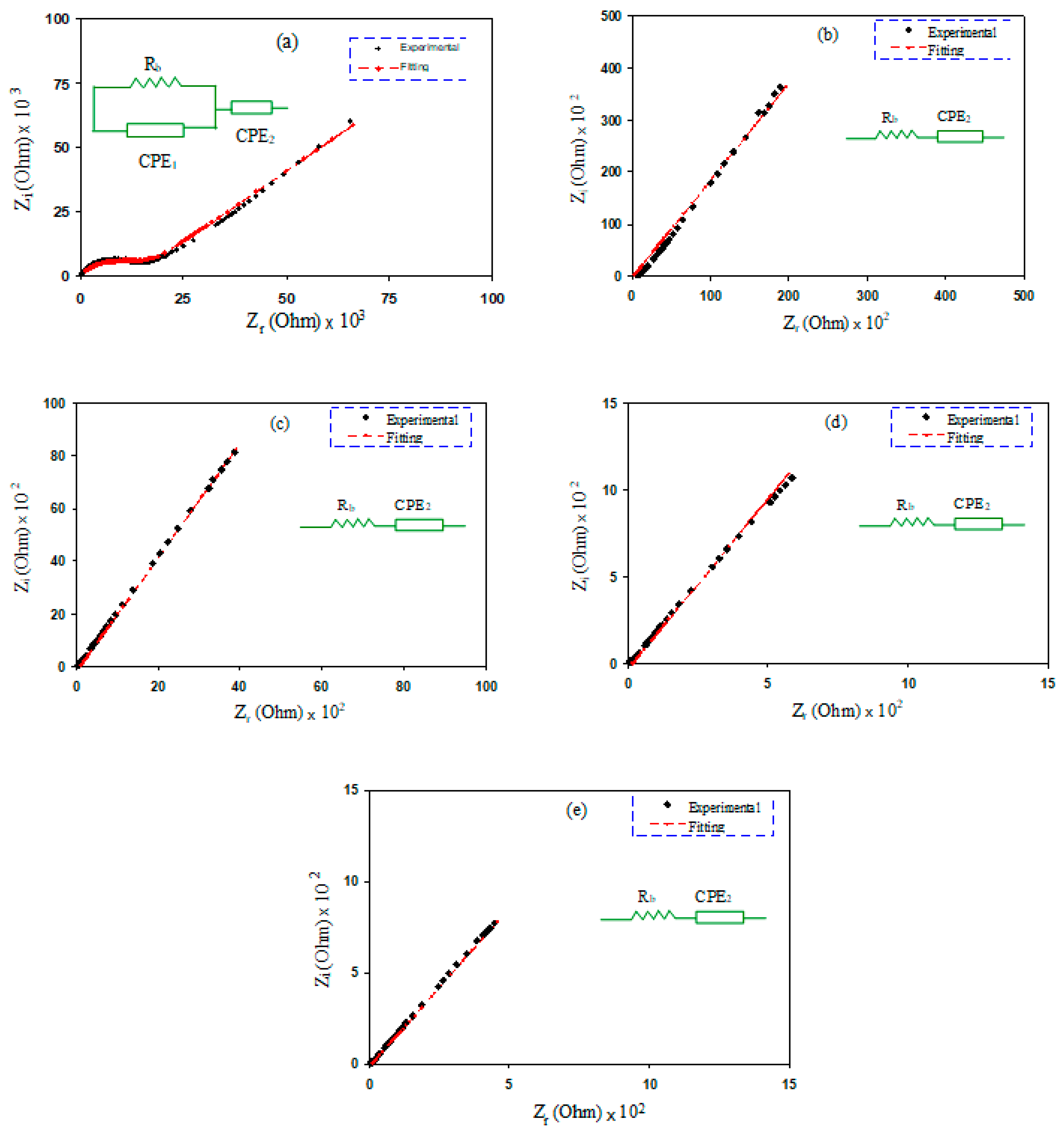
2.2. EIS Study
2.3. Dielectric Properties
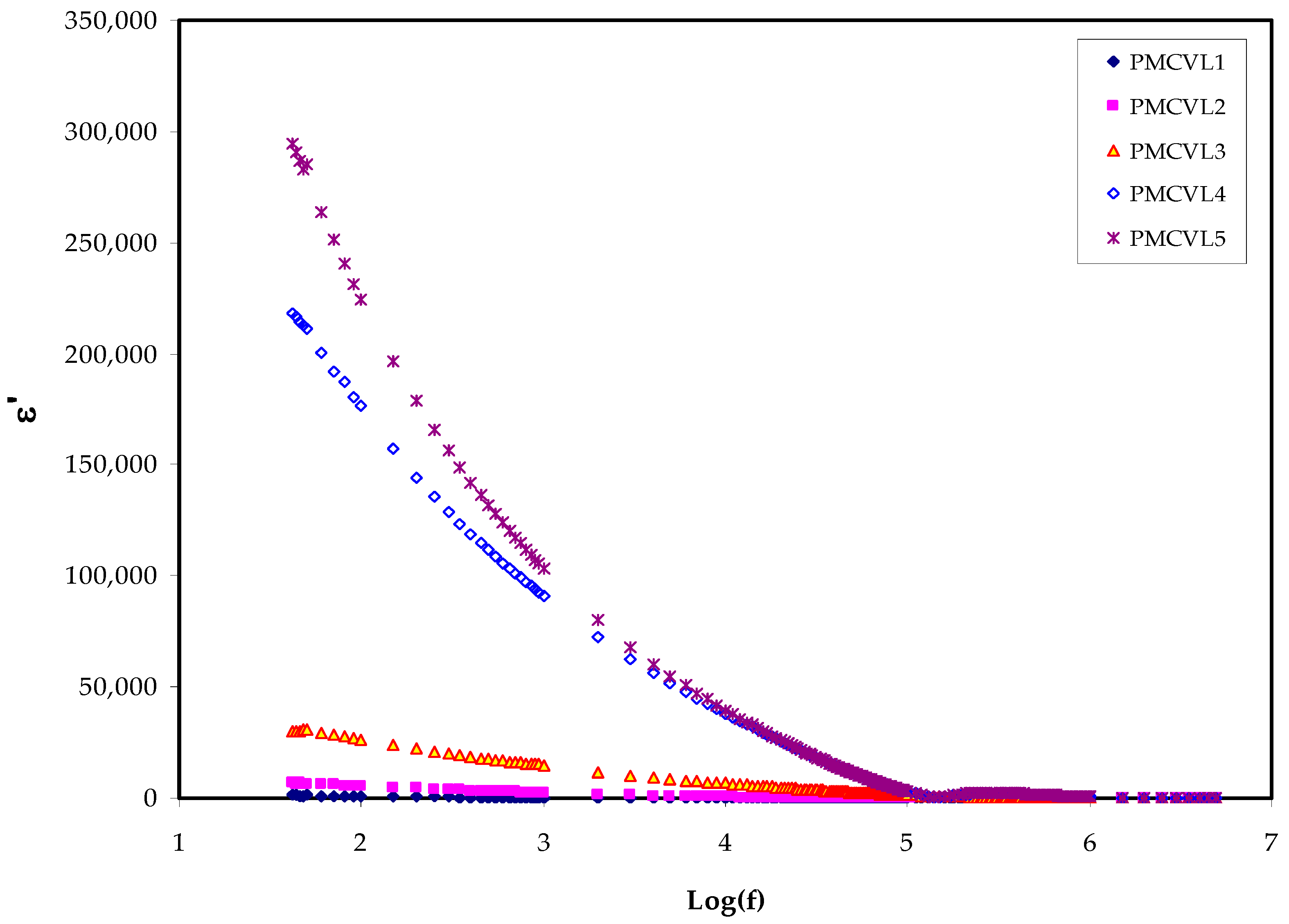
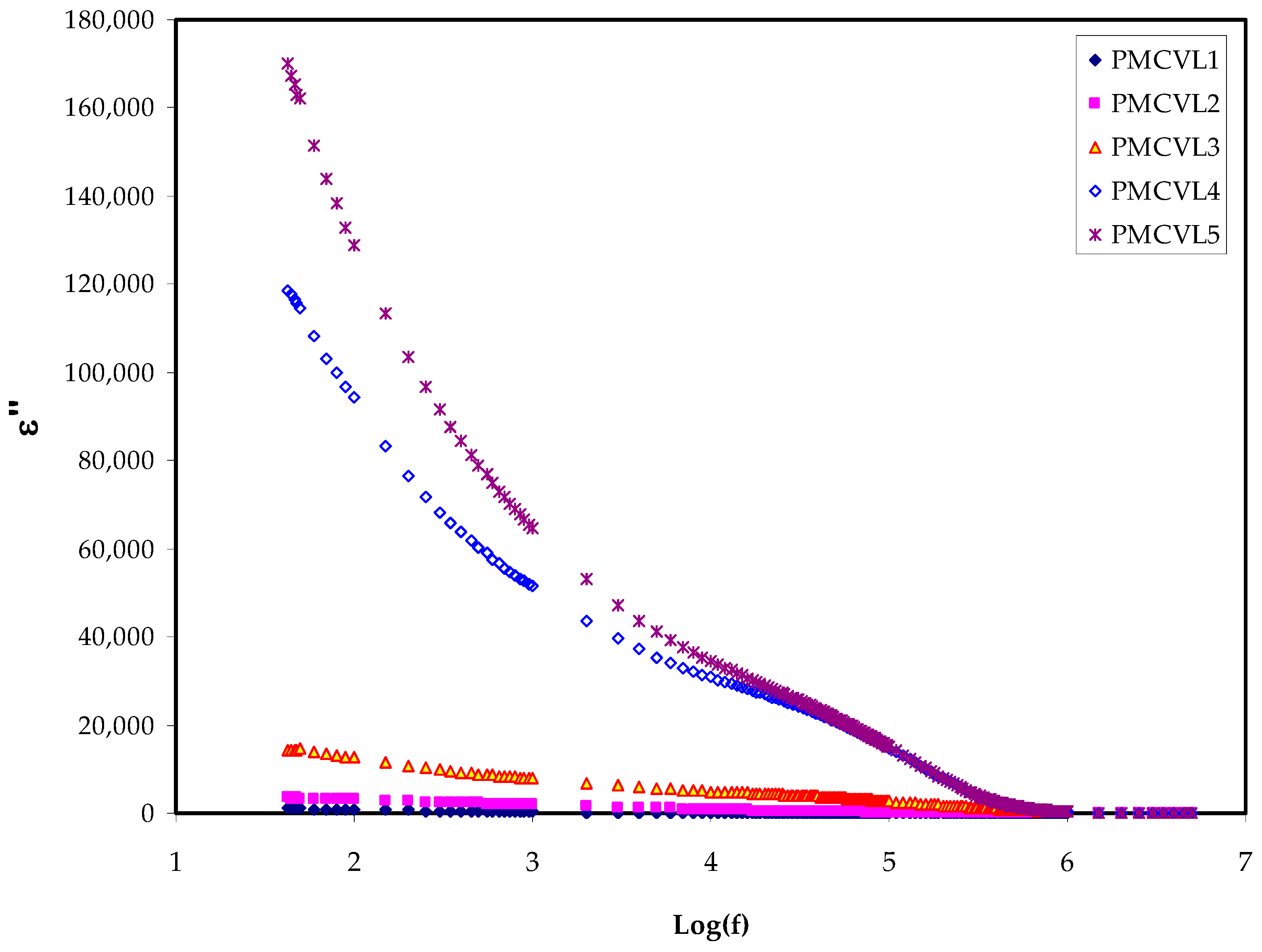
2.3.1. The Dielectric Constant and Loss Study
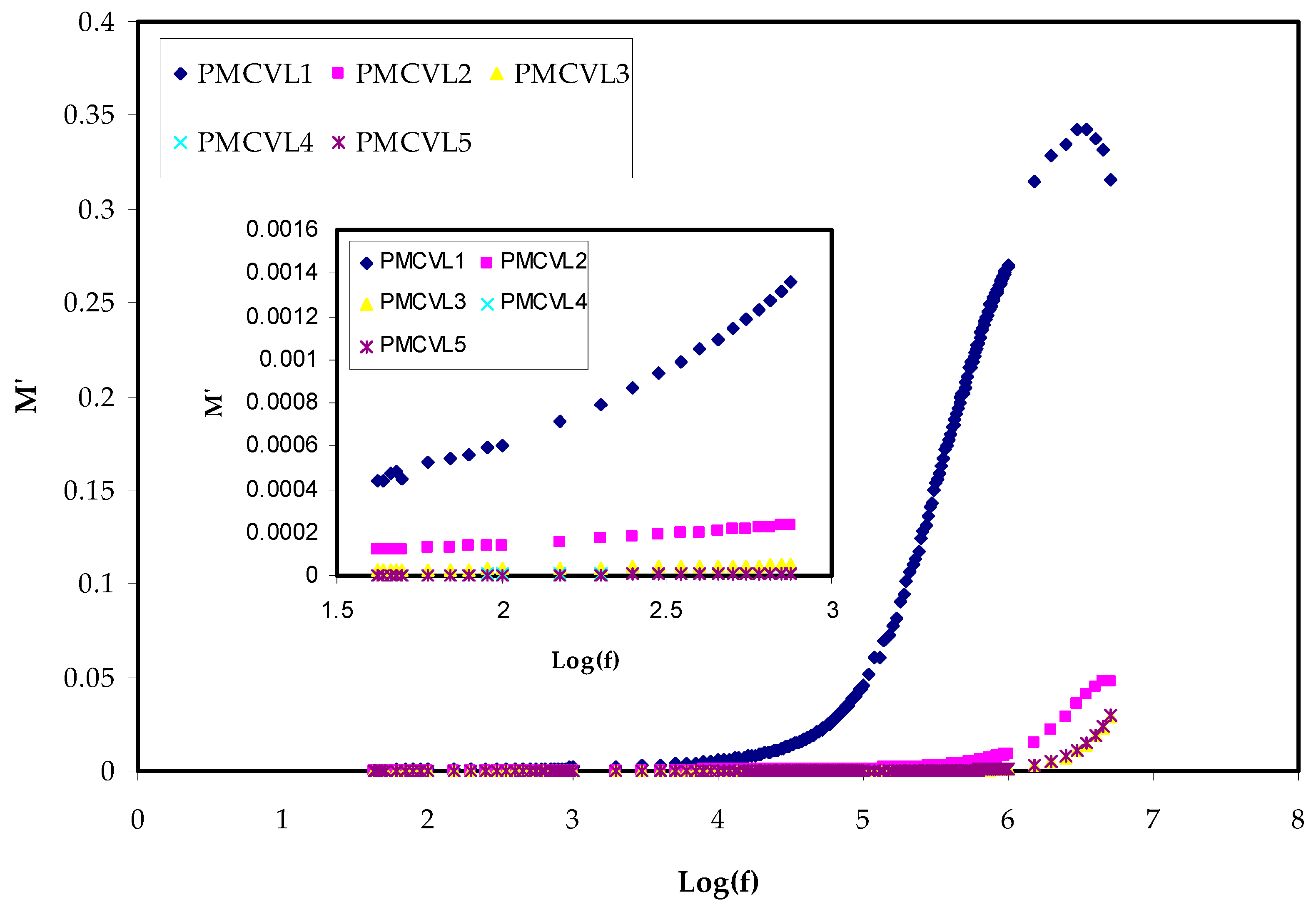
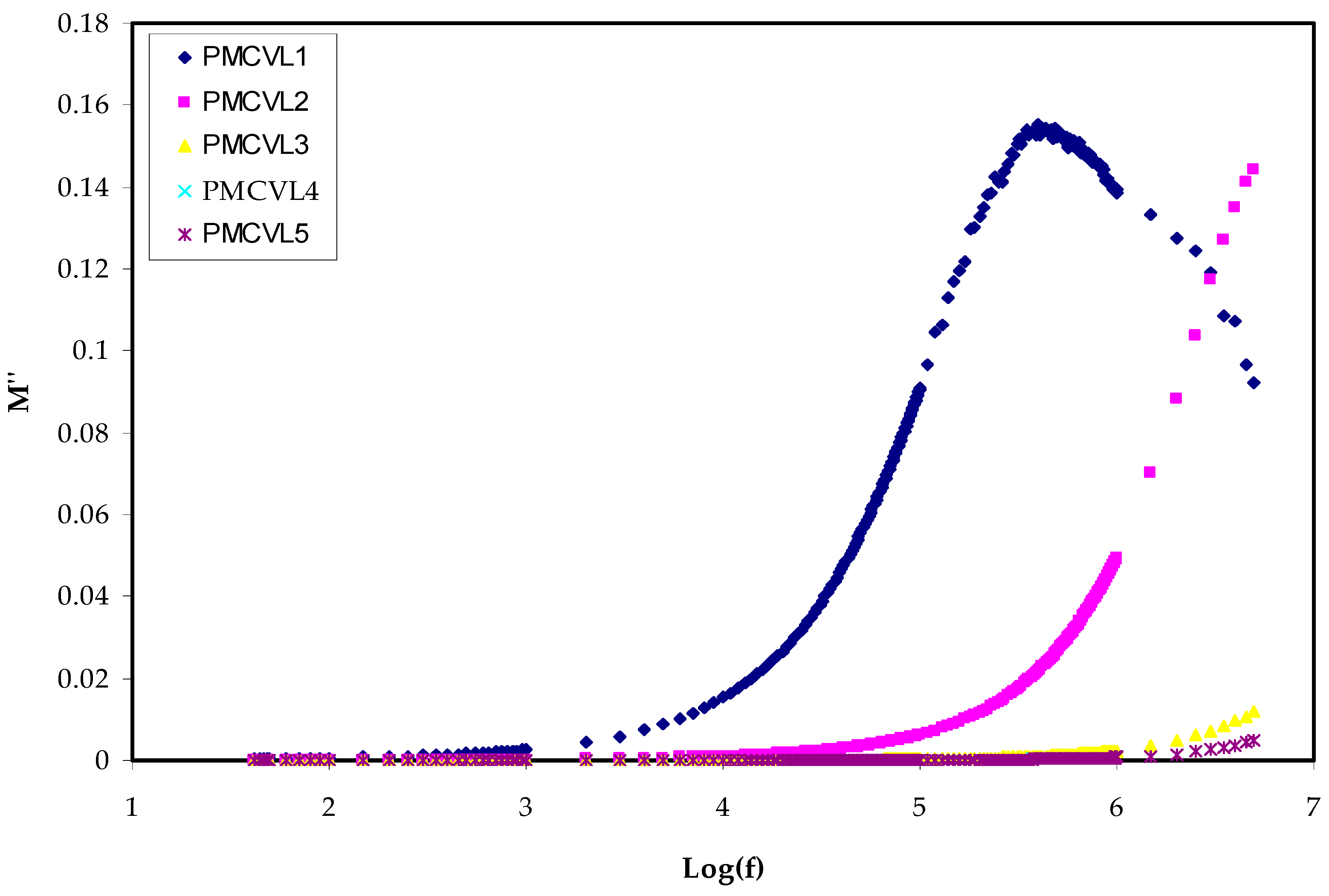
2.3.2. Electric Modulus Study
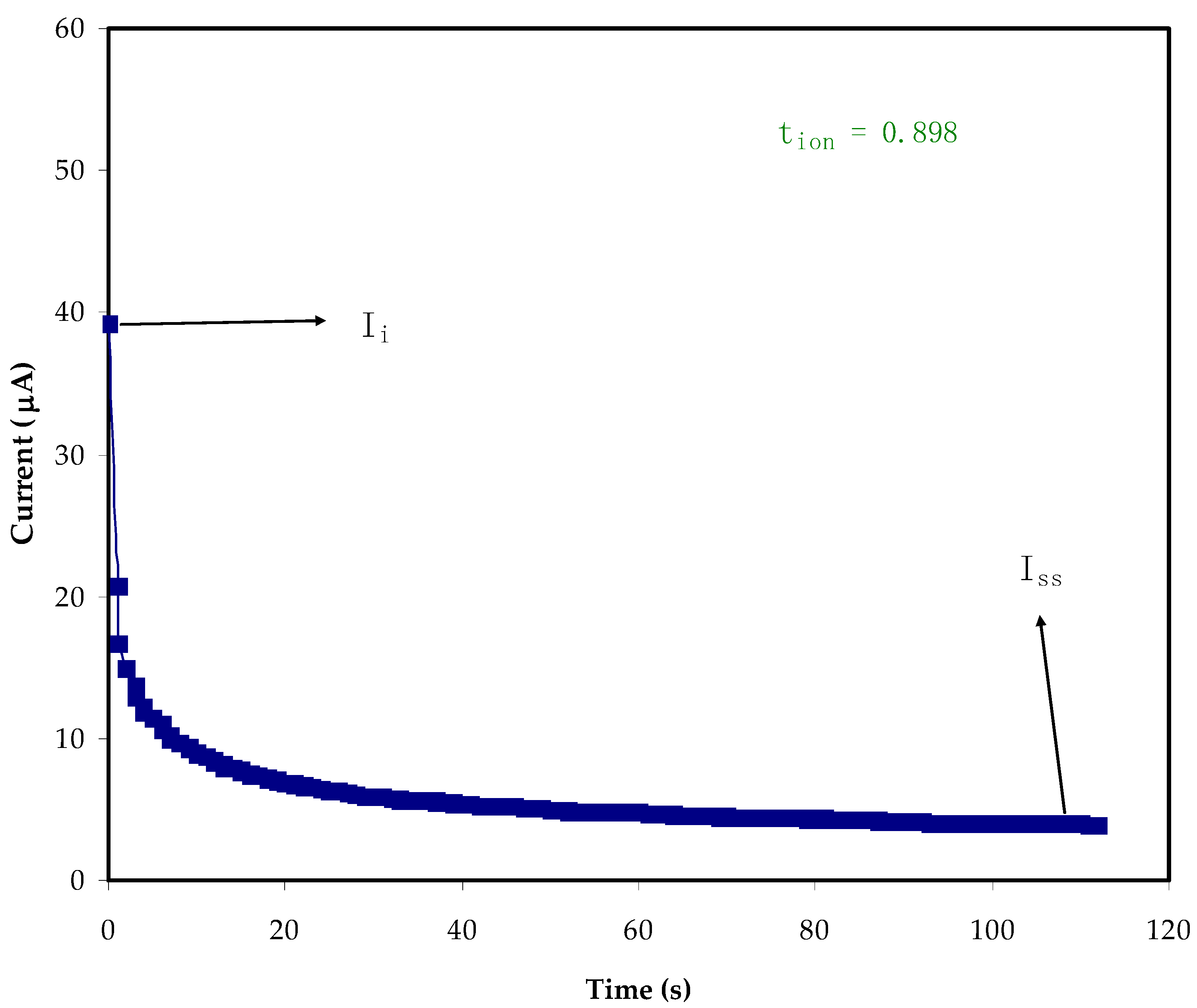
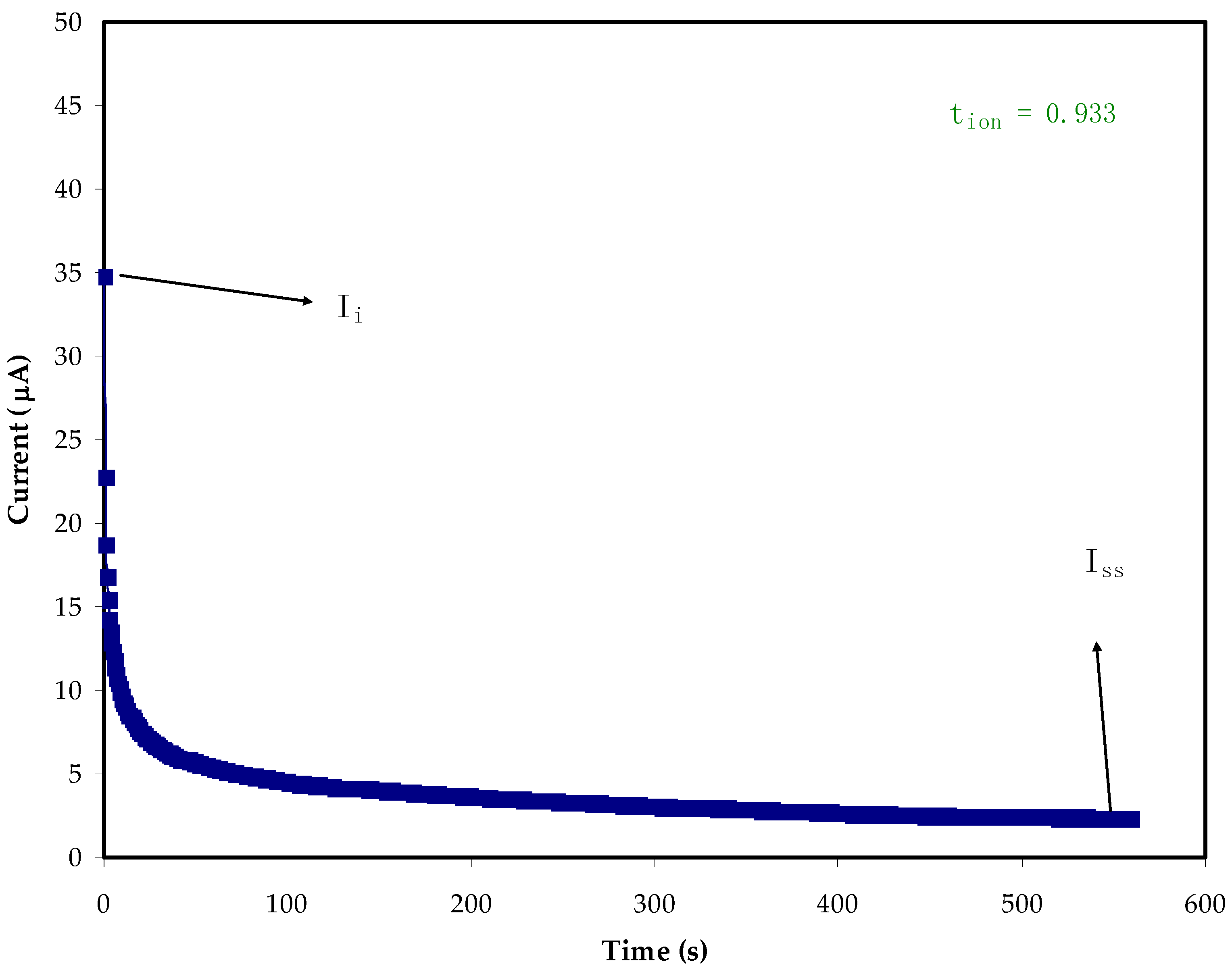
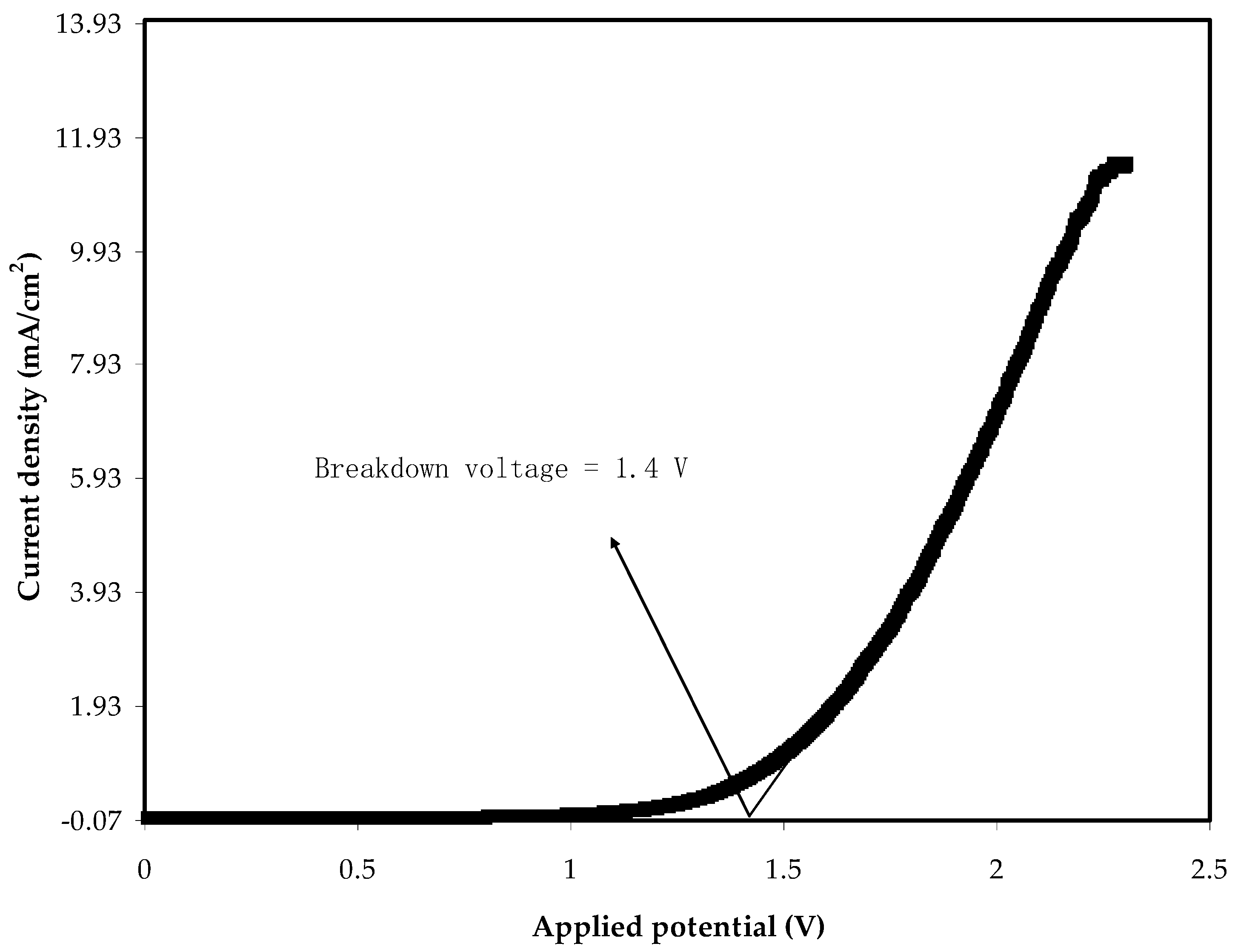
2.4. TNM and LSV Studies
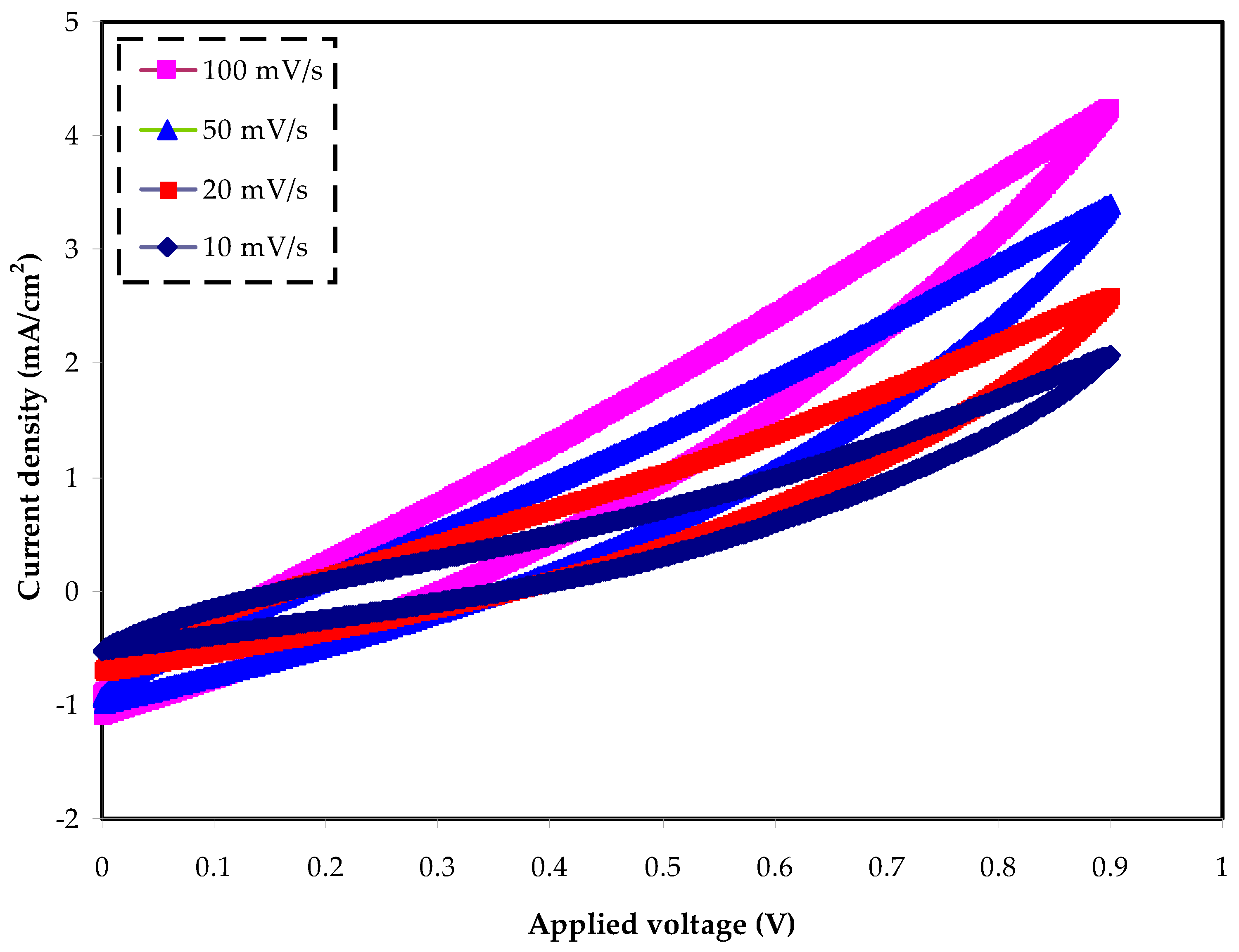
2.5. Cyclic Voltammetry Study
3. Materials and Methods
3.1. Materials and Electrolyte Preparation
3.2. Electrical Impedance Spectroscopy (EIS)
3.3. Transference Number Measurement (TNM) and Linear Sweep Voltammetry (LSV) Measurement
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dinoto, V.; Negro, E.; Lavina, S.; Vittadello, M. Hybrid inorganic–organic polymer electrolytes. In Polymer Electrolytes; Woodhead Publishing Series in Electronic and Optical Materials; Woodhead Publishing: Sawston, UK, 2010; pp. 219–277. [Google Scholar] [CrossRef]
- Aziz, S.B.; Woo, T.J.; Kadir, M.; Ahmed, H.M. A conceptual review on polymer electrolytes and ion transport models. J. Sci. Adv. Mater. Devices 2018, 3, 1–17. [Google Scholar] [CrossRef]
- Muldoon, J.; Bucur, C.B.; Boaretto, N.; Gregory, T.; di Noto, V. Polymers: Opening Doors to Future Batteries. Polym. Rev. 2015, 55, 208–246. [Google Scholar] [CrossRef]
- Nofal, M.M.; Hadi, J.M.; Aziz, S.B.; Brza, M.A.; Asnawi, A.S.F.M.; Dannoun, E.M.A.; Abdullah, A.M.; Kadir, M.F.Z. A Study of Methylcellulose Based Polymer Electrolyte Impregnated with Potassium Ion Conducting Carrier: Impedance, EEC Modeling, FTIR, Dielectric, and Device Characteristics. Materials 2021, 14, 4859. [Google Scholar] [CrossRef] [PubMed]
- Aziz, S.B. Li+ ion conduction mechanism in poly (ε-caprolactone)-based polymer electrolyte. Iran. Polym. J. 2013, 22, 877–883. [Google Scholar] [CrossRef] [Green Version]
- Itoh, T.; Mitsuda, Y.; Ebina, T.; Uno, T.; Kubo, M. Solid polymer electrolytes composed of polyanionic lithium salts and polyethers. J. Power Sources 2009, 189, 531–535. [Google Scholar] [CrossRef]
- Zhao, Y.; Wu, C.; Peng, G.; Chen, X.; Yao, X.; Bai, Y.; Wu, F.; Chen, S.; Xu, X. A new solid polymer electrolyte incorporating Li10GeP2S12 into a polyethylene oxide matrix for all-solid-state lithium batteries. J. Power Sources 2016, 301, 47–53. [Google Scholar] [CrossRef]
- Chinnam, P.R.; Zhang, H.; Wunder, S.L. Blends of Pegylated Polyoctahedralsilsesquioxanes (POSS-PEG) and Methyl Cellulose as Solid Polymer Electrolytes for Lithium Batteries. Electrochim. Acta 2015, 170, 191–201. [Google Scholar] [CrossRef]
- Ahmad, N.; Isa, M. Characterization of un-plasticized and propylene carbonate plasticized carboxymethyl cellulose doped ammonium chloride solid biopolymer electrolytes. Carbohydr. Polym. 2015, 137, 426–432. [Google Scholar] [CrossRef]
- Monisha, S.; Mathavan, T.; Selvasekarapandian, S.; Benial, A.M.F.; Aristatil, G.; Mani, N.; Premalatha, M.; Pandi, D.V. Investigation of bio polymer electrolyte based on cellulose acetate-ammonium nitrate for potential use in electrochemical devices. Carbohydr. Polym. 2017, 157, 38–47. [Google Scholar] [CrossRef]
- Fernandes, D.M.; Andrade, J.; Lima, M.; Silva, M.; Andrade, L.; Lima, S.; Hechenleitner, A.W.; Pineda, E.G. Thermal and photochemical effects on the structure, morphology, thermal and optical properties of PVA/Ni0.04Zn0.96O and PVA/Fe0.03Zn0.97O nanocomposite films. Polym. Degrad. Stab. 2013, 98, 1862–1868. [Google Scholar] [CrossRef]
- Rao, C.V.S.; Ravi, M.; Raja, V.; Bhargav, P.B.; Sharma, A.K.; Rao, V.V.R.N. Preparation and characterization of PVP-based polymer electrolytes for solid-state battery applications. Iran. Polym. J. 2012, 21, 531–536. [Google Scholar] [CrossRef]
- Kadir, M.; Aspanut, Z.; Majid, S.; Arof, A. FTIR studies of plasticized poly(vinyl alcohol)–chitosan blend doped with NH4NO3 polymer electrolyte membrane. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 78, 1068–1074. [Google Scholar] [CrossRef] [PubMed]
- Sudhakar, Y.N.; Selvakumar, M.; Bhat, D.K. LiClO4-doped plasticized chitosan and poly(ethylene glycol) blend as biodegradable polymer electrolyte for supercapacitors. Ionics 2012, 19, 277–285. [Google Scholar] [CrossRef]
- Sheha, E.; El-Mansy, M. A high voltage magnesium battery based on H2SO4-doped (PVA)0.7(NaBr)0.3 solid polymer electrolyte. J. Power Sources 2008, 185, 1509–1513. [Google Scholar] [CrossRef]
- Aziz, S.B.; Hadi, J.M.; Dannoun, E.M.A.; Abdulwahid, R.T.; Saeed, S.R.; Marf, A.S.; Karim, W.O.; Kadir, M.F. The Study of Plasticized Amorphous Biopolymer Blend Electrolytes Based on Polyvinyl Alcohol (PVA): Chitosan with High Ion Conductivity for Energy Storage Electrical Double-Layer Capacitors (EDLC) Device Application. Polymers 2020, 12, 1938. [Google Scholar] [CrossRef] [PubMed]
- Croce, F.; Persi, L.; Scrosati, B.; Serraino-Fiory, F.; Plichta, E.; Hendrickson, M.A. Role of the ceramic fillers in enhancing the transport properties of composite polymer electrolytes. Electrochi. Acta 2001, 46, 2457–2461. [Google Scholar] [CrossRef]
- Ngai, K.S.; Ramesh, S.; Ramesh, K.; Juan, J.C. A review of polymer electrolytes: Fundamental, approaches and applications. Ionics 2016, 22, 1259–1279. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abidin, Z.H.Z. Ion-transport study in nanocomposite solid polymer electrolytes based on chitosan: Electrical and dielectric analysis. J. Appl. Polym. Sci. 2014, 132, 41774. [Google Scholar] [CrossRef]
- Muthuvinayagam, M.; Sundaramahalingam, K. Characterization of proton conducting poly[ethylene oxide]: Poly[vinyl pyrrolidone] based polymer blend electrolytes for electrochemical devices. High Perform. Polym. 2020, 33, 205–216. [Google Scholar] [CrossRef]
- Aziz, S.B.; Marf, A.S.S.; Dannoun, E.M.A.; Brza, M.A.; Abdullah, R.M. The Study of the Degree of Crystallinity, Electrical Equivalent Circuit, and Dielectric Properties of Polyvinyl Alcohol (PVA)-Based Biopolymer Electrolytes. Polymers 2020, 12, 2184. [Google Scholar] [CrossRef]
- Wang, H.; Cao, S.; Yang, B.; Li, H.; Wang, M.; Hu, X.; Sun, K.; Zang, Z. NH 4 Cl-Modified ZnO for High-Performance CsPbIBr 2 Perovskite Solar Cells via Low-Temperature Process. Sol. RRL 2019, 4, 1900363. [Google Scholar] [CrossRef]
- Oku, T.; Ohishi, Y.; Ueoka, N. Highly (100)-oriented CH3NH3PbI3(Cl) perovskite solar cells prepared with NH4Cl using an air blow method. RSC Adv. 2018, 8, 10389–10395. [Google Scholar] [CrossRef] [Green Version]
- Nofal, M.M.; Aziz, S.B.; Hadi, J.M.; Abdulwahid, R.T.; Dannoun, E.M.A.; Marif, A.S.; Al-Zangana, S.; Zafar, Q.; Brza, M.A.; Kadir, M.F.Z. Synthesis of Porous Proton Ion Conducting Solid Polymer Blend Electrolytes Based on PVA: CS Polymers: Structural, Morphological and Electrochemical Properties. Materials 2020, 13, 4890. [Google Scholar] [CrossRef] [PubMed]
- Hadi, J.M.; Aziz, S.B.; Mustafa, M.S.; Brza, M.A.; Hamsan, M.H.; Kadir, M.F.Z.; Ghareeb, H.O.; Hussein, S.A. Electrochemical Impedance study of Proton Conducting Polymer Electrolytes based on PVC Doped with Thiocyanate and Plasticized with Glycerol. Int. J. Electrochem. Sci. 2020, 15, 4671–4683. [Google Scholar] [CrossRef]
- Dutta, P.; Biswas, S.; De, S.K. Dielectric relaxation in polyaniline–polyvinyl alcohol composites. Mater. Res. Bull. 2002, 37, 193–200. [Google Scholar] [CrossRef]
- Hemaa, M.; Selvasekerapandian, S.; Sakunthala, A.; Arunkumara, D.; Nithya, H. Structural, vibrational and electrical characterization of PVA–NH4Br polymer electrolyte system. Phys. B Condens. Matter 2008, 403, 2740–2747. [Google Scholar] [CrossRef]
- Aziz, S.B.; Hamsan, M.H.; Brza, M.A.; Kadir, M.F.Z.; Abdulwahid, R.T.; Ghareeb, H.O.; Woo, H.J. Fabrication of energy storage EDLC device based on CS:PEO polymer blend electrolytes with high Li+ ion transference number. Results Phys. 2019, 15, 102584. [Google Scholar] [CrossRef]
- Malathi, J.; Kumaravadivel, M.; Brahmanandhan, G.M.; Hema, M.; Baskaran, R.; Selvasekarapandian, S. Structural, thermal and electrical properties of PVA–LiCF3SO3 polymer electrolyte. J. Non. Cryst. Solids 2010, 356, 2277–2281. [Google Scholar] [CrossRef]
- Makled, M.; Sheha, E.; Shanap, T.; El-Mansy, M. Electrical conduction and dielectric relaxation in p-type PVA/CuI polymer composite. J. Adv. Res. 2012, 4, 531–538. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.A.; Lu, Y.T.; Lin, S.C.; Wang, Y.S.; Ma, C.C.M.; Hu, C.C. Designing a novel polymer electrolyte for improving the electrode/electrolyte interface in flexible all-solid-state electrical double-layer capacitors. ACS Appl. Mater. Interfaces 2018, 10, 17871–17882. [Google Scholar] [CrossRef]
- Jiang, L.; Yang, T.; Peng, L.; Dan, Y. Acrylamide modified poly(vinyl alcohol): Crystalline and enhanced water solubility. RSC Adv. 2015, 5, 86598–86605. [Google Scholar] [CrossRef]
- Hadi, J.M.; Aziz, S.B.; Saeed, S.R.; Brza, M.A.; Abdulwahid, R.T.; Hamsan, M.H.; Abdullah, R.M.; Kadir, M.F.Z.; Muzakir, S.K. Investigation of Ion Transport Parameters and Electrochemical Performance of Plasticized Biocompatible Chitosan-Based Proton Conducting Polymer Composite Electrolytes. Membranes 2020, 10, 363. [Google Scholar] [CrossRef]
- Benavente, J.; García, J.M.; Riley, R.; Lozano, A.E.; de Abajo, J. Sulfonated poly(ether ether sulfones): Characterization and study of dielectrical properties by impedance spectroscopy. J. Membr. Sci. 2000, 175, 43–52. [Google Scholar] [CrossRef]
- Aziz, S.B.; Brza, M.; Mohamed, P.A.; Kadir, M.; Hamsan, M.; Abdulwahid, R.T.; Woo, H. Increase of metallic silver nanoparticles in Chitosan:AgNt based polymer electrolytes incorporated with alumina filler. Results Phys. 2019, 13, 102326. [Google Scholar] [CrossRef]
- Hadi, J.M.; Aziz, S.B.; Kadir, M.; El-Badry, Y.A.; Ahamad, T.; Hussein, E.E.; Asnawi, A.S.; Abdullah, R.M.; Alshehri, S.M. Design of Plasticized Proton Conducting Chitosan:Dextran Based Biopolymer Blend Electrolytes for EDLC Application: Structural, Impedance and Electrochemical Studies. Arab. J. Chem. 2021, 14, 103394. [Google Scholar] [CrossRef]
- Benavente, J.; Zhang, X.; Valls, R.G. Modification of polysulfone membranes with polyethylene glycol and lignosulfate: Electrical characterization by impedance spectroscopy measurements. J. Colloid Interface Sci. 2005, 285, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Shukur, M.F.; Ithnin, R.; Kadir, M.F.Z. Electrical characterization of corn starch-LiOAc electrolytes and application in electrochemical double layer capacitor. Electrochim. Acta 2014, 136, 204–216. [Google Scholar] [CrossRef]
- Dannoun, E.M.; Aziz, S.B.; Brza, M.A.; Nofal, M.M.; Asnawi, A.S.; Yusof, Y.M.; Al-Zangana, S.; Hamsan, M.H.; Kadir, M.F.Z.; Woo, H.J. The Study of Plasticized Solid Polymer Blend Electrolytes Based on Natural Polymers and Their Application for Energy Storage EDLC Devices. Polymers 2020, 12, 2531. [Google Scholar] [CrossRef]
- Teo, L.P.; Buraidah, M.H.; Nor, A.F.M.; Majid, S.R. Conductivity and dielectric studies of Li2SnOIonics. Ionics 2012, 18, 655–665. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abdullah, R.M. Crystalline and amorphous phase identification from the tan δ relaxation peaks and impedance plots in polymer blend electrolytes based on [CS:AgNt]x:PEO(x−1) (10 ≤ x ≤ 50). Electrochim. Acta 2018, 285, 30–46. [Google Scholar] [CrossRef]
- Spectroscopy, R. Introduction to Top 20 Questions Answered.
- Aziz, S.B.; Ali, F.; Anuar, H.; Ahamad, T.; Kareem, W.O.; Brza, M.; Kadir, M.; Abu Ali, O.A.; Saleh, D.I.; Asnawi, A.; et al. Structural and electrochemical studies of proton conducting biopolymer blend electrolytes based on MC:Dextran for EDLC device application with high energy density. Alex. Eng. J. 2021, 61, 3985–3997. [Google Scholar] [CrossRef]
- Brza, M.; Aziz, S.; Anuar, H.; Ali, F. Structural, ion transport parameter and electrochemical properties of plasticized polymer composite electrolyte based on PVA: A novel approach to fabricate high performance EDLC devices. Polym. Test. 2020, 91, 106813. [Google Scholar] [CrossRef]
- Henn, F.; Durand, C.; Cerepi, A.; Brosse, E.; Giuntini, J. DC conductivity, cationic exchange capacity, and specific surface area related to chemical composition of pore lining chlorites. J. Colloid Interface Sci. 2007, 311, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Nahm, S.H. Use of dielectric spectroscopy for real-time in-situ reaction monitoring. J. Coat. Technol. Res. 2006, 3, 257–265. [Google Scholar] [CrossRef]
- Nicolau, A.; Nucci, A.M.; Martini, E.M.; Samios, D. Electrical impedance spectroscopy of epoxy systems II: Molar fraction variation, resistivity, capacitance and relaxation processes of 1,4-butanediol diglycidyl ether/succinic anhydride and triethylamine as initiator. Eur. Polym. J. 2007, 43, 2708–2717. [Google Scholar] [CrossRef]
- Rao, V.; Ashokan, P.; Shridhar, M. Studies of dielectric relaxation and a.c. conductivity in cellulose acetate hydrogen phthalate–poly(methyl methacrylate) blends. Mater. Sci. Eng. A 2000, 281, 213–220. [Google Scholar] [CrossRef]
- Khiar, A.; Puteh, R.; Arof, A. Conductivity studies of a chitosan-based polymer electrolyte. Phys. B Condens. Matter 2005, 373, 23–27. [Google Scholar] [CrossRef]
- Aziz, S.B.; Karim, W.O.; Brza, M.A.; Abdulwahid, R.T.; Saeed, S.R.; Al-Zangana, S.; Kadir, M.F.Z. Ion Transport Study in CS: POZ Based Polymer Membrane Electrolytes Using Trukhan Model. Int. J. Mol. Sci. 2019, 20, 5265. [Google Scholar] [CrossRef] [Green Version]
- Aziz, S.; Abidin, Z.H.Z. Electrical and morphological analysis of chitosan:AgTf solid electrolyte. Mater. Chem. Phys. 2014, 144, 280–286. [Google Scholar] [CrossRef]
- Michaelian, K.H. Introduction. In Photoacoustic Infrared Spectroscopy; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2003; pp. 1–6. [Google Scholar] [CrossRef]
- Filik, J. Raman spectroscopy: A the lightest touch. Spectrosc. Eur. 2005, 17, 5. [Google Scholar]
- Liao, Y.; Rao, M.; Li, W.; Yang, L.; Zhu, B.; Xu, R.; Fu, C. Fumed silica-doped poly(butyl methacrylate-styrene)-based gel polymer electrolyte for lithium ion battery. J. Membr. Sci. 2010, 352, 95–99. [Google Scholar] [CrossRef]
- Aziz, S.B.; Abdullah, O.G.; Rasheed, M.A. Structural and electrical characteristics of PVA:NaTf based solid polymer electrolytes: Role of lattice energy of salts on electrical DC conductivity. J. Mater. Sci. Mater. Electron. 2017, 28, 12873–12884. [Google Scholar] [CrossRef]
- Migahed, M.; Ishra, M.; Fahmy, T.; Barakat, A. Electric modulus and AC conductivity studies in conducting PPy composite films at low temperature. J. Phys. Chem. Solids 2004, 65, 1121–1125. [Google Scholar] [CrossRef]
- Hadi, J.M.; Aziz, S.B.; Mustafa, M.S.; Hamsan, M.H.; Abdulwahid, R.T.; Kadir, M.F.Z.; Ghareeb, H.O. Role of nano-capacitor on dielectric constant enhancement in PEO:NH4SCN:xCeO2 polymer nano-composites: Electrical and electrochemical properties. J. Mater. Res. Technol. 2020, 9, 9283–9294. [Google Scholar] [CrossRef]
- Harun, N.I.; Ali, R.M.; Ali, A.M.M.; Yahya, M.Z.A. Dielectric behaviour of cellulose acetate-based polymer electrolytes. Ionics 2011, 18, 599–606. [Google Scholar] [CrossRef]
- Smaoui, H.; Arous, M.; Guermazi, H.; Agnel, S.; Toureille, A. Study of relaxations in epoxy polymer by thermally stimulated depolarization current (TSDC) and dielectric relaxation spectroscopy (DRS). J. Alloy. Compd. 2010, 489, 429–436. [Google Scholar] [CrossRef]
- Patsidis, A.; Psarras, G. Dielectric behaviour and functionality of polymer matrix–ceramic BaTiO3 composites. Express Polym. Lett. 2008, 2, 718–726. [Google Scholar] [CrossRef]
- Berg, O.V.D.; Wübbenhorst, M.; Picken, S.; Jager, W.F. Characteristic size of molecular dynamics in polymers probed by dielectric probes of variable length. J. Non-Cryst. Solids 2005, 351, 2694–2702. [Google Scholar] [CrossRef]
- Hadi, J.M.; Aziz, S.B.; Nofal, M.M.; Hussein, S.A.; Hamsan, M.H.; Brza, M.A.; Abdulwahid, R.T.; Kadir, M.F.Z.; Woo, H.J. Electrical, Dielectric Property and Electrochemical Performances of Plasticized Silver Ion-Conducting Chitosan-Based Polymer Nanocomposites. Membranes 2020, 10, 151. [Google Scholar] [CrossRef]
- Marf, A.S.; Aziz, S.B.; Abdullah, R.M. Plasticized H+ ion-conducting PVA:CS-based polymer blend electrolytes for energy storage EDLC application. J. Mater. Sci. Mater. Electron. 2020, 31, 18554–18568. [Google Scholar] [CrossRef]
- Aziz, S.B.; Brza, M.A.; Brevik, I.; Hafiz, M.H.; Asnawi, A.S.; Yusof, Y.M.; Abdulwahid, R.T.; Kadir, M.F. Blending and Characteristics of Electrochemical Double-Layer Capacitor Device Assembled from Plasticized Proton Ion Conducting Chitosan:Dextran:NH4PF6 Polymer Electrolytes. Polymers 2020, 12, 2103. [Google Scholar] [CrossRef] [PubMed]
- Aziz, S.B.; Kadir, M.F.Z.; Abidin, Z.H.Z. Structural, Morphological and Electrochemical Impedance Study of CS:LiTf based Solid Polymer Electrolyte: Reformulated Arrhenius Equation for Ion Transport Study. Int. J. Electrochem. Sci. 2016, 11, 9228–9244. [Google Scholar] [CrossRef]
- Tripathi, M.; Tripathi, S. Electrical studies on ionic liquid-based gel polymer electrolyte for its application in EDLCs. Ionics 2017, 23, 2735–2746. [Google Scholar] [CrossRef]
- Shukur, M.F.; Ithnin, R.; Kadir, M.F.Z. Ionic conductivity and dielectric properties of potato starch-magnesium acetate biopolymer electrolytes: The effect of glycerol and 1-butyl-3-methylimidazolium chloride. Ionics 2016, 22, 1113–1123. [Google Scholar] [CrossRef]
- Shukur, M.; Ithnin, R.; Kadir, M.F.Z. Protonic Transport Analysis of Starch-Chitosan Blend Based Electrolytes and Application in Electrochemical Device. Mol. Cryst. Liq. Cryst. 2014, 603, 52–65. [Google Scholar] [CrossRef]
- Chai, M.N.; Isa, M.I.N. Novel Proton Conducting Solid Bio-polymer Electrolytes Based on Carboxymethyl Cellulose Doped with Oleic Acid and Plasticized with Glycerol. Sci. Rep. 2016, 6, 27328. [Google Scholar] [CrossRef]
- Rani, M.S.A.; Ahmad, A.; Mohamed, N.S. Influence of nano-sized fumed silica on physicochemical and electrochemical properties of cellulose derivatives-ionic liquid biopolymer electrolytes. Ionics 2018, 24, 807–814. [Google Scholar] [CrossRef]
- Brza, M.A.; Aziz, S.B.; Anuar, H.; Alshehri, S.M.; Ali, F.; Ahamad, T.; Hadi, J.M. Characteristics of a Plasticized PVA-Based Polymer Electrolyte Membrane and H+ Conductor for an Electrical Double-Layer Capacitor: Structural, Morphological, and Ion Transport Properties. Membranes 2021, 11, 296. [Google Scholar] [CrossRef]
- Aziz, S.; Nofal, M.; Kadir, M.; Dannoun, E.; Brza, M.; Hadi, J.; Abdullah, R. Bio-Based Plasticized PVA Based Polymer Blend Electrolytes for Energy Storage EDLC Devices: Ion Transport Parameters and Electrochemical Properties. Materials 2021, 14, 1994. [Google Scholar] [CrossRef]
- Shukur, M.F.; Hamsan, M.H.; Kadir, M.F.Z. Investigation of plasticized ionic conductor based on chitosan and ammonium bromide for EDLC application. Mater. Today Proc. 2019, 17, 490–498. [Google Scholar] [CrossRef]
- Francis, K.A.; Liew, C.-W.; Ramesh, S.; Ramesh, K.; Ramesh, S. Ionic liquid enhanced magnesium-based polymer electrolytes for electrical double-layer capacitors. Ionics 2016, 22, 919–925. [Google Scholar] [CrossRef]
- Sampathkumar, L.; Selvin, P.C.; Selvasekarapandian, S.; Perumal, P.; Chitra, R.; Muthukrishnan, M. Synthesis and characterization of biopolymer electrolyte based on tamarind seed polysaccharide, lithium perchlorate and ethylene carbonate for electrochemical applications. Ionics 2019, 25, 1067–1082. [Google Scholar] [CrossRef]
- Shuhaimi, N.E.A.; Alias, N.A.; Majid, S.R.; Arof, A.K. Electrical Double Layer Capacitor With Proton Conducting Κ-Carrageenan–Chitosan Electrolytes. Funct. Mater. Lett. 2008, 1, 195–201. [Google Scholar] [CrossRef]
- Bandaranayake, C.M.; Weerasinghe, W.A.D.S.S.; Vidanapathirana, K.P.; Perera, K.S. A Cyclic Voltammetry study of a gel polymer electrolyte based redox-capacitor. Sri Lankan J. Phys. 2016, 16, 19. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, H.; Morris, D. A new estimation of the lattice energies of the ammonium halides and the proton affinity of gaseous ammonia. Mol. Phys. 1976, 32, 231–236. [Google Scholar] [CrossRef]
- Kadir, M.F.Z.; Arof, A.K. Application of PVA–chitosan blend polymer electrolyte membrane in electrical double layer capacitor. Mater. Res. Innov. 2011, 15, S217–S220. [Google Scholar] [CrossRef]
- Fattah, N.F.A.; Ng, H.M.; Mahipal, Y.K.; Numan, A.; Ramesh, S.; Ramesh, K. An Approach to Solid-State Electrical Double Layer Capacitors Fabricated with Graphene Oxide-Doped, Ionic Liquid-Based Solid Copolymer Electrolytes. Materials 2016, 9, 450. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asnawi, A.; Aziz, S.; Brevik, I.; Brza, M.; Yusof, Y.; Alshehri, S.; Ahamad, T.; Kadir, M. The Study of Plasticized Sodium Ion Conducting Polymer Blend Electrolyte Membranes Based on Chitosan/Dextran Biopolymers: Ion Transport, Structural, Morphological and Potential Stability. Polymers 2021, 13, 383. [Google Scholar] [CrossRef] [PubMed]
- Muchakayala, R.; Song, S.; Wang, J.; Fan, Y.; Bengeppagari, M.; Chen, J.; Tan, M. Development and supercapacitor application of ionic liquid-incorporated gel polymer electrolyte films. J. Ind. Eng. Chem. 2018, 59, 79–89. [Google Scholar] [CrossRef]
| Designation | K1 (F−1) | K2 (F−1) | C1 (F) | C2 (F) |
|---|---|---|---|---|
| PMCVL1 | 1.60 × 108 | 1.70 × 106 | 6.25 × 10−9 | 5.88 × 10−7 |
| PMCVL2 | 2.15 × 106 | 4.65 × 10−7 | ||
| PMCVL3 | 5.90 × 105 | 1.69 × 10−6 | ||
| PMCVL4 | 6.90 × 104 | 1.45 × 10−5 | ||
| PMCVL5 | 4.20 × 104 | 2.38 × 10−5 |
| Designation | DC Conductivity (S/cm) |
|---|---|
| PMCVL1 | 1.02 × 10−6 |
| PMCVL2 | 9.62 × 10−5 |
| PMCVL3 | 1.49 × 10−4 |
| PMCVL4 | 1.03 × 10−3 |
| PMCVL5 | 1.99 × 10−3 |
| Scan Rate | V2-V1 | Capacitance (F/g) |
|---|---|---|
| 0.1 | 0.9 | 4.161 |
| 0.05 | 0.9 | 7.773 |
| 0.02 | 0.9 | 15.775 |
| 0.01 | 0.9 | 20.645 |
| PVA–MC–NH4Cl (wt.%) | Designation |
|---|---|
| 40-60-10 | PMCVL1 |
| 40-60-20 | PMCVL2 |
| 40-60-30 | PMCVL3 |
| 40-60-40 | PMCVL4 |
| 40-60-50 | PMCVL5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aziz, S.B.; Dannoun, E.M.A.; Brza, M.A.; Sadiq, N.M.; Nofal, M.M.; Karim, W.O.; Al-Saeedi, S.I.; Kadir, M.F.Z. An Investigation into the PVA:MC:NH4Cl-Based Proton-Conducting Polymer-Blend Electrolytes for Electrochemical Double Layer Capacitor (EDLC) Device Application: The FTIR, Circuit Design and Electrochemical Studies. Molecules 2022, 27, 1011. https://doi.org/10.3390/molecules27031011
Aziz SB, Dannoun EMA, Brza MA, Sadiq NM, Nofal MM, Karim WO, Al-Saeedi SI, Kadir MFZ. An Investigation into the PVA:MC:NH4Cl-Based Proton-Conducting Polymer-Blend Electrolytes for Electrochemical Double Layer Capacitor (EDLC) Device Application: The FTIR, Circuit Design and Electrochemical Studies. Molecules. 2022; 27(3):1011. https://doi.org/10.3390/molecules27031011
Chicago/Turabian StyleAziz, Shujahadeen B., Elham M. A. Dannoun, Mohamad A. Brza, Niyaz M. Sadiq, Muaffaq M. Nofal, Wrya O. Karim, Sameerahl I. Al-Saeedi, and Mohd F. Z. Kadir. 2022. "An Investigation into the PVA:MC:NH4Cl-Based Proton-Conducting Polymer-Blend Electrolytes for Electrochemical Double Layer Capacitor (EDLC) Device Application: The FTIR, Circuit Design and Electrochemical Studies" Molecules 27, no. 3: 1011. https://doi.org/10.3390/molecules27031011
APA StyleAziz, S. B., Dannoun, E. M. A., Brza, M. A., Sadiq, N. M., Nofal, M. M., Karim, W. O., Al-Saeedi, S. I., & Kadir, M. F. Z. (2022). An Investigation into the PVA:MC:NH4Cl-Based Proton-Conducting Polymer-Blend Electrolytes for Electrochemical Double Layer Capacitor (EDLC) Device Application: The FTIR, Circuit Design and Electrochemical Studies. Molecules, 27(3), 1011. https://doi.org/10.3390/molecules27031011









