Antidiabetic Potential of Commonly Available Fruit Plants in Bangladesh: Updates on Prospective Phytochemicals and Their Reported MoAs
Abstract
1. Introduction
2. Traditional Uses of Fruit Plants in the Management of Diabetes
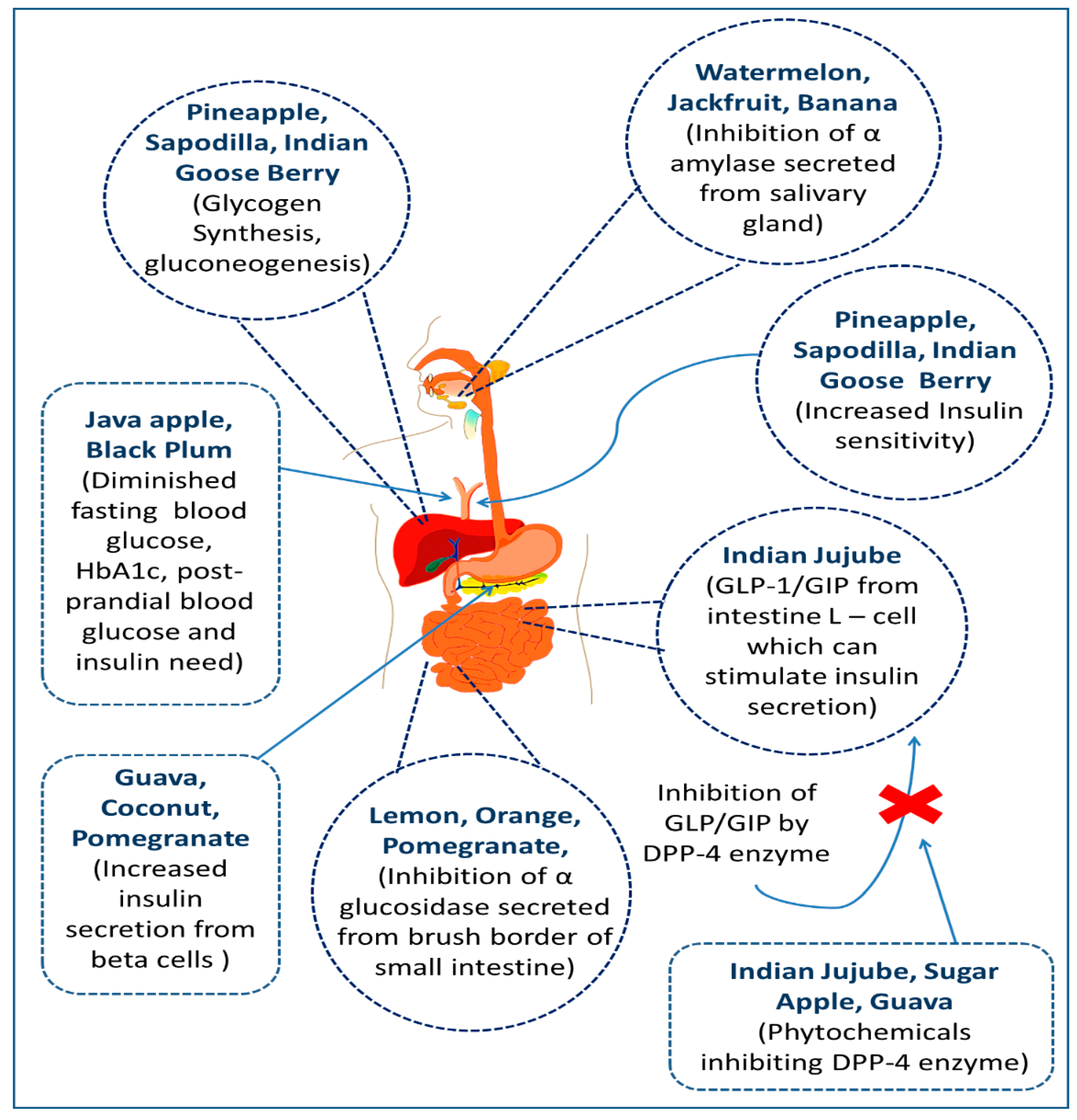
3. Antidiabetic Potentials of Commonly Available Bangladeshi Fruits
3.1. Banana (Musa sapientum L.)
3.2. Bengal Quince (Aegle marmelos (L.) Corrêa)
3.3. Black Currant (Carissa carandas L.)
3.4. Black Plum (Syzygium cumini (L.) Skeels)
3.5. Coconut (Cocos nucifera L.)
3.6. Elephant Apple (Dillenia indica L.)
3.7. Guava (Psidium guajava L.)
3.8. Hog Plum (Spondias mombin L.)
3.9. Indian Goose Berry (Phyllanthus emblica L.)
3.10. Indian Olive (Elaeocarpus floribundus Blume)
3.11. Indian Jujube (Zizyphus mauritiana Lam.)
3.12. Indian Persimmon (Diospyros malabarica (Desr.) Kostel.)
3.13. Jackfruit (Artocarpus heterophyllus Lam.)
3.14. Java Apple (Syzygium samarangense (Blume) Merr. & L.M.Perry)
3.15. Key Lime (Citrus aurantiifolia (Christm.) Swingle)
3.16. Lemon (Citrus limon (L.) Osbeck)
3.17. Lotkon (Baccaurea motleyana (Müll.Arg.))
3.18. Lychee (Litchi chinensis Sonn.)
3.19. Mango (Mangifera indica L.)
3.20. Muskmelon (Cucumis melo L.)
3.21. Orange (Citrus reticulata Blanco)
3.22. Papaya (Carica papaya L.)
3.23. Pineapple (Ananas comosus (L.) Merr.)
3.24. Pomelo (Citrus maxima (Burm.) Merr.)
3.25. Pomegranate (Puncia granatum L.)
3.26. Sapodilla (Manilkara zapota (L.) P.Royen)
3.27. Star Fruit (Averrhoa carambola L.)
3.28. Sugar apple (Annona squamosa L.)
3.29. Watermelon (Citrullus lanatus (Thunb.) Matsum. & Nakai)
3.30. Tal Palm (Borassus flabellifer L.)
3.31. Tamarind (Tamarindus indica L.)
3.32. Wood Apple (Limonia acidissima Groff)
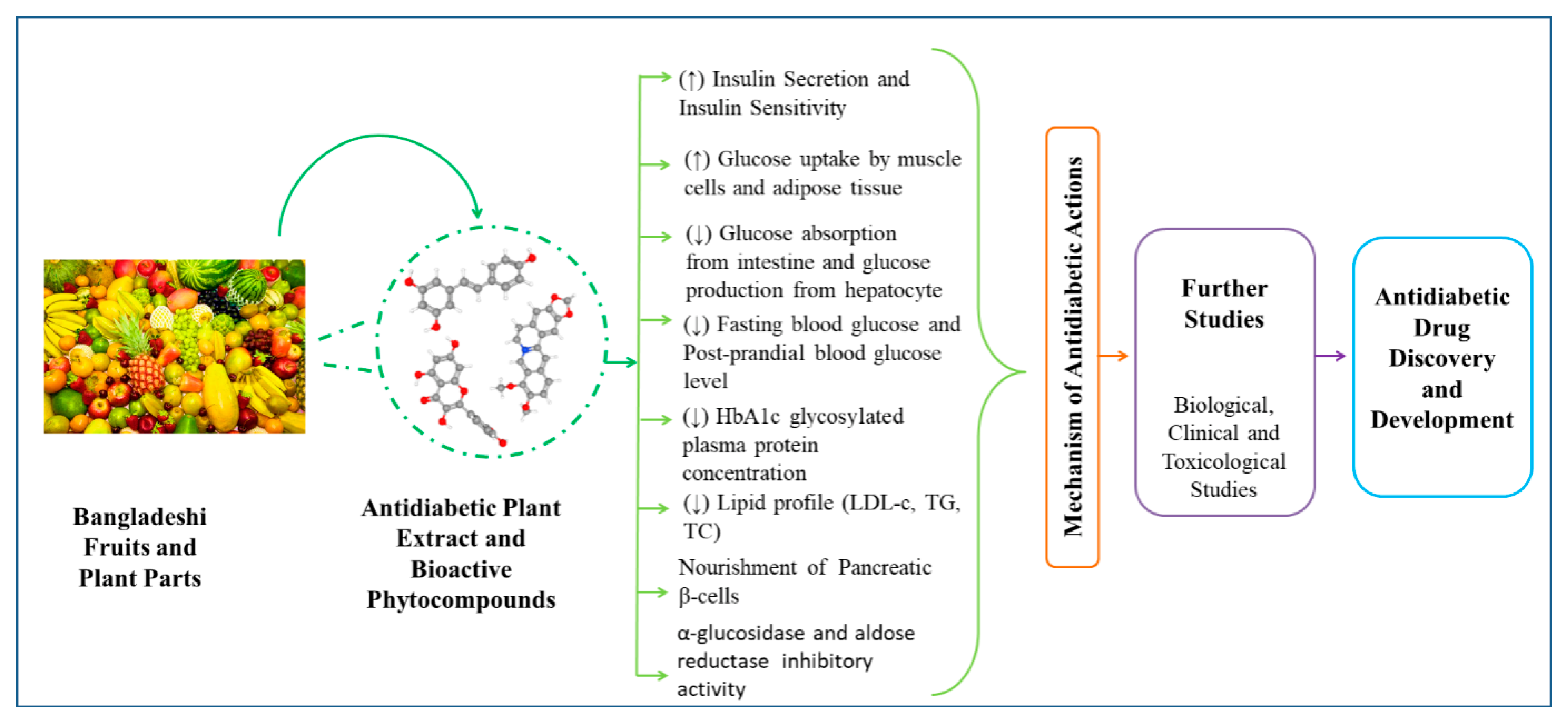
4. Phytochemicals from Fruits and Other Plant Parts
5. Clinical Trials of Prospective Fruit Plants and Their Phytochemicals to Treat Diabetes Mellitus
6. Reported Mechanism of Actions to Exert Antidiabetic Potentials
- a.
- Inhibition of α-glucosidase secreted from the brush border of the small intestine
- b.
- Inhibition of DPP-4 enzyme
- c.
- Inhibition of α-amylase secreted from the salivary gland
- d.
- Increased secretion of insulin
- e.
- Increased insulin sensitivity and improved glucose uptake by muscle cells and adipose tissue
- f.
- Nourishment of Pancreatic β-Cells
- g.
- Reduction of HbA1c and glycated plasma protein levels
- h.
- Improvement of Glucagon-like peptide-1 (GLP-1)
- i.
- Regulation of Glucose transporter type 4 (GLUT-4)
7. Pharmacokinetic and Toxicological Profiles of Phytochemicals
8. Discussion
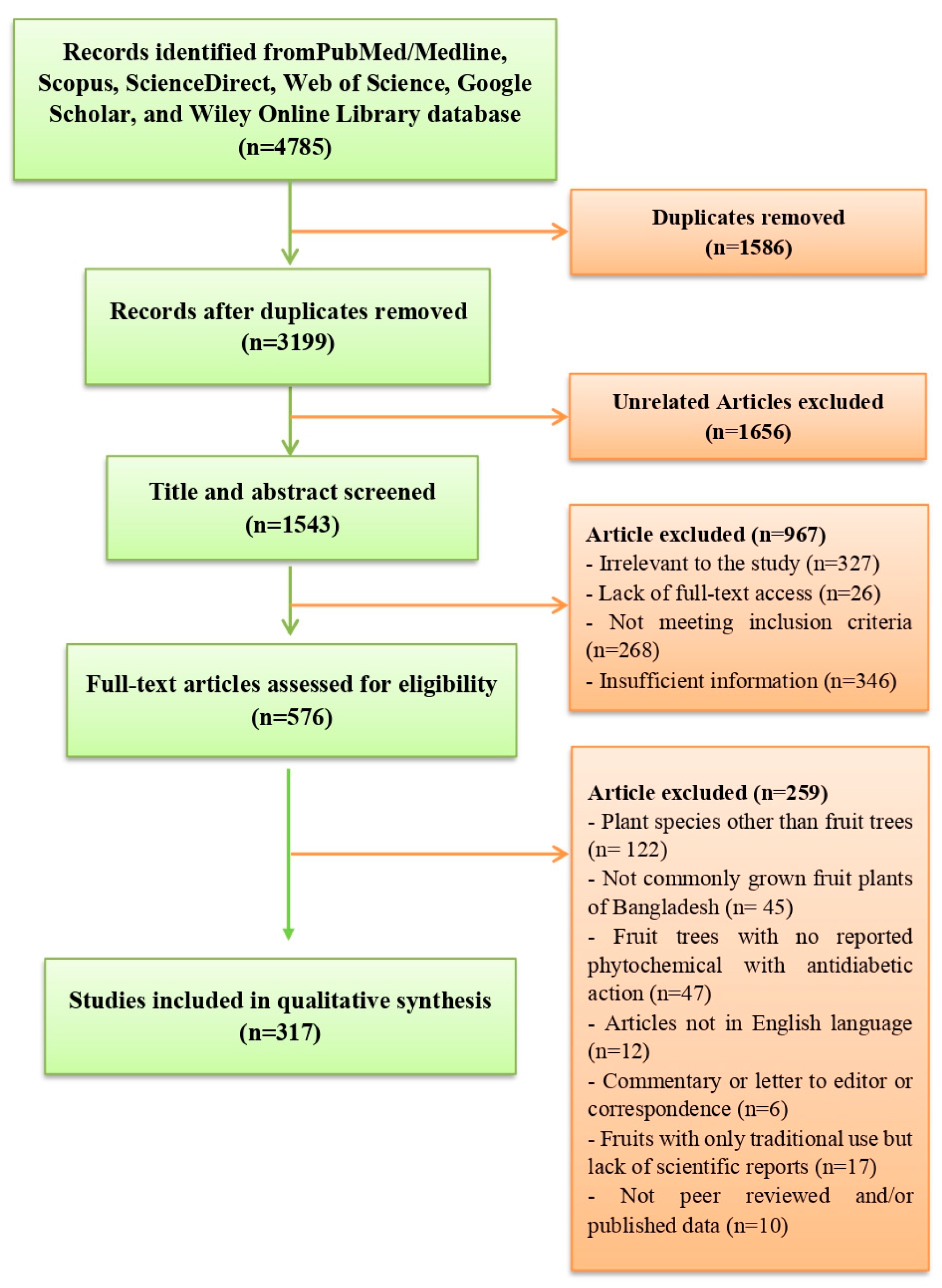
9. Materials and Methods
Article Search Strategy
10. Conclusion and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kharroubi, A.T.; Darwish, H.M. Diabetes mellitus: The epidemic of the century. World J. Diabetes 2015, 6, 850–867. [Google Scholar] [CrossRef] [PubMed]
- Amraee, S.; Bahramikia, S. Inhibitory effect of effective fraction of Salvia officinalis on aldose reductase activity: Strategy to reduce complications of type 2 diabetes. Orient. Pharm. Exp. Med. 2019, 19, 211–216. [Google Scholar] [CrossRef]
- Patel, D.; Prasad, S.K.; Kumar, R.; Hemalatha, S. An overview on antidiabetic medicinal plants having insulin mimetic property. Asian Pac. J. Trop. Biomed. 2012, 2, 320–330. [Google Scholar] [CrossRef] [PubMed]
- Rani, A.; Arora, S.; Goyal, A. Antidiabetic plants in traditional medicines: A review. Int. Res. J. Pharm 2017, 8, 17–24. [Google Scholar] [CrossRef]
- Association, A.D. Diagnosis and classification of diabetes mellitus. Diabetes Care 2009, 32, S62–S67. [Google Scholar] [CrossRef]
- Cakan, N.; Kizilbash, S.; Kamat, D. Changing spectrum of diabetes mellitus in children: Challenges with initial classification. Clin. Pediatr. 2012, 51, 939–944. [Google Scholar] [CrossRef]
- Li, G.Q.; Kam, A.; Wong, K.H.; Zhou, X.; Omar, E.A.; Alqahtani, A.; Li, K.M.; Razmovski-Naumovski, V.; Chan, K. Herbal medicines for the management of diabetes. Diabetes 2013, 771, 396–413. [Google Scholar]
- Akapelwa, T.M.; Ezaela, C.E.; Mushabati, F.; Bamitale, S.D.-K.; Kibuule, D. Commentary on the Antidiabetic Activity of Kigelia Africana. J. Prev. Rehabil. Med. 2021, 3, 21–23. [Google Scholar]
- Zamljen, T.; Medič, A.; Veberič, R.; Hudina, M.; Štampar, F.; Slatnar, A. Apple Fruit (Malus domestica Borkh.) Metabolic Response to Infestation by Invasive Brown Marmorated Stink Bug (Halyomorpha halys Stal.). Horticulturae 2021, 7, 212. [Google Scholar] [CrossRef]
- Emon, N.U.; Alam, S.; Rudra, S.; Al Haidar, I.K.; Farhad, M.; Rana, M.E.H.; Ganguly, A. Antipyretic activity of the leaves extract of Caesalpinia digyna Rottl along with phytoconstituent’s binding affinity to COX-1, COX-2 and mPGES-1 receptors: An in vivo and in silico approaches (Antipyretic activity of Caesalpinia digyna Rottl). Saudi J. Biol. Sci. 2021, 28, 5302–5309. [Google Scholar] [CrossRef] [PubMed]
- Ashrafi, S.; Alam, S.; Emon, N.U.; Ahsan, M. Isolation, Characterization and Pharmacological Investigations of a New Phenolic Compound along with Four Others Firstly Reported Phytochemicals from Glycosmis cyanocarpa (Blume) Spreng. Molecules 2022, 27, 5972. [Google Scholar] [CrossRef]
- Asif, M. The role of fruits, vegetables, and spices in diabetes. Int. J. Nutr. Pharmacol. Neurol. Dis. 2011, 1, 27. [Google Scholar] [CrossRef]
- Devalaraja, S.; Jain, S.; Yadav, H. Exotic fruits as therapeutic complements for diabetes, obesity and metabolic syndrome. Food Res. Int. 2011, 44, 1856–1865. [Google Scholar] [CrossRef] [PubMed]
- Mirunalini, S.; Krishnaveni, M. Therapeutic potential of Phyllanthus emblica (amla): The ayurvedic wonder. J. Basic Clin. Physiol. Pharmacol. 2010, 21, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Devi, S.; Kumar, D.; Kumar, M. Ethnobotanical values of antidiabetic plants of MP region India. J. Med. Plants Stud 2016, 4, 26–28. [Google Scholar]
- Kumar, B.D.; Mitra, A.; Manjunatha, M. In vitro and in vivo studies of antidiabetic Indian medicinal plants: A review. J. Herb. Med. Toxicol. 2009, 3, 9–14. [Google Scholar]
- Daimari, M.; Roy, M.K.; Swargiary, A.; Baruah, S.; Basumatary, S. An ethnobotanical survey of antidiabetic medicinal plants used by the Bodo tribe of Kokrajhar district, Assam. Indian J. Tradit. Knowl. 2019, 18, 421–429. [Google Scholar]
- Bahmani, M.; Zargaran, A.; Rafieian-Kopaei, M.; Saki, K. Ethnobotanical study of medicinal plants used in the management of diabetes mellitus in the Urmia, Northwest Iran. Asian Pac. J. Trop. Med. 2014, 7, S348–S354. [Google Scholar] [CrossRef]
- Durmuskahya, C.; Ozturk, M. Ethnobotanical survey of medicinal plants used for the treatment of diabetes in Manisa, Turkey. Sains Malays. 2013, 42, 1431–1438. [Google Scholar] [CrossRef]
- Abo, K.; Fred-Jaiyesimi, A.; Jaiyesimi, A. Ethnobotanical studies of medicinal plants used in the management of diabetes mellitus in South Western Nigeria. J. Ethnopharmacol. 2008, 115, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Édouard, K.K.; Kouassi, K. Ethnobotanical study of plants used to treat diabetes, in traditional medicine, by Abbey and Krobou people of Agboville (Côte-d’Ivoire). Am. J. Sci. Res. 2009, 4, 45–58. [Google Scholar]
- Vandebroek, I.; Balick, M.J. Lime for chest congestion, bitter orange for diabetes: Foods as medicines in the Dominican community in New York City. Econ. Bot. 2014, 68, 177–189. [Google Scholar] [CrossRef]
- Chakravarty, S.; Kalita, J. An investigation on anti diabetic medicinal plants used by villagers in Nalbari district, Assam, India. Int. J. Pharm. Sci. Res. 2012, 3, 1693. [Google Scholar]
- Dixit, P.; Kumar, V.; Shukla, R. Therapeutic and medicinal effects of different parts of Musa sapientum. Vivechan Int. J. Res. 2014, 5, 62–68. [Google Scholar]
- Rao, U.; Suryati, M.K.; Abdurrazaq, M.; Ahmad, B.A.; Mohaslinda, M.; Ali, R.M. Taxonomical, Phytochemical and Pharmacological Reviews of Musa sapientum var. Paradisiaca. Res. J. Pharm. Technol. 2014, 7, 1356–1361. [Google Scholar]
- De Oliveira Vilhena, R.; Fachi, M.M.; Marson, B.M.; Dias, B.L.; Pontes, F.L.; Tonin, F.S.; Pontarolo, R. Antidiabetic potential of Musa spp. inflorescence: A systematic review. J. Pharm. Pharmacol. 2018, 70, 1583–1595. [Google Scholar] [CrossRef]
- Dikshit, P.; Shukla, K.; Tyagi, M.K.; Garg, P.; Gambhir, J.K.; Shukla, R. Antidiabetic and antihyperlipidemic effects of the stem of Musa sapientum Linn. in streptozotocin-induced diabetic rats. J. Diabetes 2012, 4, 378–385. [Google Scholar] [CrossRef]
- Ige, A. Mechanism of Anti-Diabetic Activities of Musa Sapientum Leaf Extract in Rats Treated with Alloxan. Ph.D. Dissertation, University of Ibadan, Ibadan, Nigeria, 2014. [Google Scholar]
- Nigam, V.; Nambiar, V. Therapeutic potential of Aegle marmelos (L.) Correa leaves as an antioxidant and anti-diabetic agent: A review. Int. J. Pharma Sci. Res. 2015, 6, 611–621. [Google Scholar]
- Sekar, D.K.; Kumar, G.; Karthik, L.; Rao, K.B. A review on pharmacological and phytochemical properties of Aegle marmelos (L.) Corr. Serr. (Rutaceae). Asian J. Plant Sci. Res 2011, 1, 8–17. [Google Scholar]
- Manandhar, B.; Paudel, K.R.; Sharma, B.; Karki, R. Phytochemical profile and pharmacological activity of Aegle marmelos Linn. J. Integr. Med. 2018, 16, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Gandhi, G.R.; Ignacimuthu, S.; Paulraj, M.G. Hypoglycemic and β-cells regenerative effects of Aegle marmelos (L.) Corr. bark extract in streptozotocin-induced diabetic rats. Food Chem. Toxicol. 2012, 50, 1667–1674. [Google Scholar] [CrossRef] [PubMed]
- Sankeshi, V.; Kumar, P.A.; Naik, R.R.; Sridhar, G.; Kumar, M.P.; Gopal, V.H.; Raju, T.N. Inhibition of aldose reductase by Aegle marmelos and its protective role in diabetic cataract. J. Ethnopharmacol. 2013, 149, 215–221. [Google Scholar] [CrossRef]
- Gohil, T.; Pathak, N.; Jivani, N.; Devmurari, V.; Patel, J. Treatment with extracts of Eugenia jambolana seed and Aegle marmelos leaf prevents hyperglycemia and hyperlipidemia in alloxan induced diabetic rats. Afr. J. Pharm. Pharmacol. 2010, 4, 270–275. [Google Scholar]
- Singh, S.P.; Kumar, S.; Mathan, S.V.; Tomar, M.S.; Singh, R.K.; Verma, P.K.; Kumar, A.; Kumar, S.; Singh, R.P.; Acharya, A. Therapeutic application of Carica papaya leaf extract in the management of human diseases. DARU J. Pharm. Sci. 2020, 28, 735–744. [Google Scholar] [CrossRef]
- Anupama, N.; Madhumitha, G.; Rajesh, K. Role of dried fruits of Carissa carandas as anti-inflammatory agents and the analysis of phytochemical constituents by GC-MS. BioMed Res. Int. 2014, 2014, 512369. [Google Scholar] [CrossRef]
- Singh, A.; Uppal, G.K. A review on carissa carandas ǧ phytochemistry, ethnoǧpharmacology, and micropropagation as conservation strategy. Asian J. Pharm. Clin. Res. 2015, 8, 26–30. [Google Scholar]
- Madhuri, S.; Neelagund, S.E. Anti-oxidant, anti-diabetic activity and DNA damage inhibition activity of Carissa carandas fruit. Int. J. Adv. Res. Dev. 2019, 4, 75–82. [Google Scholar]
- Proma, N.M.; Naima, J.; Islam, M.R.; Papel, J.A.; Rahman, M.M.; Hossain, M.K. Phytochemical constituents and antidiabetic properties of Syzygium cumini Linn. Seed. Int. J. Pharm. Sci. Res. 2018, 9, 1806–1814. [Google Scholar]
- Jagetia, G.C. Phytochemical Composition and pleotropic pharmacological properties of jamun, Syzygium cumini skeels. J. Explor. Res. Pharmacol. 2017, 2, 54–66. [Google Scholar] [CrossRef]
- Agarwala, P.; Gaurb, P.K.; Tyagia, N.; Purib, D.; Kumarc, N.; Kumard, S.S. An overview of phytochemical, therapeutic, pharmacological and traditional importance of Syzygium cumini. Asian J. Pharm. 2019, 3, 5–17. [Google Scholar]
- Chhikara, N.; Kaur, R.; Jaglan, S.; Sharma, P.; Gat, Y.; Panghal, A. Bioactive compounds and pharmacological and food applications of Syzygium cumini—A review. Food Funct. 2018, 9, 6096–6115. [Google Scholar] [CrossRef] [PubMed]
- Artanti, N.; Maryani, F.; Dewi, R.T.; Handayani, S.; Dewijanti, I.D.; Meilawati, L.; Filaila, E.; Udin, L.Z. In vitro antidiabetic, antioxidant and cytotoxic activities of Syzygium cumini fractions from leaves ethanol extract. Indones. J. Cancer Chemoprev. 2019, 10, 24–29. [Google Scholar] [CrossRef]
- Rather, G.J.; Hamidudin, M.; Ikram, M.; Zaman, R. Antidiabetic potential and related activity of Jamun (Syzygium cumini Linn.) and its utilization in Unani medicine: An overview. Int. J. Herb. Med. 2019, 7, 07–11. [Google Scholar]
- Srivastava, S.; Chandra, D. Pharmacological potentials of Syzygium cumini: A review. J. Sci. Food Agric. 2013, 93, 2084–2093. [Google Scholar] [CrossRef] [PubMed]
- Gajera, H.; Gevariya, S.N.; Hirpara, D.G.; Patel, S.; Golakiya, B. Antidiabetic and antioxidant functionality associated with phenolic constituents from fruit parts of indigenous black jamun (Syzygium cumini L.) landraces. J. Food Sci. Technol. 2017, 54, 3180–3191. [Google Scholar] [CrossRef]
- Hooda, V.; Sharma, G.N.; Tyagi, N.; Hooda, A. Phytochemical and Pharmacological Profile of Cocos nucifera: An Overview. Int. J. Pharm. Ther. 2012, 3, 130–135. [Google Scholar]
- Lima, E.; Sousa, C.; Meneses, L.; Ximenes, N.; Santos, M.; Vasconcelos, G.; Lima, N.; Patrocínio, M.; Macedo, D.; Vasconcelos, S. Cocos nucifera (L.) (Arecaceae): A phytochemical and pharmacological review. Braz. J. Med. Biol. Res. 2015, 48, 953–964. [Google Scholar] [CrossRef]
- Naskar, S.; Mazumder, U.K.; Pramanik, G.; Gupta, M.; Kumar, R.S.; Bala, A.; Islam, A. Evaluation of antihyperglycemic activity of Cocos nucifera Linn. on streptozotocin induced type 2 diabetic rats. J. Ethnopharmacol. 2011, 138, 769–773. [Google Scholar] [CrossRef]
- Zohoun, E.V.; Tang, E.N.; Soumanou, M.M.; Manful, J.; Akissoe, N.H.; Bigoga, J.; Futakuchi, K.; Ndindeng, S.A. Physicochemical and nutritional properties of rice as affected by parboiling steaming time at atmospheric pressure and variety. Food Sci. Nutr. 2018, 6, 638–652. [Google Scholar] [CrossRef]
- Preetha, P.; Devi, V.G.; Rajamohan, T. Comparative effects of mature coconut water (Cocos nucifera) and glibenclamide on some biochemical parameters in alloxan induced diabetic rats. Rev. Bras. Farmacogn. 2013, 23, 481–487. [Google Scholar] [CrossRef]
- Talukdar, A.; Talukdar, N.; Deka, S.; Sahariah, B.J. Dillenia indica (OUTENGA) as anti-diabetic herb found in Assam: A review. Int. J. Pharm. Sci. Res. 2012, 3, 2482. [Google Scholar]
- Kumar, S.; Kumar, V.; Prakash, O. Enzymes inhibition and antidiabetic effect of isolated constituents from Dillenia indica. BioMed Res. Int. 2013, 2013, 382063. [Google Scholar] [CrossRef] [PubMed]
- Kaur, N.; Kishore, L.; Singh, R. Dillenia indica L. attenuates diabetic nephropathy via inhibition of advanced glycation end products accumulation in STZ-nicotinamide induced diabetic rats. J. Tradit. Complement. Med. 2018, 8, 226–238. [Google Scholar] [CrossRef]
- Kamboj, P.; Talukdar, N.C.; Banerjee, S.K. Therapeutic benefit of Dillenia indica in diabetes and its associated complications. J. Diabetes Res. 2019, 2019, 4632491. [Google Scholar] [CrossRef]
- Joseph, B.; Priya, M. Review on nutritional, medicinal and pharmacological properties of guava (Psidium guajava Linn.). Int. J. Pharma Bio Sci. 2011, 2, 53–69. [Google Scholar]
- Beidokhti, M.N.; Eid, H.M.; Villavicencio, M.L.; Jäger, A.K.; Lobbens, E.S.; Rasoanaivo, P.R.; McNair, L.M.; Haddad, P.S.; Staerk, D. Evaluation of the antidiabetic potential of Psidium guajava L. (Myrtaceae) using assays for α-glucosidase, α-amylase, muscle glucose uptake, liver glucose production, and triglyceride accumulation in adipocytes. J. Ethnopharmacol. 2020, 257, 112877. [Google Scholar] [CrossRef]
- Dakappa, S.S.; Adhikari, R.; Timilsina, S.S.; Sajjekhan, S. A review on the medicinal plant Psidium guajava Linn. (Myrtaceae). J. Drug Deliv. Ther. 2013, 3, 162–168. [Google Scholar] [CrossRef]
- Rishika, D.; Sharma, R. An update of pharmacological activity of Psidium guajava in the management of various disorders. Int. J. Pharm. Sci. Res. 2012, 3, 3577. [Google Scholar]
- Ramasamy, M.; Arumugam Vijaya, A.; Sampath, K.; Pushpa. Phytochemical and In vitro Antidiabetic Activity of Psidium guajava Leaves. Pharmacogn. J. 2016, 8, 392–394. [Google Scholar]
- Huang, C.-S.; Yin, M.-C.; Chiu, L.-C. Antihyperglycemic and antioxidative potential of Psidium guajava fruit in streptozotocin-induced diabetic rats. Food Chem. Toxicol. 2011, 49, 2189–2195. [Google Scholar] [CrossRef] [PubMed]
- Mattietto, R.; Matta, V. Cajá (Spondias mombin L.). In Postharvest Biology and Technology of Tropical and Subtropical Fruits; Elsevier: Amsterdam, The Netherlands, 2011; pp. 330–353e. [Google Scholar]
- Nkanu, E.E.; Jeje, S.O.; Ikpi, D.E.; Ujong, G.O. In vivo hypolipidemic and hypoglycemic effects of aqueous extract of Spondias mombin leaves and detoxification of reactive oxygen species in alloxan-induced diabetic rats. Int. J. Biol. Chem. Sci. 2016, 10, 1573–1579. [Google Scholar] [CrossRef][Green Version]
- Ojo, O.A.; Afon, A.A.; Ojo, A.B.; Ajiboye, B.O.; Oyinloye, B.E.; Kappo, A.P. Inhibitory effects of solvent-partitioned fractions of two nigerian herbs (spondias mombin linn. and mangifera indica L.) on α-amylase and α-glucosidase. Antioxidants 2018, 7, 73. [Google Scholar] [CrossRef] [PubMed]
- Saronee, F.; Bekinbo, M.; Ojeka, S.; Dapper, D. Comparative assessment of methanolic extracts of hog plum (Spondias mombin linn.) leaves and turmeric (Curcuma longa L.) rhizomes on blood glucose and glycosylated haemoglobin in male wistar rats. J. Appl. Sci. Environ. Manag. 2019, 23, 1631–1636. [Google Scholar] [CrossRef]
- Hasan, M.R.; Islam, M.N.; Islam, M.R. Phytochemistry, pharmacological activities and traditional uses of Emblica officinalis: A review. Int. Curr. Pharm. J. 2016, 5, 14–21. [Google Scholar] [CrossRef]
- Krishnaveni, M.; Mirunalini, S.; Karthishwaran, K.; Dhamodharan, G. Antidiabetic and antihyperlipidemic properties of Phyllanthus emblica Linn. (Euphorbiaceae) on streptozotocin induced diabetic rats. Pak. J. Nutr. 2010, 9, 43–51. [Google Scholar] [CrossRef]
- Bashir, A.; Mushtaq, A.; Mehboob, T. Evaluation of antioxidant and Antidiabetic activity of Phyllanthus emblica (fruit). Biol. Pak. 2018, 64, 85–91. [Google Scholar]
- Yang, B.; Liu, P. Composition and biological activities of hydrolyzable tannins of fruits of Phyllanthus emblica. J. Agric. Food Chem. 2014, 62, 529–541. [Google Scholar] [CrossRef]
- Sultana, Z.; Jami, S.I.; Ali, E.; Begum, M.; Haque, M. Investigation of antidiabetic effect of ethanolic extract of Phyllanthus emblica Linn. fruits in experimental animal models. Pharmacol. Pharm. 2014, 5, 11–18. [Google Scholar] [CrossRef][Green Version]
- Fatima, N.; Hafizur, R.M.; Hameed, A.; Ahmed, S.; Nisar, M.; Kabir, N. Ellagic acid in Emblica officinalis exerts anti-diabetic activity through the action on β-cells of pancreas. Eur. J. Nutr. 2017, 56, 591–601. [Google Scholar] [CrossRef]
- Tirgar, P.; Shah, K.; Patel, V.; Desai, T.; Goyal, R. Investigation into mechanism of action of anti-diabetic activity of Emblica officinalis on streptozotocin induced type I diabetic rat. Res. J. Pharm. Biol. Chem. Sci. 2010, 1, 672–682. [Google Scholar]
- Nain, P.; Saini, V.; Sharma, S.; Nain, J. Antidiabetic and antioxidant potential of Emblica officinalis Gaertn. leaves extract in streptozotocin-induced type-2 diabetes mellitus (T2DM) rats. J. Ethnopharmacol. 2012, 142, 65–71. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, J.J.; D’Souza, P.P.; Fazal, F.; Kumar, A.; Bhat, H.P.; Baliga, M.S. Anti-diabetic effects of the Indian indigenous fruit Emblica officinalis Gaertn: Active constituents and modes of action. Food Funct. 2014, 5, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Mahomoodally, M.F.; Sookhy, V. Ethnobotany and pharmacological uses of Elaeocarpus floribundus Blume (Elaeocarpaceae). In Plant and Human Health; Springer: Berlin/Heidelberg, Germany, 2018; Volume 1, pp. 125–137. [Google Scholar]
- Prasannan, P.; Jeyaram, Y.; Pandian, A.; Raju, R.; Sekar, S. A Review on Taxonomy, Phytochemistry, Pharmacology, Threats and Conservation of Elaeocarpus L. (Elaeocarpaceae). Bot. Rev. 2020, 86, 298–328. [Google Scholar] [CrossRef]
- Zaman, S. Exploring the antibacterial and antioxidant activities of Elaeocarpus floribundus leaves. Indo Am. J. Pharm. Sci. 2016, 3, 92–97. [Google Scholar]
- Okselni, T.; Santoni, A.; Dharma, A.; Efdi, M. Biological activity of methanol extract of elaeocarpus mastersii king: Antioxidant, antibacterial, and α-glucosidase inhibitor. Rasāyan J. Chem. 2019, 12, 146–151. [Google Scholar] [CrossRef]
- Deshpande, M.; Shengule, S.; Apte, K.G.; Wani, M.; Piprode, V.; Parab, P. Anti-obesity activity of Ziziphus mauritiana: A potent pancreatic lipase inhibitor. Asian J. Pharm. Clin. Res. 2013, 6, 168–173. [Google Scholar]
- Goyal, M.; Nagori, B.P.; Sasmal, D. Review on ethnomedicinal uses, pharmacological activity and phytochemical constituents of Ziziphus mauritiana (Z. jujuba Lam., non Mill). Spatula DD 2012, 2, 107–116. [Google Scholar] [CrossRef]
- Patel, D.; Kumar, R.; Laloo, D.; Hemalatha, S. Diabetes mellitus: An overview on its pharmacological aspects and reported medicinal plants having antidiabetic activity. Asian Pac. J. Trop. Biomed. 2012, 2, 411–420. [Google Scholar] [CrossRef]
- Bhatia, A.; Mishra, T. Hypoglycemic activity of Ziziphus mauritiana aqueous ethanol seed extract in alloxan-induced diabetic mice. Pharm. Biol. 2010, 48, 604–610. [Google Scholar] [CrossRef]
- Kavatagimath, S.A.; Jalalpure, S.S. Screening of ethanolic extract of Diospyros malabarica Desr. Bark for anti-diabetic and antioxidant potential. Indian J. Pharm. Educ. Res. 2016, 50, 179–189. [Google Scholar] [CrossRef]
- Sinha, B.; Bansal, S. A review of phytochemical and biological studies of Diospyros species used in folklore medicine of Jharkhand. J. Nat. Remedies 2008, 8, 11–17. [Google Scholar]
- Shubhra, R.D.; Polash, S.A.; Saha, T.; Hasan, A.; Hossain, S.; Islam, Z.; Sarker, S.R. Investigation of the Phytoconstituents and Antioxidant Activity of Diospyros malabarica Fruit Extracts. Adv. Biosci. Biotechnol. 2019, 10, 431–454. [Google Scholar] [CrossRef][Green Version]
- Kaushik, V.; Saini, V.; Pandurangan, A.; Khosa, R.L.; Parcha, V. A review of phytochemical and biological studies of Diospyros malabarica. Int. J. Pharm. Sci. Lett. 2013, 2, 167–169. [Google Scholar]
- Mondal, S.K.; Chakraborty, G.; Bhaumik, U.K.; Gupta, M.; Mazumder, U.K. Antidiabetic activity of Diospyros malabarica Kostel bark: A preliminary investigation for possible mode of action. Orient. Pharm. Exp. Med. 2008, 8, 236–242. [Google Scholar] [CrossRef]
- Zulcafli, A.S.; Lim, C.; Ling, A.P.; Chye, S.; Koh, R. Focus: Plant-based Medicine and Pharmacology: Antidiabetic Potential of Syzygium sp.: An Overview. Yale J. Biol. Med. 2020, 93, 307. [Google Scholar] [PubMed]
- Ranasinghe, R.A.S.N.; Maduwanthi, S.D.T.; Marapana, R.A.U.J. Nutritional and health benefits of jackfruit (Artocarpus heterophyllus Lam.): A review. Int. J. Food Sci. 2019, 2019, 4327183. [Google Scholar] [CrossRef]
- Baliga, M.S.; Shivashankara, A.R.; Haniadka, R.; Dsouza, J.; Bhat, H.P. Phytochemistry, nutritional and pharmacological properties of Artocarpus heterophyllus Lam (jackfruit): A review. Food Res. Int. 2011, 44, 1800–1811. [Google Scholar] [CrossRef]
- Chackrewarthy, S.; Thabrew, M.; Weerasuriya, M.; Jayasekera, S. Evaluation of the hypoglycemic and hypolipidemic effects of an ethylacetate fraction of Artocarpus heterophyllus (jak) leaves in streptozotocin-induced diabetic rats. Pharm. Mag 2010, 6, 186. [Google Scholar] [CrossRef]
- Biworo, A.; Tanjung, E.; Iskandar, K.; Suhartono, E. Antidiabetic and antioxidant activity of jackfruit (Artocarpus heterophyllus) extract. J. Med. Bioeng. 2015, 4, 318–323. [Google Scholar] [CrossRef]
- Khandaker, M.M.; Mat, N.; Boyce, A.N. Bioactive constituents, antioxidant and antimicrobial activities of three cultivars of wax apple (Syzygium samarangense L.) fruits. Res. J. Biotechnol. 2015, 10, 1. [Google Scholar]
- Khamchan, A.; Paseephol, T.; Hanchang, W. Protective effect of wax apple (Syzygium samarangense (Blume) Merr. & LM Perry) against streptozotocin-induced pancreatic ß-cell damage in diabetic rats. Biomed. Pharmacother. 2018, 108, 634–645. [Google Scholar] [PubMed]
- Shahreen, S.; Banik, J.; Hafiz, A.; Rahman, S.; Zaman, A.T.; Shoyeb, A.; Chowdhury, M.H.; Rahmatullah, M. Antihyperglycemic activities of leaves of three edible fruit plants (Averrhoa carambola, Ficus hispida and Syzygium samarangense) of Bangladesh. Afr. J. Tradit. Complement. Altern. Med. 2012, 9, 287–291. [Google Scholar] [CrossRef]
- Hu, Y.-K.; Wang, L.; Wang, J.-H.; Li, M.-J.; Li, F.; Yang, J.; Zhao, Y. Resorcinol derivatives with α-glucosidase inhibitory activities from Syzygium samarangense. Nat. Prod. Res. 2020, 35, 5948–5953. [Google Scholar] [CrossRef] [PubMed]
- Shen, S.-C.; Chang, W.-C.; Chang, C.-L. An extract from wax apple (Syzygium samarangense (Blume) Merrill and Perry) effects glycogenesis and glycolysis pathways in tumor necrosis factor-α-treated FL83B mouse hepatocytes. Nutrients 2013, 5, 455–467. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, S.Y.; Ruknuddin, G.; Prajapati, P. Ethno medicinal values of Citrus genus: A review. Med. J. Dr. DY Patil Univ. 2016, 9, 560. [Google Scholar] [CrossRef]
- Chriscensia, E.; Wibowo, E.C.; Enriko, G.; Wijaya, O.C.; Sahamastuti, A.A.T. Phytochemical Screening, Therapeutic Benefits, and Adverse Effects of Citrus aurantifolia–A Review. Indones. J. Life Sci. 2020, 2, 56–69. [Google Scholar] [CrossRef]
- Kasia Benedicta, E. Hypoglycaemic Effects of Decoction of Camelia sinensis (Lipton Tea) and Citrus aurantifolia (Lime) on Plasma Glucose Concentration and Weight of Normal Albino Rats. Sch. Int. J. Biochem. 2021, 4, 20–25. [Google Scholar] [CrossRef]
- Ibrahim, F.A.; Usman, L.A.; Akolade, J.O.; Idowu, O.A.; Abdulazeez, A.T.; Amuzat, A.O. Antidiabetic potentials of Citrus aurantifolia leaf essential oil. Drug Res. 2019, 69, 201–206. [Google Scholar] [CrossRef]
- Şeker Karatoprak, G.; Yücel Aşık, Ç.; Çakır, A.; Köngül Şafak, E. In vitro pharmacological screening of antioxidant, cytotoxic and enzyme inhibitory activities of Citrus aurantifolia Linn. Dried fruit extract. Int. J. Environ. Health Res. 2020, 31, 991–1000. [Google Scholar] [CrossRef]
- Mawarti, H.; Khotimah, M.Z.A.a.; Rajin, M. Ameliorative effect of Citrus aurantifolia and Cinnamomum burmannii extracts on diabetic complications in a hyperglycemic rat model. Trop. J. Pharm. Res. 2018, 17, 823–829. [Google Scholar] [CrossRef]
- Ramya, S.; Narayanan, V.; Ponnerulan, B.; Saminathan, E.; Veeranan, U. Potential of peel extracts of Punica granatum and Citrus aurantifolia on alloxan-induced diabetic rats. Beni-Suef Univ. J. Basic Appl. Sci. 2020, 9, 1–11. [Google Scholar] [CrossRef]
- Mohanapriya, M.; Ramaswamy, L.; Rajendran, R. Health and medicinal properties of lemon (Citrus limonum). Int. J. Ayurvedic Herb. Med. 2013, 3, 1095–1100. [Google Scholar]
- Klimek-Szczykutowicz, M.; Szopa, A.; Ekiert, H. Citrus limon (Lemon) phenomenon—A review of the chemistry, pharmacological properties, applications in the modern pharmaceutical, food, and cosmetics industries, and biotechnological studies. Plants 2020, 9, 119. [Google Scholar] [CrossRef]
- Naim, M.; Amjad, F.M.; Sultana, S.; Islam, S.N.; Hossain, M.A.; Begum, R.; Rashid, M.A.; Amran, M.S. Comparative study of antidiabetic activity of hexane-extract of lemon peel (Limon citrus) and glimepiride in alloxan-induced diabetic rats. Bangladesh Pharm. J. 2012, 15, 131–134. [Google Scholar] [CrossRef]
- Sivadasan, S.; Subathra, M.; Padmanabhan, D.; Maharani, R.; Jayalakshmi, M.; Amudha, P. An overview of phytochemical and pharmacological potential of Baccaurea species. J. Crit. Rev. 2020, 7, 2354–2362. [Google Scholar]
- Nurmayani, S.; Widodo, W.; Matra, D. Characterization of rambai (Baccaurea motleyana) genes putatively involved in sugar metabolism. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Bogor, Indonesia, 24–25 September 2020; Volume 694, p. 012067. [Google Scholar]
- Prodhan, A.S.U.; Mridu, F.S. Baccaurea motleyana (Rambai): Nutritional, phytochemical, and medicinal overview. Adv. Tradit. Med. 2021, 1–25. [Google Scholar] [CrossRef]
- Anjum, J.; Lone, R.; Wani, K.A. Lychee (Litchi chinensis): Biochemistry, Panacea, and Nutritional Value. In Lychee Disease Management; Springer: Berlin/Heidelberg, Germany, 2017; pp. 237–256. [Google Scholar]
- Upadhyaya, D.C.; Upadhyaya, C.P. Bioactive Compounds and Medicinal Importance of Litchi chinensis. In The Lychee Biotechnology; Springer: Berlin/Heidelberg, Germany, 2017; pp. 333–361. [Google Scholar]
- Lim, T. Litchi chinensis. In Edible Medicinal And Non-Medicinal Plants; Springer: Berlin/Heidelberg, Germany, 2013; pp. 45–58. [Google Scholar]
- Ibrahim, S.R.; Mohamed, G.A. Litchi chinensis: Medicinal uses, phytochemistry, and pharmacology. J. Ethnopharmacol. 2015, 174, 492–513. [Google Scholar] [CrossRef]
- Choi, S.-A.; Lee, J.E.; Kyung, M.J.; Youn, J.H.; Oh, J.B.; Whang, W.K. Anti-diabetic functional food with wasted litchi seed and standard of quality control. Appl. Biol. Chem. 2017, 60, 197–204. [Google Scholar] [CrossRef]
- Koul, B.; Singh, J. Lychee biology and biotechnology. In The Lychee Biotechnology; Springer: Berlin/Heidelberg, Germany, 2017; pp. 137–192. [Google Scholar]
- Shah, K.; Patel, M.; Patel, R.; Parmar, P. Mangifera indica (mango). Pharmacogn. Rev. 2010, 4, 42. [Google Scholar] [CrossRef]
- Parvez, G.M. Pharmacological activities of mango (Mangifera indica): A review. J. Pharmacogn. Phytochem. 2016, 5, 1. [Google Scholar]
- De la Luz Cádiz-Gurrea, M.; del Carmen Villegas-Aguilar, M.; Leyva-Jiménez, F.J.; Pimentel-Moral, S.; Fernández-Ochoa, Á.; Alañón, M.E.; Segura-Carretero, A. Revalorization of bioactive compounds from tropical fruit by-products and industrial applications by means of sustainable approaches. Food Res. Int. 2020, 138, 109786. [Google Scholar] [CrossRef] [PubMed]
- Ganogpichayagrai, A.; Palanuvej, C.; Ruangrungsi, N. Antidiabetic and anticancer activities of Mangifera indica cv. Okrong leaves. J. Adv. Pharm. Technol. Res. 2017, 8, 19. [Google Scholar] [PubMed]
- Narender, T.; Shweta, S.; Tiwari, P.; Reddy, K.P.; Khaliq, T.; Prathipati, P.; Puri, A.; Srivastava, A.; Chander, R.; Agarwal, S. Antihyperglycemic and antidyslipidemic agent from Aegle marmelos. Bioorg. Med. Chem. Lett. 2007, 17, 1808–1811. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, D.S. Neuroprotective effect of Cucumis melo Var. flexuosus leaf extract on the brains of rats with streptozotocin-induced diabetes. Metab. Brain Dis. 2017, 32, 69–75. [Google Scholar] [CrossRef]
- Chen, L.; Kang, Y.-H.; Suh, J.-K. Roasting processed oriental melon (Cucumis melo L. var. makuwa Makino) seed influenced the triglyceride profile and the inhibitory potential against key enzymes relevant for hyperglycemia. Food Res. Int. 2014, 56, 236–242. [Google Scholar] [CrossRef]
- Mandal, S.; Mandal, M. Tangerine (Citrus reticulata L. var.) oils. In Essential Oils in Food Preservation, Flavor and Safety; Elsevier: Amsterdam, The Netherlands, 2016; pp. 803–811. [Google Scholar]
- Ali, A.M.; Gabbar, M.A.; Abdel-Twab, S.M.; Fahmy, E.M.; Ebaid, H.; Alhazza, I.M.; Ahmed, O.M. Antidiabetic potency, antioxidant effects, and mode of actions of Citrus reticulata fruit peel hydroethanolic extract, hesperidin, and quercetin in nicotinamide/streptozotocin-induced wistar diabetic rats. Oxidative Med. Cell. Longev. 2020, 2020, 1730492. [Google Scholar] [CrossRef]
- Ghauri, S.; Raza, S.Q.; Imran, M.; Saeed, S.; Rashid, M.; Naseer, R. Assessment of α-amylase and α-glucosidase inhibitory potential of Citrus reticulata peel extracts in hyperglycemic/hypoglycemic rats. 3 Biotech 2021, 11, 167. [Google Scholar] [CrossRef]
- Mehmood, F.; Aurangzeb, M.; Manzoor, F.; Fazal, S. A Comparative Study of in vitro Total Antioxidant Capacity, in vivo Antidiabetic and Antimicrobial Activity of Essential Oils from Leaves and Seeds of Zanthoxylum armatum DC. Asian J. Chem. 2013, 25, 10221. [Google Scholar] [CrossRef]
- Rahmani, A.H.; Aldebasi, Y.H. Potential role of carica papaya and their active constituents in the prevention and treatment of diseases. Int. J. Pharm. Pharm. Sci. 2016, 8, 11–15. [Google Scholar]
- Nafiu, A.B.; Alli-Oluwafuyi, A.-m.; Haleemat, A.; Olalekan, I.S.; Rahman, M.T. Papaya (Carica papaya L., Pawpaw). In Nonvitamin and Nonmineral Nutritional Supplements; Elsevier: Amsterdam, The Netherlands, 2019; pp. 335–359. [Google Scholar]
- Abd Elgadir, M.; Salama, M.; Adam, A. Carica papaya as a source of natural medicine and its utilization in selected pharmacetical applications. Int. J. Pharm. Sci. 2014, 6, 868–871. [Google Scholar]
- Singh, S.; Bajpai, M.; Mishra, P. Carissa carandas L.–phyto-pharmacological review. J. Pharm. Pharmacol. 2020, 72, 1694–1714. [Google Scholar] [CrossRef] [PubMed]
- Agada, R.; Thagriki, D.; Lydia, D.E.; Khusro, A.; Alkahtani, J.; Al Shaqha, M.M.; Alwahibi, M.S.; Elshikh, M.S. Antioxidant and anti-diabetic activities of bioactive fractions of Carica papaya seeds extract. J. King Saud Univ.-Sci. 2021, 33, 101342. [Google Scholar] [CrossRef]
- Maniyar, Y.; Bhixavatimath, P. Antihyperglycemic and hypolipidemic activities of aqueous extract of Carica papaya Linn. leaves in alloxan-induced diabetic rats. J. Ayurveda Integr. Med. 2012, 3, 70. [Google Scholar] [CrossRef]
- Juárez-Rojop, I.E.; Díaz-Zagoya, J.C.; Ble-Castillo, J.L.; Miranda-Osorio, P.H.; Castell-Rodríguez, A.E.; Tovilla-Zárate, C.A.; Rodríguez-Hernández, A.; Aguilar-Mariscal, H.; Ramón-Frías, T.; Bermúdez-Ocaña, D.Y. Hypoglycemic effect of Carica papaya leaves in streptozotocin-induced diabetic rats. BMC Complement. Altern. Med. 2012, 12, 236. [Google Scholar] [CrossRef]
- Priyadarshi, A.; Ram, B. A review on Pharmacognosy, phytochemistry and pharmacological activity of Carica papaya (Linn.) leaf. Int. J. Pharm. Sci. Res. 2018, 9, 4071–4078. [Google Scholar]
- Rahman, I.A.; Mohamed, E.; Camalxaman, S.N.; Haron, N.; Rambely, A.S. Ananas comosus (L.) Merr.: A mini review of its therapeutic properties: Medicinal benefits of pineapple plant. Health Scope 2020, 3, 54–59. [Google Scholar]
- Faisal, M.; Hossa, F.M.M.; Rahman, S.; Bashar, A.; Hossan, S.; Rahmatullah, M. Effect of methanolic extract of Ananas comosus Leaves on glucose tolerance and acetic acid induced pain in Swiss albino mice. World J. Pharm. Res 2014, 3, 24–34. [Google Scholar]
- Xie, W.; Xing, D.; Sun, H.; Wang, W.; Ding, Y.; Du, L. The effects of Ananas comosus L. leaves on diabetic-dyslipidemic rats induced by alloxan and a high-fat/high-cholesterol diet. Am. J. Chin. Med. 2005, 33, 95–105. [Google Scholar] [CrossRef]
- Vijayalakshmi, P.; Radha, R. Pharmacognostical and Phytochemical Screening of the Peels of Citrus maxima. Res. J. Pharmacogn. Phytochem. 2016, 8, 25–31. [Google Scholar]
- Abirami, A.; Nagarani, G.; Siddhuraju, P. In vitro antioxidant, anti-diabetic, cholinesterase and tyrosinase inhibitory potential of fresh juice from Citrus hystrix and C. maxima fruits. Food Sci. Hum. Wellness 2014, 3, 16–25. [Google Scholar] [CrossRef]
- KunduSen, S.; Gupta, M.; Mazumder, U.K.; Haldar, P.K.; Saha, P.; Bhattacharya, S.; Kar, B.; Bala, A. Antihyperglycemic effect and antioxidant property of Citrus maxima leaf in streptozotocin-induced diabetic rats. Diabetol. Croat. 2011, 40, 113–120. [Google Scholar]
- Ani, P.N.; Ochu, K.E. Anti-diabetic, anti-hyperlipidemic and hepatoprotective potential of shaddock (Citrus maxima) peel extract. Acta Sci. Pol. Technol. Aliment. 2020, 19, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Oyedepot, A.; Babarinde, S. Effects of Shaddock (Citrus maxima) fruit juice on glucose tolerance and lipid profile in type-II Diabetic Rats. Chem. Sci. Trans. 2013, 2, 19–24. [Google Scholar] [CrossRef][Green Version]
- Ali Redha, A.; Hasan, A.; Mandeel, Q. Phytochemical determinations of Pomegranate (Punica granatum) Rind and Aril extracts and their antioxidant, antidiabetic and antibacterial activity. Nat. Prod. Chem. Res. 2018, 6, 4. [Google Scholar] [CrossRef]
- Bagri, P.; Ali, M.; Aeri, V.; Bhowmik, M.; Sultana, S. Antidiabetic effect of Punica granatum flowers: Effect on hyperlipidemia, pancreatic cells lipid peroxidation and antioxidant enzymes in experimental diabetes. Food Chem. Toxicol. 2009, 47, 50–54. [Google Scholar] [CrossRef] [PubMed]
- Gharib, E.; Kouhsari, S.M. Study of the antidiabetic activity of Punica granatum L. fruits aqueous extract on the alloxan-diabetic wistar rats. Iran. J. Pharm. Res. 2019, 18, 358. [Google Scholar] [PubMed]
- Barathikannan, K.; Venkatadri, B.; Khusro, A.; Al-Dhabi, N.A.; Agastian, P.; Arasu, M.V.; Choi, H.S.; Kim, Y.O. Chemical analysis of Punica granatum fruit peel and its in vitro and in vivo biological properties. BMC Complement. Altern. Med. 2016, 16, 264. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.H.; Peng, G.; Kota, B.P.; Li, G.Q.; Yamahara, J.; Roufogalis, B.D.; Li, Y. Anti-diabetic action of Punica granatum flower extract: Activation of PPAR-γ and identification of an active component. Toxicol. Appl. Pharmacol. 2005, 207, 160–169. [Google Scholar] [CrossRef]
- Jain, V.; Viswanatha, G.L.; Manohar, D.; Shivaprasad, H. Isolation of antidiabetic principle from fruit rinds of Punica granatum. Evid.-Based Complement. Altern. Med. 2012, 2012, 147202. [Google Scholar] [CrossRef]
- Bano, M.; Ahmed, B. Manilkara zapota (L.) P. Royen (Sapodilla): A review. Int. J. Adv. Res. Ideas Innov. Technol. 2017, 3, 1364–1371. [Google Scholar]
- Barbalho, S.M.; Bueno, P.C.d.S.; Delazari, D.S.; Guiguer, E.L.; Coqueiro, D.P.; Araújo, A.C.; de Souza, M.d.S.S.; Farinazzi-Machado, F.M.; Mendes, C.G.; Groppo, M. Antidiabetic and antilipidemic effects of Manilkara zapota. J. Med. Food 2015, 18, 385–391. [Google Scholar] [CrossRef]
- Sathishkumar, T.; Anitha, S.; Sharon, R.E.; Santhi, V.; Sukanya, M.; Kumaraesan, K.; Rapheal, V.S. Evaluation of In Vitro Invertase Inhibitory Activity of M anilkara zapota Seeds—A Novel Strategy to Manage Diabetes Mellitus. J. Food Biochem. 2015, 39, 517–527. [Google Scholar] [CrossRef]
- Fayek, N.M.; Monem, A.R.A.; Mossa, M.Y.; Meselhy, M.R.; Shazly, A.H. Chemical and biological study of Manilkara zapota (L.) Van Royen leaves (Sapotaceae) cultivated in Egypt. Pharmacogn. Res. 2012, 4, 85. [Google Scholar]
- Paul, S.R.; Hakim, M.L. In vivo hypoglycemic study of Manilkara zapota leaf and seed extracts. Bangladesh J. Pharmacol. 2015, 10, 246–250. [Google Scholar] [CrossRef][Green Version]
- Muthu, N.; Lee, S.Y.; Phua, K.K.; Bhore, S.J. Nutritional, medicinal and toxicological attributes of star-fruits (Averrhoa carambola L.): A review. Bioinformation 2016, 12, 420. [Google Scholar] [CrossRef]
- Lakmal, K.; Yasawardene, P.; Jayarajah, U.; Seneviratne, S.L. Nutritional and medicinal properties of Star fruit (Averrhoa carambola): A review. Food Sci. Nutr. 2021, 9, 1810–1823. [Google Scholar] [CrossRef]
- Islam, S.; Alam, M.B.; Ahmed, A.; Lee, S.; Lee, S.-H.; Kim, S. Identification of secondary metabolites in Averrhoa carambola L. bark by high-resolution mass spectrometry and evaluation for α-glucosidase, tyrosinase, elastase, and antioxidant potential. Food Chem. 2020, 332, 127377. [Google Scholar] [CrossRef]
- Xu, X.; Liang, T.; Wen, Q.; Lin, X.; Tang, J.; Zuo, Q.; Tao, L.; Xuan, F.; Huang, R. Protective effects of total extracts of Averrhoa carambola L. (Oxalidaceae) roots on streptozotocin-induced diabetic mice. Cell. Physiol. Biochem. 2014, 33, 1272–1282. [Google Scholar] [CrossRef]
- Pham, H.T.T.; Huang, W.; Han, C.; Li, J.; Xie, Q.; Wei, J.; Xu, X.; Lai, Z.; Huang, X.; Huang, R. Effects of Averrhoa carambola L. (Oxalidaceae) juice mediated on hyperglycemia, hyperlipidemia, and its influence on regulatory protein expression in the injured kidneys of streptozotocin-induced diabetic mice. Am. J. Transl. Res. 2017, 9, 36. [Google Scholar]
- Panda, S.; Kar, A. Antidiabetic and antioxidative effects of Annona squamosa leaves are possibly mediated through quercetin-3-O-glucoside. Biofactors 2007, 31, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Pandey, N.; Barve, D. Phytochemical and pharmacological review on Annona squamosa Linn. Int. J. Res. Pharm. Biomed. Sci. 2011, 2, 1404–1412. [Google Scholar]
- Tomar, R.S.; Sisodia, S.S. Antidiabetic activity of Annona squamosa L. in experimental induced diabetic rats. Int. J. Pharm. Biol. Arch. 2012, 3, 1492–1495. [Google Scholar]
- Marahatta, A.B.; Aryal, A.; Basnyat, R.C.; Marahatta, C.A.B. The phytochemical and nutritional analysis and biological activity of Annona squamosa Linn. Int. J. Herb. Med. 2019, 7, 19–28. [Google Scholar]
- Davis, J.A.; Sharma, S.; Mittra, S.; Sujatha, S.; Kanaujia, A.; Shukla, G.; Katiyar, C.; Lakshmi, B.; Bansal, V.S.; Bhatnagar, P.K. Antihyperglycemic effect of Annona squamosa hexane extract in type 2 diabetes animal model: PTP1B inhibition, a possible mechanism of action? Indian J. Pharm. 2012, 44, 326. [Google Scholar] [CrossRef] [PubMed]
- Sangala, R.; Kodati, D.; Burra, S.; Gopu, J.; Dubasi, A. Evaluation of antidiabetic activity of Annona squamosa Linn Seed in alloxan–induced diabetic rats. Diabetes 2011, 2, 100–106. [Google Scholar]
- Jibril, M.M.; Abdul-Hamid, A.; Ghazali, H.M.; Dek, M.S.P.; Ramli, N.S.; Jaafar, A.H.; Karrupan, J.; Mohammed, A.S. Antidiabetic antioxidant and phytochemical profile of yellow-fleshed seeded watermelon (Citrullus lanatus) extracts. J. Food Nutr. Res. 2019, 7, 82–95. [Google Scholar]
- Erhirhie, E.; Ekene, N. Medicinal values on Citrullus lanatus (watermelon): Pharmacological review. Int. J. Res. Pharm. Biomed. Sci. 2013, 4, 1305–1312. [Google Scholar]
- Ajiboye, B.O.; Shonibare, M.T.; Oyinloye, B.E. Antidiabetic activity of watermelon (Citrullus lanatus) juice in alloxan-induced diabetic rats. J. Diabetes Metab. Disord. 2020, 19, 343–352. [Google Scholar] [CrossRef]
- Arise, R.O.; Yekeen, A.A.; Ekun, O.E. In vitro antioxidant and α-amylase inhibitory properties of watermelon seed protein hydrolysates. Environ. Exp. Biol. 2016, 14, 163–172. [Google Scholar] [CrossRef]
- Rahman, S.S.; Yasmin, N.; Kamruzzaman, M.; Islam, M.R.; Karim, M.R.; Rouf, S.M. Anti-hyperglycemic effect of the immature endosperm of sugar palm (Borassus flabellifer) fruit on type 2 diabetes mellitus patients—A case study. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 1317–1322. [Google Scholar] [CrossRef] [PubMed]
- Akter, M.; Islam, K.S.; Sarkar, U.R.; Hossien, M.M.; Akter, S.; Tondra, S.F.S.; Hossen, M.I.; Hasan, M.N. Investigation of anti-diabetic properties of Borassus flabellifer L. (roots) on type-2 diabetic rats. Pharmacologyonline 2020, 1, 105–112. [Google Scholar]
- Kavatagimath, S.A.; Jalalpure, S.S.; Hiremath, R.D. Screening of ethanolic extract of Borassus flabellifer flowers for its antidiabetic and antioxidant potential. J. Nat. Remedies 2016, 16, 22–32. [Google Scholar] [CrossRef]
- Duraipandiyan, V.; Balamurugan, R.; Al-Dhabi, N.A.; Raja, T.W.; Ganesan, P.; Ahilan, B.; Arasu, M.V.; Ignacimuthu, S.; Esmail, G.A. The down regulation of PTP1B expression and attenuation of disturbed glucose and lipid metabolism using Borassus flabellifer (L) fruit methanol extract in high fat diet and streptozotocin induced diabetic rats. Saudi J. Biol. Sci. 2020, 27, 433–440. [Google Scholar] [CrossRef]
- Bhadoriya, S.S.; Ganeshpurkar, A.; Narwaria, J.; Rai, G.; Jain, A.P. Tamarindus indica: Extent of explored potential. Pharmacogn. Rev. 2011, 5, 73. [Google Scholar] [PubMed]
- Menezes, A.P.P.; Trevisan, S.C.C.; Barbalho, S.M.; Guiguer, E.L. Tamarindus indica L. A plant with multiple medicinal purposes. J. Pharmacogn. Phytochem. 2016, 5, 50. [Google Scholar]
- Krishna, R.N.; Anitha, R.; Ezhilarasan, D. Aqueous extract of Tamarindus indica fruit pulp exhibits antihyperglycaemic activity. Avicenna J. Phytomed. 2020, 10, 440. [Google Scholar] [PubMed]
- Bhutkar, M.A.; Bhinge, S.D.; Randive, D.S.; Wadkar, G.H. Hypoglycemic effects of Berberis aristata and Tamarindus indica extracts in vitro. Bull. Fac. Pharm. Cairo Univ. 2017, 55, 91–94. [Google Scholar] [CrossRef][Green Version]
- Nahar, L.; Nasrin, F.; Zahan, R.; Haque, A.; Haque, E.; Mosaddik, A. Comparative study of antidiabetic activity of Cajanus cajan and Tamarindus indica in alloxan-induced diabetic mice with a reference to in vitro antioxidant activity. Pharmacogn. Res. 2014, 6, 180. [Google Scholar]
- Agnihotri, A.; Singh, V. Effect of Tamarindus indica Linn. and Cassia fistula Linn. stem bark extracts on oxidative stress and diabetic conditions. Acta Pol. Pharm. 2013, 70, 1011–1019. [Google Scholar]
- Koyagura, N.; Jamadar, M.; Huilgol, S.V.; Nayak, N.; Yendigeri, S.M.; Shamsuddin, M. Antidiabetic and hepatoprotective activities of Tamarindus indica fruit pulp in alloxan induced diabetic rats. Int. J. Pharmacol. Clin. Sci. 2013, 2. [Google Scholar]
- Kumawat, B.K.; Chand, T.; Singh, Y. Antidiabetic and antihyperlipidemic effects of alcoholic and aqueous stem bark extracts of Limonia acidissima, linn in alloxan induced diabetic rats. Int. J. Phytomed. 2012, 4, 187. [Google Scholar]
- Bhavsar, S.; Sapra, P.; Maitreya, B.; Mankad, A.U. A Review on Potential of Medicinal Plant: Limonia acidissima L. Int. Assoc. Biol. Comput. Dig. 2019, 1, 159–165. [Google Scholar] [CrossRef]
- Murthy, H.N.; Dalawai, D. Bioactive Compounds of Wood Apple (Limonia acidissima L.). In Bioactive Compounds in Underutilized Fruits and Nuts; Springer: Cham, Switzerland, 2020; pp. 543–569. [Google Scholar]
- Priya, E.M.; Gothandam, K.; Karthikeyan, S. Antidiabetic activity of Feronia limonia and Artocarpus heterophyllus in streptozotocin induced diabetic rats. Am. J. Food Technol. 2012, 7, 43–49. [Google Scholar] [CrossRef][Green Version]
- Reddy, G.J.; Reddy, K.B.; Reddy, G. In vitro Feronia nhibitory xtracts of α-Amylase and α-Glucosidase i activity of e elephantum Paspalum scrobiculatum f G ruit and rains. Asian J. Pharm. Pharmacol. 2019, 5, 42–47. [Google Scholar] [CrossRef]
- Nigam, V.; Nambiar, V.S. Aegle marmelos leaf juice as a complementary therapy to control type 2 diabetes–Randomised controlled trial in Gujarat, India. Adv. Integr. Med. 2019, 6, 11–22. [Google Scholar] [CrossRef]
- Ansari, P.; Flatt, P.R.; Harriott, P.; Abdel-Wahab, Y.H. Evaluation of the antidiabetic and insulin releasing effects of A. squamosa, including isolation and characterization of active phytochemicals. Plants 2020, 9, 1348. [Google Scholar] [CrossRef]
- Perez, G.R.M.; Zavala, S.M.A.; Perez, G.S.; Perez, G.C. Antidiabetic effect of compounds isolated from plants. Phytomedicine 1998, 5, 55–75. [Google Scholar] [CrossRef]
- Mrabti, H.N.; Jaradat, N.; Fichtali, I.; Ouedrhiri, W.; Jodeh, S.; Ayesh, S.; Cherrah, Y.; Faouzi, M.E.A. Separation, identification, and antidiabetic activity of catechin isolated from Arbutus unedo L. Plant roots. Plants 2018, 7, 31. [Google Scholar] [CrossRef]
- Debnath, B.; Singh, W.S.; Manna, K. A phytopharmacological review on Ananas comosus. Adv. Tradit. Med. 2021, 1–8. [Google Scholar] [CrossRef]
- Kumar, S.; Kumar, V.; Prakash, O. Antidiabetic and antihyperlipidemic effects of Dillenia indica (L.) leaves extract. Braz. J. Pharm. Sci. 2011, 47, 373–378. [Google Scholar] [CrossRef]
- Variya, B.C.; Bakrania, A.K.; Patel, S.S. Antidiabetic potential of gallic acid from Emblica officinalis: Improved glucose transporters and insulin sensitivity through PPAR-γ and Akt signaling. Phytomedicine 2020, 73, 152906. [Google Scholar] [CrossRef] [PubMed]
- Amalan, V.; Vijayakumar, N.; Indumathi, D.; Ramakrishnan, A. Antidiabetic and antihyperlipidemic activity of p-coumaric acid in diabetic rats, role of pancreatic GLUT 2: In vivo approach. Biomed. Pharmacother. 2016, 84, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Narasimhan, A.; Chinnaiyan, M.; Karundevi, B. Ferulic acid exerts its antidiabetic effect by modulating insulin-signalling molecules in the liver of high-fat diet and fructose-induced type-2 diabetic adult male rat. Appl. Physiol. Nutr. Metab. 2015, 40, 769–781. [Google Scholar] [CrossRef]
- Eze, E.D. Hypoglycaemic effect of lycopene in streptozotocin-induced diabetic Wistar rats. Br. J. Med. Med. Res. 2015, 7, 762–770. [Google Scholar] [CrossRef]
- Lu, G.; Luo, X.; Liu, Z.; Yang, L.; Lin, C.; Xu, M. Protective effect of vanillin in streptozotocin-induced diabetes in neonatal rats via attenuation of oxidative stress and inflammation. Trop. J. Pharm. Res. 2019, 18, 349–355. [Google Scholar] [CrossRef]
- Prakash, O.; Kumar, R.; Mishra, A.; Gupta, R. Artocarpus heterophyllus (Jackfruit): An overview. Pharmacogn. Rev. 2009, 3, 353. [Google Scholar]
- Song, T.-J.; Park, C.-H.; In, K.-R.; Kim, J.-B.; Kim, J.H.; Kim, M.; Chang, H.J. Antidiabetic effects of betulinic acid mediated by the activation of the AMP-activated protein kinase pathway. PLoS ONE 2021, 16, e0249109. [Google Scholar] [CrossRef]
- Mlala, S.; Oyedeji, A.O.; Gondwe, M.; Oyedeji, O.O. Ursolic Acid and Its Derivatives as Bioactive Agents. Molecules 2019, 24, 2751. [Google Scholar] [CrossRef]
- Babu, S.; Jayaraman, S. An update on β-sitosterol: A potential herbal nutraceutical for diabetic management. Biomed. Pharmacother. 2020, 131, 110702. [Google Scholar] [CrossRef]
- More, T.A.; Kulkarni, B.R.; Nalawade, M.L.; Arvindekar, A.U. Antidiabetic activity of linalool and limonene in streptozotocin-induced diabetic rat: A combinatorial therapy approach. Int. J. Pharm. Pharm. Sci. 2014, 6, 159–163. [Google Scholar]
- Dej-adisai, S.; Pitakbut, T.; Wattanapiromsakul, C. Alpha-glucosidase inhibitory activity and phytochemical investigation of Borassus flabellifer Linn. Afr. J. Pharm. Pharmacol. 2017, 11, 45–52. [Google Scholar]
- Prakash, O.; Usmani, S.; Singh, R.; Singh, N.; Gupta, A.; Ved, A. A panoramic view on phytochemical, nutritional, and therapeutic attributes of Ziziphus mauritiana Lam.: A comprehensive review. Phytother. Res. 2021, 35, 63–77. [Google Scholar] [CrossRef] [PubMed]
- Bano, Z.; Begum, S.; Ali, S.S.; Kiran, Z.; Siddiqui, B.S.; Ahmed, A.; Khawaja, S.; Fatima, F.; Jabeen, A. Phytochemicals from Carissa carandas with potent cytotoxic and anti-inflammatory activities. Nat. Prod. Res. 2021, 36, 1587–1592. [Google Scholar] [CrossRef]
- Kim, J.N.; Han, S.N.; Kim, H.-K. Phytic acid and myo-inositol support adipocyte differentiation and improve insulin sensitivity in 3T3-L1 cells. Nutr. Res. 2014, 34, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, A.O.; Oliveira, Y.I.S.; Adjafre, B.L.; de Moraes, M.E.A.; Aragao, G.F. Pharmacological effects of the isomeric mixture of alpha and beta amyrin from Protium heptaphyllum: A literature review. Fundam. Clin. Pharmacol. 2019, 33, 4–12. [Google Scholar] [CrossRef] [PubMed]
- Azizi, S.; Mahdavi, R.; Vaghef-Mehrabany, E.; Maleki, V.; Karamzad, N.; Ebrahimi-Mameghani, M. Potential roles of Citrulline and watermelon extract on metabolic and inflammatory variables in diabetes mellitus, current evidence and future directions: A systematic review. Clin. Exp. Pharmacol. Physiol. 2020, 47, 187–198. [Google Scholar] [CrossRef]
- Singh, A.B.; Akanksha; Singh, N.; Maurya, R.; Srivastava, A.K. Anti-hyperglycaemic, lipid lowering and anti-oxidant properties of [6]-gingerol in db/db mice. Int. J. Med. Med. Sci. 2009, 1, 536–544. [Google Scholar]
- Ghosh, J.; Sil, P.C. Arjunolic acid: A new multifunctional therapeutic promise of alternative medicine. Biochimie 2013, 95, 1098–1109. [Google Scholar] [CrossRef]
- Gandhi, G.R.; Vasconcelos, A.B.S.; Wu, D.-T.; Li, H.-B.; Antony, P.J.; Li, H.; Geng, F.; Gurgel, R.Q.; Narain, N.; Gan, R.-Y. Citrus flavonoids as promising phytochemicals targeting diabetes and related complications: A systematic review of in vitro and in vivo studies. Nutrients 2020, 12, 2907. [Google Scholar] [CrossRef]
- Narang, N.; Jiraungkoorskul, W. Anticancer activity of key lime, Citrus aurantifolia. Pharmacogn. Rev. 2016, 10, 118. [Google Scholar] [PubMed]
- Ibitoye, O.B.; Uwazie, J.N.; Ajiboye, T.O. Bioactivity-guided isolation of kaempferol as the antidiabetic principle from Cucumis sativus L. fruits. J. Food Biochem. 2018, 42, e12479. [Google Scholar] [CrossRef]
- Panda, S.; Kar, A. Apigenin (4′, 5, 7-trihydroxyflavone) regulates hyperglycaemia, thyroid dysfunction and lipid peroxidation in alloxan-induced diabetic mice. J. Pharm. Pharmacol. 2007, 59, 1543–1548. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-D.; Gao, D.; Xu, W.J.; Li, F.; Yin, M.N.; Fu, Q.F.; Xia, Z.N. Magnetic molecularly imprinted polymer for the selective extraction of hesperetin from the dried pericarp of Citrus reticulata Blanco. Talanta 2018, 184, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.P.; Kandpal, J.B.; Sharma, R.K.; Chitme, H. Nobiletin a Biologically Active Phytoconstituent: Systematic Review. J. Biol. Act. Prod. Nat. 2021, 11, 204–211. [Google Scholar] [CrossRef]
- Xu, Y.R.; Zhang, K.F.; Xie, Q.J.; Lin, J.X.; Huan, K.X.; Liao, Y. Chemical constituents from young fruits of Citrus maxima cv. Shatian. Zhong Yao Cai = Zhongyaocai = J. Chin. Med. Mater. 2015, 38, 1879–1881. [Google Scholar]
- Den Hartogh, D.J.; Tsiani, E. Antidiabetic properties of naringenin: A citrus fruit polyphenol. Biomolecules 2019, 9, 99. [Google Scholar] [CrossRef]
- Ahmed, O.M.; Hassan, M.A.; Abdel-Twab, S.M.; Azeem, M.N.A. Navel orange peel hydroethanolic extract, naringin and naringenin have anti-diabetic potentials in type 2 diabetic rats. Biomed. Pharmacother. 2017, 94, 197–205. [Google Scholar] [CrossRef]
- Musara, C.; Aladejana, E.B.; Mudyiwa, S.M. Review of the nutritional composition, medicinal, phytochemical and pharmacological properties of Citrus reticulata Blanco (Rutaceae). F1000Research 2020, 9, 1387. [Google Scholar] [CrossRef]
- Özbek, H.; Yılmaz, B.S. Anti-inflammatory and hypoglycemic activities of alpha-pinene. Acta Pharm. Sci. 2017, 55, 7–14. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, J.; Gu, S.; Liu, Z.; Zhang, Y.; Zhang, X. Simultaneous Determination of Flavonoids in Different Parts of Citrus reticulata ‘Chachi’ Fruit by High Performance Liquid Chromatography—Photodiode Array Detection. Molecules 2010, 15, 5378–5388. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Yi, W.J.; Tan, L.; Zhang, J.H.; Xu, J.; Chen, Y.; Qin, M.; Yu, S.; Guan, J.; Zhang, R. Apigenin attenuates streptozotocin-induced pancreatic β cell damage by its protective effects on cellular antioxidant defense. Vitr. Cell. Dev. Biol.-Anim. 2017, 53, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhu, X.; Luo, F.; Sun, C.; Huang, J.; Li, X.; Chen, K. Separation and purification of neohesperidin from the albedo of Citrus reticulata cv. Suavissima by combination of macroporous resin and high-speed counter-current chromatography. J. Sep. Sci. 2012, 35, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Yong, J.W.H.; Ge, L.; Ng, Y.F.; Tan, S.N. The Chemical Composition and Biological Properties of Coconut (Cocos nucifera L.) Water. Molecules 2009, 14, 5144–5164. [Google Scholar] [CrossRef]
- Mallek-Ayadi, S.; Bahloul, N.; Kechaou, N. Phytochemical profile, nutraceutical potential and functional properties of Cucumis melo L. seeds. J. Sci. Food Agric. 2019, 99, 1294–1301. [Google Scholar] [CrossRef]
- Prabhakar, P.K.; Doble, M. Effect of natural products on commercial oral antidiabetic drugs in enhancing 2-deoxyglucose uptake by 3T3-L1 adipocytes. Ther. Adv. Endocrinol. Metab. 2011, 2, 103–114. [Google Scholar] [CrossRef]
- Peungvicha, P.; Temsiririrkkul, R.; Prasain, J.K.; Tezuka, Y.; Kadota, S.; Thirawarapan, S.S.; Watanabe, H. 4-Hydroxybenzoic acid: A hypoglycemic constituent of aqueous extract of Pandanus odorus root. J. Ethnopharmacol. 1998, 62, 79–84. [Google Scholar] [CrossRef]
- Kaur, N.; Kishore, L.; Singh, R. Antidiabetic effect of new chromane isolated from Dillenia indica L. leaves in streptozotocin induced diabetic rats. J. Funct. Foods 2016, 22, 547–555. [Google Scholar] [CrossRef]
- Ogundele, A.V.; Yadav, A.; Das, A.M. Antimicrobial and α-Amylase Inhibitory Activities of Constituents from Elaeocarpus floribundus. Rev. Bras. Farmacogn. 2021, 31, 330–334. [Google Scholar] [CrossRef]
- Alam, M.R.; Rahman, A.B.; Moniruzzaman, M.; Kadir, M.F.; Haque, M.A.; Alvi, M.R.-U.-H.; Ratan, M. Evaluation of antidiabetic phytochemicals in Syzygium cumini (L.) Skeels (Family: Myrtaceae). J. Appl. Pharm. Sci. 2012, 2, 94. [Google Scholar] [CrossRef]
- Zarga, M.H.A. Three new simple indole alkaloids from Limonia acidissima. J. Nat. Prod. 1986, 49, 901–904. [Google Scholar] [CrossRef]
- Salau, V.F.; Erukainure, O.L.; Ibeji, C.U.; Koorbanally, N.A.; Islam, M.S. Umbelliferone stimulates glucose uptake; modulates gluconeogenic and nucleotide-hydrolyzing enzymes activities, and dysregulated lipid metabolic pathways in isolated psoas muscle. J. Funct. Foods 2020, 67, 103847. [Google Scholar] [CrossRef]
- Chang, Y.-Y.; Yang, D.J.; Chiu, C.H.; Lin, Y.L.; Chen, J.W.; Chen, Y.C. Antioxidative and anti-inflammatory effects of polyphenol-rich litchi (Litchi chinensis Sonn.)-flower-water-extract on livers of high-fat-diet fed hamsters. J. Funct. Foods 2013, 5, 44–52. [Google Scholar] [CrossRef]
- Mechchate, H.; Es-Safi, I.; Bousta, D. Insight into Gentisic Acid Antidiabetic Potential Using In Vitro and In Silico Approaches. Molecules 2021, 26, 1932. [Google Scholar] [CrossRef] [PubMed]
- Prakash, M.; Basavaraj, B.; Murthy, K.C. Biological functions of epicatechin: Plant cell to human cell health. J. Funct. Foods 2019, 52, 14–24. [Google Scholar] [CrossRef]
- Amran, M.S.; Sultan, M.Z.; Rahman, A.; Rashid, M.A. Antidiabetic activity of compounds isolated from the kernel of Mangifera indica in alloxan induced diabetic rats. Dhaka Univ. J. Pharm. Sci. 2013, 12, 77–81. [Google Scholar] [CrossRef]
- Mohan, C.G.; Viswanatha, G.L.; Savinay, G.; Rajendra, C.E.; Halemani, P.D. 1,2,3,4,6 Penta-O-galloyl-β-d-glucose, a bioactivity guided isolated compound from Mangifera indica inhibits 11β-HSD-1 and ameliorates high fat diet-induced diabetes in C57BL/6 mice. Phytomedicine 2013, 20, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Pravin, K.P.; Shashikant, D.C. Manilkara zapota (L.) Royen fruit peel: A phytochemical and pharmacological review. Syst. Rev. Pharm. 2019, 10, 11–14. [Google Scholar] [CrossRef]
- Imam, M.Z.; Akter, S. Musa paradisiaca L. and Musa sapientum L.: A phytochemical and pharmacological review. J. Appl. Pharm. Sci. 2011, 1, 14–20. [Google Scholar]
- Kushwaha, P.S.; Raj, V.; Singh, A.K.; Keshari, A.K.; Saraf, S.A.; Mandal, S.C.; Yadav, R.K.; Saha, S. Antidiabetic effects of isolated sterols from Ficus racemosa leaves. RSC Adv. 2015, 5, 35230–35237. [Google Scholar] [CrossRef]
- Nandini, H.; Naik, P.R. Action of corilagin on hyperglycemia, hyperlipidemia and oxidative stress in streptozotocin-induced diabetic rats. Chem.-Biol. Interact. 2019, 299, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Poltanov, E.A.; Shikov, A.N.; Dorman, H.D.; Pozharitskaya, O.N.; Makarov, V.G.; Tikhonov, V.P.; Hiltunen, R. Chemical and antioxidant evaluation of Indian gooseberry (Emblica officinalis Gaertn., syn. Phyllanthus emblica L.) supplements. Phytother. Res. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2009, 23, 1309–1315. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, L.; Guo, X.; Li, C.; Li, H.; Lou, H.; Ren, D. Chemical constituents from Phyllanthus emblica and the cytoprotective effects on H 2 O 2-induced PC12 cell injuries. Arch. Pharmacal Res. 2016, 39, 1202–1211. [Google Scholar] [CrossRef] [PubMed]
- Adisakwattana, S. Cinnamic acid and its derivatives: Mechanisms for prevention and management of diabetes and its complications. Nutrients 2017, 9, 163. [Google Scholar] [CrossRef]
- Zhu, X.; Ouyang, W.; Lan, Y.; Xiao, H.; Tang, L.; Liu, G.; Feng, K.; Zhang, L.; Song, M.; Cao, Y. Anti-hyperglycemic and liver protective effects of flavonoids from Psidium guajava L. (guava) leaf in diabetic mice. Food Biosci. 2020, 35, 100574. [Google Scholar] [CrossRef]
- Mohammed, F.Z.; El-Shehabi, M. Antidiabetic activity of caffeic acid and 18β-glycyrrhetinic acid and its relationship with the antioxidant property. Asian J. Pharm. Clin. Res 2015, 8, 229–235. [Google Scholar]
- Baliga, M.; Shivashankara, A.; Shetty, C.; Thilakchand, K.; Periera, N.; Palatty, P. Antidiabetic effects of Punica granatum L. (Pomegranate): A review. Bioact. Food Diet. Interv. Diabetes 2012, 355–369. [Google Scholar]
- Wu, S.; Tian, L. A new flavone glucoside together with known ellagitannins and flavones with anti-diabetic and anti-obesity activities from the flowers of pomegranate (Punica granatum). Nat. Prod. Res. 2019, 33, 252–257. [Google Scholar] [CrossRef]
- Khajebishak, Y.; Payahoo, L.; Alivand, M.; Alipour, B. Punicic acid: A potential compound of pomegranate seed oil in Type 2 diabetes mellitus management. J. Cell. Physiol. 2019, 234, 2112–2120. [Google Scholar] [CrossRef]
- Jun, L.; Xue, W.; Yu-Peng, C.; Li-Fei, M.; Shang, J.; Hong-Bin, S.; Zhang, L.-Y. Maslinic acid modulates glycogen metabolism by enhancing the insulin signaling pathway and inhibiting glycogen phosphorylase. Chin. J. Nat. Med. 2014, 12, 259–265. [Google Scholar]
- Zang, Y.; Igarashi, K.; Li, Y. Anti-diabetic effects of luteolin and luteolin-7-O-glucoside on KK-A y mice. Biosci. Biotechnol. Biochem. 2016, 80, 1580–1586. [Google Scholar] [CrossRef] [PubMed]
- Bellesia, A.; Verzelloni, E.; Tagliazucchi, D. Pomegranate ellagitannins inhibit α-glucosidase activity in vitro and reduce starch digestibility under simulated gastro-intestinal conditions. Int. J. Food Sci. Nutr. 2015, 66, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Fred-Jaiyesimi, A.; Kio, A.; Richard, W. α-Amylase inhibitory effect of 3β-olean-12-en-3-yl (9Z)-hexadec-9-enoate isolated from Spondias mombin leaf. Food Chem. 2009, 116, 285–288. [Google Scholar] [CrossRef]
- Fred-Jaiyesimi, A.A.; Wilkins, R.M.; Abo, K.A. Glucose lowering activities of mombintane I and mombintane II isolated from the leaves of Spondias mombin L. Int. J. Biol. Chem. Sci. 2017, 11, 1315–1319. [Google Scholar] [CrossRef][Green Version]
- Resurreccion-Magno, M.H.C.; Villaseñor, I.M.; Harada, N.; Monde, K. Antihyperglycaemic flavonoids from Syzygium samarangense (Blume) merr. and perry. Phytother. Res. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2005, 19, 246–251. [Google Scholar]
- Huang, D.-W.; Chang, W.-C.; Wu, J.S.-B.; Shih, R.-W.; Shen, S.-C. Vescalagin from pink wax apple [Syzygium samarangense (Blume) Merrill and Perry] alleviates hepatic insulin resistance and ameliorates glycemic metabolism abnormality in rats fed a high-fructose diet. J. Agric. Food Chem. 2016, 64, 1122–1129. [Google Scholar] [CrossRef]
- Meher, B.; Dash, D.K.; Roy, A. A review on: Phytochemistry, pharmacology and traditional uses of Tamarindus indica L. WJPPS 2014, 3, 229–240. [Google Scholar]
- Bates, S.H.; Jones, R.B.; Bailey, C.J. Insulin-like effect of pinitol. Br. J. Pharmacol. 2000, 130, 1944–1948. [Google Scholar] [CrossRef]
- Gan, Q.; Wang, J.; Hu, J.; Lou, G.; Xiong, H.; Peng, C.; Zheng, S.; Huang, Q. The role of diosgenin in diabetes and diabetic complications. J. Steroid Biochem. Mol. Biol. 2020, 198, 105575. [Google Scholar] [CrossRef]
- Gao, D.; Li, Q.; Li, Y.; Liu, Z.; Liu, Z.; Fan, Y.; Han, Z.; Li, J.; Li, K. Antidiabetic potential of oleanolic acid from Ligustrum lucidum Ait. Can. J. Physiol. Pharmacol. 2007, 85, 1076–1083. [Google Scholar] [CrossRef]
- Dadheech, N.; Srivastava, A.; Paranjape, N.; Gupta, S.; Dave, A.; Shah, G.M.; Bhonde, R.R.; Gupta, S. Swertisin an anti-diabetic compound facilitate islet neogenesis from pancreatic stem/progenitor cells via p-38 MAP kinase-SMAD pathway: An in-vitro and in-vivo study. PLoS ONE 2015, 10, e0128244. [Google Scholar] [CrossRef]
- Baliga, M.S.; Fernandes, S.; Thilakchand, K.R.; D’Souza, P.; Rao, S. Scientific validation of the antidiabetic effects of Syzygium jambolanum DC (black plum), a traditional medicinal plant of India. J. Altern. Complement. Med. 2013, 19, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Deguchi, Y.; Miyazaki, K. Anti-hyperglycemic and anti-hyperlipidemic effects of guava leaf extract. Nutr. Metab. 2010, 7, 9. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Jin, D.; An, X.; Duan, L.; Duan, Y.; Lian, F. Lychee Seed as a Potential Hypoglycemic Agent and Its Underlying Mechanism Exploration. Front. Pharmacol. 2021, 12, 737803. [Google Scholar] [CrossRef] [PubMed]
- Alam, S.; Sarker, M.M.R.; Sultana, T.N.; Chowdhury, M.N.R.; Rashid, M.A.; Chaity, N.I.; Zhao, C.; Xiao, J.; Hafez, E.E.; Khan, S.A. Antidiabetic Phytochemicals from Medicinal Plants: Prospective Candidates for New Drug Discovery and Development. Front. Endocrinol. 2022, 13, 800714. [Google Scholar] [CrossRef]
- Yin, J.; Ye, J.; Jia, W. Effects and mechanisms of berberine in diabetes treatment. Acta Pharm. Sin. B 2012, 2, 327–334. [Google Scholar] [CrossRef]
- Eghtesadi, S.; Mohammadi, M.; Vafa, M.; Heidari, I.; Salehi, M.; Khadem, H.H.; Amiri, F.; Alipour, R.; Eghtesadi, M. Effects of hesperidin supplementation on glycemic control, lipid profile and inflammatory factors in patients with type 2 diabetes: A randomized, double-blind and placebo-controlled clinical trial. Endocr. Abstr. 2016, 43, 269. [Google Scholar] [CrossRef]
- Ghadimi, M.; Foroughi, F.; Hashemipour, S.; Rashidi Nooshabadi, M.; Ahmadi, M.H.; Ahadi Nezhad, B.; Khadem Haghighian, H. Randomized double-blind clinical trial examining the Ellagic acid effects on glycemic status, insulin resistance, antioxidant, and inflammatory factors in patients with type 2 diabetes. Phytother. Res. 2021, 35, 1023–1032. [Google Scholar] [CrossRef]
- Kim, H.J.; Park, K.S.; Lee, S.K.; Min, K.W.; Han, K.A.; Kim, Y.K.; Ku, B.J. Effects of pinitol on glycemic control, insulin resistance and adipocytokine levels in patients with type 2 diabetes mellitus. Ann. Nutr. Metab. 2012, 60, 1–5. [Google Scholar] [CrossRef]
- Abbaszadeh, F. The Effect of L-Citrulline Supplementation on Glycemic Status, Meta-Inflammation and Obesity Indices in Type 2 Diabetes Patients: A Randomized Controlled Clinical Trial; School of Nutrition, Tabriz University of Medical Sciences: Tabriz, Iran, 2021. [Google Scholar]
- Agu, K.C.; Eluehike, N.; Ofeimun, R.O.; Abile, D.; Ideho, G.; Ogedengbe, M.O.; Onose, P.O.; Elekofehinti, O.O. Possible anti-diabetic potentials of Annona muricata (soursop): Inhibition of α-amylase and α-glucosidase activities. Phytomedicine 2019, 5, 1–13. [Google Scholar] [CrossRef]
- Youn, J.-Y.; Park, H.-Y.; Cho, K.-H. Anti-hyperglycemic activity of Commelina communis L.: Inhibition of α-glucosidase. Diabetes Res. Clin. Pract. 2004, 66, S149–S155. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhang, Y.; Zhu, J.; Li, B.; Li, Z.; Zhu, W.; Shi, J.; Jia, Q.; Li, Y. Recent progress in natural products as DPP-4 inhibitors. Future Med. Chem. 2015, 7, 1079–1089. [Google Scholar] [CrossRef] [PubMed]
- Elya, B.; Handayani, R.; Sauriasari, R.; Hasyyati, U.S.; Permana, I.T.; Permatasari, Y.I. Antidiabetic activity and phytochemical screening of extracts from Indonesian plants by inhibition of alpha amylase, alpha glucosidase and dipeptidyl peptidase IV. Pak. J. Biol. Sci. 2015, 18, 279. [Google Scholar] [CrossRef]
- Mechchate, H.; Es-Safi, I.; Louba, A.; Alqahtani, A.S.; Nasr, F.A.; Noman, O.M.; Farooq, M.; Alharbi, M.S.; Alqahtani, A.; Bari, A. In vitro alpha-amylase and alpha-glucosidase inhibitory activity and in vivo antidiabetic activity of Withania frutescens L. Foliar extract. Molecules 2021, 26, 293. [Google Scholar] [CrossRef]
- Adisakwattana, S.; Moonsan, P.; Yibchok-Anun, S. Insulin-releasing properties of a series of cinnamic acid derivatives in vitro and in vivo. J. Agric. Food Chem. 2008, 56, 7838–7844. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Yang, C.; Wai, S.T.C.; Zhang, Y.; Portillo, M.P.; Paoli, P.; Wu, Y.; San Cheang, W.; Liu, B.; Carpéné, C. Regulation of glucose metabolism by bioactive phytochemicals for the management of type 2 diabetes mellitus. Crit. Rev. Food Sci. Nutr. 2019, 59, 830–847. [Google Scholar] [CrossRef]
- Schenk, S.; Saberi, M.; Olefsky, J.M. Insulin sensitivity: Modulation by nutrients and inflammation. J. Clin. Investig. 2008, 118, 2992–3002. [Google Scholar] [CrossRef]
- Ghorbani, A.; Rashidi, R.; Shafiee-Nick, R. Flavonoids for preserving pancreatic beta cell survival and function: A mechanistic review. Biomed. Pharmacother. 2019, 111, 947–957. [Google Scholar] [CrossRef]
- Oh, Y.S. Plant-derived compounds targeting pancreatic beta cells for the treatment of diabetes. Evid.-Based Complement. Altern. Med. 2015, 2015, 629863. [Google Scholar] [CrossRef]
- Ghazanfari-Sarabi, S.; Habibi-Rezaei, M.; Eshraghi-Naeeni, R.; Moosavi-Movahedi, A.A. Prevention of haemoglobin glycation by acetylsalicylic acid (ASA): A new view on old mechanism. PLoS ONE 2019, 14, e0214725. [Google Scholar] [CrossRef]
- Welsh, K.J.; Kirkman, M.S.; Sacks, D.B. Role of glycated proteins in the diagnosis and management of diabetes: Research gaps and future directions. Diabetes Care 2016, 39, 1299–1306. [Google Scholar] [CrossRef] [PubMed]
- Harding, J.J.; Ganea, E. Protection against glycation and similar post-translational modifications of proteins. Biochim. Biophys. Acta (BBA)-Proteins Proteom. 2006, 1764, 1436–1446. [Google Scholar] [CrossRef] [PubMed]
- Baggio, L.L.; Drucker, D.J. Biology of incretins: GLP-1 and GIP. Gastroenterology 2007, 132, 2131–2157. [Google Scholar] [CrossRef] [PubMed]
- Shaefer, C.F., Jr.; Kushner, P.; Aguilar, R. User’s guide to mechanism of action and clinical use of GLP-1 receptor agonists. Postgrad. Med. 2015, 127, 818–826. [Google Scholar] [CrossRef]
- Huang, S.; Czech, M.P. The GLUT4 glucose transporter. Cell Metab. 2007, 5, 237–252. [Google Scholar] [CrossRef]
- Manda, V.; Avula, B.; Mir, T.; Ashfaq, M.; Khan, I.; Khan, S. Pharmacokinetics of aegeline after oral administration in a mouse model. In Proceedings of the International Conference on the Science of Botanicals, Oxford, MS, USA, 4 April 2017. [Google Scholar]
- Mohammed, M.M.; Ibrahim, N.A.; El-Sakhawy, F.S.; Mohamed, K.M.; Deabes, D.A.-H. Two new cytotoxic furoquinoline alkaloids isolated from Aegle marmelos (Linn.) Correa. Nat. Prod. Res. 2016, 30, 2559–2566. [Google Scholar] [CrossRef]
- Zuo, F.; Nakamura, N.; Akao, T.; Hattori, M. Pharmacokinetics of berberine and its main metabolites in conventional and pseudo germ-free rats determined by liquid chromatography/ion trap mass spectrometry. Drug Metab. Dispos. 2006, 34, 2064–2072. [Google Scholar] [CrossRef]
- Hua, W.; Ding, L.; Chen, Y.; Gong, B.; He, J.; Xu, G. Determination of berberine in human plasma by liquid chromatography–electrospray ionization–mass spectrometry. J. Pharm. Biomed. Anal. 2007, 44, 931–937. [Google Scholar] [CrossRef]
- Liu, Y.-T.; Hao, H.-P.; Xie, H.-G.; Lai, L.; Wang, Q.; Liu, C.-X.; Wang, G.-J. Extensive intestinal first-pass elimination and predominant hepatic distribution of berberine explain its low plasma levels in rats. Drug Metab. Dispos. 2010, 38, 1779–1784. [Google Scholar] [CrossRef]
- Ma, J.Y.; Feng, R.; Tan, X.S.; Ma, C.; Shou, J.W.; Fu, J.; Huang, M.; He, C.Y.; Chen, S.N.; Zhao, Z.X. Excretion of berberine and its metabolites in oral administration in rats. J. Pharm. Sci. 2013, 102, 4181–4192. [Google Scholar] [CrossRef]
- Feng, R.; Zhao, Z.-X.; Ma, S.-R.; Guo, F.; Wang, Y.; Jiang, J.-D. Gut microbiota-regulated pharmacokinetics of berberine and active metabolites in beagle dogs after oral administration. Front. Pharmacol. 2018, 9, 214. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Zhou, Y.; Xu, P.; Wang, Y.; Yan, J.; Bin, W.; Qiu, F.; Kang, N. Berberine metabolites exhibit triglyceride-lowering effects via activation of AMP-activated protein kinase in Hep G2 cells. J. Ethnopharmacol. 2013, 149, 576–582. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Cao, S.; Wang, Y.; Xu, P.; Yan, J.; Bin, W.; Qiu, F.; Kang, N. Berberine metabolites could induce low density lipoprotein receptor up-regulation to exert lipid-lowering effects in human hepatoma cells. Fitoterapia 2014, 92, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Yang, N.; Sun, R.-B.; Chen, X.-L.; Zhen, L.; Ge, C.; Zhao, Y.-Q.; He, J.; Geng, J.-L.; Guo, J.-H.; Yu, X.-Y. In vitro assessment of the glucose-lowering effects of berberrubine-9-O-β-D-glucuronide, an active metabolite of berberrubine. Acta Pharmacol. Sin. 2017, 38, 351–361. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, H.; Mimura, M.; Sugahara, C.; Shimada, T. Catalytic roles of rat and human cytochrome P450 2A enzymes in testosterone 7α-and coumarin 7-hydroxylations. Biochem. Pharmacol. 1994, 48, 1524–1527. [Google Scholar] [CrossRef]
- Born, S.L.; Caudill, D.; Fliter, K.L.; Purdon, M.P. Identification of the cytochromes P450 that catalyze coumarin 3, 4-epoxidation and 3-hydroxylation. Drug Metab. Dispos. 2002, 30, 483–487. [Google Scholar] [CrossRef]
- Tanaka, Y.; Fujii, W.; Hori, H.; Kitagawa, Y.; Ozaki, K. Changes in coumarin kinetics and subcellular localization of CYP2E1 contribute to bile duct damage and reduce hepatocellular damage after repeated administration of coumarin in rats. Toxicol. Lett. 2017, 280, 99–105. [Google Scholar] [CrossRef]
- Velderrain-Rodríguez, G.; Palafox-Carlos, H.; Wall-Medrano, A.; Ayala-Zavala, J.; Chen, C.O.; Robles-Sánchez, M.; Astiazaran-García, H.; Alvarez-Parrilla, E.; González-Aguilar, G. Phenolic compounds: Their journey after intake. Food Funct. 2014, 5, 189–197. [Google Scholar] [CrossRef]
- Wiese, S.; Esatbeyoglu, T.; Winterhalter, P.; Kruse, H.P.; Winkler, S.; Bub, A.; Kulling, S.E. Comparative biokinetics and metabolism of pure monomeric, dimeric, and polymeric flavan-3-ols: A randomized cross-over study in humans. Mol. Nutr. Food Res. 2015, 59, 610–621. [Google Scholar] [CrossRef]
- DiNicolantonio, J.J.; O’Keefe, J.H.; Lucan, S.C. Added fructose: A principal driver of type 2 diabetes mellitus and its consequences. Mayo Clin. Proc. 2015, 90, 372–381. [Google Scholar] [CrossRef]
- Papachristos, D.; Kimbaris, A.; Papadopoulos, N.; Polissiou, M. Toxicity of citrus essential oils against Ceratitis capitata (Diptera: Tephritidae) larvae. Ann. Appl. Biol. 2009, 155, 381–389. [Google Scholar] [CrossRef]
- Unger, J.; Parkin, C.G. Type 2 diabetes: An expanded view of pathophysiology and therapy. Postgrad. Med. 2010, 122, 145–157. [Google Scholar] [CrossRef]
- Montecucco, F.; Steffens, S.; Mach, F. Insulin resistance: A proinflammatory state mediated by lipid-induced signaling dysfunction and involved in atherosclerotic plaque instability. Mediat. Inflamm. 2008, 2008, 767623. [Google Scholar] [CrossRef]
- Tatoń, J.; Czech, A.; Piątkiewicz, P. Insulin as the main regulator of cellular glucose utilization-aetiological aspects of insulin resistance. Endokrynol. Pol. 2010, 61, 388–394. [Google Scholar] [PubMed]
- Alam, S.; Sarker, M.M.R.; Afrin, S.; Richi, F.T.; Zhao, C.; Zhou, J.R.; Mohamed, I.N. Traditional herbal medicines, bioactive metabolites, and plant products against COVID-19: Update on clinical trials and mechanism of actions. Front. Pharmacol. 2021, 12, 671498. [Google Scholar] [CrossRef] [PubMed]
- Osadebe, P.O.; Odoh, E.U.; Uzor, P.F. Natural products as potential sources of antidiabetic drugs. Br. J. Pharm. Res. 2014, 4, 2075–2095. [Google Scholar] [CrossRef]
- Verspohl, E. Recommended testing in diabetes research. Planta Med. 2002, 68, 581–590. [Google Scholar] [CrossRef]
- Chopra, A.; Saluja, M.; Tillu, G. Ayurveda–modern medicine interface: A critical appraisal of studies of Ayurvedic medicines to treat osteoarthritis and rheumatoid arthritis. J. Ayurveda Integr. Med. 2010, 1, 190. [Google Scholar] [CrossRef]
- Piggott, A.M.; Karuso, P. Quality, not quantity: The role of natural products and chemical proteomics in modern drug discovery. Comb. Chem. High Throughput Screen. 2004, 7, 607–630. [Google Scholar] [CrossRef]
- Ortholand, J.-Y.; Ganesan, A. Natural products and combinatorial chemistry: Back to the future. Curr. Opin. Chem. Biol. 2004, 8, 271–280. [Google Scholar] [CrossRef]
- Kennedy, D.; Wightman, E. Herbal extracts and phytochemicals: Plant secondary metabolites and the enhancement of human brain function. Adv. Nutr. 2011, 2, 32–50. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.; Cragg, G. Natural Products, Derivatives and Mimics as Antitumour Agents. Spec. Publ.-R. Soc. Chem. 2011, 320, 3–36. [Google Scholar]
- Lane, T.; Anantpadma, M.; Freundlich, J.S.; Davey, R.A.; Madrid, P.B.; Ekins, S. The natural product eugenol is an inhibitor of the ebola virus in vitro. Pharm. Res. 2019, 36, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Subirana, M.; Solá, I.; Garcia, J.M.; Gich, I.; Urrútia, G. A nursing qualitative systematic review required MEDLINE and CINAHL for study identification. J. Clin. Epidemiol. 2005, 58, 20–25. [Google Scholar] [CrossRef] [PubMed]

| S.L. | Species | Plant Part | Phytochemicals | Mode of Actions | References |
|---|---|---|---|---|---|
| 1 | Aegle Marmelos | Leaf | Aegeline | Blood glucose-lowering activity | [121] |
| Aegelin 2; sitosterol; scopoletin | Blood glucose-lowering activity | [186] | |||
| 2 | Annona squamosa | Leaf | Rutin | Inhibition of α-glucosidase activity and improved insulin secretion | [187] |
| Quercetin | Improving insulin secretion | [187,188] | |||
| Isoquercetin | Inhibiting the activity of DPP-4 | [187] | |||
| 3 | Ananas comosus | Fruit | Catechin | α-glucosidase inhibitory actions | [189,190] |
| Epicatechin | Promoting β-cell regeneration | [190,191] | |||
| Gallic acid | Improved glucose transporters and insulin sensitivity through PPAR-γ and Akt signaling | [190,192] | |||
| p-coumaric acid | Lowering the blood glucose level, increasing the level of insulin | [190,193] | |||
| Ferulic acid | Restoring blood glucose and serum insulin level; improving insulin sensitivity, hepatic glycogenesis, glucose tolerance, and insulin tolerance along with reducing the activity of glycogen synthase and glucokinase and also increasing activity of glycogen phosphorylase and enzymes of gluconeogenesis (PEPCK and G6Pase) | [190,194] | |||
| Caffeic acid | Reducing blood glucose concentration by inhibiting the activity of glucose-6-phosphatase and increasing insulin secretion | [190,195] | |||
| Ellagic acid | Stimulating insulin secretion and decreases glucose intolerance by acting on β-cells of the pancreas | [71,190] | |||
| Vanillin | Reducing serum glucose level and increasing insulin level | [190,196] | |||
| 4 | Artocarpus heterophyllus | Whole Plant | Morin | Down-regulation of miR-29a expression level to improve insulin signaling and glucose metabolism | [197] |
| Bark from the main trunk, Root | Betulinic acid | Activating AMPK, reducing blood glucose level, and stimulating mRNA expression of GLUT-4 | [197,198] | ||
| Root | Ursolic acid | Lowering blood glucose level | [197,199] | ||
| Leaves, Stem, Root | β-sitosterol | Improving insulin resistance and insulin signaling along with reducing fasting blood glucose level and glycated hemoglobin | [197,200] | ||
| 5 | Averrhoa carambola | Fruit | 2-dodecyl-6-methoxycyclohexa-2,5-diene-1, 4-dione | Improved blood sugar level and inhibition of the TLR-4/TGF-β signaling pathway | [156] |
| 6 | Baccaurea motleyana | Fruit | Limonene | Inhibiting protein glycation | [110,201] |
| Carvacrol | α-amylase inhibitory actions | [110,201] | |||
| 7 | Borassus flabellifer | Fruit pulp | Tyrosol, Glucosyl-(6-1)-glycerol | α-glucosidase inhibitory actions | [202] |
| 8 | Carica papaya | Seed | Hexadecanoic acid, methyl ester; 11-octadecenoic acid, methyl ester; N, N-dimethyl-; n-hexadecanoic acid, and oleic acid | Inhibiting α-amylase and α-glucosidase | [132] |
| 9 | Carissa carandas | Leaf | Ursolic acid | Lowering blood glucose level | [203,204] |
| Fruit | Myo-inositol | Improved insulin-stimulated glucose uptake in mature adipocytes; increased insulin sensitivity and glucose uptake | [36,205] | ||
| β-amyrin | Improvement in blood sugar level and plasma insulin along with preservation of β cell integrity | [36,206] | |||
| 10 | Citrullus lanatus | Fruit | Citrulline | Improving insulin sensitivity | [166,207] |
| 6-gingerol | Lowering fasting blood glucose levels and improving glucose tolerance | [166,208] | |||
| Arjunolic acid | Protective action on pancreatic β-cells, Reducing blood glucose level | [166,209] | |||
| Lycopene | Reducing blood glucose level | [195] | |||
| 11 | Citrus arunattifolia | Fruit | Hesperetin | Reducing blood glucose levels and increasing plasma insulin concentration and glycogen levels, ameliorating the abnormality resulting from hyperglycemia in pancreatic β-cells, increasing the number of insulin immune-positive cells of the islets | [210,211] |
| Quercetin | Increased glucose uptake via regulation of the AMPK pathway along with improved GLUT-4 expression and regeneration of β-cells in the pancreatic islets | [210,211] | |||
| Rutin | Reducing blood glucose, elevating glycogen concentration | [210,211] | |||
| Nobiletin | Enhancing glucose uptake via the PI3K/Akt signaling pathway | [210,211] | |||
| Kaempferol | Inhibiting the activities of α-amylase and α-glucosidase along with glucose-lowering activity | [211,212] | |||
| Naringenin | lowered the increased plasma glucose concentration, increased the activity of Superoxide dismutase (SOD) | [210,211] | |||
| Apigenin | Reduction in serum glucose level, enhancing serum insulin level, nourishment of pancreatic β cell | [211,213,214] | |||
| 12 | Citrus limon | Fruit Peel | Nobiletin | Enhancing glucose uptake via the PI3K/Akt signaling pathway | [210,215] |
| Fruit, leaf | Diosmin | Improvement in fasting plasma glucose concentrations, glycosylated hemoglobin (HbA1c), C-reactive protein (CRP), and the activities of Lipoprotein lipase (LPL) and lecithin cholesterol acyl transferase (LCAT) enzymes | [210] | ||
| Fruit | Rhoifolin | Insulin mimetic activity | [210] | ||
| Didymin | Improving insulin sensitivity, inhibitory action on α-glucosidase and diminished hepatic glucose production in insulin-resistant HepG2 cells | [210] | |||
| Hesperidin | Reducing serum insulin and blood glucose level, normalizing the enzymatic activities of glucose-6-phosphatase and glucokinase enzyme | [210] | |||
| Hesperetin | Reducing blood glucose levels and increasing plasma insulin concentration and glycogen levels, ameliorating the abnormality resulting from hyperglycemia in pancreatic β-cells, increasing the number of insulin immune-positive cells of the islets | [210] | |||
| Rutin | Reducing blood glucose, elevating glycogen concentration | [210] | |||
| Quercetin | Increased glucose uptake via regulation of the AMPK pathway along with improved GLUT-4 expression and regeneration of β-cells in the pancreatic islets | [210] | |||
| 13 | Citrus maxima | Fruit | β-sitosterol | Improving insulin resistance and insulin signaling along with reducing fasting blood glucose level and glycated hemoglobin | [200,216] |
| Naringenin | Increased glucose uptake | [216,217] | |||
| Naringin | Improved serum insulin level; enhanced mRNA expression of insulin receptor β-subunit and GLUT-4 | [216,218] | |||
| 14 | Citrus reticulata | Fruit | alpha-pinene | Decreasing fasting blood glucoselevels | [219,220] |
| Limonene | Inhibiting protein glycation | [201,219] | |||
| Rutin | Reducing blood glucose, elevating glycogen concentration | [210] | |||
| Quercetin | Increased glucose uptake via regulation of the AMPK pathway along with improved GLUT-4 expression and regeneration of β-cells in the pancreatic islets | [210] | |||
| Naringin | Modifying the release of insulin from isolated islet and intestinal glucose absorption, improved expression of GLUT-4 in adipose tissue, and free circulating glucose uptake from the blood to peripheral tissues | [210,221] | |||
| Naringenin | Lowered the increased plasma glucose concentration, increased the activity of Superoxide dismutase (SOD) | [210] | |||
| Didymin | Improving insulin sensitivity, inhibitory action on α-glucosidase and diminished hepatic glucose production in insulin-resistant HepG2 cells | [210] | |||
| Hesperidin | Reducing serum insulin and blood glucose level, normalizing the enzymatic activities of glucose-6-phosphatase and glucokinase enzyme | [210] | |||
| 8-Prenylnaringenin | Regulating the expression of Galectin-3 (Gal-3) protein which is overexpressed during the diabetic state, promoting glycation end products (AGEs) production | [210] | |||
| Fruits and leaves | Diosmin | Improvement in fasting plasma glucose concentrations, glycosylated hemoglobin (HbA1c), C-reactive protein (CRP), and the activities of Lipoprotein lipase (LPL) and lecithin cholesterol acyl transferase (LCAT) enzymes | [210] | ||
| Fruit peel | Nobiletin | Enhancing glucose uptake via the PI3K/Akt signaling pathway | [210] | ||
| Tangeretin | Increasing glucose uptake, improving glycogen level and the activities of glycogen synthase and glycogen phosphorylase, regeneration of pancreatic β-cells in the islets | [210] | |||
| Dried pericarp | Hesperetin | Reducing blood glucose levels and increasing plasma insulin concentration and glycogen levels, ameliorating the abnormality resulting from hyperglycemia in pancreatic β-cells, increasing the number of insulin immune-positive cells of the islets | [210,222] | ||
| Albedo of fruit | Neohesperidin | Improved glucose tolerance and insulin sensitivity along with abatement in the blood glucose level | [210,223] | ||
| 15 | Cocos nucifera | Coconut water | Myo-inositol | Improved insulin-stimulated glucose uptake in mature adipocytes; increased insulin sensitivity and glucose uptake | [205,224] |
| 16 | Cucumis melo | Seed | Gallic acid | Improved glucose transporters and insulin sensitivity through PPAR-γ and Akt signaling | [192,225] |
| Vanillic acid | Translocation of GLUT-4 via the AMPK-dependent pathway | [225,226] | |||
| 4-Hydroxybenzoic acid | Increasing serum insulin levels and liver glycogen content | [225,227] | |||
| 17 | Dillenia indica | Leaf | 3,5,7,-Trihydroxy-2-(4-hydroxy-benzyl)-chroman-4-one | Enhanced serum insulin level and reduced fasting blood glucose level | [228] |
| Betulinic acid, Quercetin, β-sitosterol, Stigmasterol | Inhibiting activities of α-amylase and α-glucosidase | [191] | |||
| 18 | Diospyros malabarica | Leaf | Betulinic acid | Activating AMPK, reducing blood glucose level, and stimulating mRNA expression of GLUT-4 | [86,198] |
| Fruit, Leaf | β-sitosterol | Improving insulin resistance and insulin signaling along with reducing fasting blood glucose level and glycated hemoglobin | [86,200] | ||
| 19 | Elaeocarpus floribundus | Leaf | Myricitrin | α-amylase inhibitory actions | [229] |
| 20 | Limonia acidissima | Whole Plant | Stigmasterol | lowering serum glucose level | [230,231] |
| Umbelliferone | Improving glucose uptake | [231,232] | |||
| Lupeol | Improving serum insulin level, reducing glycated hemoglobin, serum glucose | [230,231] | |||
| β-sitosterol | Improving insulin resistance and insulin signaling along with reducing fasting blood glucose level and glycated hemoglobin | [200,231] | |||
| 21 | Litchi chinensis | Fruit | Epicatechin | Promoting β-cell regeneration, enhancement of glucose consumption in HepG2 cells | [122] |
| Seed | (2R)-naringenin-7-O-(3-O-α-L-rhamnopyranosyl-β -D-glucopyranoside); narirutin; quercetin; phlorizin | dose-dependent α-glucosidase inhibitory actions | |||
| Fruit | Oligonol | Ameliorating insulin resistance | |||
| Flower | Gentisic acid | Inhibiting α-amylase and α-glucosidase | [233,234] | ||
| Epicatechin | Promoting β-cell regeneration, enhancement of glucose consumption in HepG2 cells | [122,233,235] | |||
| 22 | Mangifera indica | Kernel | 6-O-galloyl-5′-hydroxy mangiferin; mangiferin; 5-hydroxy mangiferin; methyl gallate | Reducing the blood glucose levels | [236] |
| Leaf | 1,2,3,4,6 Penta-O-galloyl-β-d-glucose | Inhibiting 11-β-HSD-1 and ameliorating high-fat diet-induced diabetes | [237] | ||
| 23 | Manilkara zapota | Fruit peel | Gallic acid | Improved glucose transporters and insulin sensitivity through PPAR-γ and Akt signaling | [192,238] |
| Ellagic acid | Stimulating insulin secretion and decreases glucose intolerance by acting on β-cells of the pancreas | [71,238] | |||
| Ferulic acid | Restoring blood glucose and serum insulin level; improving insulin sensitivity, hepatic glycogenesis, glucose tolerance, and insulin tolerance along with reducing the activity of glycogen synthase and glucokinase and also increasing activity of glycogen phosphorylase and enzymes of gluconeogenesis (PEPCK and G6Pase) | [194,238] | |||
| Catechin | α-glucosidase inhibitory actions | [189,238] | |||
| Epicatechin | Promoting β-cell regeneration | [235,238] | |||
| Quercetin | Improving insulin secretion | [188,238] | |||
| 24 | Musa sapientum | Pulp | Quercetin | Improving insulin secretion | [188,239] |
| Pectin | Reducing blood glucose level, improving liver glycogen | [188,239] | |||
| Leaf | β-amyrin | Improvement in blood sugar level and plasma insulin along with preservation of β-cell integrity | [206,239] | ||
| Lanosterol | Reducing blood glucose level | [239,240] | |||
| 25 | Phyllanthus embelica | Fruit | Coliragin | Regulating fasting blood glucose, plasma insulin, and glycated hemoglobin (HbA1c) level | [241,242] |
| Gallic acid | Improved glucose transporters and insulin sensitivity through PPAR-γ and Akt signaling | [192,242] | |||
| Ellagic acid | Stimulating insulin secretion and decreases glucose intolerance by acting on β-cells of the pancreas | [71,242] | |||
| Cinnamic Acid | Stimulation of insulin secretion, enhancement of insulin signaling pathway, the activity of pancreatic β-cell and glucose uptake, inhibition of hepatic gluconeogenesis, protein glycation, and insulin fibrillation along with the delay of carbohydrate digestion and glucose absorption | [243,244] | |||
| 26 | Psidium guajava | Leaf | Guaijaverin | Inhibiting the activity of DPP-4 | [245] |
| Avicularin | Inhibiting glucose uptake mediated by GLUT-4 | ||||
| 27 | Punica granatum | Rind | Valoneic acid dilactone | Inhibiting the activity of aldose reductase and α-amylase enzyme, ameliorating blood glucose levels by inhibiting protein tyrosine phosphatase 1B (PTP1B), improved insulin secretion from pancreatic β cells or its release from the bound form along with insulin-mimetic actions along with rejuvenation of glucose utilization technique | [149] |
| Fruit | Caffeic acid | Reducing blood glucose concentration by inhibiting the activity of glucose-6-phosphatase and increasing insulin secretion | [246,247] | ||
| Fruit, pericarp | Quercetin | Increased glucose uptake via regulation of the AMPK pathway along with improved GLUT-4 expression and regeneration of β-cells in the pancreatic islets | [210,247] | ||
| Rutin | Reducing blood glucose, elevating glycogen concentration | [210,212,247] | |||
| Catechin | α-glucosidase inhibitory actions | [189,247] | |||
| Fruit, rind, flower | Gallic acid | Improved glucose transporters and insulin sensitivity through PPAR-γ and Akt signaling | [192,247] | ||
| Fruit, seed, flower | Ellagic acid | Stimulating insulin secretion and decreases glucose intolerance by acting on β-cells of the pancreas | [71,247,248] | ||
| Seed | Punicic acid | Increasing serum insulin levels and modulating glucose homeostasis | [247,249] | ||
| Flower | Tricetin 4′-O-β-glucopyranoside | Inhibiting activities of α-amylase and α-glucosidase | [248] | ||
| Tricetin | Inhibiting activities of α-amylase and α-glucosidase | [248] | |||
| Ursolic acid | Lowering blood glucose level | [197,247] | |||
| Mascilinic acid | Improving insulin resistance, hepatic glycogen content, and inhibiting glycogen phosphorylase activity in HepG2 cells | [247,250] | |||
| Luteolin | Regulating blood glucose, HbA1c, and insulin level | [248,251] | |||
| Fruit, Leaf, Root, Bark | Punicalin | α-glucosidase inhibitory actions | [247,252] | ||
| Leaf | Apigenin | Reduction in serum glucose level, enhancing serum insulin level, nourishment of pancreatic β-cell | [213,222,247] | ||
| Fruit, Root, bark | Punicalagin | α-glucosidase inhibitory actions | [247,252] | ||
| 28 | Spondias mombin | Leaf | 3β-olean-12-en-3-yl (9Z)-hexadec-9-enoate | α-amylase inhibitory actions | [253] |
| Mombintane I; mombintane II | Glucose lowering and α-amylase inhibitory actions | [254] | |||
| 29 | Syzygium cumini | Leaf | Stigmasterol | Lowering serum glucose level | [230] |
| Lupeol | Improving serum insulin level, reducing glycated hemoglobin and serum glucose | ||||
| β-sitosterol | Improving insulin resistance and insulin signaling along with reducing fasting blood glucose level and glycated hemoglobin | [200,230] | |||
| 30 | Syzygium samarangense | Leaf | 2′,4′-dihydroxy-3′, 5′-dimethyl-6′-methoxychalcone | Lowering blood glucose level | [255] |
| Fruit | Vescalagin | Improving insulin resistance and glycemic metabolism | [256] | ||
| 31 | Tamarindus indica | Leaf | Limonene | Inhibiting protein glycation | [201,257] |
| Root bark | Pinitol | Insulin mimetic activity and regulating glucose uptake | [257,258] | ||
| Seed | β-amyrin | Improvement in blood sugar level and plasma insulin along with preservation of β-cell integrity | [206,257] | ||
| Seed, Root bark | β-sitosterol | Improving insulin resistance and insulin signaling along with reducing fasting blood glucose level and glycated hemoglobin | [200,257] | ||
| Fruit | Lupeol | Improving serum insulin level, reducing glycated hemoglobin, serum glucose | [230,257] | ||
| Naringenin | Increased glucose uptake | [217,257] | |||
| Catechin | α-glucosidase inhibitory actions | [189,257] | |||
| Epicatechin | Promoting β-cell regeneration | [235,257] | |||
| 32 | Ziziphus mauritiana | Aerial Part | Berberine | Insulin mimetic activity and improving the action of insulin by activating AMPK, decreasing insulin resistance through protein kinase C-dependent up-regulation of insulin receptor expression, enhancing GLP-1 secretion and modulating its release, inhibiting actions of DPP-4 | [203] |
| Quercetin | Improving insulin secretion | [188,203] | |||
| Diosgenin | Reducing intestinal disaccharidases, blood glucose level, and activity of α-Glucosidase along with modification of hepatocyte absorption of glucose and insulin resistance and insulin secretion | [203,259] | |||
| Kaempferol | Inhibiting the activities of α-amylase and α-glucosidase along with glucose-lowering activity | [203,212] | |||
| Fruit | Ursolic acid | Lowering blood glucose level | [197,203] | ||
| Oleanolic acid | Improved blood glucose and serum insulin levels | [203,260] | |||
| Leaf | Swertisin | Generating new β-cells | [203,261] |
| Alkaloids | Coumarins | Flavonoids | Polyphenols and Phenolic Compounds | Terpenes and Triterpenoids | Fatty Acids | Tannins and Ellagitannins | Xanthones | Miscellaneous |
|---|---|---|---|---|---|---|---|---|
| Aegelin 2 | Scopoletin | Swertisin | Ferulic acid | Ursolic acid | n-hexadecanoic acid | Punicalagin | 5-hydroxy mangiferin | Pinitol (Cyclic polyol) |
| Aegeline | Umbelliferon | Apigenin | Gallic acid | Limonene | Cinnamic Acid | Punicalin | 6-O-galloyl-5′-hydroxy mangiferin | Neohesperidin (Glycoside) |
| Berberine | Avicularin | Gentisic acid | Arjunolic acid | Oleic acid | Vescalagin | Mangiferin | Citrulline (Non-essential amino acid) | |
| Catechin | p-coumaric acid | Betulinic acid | Punicic acid | Punicalagin | Diosgenin (Steroidal sapogenin) | |||
| Didymin | Carvacrol | Lanosterol | Valoneic acid dilactone | |||||
| Diosmin | Methyl gallate | Lupeol | ||||||
| Tangeretin | Vanillic acid | Mascilinic acid | ||||||
| Epicatechin | Vanillin | Oleanolic acid | ||||||
| Guaijaverin | Mombintane I | β-amyrin | ||||||
| Hesperetin | Mombintane II | α-Pinene | ||||||
| Hesperidin | Sitosterol | |||||||
| Isoquercetin | Stigmasterol | |||||||
| Kaempferol | β-sitosterol | |||||||
| Luteolin | Phlorizin | |||||||
| Morin | 6-gingerol | |||||||
| Myricitrin | Caffeic acid | |||||||
| Naringenin | Oligonol | |||||||
| Naringin | Ellagic Acid | |||||||
| Nobiletin | ||||||||
| Quercetin | ||||||||
| Rhoifolin | ||||||||
| Rutin | ||||||||
| 8-Prenylnaringenin |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alam, S.; Dhar, A.; Hasan, M.; Richi, F.T.; Emon, N.U.; Aziz, M.A.; Mamun, A.A.; Chowdhury, M.N.R.; Hossain, M.J.; Kim, J.K.; et al. Antidiabetic Potential of Commonly Available Fruit Plants in Bangladesh: Updates on Prospective Phytochemicals and Their Reported MoAs. Molecules 2022, 27, 8709. https://doi.org/10.3390/molecules27248709
Alam S, Dhar A, Hasan M, Richi FT, Emon NU, Aziz MA, Mamun AA, Chowdhury MNR, Hossain MJ, Kim JK, et al. Antidiabetic Potential of Commonly Available Fruit Plants in Bangladesh: Updates on Prospective Phytochemicals and Their Reported MoAs. Molecules. 2022; 27(24):8709. https://doi.org/10.3390/molecules27248709
Chicago/Turabian StyleAlam, Safaet, Anik Dhar, Muhib Hasan, Fahmida Tasnim Richi, Nazim Uddin Emon, Md. Abdul Aziz, Abdullah Al Mamun, Md. Nafees Rahman Chowdhury, Md. Jamal Hossain, Jin Kyu Kim, and et al. 2022. "Antidiabetic Potential of Commonly Available Fruit Plants in Bangladesh: Updates on Prospective Phytochemicals and Their Reported MoAs" Molecules 27, no. 24: 8709. https://doi.org/10.3390/molecules27248709
APA StyleAlam, S., Dhar, A., Hasan, M., Richi, F. T., Emon, N. U., Aziz, M. A., Mamun, A. A., Chowdhury, M. N. R., Hossain, M. J., Kim, J. K., Kim, B., Hasib, M. S., Zihad, S. M. N. K., Haque, M. R., Mohamed, I. N., & Rashid, M. A. (2022). Antidiabetic Potential of Commonly Available Fruit Plants in Bangladesh: Updates on Prospective Phytochemicals and Their Reported MoAs. Molecules, 27(24), 8709. https://doi.org/10.3390/molecules27248709









