Polylactide Nanoparticles as a Biodegradable Vaccine Adjuvant: A Study on Safety, Protective Immunity and Efficacy against Human Leishmaniasis Caused by Leishmania Major
Abstract
1. Introduction
2. Results
2.1. Characterization of PLA NPs
2.2. Characterization of a Recombinant H2B Protein
2.3. Adsorption Efficiency and Zeta Potential of H2B/PLA Particles
2.4. In Vitro Desorption of Adsorbed H2B
2.5. Assessment of the Antibody Response
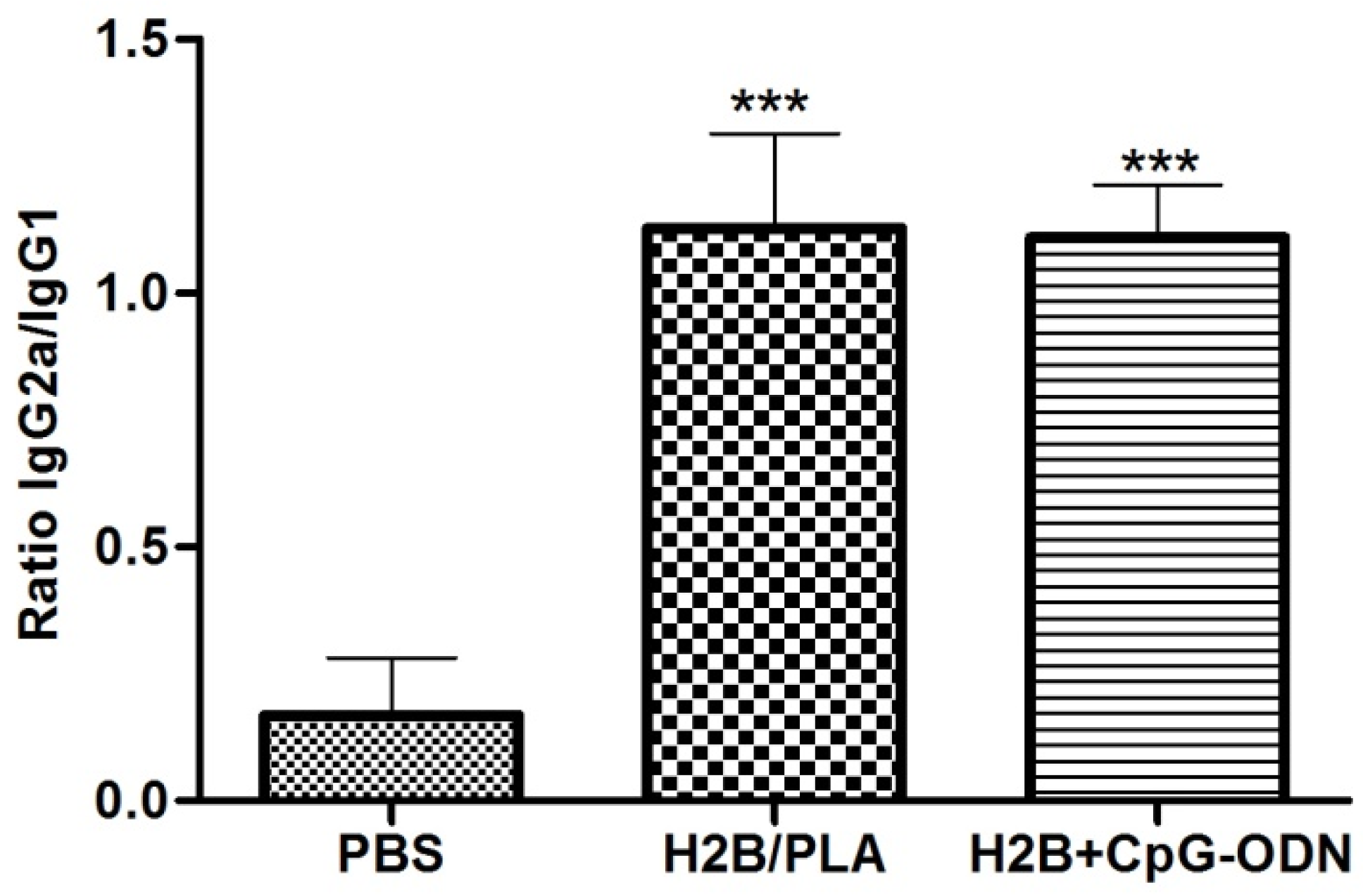
2.6. Analysis of the Anti-H2B IgG Isotype
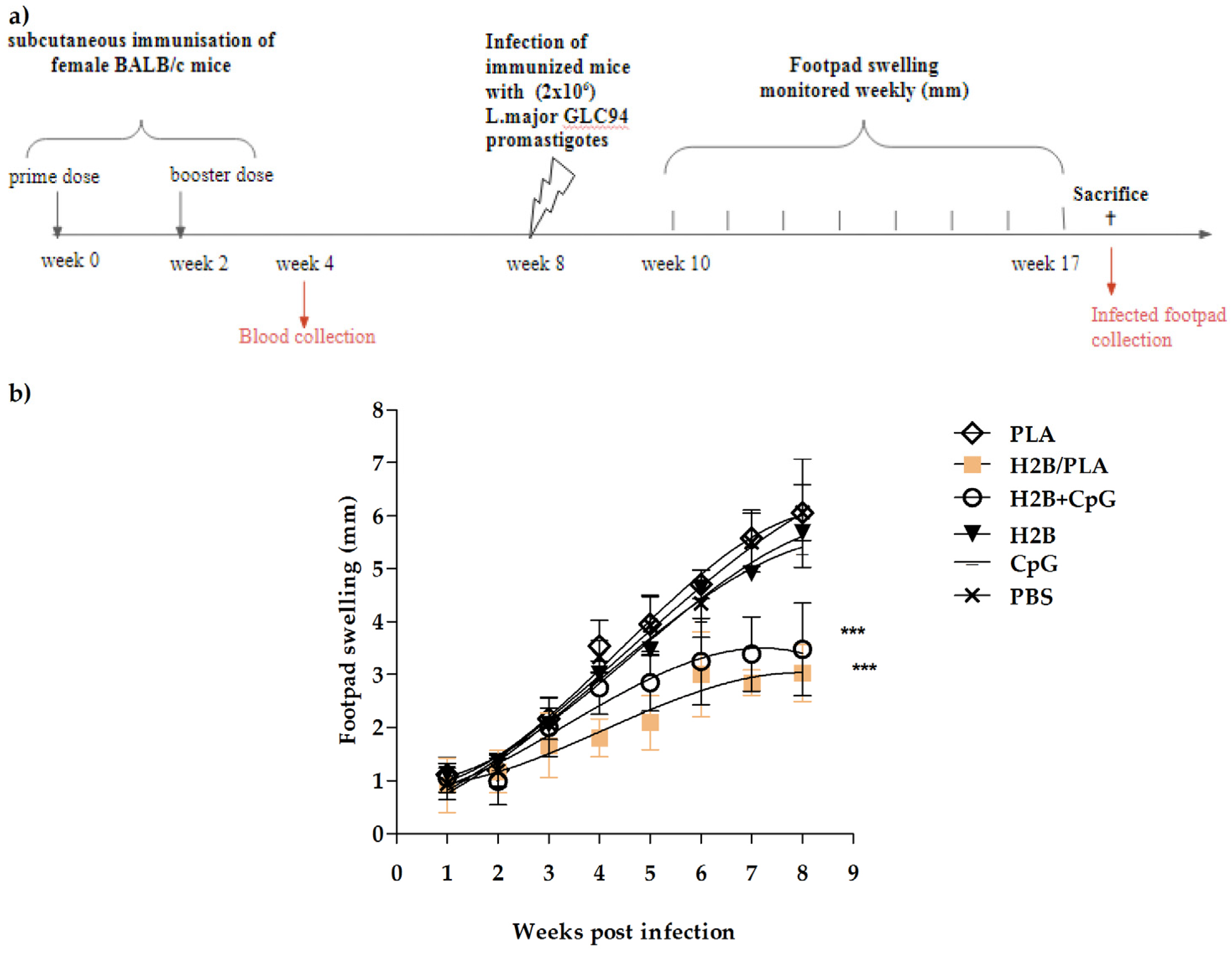
2.7. Protective Potency of H2B/PLA in Mice
2.8. Evaluating the Parasite Load in Mice
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Parasites
4.3. Mice
4.4. Preparation of PLA Nanoparticles
4.5. Expression and Purification of Recombinant Histone H2B
4.6. Synthesis of H2B/PLA, a Nanovaccine Formulation
4.7. In Vitro Release Study
4.8. In Vivo Assay
4.8.1. Immunization of BALB/c Mice
4.8.2. Identifying the IGg1 and IgG2 Isotypes
4.8.3. Parasite Infection and Development of Lesions
4.8.4. Determining the Parasite Load
5. Statistical Analysis
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Leishmaniasis. Available online: https://www.who.int/news-room/fact-sheets/detail/leishmaniasis (accessed on 8 January 2022).
- Burza, S.; Croft, S.L.; Boelaert, M. Leishmaniasis. Lancet 2018, 392, 951–970. [Google Scholar] [CrossRef] [PubMed]
- Pace, D. Leishmaniasis. J. Infect. 2014, 69 (Suppl. 1), S10–S18. [Google Scholar] [CrossRef] [PubMed]
- Bennis, I.; Thys, S.; Filali, H.; De Brouwere, V.; Sahibi, H.; Boelaert, M. Psychosocial impact of scars due to cutaneous leishmaniasis on high school students in Errachidia province, Morocco. Infect. Dis. Poverty 2017, 6, 46. [Google Scholar] [CrossRef] [PubMed]
- Himmelrich, H.; Parra-Lopez, C.; Tacchini-Cottier, F.; Louis, J.A.; Launois, P. The IL-4 rapidly produced in BALB/c mice after infection with Leishmania major down-regulates IL-12 receptor beta 2-chain expression on CD4+ T cells resulting in a state of unresponsiveness to IL-12. J. Immunol. 1998, 161, 6156–6163. [Google Scholar] [PubMed]
- Armijos, R.X.; Weigel, M.M.; Calvopina, M.; Hidalgo, A.; Cevallos, W.; Correa, J. Safety, immunogenecity, and efficacy of an autoclaved Leishmania amazonensis vaccine plus BCG adjuvant against New World cutaneous leishmaniasis. Vaccine 2004, 22, 1320–1326. [Google Scholar] [CrossRef]
- Elfaki, M.E.; Khalil, E.A.; De Groot, A.S.; Musa, A.M.; Gutierrez, A.; Younis, B.M.; Salih, K.A.; El-Hassan, A.M. Immunogenicity and immune modulatory effects of in silico predicted L. donovani candidate peptide vaccines. Hum. Vaccin. Immunother. 2012, 8, 1769–1774. [Google Scholar] [CrossRef]
- Khalil, E.A.; Musa, A.M.; Modabber, F.; El-Hassan, A.M. Safety and immunogenicity of a candidate vaccine for visceral leishmaniasis (Alum-precipitated autoclaved Leishmania major + BCG) in children: An extended phase II study. Ann. Trop. Paediatr. 2006, 26, 357–361. [Google Scholar] [CrossRef]
- Duthie, M.S.; Raman, V.S.; Piazza, F.M.; Reed, S.G. The development and clinical evaluation of second-generation leishmaniasis vaccines. Vaccine 2012, 30, 134–141. [Google Scholar] [CrossRef]
- Gillespie, P.M.; Beaumier, C.M.; Strych, U.; Hayward, T.; Hotez, P.J.; Bottazzi, M.E. Status of vaccine research and development of vaccines for leishmaniasis. Vaccine 2016, 34, 2992–2995. [Google Scholar] [CrossRef]
- Choudhury, R.; Das, P.; De, T.; Chakraborti, T. 115 kDa serine protease confers sustained protection to visceral leishmaniasis caused by Leishmania donovani via IFN-gamma induced down-regulation of TNF-alpha mediated MMP-9 activity. Immunobiology 2013, 218, 114–126. [Google Scholar] [CrossRef]
- Grimaldi, G., Jr.; Teva, A.; Dos-Santos, C.B.; Santos, F.N.; Pinto, I.D.; Fux, B.; Leite, G.R.; Falqueto, A. Field trial of efficacy of the Leish-tec(R) vaccine against canine leishmaniasis caused by Leishmania infantum in an endemic area with high transmission rates. PLoS ONE 2017, 12, e0185438. [Google Scholar] [CrossRef] [PubMed]
- Moafi, M.; Rezvan, H.; Sherkat, R.; Taleban, R. Leishmania Vaccines Entered in Clinical Trials: A Review of Literature. Int. J. Prev. Med. 2019, 10, 95. [Google Scholar] [CrossRef] [PubMed]
- Agallou, M.; Margaroni, M.; Karagouni, E. Cellular vaccination with bone marrow-derived dendritic cells pulsed with a peptide of Leishmania infantum KMP-11 and CpG oligonucleotides induces protection in a murine model of visceral leishmaniasis. Vaccine 2011, 29, 5053–5064. [Google Scholar] [CrossRef] [PubMed]
- Azmi, F.; Ahmad Fuaad, A.A.; Skwarczynski, M.; Toth, I. Recent progress in adjuvant discovery for peptide-based subunit vaccines. Hum. Vaccin Immunother. 2014, 10, 778–796. [Google Scholar] [CrossRef] [PubMed]
- Doroud, D.; Rafati, S. Leishmaniasis: Focus on the design of nanoparticulate vaccine delivery systems. Expert Rev. Vaccines 2012, 11, 69–86. [Google Scholar] [CrossRef]
- Xia, Y.; Fan, Q.; Hao, D.; Wu, J.; Ma, G.; Su, Z. Chitosan-based mucosal adjuvants: Sunrise on the ocean. Vaccine 2015, 33, 5997–6010. [Google Scholar] [CrossRef]
- Black, M.; Trent, A.; Tirrell, M.; Olive, C. Advances in the design and delivery of peptide subunit vaccines with a focus on toll-like receptor agonists. Expert Rev. Vaccines 2010, 9, 157–173. [Google Scholar] [CrossRef]
- Rice-Ficht, A.C.; Arenas-Gamboa, A.M.; Kahl-McDonagh, M.M.; Ficht, T.A. Polymeric particles in vaccine delivery. Curr. Opin. Microbiol. 2010, 13, 106–112. [Google Scholar] [CrossRef]
- Mallapragada, S.K.; Narasimhan, B. Immunomodulatory biomaterials. Int. J. Pharm. 2008, 364, 265–271. [Google Scholar] [CrossRef]
- Raman, V.S.; Duthie, M.S.; Fox, C.B.; Matlashewski, G.; Reed, S.G. Adjuvants for Leishmania vaccines: From models to clinical application. Front. Immunol. 2012, 3, 144. [Google Scholar] [CrossRef]
- Daraee, H.; Etemadi, A.; Kouhi, M.; Alimirzalu, S.; Akbarzadeh, A. Application of liposomes in medicine and drug delivery. Artif. Cells Nanomed. Biotechnol. 2016, 44, 381–391. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Jiao, Y.; Wang, Y.; Zhou, C.; Zhang, Z. Polysaccharides-based nanoparticles as drug delivery systems. Adv. Drug Deliv. Rev. 2008, 60, 1650–1662. [Google Scholar] [CrossRef] [PubMed]
- Makadia, H.K.; Siegel, S.J. Poly Lactic-co-Glycolic Acid (PLGA) as Biodegradable Controlled Drug Delivery Carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef] [PubMed]
- Ataman-Onal, Y.; Munier, S.; Ganee, A.; Terrat, C.; Durand, P.Y.; Battail, N.; Martinon, F.; Le Grand, R.; Charles, M.H.; Delair, T.; et al. Surfactant-free anionic PLA nanoparticles coated with HIV-1 p24 protein induced enhanced cellular and humoral immune responses in various animal models. J. Control. Release 2006, 112, 175–185. [Google Scholar] [CrossRef] [PubMed]
- Lamalle-Bernard, D.; Munier, S.; Compagnon, C.; Charles, M.H.; Kalyanaraman, V.S.; Delair, T.; Verrier, B.; Ataman-Onal, Y. Coadsorption of HIV-1 p24 and gp120 proteins to surfactant-free anionic PLA nanoparticles preserves antigenicity and immunogenicity. J. Control. Release 2006, 115, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Venkataprasad, N.; Coombes, A.G.; Singh, M.; Rohde, M.; Wilkinson, K.; Hudecz, F.; Davis, S.S.; Vordermeier, H.M. Induction of cellular immunity to a mycobacterial antigen adsorbed on lamellar particles of lactide polymers. Vaccine 1999, 17, 1814–1819. [Google Scholar] [CrossRef]
- Handman, E. Leishmaniasis: Current status of vaccine development. Clin. Microbiol. Rev. 2001, 14, 229–243. [Google Scholar] [CrossRef]
- Khorasanizadeh, S. The nucleosome: From genomic organization to genomic regulation. Cell 2004, 116, 259–272. [Google Scholar] [CrossRef]
- Maalej, I.A.; Chenik, M.; Louzir, H.; Ben Salah, A.; Bahloul, C.; Amri, F.; Dellagi, K. Comparative evaluation of ELISAs based on ten recombinant or purified Leishmania antigens for the serodiagnosis of Mediterranean visceral leishmaniasis. Am. J. Trop. Med. Hyg. 2003, 68, 312–320. [Google Scholar] [CrossRef]
- Chenik, M.; Louzir, H.; Ksontini, H.; Dilou, A.; Abdmouleh, I.; Dellagi, K. Vaccination with the divergent portion of the protein histone H2B of Leishmania protects susceptible BALB/c mice against a virulent challenge with Leishmania major. Vaccine 2006, 24, 2521–2529. [Google Scholar] [CrossRef]
- Meddeb-Garnaoui, A.; Toumi, A.; Ghelis, H.; Mahjoub, M.; Louzir, H.; Chenik, M. Cellular and humoral responses induced by Leishmania histone H2B and its divergent and conserved parts in cutaneous and visceral leishmaniasis patients, respectively. Vaccine 2010, 28, 1881–1886. [Google Scholar] [CrossRef] [PubMed]
- Benhnini, F.; Chenik, M.; Laouini, D.; Louzir, H.; Cazenave, P.A.; Dellagi, K. Comparative evaluation of two vaccine candidates against experimental leishmaniasis due to Leishmania major infection in four inbred mouse strains. Clin. Vaccine Immunol. 2009, 16, 1529–1537. [Google Scholar] [CrossRef] [PubMed]
- Vijaya BJoshi, S.M.G.; Salem, A.K. Biodegradable particles as vaccine antigen delivery systems for stimulating cellular immune responses. Hum. Vaccines Immunother. 2013, 9, 2584–2590. [Google Scholar] [CrossRef]
- Srivastava, S.; Shankar, P.; Mishra, J.; Singh, S. Possibilities and challenges for developing a successful vaccine for leishmaniasis. Parasites Vectors 2016, 9, 277. [Google Scholar] [CrossRef]
- Gheibi Hayat, S.M.; Darroudi, M. Nanovaccine: A novel approach in immunization. J. Cell. Physiol. 2019, 234, 12530–12536. [Google Scholar] [CrossRef]
- Yasinzai, M.; Khan, M.; Nadhman, A.; Shahnaz, G. Drug resistance in leishmaniasis: Current drug-delivery systems and future perspectives. Future Med. Chem. 2013, 5, 1877–1888. [Google Scholar] [CrossRef]
- Shive, M.S.; Anderson, J.M. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv. Drug Deliv. Rev. 1997, 28, 5–24. [Google Scholar] [CrossRef]
- Soto, M.; Requena, J.M.; Quijada, L.; Perez, M.J.; Nieto, C.G.; Guzman, F.; Patarroyo, M.E.; Alonso, C. Antigenicity of the Leishmania infantum histones H2B and H4 during canine viscerocutaneous leishmaniasis. Clin. Exp. Immunol. 1999, 115, 342–349. [Google Scholar] [CrossRef]
- Lemery, E.; Briancon, S.; Chevalier, Y.; Oddos, T.; Gohier, A.; Boyron, O.; Bolzinger, M.A. Surfactants have multi-fold effects on skin barrier function. Eur. J. Dermatol. 2015, 25, 424–435. [Google Scholar] [CrossRef]
- Ronningen, T.; Shah, A.; Oldenburg, A.R.; Vekterud, K.; Delbarre, E.; Moskaug, J.O.; Collas, P. Prepatterning of differentiation-driven nuclear lamin A/C-associated chromatin domains by GlcNAcylated histone H2B. Genome Res. 2015, 25, 1825–1835. [Google Scholar] [CrossRef]
- Foged, C.; Brodin, B.; Frokjaer, S.; Sundblad, A. Particle size and surface charge affect particle uptake by human dendritic cells in an in vitro model. Int. J. Pharm. 2005, 298, 315–322. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.K.; Asthana, S.; Jaiswal, A.K.; Kumar, V.; Verma, A.K.; Shukla, P.; Dwivedi, P.; Dube, A.; Mishra, P.R. Exploitation of lectinized lipo-polymerosome encapsulated Amphotericin B to target macrophages for effective chemotherapy of visceral leishmaniasis. Bioconjugate Chem. 2014, 25, 1091–1102. [Google Scholar] [CrossRef] [PubMed]
- Margaroni, M.; Agallou, M.; Kontonikola, K.; Karidi, K.; Kammona, O.; Kiparissides, C.; Gaitanaki, C.; Karagouni, E. PLGA nanoparticles modified with a TNFalpha mimicking peptide, soluble Leishmania antigens and MPLA induce T cell priming in vitro via dendritic cell functional differentiation. Eur. J. Pharm. Biopharm. 2016, 105, 18–31. [Google Scholar] [CrossRef]
- Kebaier, C.; Louzir, H.; Chenik, M.; Ben Salah, A.; Dellagi, K. Heterogeneity of wild Leishmania major isolates in experimental murine pathogenicity and specific immune response. Infect. Immun. 2001, 69, 4906–4915. [Google Scholar] [CrossRef]
- Pavot, V.; Rochereau, N.; Primard, C.; Genin, C.; Perouzel, E.; Lioux, T.; Paul, S.; Verrier, B. Encapsulation of Nod1 and Nod2 receptor ligands into poly(lactic acid) nanoparticles potentiates their immune properties. J. Control. Release 2013, 167, 60–67. [Google Scholar] [CrossRef]
- Banerjee, K.; Klasse, P.J.; Sanders, R.W.; Pereyra, F.; Michael, E.; Lu, M.; Walker, B.D.; Moore, J.P. IgG subclass profiles in infected HIV type 1 controllers and chronic progressors and in uninfected recipients of Env vaccines. AIDS Res. Hum. Retrovir. 2010, 26, 445–458. [Google Scholar] [CrossRef] [PubMed]
- Launois, P.; Conceicao-Silva, F.; Himmerlich, H.; Parra-Lopez, C.; Tacchini-Cottier, F.; Louis, J.A. Setting in motion the immune mechanisms underlying genetically determined resistance and susceptibility to infection with Leishmania major. Parasite Immunol. 1998, 20, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Liew, F.Y.; O’Donnell, C.A. Immunology of leishmaniasis. Adv. Parasitol. 1993, 32, 161–259. [Google Scholar] [CrossRef] [PubMed]
- Reiner, S.L.; Locksley, R.M. The regulation of immunity to Leishmania major. Annu. Rev. Immunol. 1995, 13, 151–177. [Google Scholar] [CrossRef]
- Palma, E.; Cho, M.J. Improved systemic pharmacokinetics, biodistribution, and antitumor activity of CpG oligodeoxynucleotides complexed to endogenous antibodies in vivo. J. Control. Release 2007, 120, 95–103. [Google Scholar] [CrossRef][Green Version]
- Khatik, R.; Dwivedi, P.; Khare, P.; Kansal, S.; Dube, A.; Mishra, P.R.; Dwivedi, A.K. Development of targeted 1,2-diacyl-sn-glycero-3-phospho-l-serine-coated gelatin nanoparticles loaded with amphotericin B for improved in vitro and in vivo effect in leishmaniasis. Expert Opin. Drug Deliv. 2014, 11, 633–646. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, R.F.; Ribeiro, I.F.; Miranda-Vilela, A.L.; de Souza Filho, J.; Martins, O.P.; Cintra e Silva Dde, O.; Tedesco, A.C.; Lacava, Z.G.; Bao, S.N.; Sampaio, R.N. Leishmanicidal activity of amphotericin B encapsulated in PLGA-DMSA nanoparticles to treat cutaneous leishmaniasis in C57BL/6 mice. Exp. Parasitol. 2013, 135, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Lage, D.P.; Ribeiro, P.A.F.; Dias, D.S.; Mendonca, D.V.C.; Ramos, F.F.; Carvalho, L.M.; Steiner, B.T.; Tavares, G.S.V.; Martins, V.T.; Machado, A.S.; et al. Liposomal Formulation of ChimeraT, a Multiple T-Cell Epitope-Containing Recombinant Protein, Is a Candidate Vaccine for Human Visceral Leishmaniasis. Vaccines 2020, 8, 289. [Google Scholar] [CrossRef] [PubMed]
- Katebi, A.; Varshochian, R.; Riazi-Rad, F.; Ganjalikhani-Hakemi, M.; Ajdary, S. Combinatorial delivery of antigen and TLR agonists via PLGA nanoparticles modulates Leishmania major-infected-macrophages activation. Biomed. Pharmacother. 2021, 137, 111276. [Google Scholar] [CrossRef] [PubMed]
- Noormehr, H.; Zavaran Hosseini, A.; Soudi, S.; Beyzay, F. Enhancement of Th1 immune response against Leishmania cysteine peptidase A, B by PLGA nanoparticle. Int. Immunopharmacol. 2018, 59, 97–105. [Google Scholar] [CrossRef]
- Ayari-Riabi, S.; Trimaille, T.; Mabrouk, K.; Bertin, D.; Gigmes, D.; Benlasfar, Z.; Zaghmi, A.; Bouhaouala-Zahar, B.; Elayeb, M. Venom conjugated polylactide applied as biocompatible material for passive and active immunotherapy against scorpion envenomation. Vaccine 2016, 34, 1810–1815. [Google Scholar] [CrossRef]
- Laskay, T.; Diefenbach, A.; Rollinghoff, M.; Solbach, W. Early parasite containment is decisive for resistance to Leishmania major infection. Eur. J. Immunol. 1995, 25, 2220–2227. [Google Scholar] [CrossRef]
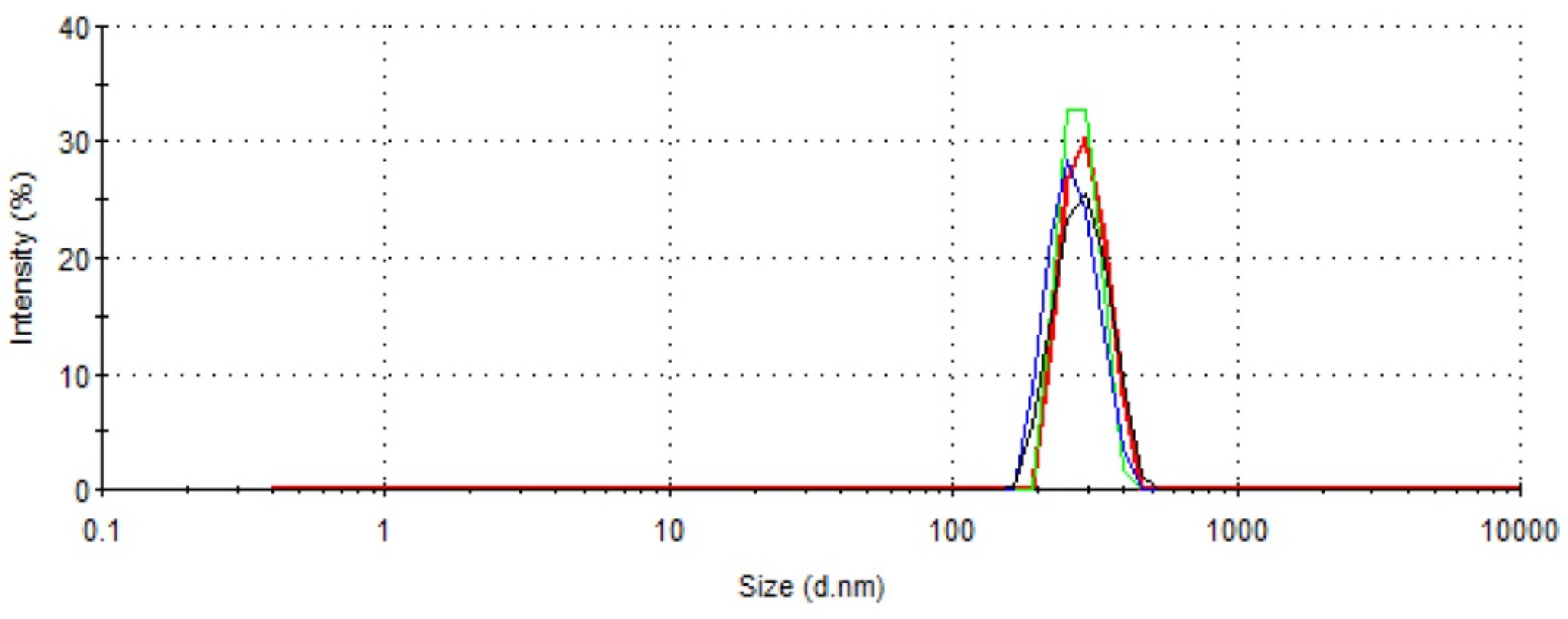
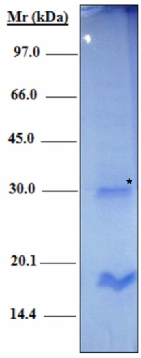
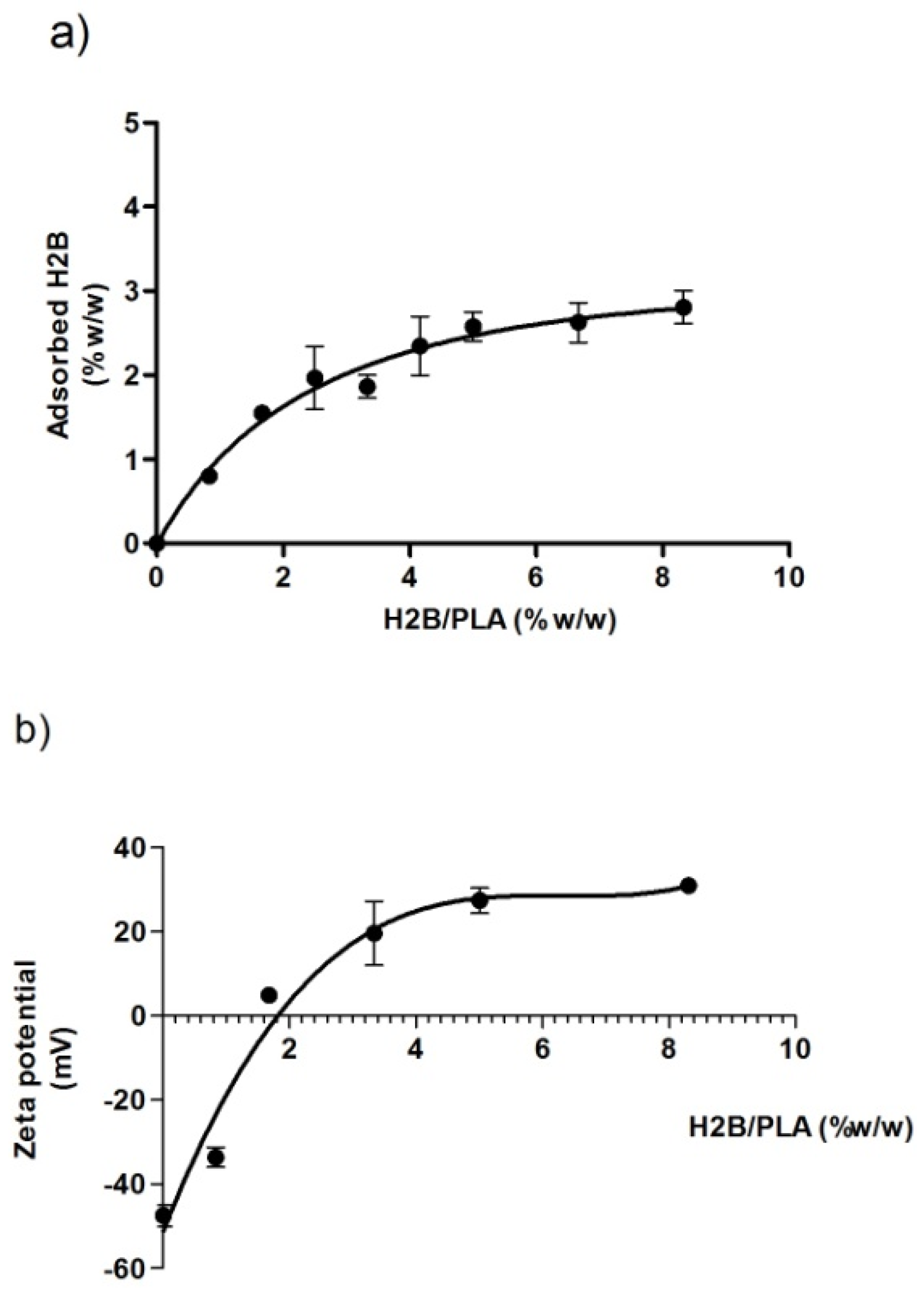
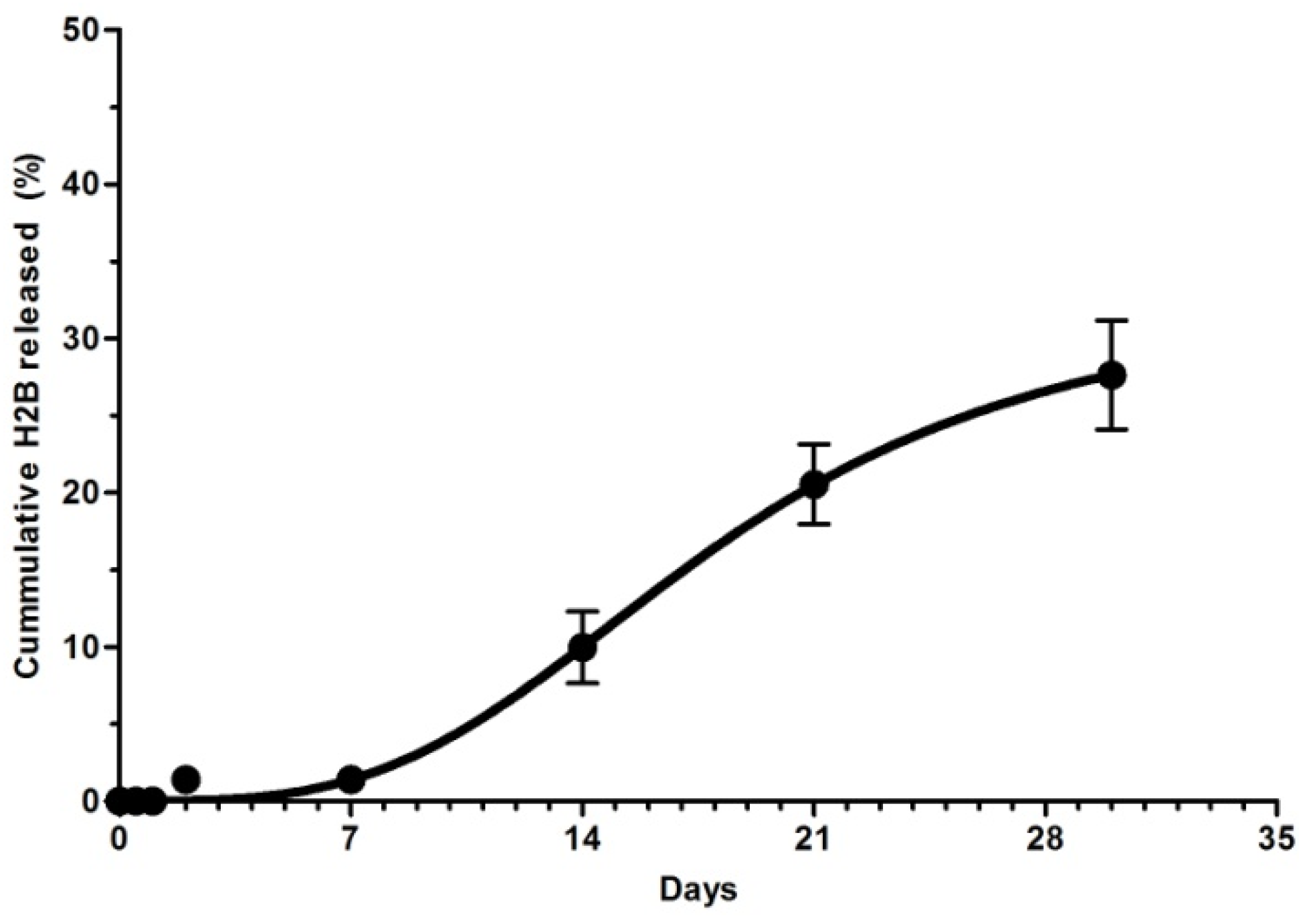


| Adsorption Capacity % w/w | Zeta Potential mV | DLS Size nm |
|---|---|---|
| 2.8 (±0.24) | 30.95 (±0.78) | 340.8 (±52) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ayari-Riabi, S.; Ben khalaf, N.; Bouhaouala-Zahar, B.; Verrier, B.; Trimaille, T.; Benlasfar, Z.; Chenik, M.; Elayeb, M. Polylactide Nanoparticles as a Biodegradable Vaccine Adjuvant: A Study on Safety, Protective Immunity and Efficacy against Human Leishmaniasis Caused by Leishmania Major. Molecules 2022, 27, 8677. https://doi.org/10.3390/molecules27248677
Ayari-Riabi S, Ben khalaf N, Bouhaouala-Zahar B, Verrier B, Trimaille T, Benlasfar Z, Chenik M, Elayeb M. Polylactide Nanoparticles as a Biodegradable Vaccine Adjuvant: A Study on Safety, Protective Immunity and Efficacy against Human Leishmaniasis Caused by Leishmania Major. Molecules. 2022; 27(24):8677. https://doi.org/10.3390/molecules27248677
Chicago/Turabian StyleAyari-Riabi, Sana, Noureddine Ben khalaf, Balkiss Bouhaouala-Zahar, Bernard Verrier, Thomas Trimaille, Zakaria Benlasfar, Mehdi Chenik, and Mohamed Elayeb. 2022. "Polylactide Nanoparticles as a Biodegradable Vaccine Adjuvant: A Study on Safety, Protective Immunity and Efficacy against Human Leishmaniasis Caused by Leishmania Major" Molecules 27, no. 24: 8677. https://doi.org/10.3390/molecules27248677
APA StyleAyari-Riabi, S., Ben khalaf, N., Bouhaouala-Zahar, B., Verrier, B., Trimaille, T., Benlasfar, Z., Chenik, M., & Elayeb, M. (2022). Polylactide Nanoparticles as a Biodegradable Vaccine Adjuvant: A Study on Safety, Protective Immunity and Efficacy against Human Leishmaniasis Caused by Leishmania Major. Molecules, 27(24), 8677. https://doi.org/10.3390/molecules27248677






