Advances in the Understanding of Two-Pore Domain TASK Potassium Channels and Their Potential as Therapeutic Targets
Abstract
1. Introduction
2. The Structure, Localization, and Electrophysiological Properties of TASK Channels
2.1. Structure
2.2. Expression and Localization
2.3. Electrophysiological Properties
3. Gating Profiles
4. The Biological Roles of TASK Channels
4.1. Breathing Rhythm
4.2. Pulmonary Artery Hypertension
4.3. Cardiac Arrhythmia
4.4. Aldosterone Secretion
4.5. Pain
4.6. Anesthetics
4.6.1. Volatile Anesthetics
4.6.2. Local Anesthetics
4.7. Cancers
4.8. Neurological Activities and/or Disorders
4.8.1. Sleep
4.8.2. Mental Retardation
4.8.3. Depression
4.9. Other Roles
5. Development of Selectively Targeted Compounds
5.1. Compounds That Target the TASK-1 Channel
5.2. Compounds That Target the TASK-3 Channel
6. Conclusions and Future Research Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sepúlveda, F.V.; Pablo Cid, L.; Teulon, J.; Niemeyer, M.I. Molecular aspects of structure, gating, and physiology of pH-sensitive background K2P and Kir K+-transport channels. Physiol. Rev. 2015, 95, 179–217. [Google Scholar] [CrossRef]
- Kuang, Q.; Purhonen, P.; Hebert, H. Structure of potassium channels. Cell. Mol. Life Sci. CMLS 2015, 72, 3677–3693. [Google Scholar] [CrossRef]
- González, C.; Baez-Nieto, D.; Valencia, I.; Oyarzún, I.; Rojas, P.; Naranjo, D.; Latorre, R. K+ channels: Function-structural overview. Compr. Physiol. 2012, 2, 2087–2149. [Google Scholar] [PubMed]
- Kim, D.M.; Nimigean, C.M. Voltage-Gated Potassium Channels: A Structural Examination of Selectivity and Gating. Cold Spring Harb. Perspect. Biol. 2016, 8, a029231. [Google Scholar] [CrossRef]
- Trombetta-Lima, M.; Krabbendam, I.E.; Dolga, A.M. Calcium-activated potassium channels: Implications for aging and age-related neurodegeneration. Int. J. Biochem. Cell Biol. 2020, 123, 105748. [Google Scholar] [CrossRef]
- Hibino, H.; Inanobe, A.; Furutani, K.; Murakami, S.; Findlay, I.; Kurachi, Y. Inwardly rectifying potassium channels: Their structure, function, and physiological roles. Physiol. Rev. 2010, 90, 291–366. [Google Scholar] [CrossRef] [PubMed]
- Lesage, F.; Lazdunski, M. Molecular and functional properties of two-pore-domain potassium channels. Am. J. Physiol. Renal Physiol. 2000, 279, F793–F801. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, S.A.; Bockenhauer, D.; O’Kelly, I.; Zilberberg, N. Potassium leak channels and the KCNK family of two-P-domain subunits. Nat. Rev. Neurosci. 2001, 2, 175–184. [Google Scholar] [CrossRef]
- Huang, L.; Xu, G.; Jiang, R.; Luo, Y.; Zuo, Y.; Liu, J. Development of Non-opioid Analgesics Targeting Two-pore Domain Potassium Channels. Curr. Neuropharmacol. 2022, 20, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Lesage, F.; Guillemare, E.; Fink, M.; Duprat, F.; Lazdunski, M.; Romey, G.; Barhanin, J. TWIK-1, a ubiquitous human weakly inward rectifying K+ channel with a novel structure. EMBO J. 1996, 15, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Duprat, F.; Lesage, F.; Fink, M.; Reyes, R.; Heurteaux, C.; Lazdunski, M. TASK, a human background K+ channel to sense external pH variations near physiological pH. EMBO J. 1997, 16, 5464–5471. [Google Scholar] [CrossRef]
- Rajan, S.; Wischmeyer, E.; Xin Liu, G.; Preisig-Müller, R.; Daut, J.; Karschin, A.; Derst, C. TASK-3, a novel tandem pore domain acid-sensitive K+ channel. An extracellular histiding as pH sensor. J. Biol. Chem. 2000, 275, 16650–16657. [Google Scholar] [CrossRef] [PubMed]
- Ashmole, I.; Goodwin, P.A.; Stanfield, P.R. TASK-5, a novel member of the tandem pore K+ channel family. Pflugers Arch. 2001, 442, 828–833. [Google Scholar] [CrossRef]
- Brohawn, S.G.; del Mármol, J.; MacKinnon, R. Crystal structure of the human K2P TRAAK, a lipid- and mechano-sensitive K+ ion channel. Science 2012, 335, 436–441. [Google Scholar] [CrossRef] [PubMed]
- Cotten, J.F. TASK-1 (KCNK3) and TASK-3 (KCNK9) tandem pore potassium channel antagonists stimulate breathing in isoflurane-anesthetized rats. Anesth. Analg. 2013, 116, 810–816. [Google Scholar] [CrossRef] [PubMed]
- Talley, E.M.; Lei, Q.; Sirois, J.E.; Bayliss, D.A. TASK-1, a two-pore domain K+ channel, is modulated by multiple neurotransmitters in motoneurons. Neuron 2000, 25, 399–410. [Google Scholar] [CrossRef]
- Medhurst, A.D.; Rennie, G.; Chapman, C.G.; Meadows, H.; Duckworth, M.D.; Kelsell, R.E.; Gloger, I.I.; Pangalos, M.N. Distribution analysis of human two pore domain potassium channels in tissues of the central nervous system and periphery. Brain Res. Mol. Brain Res. 2001, 86, 101–114. [Google Scholar] [CrossRef] [PubMed]
- Olschewski, A.; Veale, E.L.; Nagy, B.M.; Nagaraj, C.; Kwapiszewska, G.; Antigny, F.; Lambert, M.; Humbert, M.; Czirják, G.; Enyedi, P.; et al. TASK-1 (KCNK3) channels in the lung: From cell biology to clinical implications. Eur. Respir. J. 2017, 50, 1700754. [Google Scholar] [CrossRef]
- Koizumi, H.; Smerin, S.E.; Yamanishi, T.; Moorjani, B.R.; Zhang, R.; Smith, J.C. TASK channels contribute to the K+-dominated leak current regulating respiratory rhythm generation in vitro. J. Neurosci. 2010, 30, 4273–4284. [Google Scholar] [CrossRef]
- Bayliss, D.A.; Barhanin, J.; Gestreau, C.; Guyenet, P.G. The role of pH-sensitive TASK channels in central respiratory chemoreception. Pflugers Arch. 2015, 467, 917–929. [Google Scholar] [CrossRef]
- Buckler, K.J. TASK channels in arterial chemoreceptors and their role in oxygen and acid sensing. Pflügers Arch.-Eur. J. Physiol. 2015, 467, 1013–1025. [Google Scholar] [CrossRef]
- Cunningham, K.P.; MacIntyre, D.E.; Mathie, A.; Veale, E.L. Effects of the ventilatory stimulant, doxapram on human TASK-3 (KCNK9, K2P9.1) channels and TASK-1 (KCNK3, K2P3.1) channels. Acta Physiol. 2020, 228, e13361. [Google Scholar] [CrossRef]
- Kim, D.; Cavanaugh, E.J.; Kim, I.; Carroll, J.L. Heteromeric TASK-1/TASK-3 is the major oxygen-sensitive background K+ channel in rat carotid body glomus cells. J. Physiol. 2009, 587 Pt 12, 2963–2975. [Google Scholar] [CrossRef] [PubMed]
- Trapp, S.; Aller, M.I.; Wisden, W.; Gourine, A.V. A role for TASK-1 (KCNK3) channels in the chemosensory control of breathing. J. Neurosci. 2008, 28, 8844–8850. [Google Scholar] [CrossRef] [PubMed]
- Mulkey, D.K.; Talley, E.M.; Stornetta, R.L.; Siegel, A.R.; West, G.H.; Chen, X.; Sen, N.; Mistry, A.M.; Guyenet, P.G.; Bayliss, D.A. TASK channels determine pH sensitivity in select respiratory neurons but do not contribute to central respiratory chemosensitivity. J. Neurosci. 2007, 27, 14049–14058. [Google Scholar] [CrossRef]
- Olschewski, A.; Li, Y.; Tang, B.; Hanze, J.; Eul, B.; Bohle, R.M.; Wilhelm, J.; Morty, R.E.; Brau, M.E.; Weir, E.K.; et al. Impact of TASK-1 in human pulmonary artery smooth muscle cells. Circ. Res. 2006, 98, 1072–1080. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, K.P.; Holden, R.G.; Escribano-Subias, P.M.; Cogolludo, A.; Veale, E.L.; Mathie, A. Characterization and regulation of wild-type and mutant TASK-1 two pore domain potassium channels indicated in pulmonary arterial hypertension. J. Physiol. 2019, 597, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Lambert, M.; Capuano, V.; Boet, A.; Tesson, L.; Bertero, T.; Nakhleh, M.K.; Remy, S.; Anegon, I.; Pechoux, C.; Hautefort, A.; et al. Characterization of Kcnk3-Mutated Rat, a Novel Model of Pulmonary Hypertension. Circ. Res. 2019, 125, 678–695. [Google Scholar] [CrossRef]
- Ma, L.; Roman-Campos, D.; Austin, E.D.; Eyries, M.; Sampson, K.S.; Soubrier, F.; Germain, M.; Trégouët, D.A.; Borczuk, A.; Rosenzweig, E.B.; et al. A novel channelopathy in pulmonary arterial hypertension. N. Engl. J. Med. 2013, 369, 351–361. [Google Scholar] [CrossRef]
- Navas Tejedor, P.; Tenorio Castaño, J.; Palomino Doza, J.; Arias Lajara, P.; Gordo Trujillo, G.; López Meseguer, M.; Román Broto, A.; Lapunzina Abadía, P.; Escribano Subía, P. An homozygous mutation in KCNK3 is associated with an aggressive form of hereditary pulmonary arterial hypertension. Clin. Genet. 2017, 91, 453–457. [Google Scholar] [CrossRef]
- Best, D.H.; Sumner, K.L.; Smith, B.P.; Damjanovich-Colmenares, K.; Nakayama, I.; Brown, L.M.; Ha, Y.; Paul, E.; Morris, A.; Jama, M.A.; et al. EIF2AK4 Mutations in Patients Diagnosed with Pulmonary Arterial Hypertension. Chest 2017, 151, 821–828. [Google Scholar] [CrossRef] [PubMed]
- Rosenzweig, E.B.; Abman, S.H.; Adatia, I.; Beghetti, M.; Bonnet, D.; Haworth, S.; Ivy, D.D.; Berger, R.M.F. Paediatric pulmonary arterial hypertension: Updates on definition, classification, diagnostics and management. Eur. Respir. J. 2019, 53, 1801916. [Google Scholar] [CrossRef] [PubMed]
- Higasa, K.; Ogawa, A.; Terao, C.; Shimizu, M.; Kosugi, S.; Yamada, R.; Date, H.; Matsubara, H.; Matsuda, F. A burden of rare variants in BMPR2 and KCNK3 contributes to a risk of familial pulmonary arterial hypertension. BMC Pulm. Med. 2017, 17, 57. [Google Scholar] [CrossRef] [PubMed]
- Callejo, M.; Mondejar-Parreño, G.; Morales-Cano, D.; Barreira, B.; Esquivel-Ruiz, S.; Olivencia, M.A.; Manaud, G.; Perros, F.; Duarte, J.; Moreno, L.; et al. Vitamin D deficiency downregulates TASK-1 channels and induces pulmonary vascular dysfunction. Am. J. Physiol. Lung Cell Mol. Physiol. 2020, 319, L627–L640. [Google Scholar] [CrossRef]
- Callejo, M.; Morales-Cano, D.; Mondejar-Parreño, G.; Barreira, B.; Esquivel-Ruiz, S.; Olivencia, M.A.; Moreno, L.; Cogolludo, A.; Perez-Vizcaino, F. Restoration of Vitamin D Levels Improves Endothelial Function and Increases TASK-Like K+ Currents in Pulmonary Arterial Hypertension Associated with Vitamin D Deficiency. Biomolecules 2021, 11, 795. [Google Scholar] [CrossRef]
- Antigny, F.; Hautefort, A.; Meloche, J.; Belacel-Ouari, M.; Manoury, B.; Rucker-Martin, C.; Péchoux, C.; Potus, F.; Nadeau, V.; Tremblay, E.; et al. Potassium Channel Subfamily K Member 3 (KCNK3) Contributes to the Development of Pulmonary Arterial Hypertension. Circulation 2016, 133, 1371–1385. [Google Scholar] [CrossRef] [PubMed]
- Besana, A.; Barbuti, A.; Tateyama, M.A.; Symes, A.J.; Robinson, R.B.; Feinmark, S.J. Activation of protein kinase C epsilon inhibits the two-pore domain K+ channel, TASK-1, inducing repolarization abnormalities in cardiac ventricular myocytes. J. Biol. Chem. 2004, 279, 33154–33160. [Google Scholar] [CrossRef] [PubMed]
- Gurney, A.; Manoury, B. Two-pore potassium channels in the cardiovascular system. Eur. Biophys. J. 2009, 38, 305–318. [Google Scholar] [CrossRef]
- Liang, B.; Soka, M.; Christensen, A.H.; Olesen, M.S.; Larsen, A.P.; Knop, F.K.; Wang, F.; Nielsen, J.B.; Andersen, M.N.; Humphreys, D.; et al. Genetic variation in the two-pore domain potassium channel, TASK-1, may contribute to an atrial substrate for arrhythmogenesis. J. Mol. Cell. Cardiol. 2014, 67, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Donner, B.C.; Schullenberg, M.; Geduldig, N.; Hüning, A.; Mersmann, J.; Zacharowski, K.; Kovacevic, A.; Decking, U.; Aller, M.I.; Schmidt, K.G. Functional role of TASK-1 in the heart: Studies in TASK-1-deficient mice show prolonged cardiac repolarization and reduced heart rate variability. Basic Res. Cardiol. 2011, 106, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Putzke, C.; Wemhöner, K.; Sachse, F.B.; Rinné, S.; Schlichthörl, G.; Li, X.T.; Jaé, L.; Eckhardt, I.; Wischmeyer, E.; Wulf, H.; et al. The acid-sensitive potassium channel TASK-1 in rat cardiac muscle. Cardiovasc. Res. 2007, 75, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Gierten, J.; Ficker, E.; Bloehs, R.; Schweizer, P.A.; Zitron, E.; Scholz, E.; Karle, C.; Katus, H.A.; Thomas, D. The human cardiac K2P3.1 (TASK-1) potassium leak channel is a molecular target for the class III antiarrhythmic drug amiodarone. Naunyn Schmiedebergs Arch. Pharmacol. 2010, 381, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Kraft, M.; Büscher, A.; Wiedmann, F.; L’Hoste, Y.; Haefeli, W.E.; Frey, N.; Katus, H.A.; Schmidt, C. Current Drug Treatment Strategies for Atrial Fibrillation and TASK-1 Inhibition as an Emerging Novel Therapy Option. Front. Pharmacol. 2021, 12, 638445. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.; Wiedmann, F.; Zhou, X.B.; Heijman, J.; Voigt, N.; Ratte, A.; Lang, S.; Kallenberger, S.M.; Campana, C.; Weymann, A.; et al. Inverse remodelling of K2P3.1 K+ channel expression and action potential duration in left ventricular dysfunction and atrial fibrillation: Implications for patient-specific antiarrhythmic drug therapy. Eur. Heart J. 2017, 38, 1764–1774. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.; Wiedmann, F.; Voigt, N.; Zhou, X.B.; Heijman, J.; Lang, S.; Albert, V.; Kallenberger, S.; Ruhparwar, A.; Szabó, G.; et al. Upregulation of K2P3.1 K+ Current Causes Action Potential Shortening in Patients With Chronic Atrial Fibrillation. Circulation 2015, 132, 82–92. [Google Scholar] [CrossRef]
- Wiedmann, F.; Beyersdorf, C.; Zhou, X.B.; Kraft, M.; Foerster, K.I.; El-Battrawy, I.; Lang, S.; Borggrefe, M.; Haefeli, W.E.; Frey, N.; et al. The Experimental TASK-1 Potassium Channel Inhibitor A293 Can Be Employed for Rhythm Control of Persistent Atrial Fibrillation in a Translational Large Animal Model. Front. Physiol. 2020, 11, 629421. [Google Scholar] [CrossRef]
- Wiedmann, F.; Beyersdorf, C.; Zhou, X.B.; Kraft, M.; Paasche, A.; Jávorszky, N.; Rinné, S.; Sutanto, H.; Büscher, A.; Foerster, K.I.; et al. Treatment of atrial fibrillation with doxapram: TASK-1 potassium channel inhibition as a novel pharmacological strategy. Cardiovasc. Res. 2021, 118, 1728–1741. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.; Wiedmann, F.; Beyersdorf, C.; Zhao, Z.; El-Battrawy, I.; Lan, H.; Szabo, G.; Li, X.; Lang, S.; Korkmaz-Icöz, S.; et al. Genetic Ablation of TASK-1 (Tandem of P Domains in a Weak Inward Rectifying K+ Channel-Related Acid-Sensitive K+ Channel-1) (K2P3.1) K+ Channels Suppresses Atrial Fibrillation and Prevents Electrical Remodeling. Circ. Arrhythm Electrophysiol 2019, 12, e007465. [Google Scholar] [CrossRef]
- Wiedmann, F.; Beyersdorf, C.; Zhou, X.; Büscher, A.; Kraft, M.; Nietfeld, J.; Walz, T.P.; Unger, L.A.; Loewe, A.; Schmack, B.; et al. Pharmacologic TWIK-Related Acid-Sensitive K+ channel (TASK-1) Potassium Channel Inhibitor A293 Facilitates Acute Cardioversion of Paroxysmal Atrial Fibrillation in a Porcine Large Animal Model. J. Am. Heart Assoc. 2020, 9, e015751. [Google Scholar] [CrossRef] [PubMed]
- Ratte, A.; Wiedmann, F.; Kraft, M.; Katus, H.A.; Schmidt, C. Antiarrhythmic Properties of Ranolazine: Inhibition of Atrial Fibrillation Associated TASK-1 Potassium Channels. Front. Pharmacol. 2019, 10, 1367. [Google Scholar] [CrossRef]
- Wiedmann, F.; Kraft, M.; Kallenberger, S.; Büscher, A.; Paasche, A.; Blochberger, P.L.; Seeger, T.; Jávorszky, N.; Warnecke, G.; Arif, R.; et al. MicroRNAs Regulate TASK-1 and Are Linked to Myocardial Dilatation in Atrial Fibrillation. J. Am. Heart Assoc. 2022, 11, e023472. [Google Scholar] [CrossRef] [PubMed]
- Davies, L.A.; Hu, C.; Guagliardo, N.A.; Sen, N.; Chen, X.; Talley, E.M.; Carey, R.M.; Bayliss, D.A.; Barrett, P.Q. TASK channel deletion in mice causes primary hyperaldosteronism. Proc. Natl. Acad. Sci. USA 2008, 105, 2203–2208. [Google Scholar] [CrossRef] [PubMed]
- Manichaikul, A.; Rich, S.S.; Allison, M.A.; Guagliardo, N.A.; Bayliss, D.A.; Carey, R.M.; Barrett, P.Q. KCNK3 Variants Are Associated With Hyperaldosteronism and Hypertension. Hypertension 2016, 68, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Guagliardo, N.A.; Yao, J.; Stipes, E.J.; Cechova, S.; Le, T.H.; Bayliss, D.A.; Breault, D.T.; Barrett, P.Q. Adrenal Tissue-Specific Deletion of TASK Channels Causes Aldosterone-Driven Angiotensin II-Independent Hypertension. Hypertension 2019, 73, 407–414. [Google Scholar] [CrossRef]
- Penton, D.; Bandulik, S.; Schweda, F.; Haubs, S.; Tauber, P.; Reichold, M.; Cong, L.D.; El Wakil, A.; Budde, T.; Lesage, F.; et al. Task3 potassium channel gene invalidation causes low renin and salt-sensitive arterial hypertension. Endocrinology 2012, 153, 4740–4748. [Google Scholar] [CrossRef]
- Chen, A.X.; Nishimoto, K.; Nanba, K.; Rainey, W.E. Potassium channels related to primary aldosteronism: Expression similarities and differences between human and rat adrenals. Mol. Cell. Endocrinol. 2015, 417, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Barrett, P.Q.; Guagliardo, N.A.; Bayliss, D.A. Ion Channel Function and Electrical Excitability in the Zona Glomerulosa: A Network Perspective on Aldosterone Regulation. Annu. Rev. Physiol. 2021, 83, 451–475. [Google Scholar] [CrossRef]
- Li, X.Y.; Toyoda, H. Role of leak potassium channels in pain signaling. Brain Res. Bull. 2015, 119 Pt A, 73–79. [Google Scholar] [CrossRef]
- Gada, K.; Plant, L.D. Two-pore domain potassium channels: Emerging targets for novel analgesic drugs: IUPHAR Review 26. Br. J. Pharmacol. 2019, 176, 256–266. [Google Scholar] [CrossRef]
- Marsh, B.; Acosta, C.; Djouhri, L.; Lawson, S.N. Leak K⁺ channel mRNAs in dorsal root ganglia: Relation to inflammation and spontaneous pain behaviour. Mol. Cell. Neurosci. 2012, 49, 375–386. [Google Scholar] [CrossRef]
- García, G.; Noriega-Navarro, R.; Martínez-Rojas, V.A.; Gutiérrez-Lara, E.J.; Oviedo, N.; Murbartián, J. Spinal TASK-1 and TASK-3 modulate inflammatory and neuropathic pain. Eur. J. Pharmacol. 2019, 862, 172631. [Google Scholar] [CrossRef] [PubMed]
- Patel, A.J.; Honoré, E.; Lesage, F.; Fink, M.; Romey, G.; Lazdunski, M. Inhalational anesthetics activate two-pore-domain background K+ channels. Nat. Neurosci. 1999, 2, 422–426. [Google Scholar] [CrossRef]
- Talley, E.M.; Bayliss, D.A. Modulation of TASK-1 (Kcnk3) and TASK-3 (Kcnk9) potassium channels: Volatile anesthetics and neurotransmitters share a molecular site of action. J. Biol. Chem. 2002, 277, 17733–17742. [Google Scholar] [CrossRef]
- Lazarenko, R.M.; Willcox, S.C.; Shu, S.; Berg, A.P.; Jevtovic-Todorovic, V.; Talley, E.M.; Chen, X.; Bayliss, D.A. Motoneuronal TASK channels contribute to immobilizing effects of inhalational general anesthetics. J. Neurosci. 2010, 30, 7691–7704. [Google Scholar] [CrossRef] [PubMed]
- Berg, A.P.; Talley, E.M.; Manger, J.P.; Bayliss, D.A. Motoneurons express heteromeric TWIK-related acid-sensitive K+ (TASK) channels containing TASK-1 (KCNK3) and TASK-3 (KCNK9) subunits. J. Neurosci. 2004, 24, 6693–6702. [Google Scholar] [CrossRef] [PubMed]
- Conway, K.E.; Cotten, J.F. Covalent modification of a volatile anesthetic regulatory site activates TASK-3 (KCNK9) tandem-pore potassium channels. Mol. Pharmacol. 2012, 81, 393–400. [Google Scholar] [CrossRef] [PubMed]
- Kindler, C.H.; Yost, C.S.; Gray, A.T. Local anesthetic inhibition of baseline potassium channels with two pore domains in tandem. Anesthesiology 1999, 90, 1092–1102. [Google Scholar] [CrossRef] [PubMed]
- Du, G.; Chen, X.; Todorovic, M.S.; Shu, S.; Kapur, J.; Bayliss, D.A. TASK Channel Deletion Reduces Sensitivity to Local Anesthetic-induced Seizures. Anesthesiology 2011, 115, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- Dadi, P.K.; Vierra, N.C.; Jacobson, D.A. Pancreatic β-cell-specific ablation of TASK-1 channels augments glucose-stimulated calcium entry and insulin secretion, improving glucose tolerance. Endocrinology 2014, 155, 3757–3768. [Google Scholar] [CrossRef] [PubMed]
- Dadi, P.K.; Luo, B.; Vierra, N.C.; Jacobson, D.A. TASK-1 Potassium Channels Limit Pancreatic α-Cell Calcium Influx and Glucagon Secretion. Mol. Endocrinol. 2015, 29, 777–787. [Google Scholar] [CrossRef]
- Kim, Y.; Bang, H.; Kim, D. TASK-3, a new member of the tandem pore K+ channel family. J. Biol. Chem. 2000, 275, 9340–9347. [Google Scholar] [CrossRef] [PubMed]
- Ren, W.J.; Ulrich, H.; Semyanov, A.; Illes, P.; Tang, Y. TASK-3: New Target for Pain-Relief. Neurosci. Bull. 2020, 36, 951–954. [Google Scholar] [CrossRef] [PubMed]
- Pollema-Mays, S.L.; Centeno, M.V.; Ashford, C.J.; Apkarian, A.V.; Martina, M. Expression of background potassium channels in rat DRG is cell-specific and down-regulated in a neuropathic pain model. Mol. Cell. Neurosci. 2013, 57, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Liao, P.; Qiu, Y.; Mo, Y.; Fu, J.; Song, Z.; Huang, L.; Bai, S.; Wang, Y.; Zhu, J.J.; Tian, F.; et al. Selective activation of TWIK-related acid-sensitive K+ 3 subunit-containing channels is analgesic in rodent models. Sci. Transl. Med. 2019, 11, eaaw8434. [Google Scholar] [CrossRef] [PubMed]
- Mu, D.; Chen, L.; Zhang, X.; See, L.H.; Koch, C.M.; Yen, C.; Tong, J.J.; Spiegel, L.; Nguyen, K.C.; Servoss, A.; et al. Genomic amplification and oncogenic properties of the KCNK9 potassium channel gene. Cancer Cell 2003, 3, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Pei, L.; Wiser, O.; Slavin, A.; Mu, D.; Powers, S.; Jan, L.Y.; Hoey, T. Oncogenic potential of TASK3 (Kcnk9) depends on K+ channel function. Proc. Natl. Acad. Sci. USA 2003, 100, 7803–7807. [Google Scholar] [CrossRef] [PubMed]
- Zúñiga, R.; Valenzuela, C.; Concha, G.; Brown, N.; Zúñiga, L. TASK-3 Downregulation Triggers Cellular Senescence and Growth Inhibition in Breast Cancer Cell Lines. Int. J. Mol. Sci 2018, 19, 1033. [Google Scholar] [CrossRef] [PubMed]
- Zúñiga, R.; Concha, G.; Cayo, A.; Cikutović-Molina, R.; Arevalo, B.; González, W.; Catalán, M.A.; Zúñiga, L. Withaferin A suppresses breast cancer cell proliferation by inhibition of the two-pore domain potassium (K2P9) channel TASK-3. Biomed. Pharmacother. 2020, 129, 110383. [Google Scholar] [CrossRef] [PubMed]
- Innamaa, A.; Jackson, L.; Asher, V.; Van Shalkwyk, G.; Warren, A.; Hay, D.; Bali, A.; Sowter, H.; Khan, R. Expression and prognostic significance of the oncogenic K2P potassium channel KCNK9 (TASK-3) in ovarian carcinoma. Anticancer Res. 2013, 33, 1401–1408. [Google Scholar] [PubMed]
- Rusznák, Z.; Bakondi, G.; Kosztka, L.; Pocsai, K.; Dienes, B.; Fodor, J.; Telek, A.; Gönczi, M.; Szucs, G.; Csernoch, L. Mitochondrial expression of the two-pore domain TASK-3 channels in malignantly transformed and non-malignant human cells. Virchows Arch. 2008, 452, 415–426. [Google Scholar] [CrossRef] [PubMed]
- Nagy, D.; Gönczi, M.; Dienes, B.; Szöőr, Á.; Fodor, J.; Nagy, Z.; Tóth, A.; Fodor, T.; Bai, P.; Szücs, G.; et al. Silencing the KCNK9 potassium channel (TASK-3) gene disturbs mitochondrial function, causes mitochondrial depolarization, and induces apoptosis of human melanoma cells. Arch. Dermatol. Res. 2014, 306, 885–902. [Google Scholar] [CrossRef] [PubMed]
- Bachmann, M.; Rossa, A.; Antoniazzi, G.; Biasutto, L.; Carrer, A.; Campagnaro, M.; Leanza, L.; Gonczi, M.; Csernoch, L.; Paradisi, C.; et al. Synthesis and cellular effects of a mitochondria-targeted inhibitor of the two-pore potassium channel TASK-3. Pharmacol. Res. 2021, 164, 105326. [Google Scholar] [CrossRef]
- Wrzosek, A.; Gałecka, S.; Żochowska, M.; Olszewska, A.; Kulawiak, B. Alternative Targets for Modulators of Mitochondrial Potassium Channels. Molecules 2022, 27, 299. [Google Scholar] [CrossRef] [PubMed]
- Yao, J.; McHedlishvili, D.; McIntire, W.E.; Guagliardo, N.A.; Erisir, A.; Coburn, C.A.; Santarelli, V.P.; Bayliss, D.A.; Barrett, P.Q. Functional TASK-3-Like Channels in Mitochondria of Aldosterone-Producing Zona Glomerulosa Cells. Hypertension 2017, 70, 347–356. [Google Scholar] [CrossRef] [PubMed]
- Cikutović-Molina, R.; Herrada, A.A.; González, W.; Brown, N.; Zúñiga, L. TASK-3 Gene Knockdown Dampens Invasion and Migration and Promotes Apoptosis in KATO III and MKN-45 Human Gastric Adenocarcinoma Cell Lines. Int. J. Mol. Sci. 2019, 20, 6077. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Luo, L.; Lal, B.; Ma, X.; Chen, L.; Hann, C.L.; Fulton, A.M.; Leahy, D.J.; Laterra, J.; Li, M. A monoclonal antibody against KCNK9 K+ channel extracellular domain inhibits tumour growth and metastasis. Nat. Commun. 2016, 7, 10339. [Google Scholar] [CrossRef] [PubMed]
- Bedoya, M.; Rinné, S.; Kiper, A.K.; Decher, N.; González, W.; Ramírez, D. TASK Channels Pharmacology: New Challenges in Drug Design. J. Med. Chem. 2019, 62, 10044–10058. [Google Scholar] [CrossRef] [PubMed]
- Linden, A.M.; Sandu, C.; Aller, M.I.; Vekovischeva, O.Y.; Rosenberg, P.H.; Wisden, W.; Korpi, E.R. TASK-3 knockout mice exhibit exaggerated nocturnal activity, impairments in cognitive functions, and reduced sensitivity to inhalation anesthetics. J. Pharmacol. Exp. Ther. 2007, 323, 924–934. [Google Scholar] [CrossRef] [PubMed]
- Pang, D.S.; Robledo, C.J.; Carr, D.R.; Gent, T.C.; Vyssotski, A.L.; Caley, A.; Zecharia, A.Y.; Wisden, W.; Brickley, S.G.; Franks, N.P. An unexpected role for TASK-3 potassium channels in network oscillations with implications for sleep mechanisms and anesthetic action. Proc. Natl. Acad. Sci. USA 2009, 106, 17546–17551. [Google Scholar] [CrossRef]
- Gotter, A.L.; Santarelli, V.P.; Doran, S.M.; Tannenbaum, P.L.; Kraus, R.L.; Rosahl, T.W.; Meziane, H.; Montial, M.; Reiss, D.R.; Wessner, K.; et al. TASK-3 as a potential antidepressant target. Brain Res. 2011, 1416, 69–79. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Barel, O.; Shalev, S.A.; Ofir, R.; Cohen, A.; Zlotogora, J.; Shorer, Z.; Mazor, G.; Finer, G.; Khateeb, S.; Zilberberg, N.; et al. Maternally inherited Birk Barel mental retardation dysmorphism syndrome caused by a mutation in the genomically imprinted potassium channel KCNK9. Am. J. Hum. Genet. 2008, 83, 193–199. [Google Scholar] [CrossRef]
- Graham, J.M., Jr.; Zadeh, N.; Kelley, M.; Tan, E.S.; Liew, W.; Tan, V.; Deardorff, M.A.; Wilson, G.N.; Sagi-Dain, L.; Shalev, S.A. KCNK9 imprinting syndrome-further delineation of a possible treatable disorder. Am. J. Med. Genet. A 2016, 170, 2632–2637. [Google Scholar] [CrossRef] [PubMed]
- Šedivá, M.; Laššuthová, P.; Zámečník, J.; Sedláčková, L.; Seeman, P.; Haberlová, J. Novel variant in the KCNK9 gene in a girl with Birk Barel syndrome. Eur. J. Med. Genet. 2020, 63, 103619. [Google Scholar] [CrossRef]
- Veale, E.L.; Hassan, M.; Walsh, Y.; Al-Moubarak, E.; Mathie, A. Recovery of current through mutated TASK3 potassium channels underlying Birk Barel syndrome. Mol. Pharmacol. 2014, 85, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Wright, P.D.; Veale, E.L.; McCoull, D.; Tickle, D.C.; Large, J.M.; Ococks, E.; Gothard, G.; Kettleborough, C.; Mathie, A.; Jerman, J. Terbinafine is a novel and selective activator of the two-pore domain potassium channel TASK3. Biochem. Biophys. Res. Commun. 2017, 493, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.; Butto, T.; Hammer, N.; Jagannath, S.; Fend-Guella, D.L.; Akhtar, J.; Radyushkin, K.; Lesage, F.; Winter, J.; Strand, S.; et al. Inhibition of histone deacetylation rescues phenotype in a mouse model of Birk-Barel intellectual disability syndrome. Nat. Commun. 2020, 11, 480. [Google Scholar] [CrossRef] [PubMed]
- Borsotto, M.; Veyssiere, J.; Moha Ou Maati, H.; Devader, C.; Mazella, J.; Heurteaux, C. Targeting two-pore domain K+ channels TREK-1 and TASK-3 for the treatment of depression: A new therapeutic concept. Br. J. Pharmacol. 2015, 172, 771–784. [Google Scholar] [CrossRef] [PubMed]
- Wichniak, A.; Wierzbicka, A.; Jernajczyk, W. Sleep as a biomarker for depression. Int. Rev. Psychiatry 2013, 25, 632–645. [Google Scholar] [CrossRef] [PubMed]
- Steiger, A.; Kimura, M. Wake and sleep EEG provide biomarkers in depression. J. Psychiatr. Res. 2010, 44, 242–252. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.; Liao, P.; Luo, Y.; Yang, L.; Yang, H.; Liu, L.; Jiang, R. Tandem pore domain acid-sensitive K channel 3 (TASK-3) regulates visual sensitivity in healthy and aging retina. Sci. Adv. 2022, 8, eabn8785. [Google Scholar] [CrossRef] [PubMed]
- Rödström, K.E.J.; Kiper, A.K.; Zhang, W.; Rinné, S.; Pike, A.C.W.; Goldstein, M.; Conrad, L.J.; Delbeck, M.; Hahn, M.G.; Meier, H.; et al. A lower X-gate in TASK channels traps inhibitors within the vestibule. Nature 2020, 582, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Enyedi, P.; Czirják, G. Molecular background of leak K+ currents: Two-pore domain potassium channels. Physiol. Rev. 2010, 90, 559–605. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.N.; Long, S.B. Crystal Structure of the Human Two–Pore Domain Potassium Channel K2P1. Science 2012, 335, 432. [Google Scholar] [CrossRef]
- González, W.; Zúñiga, L.; Cid, L.P.; Arévalo, B.; Niemeyer, M.I.; Sepúlveda, F.V. An extracellular ion pathway plays a central role in the cooperative gating of a K2P K+ channel by extracellular pH. J. Biol. Chem. 2013, 288, 5984–5991. [Google Scholar] [CrossRef]
- Goldstein, M.; Rinné, S.; Kiper, A.K.; Ramírez, D.; Netter, M.F.; Bustos, D.; Ortiz-Bonnin, B.; González, W.; Decher, N. Functional mutagenesis screens reveal the ‘cap structure’ formation in disulfide-bridge free TASK channels. Sci. Rep. 2016, 6, 19492. [Google Scholar] [CrossRef] [PubMed]
- Feliciangeli, S.; Chatelain, F.C.; Bichet, D.; Lesage, F. The family of K2P channels: Salient structural and functional properties. J. Physiol. 2015, 593, 2587–2603. [Google Scholar] [CrossRef] [PubMed]
- Concha, G.; Bustos, D.; Zúñiga, R.; Catalán, M.A.; Zúñiga, L. The Insensitivity of TASK-3 K₂P Channels to External Tetraethylammonium (TEA) Partially Depends on the Cap Structure. Int. J. Mol. Sci 2018, 19, 2437. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, D.; Arévalo, B.; Martínez, G.; Rinné, S.; Sepúlveda, F.V.; Decher, N.; González, W. Side Fenestrations Provide an “Anchor” for a Stable Binding of A1899 to the Pore of TASK-1 Potassium Channels. Mol. Pharm. 2017, 14, 2197–2208. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, C.; Darré, L.; Oakes, V.; Torella, R.; Pryde, D.; Domene, C. Lateral Fenestrations in K+-Channels Explored Using Molecular Dynamics Simulations. Mol. Pharm. 2016, 13, 2263–2273. [Google Scholar] [CrossRef] [PubMed]
- Talley, E.M.; Solorzano, G.; Lei, Q.; Kim, D.; Bayliss, D.A. Cns distribution of members of the two-pore-domain (KCNK) potassium channel family. J. Neurosci. 2001, 21, 7491–7505. [Google Scholar] [CrossRef]
- Rusznák, Z.; Pocsai, K.; Kovács, I.; Pór, A.; Pál, B.; Bíró, T.; Szücs, G. Differential distribution of TASK-1, TASK-2 and TASK-3 immunoreactivities in the rat and human cerebellum. Cell. Mol. Life Sci. 2004, 61, 1532–1542. [Google Scholar] [CrossRef] [PubMed]
- Zilberberg, N.; Ilan, N.; Goldstein, S.A. KCNKØ: Opening and closing the 2-P-domain potassium leak channel entails “C-type” gating of the outer pore. Neuron 2001, 32, 635–648. [Google Scholar] [CrossRef] [PubMed]
- Schewe, M.; Nematian-Ardestani, E.; Sun, H.; Musinszki, M.; Cordeiro, S.; Bucci, G.; de Groot, B.L.; Tucker, S.J.; Rapedius, M.; Baukrowitz, T. A Non-canonical Voltage-Sensing Mechanism Controls Gating in K2P K+ Channels. Cell 2016, 164, 937–949. [Google Scholar] [CrossRef] [PubMed]
- O’Connell, A.D.; Morton, M.J.; Sivaprasadarao, A.; Hunter, M. Selectivity and interactions of Ba2+ and Cs+ with wild-type and mutant TASK1 K+ channels expressed in Xenopus oocytes. J. Physiol. 2005, 562 Pt 3, 687–696. [Google Scholar] [CrossRef]
- Wang, X.; Guan, R.; Zhao, X.; Zhu, D.; Song, N.; Shen, L. TASK1 and TASK3 Are Coexpressed With ASIC1 in the Ventrolateral Medulla and Contribute to Central Chemoreception in Rats. Front. Cell. Neurosci. 2018, 12, 285. [Google Scholar] [CrossRef] [PubMed]
- Clarke, C.E.; Veale, E.L.; Green, P.J.; Meadows, H.J.; Mathie, A. Selective block of the human 2-P domain potassium channel, TASK-3, and the native leak potassium current, IKSO, by zinc. J. Physiol. 2004, 560 Pt 1, 51–62. [Google Scholar] [CrossRef]
- Maingret, F.; Patel, A.J.; Lazdunski, M.; Honoré, E. The endocannabinoid anandamide is a direct and selective blocker of the background K+ channel TASK-1. EMBO J. 2001, 20, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Czirják, G.; Enyedi, P. Formation of functional heterodimers between the TASK-1 and TASK-3 two-pore domain potassium channel subunits. J. Biol. Chem. 2002, 277, 5426–5432. [Google Scholar] [CrossRef] [PubMed]
- Wilke, B.U.; Lindner, M.; Greifenberg, L.; Albus, A.; Kronimus, Y.; Bünemann, M.; Leitner, M.G.; Oliver, D. Diacylglycerol mediates regulation of TASK potassium channels by Gq-coupled receptors. Nat. Commun. 2014, 5, 5540. [Google Scholar] [CrossRef] [PubMed]
- Bista, P.; Pawlowski, M.; Cerina, M.; Ehling, P.; Leist, M.; Meuth, P.; Aissaoui, A.; Borsotto, M.; Heurteaux, C.; Decher, N.; et al. Differential phospholipase C-dependent modulation of TASK and TREK two-pore domain K+ channels in rat thalamocortical relay neurons. J. Physiol. 2015, 593, 127–144. [Google Scholar] [CrossRef]
- Lesage, F.; Barhanin, J. Molecular physiology of pH-sensitive background K2P channels. Physiology (Bethesda) 2011, 26, 424–437. [Google Scholar] [CrossRef] [PubMed]
- Yuill, K.; Ashmole, I.; Stanfield, P.R. The selectivity filter of the tandem pore potassium channel TASK-1 and its pH-sensitivity and ionic selectivity. Pflugers Arch. 2004, 448, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Chatelain, F.C.; Lesage, F. Altered and dynamic ion selectivity of K+ channels in cell development and excitability. Trends Pharmacol. Sci. 2014, 35, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Zhang, X.; Zhou, M.; Chen, H. Acid-sensitive TWIK and TASK two-pore domain potassium channels change ion selectivity and become permeable to sodium in extracellular acidification. J. Biol. Chem. 2012, 287, 37145–37153. [Google Scholar] [CrossRef] [PubMed]
- Chatelain, F.C.; Bichet, D.; Douguet, D.; Feliciangeli, S.; Bendahhou, S.; Reichold, M.; Warth, R.; Barhanin, J.; Lesage, F. TWIK1, a unique background channel with variable ion selectivity. Proc. Natl. Acad. Sci. USA 2012, 109, 5499–5504. [Google Scholar] [CrossRef]
- Bezanilla, F. Gating currents. J. Gen. Physiol. 2018, 150, 911–932. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Fu, J.; Zhang, S.; Guo, P.; Liu, S.; Shen, J.; Guo, J.; Yang, H. ‘C-type’ closed state and gating mechanisms of K2P channels revealed by conformational changes of the TREK-1 channel. J. Mol. Cell. Biol. 2022, 14, mjac002. [Google Scholar] [CrossRef]
- Bagriantsev, S.N.; Peyronnet, R.; Clark, K.A.; Honoré, E.; Minor, D.L., Jr. Multiple modalities converge on a common gate to control K2P channel function. EMBO J. 2011, 30, 3594–3606. [Google Scholar] [CrossRef] [PubMed]
- Lolicato, M.; Arrigoni, C.; Mori, T.; Sekioka, Y.; Bryant, C.; Clark, K.A.; Minor, D.L., Jr. K2P2.1 (TREK-1)-activator complexes reveal a cryptic selectivity filter binding site. Nature 2017, 547, 364–368. [Google Scholar] [CrossRef]
- Piechotta, P.L.; Rapedius, M.; Stansfeld, P.J.; Bollepalli, M.K.; Ehrlich, G.; Andres-Enguix, I.; Fritzenschaft, H.; Decher, N.; Sansom, M.S.; Tucker, S.J.; et al. The pore structure and gating mechanism of K2P channels. EMBO J. 2011, 30, 3607–3619. [Google Scholar] [CrossRef] [PubMed]
- Lolicato, M.; Natale, A.M.; Abderemane-Ali, F.; Crottès, D.; Capponi, S.; Duman, R.; Wagner, A.; Rosenberg, J.M.; Grabe, M.; Minor, D.L., Jr. K2P channel C-type gating involves asymmetric selectivity filter order-disorder transitions. Sci. Adv. 2020, 6, eabc9174. [Google Scholar] [CrossRef]
- Natale, A.M.; Deal, P.E.; Minor, D.L., Jr. Structural Insights into the Mechanisms and Pharmacology of K2P Potassium Channels. J. Mol. Biol. 2021, 433, 166995. [Google Scholar] [CrossRef]
- Wiedmann, F.; Kiper, A.K.; Bedoya, M.; Ratte, A.; Rinné, S.; Kraft, M.; Waibel, M.; Anad, P.; Wenzel, W.; González, W.; et al. Identification of the A293 (AVE1231) Binding Site in the Cardiac Two-Pore-Domain Potassium Channel TASK-1: A Common Low Affinity Antiarrhythmic Drug Binding Site. Cell. Physiol. Biochem. 2019, 52, 1223–1235. [Google Scholar] [PubMed]
- Chokshi, R.H.; Larsen, A.T.; Bhayana, B.; Cotten, J.F. Breathing Stimulant Compounds Inhibit TASK-3 Potassium Channel Function Likely by Binding at a Common Site in the Channel Pore. Mol. Pharmacol. 2015, 88, 926–934. [Google Scholar] [CrossRef]
- Flaherty, D.P.; Simpson, D.S.; Miller, M.; Maki, B.E.; Zou, B.; Shi, J.; Wu, M.; McManus, O.B.; Aubé, J.; Li, M.; et al. Potent and selective inhibitors of the TASK-1 potassium channel through chemical optimization of a bis-amide scaffold. Bioorg. Med. Chem. Lett. 2014, 24, 3968–3973. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, D.; Bedoya, M.; Kiper, A.K.; Rinné, S.; Morales-Navarro, S.; Hernández-Rodríguez, E.W.; Sepúlveda, F.V.; Decher, N.; González, W. Structure/Activity Analysis of TASK-3 Channel Antagonists Based on a 5,6,7,8 tetrahydropyrido [4,3-d]pyrimidine. Int. J. Mol. Sci 2019, 20, 2252. [Google Scholar] [CrossRef]
- Li, B.; Rietmeijer, R.A.; Brohawn, S.G. Structural basis for pH gating of the two-pore domain K+ channel TASK2. Nature 2020, 586, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Czirják, G.; Enyedi, P. Ruthenium red inhibits TASK-3 potassium channel by interconnecting glutamate 70 of the two subunits. Mol. Pharmacol. 2003, 63, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Pope, L.; Lolicato, M.; Minor, D.L., Jr. Polynuclear Ruthenium Amines Inhibit K2P Channels via a “Finger in the Dam” Mechanism. Cell Chem. Biol. 2020, 27, 511–524.e4. [Google Scholar] [CrossRef] [PubMed]
- Braun, G.; Lengyel, M.; Enyedi, P.; Czirják, G. Differential sensitivity of TREK-1, TREK-2 and TRAAK background potassium channels to the polycationic dye ruthenium red. Br. J. Pharmacol. 2015, 172, 1728–1738. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.Y.; Pike, A.C.; Mackenzie, A.; McClenaghan, C.; Aryal, P.; Dong, L.; Quigley, A.; Grieben, M.; Goubin, S.; Mukhopadhyay, S.; et al. K2P channel gating mechanisms revealed by structures of TREK-2 and a complex with Prozac. Science 2015, 347, 1256–1259. [Google Scholar] [CrossRef] [PubMed]
- Brohawn, S.G.; Campbell, E.B.; MacKinnon, R. Physical mechanism for gating and mechanosensitivity of the human TRAAK K+ channel. Nature 2014, 516, 126–130. [Google Scholar] [CrossRef]
- Lolicato, M.; Riegelhaupt, P.M.; Arrigoni, C.; Clark, K.A.; Minor, D.L., Jr. Transmembrane helix straightening and buckling underlies activation of mechanosensitive and thermosensitive K2P channels. Neuron 2014, 84, 1198–1212. [Google Scholar] [CrossRef] [PubMed]
- Brohawn, S.G.; Su, Z.; MacKinnon, R. Mechanosensitivity is mediated directly by the lipid membrane in TRAAK and TREK1 K+ channels. Proc. Natl. Acad. Sci. USA 2014, 111, 3614–3619. [Google Scholar] [CrossRef] [PubMed]
- Rinné, S.; Kiper, A.K.; Vowinkel, K.S.; Ramírez, D.; Schewe, M.; Bedoya, M.; Aser, D.; Gensler, I.; Netter, M.F.; Stansfeld, P.J.; et al. The molecular basis for an allosteric inhibition of K+-flux gating in K2P channels. Elife 2019, 8, e39476. [Google Scholar] [CrossRef] [PubMed]
- Guyenet, P.G.; Bayliss, D.A.; Stornetta, R.L.; Ludwig, M.G.; Kumar, N.N.; Shi, Y.; Burke, P.G.; Kanbar, R.; Basting, T.M.; Holloway, B.B.; et al. Proton detection and breathing regulation by the retrotrapezoid nucleus. J. Physiol. 2016, 594, 1529–1551. [Google Scholar] [CrossRef] [PubMed]
- Hayabuchi, Y. The Action of Smooth Muscle Cell Potassium Channels in the Pathology of Pulmonary Arterial Hypertension. Pediatr. Cardiol. 2017, 38, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Le Ribeuz, H.; Capuano, V.; Girerd, B.; Humbert, M.; Montani, D.; Antigny, F. Implication of Potassium Channels in the Pathophysiology of Pulmonary Arterial Hypertension. Biomolecules 2020, 10, 1261. [Google Scholar] [CrossRef] [PubMed]
- Southgate, L.; Machado, R.D.; Gräf, S.; Morrell, N.W. Molecular genetic framework underlying pulmonary arterial hypertension. Nat. Rev. Cardiol. 2019, 17, 85–95. [Google Scholar] [CrossRef]
- Dogan, M.F.; Yildiz, O.; Arslan, S.O.; Ulusoy, K.G. Potassium channels in vascular smooth muscle: A pathophysiological and pharmacological perspective. Fundam. Clin. Pharmacol. 2019, 33, 504–523. [Google Scholar] [CrossRef]
- Tennant, B.P.; Cui, Y.; Tinker, A.; Clapp, L.H. Functional expression of inward rectifier potassium channels in cultured human pulmonary smooth muscle cells: Evidence for a major role of Kir2.4 subunits. J. Membr. Biol. 2006, 213, 19–29. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wiedmann, F.; Frey, N.; Schmidt, C. Two-Pore-Domain Potassium (K2P-) Channels: Cardiac Expression Patterns and Disease-Specific Remodelling Processes. Cells 2021, 10, 2914. [Google Scholar] [CrossRef] [PubMed]
- Rinné, S.; Kiper, A.K.; Schlichthörl, G.; Dittmann, S.; Netter, M.F.; Limberg, S.H.; Silbernagel, N.; Zuzarte, M.; Moosdorf, R.; Wulf, H.; et al. TASK-1 and TASK-3 may form heterodimers in human atrial cardiomyocytes. J. Mol. Cell. Cardiol. 2015, 81, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, K.; Staudacher, I.; Ficker, E.; Seyler, C.; Gierten, J.; Kisselbach, J.; Rahm, A.K.; Trappe, K.; Schweizer, P.A.; Becker, R.; et al. Carvedilol targets human K2P 3.1 (TASK1) K+ leak channels. Br. J. Pharmacol. 2011, 163, 1099–1110. [Google Scholar] [CrossRef]
- Rahman, F.; Kwan, G.F.; Benjamin, E.J. Global epidemiology of atrial fibrillation. Nat. Rev. Cardiol. 2014, 11, 639–654. [Google Scholar] [CrossRef]
- Shvetsova, A.A.; Lazarenko, V.S.; Gaynullina, D.K.; Tarasova, O.S.; Schubert, R. TWIK-Related Acid-Sensitive Potassium Channels (TASK-1) Emerge as Contributors to Tone Regulation in Renal Arteries at Alkaline pH. Front. Physiol. 2022, 13, 982. [Google Scholar] [CrossRef]
- Shvetsova, A.A.; Gaynullina, D.K.; Schmidt, N.; Bugert, P.; Lukoshkova, E.V.; Tarasova, O.S.; Schubert, R. TASK-1 channel blockade by AVE1231 increases vasocontractile responses and BP in 1- to 2-week-old but not adult rats. Br. J. Pharmacol. 2020, 177, 5148–5162. [Google Scholar] [CrossRef]
- Meadows, H.J.; Randall, A.D. Functional characterisation of human TASK-3, an acid-sensitive two-pore domain potassium channel. Neuropharmacology 2001, 40, 551–559. [Google Scholar] [CrossRef]
- Reeh, P.W.; Kress, M. Molecular physiology of proton transduction in nociceptors. Curr. Opin. Pharmacol. 2001, 1, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Waldmann, R.; Champigny, G.; Bassilana, F.; Heurteaux, C.; Lazdunski, M. A proton-gated cation channel involved in acid-sensing. Nature 1997, 386, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Plant, L.D. A Role for K2P Channels in the Operation of Somatosensory Nociceptors. Front. Mol. Neurosci. 2012, 5, 21. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.H.; Chen, C.C. Roles of Proton-Sensing Receptors in the Transition from Acute to Chronic Pain. J. Dent. Res. 2016, 95, 135–142. [Google Scholar] [CrossRef]
- Morenilla-Palao, C.; Luis, E.; Fernández-Peña, C.; Quintero, E.; Weaver, J.L.; Bayliss, D.A.; Viana, F. Ion channel profile of TRPM8 cold receptors reveals a role of TASK-3 potassium channels in thermosensation. Cell Rep. 2014, 8, 1571–1582. [Google Scholar] [CrossRef]
- Sirois, J.E.; Lynch, C., 3rd; Bayliss, D.A. Convergent and reciprocal modulation of a leak K+ current and I(h) by an inhalational anaesthetic and neurotransmitters in rat brainstem motoneurones. J. Physiol. 2002, 541 Pt 3, 717–729. [Google Scholar] [CrossRef]
- Meuth, S.G.; Budde, T.; Kanyshkova, T.; Broicher, T.; Munsch, T.; Pape, H.C. Contribution of TWIK-related acid-sensitive K+ channel 1 (TASK1) and TASK3 channels to the control of activity modes in thalamocortical neurons. J. Neurosci. 2003, 23, 6460–6469. [Google Scholar] [CrossRef] [PubMed]
- Buljubasic, N.; Rusch, N.J.; Marijic, J.; Kampine, J.P.; Bosnjak, Z.J. Effects of halothane and isoflurane on calcium and potassium channel currents in canine coronary arterial cells. Anesthesiology 1992, 76, 990–998. [Google Scholar] [CrossRef] [PubMed]
- Decher, N.; Rinné, S.; Bedoya, M.; Gonzalez, W.; Kiper, A.K. Molecular Pharmacology of K2P Potassium Channels. Cell. Physiol. Biochem. 2021, 55, 87–107. [Google Scholar] [PubMed]
- Bista, P.; Cerina, M.; Ehling, P.; Leist, M.; Pape, H.C.; Meuth, S.G.; Budde, T. The role of two-pore-domain background K+ (K2P) channels in the thalamus. Pflugers Arch. 2015, 467, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Meuth, S.G.; Aller, M.I.; Munsch, T.; Schuhmacher, T.; Seidenbecher, T.; Meuth, P.; Kleinschnitz, C.; Pape, H.C.; Wiendl, H.; Wisden, W.; et al. The contribution of TWIK-related acid-sensitive K+-containing channels to the function of dorsal lateral geniculate thalamocortical relay neurons. Mol. Pharmacol. 2006, 69, 1468–1476. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, V.; Nestler, E.J. The molecular neurobiology of depression. Nature 2008, 455, 894–902. [Google Scholar] [CrossRef]
- Nestler, E.J.; Barrot, M.; DiLeone, R.J.; Eisch, A.J.; Gold, S.J.; Monteggia, L.M. Neurobiology of depression. Neuron 2002, 34, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.; Wiedmann, F.; Gaubatz, A.R.; Ratte, A.; Katus, H.A.; Thomas, D. New Targets for Old Drugs: Cardiac Glycosides Inhibit Atrial-Specific K2P3.1 (TASK-1) Channels. J. Pharmacol. Exp. Ther. 2018, 365, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Streit, A.K.; Netter, M.F.; Kempf, F.; Walecki, M.; Rinné, S.; Bollepalli, M.K.; Preisig-Müller, R.; Renigunta, V.; Daut, J.; Baukrowitz, T.; et al. A specific two-pore domain potassium channel blocker defines the structure of the TASK-1 open pore. J. Biol. Chem. 2011, 286, 13977–13984. [Google Scholar] [CrossRef]
- Zou, B.; Flaherty, D.P.; Simpson, D.S.; Maki, B.E.; Miller, M.R.; Shi, J.; Wu, M.; McManus, O.B.; Golden, J.E.; Aubé, J.; et al. ML365: Development of Bis-Amides as Selective Inhibitors of the KCNK3/TASK1 Two Pore Potassium Channel. In Probe Reports from the NIH Molecular Libraries Program; National Center for Biotechnology Information (US): Bethesda, MD, USA, 2010. [Google Scholar]
- Tian, F.; Qiu, Y.; Lan, X.; Li, M.; Yang, H.; Gao, Z. A Small-Molecule Compound Selectively Activates K2P Channel TASK-3 by Acting at Two Distant Clusters of Residues. Mol. Pharmacol. 2019, 96, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.R.; Zou, B.; Shi, J.; Flaherty, D.P.; Simpson, D.S.; Yao, T.; Maki, B.E.; Day, V.W.; Douglas, J.T.; Wu, M.; et al. Development of a Selective Chemical Inhibitor for the Two-Pore Potassium Channel, KCNK9. In Probe Reports from the NIH Molecular Libraries Program; National Center for Biotechnology Information (US): Bethesda, MD, USA, 2010. [Google Scholar]
- Coburn, C.A.; Luo, Y.; Cui, M.; Wang, J.; Soll, R.; Dong, J.; Hu, B.; Lyon, M.A.; Santarelli, V.P.; Kraus, R.L.; et al. Discovery of a pharmacologically active antagonist of the two-pore-domain potassium channel K2P9.1 (TASK-3). ChemMedChem 2012, 7, 123–133. [Google Scholar] [CrossRef]
- White, R.; Ho, W.S.; Bottrill, F.E.; Ford, W.R.; Hiley, C.R. Mechanisms of anandamide-induced vasorelaxation in rat isolated coronary arteries. Br. J. Pharmacol. 2001, 134, 921–929. [Google Scholar] [CrossRef] [PubMed]
- Van den Bossche, I.; Vanheel, B. Influence of cannabinoids on the delayed rectifier in freshly dissociated smooth muscle cells of the rat aorta. Br. J. Pharmacol. 2000, 131, 85–93. [Google Scholar] [CrossRef] [PubMed]
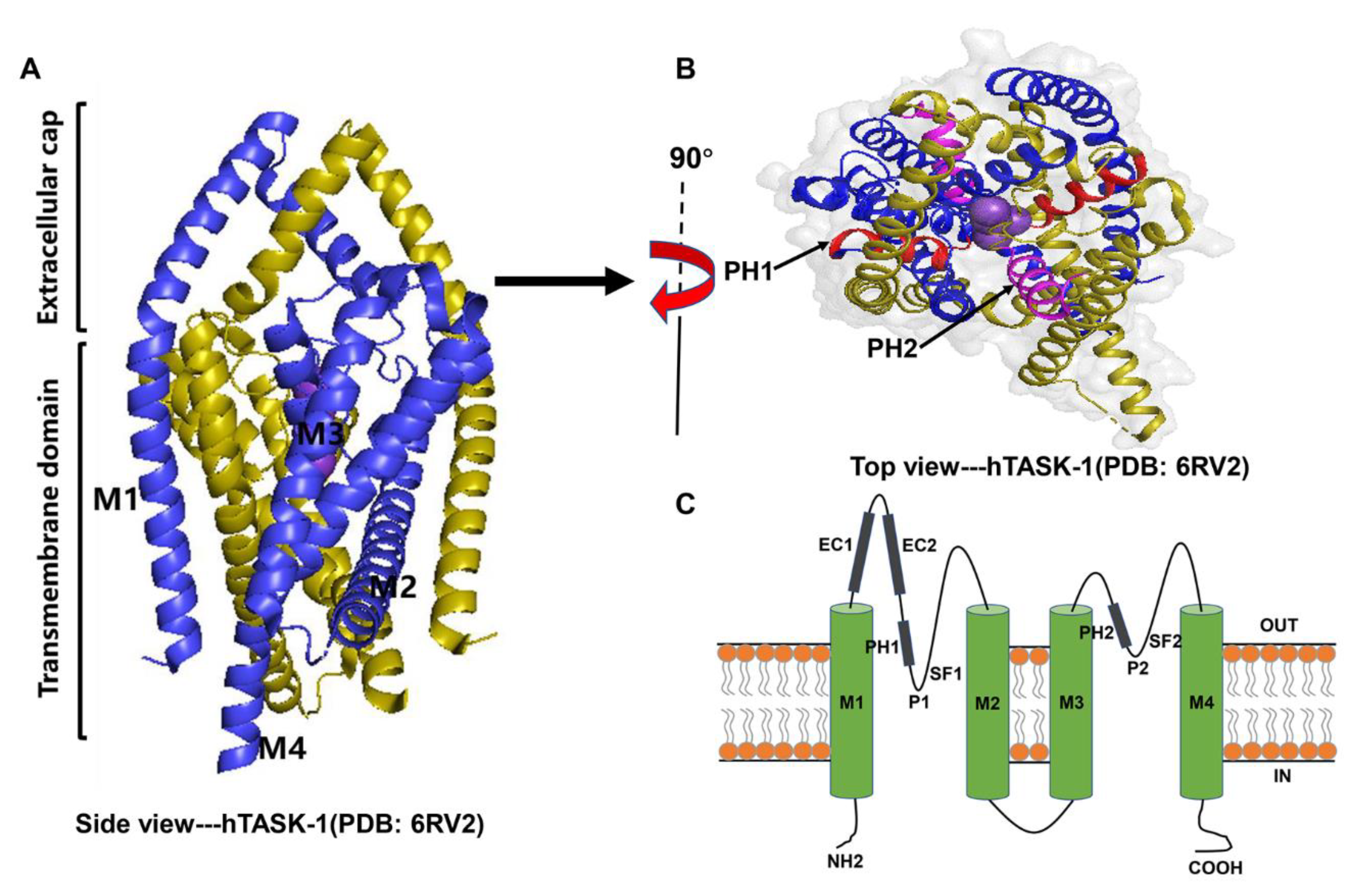
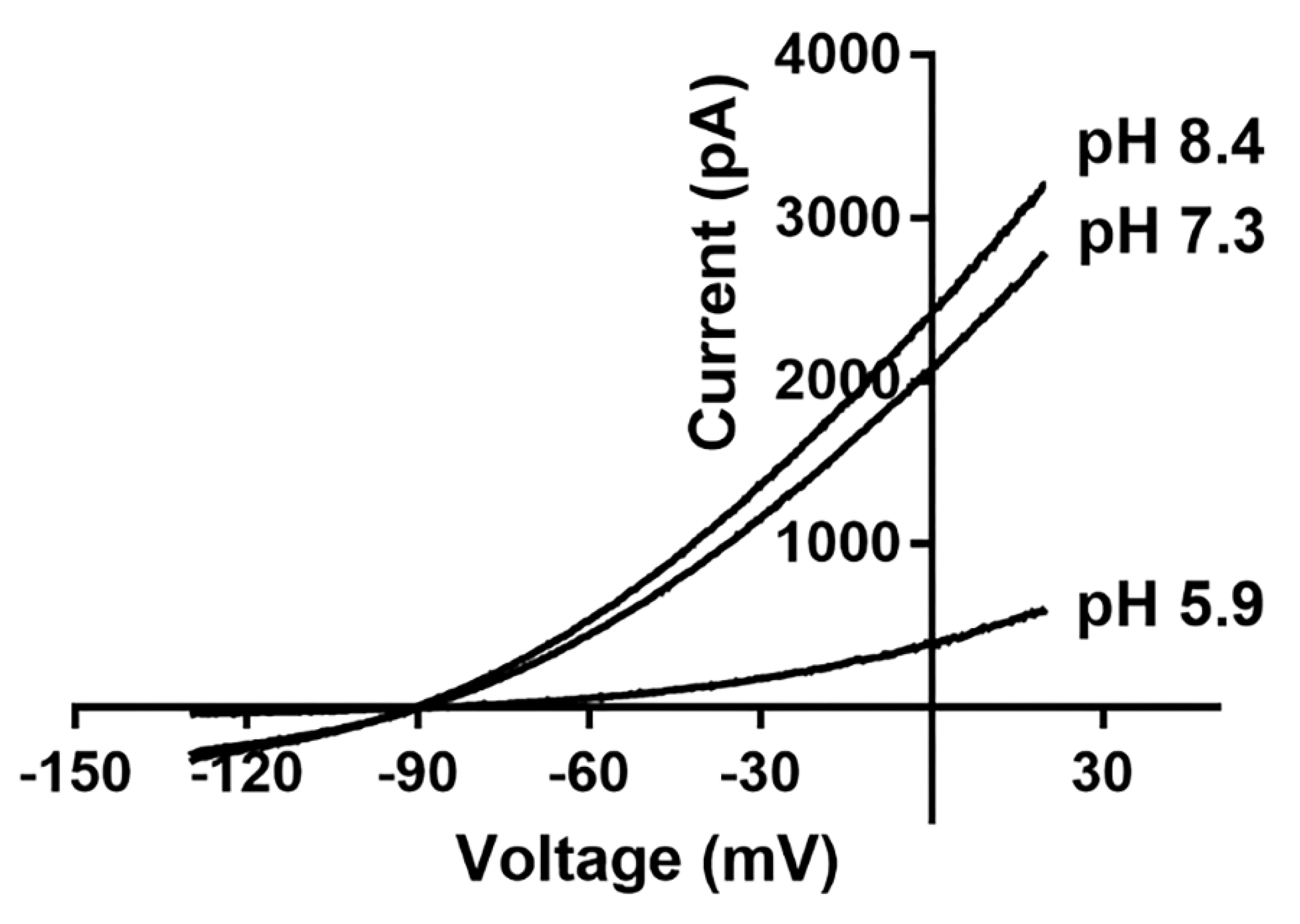
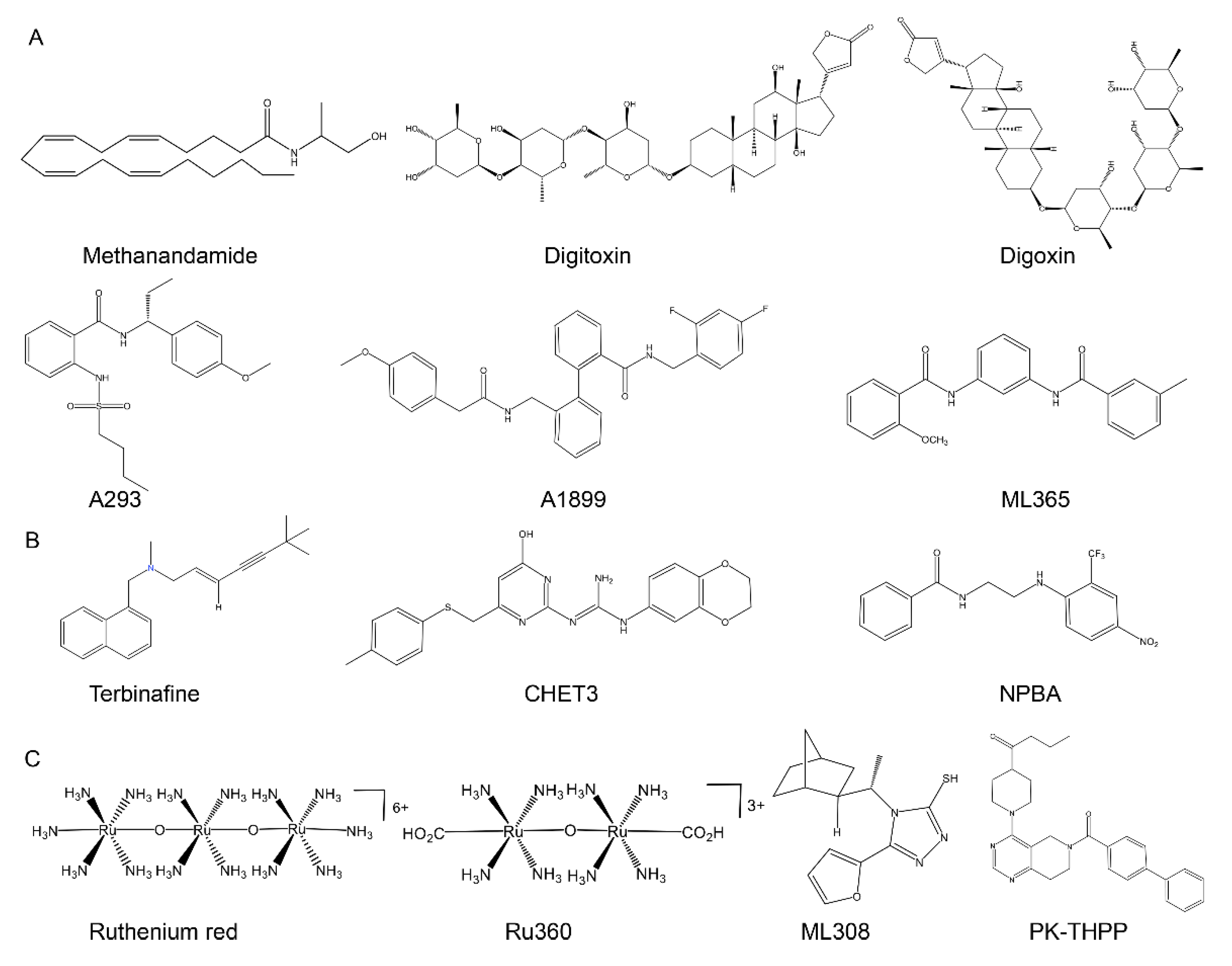
| Channel | Gene Name | Length in Humans | Distribution in Humans | Indicative Biological Roles | References |
|---|---|---|---|---|---|
| TASK-1 (K2P3.1) | KCNK3 | 394 | Abundantly expressed in the CNS and periphery. CNS: highest in the cerebellum, thalamus and pituitary gland, and lowest in the corpus callosum. Periphery: highest in the pancreas, placenta, lungs and pulmonary arteries, and lower in the prostate, stomach, small intestine, and heart, and lowest in the liver, spleen, skeletal muscle, and testis. | Chemosensation of breathing rhythm, pulmonary artery hypertension (PAH), cardiac arrhythmia (AF), aldosterone secretion, pain, general anesthesia, local anesthetic toxicity, glucose homeostasis | [11,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70] |
| TASK-3 (K2P9.1) | KCNK9 | 374 | Predominantly expressed in the CNS: highest in the cerebellum, higher in the cerebral cortex, thalamus, nucleus accumbens, hippocampus, and hypothalamus, and lowest in the spinal cord, caudate nucleus, and corpus callosum. Small amounts in the periphery: stomach, testis, skeletal muscles, uterus, kidneys, spleen, pancreas, prostate, and small intestine, and lowest in the heart, liver, and lungs. | Aldosterone secretion, pain, general anesthesia, local anesthetic toxicity, cancers, sleep, BBIDS, depression, visual sensitivity | [12,14,15,17,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88,89,90,91,92,93,94,95,96,97,98,99,100] |
| TASK-5 (K2P15.1) | KCNK15 | 330 | Highest in the pancreas, higher in the liver, kidneys, lungs, ovary, testis, and heart. | No reports | [13] |
| Channel | Activators | Compound Category | EC50 (μM) | Reference | Inhibitors | Compound Category | IC50 (μM) | Reference |
|---|---|---|---|---|---|---|---|---|
| TASK-1 | Methanandamide | Cannabinoids | 0.7 (MC) | [117] | ||||
| Digitoxin | Cardiac glycosides | 7.4 (XO) | [172] | |||||
| Digoxin | Cardiac glycosides | 0.9 (XO) | [172] | |||||
| A293 | Small molecules | 0.22 (XO) | [41] | |||||
| A1899 | Small molecules | 0.035 (XO), 0.007 (MC) | [173] | |||||
| ML365 | Small molecules | 0.016 (MC) | [135,174] | |||||
| TASK-3 | Terbinafine | Antifungals | NI | [95] | Ruthenium red | Cationic dyes | 0.114 (XO) | [139] |
| CHET3 | Small molecules | 1.4 (MC) | [74] | Ru360 | Polynuclear ruthenium amines | 15.6 (XO) | [139] | |
| NPBA | Small molecules | 6.7 (MC) | [175] | ML308 | Small molecules | 0.413 (MC) | [176] | |
| PK-THPP | Small molecules | 0.035 (MC) | [177] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, X.; Lu, Y.; Du, G.; Liu, J. Advances in the Understanding of Two-Pore Domain TASK Potassium Channels and Their Potential as Therapeutic Targets. Molecules 2022, 27, 8296. https://doi.org/10.3390/molecules27238296
Fan X, Lu Y, Du G, Liu J. Advances in the Understanding of Two-Pore Domain TASK Potassium Channels and Their Potential as Therapeutic Targets. Molecules. 2022; 27(23):8296. https://doi.org/10.3390/molecules27238296
Chicago/Turabian StyleFan, Xueming, Yongzhi Lu, Guizhi Du, and Jin Liu. 2022. "Advances in the Understanding of Two-Pore Domain TASK Potassium Channels and Their Potential as Therapeutic Targets" Molecules 27, no. 23: 8296. https://doi.org/10.3390/molecules27238296
APA StyleFan, X., Lu, Y., Du, G., & Liu, J. (2022). Advances in the Understanding of Two-Pore Domain TASK Potassium Channels and Their Potential as Therapeutic Targets. Molecules, 27(23), 8296. https://doi.org/10.3390/molecules27238296





