Benzochromenopyrimidines: Synthesis, Antiproliferative Activity against Colorectal Cancer and Physicochemical Properties
Abstract
1. Introduction
2. Results
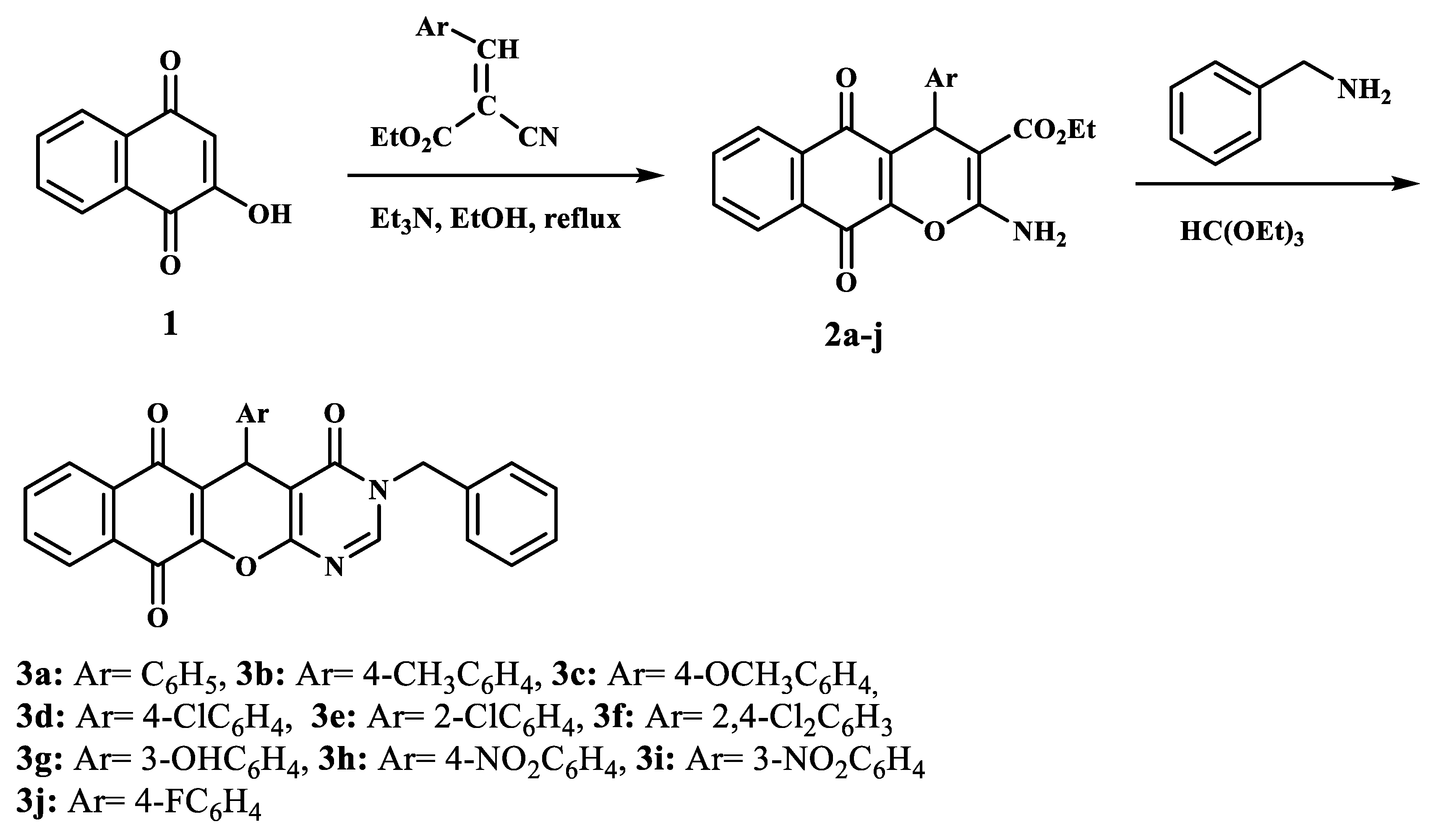
2.1. Synthesis
2.2. Biological Evaluation
2.2.1. Cytotoxicity Test
2.2.2. ADME Studies
3. Materials and Methods
3.1. Synthesis of Compounds 2a–j
3.1.1. General Procedure for the Synthesis of Ethyl 2-Amino-4-(3-hydroxyphenyl)-5,10-dioxo-5,10-dihy dro-4H-benzo[g]chromene-3-carboxylate (2a–j)
3.1.2. General Procedure for the Synthesis of 3-Benzyl-5-aryl-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidin-4,6,11-triones (3a–j)
3-Benzyl-5-phenyl-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3a)
3-Benzyl-5-(p-tolyl)-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3b)
3-Benzyl-5-(4-methoxyphenyl)-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3c)
3-Benzyl-5-(4-chlorophenyl)-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3d)
3-Benzyl-5-(2-chlorophenyl)-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3e)
3-Benzyl-5-(2,4-dichlorophenyl)-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3f)
3-Benzyl-5-(3-hydroxyphenyl)-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3g)
3-Benzyl-5-(4-nitrophenyl)-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3h)
3-Benzyl-5-(3-nitrophenyl)-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3i)
3-Benzyl-5-(4-fluorophenyl)-3,5-dihydro-4H-benzo[6,7]chromeno[2,3-d]pyrimidine-4,6,11-trione (3j)
3.2. Biological Evaluation
3.2.1. Materials and Methods
3.2.2. Cytotoxic Activity by MTT Assay
3.3. Statistical Data Analyses
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Henidi, H.A.; Al-Abd, A.M.; Al-Abbasi, F.A.; BinMahfouz, H.A.; El-Deeb, I.M. Design and Synthesis of Novel Phenylaminopyrimidines with Antiproliferative Activity against Colorectal Cancer. RSC Adv. 2019, 9, 21578–21586. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2017. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef]
- Ranjan Dwivedi, A.; Kumar, V.; Kaur, H.; Kumar, N.; Prakash Yadav, R.; Poduri, R.; Baranwal, S.; Kumar, V. Anti-Proliferative Potential of Triphenyl Substituted Pyrimidines against MDA-MB-231, HCT-116 and HT-29 Cancer Cell Lines. Bioorg. Med. Chem. Lett. 2020, 30, 127468. [Google Scholar] [CrossRef] [PubMed]
- Cancer. Available online: https://www.who.int/health-topics/cancer (accessed on 27 September 2022).
- Siripong, P.; Kanokmedakul, K.; Piyaviriyagul, S.; Yahuafai, J.; Chanpai, R.; Ruchirawat, S.; Oku, N. Antiproliferative Naphthoquinone Esters from Rhinacanthus nasutus Kurz. Roots on Various Cancer Cells. J. Tradit. Med. 2006, 23, 166–172. [Google Scholar] [CrossRef]
- Sperry, J.; Lorenzo-Castrillejo, I.; Brimble, M.A.; Machín, F. Pyranonaphthoquinone Derivatives of Eleutherin, Ventiloquinone L, Thysanone and Nanaomycin A Possessing a Diverse Topoisomerase II Inhibition and Cytotoxicity Spectrum. Bioorg. Med. Chem. 2009, 17, 7131–7137. [Google Scholar] [CrossRef]
- Abdelfattah, M.S.; Kazufumi, T.; Ishibashi, M. New Pyranonaphthoquinones and a Phenazine Alkaloid Isolated from Streptomyces Sp. IFM 11307 with TRAIL Resistance-Overcoming Activity. J. Antibiot. 2011, 64, 729–734. [Google Scholar] [CrossRef]
- Yang, Z.; Ding, J.; Ding, K.; Chen, D.; Cen, S.; Ge, M. Phomonaphthalenone A: A Novel Dihydronaphthalenone with Anti-HIV Activity from Phomopsis Sp. HCCB04730. Phytochem. Lett. 2013, 6, 257–260. [Google Scholar] [CrossRef]
- Jardosh, H.H.; Patel, M.P. Microwave-Induced CAN Promoted Atom-Economic Synthesis of 1H-Benzo[b]Xanthene and 4H-Benzo[g]Chromene Derivatives of N-Allyl Quinolone and Their Antimicrobial Activity. Med. Chem. Res. 2013, 22, 2954–2963. [Google Scholar] [CrossRef]
- Tangeti, V.; Vasundhara, D.; Kumar, M.; Mylapalli, H.; Kumar, K. Synthesis, Characterization and Cytotoxic Investigations of Novel C3-Dihydrofuran Substituted 1H-Benzo[g]Chromene-2,5,10-Triones besides Antimicrobial Study. Asian J. Chem. 2017, 29, 503–511. [Google Scholar] [CrossRef]
- Guimarães, T.T.; Pinto, M.d.C.F.R.; Lanza, J.S.; Melo, M.N.; do Monte-Neto, R.L.; de Melo, I.M.M.; Diogo, E.B.T.; Ferreira, V.F.; Camara, C.A.; Valença, W.O.; et al. Potent Naphthoquinones against Antimony-Sensitive and -Resistant Leishmania Parasites: Synthesis of Novel α- and nor-α-Lapachone-Based 1,2,3-Triazoles by Copper-Catalyzed Azide–Alkyne Cycloaddition. Eur. J. Med. Chem. 2013, 63, 523–530. [Google Scholar] [CrossRef]
- Al Nasr, I.S.; Jentzsch, J.; Shaikh, A.; Singh Shuveksh, P.; Koko, W.S.; Khan, T.A.; Ahmed, K.; Schobert, R.; Ersfeld, K.; Biersack, B. New Pyrano-4H-Benzo[g]Chromene-5,10-Diones with Antiparasitic and Antioxidant Activities. Chem. Biodivers. 2021, 18, e2000839. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Malachowski, W.P.; DuHadaway, J.B.; LaLonde, J.M.; Carroll, P.J.; Jaller, D.; Metz, R.; Prendergast, G.C.; Muller, A.J. Indoleamine 2,3-Dioxygenase Is the Anticancer Target for a Novel Series of Potent Naphthoquinone-Based Inhibitors. J. Med. Chem. 2008, 51, 1706–1718. [Google Scholar] [CrossRef][Green Version]
- da Rocha, D.R.; de Souza, A.C.G.; Resende, J.A.L.C.; Santos, W.C.; dos Santos, E.A.; Pessoa, C.; de Moraes, M.O.; Costa-Lotufo, L.V.; Montenegro, R.C.; Ferreira, V.F. Synthesis of New 9-Hydroxy-α- and 7-Hydroxy-β-Pyran Naphthoquinones and Cytotoxicity against Cancer Cell Lines. Org. Biomol. Chem. 2011, 9, 4315–4322. [Google Scholar] [CrossRef] [PubMed]
- Magedov, I.V.; Kireev, A.S.; Jenkins, A.R.; Evdokimov, N.M.; Lima, D.T.; Tongwa, P.; Altig, J.; Steelant, W.F.A.; Van Slambrouck, S.; Antipin, M.Y.; et al. Structural Simplification of Bioactive Natural Products with Multicomponent Synthesis. 4. 4H-Pyrano-[2,3-b]Naphthoquinones with Anticancer Activity. Bioorg. Med. Chem. Lett. 2012, 22, 5195–5198. [Google Scholar] [CrossRef]
- da Cruz, E.H.G.; Silvers, M.A.; Jardim, G.A.M.; Resende, J.M.; Cavalcanti, B.C.; Bomfim, I.S.; Pessoa, C.; de Simone, C.A.; Botteselle, G.V.; Braga, A.L.; et al. Synthesis and Antitumor Activity of Selenium-Containing Quinone-Based Triazoles Possessing Two Redox Centres, and Their Mechanistic Insights. Eur. J. Med. Chem. 2016, 122, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Ravichandiran, P.; Subramaniyan, S.A.; Kim, S.-Y.; Kim, J.-S.; Park, B.-H.; Shim, K.S.; Yoo, D.J. Synthesis and Anticancer Evaluation of 1,4-Naphthoquinone Derivatives Containing a Phenylaminosulfanyl Moiety. ChemMedChem 2019, 14, 532–544. [Google Scholar] [CrossRef]
- Patel, R.B.; Desai, P.S.; Desai, K.R.; Chikhalia, K.H. Synthesis of Pyrimidine Based Thiazolidinones and Azetidinones: Antimicrobial and Antitubercular Agents. IJC-B 2006, 45B, 747–751. [Google Scholar] [CrossRef]
- Rostom, S.A.F.; Ashour, H.M.A.; Abd El Razik, H.A. Synthesis and Biological Evaluation of Some Novel Polysubstituted Pyrimidine Derivatives as Potential Antimicrobial and Anticancer Agents. Archiv. Pharm. 2009, 342, 299–310. [Google Scholar] [CrossRef]
- Garavito, M.F.; Narváez-Ortiz, H.Y.; Zimmermann, B.H. Pyrimidine Metabolism: Dynamic and Versatile Pathways in Pathogens and Cellular Development. J. Genet. Genom. 2015, 42, 195–205. [Google Scholar] [CrossRef]
- Stolarczyk, M.; Wolska, A.; Mikołajczyk, A.; Bryndal, I.; Cieplik, J.; Lis, T.; Matera-Witkiewicz, A. A New Pyrimidine Schiff Base with Selective Activities against Enterococcus Faecalis and Gastric Adenocarcinoma. Molecules 2021, 26, 2296. [Google Scholar] [CrossRef]
- Sondhi, S.M.; Singh, N.; Johar, M.; Kumar, A. Synthesis, Anti-Inflammatory and Analgesic Activities Evaluation of Some Mono, Bi and Tricyclic Pyrimidine Derivatives. Bioorg. Med. Chem. 2005, 13, 6158–6166. [Google Scholar] [CrossRef] [PubMed]
- Abu-Hashem, A.A.; Gouda, M.A.; Badria, F.A. Synthesis of Some New Pyrimido[2′,1′:2,3]Thiazolo[4,5-b]Quinoxaline Derivatives as Anti-Inflammatory and Analgesic Agents. Eur. J. Med. Chem. 2010, 45, 1976–1981. [Google Scholar] [CrossRef] [PubMed]
- Abdellatif, K.R.A.; Abdelall, E.K.A.; Abdelgawad, M.A.; Ahmed, R.R.; Bakr, R.B. Synthesis and Anticancer Activity of Some New Pyrazolo[3,4-d]Pyrimidin-4-One Derivatives. Molecules 2014, 19, 3297–3309. [Google Scholar] [CrossRef] [PubMed]
- Shyyka, O.; Pokhodylo, N.; Finiuk, N.; Matiychuk, V.; Stoika, R.; Obushak, M. Anticancer Activity Evaluation of New Thieno[2,3-d]Pyrimidin-4(3H)-Ones and Thieno[3,2-d]Pyrimidin-4(3H)-One Derivatives. Sci. Pharm. 2018, 86, 28. [Google Scholar] [CrossRef]
- Fouad, M.M.; El-Bendary, E.R.; Suddek, G.M.; Shehata, I.A.; El-Kerdawy, M.M. Synthesis and in Vitro Antitumor Evaluation of Some New Thiophenes and Thieno[2,3-d]Pyrimidine Derivatives. Bioorg. Chem. 2018, 81, 587–598. [Google Scholar] [CrossRef]
- Cherukupalli, S.; Chandrasekaran, B.; Aleti, R.R.; Sayyad, N.; Hampannavar, G.A.; Merugu, S.R.; Rachamalla, H.R.; Banerjee, R.; Karpoormath, R. Synthesis of 4,6-Disubstituted Pyrazolo[3,4-d]Pyrimidine Analogues: Cyclin-Dependent Kinase 2 (CDK2) Inhibition, Molecular Docking and Anticancer Evaluation. J. Mol. Struct. 2019, 1176, 538–551. [Google Scholar] [CrossRef]
- Kilic, A.; Beyazsakal, L.; Işık, M.; Türkeş, C.; Necip, A.; Takım, K.; Beydemir, Ş. Mannich Reaction Derived Novel Boron Complexes with Amine-Bis(Phenolate) Ligands: Synthesis, Spectroscopy and in Vitro/in Silico Biological Studies. J. Organomet. Chem. 2020, 927, 121542. [Google Scholar] [CrossRef]
- Malki, A.; Ashour, H.M.A.; Elbayaa, R.Y.; Issa, D.A.E.; Aziz, H.A.; Chen, X. Novel 1,5-Diphenyl-6-Substituted 1H-Pyrazolo[3,4-d]Pyrimidin-4(5H)-Ones Induced Apoptosis in RKO Colon Cancer Cells. J. Enzym. Inhib. Med. Chem. 2016, 31, 1286–1299. [Google Scholar] [CrossRef]
- Dgachi, Y.; Martin, H.; Bonet, A.; Chioua, M.; Iriepa, I.; Moraleda, I.; Chabchoub, F.; Marco-Contelles, J.; Ismaili, L. Synthesis and Biological Assessment of Racemic Benzochromenopyrimidinetriones as Promising Agents for Alzheimer’s Disease Therapy. Future Med. Chem. 2017, 9, 715–721. [Google Scholar] [CrossRef]
- Cherif, M.; Horchani, M.; Al-Ghamdi, Y.O.; Almalki, S.G.; Alqurashi, Y.E.; Ben Jannet, H.; Romdhane, A. New Pyrano-1,2,3-Triazolopyrimidinone Derivatives as Anticholinesterase and Antibacterial Agents: Design, Microwave-Assisted Synthesis and Molecular Docking Study. J. Mol. Struct. 2020, 1220, 128685. [Google Scholar] [CrossRef]
- Khurana, J.M.; Lumb, A.; Chaudhary, A.; Nand, B. Synthesis and in Vitro Evaluation of Antioxidant Activity of Diverse Naphthopyranopyrimidines, Diazaanthra[2,3-d][1,3]Dioxole-7,9-Dione and Tetrahydrobenzo[a]Xanthen-11-Ones. RSC Adv. 2013, 3, 1844–1854. [Google Scholar] [CrossRef]
- Mobinikhaledi, A.; Foroughifar, N.; Mosleh, T.; Hamta, A. Synthesis of Some Novel Chromenopyrimidine Derivatives and Evaluation of Their Biological Activities. Iran J. Pharm. Res. 2014, 13, 873–879. [Google Scholar] [PubMed]
- Ravichandiran, P.; Sheet, S.; Premnath, D.; Kim, A.R.; Yoo, D.J. 1,4-Naphthoquinone Analogues: Potent Antibacterial Agents and Mode of Action Evaluation. Molecules 2019, 24, 1437. [Google Scholar] [CrossRef] [PubMed]
- Ravichandiran, P.; Masłyk, M.; Sheet, S.; Janeczko, M.; Premnath, D.; Kim, A.R.; Park, B.-H.; Han, M.-K.; Yoo, D.J. Synthesis and Antimicrobial Evaluation of 1,4-Naphthoquinone Derivatives as Potential Antibacterial Agents. Chem. Open 2019, 8, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Khurana, J.M.; Nand, B.; Saluja, P. DBU: A Highly Efficient Catalyst for One-Pot Synthesis of Substituted 3,4-Dihydropyrano[3,2-c]Chromenes, Dihydropyrano[4,3-b]Pyranes, 2-Amino-4H-Benzo[h]Chromenes and 2-Amino-4H Benzo[g]Chromenes in Aqueous Medium. Tetrahedron 2010, 66, 5637–5641. [Google Scholar] [CrossRef]
- Yu, Y.; Guo, H.; Li, X. An Improved Procedure for the Three-Component Synthesis of Benzo[g]Chromene Derivatives Using Basic Ionic Liquid. J. Heterocycl. Chem. 2011, 48, 1264–1268. [Google Scholar] [CrossRef]
- Khurana, J.M.; Magoo, D.; Chaudhary, A. Efficient and Green Approaches for the Synthesis of 4H-Benzo[g]Chromenes in Water, Under Neat Conditions, and Using Task-Specific Ionic Liquid. Synth. Commun. 2012, 42, 3211–3219. [Google Scholar] [CrossRef]
- Dekamin, M.G.; Eslami, M.; Maleki, A. Potassium Phthalimide-N-Oxyl: A Novel, Efficient, and Simple Organocatalyst for the One-Pot Three-Component Synthesis of Various 2-Amino-4H-Chromene Derivatives in Water. Tetrahedron 2013, 69, 1074–1085. [Google Scholar] [CrossRef]
- Rahimzadeh, G.; Tajbakhsh, M.; Daraie, M.; Mohammadi, M. Dysprosium–Balsalazide Complex Trapped between the Functionalized Halloysite and g-C3N4: A Novel Heterogeneous Catalyst for the Synthesis of Annulated Chromenes in Water. Appl. Organomet. Chem. 2022, 36, e6829. [Google Scholar] [CrossRef]
- Safaei-Ghomi, J.; Bateni, F.-S.; Babaei, P. CeO2/CuO@N-GQDs@NH2 Nanocomposite as a High-Performance Catalyst for the Synthesis of Benzo[g]Chromenes. Appl. Organomet. Chem. 2020, 34, e5657. [Google Scholar] [CrossRef]
- Safaei-Ghomi, J.; Enayat-Mehri, N.; Eshteghal, F. 4-(4′-Diamino-Di-Phenyl)-Sulfone Supported on Hollow Magnetic Mesoporous Fe3O4@SiO2 NPs: As a Reusable and Efficient Catalyst for the Synthesis of Ethyl 2-Amino-5,10-Dihydro-5,10-Dioxo-4-Phenyl-4H Benzo[g]Chromene-3-Carboxylates. J. Saudi Chem. Soc. 2018, 22, 485–495. [Google Scholar] [CrossRef]
| Entry | Product | Ar | Time (h) | Yield (%) |
|---|---|---|---|---|
| 1 | 3a | C6H5 | 3 | 59 |
| 2 | 3b | 4-CH3C6H4 | 4 | 53 |
| 3 | 3c | 4-OCH3C6H4 | 3 | 60 |
| 4 | 3d | 4-ClC6H4 | 4 | 55 |
| 5 | 3e | 2-ClC6H4 | 5 | 43 |
| 6 | 3f | 2,4-Cl2C6H3 | 5 | 37 |
| 7 | 3g | 3-OHC6H4 | 3 | 57 |
| 8 | 3h | 4-NO2C6H4 | 5 | 50 |
| 9 | 3i | 3-NO2C6H4 | 4 | 52 |
| 10 | 3j | 4-FC6H4 | 3 | 51 |
| IC50 (µM) | ||
|---|---|---|
| Compounds | LoVo | HCT-116 |
| 3a | # 14.99 ± 0.59 * | # 15.92 ± 0.30 |
| 3b | 19.55 ± 3.17 | 53.93 ± 7.52 |
| 3c | 24.35 ± 3.30 | 40.32 ± 3.28 |
| 3d | 30.11 ± 3.16 | 42.65 ± 3.41 |
| 3e | 18.49 ± 0.57 | # 13.70 ± 0.74 |
| 3f | 39.32 ± 5.13 | 56.03 ± 0.94 |
| 3g | # 11.79 ± 0.16 * | # 13.61 ± 2.51 |
| 3h | 47.12 ± 3.83 | 16.45 ± 2.32 |
| 3i | 33.14 ± 2.18 | # 7.15 ± 0.67 * |
| 3j | 49.03 ± 2.19 | 23.85 ± 3.08 |
| Oxaliplatin | 19.51 ± 2.35 | 13.48 ± 1.78 |
| 5-FU | 19.99 ± 3.21 | 26.98 ± 1.87 |
| Name | Molweight (g/mol) | CLogP | CLogS | H-Donors | H-Acceptors | Lipinsky Violations | Drug-Likeness | TPSA (Å2) |
|---|---|---|---|---|---|---|---|---|
| 3a | 446.461 | 4.237 | −7.271 | 0 | 6 | 0 | 4.8734 | 76.04 |
| 3b | 460.488 | 4.5809 | −7.615 | 0 | 6 | 0 | 4.83 | 76.04 |
| 3c | 476.487 | 4.167 | −7.289 | 0 | 7 | 0 | 4.9122 | 85.27 |
| 3d | 480.906 | 4.843 | −8.007 | 0 | 6 | 0 | 4.9183 | 76.04 |
| 3e | 480.906 | 4.843 | −8.007 | 0 | 6 | 0 | 4.9183 | 76.04 |
| 3f | 515.351 | 5.449 | −8.743 | 0 | 6 | 0 | 4.9183 | 76.04 |
| 3g | 462.46 | 3.8913 | −6.975 | 1 | 7 | 0 | 4.8783 | 96.27 |
| 3h | 491.458 | 3.3154 | −7.731 | 0 | 9 | 0 | −0.18075 | 121.86 |
| 3i | 491.458 | 3.3154 | −7.731 | 0 | 9 | 0 | −0.18075 | 121.86 |
| 3j | 464.451 | 4.3378 | −7.585 | 0 | 6 | 0 | 3.5334 | 76.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choura, E.; Elghali, F.; Bernard, P.J.; Msalbi, D.; Marco-Contelles, J.; Aifa, S.; Ismaili, L.; Chabchoub, F. Benzochromenopyrimidines: Synthesis, Antiproliferative Activity against Colorectal Cancer and Physicochemical Properties. Molecules 2022, 27, 7878. https://doi.org/10.3390/molecules27227878
Choura E, Elghali F, Bernard PJ, Msalbi D, Marco-Contelles J, Aifa S, Ismaili L, Chabchoub F. Benzochromenopyrimidines: Synthesis, Antiproliferative Activity against Colorectal Cancer and Physicochemical Properties. Molecules. 2022; 27(22):7878. https://doi.org/10.3390/molecules27227878
Chicago/Turabian StyleChoura, Emna, Fares Elghali, Paul J. Bernard, Dhouha Msalbi, José Marco-Contelles, Sami Aifa, Lhassane Ismaili, and Fakher Chabchoub. 2022. "Benzochromenopyrimidines: Synthesis, Antiproliferative Activity against Colorectal Cancer and Physicochemical Properties" Molecules 27, no. 22: 7878. https://doi.org/10.3390/molecules27227878
APA StyleChoura, E., Elghali, F., Bernard, P. J., Msalbi, D., Marco-Contelles, J., Aifa, S., Ismaili, L., & Chabchoub, F. (2022). Benzochromenopyrimidines: Synthesis, Antiproliferative Activity against Colorectal Cancer and Physicochemical Properties. Molecules, 27(22), 7878. https://doi.org/10.3390/molecules27227878







