Essential Oils and Their Compounds as Potential Anti-Influenza Agents
Abstract
1. Introduction
2. Methodology
3. Essential Oils as an Integral Part of Natural Products
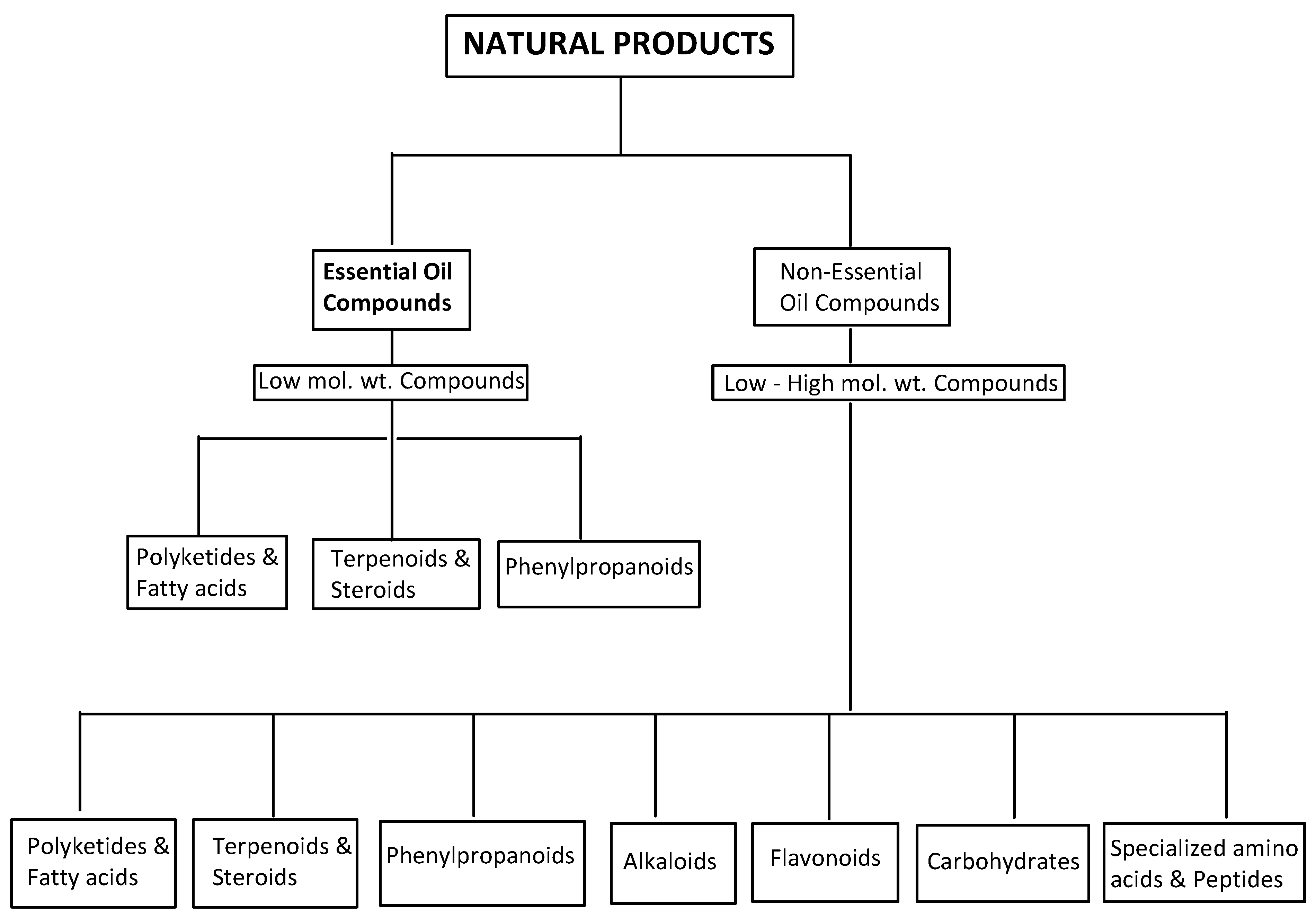
3.1. Natural Products
3.2. Plant-Derived Essential Oils
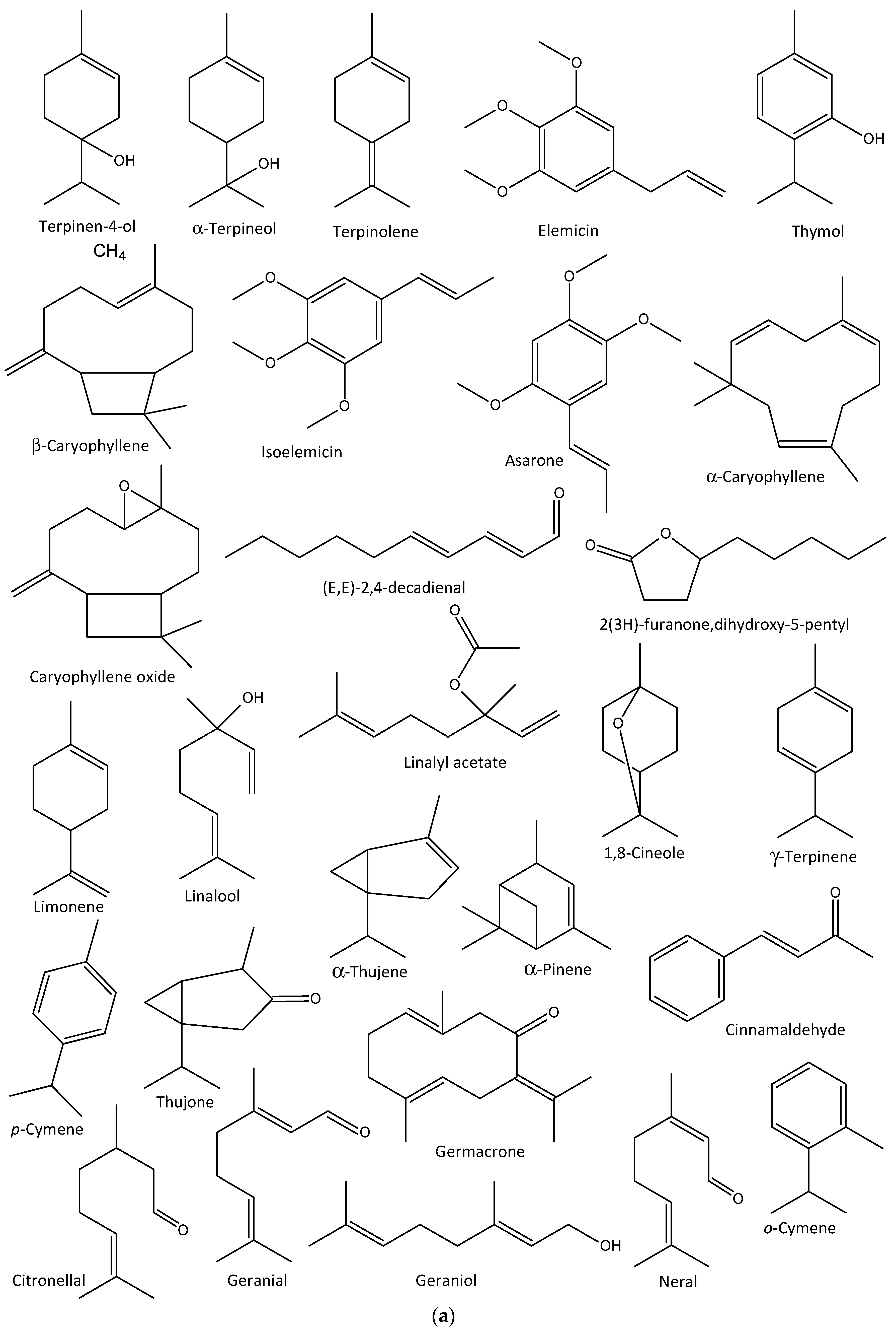
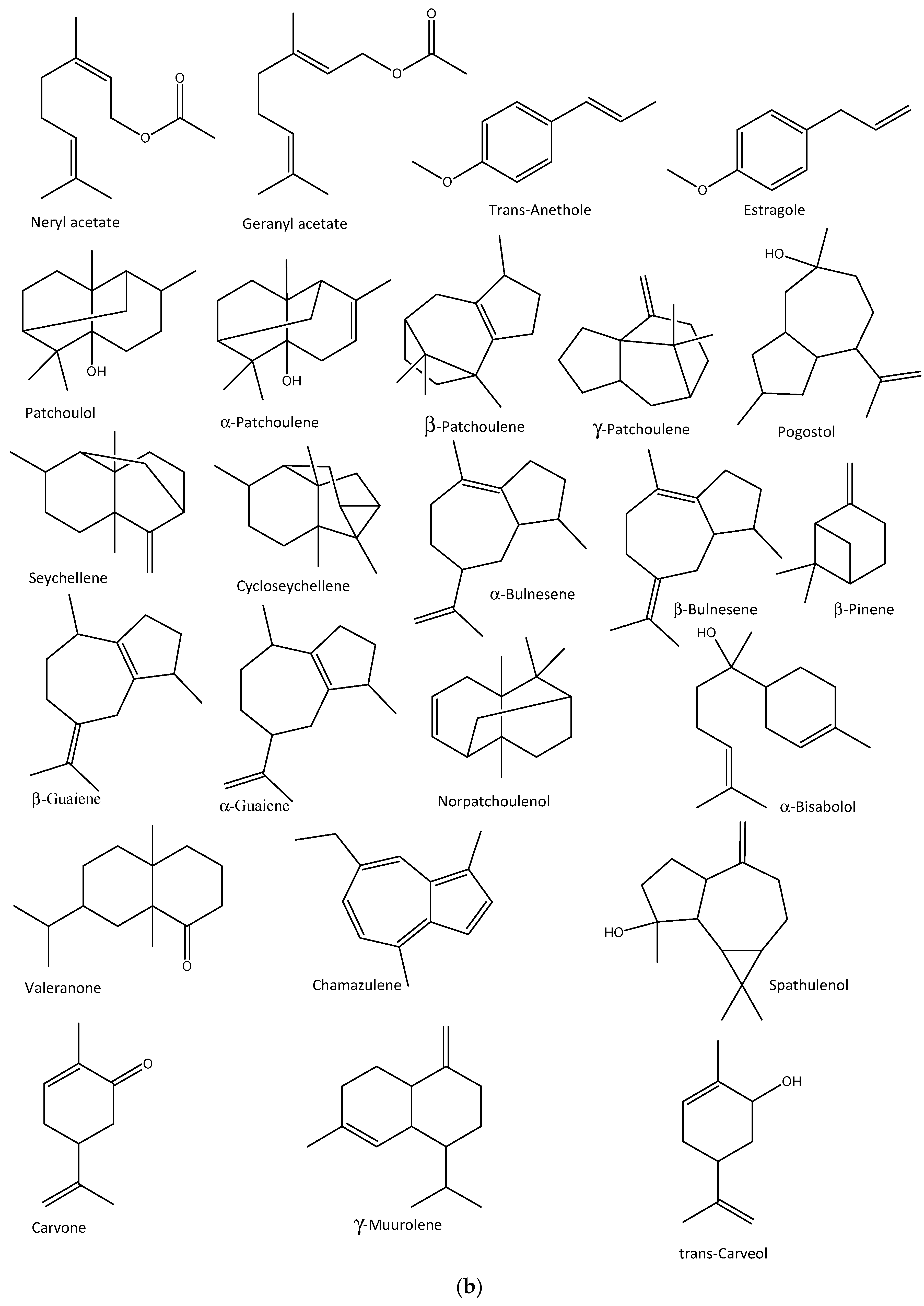
3.3. Biosynthetic Routes of Essential Oil Compounds
3.4. Classes of Essential Oils
3.5. Medicinal Applications of Plant-Derived Essential Oils
4. Influenza (Flu)
5. Essential Oils as Potential Anti-Influenza Agents
5.1. Anti-Influenza Properties of Plant-Derived Essential Oils and Their Compounds
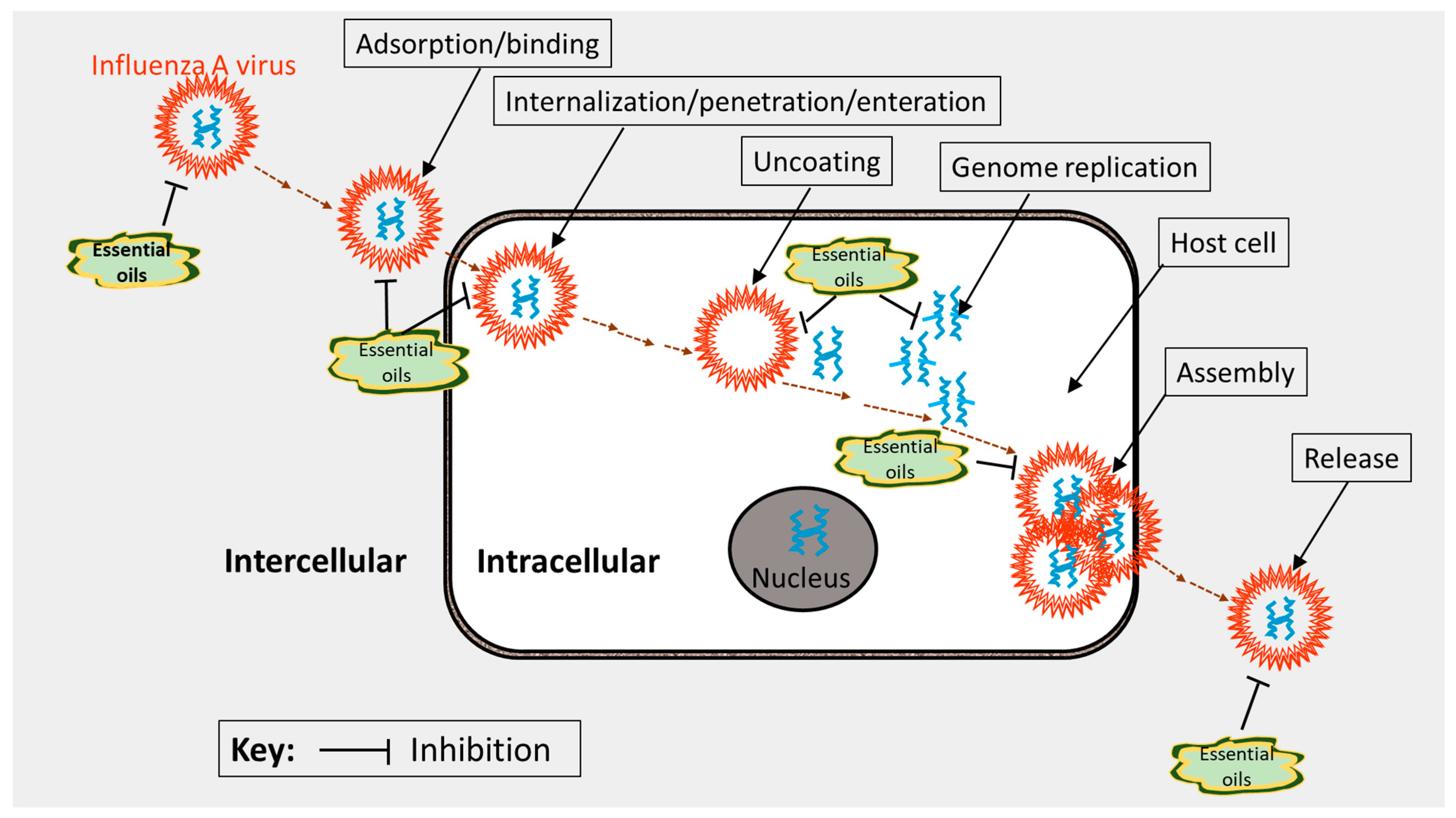
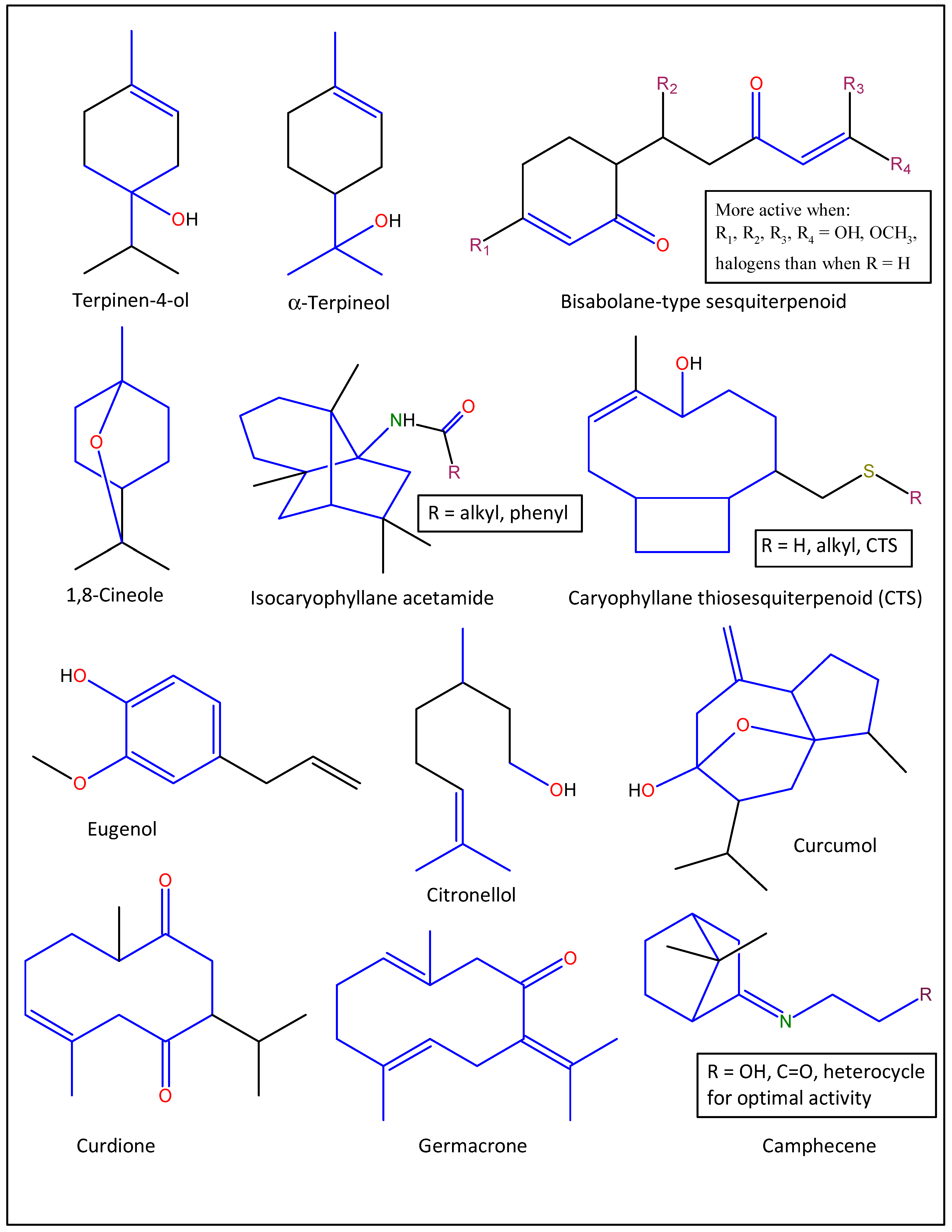
5.2. Mechanisms of Action and Structure-Activity Relationships of Some Lead Anti-Influenza Essential Oil Compounds
6. Future Perspectives and Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bruce, O.S.; Onyegbule, A.F. Biosynthesis of Natural Products. In Bioactive Compounds-Biosynthesis, Characterization and Applications; Zepka, L.Q., Nascimento, T.C.d., Jacob-Lopes, E., Eds.; IntechOpen: London, UK, 2021; 304p, Available online: https://www.intechopen.com/books/10290 (accessed on 10 October 2022).
- Ntie-Kang, F.; Svozil, D. An Enumeration of Natural Products from Microbial, Marine and Terrestrial Sources. Phys. Sci. Rev. 2020, 5, 20180121. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef] [PubMed]
- Aziz, Z.A.A.; Ahmad, A.; Setapar, S.H.M.; Karakucuk, A.; Azim, M.M.; Lokhat, D.; Rafatullah, M.; Ganash, M.; Kamal, M.A.; Ashraf, G.M. Essential Oils: Extraction Techniques, Pharmaceutical and Therapeutic Potential—A Review. Curr. Drug Metab. 2018, 19, 1100–1110. [Google Scholar] [CrossRef]
- Stephane, F.F.Y.; Jules, B.K.J. Terpenoids as Important Bioactive Constituents of Essential Oils. Essent. Oils—Bioact. Compd. New Perspect. Appl. 2020. [Google Scholar] [CrossRef]
- Dhifi, W.; Bellili, S.; Jazi, S.; Bahloul, N.; Mnif, W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines 2016, 3, 25. [Google Scholar] [CrossRef] [PubMed]
- Baser, K.H.C.; Butchbauer, G. Handbook of Essential Oils: Science, Technology and Applications; CRC Press: Boca Raton, FL, USA, 2009; p. 978042914. [Google Scholar] [CrossRef]
- Medicinal Plant Research in Africa: Pharmacology and Chemistry—Ghent University Library. Available online: https://lib.ugent.be/catalog/ebk01:2550000001064753 (accessed on 6 August 2022).
- Stratakos, A.C.; Koidis, A. Methods for Extracting Essential Oils. In Essential Oils in Food Preservation, Flavor and Safety; Academic Press: Cambridge, MA, USA, 2016; pp. 31–38. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Camele, I. An Overview of the Biological Effects of Some Mediterranean Essential Oils on Human Health. Biomed. Res. Int. 2017, 2017, 9268468. [Google Scholar] [CrossRef]
- Horváth, G.; Ács, K. Essential Oils in the Treatment of Respiratory Tract Diseases Highlighting Their Role in Bacterial Infections and Their Anti-inflammatory Action: A Review. Flavour Fragr. J. 2015, 30, 331. [Google Scholar] [CrossRef]
- Influenza (Seasonal). Available online: https://www.who.int/news-room/fact-sheets/detail/influenza-(seasonal) (accessed on 6 August 2022).
- Key Facts about Influenza (Flu)|CDC. Available online: https://www.cdc.gov/flu/about/keyfacts.htm (accessed on 6 August 2022).
- Types of Influenza Viruses|CDC. Available online: https://www.cdc.gov/flu/about/viruses/types.htm (accessed on 7 August 2022).
- Wu, P.; Presanis, A.M.; Bond, H.S.; Lau, E.H.Y.; Fang, V.J.; Cowling, B.J. A Joint Analysis of Influenza-Associated Hospitalizations and Mortality in Hong Kong, 1998–2013. Sci. Rep. 2017, 7, 929. [Google Scholar] [CrossRef]
- Senthur Nambi, P.; Suresh Kumar, D.; Gopalakrishnan, R. Influenza Vaccine. Indian J. Pract. Pediatr. 2022, 12, 372–377. [Google Scholar] [CrossRef]
- Selecting Viruses for the Seasonal Influenza Vaccine|CDC. Available online: https://www.cdc.gov/flu/prevent/vaccine-selection.htm (accessed on 7 August 2022).
- Ullah, I.; Khan, K.S.; Tahir, M.J.; Ahmed, A.; Harapan, H. Myths and Conspiracy Theories on Vaccines and COVID-19: Potential Effect on Global Vaccine Refusals. Vacunas 2021, 22, 93. [Google Scholar] [CrossRef]
- Influenza (Flu) Antiviral Drugs and Related Information|FDA. Available online: https://www.fda.gov/drugs/information-drug-class/influenza-flu-antiviral-drugs-and-related-information (accessed on 7 August 2022).
- Yin, H.; Jiang, N.; Shi, W.; Chi, X.; Liu, S.; Chen, J.L.; Wang, S. Development and Effects of Influenza Antiviral Drugs. Molecules 2021, 26, 810. [Google Scholar] [CrossRef] [PubMed]
- Bhanot, A.; Bhanot, A.; Sharma, R.; Noolvi, M.N. Natural Sources as Potential Anti-Cancer Agents: A Review. Int. J. Phytomed. 2011, 3, 9–26. [Google Scholar]
- Bernardini, S.; Tiezzi, A.; Laghezza Masci, V.; Ovidi, E. Natural Products for Human Health: An Historical Overview of the Drug Discovery Approaches. Nat. Prod. Res. 2018, 32, 1926–1950. [Google Scholar] [CrossRef] [PubMed]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. The Influence of Natural Products upon Drug Discovery. Nat. Prod. Rep. 2000, 17, 215–234. [Google Scholar] [CrossRef]
- Billowria, K.; Ali, R.; Rangra, N.K.; Kumar, R.; Chawla, P.A. Bioactive Flavonoids: A Comprehensive Review on Pharmacokinetics and Analytical Aspects. Crit. Rev. Anal. Chem. 2022, 1–15. [Google Scholar] [CrossRef]
- Shibata, M.A.; Khan, I.A.; Iinuma, M.; Shirai, T. Natural Products for Medicine. J. Biomed. Biotechnol. 2012, 2012, 147120. [Google Scholar] [CrossRef] [PubMed]
- Ekiert, H.M.; Szopa, A. Biological Activities of Natural Products. Molecules 2020, 25, 5769. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Orhan, I.E.; Banach, M.; Rollinger, J.M.; Barreca, D.; Weckwerth, W.; Bauer, R.; Bayer, E.A.; et al. Natural Products in Drug Discovery: Advances and Opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- Omrani, M.; Keshavarz, M.; Nejad Ebrahimi, S.; Mehrabi, M.; McGaw, L.J.; Ali Abdalla, M.; Mehrbod, P. Potential Natural Products Against Respiratory Viruses: A Perspective to Develop Anti-COVID-19 Medicines. Front. Pharmacol. 2021, 11, 2115. [Google Scholar] [CrossRef]
- Springob, K.; Kutchan, T.M. Introduction to the Different Classes of Natural Products. In Plant-Derived Natural Products: Synthesis, Function, and Application; Springer: New York, NY, USA, 2009; pp. 3–50. [Google Scholar] [CrossRef]
- Jabeen, S.; Hanif, M.A.; Khan, M.M.; Waseem, R.; Qadri, K. Natural Products Sources and Their Active Compounds on Disease Prevention: A Review. IJCBS 2014, 6, 76–83. [Google Scholar]
- Ríos, J.L. Essential Oils: What They Are and How the Terms Are Used and Defined. In Essential Oils in Food Preservation, Flavor and Safety; Academic Press: Cambridge, MA, USA, 2016; pp. 3–10. [Google Scholar] [CrossRef]
- Sonwa, M.M. Isolation and Structure Elucidation of Essential Oil Constituents : Comparative Study of the Oils of Cyperus Alopecuroides, Cyperus Papyrus, and Cyperus Rotundus. Ph.D. Thesis, Staats-und Universitätsbibliothek Hamburg Carl von Ossietzky, Hamburg, Germany, 2000. [Google Scholar]
- Sharifi-Rad, J.; Sureda, A.; Tenore, G.C.; Daglia, M.; Sharifi-Rad, M.; Valussi, M.; Tundis, R.; Sharifi-Rad, M.; Loizzo, M.R.; Oluwaseun Ademiluyi, A.; et al. Biological Activities of Essential Oils: From Plant Chemoecology to Traditional Healing Systems. Molecules 2017, 22, 70. [Google Scholar] [CrossRef] [PubMed]
- Fürstenberg-Hägg, J.; Zagrobelny, M.; Bak, S. Plant Defense against Insect Herbivores. Int. J. Mol. Sci. 2013, 14, 10242–10297. [Google Scholar] [CrossRef] [PubMed]
- Evans, W.C. Trease and Evans’ Pharmacognosy: Sixteenth Edition; Elsevier Health Sciences: Amsterdam, The Netherlands, 2009; pp. 1–603. [Google Scholar]
- Raut, J.S.; Karuppayil, S.M. A Status Review on the Medicinal Properties of Essential Oils. Ind. Crops Prod. 2014, 62, 250–264. [Google Scholar] [CrossRef]
- Sowndhararajan, K.; Deepa, P.; Kim, M.; Park, S.J.; Kim, S. A Review of the Composition of the Essential Oils and Biological Activities of Angelica Species. Sci. Pharm. 2017, 85, 33. [Google Scholar] [CrossRef]
- Marzouki, H.; Khaldi, A.; Falconieri, D.; Piras, A.; Marongiu, B.; Molicotti, P.; Zanetti, S. Essential Oils of Daucus carota Subsp. Carota of Tunisia Obtained by Supercritical Carbon Dioxide Extraction. NPC Nat. Prod. Commun. 2010, 5, 1955–1958. [Google Scholar] [CrossRef]
- Stanojevic, L.P.; Marjanovic-Balaban, Z.R.; Kalaba, V.D.; Stanojevic, J.S.; Cvetkovic, D.J. Chemical Composition, Antioxidant and Antimicrobial Activity of Chamomile Flowers Essential Oil (Matricaria chamomilla L.). J. Essent. Oil Bear. Plants 2016, 19, 2017–2028. [Google Scholar] [CrossRef]
- Candan, F.; Unlu, M.; Tepe, B.; Daferera, D.; Polissiou, M.; Sökmen, A.; Akpulat, H.A. Antioxidant and Antimicrobial Activity of the Essential Oil and Methanol Extracts of Achillea millefolium Subsp. millefolium Afan. (Asteraceae). J. Ethnopharmacol. 2003, 87, 215–220. [Google Scholar] [CrossRef]
- Orav, A.; Arak, E.; Raal, A. Phytochemical Analysis of the Essential Oil of Achillea millefolium L. from Various European Countries. Nat. Prod. Res. 2006, 20, 1082–1088. [Google Scholar] [CrossRef]
- Demirpolat, A.; Akman, F.; Kazachenko, A.S. An Experimental and Theoretical Study on Essential Oil of Aethionema sancakense: Characterization, Molecular Properties and RDG Analysis. Molecules 2022, 27, 6129. [Google Scholar] [CrossRef]
- Morales-López, J.; Centeno-Álvarez, M.; Nieto-Camacho, A.; López, M.G.; Pérez-Hernández, E.; Pérez-Hernández, N.; Fernández-Martínez, E. Evaluation of Antioxidant and Hepatoprotective Effects of White Cabbage Essential Oil. Pharm. Biol. 2016, 55, 233–241. [Google Scholar] [CrossRef]
- Al-Harrasi, A.; Al-Saidi, S. Phytochemical Analysis of the Essential Oil from Botanically Certified Oleogum Resin of Boswellia sacra (Omani Luban). Molecules 2008, 13, 2181–2189. [Google Scholar] [CrossRef] [PubMed]
- Marongiu, B.; Piras, A.; Porcedda, S.; Scorciapino, A. Chemical Composition of the Essential Oil and Supercritical CO2 Extract of Commiphora myrrha (Nees) Engl. and of Acorus calamus L. J. Agric. Food Chem. 2005, 53, 7939–7943. [Google Scholar] [CrossRef] [PubMed]
- Ljaljević Grbić, M.; Unković, N.; Dimkić, I.; Janaćković, P.; Gavrilović, M.; Stanojević, O.; Stupar, M.; Vujisić, L.; Jelikić, A.; Stanković, S.; et al. Frankincense and Myrrh Essential Oils and Burn Incense Fume against Micro-Inhabitants of Sacral Ambients. Wisdom of the Ancients? J. Ethnopharmacol. 2018, 219, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Selim, S.A.; Adam, M.E.; Hassan, S.M.; Albalawi, A.R. Chemical Composition, Antimicrobial and Antibiofilm Activity of the Essential Oil and Methanol Extract of the Mediterranean Cypress (Cupressus sempervirens L.). BMC Complement. Altern. Med. 2014, 14, 179. [Google Scholar] [CrossRef]
- Höferl, M.; Stoilova, I.; Schmidt, E.; Wanner, J.; Jirovetz, L.; Trifonova, D.; Krastev, L.; Krastanov, A. Chemical Composition and Antioxidant Properties of Juniper Berry (Juniperus communis L.) Essential Oil. Action of the Essential Oil on the Antioxidant Protection of Saccharomyces cerevisiae Model Organism. Antioxidants 2014, 3, 81–98. [Google Scholar] [CrossRef]
- Zhang, K.; Yao, L. The Anxiolytic Effect of Juniperus virginiana L. Essential Oil and Determination of Its Active Constituents. Physiol. Behav. 2018, 189, 50–58. [Google Scholar] [CrossRef]
- Sakai, A.; Yoshimura, H. Monoterpenes of Salvia leucophylla. Curr. Bioact. Compd. 2012, 8, 90–100. [Google Scholar] [CrossRef]
- Kathirvel, P.; Ravi, S. Chemical Composition of the Essential Oil from Basil (Ocimum basilicum Linn.) and Its in Vitro Cytotoxicity against HeLa and HEp-2 Human Cancer Cell Lines and NIH 3T3 Mouse Embryonic Fibroblasts. Nat. Prod. Res. 2012, 26, 1112–1118. [Google Scholar] [CrossRef]
- Mimica-Dukić, N.; Božin, B.; Soković, M.; Mihajlović, B.; Matavulj, M. Antimicrobial and Antioxidant Activities of Three Mentha Species Essential Oils. Planta Med. 2003, 69, 413–419. [Google Scholar] [CrossRef]
- Snoussi, M.; Noumi, E.; Trabelsi, N.; Flamini, G.; Papetti, A.; de Feo, V. Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains. Molecules 2015, 20, 14402. [Google Scholar] [CrossRef]
- Özcan, M.M.; Chalchat, J.C. Chemical Composition and Antifungal Activity of Rosemary (Rosmarinus officinalis L.) Oil from Turkey. Int. J. Food Sci. Nutr. 2008, 59, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Ben Jabeur, M.; Somai-Jemmali, L.; Hamada, W. Thyme Essential Oil as an Alternative Mechanism: Biofungicide-Causing Sensitivity of Mycosphaerella graminicola. J. Appl. Microbiol. 2017, 122, 932–939. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Geng, Z.; Zhang, W.; Liang, J.; Wang, C.; Deng, Z.; Du, S. The Chemical Composition of Essential Oils from Cinnamomum camphora and Their Insecticidal Activity against the Stored Product Pests. Int. J. Mol. Sci. 2016, 17, 1836. [Google Scholar] [CrossRef]
- Alizadeh Behbahani, B.; Falah, F.; Lavi Arab, F.; Vasiee, M.; Tabatabaee Yazdi, F. Chemical Composition and Antioxidant, Antimicrobial, and Antiproliferative Activities of Cinnamomum zeylanicum Bark Essential Oil. Evid. Based Complement. Altern. Med. 2020, 2020, 5190603. [Google Scholar] [CrossRef] [PubMed]
- Caputo, L.; Nazzaro, F.; Souza, L.F.; Aliberti, L.; de Martino, L.; Fratianni, F.; Coppola, R.; de Feo, V. Laurus nobilis: Composition of Essential Oil and Its Biological Activities. Molecules 2017, 22, 930. [Google Scholar] [CrossRef] [PubMed]
- Elaissi, A.; Rouis, Z.; Mabrouk, S.; Bel Haj Salah, K.; Aouni, M.; Khouja, M.L.; Farhat, F.; Chemli, R.; Harzallah-Skhiri, F. Correlation between Chemical Composition and Antibacterial Activity of Essential Oils from Fifteen Eucalyptus species Growing in the Korbous and Jbel Abderrahman Arboreta (North East Tunisia). Molecules 2012, 17, 3044–3057. [Google Scholar] [CrossRef] [PubMed]
- Sebei, K.; Sakouhi, F.; Herchi, W.; Khouja, M.L.; Boukhchina, S. Chemical Composition and Antibacterial Activities of Seven Eucalyptus species Essential Oils Leaves. Biol. Res. 2015, 48, 7. [Google Scholar] [CrossRef]
- Usai, M.; Marchetti, M.; Culeddu, N.; Mulas, M. Chemical Composition of Myrtle (Myrtus communis L.) Berries Essential Oils as Observed in a Collection of Genotypes. Molecules 2018, 23, 2502. [Google Scholar] [CrossRef]
- de Groot, A.C.; Schmidt, E. Tea Tree Oil: Contact Allergy and Chemical Composition. Contact Dermat. 2016, 75, 129–143. [Google Scholar] [CrossRef]
- Stevanovic, F.; Francezon, N.; Stevanovic, T. Spruce Bark Oil. Bioresources 2017, 12, 2635–2645. [Google Scholar]
- Samadi, N.; Manayi, A.; Vazirian, M.; Samadi, M.; Zeinalzadeh, Z.; Saghari, Z.; Abadian, N.; Mozaffarian, V.O.A.; Khanavi, M. Chemical Composition and Antimicrobial Activity of the Essential Oil of Anthemis altissima L. Var. altissima. Nat. Prod. Res. 2012, 26, 1931–1934. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, T.; Kafil, H.S.; Asnaashari, S.; Farajnia, S.; Delazar, A.; Baek, S.C.; Hamishehkar, H.; Kim, K.H. Chemical Composition and Antimicrobial Activity of Essential Oils from the Aerial Parts of Pinus eldarica Grown in Northwestern Iran. Molecules 2019, 24, 3203. [Google Scholar] [CrossRef] [PubMed]
- Boukhatem, M.N.; Ferhat, M.A.; Kameli, A.; Saidi, F.; Kebir, H.T. Lemon Grass (Cymbopogon citratus) Essential Oil as a Potent Anti-Inflammatory and Antifungal Drugs. Libyan J. Med. 2014, 9, 25431. [Google Scholar] [CrossRef] [PubMed]
- Dubey, V.S.; Luthra, R. Biotransformation of Geranyl Acetate to Geraniol during Palmarosa (Cymbopogon martinii, Roxb. Wats. Var. Motia) Inflorescence Development. Phytochemistry 2001, 57, 675–680. [Google Scholar] [CrossRef]
- de Toledo, L.G.; dos Santos Ramos, M.A.; Spósito, L.; Castilho, E.M.; Pavan, F.R.; de Oliveira Lopes, É.; Zocolo, G.J.; Silva, F.A.N.; Soares, T.H.; dos Santos, A.G.; et al. Essential Oil of Cymbopogon nardus (L.) Rendle: A Strategy to Combat Fungal Infections Caused by Candida species. Int. J. Mol. Sci. 2016, 17, 1252. [Google Scholar] [CrossRef]
- Ben Hsouna, A.; ben Halima, N.; Smaoui, S.; Hamdi, N. Citrus Lemon Essential Oil: Chemical Composition, Antioxidant and Antimicrobial Activities with Its Preservative Effect against Listeria monocytogenes Inoculated in Minced Beef Meat. Lipids Health Dis. 2017, 16, 146. [Google Scholar] [CrossRef]
- Miya, G.; Nyalambisa, M.; Oyedeji, O.; Gondwe, M.; Oyedeji, A. Chemical Profiling, Toxicity and Anti-Inflammatory Activities of Essential Oils from Three Grapefruit Cultivars from KwaZulu-Natal in South Africa. Molecules 2021, 26, 3387. [Google Scholar] [CrossRef]
- Hu, W.; Zhang, N.; Chen, H.; Zhong, B.; Yang, A.; Kuang, F.; Ouyang, Z.; Chun, J. Fumigant Activity of Sweet Orange Essential Oil Fractions Against Red Imported Fire Ants (Hymenoptera: Formicidae). J. Econ. Entomol. 2017, 110, 1556–1562. [Google Scholar] [CrossRef]
- Liju, V.B.; Jeena, K.; Kuttan, R. An Evaluation of Antioxidant, Anti-Inflammatory, and Antinociceptive Activities of Essential Oil from Curcuma longa L. Indian J. Pharmacol. 2011, 43, 526–531. [Google Scholar] [CrossRef]
- Noumi, E.; Snoussi, M.; Alreshidi, M.M.; Rekha, P.D.; Saptami, K.; Caputo, L.; de Martino, L.; Souza, L.F.; Msaada, K.; Mancini, E.; et al. Chemical and Biological Evaluation of Essential Oils from Cardamom species. Molecules 2018, 23, 2818. [Google Scholar] [CrossRef]
- Tarfaoui, K.; Brhadda, N.; Ziri, R.; Oubihi, A.; Imtara, H.; Haida, S.; al kamaly, O.M.; Saleh, A.; Parvez, M.K.; Fettach, S.; et al. Chemical Profile, Antibacterial and Antioxidant Potential of Zingiber officinale Roscoe and Elettaria cardamomum (L.) Maton Essential Oils and Extracts. Plants 2022, 11, 1487. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, J.; Zhang, Y. Research Progress on Chemical Constituents of Zingiber officinale Roscoe. Biomed. Res. Int. 2019, 2019, 5370823. [Google Scholar] [CrossRef] [PubMed]
- Nagegowda, D.A. Plant Volatile Terpenoid Metabolism: Biosynthetic Genes, Transcriptional Regulation and Subcellular Compartmentation. FEBS Lett. 2010, 584, 2965–2973. [Google Scholar] [CrossRef] [PubMed]
- Rehman, R.; Hanif, M.A.; Mushtaq, Z.; Al-Sadi, A.M. Food Reviews International Biosynthesis of Essential Oils in Aromatic Plants: A Review. Food Rev. Int. 2016, 32, 117–160. [Google Scholar] [CrossRef]
- Pulido, P.; Perello, C.; Rodriguez-Concepcion, M. New Insights into Plant Isoprenoid Metabolism. Mol. Plant 2012, 5, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Masango, P. Cleaner Production of Essential Oils by Steam Distillation. J. Clean. Prod. 2005, 13, 833–839. [Google Scholar] [CrossRef]
- Arroo, R. Eberhard Breitmaier. Terpenes—Flavors, Fragrances, Pharmaca, Pheromones. Wiley-VCH, 2006, 214 Pp ISBN 3-527-31786-4. Appl. Organomet. Chem. 2007, 21, 377. [Google Scholar] [CrossRef]
- Liu, Y.; Luo, S.H.; Schmidt, A.; Wang, G.D.; Sun, G.L.; Grant, M.; Kuang, C.; Yang, M.J.; Jing, S.X.; Li, C.H.; et al. A Geranylfarnesyl Diphosphate Synthase Provides the Precursor for Sesterterpenoid (C25) Formation in the Glandular Trichomes of the Mint Species Leucosceptrum canum. Plant Cell 2016, 28, 804–822. [Google Scholar] [CrossRef] [PubMed]
- Pour, P.M.; Behzad, S.; Asgari, S.; Khankandi, H.P.; Farzaei, M.H. Sesterterpenoids. In Recent Advances in Natural Products Analysis; Elsevier: Amsterdam, The Netherlands, 2020; pp. 347–391. [Google Scholar] [CrossRef]
- Islam, M.T.; da Mata, A.M.O.F.; de Aguiar, R.P.S.; Paz, M.F.C.J.; de Alencar, M.V.O.B.; Ferreira, P.M.P.; de Carvalho Melo-Cavalcante, A.A. Therapeutic Potential of Essential Oils Focusing on Diterpenes. Phytother. Res. 2016, 30, 1420–1444. [Google Scholar] [CrossRef]
- George, K.W.; Alonso-Gutierrez, J.; Keasling, J.D.; Lee, T.S. Isoprenoid Drugs, Biofuels, and Chemicals—Artemisinin, Farnesene, and Beyond. Adv. Biochem. Eng. Biotechnol. 2015, 148, 355–389. [Google Scholar] [CrossRef]
- Cincotta, F.; Verzera, A.; Tripodi, G.; Condurso, C. Determination of Sesquiterpenes in Wines by HS-SPME Coupled with GC-MS. Chromatography 2015, 2, 410–421. [Google Scholar] [CrossRef]
- Sarup, P.; Bala, S.; Kamboj, S. Pharmacology and Phytochemistry of Oleo-Gum Resin of Commiphora wightii (Guggulu). Scientifica 2015, 2015, 138039. [Google Scholar] [CrossRef]
- Ali, B.; Al-Wabel, N.A.; Shams, S.; Ahamad, A.; Khan, S.A.; Anwar, F. Essential Oils Used in Aromatherapy: A Systemic Review. Asian Pac. J. Trop. Biomed. 2015, 5, 601–611. [Google Scholar] [CrossRef]
- Borges, R.S.; Keita, H.; Ortiz, B.L.S.; dos Santos Sampaio, T.I.; Ferreira, I.M.; Lima, E.S.; de Jesus Amazonas da Silva, M.; Fernandes, C.P.; de Faria Mota Oliveira, A.E.M.; da Conceição, E.C.; et al. Anti-Inflammatory Activity of Nanoemulsions of Essential Oil from Rosmarinus officinalis L.: In Vitro and in Zebrafish Studies. Inflammopharmacology 2018, 26, 1057–1080. [Google Scholar] [CrossRef] [PubMed]
- Oriola, A.O.; Oyedeji, A.O. Plant-Derived Natural Products as Lead Agents against Common Respiratory Diseases. Molecules 2022, 27, 3054. [Google Scholar] [CrossRef]
- Liao, B.; Hu, H.; Xiao, S.; Zhou, G.; Sun, W.; Chu, Y.; Meng, X.; Wei, J.; Zhang, H.; Xu, J.; et al. Global Pharmacopoeia Genome Database Is an Integrated and Mineable Genomic Database for Traditional Medicines Derived from Eight International Pharmacopoeias. Sci. China Life Sci. 2022, 65, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Silveira, D.; Prieto-Garcia, J.M.; Boylan, F.; Estrada, O.; Fonseca-Bazzo, Y.M.; Jamal, C.M.; Magalhães, P.O.; Pereira, E.O.; Tomczyk, M.; Heinrich, M. COVID-19: Is There Evidence for the Use of Herbal Medicines as Adjuvant Symptomatic Therapy? Front. Pharmacol. 2020, 11, 1479. [Google Scholar] [CrossRef]
- Boktor, S.W.; Hafner, J.W. Influenza. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Glezen, W.P. Emerging Infections: Pandemic Influenza. Epidemiol. Rev. 1996, 18, 64–76. [Google Scholar] [CrossRef]
- Yang, L.; Chan, K.P.; Wong, C.M.; Chiu, S.S.S.; Magalhaes, R.J.S.; Thach, T.Q.; Peiris, J.S.M.; Clements, A.C.A.; Hu, W. Comparison of Influenza Disease Burden in Older Populations of Hong Kong and Brisbane: The Impact of Influenza and Pneumococcal Vaccination. BMC Infect. Dis. 2019, 19, 162. [Google Scholar] [CrossRef]
- Hagey, R.J.; Elazar, M.; Pham, E.A.; Tian, S.; Ben-Avi, L.; Bernardin-Souibgui, C.; Yee, M.F.; Moreira, F.R.; Rabinovitch, M.V.; Meganck, R.M.; et al. Programmable Antivirals to Target Conserved Essential Shapes in Pandemic Viral Genomes. Nat. Med. 2022, 28, 1944–1955. [Google Scholar] [CrossRef]
- Sellers, S.A.; Hagan, R.S.; Hayden, F.G.; Fischer, W.A. The Hidden Burden of Influenza: A Review of the Extra-Pulmonary Complications of Influenza Infection. Influenza Other Respir. Viruses 2017, 11, 372–393. [Google Scholar] [CrossRef]
- Krammer, F.; Smith, G.J.D.; Fouchier, R.A.M.; Peiris, M.; Kedzierska, K.; Doherty, P.C.; Palese, P.; Shaw, M.L.; Treanor, J.; Webster, R.G.; et al. Influenza. Nat. Rev. Dis. Prim. 2018, 4, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Alonso, W.J.; Feng, L.; Tan, Y.; Shu, Y.; Yang, W.; Viboud, C. Characterization of Regional Influenza Seasonality Patterns in China and Implications for Vaccination Strategies: Spatio-Temporal Modeling of Surveillance Data. PLoS Med. 2013, 10, e1001552. [Google Scholar] [CrossRef] [PubMed]
- Palese, P.; Tumpey, T.M.; Garcia-Sastre, A. What Can We Learn from Reconstructing the Extinct 1918 Pandemic Influenza Virus? Immunity 2006, 24, 121–124. [Google Scholar] [CrossRef]
- Iuliano, A.D.; Roguski, K.M.; Chang, H.H.; Muscatello, D.J.; Palekar, R.; Tempia, S.; Cohen, C.; Gran, J.M.; Schanzer, D.; Cowling, B.J.; et al. Estimates of Global Seasonal Influenza-Associated Respiratory Mortality: A Modelling Study. Lancet 2018, 391, 1285–1300. [Google Scholar] [CrossRef]
- Global Influenza Programme. Available online: https://www.who.int/teams/global-influenza-programme/vaccines (accessed on 13 September 2022).
- Young, K.; Gemmill, I.; Harrison, R. Summary of the NACI Seasonal Influenza Vaccine Statement for 2020–2021. Can. Commun. Dis. Rep. 2020, 46, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Seasonal Flu Vaccines|CDC. Available online: https://www.cdc.gov/flu/prevent/flushot.htm (accessed on 13 September 2022).
- Paules, C.I.; Sullivan, S.G.; Subbarao, K.; Fauci, A.S. Chasing Seasonal Influenza—The Need for a Universal Influenza Vaccine. N. Engl. J. Med. 2018, 378, 7–9. [Google Scholar] [CrossRef] [PubMed]
- Saito, N.; Komori, K.; Suzuki, M.; Morimoto, K.; Kishikawa, T.; Yasaka, T.; Ariyoshi, K. Negative Impact of Prior Influenza Vaccination on Current Influenza Vaccination among People Infected and Not Infected in Prior Season: A Test-Negative Case-Control Study in Japan. Vaccine 2017, 35, 687–693. [Google Scholar] [CrossRef]
- DiazGranados, C.A.; Denis, M.; Plotkin, S. Seasonal Influenza Vaccine Efficacy and Its Determinants in Children and Non-Elderly Adults: A Systematic Review with Meta-Analyses of Controlled Trials. Vaccine 2012, 31, 49–57. [Google Scholar] [CrossRef]
- Gasparini, R.; Amicizia, D.; Lai, P.L.; Bragazzi, N.L.; Panatto, D. Compounds with Anti-Influenza Activity: Present and Future of Strategies for the Optimal Treatment and Management of Influenza Part II: Future Compounds against Influenza Virus. J. Prev. Med. Hyg. 2014, 55, 109. [Google Scholar]
- Świerczyńska, M.; Mirowska-Guzel, D.M.; Pindelska, E. Antiviral Drugs in Influenza. Int. J. Environ. Res. Public Health 2022, 19, 3018. [Google Scholar] [CrossRef] [PubMed]
- Doll, M.K.; Winters, N.; Boikos, C.; Kraicer-Melamed, H.; Gore, G.; Quach, C. Safety and Effectiveness of Neuraminidase Inhibitors for Influenza Treatment, Prophylaxis, and Outbreak Control: A Systematic Review of Systematic Reviews and/or Meta-Analyses. J. Antimicrob. Chemother. 2017, 72, 2990–3007. [Google Scholar] [CrossRef]
- Hayden, F.G.; Shindo, N. Influenza Virus Polymerase Inhibitors in Clinical Development. Curr. Opin. Infect. Dis. 2019, 32, 176. [Google Scholar] [CrossRef] [PubMed]
- Asif, M.; Saleem, M.; Saadullah, M.; Yaseen, H.S.; al Zarzour, R. COVID-19 and Therapy with Essential Oils Having Antiviral, Anti-Inflammatory, and Immunomodulatory Properties. Inflammopharmacology 2020, 28, 1153. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.F.; Wang, W.; Dai, X.Y.; Wang, Z.Y.; Shen, Z.H.; Ying, H.Z.; Yu, C.H. Chemical Compositions and Anti-Influenza Activities of Essential Oils from Mosla dianthera. J. Ethnopharmacol. 2012, 139, 668–671. [Google Scholar] [CrossRef]
- Zai-Chang, Y.; Bo-Chu, W.; Xiao-Sheng, Y.; Qiang, W. Chemical Composition of the Volatile Oil from Cynanchum stauntonii and Its Activities of Anti-Influenza Virus. Colloids Surf. B Biointerfaces 2005, 43, 198–202. [Google Scholar] [CrossRef]
- Garozzo, A.; Timpanaro, R.; Stivala, A.; Bisignano, G.; Castro, A. Activity of Melaleuca alternifolia (Tea Tree) Oil on Influenza Virus A/PR/8: Study on the Mechanism of Action. Antivir. Res. 2011, 89, 83–88. [Google Scholar] [CrossRef]
- Lee, D.H.; Kim, S.H.; Eun, J.S.; Shin, T.Y. Mosla dianthera Inhibits Mast Cell-Mediated Allergic Reactions through the Inhibition of Histamine Release and Inflammatory Cytokine Production. Toxicol. Appl. Pharmacol. 2006, 216, 479–484. [Google Scholar] [CrossRef]
- Madia, V.N.; Toscanelli, W.; de Vita, D.; de Angelis, M.; Messore, A.; Ialongo, D.; Scipione, L.; Tudino, V.; D’auria, F.D.; di Santo, R.; et al. Ultrastructural Damages to H1N1 Influenza Virus Caused by Vapor Essential Oils. Molecules 2022, 27, 3718. [Google Scholar] [CrossRef]
- Selvarani, V.; James, H. The Activity of Cedar Leaf Oil Vapor against Respiratory Viruses: Practical Applications. J. Appl. Pharm. Sci. 2013, 3, 11–15. [Google Scholar] [CrossRef]
- Hayashi, K.; Imanishi, N.; Kashiwayama, Y.; Kawano, A.; Terasawa, K.; Shimada, Y.; Ochiai, H. Inhibitory Effect of Cinnamaldehyde, Derived from Cinnamomi Cortex, on the Growth of Influenza A/PR/8 Virus in Vitro and in Vivo. Antivir. Res. 2007, 74, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Wani, A.R.; Yadav, K.; Khursheed, A.; Rather, M.A. An Updated and Comprehensive Review of the Antiviral Potential of Essential Oils and Their Chemical Constituents with Special Focus on Their Mechanism of Action against Various Influenza and Coronaviruses. Microb. Pathog. 2021, 152, 104620. [Google Scholar] [CrossRef] [PubMed]
- Nagy, M.M.; Al-Mahdy, D.A.; Abd El Aziz, O.M.; Kandil, A.M.; Tantawy, M.A.; el Alfy, T.S.M. Chemical Composition and Antiviral Activity of Essential Oils from Citrus reshni Hort. Ex Tanaka (Cleopatra Mandarin) Cultivated in Egypt. J. Essent. Oil-Bear. Plants 2018, 21, 264–272. [Google Scholar] [CrossRef]
- Liao, Q.; Qian, Z.; Liu, R.; An, L.; Chen, X. Germacrone Inhibits Early Stages of Influenza Virus Infection. Antivir. Res. 2013, 100, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Nocera, F.P.; Mancini, S.; Najar, B.; Bertelloni, F.; Pistelli, L.; de Filippis, A.; Fiorito, F.; de Martino, L.; Fratini, F. Antimicrobial Activity of Some Essential Oils against Methicillin-Susceptible and Methicillin-Resistant Staphylococcus Pseudintermedius-Associated Pyoderma in Dogs. Animals 2020, 10, 1782. [Google Scholar] [CrossRef]
- Li, Y.; Xu, Y.; Lai, Y.; Liao, S.; Liu, N.; Xu, P. Intranasal Co-Administration of 1,8-Cineole with Influenza Vaccine Provide Cross-Protection against Influenza Virus Infection. Phytomedicine 2017, 34, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, N.A.; El-Hawary, S.S.; Mohammed, M.M.D.; Farid, M.A.; Abdel-Wahed, N.A.M.; Ali, M.A.; El-Abd, E.A.W. Chemical Composition, Antiviral against Avian Influenza (H5N1) Virus and Antimicrobial Activities of the Essential Oils of the Leaves and Fruits of Fortunella margarita, Lour. Swingle, Growing in Egypt. J. Appl. Pharm. Sci. 2015, 5, 6–12. [Google Scholar] [CrossRef]
- Garozzo, A.; Timpanaro, R.; Bisignano, B.; Furneri, P.M.; Bisignano, G.; Castro, A. In Vitro Antiviral Activity of Melaleuca alternifolia Essential Oil. Lett. Appl. Microbiol. 2009, 49, 806–808. [Google Scholar] [CrossRef]
- Shabby, A.S.; El-Gengaihi, S.; Khattab, M. Oil of Melissa officinalis L., as Affected by Storage and Herb Drying. J. Essent. Oil Res. 1995, 7, 667–669. [Google Scholar] [CrossRef]
- Pourghanbari, G.; Nili, H.; Moattari, A.; Mohammadi, A.; Iraji, A. Antiviral Activity of the Oseltamivir and Melissa officinalis L. Essential Oil against Avian Influenza A Virus (H9N2). VirusDisease 2016, 27, 170–178. [Google Scholar] [CrossRef]
- Choi, H.J. Chemical Constituents of Essential Oils Possessing Anti-Influenza A/WS/33 Virus Activity. Osong Public Health Res. Perspect. 2018, 9, 348. [Google Scholar] [CrossRef]
- Swamy, M.K.; Sinniah, U.R. A Comprehensive Review on the Phytochemical Constituents and Pharmacological Activities of Pogostemon cablin Benth.: An Aromatic Medicinal Plant of Industrial Importance. Molecules 2015, 20, 8521–8547. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Peng, S.Z.; Chen, H.M.; Zhang, F.X.; Xu, P.P.; Xie, J.H.; He, J.J.; Chen, J.N.; Lai, X.P.; Su, Z.R. Oral Administration of Patchouli Alcohol Isolated from Pogostemonis herba Augments Protection against Influenza Viral Infection in Mice. Int. Immunopharmacol. 2012, 12, 294–301. [Google Scholar] [CrossRef] [PubMed]
- De, J.; Lu, Y.; Ling, L.; Peng, N.; Zhong, Y. Essential Oil Composition and Bioactivities of Waldheimia glabra (Asteraceae) from Qinghai-Tibet Plateau. Molecules 2017, 22, 460. [Google Scholar] [CrossRef]
- Scherließ, R.; Ajmera, A.; Dennis, M.; Carroll, M.W.; Altrichter, J.; Silman, N.J.; Scholz, M.; Kemter, K.; Marriott, A.C. Induction of Protective Immunity against H1N1 Influenza A(H1N1)Pdm09 with Spray-Dried and Electron-Beam Sterilised Vaccines in Non-Human Primates. Vaccine 2014, 32, 2231–2240. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.; Torri, A.; Harte, W.E.; Danetz, S.; Cianci, C.; Tiley, L.; Day, S.; Mullaney, D.; Yu, K.L.; Ouellet, C.; et al. Molecular Mechanism Underlying the Action of a Novel Fusion Inhibitor of Influenza A Virus. J. Virol. 1997, 71, 4062–4070. [Google Scholar] [CrossRef] [PubMed]
- Yoshimoto, J.; Kakui, M.; Iwasaki, H.; Fujiwara, T.; Sugimoto, H.; Hattori, N. Identification of a Novel HA Conformational Change Inhibitor of Human Influenza Virus. Arch. Virol. 1999, 144, 865–878. [Google Scholar] [CrossRef]
- Wang, J.; Wu, Y.; Ma, C.; Fiorin, G.; Wang, J.; Pinto, L.H.; Lamb, R.A.; Klein, M.L.; DeGrado, W.F. Structure and Inhibition of the Drug-Resistant S31N Mutant of the M2 Ion Channel of Influenza A Virus. Proc. Natl. Acad. Sci. USA 2013, 110, 1315–1320. [Google Scholar] [CrossRef]
- Bahgat, M.M.; Bazejewska, P.; Schughart, K. Inhibition of Lung Serine Proteases in Mice: A Potentially New Approach to Control Influenza Infection. Virol. J. 2011, 8, 27. [Google Scholar] [CrossRef]
- Loregian, A.; Mercorelli, B.; Nannetti, G.; Compagnin, C.; Palù, G. Antiviral Strategies against Influenza Virus: Towards New Therapeutic Approaches. Cell. Mol. Life Sci. 2014, 71, 3659–3683. [Google Scholar] [CrossRef]
- Planz, O. Development of Cellular Signaling Pathway Inhibitors as New Antivirals against Influenza. Antivir. Res. 2013, 98, 457–468. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhang, D.; Zhu, X.; He, Z.; Liu, S.; Li, M.; Pang, J.; Lin, Y. Studies on Synthesis and Structure-Activity Relationship (SAR) of Derivatives of a New Natural Product from Marine Fungi as Inhibitors of Influenza Virus Neuraminidase. Mar. Drugs 2011, 9, 1887–1901. [Google Scholar] [CrossRef]
- Feldman, T.; Kabaleeswaran, V.; Jang, S.B.; Antczak, C.; Djaballah, H.; Wu, H.; Jiang, X. A Class of Allosteric Caspase Inhibitors Identified by High-Throughput Screening. Mol. Cell 2012, 47, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Muniruzzaman, S.; Pan, Y.T.; Zeng, Y.; Atkins, B.; Izumori, K.; Elbein, A.D. Inhibition of Glycoprotein Processing by L-Fructose and L-Xylulose. Glycobiology 1996, 6, 795–803. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Oguin, T.H.; Sharma, S.; Stuart, A.D.; Duan, S.; Scott, S.A.; Jones, C.K.; Daniels, J.S.; Lindsley, C.W.; Thomas, P.G.; Brown, H.A. Phospholipase D Facilitates Efficient Entry of Influenza Virus, Allowing Escape from Innate Immune Inhibition. J. Biol. Chem. 2014, 289, 25405–25417. [Google Scholar] [CrossRef]
- Hsieh, C.F.; Lo, C.W.; Liu, C.H.; Lin, S.; Yen, H.R.; Lin, T.Y.; Horng, J.T. Mechanism by Which Ma-Xing-Shi-Gan-Tang Inhibits the Entry of Influenza Virus. J. Ethnopharmacol. 2012, 143, 57–67. [Google Scholar] [CrossRef]
- Hsieh, C.F.; Yen, H.R.; Liu, C.H.; Lin, S.; Horng, J.T. Ching-Fang-Pai-Tu-San Inhibits the Release of Influenza Virus. J. Ethnopharmacol. 2012, 144, 533–544. [Google Scholar] [CrossRef]
- Dai, J.; Wang, G.; Li, W.; Zhang, L.; Yang, J.; Zhao, X.; Chen, X.; Xu, Y.; Li, K. High-Throughput Screening for Anti-Influenza A Virus Drugs and Study of the Mechanism of Procyanidin on Influenza A Virus-Induced Autophagy. J. Biomol. Screen. 2012, 17, 605–617. [Google Scholar] [CrossRef]
- Ma, L.; Yao, L. Antiviral Effects of Plant-Derived Essential Oils and Their Components: An Updated Review. Molecules 2020, 25, 2627. [Google Scholar] [CrossRef]
- Li, L.; Xie, Q.; Bian, G.; Zhang, B.; Wang, M.; Wang, Y.; Chen, Z.; Li, Y. Anti-H1N1 Viral Activity of Three Main Active Ingredients from Zedoary Oil. Fitoterapia 2020, 142, 104489. [Google Scholar] [CrossRef]
- Xia, Z.; Xu, G.; Yang, X.; Peng, N.; Zuo, Q.; Zhu, S.; Hao, H.; Liu, S.; Zhu, Y. Inducible TAP1 Negatively Regulates the Antiviral Innate Immune Response by Targeting the TAK1 Complex. J. Immunol. 2017, 198, 3690–3704. [Google Scholar] [CrossRef] [PubMed]
- Galisteo Pretel, A.; Pérez Del Pulgar, H.; Guerrero de León, E.; López-Pérez, J.L.; Olmeda, A.S.; Gonzalez-Coloma, A.; Barrero, A.F.; Quílez Del Moral, J.F. Germacrone Derivatives as New Insecticidal and Acaricidal Compounds: A Structure-Activity Relationship. Molecules 2019, 24, 2898. [Google Scholar] [CrossRef] [PubMed]
- Ti, H.; Mai, Z.; Wang, Z.; Zhang, W.; Xiao, M.; Yang, Z.; Shaw, P. Bisabolane-Type Sesquiterpenoids from Curcuma longa L. Exert Anti-Influenza and Anti-Inflammatory Activities through NF-ΚB/MAPK and RIG-1/STAT1/2 Signaling Pathways. Food Funct. 2021, 12, 6697–6711. [Google Scholar] [CrossRef] [PubMed]
- Toden, S.; Theiss, A.L.; Wang, X.; Goel, A. Essential Turmeric Oils Enhance Anti-Inflammatory Efficacy of Curcumin in Dextran Sulfate Sodium-Induced Colitis. Sci. Rep. 2017, 7, 816. [Google Scholar] [CrossRef] [PubMed]
- da Silva, A.P.; Martini, M.v.; de Oliveira, C.M.A.; Cunha, S.; de Carvalho, J.E.; Ruiz, A.L.T.G.; da Silva, C.C. Antitumor Activity of (-)-α-Bisabolol-Based Thiosemicarbazones against Human Tumor Cell Lines. Eur. J. Med. Chem. 2010, 45, 2987–2993. [Google Scholar] [CrossRef] [PubMed]
- Ait-Ouazzou, A.; Lorán, S.; Bakkali, M.; Laglaoui, A.; Rota, C.; Herrera, A.; Pagán, R.; Conchello, P. Chemical Composition and Antimicrobial Activity of Essential Oils of Thymus algeriensis, Eucalyptus globulus and Rosmarinus officinalis from Morocco. J. Sci. Food Agric. 2011, 91, 2643–2651. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lai, Y.; Wang, Y.; Liu, N.; Zhang, F.; Xu, P. 1,8-Cineol Protect Against Influenza-Virus-Induced Pneumonia in Mice. Inflammation 2016, 39, 1582–1593. [Google Scholar] [CrossRef]
- Juergens, U.R.; Stöber, M.; Vetter, H. Inhibition of Cytokine Production and Arachidonic Acid Metabolism by Eucalyptol (1.8-Cineole) in Human Blood Monocytes in Vitro. Eur. J. Med. Res. 1998, 3, 508–510. [Google Scholar]
- Mieres-Castro, D.; Ahmar, S.; Shabbir, R.; Mora-Poblete, F. Antiviral Activities of Eucalyptus Essential Oils: Their Effectiveness as Therapeutic Targets against Human Viruses. Pharmaceuticals 2021, 14, 1210. [Google Scholar] [CrossRef]
- Müller, J.; Greiner, J.F.W.; Zeuner, M.; Brotzmann, V.; Schäfermann, J.; Wieters, F.; Widera, D.; Sudhoff, H.; Kaltschmidt, B.; Kaltschmidt, C. 1,8-Cineole Potentiates IRF3-Mediated Antiviral Response in Human Stem Cells and in an Ex Vivo Model of Rhinosinusitis. Clin. Sci. 2016, 130, 1339–1352. [Google Scholar] [CrossRef]
- Villecco, M.B.; Catalán, J.v; Vega, M.I.; Garibotto, F.M.; Enriz, R.D.; Catalán, C.A.N. Synthesis and Antibacterial Activity of Highly Oxygenated 1,8-Cineole Derivatives. Nat. Prod. Commun. 2008, 3, 1934578X0800300301. [Google Scholar] [CrossRef]
- Yarovaya, O.I.; Stro, A.A.; Orshanskaya, Y.R.; Zarubaev, V.V.; Khazanov, V.A.; Salakhutdinov, N.F. (1S,3aR,4R,7aS)-N-(2,2,4,7a-Tetramethyloctahydro-1,4-ethanoindene-3a-yl)-acetamide Application as a Influenza Virus Reproduction Inhibitor. RU2616255C1, 13 April 2017. [Google Scholar]
- Gyrdymova, Y.v.; Sinegubova, E.O.; Muryleva, A.S.; Zarubaev, V.v.; Rubtsova, S.A. Anti-Influenza Activity of Several Caryophyllane Hiosesquiterpenoids. Chem. Nat. Compd. 2019, 55, 1179–1181. [Google Scholar] [CrossRef]
- Scholtissek, C.; Quack, G.; Klenk, H.D.; Webster, R.G. How to Overcome Resistance of Influenza A Viruses against Adamantane Derivatives. Antivir. Res. 1998, 37, 83–95. [Google Scholar] [CrossRef]
- Volobueva, A.S.; Yarovaya, O.I.; Kireeva, M.v.; Borisevich, S.S.; Kovaleva, K.S.; Mainagashev, I.Y.; Gatilov, Y.v.; Ilyina, M.G.; Zarubaev, V.v.; Salakhutdinov, N.F. Discovery of New Ginsenol-Like Compounds with High Antiviral Activity. Molecules 2021, 26, 6794. [Google Scholar] [CrossRef] [PubMed]
- Hayden, F.G.; Pavia, A.T. Antiviral Management of Seasonal and Pandemic Influenza. J. Infect. Dis. 2006, 194, S119–S126. [Google Scholar] [CrossRef]
- Plotch, S.J.; O’Hara, B.; Morin, J.; Palant, O.; LaRocque, J.; Bloom, J.D.; Lang, S.A.; DiGrandi, M.J.; Bradley, M.; Nilakantan, R.; et al. Inhibition of Influenza A Virus Replication by Compounds Interfering with the Fusogenic Function of the Viral Hemagglutinin. J. Virol. 1999, 73, 140–151. [Google Scholar] [CrossRef]
- Rana, V.S.; Juyal, J.P.; Blazquez, M.A. Chemical Constituents of Essential Oil of Pelargonium graveolens Leaves. Int. J. Aromather. 2002, 4, 216–218. [Google Scholar] [CrossRef]
- Paranagama, P.A.; Wimalasena, S.; Jayatilake, G.S.; Jayawardena, A.L.; Senanayake, U.M.; Mubarak, A.M. A Comparison of Essential Oil Constituents of Bark, Leaf, Root and Fruit of Cinnamon (Cinnamomum zeylanicum Blum) Grown in Sri Lanka. J. Natl. Sci. Found. 2001, 29, 147–153. [Google Scholar] [CrossRef]
- Vimalanathan, S.; Hudson, J. Anti-Influenza Virus Activity of Essential Oils and Vapors. Am. J. Essent. Oils Nat. Prod. 2014, 2, 47–53. [Google Scholar]
- Andrade-Ochoa, S.; Nevárez-Moorillón, G.V.; Sánchez-Torres, L.E.; Villanueva-García, M.; Sánchez-Ramírez, B.E.; Rodríguez-Valdez, L.M.; Rivera-Chavira, B.E. Quantitative Structure-Activity Relationship of Molecules Constituent of Different Essential Oils with Antimycobacterial Activity against Mycobacterium tuberculosis and Mycobacterium bovis. BMC Complement. Altern. Med. 2015, 15, 332. [Google Scholar] [CrossRef]
- Gülçin, I. Antioxidant Activity of Eugenol: A Structure-Activity Relationship Study. J. Med. Food 2011, 14, 975–985. [Google Scholar] [CrossRef] [PubMed]
- Zarubaev, V.v.; Pushkina, E.A.; Borisevich, S.S.; Galochkina, A.v.; Garshinina, A.v.; Shtro, A.A.; Egorova, A.A.; Sokolova, A.S.; Khursan, S.L.; Yarovaya, O.I.; et al. Selection of Influenza Virus Resistant to the Novel Camphor-Based Antiviral Camphecene Results in Loss of Pathogenicity. Virology 2018, 524, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Zarubaev, V.v.; Garshinina, A.v.; Volobueva, A.S.; Slita, A.v.; Yarovaya, O.I.; Bykov, V.v.; Leonov, K.A.; Motov, V.S.; Khazanov, V.A.; Salakhutdinov, N.F. Optimization of Application Schedule of Camphecene, a Novel Anti-Influenza Compound, Based on Its Pharmacokinetic Characteristics. Fundam Clin. Pharmacol. 2022, 36, 518–525. [Google Scholar] [CrossRef] [PubMed]
- Artyushin, O.I.; Sharova, E.v.; Vinogradova, N.M.; Genkina, G.K.; Moiseeva, A.A.; Klemenkova, Z.S.; Orshanskaya, I.R.; Shtro, A.A.; Kadyrova, R.A.; Zarubaev, V.v.; et al. Synthesis of Camphecene Derivatives Using Click Chemistry Methodology and Study of Their Antiviral Activity. Bioorg. Med. Chem. Lett. 2017, 27, 2181–2184. [Google Scholar] [CrossRef]
- Wang, X.; Kulkarni, D.; Dozier, M.; Hartnup, K.; Paget, J.; Campbell, H.; Nair, H. Influenza Vaccination Strategies for 2020–21 in the Context of COVID-19. J. Glob. Health 2020, 10, 021102. [Google Scholar] [CrossRef]
- Berlanda Scorza, F.; Tsvetnitsky, V.; Donnelly, J.J. Universal Influenza Vaccines: Shifting to Better Vaccines. Vaccine 2016, 34, 2926. [Google Scholar] [CrossRef]
- Linke, L.M.; Wilusz, J.; Pabilonia, K.L.; Fruehauf, J.; Magnuson, R.; Olea-Popelka, F.; Triantis, J.; Landolt, G.; Salman, M. Inhibiting Avian Influenza Virus Shedding Using a Novel RNAi Antiviral Vector Technology: Proof of Concept in an Avian Cell Model. AMB Express 2016, 6, 16. [Google Scholar] [CrossRef]
- Cimino, C.; Maurel, O.M.; Musumeci, T.; Bonaccorso, A.; Drago, F.; Souto, E.M.B.; Pignatello, R.; Carbone, C. Essential Oils: Pharmaceutical Applications and Encapsulation Strategies into Lipid-Based Delivery Systems. Pharmaceutics 2021, 13, 327. [Google Scholar] [CrossRef]
- Sievees, A.F. MPlKB Methods of Extracting Volatile Oils from Plant Material and the Production of Such Oils in the United States; US Department of Agriculture: Washington, DC, USA, 1928. [Google Scholar]
- Sachan, A.K.; Sachan, N.K.; Kumar, S.; Sachan, A.; Gangwar, S.S. Evaluation and Standardization of Essential Oils for Development of Alternative Dosage Forms. Eur. J. Sci. Res. 2010, 46, 194–203. [Google Scholar]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano Based Drug Delivery Systems: Recent Developments and Future Prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef]
- Campolo, O.; Cherif, A.; Ricupero, M.; Siscaro, G.; Grissa-Lebdi, K.; Russo, A.; Cucci, L.M.; di Pietro, P.; Satriano, C.; Desneux, N.; et al. Citrus Peel Essential Oil Nanoformulations to Control the Tomato Borer, Tuta absoluta: Chemical Properties and Biological Activity. Sci. Rep. 2017, 7, 13036. [Google Scholar] [CrossRef] [PubMed]
- Bilia, A.R.; Guccione, C.; Isacchi, B.; Righeschi, C.; Firenzuoli, F.; Bergonzi, M.C. Essential Oils Loaded in Nanosystems: A Developing Strategy for a Successful Therapeutic Approach. Evid.-Based Complement. Altern. Med. 2014, 2014, 651593. [Google Scholar] [CrossRef] [PubMed]


| Plant Family | Species (Part Used) | Major Essential Oil Constituents | Extraction Method | Reference |
|---|---|---|---|---|
| Apiaceae (Umbelliferae or Carrot family) | Angelica archangelica (Flowers) | α-Pinene, β-phellandrene, limonene, and ρ-cymene | Hydro-distillation (HD) for 2 h | [37] |
| Daucus carrota (Seeds) | Sabinene, carotol, (Z)-β-farnesene, elemecin, and β-bisabolene | HD for 2 h | [38] | |
| Asteraceae (Sunflower family) | Matricaria chamomilla (Flowers) | α- and β-Farnesene, α-bisabolol, chamazulene, germacrene D, and spiroether | HD for 4 h | [39] |
| Achillea millefolium (Leaves, flowers, and seeds) | Eucalyptol, camphor, α-terpineol, β-pinene, sabinene, 1,8-cineole, artemisia ketone, linalool, α- and β-thujone, camphor, borneol, bornyl acetate, (E)-β-caryophyllene, germacrene D, caryophyllene oxide, β-bisabolol, δ-cadinol, and chamazulene | HD for 10 h (industrial) HD for 3 h (small scale) | [40,41] | |
| Brassicaceae (Mustard family) | Aethionema sancakense (Aerial parts) | α-Humulene, camphene, and heptanal | HD for 3 h | [42,43] |
| Brassica oleraceae (Aerial parts) | Dimethyl disulfide, dimethyl trisulphide, allyl isothiocyanate, dimethyl tetrasulfide, and 1-hexanol | HD for 12 h (industrial) | ||
| Burseraceae (Torchwood family) | Boswellia sacra (Oleogum resin) | (E)-β-Ocimene, 1-β-pinene, 2-β-pinene, camphene, sabinene, α-thujene, limonene, myrcene, α-pinene, 2-carene, (Z)-β-ocimene, δ- and γ-cadinene and caryophyllene oxide, β-elemene, and α-copaene. | Steam distillation (SD) in water/ethylene glycol (1:9) for 2 h | [44] |
| Commiphora myrrha (Oleogum resin) | Furanoeudesma-1,3-diene, lindestrene, curzerene, β-elemene, germacrene B, and germacrone | Supercritical fluid extraction (SFE) with CO2 and vacuum extraction with dichloromethane by Ultrasonic bath | [45,46] | |
| Cupressaceae (Conifer or Cypress family) | Cupressus sempervirens (Aerial part) | α-Pinene, δ-3-carene, limonene, and α-terpinolene | HD for 3 h | [47] |
| Juniperus communis (Berries) | α- and β-Pinene, myrcene, sabinene, and limonene | Commercial SD | [48] | |
| Juniperus virginiana (Heartwood) | (-)-α- and (+)-β-Cedrene, (-)-thujopsene, and (+)-cedrol | Commercial SD | [49] | |
| Lamiaceae (Labiatae or Mint family) | Salvia leucophylla (Leaves and flowers) | 1,8-Cineole, camphor, camphene, and α- and β-pinene | HD for 2 h | [50] |
| Ocimum basilicum (Aerial part) | Methyl cinnamate, linalool, β-elemene, and camphor | HD for 3 h | [51] | |
| Mentha piperita (Aerial part) | Menthol and menthone, (±)-menthyl acetate, 1,8-cineole, limonene, β-pinene, and β-caryophyllene | HD for 3 h | [52] | |
| Mentha spicata (Aerial part) | Carvone, limonene, 1,8-cineole, β-pinene, cis-dihydrocarvone, and dihydrocarveol | HD for 3 h | [53] | |
| Rosmarinus officinalis (Aerial part) | ρ-Cymene, linalool, γ-terpinene, thymol, α- and β-pinene, and eucalyptol | HD for 3 h | [54] | |
| Thymus vulgaris (Aerial part) | Thymol, ρ-cymene, γ-terpinene, and caryophyllene oxide | HD for 3 h | [55] | |
| Lauraceae (Laurel family) | Cinnamomum camphora (Stem bark, leaves, and fruits) | D-Camphor, 1,8-cineole, α-terpineol, linalool, safrole, γ-terpinen, isoterpinolene, 1,3,8-ρ-menthatriene, terpinen-4-ol, α-terpineol, eugenol, β-cadinene, and α-cubebene | HD for 6 h | [56] |
| Cinnamomum zeylanicum (Leaves, stem bark, fruits, and, roots) | (E)-Cinnamaldehyde, linalool, β-caryophyllene, eucalyptol, eugenol, carvacrol, ρ-cymene, α-humulene, δ-cadinene, and α-pinene | HD for 3 h | [57] | |
| Laurus nobilis (Leaves) | 1,8-Cineole, sabinene, linalool, α-terpinyl acetate, α-pinene, α-terpineol, methyl-eugenol, neoiso-isopulegol, eugenol, β-pinene, and γ-terpinene | HD for 3 h | [58] | |
| Myrtaceae (Myrtle family) | Eucalyptus species (Leaves) | 1,8-Cineol, α-pinene, spathulenol, trans-pinocarveol, ρ-cymene, globulol, cryptone, β-phellandrene, viridiflorol, borneol, limonene, and isospathulenol | HD for 4 h HD for 3 h | [59,60] |
| Myrtus communis (Berries) | Geranyl acetate, 1,8-cineole, α-terpinyl acetate, methyleugenol, linalool, α-terpineol, β-caryophyllene, α-humulene, trans-caryophyllene oxide, and humulene epoxide II | HD for 4 h | [61] | |
| Melaleuca alternifolia (Leaves and terminal branch) | Terpinen-4-ol, γ-terpinene, 1,8-cineole, α-terpinene, α-terpineol, ρ-cymene, and α-pinene | Commercial SD | [62] | |
| Pinaceae (Pine family) | Picea mariana (Twigs/bark and needles) | α- and β-Pinene, β-phellandrene, 3-carene, limonene, α-terpineol, trans-pinocarveol, terpinen-4-ol, verbenone, borneol, and pinocarvone | SD and HD for 6 h each | [63] |
| Pinus eldarica and P. peuce (Twigs/bark, needles, and pollens) | D-Germacrene, α- and β-pinene, trans-(E)-caryophyllene, γ-terpinene, limonene, caryophyllene oxide, drimenol, β-myrcene, camphene, bornyl acetate, and δ-cardinene | HD for 4 h each | [64,65] | |
| Poaceae (Grass family) | Cymbopogon citratus (Leaves) | Geranial, neral, β-myrcene, geranyl acetate, isopulegol, and citral (comprising cis-isomer geranial and trans-isomer neral) | SD for 3 h | [66] |
| Cymbopogon martini (Leaves) | Linalool, α-terpineol, geranyl isobutyrate, geraniol, myrcene, β-caryophyllene, geranyl acetate, (E,Z) farnesol, and geranyl hexanoate | SD for 3 h | [67] | |
| Cymbopogon nardus (Leaves) | 6-octenal, citronellal, geranial, geraniol, citronellol, and neral | HD for 3 h | [68] | |
| Rutaceae (Citrus family) | Citrus limon (Leaves and fruit peels) | β-Pinene, limonene, linalool, α-terpineol, linalyl acetate, acetate geranyl, nerolidol, acetate neryl, and farnesol | SD for 3 h | [69] |
| Citrus paradisi (Leaves and fruit peels) | D-Limonene, β-myrcene, γ-terpinene, β-phellandrene, furanoid, caryophyllene, and cis-limonene oxide | HD for 4 h | [70] | |
| Citrus sinensis (Leaves, and fruit peels) | D-Limonene, β-pinene, 3-carene, β-phellandrene, and linalool | Cold-press molecular distillation | [71] | |
| Zingiberaceae (Ginger family) | Curcuma longa (Rhizomes) | Ar-turmerone, α-turmerone, curlone, and ar-curcumene | Commercial SD | [72] |
| Elettaria cardamomum (Leaves and rhizomes) | β-Caryophyllene, γ-terpinene, α- and β-pinene, myrcene, p-cymene, limonene, 1,8-cineole, linalool, 4-terpineol, and α-terpinyl acetate | HD for 3 h | [73] | |
| Zingiber officinale (Rhizomes) | Citral (geranial and neral), α-zingiberene, camphene, 1,8-cineol, α-farnesene, β-sesquiphellandrene, α-terpinene, α-terpineol, 4-terpineol, gingerols, zingerone, paradol, gingerdiones, gingerdiols, shogaols, and zingerines | HD for 5–6 h | [74,75] |
| Aromatic Plant (Part) | Major Essential Oil Component | Anti-influenza Activity | Reference |
|---|---|---|---|
| Cinnamomum verum (Cortex) | trans-Cinnamaldehyde | Inhibited the growth of influenza A/PR/8 virus in vitro at 200 µM. Inhalation (50 mg/cage/day) and nasal inoculation (250 microg/mouse/day) of EOs significantly increased survival rates over 8 days to 100% and 70%, respectively, in vivo. | [118] |
| Citrus reshni (Unripe fruit peels) | Limonene (82.4%) and linalool (7.2%) | In vitro antiviral activity (IC50 = 2.5 µg/mL) against Avian influenza virus A (H5N1) strain. | [119,120] |
| Curcuma aeruginosa (Rhizomes) | Germacrone | In vitro antiviral activity (EC50 = 6.0 µM) against influenza A/H1N1/H3N2 virus strains and the influenza B virus in a dose-dependent manner. Germacrone demonstrated effective protection of mice from lethal infection and reduced the virus titers in the lung at 100 mg/kg in mice. Its co-administration with oseltamivir showed an additive effect on the inhibition of influenza virus infection, both in vitro and in vivo. | [121] |
| Eucalyptus globulus (Stems and leaves) | 1,8-Cineol (84.2%) and o-cymene (8.0%) | Intranasal co-administration of 1,8-cineole with the influenza vaccine provides cross-protection against influenza virus infection in vivo. | [122,123] |
| Fortunella margarita (Fruits) | α-Terpineol (55.5%), carvone (5.7%), t-carveol (5.5%), muurolene (5.5%), and citronellal (5.01%) | In vitro antiviral activity (IC50 = 6.8 µM) against Avian influenza A (H5N1) virus. | [124] |
| Melaleuca alternifolia (Aerial part) | terpinen-4-ol (36.7%) and γ-terpinene (22.2%), and α-terpinene (10.1%) | Antiviral activity (ID50 = 0.0006%v/v) against the influenza A⁄ PR⁄ 8 virus subtype H1N1 with activity attributed to terpinen-4-ol. | [125] |
| Melissa officinalis (Leaves) | Geranial (38.3%), neral (26.1%), geraniol (8.1%), caryophyllene oxide (5.5%), citronellal (4.5%), neryl acetate (3.9%), and geranyl acetate (3.3%) | Inhibited influenza virus replication through different replication cycle steps. Co-administration of EOs with oseltamivir showed a synergistic activity with the EO, especially when oseltamivir concentration was under 0.005 mg/ml. | [126,127] |
| Pimpinella anisum (Fruits) | trans-anethole (82.8%), estragole (8.2%), and linalool (2.7%) | In vitro activity against influenza A/WS/33 virus (IC50 < 100 µg/mL) infected MDCK cells, inhibited the formation of a visible cytopathic effect. | [128] |
| Pogostemon cablin (Leaves) | patchoulol, caryophyllene, pogostol, α-, β-, γ- and δ-patchoulene, seychellene, cycloseychellene, α- and β-bulnesene, α- and β-guaiene, and norpatchoulenol | In vitro activity (IC50 of 4.0 µM) against influenza virus A (H2N2). Significant increase in the survival rate and survival time within the 20–80 mg/kg doses for the 7-day post-infection period in mice. | [129,130] |
| Salvia sclarea (Flowers) | Linalyl acetate (61.2%), linalool (22.1%), α-Terpineol (4.2%), and geranyl acetate (2.4%) | Inhibitory activity (>52%, IC50 < 100 µg/mL) against influenza A/WS/33 virus-infected MDCK cells, inhibited the formation of a visible cytopathic effect. | [128] |
| Thymus mastichina (Leaves) | 1,8-Cineole (64.6%), linalool (15.3%), β-pinene (5.8%), and α-pinene (4.2%) | ||
| Waldheimia glabra (Whole plant) | α-Bisabolol (20.2%), valeranone (11.8%), chamazulene (9.9%), spathulenol (8.2%), β-caryophyllene (6.1%), and caryophyllene oxide (5.2%) | Cytopathic effect against influenza H3N3 virus (IC50 = 88.8 µg/mL) and cytotoxicity against MDCK cells (CC50 = 252 µg/mL). | [131] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oriola, A.O.; Oyedeji, A.O. Essential Oils and Their Compounds as Potential Anti-Influenza Agents. Molecules 2022, 27, 7797. https://doi.org/10.3390/molecules27227797
Oriola AO, Oyedeji AO. Essential Oils and Their Compounds as Potential Anti-Influenza Agents. Molecules. 2022; 27(22):7797. https://doi.org/10.3390/molecules27227797
Chicago/Turabian StyleOriola, Ayodeji Oluwabunmi, and Adebola Omowunmi Oyedeji. 2022. "Essential Oils and Their Compounds as Potential Anti-Influenza Agents" Molecules 27, no. 22: 7797. https://doi.org/10.3390/molecules27227797
APA StyleOriola, A. O., & Oyedeji, A. O. (2022). Essential Oils and Their Compounds as Potential Anti-Influenza Agents. Molecules, 27(22), 7797. https://doi.org/10.3390/molecules27227797






