Bioassay-Guided Isolation of Iridoid Glucosides from Stenaria nigricans, Their Biting Deterrence against Aedes aegypti (Diptera: Culicidae), and Repellency Assessment against Imported Fire Ants (Hymenoptera: Formicidae)
Abstract
1. Introduction
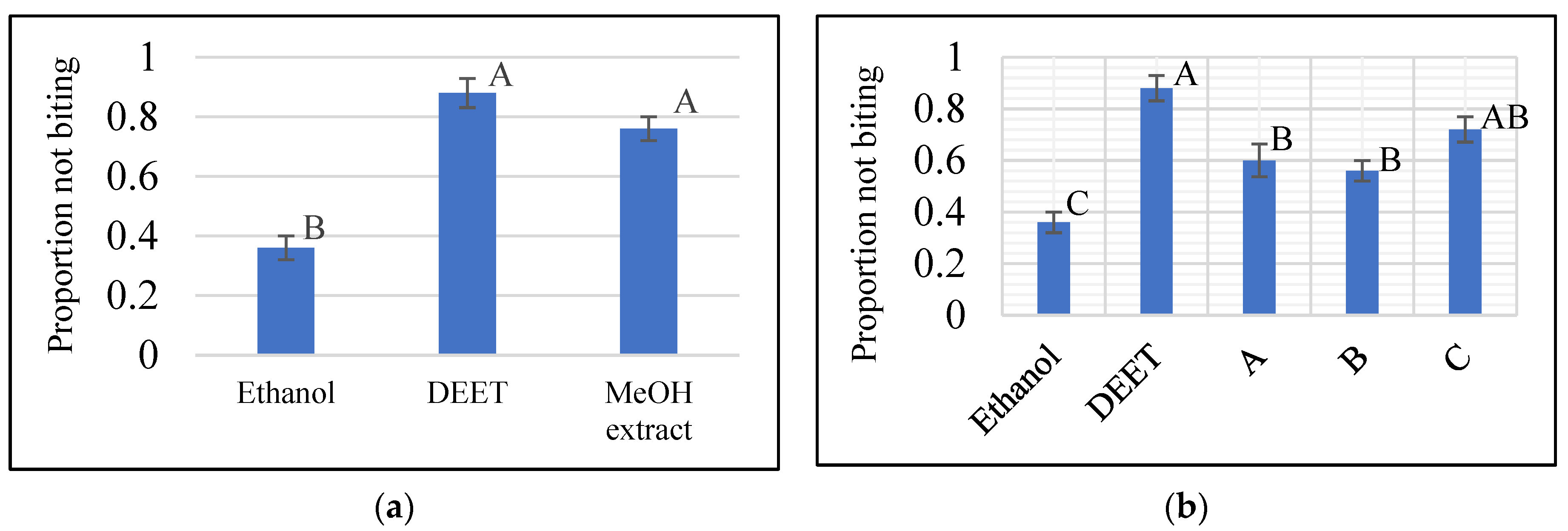
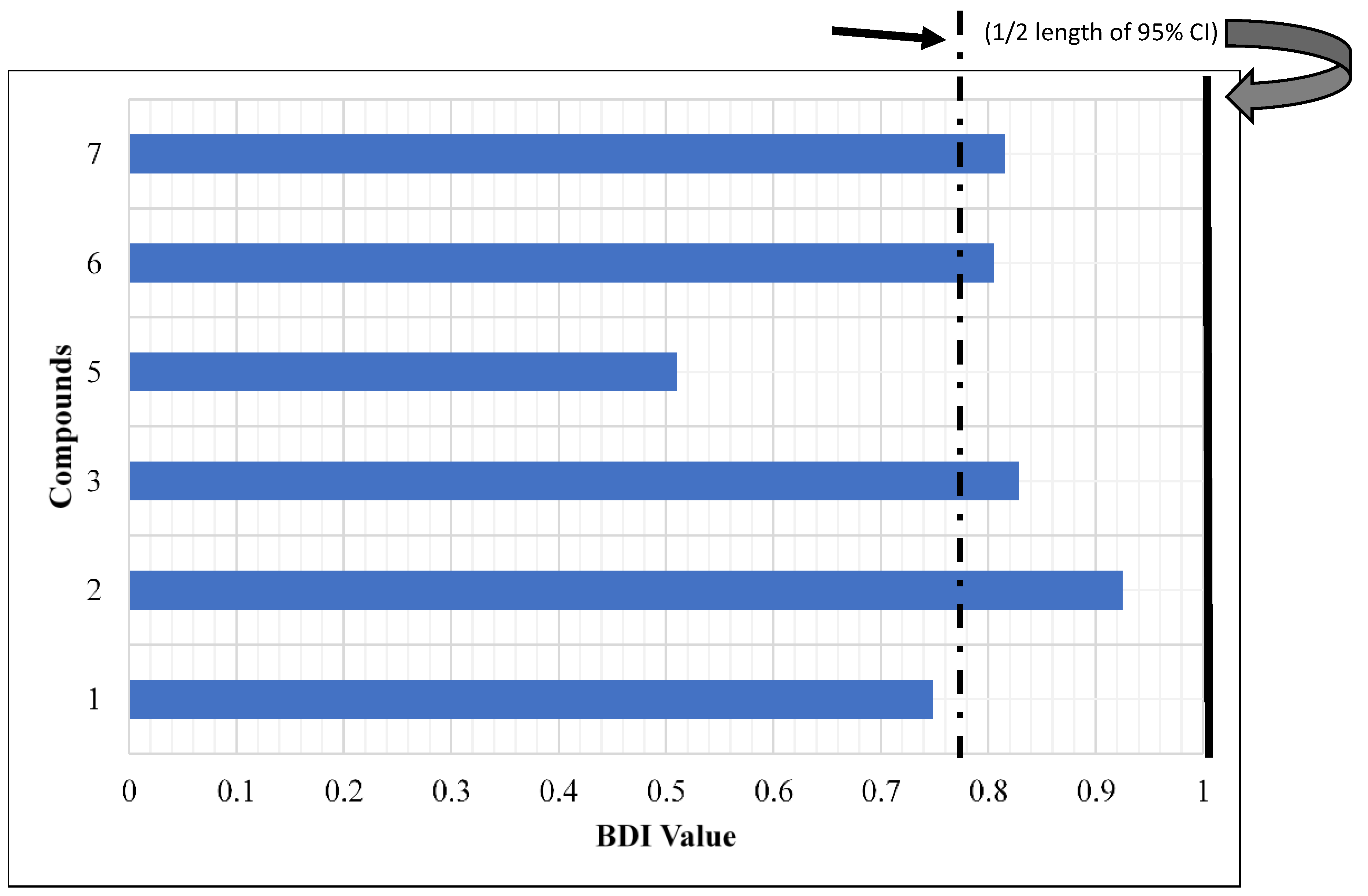
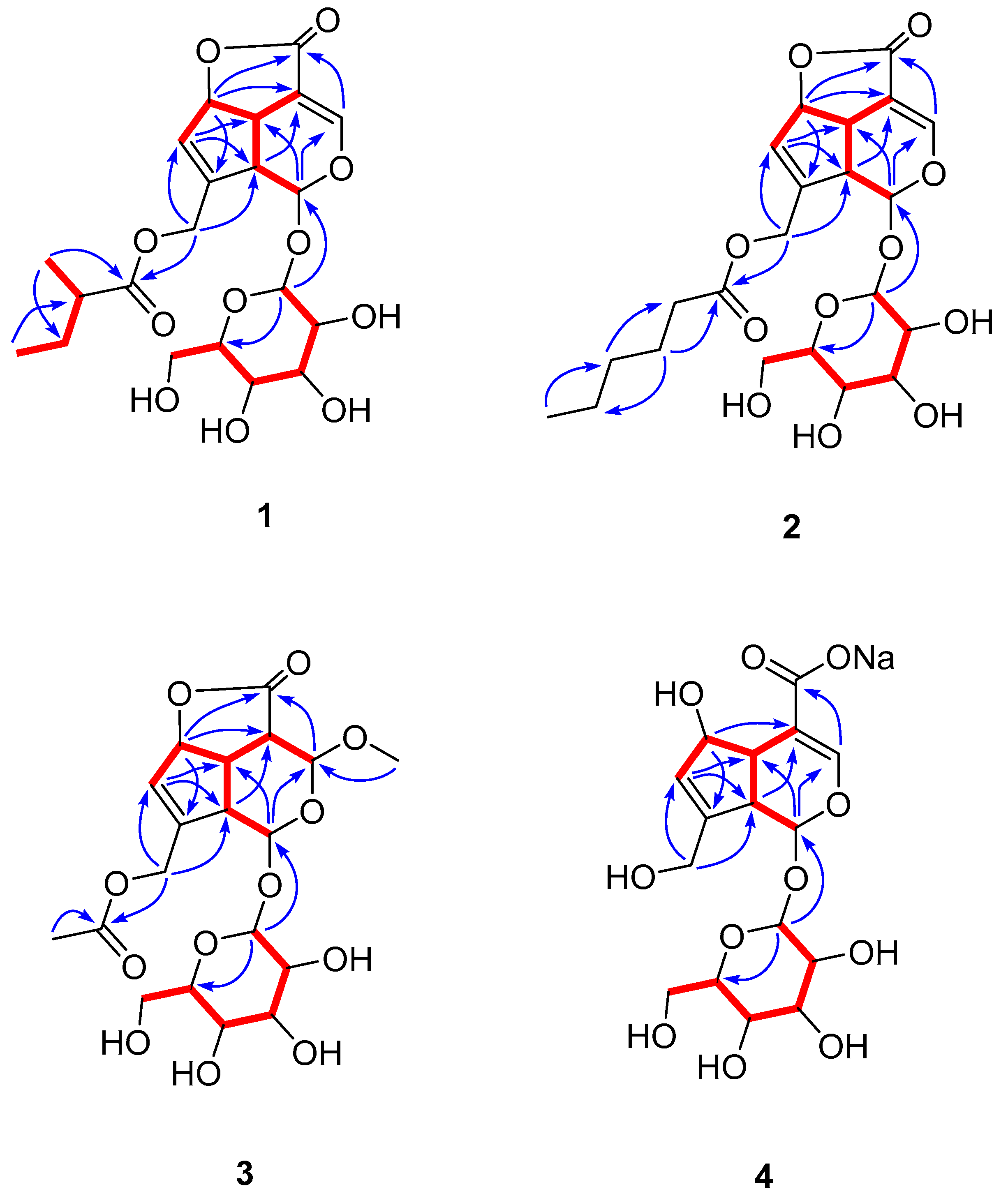
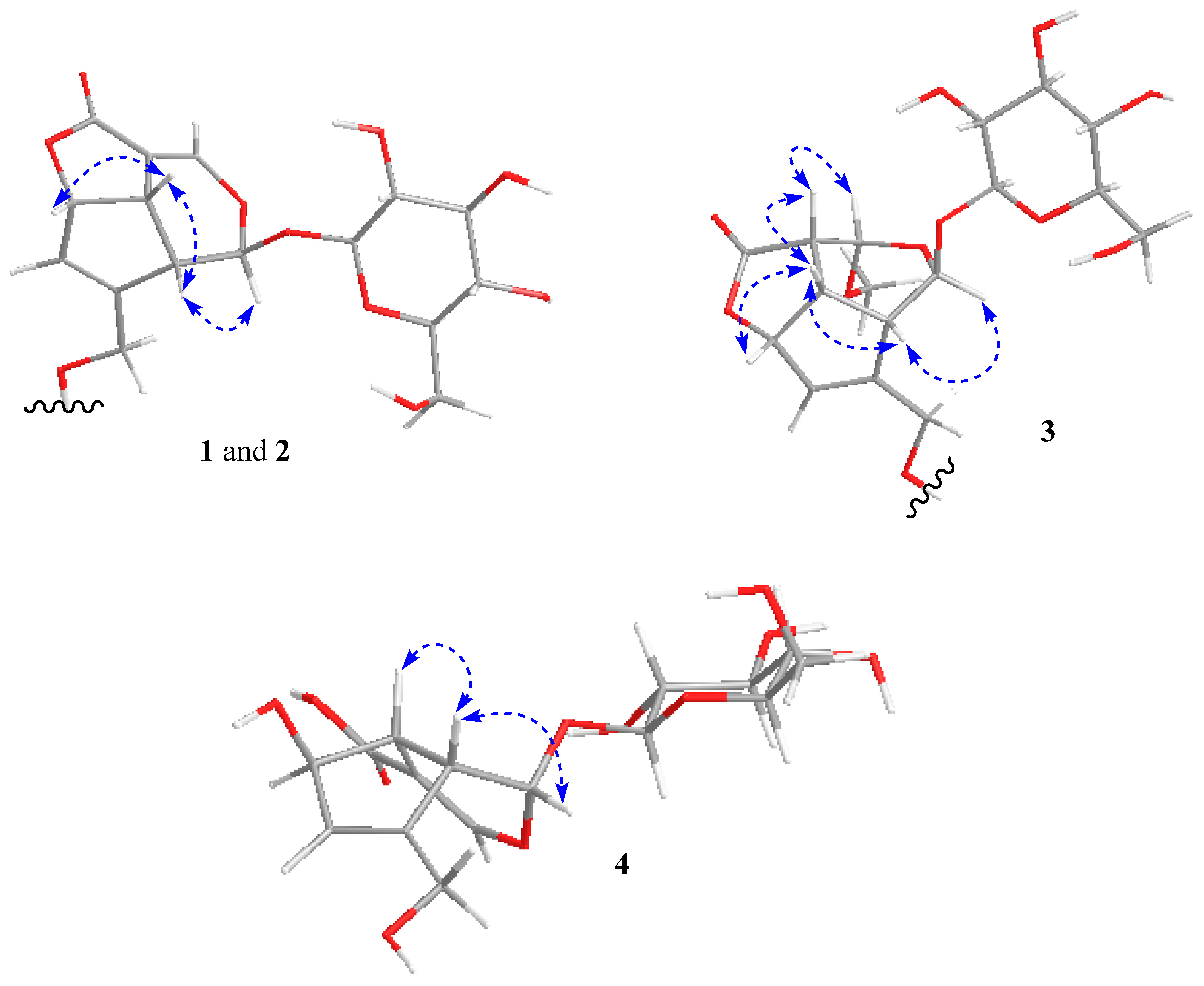
2. Results and Discussion
3. Material and Methods
3.1. General Procedures
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Spectral Data
3.5. In Vitro K & D Biting Deterrent Bioassay
3.6. In Vitro Digging Bioassay for Fire Ants
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Ali, A.; Abbas, A.; Debboun, M. Zika virus: Epidemiology, vector and sexual transmission neurological disorders and vector management—A review. Int. J. Curr. Res. 2017, 10, 58721–58737. [Google Scholar]
- Guo, C.; Zhou, Z.; Wen, Z.; Liu, Y.; Zeng, C.; Xiao, D.; Ou, M.; Han, Y.; Huang, S.; Liu, D.; et al. Global epidemiology of dengue outbreaks in 1990–2015: A systematic review and meta-analysis. Front. Cell. Infect. Microbiol. 2017, 7, 317. [Google Scholar] [CrossRef] [PubMed]
- Sanders, E.J.; Borus, P.; Ademba, G.; Kuria, G.; Tukei, P.M.; LeDuc, J.W. Sentinel surveillance for yellow fever in Kenya, 1993 to 1995. Emerg. Infect. Dis. 1996, 2, 236–238. [Google Scholar] [CrossRef] [PubMed]
- Godsey Jr, M.S.; Blackmore, M.S.; Panella, N.A.; Burkhalter, K.; Gottfried, K.; Halsey, L.A.; Rutledge, R.; Langevin, S.A.; Gates, R.; Lamonte, K.M. West Nile virus epizootiology in the southeastern United States, 2001. Vector-Borne Zoonotic Dis. 2005, 5, 82–89. [Google Scholar] [CrossRef]
- Oliver, J.B.; Vander Meer, R.K.; Ochieng, S.A.; Youssef, N.N.; Pantaleoni, E.; Mrema, F.A.; Vail, K.M.; Parkman, J.P.; Valles, S.M.; Haun, W.G. Statewide survey of imported fire ant (Hymenoptera: Formicidae) populations in Tennessee. J. Entomol. Sci. 2009, 44, 149–157. [Google Scholar] [CrossRef]
- Ross, K.G.; Meer, R.K.V.; Fletcher, D.J.; Vargo, E.L. Biochemical phenotypic and genetic studies of two introduced fire ants and their hybrid (Hymenoptera: Formicidae). Evolution 1987, 41, 280–293. [Google Scholar] [CrossRef]
- Leal, W.S. The enigmatic reception of DEET-the gold standard of insect repellents. Curr. Opin. Insect. Sci. 2014, 6, 93–98. [Google Scholar] [CrossRef]
- Chandre, F.; Darrier, F.; Manga, L.; Akogbeto, M.; Faye, O.; Mouchet, J.; Guillet, P. Status of pyrethroid resistance in Anopheles gambiae sensu lato. Bull. World Health Organ. 1999, 77, 230–234. [Google Scholar]
- Benelli, G. Research in mosquito control: Current challenges for a brighter future. Parasitol. Res. 2015, 114, 2801–2805. [Google Scholar] [CrossRef]
- Benelli, G.; Maggi, F.; Pavela, R.; Murugan, K.; Govindarajan, M.; Vaseeharan, B.; Petrelli, R.; Cappellacci, L.; Kumar, S.; Hofer, A. Mosquito control with green nanopesticides: Towards the one health approach? A review of non-target effects. Environ. Sci. Pollut. Res. 2018, 25, 10184–10206. [Google Scholar] [CrossRef]
- Brown, J.S. Chronology of a mosquito control effort after Hurricane Fran. J. Environ. Health 1997, 60, 8–13. [Google Scholar]
- Masi, M.; Cimmino, A.; Tabanca, N.; Becnel, J.J.; Bloomquist, J.R.; Evidente, A. A survey of bacterial, fungal and plant metabolites against Aedes aegypti (Diptera: Culicidae), the vector of yellow and dengue fevers and Zika virus. Open Chem. 2017, 15, 156–166. [Google Scholar] [CrossRef]
- Tabanca, N.; Ali, Z.; Bernier, U.R.; Epsky, N.; Nalbantsoy, A.; Khan, I.A.; Ali, A. Bioassay-guided isolation and identification of Aedes aegypti larvicidal and biting deterrent compounds from Veratrum lobelianum. Open Chem. 2018, 16, 324–332. [Google Scholar] [CrossRef]
- Ali, A.; Ahmed Zaki, M.; Parveen, A.; Ali, Z.; Khan, I.A. Bioassay guided isolation of mosquito biting deterrent compounds from Strumpfia maritima. Pest. Manag. Sci. 2020, 76, 2342–2346. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Cantrell, C.L.; Bernier, U.R.; Duke, S.O.; Schneider, J.C.; Agramonte, N.M.; Khan, I.A. Aedes aegypti (Diptera: Culicidae) biting deterrence: Structure-activity relationship of saturated and unsaturated fatty acids. J. Med. Entomol. 2012, 49, 1370–1378. [Google Scholar] [CrossRef]
- Ali, A.; Radwan, M.M.; Wanas, A.S.; Khan, I.A. Repellent activity of carrot seed essential oil and its pure compound, carotol, against mosquitoes. J. Am. Mosq. Control Assoc. 2018, 34, 272–280. [Google Scholar] [CrossRef]
- Ali, A.; Tabanca, N.; Demirci, B.; Baser, K.H.C.; Ellis, J.; Gray, S.; Lackey, B.R.; Murphy, C.; Khan, I.A.; Wedge, D.E. Composition, mosquito larvicidal, biting deterrent and antifungal activity of essential oils of different plant parts of Cupressus arizonica var. glabra (‘Carolina Sapphire’). Nat. Prod. Commun. 2013, 8, 257–260. [Google Scholar] [CrossRef]
- Cantrell, C.L.; Jones, A.M.P.; Ali, A. Isolation and identification of mosquito (Aedes aegypti) biting-deterrent compounds from the native American ethnobotanical remedy plant Hierochloe odorata (Sweetgrass). J. Agric. Food Chem. 2016, 64, 8352–8358. [Google Scholar] [CrossRef]
- Martins, D.; Nunez, C.V. Secondary metabolites from Rubiaceae species. Molecules 2015, 20, 13422–13495. [Google Scholar] [CrossRef]
- Govaerts, R. Stenaria in Kew Science Plants of the World online. The Board of Trustees of the Royal Botanic Gardens, Kew. 2019. Available online: https://powo.science.kew.org/taxon/urn:lsid:ipni.org:names:20001881-1 (accessed on 13 June 2019).
- Terrell, E.E. Taxonomy of Stenaria (Rubiaceae: Hedyotideae), a new genus including Hedyotis nigricans. SIDA Contrib. Bot. 2001, 591–614. [Google Scholar]
- Lajis, N.H.; Ahmad, R. Phytochemical studies and pharmacological activities of plants in genus Hedyotis/Oldenlandia. Stud. Nat. Prod. Chem. 2006, 33, 1057–1090. [Google Scholar]
- Dinda, B.; Debnath, S.; Harigaya, Y. Naturally occurring iridoids. A review, part 1. Chem. Pharm. Bull. 2007, 55, 159–222. [Google Scholar] [CrossRef] [PubMed]
- Drewes, S.E.; Horn, M.M.; Connolly, J.D.; Bredenkamp, B. Enolic iridolactone and other iridoids from Alberta magna. Phytochemistry 1998, 47, 991–996. [Google Scholar] [CrossRef]
- Li, L.; Zou, J.; Xia, Q.; Cui, H.; You, S.; Liu, Y.; Wang, Q. Anti-TMV and insecticidal potential of four iridoid glycosides from Gardenia Jasminoides fruit. Chem. Res. Chin. Univ. 2018, 34, 697–699. [Google Scholar] [CrossRef]
- Zulfiqar, F.; Khan, S.I.; Ali, Z.; Wang, Y.-H.; Ross, S.A.; Viljoen, A.M.; Khan, I.A. Norlignan glucosides from Hypoxis hemerocallidea and their potential in vitro anti-inflammatory activity via inhibition of iNOS and NF-κB. Phytochemistry 2020, 172, 112273. [Google Scholar] [CrossRef]
- Anh, N.T.H.; Tam, K.T.; Tuan, N.V.; Bao, N.C.; Thien, D.D.; Quan, T.D.; Tam, N.T.; Thuy, T.T.; Sung, T.V. Chemical constituents of Hedyotis ampliflora. Chem. Nat. Compd. 2019, 55, 379–381. [Google Scholar] [CrossRef]
- Linden, A.; Calis, I.; Neuburger, M.; Sticher, O. Asperuloside monohydrate. Acta Crystallogr. C Struct. Chem. 2000, 56, 616–618. [Google Scholar] [CrossRef]
- Bianco, A.; Passacantilli, P.; Polidori, G.; Nicoletti, M. 1H and 13CNMR data of C-6 epimeric iridoids. Org. Magn. Reson. 1983, 21, 460–461. [Google Scholar] [CrossRef]
- Iavarone, C.; Sen, A.; Trogolo, C.; Villa, S. Mollugoside, an iridoid glucoside from Galium mollugo. Phytochemistry 1983, 22, 175–178. [Google Scholar] [CrossRef]
- Van Long, L.H.; Nga, V.T.; Dam, N.P.; Hung, M.A.; Dung, T.D.; Quang, T.T.; Phung, N.K.P. Three new iridoid glucoside salts from Hedyotis tenelliflora growing in Vietnam. Nat. Prod. Commun. 2013, 8, 1507–1508. [Google Scholar] [CrossRef]
- Lee, T.G.; Kim, D.K. Iridoid compounds from the whole plant of Galium verum var. asiaticum. Nat. Prod. Sci. 2013, 19, 227–230. [Google Scholar]
- Peng, J.-N.; Feng, X.-Z.; Li, G.; Liang, X. Chemical investigation of genus Hedyotis. II. Isolation and identification of iridoids from Hedyotis chrysotricha. Acta Pharm. Sin. 1997, 32, 908–913. [Google Scholar]
- Kamiya, K.; Fujita, Y.; Saiki, Y.; Hanani, E.; Mansur, U.; Satake, T. Studies on the constituents of Indonesian Borreria latifolia. Heterocycles 2002, 56, 537–544. [Google Scholar]
- Klun, J.A.; Kramer, M.; Debboun, M. A new in vitro bioassay system for discovery of novel human-use mosquito repellents. J. Am. Mosq. Control Assoc. 2005, 21, 64–70. [Google Scholar] [CrossRef]
- Statistical Analysis System. SAS Users Guide: Statistics; SAS Institute Inc.: Cary, NC, USA, 2012. [Google Scholar]
- Chen, J.; Cantrell, C.; Duke, S.; Allen, M. Repellency of callicarpenal and intermedeol against workers of imported fire ants (Hymenoptera: Formicidae). J. Econ. Entomol. 2008, 101, 265–271. [Google Scholar] [CrossRef]
- Ali, A.; Chen, J.; Khan, I.A. Toxicity and repellency of Magnolia grandiflora seed essential oil and selected pure compounds against the workers of hybrid imported fire ants (Hymenoptera: Formicidae). J. Econ. Entomol. 2022, 115, 412–416. [Google Scholar] [CrossRef]


| Position | 1 a | 2 a | 3 a | 4 b |
|---|---|---|---|---|
| 1 | 93.1, CH | 93.1, CH | 96.3, CH | 99.4, CH |
| 3 | 149.6, CH | 149.5, CH | 97.8, CH | 150.0, CH |
| 4 | 105.7, C | 105.6, C | 43.7, CH | 116.4, C |
| 5 | 36.9, CH | 36.9, CH | 36.8, CH | 48.3, CH |
| 6 | 84.8, CH | 84.8, CH | 86.6, CH | 83.1, CH |
| 7 | 128.6, CH | 128.4, CH | 125.4, CH | 129.8, CH |
| 8 | 143.5, C | 143.5, C | 151.4, C | 147.3, C |
| 9 | 45.0, CH | 44.6, CH | 45.6, CH | 47.2, CH |
| 10 | 61.2, CH2 | 61.2, CH2 | 62.0, CH2 | 61.5, CH2 |
| 11 | 170.3, C | 170.3, C | 175.4, C | 176.0, C |
| 1′ | 101.0, CH | 101.0, CH | 100.0, CH | 100.2, CH |
| 2′ | 75.1, CH | 75.1, CH | 75.0, CH | 74.8, CH |
| 3′ | 78.8, CH | 78.7, CH | 78.5, CH | 77.6, CH |
| 4′ | 71.7, CH | 71.6, CH | 71.5, CH | 71.3, CH |
| 5′ | 79.2, CH | 79.2, CH | 78.6, CH | 78.1, CH |
| 6′ | 62.9, CH2 | 62.8, CH2 | 62.7, CH2 | 62.4, CH2 |
| 1″ | 176.1, C | 173.3, C | 171.0, C | - |
| 2″ | 41.4, CH | 34.3, CH2 | 20.8, CH3 | - |
| 3″ | 27.2, CH2 | 25.0, CH2 | - | - |
| 4″ | 12.1, CH3 | 31.7, CH2 | - | - |
| 5″ | 17.0, CH3 | 22.8, CH2 | - | - |
| 6″ | - | 14.3, CH3 | - | - |
| 3-OCH3 | - | - | 56.5 | - |
| Position | 1 a | 2 a | 3 a | 4 b |
|---|---|---|---|---|
| 1 | 6.28, d (1.5) | 6.28, br s | 5.52, d (5.9) | 4.86, d (8.7) |
| 3 | 7.61, d (2.2) | 7.61, d (2.0) | 5.40, d (3.3) | 7.34, br s |
| 4 | - | - | 3.58, dd (10.4, 3.3) | - |
| 5 | 3.51, td (6.7, 2.2) | 3.52, td (6.7, 2.0) | 3.50, ddd (10.4, 9.2, 6.6) | 2.91, dd (7.7, 6.5) |
| 6 | 5.46, dt (6.7, 1.9) | 5.45, br d (6.7) | 5.34, br d (6.7) | 4.56, br d (6.5) |
| 7 | 5.72, br d (1.9) | 5.73, br s | 5.96, br s | 5.85, br s |
| 8 | - | - | - | - |
| 9 | 3.43, dd (6.7, 1.5) | 3.45, br d (6.7) | 3.25, dd (9.2, 5.9, 1.2) | 2.81, dd (8.7, 7.7) |
| 10 | 4.66, dd (14.3, 1.3) 4.77, dd (14.3, 1.5) | 4.69, d (14.4) 4.79, d (14.4) | 4.90, d (16.0) 5.20, d (16.0) | 4.20, d (15.4) 4.39, d (15.4) |
| 11 | - | - | - | - |
| 1′ | 5.34, d (7.9) | 5.35, d (7.8) | 5.29, d (7.9) | 4.74, d (7.9) |
| 2′ | 4.07, dd (8.5, 7.9) | 4.09, dd (8.2, 7.8) | 4.02, dd (8.7, 7.9) | 3.25, dd (9.2, 7.9) |
| 3′ | 4.26, dd (8.8, 8.5) | 4.27, dd (8.7, 8.2) | 4.22, dd (9.0, 8.7) | 3.42, dd (9.2, 8.8) |
| 4′ | 4.32, dd (9.3, 8.8) | 4.31, dd (8.7, 8.2) | 4.24, t (9.0) | 3.32 c |
| 5′ | 4.00, ddd (9.3, 5.2, 2.5) | 4.01, ddd (8.2, 5.3, 2.4) | 3.87, m | 3.30, m |
| 6′ | 4.40, dd (11.8, 5.2) 4.52, dd (11.8, 2.5) | 4.40, dd (11.8, 5.4) 4.55, dd (11.8, 2.4) | 4.35, dd (11.9, 5.1) 4.50, dd (11.9, 2.5) | 3.66, dd (12.0, 5.3) 3.86, dd (12.0, 2.0) |
| 1″ | - | - | - | - |
| 2″ | 2.44, m | 2.39, t (7.4) | - | - |
| 3″ | 1.45, m1.71, m | 1.64, p (7.4) | - | - |
| 4″ | 0.88, t (7.4) | 1.22, m | - | - |
| 5″ | 1.16, d (7.0) | 1.20, m | - | - |
| 6″ | - | 0.81, t (7.0) | - | - |
| 3-OCH3 | - | - | 3.70, s | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zulfiqar, F.; Ali, A.; Ali, Z.; Khan, I.A. Bioassay-Guided Isolation of Iridoid Glucosides from Stenaria nigricans, Their Biting Deterrence against Aedes aegypti (Diptera: Culicidae), and Repellency Assessment against Imported Fire Ants (Hymenoptera: Formicidae). Molecules 2022, 27, 7053. https://doi.org/10.3390/molecules27207053
Zulfiqar F, Ali A, Ali Z, Khan IA. Bioassay-Guided Isolation of Iridoid Glucosides from Stenaria nigricans, Their Biting Deterrence against Aedes aegypti (Diptera: Culicidae), and Repellency Assessment against Imported Fire Ants (Hymenoptera: Formicidae). Molecules. 2022; 27(20):7053. https://doi.org/10.3390/molecules27207053
Chicago/Turabian StyleZulfiqar, Fazila, Abbas Ali, Zulfiqar Ali, and Ikhlas A. Khan. 2022. "Bioassay-Guided Isolation of Iridoid Glucosides from Stenaria nigricans, Their Biting Deterrence against Aedes aegypti (Diptera: Culicidae), and Repellency Assessment against Imported Fire Ants (Hymenoptera: Formicidae)" Molecules 27, no. 20: 7053. https://doi.org/10.3390/molecules27207053
APA StyleZulfiqar, F., Ali, A., Ali, Z., & Khan, I. A. (2022). Bioassay-Guided Isolation of Iridoid Glucosides from Stenaria nigricans, Their Biting Deterrence against Aedes aegypti (Diptera: Culicidae), and Repellency Assessment against Imported Fire Ants (Hymenoptera: Formicidae). Molecules, 27(20), 7053. https://doi.org/10.3390/molecules27207053














