Aptamers: Potential Diagnostic and Therapeutic Agents for Blood Diseases
Abstract
:1. Introduction
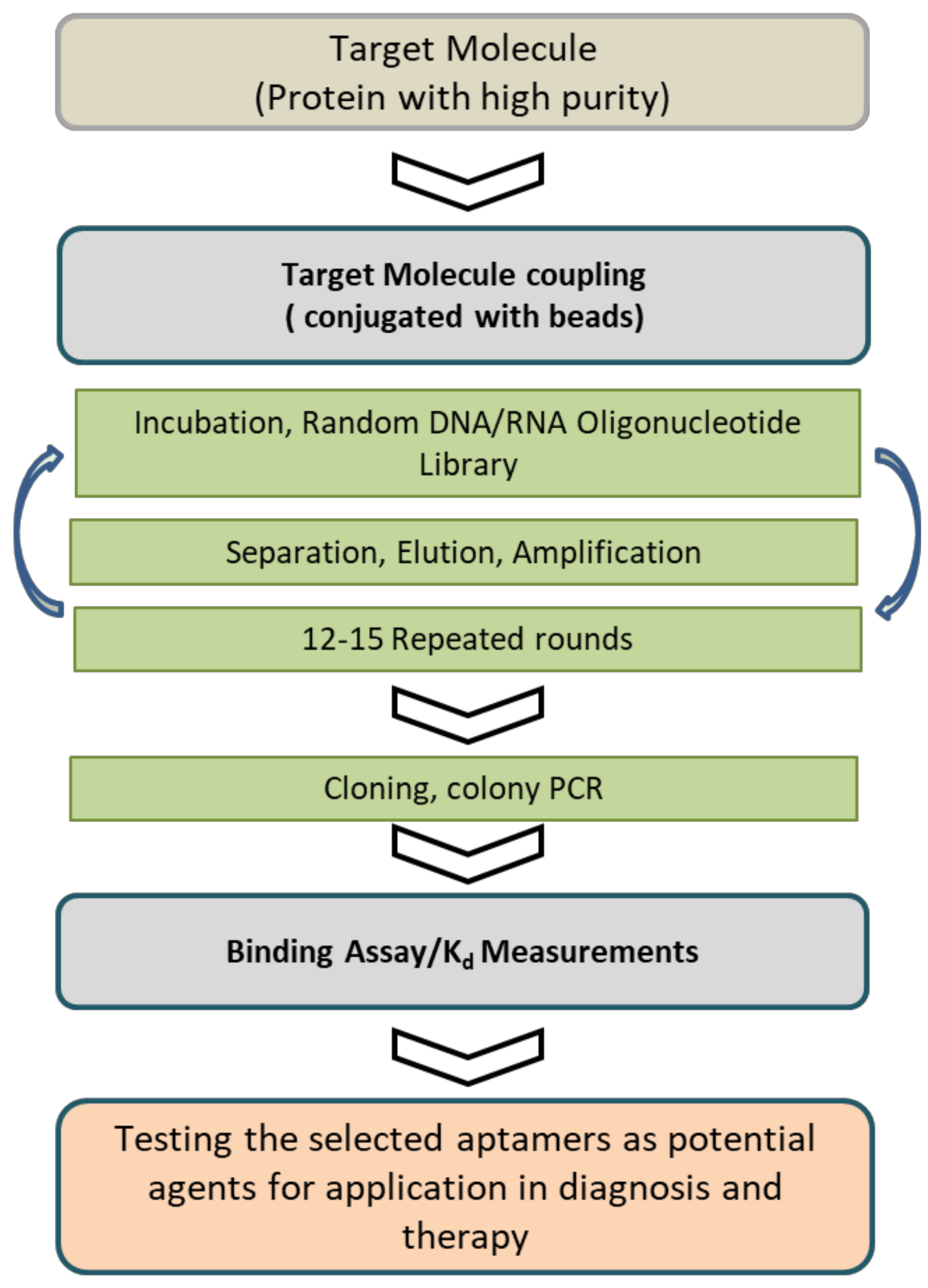
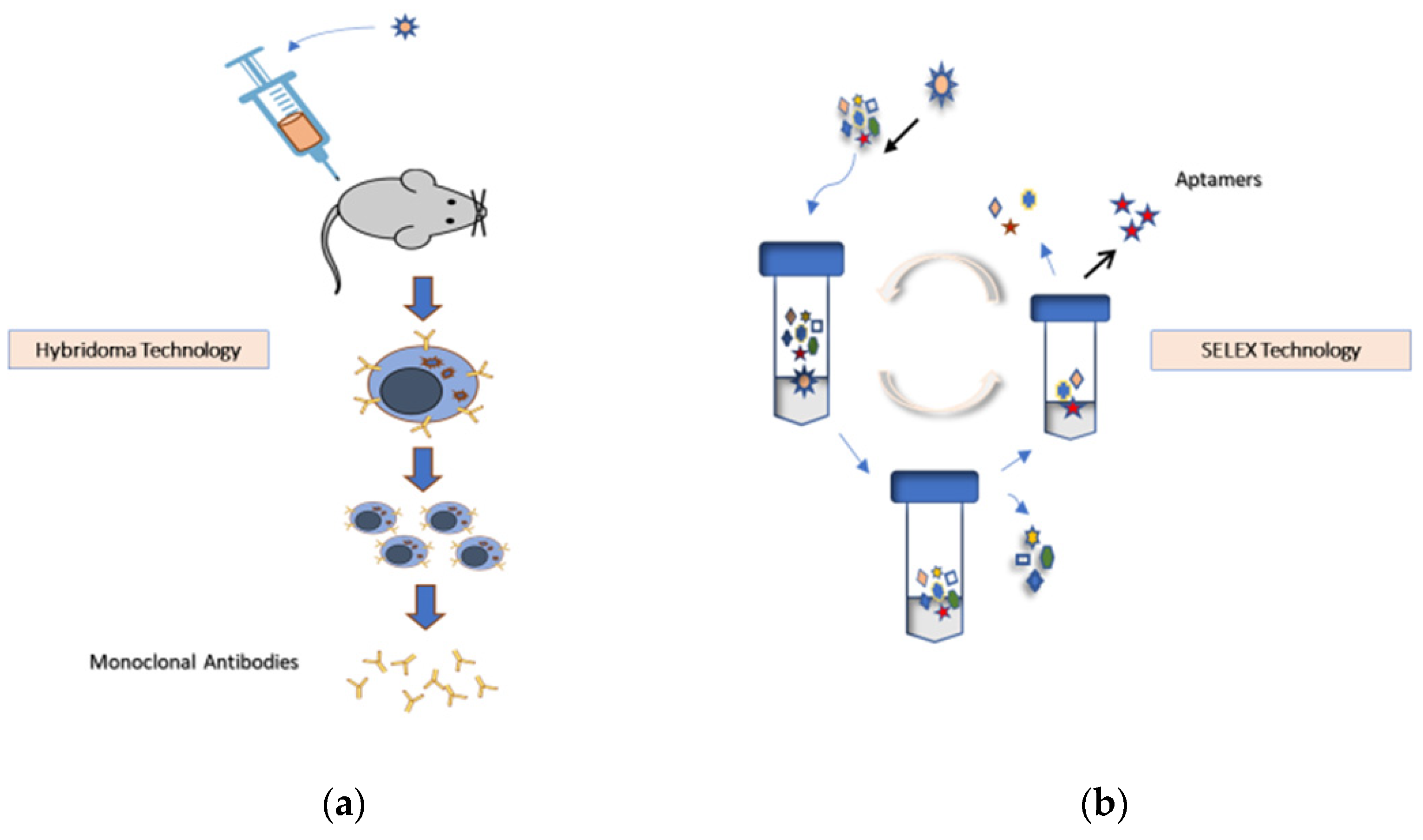
1.1. Aptamer Selection Technology
1.2. Aptamer Optimization and Modification
1.3. Aptamers for Blood Diseases
2. Aptamers for Hematologic Oncology
2.1. Anti-Nucleolin Aptamers for Acute Myeloid Leukemia (AML) Treatment
2.2. CD33-Specific Aptamer for AML Treatment
2.3. Anti-CD30 Aptamers for Diagnosing and Treating CD30-Positive Malignant Lymphomas
2.4. BAFF-R-Specific Aptamer for Non-Hodgkin’s Lymphoma (NHL)
2.5. Anti-CXCL12 Spiegelmer in Chronic Lymphocytic Leukemia and Multiple Myeloma
2.6. CD38-Specific Aptamer in Multiple Myeloma
2.7. Aptamers for B-Cell Burkitt’s Lymphoma Cells
2.8. Aptamers for T-Cell Acute Lymphoblastic Leukemia (T-ALL) Cell Lines
2.9. Aptamer–Drug Conjugates for Targeted Drug Delivery to Tumor Cells
2.10. CD117-Specific Aptamer in AML
3. Aptamers in Hemostasis Disorders
3.1. Aptamers for von Willebrand Factor-Related Diseases
3.2. Aptamers in Hemophilia
4. Aptamers for Hemoglobinopathies
4.1. Anti-P-Selectin RNA Aptamers for Sickle Cell Disease
4.2. Aptamers for Complement-Related Disorders
4.3. Aptamers for Anemia of Chronic Disease
5. Conclusions
6. Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Ellington, A.D.; Szostak, J.W. In vitro selection of RNA molecules that bind specific ligands. Nature 1990, 346, 818–822. [Google Scholar] [CrossRef] [PubMed]
- Siegel, D.L. Recombinant monoclonal antibody technology. Transfus. Clin. Biol. 2002, 9, 15–22. [Google Scholar] [CrossRef]
- Hicke, B.J.; Stephens, A.W. Escort aptamers: A delivery service for diagnosis and therapy. J. Clin. Investig. 2000, 106, 923–928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rimmele, M. Nucleic acid aptamers as tools and drugs: Recent developments. Chembiochem 2003, 4, 963–971. [Google Scholar] [CrossRef]
- Lee, J.F.; Stovall, G.M.; Ellington, A.D. Aptamer therapeutics advance. Curr. Opin. Chem. Biol. 2006, 10, 282–289. [Google Scholar] [CrossRef]
- Ouyang, W.; Yu, Z.; Zhao, X.; Lu, S.; Wang, Z. Aptamers in hematological malignancies and their potential therapeutic implications. Crit. Rev. Oncol./Hematol. 2016, 106, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Shigdar, S.; Ward, A.C.; De, A.; Yang, C.J.; Wei, M.; Duan, W. Clinical applications of aptamers and nucleic acid therapeutics in haematological malignancies. Br. J. Haematol. 2011, 155, 3–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tuerk, C.; Gold, L. Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science 1990, 249, 505–510. [Google Scholar] [CrossRef] [PubMed]
- Chinnappan, R.; Zaghloul, N.S.; AlZabn, R.; Malkawi, A.; Abdel Rahman, A.; Abu-Salah, K.M.; Zourob, M. Aptamer selection and aptasensor construction for bone density biomarkers. Talanta 2020, 224, 121818. [Google Scholar] [CrossRef] [PubMed]
- Yüce, M.; Ullah, N.; Budak, H. Trends in aptamer selection methods and applications. Analyst 2015, 140, 5379–5399. [Google Scholar] [CrossRef]
- Kong, H.Y.; Byun, J. Nucleic Acid aptamers: New methods for selection, stabilization, and application in biomedical science. Biomol. Ther. 2013, 21, 423. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, C.S.M.; Matthews, C.S.; Missailidis, S. DNA aptamers that bind to MUC1 tumour marker: Design and characterization of MUC1-binding single-stranded DNA aptamers. Tumor Biol. 2006, 27, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Lupold, S.E.; Hicke, B.J.; Lin, Y.; Coffey, D.S. Identification and characterization of nuclease-stabilized RNA molecules that bind human prostate cancer cells via the prostate-specific membrane antigen. Cancer Res. 2002, 62, 4029–4033. [Google Scholar] [PubMed]
- Gutsaeva, D.R.; Parkerson, J.B.; Yerigenahally, S.D.; Kurz, J.C.; Schaub, R.G.; Ikuta, T.; Head, C.A. Inhibition of cell adhesion by anti-P-selectin aptamer: A new potential therapeutic agent for sickle cell disease. Blood 2011, 117, 727–735. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Townshend, B.; Aubry, I.; Marcellus, R.C.; Gehring, K.; Tremblay, M.L. An RNA aptamer that selectively inhibits the enzymatic activity of protein tyrosine phosphatase 1B in vitro. ChemBioChem 2010, 11, 1583–1593. [Google Scholar] [CrossRef]
- Liu, Y.; Kuan, C.-T.; Mi, J.; Zhang, X.; Clary, B.M.; Bigner, D.D.; Sullenger, B.A. Aptamers selected against the unglycosylated EGFRvIII ectodomain and delivered intracellularly reduce membrane-bound EGFRvIII and induce apoptosis. Biol. Chem. 2009, 390, 137–144. [Google Scholar] [CrossRef] [Green Version]
- Kaur, H. Recent developments in cell-SELEX technology for aptamer selection. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2018, 1862, 2323–2329. [Google Scholar] [CrossRef]
- Chen, M.; Yu, Y.; Jiang, F.; Zhou, J.; Li, Y.; Liang, C.; Dang, L.; Lu, A.; Zhang, G. Development of cell-SELEX technology and its application in cancer diagnosis and therapy. Int. J. Mol. Sci. 2016, 17, 2079. [Google Scholar] [CrossRef] [Green Version]
- Avci-Adali, M.; Metzger, M.; Perle, N.; Ziemer, G.; Wendel, H.P. Pitfalls of cell-systematic evolution of ligands by exponential enrichment (SELEX): Existing dead cells during in vitro selection anticipate the enrichment of specific aptamers. Oligonucleotides 2010, 20, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Raddatz, M.S.L.; Dolf, A.; Endl, E.; Knolle, P.; Famulok, M.; Mayer, G. Enrichment of cell-targeting and population-specific aptamers by fluorescence-activated cell sorting. Angew. Chem. Int. Ed. 2008, 47, 5190–5193. [Google Scholar] [CrossRef]
- Kunii, T.; Ogura, S.-I.; Mie, M.; Kobatake, E. Selection of DNA aptamers recognizing small cell lung cancer using living cell-SELEX. Analyst 2011, 136, 1310–1312. [Google Scholar] [CrossRef]
- Liu, J.; Liu, H.; Sefah, K.; Liu, B.; Pu, Y.; Van Simaeys, D.; Tan, W. Selection of aptamers specific for adipose tissue. PLoS ONE 2012, 7, e37789. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eissa, S.; Siddiqua, A.; Chinnappan, R.; Zourob, M. Electrochemical selection of a DNA aptamer, and an impedimetric method for determination of the dedicator of cytokinesis 8 by self-assembly of a thiolated aptamer on a gold electrode. Microchim. Acta 2019, 186, 828. [Google Scholar] [CrossRef]
- Eissa, S.; Siddiqua, A.; Chinnappan, R.; Zourob, M. Electrochemical SELEX technique for the selection of DNA aptamers against the small molecule 11-deoxycortisol. ACS Appl. Bio Mater. 2019, 2, 2624–2632. [Google Scholar] [CrossRef]
- Wang, J.; Wei, Y.; Hu, X.; Fang, Y.-Y.; Li, X.; Liu, J.; Wang, S.; Yuan, Q. Protein activity regulation: Inhibition by closed-loop aptamer-based structures and restoration by near-IR stimulation. J. Am. Chem. Soc. 2015, 137, 10576–10584. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhao, Y.; Xu, X.; Xu, K.; Zhang, M.; Huang, K.; Kang, H.; Lin, H.C.; Yang, Y.; Han, D. An intelligent DNA nanorobot for autonomous anticoagulation. Angew. Chem. 2020, 132, 17850–17857. [Google Scholar] [CrossRef]
- Dass, C.R.; Saravolac, E.G.; Li, Y.; Sun, L.-Q. Cellular uptake, distribution, and stability of 10-23 deoxyribozymes. Antisense Nucleic Acid Drug Dev. 2002, 12, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Griffin, L.C.; Tidmarsh, G.F.; Bock, L.C.; Toole, J.J.; Leung, L.L. In vivo anticoagulant properties of a novel nucleotide-based thrombin inhibitor and demonstration of regional anticoagulation in extracorporeal circuits. Blood 1993, 81, 3271–3276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ni, S.; Yao, H.; Wang, L.; Lu, J.; Jiang, F.; Lu, A.; Zhang, G. Chemical modifications of nucleic acid aptamers for therapeutic purposes. Int. J. Mol. Sci. 2017, 18, 1683. [Google Scholar] [CrossRef] [PubMed]
- Ni, S.; Zhuo, Z.; Pan, Y.; Yu, Y.; Li, F.; Liu, J.; Wang, L.; Wu, X.; Li, D.; Wan, Y.; et al. Recent Progress in Aptamer Discoveries and Modifications for Therapeutic Applications. ACS Appl. Mater. Interfaces 2021, 13, 9500–9519. [Google Scholar] [CrossRef]
- Odeh, F.; Nsairat, H.; Alshaer, W.; Ismail, M.A.; Esawi, E.; Qaqish, B.; Bawab, A.A.; Ismail, S.I. Aptamers Chemistry: Chemical Modifications and Conjugation Strategies. Molecules 2019, 25, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmidt, K.S.; Borkowski, S.; Kurreck, J.; Stephens, A.W.; Bald, R.; Hecht, M.; Friebe, M.; Dinkelborg, L.; Erdmann, V.A. Application of locked nucleic acids to improve aptamer in vivo stability and targeting function. Nucleic Acids Res. 2004, 32, 5757–5765. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rohloff, J.C.; Gelinas, A.D.; Jarvis, T.C.; Ochsner, U.A.; Schneider, D.J.; Gold, L.; Janjic, N. Nucleic acid ligands with protein-like side chains: Modified aptamers and their use as diagnostic and therapeutic agents. Mol. Ther.-Nucleic Acids 2014, 3, e201. [Google Scholar] [CrossRef] [PubMed]
- Cotton, R.J.; Graumann, J. Readat: An R package for reading and working with SomaLogic ADAT files. BMC Bioinform. 2016, 17, 201. [Google Scholar] [CrossRef] [Green Version]
- Jhaveri, S.; Olwin, B.; Ellington, A.D. In vitro selection of phosphorothiolated aptamers. Bioorganic Med. Chem. Lett. 1998, 8, 2285–2290. [Google Scholar] [CrossRef]
- Krissanaprasit, A.; Key, C.; Fergione, M.; Froehlich, K.; Pontula, S.; Hart, M.; Carriel, P.; Kjems, J.; Andersen, E.S.; LaBean, T.H. Genetically encoded, functional single-strand RNA origami: Anticoagulant. Adv. Mater. 2019, 31, 1808262. [Google Scholar] [CrossRef]
- Wlotzka, B.; Leva, S.; Eschgfäller, B.; Burmeister, J.; Kleinjung, F.; Kaduk, C.; Muhn, P.; Hess-Stumpp, H.; Klussmann, S. In vivo properties of an anti-GnRH Spiegelmer: An example of an oligonucleotide-based therapeutic substance class. Proc. Natl. Acad. Sci. USA 2002, 99, 8898–8902. [Google Scholar] [CrossRef] [Green Version]
- Yang, S.; Li, H.; Xu, L.; Deng, Z.; Han, W.; Liu, Y.; Jiang, W.; Zu, Y. Oligonucleotide aptamer-mediated precision therapy of hematological malignancies. Mol. Ther.-Nucleic Acids 2018, 13, 164–175. [Google Scholar] [CrossRef] [Green Version]
- Giudice, V.; Mensitieri, F.; Izzo, V.; Filippelli, A.; Selleri, C. Aptamers and antisense oligonucleotides for diagnosis and treatment of hematological diseases. Int. J. Mol. Sci. 2020, 21, 3252. [Google Scholar] [CrossRef]
- Xiang, D.; Shigdar, S.; Qiao, G.; Wang, T.; Kouzani, A.Z.; Zhou, S.-F.; Kong, L.; Li, Y.; Pu, C.; Duan, W. Nucleic acid aptamer-guided cancer therapeutics and diagnostics: The next generation of cancer medicine. Theranostics 2015, 5, 23. [Google Scholar] [CrossRef] [Green Version]
- Roloff, A.; Carlini, A.S.; Callmann, C.E.; Gianneschi, N.C. Micellar thrombin-binding aptamers: Reversible nanoscale anticoagulants. J. Am. Chem. Soc. 2017, 139, 16442–16445. [Google Scholar] [CrossRef]
- Rizzieri, D.; Stockerl-Goldstein, K.; Wei, A.; Herzig, R.H.; Erlandsson, F.; Stuart, R.K. Long-term outcomes of responders in a randomized, controlled phase II trial of aptamer AS1411 in AML. J. Clin. Oncol. 2010, 28, 6557. [Google Scholar] [CrossRef]
- Zhang, S.Q.; Wang, G.P.; Zhu, P.; Liang, J.J.; Xu, Y.J.; Peng, M.Y.; Chen, Y.; Tan, S.Q.; Chen, F.P. Screening and structure analysis of nucleic acid aptamers binding to surface of CD33 (+)/CD34 (+) cells from patients with acute myeloid leukemia subtype M₂. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2011, 19, 561–565. [Google Scholar] [PubMed]
- Dürkop, H.; Latza, U.; Hummel, M.; Eitelbach, F.; Seed, B.; Stein, H. Molecular cloning and expression of a new member of the nerve growth factor receptor family that is characteristic for Hodgkin’s disease. Cell 1992, 68, 421–427. [Google Scholar] [CrossRef]
- Parekh, P.; Kamble, S.; Zhao, N.; Zeng, Z.; Portier, B.P.; Zu, Y. Immunotherapy of CD30-expressing lymphoma using a highly stable ssDNA aptamer. Biomaterials 2013, 34, 8909–8917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thompson, J.S.; Bixler, S.A.; Qian, F.; Vora, K.; Scott, M.L.; Cachero, T.G.; Hession, C.; Schneider, P.; Sizing, I.D.; Mullen, C. BAFF-R, a newly identified TNF receptor that specifically interacts with BAFF. Science 2001, 293, 2108–2111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, J.; Tiemann, K.; Chomchan, P.; Alluin, J.; Swiderski, P.; Burnett, J.; Zhang, X.; Forman, S.; Chen, R.; Rossi, J. Dual functional BAFF receptor aptamers inhibit ligand-induced proliferation and deliver siRNAs to NHL cells. Nucleic Acids Res. 2013, 41, 4266–4283. [Google Scholar] [CrossRef]
- Marasca, R.; Maffei, R. NOX-A12: Mobilizing CLL away from home. Blood J. Am. Soc. Hematol. 2014, 123, 952–953. [Google Scholar] [CrossRef] [Green Version]
- Hoellenriegel, J.; Zboralski, D.; Maasch, C.; Rosin, N.Y.; Wierda, W.G.; Keating, M.J.; Kruschinski, A.; Burger, J.A. The Spiegelmer NOX-A12, a novel CXCL12 inhibitor, interferes with chronic lymphocytic leukemia cell motility and causes chemosensitization. Blood J. Am. Soc. Hematol. 2014, 123, 1032–1039. [Google Scholar] [CrossRef]
- Bouyssou, J.M.C.; Ghobrial, I.M.; Roccaro, A.M. Targeting SDF-1 in multiple myeloma tumor microenvironment. Cancer Lett. 2016, 380, 315–318. [Google Scholar] [CrossRef]
- Ludwig, H.; Weisel, K.; Petrucci, M.T.; Leleu, X.; Cafro, A.M.; Laurent, G.; Zojer, N.; Foa, R.; Greil, R.; Yakoub-Agha, I. Final Results from the Phase IIa Study of the Anti-CXCL12 Spiegelmer® Olaptesed Pegol (NOX-A12) in Combination with Bortezomib and Dexamethasone in Patients with Multiple Myeloma; American Society of Hematology: Washington, DC, USA, 2014. [Google Scholar]
- Ludwig, H.; Weisel, K.; Petrucci, M.T.; Leleu, X.; Cafro, A.M.; Garderet, L.; Leitgeb, C.; Foa, R.; Greil, R.; Yakoub-Agha, I. Olaptesed pegol, an anti-CXCL12/SDF-1 Spiegelmer, alone and with bortezomib–dexamethasone in relapsed/refractory multiple myeloma: A Phase IIa Study. Leukemia 2017, 31, 997–1000. [Google Scholar] [CrossRef]
- Wen, J.; Tao, W.; Hao, S.; Iyer, S.P.; Zu, Y. A unique aptamer-drug conjugate for targeted therapy of multiple myeloma. Leukemia 2016, 30, 987–991. [Google Scholar] [CrossRef]
- Mallikaratchy, P.; Tang, Z.; Kwame, S.; Meng, L.; Shangguan, D.; Tan, W. Aptamer directly evolved from live cells recognizes membrane bound immunoglobin heavy mu chain in Burkitt’s lymphoma cells. Mol Cell Proteom. 2007, 6, 2230–2238. [Google Scholar] [CrossRef] [Green Version]
- Shangguan, D.; Cao, Z.; Meng, L.; Mallikaratchy, P.; Sefah, K.; Wang, H.; Li, Y.; Tan, W. Cell-specific aptamer probes for membrane protein elucidation in cancer cells. J. Proteome Res. 2008, 7, 2133–2139. [Google Scholar] [CrossRef] [Green Version]
- Jiang, G.; Zhang, M.; Yue, B.; Yang, M.; Carter, C.; Al-Quran, S.Z.; Li, B.; Li, Y. PTK7: A new biomarker for immunophenotypic characterization of maturing T cells and T cell acute lymphoblastic leukemia. Leuk. Res. 2012, 36, 1347–1353. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, Y.F.; Shangguan, D.; Liu, H.; Phillips, J.A.; Zhang, X.; Chen, Y.; Tan, W. Molecular assembly of an aptamer–drug conjugate for targeted drug delivery to tumor cells. ChemBioChem 2009, 10, 862–868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, Y.; Li, Y.; Tang, F. Nucleic Acid Aptamer: A Novel Potential Diagnostic and Therapeutic Tool for Leukemia. OncoTargets Ther. 2019, 12, 10597–10613. [Google Scholar] [CrossRef] [Green Version]
- Hans, C.P.; Finn, W.G.; Singleton, T.P.; Schnitzer, B.; Ross, C.W. Usefulness of anti-CD117 in the flow cytometric analysis of acute leukemia. Am. J. Clin. Pathol. 2002, 117, 301–305. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Pei, S.-N.; Qi, J.; Lin, P.; Zu, Y. Targeted Therapy of Acute Myeloid Leukemia by a CD117-Specific Aptamer-Drug-Conjugate; American Society of Hematology: Washington, DC, USA, 2013. [Google Scholar]
- Zhao, N.; Pei, S.N.; Qi, J.; Zeng, Z.; Iyer, S.P.; Lin, P.; Tung, C.H.; Zu, Y. Oligonucleotide aptamer-drug conjugates for targeted therapy of acute myeloid leukemia. Biomaterials 2015, 67, 42–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jilma, B.; Paulinska, P.; Jilma-Stohlawetz, P.; Gilbert, J.C.; Hutabarat, R.; Knöbl, P. A randomised pilot trial of the anti-von Willebrand factor aptamer ARC1779 in patients with type 2b von Willebrand disease. Thromb. Haemost. 2010, 104, 563–570. [Google Scholar] [PubMed]
- Gilbert, J.C.; DeFeo-Fraulini, T.; Hutabarat, R.M.; Horvath, C.J.; Merlino, P.G.; Marsh, H.N.; Healy, J.M.; BouFakhreddine, S.; Holohan, T.V.; Schaub, R.G. Clinical Perspective. Circulation 2007, 116, 2678–2686. [Google Scholar] [CrossRef]
- Markus, H.S.; McCollum, C.; Imray, C.; Goulder, M.A.; Gilbert, J.; King, A. The von Willebrand inhibitor ARC1779 reduces cerebral embolization after carotid endarterectomy: A randomized trial. Stroke 2011, 42, 2149–2153. [Google Scholar] [CrossRef] [Green Version]
- Wood, J.P.; Ellery, P.E.R.; Maroney, S.A.; Mast, A.E. Biology of tissue factor pathway inhibitor. Blood J. Am. Soc. Hematol. 2014, 123, 2934–2943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waters, E.K.; Genga, R.M.; Schwartz, M.C.; Nelson, J.A.; Schaub, R.G.; Olson, K.A.; Kurz, J.C.; McGinness, K.E. Aptamer ARC19499 mediates a procoagulant hemostatic effect by inhibiting tissue factor pathway inhibitor. Blood J. Am. Soc. Hematol. 2011, 117, 5514–5522. [Google Scholar] [CrossRef]
- Waters, E.K.; Genga, R.M.; Thomson, H.A.; Schaub, R.G.; Kurz, J.C.; McGinness, K.E. Investigation into the Mechanism of Action and Binding Site of BAX 499, an Aptamer Against Tissue Factor Pathway Inhibitor; American Society of Hematology: Washington, DC, USA, 2011. [Google Scholar]
- Dockal, M.; Pachlinger, R.; Hartmann, R.; Knappe, S.; Sorensen, B.; Wong, W.Y.; Conlan, M.; Cecerle, M.; Ewenstein, B.M.; Ehrlich, H.J. Biological Explanation of Clinically Observed Elevation of TFPI Plasma Levels after Treatment with TFPI-Antagonistic Aptamer BAX 499; American Society of Hematology: Washington, DC, USA, 2012. [Google Scholar]
- Dockal, M.; Hartmann, R.; Knappe, S.; Palige, M.; Kammlander, W.; Kunckova, K.; Ehrlich, H.J.; Scheiflinger, F. Effect of Increased Tissue Factor Pathway Inhibitor (TFPI) Levels on Factor Xa Inhibition and Global Hemostasis in the Presence of TFPI-Antagonistic Aptamer BAX 499; American Society of Hematology: Washington, DC, USA, 2012. [Google Scholar]
- Telen, M.J. Role of adhesion molecules and vascular endothelium in the pathogenesis of sickle cell disease. Hematol. Am. Soc. Hematol. Educ. Program 2007, 2007, 84–90. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Xu, C.; Manwani, D.; Frenette, P.S. Neutrophils, platelets, and inflammatory pathways at the nexus of sickle cell disease pathophysiology. Blood 2016, 127, 801–809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Biesecker, G.; Dihel, L.; Enney, K.; Bendele, R.A. Derivation of RNA aptamer inhibitors of human complement C5. Immunopharmacology 1999, 42, 219–230. [Google Scholar] [CrossRef]
- Woodruff, R.S.; Xu, Y.; Layzer, J.; Wu, W.; Ogletree, M.L.; Sullenger, B.A. Inhibiting the intrinsic pathway of coagulation with a factor XII-targeting RNA aptamer. J. Thromb. Haemost. 2013, 11, 1364–1373. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aljohani, M.M.; Chinnappan, R.; Eissa, S.; Alsager, O.A.; Weber, K.; Cialla-May, D.; Popp, J.; Zourob, M. In Vitro Selection of Specific DNA Aptamers against the Anti-Coagulant Dabigatran Etexilate. Sci. Rep. 2018, 8, 13290. [Google Scholar] [CrossRef] [PubMed]
- Aljohani, M.M.; Chinnappan, R.; Alsager, O.A.; AlZabn, R.; Alhoshani, A.; Weber, K.; Cialla-May, D.; Popp, J.; Zourob, M. Mapping the binding region of aptamer targeting small molecule: Dabigatran etexilate, an anti-coagulant. Talanta 2020, 218, 121132. [Google Scholar] [CrossRef]
- Chen, W.; Voos, K.M.; Josephson, C.D.; Li, R. Short-Acting Anti-VWF (von Willebrand Factor) Aptamer Improves the Recovery, Survival, and Hemostatic Functions of Refrigerated Platelets. Arter. Thromb. Vasc. Biol. 2019, 39, 2028–2037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giudice, V.; Biancotto, A.; Wu, Z.; Cheung, F.; Candia, J.; Fantoni, G.; Kajigaya, S.; Rios, O.; Townsley, D.; Feng, X. Aptamer-based proteomics of serum and plasma in acquired aplastic anemia. Exp. Hematol. 2018, 68, 38–50. [Google Scholar] [CrossRef]
- Çelik, H.; Lindblad, K.E.; Popescu, B.; Gui, G.; Goswami, M.; Valdez, J.; DeStefano, C.; Lai, C.; Thompson, J.; Ghannam, J.Y. Highly multiplexed proteomic assessment of human bone marrow in acute myeloid leukemia. Blood Adv. 2020, 4, 367–379. [Google Scholar] [CrossRef] [Green Version]
- Krust, B.; El Khoury, D.; Soundaramourty, C.; Nondier, I.; Hovanessian, A.G. Suppression of tumorigenicity of rhabdoid tumor derived G401 cells by the multivalent HB-19 pseudopeptide that targets surface nucleolin. Biochimie 2011, 93, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Gattoni-Celli, S.; Buckner, C.; Lazarchick, J.; Stuart, R.; Fernandes, D. Overexpression of nucleolin in engrafted acute myelogenous leukemia cells. Am. J. Hematol. 2009, 84, 535–538. [Google Scholar] [CrossRef]
- Bates, P.J.; Kahlon, J.B.; Thomas, S.D.; Trent, J.O.; Miller, D.M. Antiproliferative activity of G-rich oligonucleotides correlates with protein binding. J. Biol. Chem. 1999, 274, 26369–26377. [Google Scholar] [CrossRef] [Green Version]
- Dinndorf, P.A.; Andrews, R.G.; Benjamin, D.; Ridgway, D.; Wolff, L.; Bernstein, I.D. Expression of normal myeloid-associated antigens by acute leukemia cells. Blood 1986, 67, 1048–1053. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Liu, J.; Gu, H. Targeting nucleolin to obstruct vasculature feeding with an intelligent DNA nanorobot. J. Cell. Mol. Med. 2019, 23, 2248–2250. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Tan, J.; Zhou, L.; Shan, X.; Liu, J.; Ma, Y. Synthesis and Application of AS1411-Functionalized Gold Nanoparticles for Targeted Therapy of Gastric Cancer. ACS Omega 2020, 5, 31227–31233. [Google Scholar] [CrossRef] [PubMed]
- Bates, P.J.; Reyes-Reyes, E.M.; Malik, M.T.; Murphy, E.M.; O’toole, M.G.; Trent, J.O. G-quadruplex oligonucleotide AS1411 as a cancer-targeting agent: Uses and mechanisms. Biochim. Biophys. Acta (BBA)-Gen. Subj. 2017, 1861, 1414–1428. [Google Scholar] [CrossRef] [PubMed]
- Jurcic, J.G.; Levy, M.Y.; Park, J.H.; Ravandi, F.; Perl, A.E.; Pagel, J.M.; Smith, B.D.; Estey, E.H.; Kantarjian, H.; Cicic, D. Phase I Trial of Targeted Alpha-Particle Therapy with Actinium-225 (225Ac)-Lintuzumab and Low-Dose Cytarabine (LDAC) in Patients Age 60 or Older with Untreated Acute Myeloid Leukemia (AML); American Society of Hematology: Washington, DC, USA, 2016. [Google Scholar]
- Orava, E.W.; Cicmil, N.; Gariépy, J. Delivering cargoes into cancer cells using DNA aptamers targeting internalized surface portals. Biochim. Biophys. Acta (BBA)-Biomembr. 2010, 1798, 2190–2200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, C.; Wang, Y.; Ge, M.H.; Fu, Y.J.; Hao, R.; Islam, K.; Huang, P.; Chen, F.; Sun, J.; Naranmandura, H. Rapid identification of specific DNA aptamers precisely targeting CD33 positive leukemia cells through a paired cell-based approach. Biomater. Sci. 2019, 7, 938–950. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Zhao, N.; Zeng, Z.; Feng, Y.; Tung, C.-H.; Chang, C.-C.; Zu, Y. Using an RNA aptamer probe for flow cytometry detection of CD30-expressing lymphoma cells. Lab. Investig. 2009, 89, 1423–1432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, X.; Yang, Y.; Kong, F.; Zhang, L.; Wei, K. CD30 aptamer-functionalized PEG-PLGA nanoparticles for the superior delivery of doxorubicin to anaplastic large cell lymphoma cells. Int. J. Pharm. 2019, 564, 340–349. [Google Scholar] [CrossRef]
- Shi, H.; Tang, Z.; Kim, Y.; Nie, H.; Huang, Y.F.; He, X.; Deng, K.; Wang, K.; Tan, W. In vivo fluorescence imaging of tumors using molecular aptamers generated by cell-SELEX. Chem.-Asian J. 2010, 5, 2209–2213. [Google Scholar] [CrossRef]
- Musumeci, D.; Platella, C.; Riccardi, C.; Moccia, F.; Montesarchio, D. Fluorescence Sensing Using DNA Aptamers in Cancer Research and Clinical Diagnostics. Cancers 2017, 9, 174. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lagneaux, L.; Delforge, A.; Bron, D.; De Bruyn, C.; Stryckmans, P. Chronic lymphocytic leukemic B cells but not normal B cells are rescued from apoptosis by contact with normal bone marrow stromal cells. Blood J. Am. Soc. Hematol. 1998, 91, 2387–2396. [Google Scholar]
- D’Apuzzo, M.; Rolink, A.; Loetscher, M.; Hoxie, J.A.; Clark-Lewis, I.; Melchers, F.; Baggiolini, M.; Moser, B. The chemokine SDF-1, stromal cell-derived factor 1, attracts early stage B cell precursors via the chemokine receptor CXCR4. Eur. J. Immunol. 1997, 27, 1788–1793. [Google Scholar] [CrossRef]
- Hattori, K.; Heissig, B.; Tashiro, K.; Honjo, T.; Tateno, M.; Shieh, J.-H.; Hackett, N.R.; Quitoriano, M.S.; Crystal, R.G.; Rafii, S. Plasma elevation of stromal cell–derived factor-1 induces mobilization of mature and immature hematopoietic progenitor and stem cells. Blood J. Am. Soc. Hematol. 2001, 97, 3354–3360. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Burger, J.A.; Kipps, T.J. CXCR4: A key receptor in the crosstalk between tumor cells and their microenvironment. Blood 2006, 107, 1761–1767. [Google Scholar] [CrossRef]
- Kang, H.; O’Donoghue, M.B.; Liu, H.; Tan, W. A liposome-based nanostructure for aptamer directed delivery. Chem. Commun. 2010, 46, 249–251. [Google Scholar] [CrossRef] [PubMed]
- Paerl, H.W. Coastal eutrophication and harmful algal blooms: Importance of atmospheric deposition and groundwater as “new” nitrogen and other nutrient sources. Limnol. Oceanogr. 1997, 42, 1154–1165. [Google Scholar] [CrossRef] [Green Version]
- Diener, J.L.; Daniel Lagasse, H.A.; Duerschmied, D.; Merhi, Y.; Tanguay, J.F.; Hutabarat, R.; Gilbert, J.; Wagner, D.D.; Schaub, R. Inhibition of von Willebrand factor-mediated platelet activation and thrombosis by the anti-von Willebrand factor A1-domain aptamer ARC1779. J. Thromb. Haemost. 2009, 7, 1155–1162. [Google Scholar] [CrossRef] [PubMed]
- Cataland, S.R.; Peyvandi, F.; Mannucci, P.M.; Lämmle, B.; Hovinga, J.A.K.; Machin, S.J.; Scully, M.; Rock, G.; Gilbert, J.C.; Yang, S. Initial experience from a double-blind, placebo-controlled, clinical outcome study of ARC1779 in patients with thrombotic thrombocytopenic purpura. Am. J. Hematol. 2012, 87, 430–432. [Google Scholar] [CrossRef]
- Nimjee, S.M.; Dornbos, D., 3rd; Pitoc, G.A.; Wheeler, D.G.; Layzer, J.M.; Venetos, N.; Huttinger, A.; Talentino, S.E.; Musgrave, N.J.; Moody, H.; et al. Preclinical Development of a vWF Aptamer to Limit Thrombosis and Engender Arterial Recanalization of Occluded Vessels. Mol. Ther. 2019, 27, 1228–1241. [Google Scholar] [CrossRef] [PubMed]
- Mackman, N. Role of tissue factor in hemostasis, thrombosis, and vascular development. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1015–1022. [Google Scholar] [CrossRef]
- Maroney, S.A.; Mast, A.E. New insights into the biology of tissue factor pathway inhibitor. J. Thromb. Haemost. 2015, 13, S200–S207. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riccardi, C.; Napolitano, E.; Platella, C.; Musumeci, D.; Montesarchio, D. G-quadruplex-based aptamers targeting human thrombin: Discovery, chemical modifications and antithrombotic effects. Pharmacol. Ther. 2021, 217, 107649. [Google Scholar] [CrossRef]
- Ataga, K.I.; Kutlar, A.; Kanter, J.; Liles, D.; Cancado, R.; Friedrisch, J.; Guthrie, T.H.; Knight-Madden, J.; Alvarez, O.A.; Gordeuk, V.R. SUSTAIN: A Multicenter, Randomized, Placebo-Controlled, Double-Blind, 12-Month Study to Assess Safety and Efficacy of SelG1 with or without Hydroxyurea Therapy in Sickle Cell Disease Patients with Sickle Cell-Related Pain Crises; American Society of Hematology: Washington, DC, USA, 2016. [Google Scholar]
- Gutsaeva, D.; Parkerson, J.; Schaub, R.G.; Kurz, J.; Head, A. Single-Stranded Oligonucleotide Binding to P-Selectin Inhibits Adhesion of Sickle Red Blood Cells and Leukocytes to Endothelial Cells in Sickle Cell Model Mice: Novel Therapeutics for Vaso-Occlusive Episodes; American Society of Hematology: Washington, DC, USA, 2009. [Google Scholar]
- Sunyer, J.O.; Lambris, J.D. Evolution and diversity of the complement system of poikilothermic vertebrates. Immunol. Rev. 1998, 166, 39–57. [Google Scholar] [CrossRef] [PubMed]
- Reis, E.S.; Mastellos, D.C.; Yancopoulou, D.; Risitano, A.M.; Ricklin, D.; Lambris, J.D. Applying complement therapeutics to rare diseases. Clin. Immunol. 2015, 161, 225–240. [Google Scholar] [CrossRef] [Green Version]
- Hillmen, P.; Hall, C.; Marsh, J.C.; Elebute, M.; Bombara, M.P.; Petro, B.E.; Cullen, M.J.; Richards, S.J.; Rollins, S.A.; Mojcik, C.F.; et al. Effect of eculizumab on hemolysis and transfusion requirements in patients with paroxysmal nocturnal hemoglobinuria. N. Engl. J. Med. 2004, 350, 552–559. [Google Scholar] [CrossRef] [Green Version]
- Meyers, G.; Weitz, I.; Lamy, T.; Cahn, J.-Y.; Kroon, H.-A.; Severino, B.; Uranga, M.T.; Alonso, M.S.; Vela, J.A.G.a.; Hill, A. Disease-Related Symptoms Reported across a Broad Population of Patients with Paroxysmal Nocturnal Hemoglobinuria; American Society of Hematology: Washington, DC, USA, 2007. [Google Scholar]
- Brodsky, R.A.; Young, N.S.; Antonioli, E.; Risitano, A.M.; Schrezenmeier, H.; Schubert, J.; Gaya, A.; Coyle, L.; De Castro, C.; Fu, C.-L. Multicenter phase 3 study of the complement inhibitor eculizumab for the treatment of patients with paroxysmal nocturnal hemoglobinuria. Blood J. Am. Soc. Hematol. 2008, 111, 1840–1847. [Google Scholar] [CrossRef]
- Herper, M. The World’s Most Expensive Drugs; Forbes Media: Jersey City, NJ, USA, 2010. [Google Scholar]
- Epstein, D.; Kurz, J.C. Complement Binding Aptamers and Anti-C5 Agents Useful in the Treatment of Ocular Disorders. US20090269356A1, 29 October 2009. [Google Scholar]
- Ganz, T. Hepcidin, a key regulator of iron metabolism and mediator of anemia of inflammation. Blood 2003, 102, 783–788. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Theurl, I.; Aigner, E.; Theurl, M.; Nairz, M.; Seifert, M.; Schroll, A.; Sonnweber, T.; Eberwein, L.; Witcher, D.R.; Murphy, A.T. Regulation of iron homeostasis in anemia of chronic disease and iron deficiency anemia: Diagnostic and therapeutic implications. Blood J. Am. Soc. Hematol. 2009, 113, 5277–5286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruchala, P.; Nemeth, E. The pathophysiology and pharmacology of hepcidin. Trends Pharmacol. Sci. 2014, 35, 155–161. [Google Scholar] [CrossRef] [Green Version]
- Boyce, M.; Warrington, S.; Cortezi, B.; Zöllner, S.; Vauléon, S.; Swinkels, D.W.; Summo, L.; Schwoebel, F.; Riecke, K. Safety, pharmacokinetics and pharmacodynamics of the anti-hepcidin Spiegelmer lexaptepid pegol in healthy subjects. Br. J. Pharmacol. 2016, 173, 1580–1588. [Google Scholar] [CrossRef]
| Monoclonal Antibodies | Aptamers |
|---|---|
| Large molecule (IGG monoclonal antibody approximately 150 kDa) | Small molecule (10–100 times smaller than antibodies) |
| Produced biologically (in vivo) in animal house facilities or reactors | Produced chemically (in vitro) |
| High cost of synthesis | Low cost of synthesis, large-scale bulk production |
| Widely distributed technologies (widely used) | Limited distribution of technologies |
| Difficult to modify | Easy to modify by simple bioconjugate chemistry |
| Contamination by viral or bacterial during manufacturing process can affect product quality | Chemical/lab manufacturing process carries no risk of biological contamination |
| High batch-to-batch variation | Low batch-to-batch variation |
| Clonal variation | No clonal variation |
| Long half-life in vivo (less susceptible to serum degradation and renal filtration) | Short half-life in vivo (susceptible to serum degradation and renal filtration) |
| Often immunogenic | Less immunogenic |
| Thermally unstable | Thermally stable |
| Limited shelf life | Long shelf life |
| Poor internalization into cells/tumors | Efficient cellular internalization |
| Less susceptible to nuclease degradation, rapid elimination from plasma by renal filtration | Susceptible to nuclease degradation, rapid elimination from plasma by renal filtration |
| Antibody conjugation with one type of signaling or binding molecule, such as organic dyes, fluorescent proteins, colored particles, or enzymes, is typically achieved after antibody formation. | Aptamers can be easily conjugated to different secondary reagents such as small nanoparticles, chemotherapeutic drugs, toxins, enzymes, radionuclides, small interfering RNAs and microRNAs, etc., often during aptamer synthesis, secondary reagents. Conjugation can be readily introduced during synthesis |
| Limited ability to utilize negative selection pressure | Ability for a counter (negative) selection |
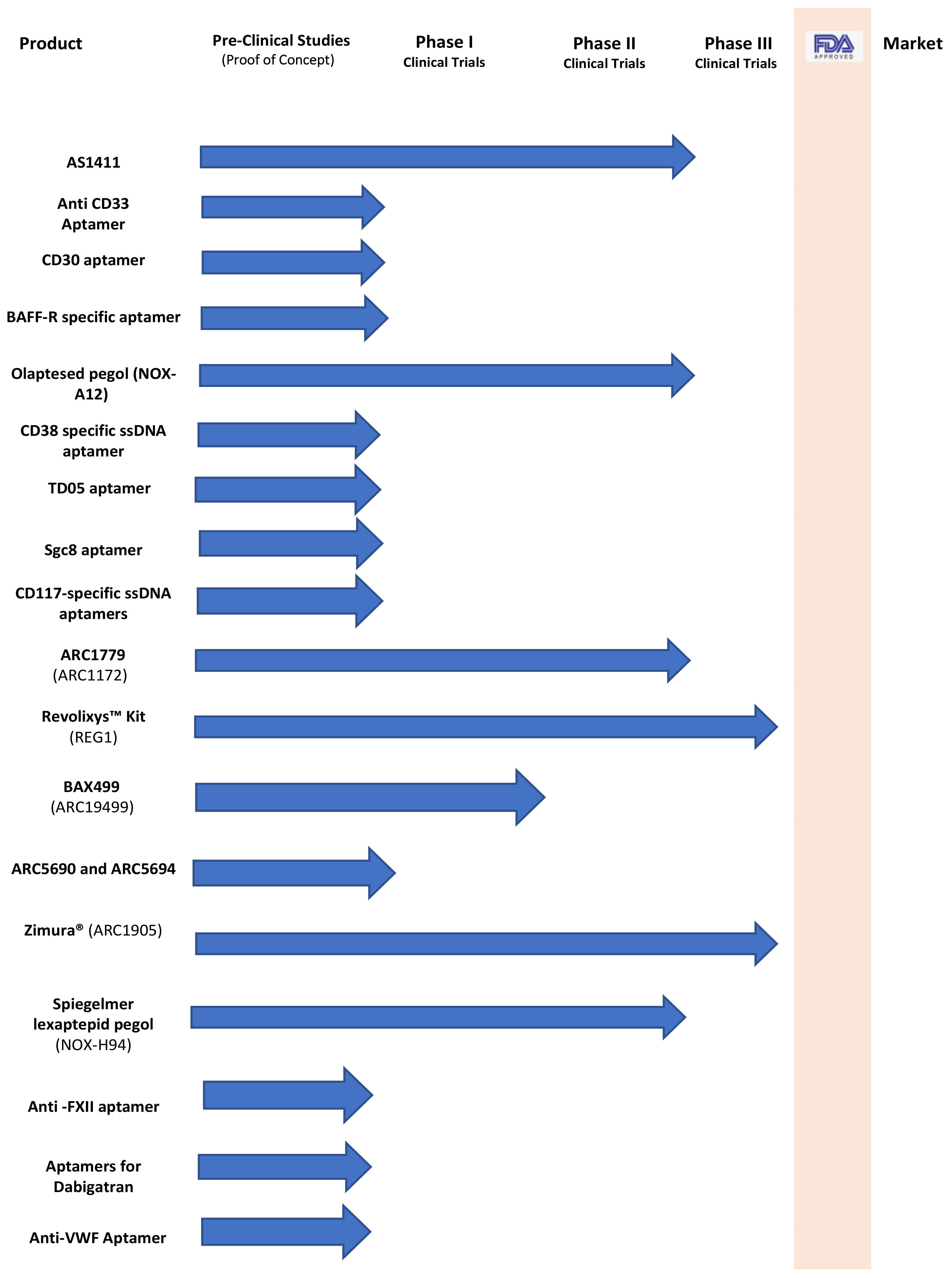
| Target | Name of Aptamer | Type of Aptamer | Hematological Indication | Phase of Testing | References |
|---|---|---|---|---|---|
| Nucleolin | AS1411 aptamer (AGRO100, later renamed AS1411 then ACT-GRO-777) | 26-ntG-rich sequence pegylated DNA aptamer | Acute myeloid leukemia (AML) | Phase II clinical trials in a combination therapy for patients with myeloid leukemia; ClinicalTrials.gov, #NCT00512083 (completed); Phase II clinical trials in a combination therapy for patients with primary refractory or relapsed AML, ClinicalTrials.gov, #NCT01034410 (terminated) | [42] |
| CD33; transmembrane protein | Anti-CD33 aptamer | DNA aptamer | AML | Pre-clinical studies (proof of concept) are binding and being internalized into CD33-positive myeloid cell lines, carrier of chemotherapeutic drugs | [43] |
| CD30; transmembrane protein | Anti-CD30 aptamer (C2NP and PS1NP/ truncated form, PS1NPD) | ssDNA aptamer | Hodgkin’s lymphoma (HL) tumor cells | Pre-clinical studies (proof of concept) | [44,45] |
| B-cell activating factor receptor (BAFF-R) | BAFF-R-specific aptamer | RNA aptamers a | BAFF-R-positive lymphoma cells, such as non-Hodgkin’s lymphoma (NHL) | Pre-clinical studies (proof of concept); specificity of this aptamer to bind and internalize to BAFF-R-positive lymphoma cells, carrier of chemotherapeutic drugs | [46,47] |
| Stromal cell-derived factor-1 (SDF-1/CXCL 12) | NOX-A12 (olaptesed pegol) | 45-nt RNA, L-ribonucleic acid, Spiegelmer, pegylated | Multiple myeloma (MM), CLL | Phase II clinical trials in a combination therapy for MM, ClinicalTrials.gov, #NCT01521533; Phase II clinical trials in a combination therapy for CLL, ClinicalTrials.gov, #NCT01486797 | [48,49,50,51,52] |
| CD38; transmembrane glycoprotein, myeloma cells | CD38-specific ssDNA aptamer | ssDNA | MM | Pre-clinical studies (proof of concept); conjugated to a cytotoxic agent to target and release this agent within MM cells and induce MM cell apoptosis | [53] |
| Immunoglobulin heavy mu chain (IGHM) | TD05 aptamer | ssDNA | Burkitt’s lymphoma | Pre-clinical studies (proof of concept); successfully recognize tumor cells with high sensitivity and specificity | [54] |
| Membrane receptor, protein tyrosine kinase 7 (PTK7) tyrosine | Sgc8 aptamer | ssDNA | T-cell acute lymphoblastic leukemia (T-ALL) | Pre-clinical studies (proof of concept); sgc8 aptamer–DOX conjugate possesses high binding affinity and the ability to be efficiently internalized by target cells; “Targeted drug delivery to PTK7 T-ALL leukemia” | [55,56,57,58] |
| CD117 (c-Kit), transmembrane receptor | CD117-specific ssDNA aptamers | ssDNA | AML | Proof of concept; this Apt-MTX used to target AML cells shows selective growth inhibition of leukemia cells and had no toxicity to normal marrow cells, potential clinical value for use in AML | [59,60,61] |
| von Willebrand factor (VWF) A1 domain to platelet GPIb receptors | ARC1779 | ARC1779 A, pegylated form of DNA aptamer (ARC1172) | von Willebrand factor-related platelet function disorders; Thrombotic thrombocytopenic purpura (TTP) and von Willebrand disease type 2B | Phase II clinical trials for cerebral microembolism in patients undergoing carotid endarterectomy, ClinicalTrials.gov, #NCT00742612 (terminated because of cessation of funding); Phase II for patients with acute TTP, ClinicalTrials.gov, #NCT00726544 (prematurely closed) | [62,63,64] |
| Factor IXa inhibitor; pegnivacogin (RB006) anivamersen (RB007), complementary active control agent | REG1 System/ Revolixys Kit; pegnivacogin (RB006) plus anivamersen (RB007), a complementary (antisense) | System consists of two RNA aptamers: 1-RB006: 2′-ribo purine/2′- fluoro pyrimidine; 2-RB007: 40 kDa PEG plus 2′-O- methyl antidote | Antithrombotic drug | Phase III clinical trial, ClinicalTrials.gov, #NCT01848106 (clinical hold because of allergic reactions) | ClinicalTrials.gov (accessed on 20 May 2021) |
| Tissue factor (TF) | BAX499/ ARC19499 | -32 nucleotides capping with a 3′ inverted dT—a 5′ 40 kDa PEG | Hemophilia | Phase 1; first-in-human and proof-of-mechanism study in hemophilia patients, ClinicalTrials.gov, #NCT01191372 (terminated) | [65,66,67,68,69] |
| P-selectin, cell adhesion molecule | Anti-P-selectin aptamers: ARC5690 and ARC5694 | ARC5690: -33-mer oligonucleotide- a 3′-inverted 2′-deoxy-thymidine—a 5′-40 kDa PEG | Sickle cell disease (SCD) | Pre-clinical studies (proof of concept); Aptamer base p-selectin increases RBC velocity and wall shear rates and reduces the adhesion of RBCs and leukocytes in SCD model mice | [70,71] |
| Complement C5 | C5-specific aptamer (ARC1905, also known as Zimura) | 38-mer 2′F RNA aptamer, 40 kDa PEG to the 5′ end and 3′-3′ linked deoxythymidine to the 3′ end | Potential for complement-related diseases, such as paroxysmal nocturnal hemoglobinuria (PNH) | Phase II/III clinical trials in age-related macular degeneration (AMD) ClinicalTrials.gov, #NCT02686658 Proof of concept of aptamer-based C5 binding and inhibitory activity Potential for study in other complement-related diseases, such as PNH | [72] |
| Hepcidin peptide | NOX-H94, (Spiegelmer® lexaptepid pegol) | 44-nt RNA L-ribonucleic acid, pegylated | Anemia of chronic disease (ACD) | Phase II clinical trials, ClinicalTrials.gov, #NCT02079896 ClinicalTrials.gov, #NCT01691040 ClinicalTrials.gov, #NCT01522794 ClinicalTrials.gov, #NCT01372137 | ClinicalTrials.gov (accessed on 20 May 2021) |
| Human FXII | Anti-FXII aptamer (Aptamer R4cXII 1t) | RNA Aptamer | Thrombosis | Pre-clinical studies (proof of concept); inhibits the intrinsic pathway of coagulation | [73] |
| Direct oral anticoagulants (dabigatran) | DGB-1, DBG-2, DBG-4, and DBG-5 | ssDNA aptamers | Direct oral anticoagulants (dabigatran) | Pre-clinical studies (proof of concept) for monitoring direct oral anticoagulants (dabigatran) | [74,75] |
| Refrigerated platelets (for platelet transfusion) | Anti-VWF aptamer | von Willebrand factor | Refrigerated platelets (for platelet transfusion) | Pre-clinical studies (proof of concept) for the use of ARC1779 to refrigerated platelets; improves post-transfusion recovery and preserves the long-term hemostatic function of refrigerated platelets | [76] |
| Serum and plasma in aplastic anemia (AA), noncellular compartment of human bone marrow in AML | SOMAscan proteomic analysis | RNA-sequencing and proteomics data set | AA, AML | Proof of concept to determine the true proteomic of serum and plasma in AA patients before and after therapy Proof of concept to determine the true proteomic composition of the extracellular soluble compartment of AML patients’ bone marrow | [77,78] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aljohani, M.M.; Cialla-May, D.; Popp, J.; Chinnappan, R.; Al-Kattan, K.; Zourob, M. Aptamers: Potential Diagnostic and Therapeutic Agents for Blood Diseases. Molecules 2022, 27, 383. https://doi.org/10.3390/molecules27020383
Aljohani MM, Cialla-May D, Popp J, Chinnappan R, Al-Kattan K, Zourob M. Aptamers: Potential Diagnostic and Therapeutic Agents for Blood Diseases. Molecules. 2022; 27(2):383. https://doi.org/10.3390/molecules27020383
Chicago/Turabian StyleAljohani, Maher M., Dana Cialla-May, Jürgen Popp, Raja Chinnappan, Khaled Al-Kattan, and Mohammed Zourob. 2022. "Aptamers: Potential Diagnostic and Therapeutic Agents for Blood Diseases" Molecules 27, no. 2: 383. https://doi.org/10.3390/molecules27020383
APA StyleAljohani, M. M., Cialla-May, D., Popp, J., Chinnappan, R., Al-Kattan, K., & Zourob, M. (2022). Aptamers: Potential Diagnostic and Therapeutic Agents for Blood Diseases. Molecules, 27(2), 383. https://doi.org/10.3390/molecules27020383








