1. Introduction
Although scientific advancement has been achieved in almost every field, the most promising area by far is nanotechnology in the 21st century, which has revolutionized almost every discipline of life.
Nanotechnology refers to the manipulation of size, shape and structure at the nanoscale in order to develop novel properties in materials in comparison to their bulk counterparts [
1]. It can be broadly defined as the manipulation, production and designing of devices and structures at the nanoscale by controlling their shape and size in order to bring forth novel properties in them. The core building blocks of nanotechnology are nanoparticles (NPs), particles that range in size from 1 to 100 nm. The extremely small size and larger surface area enable NPs to exhibit properties that are not present in their bulk counterparts. Because of their physical, chemical, optical, magnetic, electrical, catalytic and thermal characteristics, metallic nanoparticles have biomedical, agricultural and environmental applications. Antibacterial metallic nanoparticles offer a possible alternative to conventional antibiotics [
2].
There are many methods of stabilizing NPs, but they all fall into three basic groups: physical, chemical and biological methods. Physical techniques involve the practice of different techniques, including evaporation, condensation and laser ablation. Physical methods used for the synthesis of NPs are completely free of organic contaminants [
3], but they require a lot of energy, a large space for the machinery involved, release a lot of heat and take a long time [
3]. Reducing agents such as polyols, sodium citrate, ascorbic acid and Tollen’s reagents, elemental hydrogen, sodium borohydride and DMF (dimethylformamide) [
4] are employed in chemical techniques to synthesize NPs. There are also certain drawbacks of the chemical method, which include the use of noxious, non-biodegradable chemicals. These chemicals may contaminate NPs’ surfaces making the NPs useless for the majority of biomedical applications. The said chemicals also have a damaging impact on the environment [
5]. Biochemical or green synthesis of NPs is environmentally friendly and an alternative to physical or chemical techniques. It is less expensive, which makes it the method of choice. It can be executed at normal pH and room temperature, thereby reducing the time and efforts requisite to optimize temperature and pH for NPs synthesis. Biomolecules such as proteins and polypeptides, are derived from a wide variety of organisms have proved to be useful for NPs synthesis.
Nanomedicine is a sub-discipline of medicine that uses the fundamentals of nanotechnology to avoid and/or treat a number of sicknesses. In a living organism, nanomedicine entails the use of nanostructured materials for diagnosis, delivery, detection, or actuation [
6]. Nanotechnology has the potential to improve medicine delivery to regions where macromolecules could not reach. Diabetes research is increasingly relying on nanotechnology. It is possible that it will provide information that is more accurate for the diagnosis of diabetes mellitus [
7]. For the treatment of diabetes mellitus (DM) and its consequences, nanotechnology has recently emerged as one of the most important research fields. Within a range of 1–100 nm, the advantages of nano-materials include their versatility, control over size, and capacity to maintain their physiochemical properties. More surface functionalized active components can be encapsulated in nanomaterials with a higher surface area-to-volume ratio, which may facilitate cell adhesion and expedite particular regeneration processes [
8]. Nanospheres as biodegradable polymeric carriers have been used in the oral delivery of insulin, the formation of artificial beta cells, and the creation of artificial pancreas [
9].
Within the Asteraceae, the Cardueae (Thistle) is one of the largest tribes, primarily found in a temperate region. In Pakistan, this is found in Chitral, Swat, Kurram Valley, Gilgit, Astor, Baltistan, Kashmir and Northern Baluchistan. Carduus is known for its therapeutic virtues and is found primarily in Europe. The raw or cooked material of some Carduus taxa is used in traditional and modern medicines to treat liver illnesses, diuretics, digestives and rheumatism. Tonic, antispasmodic, anticancer, antibacterial and anti-inflammatory is just some of the functions of the secondary metabolites found in different species of this genus. Some of these plants are employed in Bulgarian folk medicine as diuretic, cardiotonic and antihemorroidal remedies [
10].
Due to the astounding potential of green synthesized NPs in a number of scientific disciplines, contemporary research was intended to carry out the green synthesis of AuNPs using methanol extract of aerial parts of the plant,
Carduus edelbergii. AuNPs are an important tool used in nanotechnology for a variety of purposes. AuNPs have found their applications in different areas of research, including catalysis, biosensors, bioimaging, delivery of drugs and photonics [
11]. The biocompatibility of AuNPs is their most remarkable aspect, which render them an NP of preference for biomedical applications [
12]. Different in vitro and in vivo tests are used to investigate the biological potentials of AuNPs fabricated with extract in this publication.
3. Discussion
The phytochemical analysis showed that the extract of the aerial parts of
Carduus edelbergii Rech. f. contains a mixture of bioactive compounds that were reported to have therapeutic effects [
13]. It was shown in the result that the methanol extract of
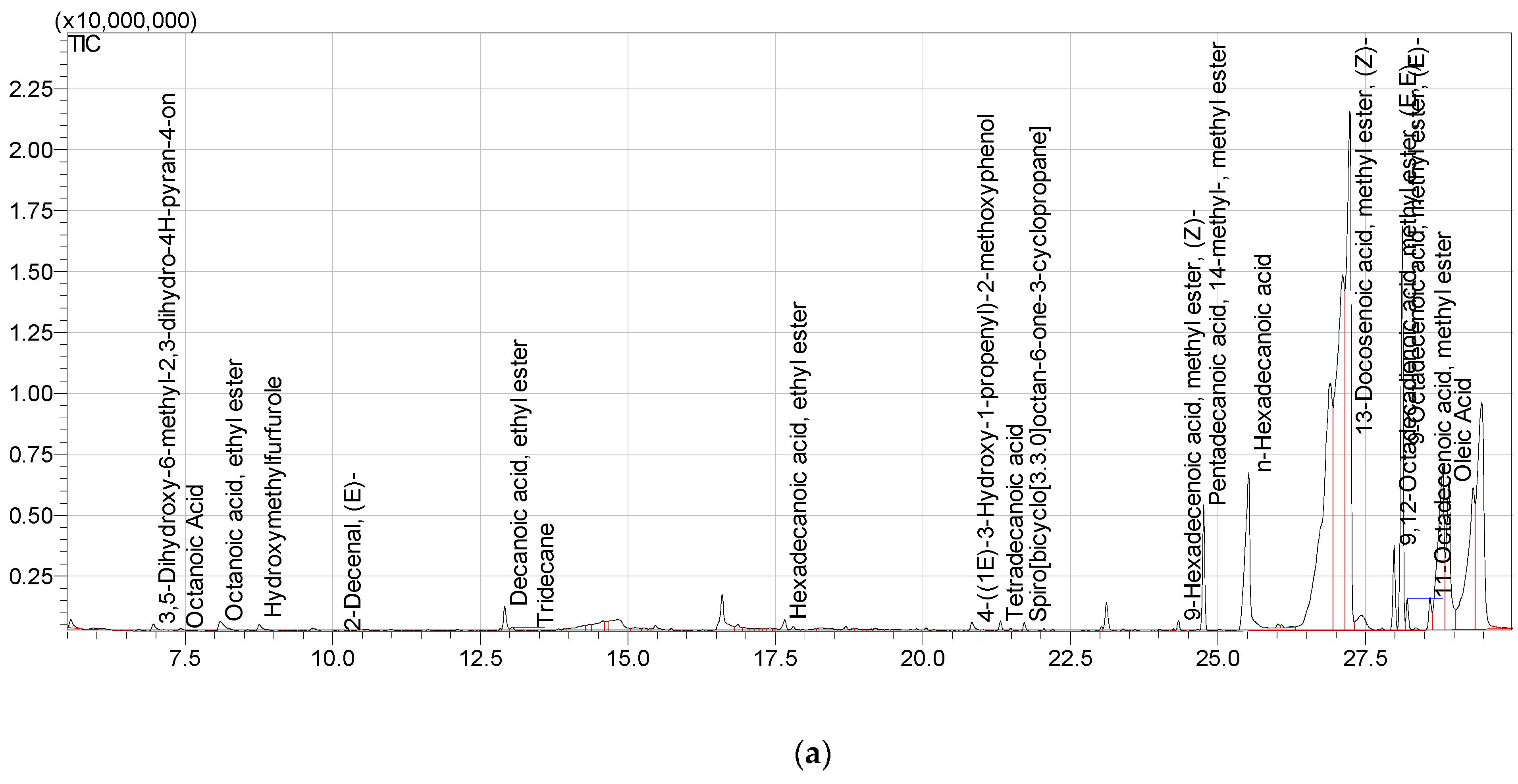
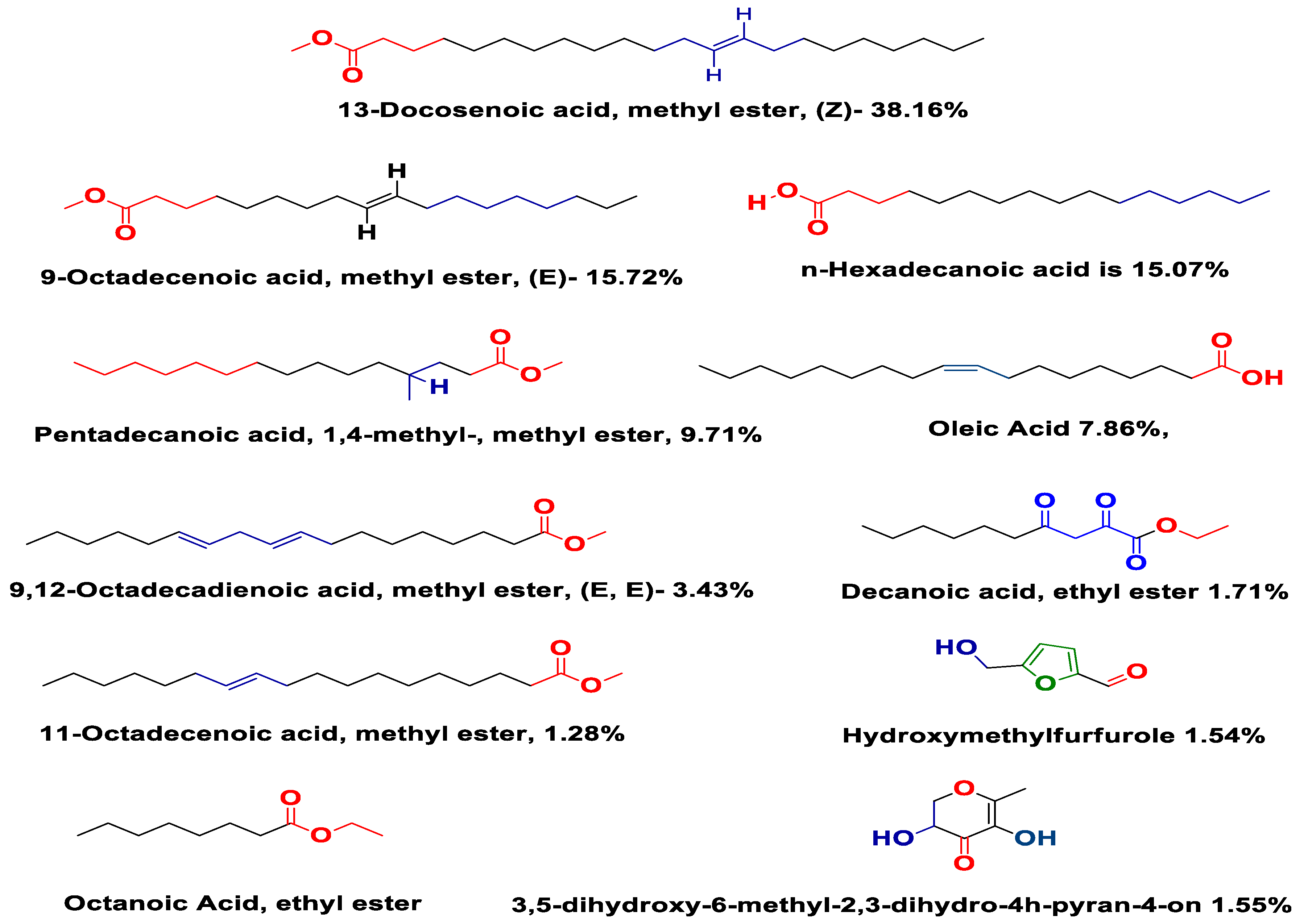
Carduus edelbergii is full of useful constituents. Among the esters, 13-Docosenoic acid, methyl ester, (Z)—(fatty acid methyl ester) has the highest concentration (38.16%), followed by 9-Octadecenoic acid, methyl ester, (E)—(15.72%) in the mass of methanol extract. On the other hand, n-Hexadecanoic acid is 15.07% in mass. The Pentadecanoic acid, 1,4-methyl-, methyl ester; Oleic Acid; and 9,12-Octadecadienoic acid, methyl ester, (E, E)—constitute 9.71%, 7.86% and 3.43% of mass, respectively. Moreover, the Decanoic acid, ethyl ester (1.72%); 11-Octadecenoic acid, methyl ester (1.28%); 3,5-Dihydroxy-6-methyl-2,3-dihudro-4H-pyran-4-on (1.55%); Octanoic Acid, ethyl ester (1.05%); and Hydroxymethylfurfurale (1.54%) were found in the sample. Congruent results were shown by
Carduus pycnocephalus which exhibited hexadecanoic acid (39.62%), Pentadecanoic acid methyl ester (0.43%), 9-octadecenoic acid methyl ester (1.44%) [
14].
Ali, R. et al., reported 13-docosenoic acid with a concentration of (34.06%) in
Acacia nilotica [
15]. All the above-given references mentioned the biological importance of identified compounds.
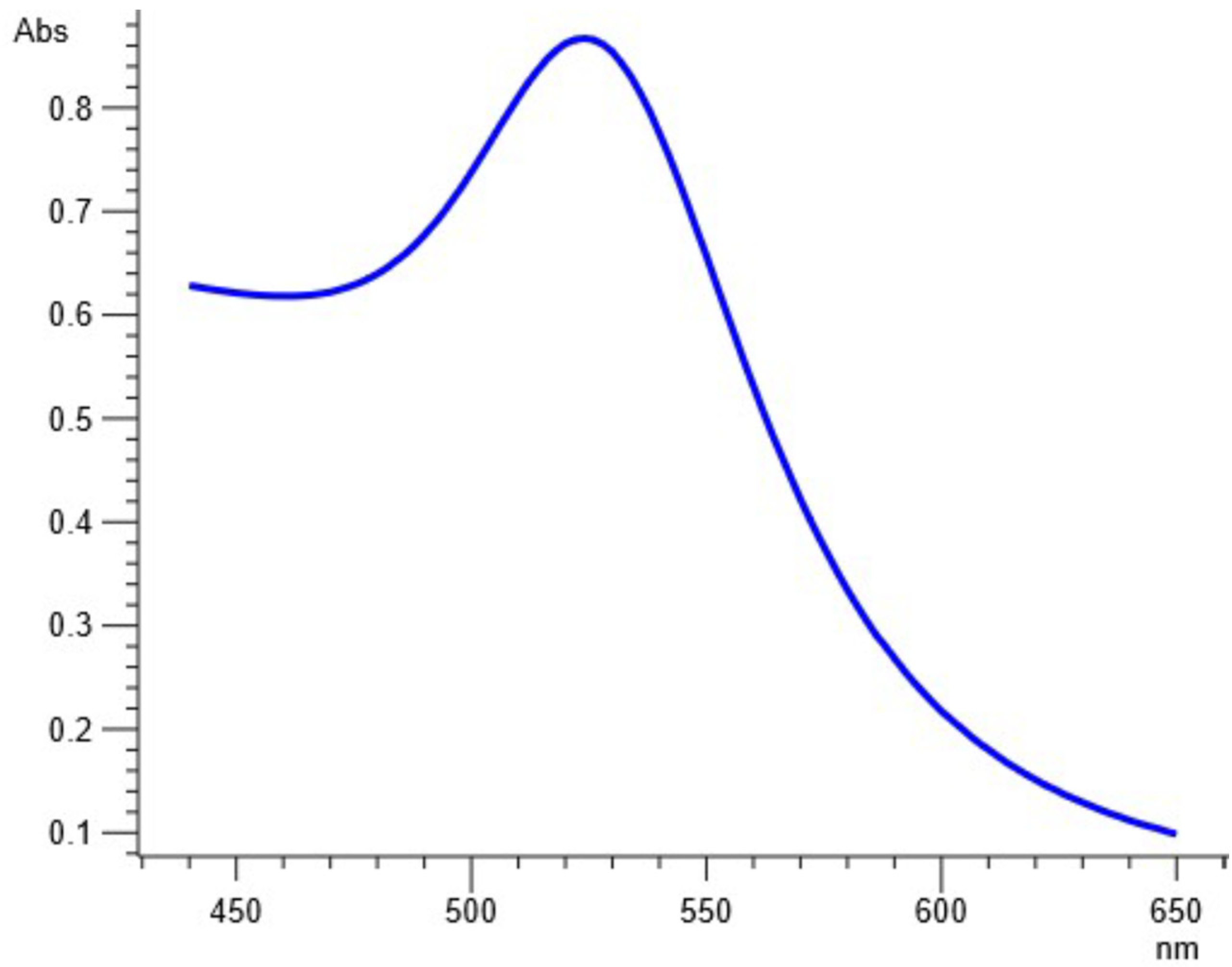
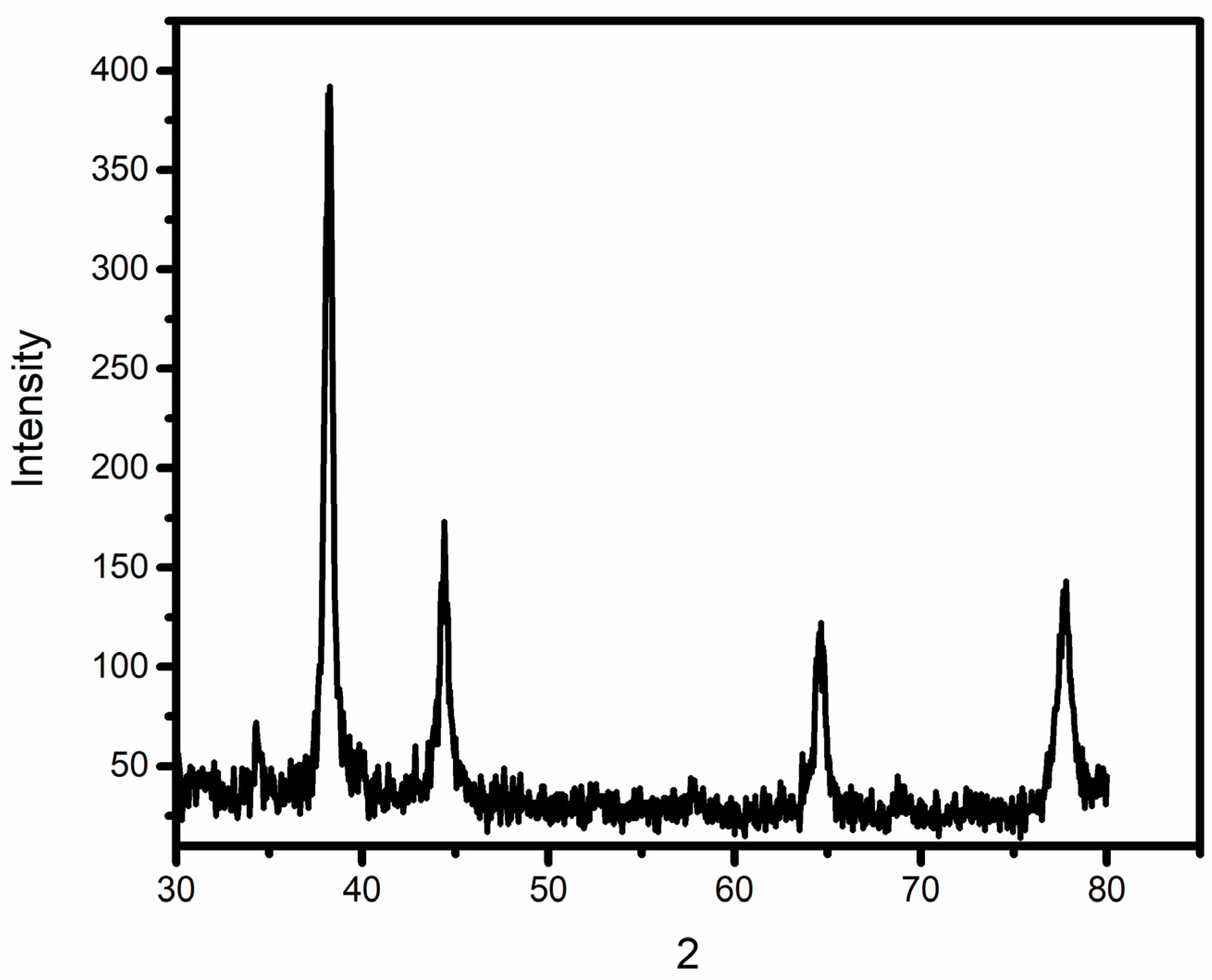
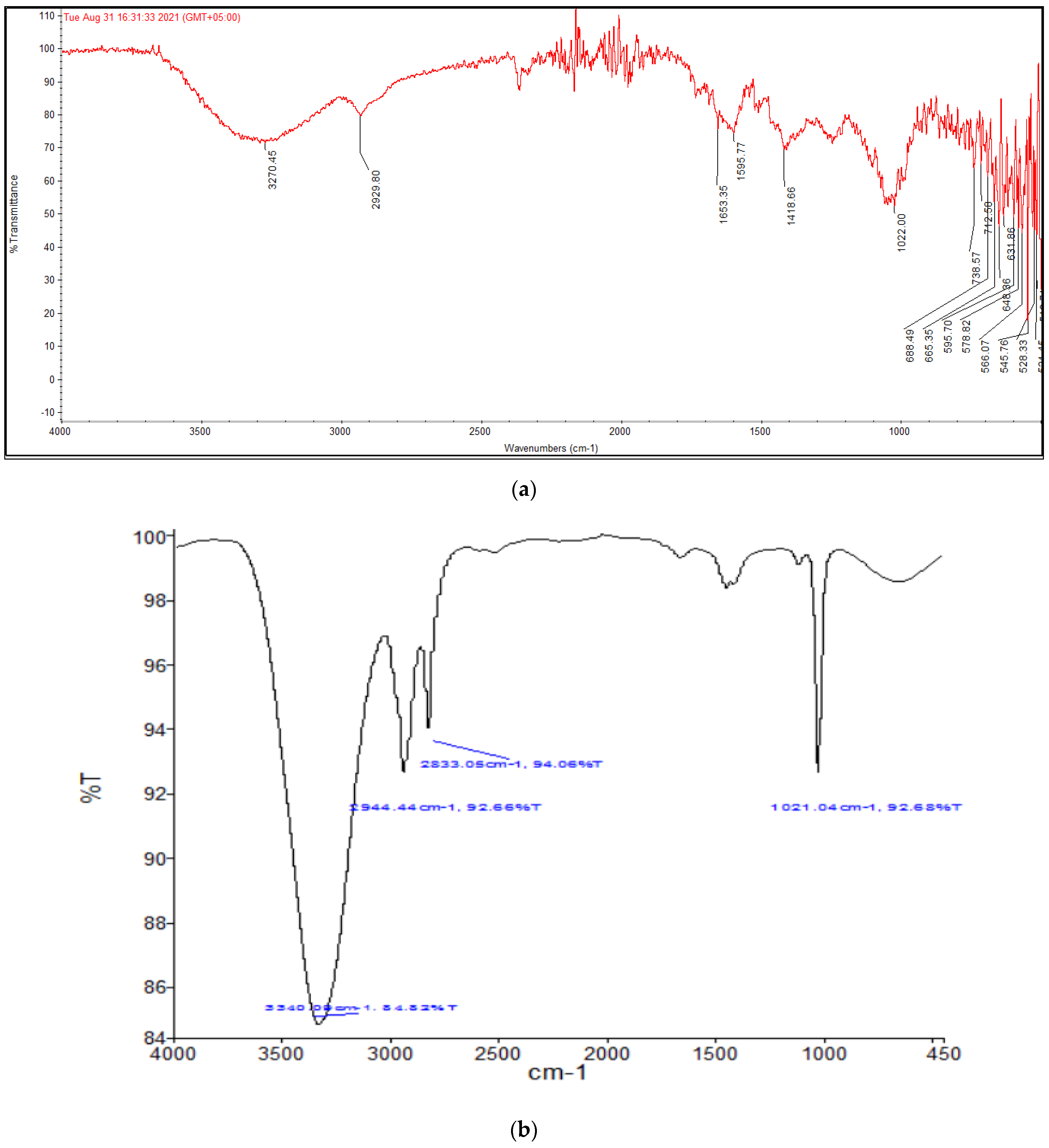
As soon as NPs are synthesized, it is critical to describe them to obtain a good picture of their physical and chemical properties. Since various factors such as the shape, size, crystallinity and morphological features of NPs influences their biological potential, it is important to have an understanding of these features and their effect on their biological potential. Various characterization techniques such as UV-vis spectrophotometry, XRD, FTIR, SEM and EDX were utilized to characterize extract-mediated AuNPs. UV-vis spectrophotometric analysis confirmed the presence of AuNPs in the crude sample, which was then isolated through centrifugation for further characterization. XRD analysis was used to determine the crystallinity and particle size. These peaks obtained in the XRD image correspond to 111, 200, 220 and 311 planes of face-centered cubic (fcc) crystal structure. The results proved that extract-mediated AuNPs were crystalline in nature, with a crystal size of approximately 15.6 nm. The XRD results of AuNPs are in concurrence with earlier studies on
Carduus crispus. From the FTIR spectral analysis, it is evident that the functional groups detected in
Carduus edelbergii extract were accountable for the capping and stabilization of AuNPs since the peaks in AuNPs samples are almost at the same position as in the plant extract sample. A small change in the position of the peaks and the intensity of the peaks may be attributed to the reaction of plant phytochemicals with the precursor salt to produce AuNPs. Relatively few peaks in AuNPs sample as compared to plant extract means that most of the phytochemicals exist in the plant extract remained unbound and did not play a part in the capping of AuNPs. The results of our study are similar to other studies [
16].
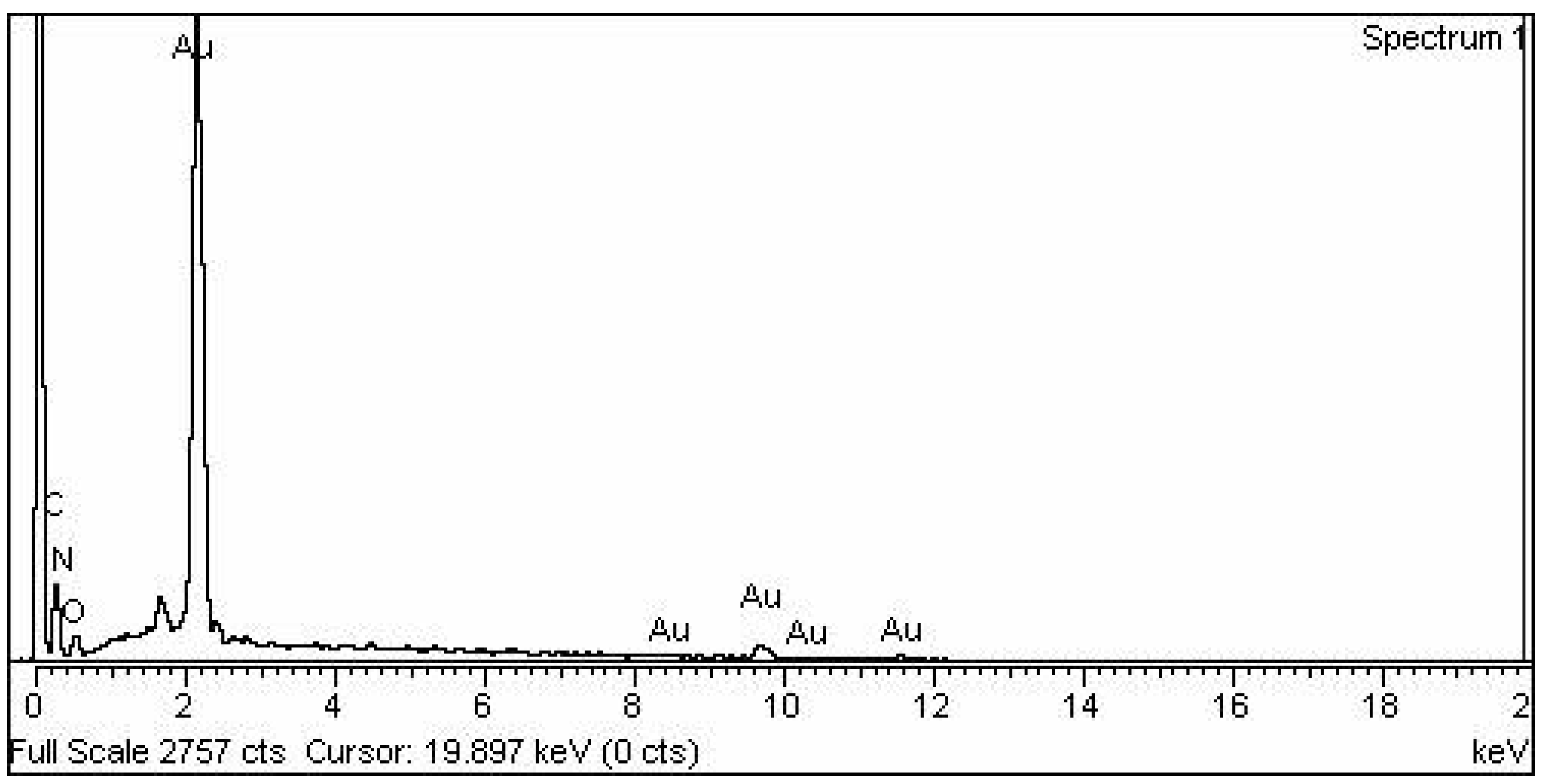
SEM and EDX analysis also gave useful insights regarding the morphology and purity of the extract-mediated AuNPs. The AuNPs obtained were morphologically spherical. A high degree of agglomeration can be seen in the XRD image, which is quite understandable considering the involvement of various phytochemicals in the capping of synthesized AuNPs. EDX elemental analysis revealed a strong peak of elemental gold, which indicates the presence of gold in the sample. The use of plant extract as a capping and reducing agent may have caused other impurities such as carbon and nitrogen.
The higher potential for improving health care that nanosized molecules have than those already found in nature could make the discovery and manipulation of new desirable molecules [
17]. In the last few years, several pharmaceutical companies have received FDA approval for the usage and development of medications based on nanotechnology. The capacity of gold compounds to decrease NF-kappa B expression and associated inflammatory reactions has garnered considerable interest as an anti-inflammatory drug [
18]. Animal studies in cerebrally ischemic rats have shown that Swarna Bhasma contains immunomodulatory, antioxidant, and restorative properties that could be useful in treating ischemia and other damage to the central nervous system [
19]. Because ionic gold is easily complexed and precipitated, it has a limited ability to perform its intended tasks in the human body. Zerovalent gold nanoparticles can be an excellent substitute for metallic gold in these situations. One-step green production of nanoparticles from metal ions can be achieved using biomolecules found in plant extracts. At room temperature and pressure, the biogenic reduction in a metal ion to a base metal can be easily scaled up. Thus, the green-produced gold nanoparticles have both zerovalent gold and a bioactive component, giving superior curative effects for the treatment of cancer. Owing to its biocompatibility and strong surface reactivity, gold nanoparticles (AuNPs), a developing nanomedicine, are known for their promising therapeutic capabilities [
20]. VPF/VEGF165-induced endothelial cell proliferation is inhibited by gold nanoparticles, providing compelling evidence that they can be used as therapeutic agents for a wide range of conditions, including rheumatoid arthritis and cancer. In light of the evidence that traditional gold has an antioxidative effect in the treatment of various inflammatory diseases and other related disorders that are context-dependent, the need for further research into the restorative effect of gold nanoparticles in conditions of hyperglycemia has been reported in previous studies [
21].
The antimicrobial potency of nanoparticles is very much successful and largely applied. In the current study, AuNPs proved to be most effective against
K. pneumoneae and S. aureus by showing 12.3 ± 0.3 mm and 12.5 ± 0.6 zone of inhibition against both the aforementioned bacterial strains, respectively. AuNPs indicated brilliant antibacterial potency against
S. epidermis,
E. coli and
Acetobacter spp. 9.3 ± 0.4, 10.7 ± 0.6 and 11.2 ± 0.6 mm each, respectively.
Aspergillus flavus and
Aspergillus niger were inhibited by AuNPs at 92.4 ± 4.3 and 70.8 ± 5.5 percent, respectively, compared to terbinafine 97.5 ± 2.5 and 98.1 ± 1.9 percent inhibition for both fungal strains. The antibacterial characteristics of AuNPs were also checked with their MICs and found that the sample with the lowest MIC had the highest inhibition zone. Congruent findings have been recorded in earlier studies [
22].
Alteration in carbohydrate metabolism may cause serious health problems such as diabetes and obesity, where a decline in function or secretion of insulin leads to an increase in blood glucose level and hence the ultimate onset of diabetes mellitus [
23]. The inhibition of alpha-amylase causes a delay in the digestion of carbohydrates and, thus, a decline in glucose uptake by the intestines. In order to determine the antidiabetic potential of plant extract, the in vitro alpha-amylase inhibitory assay was opted for. The anti-diabetic characteristics of AuNPs (63.7 ± 5.1) were higher than extract (60.8 ± 3.8) and were comparable with Glucophage (65.1 ± 3.2). This predicts that the plant extract is an effective source of antidiabetic phytochemicals. The synthesis of extract fabricated AuNPs showed improved antidiabetic activities. Analogous results were found during in vitro studies of
Carduus lanuginosus [
13] and
Carduus marianum [
24] extract. In addition, plants protect themselves with the help of their indigenous alpha-amylase inhibitors. The mentioned inhibitors alter the digestive capability of proteinases and alpha-amylases in the gut of insects and decrease their normal feeding activities [
25]. The greater alpha-amylase inhibitory potential was revealed by AuNPs and methanol extract; they were subjected to in vivo investigation and obtained desirable results. The antioxidant, antihyperglycemic and hypolipidemic properties of phytochemical compounds were reported [
26]. Enzymatic functions can also be restored by curing pancreatic islets and revamping organs such as the liver and kidneys. Diabetes-induced alterations in diabetic rats were largely reversed by
Carduus edelbergii methanol extract. The
Silybum marianum extract prevents diabetic rats from losing weight by restoring glucose levels, enzymes and tissue damage [
24].
Carduus acanthoides anti-diabetic investigations provided similar results in the past [
27]. Aminotransferase levels tend to rise in patients with diabetes and are more common than in the general population [
28]. After the introduction of alloxan in prior research, there was a large rise in the activity of specific enzymes, such as Beta-Glucuronidase, Nacetyl-bet Glucosaminidase, Leucine Aminpeptidase, Lysosomal Acid Phosphates, and Cathepsin D [
29]. Diabetes-related alterations in metabolism are closely linked to serum enzyme attenuation in animals. As a result of the increasing availability of amino acids in diabetes, transaminase activity rises, resulting in an increased rate of ketogenesis and gluconeogenesis. As a result, the better metabolism of carbohydrates, lipids and proteins led to a recovery in enzyme levels. After treatment, bilirubin and ALT levels improved, indicating that insulin secretion was restored. In previous studies, the plant extract showed restorative properties of ALT, ALP, total bilirubin and AST [
30]. As a result of diabetes, a raise in serum creatinine and urea was seen, which were considered major indicators of renal impairment [
31]. A result from anti-diabetic research of
Carduus acicularis and
Cirsium maackii was consistent with our results [
32]. Rats suffering from alloxan-induced diabetes were shown to have hypercholesterolemia, which can be attributed to a drop in fat-catalytic hormones and a reduction in the breakdown of fat deposits [
33]. There may be an association between the lipid-lowering impact and the suppression of the formation of fatty acids. Lipoprotein lipase, which hydrolyzes triglycerides, is activated by insulin during metabolism, but insulin shortage causes insulin to be inactivated, resulting in hypertriglyceridemia [
34]. A considerable decline in serum lipid concentrations in diabetic rats subsequent to treatment with methanol extract can be directly linked with an enhancement in insulin concentrations. Comparable findings were acquired in an antidiabetic study of the aerial parts of
Iphiona aucheri extracts [
25].