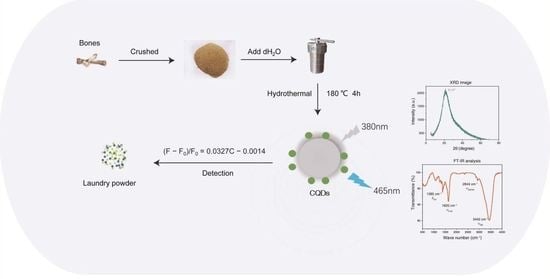
A Hydrothermal Method to Generate Carbon Quantum Dots from Waste Bones and Their Detection of Laundry Powder
Abstract
1. Introduction
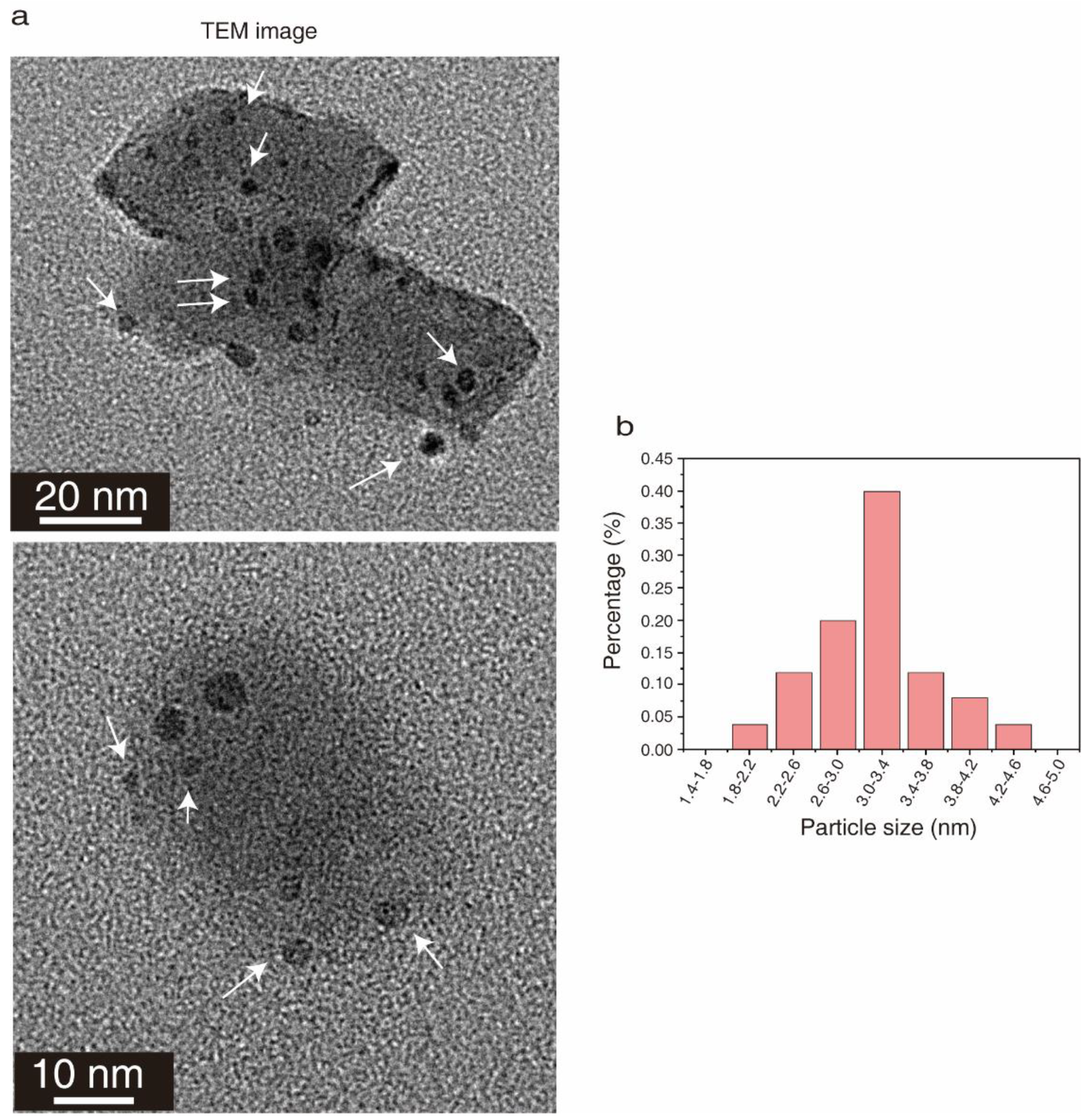
2. Results
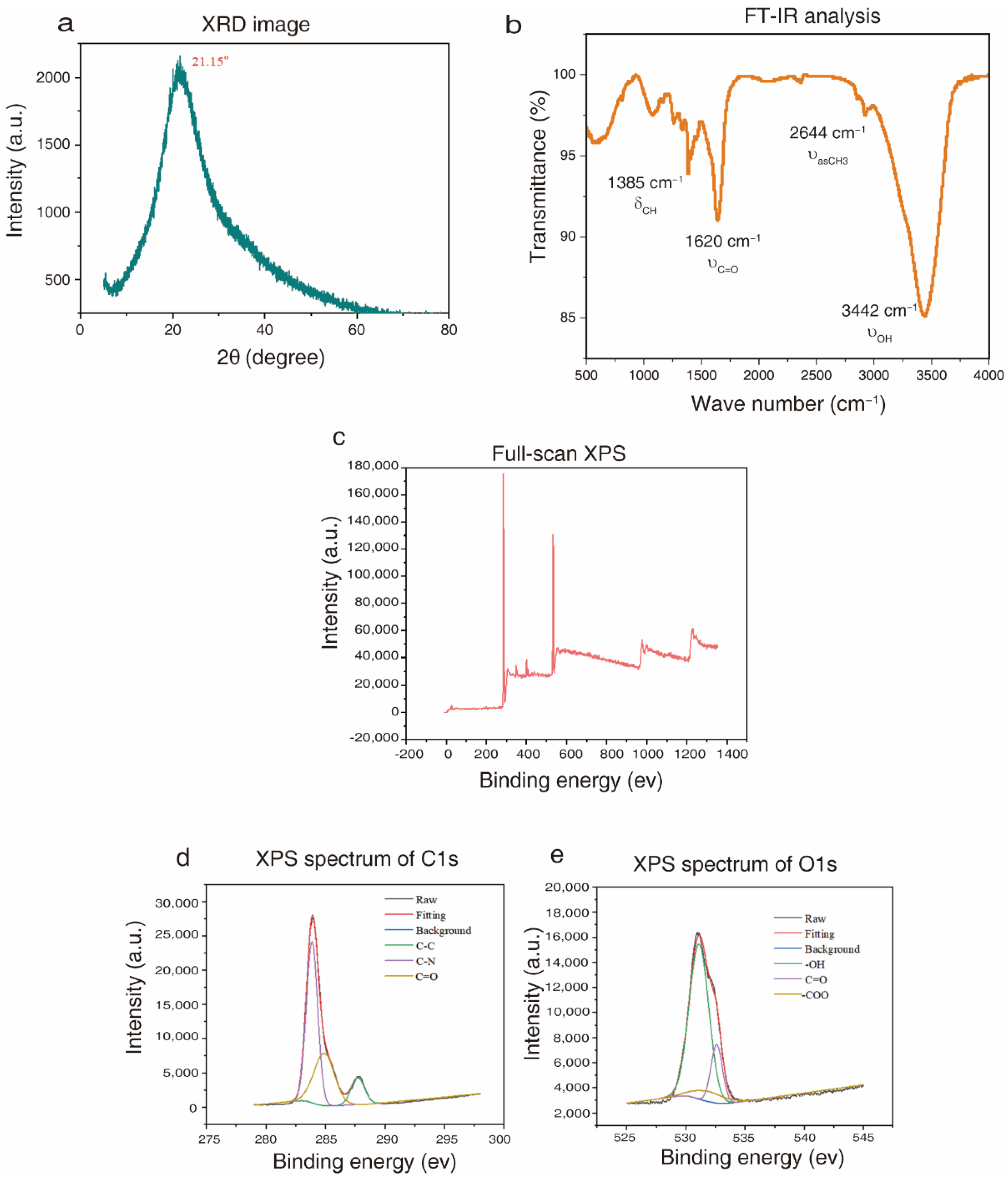
2.1. Functional Groups of the Prepared CQDs
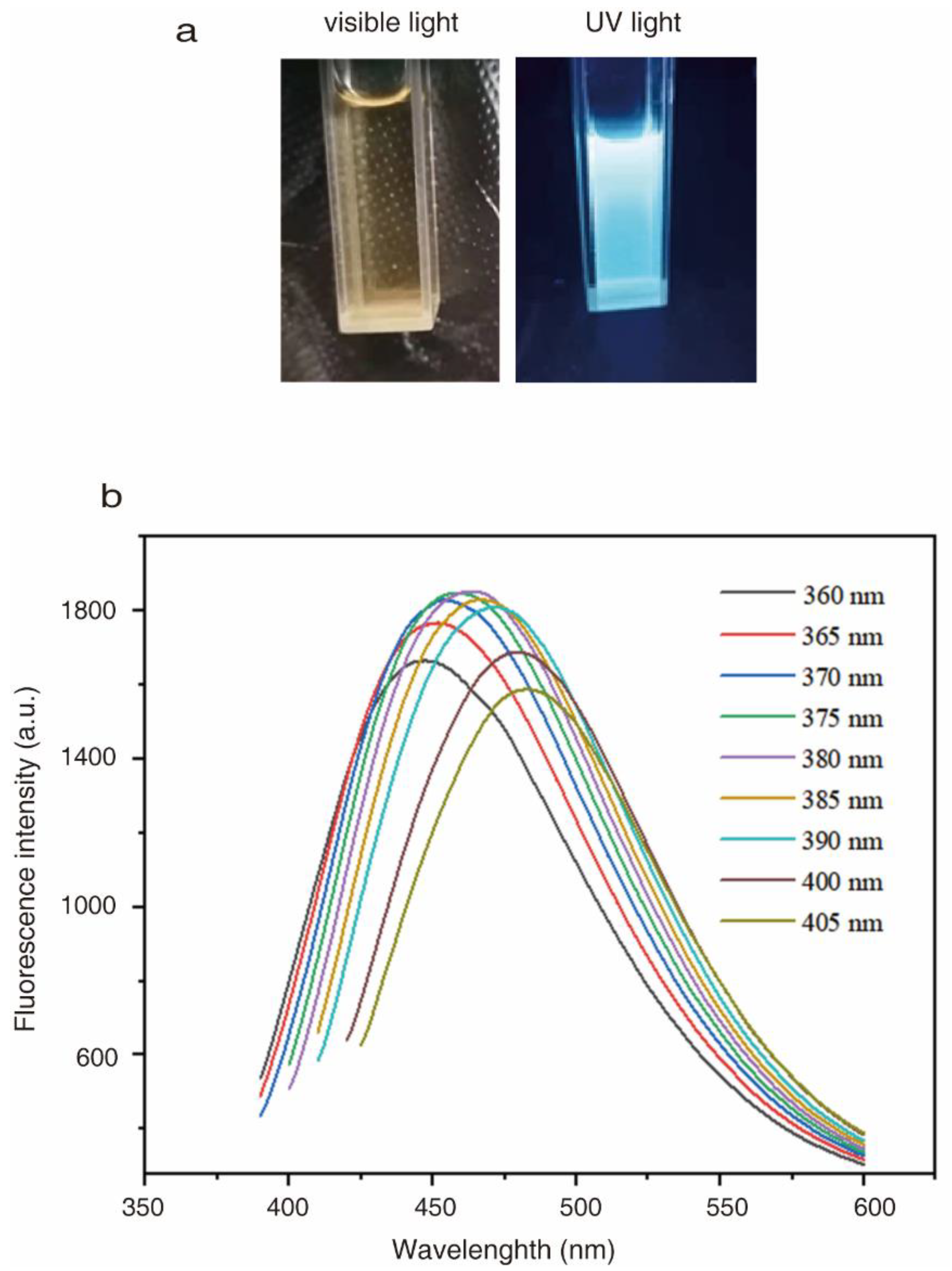
2.2. Optical Properties of CQDs
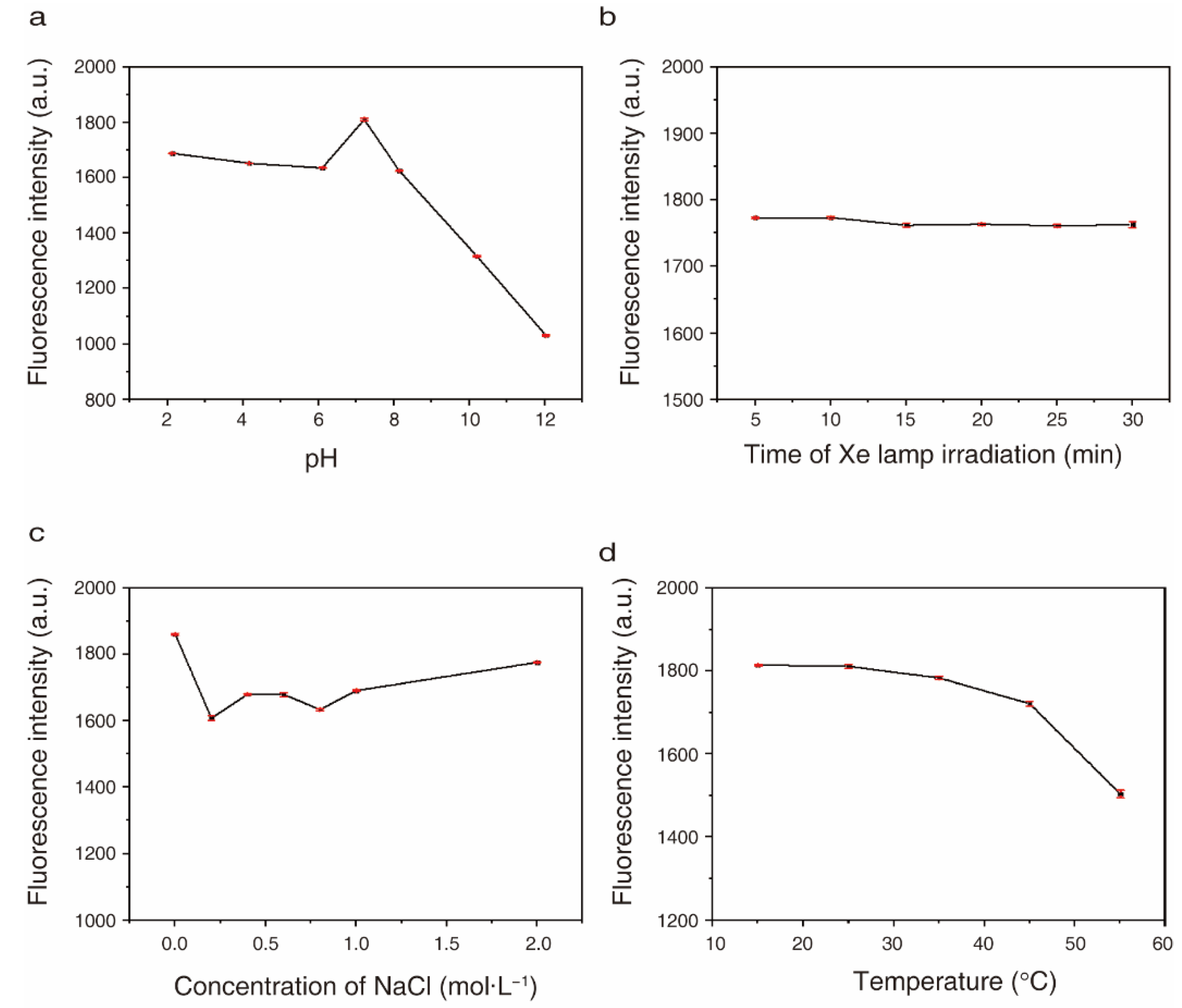
2.3. Optical Stabilities of CQDs under Different pH, Salt Concentration, Light and Temperature Conditions
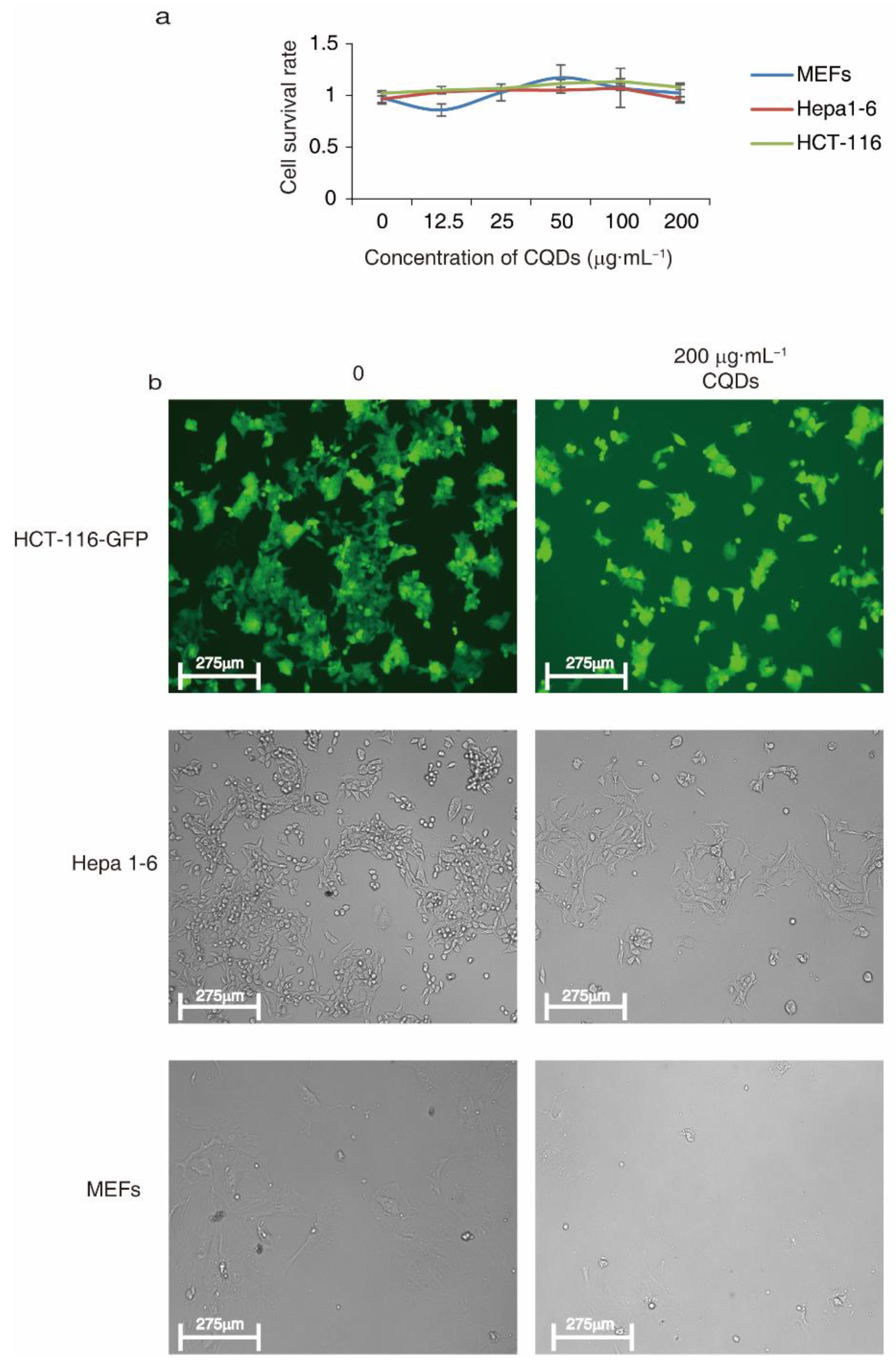
2.4. CQDs Exert Low Cytotoxicity
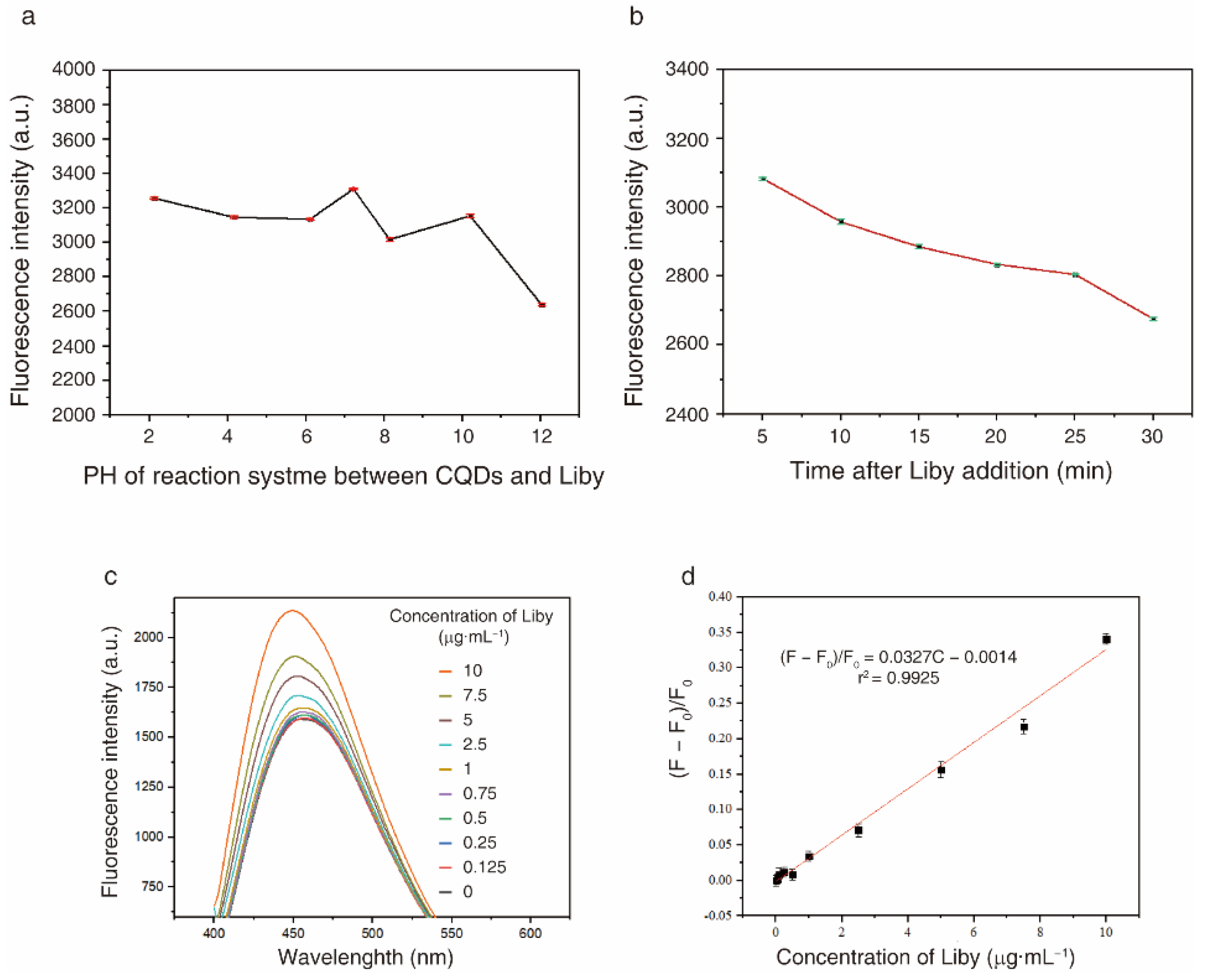
2.5. Laundry Detergents Enhance the Fluorescence Intensity of CQDs
2.6. CQDs Can Be Used as a Fluorescence Probe to Detect Laundry Powder
3. Discussion and Conclusions
4. Materials and Methods
4.1. Instruments and Materials
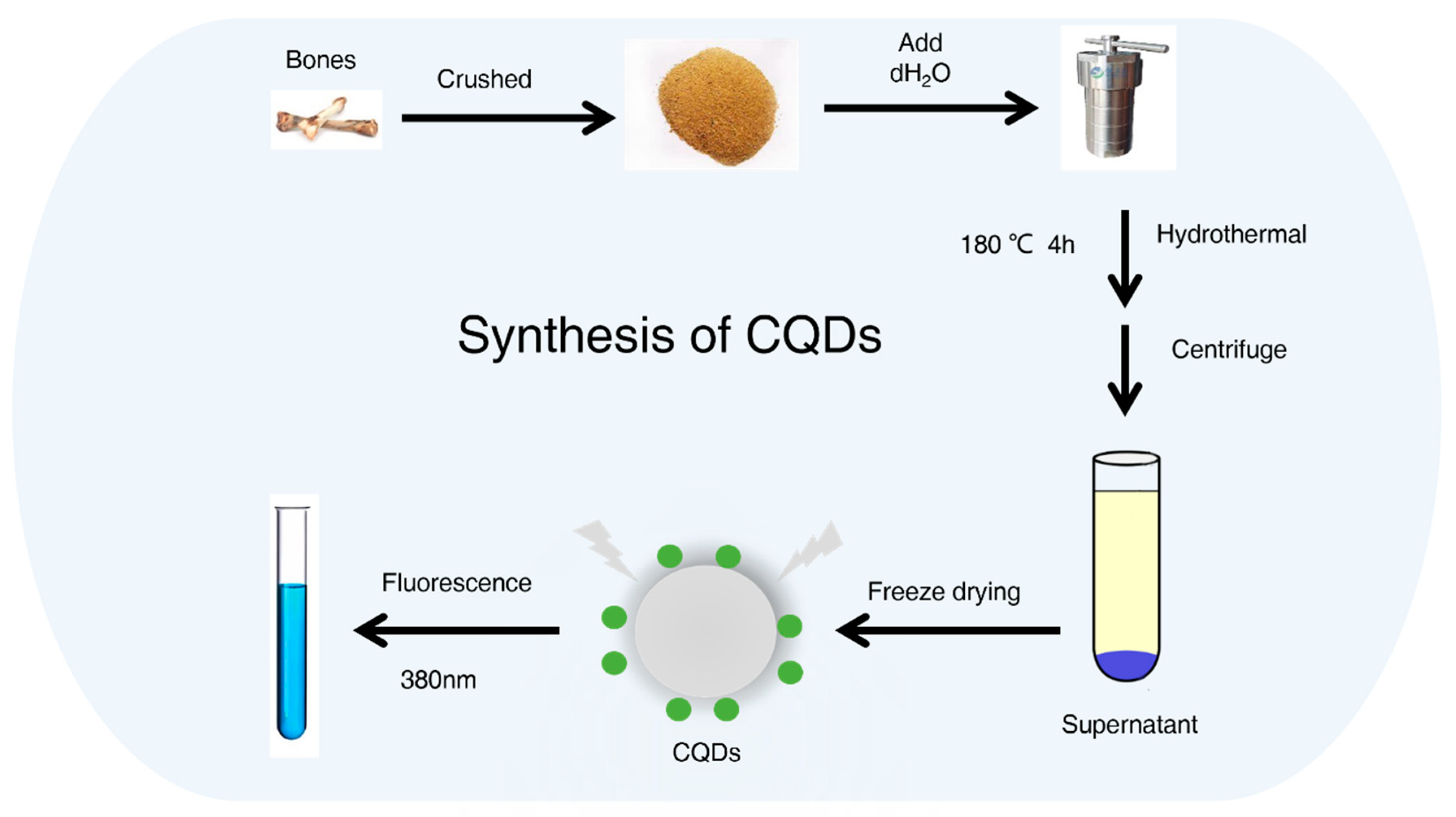
4.2. CQDs Synthesis
4.3. Fluorescence Intensity Measurement by Fluorescence Spectrophotometer
4.4. Lentivirus Preparation
4.5. Flow Cytometry
4.6. Cell Culture
4.7. Cytotoxicity Test
4.8. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Yu, Y.; Zhao, J.; Bayly, A.E. Development of Surfactants and Builders in Detergent Formulations. Chin. J. Chem. Eng. 2008, 16, 517–527. [Google Scholar] [CrossRef]
- Wang, M.; Tan, G.; Eljaszewicz, A.; Meng, Y.; Wawrzyniak, P.; Acharya, S.; Altunbulakli, C.; Westermann, P.; Dreher, A.; Yan, L.; et al. Laundry Detergents and Detergent Residue after Rinsing Directly Disrupt Tight Junction Barrier Integrity in Human Bronchial Epithelial Cells. J. Allergy Clin. Immunol. 2019, 143, 1892–1903. [Google Scholar] [CrossRef] [PubMed]
- Sobrino-Figueroa, A. Toxic Effect of Commercial Detergents on Organisms from Different Trophic Levels. Env. Sci. Pollut Res Int 2018, 25, 13283–13291. [Google Scholar] [CrossRef] [PubMed]
- Gaubert, A.; Jeudy, J.; Rougemont, B.; Bordes, C.; Lemoine, J.; Casabianca, H.; Salvador, A. Identification and Absolute Quantification of Enzymes in Laundry Detergents by Liquid Chromatography Tandem Mass Spectrometry. Anal. Bioanal. Chem. 2016, 408, 4669–4681. [Google Scholar] [CrossRef]
- Hansen, S.H.; Damhus, T.; Brask, J. Rapid and Simple Identification and Quantification of Components in Detergent Formulations by Nuclear Magnetic Resonance Spectroscopy. J. Surfactants Deterg. 2020, 23, 415–422. [Google Scholar] [CrossRef]
- Chryssou, K.; Stassinopoulou, M.; Lampi, E. Determination of Three Detergent Products and Identification of Surface Active Agents by Infrared Absorption Spectra and a Reflectance Graph. Chall. Adv. Chem. Sci. 2022, 8, 98–111. [Google Scholar] [CrossRef]
- Cui, L.; Puerto, M.; López-Salinas, J.L.; Biswal, S.L.; Hirasaki, G.J. Improved Methylene Blue Two-Phase Titration Method for Determining Cationic Surfactant Concentration in High-Salinity Brine. Anal. Chem. 2014, 86, 11055–11061. [Google Scholar] [CrossRef]
- Zheng, X.T.; Ananthanarayanan, A.; Luo, K.Q.; Chen, P. Glowing Graphene Quantum Dots and Carbon Dots: Properties, Syntheses, and Biological Applications. Small 2015, 11, 1620–1636. [Google Scholar] [CrossRef]
- Peng, Z.; Han, X.; Li, S.; Al-Youbi, A.O.; Bashammakh, A.S.; El-Shahawi, M.S.; Leblanc, R.M. Carbon Dots: Biomacromolecule Interaction, Bioimaging and Nanomedicine. Coord. Chem. Rev. 2017, 343, 256–277. [Google Scholar] [CrossRef]
- Sarkar, N.; Sahoo, G.; Das, R.; Prusty, G.; Swain, S.K. Carbon Quantum Dot Tailored Calcium Alginate Hydrogel for PH Responsive Controlled Delivery of Vancomycin. Eur. J. Pharm. Sci. 2017, 109, 359–371. [Google Scholar] [CrossRef]
- Campuzano, S.; Yáñez-Sedeño, P.; Pingarrón, J.M. Carbon Dots and Graphene Quantum Dots in Electrochemical Biosensing. Nanomaterials 2019, 9, 634. [Google Scholar] [CrossRef] [PubMed]
- Azam, N.; Najabat Ali, M.; Javaid Khan, T. Carbon Quantum Dots for Biomedical Applications: Review and Analysis. Front. Mater. 2021, 8, 1–21. [Google Scholar] [CrossRef]
- Ali, H.; Ghosh, S.; Jana, N.R. Fluorescent Carbon Dots as Intracellular Imaging Probes. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2020, 12, e1617. [Google Scholar] [CrossRef]
- Senthamizhan, A.; Fragouli, D.; Balusamy, B.; Patil, B.; Palei, M.; Sabella, S.; Uyar, T.; Athanassiou, A. Hydrochromic Carbon Dots as Smart Sensors for Water Sensing in Organic Solvents. Nanoscale Adv. 2019, 1, 4258–4267. [Google Scholar] [CrossRef] [PubMed]
- Rani, U.A.; Ng, L.Y.; Ng, C.Y.; Mahmoudi, E. A Review of Carbon Quantum Dots and Their Applications in Wastewater Treatment. Adv. Colloid Interface Sci. 2020, 278, 102124. [Google Scholar] [CrossRef] [PubMed]
- Yoo, D.; Park, Y.; Cheon, B.; Park, M.-H. Carbon Dots as an Effective Fluorescent Sensing Platform for Metal Ion Detection. Nanoscale Res. Lett. 2019, 14, 272. [Google Scholar] [CrossRef] [PubMed]
- Koç, Ö.K.; Üzer, A.; Apak, R. High Quantum Yield Nitrogen-Doped Carbon Quantum Dot-Based Fluorescent Probes for Selective Sensing of 2,4,6-Trinitrotoluene. ACS Appl. Nano Mater. 2022, 5, 5868–5881. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, Q.; Yuan, Y.; Wu, Y. Hydrothermal Synthesis of Fluorescent Carbon Dots from Sodium Citrate and Polyacrylamide and Their Highly Selective Detection of Lead and Pyrophosphate. Carbon 2017, 115, 550–560. [Google Scholar] [CrossRef]
- Yuan, X.; Zhao, H.; Bai, F.; Zhao, P.; Zhao, L.; Xiong, Z. Fluorescence and Scattering Based Dual-Optical Signals Ratiometric Sensing and Logic Gate Device for Acetylcholinesterase Activity Assay. Microchem. J. 2021, 170, 106768. [Google Scholar] [CrossRef]
- Yuan, X.; Sun, Y.; Zhao, P.; Zhao, L.; Xiong, Z. Redox-Induced Target-Dependent Ratiometric Fluorescence Sensing Strategy and Logic Gate Operation for Detection of α-Glucosidase Activity and Its Inhibitor. Dalton Trans. 2021, 50, 9426–9437. [Google Scholar] [CrossRef]
- Wang, X.; Feng, Y.; Dong, P.; Huang, J. A Mini Review on Carbon Quantum Dots: Preparation, Properties, and Electrocatalytic Application. Front. Chem. 2019, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pei, S.; Zhang, J.; Gao, M.; Wu, D.; Yang, Y.; Liu, R. A Facile Hydrothermal Approach towards Photoluminescent Carbon Dots from Amino Acids. J. Colloid Interface Sci. 2015, 439, 129–133. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Chi, Y.; Dong, Y.; Lin, J.; Wang, B. Electrochemiluminescence of Water-Soluble Carbon Nanocrystals Released Electrochemically from Graphite. J. Am. Chem. Soc. 2009, 131, 4564–4565. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.; Thongsai, N.; Chae, A.; Jo, S.; Kang, E.B.; Paoprasert, P.; Park, S.Y.; In, I. Microwave-Assisted Synthesis of Luminescent and Biocompatible Lysine-Based Carbon Quantum Dots. J. Ind. Eng. Chem. 2017, 47, 329–335. [Google Scholar] [CrossRef]
- Wang, D.M.; Lin, K.L.; Huang, C.Z. Carbon Dots-Involved Chemiluminescence: Recent Advances and Developments. Luminescence 2019, 34, 4–22. [Google Scholar] [CrossRef] [PubMed]
- Hallaj, T.; Amjadi, M.; Manzoori, J.L.; Shokri, R. Chemiluminescence Reaction of Glucose-Derived Graphene Quantum Dots with Hypochlorite, and Its Application to the Determination of Free Chlorine. Microchim. Acta 2015, 182, 789–796. [Google Scholar] [CrossRef]
- He, M.; Zhang, J.; Wang, H.; Kong, Y.; Xiao, Y.; Xu, W. Material and Optical Properties of Fluorescent Carbon Quantum Dots Fabricated from Lemon Juice via Hydrothermal Reaction. Nanoscale Res. Lett. 2018, 13, 175. [Google Scholar] [CrossRef]
- Tyagi, A.; Tripathi, K.M.; Singh, N.; Choudhary, S.; Gupta, R.K. Green Synthesis of Carbon Quantum Dots from Lemon Peel Waste: Applications in Sensing and Photocatalysis. RSC Adv. 2016, 6, 72423–72432. [Google Scholar] [CrossRef]
- Wang, C.; Shi, H.; Yang, M.; Yan, Y.; Liu, E.; Ji, Z.; Fan, J. Facile Synthesis of Novel Carbon Quantum Dots from Biomass Waste for Highly Sensitive Detection of Iron Ions. Mater. Res. Bull. 2020, 124, 110730. [Google Scholar] [CrossRef]
- Ding, S.; Gao, Y.; Ni, B.; Yang, X. Green Synthesis of Biomass-Derived Carbon Quantum Dots as Fluorescent Probe for Fe3+ Detection. Inorg. Chem. Commun. 2021, 130, 108636. [Google Scholar] [CrossRef]
- Clark, M.; Tilman, D. Comparative Analysis of Environmental Impacts of Agricultural Production Systems, Agricultural Input Efficiency, and Food Choice. Environ. Res. Lett. 2017, 12, 064016. [Google Scholar] [CrossRef]
- Oyejide, A.J.; Adetola, S.O.; Lawal, A. Machine Design Approach to Bone Waste Utilization in Slaughterhouses of Developing Countries with Focus on Nigeria. World J. Eng. Technol. 2022, 10, 444–457. [Google Scholar] [CrossRef]
- Dong, Y.; Shao, J.; Chen, C.; Li, H.; Wang, R.; Chi, Y.; Lin, X.; Chen, G. Blue Luminescent Graphene Quantum Dots and Graphene Oxide Prepared by Tuning the Carbonization Degree of Citric Acid. Carbon 2012, 50, 4738–4743. [Google Scholar] [CrossRef]
- Zhu, S.; Tang, S.; Zhang, J.; Yang, B. Control the Size and Surface Chemistry of Graphene for the Rising Fluorescent Materials. Chem. Commun. 2012, 48, 4527–4539. [Google Scholar] [CrossRef]
- Han, S.; Zhang, H.; Zhang, J.; Xie, Y.; Liu, L.; Wang, H.; Li, X.; Liu, W.; Tang, Y. Fabrication, Gradient Extraction and Surface Polarity-Dependent Photoluminescence of Cow Milk-Derived Carbon Dots. RSC Adv. 2014, 4, 58084–58089. [Google Scholar] [CrossRef]
- Salvatore, M.; Carotenuto, G.; De Nicola, S.; Camerlingo, C.; Ambrogi, V.; Carfagna, C. Synthesis and Characterization of Highly Intercalated Graphite Bisulfate. Nanoscale Res. Lett. 2017, 12, 167. [Google Scholar] [CrossRef]
- Li, M.-K.; Hu, L.-Y.; Niu, C.-G.; Huang, D.-W.; Zeng, G.-M. A Magnetic Separation Fluorescent Aptasensor for Highly Sensitive Detection of Bisphenol A. Sens. Actuators B Chem. 2018, 266, 805–811. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, Y.; Pang, G.; Zhang, Y.; Guo, S. Tuning the Aggregation/Disaggregation Behavior of Graphene Quantum Dots by Structure-Switching Aptamer for High-Sensitivity Fluorescent Ochratoxin A Sensor. Anal. Chem. 2017, 89, 1704–1709. [Google Scholar] [CrossRef]
- Wei, Q.; Zhang, P.; Pu, H.; Sun, D.-W. A Fluorescence Aptasensor Based on Carbon Quantum Dots and Magnetic Fe3O4 Nanoparticles for Highly Sensitive Detection of 17β-Estradiol. Food Chem. 2022, 373, 131591. [Google Scholar] [CrossRef]
- Teymourinia, H.; Salavati-Niasari, M.; Amiri, O.; Safardoust-Hojaghan, H. Synthesis of Graphene Quantum Dots from Corn Powder and Their Application in Reduce Charge Recombination and Increase Free Charge Carriers. J. Mol. Liq. 2017, 242, 447–455. [Google Scholar] [CrossRef]
- Irmania, N.; Dehvari, K.; Gedda, G.; Tseng, P.-J.; Chang, J.-Y. Manganese-Doped Green Tea-Derived Carbon Quantum Dots as a Targeted Dual Imaging and Photodynamic Therapy Platform. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 1616–1625. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.-Y.; Gedda, G.; Girma, W.M.; Yen, C.-L.; Ling, Y.-C.; Chang, J.-Y. Magnetofluorescent Carbon Dots Derived from Crab Shell for Targeted Dual-Modality Bioimaging and Drug Delivery. ACS Appl Mater Interfaces 2017, 9, 13887–13899. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhuo, Y.; Zhu, S.; Luo, Y.; Feng, Y.; Dou, Y. Novel and Green Synthesis of High-Fluorescent Carbon Dots Originated from Honey for Sensing and Imaging. Biosens Bioelectron 2014, 60, 292–298. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Liu, S.; Zhang, C.; Fan, L.; Zhang, J.; Chen, Q.; Sun, Y.; He, L.; Wang, Z.; Zhang, K. Coal Based Carbon Dots: Recent Advances in Synthesis, Properties, and Applications. Nano Sel. 2021, 2, 1589–1604. [Google Scholar] [CrossRef]
- Lou, Y.; Hao, X.; Liao, L.; Zhang, K.; Chen, S.; Li, Z.; Ou, J.; Qin, A.; Li, Z. Recent Advances of Biomass Carbon Dots on Syntheses, Characterization, Luminescence Mechanism, and Sensing Applications. Nano Sel. 2021, 2, 1117–1145. [Google Scholar] [CrossRef]
- Lou, Y.; Ji, J.; Qin, A.; Liao, L.; Li, Z.; Chen, S.; Zhang, K.; Ou, J. Cane Molasses Graphene Quantum Dots Passivated by PEG Functionalization for Detection of Metal Ions. ACS Omega 2020, 5, 6763–6772. [Google Scholar] [CrossRef]
- Sarkar, K.; Devi, P.; Lata, A.; Ghosh, R.; Kumar, P. Engineering Carbon Quantum Dots for Enhancing the Broadband Photoresponse in a Silicon Process-Line Compatible Photodetector. J. Mater. Chem. C 2019, 7, 13182–13191. [Google Scholar] [CrossRef]
- Liu, Z.; Zou, H.; Wang, N.; Yang, T.; Peng, Z.; Wang, J.; Li, N.; Huang, C. Photoluminescence of Carbon Quantum Dots: Coarsely Adjusted by Quantum Confinement Effects and Finely by Surface Trap States. Sci. China Chem. 2018, 61, 490–496. [Google Scholar] [CrossRef]
- De, B.; Karak, N. A Green and Facile Approach for the Synthesis of Water Soluble Fluorescent Carbon Dots from Banana Juice. RSC Adv. 2013, 3, 8286–8290. [Google Scholar] [CrossRef]
- Šafranko, S.; Goman, D.; Stanković, A.; Medvidović-Kosanović, M.; Moslavac, T.; Jerković, I.; Jokić, S. An Overview of the Recent Developments in Carbon Quantum Dots—Promising Nanomaterials for Metal Ion Detection and (Bio)Molecule Sensing. Chemosensors 2021, 9, 138. [Google Scholar] [CrossRef]
- Kang, C.; Huang, Y.; Yang, H.; Yan, X.F.; Chen, Z.P. A Review of Carbon Dots Produced from Biomass Wastes. Nanomaterials 2020, 10, 2316. [Google Scholar] [CrossRef] [PubMed]
- Das, G.S.; Shim, J.P.; Bhatnagar, A.; Tripathi, K.M.; Kim, T. Biomass-Derived Carbon Quantum Dots for Visible-Light-Induced Photocatalysis and Label-Free Detection of Fe(III) and Ascorbic Acid. Sci. Rep. 2019, 9, 15084. [Google Scholar] [CrossRef] [PubMed]
- Anmei, S.; Qingmei, Z.; Yuye, C.; Yilin, W. Preparation of Carbon Quantum Dots from Cigarette Filters and Its Application for Fluorescence Detection of Sudan I. Anal. Chim. Acta 2018, 1023, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Ensafi, A.A.; Nasr-Esfahani, P.; Rezaei, B. Synthesis of Molecularly Imprinted Polymer on Carbon Quantum Dots as an Optical Sensor for Selective Fluorescent Determination of Promethazine Hydrochloride. Sens. Actuators B Chem. 2018, C, 889–896. [Google Scholar] [CrossRef]
- Si, Q.-S.; Guo, W.-Q.; Wang, H.-Z.; Liu, B.-H.; Ren, N.-Q. Carbon Quantum Dots-Based Semiconductor Preparation Methods, Applications and Mechanisms in Environmental Contamination. Chin. Chem. Lett. 2020, 31, 2556–2566. [Google Scholar] [CrossRef]
- Zheng, G.; Li, S.; Zhang, T.; Zhu, F.; Sun, J.; Li, S.; You, L. Water Pollution Control and Treatment Based on Quantum Dot Chemical and Biological High-Sensitivity Sensing. J. Sens. 2021, 2021, e8704363. [Google Scholar] [CrossRef]
- Yan, Y.; Kuang, W.; Shi, L.; Ye, X.; Yang, Y.; Xie, X.; Shi, Q.; Tan, S. Carbon Quantum Dot-Decorated TiO2 for Fast and Sustainable Antibacterial Properties under Visible-Light. J. Alloy Compd. 2019, 777, 234–243. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, H.; Wang, A.; Liu, Y.; Hou, H.; Hu, Q. Combined Effects of Co-Exposure to Formaldehyde and Acrolein Mixtures on Cytotoxicity and Genotoxicity in Vitro. Env. Sci. Pollut. Res. Int. 2018, 25, 25306–25314. [Google Scholar] [CrossRef]

| Component | MFI ± SD |
|---|---|
| blank | 1968.33 ± 2.31 |
| Liby | 3207 ± 10.0 |
| Mg2+ | 2050 ± 4.7 |
| Na+ | 2153 ± 3.0 |
| K+ | 2080 ± 4.6 |
| Ca2+ | 1905 ± 4.0 |
| Zn+ | 2062 ± 5.0 |
| Al3+ | 2079 ± 5.2 |
| Cu2+ | 1651.67 ± 2.08 |
| Vitamin c | 2048.33 ± 3.05 |
| Samples | Added (μg·mL−1) | Found (μg·mL−1) | Recovery Rate (%) | RSD (%) |
|---|---|---|---|---|
| Mineral water | 0.50 | 0.53 | 106.00 | 5.3 |
| 2.50 | 2.22 | 88.80 | 3.3 | |
| 7.50 | 6.54 | 87.20 | 1.7 | |
| Tap water | 0.50 | 0.47 | 94.00 | 4.49 |
| 2.50 | 2.46 | 98.40 | 1.3 | |
| 7.50 | 6.39 | 85.20 | 1.23 | |
| River water | 0.50 | 0.48 | 96.00 | 2.15 |
| 2.50 | 2.54 | 101.60 | 1.01 | |
| 7.50 | 7.16 | 95.47 | 3.45 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, H.; Liu, B.; Wang, J.; Zhou, C.; Xiong, Z.; Zhao, L. A Hydrothermal Method to Generate Carbon Quantum Dots from Waste Bones and Their Detection of Laundry Powder. Molecules 2022, 27, 6479. https://doi.org/10.3390/molecules27196479
Ye H, Liu B, Wang J, Zhou C, Xiong Z, Zhao L. A Hydrothermal Method to Generate Carbon Quantum Dots from Waste Bones and Their Detection of Laundry Powder. Molecules. 2022; 27(19):6479. https://doi.org/10.3390/molecules27196479
Chicago/Turabian StyleYe, Heng, Binbin Liu, Jin Wang, Chunyu Zhou, Zhili Xiong, and Longshan Zhao. 2022. "A Hydrothermal Method to Generate Carbon Quantum Dots from Waste Bones and Their Detection of Laundry Powder" Molecules 27, no. 19: 6479. https://doi.org/10.3390/molecules27196479
APA StyleYe, H., Liu, B., Wang, J., Zhou, C., Xiong, Z., & Zhao, L. (2022). A Hydrothermal Method to Generate Carbon Quantum Dots from Waste Bones and Their Detection of Laundry Powder. Molecules, 27(19), 6479. https://doi.org/10.3390/molecules27196479