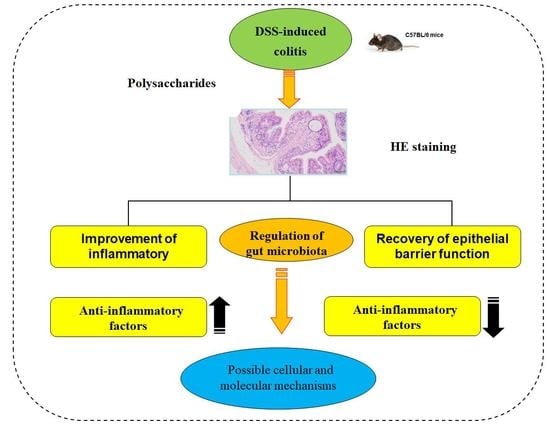
A Potential Role of Plant/Macrofungi/Algae-Derived Non-Starch Polysaccharide in Colitis Curing: Review of Possible Mechanisms of Action
Abstract
1. Introduction
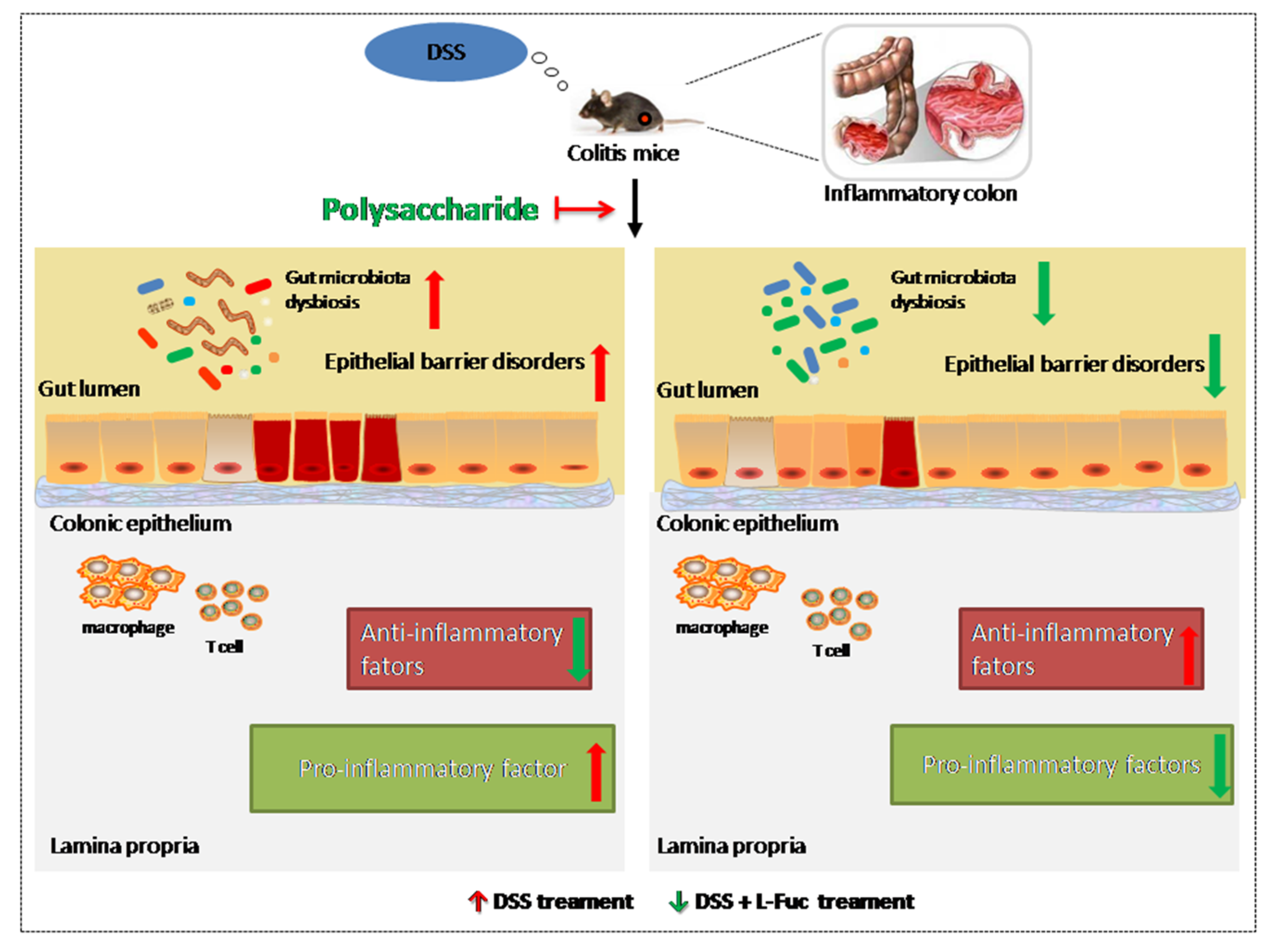
2. Improvement of Inflammatory
3. Recovery of Epithelial Barrier Function
4. Regulation of Gut Microbiota
5. Conclusion and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Orholm, M.; Munkholm, P.; Langholz, E.; Nielsen, O.H.; Sørensen, T.I.; Binder, V. Familial occurrence of inflammatory bowel disease. N. Engl. J. Med. 1991, 324, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Xavier, R.J.; Podolsky, D.K. Unravelling the pathogenesis of inflammatory bowel disease. Nature 2007, 448, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, G.G. The global burden of IBD: From 2015 to 2025. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Habens, F.; Srinivasan, N.; Oakley, F.; Mann, D.A.; Ganesan, A.; Packham, G. Novel sulfasalazine analogues with enhanced NF-kB inhibitory and apoptosis promoting activity. Apoptosis 2005, 10, 481–491. [Google Scholar] [CrossRef] [PubMed]
- Nugent, S.G.; Kumar, D.; Rampton, D.S.; Evans, D.F. Intestinal luminal pH in inflammatory bowel disease: Possible determinants and implications for therapy with aminosalicylates and other drugs. Gut 2001, 48, 571–577. [Google Scholar] [CrossRef] [PubMed]
- Rhen, T.; Cidlowski, J.A. Antiinflammatory action of glucocorticoids—New mechanisms for old drugs. N. Engl. J. Med. 2005, 353, 1711–1723. [Google Scholar] [CrossRef] [PubMed]
- Rutgeerts, P.; Sandborn, W.J.; Feagan, B.G.; Reinisch, W.; Olson, A.; Johanns, J.; Travers, S.; Rachmilewitz, D.; Hanauer, S.B.; Lichtenstein, G.R.; et al. Infliximab for induction and maintenance therapy for ulcerative colitis. N. Engl. J. Med. 2005, 353, 2462–2476. [Google Scholar] [CrossRef]
- Ratner, M. IL-17-targeting biologics aim to become standard of care in psoriasis. Nat. Biotechnol. 2015, 33, 3–4. [Google Scholar] [CrossRef]
- Neuman, M.G.; Nanau, R.M. Inflammatory bowel disease: Role of diet, microbiota, life style. Transl. Res. 2012, 160, 29–44. [Google Scholar] [CrossRef]
- Kaur, R.; Gulati, M.; Singh, S.K. Role of synbiotics in polysaccharide assisted colon targeted microspheres of mesalamine for the treatment of ulcerative colitis. Int. J. Biol. Macromol. 2017, 95, 438–450. [Google Scholar] [CrossRef]
- Ji, X.; Peng, B.; Ding, H.; Cui, B.; Nie, H.; Yan, Y. Purification, structure and biological activity of pumpkin (Cucurbita moschata) polysaccharides: A review. Food Rev. Int. 2021, 1904973. [Google Scholar] [CrossRef]
- Zhao, H.M.; Wang, Y.; Huang, X.Y.; Huang, M.F.; Xu, R.; Yue, H.Y.; Zhou, B.G.; Huang, H.Y.; Sun, Q.M.; Liu, D.Y. Astragalus polysaccharide attenuates rat experimental colitis by inducing regulatory T cells in intestinal Peyer’s patches. World J. Gastroenterol. 2016, 22, 3175–3185. [Google Scholar] [CrossRef]
- Gao, Y.J.; Zhu, F.; Qian, J.M.; Dai, J.Y. Therapeutic and immunoregulatory effect of GATA-binding protein-3/T-box expressed in T-cells ratio of astragalus polysaccharides on 2, 4, 6-trinitrobenzene sulfonic acid-induced colitis in rats. Chin. J. Integr. Med. 2016, 22, 918–924. [Google Scholar] [CrossRef]
- Yang, M.; Lin, H.B.; Gong, S.; Chen, P.Y.; Geng, L.L.; Zeng, Y.M.; Li, D.Y. Effect of Astragalus polysaccharides on expression of TNF-α, IL-1β and NFATc4 in a rat model of experimental colitis. Cytokine 2014, 70, 81–86. [Google Scholar] [CrossRef]
- Tian, T.; Wang, Z.; Zhang, J. Pathomechanisms of Oxidative Stress in Inflammatory Bowel Disease and Potential Antioxidant Therapies. Oxid. Med. Cell. Longev. 2017, 2017, 4535194. [Google Scholar] [CrossRef]
- Chen, Y.F.; Zheng, J.J.; Qu, C.; Xiao, Y.; Li, F.F.; Jin, Q.X.; Li, H.H.; Meng, F.P.; Jin, G.H.; Jin, D. Inonotus obliquus polysaccharide ameliorates dextran sulphate sodium induced colitis involving modulation of Th1/Th2 and Th17/Treg balance. Artif. Cells Nanomed. Biotechnol. 2019, 47, 757–766. [Google Scholar] [CrossRef]
- Liu, B.; Lin, Q.; Yang, T.; Zeng, L.; Shi, L.; Chen, Y.; Luo, F. Oat β-glucan ameliorates dextran sulfate sodium (DSS)-induced ulcerative colitis in mice. Food Funct. 2015, 6, 3454–3463. [Google Scholar] [CrossRef]
- Liu, L.; Yuan, S.F.; Long, Y.; Guo, Z.J.; Sun, Y.; Li, Y.H.; Niu, Y.B.; Li, C.; Mei, Q.B. Immunomodulation of Rheum tanguticum polysaccharide (RTP) on the immunosuppressive effects of dexamethasone (DEX) on the treatment of colitis in rats induced by 2, 4, 6-trinitrobenzene sulfonic acid. Int. Immunopharmacol. 2009, 9, 1568–1577. [Google Scholar] [CrossRef]
- Liu, L.; Wang, Z.P.; Xu, C.T.; Pan, B.R.; Mei, Q.B.; Long, Y.; Liu, J.Y.; Zhou, S.Y. Effects of Rheum tanguticum polysaccharide on TNBS -induced colitis and CD4+ T cells in rats. World J. Gastroenterol. 2003, 9, 2284–2288. [Google Scholar] [CrossRef]
- Liu, L.; Mei, Q.B.; Wang, Z.P.; Zhou, Y.M.; Zhang, H.; Long, Y.; Liu, J.Y. The Effects of Rheum Tanguticum Polysaccharide on the Polarization of Th1 and Th2 Cells in TNBS-Induced Colitis in Murine. Int. J. Biomed. Sci. 2005, 1, 23–32. [Google Scholar]
- Liu, L.; Guo, Z.J.; Lv, Z.G.; Sun, Y.; Cao, W.; Zhang, R.; Liu, Z.G.; Li, C.; Cao, S.S.; Mei, Q.B. The beneficial effect of Rheum tanguticum polysaccharide on protecting against diarrhea, colonic inflammation and ulceration in rats with TNBS-induced colitis: The role of macrophage mannose receptor in inflammation and immune response. Int. Immunopharmacol. 2008, 8, 1481–1492. [Google Scholar] [CrossRef] [PubMed]
- Wei, B.; Zhang, R.; Zhai, J.B.; Zhu, J.F.; Yang, F.L.; Yue, D.; Liu, X.Y.; Lu, C.L.; Sun, X. Suppression of Th17 Cell Response in the Alleviation of Dextran Sulfate Sodium-Induced Colitis by Ganoderma lucidum Polysaccharides. J. Immunol. Res. 2018, 2018, 2906494. [Google Scholar] [CrossRef] [PubMed]
- Nagai, K.; Ueno, Y.; Tanaka, S.; Hayashi, R.; Shinagawa, K.; Chayama, K. Polysaccharides derived from Ganoderma lucidum fungus mycelia ameliorate indomethacin-induced small intestinal injury via induction of GM-CSF from macrophages. Cell. Immunol. 2017, 320, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Tian, Z.Q.; Liu, Y.; Yang, B.; Zhang, J.; He, H.Y.; Ge, H.; Wu, Y.Z.; Shen, Z.G. Astagalus Polysaccharide Attenuates Murine Colitis through Inhibiton of the NLRP3 Inflammasome. Planta Med. 2017, 83, 70–77. [Google Scholar] [CrossRef]
- Popov, S.V.; Markov, P.A.; Nikitina, I.R.; Petrishev, S.; Smirnov, V.; Ovodov, Y.S. Preventive effect of a pectic polysaccharide of the common cranberry Vaccinium oxycoccos L. on acetic acid-induced colitis in mice. World J. Gastroenterol. 2006, 12, 6646–6651. [Google Scholar] [CrossRef]
- Ren, Y.L.; Geng, Y.; Du, Y.; Li, W.; Lu, Z.M.; Xu, H.Y.; Xu, G.H.; Shi, J.S.; Xu, Z.H. Polysaccharide of Hericium erinaceus attenuates colitis in C57BL/6 mice via regulation of oxidative stress, inflammation-related signaling pathways and modulating the composition of the gut microbiota. J. Nutr. Biochem. 2018, 57, 67–76. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, Y.; Yang, S.; Zhao, D.; Wang, M. A polysaccharide from cultured mycelium of Hericium erinaceus relieves ulcerative colitis by counteracting oxidative stress and improving mitochondrial function. Int. J. Biol. Macromol. 2019, 125, 572–579. [Google Scholar] [CrossRef]
- Wong, V.K.C.; Yu, L.; Cho, C.H. Protective effect of polysaccharides from Angelica sinensis on ulcerative colitis in rats. Inflammopharmacology 2018, 16, 162–167. [Google Scholar] [CrossRef]
- Liu, S.P.; Dong, W.G.; Wu, D.F.; Luo, H.S.; Yu, J.P. Protective effect of angelica sinensis polysaccharide on experimental immunological colon injury in rats. World. J. Gastroenterol. 2003, 9, 2786–2790. [Google Scholar] [CrossRef]
- Cheng, F.; Zhang, Y.; Li, Q.; Zeng, F.; Wang, K. Inhibition of Dextran Sodium Sulfate-Induced Experimental Colitis in Mice by Angelica Sinensis Polysaccharide. J. Med. Food 2020, 23, 584–592. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Li, Y.Y. Inflammatory bowel disease: Pathogenesis. World J. Gastroenterol. 2014, 20, 91–99. [Google Scholar] [CrossRef]
- Liu, L.; Liu, Z.; Zhang, T.; Shi, L.; Zhang, W.; Zhang, Y. Combined Therapy with Rheum tanguticum Polysaccharide and Low-dose 5-ASA Ameliorates TNBS-Induced Colitis in Rats by Suppression of NF-κB. Planta Med. 2015, 81, 705–712. [Google Scholar] [CrossRef]
- Żyła, E.; Dziendzikowska, K.; Gajewska, M.; Wilczak, J.; Harasym, J.; Gromadzka-Ostrowska, J. Beneficial Effects of Oat Beta-Glucan Dietary Supplementation in Colitis Depend on its Molecular Weight. Molecules 2019, 24, 3591. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, N.F.; Kan, J.; Zhang, X.; Wu, X.N.; Sun, R.; Tang, S.X.; Liu, J.; Qian, C.Q.; Jin, C.H. Structural characterization of water-soluble polysaccharide from Arctium lappa and its effects on colitis mice. Carbohydr. Polym. 2019, 213, 89–99. [Google Scholar] [CrossRef]
- Jing, Y.P.; Li, A.P.; Liu, Z.R.; Yang, P.R.; Wei, J.S.; Chen, X.J.; Zhao, T.; Bai, Y.R.; Zha, L.J.; Zhang, C.J. Absorption of Codonopsis pilosula Saponins by Coexisting Polysaccharides Alleviates Gut Microbial Dysbiosis with Dextran Sulfate Sodium-Induced Colitis in Model Mice. BioMed Res. Int. 2018, 2018, 1781036. [Google Scholar] [CrossRef]
- Sun, J.; Chen, H.; Kan, J.; Gou, Y.R.; Liu, J.; Zhang, X.; Wu, X.N.; Tang, S.X.; Sun, R.; Qian, C.L.; et al. Anti-inflammatory properties and gut microbiota modulation of an alkali-soluble polysaccharide from purple sweet potato in DSS-induced colitis mice. Int. J. Biol. Macromol. 2020, 153, 708–722. [Google Scholar] [CrossRef]
- Liu, X.; Yu, X.; Xu, X.; Zhang, X.; Zhang, X. The protective effects of Poria cocos-derived polysaccharide CMP33 against IBD in mice and its molecular mechanism. Food Funct. 2018, 9, 5936–5949. [Google Scholar] [CrossRef]
- Cui, L.; Wang, W.; Luo, Y.; Ning, Q.; Xia, Z.; Chen, J.; Feng, L.; Wang, H.; Song, J.; Tan, X.B.; et al. Polysaccharide from Scutellaria baicalensis Georgi ameliorates colitis via suppressing NF-kappaB signaling and NLRP3 inflammasome activation. Int. J. Biol. Macromol. 2019, 132, 393–405. [Google Scholar] [CrossRef]
- Cho, C.W.; Ahn, S.; Lim, T.G.; Hong, H.D.; Rhee, Y.K.; Yang, D.C.; Jang, M. Cynanchum wilfordii Polysaccharides Suppress Dextran Sulfate Sodium-Induced Acute Colitis in Mice and the Production of Inflammatory Mediators from Macrophages. Mediat. Inflamm. 2017, 2017, 3859856. [Google Scholar] [CrossRef]
- Zhu, C.H.; Zhang, S.M.; Song, C.W.; Zhang, Y.B.; Ling, Q.J.; Hoffmann, P.R.; Li, J.; Zheng, W.J.; Huang, Z. Selenium nanoparticles decorated with Ulva lactuca polysaccharide potentially attenuate colitis by inhibiting NF-κB mediated hyper inflammation. J. Nanobiotechnol. 2017, 15, 20. [Google Scholar] [CrossRef]
- Zhao, L.J.; Wu, H.B.; Zhao, A.H.; Lu, H.L.; Sun, W.; Ma, C.W.; Yang, Y.T.; Xin, X.; Zou, H.M.; Qiu, M.F.; et al. The in vivo and in vitro study of polysaccharides from a two-herb formula on ulcerative colitis and potential mechanism of action. J. Ethnopharmacol. 2014, 153, 151–159. [Google Scholar] [CrossRef]
- Hu, L.; Li, H.; Li, W.; Chen, J.; Yang, J.; Gu, J. The mechanism of alopolysaccharide protecting ulceralive colitis. Biomed. Pharmacother. 2017, 88, 145–150. [Google Scholar]
- Hu, T.; Lin, Q.L.; Guo, T.; Yang, T.; Zhou, W.H.; Deng, X.F.; Yan, J.K.; Luo, Y.; Ju, M.M.; Luo, F.J. Polysaccharide isolated from Phellinus linteus mycelia exerts anti-inflammatory effects via MAPK and PPAR signaling pathways. Carbohydr. Polym. 2018, 200, 487–497. [Google Scholar] [CrossRef]
- Zhang, R.J.; Yuan, S.J.; Ye, J.F.; Wang, X.D.; Zhang, X.D.; Shen, J.; Yuan, M.M.; Liao, W.Z. Polysaccharide from flammuliana velutipes improves colitis via regulation of colonic microbial dysbiosis and inflammatory responses. Int. J. Biol. Macromol. 2020, 14, 1252–1261. [Google Scholar] [CrossRef]
- Li, Y.; Fan, L.; Tang, T.; Tang, Y.; Xie, M.; Zeng, X.; Sun, Y.; Mei, Q. Modified apple polysaccharide prevents colitis through modulating IL-22 and IL-22BP expression. Int. J. Biol. Macromol. 2017, 103, 1217–1223. [Google Scholar] [CrossRef]
- Tao, J.H.; Duan, J.A.; Jiang, S.; Feng, N.N.; Qiu, W.Q.; Ling, Y. Polysaccharides from Chrysanthemum morifolium Ramat ameliorate colitis rats by modulating the intestinal microbiota community. Oncotarget 2017, 8, 80790–80803. [Google Scholar] [CrossRef]
- Onitake, T.; Ueno, Y.; Tanaka, S.; Sagami, S.; Hayashi, R.; Nagai, K.; Hide, M.; Chayama, K. Pulverized konjac glucomannan ameliorates oxazolone-induced colitis in mice. Eur. J. Nutr. 2015, 54, 959–969. [Google Scholar] [CrossRef]
- Miao, X.P.; Sun, X.N.; Cui, L.J.; Cao, Q.F.; Zhuang, G.F.; Deng, T.Z.; Zhang, D.Y. Suppressive effect of pectic polysaccharides extracted from Rauwolfia verticillata (Lour.) Baill.var.hainanensis Tsiang on inflammation by regulation of NF-κ B pathway and interleukin-17 in mice with dextran sulphatesodium-induced ulcerative colitis. Asian Pac. J. Trop. Med. 2015, 8, 147–152. [Google Scholar] [CrossRef]
- Miao, X.P.; Sun, X.N.; Li, Q.S.; Cui, L.J.; Wang, X.Y.; Zhuang, G.F.; Deng, T.Z. Pectic polysaccharides extracted from Rauvolfia verticillata (Lour.) Baill. var. hainanensis Tsiang ameliorate ulcerative colitis via regulating the MAPKs and NF-κB pathways in dendritic cells. Clin. Exp. Pharmacol. Physiol. 2019, 46, 48–55. [Google Scholar] [CrossRef]
- Tao, J.H.; Duan, J.A.; Jiang, S.; Guo, J.M.; Qian, Y.Y.; Qian, D.W. Simultaneous determination of six short-chain fatty acids in colonic contents of colitis mice after oral administration of polysaccharides from Chrysanthemum morifolium Ramat by gas chromatography with flame ionization detector. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2016, 1029–1030, 88–94. [Google Scholar] [CrossRef]
- Wang, Z.; Liang, Y.; Zhang, D.; Wu, X.; Yu, J.; Zhang, Z.; Li, Y.; Sun, C.; Tang, Z.; Liu, L. Protective Effects of Polysaccharide Extracted from Portulacae oleracea L. on Colitis Induced by Dextran Sulfate Sodium. J. Med. Food 2020, 23, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Yue, Y.; Wu, S.; Li, Z.; Li, J.; Li, X.; Xiang, J.; Ding, H. Wild jujube polysaccharides protect against experimental inflammatory bowel disease by enabling enhanced intestinal barrier function. Food Funct. 2015, 6, 2568–2577. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Li, Y.; Zhang, B. The effects of konjac oligosaccharide on TNBS-induced colitis in rats. Int. Immunopharmacol. 2016, 40, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Lara-Villoslada, F.; de Haro, O.; Camuesco, D.; Comalada, M.; Velasco, J.; Zarzuelo, A.; Xaus, J.; Galvez, J. Short-chain fructooligosaccharides, in spite of being fermented in the upper part of the large intestine, have anti-inflammatory activity in the TNBS model of colitis. Eur. J. Nutr. 2006, 45, 418–425. [Google Scholar] [CrossRef]
- Ji, X.; Hou, C.; Gao, Y.; Xue, Y.; Yan, Y.; Guo, X. Metagenomic analysis of gut microbiota modulatory effects of jujube (Ziziphus jujuba Mill.) polysaccharides in a colorectal cancer mouse model. Food Funct. 2020, 11, 163–173. [Google Scholar] [CrossRef]
- Suzuki, T. Regulation of intestinal epithelial permeability by tight junctions. Cell. Mol. Life Sci. 2013, 70, 631–659. [Google Scholar] [CrossRef]
- Turner, J.R. Intestinal mucosal barrier function in health and disease. Nat. Rev. Immunol. 2009, 9, 799–809. [Google Scholar] [CrossRef]
- Weber, C.R. Dynamic properties of the tight junction barrier. Ann. N. Y. Acad. Sci. 2012, 1257, 77–84. [Google Scholar] [CrossRef]
- Leppkes, M.; Roulis, M.; Neurath, M.F.; Kollias, G.; Becker, C. Pleiotropic functions of TNF-α in the regulation of the intestinal epithelial response to inflammation. Int. Immunol. 2014, 26, 509–515. [Google Scholar] [CrossRef]
- Maria-Ferreira, D.; Nascimento, A.M.; Cipriani, T.R.; Santana-Filho, A.P.; Watanabe, P.D.S.; Luciano, F.B.; Bocate, K.C.P.; van den Wijngaard, R.M.; Werner, M.F.D.P.; Baggio, C.H. Rhamnogalacturonan, a chemically-defined polysaccharide, improves intestinal barrier function in DSS-induced colitis in mice and human Caco-2 cells. Sci. Rep. 2018, 8, 12261. [Google Scholar] [CrossRef]
- Zong, S.; Ye, Z.; Zhang, X.; Chen, H.; Ye, M. Protective effect of Lachnum polysaccharide on dextran sulfate sodium-induced colitis in mice. Food Funct. 2020, 11, 846–859. [Google Scholar] [CrossRef]
- Cui, L.; Guan, X.; Ding, W.; Luo, Y.; Wang, W.; Bu, W.; Song, J.; Tan, X.; Sun, E.; Ning, Q.; et al. Scutellaria baicalensis Georgi polysaccharide ameliorates DSS-induced ulcerative colitis by improving intestinal barrier function and modulating gut microbiota. Int. J. Biol. Macromol. 2021, 166, 1035–1045. [Google Scholar] [CrossRef]
- Nagao-Kitamoto, H.; Shreiner, A.B.; Gillilland, M.; Kitamoto, S.; Ishii, C.; Hirayama, A.; Kuffa, P.; EI-Zaatari, M.; Grasberger, H.; Seekatz, A.M.; et al. Functional Characterization of Inflammatory Bowel Disease-Associated Gut Dysbiosis in Gnotobiotic Mice. Cell. Mol. Gastroenterol. Hepatol. 2016, 2, 468–481. [Google Scholar] [CrossRef]
- Shiba, T.; Aiba, Y.; Ishikawa, H.; Ushiyama, A.; Takagi, A.; Mine, T.; Koga, Y. The suppressive effect of bifidobacteria on Bacteroides vulgatus, a putative pathogenic microbe in inflammatory bowel disease. Microbiol. Immunol. 2003, 47, 371–378. [Google Scholar] [CrossRef]
- Servin, A.L. Antagonistic activities of lactobacilli and bifidobacteria against microbial pathogens. FEMS Microbiol. Rev. 2004, 28, 405–440. [Google Scholar] [CrossRef]
- Sartor, R.B. Therapeutic manipulation of the enteric microflora in inflammatory bowel diseases: Antibiotics, probiotics, and prebiotics. Gastroenterology 2004, 126, 1620–1633. [Google Scholar] [CrossRef]
- Wong, C.; Harris, P.J.; Ferguson, L.R. Potential Benefits of Dietary Fibre Intervention in Inflammatory Bowel Disease. Int. J. Mol. Sci. 2016, 17, 919. [Google Scholar] [CrossRef]
- Shao, S.; Wang, D.; Zheng, W.; Li, X.; Zhang, H.; Zhao, D.; Wang, M. A unique polysaccharide from Hericium erinaceus mycelium ameliorates acetic acid-induced ulcerative colitis rats by modulating the composition of the gut microbiota, short chain fatty acids levels and GPR41/43 respectors. Int. Immunopharmacol. 2019, 71, 411–422. [Google Scholar] [CrossRef]
- Cai, Y.; Liu, W.; Lin, Y.X.; Zhang, S.B.; Zou, B.R.; Xiao, D.; Lin, L.; Zhong, Y.P.; Zheng, H.H.; Liao, Q.F.; et al. Compound polysaccharides ameliorate experimental colitis by modulating gut microbiota composition and function. J. Gastroenterol. Hepatol. 2019, 34, 1554–1562. [Google Scholar] [CrossRef]
- Xie, J.; Liu, Y.; Chen, B.; Zhang, G.; Ou, S.; Luo, J.; Peng, X. Ganoderma lucidum polysaccharide improves rat DSS-induced colitis by altering cecal microbiota and gene expression of colonic epithelial cells. Food Nutr. Res. 2019, 63, 1–12. [Google Scholar] [CrossRef]
- Zou, Q.; Zhang, X.; Liu, X.; Li, Y.; Tan, Q.; Dan, Q.; Yuan, T.; Liu, X.; Liu, R.H.; Liu, Z. Ficus carica polysaccharide attenuates DSS-induced ulcerative colitis in C57BL/6 mice. Food Funct. 2020, 11, 6666–6679. [Google Scholar] [CrossRef]
| Name | Resource | Models | Strain | Dose | Mechanisms | References |
|---|---|---|---|---|---|---|
| Polysaccharide (EP-1) | Mycelium of Hericium erinaceus | 4% acetic acid-induced colitis | Sprague-Dawley (SD) rats | 1.2 g/kg and 2.5 g/kg | the enzyme specific activity of SOD ↑, an appropriate redox balance ↑, Bcl-2 ↑, the integrity of the mitochondrial membrane ↑, the MDA content ↓, TNF-α, IL-6, IL-8 ↓, NF-κB p-65 ↓, level of ROS ↓, the caspase-3 activation ↓, |
|
| Saponins and polysaccharide | Codonopsis pilosula Nannf (CPN) | 3% DSS-induced colitis | female C57BL/6 mice | 300 mg/kg | TGF-β and IL-10 ↑, levels of acetic acid, propionic acid, butyric acid, isobutyric acid, and isovaleric acid ↑, IL-17A, IL-17F, IL-6, IL-22, and TNF-α ↓ |
|
| An alkali-soluble polysaccharide (ASPP) | Purple sweet potato | DSS-induced colitis | female ICR mice | 400 mg/kg | Th1, Th17 or Treg cells, acetate and propionate ↑, the SCFAs levels ↑, IL-1β, IL-6, TNF-α ↓, NF-κB ↓, |
|
| Astragalus polysaccharide (APS) | Astragalus | TNBS-induced colitis | male Sprague-Dawley rats | 400 mg/kg | Treg cells ↑, STAT-5 ↑, TGF-β ↑, IL-2, IL-6, IL-17 and IL-23 ↓, ROR-gt ↓ |
|
| Astragalus polysaccharide (APS) | Astragalus | 3% DSS-induced colitis | male C57BL/6 mice | 100 mg/kg/d, 200 mg/kg/d and 500 mg/kg/d | NLRP3, ASC, and caspase-1 ↓, IL-1β and IL-18↓, caspase-1 ↓, NLRP3 inflammasome ↓ |
|
| Astragalus polysaccharide | Astragalus | TNBS-induced colitis | male Wistar rats | 100 mg/kg and 200 mg/kg | NFATc4 mRNA expression ↑, TNF-a and IL-1β mRNA expressions ↓ |
|
| Polysaccharide (APS) | Astragalus | TNBS-induced colitis | male SD rat | 0.5g/kg/day for 14 days | IL-1β, IL-6, IL-18, TNF-α and IFN-γ, IL-10 ↑, the activation of NLRP3 inflammasome, cleavage of caspase1 ↓, β-arrestin1 expression ↓, the mRNA expressions of NLRP3, ASC, caspase1 ↓, β-arrestin1 ↓ |
|
| Rheum tanguticum Polysaccharide | Rheum tanguticum | TNBS-induced colitis | Sprague-Dawley male rats | 200 mg/kg/day | Prostaglandin E2 (PGE2) ↑, NF-κBp65/Lamin B1 density ratio ↓, TNF-α ↓, the COX-2/glyceraldehyde 3-phosphate dehydrogenase (GAPDH) density ratio ↓, iNOS↓ |
|
| Rheum tanguticum polysaccharide (RTP) | Rheum tanguticum | TNBS -induced colitis | male Sprague-Dawley rats | 200 mg/kg | CD4+T cells ↓ |
|
| Rheum tanguticum polysaccharide (RTP) | Rheum tanguticum | TNBS -induced colitis | male Sprague–Dawley (SD) rats | 200 mg/kg/day | IL-5 ↑, TNF-α, IFN-γ ↓, IL-4, Th1 cell cytokine ↓, Th2 cell cytokine ↑, TLR4,NF-κB/p65, p-IκBα level ↓ |
|
| Rheum tanguticum polysaccharide (RTP) | Rheum tanguticum | UC induced by TNBS in BALB/c mice and CD induced by TNBS in SD rats | Adult SD rats, BALB/c mice | 200 mg/kg | CD4+T cell ↓, IFN-γ ↓, Il-4 ↓, |
|
| Rheum tanguticum polysaccharide (RTP) | Rheum tanguticum | TNBS-induced colitis | male Sprague–Dawley (SD) rats | 200 mg/kg/day | IFN-γ ↑ |
|
| Ganoderma lucidum polysaccharide (GLP) | Ganoderma lucidum | (DSS)-induced colitis | male C57BL/6 mice | 100 mg/kg | TNF-α, IL-1β, IL-6 ↓, IL-4 ↓, ROR-γt ↓ |
|
| Polysaccharide (MAK) | Ganoderma lucidum fungus mycelia | (TNBS)-induced colitis | C57BL/6(B6) mice | 25 μg/mL |
| |
| Cynanchum wilfordii Polysaccharide | Cynanchum wilfordii | 5% DSS-induced colitis | female BALB/c mice | 100 and 200 mg/kg | IL-6 ↓, TNF-α ↓, iNOS, COX-2 ↓, NF-κB p65 ↓, |
|
| Modified apple polysaccharide (MAP) | Apple | 2.5% DSS-induced colitis | male ICR mice | 2.5% MAP | IL-22BP ↑, IL-22 ↓, p-STAT3, Bcl-2 and cyclin D1 ↓ |
|
| β-glucan | Oat | 3% DSS-induced colitis | male, ICR mice | 500 mg/kg, 1000 mg/kg | TNF-α, IL-1β ↓ |
|
| Oat Beta-Glucan | Oat | TNBS-induced colitis | male Sprague-Dawley rats | 1% (w/w) of low molecular mass (1.7 × 106 g/mol) and high molecular mass (5.9 × 104 g/mol) | Cxcl1, Il17a, Cxcr1, Spp1 ↑, IL-6 and IL-12 ↓, TNF-α and IL-1 ↓, total cyclooxygenase (COX), prostaglandin E2 (PGE2), tromboksan A2 (TXA2), and myeloperoxidase (MPO) ↓, Ccl19, Cd40lg, Cxcr5, Il10ra, Il16, Il21, Il2rg, Il5ra, Lta, Ltb, Osm, Tnf, Tnfsf11, Tnfsf14 ↓, |
|
| Angelica Sinensis Polysaccharide (ASP) | Angelica Sinensis | 2.5% DSS-induced colitis | male BABL/C mice | 200 mg/kg | TJ proteins (ZO-1, occludin, and claudin-1) ↑, IL-6, IL-1b, and TNF-a ↓ |
|
| Inonotus obliquus polysaccharide (IOP) | Inonotus obliquus | 3% DSS-induced colitis | male BALB/c mice | 100, 200, 300 mg/kg | Treg and Th2 ↑, Foxp3 and GATA-3 ↑, IL-4 and IL-10 ↑, p-STAT6 ↑, ROR-γt and T-bet ↓, Th17 and Th1 ↓, p-STAT1 and p-STAT3 ↓ |
|
| Konjac oligosaccharide (KOS) | Konjac | TNBS-induced colitis | male SD rats, | 1.0 g/kg and 4.0 g/kg | iNOS and COX-2, TNF-α and IL-1β ↓ |
|
| Pulverized konjac glucomannan (PKGM) | Konjac | oxazolone-induced colitis | female C57BL/6(B6) mice | MF containing 5 % (w/w) PKGM powder, oral administration | IL-4 and IFN-γ, IL-13 ↓, |
|
| Pectic polysaccharide | Rauwolfia verticillata (Lour.) Baill. var. hainanensis Tsiang | DSS-induced colitis | female BALB/c mice | 200 μL | Iκ Ba↑, NF- κB p65 ↓, IL-17 and TNF- a ↓ |
|
| Pectic polysaccharide (PP) | Rauvolfia verticillata (Lour.) Baill. var. hainanensis Tsiang | (DSS)-induced colitis | female BALB/c mice | 100 mg/kg | TNF-α and IL-6 ↓ |
|
| Water-soluble polysaccharide (ALP-1) | Arctium lappa | DSS-induced colitis | male ICR mice | 300 mg/kg | IL-10↑, IL-1β, IL-6 and TNF-α ↓ |
|
| Polysaccharide | Chrysanthemum morifolium Ramat | DSS induced colitis | male C57BL/6 mice | 75, 150, 300 mg/kg | SCFAs ↑ |
|
| Polysaccharide | Portulacae oleracea L. | DSS-induced colitis | Kun Ming mice | 0.75, 0.5, and 0.25 g/mL | PGE2 and IL-6 COX-2, STAT3 ↓ |
|
| Lachnum polysaccharide (LEP) | Lachnum | 2.5% DSS-induced colitis | male ICR mice | 200 mg/kg | restore intestinal barrier integrity by regulating the expression of tight junction proteins and mucus layer protecting proteins, |
|
| Saponins and polysaccharides | Codonopsis pilosula Nannf (CPN) | 3% DSS-induced colitis | female C57BL/6 mice | 300 mg/kg | intestinal metabolism, recovery of the holistic gut microbiota ↑, gut microbial dysbiosis ↓ |
|
| An alkali-soluble polysaccharide(ASPP) | Purple sweet potato | DSS-induced colitis | female ICR mice | 400 mg/kg | regulate the composition of the gut microbiota |
|
| Yam polysaccharide and inulin | Yam | TNBS-induced colitis | SPF male Sprague-Dawley rats | 300 mg/kg/day | modulate gut microbiota composition and function |
|
| Konjac oligosaccharide (KOS) | Konjac | TNBS-induced colitis | male SD rats, | 1.0 g/kg and 4.0 g/kg | Bifidobacterium and Lactobacillus ↑, Escherichia coli and Enterococcus levels ↓ |
|
| Water-soluble polysaccharide (ALP-1) | Arctium lappa | DSS-induced colitis | male ICR mice | 300 mg/kg | Firmicutes, Ruminococcaceae, Lachnospiraceae and Lactobacillus ↑ |
|
| Short-chain fructooligosaccharides(SC-FOS) | - | TNBS-induced colitis | female Wistar rats | 50 g/kg | cecal lactobacilli and bifidobacteria counts ↑ |
|
| Ganoderma lucidum polysaccharide (GLP) | Ganoderma lucidum | 2.5% DSS-induced colitis | male Wistar rats | a basal and a GLP diet | SCFA-producing bacteria, including Ruminococcus_1, and the reduction of pathogens ↑ |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, J.; Geng, J.; Wu, J.; Wang, H.; Liu, Y.; Du, B.; Yang, Y.; Xiao, H. A Potential Role of Plant/Macrofungi/Algae-Derived Non-Starch Polysaccharide in Colitis Curing: Review of Possible Mechanisms of Action. Molecules 2022, 27, 6467. https://doi.org/10.3390/molecules27196467
Feng J, Geng J, Wu J, Wang H, Liu Y, Du B, Yang Y, Xiao H. A Potential Role of Plant/Macrofungi/Algae-Derived Non-Starch Polysaccharide in Colitis Curing: Review of Possible Mechanisms of Action. Molecules. 2022; 27(19):6467. https://doi.org/10.3390/molecules27196467
Chicago/Turabian StyleFeng, Jinxiu, Jingzhang Geng, Jinhui Wu, Huiying Wang, Yanfei Liu, Bin Du, Yuedong Yang, and Haitao Xiao. 2022. "A Potential Role of Plant/Macrofungi/Algae-Derived Non-Starch Polysaccharide in Colitis Curing: Review of Possible Mechanisms of Action" Molecules 27, no. 19: 6467. https://doi.org/10.3390/molecules27196467
APA StyleFeng, J., Geng, J., Wu, J., Wang, H., Liu, Y., Du, B., Yang, Y., & Xiao, H. (2022). A Potential Role of Plant/Macrofungi/Algae-Derived Non-Starch Polysaccharide in Colitis Curing: Review of Possible Mechanisms of Action. Molecules, 27(19), 6467. https://doi.org/10.3390/molecules27196467