Design of Deep Eutectic Systems: Plastic Crystalline Materials as Constituents
Abstract
:1. Introduction
2. Results and Discussion
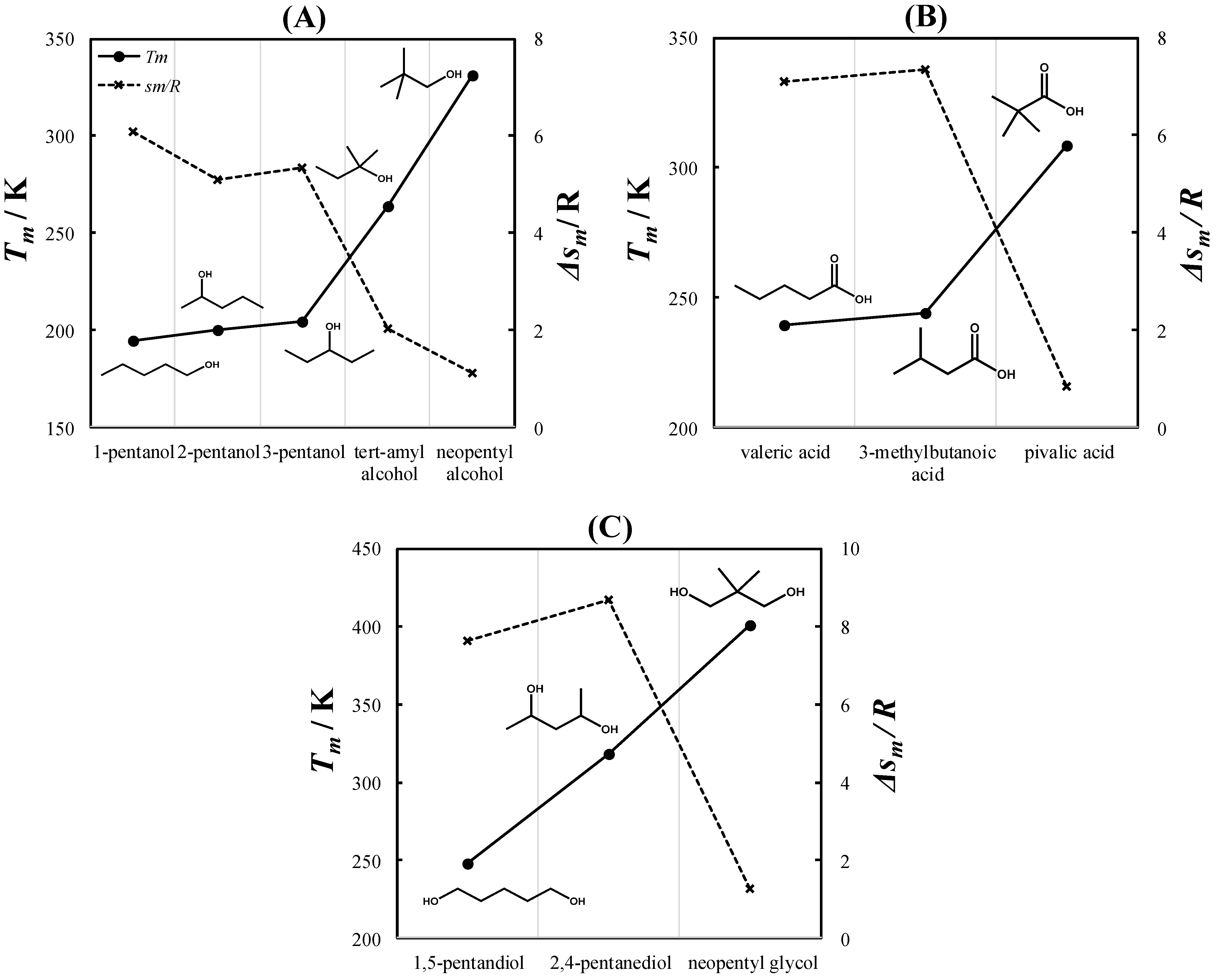
2.1. Properties of Pure PCs
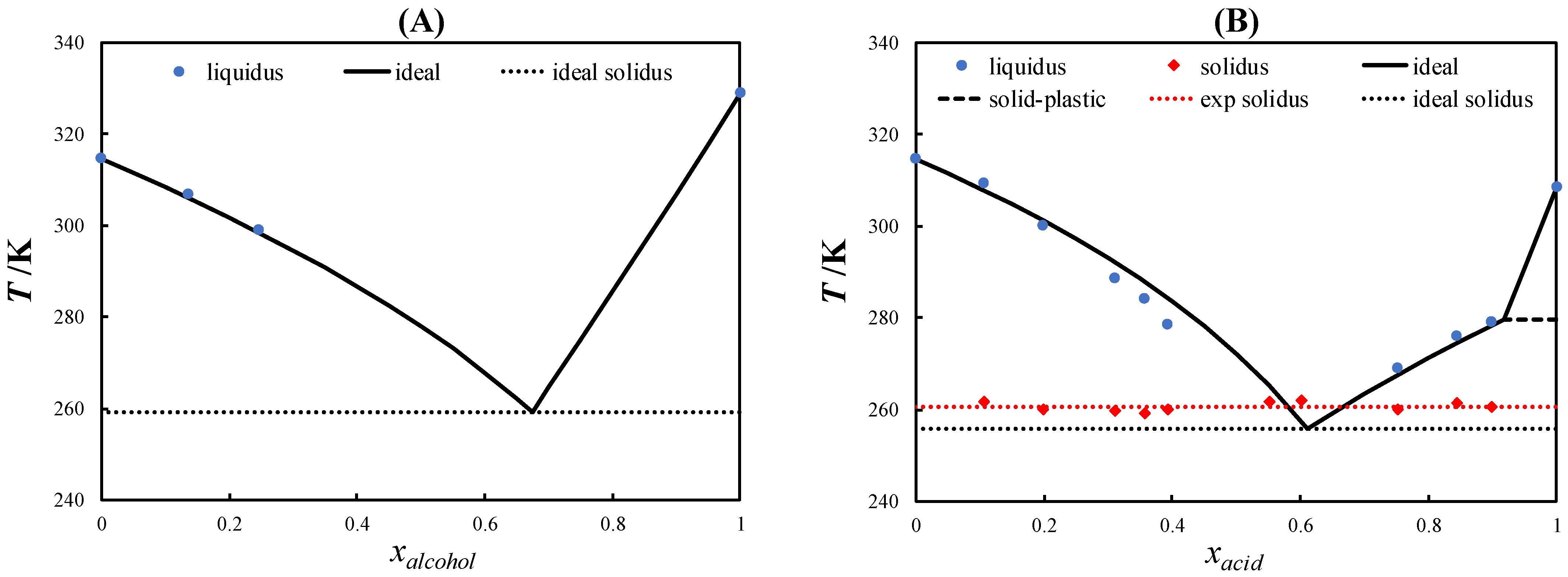
2.2. Eutectic Systems with PCs
2.3. Comparison with Other Eutectic Systems
3. Materials and Methods
3.1. Eutectic Systems
3.2. DSC
3.3. SLE Modeling
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Smith, E.L.; Abbott, A.P.; Ryder, K.S. Deep Eutectic Solvents (DESs) and Their Applications. Chem. Rev. 2014, 114, 11060–11082. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; De Oliveira Vigier, K.; Royer, S.; Jerome, F. Deep eutectic solvents: Syntheses, properties and applications. Chem. Soc. Rev. 2012, 41, 7108–7146. [Google Scholar] [CrossRef] [PubMed]
- Wazeer, I.; Hadj-Kali, M.K.; Al-Nashef, I.M. Utilization of Deep Eutectic Solvents to Reduce the Release of Hazardous Gases to the Atmosphere: A Critical Review. Molecules 2021, 26, 75. [Google Scholar] [CrossRef] [PubMed]
- Winterton, N. The green solvent: A critical perspective. Clean Technol. Environ. Policy 2021, 23, 2499–2522. [Google Scholar] [CrossRef]
- Florindo, C.; Branco, L.C.; Marrucho, I.M. Quest for Green-Solvent Design: From Hydrophilic to Hydrophobic (Deep) Eutectic Solvents. ChemSusChem 2019, 12, 1549–1559. [Google Scholar] [CrossRef]
- Abranches, D.O.; Coutinho, J.A.P. Type V deep eutectic solvents: Design and applications. Curr. Opin. Green Sustain. Chem. 2022, 35, 100612. [Google Scholar] [CrossRef]
- Perna, F.M.; Vitale, P.; Capriati, V. Deep eutectic solvents and their applications as green solvents. Curr. Opin. Green Sustain. Chem. 2020, 21, 27–33. [Google Scholar] [CrossRef]
- Dheyab, A.S.; Bakar, M.F.A.; Alomar, M.; Sabran, S.F.; Hanafi, A.F.M.; Mohamad, A. Deep eutectic solvents (DESs) as green extraction media of beneficial bioactive phytochemicals. Separations 2021, 8, 176. [Google Scholar] [CrossRef]
- Huang, J.; Guo, X.; Xu, T.; Fan, L.; Zhou, X.; Wu, S. Ionic deep eutectic solvents for the extraction and separation of natural products. J. Chromatogr. A 2019, 1598, 1–19. [Google Scholar] [CrossRef]
- Álvarez, M.S.; Zhang, Y. Sketching neoteric solvents for boosting drugs bioavailability. J. Control Release 2019, 311–312, 225–232. [Google Scholar] [CrossRef]
- Hou, Y.; Congfei, Y.; Weize, W. Deep Eutectic Solvents: Green Solvents for Separation Applications. Acta Phys.-Chim. Sin. 2018, 34, 873–885. [Google Scholar] [CrossRef]
- Florindo, C.; Romero, L.; Rintoul, I.; Branco, L.C.; Marrucho, I.M. From Phase Change Materials to Green Solvents: Hydrophobic Low Viscous Fatty Acid–Based Deep Eutectic Solvents. ACS Sustain. Chem. Eng. 2018, 6, 3888–3895. [Google Scholar] [CrossRef]
- Alhadid, A.; Jandl, C.; Mokrushina, L.; Minceva, M. Cocrystal Formation in Choline Chloride Deep Eutectic Solvents. Cryst. Growth Des. 2022, 22, 1933–1942. [Google Scholar] [CrossRef]
- Andruch, V.; Makoś-Chełstowska, P.; Płotka-Wasylka, J. Remarks on use of the term “deep eutectic solvent” in analytical chemistry. Microchem. J. 2022, 179, 107498. [Google Scholar] [CrossRef]
- Crespo, E.A.; Silva, L.P.; Martins, M.A.R.; Bülow, M.; Ferreira, O.; Sadowski, G.; Held, C.; Pinho, S.P.; Coutinho, J.A.P. The Role of Polyfunctionality in the Formation of [Ch]Cl-Carboxylic Acid-Based Deep Eutectic Solvents. Ind. Eng. Chem. Res. 2018, 57, 11195–11209. [Google Scholar] [CrossRef]
- van den Bruinhorst, A.; Kollau, L.J.B.M.; Vis, M.; Hendrix, M.M.R.M.; Meuldijk, J.; Tuinier, R.; Esteves, A.C.C. From a eutectic mixture to a deep eutectic system via anion selection: Glutaric acid + tetraethylammonium halides. J. Chem. Phys. 2021, 155, 014502. [Google Scholar] [CrossRef] [PubMed]
- Kollau, L.J.B.M.; Tuinier, R.; Verhaak, J.; den Doelder, J.; Filot, I.A.W.; Vis, M. Design of Nonideal Eutectic Mixtures Based on Correlations with Molecular Properties. J. Phys. Chem. B 2020, 124, 5209–5219. [Google Scholar] [CrossRef]
- Silva, L.P.; Martins, M.A.R.; Conceição, J.H.F.; Pinho, S.P.; Coutinho, J.A.P. Eutectic Mixtures Based on Polyalcohols as Sustainable Solvents: Screening and Characterization. ACS Sustain. Chem. Eng. 2020, 8, 15317–15326. [Google Scholar] [CrossRef]
- Silva, L.P.; Fernandez, L.; Conceição, J.H.F.; Martins, M.A.R.; Sosa, A.; Ortega, J.; Pinho, S.P.; Coutinho, J.A.P. Design and Characterization of Sugar-Based Deep Eutectic Solvents Using Conductor-like Screening Model for Real Solvents. ACS Sustain. Chem. Eng. 2018, 6, 10724–10734. [Google Scholar] [CrossRef]
- Silva, L.P.; Martins, M.A.R.; Abranches, D.O.; Pinho, S.P.; Coutinho, J.A.P. Solid-liquid phase behavior of eutectic solvents containing sugar alcohols. J. Mol. Liq. 2021, 337, 116392. [Google Scholar] [CrossRef]
- Silva, L.P.; Araújo, C.F.; Abranches, D.O.; Melle-Franco, M.; Martins, M.A.R.; Nolasco, M.M.; Ribeiro-Claro, P.J.A.; Pinho, S.P.; Coutinho, J.A.P. What a difference a methyl group makes–probing choline–urea molecular interactions through urea structure modification. PCCP 2019, 21, 18278–18289. [Google Scholar] [CrossRef] [PubMed]
- van den Bruinhorst, A.; Kollau, L.J.B.M.; Kroon, M.C.; Meuldijk, J.; Tuinier, R.; Esteves, A.C.C. A centrifuge method to determine the solid–liquid phase behavior of eutectic mixtures. J. Chem. Phys. 2018, 149, 224505. [Google Scholar] [CrossRef] [PubMed]
- Hayyan, M.; Looi, C.Y.; Hayyan, A.; Wong, W.F.; Hashim, M.A. In Vitro and In Vivo Toxicity Profiling of Ammonium-Based Deep Eutectic Solvents. PLoS ONE 2015, 10, e0117934. [Google Scholar] [CrossRef]
- Wazeer, I.; AlNashef, I.M.; Al-Zahrani, A.A.; Hadj-Kali, M.K. The subtle but substantial distinction between ammonium- and phosphonium-based deep eutectic solvents. J. Mol. Liq. 2021, 332, 115838. [Google Scholar] [CrossRef]
- Gajardo-Parra, N.F.; Cotroneo-Figueroa, V.P.; Aravena, P.; Vesovic, V.; Canales, R.I. Viscosity of Choline Chloride-Based Deep Eutectic Solvents: Experiments and Modeling. J. Chem. Eng. Data 2020, 65, 5581–5592. [Google Scholar] [CrossRef]
- Lemaoui, T.; Darwish, A.S.; Attoui, A.; Abu Hatab, F.; Hammoudi, N.E.H.; Benguerba, Y.; Vega, L.F.; Alnashef, I.M. Predicting the density and viscosity of hydrophobic eutectic solvents: Towards the development of sustainable solvents. Green Chem. 2020, 22, 8511–8530. [Google Scholar] [CrossRef]
- Al-Dawsari, J.N.; Bessadok-Jemai, A.; Wazeer, I.; Mokraoui, S.; AlMansour, M.A.; Hadj-Kali, M.K. Fitting of experimental viscosity to temperature data for deep eutectic solvents. J. Mol. Liq. 2020, 310, 113127. [Google Scholar] [CrossRef]
- Mjalli, F.S.; Naser, J. Viscosity model for choline chloride-based deep eutectic solvents. Asia-Pac. J. Chem. Eng. 2015, 10, 273–281. [Google Scholar] [CrossRef]
- van Osch, D.J.G.P.; Dietz, C.H.J.T.; van Spronsen, J.; Kroon, M.C.; Gallucci, F.; van Sint Annaland, M.; Tuinier, R. A Search for Natural Hydrophobic Deep Eutectic Solvents Based on Natural Components. ACS Sustain. Chem. Eng. 2019, 7, 2933–2942. [Google Scholar] [CrossRef]
- Aşçı, Y.S.; Lalikoglu, M. Development of New Hydrophobic Deep Eutectic Solvents Based on Trioctylphosphine Oxide for Reactive Extraction of Carboxylic Acids. Ind. Eng. Chem. Res. 2021, 60, 1356–1365. [Google Scholar] [CrossRef]
- Abdallah, M.M.; Müller, S.; González de Castilla, A.; Gurikov, P.; Matias, A.A.; Bronze, M.D.; Fernández, N. Physicochemical Characterization and Simulation of the Solid–Liquid Equilibrium Phase Diagram of Terpene-Based Eutectic Solvent Systems. Molecules 2021, 26, 1801. [Google Scholar] [CrossRef] [PubMed]
- Martins, M.A.R.; Silva, L.P.; Schaeffer, N.; Abranches, D.O.; Maximo, G.J.; Pinho, S.P.; Coutinho, J.A.P. Greener Terpene–Terpene Eutectic Mixtures as Hydrophobic Solvents. ACS Sustain. Chem. Eng. 2019, 7, 17414–17423. [Google Scholar] [CrossRef]
- Alhadid, A.; Safarov, J.; Mokrushina, L.; Müller, K.; Minceva, M. Carbon Dioxide Solubility in Nonionic Deep Eutectic Solvents Containing Phenolic Alcohols. Front. Chem. 2022, 10, 300. [Google Scholar] [CrossRef] [PubMed]
- Alhadid, A.; Mokrushina, L.; Minceva, M. Influence of the Molecular Structure of Constituents and Liquid Phase Non-Ideality on the Viscosity of Deep Eutectic Solvents. Molecules 2021, 26, 4208. [Google Scholar] [CrossRef]
- Alhadid, A.; Mokrushina, L.; Minceva, M. Design of Deep Eutectic Systems: A Simple Approach for Preselecting Eutectic Mixture Constituents. Molecules 2020, 25, 1077. [Google Scholar] [CrossRef]
- Alhadid, A.; Mokrushina, L.; Minceva, M. Modeling of Solid–Liquid Equilibria in Deep Eutectic Solvents: A Parameter Study. Molecules 2019, 24, 2334. [Google Scholar] [CrossRef]
- Das, S.; Mondal, A.; Reddy, C.M. Harnessing molecular rotations in plastic crystals: A holistic view for crystal engineering of adaptive soft materials. Chem. Soc. Rev. 2020, 49, 8878–8896. [Google Scholar] [CrossRef]
- Alarco, P.-J.; Abu-Lebdeh, Y.; Abouimrane, A.; Armand, M. The plastic-crystalline phase of succinonitrile as a universal matrix for solid-state ionic conductors. Nat. Mater. 2004, 3, 476–481. [Google Scholar] [CrossRef]
- Timmermans, J. Plastic crystals: A historical review. J. Phys. Chem. Solids 1961, 18, 1–8. [Google Scholar] [CrossRef]
- Lohmann, J.; Joh, R.; Gmehling, J. Solid–Liquid Equilibria of Viscous Binary Mixtures with Alcohols. J. Chem. Eng. Data. 1997, 42, 1170–1175. [Google Scholar] [CrossRef]
- Parks, G.S.; Huffman, H.M.; Barmore, M. Thermal data on organic compounds. XI. The heat capacities, entropies and free energies of ten compounds containing oxygen or nitrogen. J. Am. Chem. Soc. 1933, 55, 2733–2740. [Google Scholar] [CrossRef]
- Timmermans, J. Freezing points of organic compounds. VVI New determinations. Bull. Soc. Chim. Belg. 1952, 61, 393–402. [Google Scholar] [CrossRef]
- Miller, P. The Free Energy of Furfural and Some of Its Derivatives; Iowa State University: Ames, IA, USA, 1934. [Google Scholar]
- Mellan, I. Polyhydric Alcohols; Spartan Books: Washington, DC, USA, 1962. [Google Scholar]
- Jain, A.; Yang, G.; Yalkowsky, S.H. Estimation of Total Entropy of Melting of Organic Compounds. Ind. Eng. Chem. Res. 2004, 43, 4376–4379. [Google Scholar] [CrossRef]
- Sherwood, J.N. The Plastically Crystalline State: Orientationally Disordered Crystals; John Wiley & Sons: Hoboken, NJ, USA, 1979. [Google Scholar]
- Alizadeh, V.; Malberg, F.; Pádua, A.A.H.; Kirchner, B. Are There Magic Compositions in Deep Eutectic Solvents? Effects of Composition and Water Content in Choline Chloride/Ethylene Glycol from Ab Initio Molecular Dynamics. J. Phys. Chem. B 2020, 124, 7433–7443. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Deng, D.; Chen, Y.; Shan, H.; Ai, N. Solubilities and thermodynamic properties of CO2 in choline-chloride based deep eutectic solvents. J. Chem. Thermodyn. 2014, 75, 58–62. [Google Scholar] [CrossRef]
- Spittle, S.; Poe, D.; Doherty, B.; Kolodziej, C.; Heroux, L.; Haque, M.A.; Squire, H.; Cosby, T.; Zhang, Y.; Fraenza, C.; et al. Evolution of microscopic heterogeneity and dynamics in choline chloride-based deep eutectic solvents. Nat. Commun. 2022, 13, 219. [Google Scholar] [CrossRef]
- Agieienko, V.; Buchner, R. Correction: Is ethaline a deep eutectic solvent? PCCP 2022, 24, 10628. [Google Scholar] [CrossRef]
- Crespo, E.A.; Silva, L.P.; Lloret, J.O.; Carvalho, P.J.; Vega, L.F.; Llovell, F.; Coutinho, J.A.P. A methodology to parameterize SAFT-type equations of state for solid precursors of deep eutectic solvents: The example of cholinium chloride. PCCP 2019, 21, 15046–15061. [Google Scholar] [CrossRef]
- Agieienko, V.; Buchner, R. Is ethaline a deep eutectic solvent? PCCP 2022, 24, 5265–5268. [Google Scholar] [CrossRef]
- Alhadid, A.; Jandl, C.; Mokrushina, L.; Minceva, M. Experimental Investigation and Modeling of Cocrystal Formation in L-Menthol/Thymol Eutectic System. Cryst. Growth Des. 2021, 21, 6083–6091. [Google Scholar] [CrossRef]
- Alhadid, A.; Jandl, C.; Mokrushina, L.; Minceva, M. Cocrystal Formation in l-Menthol/Phenol Eutectic System: Experimental Study and Thermodynamic Modeling. Cryst. Growth Des. 2022, 22, 3973–3980. [Google Scholar] [CrossRef]
- Meng, X.; Ballerat-Busserolles, K.; Husson, P.; Andanson, J.-M. Impact of water on the melting temperature of urea + choline chloride deep eutectic solvent. New J. Chem. 2016, 40, 4492–4499. [Google Scholar] [CrossRef]
- Abranches, D.O.; Silva, L.P.; Martins, M.A.R.; Pinho, S.P.; Coutinho, J.A.P. Understanding the Formation of Deep Eutectic Solvents: Betaine as a Universal Hydrogen Bond Acceptor. ChemSusChem 2020, 13, 4916–4921. [Google Scholar] [CrossRef]
- Kazachenko, A.S.; Issaoui, N.; Medimagh, M.; Fetisova, O.Y.; Berezhnaya, Y.D.; Elsufev, E.V.; Al-Dossary, O.M.; Wojcik, M.J.; Xiang, Z.; Bousiakou, L.G. Experimental and theoretical study of the sulfamic acid-urea deep eutectic solvent. J. Mol. Liq. 2022, 363, 119859. [Google Scholar] [CrossRef]
- Granzow, B. Hydrogen bonding and phase transitions of a group of alcohols derived from 2,2-dimethylpropane. J. Mol. Struct. 1996, 381, 127–131. [Google Scholar] [CrossRef]
- Singh, N.B.; Glicksman, M.E. Physical properties of ultra-pure pivalic acid. Thermochim. Acta 1990, 159, 93–99. [Google Scholar] [CrossRef]
- Lohmann, J.; Joh, R.; Gmehling, J. Estimation of Enthalpies of Fusion, Melting Temperatures, Enthalpies of Transition, and Transition Temperatures of Pure Compounds from Experimental Binary Solid−Liquid Equilibrium Data of Eutectic Systems. J. Chem. Eng. Data 1997, 42, 1176–1180. [Google Scholar] [CrossRef]
- Prausnitz, J.M.; Lichtenthaler, R.N.; de Azevedo, E.G. Molecular Thermodynamics of Fluid-Phase Equilibria; Prentice Hall PTR: Upper Saddle River, NJ, USA, 1999. [Google Scholar]



| Component 1 | Component 2 | Tm,2/K | ||
|---|---|---|---|---|
| L-menthol | Pivalic acid a,b | 308.7 | 260.6 | −48.1 |
| Cyclohexane carboxylic acid [35] | 299.4 | 265.0 | −34.4 | |
| Capric acid [35] | 303.9 | 279.0 | −24.9 | |
| Neopentyl alcohol a,b | 328.9 | 259.2 c | −69.7 | |
| Thymol [53] | 322.7 | 271.7 | −51.0 | |
| Phenol [54] | 313.9 | 261.3 | −52.6 | |
| Neopentyl glycol a,b | 401.2 | 291.8 | −109.4 | |
| Camphor a [32] | 450.4 | 275.7 | −174.7 | |
| Borneol a [32] | 480.6 | 286.7 | −193.9 | |
| Sobrerol [32] | 420.2 | – d | – | |
| Choline chloride a | Neopentyl glycol a,b | 401.2 | 305.1 | −96.1 |
| Urea [55] | 405.2 | 297.7 | −107.5 | |
| Betaine | Urea [56] | 405.2 | 359.3 | −45.9 |
| Sulfamic acid | Urea [57] | 405.2 | 351.1 | −54.1 |
| Compound | Ttr/K | Δhtr/kJ mol−1 | Tm/K | Δhm/kJ mol−1 | ||||
|---|---|---|---|---|---|---|---|---|
| This work | Literature | This work | Literature | This work | Literature | This work | Literature | |
| Neopentyl alcohol | 235.9 ± 0.1 | 242.1 a | 4.46 ± 0.06 | 4.6 a | 328.9 ± 1.2 | 328.1 a | 4.01 ± 0.03 | 3.5 a |
| Pivalic acid | 279.7 ± 0.1 | 278.3 b | 7.99 ± 0.32 | 8.18 b | 308.7 ± 0.2 | 309.1 b | 2.15 ± 0.13 | 2.27 b |
| Neopentyl glycol | 314.7 ± 0.1 | 315.2 a | 12.86 ± 0.20 | 12.8 a | 401.2 ± 0.1 | 402.5 a | 4.16 ± 0.06 | 4.3 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alhadid, A.; Nasrallah, S.; Mokrushina, L.; Minceva, M. Design of Deep Eutectic Systems: Plastic Crystalline Materials as Constituents. Molecules 2022, 27, 6210. https://doi.org/10.3390/molecules27196210
Alhadid A, Nasrallah S, Mokrushina L, Minceva M. Design of Deep Eutectic Systems: Plastic Crystalline Materials as Constituents. Molecules. 2022; 27(19):6210. https://doi.org/10.3390/molecules27196210
Chicago/Turabian StyleAlhadid, Ahmad, Sahar Nasrallah, Liudmila Mokrushina, and Mirjana Minceva. 2022. "Design of Deep Eutectic Systems: Plastic Crystalline Materials as Constituents" Molecules 27, no. 19: 6210. https://doi.org/10.3390/molecules27196210
APA StyleAlhadid, A., Nasrallah, S., Mokrushina, L., & Minceva, M. (2022). Design of Deep Eutectic Systems: Plastic Crystalline Materials as Constituents. Molecules, 27(19), 6210. https://doi.org/10.3390/molecules27196210






