Stability Enhancement of Anthocyanins from Blackcurrant (Ribes Nigrum L.) Pomace through Intermolecular Copigmentation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Dried Blackcurrant Pomace (DBP)
2.3. Extraction of Anthocyanins from Dried Blackcurrant Pomace (DBP)
2.4. Purification of Anthocyanin Extract
2.5. Preparation of Buffer Solutions
2.6. Pigmentation Reactions and Stability Studies
2.6.1. Copigmentation Reactions
2.6.2. Stability Studies
2.7. UV-Vis Spectrophotometry and Color Measurements
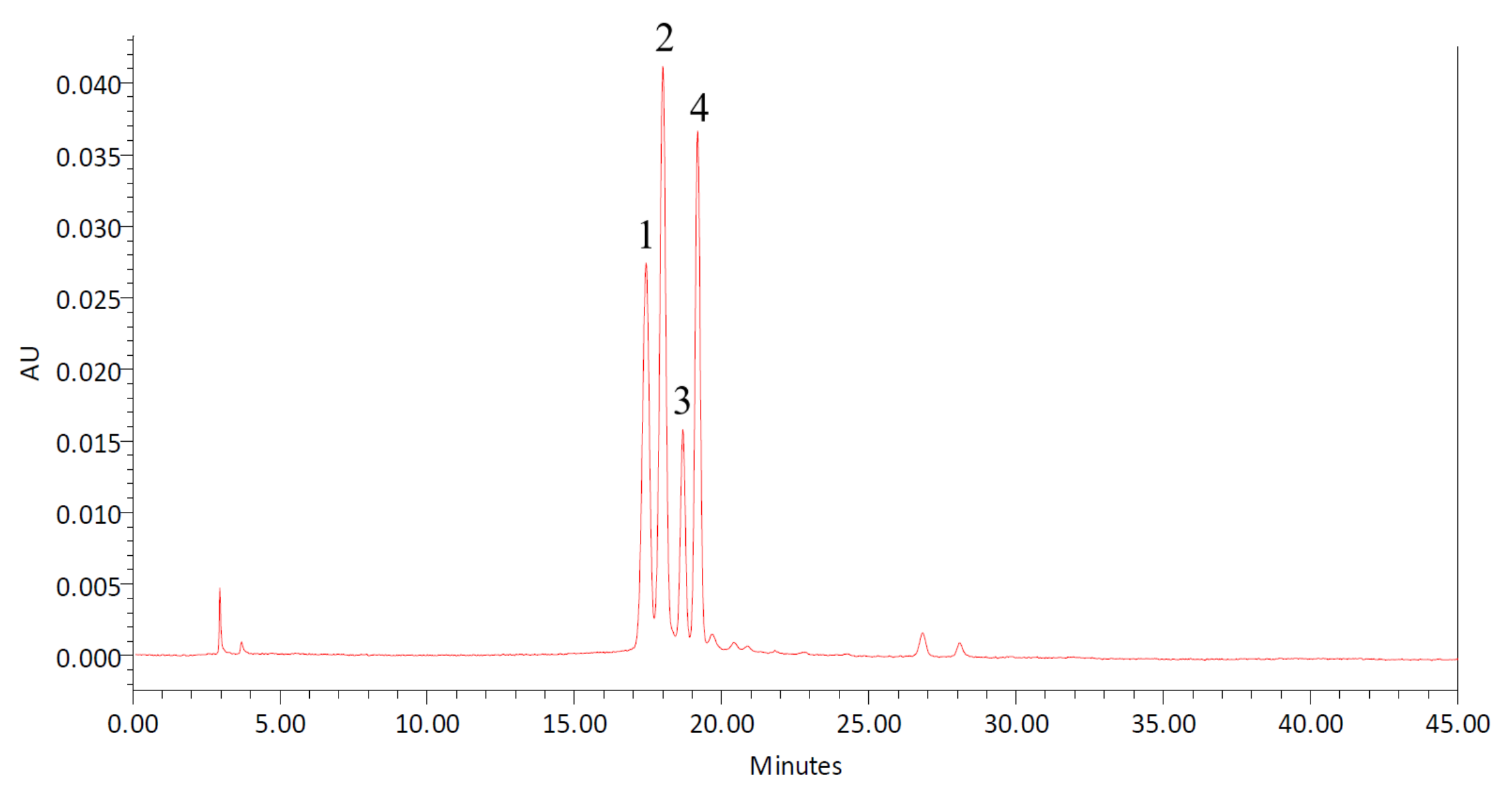
2.8. HPLC Analysis of Anthocyanins
2.9. Kinetic Study
2.10. DPPH Radical Scavenging Activity
2.11. Statistical Analysis
3. Results and Discussion
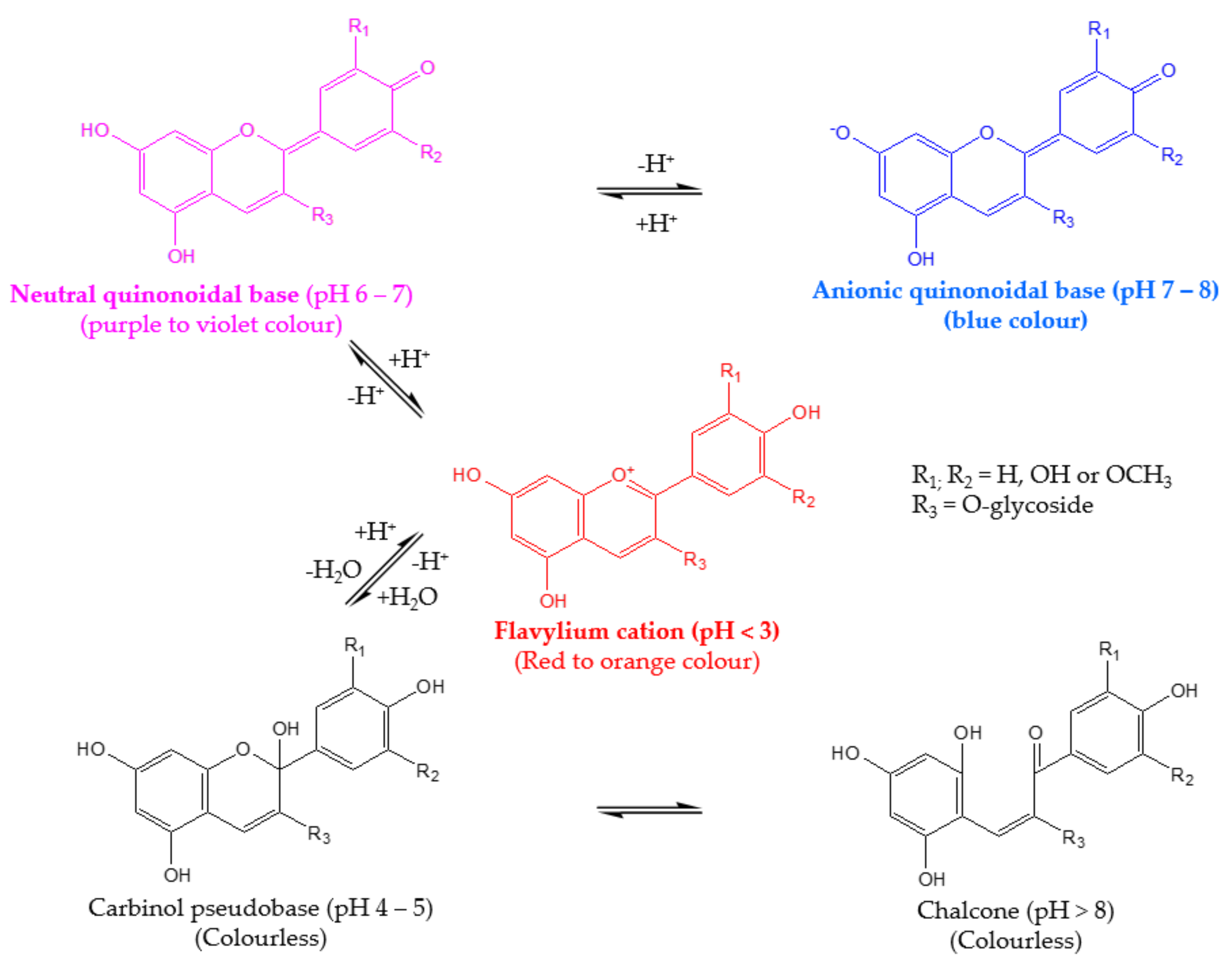
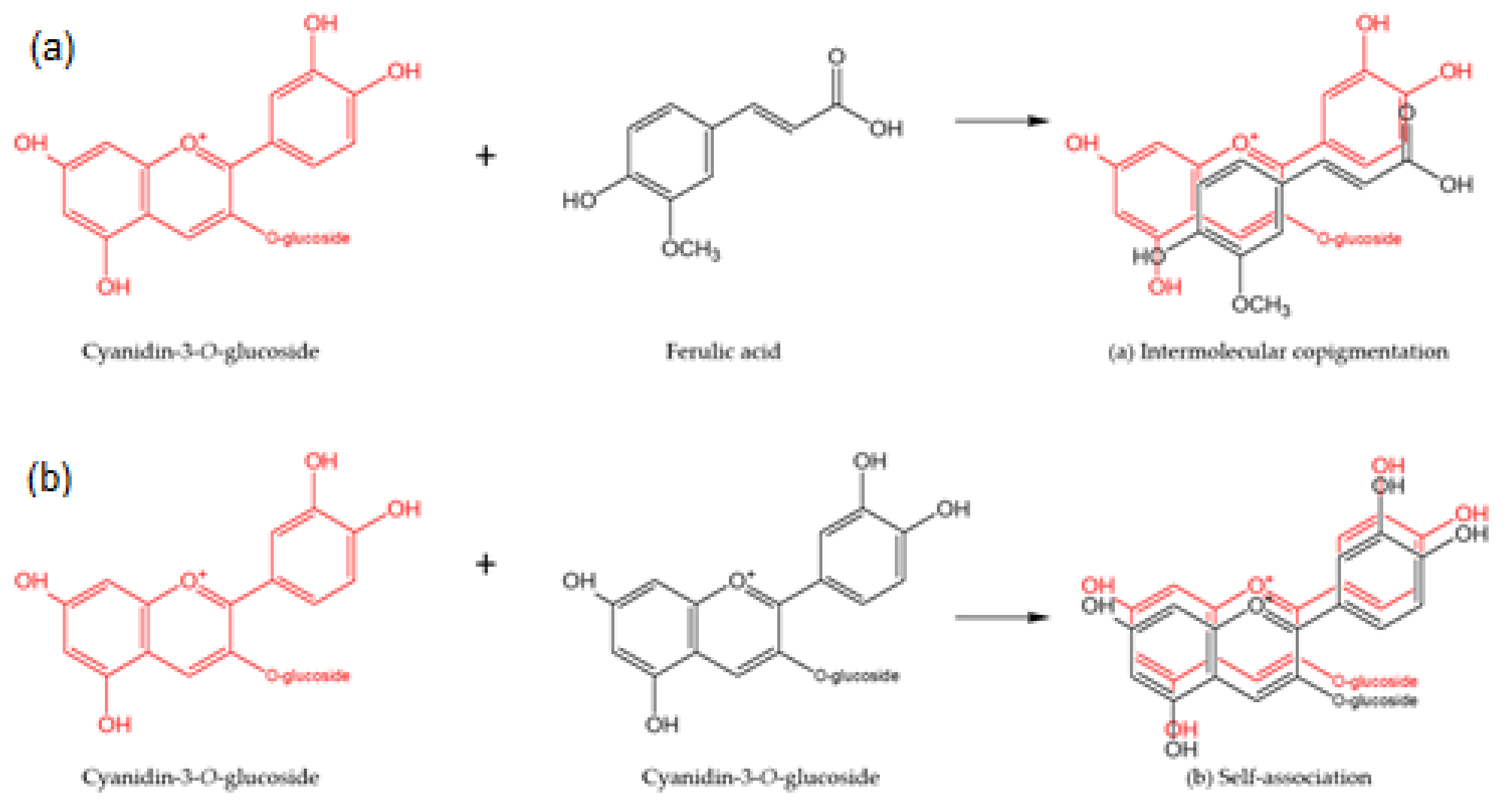
3.1. Effect of Copigmentation on UV-Vis Absorption Spectra
Copigmentation Effect in Buffer Solutions at pH 3.0 and pH 6.0
3.2. Copigmentation Stability during Storage
3.2.1. Effect on Anthocyanin Stability
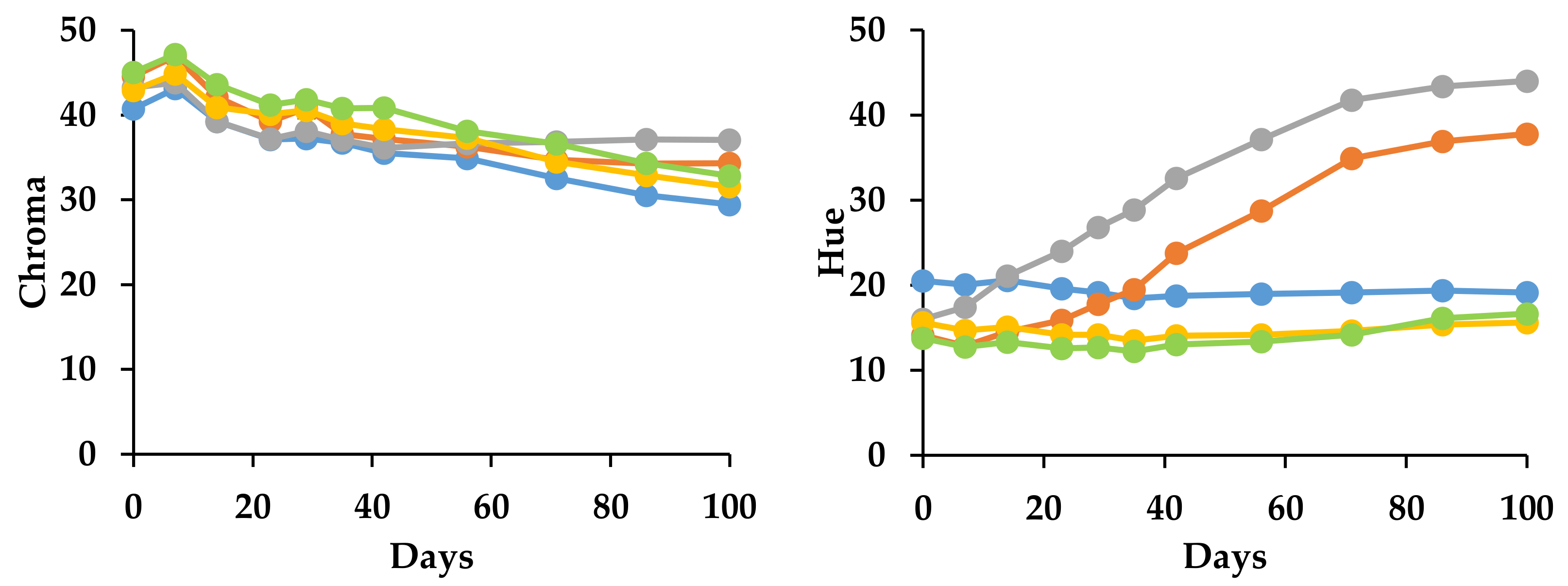
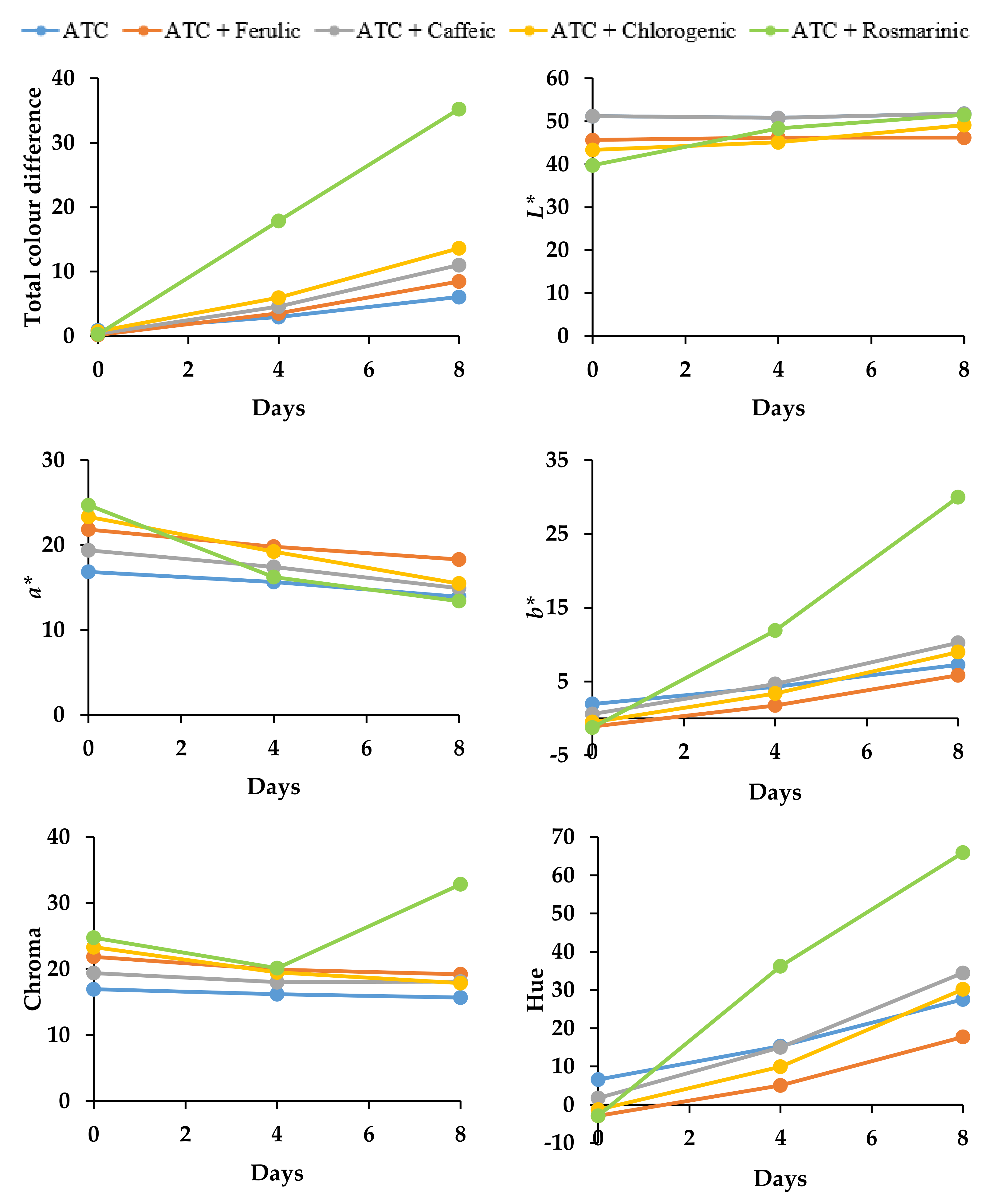
3.2.2. Effect on Color Stability
3.2.3. Effect on Antioxidant Capacity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Institute of Food Technologists. Color Goes Natural. 2018. Available online: https://www.ift.org/news-and-publications/food-technologymagazine/issues/2019/october/columns/ingredients-color-goes-natural (accessed on 18 August 2022).
- Carocho, M.; Barreiro, M.F.; Morales, P.; Ferreira, I.C. Adding molecules to food, pros and cons: A review on synthetic and natural food additives. Compr. Rev. Food Sci. Food Saf. 2014, 13, 377–399. [Google Scholar] [CrossRef]
- Cortez, R.; Luna-Vital, D.A.; Margulis, D.; Gonzalez de Mejia, E. Natural pigments: Stabilization methods of anthocyanins for food applications. Compr. Rev. Food Sci. Food Saf. 2017, 16, 180–198. [Google Scholar] [CrossRef]
- Ngamwonglumlert, L.; Devahastin, S.; Chiewchan, N. Natural colorants: Pigment stability and extraction yield enhancement via utilization of appropriate pretreatment and extraction methods. Crit. Rev. Food Sci. Nutr. 2017, 57, 3243–3259. [Google Scholar] [CrossRef]
- Ranaweera, S.J.; Ampemohotti, A.A.L.T.; Arachchige, U.S. Advantages and considerations for the applications of natural food pigments in the food industry. J. Res. Technol. Eng. 2020, 1, 8–18. [Google Scholar]
- Bueno, J.M.; Sáez-Plaza, P.; Ramos-Escudero, F.; Jiménez, A.M.; Fett, R.; Asuero, A.G. Analysis and antioxidant capacity of anthocyanin pigments. Part II: Chemical structure, colour, and intake of anthocyanins. Crit. Rev. Anal. Chem. 2012, 42, 126–151. [Google Scholar] [CrossRef]
- Khoo, H.E.; Azlan, A.; Tang, S.T.; Lim, S.M. Anthocyanidins and anthocyanins: Colored pigments as food, pharmaceutical ingredients, and the potential health benefits. Food Nutr. Res. 2017, 61, 1361779. [Google Scholar] [CrossRef]
- Cruz, L.; Basílio, N.; Mateus, N.; De Freitas, V.; Pina, F. Natural and Synthetic Flavylium-Based Dyes: The Chemistry Behind the Color. Chem. Rev. 2021, 122, 1416–1481. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef]
- Sytar, O.; Kosyan, A.; Taran, N.; Smetanska, I. Anthocyanin’s as marker for selection of buckwheat plants with high rutin content. Gesunde Pflanz. 2014, 66, 165–169. [Google Scholar] [CrossRef]
- Riaz, M.; Zia-Ul-Haq, M.; Saad, B. Biosynthesis and stability of anthocyanins. In Anthocyanins and Human Health: Biomolecular and Therapeutic Aspects; Springer: Cham, Switzerland, 2016; pp. 71–86. [Google Scholar] [CrossRef]
- Mbarki, S.; Sytar, O.; Zivcak, M.; Abdelly, C.; Cerda, A.; Brestic, M. Anthocyanins of coloured wheat genotypes in specific response to salstress. Molecules 2018, 23, 1518. [Google Scholar] [CrossRef] [Green Version]
- Enaru, B.; Drețcanu, G.; Pop, T.D.; Stǎnilǎ, A.; Diaconeasa, Z. Anthocyanins: Factors Affecting Their Stability and Degradation. Antioxidants 2021, 10, 1967. [Google Scholar] [CrossRef]
- Wahyuningsih, S.; Wulandari, L.; Wartono, M.W.; Munawaroh, H.; Ramelan, A.H. The effect of pH and color stability of anthocyanin on food colorant. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2017; Volume 193, p. 012047. [Google Scholar]
- He, J.; Giusti, M.M. Anthocyanins: Natural colorants with health-promoting properties. Annu. Rev. Food Sci. Technol. 2010, 1, 163–187. [Google Scholar] [CrossRef]
- Wrolstad, R.E.; Culver, C.A. Alternatives to those artificial FD&C food colorants. Annu. Rev. Food Sci. Technol. 2012, 3, 59–77. [Google Scholar] [CrossRef]
- Rose, P.M.; Cantrill, V.; Benohoud, M.; Tidder, A.; Rayner, C.M.; Blackburn, R.S. Application of anthocyanins from blackcurrant (Ribes nigrum L.) fruit waste as renewable hair dyes. J. Agric. Food Chem. 2018, 66, 6790–6798. [Google Scholar] [CrossRef]
- Trouillas, P.; Sancho-García, J.C.; De Freitas, V.; Gierschner, J.; Otyepka, M.; Dangles, O. Stabilizing and modulating colour by copigmentation: Insights from theory and experiment. Chem. Rev. 2016, 116, 4937–4982. [Google Scholar] [CrossRef]
- Houghton, A.; Appelhagen, I.; Martin, C. Natural blues: Structure meets function in anthocyanins. Plants 2021, 10, 726. [Google Scholar] [CrossRef]
- Harborne, J.B. The Flavonoids: Advances in Research since 1980; Springer: Berlin/Heidelberg, Germany, 2013. [Google Scholar]
- Asen, S.; Stewart, R.N.; Norris, K.H. Copigmentation of anthocyanins in plant tissues and its effect on colour. Phytochemistry 1972, 11, 1139–1144. [Google Scholar] [CrossRef]
- Brouillard, R. Chemical Structure of Anthocyanins; Academic Press: New York, NY, USA, 1982; Volume 1. [Google Scholar]
- Mazza, G.; Brouillard, R. Recent developments in the stabilization of anthocyanins in food products. Food Chem. 1987, 25, 207–225. [Google Scholar] [CrossRef]
- Castañeda-Ovando, A.; de Lourdes Pacheco-Hernández, M.; Páez-Hernández, M.E.; Rodríguez, J.A.; Galán-Vidal, C.A. Chemical studies of anthocyanins: A review. Food Chem. 2009, 113, 859–871. [Google Scholar] [CrossRef]
- Rein, M.J.; Heinonen, M. Stability and enhancement of berry juice colour. J. Agric. Food Chem. 2004, 52, 3106–3114. [Google Scholar] [CrossRef]
- Sui, X.; Dong, X.; Zhou, W. Combined effect of pH and high temperature on the stability and antioxidant capacity of two anthocyanins in aqueous solution. Food Chem. 2014, 163, 163–170. [Google Scholar] [CrossRef]
- Gras, C.C.; Nemetz, N.; Carle, R.; Schweiggert, R.M. Anthocyanins from purple sweet potato (Ipomoea batatas (L.) Lam.) and their colour modulation by the addition of phenolic acids and food-grade phenolic plant extracts. Food Chem. 2017, 235, 265–274. [Google Scholar] [CrossRef]
- Lambert, S.G.; Asenstorfer, R.E.; Williamson, N.M.; Iland, P.G.; Jones, G.P. Copigmentation between malvidin-3-glucoside and some wine constituents and its importance to colour expression in red wine. Food Chem. 2011, 125, 106–115. [Google Scholar] [CrossRef]
- Azman, E.M.; Charalampopoulos, D.; Chatzifragkou, A. Acetic acid buffer as extraction medium for free and bound phenolics from dried blackcurrant (Ribes nigrum L.) skins. J. Food Sci. 2020, 85, 3745–3755. [Google Scholar] [CrossRef]
- Toscano Martínez, H.; Gagneten, M.; Díaz-Calderón, P.; Enrione, J.; Salvatori, D.; Schebor, C.; Leiva, G. Natural food colorant from blackcurrant spray-dried powder obtained by enzymatic treatment: Characterization and acceptability. J. Food Processing Preserv. 2021, 45, e15011. [Google Scholar] [CrossRef]
- Stevenson, D.E.; Wibisono, R.; Jensen, D.J.; Stanley, R.A.; Cooney, J.M. Direct acylation of flavonoid glycosides with phenolic acids catalysed by Candida antarctica lipase B (Novozym 435®). Enzym. Microb. Technol. 2006, 39, 1236–1241. [Google Scholar] [CrossRef]
- Rodriguez-Saona, L.E.; Wrolstad, R.E. Anthocyanins. Extraction, Isolation and Purification of Anthocyanins. In Current Protocols in Food Analytical Chemistry; Wrolstad, R.E., Ed.; John Wiley & Sons: New York, NY, USA, 2001. [Google Scholar] [CrossRef]
- Sigma. Buffer Solutions (Citric Acid–Sodium Citrate Buffer Solutions). 2018. Available online: https://www.sigmaaldrich.com/MY/en/technical-documents/protocol/protein-biology/protein-concentration-and-buffer-exchange/buffer-reference-center (accessed on 20 March 2021).
- Shikov, V.; Kammerer, D.R.; Mihalev, K.; Mollov, P.; Carle, R. Heat stability of strawberry anthocyanins in model solutions containing natural copigments extracted from rose (Rosa damascena Mill.) petals. J. Agric. Food Chem. 2008, 56, 8521–8526. [Google Scholar] [CrossRef]
- Lee, J.; Durst, R.W.; Wrolstad, R.E. Determination of total monomeric anthocyanin pigment content of fruit juices, beverages, natural colourants, and wines by the pH differential method: Collaborative study. J. AOAC Int. 2005, 88, 1269–1278. [Google Scholar] [CrossRef]
- Fuleki, T.; Francis, F.J. Quantitative methods for anthocyanins. J. Food Sci. 1968, 33, 266–274. [Google Scholar] [CrossRef]
- Katsaboxakis, K.; Papanicolaou, D.; Melanitou, M. Stability of pigmented orange anthocyanins in model and real food systems [food colourants]. Ital. J. Food Sci. 1998, 10, 17–25. [Google Scholar]
- Wrolstad, R.E.; Smith, D.E. Colour analysis. In Food Analysis; Springer: Berlin/Heidelberg, Germany, 2010; pp. 573–586. [Google Scholar]
- Azman, E.M.; Nor, N.D.M.; Charalampopoulos, D.; Chatzifragkou, A. Effect of acidified water on phenolic profile and antioxidant activity of dried blackcurrant (Ribes nigrum L.) pomace extracts. LWT 2022, 154, 112733. [Google Scholar] [CrossRef]
- Remini, H.; Mertz, C.; Belbahi, A.; Achir, N.; Dornier, M.; Madani, K. Degradation kinetic modelling of ascorbic acid and colour intensity in pasteurised blood orange juice during storage. Food Chem. 2015, 173, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Mizzi, L.; Chatzitzika, C.; Gatt, R.; Valdramidis, V. HPLC analysis of phenolic compounds and flavonoids with overlapping peaks. Food Technol. Biotechnol. 2020, 58, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Juadjur, A.; Winterhalter, P. Development of a novel adsorptive membrane chromatographic method for the fractionation of polyphenols from bilberry. J. Agric. Food Chem. 2012, 60, 2427–2433. [Google Scholar] [CrossRef]
- Foster, R. Organic Charge-Transfer Complexes; Academic Press: London, UK, 1969. [Google Scholar]
- Parisa, S.; Reza, H.; Elham, G.; Rashid, J. Effect of heating, UV irradiation and pH on stability of the anthocyanin copigment complex. Pak. J. Biol. Sci. 2007, 10, 267–272. [Google Scholar] [CrossRef]
- Eiro, M.J.; Heinonen, M. Anthocyanin colour behavior and stability during storage: Effect of intermolecular copigmentation. J. Agric. Food Chem. 2002, 50, 7461–7466. [Google Scholar] [CrossRef]
- Brouillard, R.; Mazza, G.; Saad, Z.; Albrecht-Gary, A.M.; Cheminat, A. The co-pigmentation reaction of anthocyanins: A microprobe for the structural study of aqueous solutions. J. Am. Chem. Soc. 1989, 111, 2604–2610. [Google Scholar] [CrossRef]
- Boulton, R. The copigmentation of anthocyanins and its role in the colour of red wine: A critical review. Am. J. Enol. Vitic. 2001, 52, 67–87. [Google Scholar]
- Sinela, A.; Rawat, N.; Mertz, C.; Achir, N.; Fulcrand, H.; Dornier, M. Anthocyanins degradation during storage of Hibiscus sabdariffa extract and evolution of its degradation products. Food Chem. 2017, 214, 234–241. [Google Scholar] [CrossRef]
- Davies, A.J.; Mazza, G. Copigmentation of simple and acylated anthocyanins with colorless phenolic compounds. J. Agric. Food Chem. 1993, 41, 716–720. [Google Scholar] [CrossRef]
- Mazza, G.; Brouillard, R. The mechanism of copigmentation of anthocyanins in aqueous solutions. Phytochemistry 1990, 29, 1097–1102. [Google Scholar] [CrossRef]
- Brouillard, R.; Wigand, M.C.; Dangles, O.; Cheminat, A. pH and solvent effects on the copigmentation reaction of malvin with polyphenols, purine and pyrimidine derivatives. J. Chem. Soc. Perkin Trans. 2 1991, 8, 1235–1241. [Google Scholar] [CrossRef]
- Chatham, L.A.; Howard, J.E.; Juvik, J.A. A natural colorant system from corn: Flavone-anthocyanin copigmentation for altered hues and improved shelf life. Food Chem. 2020, 310, 125734. [Google Scholar] [CrossRef] [PubMed]
- Qian, B.J.; Liu, J.H.; Zhao, S.J.; Cai, J.X.; Jing, P. The effects of gallic/ferulic/caffeic acids on colour intensification and anthocyanin stability. Food Chem. 2017, 228, 526–532. [Google Scholar] [CrossRef] [PubMed]
- Kondo, T.; Ueda, M.; Isobe, M.; Goto, T. A new molecular mechanism of blue color development with protocyanin, a supramolecular pigment from cornflower, Centaurea cyanus. Tetrahedron Lett. 1998, 39, 8307–8310. [Google Scholar] [CrossRef]
- Welch, C.R.; Wu, Q.; Simon, J.E. Recent advances in anthocyanin analysis and characterization. Curr. Anal. Chem. 2008, 4, 75–101. [Google Scholar] [CrossRef]
- Jaiswal, V.; DerMarderosian, A.; Porter, J.R. Anthocyanins and polyphenol oxidase from dried arils of pomegranate (Punica granatum L.). Food Chem. 2010, 118, 11–16. [Google Scholar] [CrossRef]
- Yang, J.; He, X.; Zhao, D. Factors affecting phytochemical stability. In Handbook of Plant Food Phytochemicals: Sources, Stability and Extraction; Wiley-Blackwell: Hoboken, NJ, USA, 2013; pp. 332–374. [Google Scholar]
- Azman, E.M.; House, A.; Charalampopoulos, D.; Chatzifragkou, A. Effect of dehydration on phenolic compounds and antioxidant activity of blackcurrant (Ribes nigrum L.) pomace. Int. J. Food Sci. Technol. 2021, 56, 600–607. [Google Scholar] [CrossRef]
- Caballero, B.; Trugo, L.C.; Finglas, P.M. Encyclopedia of Food Sciences and Nutrition; Academic Press: Cambridge, MA, USA, 2003. [Google Scholar]
- Kader, F.; Rovel, B.; Girardin, M.; Metche, M. Mechanism of browning in fresh highbush blueberry fruit (Vaccinium corymbosum L.). Role of blueberry polyphenol oxidase, chlorogenic acid and anthocyanins. J. Sci. Food Agric. 1997, 74, 31–34. [Google Scholar] [CrossRef]
- Moldovan, B.; David, L. Influence of temperature and preserving agents on the stability of cornelian cherries anthocyanins. Molecules 2014, 19, 8177–8188. [Google Scholar] [CrossRef]
- Hurtado, N.H.; Morales, A.L.; González-Miret, M.L.; Escudero-Gilete, M.L.; Heredia, F.J. Colour, pH stability and antioxidant activity of anthocyanin rutinosides isolated from tamarillo fruit (Solanum betaceum Cav.). Food Chem. 2009, 117, 88–93. [Google Scholar] [CrossRef]
- Seeram, N.P.; Bourquin, L.D.; Nair, M.G. Degradation products of cyanidin glycosides from tart cherries and their bioactivities. J. Agric. Food Chem. 2001, 49, 4924–4929. [Google Scholar] [CrossRef] [PubMed]
- Terefe, N.S.; Netzel, G.A.; Netzel, M.E. Copigmentation with sinapic acid improves the stability of anthocyanins in high-pressure-processed strawberry purees. J. Chem. 2019, 2019, 3138608. [Google Scholar] [CrossRef]




| Samples | (a) pH 3.0 | (b) pH 6.0 | ||||
|---|---|---|---|---|---|---|
| λmax (nm) | Amax | ∆A (%) | λmax (nm) | Amax | ∆A (%) | |
| ATC | 519 | 0.6189 | – | 535 | 0.2641 | – |
| ATC + Ferulic | 524 | 0.7263 | 17.4 ± 0.4 b | 536 | 0.3484 | 31.9 ± 1.5 c |
| ATC + Caffeic | 522 | 0.6664 | 7.7 ± 0.2 c | 536 | 0.3098 | 17.3 ± 0.7 d |
| ATC + Chlorogenic | 523 | 0.6632 | 7.2 ± 0.1 c | 544 | 0.3956 | 49.8 ± 1.9 b |
| ATC + Rosmarinic | 525 | 0.7438 | 20.2 ± 0.3 a | 546 | 0.4591 | 73.8 ± 2.1 a |
| (a) pH 3.0 | ATC | ATC + Ferulic | ATC + Caffeic | ATC + Chlorogenic | ATC + Rosmarinic | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| k | t1/2 (days) | R2 | k | t1/2 (days) | R2 | k | t1/2 (days) | R2 | k | t1/2 (days) | R2 | k | t1/2 (days) | R2 | |
| D3G | 0.230 | 51.2 ± 0.7 Ba | 0.991 | 0.558 | 21.9 ± 0.5 Bc | 0.999 | 0.618 | 18.2 ± 0.2 Bd | 0.989 | 0.231 | 50.4 ± 0.8 Ba | 0.990 | 0.258 | 46.3 ± 1.0 Bb | 0.994 |
| D3R | 0.303 | 61.8 ± 1.3 Ba | 0.993 | 0.868 | 22.7 ± 0.1 Bd | 0.999 | 0.986 | 18.4 ± 0.1 Be | 0.988 | 0.319 | 57.9 ± 0.6 Bb | 0.995 | 0.377 | 52.5 ± 0.3 Bc | 0.996 |
| C3G | 0.082 | 208.2 ± 21.4 Aa | 0.991 | 0.192 | 90.5 ± 4.2 Ab | 0.997 | 0.184 | 90.6 ± 1.6 Ab | 0.994 | 0.085 | 216.1 ± 44.0 Aa | 0.990 | 0.077 | 221.8 ± 3.5 Aa | 0.987 |
| C3R | 0.168 | 49.0 ± 1.3 Ba | 0.992 | 0.455 | 20.0 ± 0.1 Bc | 0.998 | 0.476 | 16.3 ± 0.1 Bd | 0.999 | 0.167 | 49.1 ± 0.0 Ba | 0.995 | 0.198 | 46.0 ± 0.5 Bb | 0.999 |
| Total ATC | 0.777 | 71.8 ± 1.8 a | 0.994 | 2.065 | 28.3 ± 0.1 c | 0.999 | 2.346 | 22.9 ± 0.2 d | 0.994 | 0.781 | 71.0 ± 0.1 a | 0.995 | 0.877 | 66.1 ± 0.9 b | 0.995 |
| (b) pH 6.0 | ATC | ATC + Ferulic | ATC + Caffeic | ATC + Chlorogenic | ATC + Rosmarinic | ||||||||||
| k | t1/2 (days) | R2 | k | t1/2 (days) | R2 | k | t1/2 (days) | R2 | k | t1/2 (days) | R2 | k | t1/2 (days) | R2 | |
| D3G | 2.252 | 4.7 ± 0.1 Ba | 0.996 | 2.315 | 4.4 ± 0.1 Ca | 0.995 | 2.817 | 3.2 ± 0.1 Bc | 0.998 | 2.365 | 3.8 ± 0.1 Cb | 0.991 | 3.424 | 1.6 ± 0.1 Cd | 0.981 |
| D3R | 3.229 | 5.0 ± 0.1 Ba | 0.995 | 3.044 | 5.2 ± 0.3 Ba | 0.997 | 4.489 | 3.1 ± 0.1 Bc | 0.996 | 3.550 | 4.0 ± 0.1 Cb | 0.993 | 5.516 | 1.7 ± 0.1 Cd | 0.984 |
| C3G | 0.920 | 9.1 ± 0.6 Aa | 0.996 | 0.853 | 9.6 ± 0.1 Aa | 0.998 | 1.158 | 6.8 ± 0.2 Ab | 0.995 | 0.886 | 8.9 ± 0.2 Aa | 0.994 | 1.257 | 5.5 ± 0.1 Ac | 0.992 |
| C3R | 2.067 | 4.9 ± 0.1 Bb | 0.996 | 1.709 | 5.7 ± 0.1 Ba | 0.994 | 3.218 | 2.9 ± 0.1 Bd | 0.985 | 2.032 | 4.7 ± 0.1 Bc | 0.994 | 3.253 | 2.4 ± 0.1 Be | 1.000 |
| Total ATC | 8.459 | 5.3 ± 0.1 a | 0.996 | 8.053 | 5.5 ± 0.1 a | 0.997 | 11.158 | 3.6 ± 0.1 c | 0.996 | 8.833 | 4.6 ± 0.1 b | 0.994 | 12.577 | 2.3 ± 0.1 d | 0.989 |
| (a) pH 3.0 | ATC | Ferulic | Caffeic | Chlorogenic | Rosmarinic |
|---|---|---|---|---|---|
| Day 0 |  |  |  |  |  |
| Day 30 |  |  |  |  |  |
| Color Ret (%) at Day 30 | 82.8 ±0.5 bc | 79.8 ± 0.3 d | 81.7 ± 0.3 cd | 86.3 ± 1.3 a | 84.8 ± 0.4 ab |
| Day 90 |  |  |  |  |  |
| Color Ret (%) at Day 90 | 60.0 ± 0.3 c | 63.2 ± 0.4 b | 75.2 ± 0.0 a | 60.9 ± 0.4 c | 57.8 ± 0.9 d |
| (b) pH 6.0 | ATC | Ferulic | Caffeic | Chlorogenic | Rosmarinic |
| Day 0 |  |  |  |  |  |
| Day 4 |  |  |  |  |  |
| Color Ret (%) at Day 4 | 94.1 ± 0.3 a | 93.1 ± 0.2 a | 88.7 ± 0.0 b | 86.4 ± 1.3 b | 60.2 ± 1.4 c |
| Day 8 |  |  |  |  |  |
| Color Ret (%) at Day 8 | 84.5 ± 1.8 a | 82.6 ± 0.1 ab | 76.2 ± 2.0 b | 64.5 ± 0.4 c | 48.5 ± 2.6 d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azman, E.M.; Yusof, N.; Chatzifragkou, A.; Charalampopoulos, D. Stability Enhancement of Anthocyanins from Blackcurrant (Ribes Nigrum L.) Pomace through Intermolecular Copigmentation. Molecules 2022, 27, 5489. https://doi.org/10.3390/molecules27175489
Azman EM, Yusof N, Chatzifragkou A, Charalampopoulos D. Stability Enhancement of Anthocyanins from Blackcurrant (Ribes Nigrum L.) Pomace through Intermolecular Copigmentation. Molecules. 2022; 27(17):5489. https://doi.org/10.3390/molecules27175489
Chicago/Turabian StyleAzman, Ezzat Mohamad, Nurhayati Yusof, Afroditi Chatzifragkou, and Dimitris Charalampopoulos. 2022. "Stability Enhancement of Anthocyanins from Blackcurrant (Ribes Nigrum L.) Pomace through Intermolecular Copigmentation" Molecules 27, no. 17: 5489. https://doi.org/10.3390/molecules27175489
APA StyleAzman, E. M., Yusof, N., Chatzifragkou, A., & Charalampopoulos, D. (2022). Stability Enhancement of Anthocyanins from Blackcurrant (Ribes Nigrum L.) Pomace through Intermolecular Copigmentation. Molecules, 27(17), 5489. https://doi.org/10.3390/molecules27175489







