Synthesis of Sulfonamides Incorporating Piperidinyl-Hydrazidoureido and Piperidinyl-Hydrazidothioureido Moieties and Their Carbonic Anhydrase I, II, IX and XII Inhibitory Activity
Abstract
:1. Introduction
2. Results
2.1. Chemistry
2.2. Carbonic Anhydrase Inhibition
2.3. Molecular Docking
3. Materials and Methods
3.1. Chemistry
3.1.1. General Procedure for the Preparation of Benzenesulfonamidohydrazido Ureas (5a–u)
- N-(2-Chlorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxamide (5b). Yield 67% m.p. 214–215 °C. ESIMS (m/z): 480, 482 (M+H)+.1H NMR (DMSO-d6): δ 1.58 (m, 2H, CH2), 1.70, 1.94 (m, 2H, CH2), 2.53, 2.90 (m, 2H, CH2), 3.10, 3.51 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.03 (m, 1H, Ar), 7.28 (m, 1H, Ar), 7.43 (m, 1H, Ar), 7.46 (s, 2H, NH2), 7.57 (d, J = 7.5 Hz, 2H, Ar), 7.87 (d, J = 8.5 Hz, 2H, Ar), 8.07 (m, 1H, Ar), 8.17 (s, 1H, NH), 8.74 (s, 1H, NH), 9.83 (s, 1H, NH). IR (Nujol) 3334, 1608 cm−1. Elemental analysis: calculated for C20H22ClN5O5S (479.94) %C 50.05, %H 4.62, %N 14.59, found %C 50.09, %H 4.60 %N 14.64.
- N-(4-Chlorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxamide (5d). Yield 72% m.p. 209–210 °C. ESIMS (m/z): 497 (M+H)+. 1H NMR (DMSO-d6): δ 1.55 (m, 2H, CH2), 1.74, 2.32 (m, 2H, CH2), 2.50, 2.88 (m, 2H, CH2), 3.06, 3.49 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.29 (m, 1H, Ar), 7.44 (m, 4H, Ar and NH2), 7.57 (m, 3H, Ar), 7.87 (d, J = 8.5 Hz, 2H, Ar), 8.07 (s, 1H, NH), 9.01 (s, 1H, NH), 9.68 (s, 1H, NH). IR (Nujol) 3303, 3161, 1615 cm−1. Elemental analysis: calculated for C20H22ClN5O5S (496.00) %C 48.43, %H 4.47, %N 14.12, found %C 48.48, %H 4.46 %N 14.17.
- N-(2-fluorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxamide (5f). Yield 82% m.p. 229–230 °C. ESIMS (m/z): 464 (M+H)+. 1H NMR (DMSO-d6): δ 1.57 (br s, 2H, CH2) 1.69, 1.84 (br s, 2H, CH2), 2.53, 2.90 (br s, 2H, CH2), 3.10, 3.51 (br s, 2H, CH2), 4.44 (br s, 1H, CH), 6.99 (m, 1H, Ar), 7.13 (m, 1H, Ar), 7.22 (m, 1H, Ar), 7.45 (s, 2H, NH2), 7.56 (d, J = 8.5 Hz, 2H, Ar), 7.88 (d, J = 8.5 Hz, 2H, Ar), 8.30 (m, 1H, Ar), 8.34 (s, 1H, NH), 8.51 (s, 1H, NH), 9.78 (s, 1H, NH). IR (Nujol) 3269, 1667, 1614 cm−1. Elemental analysis: calculated for C20H22FN5O5S (463.48) %C 51.83, %H 4.78, %N 15.11, found %C 51.77, %H 4.76, %N 15.15.
- N-(3-fluorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxamide (5g). Yield 40% m.p. 214–215 °C. ESIMS (m/z): 464 (M+H)+. 1H NMR (DMSO-d6): δ 1.57 (br s, 2H, CH2) 1.71, 1.86 (br s, 2H, CH2), 2.54, 2.89 (br s, 2H, CH2), 3.08, 3.44 (br s, 2H, CH2), 4.45 (br s, 1H, CH), 6.76 (m, 1H, Ar), 7.15 (m, 1H, Ar), 7.27 (m, 1H, Ar), 7.45 (s, 3H, Ar and NH2), 7.57 (d, J = 8.5 Hz, 2H, Ar), 7.88 (d, J = 8.5 Hz, 2H, Ar), 8.13 (s, 1H, NH), 8.96 (s, 1H, NH), 9.70 (s, 1H, NH). IR (Nujol) 3355, 3264, 1673, 1615 cm−1. Elemental analysis: calculated for C20H22FN5O5S (463.48) %C 51.83, %H 4.78, %N 15.11, found %C 51.88, %H 4.77, %N 15.07.
- N-(2,6-difluorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxamide (5i). Yield 34% m.p. 194–195 °C. ESIMS (m/z): 482 (M+H)+. 1H NMR (DMSO-d6): δ 1.56 (br s, 2H, CH2) 1.70, 1.85 (br s, 2H, CH2), 2.47, 2.87 (br s, 2H, CH2), 3.08, 3.50 (br s, 2H, CH2), 4.44 (br s, 1H, CH), 7.11 (m, 2H, Ar), 7.28 (m, 1H, Ar), 7.45 (s, 2H, NH2), 7.57 (d, J = 8.5 Hz, 2H, Ar), 7.87 (d, J = 8.5 Hz, 2H, Ar), 8.22 (s, 1H, NH), 8.31 (s, 1H, NH), 9.74 (s, 1H, NH). IR (Nujol) 3321, 3220, 1645, 1613 cm−1. Elemental analysis: calculated for C20H21F2N5O5S (481,47) %C 49.89, %H 4.40, %N 14.55, found %C 49.83, %H 4.41, %N 14.59.
- 2-(1-(4-Sulfamoylbenzoyl)piperidine-4-carbonyl)-N-(o-tolyl)hydrazinecarboxamide (5j). Yield 87% m.p. 238–240 °C. ESIMS (m/z): 460 (M+H)+. 1H NMR (DMSO-d6): δ 1.57 (br s, 2H, CH2), 1.60, 1.85 (br s, 2H, CH2), 2.25 (s, 3H, CH3), 2.53, 2.89 (br s, 2H, CH2), 3.10, 3.51 (br s, 2H, CH2), 4.45 (br s, 1H, CH), 6.76 (d, J = 7.5 Hz, 1H, Ar), 7.12 (m, 1H, Ar), 7.23 (m, 2H, Ar), 7.45 (s, 2H, NH2), 7.57 (d, J = 8.5 Hz, 2H, Ar), 7.88 (d, J = 8.5 Hz, 2H, Ar), 7.95 (s, 1H, NH), 8.61 (s, 1H, NH), 9.66 (s, 1H, NH). IR (Nujol) 3358, 3243, 1639, 1615 cm−1. Elemental analysis: calculated for C21H25N5O5S (459.52) %C 54.89, %H 5.48, %N 15.24, found %C 54.94, %H 5.46, %N 15.18.
- 2-(1-(4-Sulfamoylbenzoyl)piperidine-4-carbonyl)-N-(m-tolyl)hydrazinecarboxamide (5k). Yield 91% m.p. 222–225 °C. ESIMS (m/z): 460 (M+H)+. 1H NMR (DMSO-d6): δ 1.58 (br s, 2H, CH2), 1.74, 1.86 (br s, 2H, CH2), 2.26 (s, 3H, CH3), 2.53, 2.90 (br s, 2H, CH2), 3.10, 3.51 (br s, 2H, CH2), 4.46 (br s, 1H, CH), 6.77 (d, J = 7.5 Hz, 1H, Ar), 7.13 (m, 1H, Ar), 7.23 (m, 2H, Ar), 7.46 (s, 2H, NH2), 7.58 (d, J = 8.5 Hz, 2H, Ar), 7.89 (d, J = 8.5 Hz, 2H, Ar), 8.63 (s, 1H, NH), 9.03 (s, 1H, NH), 9.68 (s, 1H, NH). IR (Nujol) 3288, 3054, 1639, 1615 cm−1. Elemental analysis: calculated for C21H25N5O5S (459.52) %C 54.89, %H 5.48, %N 15.24, found %C 54.94, %H 5.46, %N 15.18.
- N-(2,6-Dimethylphenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxanide (5m). Yield 85% m.p. 212–213 °C. ESIMS (m/z): 474 (M+H)+. 1H NMR (DMSO-d6): δ 1.57 (m, 2H, CH2), 1.71, 1.83 (m, 2H, CH2), 2.15 (s, 6H, 2CH3), 2.51, 2.87 (m, 2H, CH2), 3.08, 3.51 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.03 (m, 3H Ar) 7.45 (s, 2H, NH2), 7.58 (d, J = 8.0 Hz, 2H, Ar), 7.63 (s, 1H, NH), 7.83 (s, 1H, NH), 7.88 (m, 3H, Ar), 9.68 (s, 1H, NH). 13C NMR (DMSO-d6): δ 173.8, 167.7, 156.3, 144.6, 139.4, 136.0, 135.3, 127.5 (2C), 127.2 (2C), 126.0, 125.8 (2C), 64.9, 46.5, 40.9, 28.3, 27.8, 18.1 (2C), 15.1. IR (Nujol) 3323, 3224, 1611 cm−1. Anal. Calcd for C22H27N5O5S (473.55) %C 55.80, %H 5.75, %N 14.79, found %C 55.85, %H 5.76, %N 14.75
- N-(3,5-Dimethylphenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxamide (5n). Yield 90%. m.p. 216–217 °C. ESIMS (m/z): 474 (M+H)+. 1H NMR (DMSO-d6): δ 1.56 (m, 2H, CH2), 1.70, 1.84 (m, 2H, CH2), 2.19 (s, 6H, CH3), 2.53, 2.89 (m, 2H, CH2), 3.09, 3.50 (m, 2H, CH2), 4.45 (m, 1H, CH), 6.58 (s, 1H, Ar), 7.04 (s, 2H, Ar), 7.43 (s, 2H, NH2), 7.56 (d, J = 8.0 Hz, 2H, Ar), 7.87 (d, J = 8.5 Hz, 2H, Ar), 7.91 (s, 1H, NH), 8.51 (s, 1H, NH), 9.65 (s, 1H, NH). IR (Nujol) 3289, 3068, 1644, 1556 cm−1. Elemental analysis: calculated for C22H27N5O5S (473.55) %C 55.80, %H 5.75, %N 14.79, found %C 55.85, %H 5.76, %N 14.72.
- (2-Methoxyphenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxamide (5o). Yield 84% m.p. 219–220 °C. ESIMS (m/z): 476 (M+H)+. 1H NMR (DMSO-d6): δ 1.60 (br s, 2H, CH2), 1.70, 1.86 (br s, 2H, CH2), 2.53-2.91 (br s, 2H, CH2), 3.11, 3.52 (br s, 2H, CH2), 3.84 (s, 3H, OCH3), 4.46 (br s, 1H, CH), 6.87 (m, 1H, Ar), 6.93 (m, 1H, Ar), 7.00 (m, 1H, Ar), 7.46 (s, 2H, NH2), 7.59 (d, J = 8.5 Hz, 2H, Ar), 7.90 (d, J = 8.5 Hz, 2H, Ar), 8.04 (d, J = 8.0 Hz, 1H, Ar), 8.09 (s, 1H, NH), 8.59 (s, 1H, NH), 9.78 (s, 1H, NH). 13C NMR (DMSO-d6): δ 173.8, 167.8, 155.1, 147.5, 144.7, 139.5, 128.4, 127.4 (2C), 125.9 (2C), 121.9, 120.6, 118.0, 110.7, 55.7, 46.2, 40.9, 28.5, 27.9, 18.6. IR (Nujol) 3298, 3100, 1663, 1606 cm−1. Elemental analysis: calculated for C21H25N5O6S (475,52) %C 53.04, %H 5.30, %N 14.73, found %C 53.09, %H 5.31, %N 14.69. m/z 476.
- (3-Methoxyphenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxamide (5p). Yield 62% m.p. 218–220 °C. ESIMS (m/z): 476 (M+H)+. 1H NMR (DMSO-d6): δ 1.56 (br s, 2H, CH2), 1.70, 1.85 (br s, 2H, CH2), 2.54, 2.87 (br s, 2H, CH2), 3.09, 3.51 (br s, 2H, CH2), 3.71 (s, 3H, OCH3), 4.45 (br s, 1H, CH), 6.54 (m, 1H, Ar), 6.94 (m, 1H, Ar), 7.14 (m, 2H, Ar), 7.44 (s, 2H, NH2), 7.57 (d, J = 8.5 Hz, 2H, Ar), 7.88 (d, J = 8.5 Hz, 2H, Ar), 7.98 (s, 1H, NH), 8.70 (s, 1H, NH), 9.67 (s, 1H, NH). IR (Nujol) 3317, 1651, 1614 cm−1. Elemental analysis: calculated for C21H25N5O6S (475,52) %C 53.04, %H 5.30, %N 14.73, found %C 52.99, %H 5.31, %N 14.77.
- (4-Methoxyphenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarboxamide (5q). Yield 78% m.p. 228–230 °C. ESIMS (m/z): 476 (M+H)+. 1H NMR (DMSO-d6): δ 1.58 (br s, 2H, CH2), 1.74, 1.86 (br s, 2H, CH2), 2.54, 2.86 (br s, 2H, CH2), 3.08, 3.51 (br s, 2H, CH2), 3.70 (s, 3H, OCH3), 4.45 (br s, 1H, CH), 6.54 (d, J = 9.5 Hz, 1H, Ar), 7.33 (d, J = 8.5 Hz, 1H, Ar), 7.46 (s, 2H, NH2), 7.58 (m, 3H, Ar), 7.88 (m, 3H, Ar), 8.53 (s, 1H, NH), 9.02 (s, 1H, NH), 9.65 (s, 1H, NH). IR (Nujol) 3315, 3216, 1680, 1617 cm−1. Elemental analysis: calculated for C21H25N5O6S (475,52) %C 53.04, %H 5.30, %N 14.73, found %C 52.98, %H 5.32, %N 14.76.
3.1.2. General Procedure for the Preparation of N-aryl-4-Sulfamoylbenzoyl-piperidine-4-carbonyl-hydrazincarbothioamides (6a–y)
- N-phenyl-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazine-1-carbothioamide (6a). Yield 98% m.p. 210–211 °C. ESIMS (m/z): 462 (M+H)+. 1H NMR (DMSO-d6): δ 1.56 (m, 2H, CH2), 1.76, 1.92 (m, 2H, CH2), 2.54, 2.87 (m, 2H, CH2), 3.08, 3.51 (m, 2H, CH2), 4.44 (m, 1H, CH), 7.15 (m, 1H, Ar), 7.32 (m, 3H, Ar), 7.44 (s, 3H, Ar and NH2), 7.56 (d, J = 7.5 Hz, 2H, Ar), 7.87 (m, 3H, Ar and NH), 9.51 (s, 1H, NH), 9.92 (s, 1H, NH). IR (Nujol) 3334, 3242, 3050, 1687, 1613 cm−1. Elemental analysis: calculated for C20H23N5O4S2 (461.56) %C 52.05, %H 5.02, %N 15.17, found %C 52.11, %H 5.02, %N 15.14.
- N-(4-Nitrophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6b). Yield 85% m.p. 236–237 °C. ESIMS (m/z): 507 (M+H)+. 1H NMR (DMSO-d6) δ 1.56 (m, 2H, CH2), 1.76, 1.91 (m, 2H, CH2), 2.56, 2.89 (m, 2H, CH2), 3.09, 3.52 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.43 (s, 2H, NH2), 7.56 (d, J = 8.0 Hz, 2H, Ar), 7.87 (m, 4H, Ar), 8.19 (d, J = 9.0 Hz, 2H, Ar), 9.86 (s, 1H, NH), 9.93 (s, 1H, NH), 9.97 (s, 1H, NH). IR (Nujol) 3317, 3220, 3137, 1678, 1598 cm−1. Elemental analysis: calculated for C20H22N6O6S2 (506.46) %C 47.42, %H 4.38, %N 16.59, found %C 47.47, %H 4.39, %N 16.63.
- 4-(4-(4-(4-(Trifluoromethyl)phenyl)piperazine-1-carbonyl)piperidine-1-carbonyl)benzenesulfonamide (6c). Yield 47% m.p. 201–203 °C. ESIMS (m/z): 530 (M+H)+. 1H NMR (DMSO-d6): δ 1.56 (m, 2H, CH2), 1.76, 1.91 (m, 2H, CH2), 2.54, 2.88 (m, 2H, CH2), 3.08, 3.51 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.42 (s, 2H, NH2), 7.56 (d, J = 8.0 Hz, 2H, Ar), 7.66 (d, J = 8.5 Hz, 2H, Ar), 7.73 (d, J = 8.5 Hz, 2H, Ar), 7.86 (d, J = 8.5 Hz, 2H, Ar), 9.75 (s, 2H, NH), 9.96 (s, 1H, NH). IR (Nujol) 3300, 1686, 1619 cm−1. Elemental analysis: calculated for C21H22F3N5O4S2 (529.55) %C 47.63, %H 4.19, %N 13.23, found %C 47.58, %H 4.20, %N 13.27.
- N-(2-Chlorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6d). Yield 98% m.p. 209–210 °C. ESIMS (m/z): 497 (M+H)+. 1H NMR (DMSO-d6): δ 1.57 (m, 2H, CH2), 1.77, 1.93 (m, 2H, CH2), 2.63, 2.88 (m, 2H, CH2), 3.08, 3.53 (m, 2H, CH2), 4.46 (m, 1H, CH), 7.27 (m, 1H, Ar), 7.34 (m, 1H, Ar), 7.46 (s, 2H, NH2), 7.49 (m, 2H, Ar), 7.57 (d, J = 8.0 Hz, 2H, Ar), 7.87 (d, J = 8.5 Hz, 2H, Ar), 9.34 (s, 1H, NH), 9.69 (s, 1H, NH), 10.01 (s, 1H, NH). IR (Nujol) 3362, 3254, 1682, 1619 cm−1. Elemental analysis: calculated for C20H22ClN5O4S2 (496.00) %C 48.43, %H 4.47, %N 14.12, found %C 48.38, %H 4.45, %N 14.17.
- N-(3-Chlorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6e). Yield 61% m.p. 226–227 °C. ESIMS (m/z): 497 (M+H)+. 1H NMR (DMSO-d6): δ 1.52 (m, 2H, CH2), 1.72, 1.89 (m, 2H, CH2), 2.53, 2.85 (m, 2H, CH2), 3.05, 3.49 (m, 2H, CH2), 4.43 (m, 1H, CH), 7.19 (d, J = 7.0 Hz, 1H, Ar), 7.25 (m, 3H, Ar), 7.42 (s, 2H, NH2), 7.55 (d, J = 8.0 Hz, 2H, Ar), 7.86 (d, J = 8.5 Hz, 2H, Ar), 8.33 (s, 1H, NH), 9.24 (s, 1H, NH), 9.77 (s, 1H, NH). IR (Nujol) 3277, 3175, 3087, 1677, 1544 cm−1. Elemental analysis: calculated for C20H22ClN5O4S2 (496.00) %C 48.43, %H 4.47, %N 14.12, found %C 48.36, %H 4.48, %N 14.09.
- N-(4-Chlorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6f). Following the general procedure, the title compound was prepared starting from 4-chlorophenylisothiocyanate. Yield 79% m.p. 222–223 °C. ESIMS (m/z): 497 (M+H)+. 1H NMR (DMSO-d6): δ 1.55 (s, 2H, CH2), 1.74, 1.91 (m, 2H, CH2), 2.63, 2.88 (m, 2H, CH2), 3.08, 3.51 (m, 2H, CH2), 4.46 (m, 1H, CH), 7.36 (d, J = 8.5 Hz, 2H, Ar), 7.42 (s, 2H, NH2), 7.46 (m, 2H, Ar), 7.56 (d, J = 7.5 Hz, 2H, Ar), 7.86 (d, J = 8.0 Hz, 2H, Ar), 9.59 (s, 2H, NH), 9.91 (s, 1H, NH). IR (Nujol) 3322, 3290, 3185, 1686, 1590 cm−1. Elemental analysis: calculated for C20H22ClN5O4S2 (496.00) %C 48.43, %H 4.47, %N 14.12, found %C 48.48, %H 4.48, %N 14.08. m/z 597.
- N-(3,4-Dichlorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6g). Yield 97% m.p. 232–233 °C. ESIMS (m/z): 531 (M+H)+. 1H NMR (DMSO-d6): δ 1.55 (m, 2H, CH2), 1.76, 1.91 (m, 2H, CH2), 1.53, 2.88 (m, 2H, CH2), 3.08, 3.52 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.43 (s, 2H, NH2), 7.47 (d, J = 8.0 Hz, 2H, Ar), 7.56 (d, J = 7.0 Hz, 2H, Ar), 7.82 (s, 1H, Ar), 7.86 (d, J = 8.5 Hz, 2H, Ar), 9.64 (s, 1H, NH), 9.76 (s, 1H, NH), 9.95 (s, 1H, NH). IR (Nujol) 3343, 3271, 3149, 1683, 1540 cm−1. Elemental analysis: calculated for C20H21Cl2N5O4S2 (530.45) %C 45.29, %H 3.99, %N 13.20, found %C 45.33, %H 3.97, %N 13.25.
- N-(2,4-Dichlorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6h). Yield 81% m.p. 220–221 °C. ESIMS (m/z): 531 (M+H)+. 1H NMR (DMSO-d6): δ 1.55 (m, 2H, CH2), 1.74, 1.89 (m, 2H, CH2), 2.53, 2.87 (m, 2H, CH2), 3.07, 3.50 (m, 2H, CH2), 4.44 (m, 1H, CH), 7.39 (m, 2H, Ar), 7.42 (s, 2H, NH2), 7.54 (s, 1H, Ar), 7.55 (d, J = 8.0 Hz, 2H, Ar), 7.86 (d, J = 8.5 Hz, 2H, Ar), 9.35 (s, 1H, NH), 9.74 (s, 1H, NH), 9.99 (s, 1H, NH). IR (Nujol) 3243, 1681 cm−1. Elemental analysis: calculated for C21H21N5O5S2 (530.45) %C 45.29, %H 3.99, %N 13.20, found %C 45.23, %H 3.98, %N 13.23.
- N-(2,6-Dichlorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6i). Yield 90% m.p. 179–180 °C. ESIMS (m/z): 531 (M+H)+. 1H NMR (DMSO-d6): δ 1.57 (m, 2H, CH2), 1.79, 1.95 (m, 2H, CH2), 2.53, 2.87 (m, 2H, CH2), 3.08, 3.53 (m, 2H, CH2), 4.46 (m, 1H, CH), 7.33 (m, 1H, Ar), 7.44 (s, 2H, NH2), 7.49 (m, 2H, Ar), 7.57 (d, J = 8.0 Hz, 2H, Ar), 7.87 (d, J = 8.5 Hz, 2H, Ar), 9.42 (s, 1H, NH), 9.71 (s, 1H, NH), 10.00 (s, 1H, NH). IR (Nujol) 3451, 3228, 1691, 1619 cm−1. Elemental analysis: calculated for C21H21N5O5S2 (530.45) %C 45.29, %H 3.99, %N 13.20, found %C 45.24, %H 4.02, %N 13.17.
- N-(2-methoxyphenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazine-1-carbothioamide (6j). Yield 98% m.p. 214–215 °C. ESIMS (m/z): 492 (M+H)+. 1H NMR (DMSO-d6): δ 1.61 (m, 2H, CH2), 1.76, 1.93 (m, 2H, CH2), 2.59, 2.91 (m, 2H, CH2), 3.12, 3.54 (m, 2H, CH2), 3.80 (s, 3H, OCH3), 4.48 (m, 1H, CH), 6.93 (t, J = 8.27, 1H, Ar), 7.04 (d, J = 8.26, 1H, Ar), 7.15 (t, J = 8.50, 1H, Ar), 7.47 (s, 2H, NH2), 7.59 (d, J = 8.5 Hz, 2H, Ar), 7.90 (d, J = 8.5 Hz, 2H, Ar), 8.09 (m, 1H, Ar), 8.91 (s, 1H, NH), 9.65 (s, 1H, NH), 10.08 (s, 1H, NH). 13C NMR (DMSO-d6): δ 167.8, 151.3, 144.7, 139.4, 127.6, 127.3 (2C), 125.9 (2C), 124.6, 119.8 (2C), 111.3, 56.1, 55.8 (2C), 46.5, 40.9, 28.3, 27.8, 18.6. IR (Nujol) 3305, 3272, 3223, 1683, 1590 cm−1. Elemental analysis: calculated for C21H25N5O5S2 (491.58) %C 51.31, %H 5.13, %N 13.04, found %C 51.37, %H 5.11, %N 13.07.
- N-(3-methoxyphenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazine-1-carbothioamide (6k). Yield 80% m.p. 214–215 °C. ESIMS (m/z): 492 (M+H)+. 1H NMR (DMSO-d6): δ 1.56 (m, 2H, CH2), 1.76, 1.92 (m, 2H, CH2), 2.52, 2.89 (m, 2H, CH2), 3.09, 3.52 (m, 2H, CH2), 3.74 (s, 2H, OCH3), 4.46 (m, 1H, CH), 6.73 (m, 1H, Ar), 7.00 (m, 1H, Ar), 7.23 (m, 2H, Ar), 7.45 (s, 2H, NH2), 7.57 (d, J = 8.5 Hz, 2H, Ar), 7.88 (d, J = 8.5 Hz, 2H, Ar), 9.53 (s, 2H, NH), 9.91 (s, 1H, NH). IR (Nujol) 3312, 3277, 3205, 1684, 1601, 1591 cm−1. Elemental analysis: calculated for C21H25N5O5S2 (491.58) %C 51.31, %H 5.13, %N 13.04, found %C 51.38, %H 5.11, %N 13.06.
- N-(4-Methoxyphenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6l). Yield 84% m.p. >250 °C. ESIMS (m/z): 492 (M+H)+. 1H NMR (DMSO-d6) δ 1.54 (m, 2H, CH2), 1.75, 1.91 (m, 2H, CH2), 1.54, 1.87 (m, 2H, CH2), 3.07, 3.51 (m, 2H, CH2), 3.73 (s, 3H, OCH3), 4.44 (m, 1H, CH), 6.87 (d, J = 8.0 Hz, 2H, Ar), 7.24 (d, J = 7.5 Hz, 2H, Ar), 7.43 (s, 2H, NH2), 7.56 (d, J = 8.0 Hz, 2H, Ar), 7.86 (d, J = 8.5 Hz, 2H, Ar), 9.38 (s, 1H, NH), 9.43 (s, 1H, NH), 9.86 (s, 1H, NH). IR (Nujol) 3335, 323, 1680, 1543 cm−1. Elemental analysis: calculated for C21H25N5O5S2 (491.58) %C 51.31, %H 5.13, %N 13.05, found %C 51.36, %H 5.11, %N 13.01. m/z 492.
- 2-(1-(4-Sulfamoylbenzoyl)piperidine-4-carbonyl)-N-(3,4,5-trimethoxyphenyl)hydrazinecarbothioamide (6m). Yield 44% m.p. >250 °C. ESIMS (m/z): 552 (M+H)+. 1H NMR (DMSO-d6): δ 1.55 (m, 2H, CH2), 1.75, 1.90 (m, 2H, CH2), 2.53, 2.87 (m, 2H, CH2), 3.08, 3.52 (m, 2H, CH2), 3.63 (s, 3H, OCH3), 3.73 (s, 6H, OCH3), 4.44 (m, 1H, CH), 6.82 (s, 2H, Ar), 7.43 (s, 2H, NH2), 7.56 (d, J = 8 Hz, 2H, Ar), 7.86 (d, J = 8.5 Hz, 2H, Ar), 9.48 (s, 2H, NH), 9.88 (s, 1H, NH). IR (Nujol) 3533, 3284, 3168, 1692, 1565 cm−1. Elemental analysis: calculated for C23H29N5O7S2 (551.64) %C 50.08, %H 5.30, %N 12.70, found %C 50.01, %H 5.32, %N 12.66.
- N-(2-Fluorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6n). Yield 98% m.p. 219–220 °C. ESIMS (m/z): 480 (M+H)+.1H NMR (DMSO-d6): δ 1.56 (m, 2H, CH2), 1.77, 1.93 (m, 2H, CH2), 2.63, 2.88 (m, 2H, CH2), 3.09, 3.52 (m, 2H, CH2), 4.46 (m, 1H, CH), 7.17 (m, 1H, Ar), 7.21 (m, 1H, Ar), 7.44 (s, 2H, NH2), 7.25 (m, 2H, Ar), 7.57 (d, J = 8.0 Hz, 2H, Ar), 7.88 (d, J = 8.5 Hz, 2H, Ar), 9.32 (s, 1H, NH), 9.70 (s, 1H, NH), 9.96 (s, 1H, NH). IR (Nujol) 3305, 3199, 1683, 1592 cm−1. Elemental analysis: calculated for C20H22FN5O4S2 (479.55) %C 50.09, %H 4.62, %N 14.60, found %C 50.17, %H 4.60, %N 14.63.
- N-(3-Fluorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6o). Yield 46% m.p. 214–215 °C. ESIMS (m/z): 480 (M+H)+.1H NMR (DMSO-d6): δ 1.56 (m, 2H, CH2), 1.76, 1.92 (m, 2H, CH2), 2.55, 2.89 (m, 2H, CH2), 3.10, 3.53 (m, 2H, CH2), 4.46 (m, 1H, CH), 6.97 (s, 1H, Ar), 7.26 (m, 1H, Ar), 7.35 (m, 2H, Ar), 7.45 (s, 2H, NH2), 7.57 (d, J = 8.0 Hz, 2H, Ar), 7.88 (d, J = 8.5 Hz, 2H, Ar), 9.68 (s, 2H, NH), 9.96 (s, 1H, NH). IR (Nujol) 3333, 3266, 1686, 1620 cm−1. Elemental analysis: calculated for C20H22FN5O4S2 (479.55) %C 50.09, %H 4.62, %N 14.60, found %C 50.02, %H 4.64, %N 14.64. m/z 480.
- N-(4-Fluorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6p). Yield 85% m.p. 215–216 °C. ESIMS (m/z): 480 (M+H)+. 1H NMR (DMSO-d6) δ 1.55 (m, 2H, CH2), 1.76, 1.91 (m, 2H, CH2), 2.53, 2.87 (m, 2H, CH2), 3.08, 3.51 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.14 (d, J = 8.5 Hz, 2H, Ar), 7.39 (m, 2H, Ar), 7.45 (s, 2H, NH2), 7.56 (d, J = 8.0 Hz, 2H, Ar), 7.87 (d, J = 8.0 Hz, 2H, Ar), 9.54 (s, 2H, NH), 9.90 (s, 1H, NH). IR (Nujol) 3320, 3175, 1685, 1563 cm−1. Elemental analysis: calculated for C20H22FN5O4S2 (479.55) %C 50.09, %H 4.62, %N 14.60, found %C 50.03, %H 4.60, %N 14.64.
- N-(2,6-Difluorophenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6q). Yield 77% m.p. 242–243 °C. ESIMS (m/z): 498 (M+H)+. 1H NMR (DMSO-d6) δ 1.55 (m, 2H, CH2), 1.76, 1.92 (m, 2H, CH2), 2.55, 2.86 (m, 2H, CH2), 3.07, 3.38 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.11 (d, J = 8.0 Hz, 2H, Ar), 7.33 (d, J = 7.0 Hz, 1H, Ar), 7.43 (s, 2H, NH2), 7.56 (d, J = 8.0 Hz, 2H, Ar), 7.86 (d, J = 8.5 Hz, 2H, Ar), 9.15 (s, 1H, NH), 9.83 (s, 1H, NH), 10.02 (s, 1H, NH). IR (Nujol) 3314, 3279, 3201, 1658, 1566 cm−1. Elemental analysis: calculated for C20H21F2N5O4S2 (497.54) %C 48.28, %H 4.25, %N 14.08, found %C 48.33, %H 4.26, %N 14.05.
- 2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)-N-(o-tolyl)hydrazine-1-carbothioamide (6r). Yield 98% m.p. 209–210 °C. ESIMS (m/z): 476 (M+H)+. 1H NMR (DMSO-d6): δ 1.56 (m, 2H, CH2), 1.77, 1.92 (m, 2H, CH2), 2.14 (s, 2H, CH3), 2.52, 2.87 (m, 2H, CH2), 3.08, 3.52 (m, 2H, CH2), 4.46 (m, 1H, CH), 7.16 (m, 4H, Ar), 7.45 (s, 2H, NH2), 7.57 (d, J = 8.5 Hz, 2H, Ar), 7.87 (d, J = 8.5 Hz, 2H, Ar), 9.29 (s, 1H, NH), 9.42 (s, 1H, NH), 9.92 (s, 1H, NH). IR (Nujol) 3300, 3192, 1685, 1591 cm−1. Elemental analysis: calculated for C21H25N5O4S2 (475.58) %C 53.04, %H 5.30, %N 14.73, found %C 52.98, %H 5.32, %N 14.77.
- 2-(1-(3-sulfamoylbenzoyl)piperidine-4-carbonyl)-N-(p-tolyl)hydrazine-1-carbothioamide (6s). Yield 72% m.p. 214–215 °C. ESIMS (m/z): 476 (M+H)+. 1H NMR (DMSO-d6): δ 1.56 (m, 2H, CH2), 1.77, 1.92 (m, 2H, CH2), 2.29 (s, 2H, CH3), 2.52, 2.87 (m, 2H, CH2), 3.08, 3.52 (m, 2H, CH2), 4.46 (m, 1H, CH), 6.99 (m, 1H, Ar), 7.19 (m, 3H Ar), 7.46 (s, 2H, NH2), 7.59 (d, J = 8.5 Hz, 2H, Ar), 7.87 (d, J = 8.5 Hz, 2H, Ar), 9.49 (s, 2H, NH), 9.91 (s, 1H, NH). IR (Nujol) 3316, 3291, 3197, 1684, 1591 cm−1. Elemental analysis: calculated for C21H25N5O4S2 (475.58) %C 53.04, %H 5.30, %N 14.73, found %C 52.98, %H 5.32, %N 14.76.
- 2-(1-(3-sulfamoylbenzoyl)piperidine-4-carbonyl)-N-(p-tolyl)hydrazine-1-carbothioamide (6t). Yield 80% m.p. 234–235 °C. ESIMS (m/z): 476 (M+H)+. 1H NMR (DMSO-d6): δ 1.56 (m, 2H, CH2), 1.76, 1.92 (m, 2H, CH2), 2.28 (s, 2H, CH3), 2.53, 2.88 (m, 2H, CH2), 3.09, 3.52 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.13 (d, J = 7.5 Hz, 2H, Ar), 7.28 (d, J = 7.5 Hz, 2H, Ar), 7.45 (s, 2H, NH2), 7.57 (d, J = 8.5 Hz, 2H, Ar), 7.87 (d, J = 8.5 Hz, 2H, Ar), 9.44 (s, 2H, NH), 9.89 (s, 1H, NH). IR (Nujol) 3304, 3272, 3151, 1686, 1621 cm−1. Elemental analysis: calculated for C21H25N5O4S2 (475.58) %C 53.04, %H 5.30, %N 14.73, found %C 53.09, %H 5.28, %N 14.21.
- N-(2,6-Dimethylphenyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6u). Yield 64% m.p. 233–234 °C. ESIMS (m/z): 490 (M+H)+. 1H NMR (DMSO-d6): δ 1.57 (m, 2H, CH2), 1.71, 1.83 (m, 2H, CH2), 2.15 (s, 6H, 2CH3), 2.51, 2.87 (m, 2H, CH2), 3.08, 3.51 (m, 2H, CH2), 4.45 (m, 1H, CH), 7.03 (m, 3H Ar) 7.45 (s, 2H, NH2), 7.58 (d, J = 8.0 Hz, 2H, Ar), 7.83 (s, 1H, NH), 7.88 (m, 3H, Ar and NH), 9.68 (s, 1H, NH). 13C NMR (DMSO-d6): δ 173.22, 167.8, 167.7, 144.6, 139.5, 136.9, 136.6, 127.5, 127.3 (2C), 127.2 (2C), 126.8, 125.9 (2C), 46.5, 41.0, 40.0, 28.6, 28.0, 17.9 (2C) IR (Nujol) 3330, 3238, 1689 cm−1. Elemental analysis: calculated for C22H27N5O4S2 (489.61) %C 53.97, %H 5.56, %N 14.30, found %C 54.03, %H 5.55, %N 14.26.
- N-Benzyl-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6v). Following the general procedure, the title compound was prepared starting from benzylisothiocyanate. Yield 83% m.p. >250 °C. ESIMS (m/z): 476 (M+H)+. 1H NMR (DMSO-d6): δ 1.52 (m, 2H, CH2), 1.73, 1.88 (m, 2H, CH2), 2.45, 2.84 (m, 2H, CH2), 3.10, 3.49 (s, 2H, CH2), 4.42 (m, 1H, CH), 4.70 (s, 2H, CH2), 7.19 (m, 1H, Ar), 7.24 (m, 4H, Ar), 7.46 (s, 2H, NH2), 7.59 (d, J = 8.0 Hz, 2H, Ar), 7.88 (d, J = 8.5 Hz, 2H, Ar), 8.33 (s, 1H, NH), 9.24 (s, 1H, NH), 9.77 (s, 1H, NH). IR (Nujol) 3333, 3244, 1688, 1560 cm−1. Elemental analysis: calculated for C21H25N5O4S2 (475.58) %C 53.03, %H 5.30, %N 14.73, found %C 52.96, %H 5.29, %N 14.76.
- N-(4-Methoxybenzyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6w). Yield 64% m.p. 240–241 °C. ESIMS (m/z): 506 (M+H)+. 1H NMR (DMSO-d6): δ 1.51 (m, 2H, CH2), 1.72, 1.88 (m, 2H, CH2), 2.54, 2.85 (m, 2H, CH2), 3.04, 3.49 (m, 2H, CH2), 3.71 (s, 3H, OCH3), 4.42 (m, 1H, CH), 4.62 (s, 2H, CH2), 6.84 (d, J = 8.5 Hz, 2H, Ar), 7.19 (d, J = 8.5 Hz, 2H, Ar), 7.42 (s, 2H, NH2), 7.54 (d, J = 8.5 Hz, 2H, Ar), 7.85 (d, J = 8 Hz, 2H, Ar), 8.23 (s, 1H, NH), 9.18 (s, 1H, NH), 9.73 (s, 1H, NH). IR (Nujol) 3347, 3249, 3150, 1669, 1548 cm−1. Elemental analysis: calculated for C22H27N5O5S2 (505.61) %C 52.26, %H 5.38, %N 13.85, found %C 52.31, %H 5.36, %N 13.82.
- N-(1-Phenylethyl)-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6x). Yield 49% m.p. 196–197 °C. ESIMS (m/z): 490 (M+H)+. 1H NMR (DMSO-d6): δ 1.41 (d, J = 7.0, 3H, CH3), 1.53 (m, 2H, CH2), 1.72, 1.88 (m, 2H, CH2), 2.62, 2.86 (m, 2H, CH2), 3.06, 3.49 (m, 2H, CH2), 4.43 (m, 1H, CH), 5.57 (m, 1H, CH), 7.20 (m, 1H, Ar), 7.29 (m, 4H, Ar), 7.42 (s, 2H, NH2), 7.55 (d, J = 8.5 Hz, 2H, Ar), 7.86 (d, J = 8.5 Hz, 2H, Ar), 7.99 (s, 1H, NH), 9.14 (s, 1H, NH), 9.71 (s, 1H, NH). IR (Nujol) 3335, 3230, 3087, 1688, 1597 cm−1. Elemental analysis: calculated for C22H27N5O4S2 (489.61) %C 53.97, %H 5.56, %N 14.30, found %C 53.92, %H 5.58, %N 14.32.
- N-Cyclohexyl-2-(1-(4-sulfamoylbenzoyl)piperidine-4-carbonyl)hydrazinecarbothioamide (6y). Yield 67% m.p. 183–184 °C. ESIMS (m/z): 468 (M+H)+. 1H NMR (DMSO-d6) δ 1.06 (m, 2H, CH2), 1.22 (m, 4H, CH2), 1.54 (m, 4H, CH2), 1.56 (m, 2H, CH2), 1.77, 1.86 (m, 2H, CH2), 2.53, 2.86 (m, 2H, CH2), 3.06, 3.50 (m, 2H, CH2), 4.03 (m, 1H, CH), 4.43 (m, 1H, CH), 7.36 (s, 1H, NH), 7.43 (s, 2H, NH2), 7.55 (d, J = 7.5 Hz, 2H, Ar), 7.86 (d, J = 8.0 Hz, 2H, Ar), 8.99 (s, 1H, NH), 9.64 (s, 1H, NH). IR (Nujol) 3324, 3177, 1672, 1555 cm−1. Elemental analysis: calculated for C20H29N5O4S2 (467.61) %C 51.37, %H 6.25, %N 14.98, found %C 51.42, %H 6.26, %N 14.94.
3.2. Carbonic Anhydrase Inhibition
3.3. Molecular Docking
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Nocentini, A.; Supuran, C.T. Carbonic anhydrase inhibitors as antitumor/antimetastatic agents: A patent review (2008–2018). Expert Opin. Ther. Pat. 2018, 28, 729–740. [Google Scholar] [CrossRef] [PubMed]
- Alterio, V.; Di Fiore, A.; D’Ambrosio, K.; Supuran, C.T.; De Simone, G. Multiple Binding Modes of Inhibitors to Carbonic Anhydrases: How to Design Specific Drugs Targeting 15 Different Isoforms? Chem. Rev. 2012, 112, 4421–4468. [Google Scholar] [CrossRef] [PubMed]
- Supuran, C.T.; Scozzafava, A.; Conway, J. Carbonic Anhydrase: Its Inhibitors and Activators. J. Am. Chem. Soc. 2005, 127, 3643. [Google Scholar] [CrossRef]
- D’Ambrosio, K.; De Simone, G.; Supuran, C.T. Human Carbonic Anhydrases: Catalytic Properties, Structural Features, and Tissue Distribution. Carbon. Anhydrases Biocatal. 2015, 2, 17–30. [Google Scholar] [CrossRef]
- Stams, T.; Christianson, D.W. X-ray crystallographic studies of mammalian carbonic anhydrase isozymes. In The Carbonic Anhydrases; Birkhäuser: Basel, Switzerland, 2000; Volume 90, pp. 159–174. [Google Scholar] [CrossRef]
- McDonald, P.C.; Winum, J.Y.; Supuran, C.T.; Dedhar, S. Recent developments in targeting carbonic anhydrase IX for cancer therapeutics. Oncotarget 2012, 3, 84–97. [Google Scholar] [CrossRef] [PubMed]
- Ulmasov, B.; Waheed, A.; Shah, G.N.; Grubb, J.H.; Sly, W.S.; Tu, C.; Silverman, D.N. Purification and kinetic analysis of recombinant CA XII, a membrane carbonic anhydrase overexpressed in certain cancers. Proc. Natl. Acad. Sci. USA 2000, 97, 14212–14217. [Google Scholar] [CrossRef]
- Angeli, A.; Carta, F.; Nocentini, A.; Winum, J.Y.; Zalubovskis, R.; Akdemir, A.; Onnis, V.; Eldehna, W.M.; Capasso, C.; Simone, G.; et al. Carbonic Anhydrase Inhibitors Targeting Metabolism and Tumor Microenvironment. Metabolites 2020, 10, 412. [Google Scholar] [CrossRef]
- Supuran, C.T. Carbonic anhydrase inhibitors: An update on experimental agents for the treatment and imaging of hypoxic tumors. Expert Opin. Investig. Drugs 2021, 30, 1197–1208. [Google Scholar] [CrossRef]
- Tanini, D.; Carradori, S.; Capperucci, A.; Lupori, L.; Zara, S.; Ferraroni, M.; Ghelardini, C.; Mannelli, L.; Micheli, L.; Lucarini, E.; et al. Chalcogenides-incorporating carbonic anhydrase inhibitors concomitantly reverted oxaliplatin-induced neuropathy and enhanced antiproliferative action. Eur. J. Med. Chem. 2021, 225, 113793. [Google Scholar] [CrossRef]
- D’Ascenzio, M.; Secci, D.; Carradori, S.; Zara, S.; Guglielmi, P.; Cirilli, R.; Pierini, M.; Poli, G.; Tuccinardi, T.; Angeli, A.; et al. 1,3-Dipolar Cycloaddition, HPLC Enantioseparation, and Docking Studies of Saccharin/Isoxazole and Saccharin/Isoxazoline Derivatives as Selective Carbonic Anhydrase IX and XII Inhibitors. J. Med. Chem. 2020, 63, 2470–2488. [Google Scholar] [CrossRef] [PubMed]
- Kroemer, G.; Pouyssegur, J. Tumor cell metabolism: Cancer’s Achilles’ heel. Cancer Cell 2008, 6, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Di Fiore, A.; Monti, S.M.; Hilvo, M.; Parkkila, S.; Romano, V.; Scaloni, A.; Pedone, C.; Scozzafava, A.; Supuran, C.T.; De Simone, G. Crystal structure of human carbonic anhydrase XIII and its complex with the inhibitor acetazolamide. Proteins 2009, 74, 164–175. [Google Scholar] [CrossRef]
- Svastová, E.; Hulíková, A.; Rafajová, M.; Zat’ovicová, A.; Gibadulinová, A.; Casini, A.; Cecchi, A.; Scozzafava, A.; Supuran, C.T.; Pastorek, J.; et al. Hypoxia activates the capacity of tumor-associated carbonic anhydrase IX to acidify extracellular pH. FEBS Lett. 2004, 577, 439–445. [Google Scholar] [CrossRef]
- Neri, D.; Supuran, C.T. Interfering with pH regulation in tumours as a therapeutic strategy. Nat. Rev. Drug Discov. 2011, 10, 767–777. [Google Scholar] [CrossRef]
- Petreni, A.; Osman, S.M.; Alasmary, F.A.; Almutairi, T.M.; Nocentini, A.; Supuran, C.T. Binding site comparison for coumarin inhibitors and amine/amino acid activators of human carbonic anhydrases. Eur. J. Med. Chem. 2021, 226, 113875. [Google Scholar] [CrossRef] [PubMed]
- Citarella, A.; Moi, D.; Pinzi, L.; Bonanni, D.; Rastelli, G. Hydroxamic Acid Derivatives: From Synthetic Strategies to Medicinal Chemistry Applications. ACS Omega 2021, 6, 21843–21849. [Google Scholar] [CrossRef]
- Moi, D.; Nocentini, A.; Deplano, A.; Osman, S.M.; AlOthman, Z.A.; Piras, V.; Balboni, G.; Supuran, C.T.; Onnis, V. Appliance of the piperidinyl-hydrazidoureido linker to benzenesulfonamide compounds: Synthesis, in vitro and in silico evaluation of potent carbonic anhydrase II, IX and XII inhibitors. Bioorg. Chem. 2020, 98, 103728. [Google Scholar] [CrossRef]
- Nocentini, A.; Moi, D.; Deplano, A.; Osman, S.M.; AlOthman, Z.A.; Balboni, G.; Supuran, C.T.; Onnis, V. Sulfonamide/sulfamate switch with a series of piperazinylureido derivatives: Synthesis, kinetic and in silico evaluation as carbonic anhydrase isoforms I, II, IV, and IX inhibitors. Eur. J. Med. Chem. 2020, 186, 111896. [Google Scholar] [CrossRef]
- Moi, D.; Nocentini, A.; Deplano, A.; Balboni, G.; Supuran, C.T.; Onnis, V. Structure-activity relationship with pyrazoline-based aromatic sulfamates as carbonic anhydrase isoforms I, II, IX and XII inhibitors: Synthesis and biological evaluation. Eur. J. Med. Chem. 2019, 182, 111638. [Google Scholar] [CrossRef]
- Nocentini, A.; Moi, D.; Balboni, G.; Onnis, V.; Supuran, C.T. Discovery of thiazolin-4-one-based aromatic sulfamates as a new class of carbonic anhydrase isoforms I, II, IV, and IX inhibitors. Bioorg. Chem. 2018, 77, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Nocentini, A.; Moi, D.; Balboni, G.; Salvadori, S.; Onnis, V.; Supuran, C.T. Synthesis and biological evaluation of novel pyrazoline-based aromatic sulfamates with potent carbonic anhydrase isoforms II, IV and IX inhibitory efficacy. Bioorg. Chem. 2018, 77, 633–639. [Google Scholar] [CrossRef] [PubMed]
- Khalifah, R.G. The carbon dioxide hydration activity of carbonic anhydrase. I. Stop-flow kinetic studies on the native human isoenzymes B and C. J. Biol. Chem. 1971, 246, 2561–2573. [Google Scholar] [CrossRef]
- Kiss, L.E.; Ferreira, H.S.; Torrão, L.; Bonifácio, M.J.; Palma, P.N.; Soares-da-Silva, P.; Learmonth, D.A. Discovery of a long-acting, peripherally selective inhibitor of catechol-O-methyltransferase. J. Med. Chem. 2010, 53, 3396–3411. [Google Scholar] [CrossRef]
- Vullo, D.; Del Prete, S.; Nocentini, A.; Osman, S.M.; AlOthman, Z.A.; Capasso, C.; Bozdag, M.; Carta, F.; Gratteri, P.; Supuran, C.T. Dithiocarbamates effectively inhibit the β-carbonic anhydrase from the dandruff-producing fungus Malassezia globose. Bioorg. Med. Chem. 2017, 25, 1260–1265. [Google Scholar] [CrossRef]
- Del Prete, S.; Angeli, A.; Ghobril, C.; Hitce, J.; Clavaud, C.; Marat, X.; Supuran, C.T.; Capasso, C. Sulfonamide Inhibition Profile of the β-Carbonic Anhydrase from Malassezia restricta, An Opportunistic Pathogen Triggering Scalp Conditions. Metabolites 2020, 10, 39. [Google Scholar] [CrossRef]
- Nocentini, A.; Bonardi, A.; Gratteri, P.; Cerra, B.; Gioiello, A.; Supuran, C.T. Steroids interfere with human carbonic anhydrase activity by using alternative binding mechanisms. J. Enzyme Inhib. Med. Chem. 2018, 33, 1453–1459. [Google Scholar] [CrossRef]
- Bonardi, A.; Vermelho, A.B.; da Silva Cardoso, V.; de Souza Pereira, M.C.; da Silva Lara, L.; Selleri, S.; Gratteri, P.; Supuran, C.T.; Nocentini, A. N-Nitrosulfonamides as Carbonic Anhydrase Inhibitors: A Promising Chemotype for Targeting Chagas Disease and Leishmaniasis. ACS Med. Chem. Lett. 2018, 10, 413–418. [Google Scholar] [CrossRef]
- Glide, version 6.7; Schrödinger LLC: New York, NY, USA, 2015.
- Maestro, version 10.2; Schrödinger LLC: New York, NY, USA, 2015.
- RDKit. Cheminformatics and Machine Learning Software. 2013. Available online: http://www.rdkit.Org (accessed on 1 March 2022).
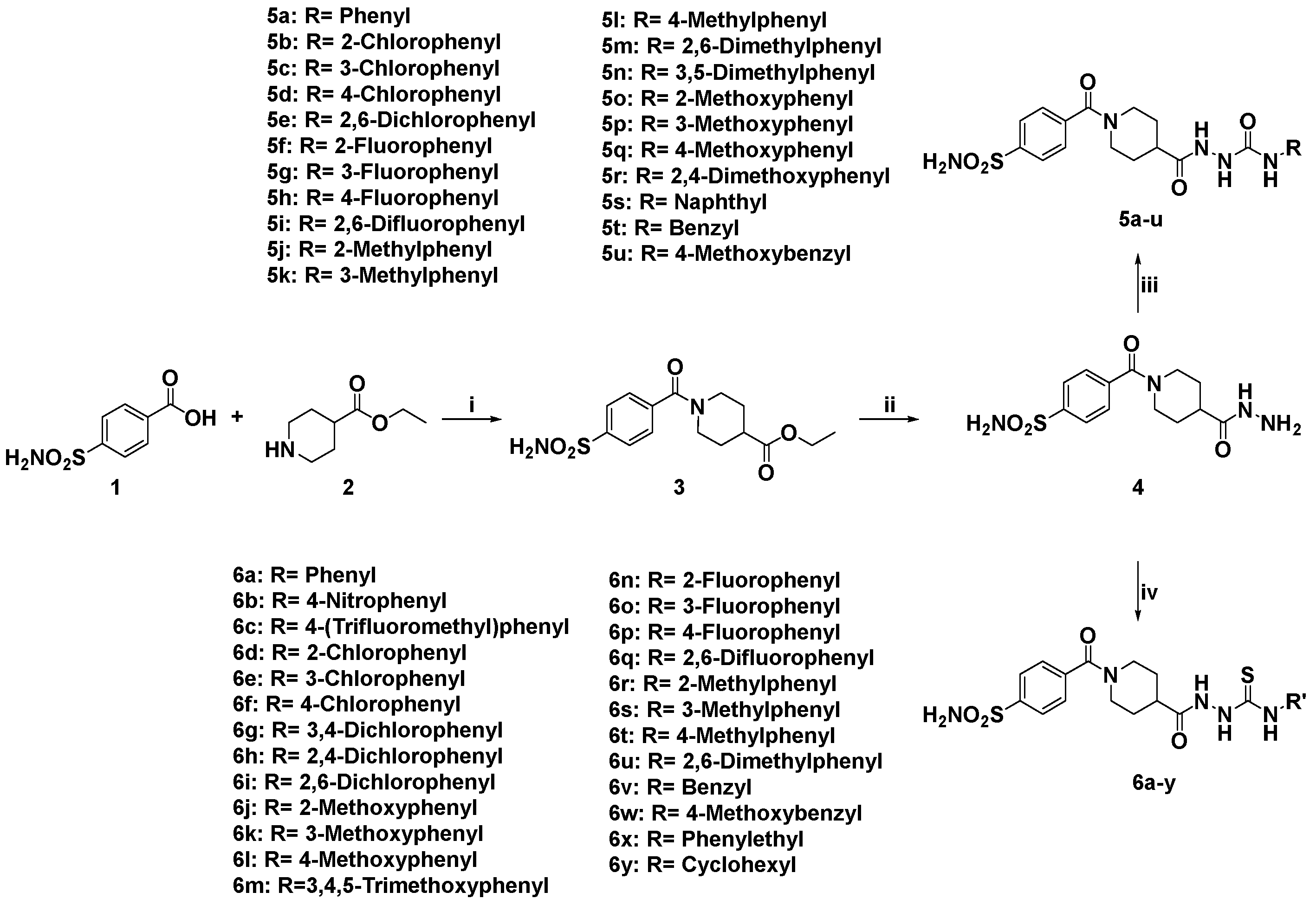
 | |||||
|---|---|---|---|---|---|
| Compound | R | Ki (nM) | |||
| hCA I | hCA II | hCA IX | hCA XII | ||
| 5a | Phenyl | 93.3 | 29.3 | 16.5 | 22.8 |
| 5b | 2-Chlorophenyl | 163.8 | 15.3 | 25.1 | 13.6 |
| 5c | 3-Chlorophenyl | 217.9 | 5.1 | 37.8 | 44.6 |
| 5d | 4-Chlorophenyl | 447.3 | 108.6 | 93.7 | 100.8 |
| 5e | 2,6-Dichlorophenyl | 259.4 | 68.8 | 28.9 | 16.4 |
| 5f | 2-Fluorophenyl | 562.1 | 24.6 | 22.5 | 8.4 |
| 5g | 3-Fluorophenyl | 277.2 | 17.7 | 89.7 | 19.4 |
| 5h | 4-Fluorophenyl | 60.6 | 12.1 | 2.1 | 24.0 |
| 5i | 2,6-Difluorophenyl | 184.1 | 8.6 | 10.1 | 12.7 |
| 5j | 2-Methylphenyl | 67.6 | 25.4 | 34.5 | 60.4 |
| 5k | 3-Methylphenyl | 94.5 | 114.3 | 8.2 | 15.6 |
| 5l | 4-Methylphenyl | 81.7 | 22.0 | 8.1 | 36.7 |
| 5m | 2,6-Dimethylphenyl | 363.6 | 62.1 | 25.1 | 51.2 |
| 5n | 3,5-Dimethylphenyl | 165.4 | 47.8 | 34.4 | 44.7 |
| 5o | 2-Methoxyphenyl | 451.5 | 185.0 | 8.6 | 19.6 |
| 5p | 3-Methoxyphenyl | 280.7 | 45.6 | 24.0 | 17.0 |
| 5q | 4-Methoxyphenyl | 487.2 | 99.7 | 8.9 | 9.1 |
| 5r | 2,4-Dimethoxyphenyl | 321.8 | 19.8 | 23.2 | 15.6 |
| 5s | Naphthyl | 521.5 | 101.3 | 26.7 | 60.6 |
| 5t | Benzyl | 77.8 | 48.2 | 40.0 | 20.1 |
| 5u | 4-Methoxybenzyl | 129.8 | 40.7 | 45.3 | 6.4 |
| AAZ | / | 250 | 12.5 | 25 | 5.7 |
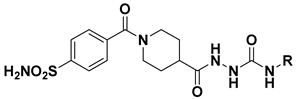
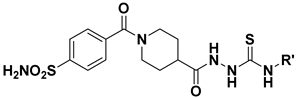 | |||||
|---|---|---|---|---|---|
| Compound | R | Ki (nM) | |||
| hCAI | hCAII | hCAIX | hCAXII | ||
| 6a | Phenyl | 30.1 | 26.5 | 28.8 | 3.2 |
| 6b | 4-Nitrophenyl | 297.9 | 60.4 | 13.6 | 45.0 |
| 6c | 4-(Trifluoromethyl)phenyl | 52.0 | 3.2 | 7.2 | 4.5 |
| 6d | 2-Chlorophenyl | 172.9 | 20.6 | 8.2 | 27.1 |
| 6e | 3-Chlorophenyl | 322.8 | 16.6 | 18.0 | 9.7 |
| 6f | 4-Chlorophenyl | 130.9 | 41.6 | 26.1 | 17.7 |
| 6g | 3,4-Dichlorophenyl | 442.3 | 22.4 | 4.7 | 26.9 |
| 6h | 2,4-Dichlorophenyl | 228.6 | 53.4 | 18.0 | 2.6 |
| 6i | 2,6-Dichlorophenyl | 1337 | 21.5 | 24.2 | 10.0 |
| 6j | 2-Methoxyphenyl | 160.3 | 13.1 | 4.2 | 4.6 |
| 6k | 3-Methoxyphenyl | 145.6 | 11.2 | 25.1 | 29.9 |
| 6l | 4-Methoxyphenyl | 211.4 | 15.1 | 22.1 | 27.8 |
| 6m | 3,4,5-Trimethoxyphenyl | 428.1 | 36.8 | 16.6 | 27.3 |
| 6n | 2-Fluorophenyl | 430.5 | 15.2 | 6.9 | 4.9 |
| 6o | 3-Fluorophenyl | 590.1 | 101.7 | 5.6 | 4.2 |
| 6p | 4-Fluorophenyl | 79.6 | 23.0 | 20.1 | 15.9 |
| 6q | 2,6-Difluorophenyl | 65.4 | 28.2 | 5.9 | 5.6 |
| 6r | 2-Methylphenyl | 277.2 | 38.5 | 9.0 | 18.4 |
| 6s | 3-Methylphenyl | 56.5 | 4.3 | 9.3 | 2.6 |
| 6t | 4-Methylphenyl | 613.2 | 58.0 | 28.4 | 31.0 |
| 6u | 2,6-Dimethylphenyl | 307.9 | 89.6 | 4.7 | 9.5 |
| 6v | Benzyl | 198.1 | 71.4 | 24.2 | 25.0 |
| 6w | 4-Methoxybenzyl | 178.3 | 29.3 | 31.7 | 29.9 |
| 6x | Phenylethyl | 76.6 | 7.6 | 2.9 | 10.4 |
| 6y | Cyclohexyl | 117.6 | 5.7 | 1.7 | 19.6 |
| AAZ | / | 250 | 12.5 | 25 | 5.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moi, D.; Deplano, A.; Angeli, A.; Balboni, G.; Supuran, C.T.; Onnis, V. Synthesis of Sulfonamides Incorporating Piperidinyl-Hydrazidoureido and Piperidinyl-Hydrazidothioureido Moieties and Their Carbonic Anhydrase I, II, IX and XII Inhibitory Activity. Molecules 2022, 27, 5370. https://doi.org/10.3390/molecules27175370
Moi D, Deplano A, Angeli A, Balboni G, Supuran CT, Onnis V. Synthesis of Sulfonamides Incorporating Piperidinyl-Hydrazidoureido and Piperidinyl-Hydrazidothioureido Moieties and Their Carbonic Anhydrase I, II, IX and XII Inhibitory Activity. Molecules. 2022; 27(17):5370. https://doi.org/10.3390/molecules27175370
Chicago/Turabian StyleMoi, Davide, Alessandro Deplano, Andrea Angeli, Gianfranco Balboni, Claudiu T. Supuran, and Valentina Onnis. 2022. "Synthesis of Sulfonamides Incorporating Piperidinyl-Hydrazidoureido and Piperidinyl-Hydrazidothioureido Moieties and Their Carbonic Anhydrase I, II, IX and XII Inhibitory Activity" Molecules 27, no. 17: 5370. https://doi.org/10.3390/molecules27175370
APA StyleMoi, D., Deplano, A., Angeli, A., Balboni, G., Supuran, C. T., & Onnis, V. (2022). Synthesis of Sulfonamides Incorporating Piperidinyl-Hydrazidoureido and Piperidinyl-Hydrazidothioureido Moieties and Their Carbonic Anhydrase I, II, IX and XII Inhibitory Activity. Molecules, 27(17), 5370. https://doi.org/10.3390/molecules27175370









