Evaluation of Toxicity and Efficacy of Inotodiol as an Anti-Inflammatory Agent Using Animal Model
Abstract
:1. Introduction
2. Results
2.1. Repeated dose Toxicity Study
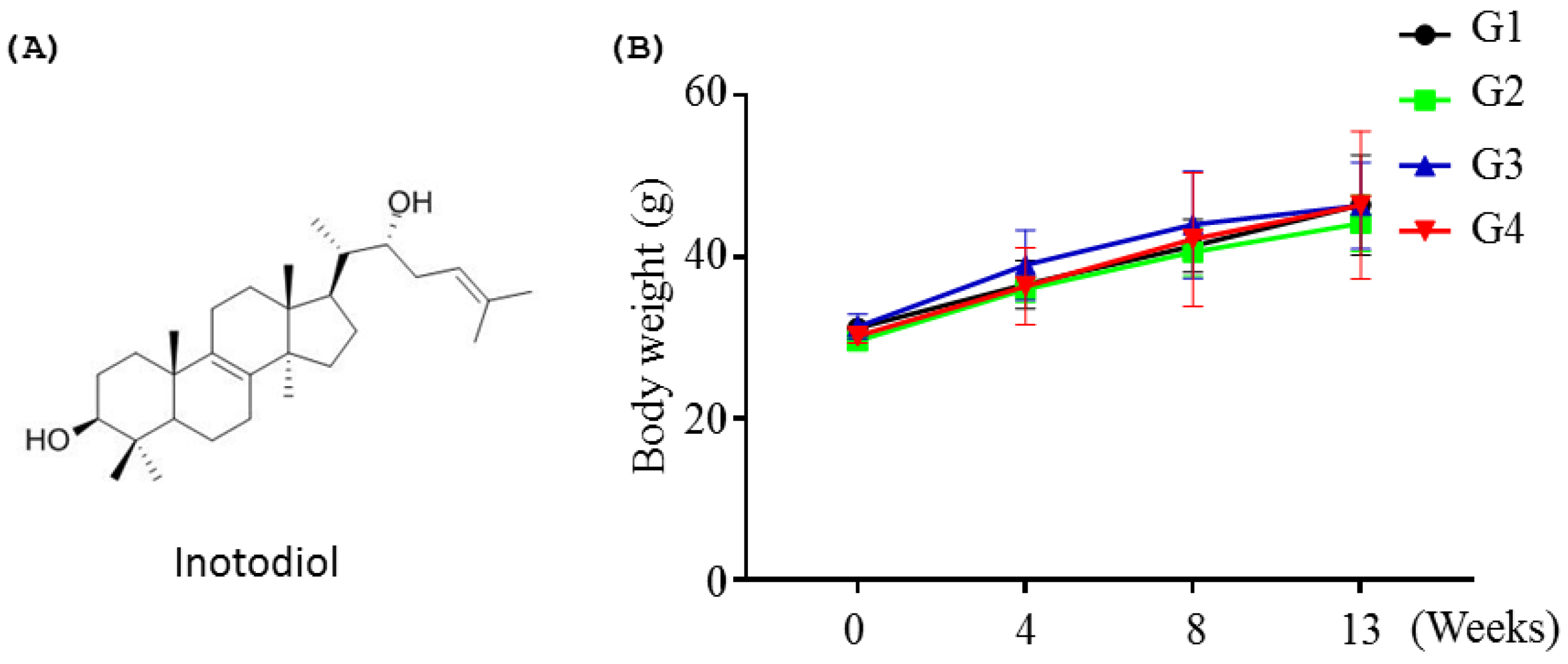
2.1.1. Effects of Inotodiol on Body Weights
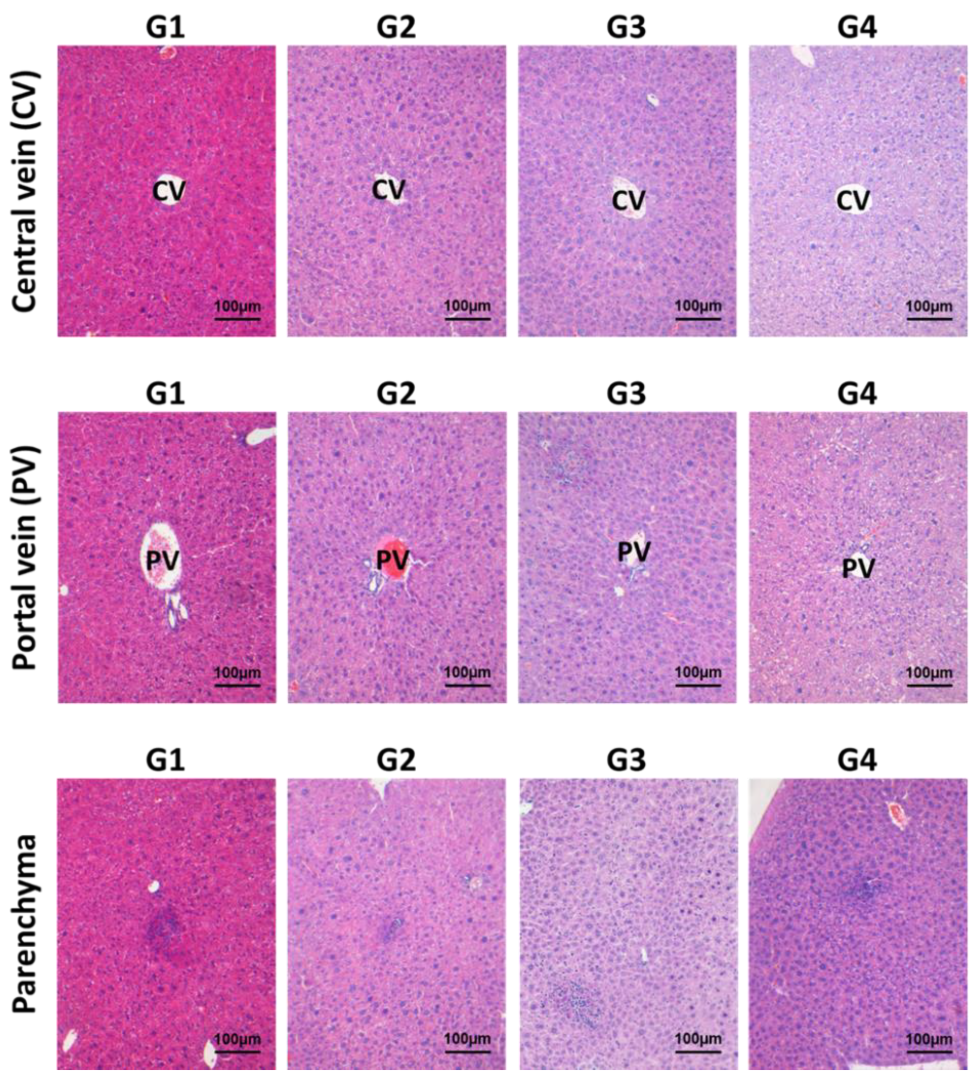
2.1.2. Effects of Inotodiol on Hepatopoietic System
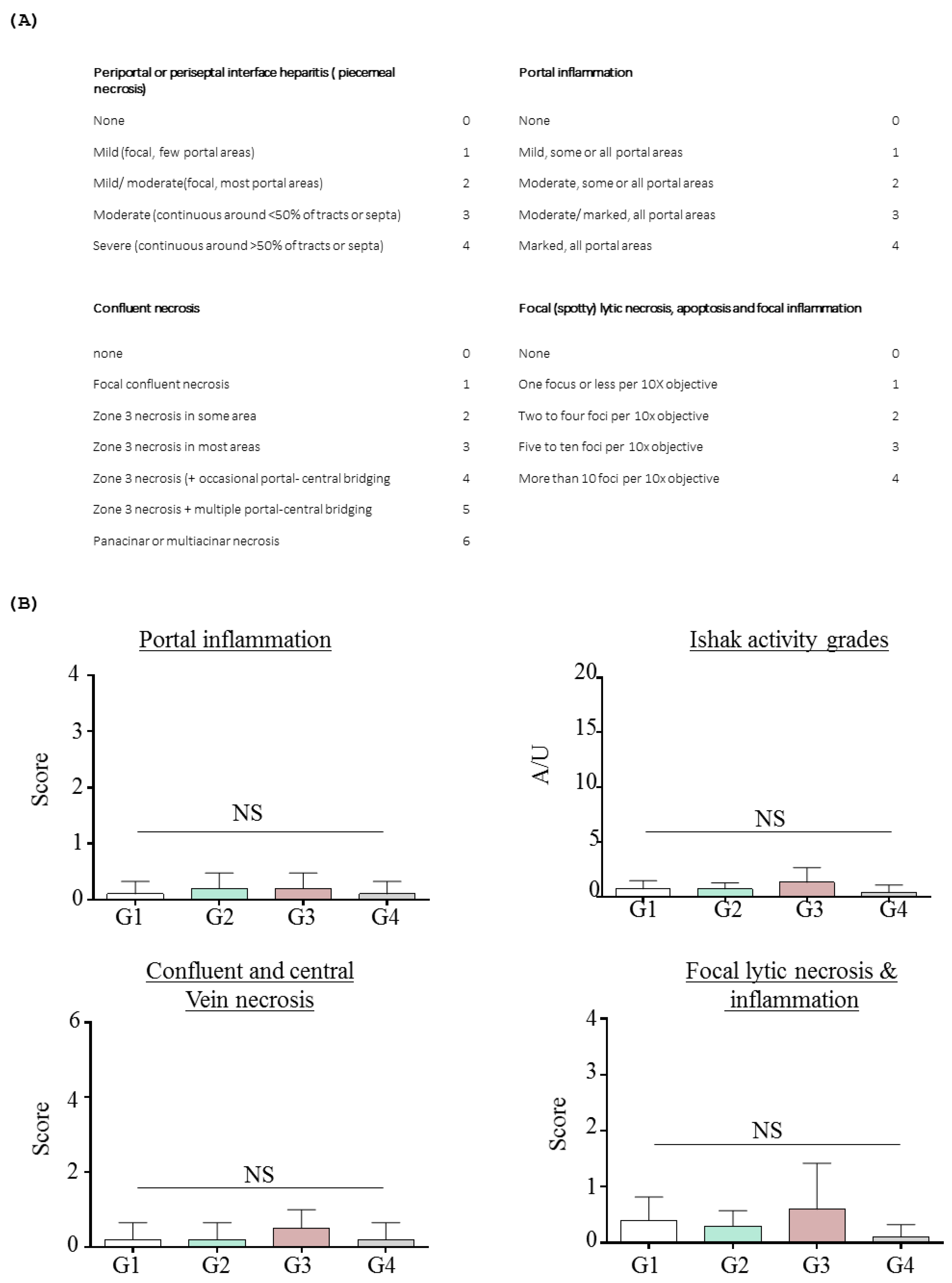
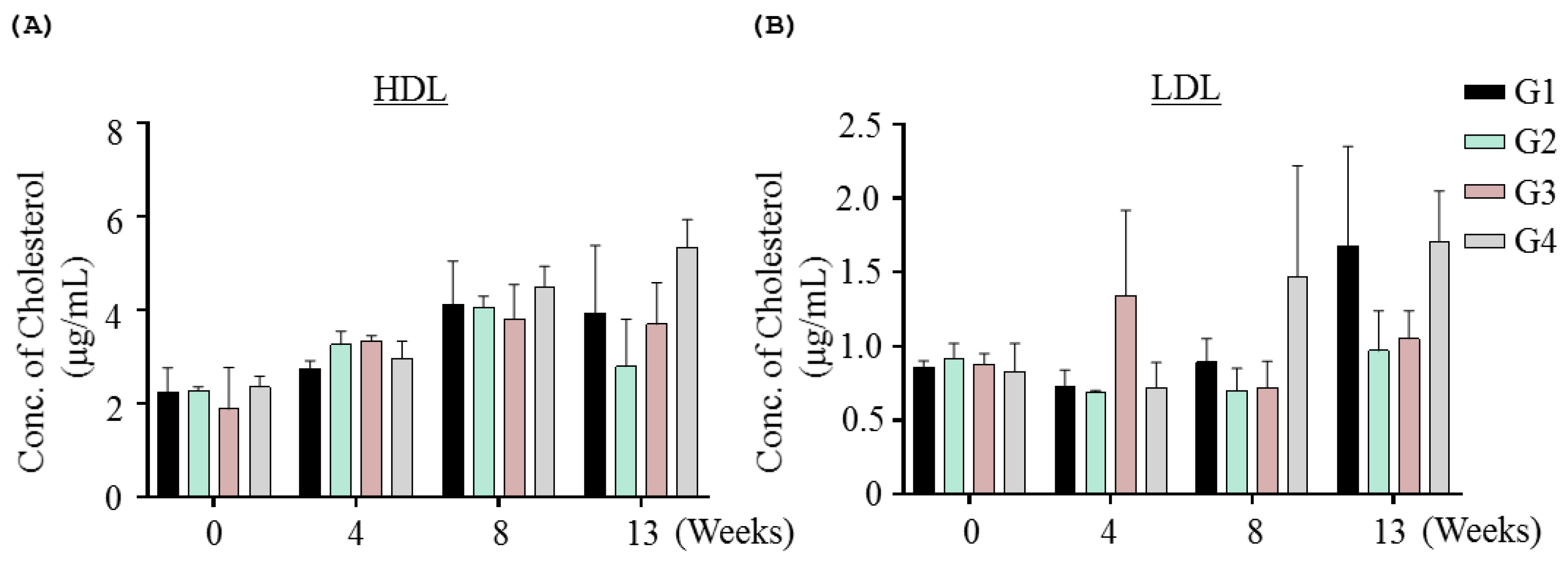
2.1.3. Effects of Inotodiol on Biochemical Parameters and Histology
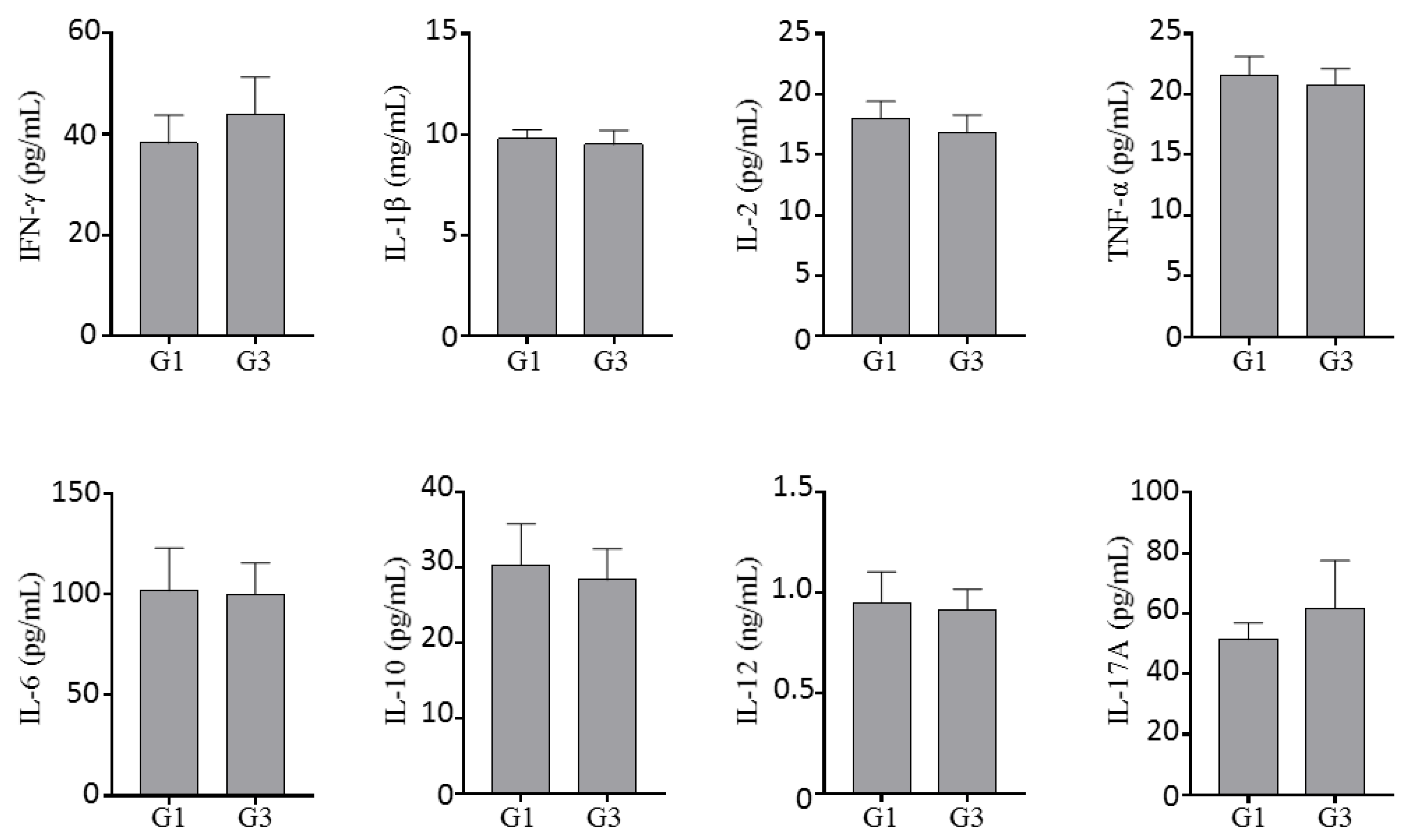
2.1.4. Effect of Inotodiol on Cytokine Production
2.1.5. Prediction of Toxicity and ADME of Inotodiol
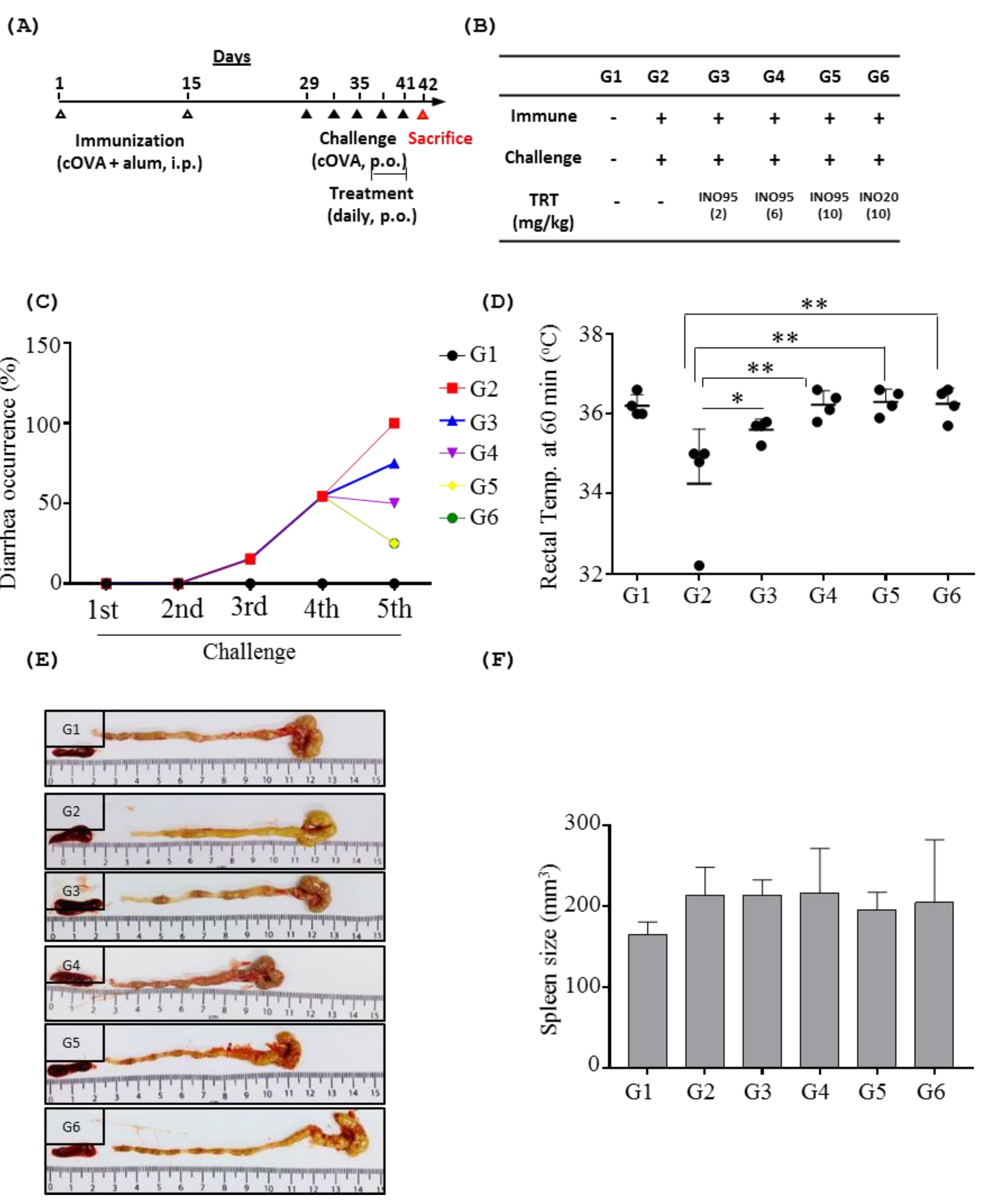
2.2. Effects of Inotodiol on Food Allergic Symptoms
2.2.1. Overall Symptoms
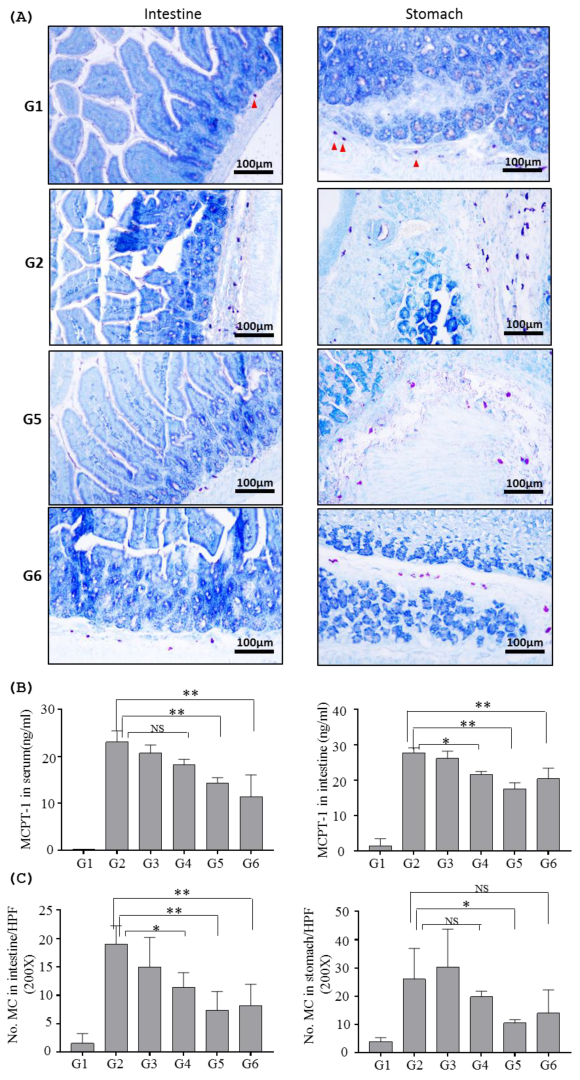
2.2.2. Effects of Inotodiol on Mast Cells
3. Discussion
4. Materials and Methods
4.1. Preparation of Inotodiol
4.2. Animals
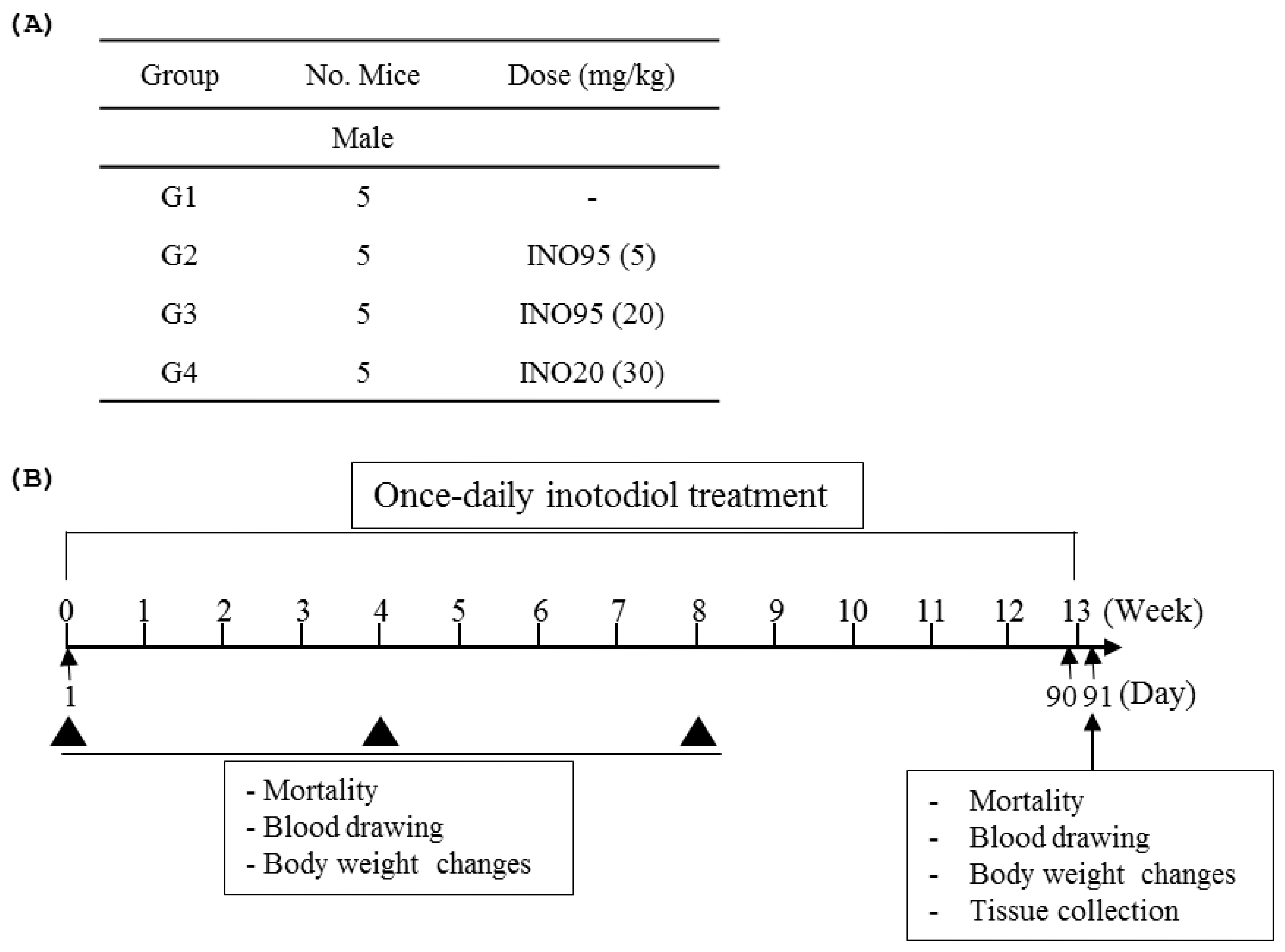
4.3. Repeated dose Toxicity Evaluation
4.3.1. Hematology and Clinical Biochemistry
4.3.2. Measuring Blood Cytokines Using Multiplex Immunoassay
4.4. Pharmacokinetic Prediction
4.4.1. Cardiotoxicity Prediction
4.4.2. Pharmacokinetic of Prediction
4.5. Experimental Mouse Model of Food Allergy
4.5.1. Preparation of the Tissue (Small Intestine) Extract
4.5.2. Measurement of the Levels of MCPT-1
4.6. Histology
4.7. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Balandaykin, M.E.; Zmitrovich, I.V. Review on Chaga Medicinal Mushroom, Inonotus obliquus (Higher Basidiomycetes): Realm of Medicinal Applications and Approaches on Estimating its Resource Potential. Int. J. Med. Mushrooms 2015, 17, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Yang, S.C.; Hwang, A.Y.; Cho, H.; Hwang, K.T. Composition of Triterpenoids in Inonotus obliquus and Their Anti-Proliferative Activity on Cancer Cell Lines. Molecules 2020, 25, 4066. [Google Scholar] [CrossRef] [PubMed]
- Du, N.; Wu, K.; Zhang, J.; Wang, L.; Pan, X.; Zhu, Y.; Wu, X.; Liu, J.; Chen, Y.; Ye, Y.; et al. Inonotsuoxide B regulates M1 to M2 macrophage polarization through sirtuin-1/endoplasmic reticulum stress axis. Int. Immunopharmacol. 2021, 96, 107603. [Google Scholar] [CrossRef]
- Li, J.; Qu, C.; Li, F.; Chen, Y.; Zheng, J.; Xiao, Y.; Jin, Q.; Jin, G.; Huang, X.; Jin, D. Inonotus obliquus Polysaccharide Ameliorates Azoxymethane/Dextran Sulfate Sodium-Induced Colitis-Associated Cancer in Mice via Activation of the NLRP3 Inflammasome. Front. Pharmacol. 2021, 11, 621835. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zheng, W. Deciphering the antitumoral potential of the bioactive metabolites from medicinal mushroom Inonotus obliquus. J. Ethnopharmacol. 2021, 265, 113321. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Wang, M.; Zhao, Z.; Hu, J.; Zhang, J.; Liu, P. Effect of Different Pretreatment of Birch Sawdust on the Production of Active Polysaccharides by Inonotus obliquus Under Submerged Fermentation and Its Structural Mechanism. Appl. Biochem. Biotechnol. 2021, 193, 1545–1557. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Yu, Y.; Sang, R.; Ge, B.; Wang, M.; Zhou, H.; Zhang, X. Inonotus obliquus polysaccharide protects against adverse pregnancy caused by Toxoplasma gondii infection through regulating Th17/Treg balance via TLR4/NF-κB pathway. Int. J. Biol. Macromol. 2020, 146, 832–840. [Google Scholar] [CrossRef]
- Wold, C.W.; Gerwick, W.H.; Wangensteen, H.; Inngjerdingen, K.T. Bioactive triterpenoids and water-soluble melanin from Inonotus obliquus (Chaga) with immunomodulatory activity. J. Funct. Foods 2020, 71, 104025. [Google Scholar] [CrossRef]
- Xu, T.; Lv, C.; Wang, X.; Du, X.; Wang, T. Optimization of extraction of polysaccharides from Inonotus obliquus by response surface methodology and antioxidant activity. Food Res. Dev. 2021, 42, 143–148. [Google Scholar]
- Park, Y.-M.; Won, J.-H.; Kim, Y.-H.; Choi, J.-W.; Park, H.-J.; Lee, K.-T. In vivo and in vitro anti-inflammatory and anti-nociceptive effects of the methanol extract of Inonotus obliquus. J. Ethnopharmacol. 2005, 101, 120–128. [Google Scholar] [CrossRef]
- Kim, H.G.; Kim, C.H.; Shrestha, B.; Chang, W.C.; Lim, S.Y.; Lee, W.H.; Han, S.G.; Lee, J.O.; Lim, M.H.; Kim, G.Y.; et al. Ethanol Extract of Inonotus obliquus Inhibits Lipopolysaccharide-Induced Inflammation in RAW 264.7 Macrophage Cells. J. Med. Food 2007, 10, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Hyun, C.-K. Insulin-Sensitizing and Beneficial Lipid-Metabolic Effects of the Water-Soluble Melanin Complex Extracted from Inonotus obliquus. Phytother. Res. 2014, 28, 1320–1328. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.K.; Kang, J.-H.; Kim, D.-K.; Oh, S.H.; Kim, M.K. Orally administered aqueous extract of Inonotus obliquus ameliorates acute inflammation in dextran sulfate sodium (DSS)-induced colitis in mice. J. Ethnopharmacol. 2012, 143, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Yoon, T.J.; Lee, S.J.; Kim, E.Y.; Cho, E.H.; Kang, T.B.; Yu, K.-W.; Suh, H.J. Inhibitory effect of chaga mushroom extract on compound 48/80-induced anaphylactic shock and IgE production in mice. Int. Immunopharmacol. 2013, 15, 666–670. [Google Scholar] [CrossRef] [PubMed]
- Nguyet, T.M.; Lomunova, M.; Le, B.V.; Lee, J.S.; Park, S.K.; Kang, J.S.; Kim, Y.H.; Hwang, I. The mast cell stabilizing activity of Chaga mushroom critical for its therapeutic effect on food allergy is derived from inotodiol. Int. Immunopharmacol. 2018, 54, 286–295. [Google Scholar] [CrossRef]
- Nguyen, T.M.N.; Le, H.S.; Le, B.V.; Kim, Y.H.; Hwang, I. Anti-allergic effect of inotodiol, a lanostane triterpenoid from Chaga mushroom, via selective inhibition of mast cell function. Int. Immunopharmacol. 2020, 81, 106244. [Google Scholar] [CrossRef]
- Maza, P.A.M.A.; Lee, J.-H.; Kim, Y.-S.; Sun, G.-M.; Sung, Y.-J.; Ponomarenko, L.P.; Stonik, V.A.; Ryu, M.; Kwak, J.-Y. Inotodiol From Inonotus obliquus Chaga Mushroom Induces Atypical Maturation in Dendritic Cells. Front. Immunol. 2021, 12, 650841. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [Green Version]
- Ul Hassan, S.S.; Muhammad, I.; Abbas, S.Q.; Hassan, M.; Majid, M.; Jin, H.-Z.; Bungau, S. Stress Driven Discovery of Natural Products From Actinobacteria with Anti-Oxidant and Cytotoxic Activities Including Docking and ADMET Properties. Int. J. Mol. Sci. 2021, 22, 11432. [Google Scholar] [CrossRef]
- Elsayed, E.A.; El Enshasy, H.; Wadaan, M.A.M.; Aziz, R. Mushrooms: A Potential Natural Source of Anti-Inflammatory Compounds for Medical Applications. Mediators Inflamm. 2014, 2014, 805841. [Google Scholar] [CrossRef]
- Warren, C.M.; Jiang, J.; Gupta, R.S. Epidemiology and Burden of Food Allergy. Curr. Allergy Asthma Rep. 2020, 20, 6. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Y.; Ma, Y.; Deng, Z.; Wang, Z.; Ji, L. Cytokines as potential biomarkers of liver toxicity induced by Dioscorea bulbifera L. Biosci. Trends 2014, 8, 32–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, M.; Gao, P.; Wu, X.; Chen, Y.; Feng, Y.; Yang, Q.; Xu, Y.; Zhao, J.; Xie, J. Impaired anti-inflammatory action of glucocorticoid in neutrophil from patients with steroid-resistant asthma. Respir. Res. 2016, 17, 153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macfarlane, E.; Seibel, M.J.; Zhou, H. Arthritis and the role of endogenous glucocorticoids. Bone Res. 2020, 8, 33. [Google Scholar] [CrossRef]
- Stevens, L.; Bowden, A.; Morton, M.; Tepper, J.; Tomkinson, A. Effects of Cyclosporine and Dexamethasone in a Mouse Model of Atopic Dermatitis. J. Allergy Clin. Immunol. 2008, 121, 35. [Google Scholar] [CrossRef]
- Nguyen, T.M.N.; Lomunova, M.; Shin, H.S.; Shon, D.-H.; Kim, Y.H.; Hwang, I. Anti-allergic effects of Rosae multiflorae fructus via inhibition of T cell proliferation and the mast cell function. Appl. Biol. Chem. 2017, 60, 391–402. [Google Scholar] [CrossRef]
- Stournaras, E.; Tziomalos, K. Herbal medicine-related hepatotoxicity. World J. Hepatol. 2015, 7, 2189–2193. [Google Scholar] [CrossRef]
- Venkateswaran, A.; Laffitte, B.A.; Joseph, S.B.; Mak, P.A.; Wilpitz, D.C.; Edwards, P.A.; Tontonoz, P. Control of cellular cholesterol efflux by the nuclear oxysterol receptor LXRα. Proc. Natl. Acad. Sci. USA 2000, 97, 12097–12102. [Google Scholar] [CrossRef] [Green Version]
- Kirchgessner, T.G.; Sleph, P.; Ostrowski, J.; Lupisella, J.; Ryan, C.S.; Liu, X.; Fernando, G.; Grimm, D.; Shipkova, P.; Zhang, R.; et al. Beneficial and Adverse Effects of an LXR Agonist on Human Lipid and Lipoprotein Metabolism and Circulating Neutrophils. Cell Metab. 2016, 24, 223–233. [Google Scholar] [CrossRef] [Green Version]
- Sun, J.-E.; Ao, Z.-H.; Lu, Z.-M.; Xu, H.-Y.; Zhang, X.-M.; Dou, W.-F.; Xu, Z.-H. Antihyperglycemic and antilipidperoxidative effects of dry matter of culture broth of Inonotus obliquus in submerged culture on normal and alloxan-diabetes mice. J. Ethnopharmacol. 2008, 118, 7–13. [Google Scholar] [CrossRef]
- Géry, A.; Dubreule, C.; André, V.; Rioult, J.-P.; Bouchart, V.; Heutte, N.; Eldin de Pécoulas, P.; Krivomaz, T.; Garon, D. Chaga (Inonotus obliquus), a Future Potential Medicinal Fungus in Oncology? A Chemical Study and a Comparison of the Cytotoxicity Against Human Lung Adenocarcinoma Cells (A549) and Human Bronchial Epithelial Cells (BEAS-2B). Integr. Cancer Ther. 2018, 17, 832–843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amiri, S.; Dastghaib, S.; Ahmadi, M.; Mehrbod, P.; Khadem, F.; Behrouj, H.; Aghanoori, M.-R.; Machaj, F.; Ghamsari, M.; Rosik, J.; et al. Betulin and its derivatives as novel compounds with different pharmacological effects. Biotechnol. Adv. 2020, 38, 107409. [Google Scholar] [CrossRef] [PubMed]
- Collington, S.J.; Williams, T.J.; Weller, C.L. Mechanisms underlying the localisation of mast cells in tissues. Trends Immunol. 2011, 32, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Yamaki, K.; Yoshino, S. Preventive and therapeutic effects of rapamycin, a mammalian target of rapamycin inhibitor, on food allergy in mice. Allergy 2012, 67, 1259–1270. [Google Scholar] [CrossRef] [PubMed]
- Ando, T.; Kashiwakura, J.I.; Itoh-Nagato, N.; Yamashita, H.; Baba, M.; Kawakami, Y.; Tsai, S.H.; Inagaki, N.; Takeda, K.; Iwata, T.; et al. Histamine-releasing factor enhances food allergy. J. Clin. Investig. 2017, 127, 4541–4553. [Google Scholar] [CrossRef] [Green Version]
- Srivastava, K.D.; Kattan, J.D.; Zou, Z.M.; Li, J.H.; Zhang, L.; Wallenstein, S.; Goldfarb, J.; Sampson, H.A.; Li, X.-M. The Chinese herbal medicine formula FAHF-2 completely blocks anaphylactic reactions in a murine model of peanut allergy. J. Allergy Clin. Immunol. 2005, 115, 171–178. [Google Scholar] [CrossRef]
| WBC | Unit | G1 | G2 | G3 | G4 |
|---|---|---|---|---|---|
| Neutrophils | % | 36.9 ± 17.4 | 31.0 ± 8.8 | 35.6 ± 11.8 | 29.3 ± 11.2 |
| Lymphocytes | % | 54.1 ± 17.4 | 60.9 ± 7.4 | 55.1 ± 11.2 | 63.9 ± 8.9 |
| Monocytes | % | 5.4 ± 2.7 | 3.9 ± 0.9 | 5.4 ± 1.9 | 2.5 ± 1.0 |
| Eosinophils | % | 3.2 ± 0.7 | 4.2 ± 2.2 | 3.7 ± 3.5 | 4.3 ± 2.6 |
| Basophils | % | 0.38 ± 0.08 | 0 | 0.40 ± 0.01 | 0 |
| Unit | G1 | G2 | G3 | G4 | |
|---|---|---|---|---|---|
| Liver function | |||||
| TP | g/DL | 6.49 ± 0.6 | 6.24 ± 0.2 | 6.21 ± 0.1 | 6.24 ± 0.40 |
| ALB | g/DL | 2.33 ± 0.2 | 2.18 ± 0.1 | 2.12 ± 0.1 | 2.17 ± 0.20 |
| Total bilirubin | mg/DL | 0.3 ± 0.06 | 0.26 ± 0.02 | 0.25 ± 0.06 | 0.26 ± 0.03 |
| TG | mg/DL | 227.5 ± 52.2 | 198.2 ± 49.7 | 220.9 ± 80.7 | 230.4 ± 102.7 |
| ALP | U/L | 162.9 ± 26.7 | 151.7 ± 35.4 | 130.1 ± 21.7 | 130.1 ± 21.7 |
| AST | U/L | 112.4 ± 22.2 | 123.1 ± 39.9 | 130.0 ± 29.1 | 108.5 ± 17.2 |
| ALT | U/L | 42.5 ± 8.6 | 33.4 ± 4.1 | 41.8 ± 6.8 | 35.3 ± 7.8 |
| CHO | mg/DL | 196.3 ± 40.1 | 206.6 ± 28.7 | 198.3 ± 23.2 | 209.9 ± 52.6 |
| GLU | mg/DL | 181.3 ± 19.4 | 201.7 ± 19.5 | 201.3 ± 29.9 | 170.0 ± 19.4 |
| Kidney function | |||||
| SCr | mg/DL | 0.49 ± 0.14 | 0.46 ± 0.04 | 0.50 ± 0.05 | 0.48 ± 0.07 |
| BUN | mg/DL | 31.1 ± 10.7 | 31.9 ± 1.1 | 31.5 ± 4.1 | 31.0 ± 3.1 |
| Ca | mg/DL | 11.5 ± 0.5 | 11.7 ± 0.31 | 12.3 ± 0.9 | 12.3 ± 0.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nguyen, T.M.N.; Ban, S.-Y.; Park, K.-B.; Lee, C.-K.; Lee, S.-W.; Lee, Y.-J.; Baek, S.-M.; Park, J.-K.; Nguyen, M.T.T.; Kim, J.; et al. Evaluation of Toxicity and Efficacy of Inotodiol as an Anti-Inflammatory Agent Using Animal Model. Molecules 2022, 27, 4704. https://doi.org/10.3390/molecules27154704
Nguyen TMN, Ban S-Y, Park K-B, Lee C-K, Lee S-W, Lee Y-J, Baek S-M, Park J-K, Nguyen MTT, Kim J, et al. Evaluation of Toxicity and Efficacy of Inotodiol as an Anti-Inflammatory Agent Using Animal Model. Molecules. 2022; 27(15):4704. https://doi.org/10.3390/molecules27154704
Chicago/Turabian StyleNguyen, Thi Minh Nguyet, So-Young Ban, Kyu-Been Park, Chang-Kyu Lee, Seoung-Woo Lee, Young-Jin Lee, Su-Min Baek, Jin-Kyu Park, My Tuyen Thi Nguyen, Jaehan Kim, and et al. 2022. "Evaluation of Toxicity and Efficacy of Inotodiol as an Anti-Inflammatory Agent Using Animal Model" Molecules 27, no. 15: 4704. https://doi.org/10.3390/molecules27154704
APA StyleNguyen, T. M. N., Ban, S.-Y., Park, K.-B., Lee, C.-K., Lee, S.-W., Lee, Y.-J., Baek, S.-M., Park, J.-K., Nguyen, M. T. T., Kim, J., Park, J., & Park, J.-T. (2022). Evaluation of Toxicity and Efficacy of Inotodiol as an Anti-Inflammatory Agent Using Animal Model. Molecules, 27(15), 4704. https://doi.org/10.3390/molecules27154704








