Determination of 2-Acetyl-1-pyrroline via a Color-Change Reaction Using Chromium Hexacarbonyl
Abstract
:1. Introduction
2. Results and Discussion
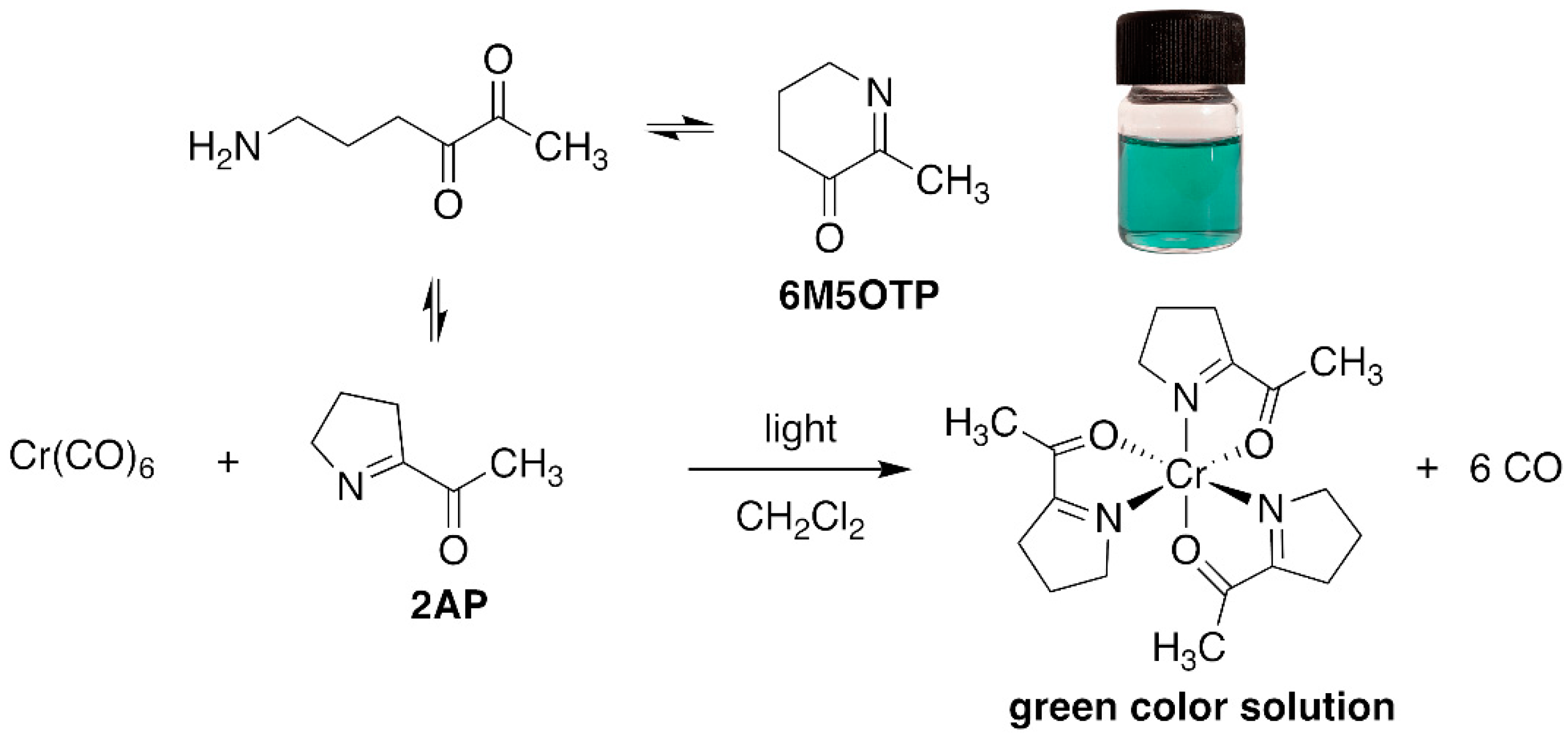
2.1. Synthetic 2AP and Its Reaction with Reagents
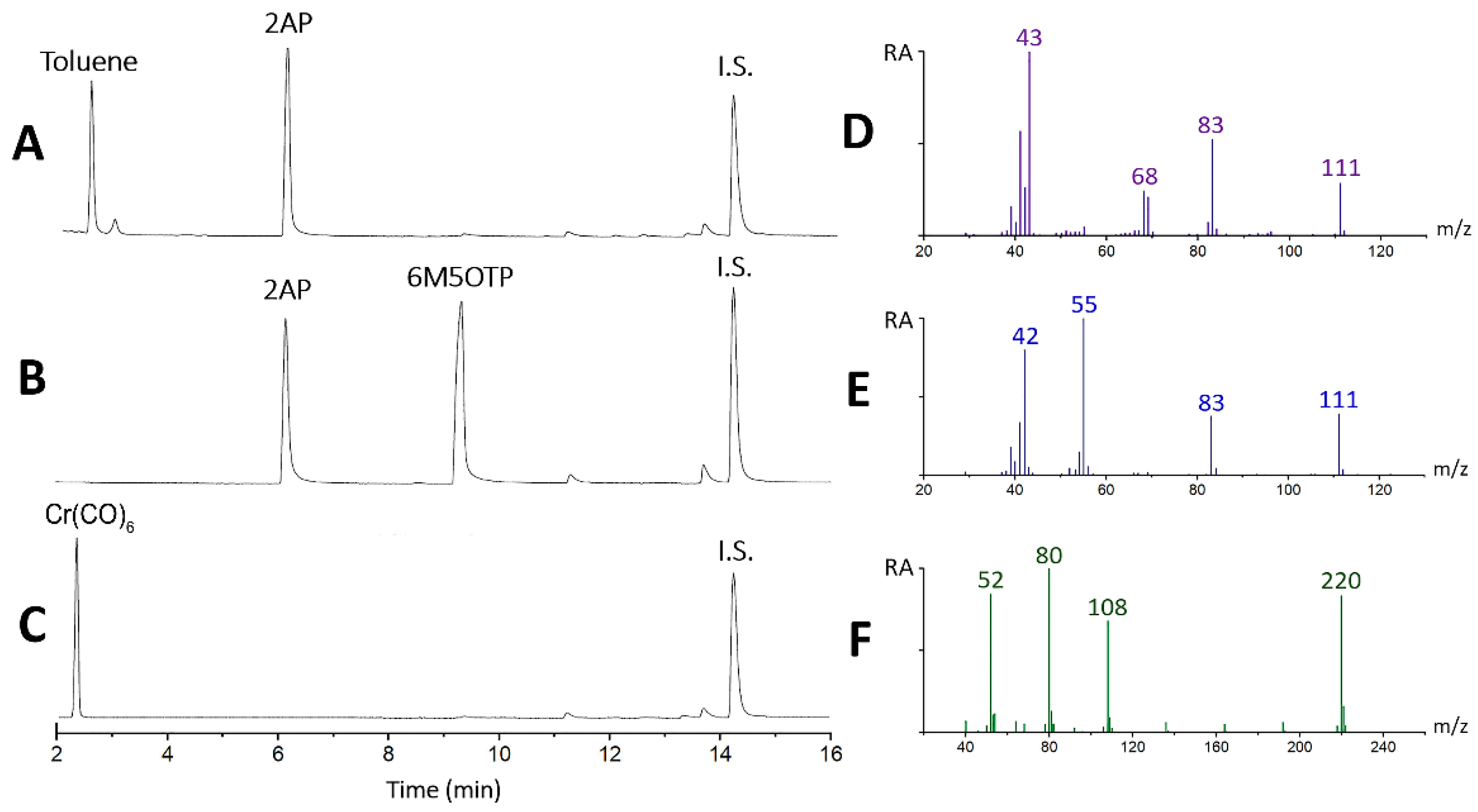
2.2. Confirmation of the Interaction between 2AP and Cr(CO)6 via GC–MS
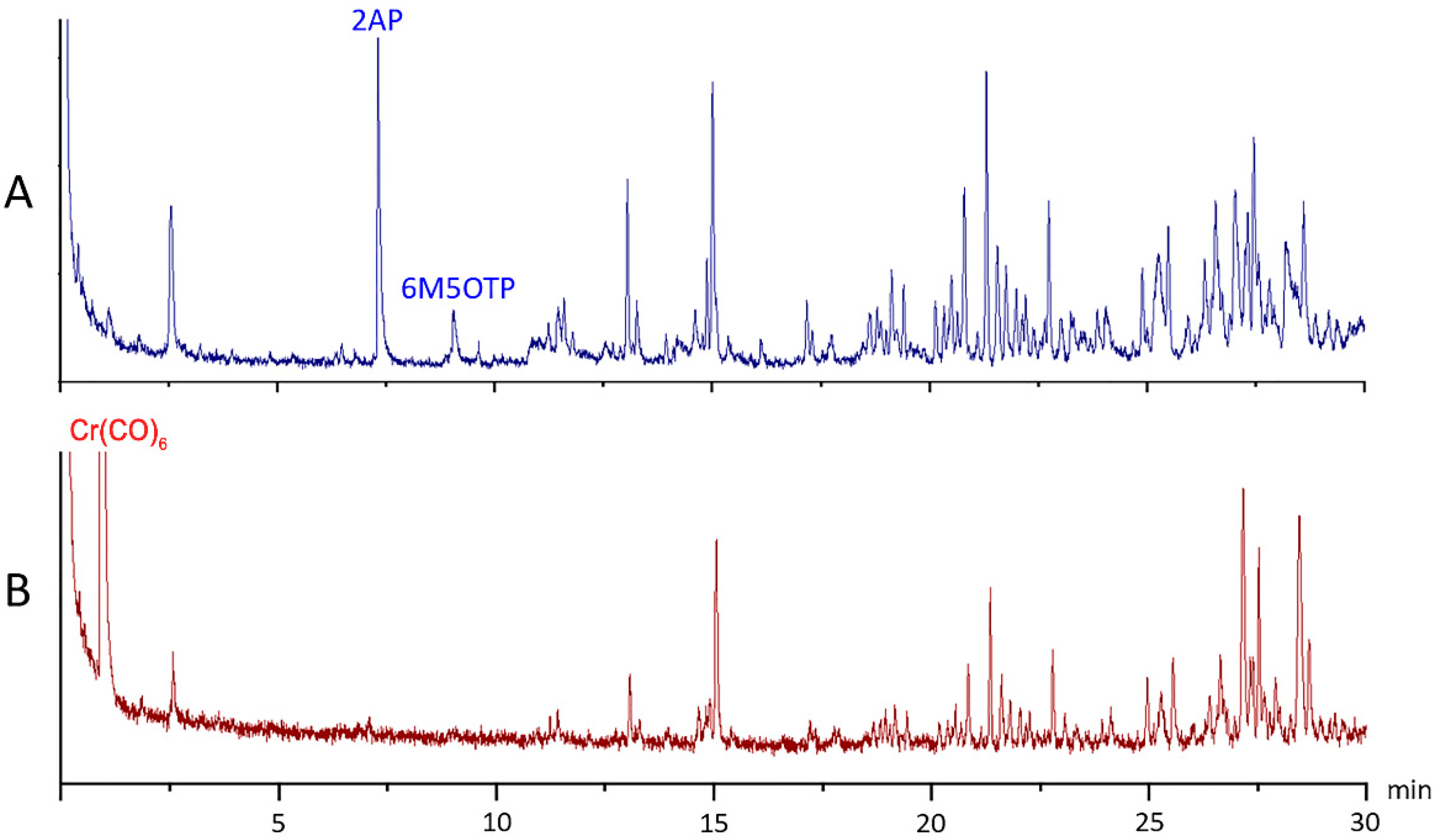
2.3. Specificity of the Colorimetric Method for the Determination of 2AP
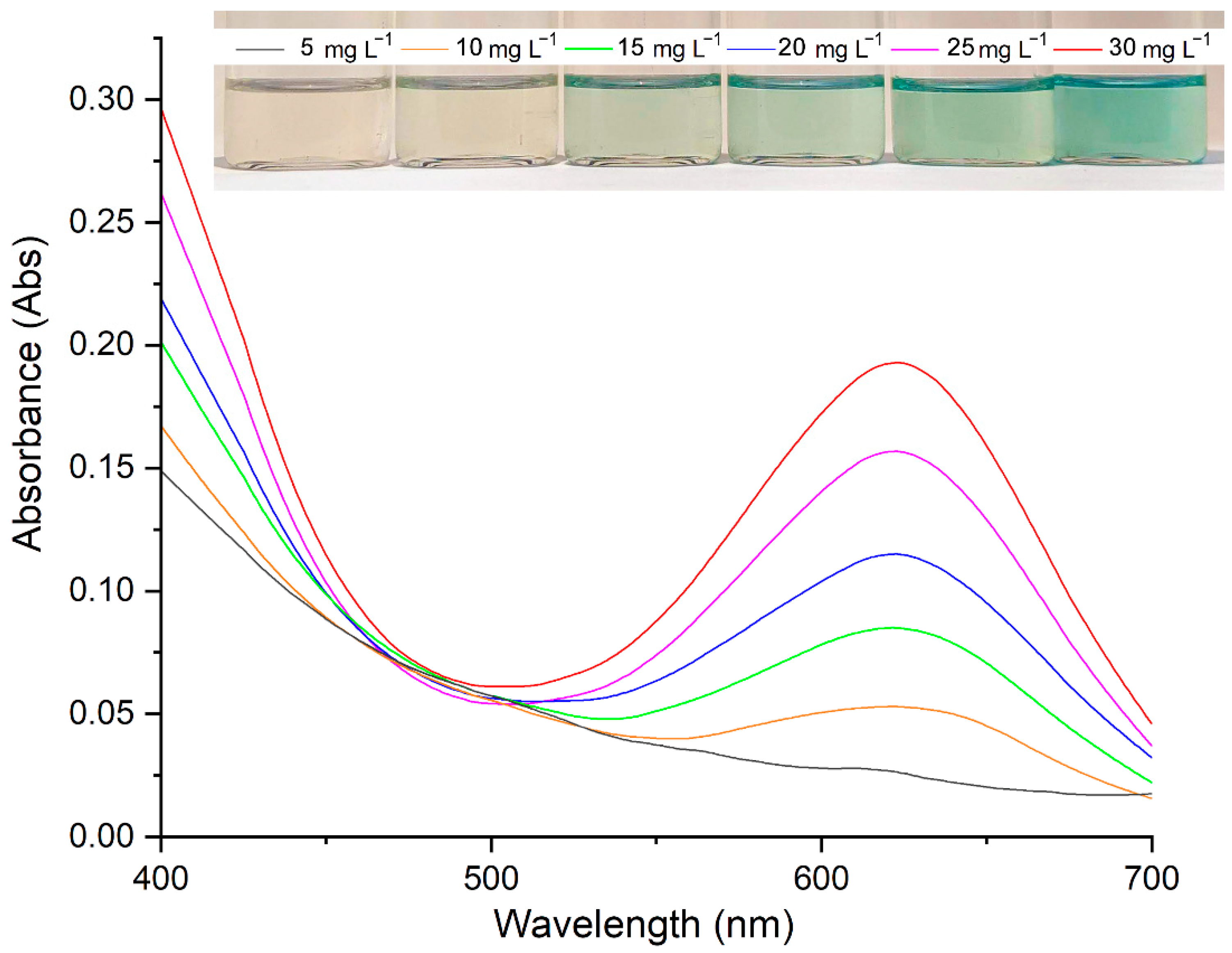
2.4. Analytical Performance and Validation
2.5. Comparison of the 2AP Content Determined by SHS-GC–NPD and the Colorimetric Method
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Synthesis of 2-Acetyl-1-pyrroline
3.2.1. Silver (I) Carbonate on Celite
3.2.2. 1-(Pyrrolidin-2-yl)ethan-1-ol
3.2.3. 2-Acetyl-1-pyrroline (2AP)
3.3. Plant Materials and Extraction
3.4. GC–MS Analysis
3.5. Comprehensive Two-Dimensional Gas Chromatography–Mass Spectrometry (GC×GC–MS) Analysis
3.6. Chemical Reaction of 2AP with Reagents to form Colored Products
3.7. Colorimetric Method
3.8. SHS-GC–NPD
3.9. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Wongpornchai, S.; Sriseadka, T.; Choonvisase, S. Identification and Quantitation of the Rice Aroma Compound, 2-Acetyl-1-pyrroline, in Bread Flowers (Vallaris glabra Ktze). J. Agric. Food Chem. 2003, 51, 457–462. [Google Scholar] [CrossRef] [PubMed]
- Adams, A.; Kimpe, N. Chemistry of 2-Acetyl-1-Pyrroline, 6-Acetyl-1,2,3,4- Tetrahydropyridine, 2-Acetyl-2-Thiazoline, and 5-Acetyl-2,3-Dihydro-4H-Thiazine: Extraordinary Maillard Flavor Compounds. Chem. Rev. 2006, 106, 2299–2319. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, M.; Warita, Y.; Takahisa, E.; Ohkubo, Y. Human-Environmental interactions: Flavor and fragrance. In Comprehensive Natural Products II; Elsiver: Amsterdam, The Netherlands, 2010; pp. 595–629. [Google Scholar]
- Juwattanasomran, R.; Somta, P.; Chankaew, S.; Shimizu, T.; Wongpornchai, S.; Kaga, A.; Srinives, P. A SNP in GmBADH2 gene associates with fragrance in vegetable soybean variety “Kaori” and SNAP marker development for the fragrance. Theor. Appl. Genet. 2011, 122, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Yundaeng, C.; Somta, P.; Tangphatsornruang, S.; Wongpornchai, S.; Srinives, P. Gene discovery and fuctional marker development for fragrance in sorghum (Sorghum bicolour (L.) Moench). Theor. Appl. Genet. 2013, 126, 2897–2906. [Google Scholar] [CrossRef]
- Saensuk, C.; Wanchana, S.; Choowongkomon, K.; Wongpornchai, S.; Kraithong, T.; Imsabai, W.; Chaichoompu, E.; Ruanjaichon, V.; Toojinda, T.; Vanavichit, A.; et al. De novo transciptome assembly and identification of the gene conferring a “pandan-like” aroma in coconut (Cocos nucifera L.). Plant Sci. 2016, 252, 324–334. [Google Scholar] [CrossRef]
- Ruangnam, S.; Wanchana, S.; Phoka, N.; Saeansuk, C.; Mahatheeranont, S.; Jan de Hoop, S.; Toojinda, T.; Vanavichit, A.; Arikit, S. A deletion of the gene encoding amino aldehyde dehydrogenase enhances the “pandan-like” aroma of winter melon (Benincasa hispida) and is a functional marker for the development of the aroma. Theor. Appl. Genet. 2017, 130, 2557–2565. [Google Scholar] [CrossRef]
- Dumhai, R.; Wanchana, S.; Saensuk, C.; Choowongkomon, K.; Mahatheeranont, S.; Kraithong, T.; Toojinda, T.; Vanavichit, A.; Arikit, S. Discovery of a novel CnAMADH2 allele associated with higher levels of 2-acetyl-1-pyrroline (2AP) in yellow dwarf coconut (Cocos nucifera L.). Sci. Hortic. 2019, 243, 490–497. [Google Scholar] [CrossRef]
- Jost, T.; Heymann, T.; Glomb, M. Efficient Analysis of 2 Acetyl-1-pyrroline in Foods Using a Novel Derivatization Strategy and LC-MS/MS. J. Agric. Food Chem. 2019, 67, 3046−3054. [Google Scholar] [CrossRef]
- Verma, D.K.; Srivastav, P.P. Extraction, identificatino and quantification methods of rice aroma compounds with emphasis on 2acetyl-1-pyrroline (2AP) and its relationship with rice quality: A comprehensive review. Food Rev. Int. 2022, 38, 111–162. [Google Scholar] [CrossRef]
- Sriseadka, T.; Wongpornchai, S.; Kitsawatpaiboon, P. Rapid method for quantitative analysis of the aroma impact compound, 2-acetyl-1-pyrroline, in fragrant rice using automated headspace gas chromatography. J. Agric. Food Chem. 2006, 54, 8183–8189. [Google Scholar] [CrossRef]
- Bryant, R.J.; McClung, A.M. Volatile profiles of aromatic and non-aromatic rice cultivars using SPME/GC-MS. Food Chem. 2011, 124, 501–513. [Google Scholar] [CrossRef]
- Zeng, Z.; Zhang, H.; Zhang, T.; Tamogami, S.; Chen, J.Y. Analysis of flavor volatiles of glutinous rice during cooking by combined gas chromatography-mass spectrometry with modified headspace solid-phase microextraction method. J. Food Compost. Anal. 2009, 22, 347–353. [Google Scholar] [CrossRef]
- Nadaf, A.B.; Krishnan, S.; Wakte, K.V. Histochemical and biochemical analysis of major aroma compound (2-acetyl-1-pyrroline) in basmati and other scented rice (Oryza sativa L.). Curr Sci. 2006, 91, 1533–1536. [Google Scholar]
- Lin, H.; Man, Z.; Kang, W.; Guan, B.; Chen, Q.; Xue, Z. A novel colorimetric sensor array based on boron-dipyrromethene dyes for monitoring the storage time of rice. Food Chem. 2018, 268, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Arslan, M.; Zareef, M.; Tahir, H.E.; Guo, Z.; Rakha, A.; Xuetao, H.; Shi, J.; Zhihua, L.; Xiaobo, Z.; Khan, M.R. Discrimination of rice varieties using smartphone-based colorimetric sensor arrays and gas chromatography techniques. Food Chem. 2022, 368, 130783. [Google Scholar] [CrossRef] [PubMed]
- Buttery, R.; Ling, L.; Juliano, B.; Turnbaugh, J. Cooked rice aroma and 2-acetyl-1-pyrroline. J. Agric. Food Chem. 1983, 31, 823–826. [Google Scholar] [CrossRef]
- De Kimpe, N.; Stevens, C.; Keppens. Synthesis of 2-acetyl-1-pyrroline, the principal rice flavor component. J. Agric. Food Chem. 1993, 41, 1458–1461. [Google Scholar] [CrossRef]
- Kolliker, S.; Oehme, M.; Dye, C. Structure elucidation of 2,4-dinitrophenylhydrazone derivatives of carbonyl compounds in ambient air by HPLC/MS and multiple MS/MS using atmospheric chemical ionization in the negative ion mode. Anal. Chem. 1998, 70, 1979–1985. [Google Scholar] [CrossRef]
- Xiong, B.; Wang, Y.; Liu, Y.; Bao, Y.; Liu, Z.; Zhang, Y.; Ling, Y. Straightforward synthesis of quinolines from enones and 2-aminobenzyl alcohols using an iridium-catalysed transfer hydrogenative strategy. Org. Biomol. Chem. 2018, 16, 5707–5711. [Google Scholar] [CrossRef]
- Soliman, A.A.; Ali, S.A.; Orabi, A. Spectral and thermal studies of some chromium and molybdenum complexes with ONO donor Schiff bases. Spectrochim. Acta A Mol. Biomol. 2006, 65, 841–845. [Google Scholar] [CrossRef]
- Fang, M.C.; Cadwallader, K.R. Stabilization of the potent odorant 2-acetyl-1-pyrroline and structural analogues by complexation with zinc halides. J. Agric. Food Chem. 2014, 62, 8808–8813. [Google Scholar] [CrossRef] [PubMed]
- El-Medani, S.M.; Sharaby, C.M.; Ramadan, R.M. Reactions and spectroscopic studies of group 6 metal carbonyls with pyrazinecarboxamide and certain phosphine ligands. J Coord. Chem. 2005, 58, 1241–1248. [Google Scholar] [CrossRef]
- Daygon, V.D.; Calingacion, M.; Forster, L.C.; De Voss, J.J.; Schwartz, B.D.; Ovenden, B.; Alonso, D.E.; McCouch, S.R.; Garson, M.J.; Fitzgerald, M.A. Metabolomics and genomics combine to unravel the pathway for the presence of fragrance in ice. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jianming, G.; Xiaohuei, X. Identification of 2-acetylpyridine in Xiangjing-8618 rice and in Yahonkaoluo leaves. Food Chem. 2002, 78, 163–166. [Google Scholar] [CrossRef]
- Noenplab, A.; Immark, S.; Palawisut, S.; Srivantaneeyaku, S.; Noenplab, A.; Patirupanusara, P.; Patirupanusara, T.; Chiengwattana, N.; Suwanthada, S.; Chairinte, S.; et al. Non-glutinous rice variety Phitsanulok 80. Thai Rice Res. J. 2008, 2, 59–70. [Google Scholar]
- Fétizon, M. Silver (I) Carbonate on Celite. EROS. In Encyclopedia of Reagents for Organic Synthesis; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2001. [Google Scholar] [CrossRef]
- Boontakham, P.; Sookwong, P.; Jongkaewwattana, S.; Wangtueai, S.; Mahatheeranont, S. Comparison of grain yield and 2-acetyl-1-pyrroline (2AP) content in leaves and grain of two Thai fragrant rice cultivars cultivated at greenhouse and open-air conditions. Aust. J. Crop Sci. 2019, 13, 159–169. [Google Scholar] [CrossRef]

| Reagent | Structure | λmax (nm) of the Colored Product | Product Color |
|---|---|---|---|
| Aniline |  | 244 | - |
| Aniline hydrochloride | 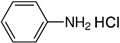 | 254 | - |
| Aniline sulphate | 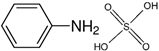 | 253 | - |
| p-Nitroaniline |  | 226 | - |
| p-Nitroaniline hydrochloride | 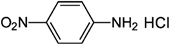 | 255 | - |
| Phenyl hydrazine | 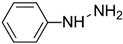 | 244 | - |
| Phenyl hydrazine hydrochloride | 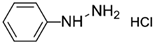 | 230 | - |
| 2,4-Dinitrophenyl hydrazine | 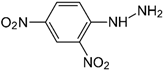 | 380 | - |
| 2-Aminobenzoic acid | 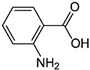 | 342 | - |
| 2-Aminobenzonitrile |  | 314 | - |
| 2-Aminobenzaldehyde | 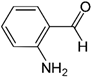 | 430 | Light yellow |
| Dicobalt octacarbonyl (Co2(CO)8) | 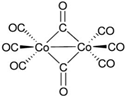 | - | - |
| Chromium hexacarbonyl (Cr(CO)6) | 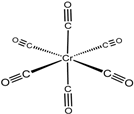 | 623 | Green |
| Sample | Concentration of 2AP, µg g−1 (Mean ± SD) | |
|---|---|---|
| Colorimetry | SHS-GC–NPD | |
| Khao dawk mali 105 (KDML105) | 3.83 ± 0.09 | 3.68 ± 0.07 |
| Phitsanulok 80 (PSL80) | 2.11 ± 0.14 | 1.95 ± 0.10 |
| Phitsanulok 2 (PSL2) | ND | ND |
| Rice department 79 (RD79) | ND | ND |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bennett, C.; Sriyotai, W.; Wiratchan, S.; Semakul, N.; Mahatheeranont, S. Determination of 2-Acetyl-1-pyrroline via a Color-Change Reaction Using Chromium Hexacarbonyl. Molecules 2022, 27, 3957. https://doi.org/10.3390/molecules27123957
Bennett C, Sriyotai W, Wiratchan S, Semakul N, Mahatheeranont S. Determination of 2-Acetyl-1-pyrroline via a Color-Change Reaction Using Chromium Hexacarbonyl. Molecules. 2022; 27(12):3957. https://doi.org/10.3390/molecules27123957
Chicago/Turabian StyleBennett, Chonlada, Woraprapa Sriyotai, Sirakorn Wiratchan, Natthawat Semakul, and Sugunya Mahatheeranont. 2022. "Determination of 2-Acetyl-1-pyrroline via a Color-Change Reaction Using Chromium Hexacarbonyl" Molecules 27, no. 12: 3957. https://doi.org/10.3390/molecules27123957
APA StyleBennett, C., Sriyotai, W., Wiratchan, S., Semakul, N., & Mahatheeranont, S. (2022). Determination of 2-Acetyl-1-pyrroline via a Color-Change Reaction Using Chromium Hexacarbonyl. Molecules, 27(12), 3957. https://doi.org/10.3390/molecules27123957






