Oxidative Stress and Antioxidative Therapy in Pulmonary Arterial Hypertension
Abstract
1. Introduction
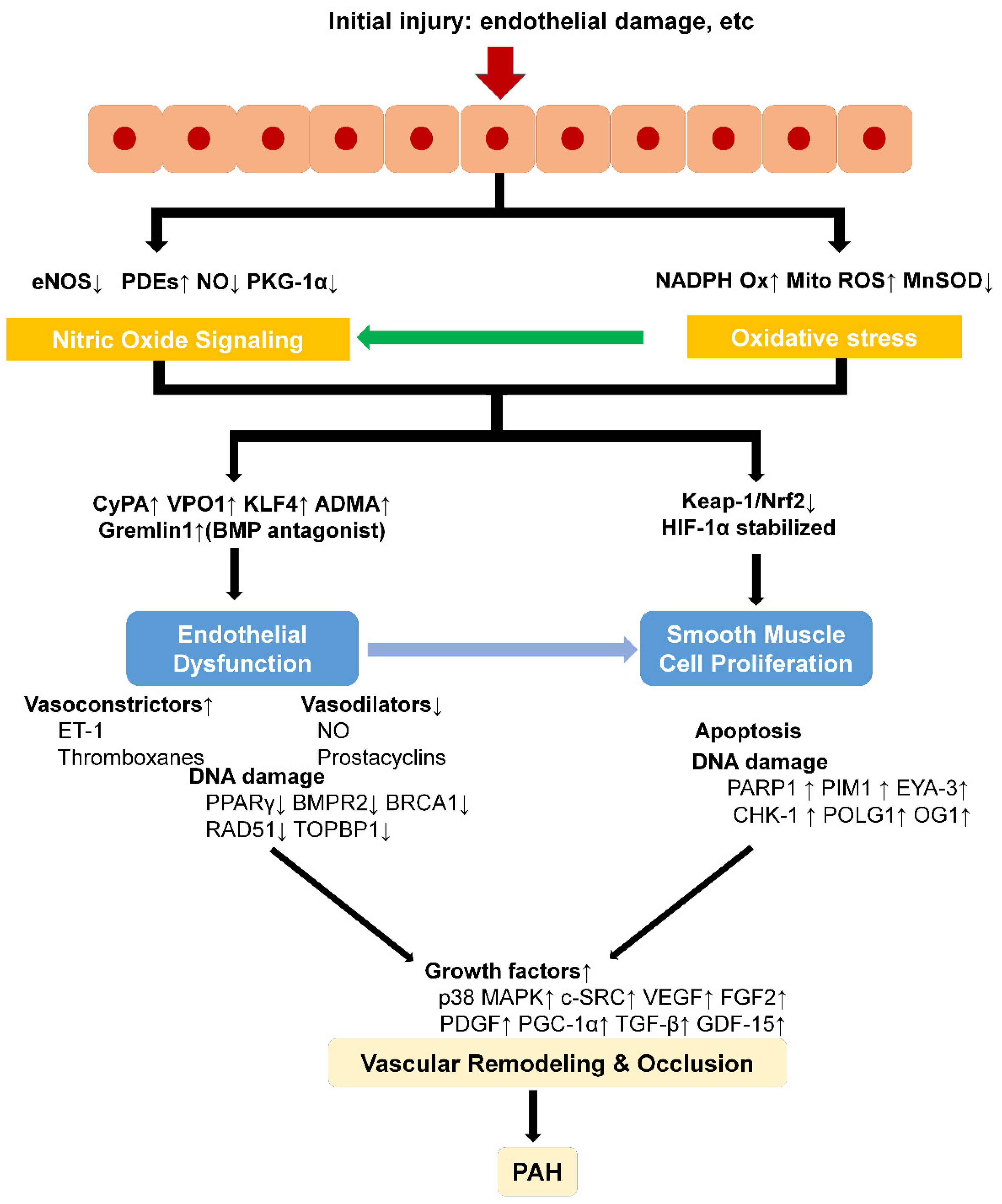
2. Oxidative Stress, DNA Damage, and Oxidative Metabolism in PAH
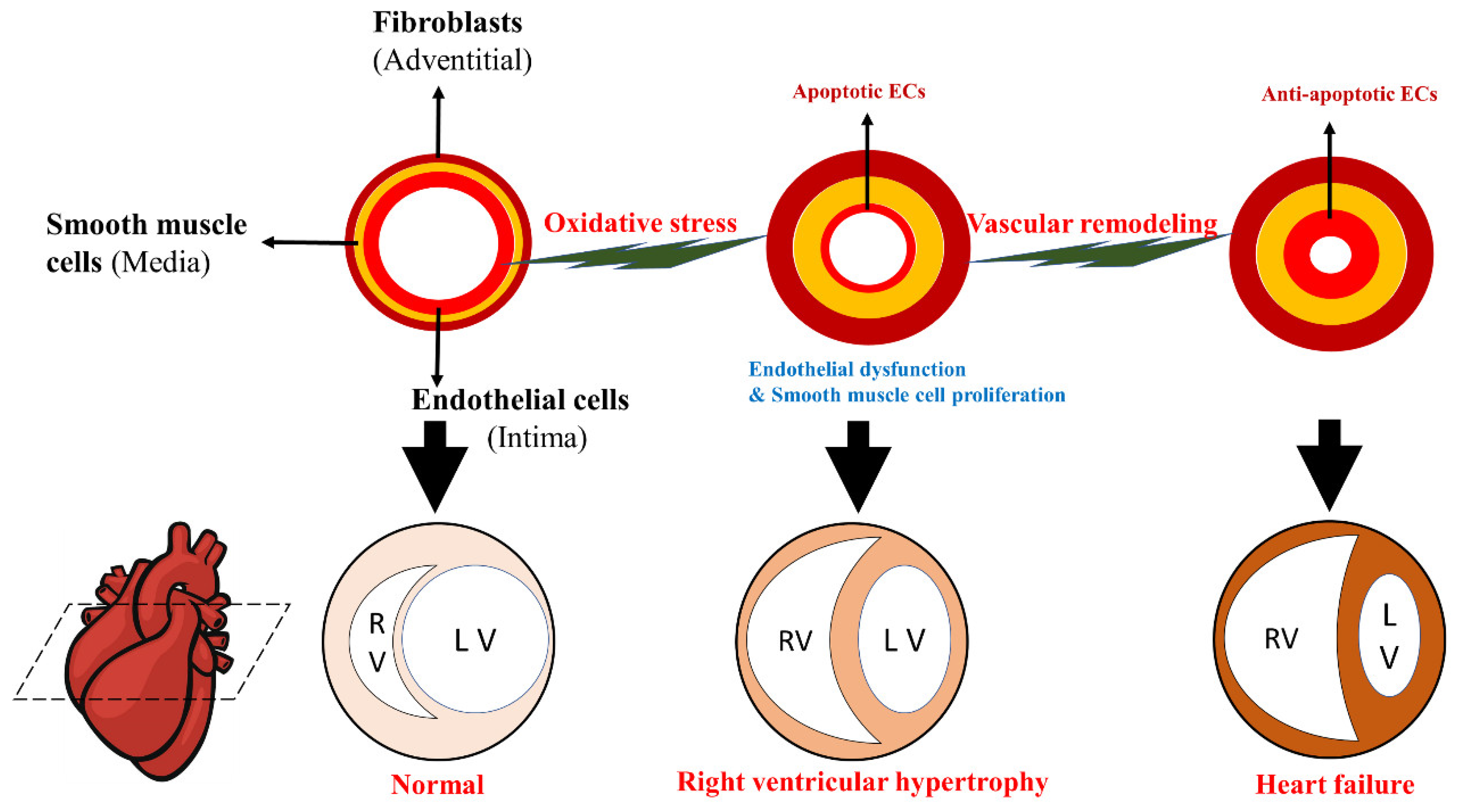
3. Oxidative Stress Signal Transduction in Pulmonary Hypertension Vascular Remodeling
4. Oxidative Stress-Induced Dysfunction of Pulmonary Artery ECs
5. Oxidative Stress Signaling-Associated Proliferation of PASMCs
6. Oxidative Stress and Its Signal Transduction in Right Ventricular Hypertrophy
7. Oxidative Stress Is Involved in the Formation of Pulmonary Hypertension through Growth Factors and Their Signal Transduction Systems
8. General Advancements in the Antioxidant Treatment of PAH
8.1. Current PAH Treatments
8.2. Latest Strategies for Oxidative Stress in PAH
8.2.1. Preclinical Strategies: Antioxidants
8.2.2. Preclinical Strategies: Herbal Treatments
8.2.3. Clinical Trials
9. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Galiè, N.; Humbert, M.; Vachiery, J.; Gibbs, S.; Lang, I.; Torbicki, A.; Simonneau, G.; Peacock, A.; Vonk Noordegraaf, A.; Beghetti, M.; et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur. Heart J. 2016, 37, 67–119. [Google Scholar] [CrossRef]
- Hoeper, M.M.; Ghofrani, H.A.; Grünig, E.; Klose, H.; Olschewski, H.; Rosenkranz, S. Pulmonary Hypertension. Dtsch. Arztebl. Int. 2017, 114, 73–84. [Google Scholar] [CrossRef]
- Bourgeois, A.; Omura, J.; Habbout, K.; Bonnet, S.; Boucherat, O. Pulmonary arterial hypertension: New pathophysiological insights and emerging therapeutic targets. Int. J. Biochem. Cell Biol. 2018, 104, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Foshat, M.; Boroumand, N. The Evolving Classification of Pulmonary Hypertension. Arch. Pathol. Lab. Med. 2017, 141, 696–703. [Google Scholar] [CrossRef] [PubMed]
- Sahay, S. Evaluation and classification of pulmonary arterial hypertension. J. Thorac. Dis. 2019, 11, S1789–S1799. [Google Scholar] [CrossRef] [PubMed]
- Coons, J.C.; Pogue, K.; Kolodziej, A.R.; Hirsch, G.A.; George, M.P. Pulmonary Arterial Hypertension: A Pharmacotherapeutic Update. Curr. Cardiol. Rep. 2019, 21, 141. [Google Scholar] [CrossRef] [PubMed]
- Simonneau, G.; Montani, D.; Celermajer, D.S.; Denton, C.P.; Gatzoulis, M.A.; Krowka, M.; Williams, P.G.; Souza, R. Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur. Respir. J. 2019, 53, 1801913. [Google Scholar] [CrossRef] [PubMed]
- Vonk Noordegraaf, A.; Groeneveldt, J.A.; Bogaard, H.J. Pulmonary hypertension. Eur. Respir. Rev. 2016, 25, 4–11. [Google Scholar] [CrossRef] [PubMed]
- Mandras, S.A.; Mehta, H.S.; Vaidya, A. Pulmonary Hypertension: A Brief Guide for Clinicians. Mayo Clin. Proc. 2020, 95, 1978–1988. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Ahn, M.J.; Chan, A.; Wang, C.H.; Kang, J.H.; Kim, S.B.; Bello, M.; Arora, R.S.; Zhang, Q.; He, X.; et al. Afatinib versus methotrexate as second-line treatment in Asian patients with recurrent or metastatic squamous cell carcinoma of the head and neck progressing on or after platinum-based therapy (LUX-Head & Neck 3): An open-label, randomised phase III trial. Ann. Oncol. 2019, 20, 1831–1839. [Google Scholar] [CrossRef]
- Dai, Z.; Zhu, M.M.; Peng, Y.; Jin, H.; Machireddy, N.; Qian, Z.; Zhang, X.; Zhao, Y. Endothelial and Smooth Muscle Cell Interaction via FoxM1 Signaling Mediates Vascular Remodeling and Pulmonary Hypertension. Am. J. Respir. Crit. Care Med. 2018, 198, 788–802. [Google Scholar] [CrossRef] [PubMed]
- Ranchoux, B.; Harvey, L.D.; Ayon, R.J.; Babicheva, A.; Bonnet, S.; Chan, S.Y.; Yuan, J.X.; Perez, V.J. Endothelial dysfunction in pulmonary arterial hypertension: An evolving landscape (2017 Grover Conference Series). Pulm. Circ. 2018, 8, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Le Ribeuz, H.; Dumont, F.; Ruellou, G.; Lambert, M.; Balliau, T.; Quatredeniers, M.; Girerd, B.; Cohen-Kaminsky, S.; Mercier, O.; Yen-Nicolay, S.; et al. Proteomic Analysis of KCNK3 Loss of Expression Identified Dysregulated Pathways in Pulmonary Vascular Cells. Int. J. Mol. Sci. 2020, 21, 7400. [Google Scholar] [CrossRef] [PubMed]
- Sakao, S.; Tatsumi, K.; Voelkel, N.F. Endothelial cells and pulmonary arterial hypertension: Apoptosis, proliferation, interaction and transdifferentiation. Respir. Res. 2009, 10, 95. [Google Scholar] [CrossRef] [PubMed]
- Humbert, M.; Montani, D.; Perros, F.; Dorfmüller, P.; Adnot, S.; Eddahibi, S. Endothelial cell dysfunction and cross talk between endothelium and smooth muscle cells in pulmonary arterial hypertension. Vasc. Pharm. 2008, 49, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Blanco, F.J.; Stevens, H.; Lu, R.; Caudrillier, A.; McBride, M.; McClure, J.D.; Grant, J.; Thomas, M.; Frid, M.; et al. MicroRNA-143 Activation Regulates Smooth Muscle and Endothelial Cell Crosstalk in Pulmonary Arterial Hypertension. Circ. Res. 2015, 117, 870–883. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.F.; Zhao, F.Y.; Xu, S.L.; Liu, J.; Xing, X.Q.; Yang, J. Autophagy in pulmonary hypertension: Emerging roles and therapeutic implications. J. Cell. Physiol. 2019, 234, 16755–16767. [Google Scholar] [CrossRef] [PubMed]
- Schlüter, K.-D.; Kutsche, H.S.; Hirschhäuser, C.; Schreckenberg, R.; Schulz, R. Review on Chamber-Specific Differences in Right and Left Heart Reactive Oxygen Species Handling. Front. Physiol. 2018, 9, 1799. [Google Scholar] [CrossRef] [PubMed]
- Bottje, W.G. Oxidative metabolism and efficiency: The delicate balancing act of mitochondria. Poult. Sci. 2019, 98, 4223–4230. [Google Scholar] [CrossRef] [PubMed]
- Marushchak, M.; Maksiv, K.; Krynytska, I.; Dutchak, O.; Behosh, N. The Severity of Oxidative Stress in Comorbid Chronic Obstructive Pulmonary Disease (COPD) and Hypertension: Does it Depend On ACE and AGT Gene Polymorphisms? J. Med. Life 2019, 12, 426–434. [Google Scholar] [CrossRef]
- Perez, M.; Robbins, M.E.; Revhaug, C.; Saugstad, O.D. Oxygen radical disease in the newborn, revisited: Oxidative stress and disease in the newborn period. Free Radic. Biol. Med. 2019, 142, 61–72. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Pan, W.; Wang, C.; Dong, H.; Xing, L.; Hou, J.; Fang, S.; Li, H.; Yang, F.; Yu, B. H2S attenuates endoplasmic reticulum stress in hypoxia-induced pulmonary artery hypertension. Biosci. Rep. 2019, 39, BSR20190304. [Google Scholar] [CrossRef] [PubMed]
- Scioli, M.G.; Storti, G.; D’Amico, F.; Rodriguez Guzman, R.; Centofanti, F.; Doldo, E.; Cespedes Miranda, E.M.; Orlandi, A. Oxidative Stress and New Pathogenetic Mechanisms in Endothelial Dysfunction: Potential Diagnostic Biomarkers and Therapeutic Targets. J. Clin. Med. 2020, 9, 1995. [Google Scholar] [CrossRef] [PubMed]
- Ranchoux, B.; Meloche, J.; Paulin, R.; Boucherat, O.; Provencher, S.; Bonnet, S. DNA Damage and Pulmonary Hypertension. Int. J. Mol. Sci. 2016, 17, 990. [Google Scholar] [CrossRef]
- Sharma, S.; Aldred, M.A. DNA Damage and Repair in Pulmonary Arterial Hypertension. Genes 2020, 11, 1224. [Google Scholar] [CrossRef]
- Leopold, J.A.; Maron, B.A. Molecular Mechanisms of Pulmonary Vascular Remodeling in Pulmonary Arterial Hypertension. Int. J. Mol. Sci. 2016, 17, 761. [Google Scholar] [CrossRef]
- Wauchope, O.R.; Mitchener, M.M.; Beavers, W.N.; Galligan, J.J.; Camarillo, J.M.; Sanders, W.D.; Kingsley, P.J.; Shim, H.N.; Blackwell, T.; Luong, T.; et al. Oxidative stress increases M1dG, a major peroxidation-derived DNA adduct, in mitochondrial DNA. Nucleic Acids Res. 2018, 46, 3458–3467. [Google Scholar] [CrossRef]
- Boucherat, O.; Peterlini, T.; Bourgeois, A.; Nadeau, V.; Breuils-Bonnet, S.; Boilet-Molez, S.; Potus, F.; Meloche, J.; Chabot, S.; Lambert, C.; et al. Mitochondrial HSP90 Accumulation Promotes Vascular Remodeling in Pulmonary Arterial Hypertension. Am. J. Respir. Crit. Care Med. 2018, 198, 90–103. [Google Scholar] [CrossRef]
- Wu, D.; Dasgupta, A.; Read, A.D.; Bentley, R.E.T.; Motamed, M.; Chen, K.H.; Al-Qazazi, R.; Mewburn, J.D.; Dunham-Snary, K.J.; Alizadeh, E.; et al. Oxygen sensing, mitochondrial biology and experimental therapeutics for pulmonary hypertension and cancer. Free Radic. Biol. Med. 2021, 170, 156–178. [Google Scholar] [CrossRef]
- Archer, S.L.; Marsboom, G.; Kim, G.H.; Zhang, H.J.; Toth, P.T.; Svensson, E.C.; Dyck, J.R.; Gomberg-Maitland, M.; Thebaud, B.; Husain, A.N.; et al. Epigenetic attenuation of mitochondrial superoxide dismutase 2 in pulmonary arterial hypertension: A basis for excessive cell proliferation and a new therapeutic target. Circulation 2010, 121, 2661–2671. [Google Scholar] [CrossRef]
- Yu, W.-C.; Chen, H.-Y.; Yang, H.-L.; Xia, P.; Zou, C.-W.; Sun, T.-W.; Wang, L.-X. rBMSC/Cav-1F92A Mediates Oxidative Stress in PAH Rat by Regulating SelW/14-3-3η and CA1/Kininogen Signal Transduction. Stem Cells Int. 2019, 2019, 6768571. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, A.; Teixeira, R.B.; Constantin, R.L.; Campos-Carraro, C.; Aparicio Cordero, E.A.; Ortiz, V.D.; Donatti, L.; Gonzalez, E.; Bahr, A.C.; Visioli, F.; et al. The progression of pulmonary arterial hypertension induced by monocrotaline is characterized by lung nitrosative and oxidative stress, and impaired pulmonary artery reactivity. Eur. J. Pharmacol. 2021, 891, 173699. [Google Scholar] [CrossRef] [PubMed]
- Rochette, L.; Lorin, J.; Zeller, M.; Guilland, J.C.; Lorgis, L.; Cottin, Y.; Vergely, C. Nitric oxide synthase inhibition and oxidative stress in cardiovascular diseases: Possible therapeutic targets? Pharmacol. Ther. 2013, 140, 239–257. [Google Scholar] [CrossRef]
- Tabima, D.M.; Frizzell, S.; Gladwin, M.T. Reactive oxygen and nitrogen species in pulmonary hypertension. Free Radic. Biol. Med. 2012, 52, 1970–1986. [Google Scholar] [CrossRef] [PubMed]
- Zolty, R. Pulmonary arterial hypertension specific therapy: The old and the new. Pharmacol. Ther. 2020, 214, 107576. [Google Scholar] [CrossRef] [PubMed]
- Crosswhite, P.; Sun, Z. Nitric oxide, oxidative stress and inflammation in pulmonary arterial hypertension. J. Hypertens. 2010, 28, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, S.; Sharma, H.; Chen, L.; Dhillon, N.K. NADPH oxidase-mediated endothelial injury in HIV- and opioid-induced pulmonary arterial hypertension. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 318, L1097–L1108. [Google Scholar] [CrossRef] [PubMed]
- Novelli, E.M.; Little-Ihrig, L.; Knupp, H.E.; Rogers, N.M.; Yao, M.; Baust, J.J.; Meijles, D.; St Croix, C.M.; Ross, M.A.; Pagano, P.J.; et al. Vascular TSP1-CD47 signaling promotes sickle cell-associated arterial vasculopathy and pulmonary hypertension in mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 2019, 316, L1150–L1164. [Google Scholar] [CrossRef]
- Wang, X.; Xiao, D.; Ma, C.; Zhang, L.; Duan, Q.; Zheng, X.; Mao, M.; Zhu, D.; Li, Q. The effect of honokiol on pulmonary artery endothelium cell autophagy mediated by cyclophilin A in hypoxic pulmonary arterial hypertension. J. Pharmacol. Sci. 2019, 139, 158–165. [Google Scholar] [CrossRef]
- Ornatowski, W.; Lu, Q.; Yegambaram, M.; Garcia, A.E.; Zemskov, E.A.; Maltepe, E.; Fineman, J.R.; Wang, T.; Black, S.M. Complex interplay between autophagy and oxidative stress in the development of pulmonary disease. Redox Biol. 2020, 36, 101679. [Google Scholar] [CrossRef]
- Xue, C.; Senchanthisai, S.; Sowden, M.; Pang, J.; White, J.; Berk, B.C. Endothelial-to-Mesenchymal Transition and Inflammation Play Key Roles in Cyclophilin A–Induced Pulmonary Arterial Hypertension. Hypertension 2020, 76, 1113–1123. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.L.; Jia, M.-M.; Luo, F.-M.; Li, T.; Peng, J.-J.; Luo, X.-J.; Song, F.-L.; Yang, J.-F.; Peng, J.; Liu, B. Coordination between NADPH oxidase and vascular peroxidase 1 promotes dysfunctions of endothelial progenitor cells in hypoxia-induced pulmonary hypertensive rats. Eur. J. Pharmacol. 2019, 857, 172459. [Google Scholar] [CrossRef] [PubMed]
- Ghouleh, I.A.; Sahoo, S.; Meijles, D.N.; Amaral, J.H.; de Jesus, D.S.; Sembrat, J.; Rojas, M.; Goncharov, D.A.; Goncharova, E.A.; Pagano, P.J. Endothelial Nox1 oxidase assembly in human pulmonary arterial hypertension; driver of Gremlin1-mediated proliferation. Clin. Sci. 2017, 131, 2019–2035. [Google Scholar] [CrossRef] [PubMed]
- Ban, Y.; Liu, Y.; Li, Y.; Zhang, Y.; Xiao, L.; Gu, Y.; Chen, S.; Zhao, B.; Chen, C.; Wang, N. S-nitrosation impairs KLF4 activity and instigates endothelial dysfunction in pulmonary arterial hypertension. Redox Biol. 2019, 21, 101099. [Google Scholar] [CrossRef]
- Ochoa, C.D.; Wu, R.F.; Terada, L.S. ROS signaling and ER stress in cardiovascular disease. Mol. Asp. Med. 2018, 63, 18–29. [Google Scholar] [CrossRef]
- Kikuchi, N.; Satoh, K.; Kurosawa, R.; Yaoita, N.; Elias-Al-Mamun, M.; Siddique, M.A.H.; Omura, J.; Satoh, T.; Nogi, M.; Sunamura, S.; et al. Selenoprotein P Promotes the Development of Pulmonary Arterial Hypertension. Circulation 2018, 138, 600–623. [Google Scholar] [CrossRef]
- Pena, E.; Brito, J.; El Alam, S.; Siques, P. Oxidative Stress, Kinase Activity and Inflammatory Implications in Right Ventricular Hypertrophy and Heart Failure under Hypobaric Hypoxia. Int. J. Mol. Sci. 2020, 21, 6421. [Google Scholar] [CrossRef]
- Shults, N.; Melnyk, O.; Suzuki, D.; Suzuki, Y. Redox Biology of Right-Sided Heart Failure. Antioxidants 2018, 7, 106. [Google Scholar] [CrossRef]
- Stam, K.; Cai, Z.; van der Velde, N.; van Duin, R.; Lam, E.; van der Velden, J.; Hirsch, A.; Duncker, D.J.; Merkus, D. Cardiac remodelling in a swine model of chronic thromboembolic pulmonary hypertension: Comparison of right vs. left ventricle. J. Physiol. 2019, 597, 4465–4480. [Google Scholar] [CrossRef]
- Ren, X.; Johns, R.A.; Gao, W.D. Right heart in pulmonary hypertension: From adaptation to failure. Pulm. Circ. 2019, 9, 1–20. [Google Scholar] [CrossRef]
- Boehm, M.; Novoyatleva, T.; Kojonazarov, B.; Veit, F.; Weissmann, N.; Ghofrani, H.A.; Seeger, W.; Schermuly, R.T. Nitric Oxide Synthase 2 Induction Promotes Right Ventricular Fibrosis. Am. J. Respir. Cell Mol. Biol. 2019, 60, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Li, H.; Weir, E.K.; Xu, Y.; Xu, D.; Chen, Y. Dimethylarginine dimethylaminohydrolase 1 deficiency aggravates monocrotaline-induced pulmonary oxidative stress, pulmonary arterial hypertension and right heart failure in rats. Int. J. Cardiol. 2019, 295, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Rudyk, O.; Rowan, A.; Prysyazhna, O.; Krasemann, S.; Hartmann, K.; Zhang, M.; Shah, A.M.; Ruppert, C.; Weiss, A.; Schermuly, R.T.; et al. Oxidation of PKGIα mediates an endogenous adaptation to pulmonary hypertension. Proc. Natl. Acad. Sci. USA 2019, 116, 13016–13025. [Google Scholar] [CrossRef] [PubMed]
- Ayinapudi, K.; Singh, T.; Motwani, A.; Le Jemtel, T.H.; Oparil, S. Obesity and Pulmonary Hypertension. Curr. Hypertens. Rep. 2018, 20, 99. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.; Galougahi, K.-K.; Celermajer, D.; Rasko, N.; Tang, O.; Bubb, K.J.; Figtree, G. Oxidative and nitrosative signalling in pulmonary arterial hypertension—Implications for development of novel therapies. Pharmacol. Ther. 2016, 165, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Larissi, K.; Politou, M.; Margeli, A.; Poziopoulos, C.; Flevari, P.; Terpos, E.; Papassotiriou, I.; Voskaridou, E. The Growth Differentiation Factor-15 (GDF-15) levels are increased in patients with compound heterozygous sickle cell and beta-thalassemia (HbS/βthal), correlate with markers of hemolysis, iron burden, coagulation, endothelial dysfunction and pulmonary hypertension. Blood Cells Mol. Dis. 2019, 77, 137–141. [Google Scholar] [CrossRef]
- Xu, T.; Shao, L.; Wang, A.; Liang, R.; Lin, Y.; Wang, G.; Zhao, Y.; Hu, J.; Liu, S. CD248 as a novel therapeutic target in pulmonary arterial hypertension. Clin. Transl. Med. 2020, 10, E175. [Google Scholar] [CrossRef]
- Weise-Cross, L.; Resta, T.C.; Jernigan, N.L. Redox Regulation of Ion Channels and Receptors in Pulmonary Hypertension. Antioxid. Redox Signal. 2019, 31, 898–915. [Google Scholar] [CrossRef]
- Mathew, R. Signaling Pathways Involved in the Development of Bronchopulmonary Dysplasia and Pulmonary Hypertension. Children 2020, 7, 100. [Google Scholar] [CrossRef]
- Fallah, F. Recent Strategies in Treatment of Pulmonary Arterial Hypertension, A Review. Glob. J. Health Sci. 2015, 7, 307. [Google Scholar] [CrossRef]
- Feng, W.; Hu, Y.; An, N.; Feng, Z.; Liu, J.; Mou, J.; Hu, T.; Guan, H.; Zhang, D.; Mao, Y. Alginate Oligosaccharide Alleviates Monocrotaline-Induced Pulmonary Hypertension via Anti-Oxidant and Anti-Inflammation Pathways in Rats. Int. Heart J. 2020, 61, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Kang, Y.; Zhang, G.; Huang, E.C.; Huang, J.; Cai, J.; Cai, L.; Wang, S.; Keller, B.B. Sulforaphane prevents right ventricular injury and reduces pulmonary vascular remodeling in pulmonary arterial hypertension. Am. J. Physiol. Heart Circ. Physiol. 2020, 318, H853–H866. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, Z.; Dianat, M.; Radan, M.; Badavi, M. Ellagic Acid Ameliorates Lung Inflammation and Heart Oxidative Stress in Elastase-Induced Emphysema Model in Rat. Inflammation 2020, 43, 1143–1156. [Google Scholar] [CrossRef]
- Gonzalez-Candia, A.; Veliz, M.; Carrasco-Pozo, C.; Castillo, R.L.; Cárdenas, J.C.; Ebensperger, G.; Reyes, R.V.; Llanos, A.J.; Herrera, E.A. Antenatal melatonin modulates an enhanced antioxidant/pro-oxidant ratio in pulmonary hypertensive newborn sheep. Redox Biol. 2019, 22, 101128. [Google Scholar] [CrossRef]
- Astorga, C.R.; González-Candia, A.; Candia, A.A.; Figueroa, E.G.; Cañas, D.; Ebensperger, G.; Reyes, R.V.; Llanos, A.J.; Herrera, E.A. Melatonin Decreases Pulmonary Vascular Remodeling and Oxygen Sensitivity in Pulmonary Hypertensive Newborn Lambs. Front. Physiol. 2018, 9, 185. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Chang, Z.; Zhao, F.; Zhang, P.; Hao, Y.-J.; Yan, L.; Liu, N.; Wang, J.-L.; Bo, L.; Ma, P.; et al. Protective Effects of 18β-Glycyrrhetinic Acid on Monocrotaline-Induced Pulmonary Arterial Hypertension in Rats. Front. Pharmacol. 2019, 10, 13. [Google Scholar] [CrossRef] [PubMed]
- Budas, G.R.; Boehm, M.; Kojonazarov, B.; Viswanathan, G.; Tian, X.; Veeroju, S.; Novoyatleva, T.; Grimminger, F.; Hinojosa-Kirschenbaum, F.; Ghofrani, H.A.; et al. ASK1 Inhibition Halts Disease Progression in Preclinical Models of Pulmonary Arterial Hypertension. Am. J. Respir. Crit. Care Med. 2018, 197, 373–385. [Google Scholar] [CrossRef] [PubMed]
- Boucherat, O.; Provencher, S.; Bonnet, S. Therapeutic Value of ASK1 Inhibition in Pulmonary Arterial Hypertension. Am. J. Respir. Crit. Care Med. 2018, 197, 284–286. [Google Scholar] [CrossRef] [PubMed]
- Lampron, M.C.; Vitry, G.; Nadeau, V.; Grobs, Y.; Paradis, R.; Samson, N.; Tremblay, E.; Boucherat, O.; Meloche, J.; Bonnet, S.; et al. PIM1 (Moloney Murine Leukemia Provirus Integration Site) Inhibition Decreases the Nonhomologous End-Joining DNA Damage Repair Signaling Pathway in Pulmonary Hypertension. Arterioscler. Thromb. Vasc. Biol. 2020, 40, 783–801. [Google Scholar] [CrossRef]
- Kurosawa, R.; Satoh, K.; Kikuchi, N.; Kikuchi, H.; Saigusa, D.; Al-Mamun, M.E.; Siddique, M.A.H.; Omura, J.; Satoh, T.; Sunamura, S.; et al. Identification of Celastramycin as a Novel Therapeutic Agent for Pulmonary Arterial Hypertension. Circ. Res. 2019, 125, 309–327. [Google Scholar] [CrossRef]
- Becker, C.U.; Sartório, C.L.; Campos-Carraro, C.; Siqueira, R.; Colombo, R.; Zimmer, A.; Belló-Klein, A. Exercise training decreases oxidative stress in skeletal muscle of rats with pulmonary arterial hypertension. Arch. Physiol. Biochem. 2020, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.-F.; Zhang, R.; Jiang, X.; Wen, L.; Wu, D.-C.; Liu, D.; Yuan, P.; Wang, Y.-L.; Jing, Z.-C. The phosphodiesterase-5 inhibitor vardenafil reduces oxidative stress while reversing pulmonary arterial hypertension. Cardiovasc. Res. 2013, 99, 395–403. [Google Scholar] [CrossRef] [PubMed]
- Steven, S.; Oelze, M.; Brandt, M.; Ullmann, E.; Kröller-Schön, S.; Heeren, T.; Tran, L.P.; Daub, S.; Dib, M.; Stalleicken, D.; et al. Pentaerythritol Tetranitrate In Vivo Treatment Improves Oxidative Stress and Vascular Dysfunction by Suppression of Endothelin-1 Signaling in Monocrotaline-Induced Pulmonary Hypertension. Oxid. Med. Cell. Longev. 2017, 2017, 4353462. [Google Scholar] [CrossRef] [PubMed]
- Dianat, M.; Radan, M.; Mard, S.A.; Sohrabi, F.; Saryazdi, S.S.N. Contribution of reactive oxygen species via the OXR1 signaling pathway in the pathogenesis of monocrotaline-induced pulmonary arterial hypertension: The protective role of Crocin. Life Sci. 2020, 256, 117848. [Google Scholar] [CrossRef]
- Xu, D.; Li, Y.; Zhang, B.; Wang, Y.; Liu, Y.; Luo, Y.; Niu, W.; Dong, M.; Liu, M.; Dong, H.; et al. Resveratrol alleviate hypoxic pulmonary hypertension via anti-inflammation and anti-oxidant pathways in rats. Int. J. Med. Sci. 2016, 13, 942–954. [Google Scholar] [CrossRef]
- Sunamura, S.; Satoh, K.; Kurosawa, R.; Ohtsuki, T.; Kikuchi, N.; Elias-Al-Mamun, M.; Shimizu, T.; Ikeda, S.; Suzuki, K.; Satoh, T.; et al. Different roles of myocardial ROCK1 and ROCK2 in cardiac dysfunction and postcapillary pulmonary hypertension in mice. Proc. Natl. Acad. Sci. USA 2018, 115, E7129–E7138. [Google Scholar] [CrossRef]
- Cheng, Y.; Gong, Y.; Qian, S.; Mou, Y.; Li, H.; Chen, X.; Kong, H.; Xie, W.; Wang, H.; Zhang, Y.; et al. Identification of a Novel Hybridization from Isosorbide 5-Mononitrate and Bardoxolone Methyl with Dual Activities of Pulmonary Vasodilation and Vascular Remodeling Inhibition on Pulmonary Arterial Hypertension Rats. J. Med. Chem. 2018, 61, 1474–1482. [Google Scholar] [CrossRef]
- Li, T.; Li, S.; Feng, Y.; Zeng, X.; Dong, S.; Li, J.; Zha, L.; Luo, H.; Zhao, L.; Liu, B.; et al. Combination of Dichloroacetate and Atorvastatin Regulates Excessive Proliferation and Oxidative Stress in Pulmonary Arterial Hypertension Development via p38 Signaling. Oxid. Med. Cell. Longev. 2020, 2020, 6973636. [Google Scholar] [CrossRef]
- Shi, R.; Wei, Z.; Zhu, D.; Fu, N.; Wang, C.; Yin, S.; Liang, Y.; Xing, J.; Wang, X.; Wang, Y. Baicalein attenuates monocrotaline-induced pulmonary arterial hypertension by inhibiting vascular remodeling in rats. Pulm. Pharmacol. Ther. 2018, 48, 124–135. [Google Scholar] [CrossRef]
- Liu, Z.; Duan, Y.L.; Ge, S.L.; Zhang, C.X.; Gong, W.H.; Xu, J.J. Effect of estrogen on right ventricular remodeling of monocrotaline-induced pulmonary arterial hypertension in rats and its mechanism. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 1742–1750. [Google Scholar] [CrossRef]
- Campos-Carraro, C.; Turck, P.; de Lima-Seolin, B.G.; Tavares, A.M.V.; Dos Santos Lacerda, D.; Corssac, G.B.; Teixeira, R.B.; Hickmann, A.; Llesuy, S.; da Rosa Araujo, A.S.; et al. Copaiba Oil Attenuates Right Ventricular Remodeling by Decreasing Myocardial Apoptotic Signaling in Monocrotaline-Induced Rats. J. Cardiovasc. Pharmacol. 2018, 72, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Grzegorzewska, A.P.; Seta, F.; Han, R.; Czajka, C.A.; Makino, K.; Stawski, L.; Isenberg, J.S.; Browning, J.L.; Trojanowska, M. Dimethyl Fumarate ameliorates pulmonary arterial hypertension and lung fibrosis by targeting multiple pathways. Sci. Rep. 2017, 7, 41605. [Google Scholar] [CrossRef]
- de Lima-Seolin, B.G.; Hennemann, M.M.; Fernandes, R.O.; Colombo, R.; Bonetto, J.H.P.; Teixeira, R.B.; Khaper, N.; Godoy, A.E.G.; Litvin, I.E.; Sander da Rosa Araujo, A.; et al. Bucindolol attenuates the vascular remodeling of pulmonary arteries by modulating the expression of the endothelin-1 A receptor in rats with pulmonary arterial hypertension. Biomed. Pharmacother. 2018, 99, 704–714. [Google Scholar] [CrossRef] [PubMed]
- DeMarco, V.G.; Habibi, J.; Whaley-Connell, A.T.; Schneider, R.I.; Sowers, J.R.; Andresen, B.T.; Gutweiler, A.A.; Ma, L.; Johnson, M.S.; Ferrario, C.M.; et al. Rosuvastatin ameliorates the development of pulmonary arterial hypertension in the transgenic (mRen2)27 rat. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H1128–H1139. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zhang, Q.; Fan, K.; Wang, P.; Yu, J.; Liu, R.; Qi, H.; Sun, H.; Cao, Y. Carvacrol induces the apoptosis of pulmonary artery smooth muscle cells under hypoxia. Eur. J. Pharmacol. 2016, 770, 134–146. [Google Scholar] [CrossRef] [PubMed]
- Türck, P.; Lacerda, D.S.; Carraro, C.C.; de Lima-Seolin, B.G.; Teixeira, R.B.; Poletto Bonetto, J.H.; Colombo, R.; Schenkel, P.C.; Belló-Klein, A.; da Rosa Araujo, A.S. Trapidil improves hemodynamic, echocardiographic and redox state parameters of right ventricle in monocrotaline-induced pulmonary arterial hypertension model. Biomed. Pharmacother. 2018, 103, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yang, Y.; Yang, D.; Tong, G.; Lv, S.; Lin, X.; Chen, C.; Dong, W. Tetrandrine prevents monocrotaline-induced pulmonary arterial hypertension in rats through regulation of the protein expression of inducible nitric oxide synthase and cyclic guanosine monophosphate-dependent protein kinase type 1. J. Vasc. Surg. 2016, 64, 1468–1477. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Luo, X.-J.; Yang, Z.-B.; Zhang, J.-J.; Li, T.-B.; Zhang, X.-J.; Ma, Q.-L.; Zhang, G.-G.; Hu, C.-P.; Peng, J. Inhibition of NOX/VPO1 pathway and inflammatory reaction by trimethoxystilbene in prevention of cardiovascular remodeling in hypoxia-induced pulmonary hypertensive rats. J. Cardiovasc. Pharmacol. 2014, 63, 567–576. [Google Scholar] [CrossRef]
- Kishimoto, Y.; Kato, T.; Ito, M.; Azuma, Y.; Fukasawa, Y.; Ohno, K.; Kojima, S. Hydrogen ameliorates pulmonary hypertension in rats by anti-inflammatory and antioxidant effects. J. Thorac. Cardiovasc. Surg. 2015, 150, 645–654. [Google Scholar] [CrossRef]
- Türck, P.; Fraga, S.; Salvador, I.; Campos-Carraro, C.; Lacerda, D.; Bahr, A.; Ortiz, V.; Hickmann, A.; Koetz, M.; Belló-Klein, A.; et al. Blueberry extract decreases oxidative stress and improves functional parameters in lungs from rats with pulmonary arterial hypertension. Nutrition 2020, 70, 110579. [Google Scholar] [CrossRef]
- Meghwani, H.; Prabhakar, P.; Mohammed, S.; Dua, P.; Seth, S.; Hote, M.; Banerjee, S.; Arava, S.; Ray, R.; Maulik, S. Beneficial Effect of Ocimum sanctum (Linn) against Monocrotaline-Induced Pulmonary Hypertension in Rats. Medicines 2018, 5, 34. [Google Scholar] [CrossRef] [PubMed]
- Black, S.M.; Nozik-Grayck, E. Compartmentalization of Redox-Regulated Signaling in the Pulmonary Circulation. Antioxid. Redox Signal. 2019, 31, 801–803. [Google Scholar] [CrossRef]
- Spiekerkoetter, E.; Kawut, S.M.; de Jesus Perez, V.A. New and Emerging Therapies for Pulmonary Arterial Hypertension. Annu. Rev. Med. 2019, 70, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.; Baird, G.; Atalay, M.; Agarwal, S.; Arcuri, D.; Klinger, J.; Mullin, C.; Morreo, H.; Normandin, B.; Shiva, S.; et al. Experimental design of the Effects of Dehydroepiandrosterone in Pulmonary Hypertension (EDIPHY) trial. Pulm. Circ. 2021, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Mi, Y.; Zhang, F.; Cheng, Y.; Wu, X. Suppression of bromodomain-containing protein 4 protects trophoblast cells from oxidative stress injury by enhancing Nrf2 activation. Hum. Exp. Toxicol. 2021, 40, 742–753. [Google Scholar] [CrossRef] [PubMed]
- Valenca, S.; Dong, B.; Gordon, E.; Sun, R.; Waters, C. ASK1 Regulates Bleomycin-induced Pulmonary Fibrosis. Am. J. Respir. Cell Mol. Biol. 2022, 66, 484–496. [Google Scholar] [CrossRef] [PubMed]

| Drugs | Type of Drug | Animals | Model | Biological Indicators | Administration | Therapeutic Effect | References |
|---|---|---|---|---|---|---|---|
| Alginate Oligosaccharide (AOS) | biodegradable polymer | Sprague-Dawley rats | MCT (i.p.) | p47-phox, p67-phox, and gp91-phox, subunits of NADPH oxidase, MDA | i.p. | Down-regulate the expressions of malondialdehyde and NADPH by inhibiting the TGF-β1 /p-Smad2 signaling pathway to prevent the pulmonary vascular remodeling induced by MCT | [61] |
| Vardenafil | Phosphodiesterase-5 inhibitor | Sprague-Dawley rats | MCT (i.p.) | 8-iso-prostaglandin-F2a, 3-nitrotyrosine, eNOS, NO, MDA, SOD, Nox2, Nox4 | i.g. | Suppress proliferation and enhanced apoptosis of pulmonary artery smooth muscle cells, attenuating small pulmonary artery remodeling, and right ventricular hypertrophy | [72] |
| Pentaerythritol Tetranitrate | —— | Wistar rats | MCT (i.v.) | HO-1, ICAM-1 | i.g. | PETN therapy improved endothelium-dependent relaxation in pulmonary arteries and reduced oxidative stress | [73] |
| Sulforaphane | Nrf2 activator | Male mice | SU5416 and 10% hypoxia (SuHx) | Nrf2, NQO1, NLRP3 | i.g. | Reduce SuHx-induced pulmonary vascular remodeling, inflammation, and fibrosis | [62] |
| Crocin | —— | Sprague-Dawley rats | MCT (i.p.) | OXR1, P21, Nrf2 | i.p. | Crocin co-treatment significantly improved the hemodynamic, oxidative stress biomarkers and histological data of the PAH rats | [74] |
| Melatonin | —— | Newborn sheep | Chronic hypobaric hypoxia (Putre, 3600 m) | SOD2, CAT, GPx1, VDAC, p47-phox, Xantine Oxidase, 8-isoprostanes, 4HNE, and nitrotyrosine | i.g. | Reduced major sources of pro-oxidative ROS at the cellular level, reduced oxidative stress and enhanced antioxidant status at the pulmonary level of neonatal PAH | [64,65] |
| Resveratrol | Polyphenolic compound | Sprague-Dawley rats | Hypoxia | Nrf2, HIF-1 α | i.g. | Exert antiproliferation, antioxidant, and anti-inflammation effects | [75] |
| Ellagic Acid | —— | Male Sprague-Dawley rats | Porcine pancreatic elastase(intratracheal) | SOD, catalase, and glutathione | i.g. | Reduce oxidative stress and prevent PAH | [63] |
| 18β-Glycyrrhetinic Acid | —— | Male Sprague-Dawley rats | MCT (i.p.) | Nox2, Nox4 | i.g. | Reduce the changes in oxidative stress biomarkers and inhibit Nox2 and Nox4 expression | [66] |
| Celastramycin | —— | Wild-type mice; SD rats | 3 wk of hypoxic exposure (10% O2); SU5416, s.c. | ROS, Nrf2, Nox, GSH/GSSG, SOD2 | Osmotic pump; i.p. | Increase protein levels of Nrf2 (nuclear factor erythrocyte-related factor 2) and improve pulmonary hypertension | [70] |
| Celastrol | Tripterygium wilfordii extractive | cROCK1−/− and cROCK2−/− mice | TAC | CyPA, Bsg, Nox2, Nox4 | i.p. | Inhibit CyPA/Bsg-NF-κB axis and enhance ROS production | [76] |
| Hybridization of Isosorbide 5 Mononitrate and Bardoxolone Methyl | A NO donor and a semisynthetic derivative of oleanolic acid | Male Sprague-Dawley rats | MCT (i.h.) | NO, Nox4 | i.t. | By inactivating Nox4, excessive proliferation of vascular pericytes was inhibited, macrophage infiltration and oxidative stress were reduced, and cardiac hypertrophy and fibrosis were significantly reduced in rats with pulmonary hypertension | [77] |
| Combination of Dichloroacetate and Atorvastatin | —— | Male Sprague-Dawley rats | MCT (i.h.) | CHOP, Bcl2 | i.g. | The combined treatment of DCA/ATO significantly reduces the right ventricular systolic blood pressure accompanied by a decrease in right heart hypertrophy and reduces vascular remodeling, thereby inhibiting excessive PASMC proliferation | [78] |
| Baicalein | Natural flavonoid | Male Sprague-Dawley rats | MCT (i.h.) | MDA, SOD, GSH-Px, Bax, Bcl-2 | i.g. | Inhibit oxidative stress and alleviated pulmonary vascular remodeling in MCT-induced PAH | [79] |
| 17-β estradiol | Estrogen | Male Sprague-Dawley rats | MCT (i.h.) | T-AOC, MDA, Nox4 | i.p. | Inhibit Nox4-mediated oxidative stress and alleviated MCT-induced right ventricular remodeling of PAH rats | [80] |
| Copaiba Oil | —— | Male Wistar rats | MCT (i.p.) | eNOS | i.g. | Reduce oxidative stress and apoptosis signaling in RV of rats with PAH | [81] |
| Dimethyl Fumarate | Antioxidative and anti-inflammatory agent | Male C57BL/6 mice | Hypoxic chamber | HO-1, NOX4 | i.p. | Mitigate oxidative stress damage and inflammation in lung | [82] |
| Bucindolol | β-adrenergic blocker | Male Wistar rats | MCT (i.p.) | eNOS, SOD-1 | i.p. | Decrease (21%) PVR and increase RV workload, thereby improving the vascular remodeling of the pulmonary artery | [83] |
| Rosuvastatin | —— | Male Ren2 and Sprague-Dawley rats | Transgenic (mRen2) 27 rats | 3-NT, NO(x), Nox, and endothelial NO synthase expression | i.p. | Improve cardiovascular outcomes/risk by restoring endothelial and SMC function, inhibiting SMC proliferation, reducing oxidative stress and inflammation in the vascular wall | [84] |
| Carvacrol | —— | Male Wistar rats | Hypoxia | SOD, GSH, MDA, caspase-3 | i.p. | Attenuate the pulmonary vascular remodeling and promotes PASMC apoptosis | [85] |
| Trapidil | —— | Male Wistar rats | MCT (i.p.) | NADPH oxidases, glutathiones/total glutathiones | i.p. | Improve hemodynamic, echocardiographic, and redox state parameters of right ventricle | [86] |
| Tetrandrine | Bisbenzylisoquinoline alkaloid | Male Sprague-Dawley rats | MCT (i.p.) | cGMP, PKG-1, iNOS | i.p. | Alleviate MCT-induced PAH through regulation of NO signaling pathway and antioxidant and antiproliferation effects | [87] |
| Trimethoxystilbene | Resveratrol analog | Male Sprague-Dawley rats | Hypoxic chamber | Nox2, Nox4, VPO1 | i.g. | Attenuate hypoxia-induced pulmonary vascular remodeling and right ventricle hypertrophy accompanied by downregulation of Nox2, Nox4, and VPO1 expression | [88] |
| Hydrogen | —— | Male Sprague-Dawley rats | MCT (i.h.) | STAT3, NFAT | Housed ad libitum to hydrogen-saturated water | Ameliorate MCT-induced PAH in rats by suppressing macrophage accumulation, reducing oxidative stress, and modulating the STAT3/NFAT axis | [89] |
| Blueberry extract | herb | Male Wistar rats | MCT (i.p.) | NADPH, SOD, GPx, ETA/ETB | i.g. | Decrease the mean pulmonary artery pressure and total reactive species concentration and lipid oxidation | [90] |
| Ocimum Sanctum (Linn) | herb | Male Wistar rats | MCT (i.h.) | Thiobarbituric Acid Reactive Substances (TBARS); GSH; Catalase; SOD; Nox1 | i.g. | Decrease Nox-1 expression and increase expression of Bcl2/Bax ratio caused by MCT | [91] |
| Honokiol | herb | Male Sprague-Dawley rats | MCT (i.p.) | CyPA | i.g. | Alleviate autophagy and PAH regulated by CyPA in PAECs | [39] |
| GS-444217/Selonsertib | ASK1 inhibitor | SD rats | MCT (i.h)/Sugen/hypoxia | phosphorylation of p38 and JNK | i.g. | Reduce pulmonary arterial pressure and RV hypertrophy in PAH models associated with reduced ASK1 phosphorylation, reduced muscularization of the pulmonary arteries, and reduced fibrotic gene expression in the RV | [67] |
| SGI-1776, TP-3654 | Pim1 inhibitor | Male SD rats | MCT (i.h)/Fawn-Hooded Rats (FHR) | Repair of DNA damage | i.g. | Improve significantly pulmonary hemodynamics (right heart catheterization) and vascular remodeling (Elastica van Gieson) | [69] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, D.; Hu, Y.-H.; Gou, X.; Li, F.-Y.; Yang, X.-Y.-C.; Li, Y.-M.; Chen, F. Oxidative Stress and Antioxidative Therapy in Pulmonary Arterial Hypertension. Molecules 2022, 27, 3724. https://doi.org/10.3390/molecules27123724
Xu D, Hu Y-H, Gou X, Li F-Y, Yang X-Y-C, Li Y-M, Chen F. Oxidative Stress and Antioxidative Therapy in Pulmonary Arterial Hypertension. Molecules. 2022; 27(12):3724. https://doi.org/10.3390/molecules27123724
Chicago/Turabian StyleXu, Dan, Ya-Hui Hu, Xue Gou, Feng-Yang Li, Xi-Yu-Chen Yang, Yun-Man Li, and Feng Chen. 2022. "Oxidative Stress and Antioxidative Therapy in Pulmonary Arterial Hypertension" Molecules 27, no. 12: 3724. https://doi.org/10.3390/molecules27123724
APA StyleXu, D., Hu, Y.-H., Gou, X., Li, F.-Y., Yang, X.-Y.-C., Li, Y.-M., & Chen, F. (2022). Oxidative Stress and Antioxidative Therapy in Pulmonary Arterial Hypertension. Molecules, 27(12), 3724. https://doi.org/10.3390/molecules27123724





