Mechanochemistry and Eco-Bases for Sustainable Michael Addition Reactions
Abstract
:1. Introduction
2. Methods
2.1. General Information
2.2. General Procedures
2.2.1. Harvest and Preparation of the Eco-bases
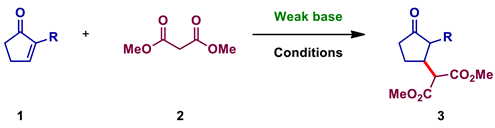
2.2.2. Procedures for Dimethyl-2-(3-oxocyclopentyl) Malonate 3
3. Results and Discussion
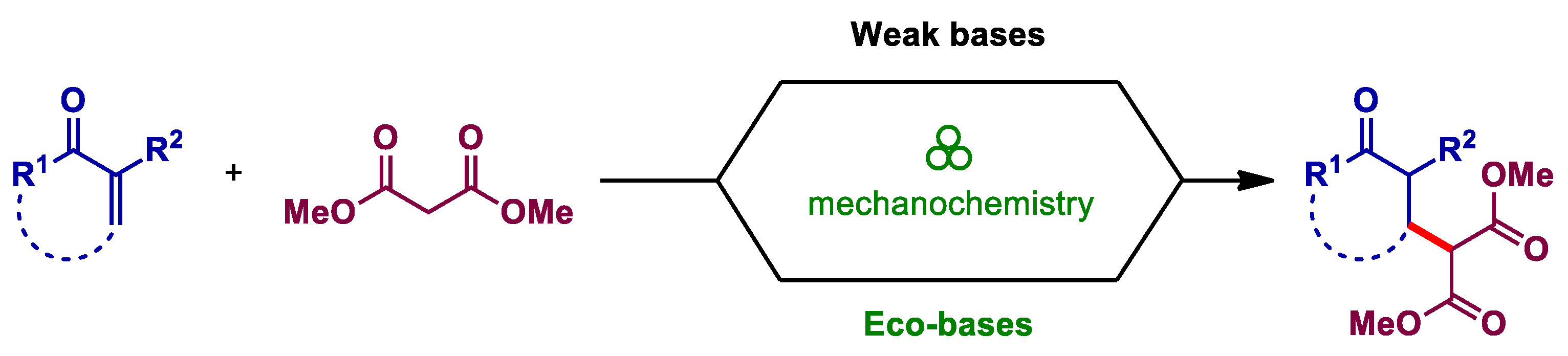
3.1. Mechanochemical Michael Addition Promoted by Weak Bases
3.2. Mechanochemical Michael Addition Promoted by Eco-Bases
3.2.1. Preparation of Eco-bases
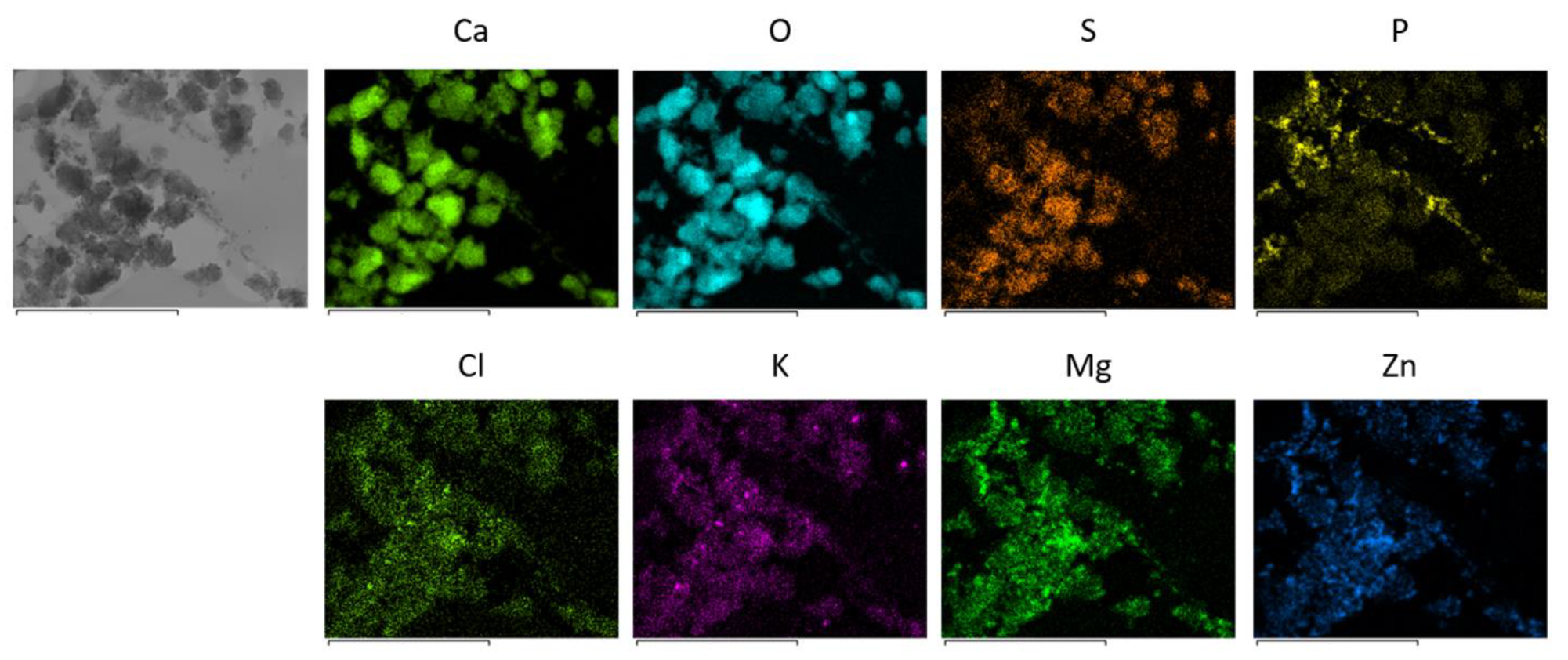
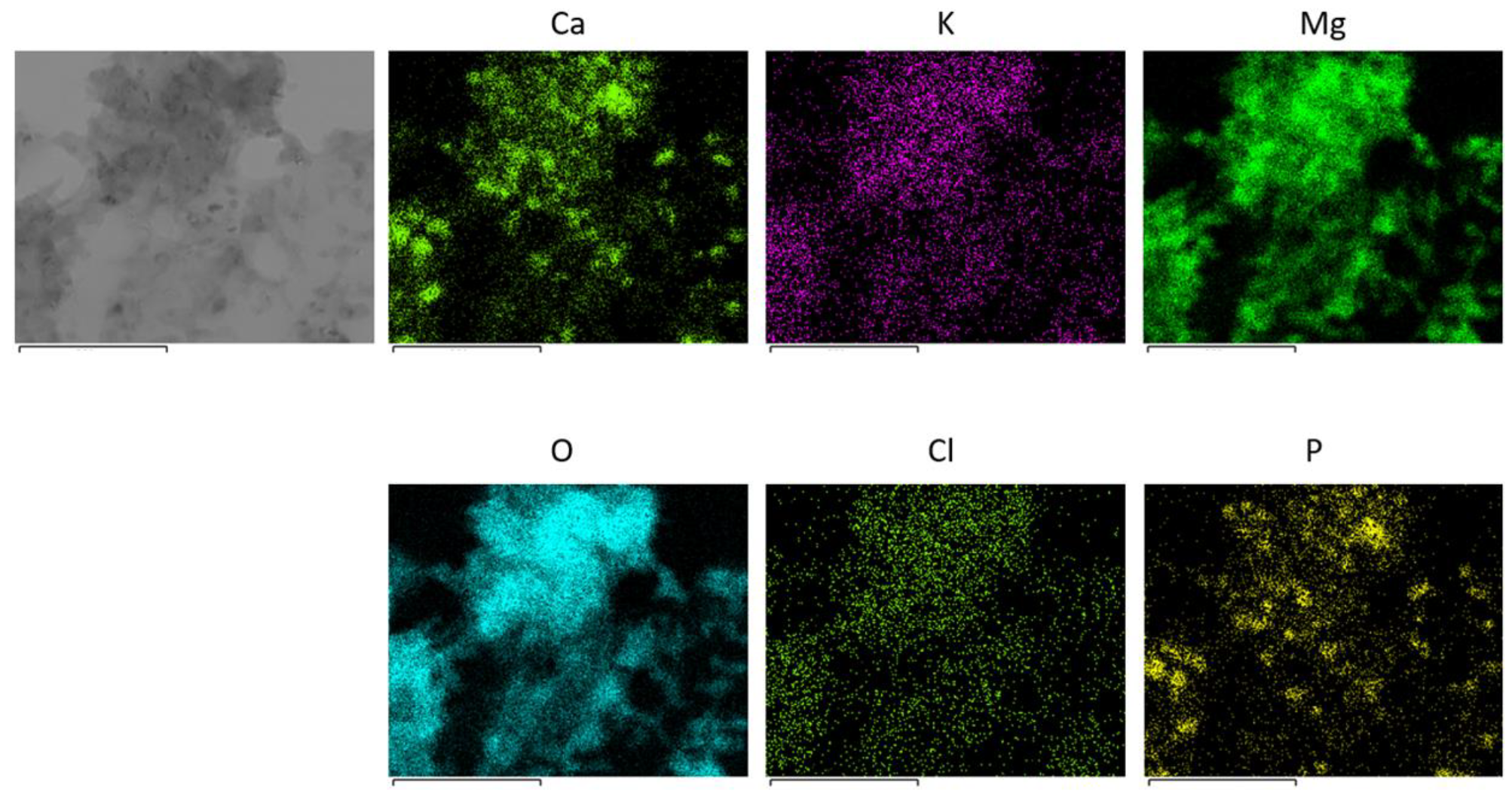
3.2.2. Characterization of Eco-bases
3.2.3. Eco-bases Reactivity in the Mechanochemical Michael Addition
Synthesis of Dimethyl 2-(3-oxocyclopentyl) Malonate 3
Recyclability of Eco-base-Fj
Scope of Michael Addition Promoted by Eco-base and Mechanochemistry
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mather, B.D.; Viswanathan, K.; Miller, K.M.; Long, T.E. Michael Addition Reactions in Macromolecular Design for Emerging Technologies. Prog. Polym. Sci. 2006, 31, 487–531. [Google Scholar] [CrossRef]
- Vasiloiu, M.; Gaertner, P.; Bica, K. Iron Catalyzed Michael Addition: Chloroferrate Ionic Liquids as Efficient Catalysts under Microwave Conditions. Sci. China Chem. 2012, 55, 1614–1619. [Google Scholar] [CrossRef]
- Lancaster, M. Green Chemistry: An Introductory Text, 3rd ed.; Royal Society of Chemistry: Cambridge, UK, 2016. [Google Scholar]
- Gamba-Sánchez, D.; Prunet, J. Synthesis of 1,3-Diols by O-Nucleophile Additions to Activated Alkenes. Synthesis 2018, 50, 3997–4007. [Google Scholar] [CrossRef] [Green Version]
- Smith, M.; March, J.; March, J. March’s Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 5th ed.; Wiley: New York, USA, 2001. [Google Scholar]
- Oare, D.A.; Heathcock, C.H. Acyclic Stereocontrol in Michael Addition Reactions of Enamines and Enol Ethers. In Topics in stereochemistry; Eliel, E.L., Wilen, S.H., Eds.; John Wiley & Sons, Ltd.: NJ, USA, 1991; pp. 87–170. [Google Scholar] [CrossRef]
- Oare, D.A.; Heathcock, C.H. Stereochemistry of the Base-Promoted Michael Addition Reaction. In Topics in Stereochemistry; Eliel, E.L., Wilen, S.H., Eds.; John Wiley & Sons Ltd.: NJ, USA, 1989; pp. 227–407. [Google Scholar] [CrossRef]
- Takatsuna, K.; Tachiwaka, M.; Shiozawa, K.; Okamoto, N.; Okumura, Y. Process for Preparing Aminopropyl Alkoxy Silanes. JP Patent 192263/87, 08 February 1989. [Google Scholar]
- Kumar, B.; Reddy, M.S.; Dwivedi, K.D.; Dahiya, A.; Babu, J.N.; Chowhan, L.R. Synthesis of in Situ Immobilized Iron Oxide Nanoparticles (Fe3O4) on Microcrystalline Cellulose: Ecofriendly and Recyclable Catalyst for Michael Addition. Appl. Organomet. Chem. 2022, 36, 1–10. [Google Scholar] [CrossRef]
- Chapuis, C.; Skuy, D.; Richard, C.-A. Syntheses of Methyl Jasmonate and Analogues. Chimia 2019, 73, 194–204. [Google Scholar] [CrossRef]
- The Chemistry of Fragrances: From Perfumer to Consumer, 2nd ed.; Pybus, D.; Sell, C. (Eds.) Royal Society of Chemistry: Cambridge, UK, 2007. [Google Scholar] [CrossRef] [Green Version]
- Grison, C. Use of Certain Organic Materials, Containing Alkali or Alkaline-Earth Metals, for Implementing Organic Chemical Reactions. Patent US2016228859A1, 24 May 2016. [Google Scholar]
- Büchel, K.H.; Moretto, H.-H.; Woditsch, P. Industrial Inorganic Chemistry, 2nd ed.; Wiley-VCH: NJ, USA, 2008. [Google Scholar]
- Grison, C.; Lock Toy Ki, Y. Ecocatalysis, a New Vision of Green and Sustainable Chemistry. Curr. Opin. Green Sustain. Chem. 2021, 29, 100461. [Google Scholar] [CrossRef]
- Bihanic, C.; Richards, K.; Olszewski, T.K.; Grison, C. Eco-Mn Ecocatalysts: Toolbox for Sustainable and Green Lewis Acid Catalysis and Oxidation Reactions. ChemCatChem 2020, 12, 1529–1545. [Google Scholar] [CrossRef]
- Garel, C.; Fonda, E.; Michalowicz, A.; Diliberto, S.; Boulanger, C.; Petit, E.; Legrand, Y.-M.; Poullain, Y.M. Structure and Composition of the First Biosourced Mn-Rich Catalysts with a Unique Vegetal Footprint. Mat. Tod. Sust. 2019, 5, 100020. [Google Scholar] [CrossRef]
- Deyris, P.-A.; Grison, C. Nature, Ecology and Chemistry: An Unusual Combination for a New Green Catalysis, Ecocatalysis. Curr. Opin. Green Sustain. Chem. 2018, 10, 6–10. [Google Scholar] [CrossRef]
- Deyris, P.-A.; Bert, V.; Diliberto, S.; Boulanger, C.; Petit, E.; Legrand, Y.-M.; Grison, C. Biosourced Polymetallic Catalysis: A Surprising and Efficient Means to Promote the Knoevenagel Condensation. Front. Chem. 2018, 6, 48. [Google Scholar] [CrossRef] [Green Version]
- Escande, V.; Poullain, C.; Clavé, G.; Petit, E.; Masquelez, N.; Hesemann, P.; Grison, C. Bio-Based and Environmental Input for Transfer Hydrogenation Using EcoNi(0) Catalyst in Isopropanol. Appl. Catal. B Environ. 2017, 210, 495–503. [Google Scholar] [CrossRef]
- Grison, C.; Escande, V.; Bitton, J. Ecocatalysis: New Integrated Approach to Scientific Ecology, 1st ed.; ISTE Press Ltd. - Elsevier Inc: London, UK, 2015. [Google Scholar]
- Grison, C. Combining Phytoextraction and Ecocatalysis: A Novel Concept for Greener Chemistry, an Opportunity for Remediation. Environ. Sci. Pollut. Res. 2015, 22, 5589–5591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Escande, V.; Velati, A.; Garel, C.; Renard, B.-L.; Petit, E.; Grison, C. Phytoextracted Mining Wastes for Ecocatalysis: Eco-Mn®, an Efficient and Eco-Friendly Plant-Based Catalyst for Reductive Amination of Ketones. Green Chem. 2015, 17, 2188–2199. [Google Scholar] [CrossRef]
- Escande, V.; Olszewski, T.K.; Petit, E.; Grison, C. Biosourced Polymetallic Catalysts: An Efficient Means To Synthesize Underexploited Platform Molecules from Carbohydrates. ChemSusChem 2014, 7, 1915–1923. [Google Scholar] [CrossRef]
- Escande, V.; Olszewski, T.K.; Grison, C. Preparation of Ecological Catalysts Derived from Zn Hyperaccumulating Plants and Their Catalytic Activity in Diels–Alder Reaction. Comptes Rendus Chim. 2014, 17, 731–737. [Google Scholar] [CrossRef]
- Escande, V.; Garoux, L.; Grison, C.; Thillier, Y.; Debart, F.; Vasseur, J.-J.; Boulanger, C.; Grison, C. Ecological Catalysis and Phytoextraction: Symbiosis for Future. Appl. Catal. B Environ. 2014, 146, 279–288. [Google Scholar] [CrossRef]
- Grison, C.; Escande, V.; Petit, E.; Garoux, L.; Boulanger, C.; Grison, C. Psychotriadouarrei and Geissois Pruinosa, Novel Resources for the Plant-Based Catalytic Chemistry. RSC Adv. 2013, 3, 22340–22345. [Google Scholar] [CrossRef]
- Losfeld, G.; Escande, V.; Vidal de La Blache, P.; L’Huillier, L.; Grison, C. Design and Performance of Supported Lewis Acid Catalysts Derived from Metal Contaminated Biomass for Friedel–Crafts Alkylation and Acylation. Catal. Today 2012, 189, 111–116. [Google Scholar] [CrossRef]
- Garel, C.; Renard, B.-L.; Escande, V.; Galtayries, A.; Hesemann, P.; Grison, C. C-C Bond Formation Strategy through Ecocatalysis: Insights from Structural Studies and Synthetic Potential. Appl. Catal. Gen. 2015, 504, 272–286. [Google Scholar] [CrossRef]
- Clavé, G.; Garel, C.; Poullain, C.; Renard, B.-L.; Olszewski, T.K.; Lange, B.; Shutcha, M.; Faucon, M.-P.; Grison, C. Ullmann Reaction through Ecocatalysis: Insights from Bioresource and Synthetic Potential. RSC Adv. 2016, 6, 59550–59564. [Google Scholar] [CrossRef]
- Clavé, G.; Pelissier, F.; Campidelli, S.; Grison, C. Ecocatalyzed Suzuki Cross Coupling of Heteroaryl Compounds. Green Chem. 2017, 19, 4093–4103. [Google Scholar] [CrossRef]
- Olszewski, T.K.; Adler, P.; Grison, C. Bio-Based Catalysts from Biomass Issued after Decontamination of Effluents Rich in Copper—an Innovative Approach towards Greener Copper-Based Catalysis. Catalysts 2019, 9, 214. [Google Scholar] [CrossRef] [Green Version]
- Grison, C.; Adler, P.; Deyris, P.-A.; Diliberto, S.; Boulanger, C. A Green Approach for the Reduction of Representative Aryl Functional Groups Using Palladium Ecocatalysts. Green Chem. Lett. Rev. 2021, 14, 234–245. [Google Scholar] [CrossRef]
- Cases, L.; Adler, P.; Pelissier, F.; Diliberto, S.; Boulanger, C.; Grison, C. New Biomaterials for Ni Biosorption Turned into Catalysts for Suzuki–Miyaura Cross Coupling of Aryl Iodides in Green Conditions. RSC Adv. 2021, 11, 28085–28091. [Google Scholar] [CrossRef] [PubMed]
- Adler, P.; Dumas, T.; Deyris, P.-A.; Petit, E.; Diliberto, S.; Boulanger, C.; Grison, C. II- from Ecological Recycling of Pd to Greener Sonogashira Cross-Coupling Reactions. J. Clean. Prod. 2021, 293, 126164. [Google Scholar] [CrossRef]
- Bihanic, C.; Diliberto, S.; Pelissier, F.; Petit, E.; Boulanger, C.; Grison, C. Eco-CaMnOx: A Greener Generation of Eco-Catalysts for Eco-Friendly Oxidation Processes. ACS Sustain. Chem. Eng. 2020, 8, 4044–4057. [Google Scholar] [CrossRef]
- Bihanic, C.; Lasbleiz, A.; Regnier, M.; Petit, E.; Le Blainvaux, P.; Grison, C. New Sustainable Synthetic Routes to Cyclic Oxyterpenes Using the Ecocatalyst Toolbox. Molecules 2021, 26, 7194. [Google Scholar] [CrossRef]
- Liodakis, S.; Katsigiannis, G.; Kakali, G. Ash Properties of Some Dominant Greek Forest Species. Thermochim. Acta 2005, 437, 158–167. [Google Scholar] [CrossRef]
- Hwang, G.C.; Hwang, H.; Bang, Y.; Choi, J.; Park, Y.; Jeon, T.-Y.; Chae, B.; Jung, H.; Lee, Y. A Role for Subducted Albite in the Water Cycle and Alkalinity of Subduction Fluids. Nat. Commun. 2021, 12, 1155. [Google Scholar] [CrossRef]
- Hedayati, A.; Lestander, T.A.; Rudolfsson, M.; Thyrel, M.; Öhman, M. Fate of Phosphorus and Potassium in Single-Pellet Thermal Conversion of Forest Residues with a Focus on the Char Composition. Biomass Bioenergy 2021, 150, 106124. [Google Scholar] [CrossRef]
- Hedayati, A.; Sefidari, H.; Boman, C.; Skoglund, N.; Kienzl, N.; Öhman, M. Ash Transformation during Single-Pellet Gasification of Agricultural Biomass with Focus on Potassium and Phosphorus. Fuel Process. Technol. 2021, 217, 106805. [Google Scholar] [CrossRef]
- Garcia, S.D. Les Hydroxyapatites, un système basique atypique modulable par la synthèse: Vers l’identification des sites actifs. Ph.D. Thesis, Université Pierre et Marie Curie, Paris, France, October 2012. [Google Scholar]


 | |||||
|---|---|---|---|---|---|
| Entry | Base | R1 | Conditions | Conv. (%) c | Yield (%) |
| 1 | - | - | Mechano- catalysis a | 2 | 1 |
| 2 | ZnO | H | Mechano- chemistry a | 10 | 1 |
| 3 | MgO | H | Mechano- chemistry a | 4 | 1 |
| 4 | CaCO3 | H | Mechano- chemistry a | 0 | 0 |
| 5 | CaO | H | Mechano- chemistry a | >99 | 95d |
| 6 | Eco-CaOx | H | Mechano- chemistry a | >99 | 84c |
| 7 | K2CO3 | H | Mechano- chemistry a | 94 | 91d |
| 8 | K2CO3 | H | Batch a | 98 | 93c |
| 9 | K2CO3 + CaO | H | Mechano- chemistry a | >99 | 95c |
| 10 | K2CO3 + CaCO3 | H | Mechano- chemistry a | >99 | 93d |
| 11 | K2CO3 | C5H11 | Mechano- chemistry b | 91 | 89c |
| 12 | K2CO3 | C5H11 | Batch b | 11 | 0 |
| 13 | KHCO3 | C5H11 | Mechano- chemistry b | 0 | 0 |
| Composition (Weight % (± %RSD)) | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Entry | Eco-base | Al | Ca | Fe | K | Mg | Mn | Na | Zn |
| 1 | Eco-base-Av | 0.53 (0.49) | 19.65 (1.82) | 1.02 (1.60) | 10.46 (0.67) | 2.17 (1.90) | 0.25 (0.75) | 1.74 (0.31) | 5.02 (1.94) |
| 2 | Eco-base-Nc | 0.60 (4.07) | 7.76 (7.59) | 1.21 (1.19) | 16.99 (4.48) | 2.24 (5.20) | 0.02 (8.30) | 0.01 (115.9) | 9.13 (3.38) |
| 3 | Eco-base-Ah | 0.10 (2.79) | 14.06 (0.37) | 0.16 (10.03) | 15.82 (0.63) | 5.25 (0.18) | 0.07 (0.27) | 0.14 (1.11) | 10.16 (1.06) |
| 4 | Eco-base-Fj | 0.74 (0.99) | 13.57 (1.01) | 0.54 (2.77) | 20.13 (1.82) | 6.21 (0.09) | 0.20 (0.70) | 0.21 (0.60) | 0.12 (2.43) |
| 5 | Eco-base-Sa | 0.50 (1.45) | 16.90 (0.72) | 1.14 (3.76) | 5.95 (2.98) | 1.62 (3.21) | 0.16 (1.35) | 0.19 (2.18) | 0.59 (1.42) |
| Entry | Eco- base | Na | Al | Mg | K | Ca | Zn | Si |
|---|---|---|---|---|---|---|---|---|
| 1 | Eco-base- Ah | NaAl2(Si3Al)O10 (OH)2 | (Mg0.9Fe0.1)O (Mg0.9Zn0.1)O | K2SO4 KCl | CaCO3 | ZnO K2ZnSiO4 ZnSiO3 | SiO2 | |
| 2 | Eco-base- Fj | Na2SO4 | CaMgSi2O6 | K2SO4 KCl K2Ca(CO3)2 | CaCO3 | SiO2 | ||
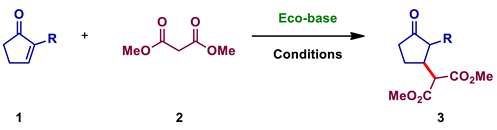 | |||||||
|---|---|---|---|---|---|---|---|
| Entry | Eco-base | R | Conditions | Time (h) | Equiv. of 2 | Conv. (%) c | Yield (%) c |
| 1 | Eco-base- Ah | H | Batch a | 2 | 1 | 0 | 0 |
| 2 | Eco-base- Ah | H | Batch a | 2 | 10 | >99 | 83 |
| 3 | Eco-base- Ah | H | Mechano- chemistry a | 2 | 1 | 99 | 85 |
| 4 | Eco-base- Nc | H | Mechano- chemistry a | 2 | 1 | >99 | 93 |
| 5 | Eco-base- Av | H | Mechano- chemistry a | 2 | 1 | >99 | 81 |
| 6 | Eco-base- Fj | H | Mechano- chemistry a | 2 | 1 | >99 | 97 |
| 7 | Eco-base- Ah | C5H11 | Batch b | 24 | 10 | 0 | 0 |
| 8 | Eco-base- Ah | C5H11 | Mechano- chemistry b | 6 24 | 10 10 | 9 72 | 1 - |
| 9 | Eco-base- Sa | C5H11 | Mechano- chemistry b | 6 16 | 10 10 | 26 67 | - - |
| 10 | Eco-base- Fj | C5H11 | Mechano- chemistry b | 2 6 | 10 10 | 64 84 | 61 - |
 | |||
|---|---|---|---|
| Entry | Number of runs | Conv. (%) | Yield (%) |
| 1 | 1 | >99 | 97 |
| 2 | 2 | >99 | 93 |
| 3 | 3 | >99 | 91 |
| 4 | 4 | >99 | 98 |
 | ||||
|---|---|---|---|---|
| Entry | Michael Acceptor | Product | Conv. (%)a | Isolated Yield (%)a |
| 1 | 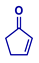 |  | >99 | 95 |
| 2 | 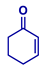 | 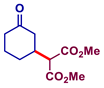 | >99 | 97 |
| 3 |  | 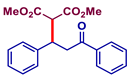 | 76 | 61 |
| 4 |  | 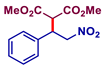 | >99 | 38 |
| 5 |  |  | >99 | 61 |
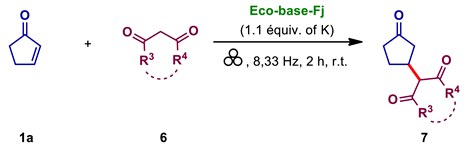 | |||
|---|---|---|---|
| Entry | Michael Donor | Product | Conv. (%) |
| 1 | 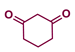 | 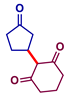 | 75 |
| 2 | 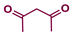 | 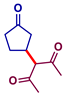 | 24 |
| 3 | 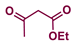 | 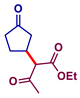 | 95 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lock Toy Ki, Y.; Garcia, A.; Pelissier, F.; Olszewski, T.K.; Babst-Kostecka, A.; Legrand, Y.-M.; Grison, C. Mechanochemistry and Eco-Bases for Sustainable Michael Addition Reactions. Molecules 2022, 27, 3306. https://doi.org/10.3390/molecules27103306
Lock Toy Ki Y, Garcia A, Pelissier F, Olszewski TK, Babst-Kostecka A, Legrand Y-M, Grison C. Mechanochemistry and Eco-Bases for Sustainable Michael Addition Reactions. Molecules. 2022; 27(10):3306. https://doi.org/10.3390/molecules27103306
Chicago/Turabian StyleLock Toy Ki, Yvette, Armelle Garcia, Franck Pelissier, Tomasz K. Olszewski, Alicja Babst-Kostecka, Yves-Marie Legrand, and Claude Grison. 2022. "Mechanochemistry and Eco-Bases for Sustainable Michael Addition Reactions" Molecules 27, no. 10: 3306. https://doi.org/10.3390/molecules27103306
APA StyleLock Toy Ki, Y., Garcia, A., Pelissier, F., Olszewski, T. K., Babst-Kostecka, A., Legrand, Y.-M., & Grison, C. (2022). Mechanochemistry and Eco-Bases for Sustainable Michael Addition Reactions. Molecules, 27(10), 3306. https://doi.org/10.3390/molecules27103306







