Insight into the Progress on Natural Dyes: Sources, Structural Features, Health Effects, Challenges, and Potential
Abstract
:1. Introduction
2. Results
2.1. Literature Search Results
2.2. Resources
2.2.1. Plants
2.2.2. Animals
2.2.3. Microorganisms
2.2.4. Minerals
2.3. Structural Features of Natural Dyes
2.3.1. Carotenoids
2.3.2. Polyphenols
2.3.3. Porphyrins
2.3.4. Alkaloids
2.3.5. Quinones
2.4. Pharmacological Activities of Natural Dyes and Related Mechanisms
2.4.1. Antioxidant activities
2.4.2. Anti-Inflammatory Activities
2.4.3. Anti-Cancer Activities
2.4.4. Anti-Obesity and Anti-Diabetic Activities
2.4.5. Anti-Cardiovascular Disease Effects
2.4.6. Anti-Microbial Activity
2.4.7. Anti-Viral Activities
2.4.8. Neuroprotective Effect
2.4.9. Biological Effects of Dyes Regarding Illumination Conditions
2.5. Challenges and Potential of Natural Dyes
2.5.1. Resources
2.5.2. Biotechnology
2.5.3. Efficient Extraction and Separation Strategy
2.5.4. Improvement of Dye Stability
Co-Pigmentation
Encapsulation
3. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Reyes, F.G.; Valim, M.F.; Vercesi, A.E. Effect of Organic Synthetic Food Colours on Mitochondrial Respiration. Food Addit. Contam. 1996, 13, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Giulieri, F.; Ovarlez, S.; Chaze, A.M. Indigo/Sepiolite Nanohybrids: Stability of Natural Pigments Inspired by Maya Blue. Int. J. Nanotechnol. 2012, 9, 605–617. [Google Scholar] [CrossRef]
- Wrolstad, R.E.; Culver, C.A. Alternatives to Those Artificial FD&C Food Colorants. Annu. Rev. Food Sci. Technol. 2012, 3, 59–77. [Google Scholar] [PubMed]
- Newsome, A.G.; Culver, C.A.; Van Breemen, R.B. Nature’s Palette: The Search for Natural Blue Colorants. J. Agric. Food Chem. 2014, 62, 6498–6511. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2020, 372, n71. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. J. Clin. Epidemiol. 2021, 134, 178–189. [Google Scholar] [CrossRef]
- Tian, L.; Tan, Y.; Chen, G.; Wang, G.; Sun, J.; Ou, S.; Chen, W.; Bai, W. Metabolism of Anthocyanins and Consequent Effects on the Gut Microbiota. Crit. Rev. Food Sci. Nutr. 2019, 59, 982–991. [Google Scholar] [CrossRef]
- Maoka, T. Carotenoids as Natural Functional Pigments. J. Nat. Med. 2020, 74, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Li, S.; Mu, B.; Wang, X.; Wang, A. Recent Researches on Natural Pigments Stabilized by Clay Minerals: A Review. Dye. Pigment. 2021, 190, 109322. [Google Scholar] [CrossRef]
- Bayer, E.; Egeter, H.; Fink, A.; Nether, K.; Wegmann, K. Complex Formation and Flower Colors. Angew. Chemie Int. Ed. Engl. 1966, 5, 791–798. [Google Scholar] [CrossRef]
- Yungyuen, W.; Vo, T.T.; Uthairatanakij, A.; Ma, G.; Zhang, L.C.; Tatmala, N.; Kaewsuksaeng, S.; Jitareerat, P.; Kato, M. Carotenoid Accumulation and the Expression of Carotenoid Metabolic Genes in Mango during Fruit Development and Ripening. Appl. Sci. 2021, 11, 4249. [Google Scholar] [CrossRef]
- Yu, J.H.; Gleize, B.; Zhang, L.F.; Caris-Veyrat, C.; Renard, C. A D-Optimal Mixture Design of Tomato-Based Sauce Formulations: Effects of Onion and EVOO on Lycopene Isomerization and Bioaccessibility. Food Funct. 2019, 10, 3589–3602. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Meng, X.; Zhu, M.; Li, Z. Research Progress of Betalain in Response to Adverse Stresses and Evolutionary Relationship Compared with Anthocyanin. Molecules 2019, 119, 3078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Landi, M.; Tattini, M.; Gould, K.S. Multiple Functional Roles of Anthocyanins in Plant-Environment Interactions. Environ. Exp. Bot. 2015, 119, 4–17. [Google Scholar] [CrossRef]
- Gong, K.; Pan, Y.; Rather, L.J.; Wang, W.; Zhou, Q.; Zhang, T.; Li, Q. Natural Pigment during Flora Leaf Senescence and Its Application in Dyeing and UV Protection Finish of Silk and Wool—A Case Study of Cinnamomum Camphora. Dye. Pigment. 2019, 166, 114–121. [Google Scholar] [CrossRef]
- Borges, M.E.; Tejera, R.L.; Díaz, L.; Esparza, P.; Ibáñez, E. Natural Dyes Extraction from Cochineal (Dactylopius Coccus). New Extraction Methods. Food Chem. 2012, 132, 1855–1860. [Google Scholar] [CrossRef]
- Fernández-López, J.A.; Angosto, J.M.; Giménez, P.J.; León, G. Thermal Stability of Selected Natural Red Extracts Used as Food Colorants. Plant Foods Hum. Nutr. 2013, 68, 11–17. [Google Scholar] [CrossRef]
- Scurria, A.; Tixier, A.S.F.; Lino, C.; Pagliaro, M.; D’Agostino, F.; Avellone, G.; Chemat, F.; Ciriminna, R. High Yields of Shrimp Oil Rich in Omega-3 and Natural Astaxanthin from Shrimp Waste. ACS OMEGA 2020, 5, 17500–17505. [Google Scholar] [CrossRef]
- Tizkar, B.; Seidavi, A.; Ponce-Palafox, J.T.; Pourashoori, P. The Effect of Astaxanthin on Resistance of Juvenile Prawns Macrobrachium Nipponense (Decapoda: Palaemonidae) to Physical and Chemical Stress. Rev. Biol. Trop. 2014, 62, 1331–1341. [Google Scholar] [CrossRef] [Green Version]
- Calvo, N.S.; Reynoso, C.M.; Resnik, S.; Cortés-Jacinto, E.; Collins, P. Thermal Stability of Astaxanthin in Oils for Its Use in Fish Food Technology. Anim. Feed Sci. Technol. 2020, 270, 114668. [Google Scholar] [CrossRef]
- Santos, R.; Hallett, J.; Oliveira, M.C.; Sousa, M.M.; Sarraguça, J.; Simmonds, M.S.J.; Nesbitt, M. HPLC-DAD-MS Analysis of Colorant and Resinous Components of Lac-Dye: A Comparison between Kerria and Paratachardina Genera. Dye. Pigment. 2015, 118, 129–136. [Google Scholar] [CrossRef]
- Berbers, S.V.J.; Tamburini, D.; van Bommel, M.R.; Dyer, J. Historical Formulations of Lake Pigments and Dyes Derived from Lac: A Study of Compositional Variability. Dye. Pigment. 2019, 170, 107579. [Google Scholar] [CrossRef]
- Vemulapalli, S.P.B.; Fuentes-Monteverde, J.C.; Karschin, N.; Oji, T.; Griesinger, C.; Wolkenstein, K. Structure and Absolute Configuration of Phenanthro-Perylene Quinone Pigments from the Deep-Sea Crinoid Hypalocrinus Naresianus. Mar. Drugs 2021, 19, 445. [Google Scholar] [CrossRef] [PubMed]
- Wolkenstein, K.; Fuentes-Monteverde, J.C.; Nath, N.; Oji, T.; Griesinger, C. Hypalocrinins, Taurine-Conjugated Anthraquinone and Biaryl Pigments from the Deep Sea Crinoid Hypalocrinus Naresianus. J. Nat. Prod. 2019, 82, 163–167. [Google Scholar] [CrossRef] [Green Version]
- Aylward, F.O.; Eppley, J.M.; Smith, J.M.; Chavez, F.P.; Scholin, C.A.; DeLong, E.F. Microbial Community Transcriptional Networks Are Conserved in Three Domains at Ocean Basin Scales. Proc. Natl. Acad. Sci. USA 2015, 112, 5443–5448. [Google Scholar] [CrossRef] [Green Version]
- Soliev, A.B.; Hosokawa, K.; Enomoto, K. Bioactive Pigments from Marine Bacteria: Applications and Physiological Roles. Evid.-Based Complement. Altern. Med. 2011, 2011, 670349. [Google Scholar] [CrossRef]
- Numan, M.; Bashir, S.; Mumtaz, R.; Tayyab, S.; Rehman, N.U.; Khan, A.L.; Shinwari, Z.K.; Al-Harrasi, A. Therapeutic Applications of Bacterial Pigments: A Review of Current Status and Future Opportunities. 3 Biotech 2018, 8, 207. [Google Scholar] [CrossRef]
- Venil, C.K.; Zakaria, Z.A.; Ahmad, W.A. Bacterial Pigments and Their Applications. Process Biochem. 2013, 48, 1065–1079. [Google Scholar] [CrossRef]
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine Natural Products. Nat. Prod. Rep. 2013, 30, 237–323. [Google Scholar] [CrossRef]
- Ramesh, C.; Vinithkumar, N.V.; Kirubagaran, R.; Venil, C.K.; Dufossé, L. Multifaceted Applications of Microbial Pigments: Current Knowledge, Challenges and Future Directions for Public Health Implications. Microorganisms 2019, 7, 186. [Google Scholar] [CrossRef] [Green Version]
- Gmoser, R.; Ferreira, J.A.; Lennartsson, P.R.; Taherzadeh, M.J. Filamentous Ascomycetes Fungi as a Source of Natural Pigments. Fungal Biol. Biotechnol. 2017, 4, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Souza, P.N.D.; Grigoletto, T.L.B.; de Moraes, L.A.B.; Abreu, L.M.; Guimaraes, L.H.S.; Santos, C.; Galvao, L.R.; Cardoso, P.G. Production and Chemical Characterization of Pigments in Filamentous Fungi. Microbiology 2016, 162, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Dufosse, L. Microbial Production of Food Grade Pigments. Food Technol. Biotechnol. 2006, 44, 313–321. [Google Scholar]
- Kim, D.; Ku, S. Beneficial Effects of Monascus Sp. KCCM 10093 Pigments and Derivatives: A Mini Review. Molecules 2018, 23, 98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Joshi, V.K.; Attri, D.; Bala, A.; Bhushan, S. Microbial Pigments. Indian J. Biotechnol. 2003, 2, 362–369. [Google Scholar]
- Feng, Y.; Shao, Y.; Chen, F. Monascus Pigments. Appl. Microbiol. Biotechnol. 2012, 96, 1421–1440. [Google Scholar] [CrossRef]
- Manikprabhu, D.; Lingappa, K. Gamma Actinorhodin a Natural and Attorney Source for Synthetic Dye to Detect Acid Production of Fungi. Saudi J. Biol. Sci. 2013, 20, 163–168. [Google Scholar] [CrossRef] [Green Version]
- Neiman, M.K.; Balonis, M.; Kakoulli, I. Cinnabar Alteration in Archaeological Wall Paintings: An Experimental and Theoretical Approach. Appl. Phys. A Mater. Sci. Process. 2015, 121, 915–938. [Google Scholar] [CrossRef] [Green Version]
- Pérez-Arantegui, J.; Pardos, C.; Abad, J.L.; García, J.R. Microcharacterization of a Natural Blue Pigment Used in Wall Paintings during the Romanesque Period in Northern Spain. Microsc. Microanal. 2013, 19, 1645–1652. [Google Scholar] [CrossRef]
- Škvarlová, A.; Kaňuchová, M.; Kozáková, Ľ.; Valušová, E.; Holub, M.; Škvarla, J. Preparation and Characterization of Ultramarine Blue Pigments from Fly Ash by Using the X-Ray Photoelectron Spectroscopy (XPS) for the Determination of Chemical States of Sulphur in Chromophores. Microporous Mesoporous Mater. 2019, 284, 283–288. [Google Scholar] [CrossRef]
- Borhade, A.V.; Kshirsagar, T.A.; Dholi, A.G. Novel Synthesis of Ultramarine Blue from Waste Coal Fly Ash via Thiocyanate Aluminosilicate Sodalite. J. Sulfur Chem. 2016, 37, 632–645. [Google Scholar] [CrossRef]
- Grabowska, M.; Wawrzyniak, D.; Rolle, K.; Chomczyński, P.; Oziewicz, S.; Jurga, S.; Barciszewski, J. Let Food Be Your Medicine: Nutraceutical Properties of Lycopene. Food Funct. 2019, 10, 3090–3102. [Google Scholar] [CrossRef] [PubMed]
- Heider, S.A.E.; Peters-Wendisch, P.; Wendisch, V.F.; Beekwilder, J.; Brautaset, T. Metabolic Engineering for the Microbial Production of Carotenoids and Related Products with a Focus on the Rare C50 Carotenoids. Appl. Microbiol. Biotechnol. 2014, 98, 4355–4368. [Google Scholar] [CrossRef]
- Avalos, J.; Limon, M.C. Biological Roles of Fungal Carotenoids. Curr. Genet. 2015, 61, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Britton, G.; Liaanen-Jensen, S.; Pfander, H. Abstract. In Carotenoids Handbook; Birkhäuser: Basel, Switzerland, 2004; ISBN 978-3-7643-6180-8. [Google Scholar]
- Grune, T.; Lietz, G.; Palou, A.; Ross, A.C.; Stahl, W.; Tang, G.W.; Thurnham, D.; Yin, S.A.; Biesalski, H.K. Beta-Carotene is an Important Vitamin A Source for Humans. J. Nutr. 2010, 140, 2268S–2285S. [Google Scholar] [CrossRef] [Green Version]
- Pashkow, F.J.; Watumull, D.G.; Campbell, C.L. Astaxanthin: A Novel Potential Treatment for Oxidative Stress and Inflammation in Cardiovascular Disease. Am. J. Cardiol. 2008, 101, S58–S68. [Google Scholar] [CrossRef]
- Hojjati, M.; Razavi, S.H.; Rezaei, K.; Gilani, K. Stabilization of Canthaxanthin Produced by Dietzia Natronolimnaea HS-1 with Spray Drying Microencapsulation. J. Food Sci. Technol. 2014, 51, 2134–2140. [Google Scholar] [CrossRef] [Green Version]
- Das, A.; Yoon, S.H.; Lee, S.H.; Kim, J.Y.; Oh, D.K.; Kim, S.W. An Update on Microbial Carotenoid Production: Application of Recent Metabolic Engineering Tools. Appl. Microbiol. Biotechnol. 2007, 77, 505–512. [Google Scholar] [CrossRef]
- Gammone, M.A.; Riccioni, G.; D’Orazio, N. Marine Carotenoids against Oxidative Stress: Effects on Human Health. Mar. Drugs 2015, 13, 6226–6246. [Google Scholar] [CrossRef]
- Maoka, T. New Acetylenic Carotenoid 6′-Epimonadoxanthin from the Rosary Goby Gymnogobius Castaneus. J. Oleo Sci. 2018, 67, 1259–1263. [Google Scholar] [CrossRef] [Green Version]
- Murillo, E.; Mosquera, Y.; Kurtán, T.; Gulyás-Fekete, G.; Nagy, V.; Deli, J. Isolation and Characterization of Novel Capsorubin-like Carotenoids from the Red Mamey (Pouteria sapota). Helv. Chim. Acta 2012, 95, 983–988. [Google Scholar] [CrossRef]
- Shindo, K.; Misawa, N. New and Rare Carotenoids Isolated from Marine Bacteria and Their Antioxidant Activities. Mar. Drugs 2014, 12, 1690–1698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Long, J.T.; Fan, H.X.; Zhou, Z.Q.; Sun, W.Y.; Li, Q.W.; Wang, Y.; Ma, M.; Gao, H.; Zhi, H. The Major Zeaxanthin Dipalmitate Derivatives from Wolfberry. J. Asian Nat. Prod. Res. 2020, 22, 746–753. [Google Scholar] [CrossRef] [PubMed]
- Su, X.; Xu, J.; Rhodes, D.; Shen, Y.; Song, W.; Katz, B.; Tomich, J.; Wang, W. Identification and Quantification of Anthocyanins in Transgenic Purple Tomato. Food Chem. 2016, 202, 184–188. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Sun, S.; Zhou, Z.; Qiu, Z.; Cui, X. Rapid Analysis of Anthocyanin and Its Structural Modi Fi Cations in Fresh Tomato Fruit. Food Chem. 2020, 333, 127439. [Google Scholar] [CrossRef]
- Osorio, C.; Hurtado, N.; Dawid, C.; Hofmann, T.; Heredia-Mira, F.J.; Morales, A.L. Chemical Characterisation of Anthocyanins in Tamarillo (Solanum betaceum Cav.) and Andes Berry (Rubus glaucus Benth.) Fruits. Food Chem. 2012, 132, 1915–1921. [Google Scholar] [CrossRef]
- Wang, Y.M.; Yu, C.H.; Zhao, X.J.; Zhao, J.Q. A Rapid High-Performance Liquid Chromatography Separation of a New Anthocyanin from Nitraria Tangutorum. J. Asian Nat. Prod. Res. 2020, 22, 503–507. [Google Scholar] [CrossRef]
- Engmann, N.F.; Ma, Y.K.; Ying, X.; Qing, Y. Investigating the Effect of High Hydrostatic Pressure Processing on Anthocyanins Composition of Mulberry (Morus moraceae) Juice. Czech J. Food Sci. 2013, 31, 72–80. [Google Scholar] [CrossRef] [Green Version]
- Tatsuzawa, F.; Kato, K.; Sato, M.; Ito, S.; Muraoka, H.; Takahata, Y.; Ogawa, S. Acylated Cyanidin 3-Sophoroside-5-Glucoside in Purple-Violet Flowers of Moricandia Arvensis (Brassicaceae). Nat. Prod. Commun. 2015, 10, 457–459. [Google Scholar] [CrossRef] [Green Version]
- Skaar, I.; Jordheim, M.; Byamukama, R.; Mbabazi, A.; Wubshet, S.G.; Kiremire, B.; Andersen, Ø.M. New Anthocyanidin and Anthocyanin Pigments from Blue Plumbago. J. Agric. Food Chem. 2012, 60, 1510–1515. [Google Scholar] [CrossRef]
- Jin, H.; Liu, Y.; Guo, Z.; Yang, F.; Wang, J.; Li, X.; Peng, X.; Liang, X. High-Performance Liquid Chromatography Separation of Cis—Trans Anthocyanin Isomers from Wild Lycium Ruthenicum Murr. Employing a Mixed-Mode Reversed-Phase/Strong Anion-Exchange Stationary Phase. J. Agric. Food Chem. 2015, 63, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Kondo, T.; Hagihara, S.; Takaya, Y.; Yoshida, K. Polyacylated Anthocyanins in Bluish-Purple Petals of Chinese Bellflower, Platycodon Grandiflorum. Int. J. Mol. Sci. 2021, 22, 4044. [Google Scholar] [CrossRef] [PubMed]
- Moriya, C.; Hosoya, T.; Agawa, S.; Sugiyama, Y.; Kozone, I.; Shin-Ya, K.; Terahara, N.; Kumazawa, S. New Acylated Anthocyanins from Purple Yam and Their Antioxidant Activity. Biosci. Biotechnol. Biochem. 2015, 79, 1484–1492. [Google Scholar] [CrossRef] [PubMed]
- Nabuurs, M.H.; McCallum, J.L.; Brown, D.C.; Kirby, C.W. NMR Characterization of Novel Pyranoanthocyanins Derived from the Pulp of Panax Quinquefolius L. (North American Ginseng). Magn. Reson. Chem. 2017, 55, 177–182. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.W.; Song, C.W.; Zhang, Y.; Khan, A.; Jiang, L.P.; Chen, Y.B.; Liu, Y.P.; Luo, X.D. Alstoscholarisines F and G, Two Unusual Monoterpenoid Indole Alkaloids from the Leaves of Alstonia Scholaris. Tetrahedron Lett. 2015, 56, 6715–6718. [Google Scholar] [CrossRef]
- Nakano, H.; Yoshida, M.; Kaji, R.; Sakai, M.; Doi, S.; Kosemura, S. Oryzamutaic Acid K and Oryzadiamine C, Alkaloids from an Oryza Sativa Mutant with Yellow Endosperm. Tetrahedron Lett. 2020, 61, 152381. [Google Scholar] [CrossRef]
- Nakano, H.; Ono, H.; Kaji, R.; Sakai, M.; Doi, S.; Kosemura, S. Oryzadiamines A and B, Alkaloids from Oryza Sativa with Yellow Grain. Tetrahedron Lett. 2020, 61, 151519. [Google Scholar] [CrossRef]
- Lohmann, J.S.; von Nussbaum, M.; Brandt, W.; Mülbradt, J.; Steglich, W.; Spiteller, P. Rosellin A and B, Two Red Diketopiperazine Alkaloids from the Mushroom Mycena Rosella. Tetrahedron 2018, 74, 5113–5118. [Google Scholar] [CrossRef]
- Lünne, F.; Köhler, J.; Stroh, C.; Müller, L.; Daniliuc, C.G.; Mück-Lichtenfeld, C.; Würthwein, E.U.; Esselen, M.; Humpf, H.U.; Kalinina, S.A. Insights into Ergochromes of the Plant Pathogen Claviceps Purpurea. J. Nat. Prod. 2021, 84, 2630–2643. [Google Scholar] [CrossRef]
- Abdelfattah, M.S.; Toume, K.; Arai, M.A.; Masu, H.; Ishibashi, M. Katorazone, a New Yellow Pigment with a 2-Azaquinone-Phenylhydrazone Structure Produced by Streptomyces Sp. IFM 11299. Tetrahedron Lett. 2012, 53, 3346–3348. [Google Scholar] [CrossRef]
- Pradeep, F.S.; Palaniswamy, M.; Ravi, S.; Thangamani, A.; Pradeep, B.V. Larvicidal Activity of a Novel Isoquinoline Type Pigment from Fusarium Moniliforme KUMBF1201 against Aedes Aegypti and Anopheles Stephensi. Process Biochem. 2015, 50, 1479–1486. [Google Scholar] [CrossRef]
- Lystvan, K.; Kumorkiewicz, A.; Szneler, E.; Wybraniec, S. Study on Betalains in Celosia Cristata Linn. Callus Culture and Identification of New Malonylated Amaranthins. J. Agric. Food Chem. 2018, 66, 3870–3879. [Google Scholar] [CrossRef] [PubMed]
- Basnet, B.B.; Liu, L.; Zhao, W.; Liu, R.; Ma, K.; Bao, L.; Ren, J.; Wei, X.; Yu, H.; Wei, J.; et al. New 1, 2-Naphthoquinone-Derived Pigments from the Mycobiont of Lichen Trypethelium Eluteriae Sprengel. Nat. Prod. Res. 2019, 33, 2044–2050. [Google Scholar] [CrossRef] [PubMed]
- Chagas, F.O.; Dias, L.G.; Pupo, M.T. New Perylenequinone Derivatives from the Endophytic Fungus Alternaria Tenuissima SS77. Tetrahedron Lett. 2016, 57, 3185–3189. [Google Scholar] [CrossRef]
- Ben Ammar, R.; Miyamoto, T.; Chekir-Ghedira, L.; Ghedira, K.; Lacaille-Dubois, M.A. Isolation and Identification of New Anthraquinones from Rhamnus Alaternus L and Evaluation of Their Free Radical Scavenging Activity. Nat. Prod. Res. 2019, 33, 280–286. [Google Scholar] [CrossRef]
- Khokhar, S.; Pierens, G.K.; Hooper, J.N.A.; Ekins, M.G.; Feng, Y.; Davis, R.A. Rhodocomatulin-Type Anthraquinones from the Australian Marine Invertebrates Clathria Hirsuta and Comatula Rotalaria. J. Nat. Prod. 2016, 79, 946–953. [Google Scholar] [CrossRef]
- Kopa, T.K.; Tchinda, A.T.; Tala, M.F.; Zofou, D.; Jumbam, R.; Wabo, H.K.; Titanji, V.P.K.; Frédérich, M.; Tan, N.H.; Tane, P. Antiplasmodial Anthraquinones and Hemisynthetic Derivatives from the Leaves of Tectona Grandis (Verbenaceae). Phytochem. Lett. 2014, 8, 41–45. [Google Scholar] [CrossRef]
- Klaiklay, S.; Rukachaisirikul, V.; Phongpaichit, S.; Pakawatchai, C.; Saithong, S.; Buatong, J.; Preedanon, S.; Sakayaroj, J. Anthraquinone Derivatives from the Mangrove-Derived Fungus Phomopsis Sp. PSU-MA214. Phytochem. Lett. 2012, 5, 738–742. [Google Scholar] [CrossRef]
- Chen, J.; Xu, B.; Sun, J.; Jiang, X.; Bai, W. Anthocyanin Supplement as a Dietary Strategy in Cancer Prevention and Management: A Comprehensive Review. Crit. Rev. Food. Sci. 2021, 1–13. [Google Scholar] [CrossRef]
- Kang, S.; Wang, H.; Guo, M.; Zhang, L.; Chen, M.; Jiang, S.; Li, X.; Jiang, S. Ethylene-Vinyl Alcohol Copolymer-Montmorillonite Multilayer Barrier Film Coated with Mulberry Anthocyanin for Freshness Monitoring. J. Agric. Food Chem. 2018, 66, 13268–13276. [Google Scholar] [CrossRef]
- Zhang, J.; Zou, X.; Zhai, X.; Huang, X.W.; Jiang, C.; Holmes, M. Preparation of an Intelligent PH Film Based on Biodegradable Polymers and Roselle Anthocyanins for Monitoring Pork Freshness. Food Chem. 2019, 272, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Bueno, J.M.; Saez-Plaza, P.; Ramos-Escudero, F.; Jimenez, A.M.; Fett, R.; Asuero, A.G. Analysis and Antioxidant Capacity of Anthocyanin Pigments. Part II: Chemical Structure, Color, and Intake of Anthocyanins. Crit. Rev. Anal. Chem. 2012, 42, 126–151. [Google Scholar] [CrossRef]
- De Freitas, V.; Mateus, N. Chemical Transformations of Anthocyanins Yielding a Variety of Colours (Review). Environ. Chem. Lett. 2006, 4, 175–183. [Google Scholar] [CrossRef]
- Pereira, D.M.; Valentao, P.; Pereira, J.A.; Andrade, P.B. Phenolics: From Chemistry to Biology. Molecules 2009, 14, 2202–2211. [Google Scholar] [CrossRef]
- Cardoso, A.L.; Di Pietro, P.F.; Vieira, F.G.K.; Boaventura, B.C.B.; de Liz, S.; Borges, G.D.C.; Fett, R.; de Andrade, D.F.; da Silva, E.L. Acute Consumption of Jucara Juice (Euterpe Edulis) and Antioxidant Activity in Healthy Individuals. J. Funct. Foods 2015, 17, 152–162. [Google Scholar] [CrossRef]
- Tsuda, T. Curcumin as a Functional Food-Derived Factor: Degradation Products, Metabolites, Bioactivity, and Future Perspectives. Food Funct. 2018, 9, 705–714. [Google Scholar] [CrossRef]
- Aliabbasi, N.; Fathi, M.; Emam-Djomeh, Z. Curcumin: A Promising Bioactive Agent for Application in Food Packaging Systems. J. Environ. Chem. Eng. 2021, 9, 105520. [Google Scholar] [CrossRef]
- Tezcan, A.; Aslan, G.E.; Kaman, H. Evaluation of drought stress on the chlorophyll content of the plants: A review of the solanaceae family. Fresenius Environ. Bull. 2019, 28, 4636–4641. [Google Scholar]
- Manivasagan, P.; Bharathiraja, S.; Moorthy, M.S.; Mondal, S.; Seo, H.; Lee, K.D.; Oh, J. Marine Natural Pigments as Potential Sources for Therapeutic Applications. Crit. Rev. Biotechnol. 2018, 38, 745–761. [Google Scholar] [CrossRef]
- Wang, Y.Q.; Gao, Z.Q. Contrasting Chlorophyll-a Seasonal Patterns between Nearshore and Offshore Waters in the Bohai and Yellow Seas, China: A New Analysis Using Improved Satellite Data. Cont. Shelf Res. 2020, 203, 104173. [Google Scholar] [CrossRef]
- Ito, H.; Ohtsuka, T.; Tanaka, A. Conversion of Chlorophyll b to Chlorophyll a via 7-Hydroxymethyl Chlorophyll. J. Biol. Chem. 1996, 271, 1475–1479. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, M. Chapter Four—Chlorophylls d and f: Synthesis, Occurrence, Light-Harvesting, and Pigment Organization in Chlorophyll-Binding Protein Complexes. In Metabolism, Structure and Function of Plant Tetrapyrroles: Introduction, Microbial and Eukaryotic Chlorophyll Synthesis and Catabolism; Grimm, B., Ed.; Academic Press: Cambridge, MA, USA, 2019; Volume 90, pp. 121–139. ISBN 0065-2296. [Google Scholar]
- Tutunchi, P.; Roufegarinejad, L.; Hamishehkar, H.; Alizadeh, A. Extraction of Red Beet Extract with β-Cyclodextrin-Enhanced Ultrasound Assisted Extraction: A Strategy for Enhancing the Extraction Efficacy of Bioactive Compounds and Their Stability in Food Models. Food Chem. 2019, 297, 124994. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.I. Plant Betalains: Safety, Antioxidant Activity, Clinical Efficacy, and Bioavailability. Compr. Rev. Food Sci. Food Saf. 2016, 15, 316–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roriz, C.L.; Barreira, J.C.M.; Morales, P.; Barros, L.; Ferreira, I.C.F.R. Gomphrena Globosa L. as a Novel Source of Food-Grade Betacyanins: Incorporation in Ice-Cream and Comparison with Beet-Root Extracts and Commercial Betalains. LWT 2018, 92, 101–107. [Google Scholar] [CrossRef] [Green Version]
- Deladino, L.; Alvarez, I.; De Ancos, B.; Sánchez-Moreno, C.; Molina-García, A.D.; Schneider Teixeira, A. Betalains and Phenolic Compounds of Leaves and Stems of Alternanthera Brasiliana and Alternanthera Tenella. Food Res. Int. 2017, 97, 240–249. [Google Scholar] [CrossRef] [Green Version]
- Escribano, J.; Cabanes, J.; Jiménez-Atiénzar, M.; Ibañez-Tremolada, M.; Gómez-Pando, L.R.; García-Carmona, F.; Gandía-Herrero, F. Characterization of Betalains, Saponins and Antioxidant Power in Differently Colored Quinoa (Chenopodium quinoa) Varieties. Food Chem. 2017, 234, 285–294. [Google Scholar] [CrossRef]
- Ferreira, E.S.B.; Hulme, A.N.; McNab, H.; Quye, A. The Natural Constituents of Historical Textile Dyes. Chem. Soc. Rev. 2004, 33, 329–336. [Google Scholar] [CrossRef]
- Clark, R.J.H.; Cooksey, C.J.; Daniels, M.A.M.; Withnall, R. Indigo, Woad, and Tyrian Purple: Important Vat Dyes from Antiquity to the Present. Endeavour 1993, 17, 191–199. [Google Scholar] [CrossRef]
- De Melo, J.S.; Moura, A.P.; Melo, M.J. Photophysical and Spectroscopic Studies of Indigo Derivatives in Their Keto and Leuco Forms. J. Phys. Chem. A 2004, 108, 6975–6981. [Google Scholar] [CrossRef] [Green Version]
- Hong, H.; Conover, C.M.; Hofsommer, D.T.; Sanz, C.A.; Hicks, R.G. Discovery and Properties of a New Indigoid Structure Type Based on Dimeric: Cis -Indigos. Org. Biomol. Chem. 2020, 18, 5838–5842. [Google Scholar] [CrossRef]
- Sharma, V.; McKone, H.T.; Markow, P.G. A Global Perspective on the History, Use, and Identification of Synthetic Food Dyes. J. Chem. Educ. 2011, 88, 24–28. [Google Scholar] [CrossRef]
- Lai, J.L.; Liu, Y.H.; Liu, C.; Qi, M.P.; Liu, R.N.; Zhu, X.F.; Zhou, Q.G.; Chen, Y.Y.; Guo, A.Z.; Hu, C.M. Indirubin Inhibits LPS-Induced Inflammation via TLR4 Abrogation Mediated by the NF-KB and MAPK Signaling Pathways. Inflammation 2017, 40, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Niu, T.C.; Wang, Z.H.; Tan, W.S.; Liu, F.; Kong, Y. Electrochemical Polymerization of Alizarin and the Electrochemical Properties of Poly (Alizarin). Ionics 2018, 24, 1391–1397. [Google Scholar] [CrossRef]
- Gao, J.Q.; Guo, Y.W.; Wang, J.; Jin, X.D.; Wang, Z.Q.; Fan, T.T.; Li, K.; Xu, Y.N. Spectroscopic Analysis of the Interactions of Anthraquinone Derivatives (Alizarin, Alizarin-DA and Alizarin-DA-Fe) with Bovine Serum Albumin (BSA). J. Solut. Chem. 2011, 40, 876–888. [Google Scholar] [CrossRef]
- Liu, J.M.; Gao, H.; Li, F.M.; Liu, Y.L.; Liu, J.Q.; Ou-yang, M.L.; Wang, H.X.; Lin, S.Q.; Lin, C.Q.; Li, Z.M. Determination of trace deoxyribonucleic acid based on a room temperature phosphorescent probe of alizarin red-piperidine self-ordered ring. Anal. Lett. 2010, 43, 300–311. [Google Scholar] [CrossRef]
- Yao, G.Y.; Dai, W.L.; Ye, M.Y.; Huang, R.Z.; Pan, Y.M.; Liao, Z.X.; Wang, H.S. Synthesis and Antitumor Properties of Novel Alizarin Analogs. Med. Chem. Res. 2014, 23, 5031–5042. [Google Scholar] [CrossRef]
- Zou, Z.; He, D.G.; Cai, L.L.; He, X.X.; Wang, K.M.; Yang, X.; Li, L.L.; Li, S.Q.; Su, X.Y. Alizarin Complexone Functionalized Mesoporous Silica Nanoparticles: A Smart System Integrating Glucose-Responsive Double-Drugs Release and Real-Time Monitoring Capabilities. ACS Appl. Mater. Interfaces 2016, 8, 8358–8366. [Google Scholar] [CrossRef]
- Auber, R.P.; Suttiyut, T.; McCoy, R.M.; Ghaste, M.; Crook, J.W.; Pendleton, A.L.; Widhalm, J.R.; Wisecaver, J.H. Hybrid de Novo Genome Assembly of Red Gromwell (Lithospermum Erythrorhizon) Reveals Evolutionary Insight into Shikonin Biosynthesis. Hortic. Res. 2020, 7, 82. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, X.; Song, H.; Zhou, S.; Li, S. Synthesis and Evaluation of Novel Alkannin and Shikonin Oxime Derivatives as Potent Antitumor Agents. Bioorganic Med. Chem. Lett. 2014, 24, 4304–4307. [Google Scholar] [CrossRef]
- Ho, X.L.; Liu, J.J.H.; Loke, W.M. Plant Sterol-Enriched Soy Milk Consumption Modulates 5-Lipoxygenase, 12-Lipoxygenase, and Myeloperoxidase Activities in Healthy Adults—A Randomized-Controlled Trial. Free Radic. Res. 2016, 50, 1396–1407. [Google Scholar] [CrossRef]
- Kim, S.; Sieburth, D. Sphingosine Kinase Regulates Neuropeptide Secretion During the Oxidative Stress-Response Through Intertissue Signaling. J. Neurosci. 2018, 38, 8160–8176. [Google Scholar] [CrossRef] [PubMed]
- Patel, R.S.; Ghasemzadeh, N.; Eapen, D.J.; Sher, S.; Arshad, S.; Ko, Y.A.; Veledar, E.; Samady, H.; Zafari, A.M.; Sperling, L.; et al. Novel Biomarker of Oxidative Stress Is Associated with Risk of Death in Patients with Coronary Artery Disease. Circulation 2016, 133, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Bohn, T.; Bonet, M.L.; Borel, P.; Keijer, J.; Landrier, J.F.; Milisav, I.; Ribot, J.; Riso, P.; Winklhofer-Roob, B.; Sharoni, Y.; et al. Mechanistic Aspects of Carotenoid Health Benefits—Where Are We Now? Nutr. Res. Rev. 2021, 34, 276–302. [Google Scholar] [CrossRef]
- Dose, J.; Matsugo, S.; Yokokawa, H.; Koshida, Y.; Okazaki, S.; Seidel, U.; Eggersdorfer, M.; Rimbach, G.; Esatbeyoglu, T. Free Radical Scavenging and Cellular Antioxidant Properties of Astaxanthin. Int. J. Mol. Sci. 2016, 17, 103. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, D.; Sousa, A.; Nicola, P.; de Oliveira, J.; Rufino, A.T.; Silva, M.; Freitas, M.; Carvalho, F.; Fernandes, E. Beta-Carotene and Its Physiological Metabolites: Effects on Oxidative Status Regulation and Genotoxicity in in Vitro Models. Food Chem. Toxicol. 2020, 141, 111392. [Google Scholar] [CrossRef]
- Neves, D.; Valentao, P.; Bernardo, J.; Oliveira, M.C.; Ferreira, J.M.G.; Pereira, D.M.; Andrade, P.B.; Videira, R.A. A New Insight on Elderberry Anthocyanins Bioactivity: Modulation of Mitochondrial Redox Chain Functionality and Cell Redox State. J. Funct. Foods 2019, 56, 145–155. [Google Scholar] [CrossRef]
- Shen, Y.B.; Zhang, H.; Cheng, L.L.; Wang, L.; Qian, H.F.; Qi, X.G. In Vitro and in Vivo Antioxidant Activity of Polyphenols Extracted from Black Highland Barley. Food Chem. 2016, 194, 1003–1012. [Google Scholar] [CrossRef]
- Ma, T.; Hu, N.; Ding, C.; Zhang, Q.; Li, W.; Suo, Y.; Wang, H.; Bai, B.; Ding, C. In Vitro and in Vivo Biological Activities of Anthocyanins from Nitraria Tangutorun Bobr. Fruits. Food Chem. 2016, 194, 296–303. [Google Scholar] [CrossRef]
- Li, L.; Li, J.; Xu, H.; Zhu, F.; Li, Z.; Lu, H.; Zhang, J.; Yang, Z.; Liu, Y. The Protective Effect of Anthocyanins Extracted from Aronia Melanocarpa Berry in Renal Ischemia-Reperfusion Injury in Mice. Mediators Inflamm. 2021, 2021, 7372893. [Google Scholar] [CrossRef]
- Ereminas, G.; Majiene, D.; Sidlauskas, K.; Jakstas, V.; Ivanauskas, L.; Vaitiekaitis, G.; Liobikas, J. Neuroprotective Properties of Anthocyanidin Glycosides against H2O2-Induced Glial Cell Death Are Modulated by Their Different Stability and Antioxidant Activity in Vitro. Biomed. Pharmacother. 2017, 94, 188–196. [Google Scholar] [CrossRef]
- Chen, S.; Hu, N.; Wang, H.; Wu, Y.; Li, G. Bioactivity-Guided Isolation of the Major Anthocyanin from Lycium Ruthenicum Murr. Fruit and Its Antioxidant Activity and Neuroprotective Effects in Vitro and in Vivo. Food Funct. 2022, 13, 3247–3257. [Google Scholar] [CrossRef] [PubMed]
- Momeni, H.R.; Eskandari, N. Effect of Curcumin on Kidney Histopathological Changes, Lipid Peroxidation and Total Antioxidant Capacity of Serum in Sodium Arsenite-Treated Mice. Exp. Toxicol. Pathol. 2017, 69, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Hassanzadeh, S.; Read, M.I.; Bland, A.R.; Majeed, M.; Jamialahmadi, T.; Sahebkar, A. Curcumin: An Inflammasome Silencer. Pharmacol. Res. 2020, 159, 104921. [Google Scholar] [CrossRef] [PubMed]
- Niu, T.; Xuan, R.; Jiang, L.; Wu, W.; Zhen, Z.; Song, Y.; Hong, L.; Zheng, K.; Zhang, J.; Xu, Q.; et al. Astaxanthin Induces the Nrf2/HO-1 Antioxidant Pathway in Human Umbilical Vein Endothelial Cells by Generating Trace Amounts of ROS. J. Agric. Food Chem. 2018, 66, 1551–1559. [Google Scholar] [CrossRef]
- Xie, S.; Yin, P.; Tian, L.; Yu, Y.; Liu, Y.; Niu, J. Dietary Supplementation of Astaxanthin Improved the Growth Performance, Antioxidant Ability and Immune Response of Juvenile Largemouth Bass (Micropterus salmoides) Fed High-Fat Diet. Mar. Drugs 2020, 18, 642. [Google Scholar] [CrossRef]
- Bandeira, A.C.B.; da Silva, T.P.; de Araujo, G.R.; Araujo, C.M.; da Silva, R.C.; Lima, W.G.; Bezerra, F.S.; Costa, D.C. Lycopene Inhibits Reactive Oxygen Species Production in SK-Hep-1 Cells and Attenuates Acetaminophen-Induced Liver Injury in C57BL/6 Mice. Chem. Biol. Interact. 2017, 263, 7–17. [Google Scholar] [CrossRef]
- Sharavana, G.; Joseph, G.S.; Baskaran, V. Lutein Attenuates Oxidative Stress Markers and Ameliorates Glucose Homeostasis through Polyol Pathway in Heart and Kidney of STZ-Induced Hyperglycemic Rat Model. Eur. J. Nutr. 2017, 56, 2475–2485. [Google Scholar] [CrossRef]
- El-Akabawy, G.; El-Sherif, N.M. Zeaxanthin Exerts Protective Effects on Acetic Acid-Induced Colitis in Rats via Modulation of pro-Inflammatory Cytokines and Oxidative Stress. Biomed. Pharmacother. 2019, 111, 841–851. [Google Scholar] [CrossRef]
- Zhang, Y.; Yin, L.; Huang, L.; Tekliye, M.; Xia, X.; Li, J.; Dong, M. Composition, Antioxidant Activity, and Neuroprotective Effects of Anthocyanin-Rich Extract from Purple Highland Barley Bran and Its Promotion on Autophagy. Food Chem. 2021, 339, 127849. [Google Scholar] [CrossRef]
- Gao, Y.; Liang, X.; Tian, Z.; Ma, Y.; Sun, C. Betalain Exerts Cardioprotective and Anti-Inflammatory Effects against the Experimental Model of Heart Failure. Hum. Exp. Toxicol. 2021, 40, S16–S28. [Google Scholar] [CrossRef]
- Da Silva, D.V.T.; dos Santos Baião, D.; de Oliveira Silva, F.; Alves, G.; Perrone, D.; Del Aguila, E.M.; Flosi Paschoalin, V.M. Betanin, a Natural Food Additive: Stability, Bioavailability, Antioxidant and Preservative Ability Assessments. Molecules 2019, 24, 458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pagliaro, P.; Penna, C. Editorial: Alteration of Redox Equilibrium, Inflammation and Progression of Disease. Curr. Med. Chem. 2018, 25, 1272–1274. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, P.P.; Jena, G.B. Mechanistic Insight into Beta-Carotene-Mediated Protection against Ulcerative Colitis-Associated Local and Systemic Damage in Mice. Eur. J. Nutr. 2015, 54, 639–652. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.X.; Xiong, F. Astaxanthin and Its Effects in Inflammatory Responses and Inflammation-Associated Diseases: Recent Advances and Future Directions. Molecules 2020, 25, 5342. [Google Scholar] [CrossRef]
- Abdin, M.; Hamed, Y.S.; Akhtar, H.M.S.; Chen, D.; Chen, G.; Wan, P.; Zeng, X. Antioxidant and Anti-Inflammatory Activities of Target Anthocyanins Di-Glucosides Isolated from Syzygium Cumini Pulp by High Speed Counter-Current Chromatography. J. Food Biochem. 2020, 44, 1050–1062. [Google Scholar] [CrossRef]
- Lee, S.G.; Brownmiller, C.R.; Lee, S. Anti-Inflammatory and Antioxidant Effects of Anthocyanins of Trifolium Pratense (Red Clover) in Lipopolysaccharide-Stimulated RAW-267.4 Macrophages. Nutrients 2020, 12, 1089. [Google Scholar] [CrossRef] [Green Version]
- Vendrame, S.; Klimis-Zacas, D. Anti-Inflammatory Effect of Anthocyanins via Modulation of Nuclear Factor-ΚB and Mitogen-Activated Protein Kinase Signaling Cascades. Nutr. Rev. 2015, 73, 348–358. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, T.; Wang, X.; Wei, X.; Chen, Y.; Guo, L.; Zhang, J.; Wang, C. Curcumin Modulates Macrophage Polarization through the Inhibition of the Toll-like Receptor 4 Expression and Its Signaling Pathways. Cell. Physiol. Biochem. 2015, 36, 631–641. [Google Scholar] [CrossRef]
- Gyamfi, J.; Kim, J.; Choi, J. Cancer as a Metabolic Disorder. Int. J. Mol. Sci. 2022, 23, 1155. [Google Scholar] [CrossRef]
- Liu, J.; Zhou, H.; Song, L.; Yang, Z.; Qiu, M.; Wang, J.; Shi, S. Anthocyanins: Promising Natural Products with Diverse Pharmacological Activities. Molecules 2021, 26, 3807. [Google Scholar] [CrossRef]
- McCall, B.; McPartland, C.K.; Moore, R.; Frank-Kamenetskii, A.; Booth, B.W. Effects of Astaxanthin on the Proliferation and Migration of Breast Cancer Cells in Vitro. Antioxidants 2018, 7, 135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahn, Y.T.; Kim, M.S.; Kim, Y.S.; An, W.G. Astaxanthin Reduces Stemness Markers in BT20 and T47D Breast Cancer Stem Cells by Inhibiting Expression of Pontin and Mutant P53. Mar. Drugs 2020, 18, 577. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Ahn, Y.T.; Lee, C.W.; Kim, H.; An, W.G. Astaxanthin Modulates Apoptotic Molecules to Induce Death of SKBR3 Breast Cancer Cells. Mar. Drugs 2020, 18, 266. [Google Scholar] [CrossRef] [PubMed]
- Charepalli, V.; Reddivari, L.; Radhakrishnan, S.; Vadde, R.; Agarwal, R.; Vanamala, J.K.P. Anthocyanin-Containing Purple-Fleshed Potatoes Suppress Colon Tumorigenesis via Elimination of Colon Cancer Stem Cells. J. Nutr. Biochem. 2015, 26, 1641–1649. [Google Scholar] [CrossRef]
- Wang, Y.; Lin, J.; Tian, J.; Si, X.; Jiao, X.; Zhang, W.; Gong, E.; Li, B. Blueberry Malvidin-3-Galactoside Suppresses Hepatocellular Carcinoma by Regulating Apoptosis, Proliferation, and Metastasis Pathways in Vivo and in Vitro. J. Agric. Food Chem. 2019, 67, 625–636. [Google Scholar] [CrossRef]
- Xu, Y.; Huang, Y.; Chen, Y.; Cao, K.; Liu, Z.; Wan, Z.; Liao, Z.; Li, B.; Cui, J.; Yang, Y.; et al. Grape Seed Proanthocyanidins Play the Roles of Radioprotection on Normal Lung and Radiosensitization on Lung Cancer via Differential Regulation of the MAPK Signaling Pathway. J. Cancer 2021, 12, 2844–2854. [Google Scholar] [CrossRef]
- Giordano, A.; Tommonaro, G. Curcumin and Cancer. Nutrients 2019, 11, 2376. [Google Scholar] [CrossRef] [Green Version]
- Wu, L.; Guo, L.; Liang, Y.; Liu, X.; Jiang, L.; Wang, L. Curcumin Suppresses Stem-like Traits of Lung Cancer Cells via Inhibiting the JAK2/STAT3 Signaling Pathway. Oncol. Rep. 2015, 34, 3311–3317. [Google Scholar] [CrossRef] [Green Version]
- Zhang, B.Y.; Shi, Y.Q.; Chen, X.; Dai, J.; Jiang, Z.F.; Li, N.; Zhang, Z.B. Protective Effect of Curcumin against Formaldehyde-Induced Genotoxicity in A549 Cell Lines. J. Appl. Toxicol. 2013, 33, 1468–1473. [Google Scholar] [CrossRef]
- Jin, H.; Qiao, F.; Wang, Y.; Xu, Y.; Shang, Y. Curcumin Inhibits Cell Proliferation and Induces Apoptosis of Human Non-Small Cell Lung Cancer Cells through the Upregulation of MiR-192-5p and Suppression of PI3K/Akt Signaling Pathway. Oncol. Rep. 2015, 34, 2782–2789. [Google Scholar] [CrossRef] [Green Version]
- Lu, Y.; Wei, C.; Xi, Z. Curcumin Suppresses Proliferation and Invasion in Non-Small Cell Lung Cancer by Modulation of MTA1-Mediated Wnt/β-Catenin Pathway. Vitr. Cell. Dev. Biol. Anim. 2014, 50, 840–850. [Google Scholar] [CrossRef] [PubMed]
- Takeshima, M.; Ono, M.; Higuchi, T.; Chen, C.; Hara, T.; Nakano, S. Anti-Proliferative and Apoptosis-Inducing Activity of Lycopene against Three Subtypes of Human Breast Cancer Cell Lines. Cancer Sci. 2014, 105, 252–257. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shree, G.S.; Prasad, K.Y.; Arpitha, H.S.; Deepika, U.R.; Kumar, K.N.; Mondal, P.; Ganesan, P. Beta-Carotene at Physiologically Attainable Concentration Induces Apoptosis and down-Regulates Cell Survival and Antioxidant Markers in Human Breast Cancer (MCF-7) Cells. Mol. Cell. Biochem. 2017, 436, 1–12. [Google Scholar] [CrossRef]
- Gloria, N.F.; Soares, N.; Brand, C.; Oliveira, F.L.; Borojevic, R.; Teodoro, A.J. Lycopene and Beta-Carotene Induce Cell-Cycle Arrest and Apoptosis in Human Breast Cancer Cell Lines. Anticancer Res. 2014, 34, 1377–1386. [Google Scholar] [PubMed]
- Chang, J.; Zhang, Y.; Li, Y.; Lu, K.; Shen, Y.; Guo, Y.; Qi, Q.; Wang, M.; Zhang, S. NrF2/ARE and NF-ΚB Pathway Regulation May Be the Mechanism for Lutein Inhibition of Human Breast Cancer Cell. Future Oncol. 2018, 14, 719–726. [Google Scholar] [CrossRef]
- Gong, X.; Smith, J.R.; Swanson, H.M.; Rubin, L.P. Carotenoid Lutein Selectively Inhibits Breast Cancer Cell Growth and Potentiates the Effect of Chemotherapeutic Agents through ROS-Mediated Mechanisms. Molecules 2018, 23, 905. [Google Scholar] [CrossRef] [Green Version]
- Hire, R.R.; Srivastava, S.; Davis, M.B.; Kumar Konreddy, A.; Panda, D. Antiproliferative Activity of Crocin Involves Targeting of Microtubules in Breast Cancer Cells. Sci. Rep. 2017, 7, 44984. [Google Scholar] [CrossRef]
- Arzi, L.; Farahi, A.; Jafarzadeh, N.; Riazi, G.; Sadeghizadeh, M.; Hoshyar, R. Inhibitory Effect of Crocin on Metastasis of Triple-Negative Breast Cancer by Interfering with Wnt/β-Catenin Pathway in Murine Model. DNA Cell Biol. 2018, 37, 1068–1075. [Google Scholar] [CrossRef]
- Tan, J.; Li, Q.; Xue, H.; Tang, J. Ultrasound-Assisted Enzymatic Extraction of Anthocyanins from Grape Skins: Optimization, Identification, and Antitumor Activity. J. Food Sci. 2020, 85, 3731–3744. [Google Scholar] [CrossRef]
- Mazzoni, L.; Giampieri, F.; Alvarez Suarez, J.M.; Gasparrini, M.; Mezzetti, B.; Forbes Hernandez, T.Y.; Battino, M.A. Isolation of Strawberry Anthocyanin-Rich Fractions and Their Mechanisms of Action against Murine Breast Cancer Cell Lines. Food Funct. 2019, 10, 7103–7120. [Google Scholar] [CrossRef]
- Kim, J.M.; Noh, E.M.; Kwon, K.B.; Kim, J.S.; You, Y.O.; Hwang, J.K.; Hwang, B.M.; Kim, B.S.; Lee, S.H.; Lee, S.J.; et al. Curcumin Suppresses the TPA-Induced Invasion through Inhibition of PKCα-Dependent MMP-Expression in MCF-7 Human Breast Cancer Cells. Phytomedicine 2012, 19, 1085–1092. [Google Scholar] [CrossRef] [PubMed]
- Yim-Im, W.; Sawatdichaikul, O.; Semsri, S.; Horata, N.; Mokmak, W.; Tongsima, S.; Suksamrarn, A.; Choowongkomon, K. Computational Analyses of Curcuminoid Analogs against Kinase Domain of HER2. BMC Bioinformatics 2014, 15, 261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guan, F.; Ding, Y.; Zhang, Y.; Zhou, Y.; Li, M.; Wang, C. Curcumin Suppresses Proliferation and Migration of MDA-MB-231 Breast Cancer Cells through Autophagy-Dependent Akt Degradation. PLoS ONE 2016, 11, e0146553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Akkoç, Y.; Berrak, Ö.; Arisan, E.D.; Obakan, P.; Çoker-Gürkan, A.; Palavan-Ünsal, N. Inhibition of PI3K Signaling Triggered Apoptotic Potential of Curcumin Which Is Hindered by Bcl-2 through Activation of Autophagy in MCF-7 Cells. Biomed. Pharmacother. 2015, 71, 161–171. [Google Scholar] [CrossRef]
- Sun, X.D.; Liu, X.E.; Huang, D.S. Curcumin Induces Apoptosis of Triple-Negative Breast Cancer Cells by Inhibition of EGFR Expression. Mol. Med. Rep. 2012, 6, 1267–1270. [Google Scholar] [CrossRef] [Green Version]
- Nowacki, L.; Vigneron, P.; Rotellini, L.; Cazzola, H.; Merlier, F.; Prost, E.; Ralanairina, R.; Gadonna, J.P.; Rossi, C.; Vayssade, M. Betanin-Enriched Red Beetroot (Beta Vulgaris L.) Extract Induces Apoptosis and Autophagic Cell Death in MCF-7 Cells. Phytother. Res. 2015, 29, 1964–1973. [Google Scholar] [CrossRef]
- Kim, H.Y.; Kim, Y.M.; Hong, S. Astaxanthin Suppresses the Metastasis of Colon Cancer by Inhibiting the MYC-Mediated Downregulation of MicroRNA-29a-3p and MicroRNA-200a. Sci. Rep. 2019, 9, 9457. [Google Scholar] [CrossRef]
- Hormozi, M.; Ghoreishi, S.; Baharvand, P. Astaxanthin Induces Apoptosis and Increases Activity of Antioxidant Enzymes in LS-180 Cells. Artif. Cells Nanomed. Biotechnol. 2019, 47, 891–895. [Google Scholar] [CrossRef] [Green Version]
- Neumann, U.; Derwenskus, F.; Flister, V.F.; Schmid-Staiger, U.; Hirth, T.; Bischoff, S.C. Fucoxanthin, a Carotenoid Derived from Phaeodactylum Tricornutum Exerts Antiproliferative and Antioxidant Activities in Vitro. Antioxidants 2019, 8, 183. [Google Scholar] [CrossRef] [Green Version]
- Lopes-Costa, E.; Abreu, M.; Gargiulo, D.; Rocha, E.; Ramos, A.A. Anticancer Effects of Seaweed Compounds Fucoxanthin and Phloroglucinol, Alone and in Combination with 5-Fluorouracil in Colon Cells. J. Toxicol. Environ. Health A Curr. Issues 2017, 80, 776–787. [Google Scholar] [CrossRef]
- Amin, A.; Farrukh, A.; Murali, C.; Soleimani, A.; Praz, F.; Graziani, G.; Brim, H.; Ashktorab, H. Saffron and Its Major Ingredients’ Effect on Colon Cancer Cells with Mismatch Repair Deficiency and Microsatellite Instability. Molecules 2021, 26, 3855. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Jiang, B.; Zhong, C.; Guo, J.; Zhang, L.; Mu, T.; Zhang, Q.; Bi, X. Chemoprevention of Colorectal Cancer by Black Raspberry Anthocyanins Involved the Modulation of Gut Microbiota and SFRP2 Demethylation. Carcinogenesis 2018, 39, 471–481. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Guo, J.; Mao, L.; Li, Q.; Guo, M.; Mu, T.; Zhang, Q.; Bi, X. Up-Regulation of MIR-24-1-5p Is Involved in the Chemoprevention of Colorectal Cancer by Black Raspberry Anthocyanins. Br. J. Nutr. 2019, 122, 518–526. [Google Scholar] [CrossRef] [PubMed]
- López De Las Hazas, M.C.; Mosele, J.I.; Macià, A.; Ludwig, I.A.; Motilva, M.J. Exploring the Colonic Metabolism of Grape and Strawberry Anthocyanins and Their in Vitro Apoptotic Effects in HT-29 Colon Cancer Cells. J. Agric. Food Chem. 2017, 65, 6477–6487. [Google Scholar] [CrossRef] [PubMed]
- Rajitha, B.; Belalcazar, A.; Nagaraju, G.P.; Shaib, W.L.; Snyder, J.P.; Shoji, M.; Pattnaik, S.; Alam, A.; El-Rayes, B.F. Inhibition of NF-ΚB Translocation by Curcumin Analogs Induces G0/G1 Arrest and Downregulates Thymidylate Synthase in Colorectal Cancer. Cancer Lett. 2016, 373, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.D.; Chen, X.J.; Hu, Y.H.; Yu, Z.J.; Wang, D.; Liu, J.Z. Curcumin Inhibits Proliferation and Induces Apoptosis of Human Colorectal Cancer Cells by Activating the Mitochondria Apoptotic Pathway. Phytother. Res. 2013, 27, 422–430. [Google Scholar] [CrossRef]
- Farabegoli, F.; Scarpa, E.S.; Frati, A.; Serafini, G.; Papi, A.; Spisni, E.; Antonini, E.; Benedetti, S.; Ninfali, P. Betalains Increase Vitexin-2-O-Xyloside Cytotoxicity in CaCo-2 Cancer Cells. Food Chem. 2017, 218, 356–364. [Google Scholar] [CrossRef]
- Zhou, Y.; Xu, Q.; Shang, J.; Lu, L.; Chen, G. Crocin Inhibits the Migration, Invasion, and Epithelial-Mesenchymal Transition of Gastric Cancer Cells via MiR-320/KLF5/HIF-1α Signaling. J. Cell. Physiol. 2019, 234, 17876–17885. [Google Scholar] [CrossRef]
- Park, Y.; Choi, J.; Lim, J.W.; Kim, H. Beta-Carotene-Induced Apoptosis Is Mediated with Loss of Ku Proteins in Gastric Cancer AGS Cells. Genes Nutr. 2015, 10, 467. [Google Scholar] [CrossRef]
- Zhu, Y.; Cheng, J.; Min, Z.; Yin, T.; Zhang, R.; Zhang, W.; Hu, L.; Cui, Z.; Gao, C.; Xu, S.; et al. Effects of Fucoxanthin on Autophagy and Apoptosis in SGC-7901cells and the Mechanism. J. Cell. Biochem. 2018, 119, 7274–7284. [Google Scholar] [CrossRef]
- Yu, R.-X.; Yu, R.-T.; Liu, Z. Inhibition of Two Gastric Cancer Cell Lines Induced by Fucoxanthin Involves Downregulation of Mcl-1 and STAT3. Hum. Cell 2018, 31, 50–63. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Park, J.J.; Lee, B.J.; Joo, M.K.; Chun, H.J.; Lee, S.W.; Bak, Y.T. Astaxanthin Inhibits Proliferation of Human Gastric Cancer Cell Lines by Interrupting Cell Cycle Progression. Gut Liver 2016, 10, 369–374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.; Lee, H.; Lim, J.W.; Kim, H. Astaxanthin Induces NADPH Oxidase Activation and Receptor-interacting Protein Kinase 1-mediated Necroptosis in Gastric Cancer AGS Cells. Mol. Med. Rep. 2021, 24, 837. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Ni, Y.; Yang, J.; Lin, X.; Li, J.; Zhang, L. Astaxanthin Inhibits Proliferation and Induces Apoptosis and Cell Cycle Arrest of Mice H22 Hepatoma Cells. Med. Sci. Monit. 2016, 22, 2152–2160. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Dai, W.; Xia, Y.; Chen, K.; Li, S.; Liu, T.; Zhang, R.; Wang, J.; Lu, W.; Zhou, Y.; et al. Astaxanthin Inhibits Proliferation and Induces Apoptosis of Human Hepatocellular Carcinoma Cells via Inhibition of Nf-Κb P65 and Wnt/B-Catenin in Vitro. Mar. Drugs 2015, 13, 6064–6081. [Google Scholar] [CrossRef]
- Ohno, T.; Shimizu, M.; Shirakami, Y.; Miyazaki, T.; Ideta, T.; Kochi, T.; Kubota, M.; Sakai, H.; Tanaka, T.; Moriwaki, H. Preventive Effects of Astaxanthin on Diethylnitrosamine-Induced Liver Tumorigenesis in C57/BL/KsJ-Db/Db Obese Mice. Hepatol. Res. 2016, 46, E201–E209. [Google Scholar] [CrossRef]
- Amin, A.; Hamza, A.A.; Daoud, S.; Khazanehdari, K.; Al Hrout, A.; Baig, B.; Chaiboonchoe, A.; Adrian, T.E.; Zaki, N.; Salehi-Ashtiani, K. Saffron-Based Crocin Prevents Early Lesions of Liver Cancer: In Vivo, In Vitro and Network Analyses. Recent Pat. Anticancer. Drug Discov. 2015, 11, 121–133. [Google Scholar] [CrossRef]
- Sangavi, P.; Langeswaran, K.; Kumar, S.G. Anticarcinogenic Efficacy of Fucoxanthin on HepG2 Cell Lines. J. Clin. Diagn. Res. 2022, 16, 5–9. [Google Scholar] [CrossRef]
- Jin, X.; Zhao, T.T.; Shi, D.; Ye, M.B.; Yi, Q. Protective Role of Fucoxanthin in Diethylnitrosamine-Induced Hepatocarcinogenesis in Experimental Adult Rats. Drug Dev. Res. 2019, 80, 209–217. [Google Scholar] [CrossRef]
- Dokkaew, A.; Punvittayagul, C.; Insuan, O.; Limtrakul, P.; Wongpoomchai, R. Protective Effects of Defatted Sticky Rice Bran Extracts on the Early Stages of Hepatocarcinogenesis in Rats. Molecules 2019, 24, 2142. [Google Scholar] [CrossRef] [Green Version]
- Liao, S.; Liu, J.; Xu, M.; Zheng, J. Evaluation of the Liver Cancer Prevention of Anthocyanin Extracts from Mulberry (Morus alba L.) Variety PR-01. Adv. Biosci. Biotechnol. 2018, 9, 423–442. [Google Scholar] [CrossRef] [Green Version]
- Tsai, C.F.; Hsieh, T.H.; Lee, J.N.; Hsu, C.Y.; Wang, Y.C.; Kuo, K.K.; Wu, H.L.; Chiu, C.C.; Tsai, E.M.; Kuo, P.L. Curcumin Suppresses Phthalate-Induced Metastasis and the Proportion of Cancer Stem Cell (CSC)-like Cells via the Inhibition of AhR/ERK/SK1 Signaling in Hepatocellular Carcinoma. J. Agric. Food Chem. 2015, 63, 10388–10398. [Google Scholar] [CrossRef] [PubMed]
- Krajka-Kuźniak, V.; Paluszczak, J.; Szaefer, H.; Baer-Dubowska, W. Betanin, a Beetroot Component, Induces Nuclear Factor Erythroid-2-Related Factor 2-Mediated Expression of Detoxifying/Antioxidant Enzymes in Human Liver Cell Lines. Br. J. Nutr. 2013, 110, 2138–2149. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ko, J.C.; Chen, J.C.; Wang, T.J.; Zheng, H.Y.; Chen, W.C.; Chang, P.Y.; Lin, Y.W. Astaxanthin Down-Regulates Rad51 Expression via Inactivation of AKT Kinase to Enhance Mitomycin C-Induced Cytotoxicity in Human Non-Small Cell Lung Cancer Cells. Biochem. Pharmacol. 2016, 105, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Zhao, S.; Wang, X.; Zhang, L.; Jiang, E.; Gu, Y.; Shangguan, A.J.; Zhao, H.; Lv, T.; Yu, Z. Crocin Inhibits Cell Proliferation and Enhances Cisplatin and Pemetrexed Chemosensitivity in Lung Cancer Cells. Transl. Lung Cancer Res. 2015, 4, 775–783. [Google Scholar] [CrossRef]
- Zhang, W.L.; Zhao, Y.N.; Shi, Z.Z.; Cong, D.; Bai, Y.S. Lutein Inhibits Cell Growth and Activates Apoptosis via the PI3K/AKT/MTOR Signaling Pathway in A549 Human Non-Small-Cell Lung Cancer Cells. J. Environ. Pathol. Toxicol. Oncol. 2018, 37, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Amararathna, M.; Hoskin, D.W.; Rupasinghe, H.P.V. Cyanidin-3-O-Glucoside-Rich Haskap Berry Administration Suppresses Carcinogen-Induced Lung Tumorigenesis in A/JCr Mice. Molecules 2020, 25, 3823. [Google Scholar] [CrossRef]
- Yin, Z.; Yang, Y.; Guo, T.; Veeraraghavan, V.P.; Wang, X. Potential Chemotherapeutic Effect of Betalain against Human Non-Small Cell Lung Cancer through PI3K/Akt/MTOR Signaling Pathway. Environ. Toxicol. 2021, 36, 1011–1020. [Google Scholar] [CrossRef]
- Ni, X.; Yu, H.; Wang, S.; Zhang, C.; Shen, S. Astaxanthin Inhibits PC-3 Xenograft Prostate Tumor Growth in Nude Mice. Mar. Drugs 2017, 15, 66. [Google Scholar] [CrossRef] [Green Version]
- D’Alessandro, A.M.; Mancini, A.; Lizzi, A.R.; De Simone, A.; Marroccella, C.E.; Gravina, G.L.; Tatone, C.; Festuccia, C. Crocus Sativus Stigma Extract and Its Major Constituent Crocin Possess Significant Antiproliferative Properties against Human Prostate Cancer. Nutr. Cancer 2013, 65, 930–942. [Google Scholar] [CrossRef]
- Tyagi, A.; Kumar, S.; Raina, K.; Wempe, M.F.; Maroni, P.D.; Agarwal, R.; Agarwal, C. Differential Effect of Grape Seed Extract and Its Active Constituent Procyanidin B2 3,3″-Di-O-Gallate against Prostate Cancer Stem Cells. Mol. Carcinog. 2019, 58, 1105–1117. [Google Scholar] [CrossRef] [PubMed]
- Jongsomchai, K.; Leardkamolkarn, V.; Mahatheeranont, S. A Rice Bran Phytochemical, Cyanidin 3-Glucoside, Inhibits the Progression of PC3 Prostate Cancer Cell. Anat. Cell Biol. 2020, 53, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Ide, H.; Lu, Y.; Noguchi, T.; Muto, S.; Okada, H.; Kawato, S.; Horie, S. Modulation of AKR1C2 by Curcumin Decreases Testosterone Production in Prostate Cancer. Cancer Sci. 2018, 109, 1230–1238. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, M.; Zheng, Z.; Huang, J.; Ma, X.; Huang, C.; Wu, R.; Li, X.; Liang, Z.; Deng, F.; Wu, J.; et al. Modulation of MiR-34a in Curcumin-Induced Antiproliferation of Prostate Cancer Cells. J. Cell. Biochem. 2019, 120, 15616–15624. [Google Scholar] [CrossRef] [PubMed]
- Gopal, S.S.; Maradgi, T.; Ponesakki, G. 3-Antiobese Properties of Carotenoids: An Overview of Underlying Molecular Mechanisms. In Carotenoids: Properties, Processing and Applications; Charis, M.G., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 75–105. ISBN 978-0-12-817067-0. [Google Scholar]
- Wang, J.; Liu, S.; Wang, H.; Xiao, S.; Li, C.; Li, Y.; Liu, B. Xanthophyllomyces Dendrorhous-Derived Astaxanthin Regulates Lipid Metabolism and Gut Microbiota in Obese Mice Induced by a High-Fat Diet. Mar. Drugs 2019, 17, 337. [Google Scholar] [CrossRef] [Green Version]
- Wu, T.; Gao, Y.F.; Hao, J.Y.; Geng, J.T.; Zhang, J.J.; Yin, J.J.; Liu, R.; Sui, W.J.; Gong, L.X.; Zhang, M. Capsanthin Extract Prevents Obesity, Reduces Serum TMAO Levels and Modulates the Gut Microbiota Composition in High-Fat-Diet Induced Obese C57BL/6J Mice. Food Res. Int. 2020, 128, 108774. [Google Scholar] [CrossRef]
- Fen, Z.G.; Ni, Y.H.; Wan, C.Y.; Liu, F.; Fu, Z.W. Anti-Diabetic Effects of Astaxanthin on an STZ-Induced Diabetic Model in Rats. Endocr. J. 2021, 68, 451–459. [Google Scholar]
- Xie, L.; Su, H.; Sun, C.; Zheng, X.; Chen, W. Recent Advances in Understanding the Anti-Obesity Activity of Anthocyanins and Their Biosynthesis in Microorganisms. Trends Food Sci. Technol. 2018, 72, 13–24. [Google Scholar] [CrossRef]
- Lee, B.; Lee, M.; Lefevre, M.; Kim, H.R. Anthocyanins Inhibit Lipogenesis During Adipocyte Differentiation of 3T3-L1 Preadipocytes. Plant Foods Hum. Nutr. 2014, 69, 137–141. [Google Scholar] [CrossRef]
- Skates, E.; Overall, J.; DeZego, K.; Wilson, M.; Esposito, D.; Lila, M.A.; Komarnytsky, S. Berries Containing Anthocyanins with Enhanced Methylation Profiles Are More Effective at Ameliorating High Fat Diet-Induced Metabolic Damage. Food Chem. Toxicol. 2018, 111, 445–453. [Google Scholar] [CrossRef]
- Li, Q.; Yang, G.; Xu, H.; Tang, S.; Lee, W.Y. Effects of Resveratrol Supplementation on Bone Quality: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. BMC Complement. Med. Ther. 2021, 21, 214. [Google Scholar] [CrossRef] [PubMed]
- Jang, H.H.; Kim, H.W.; Kim, S.Y.; Kim, S.M.; Kim, J.B.; Lee, Y.M. In Vitro and in Vivo Hypoglycemic Effects of Cyanidin 3-Caffeoyl-p-Hydroxybenzoylsophoroside-5-Glucoside, an Anthocyanin Isolated from Purple-Fleshed Sweet Potato. Food Chem. 2019, 272, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Matsukawa, T.; Inaguma, T.; Han, J.; Villareal, M.O.; Isoda, H. Cyanidin-3-Glucoside Derived from Black Soybeans Ameliorate Type 2 Diabetes through the Induction of Differentiation of Preadipocytes into Smaller and Insulin-Sensitive Adipocytes. J. Nutr. Biochem. 2015, 26, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Kasprzak-Drozd, K.; Oniszczuk, T.; Gancarz, M.; Kondracka, A.; Rusinek, R.; Oniszczuk, A. Curcumin and Weight Loss: Does It Work? Int. J. Mol. Sci. 2022, 23, 639. [Google Scholar] [CrossRef]
- Song, H.Z.; Chu, Q.; Xu, D.D.; Xu, Y.; Zheng, X.D. Purified Betacyanins from Hylocereus Undatus Peel Ameliorate Obesity and Insulin Resistance in High-Fat-Diet-Fed Mice. J. Agric. Food Chem. 2016, 64, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Ridker, P.M.; Hansson, G.K. Progress and Challenges in Translating the Biology of Atherosclerosis. Nature 2011, 473, 317–325. [Google Scholar] [CrossRef]
- Yao, Y.; Goh, H.M.; Kim, J.E. The Roles of Carotenoid Consumption and Bioavailability in Cardiovascular Health. Antioxidants 2021, 10, 1978. [Google Scholar] [CrossRef]
- Ouyang, B.; Li, Z.L.; Ji, X.Y.; Huang, J.W.; Zhang, H.S.; Jiang, C.R. The Protective Role of Lutein on Isoproterenol-Induced Cardiac Failure Rat Model through Improving Cardiac Morphology, Antioxidant Status via Positively Regulating Nrf2/HO-1 Signalling Pathway. Pharm. Biol. 2019, 57, 529–535. [Google Scholar] [CrossRef] [Green Version]
- Cassidy, A.; Mukamal, K.J.; Liu, L.; Franz, M.; Eliassen, A.H.; Rimm, E.B. High Anthocyanin Intake is Associated with a Reduced Risk of Myocardial Infarction in Young and Middle-Aged Women. Circulation 2013, 127, 188–196. [Google Scholar] [CrossRef] [Green Version]
- Cassidy, A.; Bertoia, M.; Chiuve, S.; Flint, A.; Forman, J.; Rimm, E.B. Habitual Intake of Anthocyanins and Flavanones and Risk of Cardiovascular Disease in Men. Am. J. Clin. Nutr. 2016, 104, 587–594. [Google Scholar] [CrossRef] [Green Version]
- Krga, I.; Milenkovic, D. Anthocyanins: From Sources and Bioavailability to Cardiovascular-Health Benefits and Molecular Mechanisms of Action. J. Agric. Food Chem. 2019, 67, 1771–1783. [Google Scholar] [CrossRef] [PubMed]
- Pourbagher-Shahri, A.M.; Farkhondeh, T.; Ashrafizadeh, M.; Talebi, M.; Samargahndian, S. Curcumin and Cardiovascular Diseases: Focus on Cellular Targets and Cascades. Biomed. Pharmacother. 2021, 136, 111214. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.J.; Zhang, K.; Ma, J.J.; Wang, L.; Zhuang, Y. Mechanism of Curcumin against Myocardial Ischaemia-Reperfusion Injury Based on the P13K/Akt/MTOR Signalling Pathway. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 5490–5499. [Google Scholar] [CrossRef] [PubMed]
- Demirdöven, A.; Karabiyikli, Ş.; Tokatli, K.; Öncül, N. Inhibitory Effects of Red Cabbage and Sour Cherry Pomace Anthocyanin Extracts on Food Borne Pathogens and Their Antioxidant Properties. LWT 2015, 63, 8–13. [Google Scholar] [CrossRef]
- Moghadamtousi, S.Z.; Kadir, H.A.; Hassandarvish, P.; Tajik, H.; Abubakar, S.; Zandi, K. A Review on Antibacterial, Antiviral, and Antifungal Activity of Curcumin. Biomed Res. Int. 2014, 2014, 186864. [Google Scholar] [CrossRef]
- Batista de Andrade Neto, J.; Pessoa de Farias Cabral, V.; Brito Nogueira, L.F.; Rocha da Silva, C.; Gurgel do Amaral Valente Sá, L.; Ramos da Silva, A.; Barbosa da Silva, W.M.; Silva, J.; Marinho, E.S.; Cavalcanti, B.C.; et al. Anti-MRSA Activity of Curcumin in Planktonic Cells and Biofilms and Determination of Possible Action Mechanisms. Microb. Pathog. 2021, 155, 104892. [Google Scholar] [CrossRef]
- Larussa, T.; Gervasi, S.; Liparoti, R.; Suraci, E.; Marasco, R.; Imeneo, M.; Luzza, F. Downregulation of Interleukin- (IL-) 17 through Enhanced Indoleamine 2,3-Dioxygenase (IDO) Induction by Curcumin: A Potential Mechanism of Tolerance towards Helicobacter Pylori. J. Immunol. Res. 2018, 2018, 3739593. [Google Scholar] [CrossRef] [Green Version]
- Da Silva Souza Campanholi, K.; Jaski, J.M.; da Silva Junior, R.C.; Zanqui, A.B.; Lazarin-Bidóia, D.; da Silva, C.M.; da Silva, E.A.; Hioka, N.; Nakamura, C.V.; Cardozo-Filho, L.; et al. Photodamage on Staphylococcus Aureus by Natural Extract from Tetragonia Tetragonoides (Pall.) Kuntze: Clean Method of Extraction, Characterization and Photophysical Studies. J. Photochem. Photobiol. B Biol. 2020, 203, 111763. [Google Scholar] [CrossRef]
- Canadanovic-Brunet, J.M.; Savatovic, S.S.; Cetkovic, G.S.; Vulic, J.J.; Djilas, S.M.; Markov, S.L.; Cvetkovic, D.D. Antioxidant and Antimicrobial Activities of Beet Root Pomace Extracts. Czech J. Food Sci. 2011, 29, 575–585. [Google Scholar] [CrossRef] [Green Version]
- Tenore, G.C.; Novellino, E.; Basile, A. Nutraceutical Potential and Antioxidant Benefits of Red Pitaya (Hylocereus polyrhizus) Extracts. J. Funct. Foods 2012, 4, 129–136. [Google Scholar] [CrossRef]
- Yolmeh, M.; Hamedi, H.; Khomeiri, M. Antimicrobial Activity of Pigments Extracted from Rhodotorula Glutinis Against Some Bacteria and Fungi. Zahedan J. Res. Med. Sci. 2016, in press. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.Y.; Liao, Y.Y.; Chen, R.X.; Hou, Y.P.; Ke, W.Q.; Zhang, B.B.; Gao, M.L.; Shao, Z.Z.; Chen, J.M.; Li, F. Chlorinated Azaphilone Pigments with Antimicrobial and Cytotoxic Activities Isolated from the Deep Sea Derived Fungus Chaetomium Sp NA-S01-R1. Mar. Drugs 2018, 16, 61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manickam, P.; Thilagavathi, G. Development of Antibacterial Silk Sutures Using Natural Fungal Extract for Healthcare Applications. J. Text. Sci. Eng. 2016, 6, 249. [Google Scholar] [CrossRef]
- Hegazy, G.E.; Abu-Serie, M.M.; Abo-Elela, G.M.; Ghozlan, H.; Sabry, S.A.; Soliman, N.A.; Abdel-Fattah, Y.R. In Vitro Dual (Anticancer and Antiviral) Activity of the Carotenoids Produced by Haloalkaliphilic Archaeon Natrialba Sp. M6. Sci. Rep. 2020, 10, 5986. [Google Scholar] [CrossRef] [PubMed]
- Yim, S.K.; Kim, I.; Warren, B.; Kim, J.; Jung, K.; Ku, B. Antiviral Activity of Two Marine Carotenoids against Sars-Cov-2 Virus Entry in Silico and in Vitro. Int. J. Mol. Sci. 2021, 22, 6481. [Google Scholar] [CrossRef] [PubMed]
- Nikolaeva-Glomb, L.; Mukova, L.; Nikolova, N.; Badjakov, I.; Dincheva, I.; Kondakova, V.; Doumanova, L.; Galabov, A.S. In Vitro Antiviral Activity of a Series of Wild Berry Fruit Extracts against Representatives of Picorna-, Orthomyxo- and Paramyxoviridae. Nat. Prod. Commun. 2014, 9, 51–54. [Google Scholar] [CrossRef] [Green Version]
- Calland, N.; Sahuc, M.E.; Belouzard, S.; Pene, V.; Bonnafous, P.; Mesalam, A.A.; Deloison, G.; Descamps, V.; Sahpaz, S.; Wychowski, C.; et al. Polyphenols Inhibit Hepatitis C Virus Entry by a New Mechanism of Action. J. Virol. 2015, 89, 10053–10063. [Google Scholar] [CrossRef] [Green Version]
- Mathew, D.; Hsu, W.L. Antiviral Potential of Curcumin. J. Funct. Foods 2018, 40, 692–699. [Google Scholar] [CrossRef]
- Qin, Y.; Lin, L.; Chen, Y.; Wu, S.; Si, X.; Wu, H.; Zhai, X.; Wang, Y.; Tong, L.; Pan, B.; et al. Curcumin Inhibits the Replication of Enterovirus 71 in Vitro. Acta Pharm. Sin. B 2014, 4, 284–294. [Google Scholar] [CrossRef] [Green Version]
- Marín-Palma, D.; Tabares-Guevara, J.H.; Zapata-Cardona, M.I.; Flórez-álvarez, L.; Yepes, L.M.; Rugeles, M.T.; Zapata-Builes, W.; Hernandez, J.C.; Taborda, N.A. Curcumin Inhibits in Vitro Sars-Cov-2 Infection in Vero E6 Cells through Multiple Antiviral Mechanisms. Molecules 2021, 26, 6900. [Google Scholar] [CrossRef]
- Vicente Miranda, H.; El-Agnaf, O.M.A.; Outeiro, T.F. Glycation in Parkinson’s Disease and Alzheimer’s Disease. Mov. Disord. 2016, 31, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Lakey-Beitia, J.; Doens, D.; Jagadeesh Kumar, D.; Murillo, E.; Fernandez, P.L.; Rao, K.S.; Durant-Archibold, A.A. Anti-Amyloid Aggregation Activity of Novel Carotenoids: Implications for Alzheimer’s Drug Discovery. Clin. Interv. Aging 2017, 12, 815–822. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nataraj, J.; Manivasagam, T.; Justin, A.; Essa, M.M. Lutein Protects Dopaminergic Neurons against MPTP-Induced Apoptotic Death and Motor Dysfunction by Ameliorating Mitochondrial Disruption and Oxidative Stress. Nutr. Neurosci. 2015, 19, 237–246. [Google Scholar] [CrossRef] [PubMed]
- Ali, T.; Kim, T.; Rehman, S.U.; Khan, M.S.; Amin, F.U.; Khan, M.; Ikram, M.; Kim, M.O. Natural Dietary Supplementation of Anthocyanins via PI3K/Akt/Nrf2/HO-1 Pathways Mitigate Oxidative Stress, Neurodegeneration, and Memory Impairment in a Mouse Model of Alzheimer’s Disease. Mol. Neurobiol. 2018, 55, 6076–6093. [Google Scholar] [CrossRef]
- Li, J.; Zhao, R.; Jiang, Y.; Xu, Y.; Zhao, H.; Lyu, X.; Wu, T. Bilberry Anthocyanins Improve Neuroinflammation and Cognitive Dysfunction in APP/PSEN1 Mice: Via the CD33/TREM2/TYROBP Signaling Pathway in Microglia. Food Funct. 2020, 11, 1572–1584. [Google Scholar] [CrossRef]
- Sohanaki, H.; Baluchnejadmojarad, T.; Nikbakht, F.; Roghani, M. Pelargonidin Improves Memory Deficit in Amyloid Β25-35 Rat Model of Alzheimer’s Disease by Inhibition of Glial Activation, Cholinesterase, and Oxidative Stress. Biomed. Pharmacother. 2016, 83, 85–91. [Google Scholar] [CrossRef]
- Chen, M.; Du, Z.Y.; Zheng, X.; Li, D.L.; Zhou, R.P.; Zhang, K. Use of Curcumin in Diagnosis, Prevention, and Treatment of Alzheimer’s Disease. Neural Regen. Res. 2018, 13, 742–752. [Google Scholar] [CrossRef]
- Qian, W.; Li, H.; Pan, N.; Zhang, C. Curcumin Treatment Is Associated with Increased Expression of the N-Methyl-D-Aspartate Receptor (NMDAR) Subunit, NR2A, in a Rat PC12 Cell Line Model of Alzheimer’s Disease Treated with the Acetyl Amyloid-β Peptide, Aβ(25–35). Med. Sci. Monit. 2018, 24, 2693–2699. [Google Scholar] [CrossRef] [Green Version]
- Shunan, D.; Yu, M.; Guan, H.; Zhou, Y. Neuroprotective Effect of Betalain against AlCl3-Induced Alzheimer’s Disease in Sprague Dawley Rats via Putative Modulation of Oxidative Stress and Nuclear Factor Kappa B (NF-ΚB) Signaling Pathway. Biomed. Pharmacother. 2021, 137, 111369. [Google Scholar] [CrossRef]
- Teerakapong, A.; Damrongrungruang, T.; Sattayut, S.; Morales, N.P.; Tantananugool, S. Efficacy of Erythrosine and Cyanidin-3-Glucoside Mediated Photodynamic Therapy on Porphyromonas Gingivalis Biofilms Using Green Light Laser. Photodiagn. Photodyn. Ther. 2017, 20, 154–158. [Google Scholar] [CrossRef]
- Huang, P.; Zhang, B.; Yuan, Q.; Zhang, X.; Leung, W.; Xu, C. Photodynamic Treatment with Purpurin 18 Effectively Inhibits Triple Negative Breast Cancer by Inducing Cell Apoptosis. Lasers Med. Sci. 2021, 36, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Leung, A.W.; Hua, H.; Rao, X.; Xu, C. Photodynamic Action of LED-Activated Curcumin against Staphylococcus Aureus Involving Intracellular ROS Increase and Membrane Damage. Int. J. Photoenergy 2014, 2014, 9–11. [Google Scholar] [CrossRef] [Green Version]
- Carrion-Gutierrez, M.; Ramirez-Bosca, A.; Navarro-Lopez, V.; Martinez-Andres, A.; Asín-Llorca, M.; Bernd, A.; Horga De La Parte, J.F. Effects of Curcuma Extract and Visible Light on Adults with Plaque Psoriasis. Eur. J. Dermatol. 2015, 25, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Song, B.H.; Lee, D.H.; Kim, B.C.; Ku, S.H.; Park, E.J.; Kwon, I.H.; Kim, K.H.; Kim, K.J. Photodynamic Therapy Using Chlorophyll-a in the Treatment of Acne Vulgaris: A Randomized, Single-Blind, Split-Face Study. J. Am. Acad. Dermatol. 2014, 71, 764–771. [Google Scholar] [CrossRef]
- Suvorov, N.; Pogorilyy, V.; Diachkova, E.; Vasil’ev, Y.; Mironov, A.; Grin, M. Derivatives of Natural Chlorophylls as Agents for Antimicrobial Photodynamic Therapy. Int. J. Mol. Sci. 2021, 22, 6392. [Google Scholar] [CrossRef]
- Saide, A.; Lauritano, C.; Ianora, A. Pheophorbide A: State of the Art. Mar. Drugs 2020, 18, 257. [Google Scholar] [CrossRef]
- Nelson, K.M.; Dahlin, J.L.; Bisson, J.; Graham, J.; Pauli, G.F.; Walters, M.A. The Essential Medicinal Chemistry of Curcumin. J. Med. Chem. 2017, 60, 1620–1637. [Google Scholar] [CrossRef]
- Hejazi, J.; Rastmanesh, R.; Taleban, F.A.; Molana, S.H.; Hejazi, E.; Ehtejab, G.; Hara, N. Effect of Curcumin Supplementation During Radiotherapy on Oxidative Status of Patients with Prostate Cancer: A Double Blinded, Randomized, Placebo-Controlled Study. Nutr. Cancer Int. J. 2016, 68, 77–85. [Google Scholar] [CrossRef]
- Sadraei, M.R.; Tavalaee, M.; Forouzanfar, M.; Nasr-Esfahani, M.H. Effect of Curcumin, and Nano-Curcumin on Sperm Function in Varicocele Rat Model. Andrologia 2022, 54, e14282. [Google Scholar] [CrossRef]
- Kumar, V.; Prakash, C.; Singh, R.; Sharma, D. Curcumin’s Antiepileptic Effect, and Alterations in Na v 1.1 and Na v 1.6 Expression in Iron-Induced Epilepsy. Epilepsy Res. 2019, 150, 7–16. [Google Scholar] [CrossRef]
- Akomolafe, S.F.; Olasehinde, T.A.; Oyeleye, S.I.; Aluko, T.B.; Adewale, O.O.; Ijomone, O.M. Curcumin Administration Mitigates Cyclophosphamide-Induced Oxidative Damage and Restores Alteration of Enzymes Associated with Cognitive Function in Rats’ Brain. Neurotox. Res. 2020, 38, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Mahammedi, H.; Planchat, E.; Pouget, M.; Durando, X.; Cure, H.; Guy, L.; Van-Praagh, I.; Savareux, L.; Atger, M.; Bayet-Robert, M.; et al. The New Combination Docetaxel, Prednisone and Curcumin in Patients with Castration-Resistant Prostate Cancer: A Pilot Phase II Study. Oncology 2016, 90, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Scazzocchio, B.; Minghetti, L.; D’Archivio, M. Interaction between Gut Microbiota and Curcumin: A New Key of Understanding for the Health Effects of Curcumin. Nutrients 2020, 12, 2499. [Google Scholar] [CrossRef] [PubMed]
- Rutz, J.; Maxeiner, S.; Justin, S.; Bachmeier, B.; Bernd, A.; Kippenberger, S.; Zöller, N.; Chun, F.K.H.; Blaheta, R.A. Low Dosed Curcumin Combined with Visible Light Exposure Inhibits Renal Cell Carcinoma Metastatic Behavior in Vitro. Cancers 2020, 12, 302. [Google Scholar] [CrossRef] [Green Version]
- Roos, F.; Binder, K.; Rutz, J.; Maxeiner, S.; Bernd, A.; Kippenberger, S.; Zöller, N.; Chun, F.K.H.; Juengel, E.; Blaheta, R.A. The Antitumor Effect of Curcumin in Urothelial Cancer Cells Is Enhanced by Light Exposure in Vitro. Evid. Based Complement. Altern. Med. 2019, 2019, 6374940. [Google Scholar] [CrossRef] [Green Version]
- Shlar, I.; Droby, S.; Rodov, V. Modes of Antibacterial Action of Curcumin under Dark and Light Conditions: A Toxicoproteomics Approach. J. Proteomics 2017, 160, 8–20. [Google Scholar] [CrossRef]
- Zhang, C.; Su, F.; Li, S.; Yu, Y.; Xiang, J.; Liu, J.; Li, F. Isolation and Identification of the Main Carotenoid Pigment from a New Variety of the Ridgetail White Prawn Exopalaemon Carinicauda. Food Chem. 2018, 269, 450–454. [Google Scholar] [CrossRef]
- Singh, D.; Puri, M.; Wilkens, S.; Mathur, A.S.; Tuli, D.K.; Barrow, C.J. Characterization of a New Zeaxanthin Producing Strain of Chlorella Saccharophila Isolated from New Zealand Marine Waters. Bioresour. Technol. 2013, 143, 308–314. [Google Scholar] [CrossRef]
- Hu, C.W.; Chuang, L.T.; Yu, P.C.; Chen, C.N.N. Pigment Production by a New Thermotolerant Microalga Coelastrella Sp. F50. Food Chem. 2013, 138, 2071–2078. [Google Scholar] [CrossRef]
- Igreja, W.S.; Maia, F.d.A.; Lopes, A.S.; Chisté, R.C. Biotechnological Production of Carotenoids Using Low Cost-Substrates Is Influenced by Cultivation Parameters: A Review. Int. J. Mol. Sci. 2021, 22, 8819. [Google Scholar] [CrossRef]
- Darwesh, O.M.; Matter, I.A.; Almoallim, H.S.; Alharbi, S.A.; Oh, Y.K. Isolation and Optimization of Monascus Ruber OMNRC45 for Red Pigment Production and Evaluation of the Pigment as a Food Colorant. Appl. Sci. 2020, 10, 8867. [Google Scholar] [CrossRef]
- Sunil, L.; Shetty, N.P. Biosynthesis and Regulation of Anthocyanin Pathway Genes. Appl. Microbiol. Biotechnol. 2022, 106, 1783–1798. [Google Scholar] [CrossRef] [PubMed]
- Rapoport, A.; Guzhova, I.; Bernetti, L.; Buzzini, P.; Kieliszek, M.; Kot, A.M. Carotenoids and Some Other Pigments from Fungi and Yeasts. Metabolites 2021, 11, 92. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; He, P.; Zhao, D.; Ye, L.; Dai, L.; Zhang, X.; Sun, Y.; Zheng, J.; Bi, C. Construction of Escherichia Coli Cell Factories for Crocin Biosynthesis. Microb. Cell Fact. 2019, 18, 120. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Binkley, R.M.; Kim, W.J.; Lee, M.H.; Lee, S.Y. Metabolic Engineering of Escherichia Coli for High-Level Astaxanthin Production with High Productivity. Metab. Eng. 2018, 49, 105–115. [Google Scholar] [CrossRef]
- Shrestha, B.; Pandey, R.P.; Darsandhari, S.; Parajuli, P.; Sohng, J.K. Combinatorial Approach for Improved Cyanidin 3-O-Glucoside Production in Escherichia Coli. Microb. Cell Fact. 2019, 18, 7. [Google Scholar] [CrossRef]
- Cress, B.F.; Leitz, Q.D.; Kim, D.C.; Amore, T.D.; Suzuki, J.Y.; Linhardt, R.J.; Koffas, M.A.G. CRISPRi-Mediated Metabolic Engineering of E. Coli for O-Methylated Anthocyanin Production. Microb. Cell Fact. 2017, 16, 10. [Google Scholar] [CrossRef] [Green Version]
- Eichenberger, M.; Hansson, A.; Fischer, D.; Dürr, L.; Naesby, M. De Novo Biosynthesis of Anthocyanins in Saccharomyces Cerevisiae. FEMS Yeast Res. 2018, 18, foy046. [Google Scholar] [CrossRef]
- Du, Y.; Yang, B.; Yi, Z.; Hu, L.; Li, M. Engineering Saccharomyces Cerevisiae Coculture Platform for the Production of Flavonoids. J. Agric. Food Chem. 2020, 68, 2146–2154. [Google Scholar] [CrossRef]
- Espada-Bellido, E.; Ferreiro-González, M.; Carrera, C.; Palma, M.; Barroso, C.G.; Barbero, G.F. Optimization of the Ultrasound-Assisted Extraction of Anthocyanins and Total Phenolic Compounds in Mulberry (Morus Nigra) Pulp. Food Chem. 2017, 219, 23–32. [Google Scholar] [CrossRef]
- Tiwari, S.; Upadhyay, N.; Singh, A.K.; Meena, G.S.; Arora, S. Organic Solvent-Free Extraction of Carotenoids from Carrot Bio-Waste and Its Physico-Chemical Properties. J. Food Sci. Technol. 2019, 56, 4678–4687. [Google Scholar] [CrossRef]
- Elik, A.; Yanık, D.K.; Göğüş, F. Microwave-Assisted Extraction of Carotenoids from Carrot Juice Processing Waste Using Flaxseed Oil as a Solvent. Lwt 2020, 123, 109100. [Google Scholar] [CrossRef]
- Elez Garofulić, I.; Dragović-Uzelac, V.; Režek Jambrak, A.; Jukić, M. The Effect of Microwave Assisted Extraction on the Isolation of Anthocyanins and Phenolic Acids from Sour Cherry Marasca (Prunus Cerasus Var. Marasca). J. Food Eng. 2013, 117, 437–442. [Google Scholar] [CrossRef]
- De Andrade Lima, M.; Kestekoglou, I.; Charalampopoulos, D.; Chatzifragkou, A. Supercritical Fluid Extraction of Carotenoids from Vegetable Waste Matrices. Molecules 2019, 24, 466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gong, Y.; Huang, X.Y.; Liu, J.F.; Pei, D.; Sun, X.; Di, D.L. Development of an Effective Method Based upon Second-Order Overlapping Repeated Sample Injections for Isolation of Carotenoids from Lycium Barbarum L. Fruits with Elution-Extrusion Counter-Current Chromatography. J. Chromatogr. A 2021, 1645, 462026. [Google Scholar] [CrossRef]
- Xue, H.; Tan, J.; Li, Q.; Tang, J.; Cai, X. Optimization Ultrasound-Assisted Deep Eutectic Solvent Extraction of Anthocyanins from Raspberry Using Response Surface Methodology Coupled with Genetic Algorithm. Foods 2020, 9, 1409. [Google Scholar] [CrossRef]
- Trouillas, P.; Sancho-Garcia, J.C.; De Freitas, V.; Gierschner, J.; Otyepka, M.; Dangles, O. Stabilizing and Modulating Color by Copigmentation: Insights from Review Theory and Experiment. Chem. Rev. 2016, 116, 4937–4982. [Google Scholar] [CrossRef] [Green Version]
- Klisurova, D.; Petrova, I.; Ognyanov, M.; Georgiev, Y.; Kratchanova, M.; Denev, P. Co-Pigmentation of Black Chokeberry (Aronia Melanocarpa) Anthocyanins with Phenolic Co-Pigments and Herbal Extracts. Food Chem. 2019, 279, 162–170. [Google Scholar] [CrossRef]
- Khalifa, I.; Du, J.; Nawaz, A.; Li, C. Multiple Co-Pigments of Quercetin and Chlorogenic Acid Blends Intensify the Color of Mulberry Anthocyanins: Insights from Hyperchromicity, Kinetics, and Molecular Modeling Investigations. J. Sci. Food Agric. 2021, 101, 1579–1588. [Google Scholar] [CrossRef]
- Sun, X.; Shokri, S.; Gao, B.; Xu, Z.; Li, B.; Zhu, T.; Wang, Y.; Zhu, J. Improving Effects of Three Selected Co-Pigments on Fermentation, Color Stability, and Anthocyanins Content of Blueberry Wine. Lwt 2022, 156, 113070. [Google Scholar] [CrossRef]
- Ertan, K.; Türkyılmaz, M.; Özkan, M. Color and Stability of Anthocyanins in Strawberry Nectars Containing Various Co-Pigment Sources and Sweeteners. Food Chem. 2020, 310, 125856. [Google Scholar] [CrossRef] [PubMed]
- Patil, V.M.; Das, S.; Balasubramanian, K. Quantum Chemical and Docking Insights into Bioavailability Enhancement of Curcumin by Piperine in Pepper. J. Phys. Chem. A 2016, 120, 3643–3653. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.S.; Xin, Z.; Li, N.N.; Chang, S.Y.; Chen, Y.D.; Geng, L.N.; Chang, H.R.; Shi, H.L.; Chang, Y.Z. Nano-Liposomes of Lycopene Reduces Ischemic Brain Damage in Rodents by Regulating Iron Metabolism. Free Radic. Biol. Med. 2018, 124, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.K.; Wang, D.; Liu, F.G.; Gao, Y.X. Emulsion Design for the Delivery of Beta-Carotene in Complex Food Systems. Crit. Rev. Food Sci. Nutr. 2018, 58, 770–784. [Google Scholar] [CrossRef]
- Toragall, V.; Jayapala, N.; Vallikannan, B. Chitosan-Oleic Acid-Sodium Alginate a Hybrid Nanocarrier as an Efficient Delivery System for Enhancement of Lutein Stability and Bioavailability. Int. J. Biol. Macromol. 2020, 150, 578–594. [Google Scholar] [CrossRef]
- Hoshyar, R.; Khayati, G.R.; Poorgholami, M.; Kaykhaii, M. A Novel Green One-Step Synthesis of Gold Nanoparticles Using Crocin and Their Anti-Cancer Activities. J. Photochem. Photobiol. B Biol. 2016, 159, 237–242. [Google Scholar] [CrossRef]
- Pattni, B.S.; Chupin, V.V.; Torchilin, V.P. New Developments in Liposomal Drug Delivery. Chem. Rev. 2015, 115, 10938–10966. [Google Scholar] [CrossRef]
- Tan, C.; Xue, J.; Abbas, S.; Feng, B.; Zhang, X.; Xia, S. Liposome as a Delivery System for Carotenoids: Comparative Antioxidant Activity of Carotenoids as Measured by Ferric Reducing Antioxidant Power, DPPH Assay and Lipid Peroxidation. J. Agric. Food Chem. 2014, 62, 6726–6735. [Google Scholar] [CrossRef]
- Butnariu, M.V.; Giuchici, C.V. The Use of Some Nanoemulsions Based on Aqueous Propolis and Lycopene Extract in the Skin’s Protective Mechanisms against UVA Radiation. J. Nanobiotechnol. 2011, 9, 3. [Google Scholar] [CrossRef] [Green Version]
- Zardini, A.A.; Mohebbi, M.; Farhoosh, R.; Bolurian, S. Production and Characterization of Nanostructured Lipid Carriers and Solid Lipid Nanoparticles Containing Lycopene for Food Fortification. J. Food Sci. Technol. 2018, 55, 287–298. [Google Scholar] [CrossRef]
- Zhao, L.S.; Temelli, F. Preparation of Anthocyanin-Loaded Liposomes Using an Improved Supercritical Carbon Dioxide Method. Innov. Food Sci. Emerg. Technol. 2017, 39, 119–128. [Google Scholar] [CrossRef]
- Lee, C.; Na, K. Anthocyanin-Loaded Liposomes Prepared by the PH-Gradient Loading Method to Enhance the Anthocyanin Stability, Antioxidation Effect and Skin Permeability. Macromol. Res. 2020, 28, 289–297. [Google Scholar] [CrossRef]
- Kontogiannopoulos, K.N.; Assimopoulou, A.N.; Dimas, K.; Papageorgiou, V.P. Shikonin-Loaded Liposomes as a New Drug Delivery System: Physicochemical Characterization and in Vitro Cytotoxicity. Eur. J. Lipid Sci. Technol. 2011, 113, 1113–1123. [Google Scholar] [CrossRef]
- Feng, T.; Wei, Y.; Lee, R.J.; Zhao, L. Liposomal Curcumin and Its Application in Cancer. Int. J. Nanomed. 2017, 12, 6027–6044. [Google Scholar] [CrossRef] [Green Version]
- Amjadi, S.; Ghorbani, M.; Hamishehkar, H.; Roufegarinejad, L. Improvement in the Stability of Betanin by Liposomal Nanocarriers: Its Application in Gummy Candy as a Food Model. Food Chem. 2018, 256, 156–162. [Google Scholar] [CrossRef]
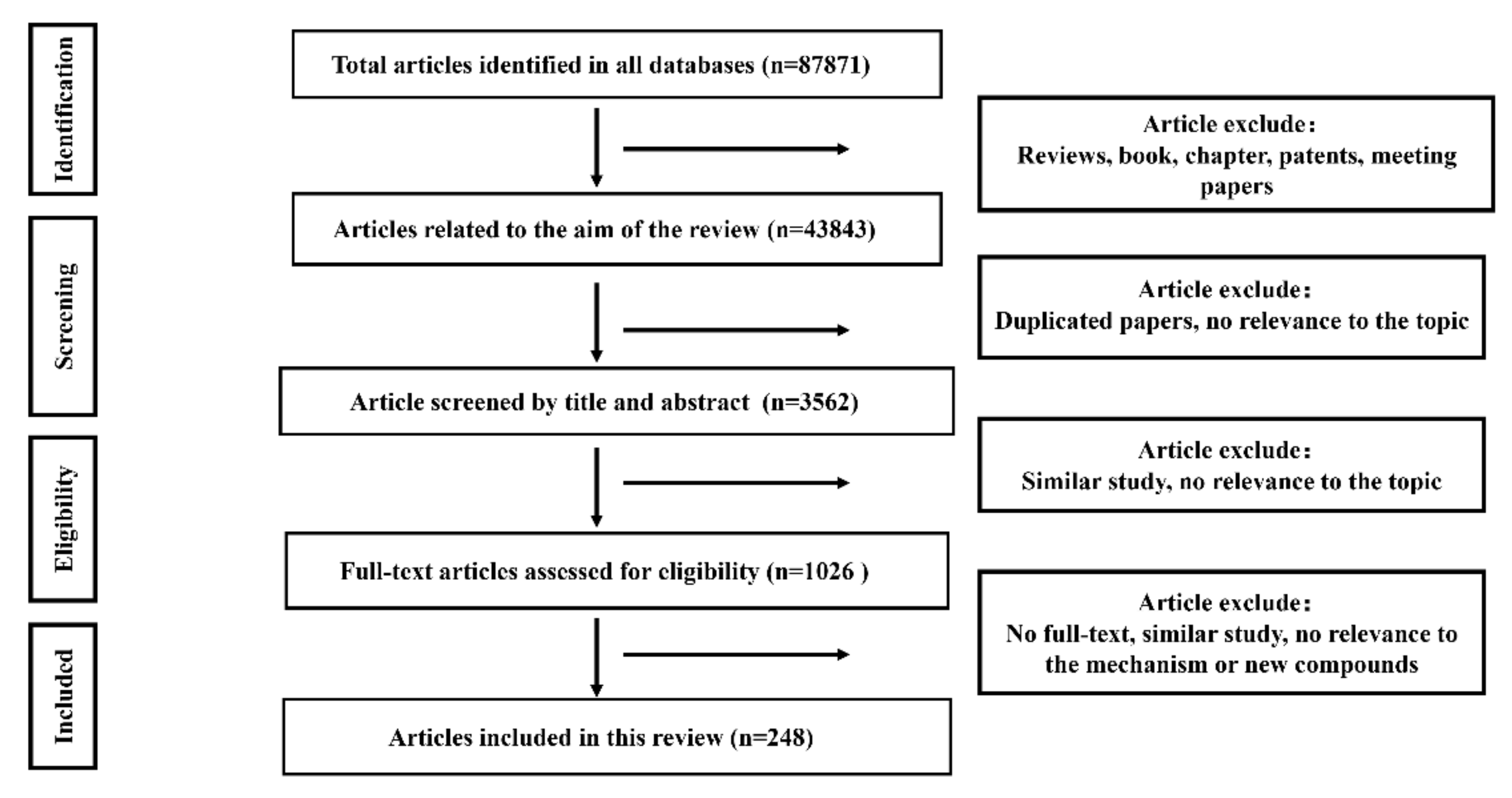
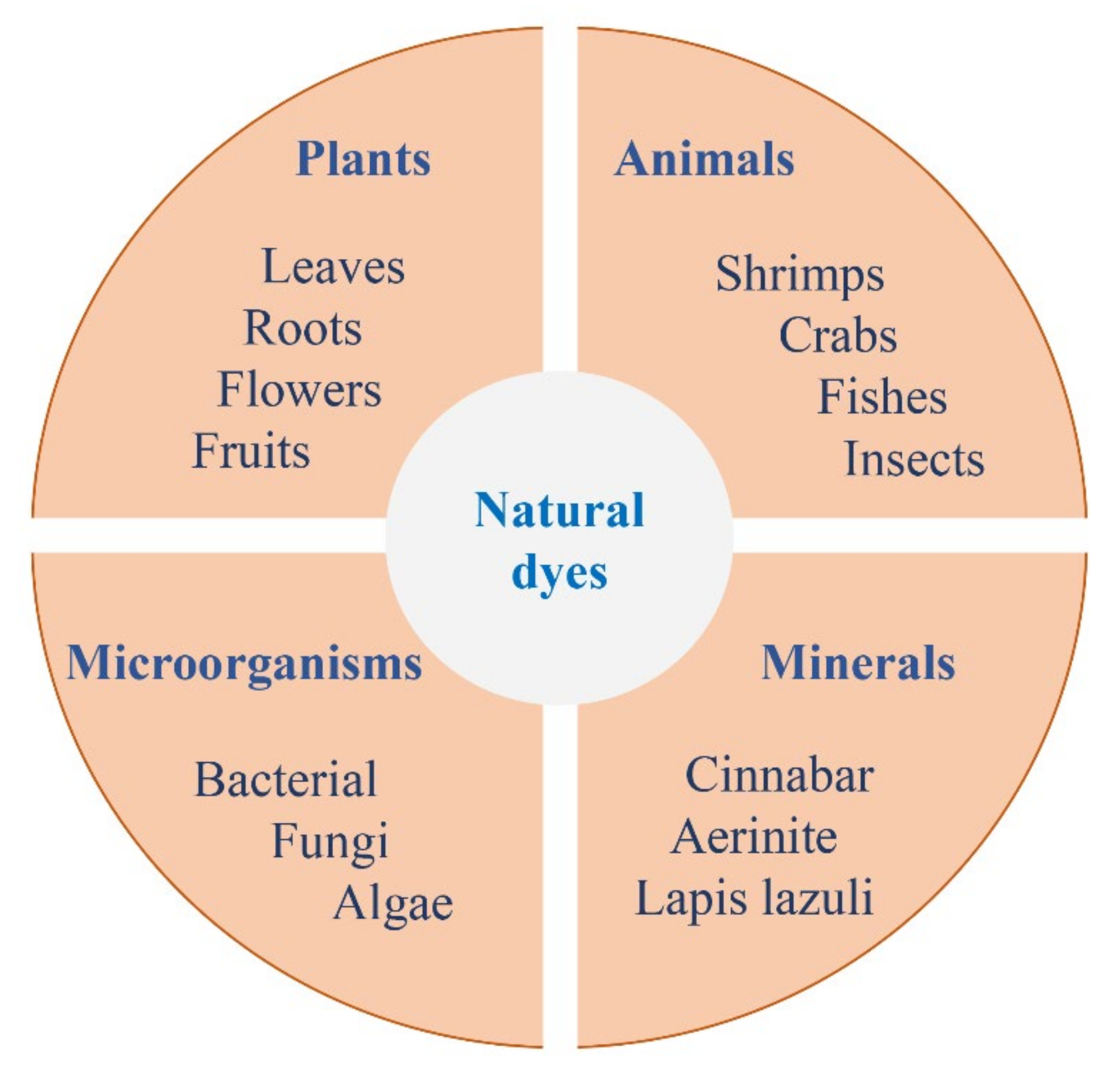
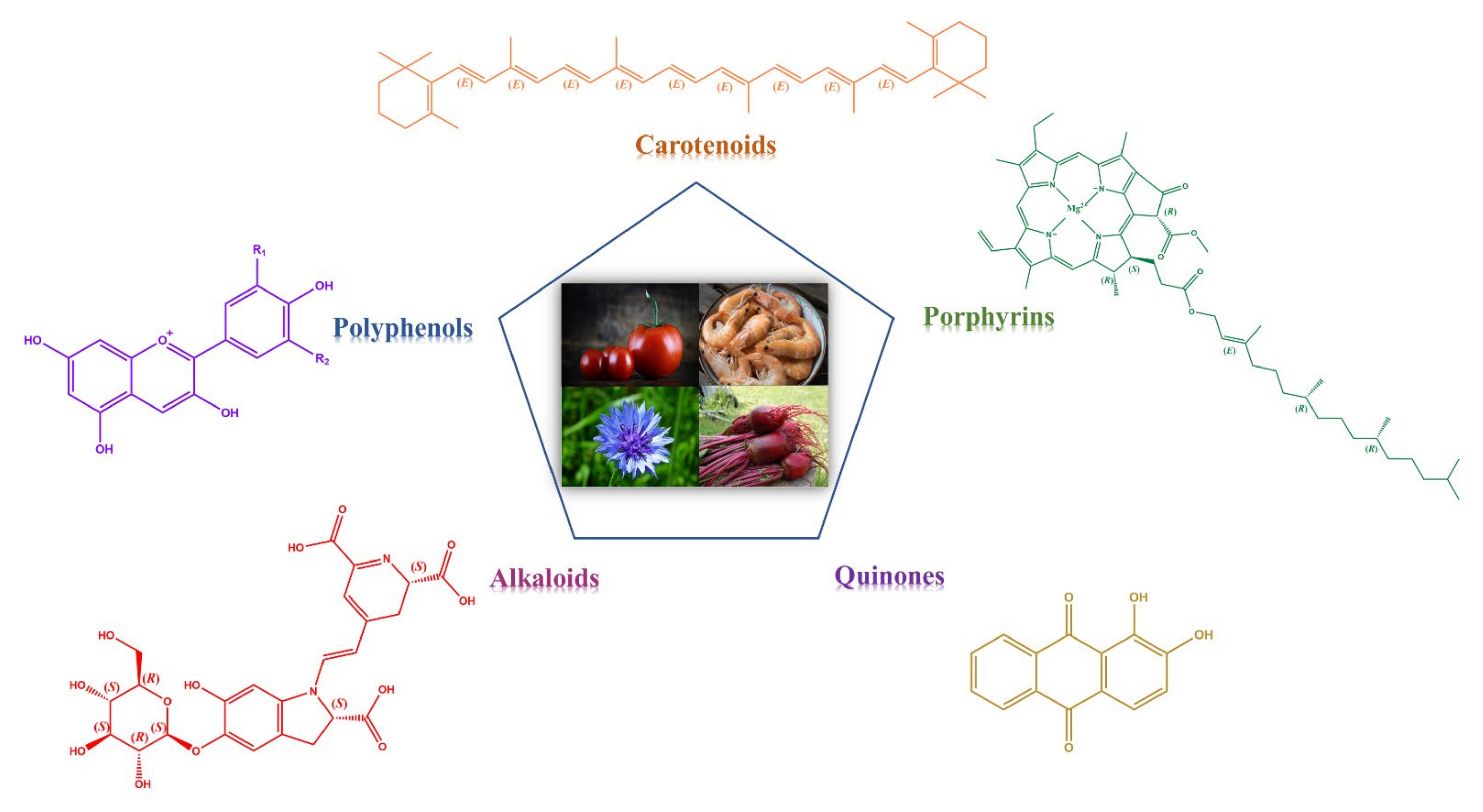
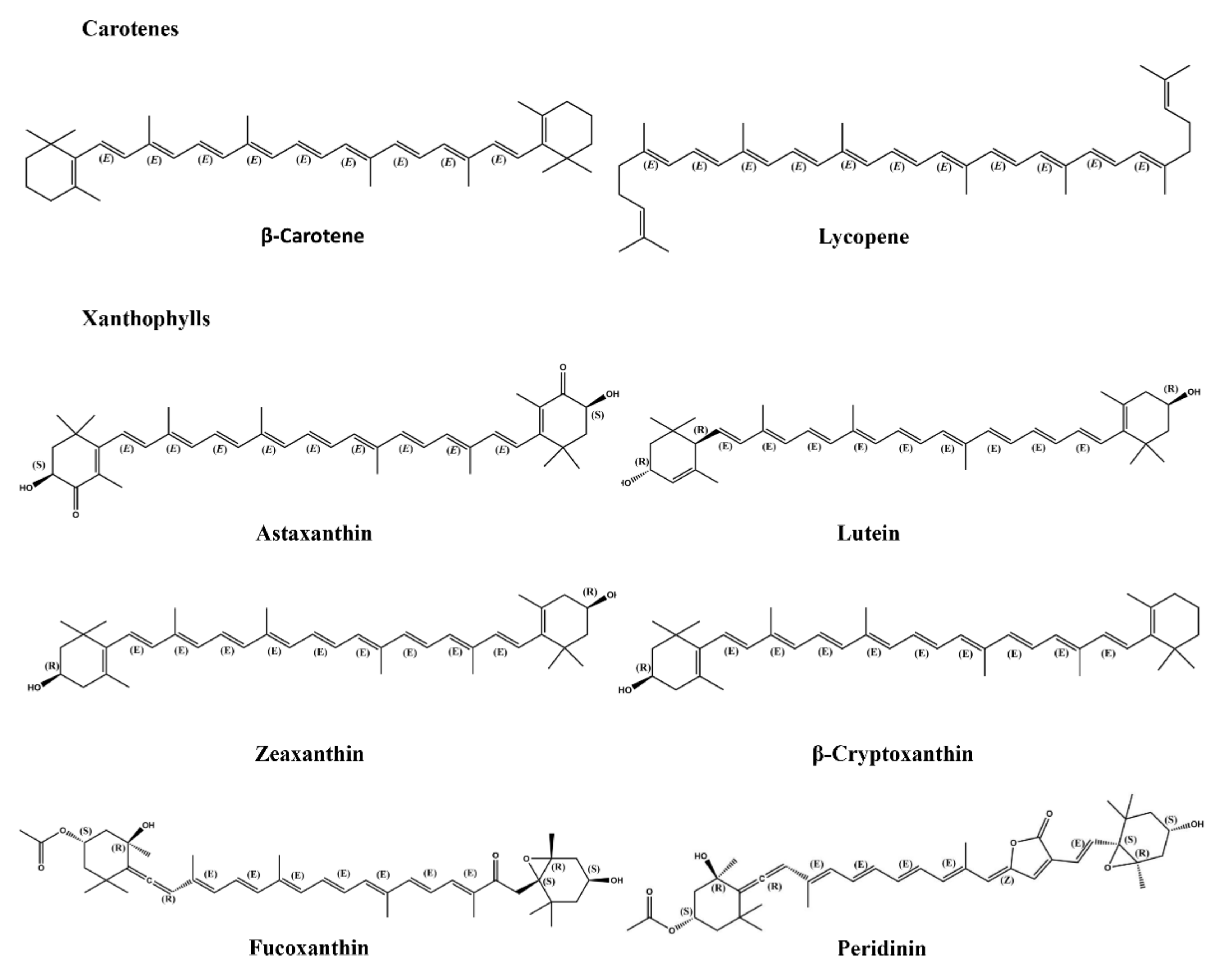
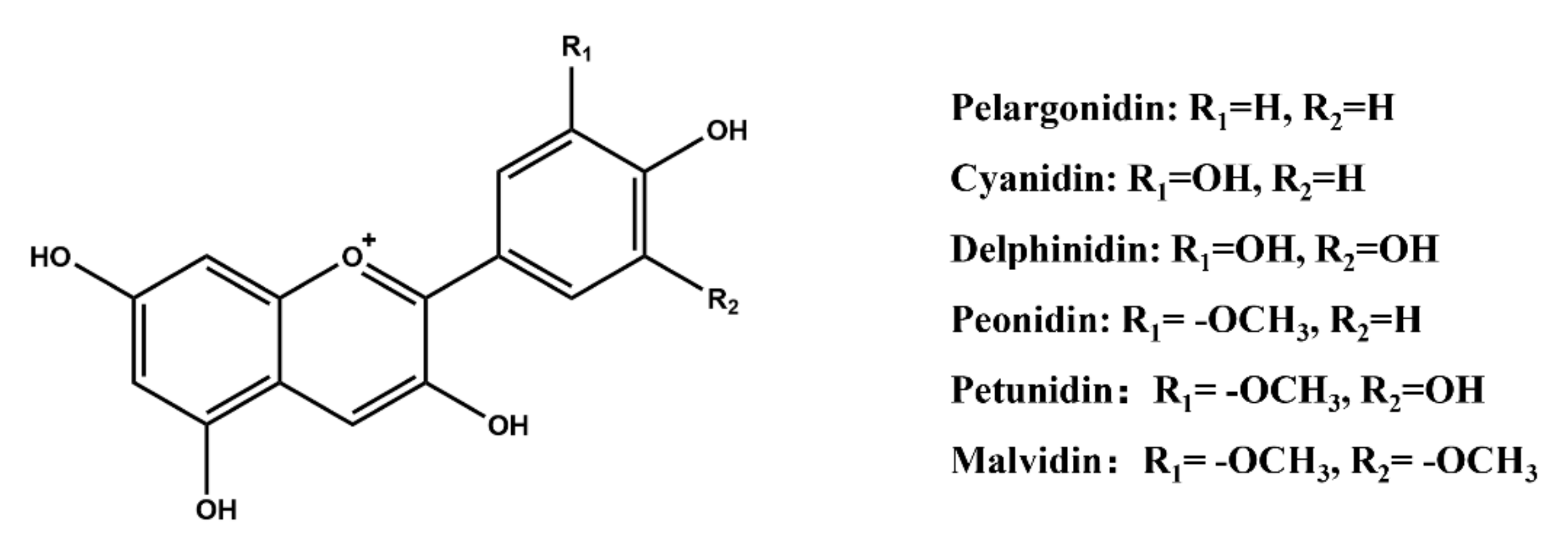
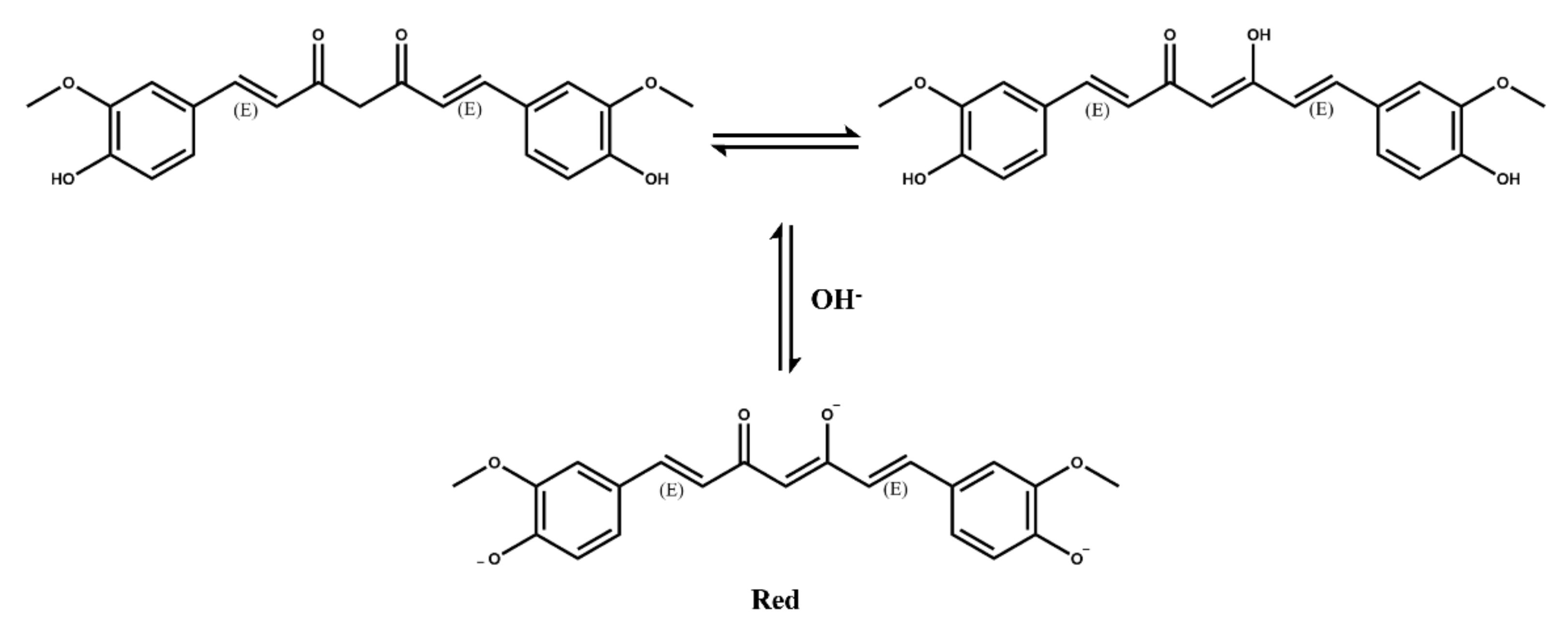
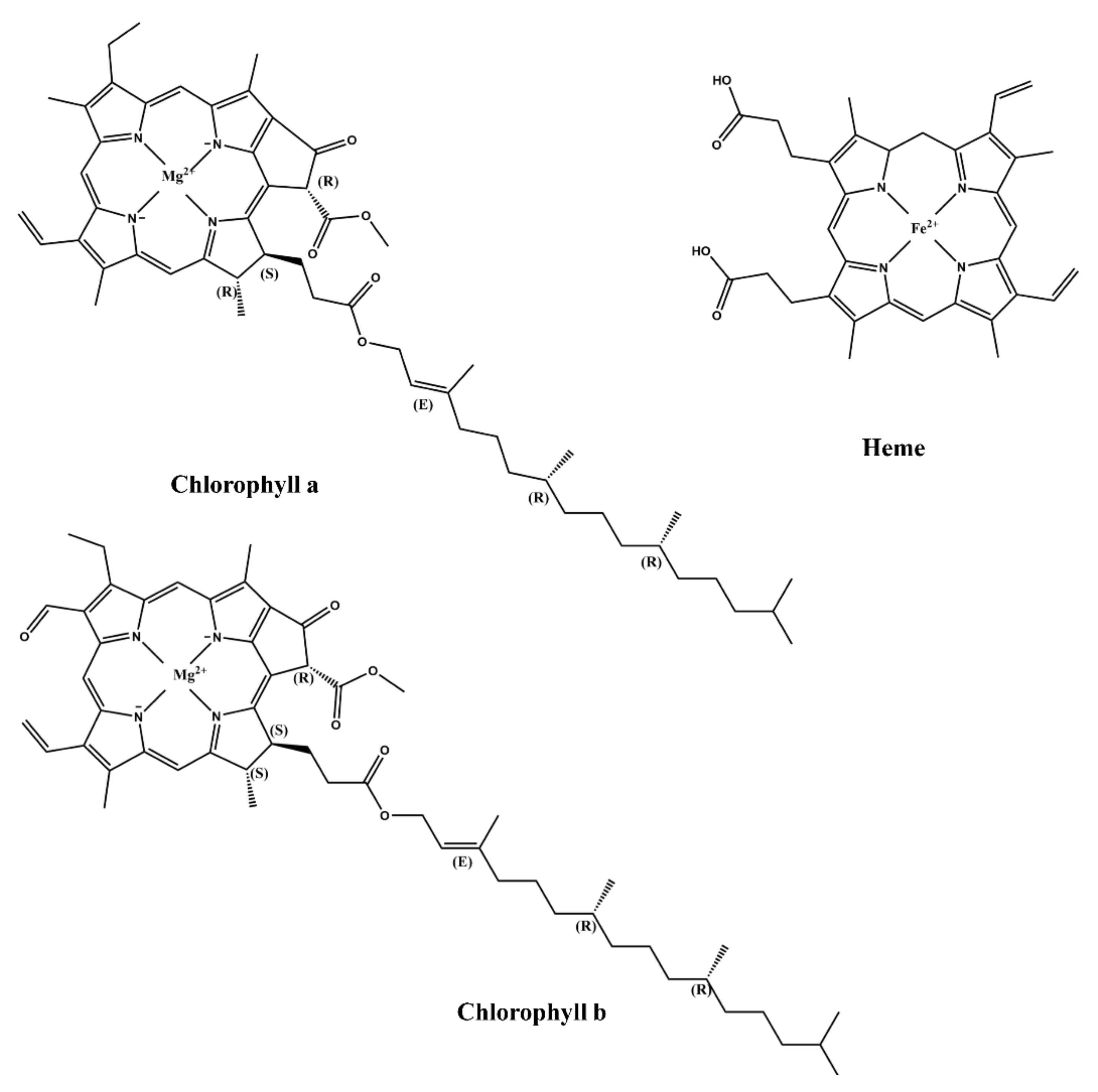
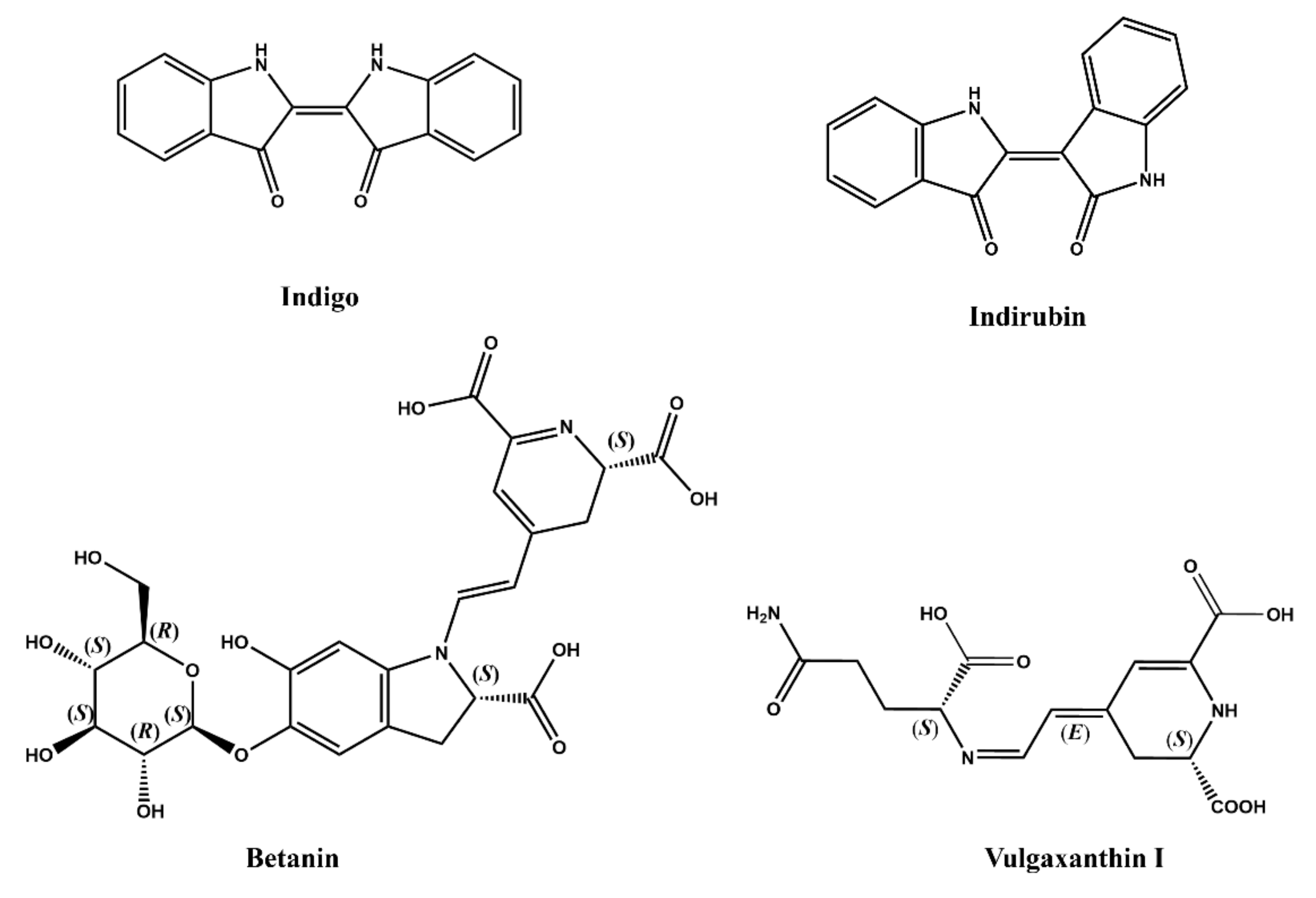
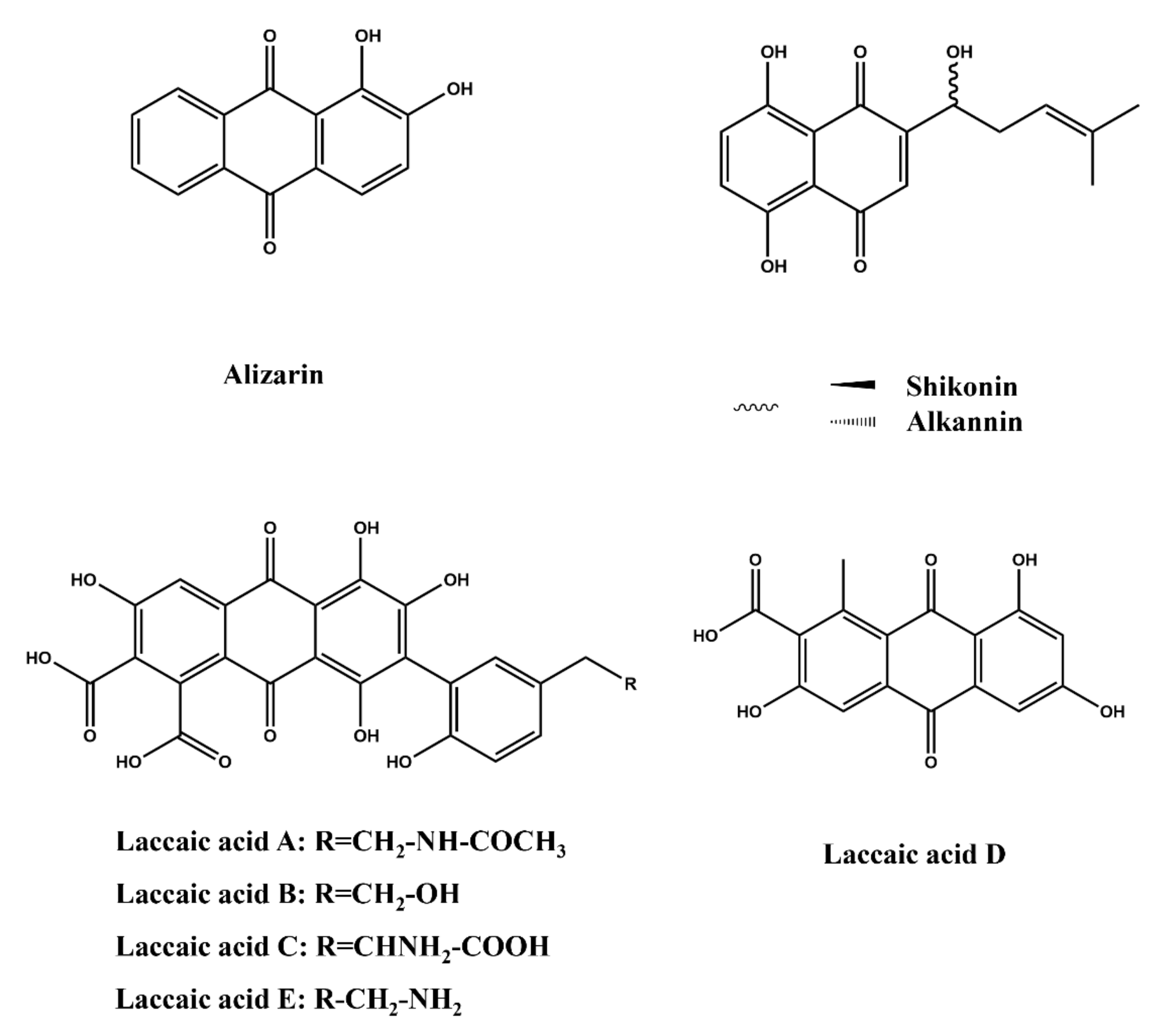
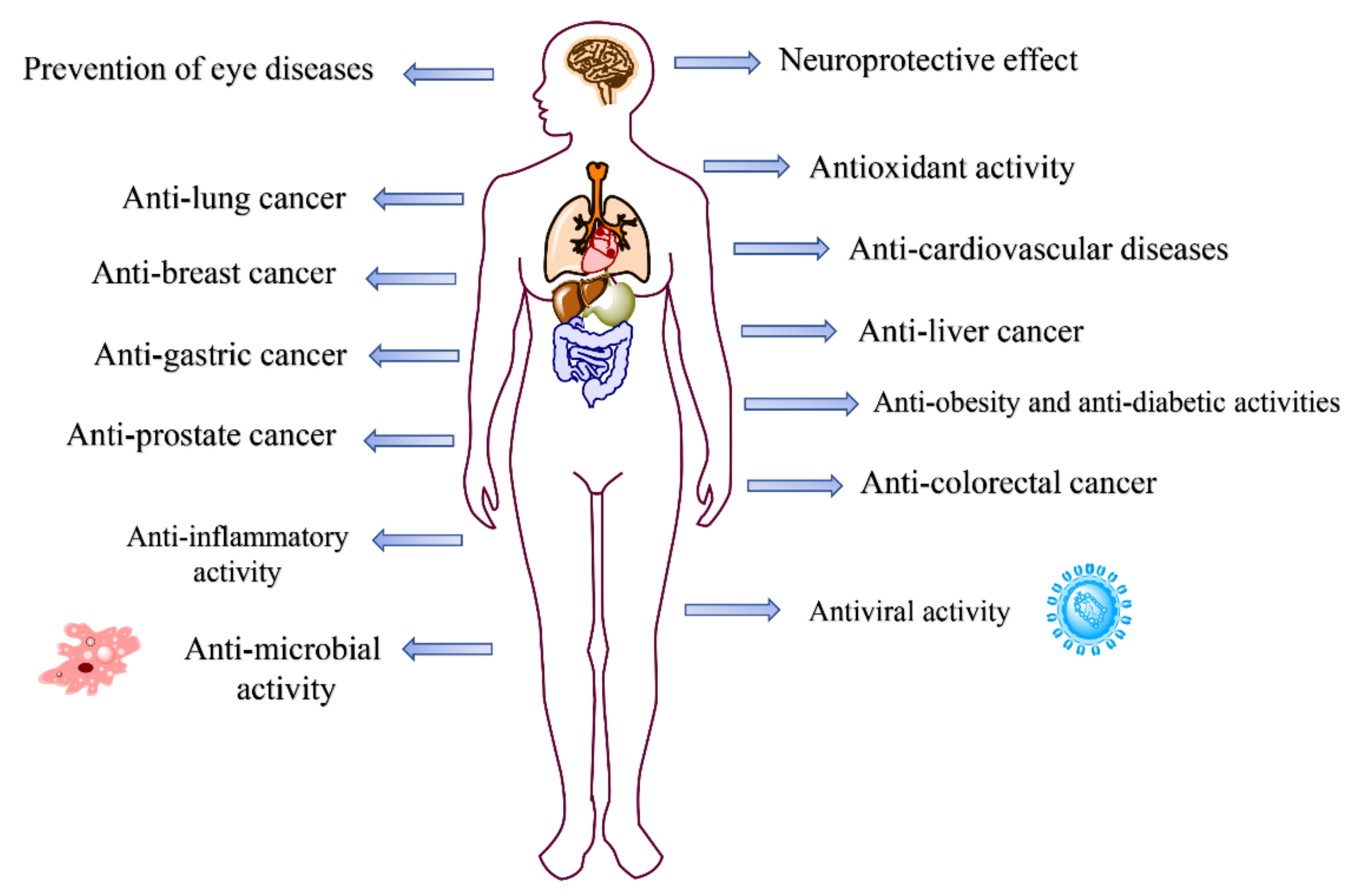
| Electronic Database | Search and Terms |
|---|---|
| Web of Science PubMed | #1 (“Natural dye” OR “Natural pigment” OR “Natural colorants”) AND (“Carotenoids” OR “Anthocyanins” OR “Curcumin” OR “Chlorophylls” OR “Alkaloid” OR “Quinone”) #2 “New” And “Pigment” AND (“Carotenoids” OR “Anthocyanins” OR “Curcumin” OR “Alkaloid” OR “Quinone”) AND “Isolated” #3 (“Carotenoids” OR “Anthocyanins” OR “Curcumin” OR “Betalain”) AND (“Antioxidant” OR “Inflammatory” OR “Anti-cancer” OR “Cancer” OR “Anti-bacterial” OR “Antimicrobial” OR “Obesity” OR “Anti-obesity” OR “Diabetes” OR “Cardiovascular” OR “Anti-viral” OR “Neuroprotective” OR “Alzheimer’s disease”) #4 (“Extraction” OR “Isolation” OR “Extracted” OR “Isolated”) AND (“Carotenoids” OR “Anthocyanins”) AND “New method” |
| Category | Compounds | Source | Ref. |
|---|---|---|---|
| Carotenoids | 6′-Epimonadoxanthin | Rosary goby (Gymnogobius castaneus) | [51] |
| 3′-Deoxycapsorubin | Red mamey (Pouteria sapota) | [52] | |
| 3,3′-Dideoxycapsorubin | Red mamey (Pouteria sapota) | [52] | |
| Methyl 5-glucosyl-5,6-dihydro-apo-4,4′-lycopenoate | Planococcus maritimus strain iso-3 | [53] | |
| Diapolycopenedioc Acid Xylosylesters A/B/C | Rubritalea squalenifaciens | [53] | |
| 13Z-zeaxanthin dipalmitate | Wolfberry | [54] | |
| Anthocyanins | Malvidin-3-(p-coumaroyl)-rutinoside-5-glucoside | Transgenic Del/Ros1 tomato fruit | [55] |
| Malvidin-3-(feruloyl)-rutinoside-5-glucoside | Transgenic Del/Ros1 tomato fruit | [55] | |
| Petunidin-3-(cis-p-coumaroyl)-rutinoside-5-glucoside | Tomato cultivar Indigo Rose | [56] | |
| Malvidin-3-(cis-p-coumaroyl)-rutinoside-5-glucoside | Tomato cultivar Indigo Rose | [56] | |
| Petunidin-3-(trans-p-coumaroyl-rhamonside)-glucoside-5-glucoside | Tomato cultivar Indigo Rose | [56] | |
| Malvidin-3-(p-methoxy-trans-coumaroyl)-rutinoside-5-glucosid | Tomato cultivar Indigo Rose | [56] | |
| Delphinidin 3-O-a-l-rhamnopyranosyl-(1→6)-b-d-glucopyranoside-30-O-b-d-glucopyranoside | Tamarillo fruit | [57] | |
| Cyanidin 3-[2′’-(6′’’-coumaroyl)-glucosyl]-glucoside | Nitraria tangutorum | [58] | |
| Pelargonidin-3-O-coumaroylglucoside | Mulberry (Morus moraceae) juice | [59] | |
| Delphinidin-3-O-coumaroylglucoside | Mulberry (Morus moraceae) juice | [59] | |
| Cyanidin 3-O-[2-O-(2-O-(4-O-(6-O-(4-O-(β-glucopyranosyl)-trans-caffeoyl)-β-glucopyranosyl)-trans-caffeoyl)-β-glucopyranosyl)-6-O-(trans-sinapoyl)-β-glucopyranoside]-5-O-[6-O-(malonyl)- glucopyranoside] | Purple-violet flowers of Moricandia arvensis | [60] | |
| 5,7-Dimethylmalvidin 3-O-β-galactopyranoside | Blue Plumbago flower | [61] | |
| 5,7-Di-methylpetunidin 3-O-β-galactopyranoside | Blue Plumbago flower | [61] | |
| 5,7-Di-methyldelphinidin 3-O-β-galactopyranoside | Blue Plumbago flower | [56] | |
| 5,7-Dimethylmalvidin 3-O-α-rhamnopyranoside | Blue Plumbago flower | [61] | |
| 5,7-Dimethyldelphinidin 3-O-α- rhamnopyranoside | Blue Plumbago flower | [61] | |
| 5,7-Dimethylpetunidin 3-O-α-rhamnopyranoside | Blue Plumbago flower | [61] | |
| petunidin 3-O-[6-O-(4-O-(4-O-cis-(β-d-glucopyranoside)-p-coumaroyl)-α-l-rhamnopyranosyl)-β-d-glucopyranoside] -5-O-[β-d-glucopyranoside] | Wild Lycium ruthenicum Murr. | [62] | |
| 3-O-(6-O-α-l-Rhamnopyranosyl-β-d-glucopyranosyl)-7-O-(6-O-(4-O-(6-O-(E)-caffeoyl-β-d-glucopyranosyl)-(E)-caffeoyl)-β-d-glucopyranosyl) del-phinidin | Bluish-purple petals of Chinese bellflower (Platycodon grandifloru) | [63] | |
| 3-O-(6-O-α-l-Rhamnopyranosyl-β-d-glucopyranosyl)-7-O-(6-O-(4-O-(6-O-(4-O-β-d-glucopyranosyl-(E)-p-coumaroyl)-β-d-glucopyranosyl)-(E)-caffeoyl)-β-d-glucopyranosyl) delphinidin | Bluish-purple petals of Chinese bellflower (Platycodon grandifloru) | [63] | |
| 3-O-(6-O-α-l-Rhamnopyranosyl-β-d-glucopyranosyl)-7-O-(6-O-(4-O-(6-O-(4-O-β-d-glucopyranosyl-(E)-caffeoyl)-β-d-glucopyranosyl)-(E)-p-coumaroyl)-β-d-glucopyranosyl) delphinidin | Bluish-purple petals of Chinese bellflower (Platycodon grandifloru) | [63] | |
| Alatanin D | Purple yam (Dioscorea alata L.) | [64] | |
| Alatanin E | Purple yam (Dioscorea alata L.) | [64] | |
| Alatanin F | Purple yam (Dioscorea alata L.) | [64] | |
| Alatanin G | Purple yam (Dioscorea alata L.) | [64] | |
| Panaxidin A (pelaragonidin-4-vinylcatechol) | Panax quinquefolius L. | [65] | |
| Panaxidin B (pelargonidin-4-vinylphenol) | Panax quinquefolius L. | [65] | |
| Alkaloid | Alstoscholarisine F/G | Alstonia scholaris | [66] |
| Oryzadiamine C | Oryza sativa mutant | [67] | |
| Oryzadiamine A | Oryza sativa with yellow grain | [68] | |
| Rosellin A | Mushroom Mycena rosella | [69] | |
| Rosellin B | Mushroom Mycena rosella | [69] | |
| Ergopigment 8/9/10 | Claviceps purpurea | [70] | |
| Katorazone | Streptomyces sp. IFM 11299 | [71] | |
| 2-(4-((3E,5E)-14-Aminotetradeca-3,5-dienyloxy) butyl)-1,2,3,4-tetrahydroisoquinolin-4-ol | Fusarium moniliforme KUMBF1201 | [72] | |
| 6′-O-malonyl-amaranthin | Callus culture of Celosia cristata L. | [73] | |
| Quinone | Hypalocrinins A/B/C/D/E/F/G | Deep-sea crinoid Hypalocrinus naresianus | [24] |
| 5′-Hydroxytrypethelone | The mycobiont of lichen Trypethelium eluteriae Sprengel | [74] | |
| Gymnochrome A/H | Deep-sea crinoid Hypalocrinus naresianus | [23] | |
| 1,4,6b,7,10-Pentahydroxy-1,2,6b,7,8,12b-hexahydroperylene-3,9-dione | Endophytic fungus Alternaria tenuissima SS77 | [75] | |
| 1,4,9,12a-Tetrahydroxy-12-methoxy-1,2,11,12,12a,12b-Hexahydroperylene-3,10-dione | Endophytic fungus Alternaria tenuissima SS77 | [75] | |
| 1,4,9-tri-hydroxy-1,2-Dihydroperylene-3,10-dione | Endophytic fungus Alternaria tenuissima SS77 | [75] | |
| Alaternosides A/C | Rhamnus alaternus L | [76] | |
| 6-Methoxy-rhodocomatulin 7-methyl ether | Australian sponge Clathria hirsuta | [77] | |
| 3-Bromo-6-methoxy-12-desethyl- rhodocomatulin 7-methyl ether | Australian sponge Clathria hirsuta | [77] | |
| 3-Bromo-6-methoxy-rhodocomatulin 7-methyl ether | Australian sponge Clathria hirsuta | [77] | |
| 3-Bromorhodocomatulin 7-methyl ether | Australian sponge Clathria hirsuta | [77] | |
| Grandiquinone A | Leaves of Tectona grandis | [78] | |
| Phomopsanthraquinone | Fungus Phomopsis sp. PSU-MA214 | [79] |
| Category | Compounds Name | Mechanism | Refs. |
|---|---|---|---|
| Carotenoids | Astaxanthin | Scavenged free radicals, quenched singlet oxygen, ↑ antioxidant enzyme paroxoanase-1, ↑ glutathione concentrations, ↓ lipid peroxidation. | [116] |
| Activated the Nrf-2/HO-1 antioxidant pathway by generating small amounts of ROS in HUVEC model. | [126] | ||
| ↓ Oxidative stress, ↓ MDA content, ↑ SOD | [127] | ||
| Lycopene | ↓ NADPH oxidase, ↓ ROS production | [128] | |
| Lutein | ↑ SOD, ↓ ROS level, ↑ CAT, ↑ GPx, ↓ GR, ↓ MDA, ↑ reduced glutathione level | [129] | |
| Zeaxanthin | ↓ Myeloperoxidase, ↓ MDA, ↑ SOD, ↑ CAT, ↑ glutathione level | [130] | |
| Polyphenols | Anthocyanins | Scavenged free radicals, ↑ SOD, ↑ total antioxidant activity | [120] |
| Cyanidin-3-arabinoside | ↓ Renal oxidative stress (↑ SOD, ↑ CAT), ↓ lipid peroxidation (↓ TBARS and ↓ MDA) | [121] | |
| Gy3G, Mv3G | ↓ ROS, sustained the level of GSH and glutathione antioxidant defense system | [122] | |
| Petunidin-3,5-O-diglucoside | Scavenged free radicals, ↓ ROS, ↓ MDA level and GSH consumption | [123] | |
| Anthocyanin extract from purple highland barley | Scavenged free radicals, ↓ ROS, ↑ SOD, ↑ CAT | [131] | |
| Curcumin | ↓ Serum MDA, ↑ total antioxidant activity, ↑ transcription and expression levels of antioxidant enzymes, ↑ mitochondrial function | [124,125] | |
| Alkaloids | Betalain | ↓ MDA, ↑ CAT, ↑ SOD, ↑ GPx, ↑ xanthine oxidase | [132] |
| Betanin | Scavenged free radicals, ↓ MDA, ↑ total antioxidant activity | [133] |
| Cancers | Compounds Name | Category | Mechanism | Refs. |
|---|---|---|---|---|
| Breast cancer | Lycopene | Carotenoids | Activation of ERK1/2, ↓ cyclin D1 ↑ p21 ↓ phosphorylation of Akt and its downstream molecule mTOR ↑ Bax | [154] |
| β-Carotene | Carotenoids | ↑ Apoptosis ↓ cell cycle ↓ PI3K/Akt ↓ ERK | [155,156] | |
| Lutein | Carotenoids | ↓ Breast cancer cell proliferation, ↑ expression of cellular antioxidant enzymes, ↓ ROS, ↑ NrF2/ARE pathway, ↓ NF-κB signaling pathway ↑ p53, ↓ HSP60 | [157,158] | |
| Crocin | Carotenoids | ↑ Disrupting the microtubule network ↓ Wnt/β-catenin target genes | [159,160] | |
| Astaxanthin | Carotenoids | ↓ Cellular migration, ↓ cell number ↓ Expression levels of pontin, mutp53, Oct4, and Nanog, ↓ proliferation Activation of Bax/Bcl2, cleaved caspase-3, and cleaved caspase-9 as well as the phosphorylation of ERK1/2, JNK, and p38 | [143,144,145] | |
| D-3-5-D, C3R | Polyphenols | ↑ Intracellular reactive oxygen, ↑ apoptosis ↓ MCF-7 cells in the G2/M phases | [161] | |
| C3G, Pg-3-G | Polyphenols | ↓ AMPK, ↑ apoptosis ↑ Oxidative stress | [162] | |
| Curcumin | Polyphenols | ↓ NF-κB signaling pathway ↓ HER2-TK ↓ Akt protein, ↓ ubiquitin-proteasome pathway ↓ PI3K/Akt signaling pathway ↓ EGFR signaling | [163,164,165,166,167] | |
| Betanin | Alkaloids | ↑ Apoptosis-related proteins (Bad, TRAILR4, FAS, p53) | [168] | |
| Colorectal Cancer | Astaxanthin | Carotenoids | ↓ Invadopodia, ↓ EMT, ↑ E-cadherin, ↓ vimentin, ↓ cortactin, ↓ MMP2, ↑ miR-29a-3p, ↓ ZEB1, ↓ MYC ↑ Apoptosis, ↑ Bax, ↑ caspase-3, ↓ Bcl2 | [169,170] |
| Fucoxanthin | Carotenoids | ↓ Proliferation ↑ DNA damage | [171,172] | |
| Crocin | Carotenoids | ↑ Caspase-3 and -7, ↓ proliferation | [173] | |
| C3G, C3XR, C3R | Polyphenols | ↑ Probiotics, ↓ inflammation ↓ Pathogenic bacteria | [174] | |
| C3G, C3XR, C3R | Polyphenols | ↑ MiR-24-1-5p, ↓ β-catenin | [175] | |
| Pg-3-G | Polyphenols | ↓ HT-29 colon cancer cells | [176] | |
| Anthocyanin extract | Polyphenols | ↓ Wnt/β-catenin ↓ Mitochondrion-mediated apoptosis | [146] | |
| Curcumin | Polyphenols | ↓ NF-κB pathway, ↓ cell cycle ↑ Cytochrome c, ↑ Bax and p53, ↓ Bcl-2 | [177,178] | |
| Betaxanthin and betacyanin | Alkaloids | ↓ Bcl2-like protein 4, ↓ cleaved poly ADP-ribosyl polymerase 1, ↓ cleaved caspase-3 ↓ Anti-apoptotic protein B-cell leukemia/lymphoma 2 levels | [179] | |
| Gastric cancer | Crocin | Carotenoids | ↓ KLF5 HIF-1, ↑ miR-320, ↓ epithelial–mesenchymal transition, ↓ migration | [180] |
| β-Carotene | Carotenoids | ↓ Cell viability, ↑ DNA damage, ↑ apoptotic indices, ↑ caspase-3, ↓ Ku70/80 | [181] | |
| Fucoxanthin | Carotenoids | ↑ Beclin-1, ↑ LC3, ↑ cleaved caspase-3 (CC3), ↓ Bcl-2, ↓ cell cycle, ↑ apoptosis, ↓ Mcl-1, STAT3, and p-STAT3 | [182,183] | |
| Astaxanthin | Carotenoids | ↓ Cell cycle ↑ NADPH oxidase activity, ↑ ROS levels, ↑ LDH release, ↑ the number of propidium iodide-positive cells ↑ RIP1/RIP3/MLKL signaling pathway | [184,185] | |
| Curcumin | Polyphenols | ↓ STAT3 pathway | [178] | |
| Liver cancer | Astaxanthin | Carotenoids | ↑ Cell number in G2 phase ↑ Cell number in G2/M phase ↑ Apoptosis ↑ Oxidative stress, ↑ adiponectin | [186,187,188] |
| Crocin | Carotenoids | ↓ NF-κB, ↓ inflammation, ↓ cell cycle, ↑ apoptosis | [189] | |
| Fucoxanthin | Carotenoids | ↓ Glutathione (GSH) content, ↓ proliferation Reverting body weight, serum albumin, antioxidant enzymes, all the liver enzymes, serum bilirubin, and stress markers to normal levels in hepatocellular carcinoma rats | [190,191] | |
| C3G, Pn-3-G | Polyphenols | ↓ TNF-α, iNOS, NF-κB ↓ Cell proliferation | [192] | |
| C3G, C3R | Polyphenols | ↓ Lipid peroxidation, ↓ COX-2 ↑ Nrf2-mediated antioxidant enzymes | [193] | |
| M3G | Polyphenols | ↓ Proliferation, ↑ apoptosis, ↓ ROS, ↑ JNK/p38 MAPK pathways, ↓ AKT phosphorylation, ↓ migration, ↓ invasion | [147] | |
| Curcumin | Polyphenols | ↓ Migration, ↓ invasion, ↓ epithelial–mesenchymal transition, ↓ aryl hydrocarbon receptor/ERK/SK1/S1P3 signaling pathway | [194] | |
| Betanin | Alkaloids | ↑ Nrf2, ↑ mitogen-activated protein kinases | [195] | |
| Lung cancer | Astaxanthin | Carotenoids | ↑ Cell number in G0/G1 phase ↑ p38 MAPK ↑ Apoptosis | [196] |
| Crocin | Carotenoids | ↑ G0/G1 arrest, ↑ mRNA levels of p53 and Bax, ↓ Bcl-2, ↑ apoptosis | [197] | |
| Lutein | Carotenoids | ↓ PI3K/AKT, ↑ apoptosis | [198] | |
| C3G | Polyphenols | ↓ Lung tumor multiplicity and tumor area, ↓ expression of proliferative cell nuclear antigen (PCNA) and Ki-67 | [199] | |
| Curcumin | Polyphenols | ↓ NF-κB, ↓ JAK2/STAT3 signaling pathway, ↓ JAK2 ↓ Cell proliferation, ↑ apoptosis ↑ microRNA-192-5p, ↓ PI3K/Akt signaling pathway ↓ Wnt/β-catenin pathway | [150,151,152,153] | |
| Betalain | Alkaloids | ↑ Proliferation, ↓ cell cycles, ↑ p53/p21, ↓ levels of cyclin-D1 complex, ↓ levels of p-PI3K, ↓ p-Akt, ↓ mammalian target of rapamycin | [200] | |
| Prostate cancer | Astaxanthin | Carotenoids | ↑ Apoptosis, ↑ cleaved caspase-3; ↑ miR-375 and miR-487b | [201] |
| Crocin | Carotenoids | ↓ Proliferation, ↓ cell cycle, ↑ apoptosis ↓ Bcl-2, ↓ Bax | [202] | |
| Proanthocyanidins | Polyphenols | ↓ Notch1 pathway | [203] | |
| C3G | Polyphenols | ↓ Epithelial–mesenchymal transition | [204] | |
| Curcumin | Polyphenols | ↓ Expression of CYP11A1 and HSD3B2, ↑ AKR1C2, ↓ dihydrotestos terone ↑ miR-34a, ↓ β-catenin, ↓ c-myc | [205,206] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, N.; Wang, Q.; Zhou, J.; Li, S.; Liu, J.; Chen, H. Insight into the Progress on Natural Dyes: Sources, Structural Features, Health Effects, Challenges, and Potential. Molecules 2022, 27, 3291. https://doi.org/10.3390/molecules27103291
Li N, Wang Q, Zhou J, Li S, Liu J, Chen H. Insight into the Progress on Natural Dyes: Sources, Structural Features, Health Effects, Challenges, and Potential. Molecules. 2022; 27(10):3291. https://doi.org/10.3390/molecules27103291
Chicago/Turabian StyleLi, Nannan, Qirou Wang, Jingna Zhou, Shuqin Li, Junyu Liu, and Haixia Chen. 2022. "Insight into the Progress on Natural Dyes: Sources, Structural Features, Health Effects, Challenges, and Potential" Molecules 27, no. 10: 3291. https://doi.org/10.3390/molecules27103291
APA StyleLi, N., Wang, Q., Zhou, J., Li, S., Liu, J., & Chen, H. (2022). Insight into the Progress on Natural Dyes: Sources, Structural Features, Health Effects, Challenges, and Potential. Molecules, 27(10), 3291. https://doi.org/10.3390/molecules27103291







