ICP-MS Determination of Antimicrobial Metals in Microcapsules
Abstract
:1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. Instrumentation
3.2. Reagents and Solutions
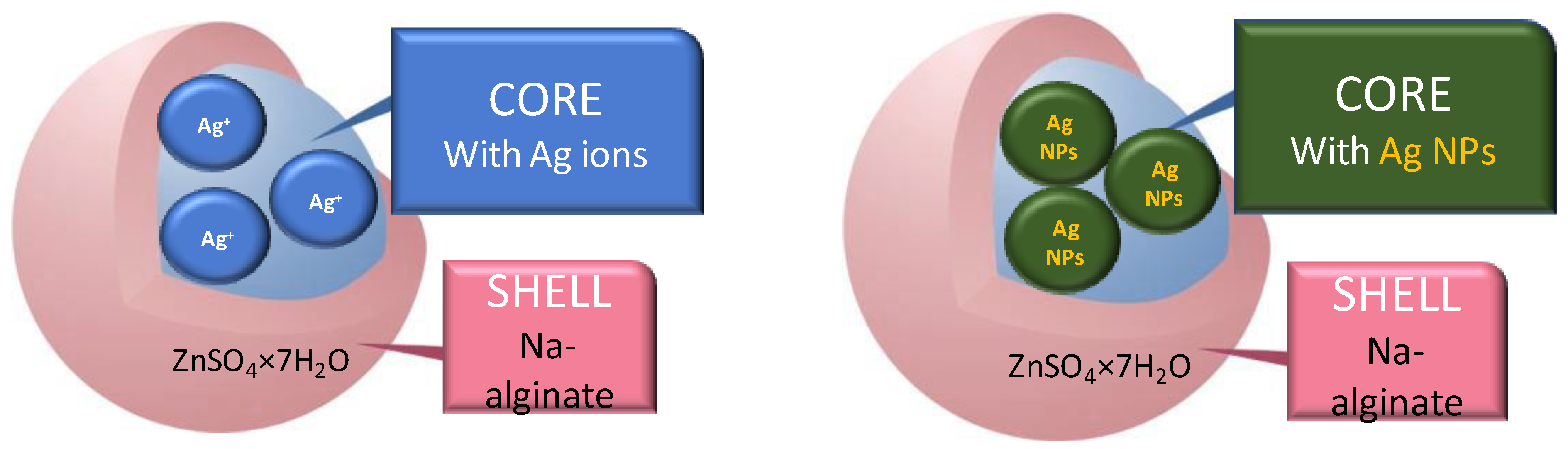
3.3. Microencapsulation
3.4. Microwave Digestion
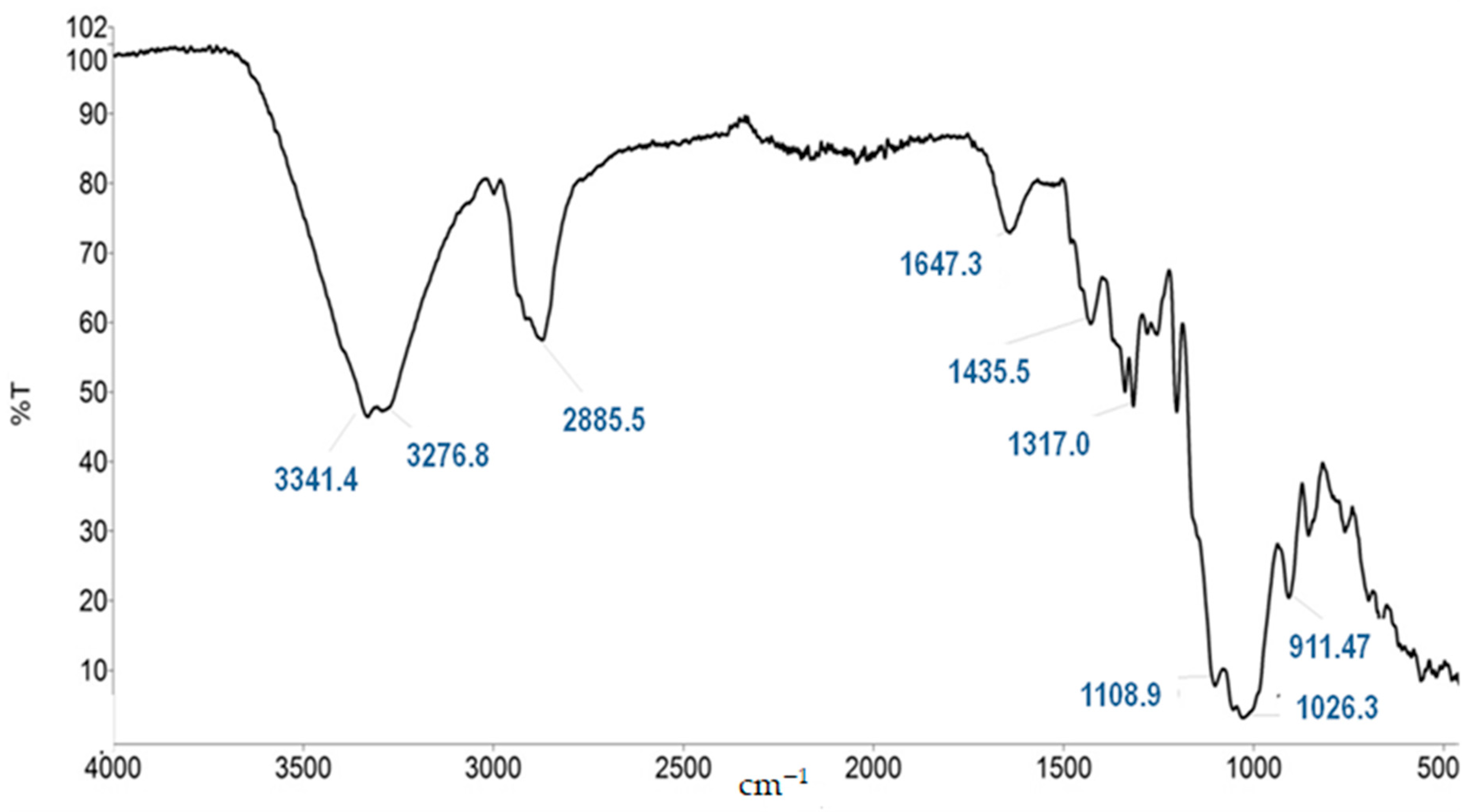
3.5. Sol-Gel Procedure
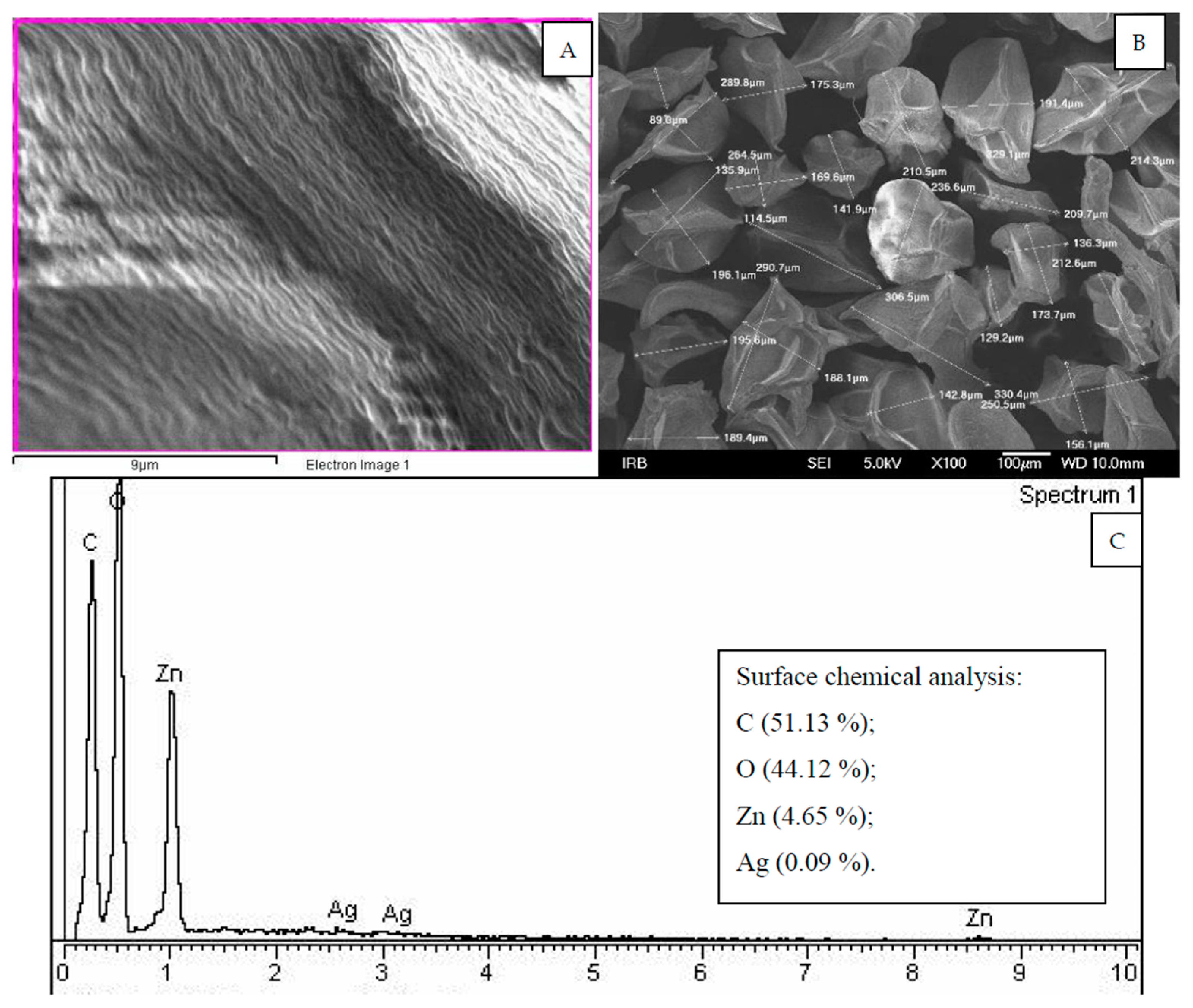
3.6. SEM-EDX Characterization
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Seil, J.T.; Webster, T.J. Antimicrobial applications of nanotechnology. Int. J. Nanomed. 2012, 7, 2767–2781. [Google Scholar]
- Azam, A.; Ahmed, A.S.; Oves, M.; Khan, M.S.; Habib, S.S.; Memic, A. Antimicrobial activity of metal oxide nanoparticles against Gram-positive and Gram-negative bacteria: A comparative study. Int. J. Nanomed. 2011, 7, 6003–6009. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirin, P. The Analysis of Antimicrobial Surface by Thin Layer Chromatography. Master’s Thesis, Faculty of Textile Technology, University of Zagreb, Zagreb, Croatia, 2019. [Google Scholar]
- Tago, A.; Yanase, M.; Yamauchi, N.; Nakashima, K.; Nagao, D.; Kobayashi, Y. Preparation and properties of silica-coated metallic nickel particles. Coll. Surf. A Physicochem. Eng. Asp. 2021, 629, 127524–127529. [Google Scholar] [CrossRef]
- Batcheller, J.; Hacke, A.M.; Mitchell, R.; Carr, C.M. Investigation into the nature of historical tapestries using time of flight secondary ion mass spectrometry (ToF-SIMS). Appl. Surf. Sci. 2006, 19, 7113–7116. [Google Scholar] [CrossRef]
- Urstoeger, A.; Zacherl, L.; Muhr, M.; Selic, Y.; Wenisch, M.; Klotz, M.; Schuster, M. Using a Cation-Exchange Resin. Variables Affecting Their Separation and Improvements of AgNP Characterization by SP-ICPMS. Talanta 2021, 225, 122028. [Google Scholar] [CrossRef]
- Iglesias, M.; Torrent, L. Silver Nanoparticles and Ionic Silver Separation. Nanomaterials 2021, 11, 2626. [Google Scholar] [CrossRef]
- Rezić, I.; Steffan, I. ICP-OES determination of metals present in textile materials. Microchem. J. 2007, 85, 46–49. [Google Scholar] [CrossRef]
- Guan, M.; Kang, X.; Wei, L.; Hu, X.; Han, C.; Li, X.; Liu, H.; Qu, L.; Zhao, Z. Ligand Effects on Intramolecular Configuration, Intermolecular Packing, and Optical Properties of Metal Nanoclusters. Talanta 2022, 241, 123277. [Google Scholar] [CrossRef]
- Nölte, J. ICP Emission Spectrometry, A Practical Guide; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Silva, F.V.; Trevizan, L.C.; Silva, C.S.; Nogueira, A.R.A.; Nóbrega, J.A. Evaluation of inductively coupled plasma optical emission spectrometers with axially and radially viewed configurations. Spectrochim. Acta B 2002, 57, 1905–1913. [Google Scholar] [CrossRef]
- Brenner, I.B.; Zander, A.T. Axially and radially viewed inductively coupled plasmas a critical review. Spectrochim. Acta Part B 2000, 51, 1195–1240. [Google Scholar] [CrossRef]
- Gruszka, J.; Martyna, A.; Godlewska-Żyłkiewicz, B. Chemometric approach to discrimination and determination of binary mixtures of silver ions and nanoparticles in consumer products by graphite furnace atomic absorption spectrometry. Talanta 2021, 230, 122319. [Google Scholar] [CrossRef] [PubMed]
- Adelantado, J.V.G.; Eres, M.A.F.; Algarra, F.M.V.; Vicente, J.P.; Reig, F.B. The Dor 2002/2 Shipwreck, Israel: Characterization of Surviving Ironwork. Talanta 2003, 60, 895–910. [Google Scholar]
- Mazzocchin, G.A.; Agnoli, F.; Salvadori, M. Analysis of Roman age wall paintings found in Pordenone, Trieste and Montegrotto. Talanta 2005, 64, 732–741. [Google Scholar] [CrossRef] [PubMed]
- Mazzocchin, G.A.; Agnoli, F.; Mazzocchin, S.; Colpo, I. Analysis of pigments from Roman wall paintings found in Vicenza. Talanta 2003, 61, 565–572. [Google Scholar] [CrossRef]
- Giménez, R.G.; Villa, R.V.; Rosa, P.R.; Domínguez, M.D.P.; Rucandio, M.I. Analytical and multivariate study of roman age architectural terracotta from northeast of Spain. Talanta 2005, 65, 861–868. [Google Scholar] [CrossRef] [PubMed]
- Coedo, A.G.; Dorado, T.; Padilla, I.; Fariñas, J.C. Study of heterogeneities in steels and determination of soluble and total aluminium and titanium concentration by laser ablation inductively coupled plasma mass spectrometry. Talanta 2007, 71, 2108–2120. [Google Scholar] [CrossRef]
- Rezić, I.; Ćurković, L.; Ujević, M. Metal ion release from electric guitar strings in artificial sweat. Corros. Sci. 2009, 51, 1985–1989. [Google Scholar] [CrossRef]
- Smichowski, P. Comparison of three different sample preparation procedures for the determination of traffic-related elements in airborne particulate matter collected on glass fiber filters. Talanta 2008, 75, 2–14. [Google Scholar] [CrossRef]
- Rezić, I.; Zeiner, M.; Steffan, I. Determination of allergy-causing metals from coins. Monatsh. Chem. 2009, 140, 147–151. [Google Scholar] [CrossRef]
- Marguí, E.; Queralt, I.; Carvalho, M.L.; Hidalgo, M. omparison of EDXRF and ICP-OES after microwave digestion for element determination in plant specimens from an abandoned mining area. Anal. Chim. Acta 2005, 549, 197–204. [Google Scholar] [CrossRef]
- Rezić, I.; Bokić, L.; Horvat, A.J.M. Quantitative Analysis and Validation of Method Using HPTLC. J. Planar. Chromatogr. Mod. TLC 2004, 17, 305–308. [Google Scholar] [CrossRef]
- Suzuki, K. Atmos Characterisation of airborne particulates and associated trace metals deposited on tree bark by ICP-OES, ICP-MS, SEM-EDX and laser ablation ICP-MS. Atmos. Environ. 2006, 40, 2626–2634. [Google Scholar] [CrossRef]
- Bai, R.; Wan, L.; Li, H.; Zhang, Z.; Ma, Z. The detection of metallic residues in skin stab wounds by y means of SEM-EDS: A pilot study. Forensic. Sci. Int. 2007, 166, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Rezić, I. Optimization of ultrasonic extraction of 23 elements from cotton. Ultrason. Sonochem. 2009, 16, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Karatzani, A. Study and analytical investigation of metal threads from Byzantine/Greek ecclesiastical textiles. X-ray Spectrom. 2008, 37, 410–417. [Google Scholar] [CrossRef]
- Karatzani, A.; Rehren, T. The use of metal threads and decorations in Byzantine-Greek Orthodox ecclesiastical textiles. JOM 2006, 58, 34–37. [Google Scholar] [CrossRef]
- Hacke, A.M.; Carr, C.M.; Brown, A. Characterisation of metal threads in Renaissance tapestries. Metal 2004, 4, 415. [Google Scholar]
- Rujido-Santos, I.; Naveiro-Seijo, L.; Herbello-Hermelo, P.; Barciela-Alonso, M.C.; Bermejo-Barrera, P.; Moreda-Piñeiro, A. Nanotechnology meets atopic dermatitis: Current solutions. Talanta 2019, 197, 530–538. [Google Scholar] [CrossRef]
- Parham, H.; Saeed, S. Resonance Rayleigh scattering method for determination of ethion using silver nanoparticles as probe. Talanta 2015, 131, 570–576. [Google Scholar] [CrossRef]
- Bolea, E.; Jimenez, M.S.; Perez-Arantegui, J.; Vidal, J.C.; Bakir, M.; Ben-Jeddou, K.; Gimenez-Ingalaturre, A.C.; Ojeda, D.; Trujillo, C.; Laborda, F. Analytical applications of single particle inductively coupled plasma mass spectrometry: A comprehensive and critical review. Anal. Methods 2021, 13, 2742–2795. [Google Scholar] [CrossRef]
- Nord, A.G.; Troner, K. A note on the analysis of gilded metal embroidery threads. Stud. Conserv. 2000, 45, 274–282. [Google Scholar]
- Enguita, O.; Climent-Font, A.; García, G.; Montero, I.; Fedi, M.E.; Chiari, M.; Lucarelli, F. Characterization of metal threads using differential PIXE analysis. Nucl. Instrum. Methods Phys. Res. B 2002, 189, 328–333. [Google Scholar] [CrossRef]
- Lee, J.M.; Yu, J.E.; Koh, Y.S. Experimental study on the effect of wavelength in the laser cleaning of silver threads. J. Cult. Herit. 2003, 4, 157–161. [Google Scholar] [CrossRef]
- Laborda, F.; Gimenez-Ingalaturre, A.C.; Bolea, E. Suspect Screening of Fentanyl Analogs Using Matrix-Assisted Ionization and a Miniature Mass Spectrometer with a Custom Expandable Mass Spectral Library. Compr. Anal. Chem. 2021, 93, 35–67. [Google Scholar]
- Wang, X.; Yang, H.; Li, K.; Xiang, Y.; Sha, Y.; Zhang, M.; Yuan, X.; Huang, K. Gold Nanoparticles in Chemical and Biological Sensing—NCBI. Appl. Spectr. Rev. 2020, 6, 509–524. [Google Scholar] [CrossRef]
- Rezić, I.; Steffan, I. ICP-OES Monitoring of aluminum, copper, cobalt, iron and manganese during bleaching. AATCC Rev. 2008, 83, 44–48. [Google Scholar]
- Rezić, I.; Zeiner, M.; Steffan, I. Determination of 28 selected elements in textiles by axially viewed inductively coupled plasma optical emission spectrometry. Talanta 2011, 83, 865–871. [Google Scholar] [CrossRef]
- Zeiner, M.; Rezić, I.; Steffan, I. Analytical Methods for the Determination of Heavy Metals in Textile. Kem. Ind. 2007, 56, 587–595. [Google Scholar]
- Bokić, L.J.; Rezić, I.; Flinčec, S. Determination of Alkaline and Earth Alkaline Elements in Natural Cellulose Textile Materials by Atom Absorption Spectrometry. Tekstil 2003, 52, 503–511. [Google Scholar]
- Rezić, I.; Ćurković, L.; Ujević, M. Simple methods for characterization of metals in historical textile threads. Talanta 2010, 82, 237–244. [Google Scholar] [CrossRef]
- Pintarić, L.M.; Škoc, M.S.; Bilić, V.L.; Pokrovac, I.; Kosalec, I.; Rezić, I. Synthesis, Modification and Characterization of Antimicrobial Textile Surface Containing ZnO Nanoparticles. Polymers 2020, 12, 1210. [Google Scholar] [CrossRef] [PubMed]
- Rezić, I.; Kiš, A. Design of Experiment Approach to Optimize Hydrophobic surfaces. Polymers 2020, 12, 2131. [Google Scholar] [CrossRef] [PubMed]
- Rezić, I.; Majdak, M.; Bilić, V.L.; Pokrovac, I.; Martinaga, L.; Škoc, M.S.; Kosalec, I. Development of Antibacterial Protective Coatings Active against MSSA and MRSA. Polymers 2021, 13, 659. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Liang, X.; Chen, N.; Tang, J.; Shao, W.; Gao, Q.; Teng, Z.J. 2D inorganic nanosheet-based hybrid photocatalysts. Coll. Interf. Sci. 2017, 507, 353–359. [Google Scholar] [CrossRef]
- Neri, G.; Corsaro, C.; Fazio, E. Plasmon-Enhanced Controlled Drug Release from Ag-PMA capsules. Molecules 2020, 25, 2267. [Google Scholar] [CrossRef]
- Wang, B.H.; Hu, T.J.S.L. Drug Delivery: Principles and Applications, 2nd ed.; John Wiley and Sons: Hoboken, NJ, USA, 2016; p. 509. [Google Scholar]
- Lengyel, M.; Kállai-Szabó, N.; Antal, V.; Laki, A.J.; Antal, I. Microparticles, microspheres, and microcapsules for advanced drug delivery. Sci. Pharm. 2019, 87, 20. [Google Scholar] [CrossRef] [Green Version]
- Shekunov, B.Y.; Chattopadhyay, P.; Tong, H.H.Y.; Chow, A.H.L. Particle Size Analysis in Pharmaceutics: Principles, Methods and Applications. Pharm. Res. 2007, 24, 203–227. [Google Scholar] [CrossRef]
- Shahi, S. Micro particles: An approach for betterment of drug delivery system. Int. J. Pharm. Res. Dev. 2014, 1, 99–115. [Google Scholar]
- Khan, M.R.; Ahmad, N.; Ouladsmane, M. Azam Heavy Metals in Acrylic Color Paints Intended for the School Children Use: A Potential Threat to the Children of Early Age. Molecules 2021, 19, 2375. [Google Scholar] [CrossRef]
- Szymanska-Chargot, M.; Gruszecka, A.; Smolira, A.; Bederski, K. Mass-Spectrometric Investigations of Silver Cluters. Acta Phys. Pol. A 2011, 120, 1012–1017. [Google Scholar] [CrossRef]

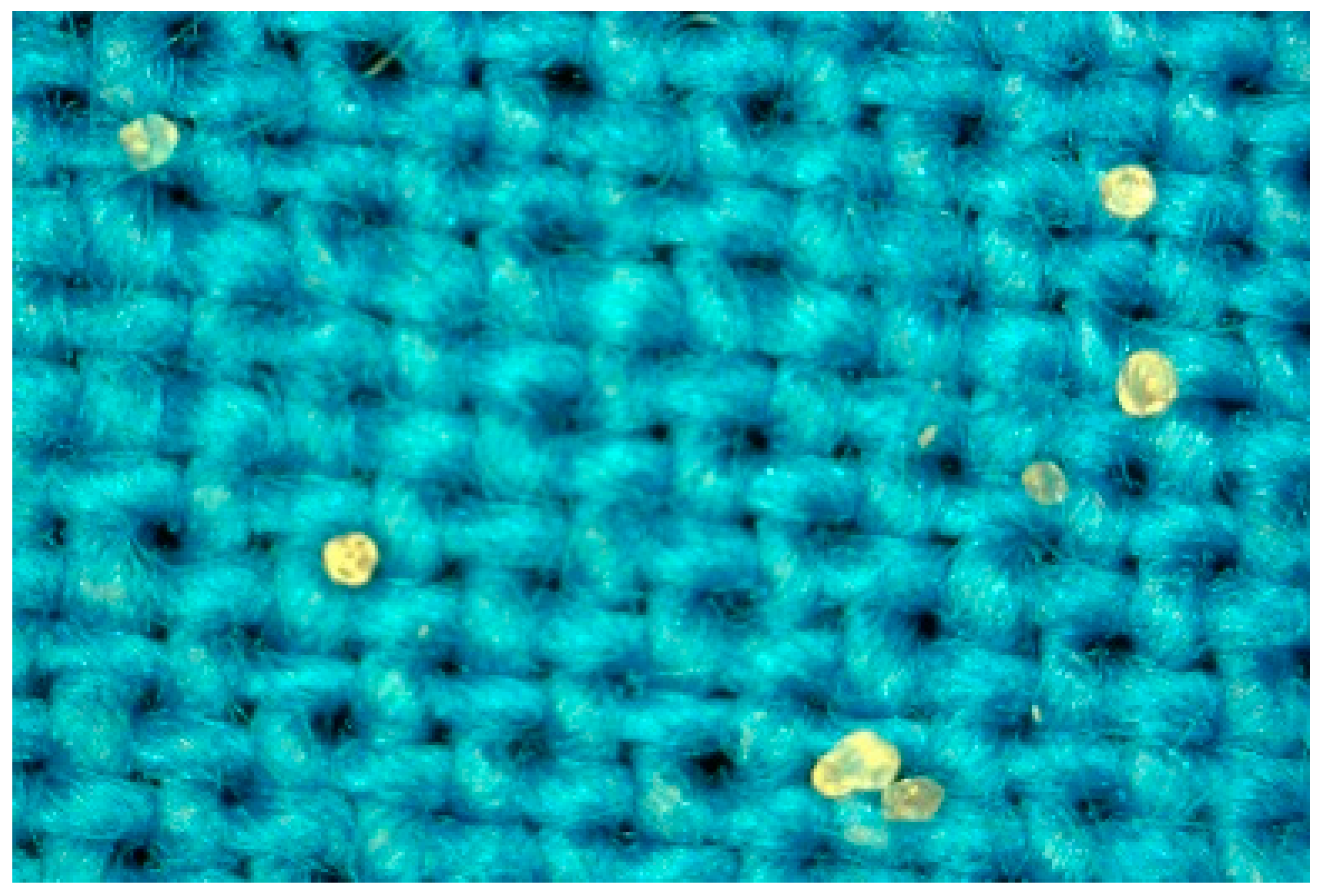
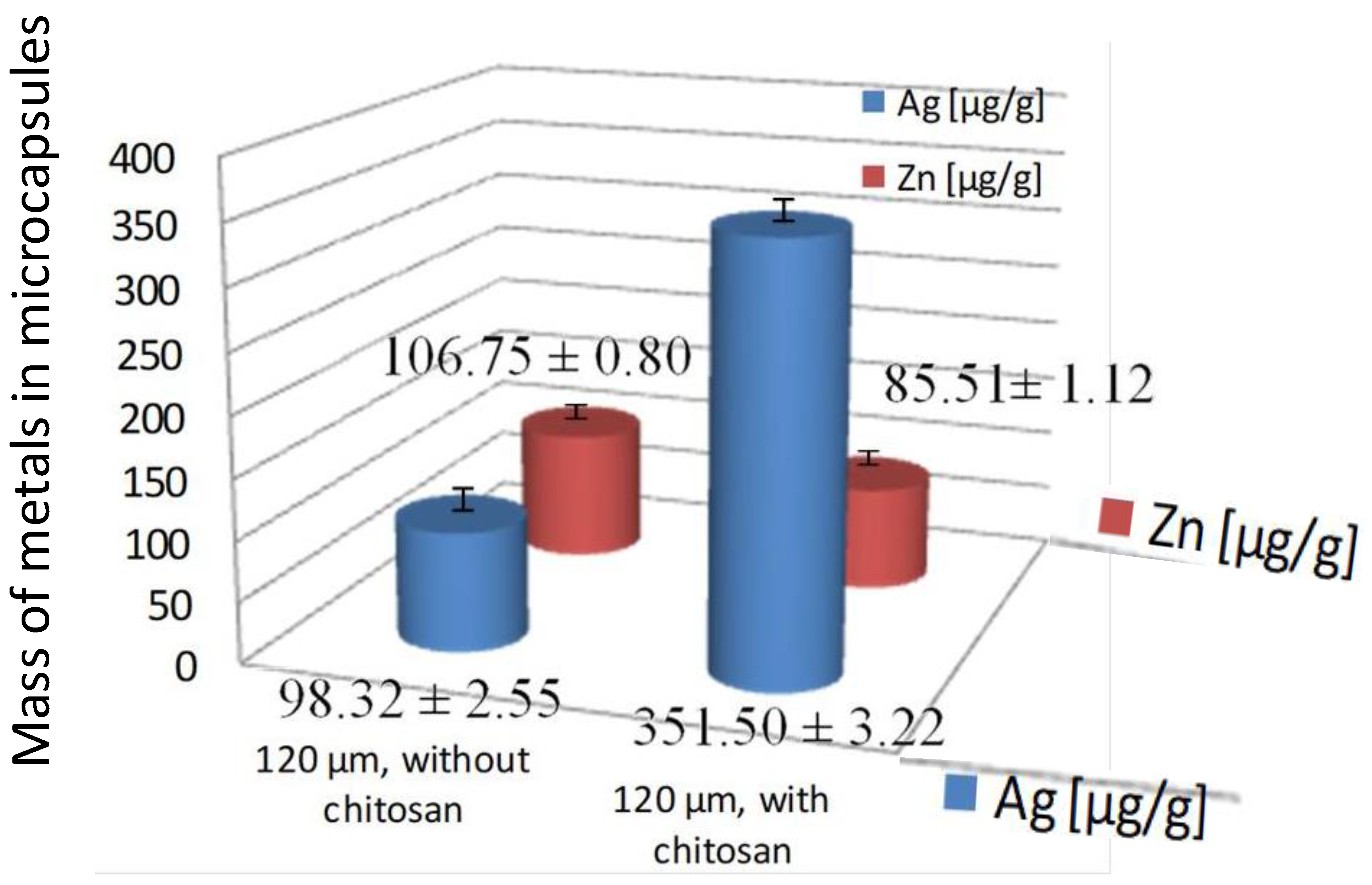
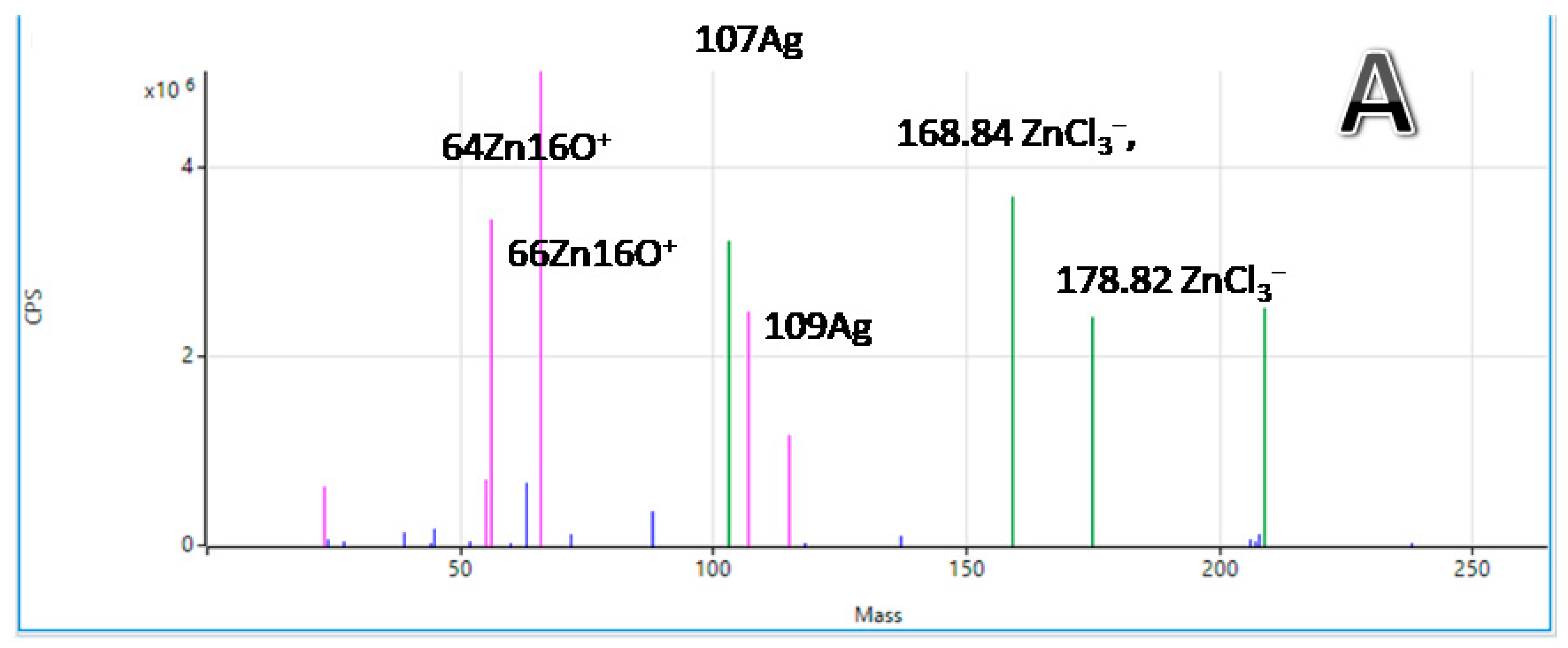
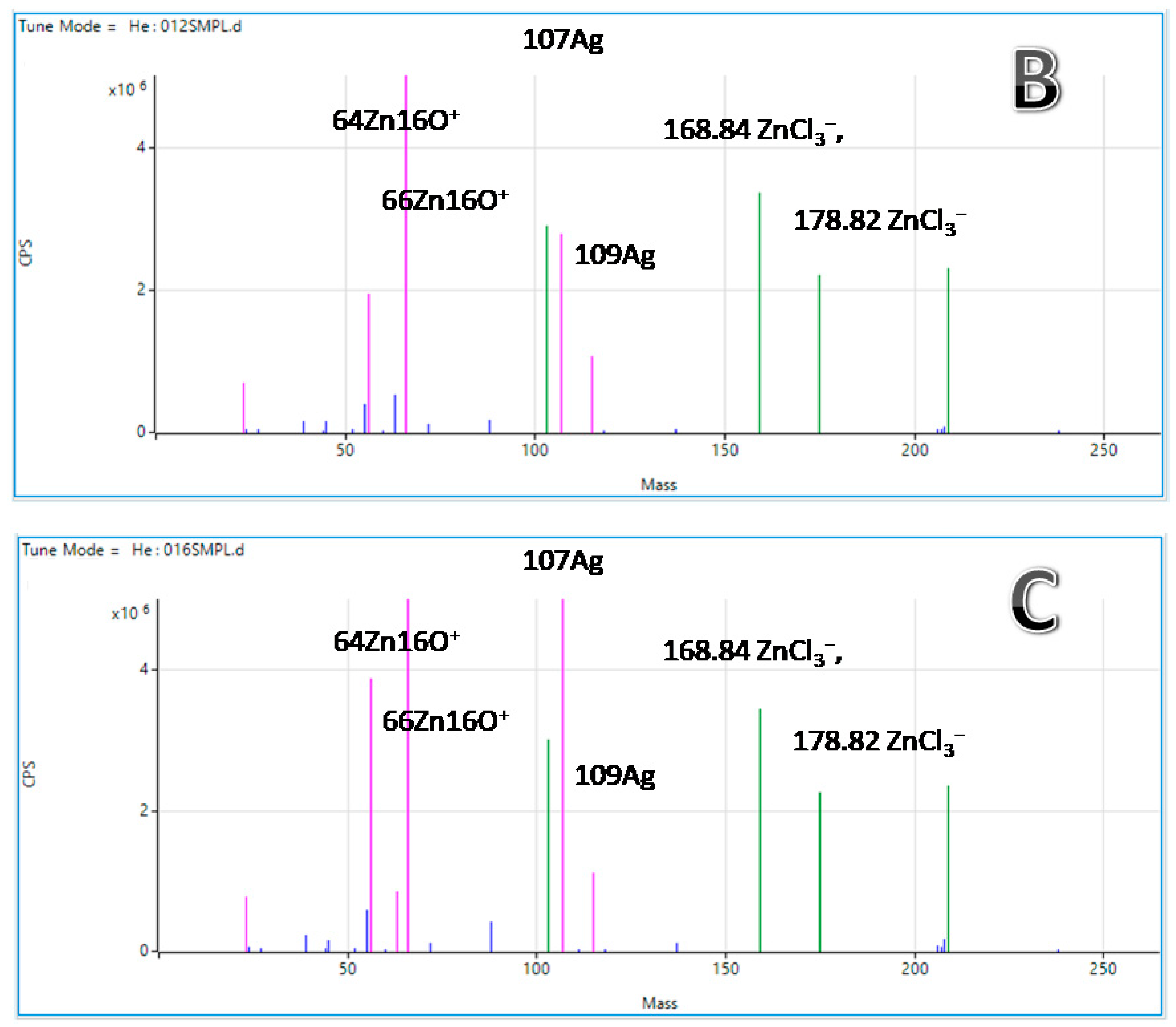
| 120 μm, without Chitosan | 450 μm, without Chitosan | 120 μm, with Chitosan | |||
|---|---|---|---|---|---|
| Element | w [μg/g] | Element | w [μg/g] | Element | w [μg/g] |
| Ag | 98.32 ± 2.55 | Ag | 190.98 ± 4.87 | Ag | 351.50 ± 3.22 |
| Zn | 106.75 ± 0.80 | Zn | 121.35 ± 1.65 | Zn | 85.51 ± 1.12 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rezić, I.; Škoc, M.S.; Majdak, M.; Jurić, S.; Stracenski, K.S.; Vlahoviček-Kahlina, K.; Vinceković, M. ICP-MS Determination of Antimicrobial Metals in Microcapsules. Molecules 2022, 27, 3219. https://doi.org/10.3390/molecules27103219
Rezić I, Škoc MS, Majdak M, Jurić S, Stracenski KS, Vlahoviček-Kahlina K, Vinceković M. ICP-MS Determination of Antimicrobial Metals in Microcapsules. Molecules. 2022; 27(10):3219. https://doi.org/10.3390/molecules27103219
Chicago/Turabian StyleRezić, Iva, Maja Somogyi Škoc, Mislav Majdak, Slaven Jurić, Katarina Sopko Stracenski, Kristina Vlahoviček-Kahlina, and Marko Vinceković. 2022. "ICP-MS Determination of Antimicrobial Metals in Microcapsules" Molecules 27, no. 10: 3219. https://doi.org/10.3390/molecules27103219
APA StyleRezić, I., Škoc, M. S., Majdak, M., Jurić, S., Stracenski, K. S., Vlahoviček-Kahlina, K., & Vinceković, M. (2022). ICP-MS Determination of Antimicrobial Metals in Microcapsules. Molecules, 27(10), 3219. https://doi.org/10.3390/molecules27103219








