Bio-Active Compounds from Teucrium Plants Used in the Traditional Medicine of Kurdistan Region, Iraq
Abstract
1. Introduction
2. Ethnobotanical Data about the Teucrium Species Growing in the Kurdistan Region, Iraq
3. Phytochemistry and Ethnopharmacology of Teucrium Taxa Used in the Traditional Medicine of the Kurdistan Region—Iraq Methods for the Literature Search
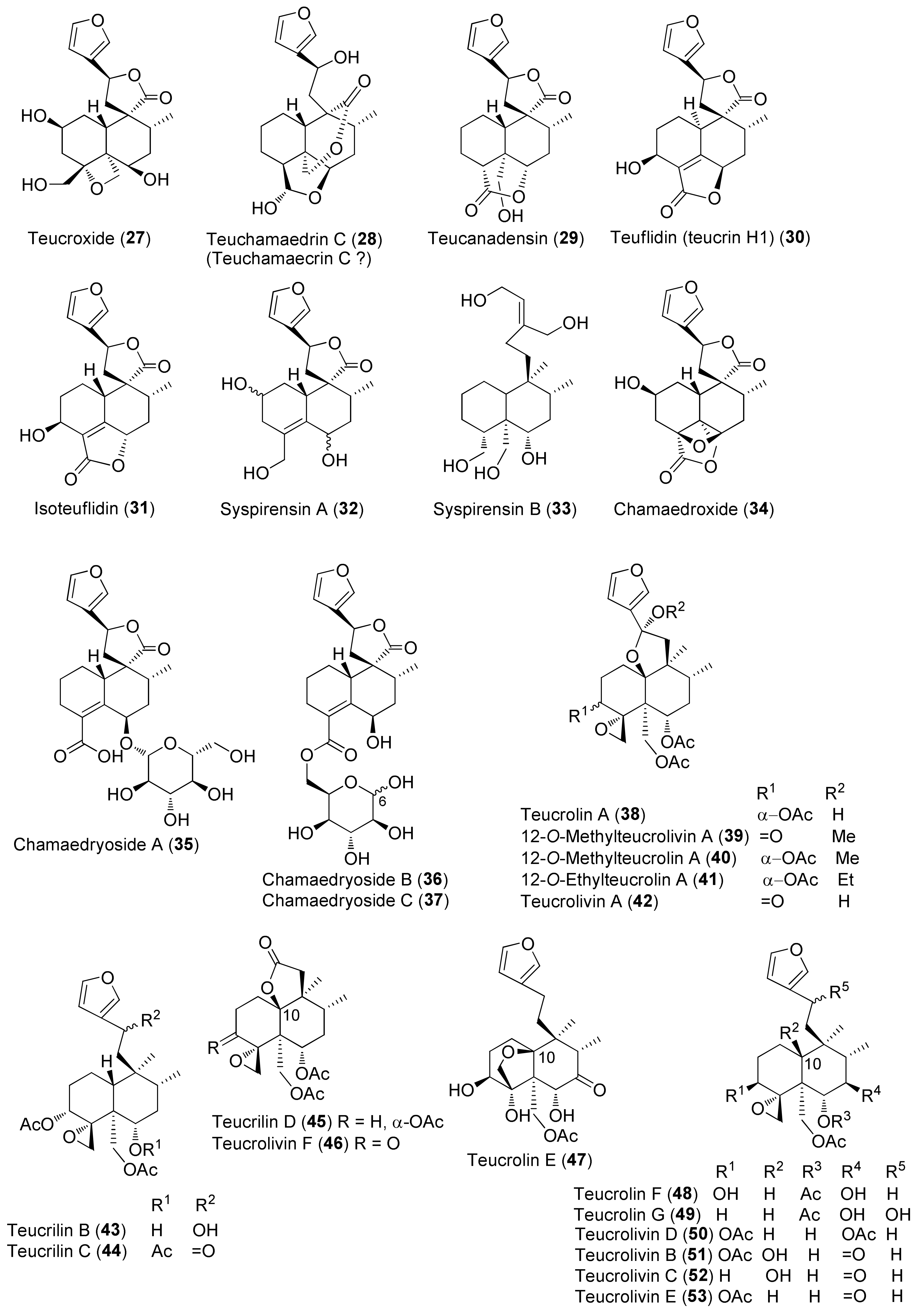
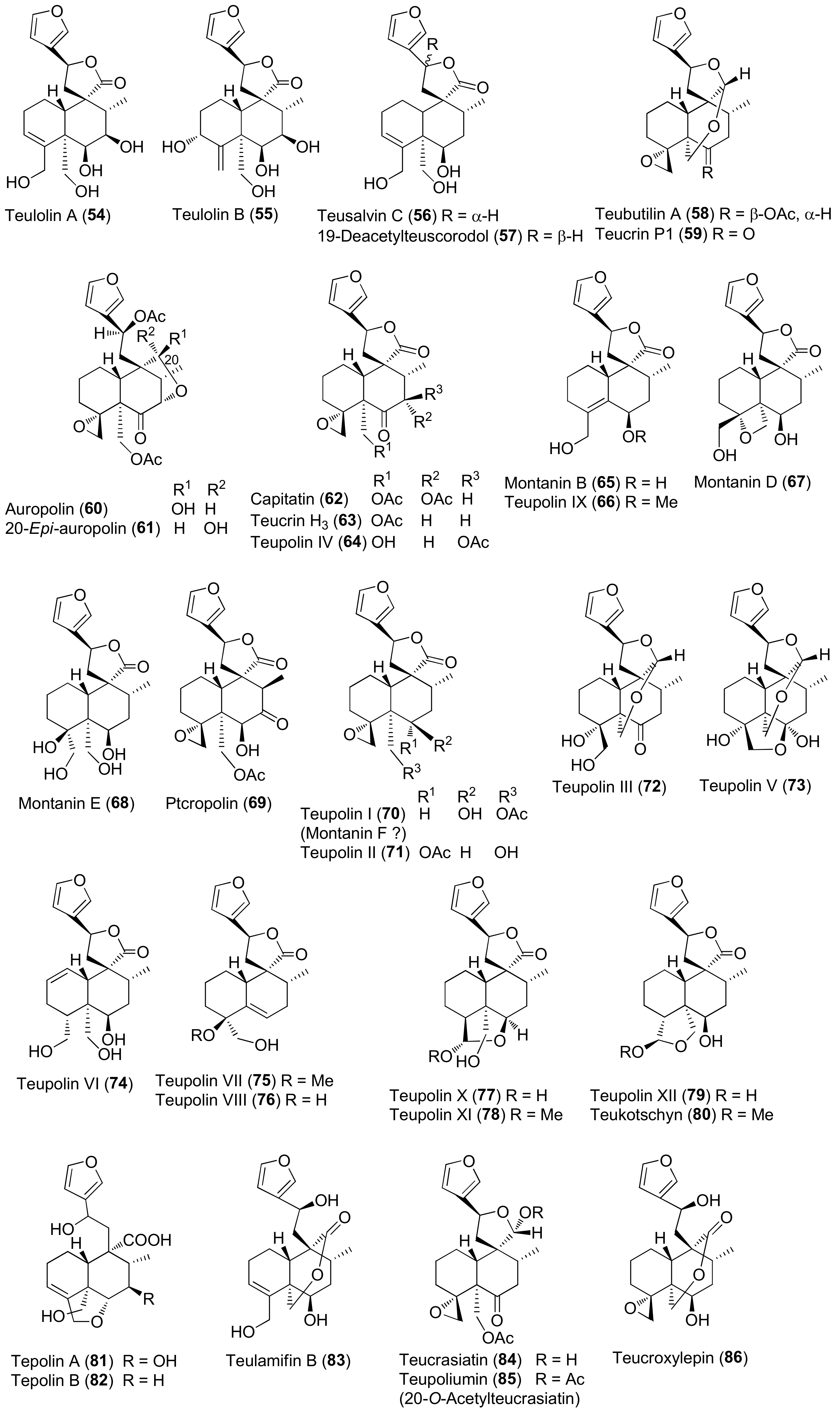
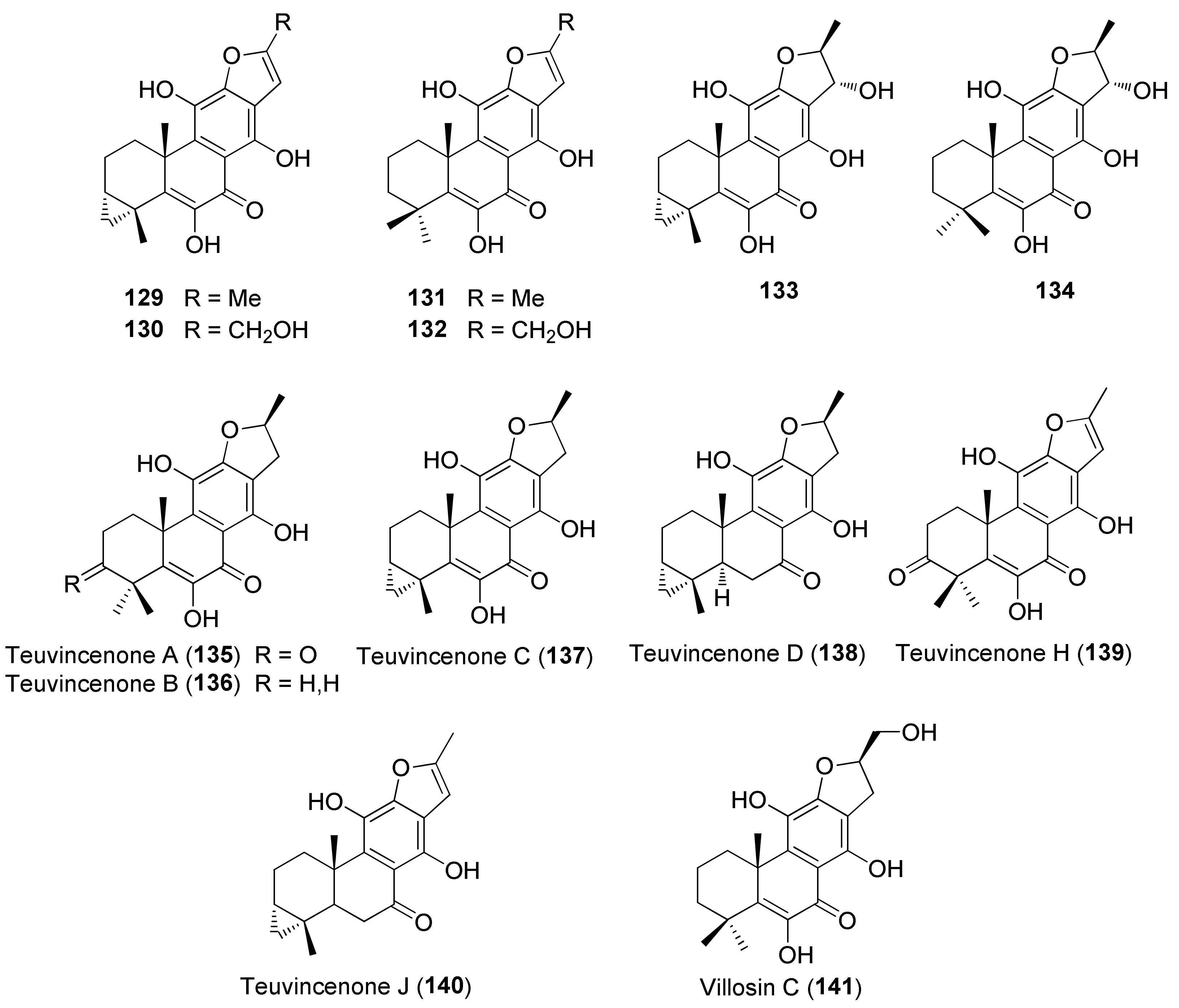
3.1. Phytochemical Aspects
3.2. Bioactivity and Pharmacological Properties
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mathur, S.; Hoskins, C. Drug development: Lessons from nature. Biomed. Rep. 2017, 6, 612–614. [Google Scholar] [CrossRef] [PubMed]
- Mati, E.; de Boer, H. Ethnobotany and trade of medicinal plants in the Qaysari Market, Kurdish Autonomous Region, Iraq. J. Ethnopharmacol. 2011, 133, 490–510. [Google Scholar] [CrossRef] [PubMed]
- Brusotti, G.; Ibrahim, M.F.; Dentamaro, A.; Gilardoni, G.; Tosi, S.; Grisoli, P.; Cesare Dacarro, C.; Guglielminetti, M.L.; Hussain, F.H.S.; Caccialanza, G.; et al. Chemical composition and antimicrobial activity of the volatile fractions from leaves and flowers of the wild Iraqi Kurdish plant Prangos peucedanifolia Fenzl. Chem. Biodivers. 2013, 10, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.A.; Askari, A.A. Ethnobotany of the Hawraman region of Kurdistan Iraq. Harv. Pap. Bot. 2015, 20, 85–89. [Google Scholar] [CrossRef]
- Mahmud, S.A.; Al-Habib, O.A.M.; Bugonl, S.; Clericuzio, M.; Vidari, G. A new ursane-type triterpenoid and other constituents from the leaves of Crataegus azarolus var. aronia. Nat. Prod. Commun. 2016, 11, 1637–1639. [Google Scholar] [CrossRef]
- Amin, H.I.M.; Ibrahim, M.F.; Hussain, F.H.S.; Sardar, A.S.; Vidari, G. Phytochemistry and Ethnopharmacology of Some Medicinal Plants Used in the Kurdistan Region of Iraq. Nat. Prod. Commun. 2016, 11, 291–296. [Google Scholar]
- Abdullah, F.O.; Hussain, F.H.S.; Abdullah, S.S.; Vita-Finzi, P.; Vidari, G. Phytochemistry and Ethnopharmacology of Medicinal Plants Used on Safeen Mountain in the Kurdistan Region of Iraq. Nat. Prod. Commun. 2016, 11, 1923–1927. [Google Scholar]
- Ahmed, H.M. Ethnopharmacobotanical study on the medicinal plants used by herbalists in Sulaymaniyah Province, Kurdistan, Iraq. J. Ethnobiol. Ethnomed. 2016, 12, 1–17. [Google Scholar] [CrossRef]
- Ibrahim, M.F.; Hussain, F.H.S.; Zanoni, G.; Vidari, G. The main constituents of Tulipa systola Stapf. roots and flowers; their antioxidant activities. Nat. Prod. Res. 2017, 31, 2001–2007. [Google Scholar] [CrossRef]
- Abdullah, F.O.; Hussain, F.H.; Clericuzio, M.; Porta, A.; Vidari, G. A new iriDOId dimer and other constituents from the traditional Kurdish plant Pterocephalus nestorianus Nábělek. Chem. Biodivers. 2017, 14, e1600281. [Google Scholar] [CrossRef]
- Amin, H.I.M.; Amin, A.A.; Tosi, S.; Mellerio, G.G.; Hussain, F.H.S.; Picco, A.M.; Vidari, G. Chemical composition and antifungal activity of essential oils from flowers, leaves, rhizomes, and bulbs of the wild Iraqi Kurdish plant Iris persica. Nat. Prod. Commun. 2017, 12, 441–444. [Google Scholar] [CrossRef]
- Abdullah, F.O.; Hussain, F.H.S.; Mannucci, B.; Lappano, R.; Tosi, S.; Maggiolini, M.; Vidari, G. Composition, antifungal and antiproliferative activities of the hydrodistilled oils from leaves and flower heads of Pterocephalus nestorianus Nábělek. Chem. Biodivers. 2017, 14, e1700009. [Google Scholar] [CrossRef]
- Abdullah, F.O.; Hussain, F.S.H.; Cucca, L.; Vidari, G. Phytochemical investigation and antioxidant effects of different solvent extracts of Pterocephalus nestorianus Nab. growing in Kurdistan Region-Iraq. SJUOZ 2018, 6, 21–25. [Google Scholar] [CrossRef]
- Amin, H.I.M.; Hussain, F.H.S.; Maggiolini, M.; Vidari, G. Bioactive constituents from the traditional Kurdish plant Iris persica. Nat. Prod. Commun. 2018, 13, 1934578X1801300907. [Google Scholar] [CrossRef]
- Kheder, D.A.; Al-Habib, O.A.M.; Gilardoni, G.; Vidari, G. Components of volatile fractions from Eucalyptus camaldulensis leaves from Iraqi-Kurdistan and their potent spasmolytic effects. Molecules 2020, 25, 804. [Google Scholar] [CrossRef]
- Hi, M.A.; Hussain, F.H.S.; Gilardoni, G.; Thu, Z.M.; Clericuzio, M.; Vidari, G. Phytochemistry of Verbascum Species Growing in Iraqi Kurdistan and Bioactive Iridoids from the Flowers of Verbascum calvum. Plants 2020, 9, 1066. [Google Scholar] [CrossRef]
- Amin, H.I.M.; Hussain, F.H.S.; Najmaldin, S.K.; Thu, Z.M.; Ibrahim, M.F.; Gilardoni, G.; Vidari, G. Phytochemistry and biological activities of Iris species growing in Iraqi Kurdistan and phenolic constituents of the traditional plant Iris postii. Molecules 2021, 26, 264. [Google Scholar] [CrossRef]
- Thomas, N.V.; Ismail, R.R.; Diyya, A.S.M.; Ghafour, D.D.; Jalal, L.K. Antibacterial effects of the organic crude extracts of freshwater algae of Sulaymaniyah, Kurdistan Region, Iraq. J. Med. Plant Res. 2021, 15, 178–187. [Google Scholar]
- Abdullah, F.O.; Hamahameen, B.A.; Dastan, D. Chemical constituents of the volatile and nonvolatile, cytotoxic and free radical scavenging activities of medicinal plant: Ranunculus millefoliatus and Acanthus dioscoridis. Pol. J. Environ. Stud. 2021, 30, 1981–1989. [Google Scholar] [CrossRef]
- WFO. World Flora Online. An Online Flora of All Known Plants. 2021. Available online: http://www.worldfloraonline.org (accessed on 30 November 2021).
- Govaerts, R.A.; Paton, A.; Harvey, Y.; Navarro, T.; Del Rosario Garcia Pena, M. World Checklist of Lamiaceae; The Board of Trustees of the Royal Botanic Gardens, Kew: London, UK, 2013. [Google Scholar]
- Navarro, T. Systematics and Biogeography of the Genus Teucrium (Lamiaceae). In Teucrium Species: Biology and Applications; Stanković, M., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–38. [Google Scholar] [CrossRef]
- Jakovljević, D.; Stanković, M. Application of Teucrium Species: Current Challenges and Further Perspectives. In Teucrium Species: Biology and Applications; Stanković, M., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 413–432. [Google Scholar] [CrossRef]
- Jarić, S.; Mitrović, M.; Pavlović, P. Ethnobotanical Features of Teucrium Species. In Teucrium Species: Biology and Applications; Stanković, M., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 111–142. [Google Scholar] [CrossRef]
- Ulubelen, A.; Topçu, G.; Sõnmez, U. Chemical and biological evaluation of genus Teucrium. In Studies in Natural Products Chemistry, Vol. 23 (Bioactive Natural Products, Part D); Atta-ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 2000; pp. 591–648. [Google Scholar] [CrossRef]
- Gagliano Candela, R.; Rosselli, S.; Bruno, M.; Fontana, G. A review of the phytochemistry, traditional uses and biological activities of the essential oils of genus Teucrium. Planta Med. 2021, 87, 432–479. [Google Scholar] [CrossRef]
- Stankovic, M.S.; Zlatic, N.-M. Ethnobotany of Teucrium Species; CRC Press: Boca Raton, FL, USA, 2019; pp. 214–231. [Google Scholar]
- Sadeghi, Z.; Yang, J.L.; Venditti, A.; Mahdi Moridi Farimani, M.M. A review of the phytochemistry, ethnopharmacology and biological activities of Teucrium genus (Germander). Nat. Prod. Res. 2022, 1–18. [Google Scholar] [CrossRef]
- Al-Rawi, A. Wild Plants of Iraq with Their Distribution; Iraq Ministry of Agriculture: Baghdad, Iraq, 1968.
- Ridda, T.J.; Daood, W.H. Geographical Distribution of Wild Vascular Plants of Iraq; National Herbarium of Iraq: Baghdad, Iraq, 1982. [Google Scholar]
- Avula, B.; Manyam, R.B.; Bedir, E.; Khan, I.A. HPLC analysis of neo-clerodane diterpenoids from Teucrium chamaedrys. Die Pharmazie 2003, 58, 494–496. [Google Scholar]
- Frezza, C.; Venditti, A.; Matrone, G.; Serafini, I.; Foddai, S.; Bianco, A.; Serafini, M. Iridoid glycosides and polyphenolic compounds from Teucrium chamaedrys L. Nat. Prod. Res. 2018, 32, 1583–1589. [Google Scholar] [CrossRef]
- Prescott, T.A.K.; Veitch, N.C.; Simmonds, M.S.J. Direct inhibition of calcineurin by caffeoyl phenylethanoid glycosides from Teucrium chamaedrys and Nepeta cataria. J. Ethnopharmacol. 2011, 137, 1306–1310. [Google Scholar] [CrossRef]
- Özel, M.Z.; Göğüş, F.; Lewis, A.C. Determination of Teucrium chamaedrys volatiles by using direct thermal desorption-comprehensive two-dimensional gas chromatography-time-of-flight mass spectrometry. J. Chromatogr. A 2006, 1114, 164–169. [Google Scholar] [CrossRef]
- Larrey, D.; Vial, T.; Pauwels, A.; Castot, A.; Biour, M.; David, M.; Michel, H. Hepatitis after germander (Teucrium chamaedrys) administration: Another instance of herbal medicine hepatotoxicity. Ann. Intern. Med. 1992, 117, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Djabou, N.; Lorenzi, V.; Guinoiseau, E.; Andreani, S.; Giuliani, M.C.; Desjobert, J.M.; Bolla, J.M.; Costa, J.; Berti, L.; Luciani, A.; et al. Phytochemical composition of Corsican Teucrium essential oils and antibacterial activity against foodborne or toxi-infectious pathogens. Food Control 2013, 30, 354–363. [Google Scholar] [CrossRef]
- Vlase, L.; Benedec, D.; Hanganu, D.; Damian, G.; Csillag, I.; Sevastre, B.; Mot, A.C.; Silaghi-Dumitrescu, R.; Tilea, I. Evaluation of antioxidant and antimicrobial activities and phenolic profile for Hyssopus officinalis, Ocimum basilicum and Teucrium chamaedrys. Molecules 2014, 19, 5490–5507. [Google Scholar] [CrossRef] [PubMed]
- Pacifico, S.; D’Abrosca, B.; Pascarella, M.T.; Letizia, M.; Uzzo, P.; Piscopo, V.; Fiorentino, A. Antioxidant efficacy of iridoid and phenylethanoid glycosides from the medicinal plant Teucrium chamaedrys in cell-free systems. Bioorg. Med. Chem. 2009, 17, 6173–6179. [Google Scholar] [CrossRef]
- Fiorentino, A.; D’Abrosca, B.; Esposito, A.; Izzo, A.; Pascarella, M.T.; D’Angelo, G.; Monaco, P. Potential allelopathic effect of neo-clerodane diterpenes from Teucrium chamaedrys (L.) on stenomediterranean and weed cosmopolitan species. Biochem. Syst. Ecol. 2009, 37, 349–353. [Google Scholar] [CrossRef]
- Malakov, P.Y.; Papanov, G.Y. Teuchamaedrin C, a neo-clerodane diterpenoid from Teucrium chamaedrys. Phytochemistry 1985, 24, 301–303. [Google Scholar] [CrossRef]
- Savona, G.; García-Alvarezt, M.C.; Rodríguez, B. Dihydroteugin, a neo-clerodane diterpenoid from Teucrium chamaedrys. Phytochemistry 1982, 21, 721–723. [Google Scholar] [CrossRef]
- Lekehal, M.; Pessayre, D.; Lereau, J.M.; Moulis, C.; Fourasté, I.; Fau, D. Hepatotoxicity of the herbal medicine germander: Metabolic activation of its furano diterpenoids by cytochrome P450 3A depletes cytoskeleton-associated protein thiols and forms plasma membrane blebs in rat hepatocytes. Hepatology 1996, 24, 212–218. [Google Scholar] [CrossRef]
- Elmastas, M.; Erenler, R.; Isnac, B.; Aksit, H.; Sen, O.; Genc, N.; Demirtas, I. Isolation and identification of a new neo-clerodane diterpenoid from Teucrium chamaedrys L. Nat. Prod. Res. 2016, 30, 299–304. [Google Scholar] [CrossRef]
- Rodríguez, M.-C.; Barluenga, J.; Pascual, C.; Rodríguez, B.; Savona, G.; Piozzi, F. Neo-clerodane diterpenoids from Teucrium chamaedrys: The identity of teucrin B with dihydroteugin. Phytochemistry 1984, 23, 2960–2961. [Google Scholar] [CrossRef]
- Sundaresan, P.R.; Slavoff, S.A.; Grundel, E.; White, K.D.; Mazzola, E.; Koblenz, D.; Rader, J.I. Isolation and characterisation of selected germander diterpenoids from authenticated Teucrium chamaedrys and T. canadense by HPLC, HPLC-MS and NMR. Phytochem. Anal. 2006, 17, 243–250. [Google Scholar] [CrossRef]
- Fernández-Gadea, F.; Pascual, C.; Rodríguez, B.; Savona, G. 6-epiteucrin A, a neo-clerodane diterpenoid from Teucrium chamaedrys. Phytochemistry 1983, 22, 723–725. [Google Scholar] [CrossRef]
- Ricci, A.; Piccolella, S.; Fiorentino, A.; Pepi, F.; D’Abrosca, B.; Monaco, P. A tandem mass spectrometric investigation of the low-energy collision-activated fragmentation of neo-clerodane diterpenes. Rapid Commun. Mass Spectrom. 2010, 24, 1543–1556. [Google Scholar] [CrossRef]
- Papanov, G.Y.; Malakov, P.Y. Furanoid diterpenes in the bitter fraction of Teucrium chamaedrys L. Zeitschrift für Naturforschung B 1980, 35, 764–766. [Google Scholar] [CrossRef]
- Reinbol’d, A.M.; Popa, D.P. Minor diterpenoids of Teucrium chamaedrys. Chem. Nat. Compd. 1974, 10, 600–606. [Google Scholar] [CrossRef]
- Ulubelen, A.; Topcu, G.; Kaya, Ü. Steroidal compounds from Teucrium chamaedrys subsp. chamaedrys. Phytochemistry 1994, 36, 171–173. [Google Scholar] [CrossRef]
- Popa, D.P.; Reinbol’d, A.M. Bitter substances from Teucrium chamaedrys. Chem. Nat. Compd. 1972, 8, 60–62. [Google Scholar] [CrossRef]
- Bedir, E.; Manyam, R.; Khan, I.A. Neo-clerodane diterpenoids and phenylethanoid glycosides from Teucrium chamaedrys L. Phytochemistry 2003, 63, 977–983. [Google Scholar] [CrossRef]
- Liu, X.; Lee, C.S. Total synthesis of (−)-teucvidin. Org. Lett. 2012, 14, 2886–2889. [Google Scholar] [CrossRef]
- Kouzi, S.A.; McMurtry, R.J.; Nelson, S.D. Hepatotoxicity of germander (Teucrium chamaedrys L.) and one of its onstituent neoclerodane diterpenes teucrin A in the mouse. Chem. Res. Toxicol. 1994, 7, 850–856. [Google Scholar] [CrossRef]
- Çalis, I.; Bedir, E.; Wright, A.D.; Sticher, O. Neoclerodane diterpenoids from Teucrium chamaedrys subsp. syspirense. J. Nat. Prod. 1996, 59, 457–460. [Google Scholar] [CrossRef]
- Eguren, L.; Perales, A.; Fayos, J.; Rodriquez, B.; Savona, G.; Piozzi, F. New neoclerodane diterpenoid containing an oxetane ring isolated from Teucrium chamaedrys. X-ray structure determination. J. Org. Chem. 1982, 47, 4157–4160. [Google Scholar] [CrossRef]
- Fiorentino, A.; D’Abrosca, B.; Ricci, A.; Pacifico, S.; Piccolella, S.; Monaco, P. Structure determination of chamaedryosides A-C, three novel nor-neo-clerodane glucosides from Teucrium chamaedrys, by NMR spectroscopy. Magn. Reson. Chem. 2009, 47, 1007–1012. [Google Scholar] [CrossRef]
- Geçibesler, I.H.; Demirtas, I.; Koldaş, S.; Behçet, L.; Gül, F.; Altun, M. Bioactivity-guided isolation of compounds with antiproliferative activity from Teucrium chamaedrys L. subsp. sinuatum (Celak.) Rech. F. Progr. Nutr. 2019, 21, 458–470. [Google Scholar] [CrossRef]
- Harborne, J.B.; Tomás-Barberán, F.A.; Williams, C.A.; Gil, M.I. A chemotaxonomic study of flavonoids from European Teucrium species. Phytochemistry 1986, 25, 2811–2816. [Google Scholar] [CrossRef]
- Antognoni, F.; Iannello, C.; Mandrone, M.; Scognamiglio, M.; Fiorentino, A.; Giovannini, P.P.; Poli, F. Elicited Teucrium chamaedrys cell cultures produce high amounts of teucrioside, but not the hepatotoxic neo-clerodane diterpenoids. Phytochemistry 2012, 81, 50–59. [Google Scholar] [CrossRef]
- Gross, G.A.; Lahloub, M.F.; Anklin, C.; Schulten, H.R.; Sticher, O. Teucrioside, a phenylpropanoid glycoside from Teucrium chamaedrys. Phytochemistry 1988, 27, 1459–1463. [Google Scholar] [CrossRef]
- Muselli, A.; Desjobert, J.-M.; Paolini, J.; Bernardini, A.-F.; Costa, J.; Rosa, A.; Dessi, M.A. Chemical composition of the essential oils of Teucrium chamaedrys L. from Corsica and Sardinia. J. Essent. Oil Res. 2009, 21, 138–143. [Google Scholar] [CrossRef]
- Bezić, N.; Vuko, E.; Dunkić, V.; Ruščić, M.; Blažević, I.; Burčul, F. Antiphytoviral activity of sesquiterpene-rich essential oils from four Croatian Teucrium species. Molecules 2011, 16, 8119–8129. [Google Scholar] [CrossRef]
- Morteza-Semnani, K.; Akbarzadeh, M.; Rostami, B. The essential oil composition of Teucrium chamaedrys L. from Iran. Flavour Fragr. J. 2005, 20, 544–546. [Google Scholar] [CrossRef]
- Hajdari, A.; Mustafa, B.; Hyseni, L.; Bajrami, A.; Mustafa, G.; Quave, C.L.; Nebija, D. Phytochemical study of eight medicinal plants of the Lamiaceae family traditionally used as tea in the Sharri Mountains region of the Balkans. Sci. World J. 2020, 2020, 4182064. [Google Scholar] [CrossRef][Green Version]
- Kovacevic, N.N.; Lakusic, B.S.; Ristic, M.S. Composition of the essential oils of seven Teucrium species from Serbia and Montenegro. J. Essent. Oil Res. 2001, 13, 163–165. [Google Scholar] [CrossRef]
- Bagci, E.; Yazgın, A.; Hayta, S.; Cakılcıoglu, U. Composition of the essential oil of Teucrium chamaedrys L. (Lamiaceae) from Turkey. J. Med. Plant Res. 2010, 4, 2588–2590. [Google Scholar]
- Ahmadi, L.; Mirza, M.; Shahmir, F. Essential oil of Teucrium melissoides Boiss. et Hausskn. ex Boiss. J. Essent. Oil Res. 2002, 14, 355–356. [Google Scholar] [CrossRef]
- Ajabnoor, M.A.; Al-Yahya, M.A.; Tariq, M.; Jayyab, A.A. Antidiabetic activity of Teucrium oliverianum. Fitoterapia 1984, 55, 227–230. [Google Scholar]
- Al-Yahya, M.A.; El-Feraly, F.S.; Chuck Dunbar, D.; Muhammad, I. Neo-clerodane diterpenoids from Teucrium oliverianum and structure revision of teucrolin E. Phytochemistry 2002, 59, 409–414. [Google Scholar] [CrossRef]
- Bruno, M.; Omar, A.A.; Perales, A.; Piozzi, F.; Rodríguez, B.; Savona, G.; Torre, M.C.D.l. Neo-clerodane diterpenoids from Teucrium oliverianum. Phytochemistry 1991, 30, 275–282. [Google Scholar] [CrossRef]
- Al-Yahya, M.A.; Muhammad, I.; Mirza, H.H.; El-Feraly, F.S.; McPhail, A.T. Neoclerodane diterpenoids and their artifacts from Teucrium oliverianum. J. Nat. Prod. 1993, 56, 830–842. [Google Scholar] [CrossRef]
- Shahat, A.A.; Alsaid, M.S.; Khan, J.A.; Higgins, M.; Dinkova-Kostova, A.T. Chemical constituents and NAD(P)H:Quinone oxidoreductase 1 (NQO1) inducer activity of Teucrium oliverianum Ging. ex Benth. Indian J. Tradit. Knowl. 2016, 15, 232–236. [Google Scholar]
- De La Torre, M.C.; Bruno, M.; Piozzi, F.; Savona, G.; Rodríguez, B.; Omar, A.A. Teucrolivins D–F, neo-clerodane derivatives from Teucrium oliverianum. Phytochemistry 1991, 30, 1603–1606. [Google Scholar] [CrossRef]
- Erbay, M.Ş.; Sarı, A. Plants used in traditional treatment against hemorrhoids in Turkey. Marmara Pharm. J. 2018, 22, 110–132. [Google Scholar] [CrossRef]
- Türkoğlu, S.; Çelik, S.; Türkoğlu, I.; Çakılcıoğlu, U.; Bahsi, M. Determination of the antioxidant properties of ethanol and water extracts from different parts of Teucrium parviflorum Schreber. Afr. J. Biotechnol. 2010, 9, 6797–6805. [Google Scholar] [CrossRef]
- Bagci, E.; Hayta, S.; Yazgin, A.; Dogan, G. Composition of the essential oil of Teucrium parviflorum L. (Lamiaceae) from Turkey. J. Med. Plant Res. 2011, 5, 3457–3460. [Google Scholar] [CrossRef]
- Tariq, M.; Ageel, A.M.; al-Yahya, M.A.; Mossa, J.S.; al-Said, M.S. Anti-inflammatory activity of Teucrium polium. Int. J. Tissue React. 1989, 11, 185–188. [Google Scholar] [PubMed]
- Bahramikia, S.; Yazdanparast, R. Phytochemistry and medicinal properties of Teucrium polium L. (Lamiaceae). Phytother. Res. 2012, 26, 1581–1593. [Google Scholar] [CrossRef] [PubMed]
- Bedir, E.; Tasdemir, D.; Çalis, I.; Zerbe, O.; Otto, S. Neo-clerodane diterpenoids from Teucrium polium. Phytochemistry 1999, 51, 921–925. [Google Scholar] [CrossRef]
- Kamel, A. 7-epi-Eudesmanes from Teucrium polium. J. Nat. Prod. 1995, 58, 428–431. [Google Scholar] [CrossRef]
- Ljubuncic, P.; Dakwar, S.; Portnaya, I.; Cogan, U.; Azaizeh, H.; Bomzon, A. Aqueous extracts of Teucrium polium possess remarkable antioxidant activity in vitro. Evid.-Based Complementary Altern. Med. 2006, 3, 436479. [Google Scholar] [CrossRef]
- Noumi, E.; Snoussi, M.; Anouar, E.H.; Alreshidi, M.; Veettil, V.N.; Elkahoui, S.; Adnan, M.; Patel, M.; Kadri, A.; Aouadi, K.; et al. HR-LCMS-based metabolite profiling, antioxidant, and anticancer properties of Teucrium polium L. methanolic extract: Computational and in vitro study. Antioxidants 2020, 9, 1089. [Google Scholar] [CrossRef]
- Ardestani, A.; Yazdanparast, R. Inhibitory effects of ethyl acetate extract of Teucrium polium on in vitro protein glycoxidation. Food Chem. Toxicol. 2007, 45, 2402–2411. [Google Scholar] [CrossRef]
- De Marino, S.; Festa, C.; Zollo, F.; Incollingo, F.; Raimo, G.; Evangelista, G.; Iorizzi, M. Antioxidant activity of phenolic and phenylethanoid glycosides from Teucrium polium L. Food Chem. 2012, 133, 21–28. [Google Scholar] [CrossRef]
- Sharififar, F.; Dehghn-Nudeh, G.; Mirtajaldini, M. Major flavonoids with antioxidant activity from Teucrium polium L. Food Chem. 2009, 112, 885–888. [Google Scholar] [CrossRef]
- Goulas, V.; Gomez-Caravaca, A.M.; Exarchou, V.; Gerothanassis, I.P.; Segura-Carretero, A.; Gutiérrez, A.F. Exploring the antioxidant potential of Teucrium polium extracts by HPLC–SPE–NMR and on-line radical-scavenging activity detection. LWT-Food Sci. Technol. 2012, 46, 104–109. [Google Scholar] [CrossRef]
- Elmasri, W.A.; Hegazy, M.-E.F.; Mechref, Y.; Paré, P.W. Structure-antioxidant and anti-tumor activity of Teucrium polium phytochemicals. Phytochem. Lett. 2016, 15, 81–87. [Google Scholar] [CrossRef]
- Esmaeili, M.A.; Zohari, F.; Sadeghi, H. Antioxidant and protective effects of major flavonoids from Teucrium polium on β-cell destruction in a model of streptozotocin-induced diabetes. Planta Med. 2009, 75, 1418–1420. [Google Scholar] [CrossRef]
- Fiorentino, A.; D’Abrosca, B.; Pacifico, S.; Scognamiglio, M.; D’Angelo, G.; Monaco, P. abeo-Abietanes from Teucrium polium roots as protective factors against oxidative stress. Bioorg. Med. Chem. 2010, 18, 8530–8536. [Google Scholar] [CrossRef] [PubMed]
- Pacifico, S.; D’Abrosca, B.; Scognamiglio, M.; D’Angelo, G.; Gallicchio, M.; Galasso, S.; Monaco, P.; Fiorentino, A. NMR-based metabolic profiling and in vitro antioxidant and hepatotoxic assessment of partially purified fractions from Golden germander (Teucrium polium L.) methanolic extract. Food Chem. 2012, 135, 1957–1967. [Google Scholar] [CrossRef] [PubMed]
- D’Abrosca, B.; Pacifico, S.; Scognamiglio, M.; D’Angelo, G.; Galasso, S.; Monaco, P.; Fiorentino, A. A new acylated flavone glycoside with antioxidant and radical scavenging activities from Teucrium polium leaves. Nat. Prod. Res. 2013, 27, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Autore, G.; Capasso, F.; De Fusco, R.; Fasulo, M.P.; Lembo, M.; Mascolo, N.; Menghini, A. Antipyretic and antibacterial actions of Teucrium polium (L.). Pharmacol. Res. Commun. 1984, 16, 21–29. [Google Scholar] [CrossRef]
- Darabpour, E.; Motamedi, H.; Nejad, S.M.S. Antimicrobial properties of Teucrium polium against some clinical pathogens. Asian Pac. J. Trop. Med. 2010, 3, 124–127. [Google Scholar] [CrossRef]
- Elmasri, W.A.; Hegazy, M.-E.F.; Aziz, M.; Koksal, E.; Amor, W.; Mechref, Y.; Hamood, A.N.; Cordes, D.B.; Paré, P.W. Biofilm blocking sesquiterpenes from Teucrium polium. Phytochemistry 2014, 103, 107–113. [Google Scholar] [CrossRef]
- BozovElmasri, W.A.; Yang, T.; Tran, P.; Hegazy, M.-E.F.; Hamood, A.N.; Mechref, Y.; Paré, P.W. Teucrium polium phenylethanol and iridoid glycoside characterization and flavonoid inhibition of biofilm-forming Staphylococcus aureus. J. Nat. Prod. 2015, 78, 2–9. [Google Scholar] [CrossRef]
- Oganesyan, G.B.; Galstyan, A.M.; Mnatsakanyan, V.A.; Shashkov, A.S.; Agababyan, P.V. Phenylpropanoid glycosides of Teucrium polium. Chem. Nat. Compd. 1991, 27, 556–559. [Google Scholar] [CrossRef]
- Elmasri, W.A.; Hegazy, M.-E.F.; Mechref, Y.; Paré, P.W. Cytotoxic saponin poliusaposide from Teucrium polium. RSC Adv. 2015, 5, 27126–27133. [Google Scholar] [CrossRef]
- Nematollahi-Mahani, S.N.; Rezazadeh-Kermani, M.; Mehrabani, M.; Nakhaee, N. Cytotoxic effects of Teucrium polium. on some established cell lines. Pharm. Biol. 2007, 45, 295–298. [Google Scholar] [CrossRef]
- Fiorentino, A.; D’Abrosca, B.; Pacifico, S.; Scognamiglio, M.; D’Angelo, G.; Gallicchio, M.; Chambery, A.; Monaco, P. Structure elucidation and hepatotoxicity evaluation against HepG2 human cells of neo-clerodane diterpenes from Teucrium polium L. Phytochemistry 2011, 72, 2037–2044. [Google Scholar] [CrossRef]
- Twaij, H.A.A.; Albadr, A.A.; Abul-Khail, A. Anti-ulcer activity of Teucrium polium. Int. J. Crude Drug Res. 1987, 25, 125–128. [Google Scholar] [CrossRef]
- Abdollahi, M.; Karimpour, H.; Monsef-Esfehani, H.R. Antinociceptive effects of Teucrium polium L. total extract and essential oil in mouse writhing test. Pharmacol. Res. 2003, 48, 31–35. [Google Scholar] [CrossRef]
- Bruno, M.; Maggio, A.M.; Piozzi, F.; Puech, S.; Rosselli, S.; Simmonds, M.S.J. Neoclerodane diterpenoids from Teucrium polium subsp. polium and their antifeedant activity. Biochem. Syst. Ecol. 2003, 31, 1051–1056. [Google Scholar] [CrossRef]
- Gharaibeh, M.N.; Elayan, H.H.; Salhab, A.S. Hypoglycemic effects of Teucrium polium. J. Ethnopharmacol. 1988, 24, 93–99. [Google Scholar] [CrossRef]
- Esmaeili, M.A.; Yazdanparast, R. Hypoglycaemic effect of Teucrium polium: Studies with rat pancreatic islets. J. Ethnopharmacol. 2004, 95, 27–30. [Google Scholar] [CrossRef]
- Rasekh, H.R.; Khoshnood-Mansourkhani, M.J.; Kamalinejad, M. Hypolipidemic effects of Teucrium polium in rats. Fitoterapia 2001, 72, 937–939. [Google Scholar] [CrossRef]
- Milošević-Djordjević, O.; Radović Jakovljević, M.; Marković, A.; Stanković, M.; Ćirić, A.; Marinković, D.; Grujičić, D. Polyphenolic contents of Teucrium polium L. and Teucrium scordium L. associated with their protective effects against MMC-induced chromosomal damage in cultured human peripheral blood lymphocytes. Turk. J. Biol. 2018, 42, 152–162. [Google Scholar] [CrossRef]
- Malakov, P.Y.; Papanov, G.Y.; Ziesche, J. Teupolin III, a furanoid diterpene from Teucrium polium. Phytochemistry 1982, 21, 2597–2598. [Google Scholar] [CrossRef]
- Malakov, P.Y.; Papanov, G.Y.; Mollov, N.M. Furanoid diterpenes in the bitter fraction of Teucrium polium L. Zeitschrift für Naturforschung B 1979, 34, 1570–1572. [Google Scholar] [CrossRef]
- Malakov, P.Y.; Papanov, G.Y. Furanoid diterpenes from Teucrium polium. Phytochemistry 1983, 22, 2791–2793. [Google Scholar] [CrossRef]
- Brieskorn, C.H.; Pfeuffer, T. Labiate bitter principles: Picropoline and similar diterpenoids from poleigamander. Chem. Ber. 1967, 100, 1998–2010. [Google Scholar] [CrossRef]
- Galstyan, A.M.; Shashkov, A.S.; Oganesyan, G.B.; Mnatsakanyan, V.A.; Serebryakov, É.P. Structures of two new diterpenoids from Teucrium polium. Chem. Nat. Compd. 1992, 28, 439–443. [Google Scholar] [CrossRef]
- Malakov, P.Y.; Boneva, I.M.; Papanov, G.Y.; Spassov, S.L. Teulamifin B, a neo-clerodane diterpenoid from Teucrium lamiifolium and T. polium. Phytochemistry 1988, 27, 1141–1143. [Google Scholar] [CrossRef]
- Venditti, A.; Frezza, C.; Trancanella, E.; Zadeh, S.M.M.; Foddai, S.; Sciubba, F.; Delfini, M.; Serafini, M.; Bianco, A. A new natural neo-clerodane from Teucrium polium L. collected in Northern Iran. Ind. Crops Prod. 2017, 97, 632–638. [Google Scholar] [CrossRef]
- Rizk, A.M.; Hammouda, F.M.; Rimpler, H.; Kamel, A. Iridoids and flavonoids of Teucrium polium herb1. Planta Med. 1986, 52, 87–88. [Google Scholar] [CrossRef]
- Verykokidou-Vitsaropoulou, E.; Vajias, C. Methylated flavones from Teucrium polium. Planta Med. 1986, 52, 401–402. [Google Scholar] [CrossRef]
- Al-Shalabi, E.; Alkhaldi, M.; Sunoqrot, S. Development and evaluation of polymeric nanocapsules for cirsiliol isolated from Jordanian Teucrium polium L. as a potential anticancer nanomedicine. J. Drug Deliv. Sci. Technol. 2020, 56, 101544. [Google Scholar] [CrossRef]
- Kawashty, S.A.; Gamal El-Din, E.M.; Saleh, N.A.M. The flavonoid chemosystematics of two Teucrium species from Southern Sinai, Egypt. Biochem. Syst. Ecol. 1999, 27, 657–666. [Google Scholar] [CrossRef]
- Hachicha, S.F.; Barrek, S.; Skanji, T.; Zarrouk, H.; Ghrabi, Z.G. Fatty acid, tocopherol, and sterol content of three Teucrium species from Tunisia. Chem. Nat. Comp. 2009, 45, 304–308. [Google Scholar] [CrossRef]
- Capasso, F.; Cerri, R.; Morrica, P.; Senatore, F. Chemical composition and anti-inflammatory activity of an alcoholic extract of Teucrium polium L. Boll. Soc. Ital. Bio. Sper. 1983, 59, 1639–1643. [Google Scholar]
- Vokou, D.; Bessiere, J.-M. Volatile constituents of Teucrium polium. J. Nat. Prod. 1985, 48, 498–499. [Google Scholar] [CrossRef]
- Wassel, G.M.; Ahmed, S.S. Essential oil of Teucrium polium L. Die Pharmazie 1974, 29, 351–352. [Google Scholar] [PubMed]
- Sayyad, R.; Farahmandfar, R. Influence of Teucrium polium L. essential oil on the oxidative stability of canola oil during storage. J. Food Sci. Technol. 2017, 54, 3073–3081. [Google Scholar] [CrossRef] [PubMed]
- Aburjai, T.; Hudaib, M.; Cavrini, V. Composition of the essential oil from Jordanian germander (Teucrium polium L.). J. Essent. Oil Res. 2006, 18, 97–99. [Google Scholar] [CrossRef]
- Eikani, M.H.; Goodarznia, I.; Mirza, M. Comparison between the essential oil and supercritical carbon dioxide extract of Teucrium polium L. J. Essent. Oil Res. 1999, 11, 470–472. [Google Scholar] [CrossRef]
- Sabzeghabaie, A.; Asgarpanah, J. Essential oil composition of Teucrium polium L. fruits. J. Essent. Oil Res. 2016, 28, 77–80. [Google Scholar] [CrossRef]
- Sadeghi, H.; Jamalpoor, S.; Shirzadi, M.H. Variability in essential oil of Teucrium polium L. of different latitudinal populations. Ind. Crops Prod. 2014, 54, 130–134. [Google Scholar] [CrossRef]
- Farahbakhsh, J.; Najafian, S.; Hosseinifarahi, M.; Gholipour, S. The effect of time and temperature on shelf life of essential oil composition of Teucrium polium L. Nat. Prod. Res. 2020, 36, 424–428. [Google Scholar] [CrossRef]
- Çakir, A.; Duru, M.E.; Harmandar, M.; Ciriminna, R.; Passannanti, S. Volatile constituents of Teucrium polium L. from Turkey. J. Essent. Oil Res. 1998, 10, 113–115. [Google Scholar] [CrossRef]
- Ashnagar, A.; Naseri, N.G.; Foroozanfar, S. Isolation and identification of the major chemical components found in the upper parts of Teucrium polium plants grown in Khuzestan province of Iran. Chin. J. Chem. 2007, 25, 1171–1173. [Google Scholar] [CrossRef]
- Bozov, P.I.; Penchev, P.N.; Girova, T.D.; Gochev, V.K. Diterpenoid constituents of Teucrium scordium L. subsp. scordioides (Shreb.) Maire Et Petitmengin. Nat. Prod. Commun. 2020, 15, 1934578X20959525. [Google Scholar] [CrossRef]
- Radulović, N.; Dekić, M.; Joksović, M.; Vukićević, R. Chemotaxonomy of Serbian Teucrium species inferred from essential oil chemical composition: The case of Teucrium scordium L. ssp. scordioides. Chem. Biodivers. 2012, 9, 106–122. [Google Scholar] [CrossRef]
- Gagliano Candela, R.; Ilardi, V.; Badalamenti, N.; Bruno, M.; Rosselli, S.; Maggi, F. Essential oil compositions of Teucrium fruticans, T. scordium subsp. scordioides and T. siculum growing in Sicily and Malta. Nat. Prod. Res. 2020, 35, 3460–3469. [Google Scholar] [CrossRef]
- Piozzi, F.; Bruno, M.; Rosselli, S.; Maggio, A. Advances on the chemistry of furano-diterpenoids from Teucrium genus. Heterocycles 2005, 65, 1221–1234. [Google Scholar] [CrossRef]
- Simmonds, M.S.J.; Blaney, W.M.; Ley, S.V.; Savona, G.; Bruno, M.; Rodriguez, B. The antifeedant activity of clerodane diterpenoids from Teucrium. Phytochemistry 1989, 28, 1069–1071. [Google Scholar] [CrossRef]
- Frezza, C.; de Vita, D.; Toniolo, C.; Ventrone, A.; Tomassini, L.; Foddai, S.; Nicoletti, M.; Guiso, M.; Bianco, A.; Serafini, M. Harpagide: Occurrence in plants and biological activities—A review. Fitoterapia 2020, 147, 104764. [Google Scholar] [CrossRef]
- Breschi, M.C.; Martinotti, E.; Catalano, S.; Flamini, G.; Morelli, I.; Pagni, A.M. Vasoconstrictor activity of 8-O-acetylharpagide from Ajuga reptans. J. Nat. Prod. 1992, 55, 1145–1148. [Google Scholar] [CrossRef]
- Herbert, J.M.; Maffrand, J.P.; Taoubi, K.; Augereau, J.M.; Fouraste, I.; Gleye, J.; Shi, C.; Hui, N.; Liu, Y.; Ling, M.; et al. Verbascoside isolated from Lantana camara, an inhibitor of protein kinase C. J. Nat. Prod. 1991, 54, 1595–1600. [Google Scholar] [CrossRef]
- Yang, D.; Li, J.; Liang, C.; Tian, L.; Shi, C.; Hui, N.; Liu, Y.; Ling, M.; Xin, L.; Wan, M.; et al. Pacifico. Syringa microphylla Diels: A comprehensive review of its phytochemical, pharmacological, pharmacokinetic, and toxicological characteristics and an investigation into its potential health benefits. Phytomedicine 2021, 93, 153770. [Google Scholar] [CrossRef]
- Shawky, E. Prediction of potential cancer-related molecular targets of North African plants constituents using network pharmacology-based analysis. J. Ethnopharmacol. 2019, 238, 111826. [Google Scholar] [CrossRef]
- Loeper, J.; Descatoire, V.; Letteron, P.; Moulis, C.; Degott, C.; Dansette, P.; Fau, D.; Pessayre, D. Hepatotoxicity of germander in mice. Gastroenterology 1994, 106, 464–472. [Google Scholar] [CrossRef]
- Chitturi, S.; Farrell, G.C. Hepatotoxic slimming aids and other herbal hepatotoxins. J. Gastroenterol. Hepatol. 2008, 23, 366–373. [Google Scholar] [CrossRef]
- Druckova, A.; Mernaugh, R.L.; Ham, A.-J.L.; Marnett, L.J. Identification of the protein targets of the reactive metabolite of teucrin A in vivo in the rat. Chem. Res. Toxicol. 2007, 20, 1393–1408. [Google Scholar] [CrossRef]
- Polymeros, D.; Kamberoglou, D.; Tzias, V. Acute cholestatic hepatitis caused by Teucrium polium (golden germander) with transient appearance of antimitochondrial antibody. J. Clin. Gastroenterol. 2002, 34, 100–101. [Google Scholar] [CrossRef]
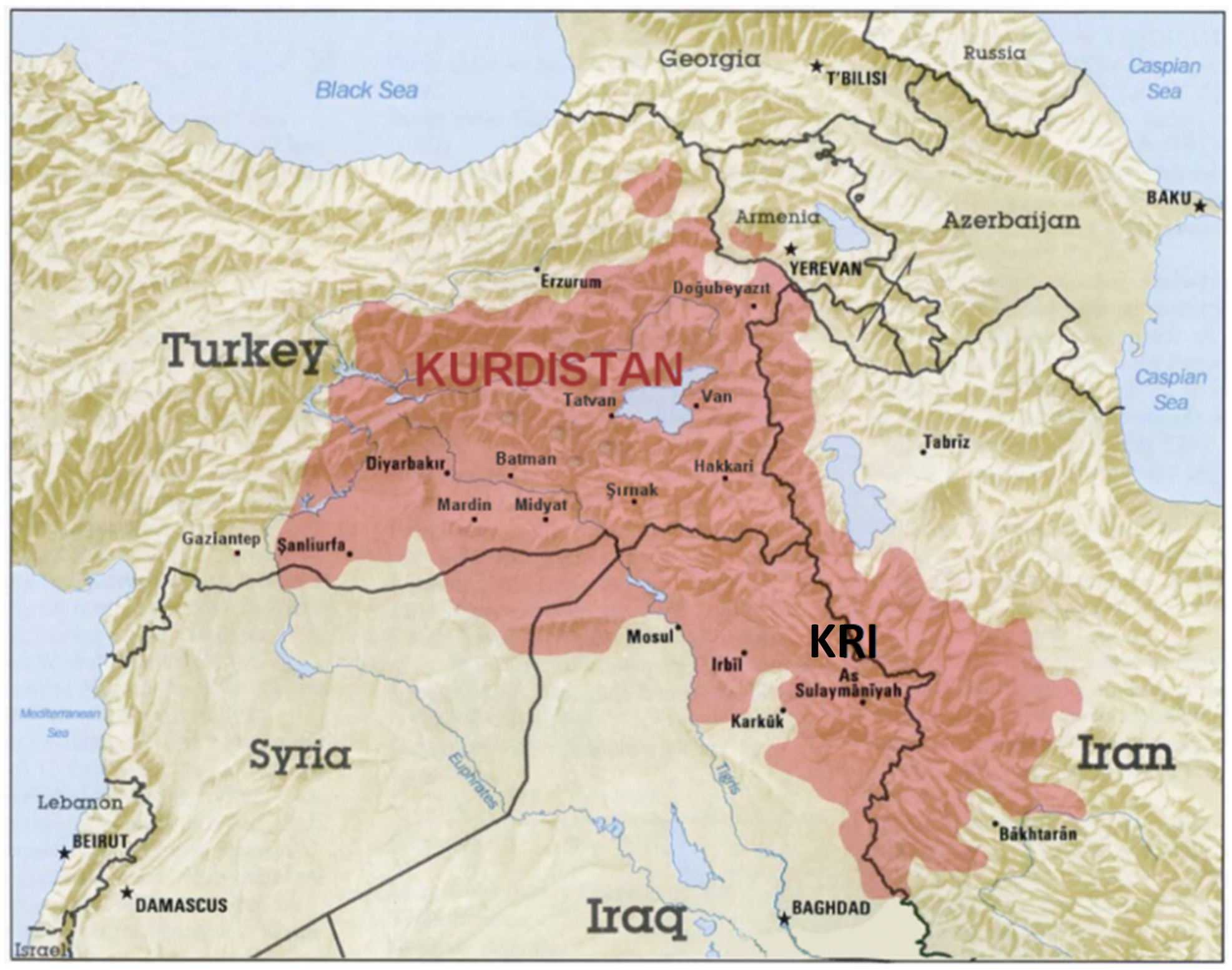







| Botanical Name | Traditional Uses in the Kurdistan Region, Iraq | Growth Places (KRI Districts) |
|---|---|---|
| T. chamaedrys L. | Eaten as a digestive | Baradost, Mosul, Duhok, Gara, Sirsang, Sharanish, Zawita, Khantur, Atrush, Suwara Tuka, Ser Amadiyah, Qara Dagh |
| T. melissoides Boiss. & Hausskn. ex Boiss. | To treat abdominal diseases | Qandil, Pushtashan, Rowanduz, Avroman, Zawita |
| T. oliverianum Ging. ex Benth. | Antidiabetic remedy | Kirkuk, Hamrin |
| T. parviflorum Schreb. | To treat jaundice, liver disorders, stomachache and to reduce cholesterol level in blood | Amadiyah, Sulaimani, Rowandruz |
| T. polium L. | Antirheumatic and to treat abdominal pain | Arbil, Safin, Shaqlawa, Kirkuk, Jarmo, Mosul, Sharanish, Suwara Tuka, Zakho, Khantur, Gara, Atrush, Sirsang |
| T. rigidum Benth. | To treat abdominal problems | Darband-i Bazian |
| Teucrium scordium subsp. scordioides (Schreb.) Arcang. (synonym T. scordioides Schreb.) | Anti-inflammatory | Safin, Baradost, Zawita, Gara |
| Taxon | Traditional Uses | Biological Activities | Secondary Metabolites |
|---|---|---|---|
| Teucrium chamaedrys L. | Astringent, antirheumatic, digestive, antispasmodic, anti-inflammatory, diuretic, diaphoretic, tonic, to treat wounds, fever, coughs, asthma, dyspepsia, anorexia, nasal catarrh, chronic bronchitis, gout, rheumatoid arthritis, abscesses, conjunctivitis, intermittent fever, uterine infections, to reduce body weight [29,31,32,33,34]. | Hepatotoxicity [35], antimicrobial [36,37], antioxidant [37,38], phytotoxic [39] activities. | Iridoids 1 [32] and 2 [32]. Neo-clerodane Diterpenoids 10 [32], 11 [40], 12 [41], 13 [40,41,42,43,44,45], 14 [39,46,47,48,49], 15 [46,48], 16 [47], 13 [31,40,44,45,49,50,51,52], 18 [31,45,52,53], 19 [31,39,41,43,45,47,48,51,52,54], 20 [31,39,45,46,47,48,52], teucrin C [51], teucrin D [51], 21 [46], 22 [39,47,49], 23 [31,39,45,47,49,52], 24 [39], 26 [43], 27 [43], 28 [40], 29 [31,39,52], 30 [31,39,45,46,47], 31 [31,45,52], 32 [43,55], 33 [55], 34 [39,56], 35 [47,57], 36 [47,57], 37 [57]. Flavonoids 87 [32], 88 [58], 89 [58], 90 [50], 91 [40], 98 [59], 99 [50]. Verbascoside Derivatives 107 [33,43,52,55,60,61], 108 [52] 109 [52], 111 [32], 112 [32], 113 [32], 114 [32,43,51]. Phenolic Compounds (E)-p-Coumaroyl-O-β-D-glucoside (118) [58]. Triterpenoids and Steroids 24α-Ethylcholesta-5,25-dien-3β-ol, β-sitosterol, α-amyrin, ursolic acid, 3β-hydroxystigmast-24(241),25-dien-242-al, 3β-hydroxy-24α-ethylcholesta-5,25-dien-7-one [50]. Essential Oil main components (%) from aerial parts collected in Corsica (France) 166 (29.0), 167 (19.4), 168 (6.8), 169 (5.4), 170 (4.4), 171 (3.2) [62]. 166 (33.9), 167 (18.5), 168 (7.5), 170 (5.1), 169 (4.6), 171 (3.1) [36]. Essential Oil main components (%) from aerial parts collected in Croatia 166 (47.6), 167 (29.0), 172 (5.7), 171 (4.5) [63]. Essential Oil main components (%) from aerial parts collected in Iran 167 (16.5), 173 (12.2), 166 (10.5), 174 (9.1), 169 (7.4), 175 (4.8), 176 (3.8), 177 (3.5) [64]. Essential Oil main components (%) from aerial parts collected in Kossovo 167 (24.1), hexadecanoic acid (12.1), 169 (7.0), linoleic acid (6.0), 166 (4.0), 178 (3.3), n-docosane (2.9) [65]. Essential Oil main components (%) from aerial parts collected in Sardinia (Italy) 166 (27.4), 167 (13.5), 171 (12.3), 168 (6.5), 174 (4.4), 175 (3.4), 176 (3.0) [62]. Essential Oil main components (%) from aerial parts collected in Serbia-Montenegro 166 (26.9), 167 (22.8), 168 (6.7), 171 (5.5), 174 (5.3), 3-octanol (3.7), 169 (3.1) [66]. Essential Oil main components (%) from leaves collected in Turkey 175 (13.1), 167 (9.5), 174 (8.9), 179 (8.0), 180 (7.8), 181 (7.4), 182 (6.4), heptacosane (4.8) [34]. Essential Oil main components (%) from aerial parts collected in Turkey 167 (32.1), 168 (14.2), 169 (13.1), 183 (6.7), 170 (4.3), 184 (4.1) [67]. |
| Teucrium melissoides Boiss. & Hausskn. ex Boiss. | No traditional uses are reported in addition to the use indicated in Table 1. | No data have been reported | Essential Oil main components (%) from aerial parts collected in Iran 174 (27.7), 175 (16.4), 185 (12.4), 167 (10.2), 166 (8.9), 186 (7.5), 168 (4.5) [68]. |
| Teucrium oliverianum Ging. ex Benth. | Against diabetes [69,70,71,72]. | Hypoglycemic [72]. Cytoprotective enzyme NAD(P)H: quinone oxidoreductase (NQO1) inducer activity [73]. | Iridoids 2 [72,73]. Neo-clerodane Diterpenoids 38 [72], 39 [72], 40 [72,73], 41 [72], 42 [71,72,73], 43–45 [72], 46 [74], 47 [70,72], 48 [70], 49 [70], 50 [74], 51 [71,72,73], 52 [71,72], 53 [74]. Flavonoids 87 [72], 92 [72,73]. Sterols 24(S)-Stigmasta-5,22,25-trien-3β-ol [72,73]. |
| Teucrium parviflorum Schreb. | A decoction of the aerial parts is utilized in Turkey against hemorrhoids [75]. | Antioxidant activity [76]. | Essential Oil main components (%) from aerial parts collected in Turkey 168 (18.6), 167 (9.2), 171 (8.8), 183 (6.0), 169 (4.5), 174 (4.4), 187 (4.4), 170 (3.7) [77]. |
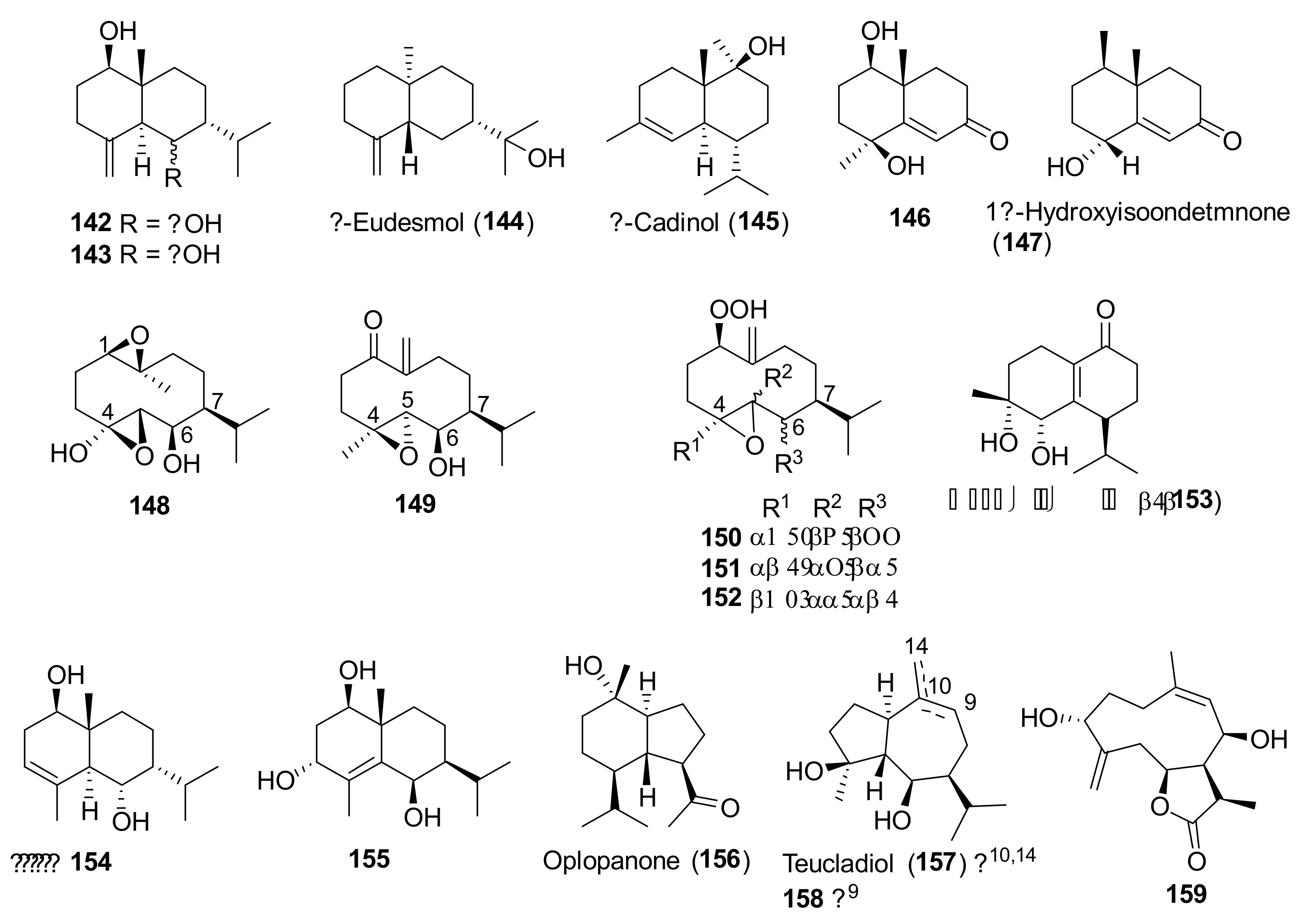
| Teucrium polium L. | Antiulcer, hypotensive, antispasmodic, anorexic and antipyretic agent, hypoglycemic, antidiabetic, hypolipidemic, anti-inflammatory, analgesic, antibacterial, antioxidant, digestive, antidiarrheal, anti-eczema, tonic, to treat skin diseases, stomach pain and disorders, gynecological diseases, kidney and liver diseases, hemorrhoids, menstruation disorders, toothache, body and joint pain, abortion, carminative, antimalarial, against rheumatism, cold and other diseases [26,78,79,80,81,82]. | Anti-inflammatory [78,79], antispasmodic [79], hypotensive [69], hepatoprotective [79], acetylcholinesterase inhibitor and memory enhancer [79], antimutagenic [79], antioxidant and antiradical [79,82,83,84,85,86,87,88,89,90,91,92], antipyretic [93], antimicrobial [79,93,94,95,96,97], anticancer/cytotoxic [79,83,88,91,98,99,100], anti-ulcer [79,101], anti-nociceptive [79,102], antifeedant [103], hypoglycemic [79,89,104,105] hypolipidemic [79,106], proapoptotic [107], immunomodulatory [28], anticonvulsant [28] effects. Antioxidant [98] and references 210, 211, 226, and 255 in reference [26] of this review], antimicrobial [references 213, 214, 229, and 336] in reference [26] of this review]. | Iridoids and Iridoid Glycosides 2 [88,91], 3 [88,91,98,108], 4 [99], 1α-(β-D-glucopyranosyloxy)- 6α,7α-epoxy-4aβ,5α-dihydroxy-7-methyl-1,4a,5,6,7,7aβ-hexahydrocyclopenta[c]pyran (5) [99], 4-[(β-D-glucopyranosyloxy)methylene]-5α-(2-hydroxyethyl)-5-(α-L-rhamnopyranosyloxy)-3-methylcyclopent-2-en-1-one (6) [96], 4α-[(β-D-glucopyranosyloxy)methyl]-5α-(2-hydroxyethyl)-3-methylcyclopent-2-en-1-one (7) [96], 5α-[2-(β-D-glucopyranosyloxy)ethyl]-4α-hydroxymethyl-3-methylcyclopent-2-en-1-one (8) [96], 5α-(2-hydroxyethyl)-4α-hydroxymethyl-3-methylcyclopent-2-en-1-one (9) [96]. Neo-clerodane Diterpenoids 28 [91,100], 54 [80], 55 [80], 56 [85,100], 57 [100], 58 [91,100], 59 [104], teucrin P2 [109], 60 [103], 61 [103], 62 [103], 63 [109], 64 [110], 65 [100,109], 66 [91], 67 [91,100], 68 [91,100], 69 [109,111], 70 [100,109], 71 [109], 72 [108], 73 [110], 74–79 [91,100], 80 [100], 81 [112], 82 [112], 83 [85,91,100,113], 84 [114], 85 [114], 86 [100]. Abeo-abietane Diterpenes 12,16-Epoxy-6,11,14-trihydroxy-17(15→16)-abeo-3α,18-cyclo-5,8,11,13,15-abietapentaen-7-one (129) [90], 12,16-epoxy-6,11,14,17-tetrahydroxy-17(15→16)-abeo-3α,18-cyclo-5,8,11,13,15-abietapentaen-7-one (130) [90], 12,16-epoxy-6,11,14-trihydroxy-17(15→16)-abeo-5,8,11,13,15-abietapentaen-7-one (131) [90], 12,16-epoxy-6,11,14,17-tetrahydroxy-17(15→16)-abeo-5,8,11,13,15-abietapentaen-7-one (132) [90], 12,16-epoxy-6,11,14,15-tetrahydroxy-17(15→16)-abeo-3α,18-cyclo-5,8,11,13-abietatetraen-7-one (133) [90], 12,16-epoxy-6,11,14,15-tetrahydroxy-17(15→16)-abeo-5,8,11,13-abietatetraen-7-one (134) [91], 135–141 [90]. Sesquiterpenoids 7-epi-Eudesm-4(15)-ene-1β,6α-diol (142) [81], 7-epi-eudesrn-4(15)-ene-lβ,6β-diol (143) [81], 144 [81], 145 [81], (1R,4S,10R)-10,11-dimethyl-dicyclohex-5(6)-en-1,4-diol-7-one (146) [88], 147 [88], (10R,1R,4S,5S,6R,7S)-4,10-diepoxygermacran-6-ol (148) [88,95], 4β,5α-epoxy-7αH-germacr-10(14)-en-6β-ol-1-one (149) [95], 4β,5α-epoxy-7αH-germacr-10(14)-en,1β-hydroperoxyl,6β-ol (150) [95], 4β,5β-epoxy-7αH-germacr-10(14)-en,1β-hydroperoxyl,6β-ol (151) [95], 4α,5β-epoxy-7αH-germacr-10(14)-en,1β-hydroperoxyl,6α-ol (152) [95], 153 [95], eudesm-3-ene-1,6-diol (154) [95], rel-1β,3α,6β-trihydroxyeudesm-4-ene (155) [95], 156 [95], 157 [95], 4β,6β-dihydroxy-1α,5β(H)-guai-9-ene (158) [95], (1R,6R,7R,8S,11R)-1,6-dihydroxy-4,11-dimethylgermacra-4(5),10(14)-dien-8,12-olide (159) [88]. Flavonoids 87 [59,115,116,117], 90 [86,89,91,92,114], 3′,6-dimethoxy apigenin [86], 4′,7-O-dimethyl apigenin [86], 92 [116], 93 [85], 94 [95,115], 95 [59,91,92,114,116], 96 [114], 97 [114], 98 [58,96,114,116,118], 100 [86,89], 101 [85], 102 [85], 103 [85,92,118], 104 [59], 5,6-dihydroxy-7,4′-dimethoxyflavone [95], 5-hydroxy-7,4′-dimethoxyflavone [116], 105 [59], 3′,4′,5-trihydroxy-6,7-dimethoxyflavone [97], 5,6,7,3′,4′-pentahydroxyflavone [96], (106) [92], 5,7,4′-trihydroxyflavone 6,8-di-C-glucoside (vicenin-2) [118], apigenin 5-galloylglucoside [118]. Verbascoside Derivatives 110 [85], 114 [88,97,114], 2-(3-hydroxy-4-methoxyphenyl)-ethyl-O-(α-L-rhamnosyl)-(1→3)-O-(α-L-rhamnosyl)-(1→6)-4-O-(E)-feruloyl-β-D-glucopyranoside (115) [88], 2-(3,4-dihydroxyphenyl)-ethyl-O-(α-L-rhamnopyranosyl)-(1→3)-O-(α-L-rhamnopyranosyl)-(1→6)-4-[O-(E)-3-(4-hydroxy-3-methoxyphenyl)]-2-propenoate-β-D-glucopyranoside [88], 116 [85,88,91,95,97], 117 [97]. Phenolics, Lignans and Miscellaneous Compounds 119 [88], (R)-mandelonitrile-β-laminaribioside (120) [88], 121 [95], 2-(3,4-dihydroxyphenyl)ethanol (122) [96], 3,4-dihydroxy-3-(O-β-D-glucopyranosyl)phenylethanol (123) [96], 3-(O-β-D-glucopyranosyl)-α-(O-β-D-glucopyranosyl)-4-hydroxyphenylethanol (124) [96], (7S,8R)-4-(O-β-D-glucopyranosyl)-dehydrodiconiferyl alcohol (125) [96], (7S,8R)-5-methoxy-4-(O-β-D-glucopyranosyl)dehydrodiconiferyl alcohol (126) [96], 127 [114], 128 [114], α-tocopherol [119], β-arbutin [32]. Triterpenes, Sterols, Fatty Acids and Saponins Oleanolic acid [114], 160 [114], β-sitosterol [119,120], stigmasterol [119,120], campesterol [120], brassicasterol [120], 161 [119,120], 162 [119], linoleic acid [119], linolenic acid [119], palmitic acid [119], lauric acid [119], 163–165 [98]. Essential Oils The compositions of the essential oils isolated from aerial parts, leaves and flowers of T. polium collected in Algeria, Croatia, Francia, Greece, Iran, Jordan, Montenegro, Morocco, Oman, Russia, Saudi Arabia, Serbia, Syria, Tunisia and Turkey have been reported in several papers. See references [63,66,79,102,121,122,123,124,125,126,127,128,129,130] in this paper and references 210–259 in reference [26] of this review. |
| Teucrium scordium subsp. scordioides (Schreb.) Arcang. | Antimicrobial activity [131]. | Neo-clerodane Diterpenoids 17 [131], 19 [131], 25 [131]. Essential Oil main components (%) from aerial parts collected in Serbia 188 (11.9), (Z)-octadec-9-enoic (oleic) acid (11.5), (Z,Z)-octadeca-9,12-dienoic (linoleic) acid (7.9), hexadecanoic (palmitic) acid (6.4), 166 (3.5), 127 (3.5) [132]. Essential Oil main components (%) from aerial parts collected in Sicily (Italy) 171 (25.8), 174 (19.4), 175 (8.5), 4-(1,5-dimethylhex-4-enyl)-cyclohex-2-enone (6.4), 189 (5.9), 166 (4.4), 190 (3.8), 191 (3.4) [133]. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abdullah, F.O.; Hussain, F.H.S.; Sardar, A.S.; Gilardoni, G.; Thu, Z.M.; Vidari, G. Bio-Active Compounds from Teucrium Plants Used in the Traditional Medicine of Kurdistan Region, Iraq. Molecules 2022, 27, 3116. https://doi.org/10.3390/molecules27103116
Abdullah FO, Hussain FHS, Sardar AS, Gilardoni G, Thu ZM, Vidari G. Bio-Active Compounds from Teucrium Plants Used in the Traditional Medicine of Kurdistan Region, Iraq. Molecules. 2022; 27(10):3116. https://doi.org/10.3390/molecules27103116
Chicago/Turabian StyleAbdullah, Fuad O., Faiq H. S. Hussain, Abdullah Sh. Sardar, Gianluca Gilardoni, Zaw Min Thu, and Giovanni Vidari. 2022. "Bio-Active Compounds from Teucrium Plants Used in the Traditional Medicine of Kurdistan Region, Iraq" Molecules 27, no. 10: 3116. https://doi.org/10.3390/molecules27103116
APA StyleAbdullah, F. O., Hussain, F. H. S., Sardar, A. S., Gilardoni, G., Thu, Z. M., & Vidari, G. (2022). Bio-Active Compounds from Teucrium Plants Used in the Traditional Medicine of Kurdistan Region, Iraq. Molecules, 27(10), 3116. https://doi.org/10.3390/molecules27103116









