Pressurized Solvent Extraction of Paulownia Bark Phenolics
Abstract
1. Introduction
2. Results and Discussion
2.1. Composition
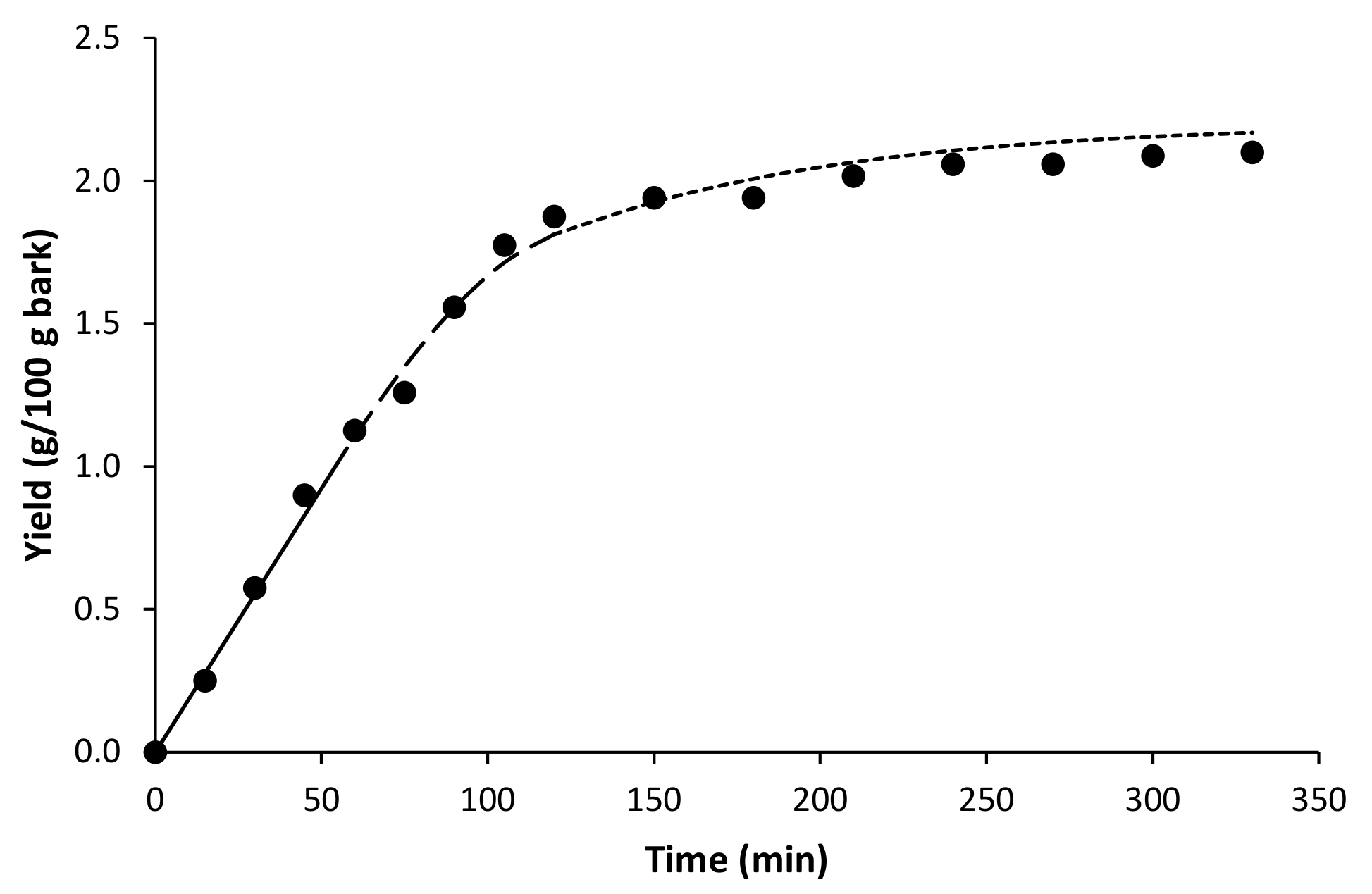
2.2. Subcritical Water Extraction
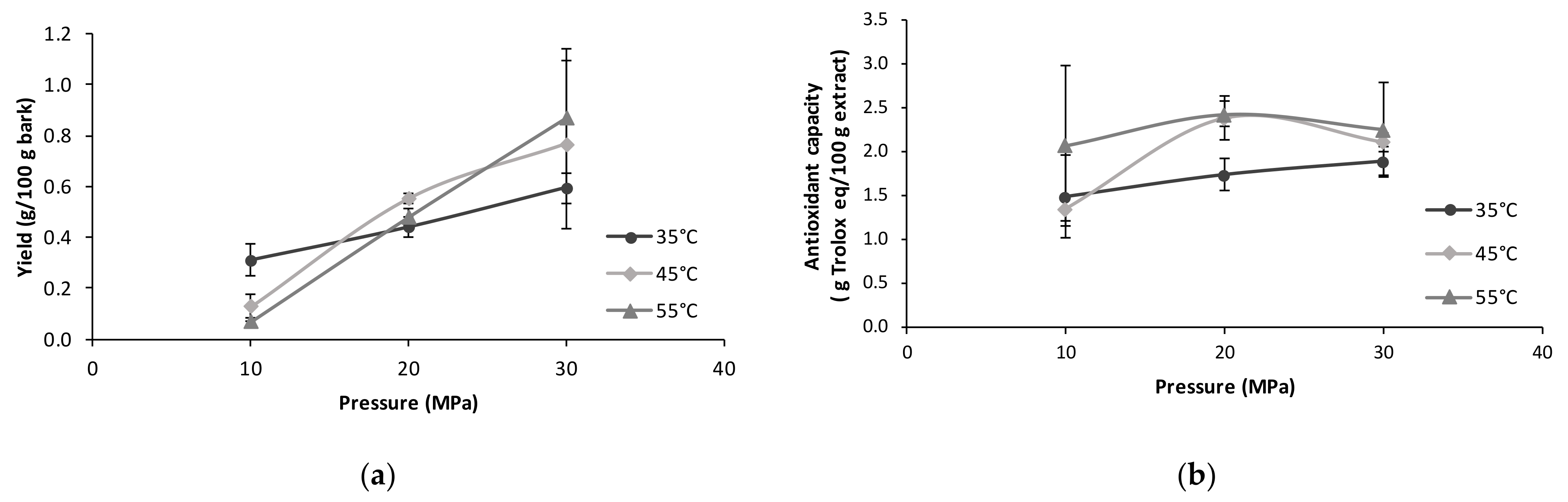
2.3. Supercritical Fluid Extraction
3. Materials and Methods
3.1. Raw Material
3.2. Extraction
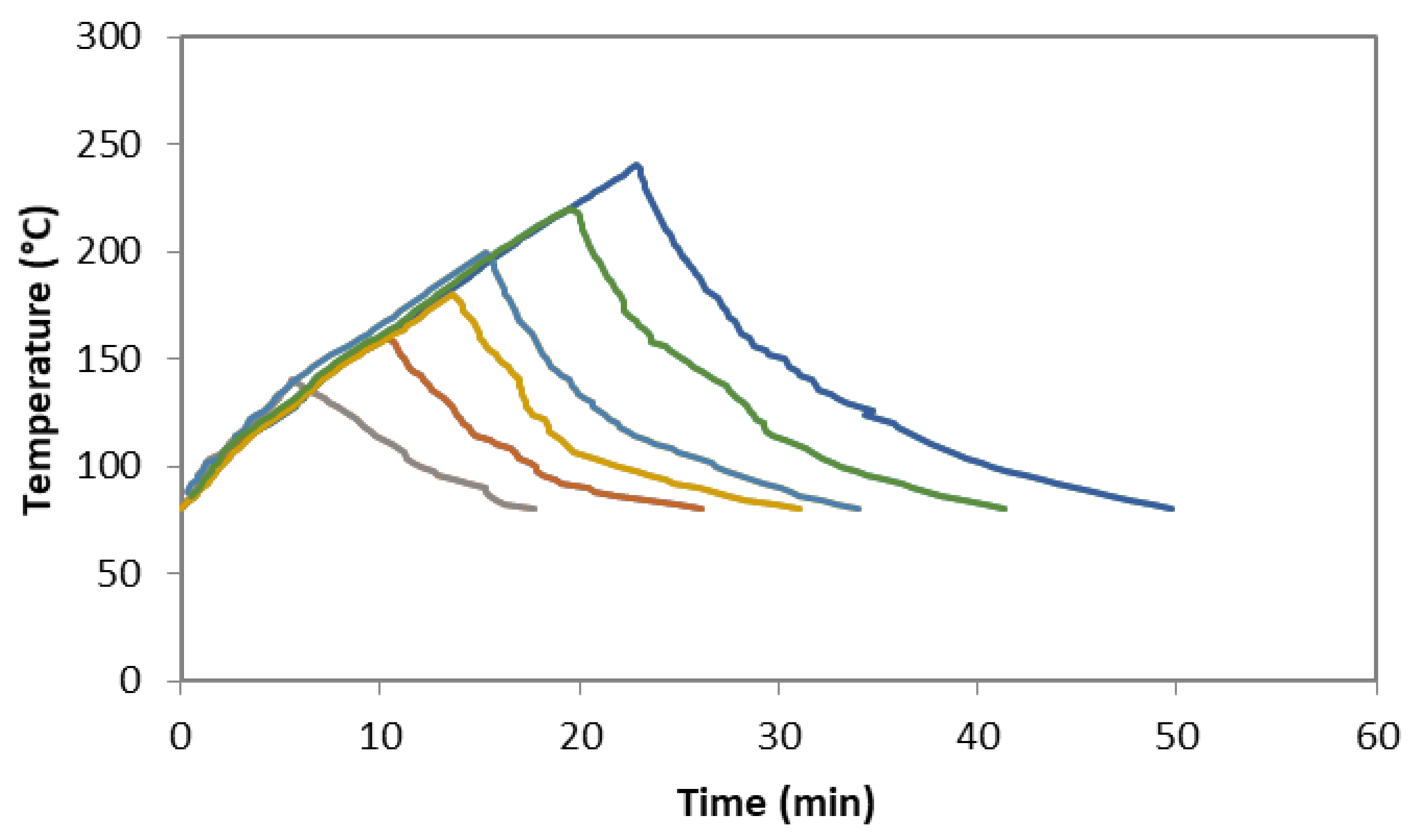
3.2.1. Subcritical Water Extraction
3.2.2. Supercritical Carbon Dioxide Extraction
3.3. Analytical Methods
3.3.1. Raw Material Characterization
3.3.2. Extraction Yield
3.3.3. Total Phenolic Content and Antioxidant Profile
3.3.4. Saccharidic Fraction of Hydrothermal Extracts
3.4. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Schneiderová, K.; Šmejkal, K. Phytochemical profile of Paulownia tomentosa (Thunb). Steud. Phytochem. Rev. 2015, 14, 799–833. [Google Scholar] [CrossRef]
- He, T.; Vaidya, B.; Perry, Z.; Parajuli, P.; Joshee, N. Paulownia as a Medicinal Tree: Traditional Uses and Current Advances. Eur. J. Med. Plants 2016, 14, 1–15. [Google Scholar] [CrossRef]
- Si, C.-L.; Wu, L.; Zhu, Z.-Y.; Kim, J.-K.; Kwon, D.-J.; Bae, Y.-S. Apigenin derivatives from Paulownia tomentosa Steud. var. tomentosa stem barks. Holzforschung 2009, 63, 440–442. [Google Scholar] [CrossRef]
- Kang, K.H.; Huh, H.; Kim, K.; Lee, L. An antiviral furanoquinone from Paulownia tomentosa Steud. Phyther. Res. 1999, 13, 624–626. [Google Scholar] [CrossRef]
- Cheng, C.-L.; Jia, X.-H.; Xiao, C.-M.; Tang, W.-Z. Paulownia C-geranylated flavonoids: Their structural variety, biological activity and application prospects. Phytochem. Rev. 2019, 18, 549–570. [Google Scholar] [CrossRef] [PubMed]
- Si, C.L.; Lu, Y.Y.; Qin, P.P.; Sun, R.C.; Ni, Y.H. Phenolic extractives with chemotaxonomic significance from the bark of Paulownia tomentosa var. Tomentosa. BioResources 2011, 6, 5086–5098. [Google Scholar] [CrossRef]
- Lee, J.-W.; Seo, K.-H.; Ryu, H.W.; Yuk, H.J.; Park, H.A.; Lim, Y.; Ahn, K.-S.; Oh, S.-R. Anti-inflammatory effect of stem bark of Paulownia tomentosa Steud. in lipopolysaccharide (LPS)-stimulated RAW264.7 macrophages and LPS-induced murine model of acute lung injury. J. Ethnopharmacol. 2018, 210, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Sticher, O.; Lahloub, M.F. Phenolic glycosides of Paulownia tomentosa bark. Planta Med. 1982, 46, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-K.; Si, C.-L.; Bae, Y.-S. Epimeric phenylpropanoid glycosides from inner bark of Paulownia coreana Uyeki. Holzforschung 2007, 61, 161–164. [Google Scholar] [CrossRef]
- Si, C.-L.; Liu, S.-C.; Hu, H.-Y.; Jiang, J.-Z.; Yu, G.-J.; Ren, X.-D.; Xu, G.-H. Activity-guided screening of the antioxidants from Paulownia tomentosa var. Tomentosa bark. BioResources 2013, 8, 628–637. [Google Scholar] [CrossRef]
- Bukhanko, N.; Attard, T.; Arshadi, M.; Eriksson, D.; Budarin, V.; Hunt, A.J.; Geladi, P.; Bergsten, U.; Clark, J. Extraction of cones, branches, needles and bark from Norway spruce (Picea abies) by supercritical carbon dioxide and soxhlet extractions techniques. Ind. Crops Prod. 2020, 145, 112096. [Google Scholar] [CrossRef]
- Santos, S.A.O.; Villaverde, J.J.; Silva, C.M.; Neto, C.P.; Silvestre, A.J.D. Supercritical fluid extraction of phenolic compounds from Eucalyptus globulus Labill bark. J. Supercrit. Fluids 2012, 71, 71–79. [Google Scholar] [CrossRef]
- Lachos-Perez, D.; Brown, A.B.; Mudhoo, A.; Martinez, J.; Timko, M.T.; Rostagno, M.A.; Forster-Carneiro, T. Applications of subcritical and supercritical water conditions for extraction, hydrolysis, gasification, and carbonization of biomass: A critical review. Biofuel Res. J. 2017, 14, 611–626. [Google Scholar] [CrossRef]
- Munir, M.T.; Kheirkhah, H.; Baroutian, S.; Quek, S.Y.; Young, B.R. Subcritical water extraction of bioactive compounds from waste onion skin. J. Clean. Prod. 2018, 183, 487–494. [Google Scholar] [CrossRef]
- Yan, Z.; Zhang, H.; Dzah, C.S.; Zhang, J.; Diao, C.; Ma, H.; Duan, Y. Subcritical water extraction, identification, antioxidant and antiproliferative activity of polyphenols from lotus seedpod. Sep. Purif. Technol. 2020, 236, R713–R715. [Google Scholar] [CrossRef]
- Wiboonsirikul, J.; Adachi, S. Extraction of functional substances from agricultural products or by-products by subcritical water treatment. Food Sci. Technol. Res. 2008, 14, 319–328. [Google Scholar] [CrossRef]
- Díaz-Reinoso, B.; Moure, A.; Domínguez, H.; Parajó, J.C. Supercritical CO2 extraction and purification of compounds with antioxidant activity. J. Agric. Food Chem. 2006, 54, 2441–2469. [Google Scholar] [CrossRef]
- Chai, Y.H.; Yusup, S.; Ruslan, M.S.H.; Chin, B.L.F. Supercritical fluid extraction and solubilization of Carica papaya Linn. leaves in ternary system with CO2 + ethanol solvents. Chem. Eng. Res. Des. 2020, 156, 31–42. [Google Scholar] [CrossRef]
- Barbini, S.; Jaxel, J.; Karlström, K.; Rosenau, T.; Potthast, A. Multistage fractionation of pine bark by liquid and supercritical carbon dioxide. Bioresour. Technol. 2021, 341, 125862. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Yang, C.; Hidayat, W.; Jang, J.H.; Kim, N.H. Solid bioenergy properties of Paulownia tomentosa grown in Korea. J. Korean Wood Sci. Technol. 2016, 44, 890–896. [Google Scholar] [CrossRef]
- Domingues, R.M.A.; De Melo, M.M.R.; Neto, C.P.; Silvestre, A.J.D.; Silva, C.M. Measurement and modeling of supercritical fluid extraction curves of Eucalyptus globulus bark: Influence of the operating conditions upon yields and extract composition. J. Supercrit. Fluids 2012, 72, 176–185. [Google Scholar] [CrossRef]
- Yesil-Celiktas, O.; Otto, F.; Gruener, S.; Parlar, H. Determination of Extractability of Pine Bark Using Supercritical CO2 Extraction and Different Solvents: Optimization and Prediction. J. Agric. Food Chem. 2008, 57, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Sovová, H. Rate of the vegetable oil extraction with supercritical CO2—I. Modelling of extraction curves. Chem. Eng. Sci. 1994, 49, 409–414. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]


| Component | Content (%, d.w.) |
|---|---|
| Ash | 3.00 ± 0.17 |
| Ethanol extractives | 21.32 ± 0.28 |
| Protein | 4.95 ± 0.51 |
| Acid insoluble residue | 25.62 ± 0.79 |
| Carbohydrates | |
| Galacturonic acid | 5.12 ± 0.13 |
| Glucose | 20.63 ± 0.15 |
| Xylose | 5.34 ± 0.30 |
| Mannose | 0.43 ± 0.11 |
| Galactose | 3.81 ± 0.09 |
| Rhamnose | 1.93 ± 0.16 |
| Arabinose | 1.83 ± 0.02 |
| (a) | ||||||
|---|---|---|---|---|---|---|
| Monomers (g/100 g Extract) | Temperature (°C) | |||||
| 140 | 160 | 180 | 200 | 220 | 240 | |
| Trehalose | 7.5 | 6.07 | 5.29 | 2.93 | - | - |
| Glucuronic acid | - | - | - | - | - | - |
| Galacturonic acid | - | - | - | - | - | - |
| Glucose | 11.63 | 10.1 | 12.72 | 11.36 | 3.08 | 2 |
| Xylose | 0.7 | 0.51 | 0.21 | 0.25 | 1.29 | 0.87 |
| Galactose | 8.36 | 1.91 | 1.57 | 1.9 | 1.57 | 0.6 |
| Rhamnose | 0.22 | 0.15 | 0.65 | 2.55 | 1.29 | 0.5 |
| Arabinose | - | 0.08 | 0.84 | 3.2 | 1.42 | 0.92 |
| Mannose | 2.34 | 2.32 | 4.12 | 2.95 | 0.92 | 0.86 |
| Mannitol | 0.14 | 0.12 | 0.12 | 0.14 | 0.16 | 0.59 |
| Formic acid | 2.72 | - | 0.8 | 3.61 | 1.99 | 15.16 |
| Acetic acid | - | 0.27 | 0.61 | 2.36 | 8.2 | 20.57 |
| (b) | ||||||
| Oligomers (g/100 g Extract) | Temperature (°C) | |||||
| 140 | 160 | 180 | 200 | 220 | 240 | |
| Trehalose | - | - | - | - | - | - |
| Glucuronic acid | 0.88 | 1.17 | 1.39 | 1 | 1.06 | 1.47 |
| Galacturonic acid | 8.41 | 11.57 | 5.65 | 1.48 | 0.05 | 0.07 |
| Glucose | 20.91 | 18.25 | 15.9 | 14.73 | 15.19 | 11.37 |
| Xylose | 0.7 | 0.37 | 1.26 | 9.07 | 16.15 | 1.25 |
| Galactose | 8.36 | 8.04 | 9.13 | 9.2 | 5.56 | 0.83 |
| Rhamnose | 6.72 | 6.69 | 6.83 | 5.13 | 1.76 | - |
| Arabinose | 3.24 | 13.01 | 17.74 | 13.28 | 1.8 | - |
| Mannose | - | - | - | - | 2.73 | 2.03 |
| Formic acid | 2.72 | 2.33 | 3.35 | 2.29 | 7.34 | - |
| Acetic acid | 0.94 | 0.88 | 1.34 | 2.97 | 3.88 | - |
| Parameter | Value |
|---|---|
| kxa (min−1) | 6.22 × 10−3 |
| kya (min−1) | 1.95 |
| r | 0.74 |
| Ys (kg/kg) | 1.09 × 10−4 |
| xo | 0.022 |
| tCER (min) | 53.3 |
| tFER (min) | 117.7 |
| AARD (%) | 7.09 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Seoane, P.; Díaz-Reinoso, B.; Domínguez, H. Pressurized Solvent Extraction of Paulownia Bark Phenolics. Molecules 2022, 27, 254. https://doi.org/10.3390/molecules27010254
Rodríguez-Seoane P, Díaz-Reinoso B, Domínguez H. Pressurized Solvent Extraction of Paulownia Bark Phenolics. Molecules. 2022; 27(1):254. https://doi.org/10.3390/molecules27010254
Chicago/Turabian StyleRodríguez-Seoane, Paula, Beatriz Díaz-Reinoso, and Herminia Domínguez. 2022. "Pressurized Solvent Extraction of Paulownia Bark Phenolics" Molecules 27, no. 1: 254. https://doi.org/10.3390/molecules27010254
APA StyleRodríguez-Seoane, P., Díaz-Reinoso, B., & Domínguez, H. (2022). Pressurized Solvent Extraction of Paulownia Bark Phenolics. Molecules, 27(1), 254. https://doi.org/10.3390/molecules27010254






