Antibacterial Activity of Clay Soils against Food-Borne Salmonella typhimurium and Staphylococcus aureus
Abstract
1. Introduction
2. Results
2.1. Preliminary Screening of Clay Soils for Antibacterial Activity
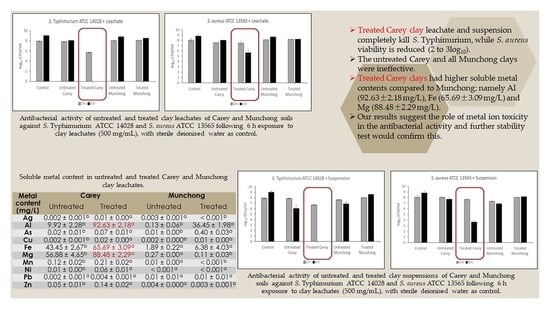
2.2. Antibacterial Effect of Untreated and Treated Clay Leachate and Suspension
2.3. Cation-Exchange Capacity of Untreated and Treated Clays of Carey and Munchong Soils
2.4. pH of Untreated and Treated Clays of Carey and Munchong Soils
2.5. Mineralogy of Untreated and Treated Clays of Carey and Munchong Soils
2.6. Soluble and Total Metals of Untreated and Treated Clays of Carey and Munchong Soils
3. Discussion
4. Materials and Methods
4.1. Soil Sampling and Preparation
4.2. Preparation of Untreated and Treated Clay Leachate and Suspension
4.2.1. Untreated Clay
4.2.2. Treated Clay
4.2.3. Clay Leachate
4.2.4. Clay Suspension
4.3. Antibacterial Testing
4.3.1. Bacterial Strains and Growth Condition
4.3.2. Antibacterial Screening of Clay Soils
4.3.3. Antibacterial Effect of Untreated and Treated Clay Leachate and Suspension
4.4. Physicochemical Properties of Soil Clays
4.4.1. Soil Particle Analysis
4.4.2. pH Analysis
4.4.3. Cation-Exchange Capacity
4.5. Mineralogical Identification
4.6. Metal Composition Analysis
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
Appendix A

References
- Williams, L.B.; Holland, M.; Eberl, D.D.; Brunet, T.; Brunet de Courrsou, L. Killer Clays! Natural antibacterial clay minerals. Mineral. Soc. Bull. 2004, 139, 3–8. [Google Scholar]
- Otto, C.C.; Koehl, J.L.; Solanky, D.; Haydel, S.E. Metal ions, not metal-catalyzed oxidative stress, cause clay leachate antibacterial activity. PLoS ONE 2014, 9, e115172. [Google Scholar] [CrossRef]
- Redmond, E.C.; Griffith, C.J. Consumer food handling in the home: A review of food safety studies. J. Food Prot. 2003, 66, 130–161. [Google Scholar] [CrossRef] [PubMed]
- Di Ciccio, P.; Vergara, A.; Festino, A.R.; Paludi, D.; Zanardi, E.; Ghidini, S.; Ianieri, A. Biofilm formation by Staphylococcus aureus on food contact surfaces: Relationship with temperature and cell surface hydrophobicity. Food Control 2015, 50, 930–936. [Google Scholar] [CrossRef]
- Williams, L.B.; Metge, D.W.; Eberl, D.D.; Harvey, R.W.; Turner, A.G.; Prapaipong, P.; Poret-Peterson, A.T. What makes a natural clay antibacterial? Environ. Sci. Technol. 2011, 45, 3768–3773. [Google Scholar] [CrossRef]
- Morrison, K.D.; Misra, R.; Williams, L.B. Unearthing the antibacterial mechanism of medicinal clay: A geochemical approach to combating antibiotic resistance. Sci. Rep. 2016, 6, 19043. [Google Scholar] [CrossRef]
- Londono, S.C.; Hartnett, H.E.; Williams, L.B. Antibacterial activity of aluminum in clay from the Colombian Amazon. Environ. Sci. Technol. 2017, 51, 2401–2408. [Google Scholar] [CrossRef] [PubMed]
- Paramananthan, S. Soils in Malaysia: Their Characteristics and Identification; ASM Publications: Kuala Lumpur, Malaysia, 2000. [Google Scholar]
- Cunningham, T.M.; Koehl, J.L.; Summers, J.S.; Haydel, S.E. pH-dependent metal ion toxicity influences the antibacterial activity of two natural mineral mixtures. PLoS ONE 2010, 5, e9456. [Google Scholar] [CrossRef]
- Williams, L.B. Geomimicry: Harnessing the antibacterial action of clays. Clay Miner. 2017, 52, 1–24. [Google Scholar] [CrossRef][Green Version]
- Otto, C.C.; Kilbourne, J.; Haydel, S.E. Natural and ion-exchanged illite clays reduce bacterial burden and inflammation in cutaneous meticillin-resistant Staphylococcus aureus infections in mice. J. Med. Microbiol. 2016, 65, 19–27. [Google Scholar] [CrossRef]
- Bagchi, B.; Kar, S.; Kr, S.; Bhandary, S.; Roy, D.; Kr, T.; Das, S.; Nandy, P. In situ synthesis and antibacterial activity of copper nanoparticle loaded natural montmorillonite clay based on contact inhibition and ion release. Colloids Surf. B Biointerfaces 2013, 108, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Zhang, C.; Zeng, G.M.; Gong, J.L.; Chang, Y.N.; Song, B.; Deng, C.H.; Liu, H.Y. The disinfection performance and mechanisms of Ag/lysozyme nanoparticles supported with montmorillonite clay. J. Hazard. Mater. 2016, 317, 416–429. [Google Scholar] [CrossRef] [PubMed]
- Berger, T.J.; Spadaro, J.A.; Chapin, S.E.; Becker, R. Electrically generated silver ions: Quantitative effects on bacterial and mammalian cells. Antimicrob Agents Chemother. 1976, 9, 357–358. [Google Scholar] [CrossRef]
- Domek, M.J.; Lechevallier, M.W.; Cameron, S.C.; Mcfeters, G.A. Evidence for the role of copper in the injury process of coliform bacteria in drinking water. Appl. Environ. Microbiol. 1984, 48, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Gordon, A.S.; Howell, L.D.; Harwood, V. Responses of diverse heterotrophic bacteria to elevated copper concentrations. Can. J. Microbiol. 1994, 40, 5–8. [Google Scholar] [CrossRef][Green Version]
- Nies, D.H. Microbial heavy-metal resistance. Appl. Microbiol. Biotechnol. 1999, 51, 730–750. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.H.; Xia, M.S. Adsorption and antibacterial effect of copper-exchanged montmorillonite on Escherichia coli K88. Appl. Clay Sci. 2006, 31, 180–184. [Google Scholar] [CrossRef]
- Magaña, S.M.; Quintana, P.; Aguilar, D.H.; Toledo, J.A.; Ángeles-Chávez, C.; Cortés, M.A.; León, L.; Freile-Pelegrín, Y.; López, T.; Sánchez, R.M.T. Antibacterial activity of montmorillonites modified with silver. J. Mol. Catal. A Chem. 2008, 281, 192–199. [Google Scholar] [CrossRef]
- USDA. Soil Mechanics Level 1. Module 3-USDA Textural Soil Classification Study Guide; U.S. Government Printing Office: Washington, DC, USA, 1987.
- Hazelton, P.; Murphy, B. Interpreting Soil Test Results: What do All the Numbers Mean? Csiro Publishing: Collingwood, VIC, Australia, 2007. [Google Scholar]
- Shamshuddin, J. Acid Sulfate Soils in Malaysia; Universiti Putra Malaysia Press: Serdang, Malaysia, 2006. [Google Scholar]
- Williams, L.B.; Haydel, S.E.; Giese, R.F., Jr.; Eberl, D.D. Chemical and mineralogical characteristics of French green clays used for healing. Clays Clay Min. 2008, 56, 437–452. [Google Scholar] [CrossRef]
- Morrison, K.D.; Underwood, J.C.; Metge, D.W.; Eberl, D.D.; Williams, L.B. Mineralogical variables that control the antibacterial effectiveness of a natural clay deposit. Environ. Geochem. Health 2013, 36, 613–631. [Google Scholar] [CrossRef]
- Wang, L.K.; Hung, Y.T.; Shammas, N.K. Handbook of Advanced Industrial and Hazardous Wastes Treatment; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Haydel, S.E.; Remenih, C.M.; Williams, L.B. Broad-spectrum in vitro antibacterial activities of clay minerals against antibiotic-susceptible and antibiotic-resistant bacterial pathogens. J. Antimicrob. Chemother. 2008, 61, 353–361. [Google Scholar] [CrossRef]
- Ibbini, J.H.; Al-qinna, M.I.; Mashal, K.Y.; Abuidhail, J.; Alzoubi, K.H.; Masadeh, M.M. Are clay minerals in Jordanian soils antibacterial? Jordan J. Earth Environ. Sci. 2018, 9, 108–115. [Google Scholar]
- Jensen, J.L.; Schj, P.; Watts, C.W.; Christensen, B.T.; Munkholm, L.J. Soil texture analysis revisited: Removal of organic matter matters more than ever. PLoS ONE 2017, 12, e0178039. [Google Scholar] [CrossRef]
- Otto, C.C.; Cunningham, T.M.; Hansen, M.R.; Haydel, S.E. Effects of antibacterial mineral leachates on the cellular ultrastructure, morphology, and membrane integrity of Escherichia coli and methicillin-resistant Staphylococcus aureus. Ann. Clin. Microbiol. Antimicrob. 2010, 9, 26. [Google Scholar] [CrossRef] [PubMed]
- Caflisch, K.M.; Schmidt-Malan, S.M.; Mandrekar, J.N.; Karau, M.J.; Nicklas, J.P.; Williams, L.B.; Patel, R. Antibacterial activity of reduced iron clay against pathogenic bacteria. Int. J. Antimicrob. Agents 2018, 52, 692–696. [Google Scholar] [CrossRef] [PubMed]
- Otto, C.C.; Haydel, S.E. Exchangeable ions are responsible for the in vitro antibacterial properties of natural clay mixtures. PLoS ONE 2013, 8, e64068. [Google Scholar] [CrossRef]
- Lund, B.; Baird-Parker, A.C.; Baird-Parker, T.C.; Gould, G.W.; Gould, G.W. Microbiological Safety and Quality of Food; Aspen Publishers: Gaithersburg, MD, USA, 2000. [Google Scholar]
- Jay, J.M.; Loessner, M.J.; Golden, D.A. Modern Food Microbiology; Springer Science & Business Media: New York, NY, USA, 2008. [Google Scholar]
- Foster, J.W. The acid tolerance response of Salmonella typhimurium involves transient synthesis of key acid shock proteins. J. Bacteriol. 1993, 175, 1981–1987. [Google Scholar] [CrossRef]
- Borrok, D.; Fein, J.B.; Tischler, M.; Loughlin, E.O.; Meyer, H.; Liss, M. The effect of acidic solutions and growth conditions on the adsorptive properties of bacterial surfaces. Chem. Geol. 2004, 209, 107–119. [Google Scholar] [CrossRef]
- Gee, G.W.; Baulder, J.W. Particle Size Analysis. In Method of Soil Analysis; American Society of Agronomy: Madison, WI, USA, 1986; Volume 9. [Google Scholar]
- Borquaye, L.S.; Ocansey, E.; Semenya, J. Inhibitory effect of selected Ghanaian clay leachates on some pathogenic microbes. Am. J. Microbiol. Immunol. 2016, 1, 1–5. [Google Scholar]
- Casman, E.P.; Bergdoll, M.S.; Robinson, J. Designation of Staphylococcal enterotoxins. J. Bacteriol. 1963, 85, 715–716. [Google Scholar] [CrossRef]
- CLSI. Performance standards for antimicrobial susceptibility testing. Clin. Lab Stand. Institute. 2016, 26, 53–79. [Google Scholar]
- Page, A.L. Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; American Society of Agronomy, Soil Science Society of America: Madison, WI, USA, 1982. [Google Scholar]



| Soil Series | Zone of Inhibition (mm) c | |
|---|---|---|
| Salmonella typhimurium | S. aureus | |
| Batu Anam | 8.82 ± 0.98 b | 6.36 ± 0.16 b |
| Bernam | 8.50 ± 0.58 b | 6.29 ± 0.15 b |
| Carey | 8.06 ± 0.43 b | 7.63 ± 0.48 a |
| Melaka | 7.83 ± 1.04 b | 6.32 ± 0.16 b |
| Munchong | 11.00 ± 0.71 a | 6.30 ± 0.11 b |
| Serkat | 9.12 ± 0.79 b | 6.26 ± 0.13 b |
| Soil Series | Soil Particle Size (%) | Texture Class 1 | ||
|---|---|---|---|---|
| Clay | Silt | Sand | ||
| Batu Anam | 77.84 | 8.51 | 13.96 | Clay |
| Bernam | 61.89 | 37.32 | 0.79 | Clay |
| Carey | 34.62 | 50.28 | 15.11 | Silty clay loam |
| Melaka | 52.40 | 7.56 | 39.96 | Clay |
| Munchong | 78.11 | 9.03 | 12.85 | Clay |
| Serkat | 58.08 | 13.41 | 29.73 | Clay |
| Soil Series | pH |
|---|---|
| Batu Anam | 5.13 ± 0.11 |
| Bernam | 5.14 ± 0.02 |
| Carey | 2.81± 0.10 |
| Melaka | 4.79 ± 0.02 |
| Munchong | 3.60 ± 0.18 |
| Serkat | 2.98 ± 0.04 |
| Sample | CEC (cmol(+)/kg) | Classification a |
|---|---|---|
| Untreated Carey | 9.67 ± 0.16 | Low |
| Treated Carey | 15.00 ± 0.63 | Moderate |
| Untreated Munchong | 7.11 ± 0.08 | Low |
| Treated Munchong | 21.33 ± 0.43 | Moderate |
| Sample | pH | |
|---|---|---|
| Leachate | Suspension | |
| Untreated Carey | 2.80 ± 0.09 | 2.84 ± 0.01 |
| Treated Carey | 2.51 ± 0.05 | 2.75 ± 0.04 |
| Untreated Munchong | 3.66 ± 0.05 | 3.48 ± 0.07 |
| Treated Munchong | 6.19 ± 0.01 | 6.10 ± 0.03 |
| Metal Content (mg/L) | Carey | Munchong | ||
|---|---|---|---|---|
| Untreated | Treated | Untreated | Treated | |
| Ag | 0.002 ± 0.001 b | 0.01 ± 0.00 a | 0.003 ± 0.001 a | <0.001 b |
| Al | 9.92 ± 2.28 b | 92.63 ± 2.18 a | 0.13 ± 0.06 b | 36.45 ± 1.98 a |
| As | 0.02 ± 0.01 b | 0.07 ± 0.01 a | 0.01 ± 0.00 b | 0.40 ± 0.03 a |
| Cu | 0.002 ± 0.001 b | 0.02 ± 0.00 a | 0.002 ± 0.000 b | 0.01 ± 0.00 a |
| Fe | 43.45 ± 2.67 b | 65.69 ± 3.09 a | 1.89 ± 0.22 a | 6.38 ± 4.03 a |
| Mg | 56.88 ± 4.65 b | 88.48 ± 2.29 a | 0.27 ± 0.00 a | 0.11 ± 0.03 b |
| Mn | 0.12 ± 0.02 b | 0.21 ± 0.02 a | 0.01 ± 0.00 a | <0.001 b |
| Ni | 0.01 ± 0.00 b | 0.06 ± 0.01 a | <0.001 a | <0.001 a |
| Pb | 0.002 ± 0.001 a | 0.004 ± 0.001 a | 0.01 ± 0.01 a | 0.01 ± 0.01 a |
| Zn | 0.05 ± 0.01 b | 0.14 ± 0.02 a | 0.004 ± 0.000 a | 0.003 ± 0.001 b |
| Metal Content (mg/L) | Carey | Munchong | ||
|---|---|---|---|---|
| Untreated | Treated | Untreated | Treated | |
| Ag | 0.32 ± 0.04 a | 0.48 ± 0.24 a | 0.04 ± 0.00 b | 0.18 ± 0.06 a |
| Al | 29,450 ± 678 b | 71,660 ± 700 a | 123,620 ± 17740 b | 213,680 ± 6280 a |
| As | 2.22 ± 0.54 b | 3.82 ± 0.26 a | 14.94 ± 3.70 b | 25.52 ± 1.28 a |
| Cu | 4.04 ± 0.08 a | 6.64 ± 5.60 a | 8.20 ± 0.28 a | 7.96 ± 0.44 a |
| Fe | 3800 ± 109 b | 4924 ± 16 a | 5382 ± 98 a | 5502 ± 26 a |
| Mg | 261.52 ± 1.12 b | 353.92 ± 4.64 a | 13.76 ± 0.64 a | 23.92 ± 9.60 a |
| Mn | 18.76 ± 1.56 b | 40.88 ± 0.56 a | 30.90 ± 0.74 a | 12.76 ± 0.68 b |
| Ni | 0.64 ± 0.00 b | 2.76 ± 0.00 a | 1.48 ± 0.24 b | 2.60 ± 0.08 a |
| Pb | 3.46 ± 0.02 b | 7.04 ± 0.60 a | 1.58 ± 0.26 b | 3.78 ± 0.42 a |
| Zn | 11.60 ± 2.00 b | 54.02 ± 1.82 a | 13.62 ± 1.78 a | 13.10 ± 1.22 a |
| Soluble Metals (mg/L) | Leachate | Suspension |
|---|---|---|
| Ag | 0.01 ± 0.00 b | 0.28 ± 0.00 a |
| Al | 92.63 ± 2.18 a | 59.48 ± 4.05 b |
| As | 0.07 ± 0.01 | n.d. |
| Cu | 0.02 ± 0.00 a | 0.01 ± 0.00 b |
| Fe | 65.69 ± 3.09 a | 25.64 ± 2.92 b |
| Mg | 88.48 ± 2.29 b | 108.34 ± 2.36 a |
| Mn | 0.21 ± 0.02 a | 0.25 ± 0.03 a |
| Ni | 0.06 ± 0.01 a | 0.05 ± 0.01 a |
| Pb | 0.004 ± 0.001 a | 0.004 ± 0.001 a |
| Zn | 0.14 ± 0.02 a | 0.13 ± 0.02 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azmi, N.N.; Mahyudin, N.A.; Wan Omar, W.H.; Mahmud Ab Rashid, N.-K.; Ishak, C.F.; Abdullah, A.H.; Sharples, G.J. Antibacterial Activity of Clay Soils against Food-Borne Salmonella typhimurium and Staphylococcus aureus. Molecules 2022, 27, 170. https://doi.org/10.3390/molecules27010170
Azmi NN, Mahyudin NA, Wan Omar WH, Mahmud Ab Rashid N-K, Ishak CF, Abdullah AH, Sharples GJ. Antibacterial Activity of Clay Soils against Food-Borne Salmonella typhimurium and Staphylococcus aureus. Molecules. 2022; 27(1):170. https://doi.org/10.3390/molecules27010170
Chicago/Turabian StyleAzmi, Nur Naqiyah, Nor Ainy Mahyudin, Wan Hasyera Wan Omar, Nor-Khaizura Mahmud Ab Rashid, Che Fauziah Ishak, Abdul Halim Abdullah, and Gary J. Sharples. 2022. "Antibacterial Activity of Clay Soils against Food-Borne Salmonella typhimurium and Staphylococcus aureus" Molecules 27, no. 1: 170. https://doi.org/10.3390/molecules27010170
APA StyleAzmi, N. N., Mahyudin, N. A., Wan Omar, W. H., Mahmud Ab Rashid, N.-K., Ishak, C. F., Abdullah, A. H., & Sharples, G. J. (2022). Antibacterial Activity of Clay Soils against Food-Borne Salmonella typhimurium and Staphylococcus aureus. Molecules, 27(1), 170. https://doi.org/10.3390/molecules27010170