Monomers and Macromolecular Materials from Renewable Resources: State of the Art and Perspectives †
Abstract
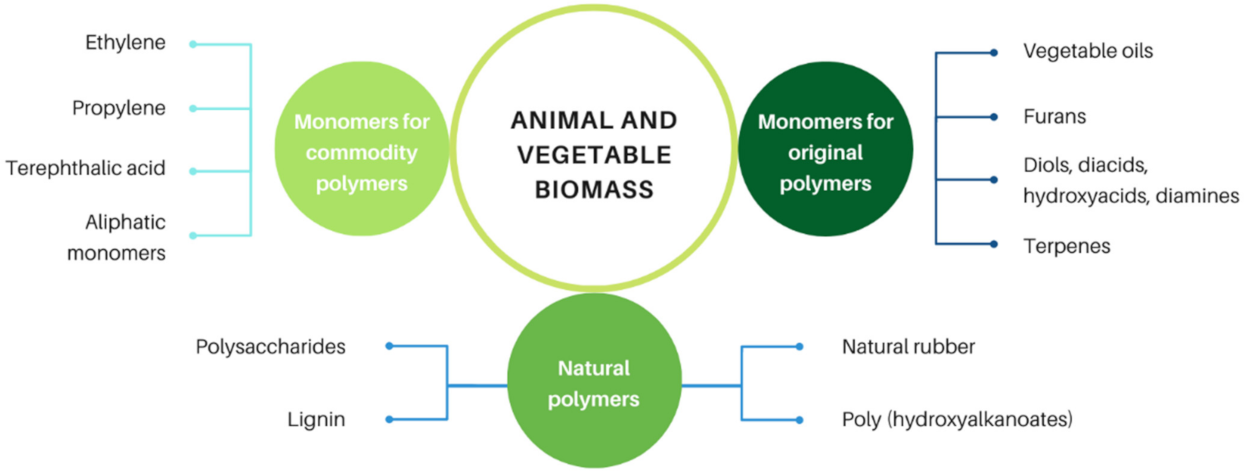
:1. Introduction
2. It All Started with Natural Polymers
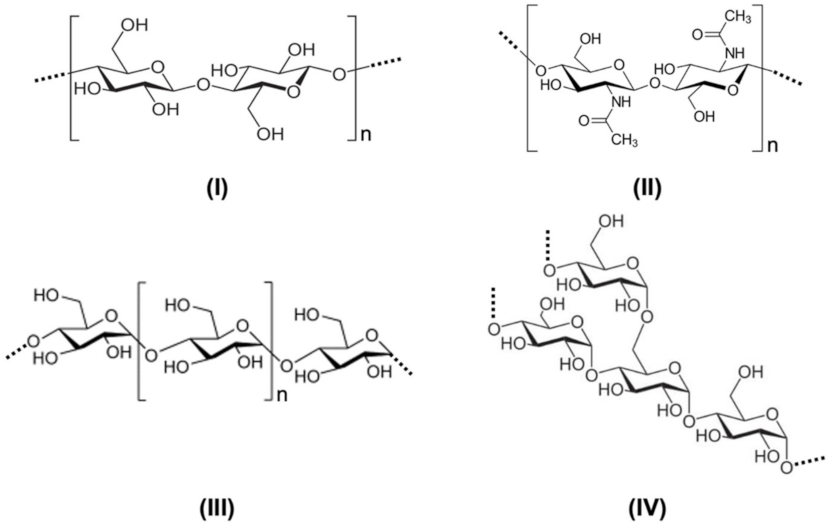
2.1. Polysaccharides
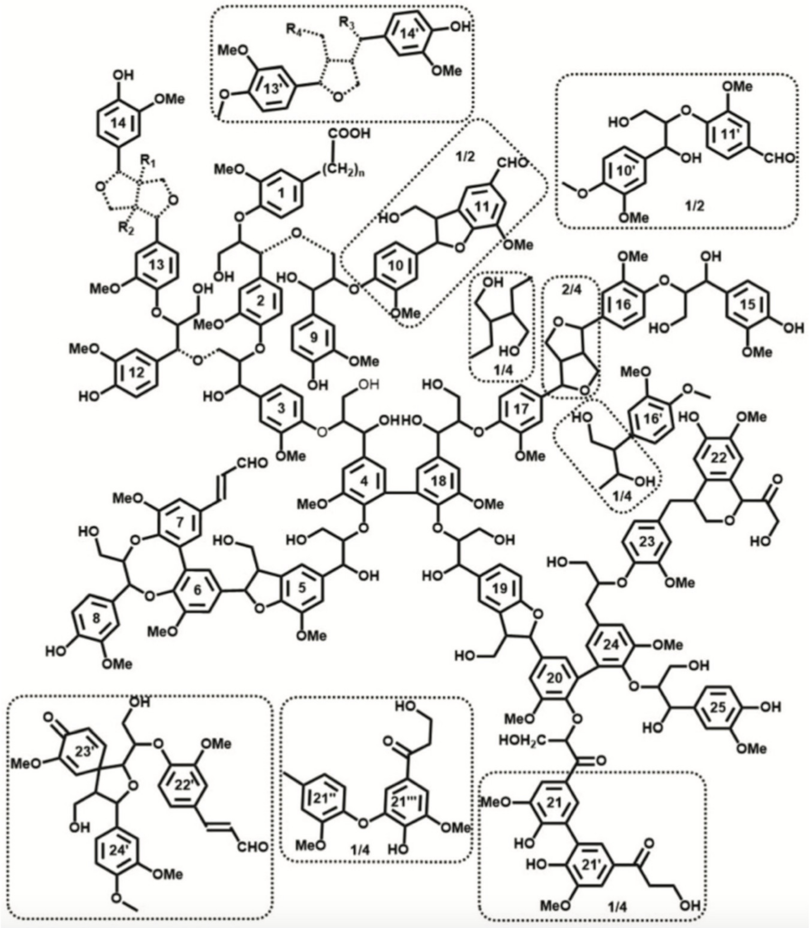
2.2. Lignin
2.3. Natural Rubber
3. Bio-Based Routes for Commodity Polymers
4. Broadening the Horizon: Bio-Based Routes for Original Polymers
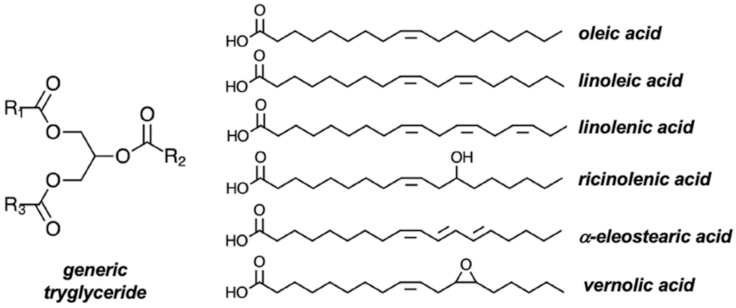
4.1. Vegetable Oils and Glycerol
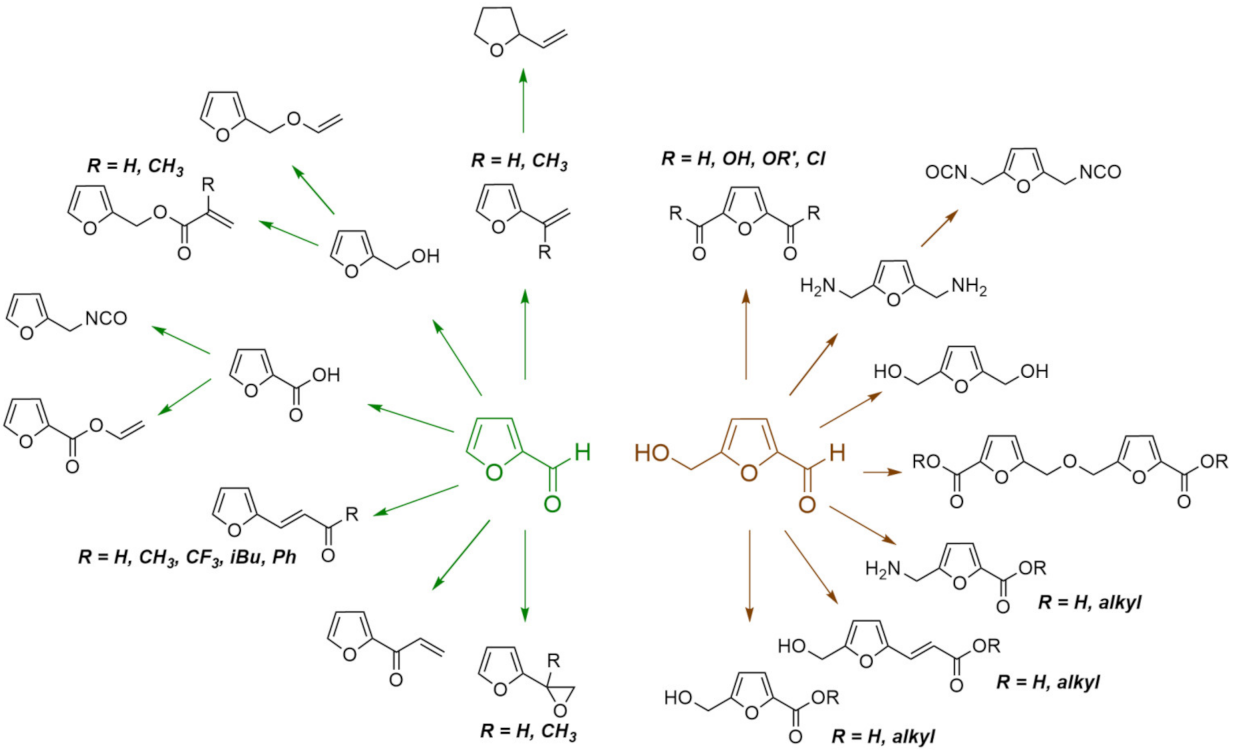
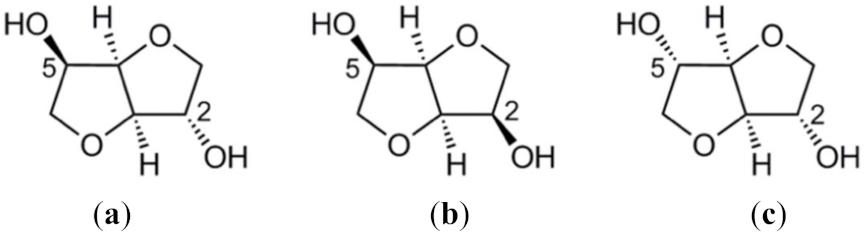
4.2. Furans
4.3. Sugars and Terpenes
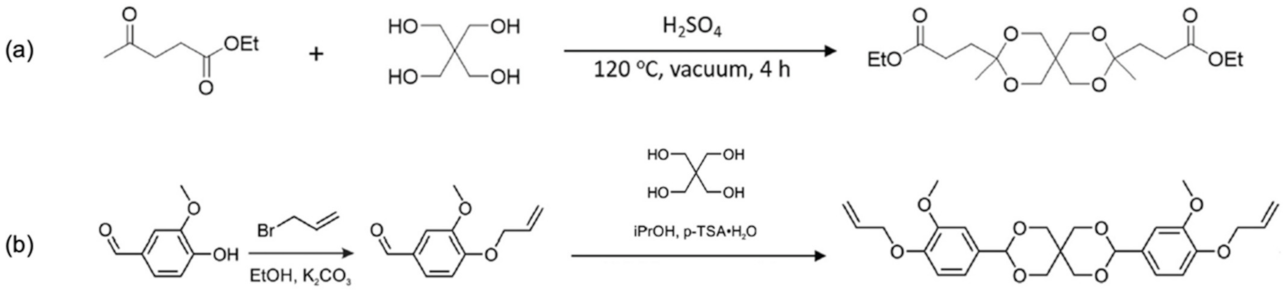
4.4. Miscellaneous: Other Relevant Bio-Based Monomers
5. Overview on Biodegradable Bio-Based Polymers
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Statista. Available online: https://www.statista.com/statistics/282732/global-production-of-plastics-since-1950/ (accessed on 21 September 2021).
- Zhu, Y.; Romain, C.; Williams, C.K. Sustainable polymers from renewable resources. Nature 2016, 540, 354–362. [Google Scholar] [CrossRef]
- Ramesh, P.; Vinodh, S. State of art review on life cycle assessment of polymers. Int. J. Sust. Eng. 2020, 13, 411–422. [Google Scholar] [CrossRef]
- Tschan, M.J.-L.; Brulé, E.; Haquette, P.; Thomas, C.M. Synthesis of biodegradable polymers from renewable resources. Polym. Chem. 2012, 3, 836–851. [Google Scholar] [CrossRef]
- Belgacem, M.N.; Gandini, A. (Eds.) Monomers Polymers and Composites from Renewable Resources; Elsevier: Amsterdam, The Neterlands, 2008; p. 552. [Google Scholar]
- Gandini, A.; Lacerda, T.M. Polymers from Renewable Resources: Macromolecular Materials for the XXI Century? In Macromolecular Engineering: From Precise Synthesis to Macroscopic Materials and Applications, 2nd ed.; Matyjaszewski, K., Gnanou, Y., Hadjichristidis, N., Muthukumar, M., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2022. [Google Scholar]
- Lange, J.-P. Managing plastic waste–Sorting, recycling, disposal, and product redesign. ACS Sustain. Chem. Eng. 2021, 9, 15722–15738. [Google Scholar] [CrossRef]
- Tardy, B.L.; Mattos, B.D.; Otoni, C.G.; Beaumont, M.; Majoinen, J.; Kämäräinen, T.; Rojas, O. Deconstruction and reassembly of renewable polymers and biocolloids into next generation structured materials. Chem. Rev. 2021, 121, 14088–14188. [Google Scholar] [CrossRef] [PubMed]
- Kalia, S.; Avérous, L. (Eds.) Biodegradable and Biobased Polymers for Environmental and Biomedical Applications; Scrivener Publishing: Salem, MA, USA, 2011. [Google Scholar]
- Mittal, V. (Ed.) Renewable Polymers: Synthesis, Processing, and Technology; Scrivener Publishing: Salem, MA, USA, 2012. [Google Scholar]
- Ebnesajjad, S. (Ed.) Handbook of Biopolymers and Biodegradable Plastics; Elsevier: Amsterdam, The Neterlands, 2012; p. 472. [Google Scholar]
- Cavani, F.; Albonetti, S.; Basile, F.; Gandini, A. (Eds.) Chemicals and Fuels from Bio-Based Building Blocks; Wiley-VCH: Weinheim, Germany, 2016; Volume 1–2. [Google Scholar]
- Gandini, A. Polymers from renewable resources: A challenge for the future of macromolecular materials. Macromolecules 2008, 41, 9491–9504. [Google Scholar] [CrossRef]
- Gandini, A. The irruption of polymers from renewable resources on the scene of macromolecular science and technology. Green Chem. 2011, 13, 1061–1083. [Google Scholar] [CrossRef]
- Gandini, A.; Lacerda, T.M. From monomers to polymers from renewable resources: Recent advances. Progr. Polym. Sci. 2015, 48, 1–39. [Google Scholar] [CrossRef]
- Gandini, A.; Lacerda, T.M.; Carvalho, A.J.F.; Trovatti, E. Progress of polymers from renewable resources: Furans, vegetable oils, and polysaccharides. Chem. Rev. 2016, 116, 1637–1669. [Google Scholar] [CrossRef]
- Delidovich, I.; Hausoul, P.J.C.; Deng, L.; Pfützenreuter, R.; Rose, M.; Palkovits, R. Alternative monomers based on lignocellulose and their use for polymer production. Chem. Rev. 2016, 116, 1540–1599. [Google Scholar] [CrossRef]
- Schneiderman, D.K.; Hillmyer, M.A. 50th Anniversary perspective: There is a great future in sustainable polymers. Macromolecules 2017, 50, 3733–3749. [Google Scholar] [CrossRef]
- John, G.; Nagarajan, S.; Vemula, P.K.; Silverman, J.R.; Pillai, C.K.S. Natural monomers: A mine for functional and sustainable materials–Occurrence, chemical modification and polymerization. Progr. Polym. Sci. 2019, 92, 158–209. [Google Scholar] [CrossRef]
- Scholten, B.V.; Moatsou, D.; Detrembleur, C.; Meier, M.A.R. Progress toward sustainable reversible deactivation radical polymerization. Macromol. Rapid Commun. 2020, 41, 2000266. [Google Scholar] [CrossRef] [PubMed]
- O’Dea, R.M.; Willie, J.A.; Epps, T.H. 100th Anniversary of macromolecular science viewpoint: Polymers from lignocellulosic biomass: Current challenges and future opportunities. ACS Macro Lett. 2020, 9, 476–493. [Google Scholar] [CrossRef]
- Wang, Z.; Ganewatta, M.S.; Tang, C. Sustainable polymers from biomass: Bridging chemistry with materials and processing. Progr. Polym. Sci. 2020, 101, 101197. [Google Scholar] [CrossRef]
- Yang, F.; Antonietti, M. The sleeping giant: A polymer view on humic matter in synthesis and applications. Progr. Polym. Sci. 2020, 100, 101182. [Google Scholar] [CrossRef]
- Carvalho, L.T.; Vieira, T.A.; Zhao, Y.; Celli, A.; Medeiros, S.F.; Lacerda, T.M. Recent advances in the production of biomedical systems based on polyhydroxyalkanoates and exopolysaccharides. Int. J. Biol. Macromol. 2021, 183, 1514–1539. [Google Scholar] [CrossRef]
- Klemm, D.; Kramer, F.; Lindström, T.; Ankerfors, M.; Gray, D.; Dorris, A. Nanocelluloses: A new family of nature-based materials. Angew. Chem. Int. Ed. 2011, 50, 5438–5466. [Google Scholar] [CrossRef]
- Dufresne, A. Nanocellulose, from Nature to High Performance Tailored Materials, 1st ed.; De Gruyter: Berlin, Germany, 2012; p. 460. [Google Scholar]
- Trovatti, E. The future of bacterial cellulose and other microbial polysaccharides. J. Renew. Mater. 2013, 1, 28–41. [Google Scholar] [CrossRef]
- Thompson, L.; Azadmanjiri, J.; Nikzad, M.; Sbarski, I.; Wang, J.; Yu, A. Cellulose nanocrystals: Production, functionalization and advanced applications. Rev. Adv. Mater. Sci. 2019, 58, 1–16. [Google Scholar] [CrossRef]
- Calvino, C.; Macke, N.; Kato, R.; Rowan, S.J. Development, processing and applications of bio-sourced cellulose nanocrystal composites. Progr. Polym. Sci. 2020, 103, 101221. [Google Scholar] [CrossRef]
- Kamel, R.; El-Wakil, N.A.; Dufresne, A.; Elkasabgy, N.A. Nanocellulose: From an agricultural waste to a valuable pharmaceutical ingredient. Int. J. Biol. Macromol. 2020, 163, 1579–1590. [Google Scholar] [CrossRef]
- Sai, T.; Fujita, K. A review of pulmonary toxicity studies of nanocellulose. Inhal. Toxicol. 2020, 32, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Clarkson, C.M.; El Awad Azrak, S.M.; Forti, E.S.; Schueneman, G.T.; Moon, R.J.; Youngblood, J.P. Recent developments in cellulose nanomaterial composites. Adv. Mater. 2021, 33, 2000718. [Google Scholar] [CrossRef]
- Sezali, N.A.A.; Ong, H.L.; Jullok, N.; Villagracia, A.R.; Doong, R.-A. A Review on nanocellulose and its application in supercapacitors. Macromol. Mater. Eng. 2021, 306, 2100556. [Google Scholar] [CrossRef]
- Ahankari, S.S.; Subhedar, A.R.; Bhadauria, S.S.; Dufresne, A. Nanocellulose in food packaging: A review. Carbohydr. Polym. 2021, 255, 117479. [Google Scholar] [CrossRef]
- Hossain, L.; Raghuwanshi, V.S.; Tanner, J.; Garnier, G. Modulating nanocellulose hydrogels and cryogels strength by crosslinking and blending. Colloid. Surf. A 2021, 630, 127608. [Google Scholar] [CrossRef]
- Tie, J.; Chai, H.; Mao, Z.; Zhang, L.; Zhong, Y.; Sui, X.; Xu, H. Nanocellulose-mediated transparent high strength conductive hydrogel based on in-situ formed polypyrrole nanofibrils as a multimodal sensor. Carbohydr. Polym. 2021, 273, 118600. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Liang, Y.; Mei, C.; Cai, L.; Nadda, A.; Van Le, Q.; Peng, Y.; Lam, S.S.; Sonne, C.; Xia, C. Advanced nanocellulose-based gas barrier materials: Present status and prospects. Chemosphere 2022, 286, 131891. [Google Scholar] [CrossRef]
- Norrrahim, M.N.F.; Mohd Kasim, N.A.; Knight, V.F.; Ong, K.K.; Mohd Noor, S.A.; Abdul Halim, N.; Ahmad Shah, N.A.; Jamal, S.H.; Janudin, N.; Misenan, M.S.M.; et al. Emerging developments regarding nanocellulose-based membrane filtration material against microbes. Polymers 2021, 13, 3249. [Google Scholar] [CrossRef] [PubMed]
- Hitam, C.N.C.; Jalil, A.A. Recent advances on nanocellulose biomaterials for environmental health photoremediation: An overview. Environ. Res. 2022, 204, 111964. [Google Scholar] [CrossRef]
- Zhu, J.; Xie, S.; Yang, Z.; Li, X.; Chen, J.; Zhang, X.; Zheng, N. A review of recent advances and prospects on nanocellulose properties and its applications in oil and gas production. J. Nat. Gas Sci. Eng. 2021, 96, 104253. [Google Scholar] [CrossRef]
- Do, T.T.A.; Grijalvo, S.; Imae, T.; Garcia-Celma, M.J.; Rodríguez-Abreu, C. A nanocellulose-based platform towards targeted chemo-photodynamic/photothermal cancer therapy. Carbohydr. Polym. 2021, 270, 118366. [Google Scholar] [CrossRef]
- Marakana, P.G.; Dey, A.; Saini, B. Isolation of nanocellulose from lignocellulosic biomass: Synthesis, characterization, modification and potential applications. J. Environ. Chem. Eng. 2021, 9, 106606. [Google Scholar] [CrossRef]
- Gandini, A.; Belgacem, M.N. Surface and in-depth modification of cellulose fibers. In Cellulose Chemistry and Properties: Materials, Fibers and Composites; Rojas, O., Ed.; A Special Issue of Advances in Polymer Science; Springer: Berlin, Germany, 2016; p. 271. [Google Scholar]
- Rol, F.; Belgacem, M.N.; Gandini, A.; Bras, J. Recent advances in surface-modified cellulose nanofibrils. Progr. Polym. Sci. 2019, 88, 241–264. [Google Scholar] [CrossRef]
- Zhou, L.; Ke, K.; Yang, M.-B.; Yang, W. Recent progress on chemical modification of cellulose for high mechanical-performance poly (lactic acid)/cellulose composite: A review. Comp. Commun. 2021, 23, 100548. [Google Scholar] [CrossRef]
- Onwukamike, K.N.; Grelier, S.; Grau, E.; Cramail, H.; Meier, M.A.R. Critical review on sustainable homogeneous cellulose modification: Why renewability is not enough. ACS Sustain. Chem. Eng. 2019, 7, 1826–1840. [Google Scholar] [CrossRef] [Green Version]
- Samyn, P. Plasma-assisted fibrillation and surface-modification of microfibrillar cellulose. Mater. Lett. 2021, 304, 130615. [Google Scholar] [CrossRef]
- Martinelli, A.; Giannini, L.; Branduardi, P. Enzymatic modification of cellulose to unlock its exploitation in advanced materials. ChemBioChem 2021, 22, 974. [Google Scholar] [CrossRef]
- Heise, K.; Delepierre, G.; King, A.W.T.; Kostiainen, M.A.; Zoppe, J.; Weder, C.; Kontturi, E. Chemical modification of reducing end-groups in cellulose nanocrystals. Angew. Chem. Int. Ed. 2021, 60, 66. [Google Scholar] [CrossRef]
- Avérous, L. Biodegradable multiphase systems based on plasticized starch: A review. J. Macromol. Sci. Pol. R. 2004, 44, 231–274. [Google Scholar] [CrossRef]
- Dang, K.M.; Yoksan, R. Thermoplastic starch blown films with improved mechanical and barrier properties. Int. J. Biol. Macromol. 2021, 188, 290–299. [Google Scholar] [CrossRef]
- Montilla-Buitrago, C.E.; Gómez-López, R.A.; Solanilla-Duque, J.F.; Serna-Cock, L.; Villada-Castillo, H.S. Effect of plasticizers on properties, retrogradation, and processing of extrusion-obtained thermoplastic starch: A review. Starch-Stärke 2021, 73, 2100060. [Google Scholar] [CrossRef]
- Rivadeneira-Velasco, K.E.; Utreras-Silva, C.A.; Díaz-Barrios, A.; Sommer-Márquez, A.E.; Tafur, J.P.; Michell, R.M. Green nanocomposites based on thermoplastic starch: A review. Polymers 2021, 13, 3227. [Google Scholar] [CrossRef]
- Gray, N.; Hamzeh, Y.; Kaboorani, A.; Abdulkhani, A. Influence of cellulose nanocrystal on strength and properties of low density polyethylene and thermoplastic starch composites. Ind. Crops Prod. 2018, 115, 298–305. [Google Scholar] [CrossRef]
- Palai, B.; Biswal, M.; Mohanty, S.; Nayak, S.K. In situ reactive compatibilization of polylactic acid (PLA) and thermoplastic starch (TPS) blends; synthesis and evaluation of extrusion blown films thereof. Ind. Crops Prod. 2019, 141, 111748. [Google Scholar] [CrossRef]
- Cai, Z.; Čadek, D.; Šmejkalová, P.; Kadeřábková, A.; Nová, M.; Kuta, A. The modification of properties of thermoplastic starch materials: Combining potato starch with natural rubber and epoxidized natural rubber. Mater. Today Commun. 2021, 26, 101912. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and chitosan: Properties and applications. Progr. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Dash, M.; Chiellini, F.; Ottenbrite, R.M.; Chiellini, E. Chitosan—A versatile semi-synthetic polymer in biomedical applications. Prog. Polym. Sci. 2011, 36, 981–1014. [Google Scholar] [CrossRef]
- Aranaz, I.; Alcántara, A.R.; Civera, M.C.; Arias, C.; Elorza, B.; Heras Caballero, A.; Acosta, N. Chitosan: An overview of its properties and applications. Polymers 2021, 13, 3256. [Google Scholar] [CrossRef] [PubMed]
- Federer, C.; Kurpiers, M.; Bernkop-Schnürch, A. Thiolated chitosans: A multi-talented class of polymers for various applications. Biomacromolecules 2021, 22, 24–56. [Google Scholar] [CrossRef]
- Burton, R.A.; Gidley, M.J.; Fincher, G.B. Heterogeneity in the chemistry, structure and function of plant cell walls. Nat. Chem. Biol. 2010, 6, 724–741. [Google Scholar] [CrossRef]
- Schutyser, W.; Renders, T.; Van den Bosch, S.; Koelewijn, S.-F.; Beckham, G.T.; Sels, B.F. Chemicals from lignin: An interplay of lignocellulose fractionation, depolymerisation, and upgrading. Chem. Soc. Rev. 2018, 47, 852–908. [Google Scholar] [CrossRef] [PubMed]
- Balakshin, M.; Capanema, E.A.; Zhu, X.; Sulaeva, I.; Potthast, A.; Rosenau, T.; Rojas, O.J. Spruce milled wood lignin: Linear, branched or cross-linked? Green Chem. 2020, 22, 3985–4001. [Google Scholar] [CrossRef]
- Luo, H.; Weeda, E.P.; Alherec, M.; Anson, C.W.; Karlen, S.D.; Cui, Y.; Foster, C.E.; Stahl, S.S. Oxidative catalytic fractionation of lignocellulosic biomass under non-alkaline conditions. J. Am. Chem. Soc. 2021, 143, 15462–15470. [Google Scholar] [CrossRef]
- Crestini, C.; Melone, F.; Sette, M.; Saladino, R. Milled wood lignin: A linear oligomer. Biomacromolecules 2011, 12, 3928–3935. [Google Scholar] [CrossRef]
- McDonough, T.J. The chemistry of organosolv delignification. IPST Tech. Pap. Ser. 1992, 455, 1–17. Available online: http://hdl.handle.net/1853/2069 (accessed on 18 October 2021).
- Chakar, F.S.; Ragauskas, A.J. Review of current and future softwood kraft lignin process chemistry. Ind. Crops Prod. 2004, 20, 131–141. [Google Scholar] [CrossRef]
- Rinaldi, R.; Woodward, R.T.; Ferrini, P.; Rivera, H.J.E. Lignin-first biorefining of lignocellulose: The impact of process severity on the uniformity of lignin oil composition. J. Braz. Chem. Soc. 2019, 30, 479–491. [Google Scholar] [CrossRef]
- Renders, T.; Van den Bosch, S.; Koelewijn, S.-F.; Schutyser, W.; Sels, B.F. Lignin-first biomass fractionation: The advent of active stabilisation strategies. Energ. Environ. Sci. 2017, 10, 1551–1557. [Google Scholar] [CrossRef]
- Alherech, M.; Omolabake, S.; Holland, C.M.; Klinger, G.E.; Hegg, E.L.; Stahl, S.S. From lignin to valuable aromatic chemicals: Lignin depolymerization and monomer separation via centrifugal partition chromatography. ACS Cent. Sci. 2021, 7, 1831–1837. [Google Scholar] [CrossRef] [PubMed]
- Fache, M.; Boutevin, B.; Caillol, S. Vanillin, a key-intermediate of biobased polymers. Eur. Polym. J. 2015, 68, 488–502. [Google Scholar] [CrossRef]
- Gioia, C.; Banella, M.B.; Vannini, M.; Celli, A.; Colonna, M.; Caretti, D. Resorcinol: A potentially bio-based building block for the preparation of sustainable polyesters. Eur. Polym. J. 2015, 73, 38–49. [Google Scholar] [CrossRef]
- Parit, M.; Jiang, Z. Towards lignin derived thermoplastic polymers. Int. J. Biol. Macromol. 2020, 165, 3180–3197. [Google Scholar] [CrossRef] [PubMed]
- Grossman, A.; Vermerris, W. Lignin-based polymers and nanomaterials. Curr. Opin. Biotech. 2019, 56, 112–120. [Google Scholar] [CrossRef] [PubMed]
- Guerra, N.B.; Sant’Ana Pegorin, G.; Boratto, M.H.; Barros, N.R.; Graeff, C.F.O.; Herculano, R.D. Biomedical applications of natural rubber latex from the rubber tree Hevea brasiliensis. Mat. Sci. Eng. C-Mater. 2021, 126, 112126. [Google Scholar] [CrossRef]
- Yamashita, S.; Takahashi, S. Molecular mechanisms of natural rubber biosynthesis. Annu. Rev. Biochem. 2020, 89, 821–851. [Google Scholar] [CrossRef]
- Rippel, M.M.; Galembeck, F. Nanostructures and adhesion in natural rubber: New era for a classic. J. Braz. Chem. Soc. 2009, 20, 1024–1030. [Google Scholar] [CrossRef] [Green Version]
- Roy, K.; Debnath, S.C.; Pongwisuthiruchte, A.; Potiyaraj, P. Recent advances of natural fibers based green rubber composites: Properties, current status, and future perspectives. J. Appl. Polym. Sci. 2021, 138, e50866. [Google Scholar] [CrossRef]
- Wichaita, W.; Promlok, D.; Sudjaipraparat, N.; Sripraphot, S.; Suteewong, T.; Tangboriboonrat, P. A concise review on design and control of structured natural rubber latex particles as engineering nanocomposites. Eur. Polym. J. 2021, 159, 110740. [Google Scholar] [CrossRef]
- Yang, D.; Ni, Y.; Kong, X.; Gao, D.; Wang, Y.; Hu, T.; Zhang, L. Mussel-inspired modification of boron nitride for natural rubber composites with high thermal conductivity and low dielectric constant. Comp. Sci. Technol. 2019, 177, 18–25. [Google Scholar] [CrossRef]
- Wan, L.; Deng, C.; Zhao, Z.-Y.; Chen, H.; Wang, Y.-Z. Flame retardation of natural rubber: Strategy and recent progress. Polymers 2020, 12, 429. [Google Scholar] [CrossRef] [Green Version]
- Ansari, A.H.; Jakarni, F.M.; Muniandy, R.; Hassim, S.; Elahi, Z. Natural rubber as a renewable and sustainable bio-modifier for pavement applications: A review. J. Clean. Prod. 2021, 289, 125727. [Google Scholar] [CrossRef]
- Siracusa, V.; Blanco, I. Bio-polyethylene (bio-PE), bio-polypropylene (bio-PP) and bio-poly(ethylene terephthalate) (bio-PET): Recent developments in bio-based polymers analogous to petroleum-derived ones for packaging and engineering applications. Polymers 2020, 12, 1641. [Google Scholar] [CrossRef]
- Machado, P.G.; Walter, A.; Cunha, M. Bio-based propylene production in a sugarcane biorefinery: A techno-economic evaluation for Brazilian conditions. Biofuels, Bioprod. Bioref. 2016, 10, 623–633. [Google Scholar] [CrossRef]
- Pang, J.; Zheng, M.; Sun, R.; Wang, A.; Wang, X.; Zhang, T. Synthesis of ethylene glycol and terephthalic acid from biomass for producing PET. Green Chem. 2016, 18, 342–359. [Google Scholar] [CrossRef]
- Lee, J.A.; Ahn, J.H.; Kim, I.; Li, S.; Lee, S.Y. Synthesis, characterization, and application of fully biobased and biodegradable nylon-4,4 and -5,4. ACS Sustain. Chem. Eng. 2020, 8, 5604–5614. [Google Scholar] [CrossRef]
- Stouten, J.; Wróblewska, A.A.; Grit, G.; Noordijk, J.; Gebben, B.; Meeusen-Wierts, M.H.M.; Bernaerts, K.V. Polyamides containing a biorenewable aromatic monomer based on coumalate esters: From synthesis to evaluation of the thermal and mechanical properties. Polym. Chem. 2021, 12, 2379–2388. [Google Scholar] [CrossRef]
- Veith, C.; Diot-Néant, F.; Miller, S.A.; Allais, F. Synthesis and polymerization of bio-based acrylates: A review. Polym. Chem. 2020, 11, 7452–7470. [Google Scholar] [CrossRef]
- United States Department of Agriculture. Available online: https://apps.fas.usda.gov/psdonline (accessed on 19 October 2021).
- Gandini, A.; Lacerda, T.M. Polymers from Plant Oils, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA; Scrivener Publishing: Beverly, CA, USA, 2019. [Google Scholar]
- Conti Silva, J.A.; Grilo, L.M.; Gandini, A.; Lacerda, T.M. The prospering of macromolecular materials based on plant oils within the blooming field of polymers from renewable resources. Polymers 2021, 13, 1722. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Huang, J.; Zhu, G.; Yu, X.; Cheng, J.; Liu, Z.; Hu, Y.; Shang, Q.; Liu, C.; Hu, L.; et al. Self-healing, recyclable, and removable UV-curable coatings derived from tung oil and malic acid. Green Chem. 2021, 23, 5875–5886. [Google Scholar] [CrossRef]
- Llevot, A. Sustainable synthetic approaches for the preparation of plant oil-based thermosets. J. Am. Oil Chem. Soc. 2017, 94, 169–186. [Google Scholar] [CrossRef]
- Allasia, M.; Estevez, V.G.; Chesta, A.A.; Baccifava, R.; Gugliotta, L.M.; Igarzabal, C.I.A.; Picchio, M.L.; Minari, R.J. New insights into the properties of alkali-degradable thermosets based on epoxidized soy oil and plant-derived dicarboxylic acids. Polymer 2021, 232, 124143. [Google Scholar] [CrossRef]
- Di Mauro, C.; Genua, A.; Mija, A. Building thermally and chemically reversible covalent bonds in vegetable oil based epoxy thermosets. Influence of epoxy–hardener ratio in promoting recyclability. Mater. Adv. 2020, 1, 1788–1798. [Google Scholar] [CrossRef]
- Di Mauro, C.; Malburet, S.; Genua, A.; Graillot, A.; Mija, A. Sustainable series of new epoxidized vegetable oil-based thermosets with chemical recycling properties. Biomacromolecules 2020, 21, 3923–3935. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.-Z.; Fu, P.; Dai, S.-L.; Zhang, H.-B.; Bi, L.-W.; Jiang, J.-X.; Chen, Y.-X. Catalyst-free self-healing fully bio-based vitrimers derived from tung oil: Strong mechanical properties, shape memory, and recyclability. Ind. Crops Prod. 2021, 171, 113978. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, X.; Liang, D.; Deng, H.; Lin, Z.; Feng, P.; Wang, Q. Rapid self-healing, multiple recyclability and mechanically robust plant oil-based epoxy resins enabled by incorporating tri-dynamic covalent bonding. J. Mater. Chem. A 2021, 9, 18431–18439. [Google Scholar] [CrossRef]
- Lomège, J.; Lapinte, V.; Negrell, C.; Robin, J.-J.; Caillol, S. Fatty acid-based radically polymerizable monomers: From novel poly (meth) acrylates to cutting-edge properties. Biomacromolecules 2019, 20, 4–26. [Google Scholar] [CrossRef]
- Nomura, K.; Awang, N.W.B. Synthesis of bio-based aliphatic polyesters from plant oils by efficient molecular catalysis: A selected survey from recent reports. ACS Sustain. Chem. Eng. 2021, 9, 5486–5505. [Google Scholar] [CrossRef]
- Ahmadi, R.; Ullah, A. Synthesis and characterization of unsaturated biobased-polyamides from plant oil. ACS Sustain. Chem. Eng. 2020, 8, 8049–8058. [Google Scholar] [CrossRef]
- Liang, B.; Zhao, J.; Li, G.; Huang, Y.; Yang, Z.; Yuan, T. Facile synthesis and characterization of novel multi-functional bio-based acrylate prepolymers derived from tung oil and its application in UV-curable coatings. Ind. Crops Prod. 2019, 138, 111585. [Google Scholar] [CrossRef]
- Chu, Z.; Feng, Y.; Xie, B.; Yang, Y.; Hu, Y.; Zhou, X.; Yuan, T.; Yang, Z. Bio-based polyfunctional reactive diluent derived from tung oil by thiol-ene click reaction for high bio-content UV-LED curable coatings. Ind. Crops Prod. 2021, 160, 113117. [Google Scholar] [CrossRef]
- Hu, H.; Chen, M.; Cao, M. TiO2 antifouling coating based on epoxy-modified tung oil waterborne resin. Polym. Polym. Compos. 2021, 29 (Suppl. 9), S521–S529. [Google Scholar] [CrossRef]
- Ismail, N.A.; Khan, A.; Fayyad, E.; Kahraman, R.; Abdullah, A.M.; Shakoor, R.A. Self-healing performance of smart polymeric coatings modified with tung oil and linalyl acetate. Polymers 2021, 13, 1609. [Google Scholar] [CrossRef]
- Ribeiro, B.O.; Valério, V.S.; Gandini, A.; Lacerda, T.M. Copolymers of xylan-derived furfuryl alcohol and natural oligomeric tung oil derivatives. Int. J. Biol. Macromol. 2020, 164, 2497–2511. [Google Scholar] [CrossRef]
- Lacerda, T.M.; Gandini, A. The cationic polymerization of tung oil and its fatty-acid methyl ester. Ind. Crops Prod. 2020, 157, 112886. [Google Scholar] [CrossRef]
- Andreeva, I.V.; Zaitsau, D.H.; Qian, S.; Turovtzev, V.V.; Pimerzin, A.A.; Bara, J.E.; Verevkin, S.P. Glycerol valorisation towards biofuel additivities: Thermodynamic studies of glycerol ethers. Chem. Eng. Sci. 2022, 247, 117032. [Google Scholar] [CrossRef]
- Ripoll, M.; Betancor, L. Oportunities for the valorization of industrial glycerol via biotransformations. Curr. Opp. Green Sust. Chem. 2021, 28, 100430. [Google Scholar] [CrossRef]
- Hejna, A.; Kosmela, P.; Formela, K.; Piszczyk, Ł.; Haponiuk, J.T. Potential applications of crude glycerol in polymer technology–Current state and perspectives. Renew. Sust. Energy Rev. 2016, 66, 449–475. [Google Scholar] [CrossRef]
- Luo, X.; Ge, X.; Cui, S.; Li, Y. Value-added processing of crude glycerol into chemicals and polymers. Bioresour. Technol. 2016, 215, 144–154. [Google Scholar] [CrossRef] [Green Version]
- Ebadipour, N.; Paul, S.; Katryniok, B.; Dumeignil, F. Alkaline-based catalysts for glycerol polymerization reaction: A review. Catalysts 2020, 10, 1021. [Google Scholar] [CrossRef]
- Zhang, H.; Li, H.; Wang, A.; Xu, C.; Yang, S. Progress of catalytic valorization of bio-glycerol with urea into glycerol carbonate as a monomer for polymeric materials. Adv. Polym. Tech. 2020, 2020, 7207068. [Google Scholar] [CrossRef] [Green Version]
- Lang, K.; Bhattacharya, S.; Ning, Z.; Sánchez-Leija, R.J.; Bramson, M.T.K.; Centore, R.; Corr, D.T.; Linhardt, R.J.; Gross, R.A. Enzymatic polymerization of poly (glycerol-1,8-octanediol-sebacate): Versatile poly (glycerol sebacate) analogues that form monocomponent biodegradable fiber scaffolds. Biomacromolecules 2020, 21, 3197–3206. [Google Scholar] [CrossRef]
- Pounds, K.; Jairam, S.; Bao, H.; Meng, S.; Zhang, L.; Godinez, S.; Savin, D.A.; Pelletier, W.; Correll, M.J.; Tong, Z. Glycerol-based dendrimer nanocomposite film as a tunable pH-Sensor for food packaging. ACS Appl. Mater. Inter. 2021, 13, 23268–23281. [Google Scholar] [CrossRef]
- Goyal, S.; Lin, F.-Y.; Forrester, M.; Henrichsen, W.; Murphy, G.; Shen, L.; Wang, T.-P.; Cochran, E.W. Glycerol ketals as building blocks for a new class of biobased (meth) acrylate polymers. ACS Sustain. Chem. Eng. 2021, 9, 10620–10629. [Google Scholar] [CrossRef]
- Yang, M.; Rosentrater, K.A. Cradle-to-gate life cycle assessment of structural bio-adhesives derived from glycerol. Int. J. Life Cycle Assess. 2021, 26, 799–806. [Google Scholar] [CrossRef]
- Tong, X.; Ma, Y.; Li, Y. Biomass into chemicals: Conversion of sugars to furan derivatives by catalytic processes. Appl. Catal. A-Gen. 2010, 385, 1–13. [Google Scholar] [CrossRef]
- Gandini, A.; Belgacem, M.N. Furans in polymer chemistry. Progr. Polym. Sci. 1997, 22, 1203–1379. [Google Scholar] [CrossRef]
- Moreau, C.; Belgacem, M.N.; Gandini, A. Recent catalytic advances in the chemistry of substituted furans from carbohydrates and in the ensuing polymers. Top. Catal. 2004, 27, 11–30. [Google Scholar] [CrossRef]
- Gandini, A.; Belgacem, M.N. Furan Derivatives and Furan Chemistry at the Service of Macromolecular Materials. In Monomers, Polymers and Composites from Renewable Resources; Elsevier: Amsterdam, The Netherlands, 2008; pp. 115–152. [Google Scholar]
- Gandini, A. Furans as offspring of sugars and polysaccharides and progenitors of a family of remarkable polymers: A review of recent progress. Polym. Chem. 2010, 1, 245–251. [Google Scholar] [CrossRef]
- Decostanzi, M.; Auvergne, R.; Boutevin, B.; Caillol, S. Biobased phenol and furan derivative coupling for the synthesis of functional monomers. Green Chem. 2019, 21, 724–747. [Google Scholar] [CrossRef]
- Li, C.; Li, J.; Qin, L.; Yang, P.; Vlachos, D.G. Recent advances in the photocatalytic conversion of biomass-derived furanic compounds. ACS Catal. 2021, 11, 11336–11359. [Google Scholar] [CrossRef]
- Lalanne, L.; Nyanhongo, G.S.; Guebitz, G.M.; Pellis, A. Biotechnological production and high potential of furan-based renewable monomers and polymers. Biotechnol. Adv. 2021, 48, 107707. [Google Scholar] [CrossRef]
- Lee, J.; Baek, S.; Moon, H.H.; Son, S.U.; Song, C. Furandiacylazide: A biomass-derived versatile polymer platform toward photodegradable and nonflammable polyurethanes. ACS Appl. Polym. Mater. 2021, 3, 5767–5777. [Google Scholar] [CrossRef]
- Zhu, J.; Lei, Y.; Chen, Z.; Liao, Y.; Yin, G. Feasible synthesis of bifurfural from renewable furfural derived 5-bromofurfural for polymerization. Mol. Catal. 2021, 513, 111814. [Google Scholar] [CrossRef]
- Singhal, S.; Agarwal, S.; Mudoi, M.P.; Singhal, N.; Singh, R. Chemical conversion of furan dicarboxylic acid to environmentally benign polyesters: An overview. Biomass Conv. Bioref. 2021. [Google Scholar] [CrossRef]
- Ma, K.; Jiang, H.; Chen, G.; Wang, W.; Zhang, Y. Polyimides from 2,5-bis[4-(4-aminophenoxy)benzoyl]furan and their thermal crosslinking reaction. High Perform. Polym. 2021. [Google Scholar] [CrossRef]
- Bianchi, E.; Soccio, M.; Siracusa, V.; Gazzano, M.; Thiyagarajan, S.; Lotti, N. Poly (butylene 2,4-furanoate), an added member to the class of smart furan-based polyesters for sustainable packaging: Structural isomerism as a key to tune the final properties. ACS Sustain. Chem. Eng. 2021, 9, 11937–11949. [Google Scholar] [CrossRef]
- Morales-Huerta, J.C.; Ilarduya, A.M.; León, S.; Muñoz-Guerra, S. Isomannide-containing poly (butylene 2,5-furandicarboxylate) copolyesters via ring-opening polymerization. Macromolecules 2018, 51, 3340–3350. [Google Scholar] [CrossRef]
- Gandini, A. The furan/maleimide Diels-Alder reaction: A versatile clink-unclick tool in macromolecular synthesis. Prog. Polym. Sci. 2013, 38, 1–29. [Google Scholar] [CrossRef]
- Thiyagarajan, S.; Genuino, H.C.; van der Waal, J.C.; de Jong, E.; Weckhuysen, B.M.; van Haveren, J.; Bruijnincx, P.C.A.; van Es, D.S. A facile solid-phase route to renewable aromatic chemicals from biobased furanics. Angew. Chem. Int. Ed. 2016, 55, 1368. [Google Scholar] [CrossRef]
- Cioc, R.C.; Smak, T.J.; Crockatt, M.; van der Waal, J.C.; Bruijnincx, P.C.A. Furoic acid and derivatives as atypical dienes in Diels-Alder reactions. Green Chem. 2021, 23, 5503–5510. [Google Scholar] [CrossRef]
- Shao, X.; Su, L.; Zhang, J.; Tian, Z.; Zhang, N.; Wang, Y.; Wang, H.; Cui, X.; Hou, X.; Deng, T. Green production of phthalic anhydride from biobased furan and maleic anhydride by an acid resin catalyst. ACS Sustain. Chem. Eng. 2021, 9, 14385–14394. [Google Scholar] [CrossRef]
- Gevrek, T.N.; Sanyal, A. Furan-containing polymeric materials: Harnessing the Diels-Alder chemistry for biomedical applications. Eur. Polym. J. 2021, 153, 110514. [Google Scholar] [CrossRef]
- Nossa, T.S.; Belgacem, N.M.; Gandini, A.; Carvalho, A.J. Thermoreversible crosslinked thermoplastic starch. Polym. Int. 2015, 64, 1366–1372. [Google Scholar] [CrossRef]
- Kramer, R.K.; Belgacem, M.N.; Carvalho, A.J.F.; Gandini, A. Thermally reversible nanocellulose hydrogels synthesized via the furan/maleimide Diels-Alder click reaction in water. Int. J. Biol. Macromol. 2019, 141, 493–498. [Google Scholar] [CrossRef] [PubMed]
- Trovatti, E.; Cunha, A.G.; Carvalho, A.J.F.; Gandini, A. Furan-modified natural rubber: A substrate for its reversible crosslinking and for clicking it onto nanocellulose. Int. J. Biol. Macromol. 2017, 95, 762–768. [Google Scholar] [CrossRef]
- Trovatti, E.; Lacerda, T.M.; Carvalho, A.J.F.; Gandini, A. Recycling tires? Reversible crosslinking of poly (butadiene). Adv. Mater. 2015, 27, 2242–2245. [Google Scholar] [CrossRef] [PubMed]
- Gandini, A.; Carvalho, A.J.F.; Trovatti, E.; Kramer, R.K.; Lacerda, T.M. Macromolecular materials based on the application of the Diels-Alder reaction to natural polymers and plant oil. Eur. J. Lipid Sci. Technol. 2018, 120, 1700091. [Google Scholar] [CrossRef]
- Banella, M.B.; Giacobazzi, G.; Vannini, M.; Marchese, P.; Colonna, M.; Celli, A.; Gandini, A.; Gioia, C. A novel approach for the synthesis of thermo-responsive co-polyesters incorporating reversible Diels-Alder adducts. Macromol. Chem. Phys. 2019, 220, 1900247. [Google Scholar] [CrossRef]
- Pandey, S.; Rajput, B.S.; Chikkali, S.H. Refining plant oils and sugars to platform chemicals, monomers, and polymers. Green Chem. 2021, 23, 4255–4295. [Google Scholar] [CrossRef]
- Galbis, J.A.; García-Martín, M.G.; Violante de Paz, M.; Galbis, E. Synthetic polymers from sugar-based monomers. Chem. Rev. 2016, 116, 1600–1636. [Google Scholar] [CrossRef]
- Wang, J.; Wang, D.; Zhang, Y.; Dong, J. Synthesis and biopharmaceutical applications of sugar-based polymers: New advances and future prospects. ACS Biomater. Sci. Eng. 2021, 7, 963–982. [Google Scholar] [CrossRef] [PubMed]
- Rose, M.; Palkovits, R. Isosorbide as a renewable platform chemical for versatile applications–quo vadis? ChemSusChem 2012, 5, 167–176. [Google Scholar] [CrossRef]
- Saxon, D.J.; Luke, A.M.; Sajjad, H.; Tolman, W.B.; Reineke, T.M. Next-generation polymers: Isosorbide as a renewable alternative. Progr. Polym. Sci. 2020, 101, 101196. [Google Scholar] [CrossRef]
- Saxon, D.J.; Nasiri, M.; Mandal, M.; Maduskar, S.; Dauenhauer, P.J.; Cramer, C.J.; LaPointe, A.M.; Reineke, T.M. Architectural control of isosorbide-based polyethers via ring-opening polymerization. J. Am. Chem. Soc. 2019, 141, 5107–5111. [Google Scholar] [CrossRef] [Green Version]
- Matt, L.; Liblikas, I.; Bonjour, O.; Jannasch, P.; Vares, L. Synthesis and anionic polymerization of isosorbide mono-epoxides for linear biobased polyethers. Polym. Chem. 2021, 12, 5937–5941. [Google Scholar] [CrossRef]
- Marie, B.; Clark, R.; Gillece, T.; Ozkan, S.; Jaffe, M.; Ravindra, N.M. Hydrophobically modified isosorbide dimethacrylates as a bisphenol-A (BPA)-free dental filling material. Materials 2021, 14, 2139. [Google Scholar] [CrossRef] [PubMed]
- Saska, J.; Dutta, S.; Kindler, A.; Zuend, S.J.; Mascal, M. Efficient and scalable production of isoidide from isosorbide. ACS Sust. Chem. Eng. 2021, 9, 11565–11570. [Google Scholar] [CrossRef]
- Silvestre, J.D.; Gandini, A. Terpenes: Major Sources, Properties and Applications. In Monomers, Polymers and Composites from Renewable Resources; Elsevier: Amsterdam, The Netherlands, 2008; pp. 17–38. [Google Scholar]
- Yarolimek, M.R.; Bookbinder, H.R.; Coia, B.M.; Kennemur, J.G. Ring-opening metathesis polymerization of δ-pinene: Well-defined polyolefins from pine sap. ACS Macro Lett. 2021, 10, 760–766. [Google Scholar] [CrossRef]
- Yarolimek, M.R.; Coia, B.M.; Bookbinder, H.R.; Kennemur, J.G. Investigating the effect of α-pinene on the ROMP of δ-pinene. Polym. Chem. 2021, 12, 5048–5058. [Google Scholar] [CrossRef]
- Atkinson, R.L.; Monaghan, O.R.; Elsmore, M.T.; Topham, P.D.; Toolan, D.T.W.; Derry, M.J.; Taresco, V.; Stockman, R.A.; De Focatiis, D.S.A.; Irvine, D.J.; et al. RAFT polymerisation of renewable terpene (meth) acrylates and the convergent synthesis of methacrylate–acrylate–methacrylate triblock copolymers. Polym. Chem. 2021, 12, 3177–3189. [Google Scholar] [CrossRef]
- Dev, A.; Rösler, A.; Schlaad, H. Limonene as a renewable unsaturated hydrocarbon solvent for living anionic polymerization of β-myrcene. Polym. Chem. 2021, 12, 3084–3087. [Google Scholar] [CrossRef]
- Della Monica, F.; Kleij, A.W. From terpenes do sustainable and functional polymers. Polym. Chem. 2020, 11, 5109–5127. [Google Scholar] [CrossRef]
- Li, H.; Shang, Y.; Huang, W.; Xue, B.; Zhang, X.; Cui, Z.; Fu, P.; Pang, X.; Zhao, Q.; Liu, M. Synthesis of succinic acid-based polyamide through direct solid-state polymerization method: Avoiding cyclization of succinic acid. J. Appl. Polym. Sci. 2021, 138, e51017. [Google Scholar] [CrossRef]
- Verma, M.; Mandyal, P.; Singh, D.; Gupta, N. Recent developments in heterogeneous catalytic routes for the sustainable production of succinic acid from biomass resources. ChemSusChem 2020, 13, 4026–4034. [Google Scholar] [CrossRef] [PubMed]
- Pyo, S.-H.; Park, J.H.; Srebny, V.; Hatti-Kaul, R. A sustainable synthetic route for biobased 6-hydroxyhexanoic acid, adipic acid and ε-caprolactone by integrating bio- and chemical catalysis. Green Chem. 2020, 22, 4450–4455. [Google Scholar] [CrossRef]
- Teleky, B.-E.; Vodnar, D.C. Biomass-derived production of itaconic acid as a building block in specialty polymers. Polymers 2019, 11, 1035. [Google Scholar] [CrossRef] [Green Version]
- Sollka, L.; Lienkamp, K. Progress in the free and controlled radical homo- and co-polymerization of itaconic acid derivatives: Toward functional polymers with controlled molar mass distribution and architecture. Macromol. Rapid Commun. 2021, 42, 2000546. [Google Scholar] [CrossRef]
- Wang, X.; Gao, S.; Wang, J.; Xu, S.; Li, H.; Chen, K.; Ouyang, P. The production of biobased diamines from renewable carbon sources: Current advances and perspectives. Chin. J. Chem. Eng. 2021, 30, 4–13. [Google Scholar] [CrossRef]
- Olmo, C.; Casas, M.T.; Martínez, J.C.; Franco, L.; Puiggalí, J. Crystalline structures and structural transitions of copolyamides derived from 1,4-diaminobutane and different ratios of glutaric and azelaic acids. Polymers 2019, 11, 572. [Google Scholar] [CrossRef] [Green Version]
- Song, L.; Wang, R.; Che, L.; Jiang, Y.; Zhou, M.; Zhao, Y.; Pang, J.; Jiang, M.; Zhou, G.; Zheng, M.; et al. Catalytic aerobic oxidation of lignocellulose-derived levulinic acid in aqueous solution: A novel route to synthesize dicarboxylic acids for bio-based polymers. ACS Catal. 2021, 11, 11588–11596. [Google Scholar] [CrossRef]
- Valsange, N.G.; Gonzalez, M.N.G.; Warlin, N.; Mankar, S.V.; Rehnberg, N.; Lundmark, S.; Zhang, B.; Jannasch, P. Biobased aliphatic polyesters from a spirocyclic dicarboxylate monomer derived from levulinic acid. Green Chem. 2021, 23, 5706–5723. [Google Scholar] [CrossRef]
- Ge, M.; Miao, J.-T.; Zhang, K.; Wu, Y.; Zheng, L.; Wu, L. Building biobased, degradable, flexible polymer networks from vanillin via thiol–ene “click” photopolymerization. Polym. Chem. 2021, 12, 564–571. [Google Scholar] [CrossRef]
- Gallos, A.; Crowet, J.-M.; Michely, L.; Raghuwanshi, V.S.; Mention, M.M.; Langlois, V.; Dauchez, M.; Garnier, G.; Allais, F. Blending ferulic acid derivatives and polylactic acid into biobased and transparent elastomeric materials with shape memory properties. Biomacromolecules 2021, 22, 1568–1578. [Google Scholar] [CrossRef]
- Pospiech, D.; Korwitz, A.; Komber, H.; Jehnichen, D.; Arnhold, K.; Brünig, H.; Scheibner, H.; Müller, M.T.; Voit, B. Polyesters with bio-based ferulic acid units: Crosslinking paves the way to property consolidation. Polym. Chem. 2021, 12, 5139–5148. [Google Scholar] [CrossRef]
- Dreava, D.M.; Benea, I.C.; Bîtcan, I.; Todea, A.; Sisu, E.; Puiu, M.; Peter, F. Biocatalytic approach for novel functional oligoesters of ε-caprolactone and malic acid. Processes 2021, 9, 232. [Google Scholar] [CrossRef]
- Yang, R.; Li, M.; Zhang, X.; Li, J. Preparation of a catalyst-free and water-blown rigid polyurethane foam from malic-co-citric acid-based polyols. Ind. Crops Prod. 2021, 169, 113648. [Google Scholar] [CrossRef]
- Plastics Europe, Plastics-the Facts 2020. Available online: https://plasticseurope.org/sustainability/climate/sustainable-feedstocks/renewable-resources-bioplastics/ (accessed on 8 November 2021).
- Meereboer, K.W.; Misra, M.; Mohanty, A.K. Review of recent advances in the biodegradability of polyhydroxyalkanoate (PHA) bioplastics and their composites. Green Chem. 2020, 22, 5519–5558. [Google Scholar] [CrossRef]
- Nakayama, A.; Yamano, N.; Kawasaki, N. Biodegradation in seawater of aliphatic polyesters. Polym. Degrad. Stabil. 2019, 166, 290–299. [Google Scholar] [CrossRef]
- Vikhareva, I.N.; Buylova, E.A.; Yarmuhametova, G.U.; Aminova, G.K.; Mazitova, A.K. An overview of the main trends in the creation of biodegradable polymer materials. J. Chem. 2021, 2021, 5099705. [Google Scholar] [CrossRef]
- Rai, P.; Mehrotra, S.; Priya, S.; Gnansounou, E.; Sharma, S.K. Recent advances in the sustainable design and applications of biodegradable polymers. Biores. Technol. 2021, 325, 124739. [Google Scholar] [CrossRef] [PubMed]
- Colnik, M.; Hrncic, M.K.; Skerget, M.; Knez, Z. Biodegradable polymers, current trends of research and their applications, a review. Chem. Ind. Chem. Eng. Q. 2020, 26, 401–418. [Google Scholar] [CrossRef]
- RameshKumar, S.; Shaiju, P.; O’Connor, K.E.; Babu P, R. Bio-based and biodegradable polymers–state-of-the-art, challenges and emerging trends. Curr. Opin. Green Sustain. Chem. 2020, 21, 75–81. [Google Scholar] [CrossRef]
- Wu, F.; Misra, M.; Mohanty, A.K. Challenges and new opportunities on barrier performance of biodegradable polymers for sustainable packaging. Prog. Polym. Sci. 2021, 117, 101395. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gandini, A.; M. Lacerda, T. Monomers and Macromolecular Materials from Renewable Resources: State of the Art and Perspectives. Molecules 2022, 27, 159. https://doi.org/10.3390/molecules27010159
Gandini A, M. Lacerda T. Monomers and Macromolecular Materials from Renewable Resources: State of the Art and Perspectives. Molecules. 2022; 27(1):159. https://doi.org/10.3390/molecules27010159
Chicago/Turabian StyleGandini, Alessandro, and Talita M. Lacerda. 2022. "Monomers and Macromolecular Materials from Renewable Resources: State of the Art and Perspectives" Molecules 27, no. 1: 159. https://doi.org/10.3390/molecules27010159
APA StyleGandini, A., & M. Lacerda, T. (2022). Monomers and Macromolecular Materials from Renewable Resources: State of the Art and Perspectives. Molecules, 27(1), 159. https://doi.org/10.3390/molecules27010159