Synthesis and Evaluation of Chalcone Derivatives as Novel Sunscreen Agent
Abstract
1. Introduction
2. Results and Discussion
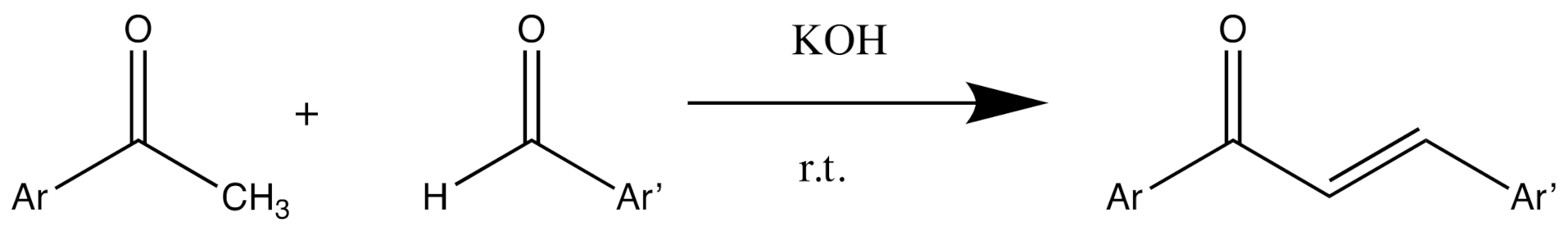
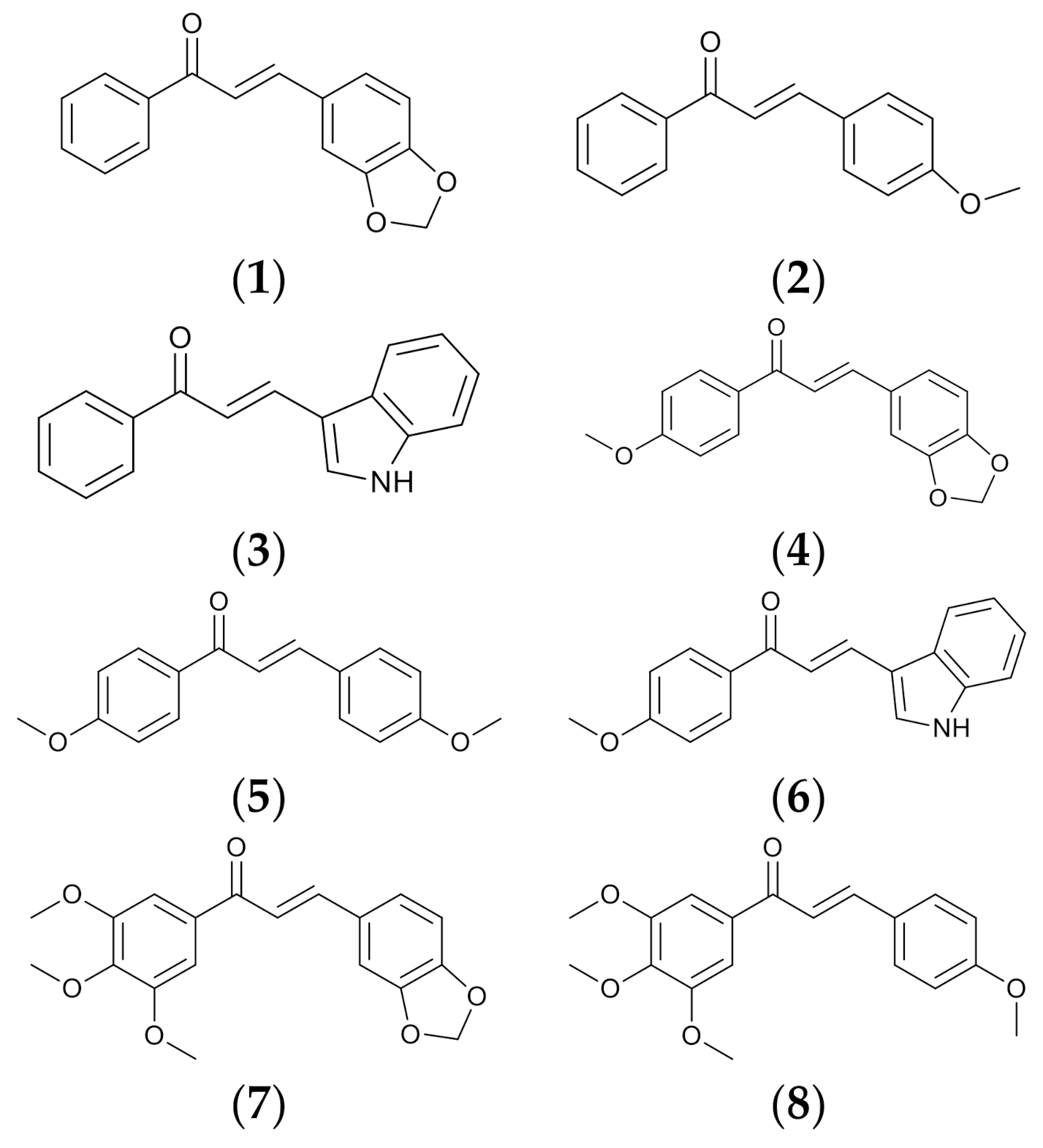
2.1. Synthesis of Chalcone Derivatives
2.2. UV Absorbance Profile of Chalcone Derivatives
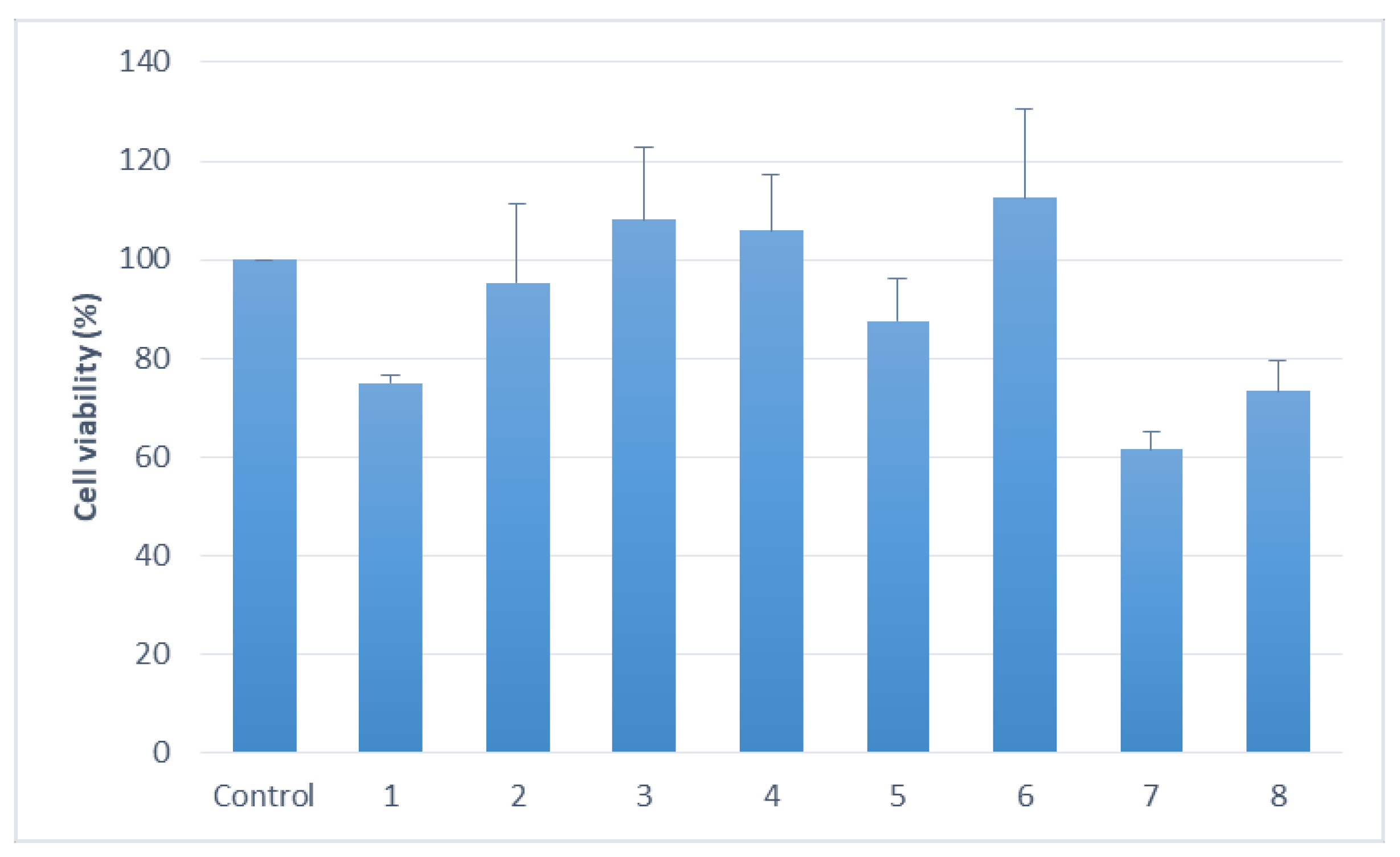
2.3. Cytotoxicity Assay
3. Materials and Methods
3.1. Materials
3.2. Instruments Analysis
3.3. General Procedure Synthesis of Chalcone Derivatives
3.4. Determination of the UV–Vis Absorbance Profile of Chalcone Derivatives
3.5. Computational Study
3.6. Cytotoxicity Assay
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Sisto, R.; Borra, M.; Casale, G.R.; Militello, A.; Siani, A.M. Quantitative evaluation of personal exposure to UV radiation of workers and general public. Radiat. Prot. Dosim. 2009, 137, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Scientific Committee on Emerging and Newly Identified Health Risks Preliminary. Opinion on Biological Effects of Ultraviolet Radiation Relevant to Health with Particular Reference to Sunbeds for Cosmetic Purposes; European Commission: Brussels, Belgium, 2015; pp. 1–97. [Google Scholar]
- Ateenyi-Agaba, C.; Dai, M.; Calvez, F.L.; Katongole-Mbidde, E.; Smet, A.; Tommasino, M.; Franceschi, S.; Hainaut, P.; Weiderpass, E. TP53 mutations in squamous-cell carcinomas of the conjunctiva: Evidence for UV-induced mutagenesis. Mutagenesis 2004, 19, 399–401. [Google Scholar] [CrossRef] [PubMed]
- D’Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV radiation and the skin. Int. J. Mol. Sci. 2013, 14, 12222–12248. [Google Scholar] [CrossRef] [PubMed]
- Black, A.T.; Gray, J.P.; Shakarjian, M.P.; Laskin, D.L.; Heck, D.E.; Laskin, J.D. Distinct effects of ultraviolet B light on antioxidant expression in undifferentiated and differentiated mouse keratinocytes. Carcinogenesis 2008, 29, 219–225. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kelly, J.; Murphy, J.E.J. Mitochondrial tolerance to single and repeat exposure to simulated sunlight in human epidermal and dermal skin cells. J. Photochem. Photobiol. B Biol. 2016, 165, 298–304. [Google Scholar] [CrossRef]
- Lawrence, K.P.; Douki, T.; Sarkany, R.P.E.; Acker, S.; Herzog, B.; Young, A.R. The UV/visible radiation boundary region (385–405 nm) damages skin cells and induces ‘dark’ cyclobutane pyrimidine dimers in human skin In Vivo. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Day, C.P.; Marchalik, R.; Merlino, G.; Michael, H. Mouse models of UV-induced melanoma: Genetics, pathology, and clinical relevance. Lab. Investig. 2017, 97, 698–705. [Google Scholar] [CrossRef]
- Geoffrey, K.; Mwangi, A.N.; Maru, S.M. Sunscreen products: Rationale for use, formulation development and regulatory considerations. Saudi Pharm. J. 2019, 27, 1009–1018. [Google Scholar] [CrossRef]
- Kohli, I.; Nicholson, C.L.; Williams, J.D.; Lyons, A.B.; Seo, I.; Maitra, P.; Tian, X.; Atillasoy, E.; Lim, H.W.; Hamzavi, I.H. Greater efficacy of SPF 100+ sunscreen compared with SPF 50+ in sunburn prevention during 5 consecutive days of sunlight exposure: A randomized, double-blind clinical trial. J. Am. Acad. Dermatol. 2020, 82, 869–877. [Google Scholar] [CrossRef]
- Aspinwall, L.G.; Taber, J.M.; Kohlmann, W.; Leaf, S.L.; Leachman, S.A. Unaffected family members report improvements in daily routine sun protection 2 years following melanoma genetic testing. Genet. Med. 2014, 16, 846–853. [Google Scholar] [CrossRef]
- Anonymous. OTC Active Ingredients. Available online: https://www.fda.gov/media/75758/download (accessed on 14 January 2021).
- Lionetti, N.; Rigano, L. The new sunscreens among formulation strategy, stability issues, changing norms, safety and efficacy evaluations. Cosmetics 2017, 4, 15. [Google Scholar] [CrossRef]
- Mota, M.D.; Morte, A.N.B.; Silva, L.C.R.C.; Chinalia, F.A. Sunscreen protection factor enhancement through supplementation with Rambutan (Nephelium lappaceum L.) ethanolic extract. J. Photochem. Photobiol. B Biol. 2019, 205, 111837. [Google Scholar] [CrossRef]
- Twilley, D.; Moodley, D.; Rolfes, H.; Moodley, I.; McGaw, L.J.; Madikizela, B.; Summers, B.; Raaff, L.; Lategan, M.; Kgatuke, L.; et al. Ethanolic extracts of South African plants, Buddleja saligna Willd. and Helichrysum odoratissimum (L.) Sweet, as multifunctional ingredients in sunscreen formulations. S. Afr. J. Bot. 2021, 137, 171–182. [Google Scholar] [CrossRef]
- Da Silva, A.C.P.; Paiva, J.P.; Diniz, R.R.; Anjos, V.M.; Silva, A.B.S.M.; Pinto, A.V.; Santos, E.P.; Leitao, A.C.; Cabral, L.M.; Rodrigues, C.R.; et al. Photoprotection assessment of olive (Olea europaea L.) leaves extract standardized to oleuropein: In vitro and in silico approach for improved sunscreens. J. Photochem. Photobiol. B Biol. 2019, 193, 162–171. [Google Scholar] [CrossRef]
- Santhanam, R.L.; Akhtar, M.T.; Ahmad, S.; Abas, F.; Ismail, I.S.; Rukayadi, Y.; Shaari, K. Utilization of the ethyl acetate fraction of Zanthoxylum rhetsa bark extract as an active ingredient in natural sunscreen formulations. Ind. Crop. Prod. 2017, 96, 165–172. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Sherje, A.P. Development of resveratrol and green tea sunscreen formulation for combined photoprotective and antioxidant properties. J. Drug Deliv. Sci. Technol. 2020, 60, 1–6. [Google Scholar] [CrossRef]
- Amador-Castro, F.; Rodriguez-Martinez, V.; Carrillo-Nieves, D. Robust natural ultraviolet filters from marine ecosystems for the formulation of environmental friendlier bio-sunscreens. Sci. Total Environ. 2020, 749, 141576. [Google Scholar] [CrossRef]
- Lavakumar, S.; Vivekanand, P.A.; Prince, A.A.M. Simultaneous analysis of octylmethoxycinnamate and butylmethoxydibenzoylmethane in sunscreen products by a validated UV-spectrophotometric method. Mater. Today Proc. 2021, 36, 893–897. [Google Scholar] [CrossRef]
- Cocci, P.; Mosconi, G.; Palermo, F.A. Sunscreen active ingredients in loggerhead turtles (Caretta caretta) and their relation to molecular markers of inflammation, oxidative stress and hormonal activity in wild populations. Mar. Pollut. Bull. 2020, 153, 111012. [Google Scholar] [CrossRef]
- Calafat, A.M.; Wong, L.Y.; Ye, X.; Reidy, J.A.; Needham, L.L. Concentrations of the sunscreen agent benzophenone-3 in residents of the United States: National Health and Nutrition Examination Survey 2003-2004. Environ. Health Perspect. 2008, 116, 893–897. [Google Scholar] [CrossRef]
- Aksöz, B.E.; Ertan, R. Spectral properties of chalcones II. FABAD J. Pharm. Sci. 2012, 37, 205–216. [Google Scholar]
- Borges, I.D.; Danielli, J.A.V.; Silva, V.E.G.; Sallum, L.O.; Queiroz, J.E.; Dias, L.D.; Iermak, I.; Aquino, G.L.B.; Camargo, A.J.; Valverde, C.; et al. Synthesis and structural studies on (: E)-3-(2,6-difluorophenyl)-1-(4-fluorophenyl) prop-2-en-1-one: A promising nonlinear optical material. RSC Adv. 2020, 10, 22542–22555. [Google Scholar] [CrossRef]
- Wheeler, O.H.; Gore, P.H.; Santiago, M.; Baez, R. Ultraviolet absorption of substituted phenyl and polycyclic aryl chalcones. Can. J. Chem. 1964, 42, 2580–2583. [Google Scholar] [CrossRef]
- Ismiyarto, I.; Matsjeh, S.; Anwar, C. Synthesis of chalcone and flavanone compound using raw material of acetophenone and benzaldehyde derivative. Indones. J. Chem. 2001, 1, 81–89. [Google Scholar] [CrossRef]
- Desai, V.; Desai, S.; Naik, S.; Palyekar, U.; Joshi, S.D.; Dixit, S.K. Novel quinoxalinyl chalcone hybrid scaffolds as enoyl ACP reductase inhibitors: Synthesis, molecular docking and biological evaluation. Bioorg. Med. Chem. Lett. 2017, 27, 2174–2180. [Google Scholar] [CrossRef]
- Khairul, W.M.; Izzati, A.; Augustine, E.; Arshad, S.; Abdul, I. FT-IR, NMR and X-ray crystallography dataset for newly synthesized alkoxy-chalcone featuring phenyl)prop–2-en-1-one. Chem. Data Collect. 2020, 28, 100473. [Google Scholar] [CrossRef]
- Wang, L.; Wang, Y.; Tian, Y.; Shang, J.; Sun, X.; Chen, H.; Wang, H.; Tan, W. Design, synthesis, biological evaluation, and molecular modeling studies of chalcone-rivastigmine hybrids as cholinesterase inhibitors. Bioorg. Med. Chem. 2016, 25, 360–371. [Google Scholar] [CrossRef]
- Nuti, E.; Bassani, B.; Camodeca, C.; Rosalia, L.; Cantelmo, A.; Gallo, C.; Baci, D.; Bruno, A.; Orlandini, E.; Nencetti, S.; et al. Synthesis and antiangiogenic activity study of new hop chalcone Xanthohumol analogues. Eur. J. Med. Chem. 2017, 138, 890–899. [Google Scholar] [CrossRef]
- Ibnaouf, K.H.; Elzupir, A.O.; Alsalhi, M.S.; Alaamer, A.S. Influence of functional groups on the photophysical properties of dimethylamino chalcones as laser dyes. Opt. Mater. 2018, 76, 216–221. [Google Scholar] [CrossRef]
- Coskun, D.; Erkisa, M.; Ulukaya, E.; Fatih, M.; Ari, F. Novel 1-(7-ethoxy-1-benzofuran-2-yl) substituted chalcone derivatives: Synthesis, characterization and anticancer activity. Eur. J. Med. Chem. 2017, 136, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Trujillo, A.; Ocayo, F.; Artigas, V.; Santos, J.C.; Ulloa, P.J.; Kahlal, S.; Saillard, J.Y.; Fuentealba, M.; Escobar, C.A. New ferrocenyl-chalcones and bichalcones: Synthesis and characterization. Tetrahedron Lett. 2017, 58, 437–441. [Google Scholar] [CrossRef]
- Prabhu, S.R.; Jayarama, A.; Chandrasekharan, K.; Upadhyaya, V. Synthesis, growth, structural characterization, Hirshfeld analysis and nonlinear optical studies of a methyl substituted chalcone. J. Mol. Struct. 2017, 1136, 244–252. [Google Scholar] [CrossRef]
- Ali, M.K.M.; Elzupir, A.O.; Ibrahem, M.A.; Suliman, I.I.; Modwi, A.; Idriss, H.; Ibnaouf, K.H. Characterization of optical and morphological properties of chalcone thin films for optoelectronics applications. Optik 2017, 145, 529–533. [Google Scholar] [CrossRef]
- Jumina, J.; Styaningrum, R.W.; Siswanta, D.; Triono, S.; Priastomo, Y.; Harizal, H.; Sholikhah, E.N.; Zulkarnain, A.K. Synthesis and preliminary evaluation of several chalcone derivatives as sunscreen compounds. Chem. J. Mold. 2019, 14, 90–96. [Google Scholar] [CrossRef]
- Riss, T.L.; Moravec, R.A.; Niles, A.L.; Duellman, S.; Benink, H.A.; Worzella, T.J. Cell viability assays. In Assay Guidance Manual; Eli Lily & Co.; The National Center for Advancing Translational Sciences: Bethesda, MD, USA, 2016; pp. 335–359. [Google Scholar]
- Kim, H.I.; Jeong, Y.U.; Kim, J.H.; Park, Y.J. 3,5,6,7,8,30,40-Heptamethoxyflavone, a citrus flavonoid, inhibits collagenase activity and induces type I procollagen synthesis in HDFn cells. Int. J. Mol. Sci. 2018, 19, 620. [Google Scholar]
- Kizhedath, A.; Wilkinson, S.; Glassey, J. Assessment of hepatotoxicity and dermal toxicity of butyl paraben and methyl paraben using HepG2 and HDFn in vitro models. Toxicol. Vitro 2019, 55, 108–115. [Google Scholar] [CrossRef]
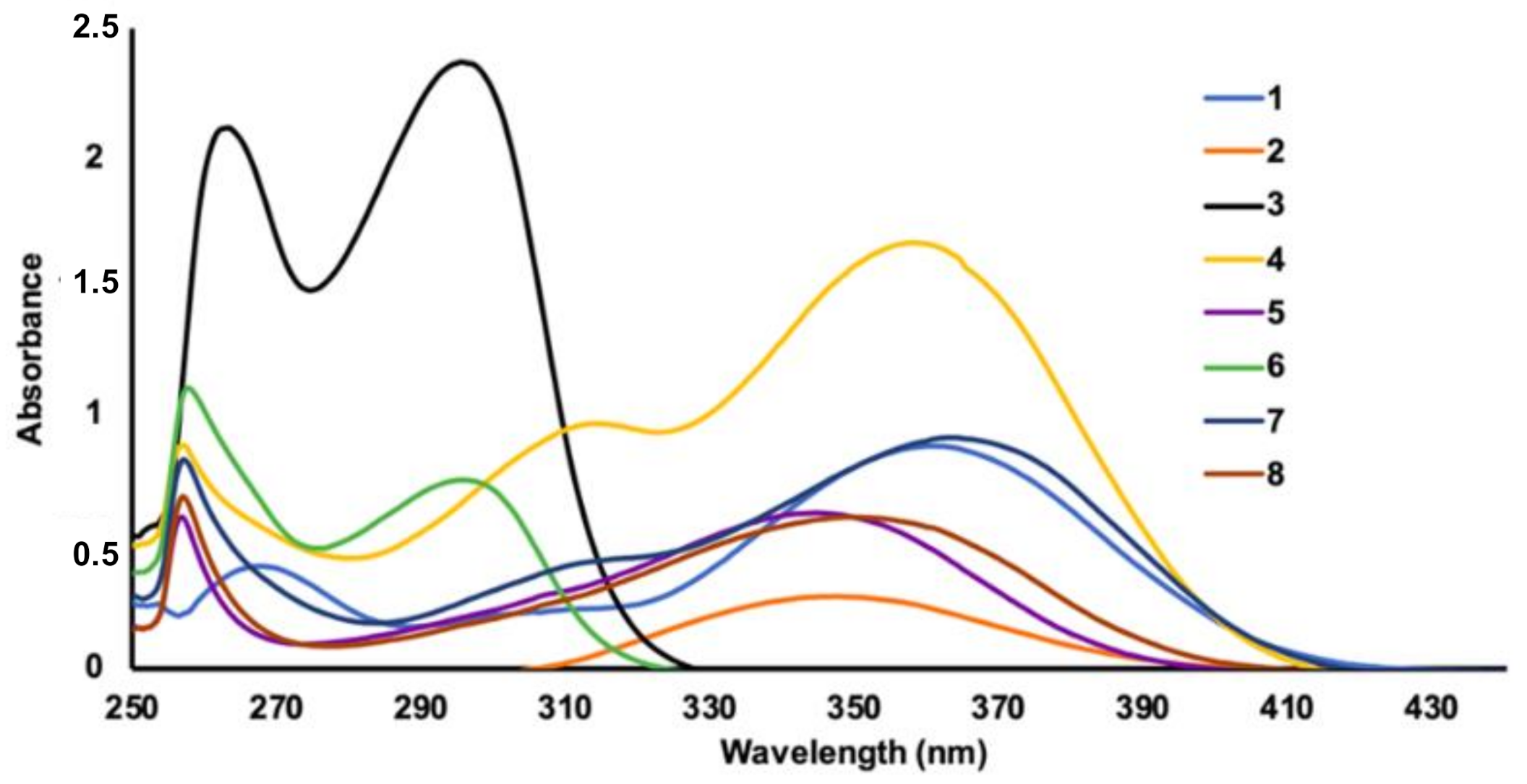
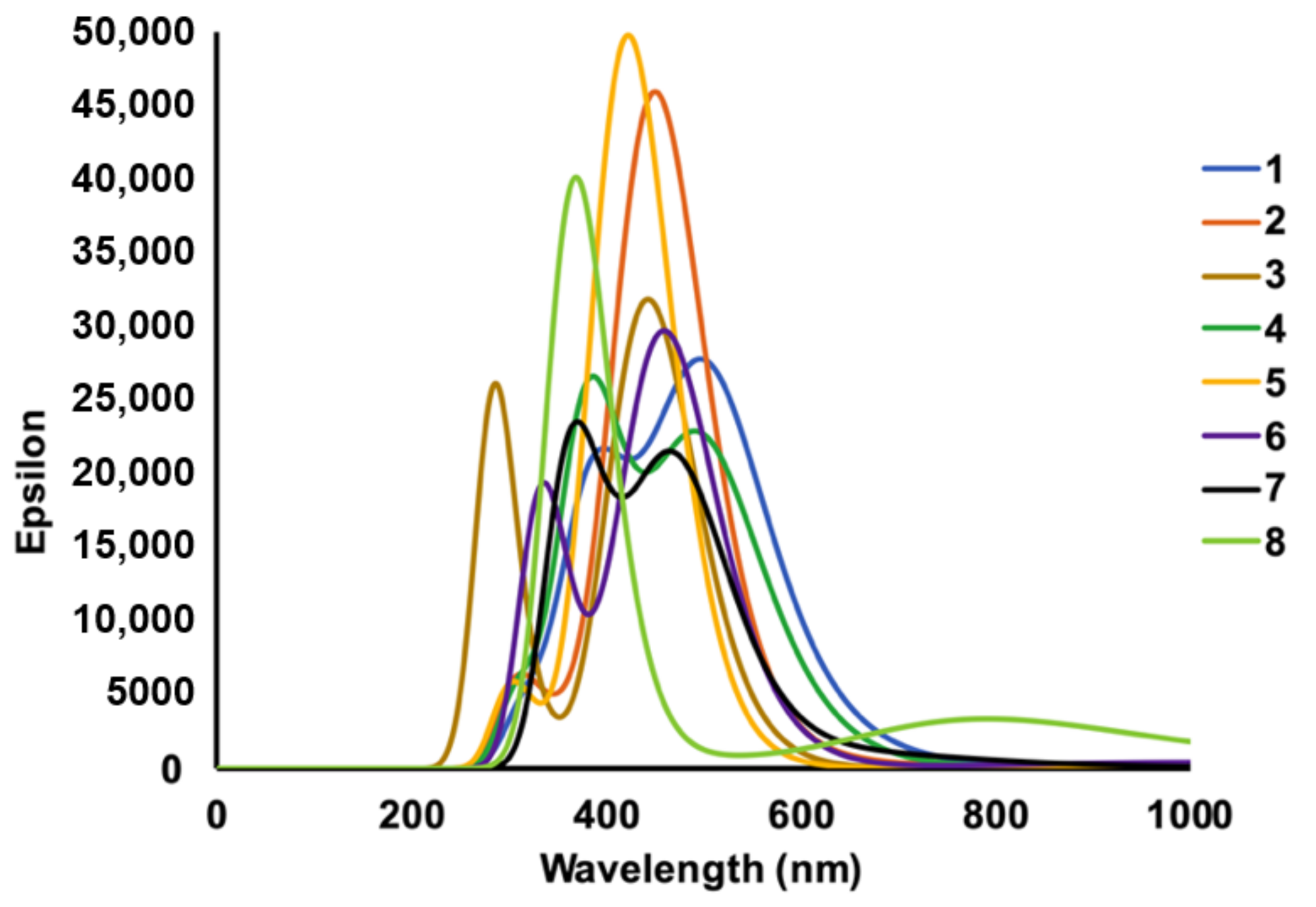
| Chalcone | Experimental | Theoretical | ||||
|---|---|---|---|---|---|---|
| λmax (nm) | ε (L mol−1 cm−1) | λcut off (nm) | λmax (nm) | ε (L mol−1 cm−1) | λcut off (nm) | |
| 1 | 269 | 34,720 | - | 405 | 27,627 | 277 |
| 363 | 426 | 506 | 866 | |||
| 2 | - | 56,400 | 306 | 221 | 45,473 | 192 |
| 352 | 406 | 276 | 397 | |||
| 3 | 265 | 47,500 | - | 209 | 31,282 | 160 |
| 299 | 326 | 323 | 473 | |||
| 4 | 258 | 33,840 | - | 235 | 25,753 | 211 |
| 363 | 415 | 253 | 302 | |||
| 5 | 258 | 46,400 | - | 166 | 49,405 | 152 |
| 349 | 398 | 190 | 241 | |||
| 6 | 259 | 21,400 | - | 171 | 29,139 | 156 |
| 299 | 324 | 196 | 262 | |||
| 7 | 258 | 21,600 | - | 199 | 23,529 | 185 |
| 367 | 418 | 213 | 286 | |||
| 8 | 257 | 32,080 | - | 183 | 38,716 | 153 |
| 355 | 405 | 319 | 497 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wijayanti, L.W.; Swasono, R.T.; Lee, W.; Jumina, J. Synthesis and Evaluation of Chalcone Derivatives as Novel Sunscreen Agent. Molecules 2021, 26, 2698. https://doi.org/10.3390/molecules26092698
Wijayanti LW, Swasono RT, Lee W, Jumina J. Synthesis and Evaluation of Chalcone Derivatives as Novel Sunscreen Agent. Molecules. 2021; 26(9):2698. https://doi.org/10.3390/molecules26092698
Chicago/Turabian StyleWijayanti, Lucia Wiwid, Respati Tri Swasono, Wonkoo Lee, and Jumina Jumina. 2021. "Synthesis and Evaluation of Chalcone Derivatives as Novel Sunscreen Agent" Molecules 26, no. 9: 2698. https://doi.org/10.3390/molecules26092698
APA StyleWijayanti, L. W., Swasono, R. T., Lee, W., & Jumina, J. (2021). Synthesis and Evaluation of Chalcone Derivatives as Novel Sunscreen Agent. Molecules, 26(9), 2698. https://doi.org/10.3390/molecules26092698





