Survey of Phenolic Acids, Flavonoids and In Vitro Antioxidant Potency Between Fig Peels and Pulps: Chemical and Chemometric Approach
Abstract
1. Introduction
2. Results and Discussion
2.1. Peels and Pulps Color
2.2. Spectrophotometric Assays
2.3. In Vitro Antioxidant Activity
2.4. The Half Maximal Inhibitory Concentration (I50)
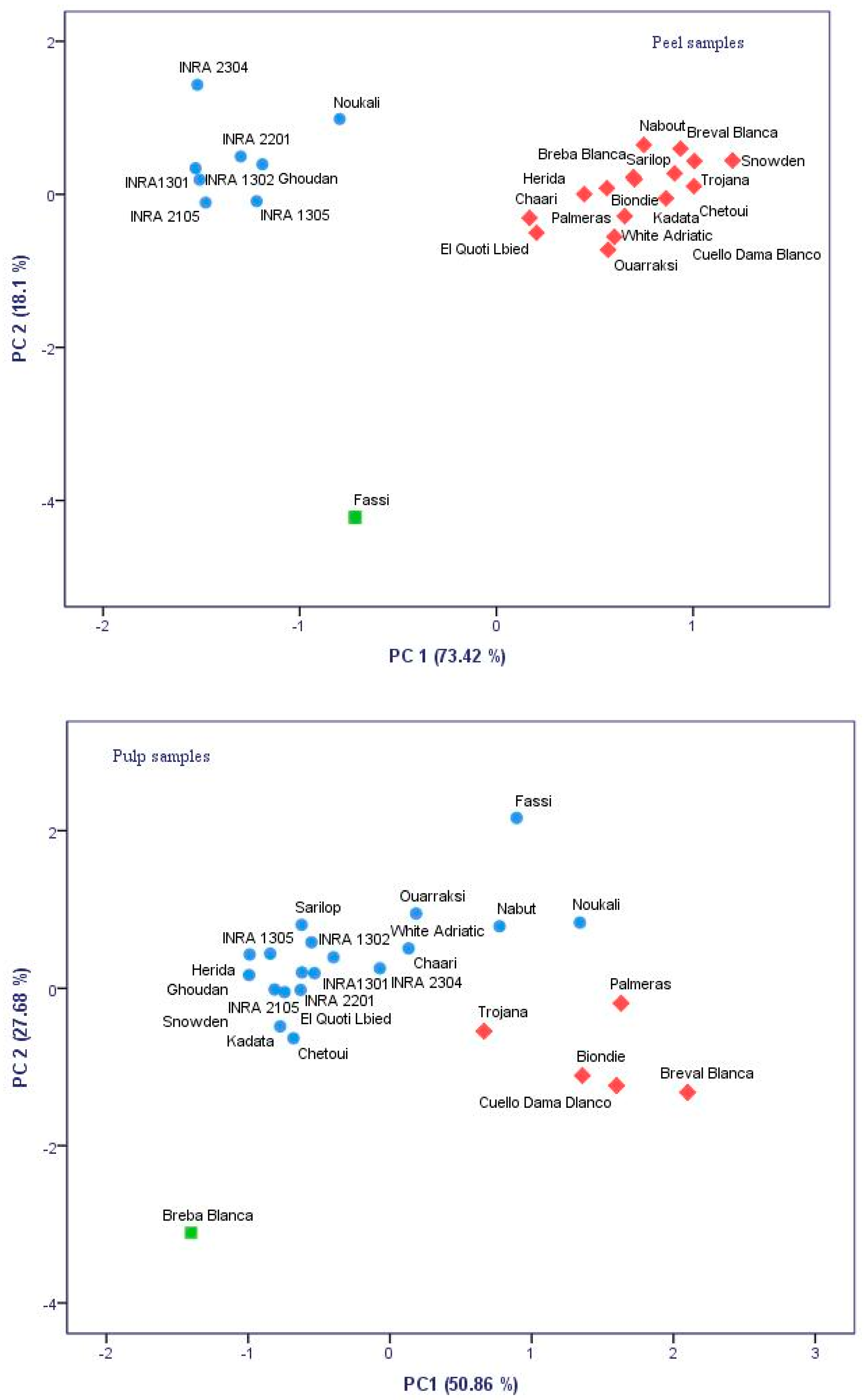
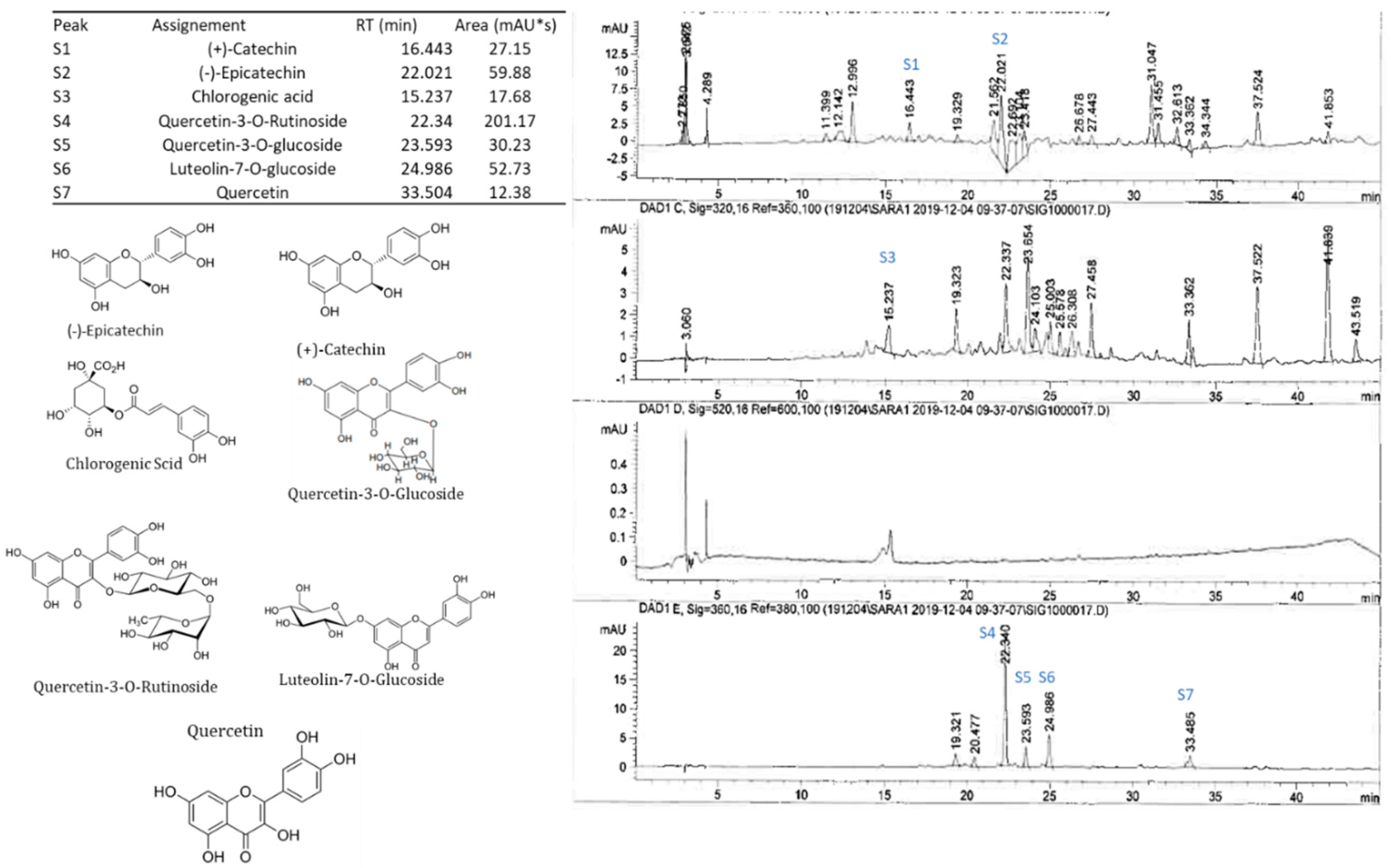
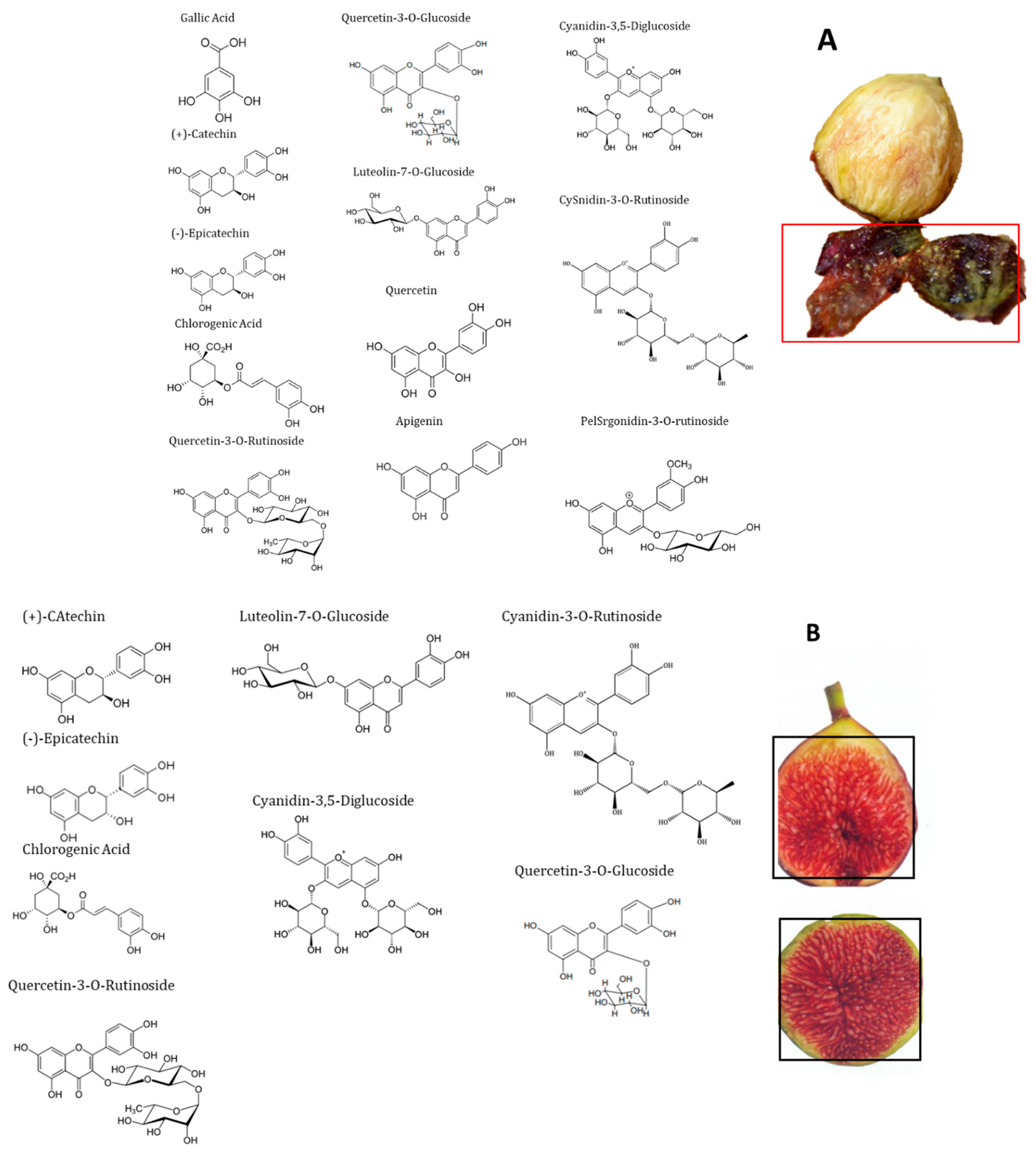
2.5. Polyphenolic Profile
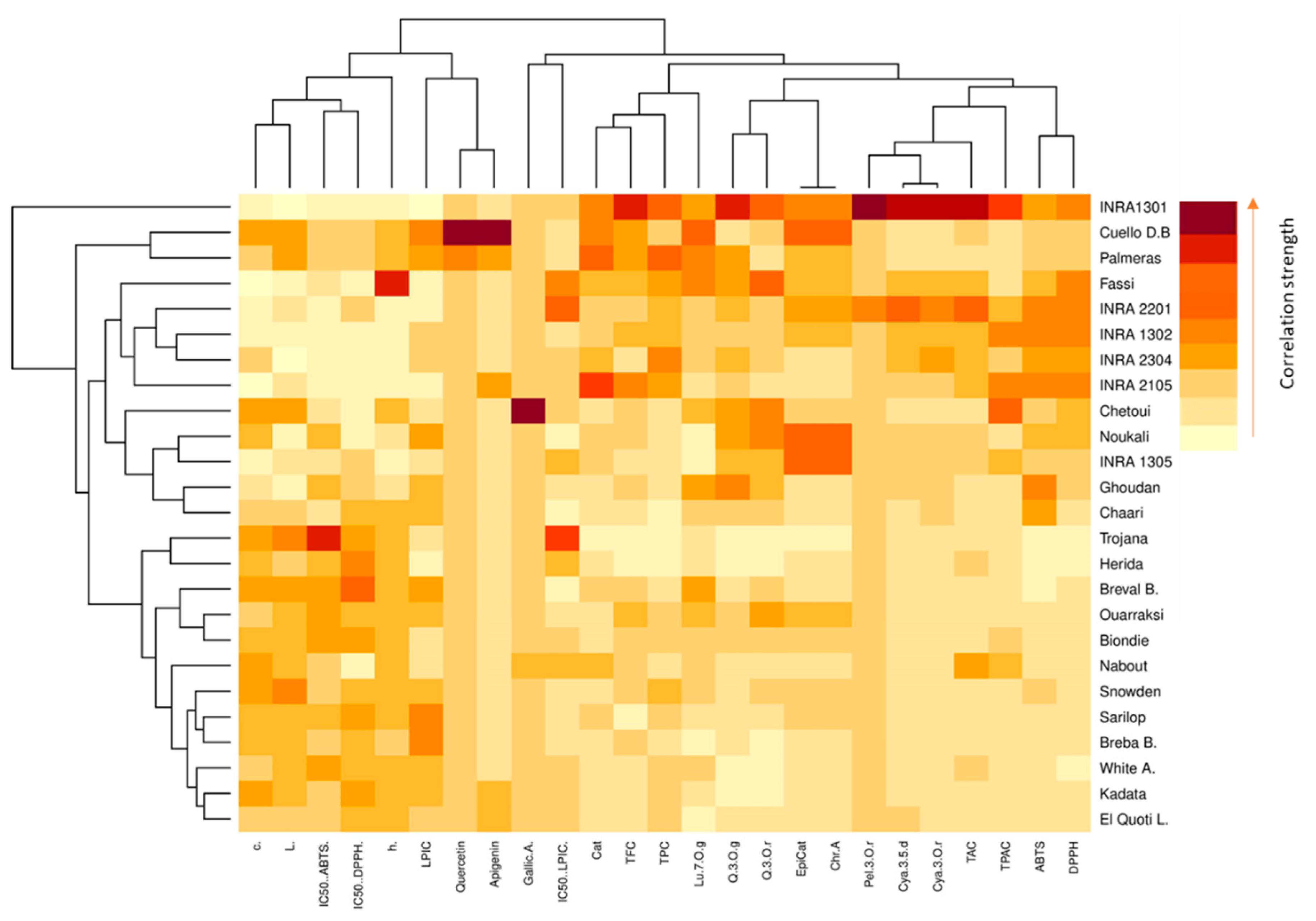
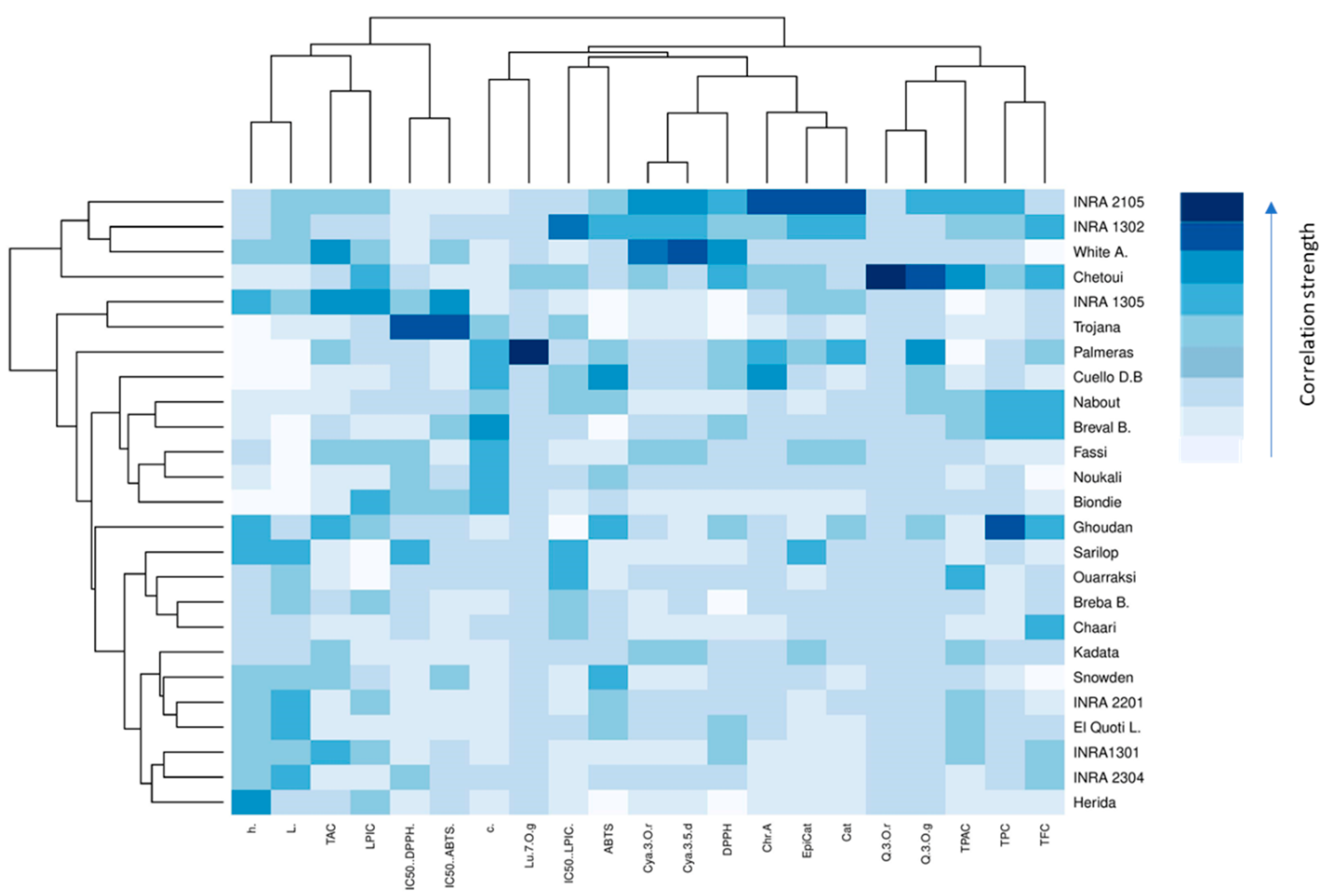
2.6. Heat Map Analysis
3. Materials and Methods
3.1. Plant Material
3.2. Growing Conditions
3.3. Fruit Peel and Pulp Color
3.4. Spectrophotometric Analysis
3.4.1. Total Phenolics Content (TPC)
3.4.2. Total Flavonoids Content (TFC)
3.4.3. Total Anthocyanins Content (TAC)
3.4.4. Total Proanthocyanidins Content (TPAC)
3.4.5. Antioxidant Activity (AA)
3.5. Polyphenolic Compounds Analysis (PCs)
3.5.1. Extraction Method
3.5.2. Determination of PCs
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Česonienė, L.; Jasutienė, I.; Šarkinas, A. Phenolics and anthocyanins in berries of European cranberry and their antimicrobial activity. Medicina 2009, 45, 992. [Google Scholar] [CrossRef]
- Tsimogiannis, D.; Oreopoulou, V. Classification of Phenolic Compounds in Plants. In Polyphenols in Plants; Academic Press: Cambridge, MA, USA, 2019; pp. 263–284. [Google Scholar]
- Rispail, N.; Morris, P.; Webb, K.J. Phenolic compounds: Extraction and analysis. In Lotus Japonicus Handbook; Springer: Dordrecht, The Netherlands, 2005; pp. 349–354. [Google Scholar]
- Laura, A.; Moreno-Escamilla, J.O.; Rodrigo-García, J.; Alvarez-Parrilla, E. Phenolic compounds. In Postharvest Physiology and Biochemistry of Fruits and Vegetables; Woodhead Publishing: Cambridge, UK, 2019; pp. 253–271. [Google Scholar]
- Winkel-Shirley, B. Biosynthesis of flavonoids and effects of stress. Curr. Plant Biol. 2002, 5, 218–223. [Google Scholar] [CrossRef]
- Gould, K.S. Muriel wheldale onslow and the rediscovery of anthocyanin function in plants. Recent Adv. Polyphen. Res. 2010, 2, 206–225. [Google Scholar]
- Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for analysis of plant phenolic compounds. Molecules 2013, 18, 2328–2375. [Google Scholar] [CrossRef]
- Shimazu, T.; Inoue, M.; Sasazuki, S.; Iwasaki, M.; Sawada, N.; Yamaji, T. Isoflavone intake and risk of lung cancer: A prospective cohort study in Japan. Japan Public Health Center–based Prospective Study Group. Am. J. Clin. Nutr. 2010, 91, 722–728. [Google Scholar] [CrossRef] [PubMed]
- Redman, L.M.; Smith, S.R.; Burton, J.H.; Martin, C.K.; Il’yasova, D.; Ravussin, E. Metabolic slowing and reduced oxidative damage with sustained caloric restriction support the rate of living and oxidative damage theories of aging. Cell Metab. 2018, 27, 805–815. [Google Scholar] [CrossRef]
- Lima, C.F.; Fernandes-Ferreira, M.; Pereira-Wilson, C. Phenolic compounds protect HepG2 cells from oxidative damage: Relevance of glutathione levels. Life Sci. 2006, 79, 2056–2068. [Google Scholar] [CrossRef]
- Afonso, M.S.; De O Silva, A.M.; Carvalho, E.B.; Rivelli, D.P.; Barros, S.B.; Rogero, M.M.; Mancini-Filho, J. Phenolic compounds from Rosemary (Rosmarinus officinalis L.) attenuate oxidative stress and reduce blood cholesterol concentrations in diet-induced hypercholesterolemic rats. Nutr. Metab. 2013, 10, 19. [Google Scholar] [CrossRef]
- Le Lay, S.; Simard, G.; Martinez, M.C.; Andriantsitohaina, R. Oxidative stress and metabolic pathologies: From an adipocentric point of view. Oxidative Med. Cell Longev. 2014, 908539. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Tawfeek, N.; Sobeh, M.; Hamdan, D.I.; Farrag, N.; Roxo, M.; El-Shazly, A.M.; Wink, M. Phenolic compounds from Populus alba L. and Salix subserrata Willd. (Salicaceae) counteract oxidative stress in Caenorhabditis elegans. Molecules 2019, 24, 1999. [Google Scholar] [CrossRef] [PubMed]
- Bautista, I.; Boscaiu, M.; Lidón, A.; Llinares, J.V.; Lull, C.; Donat, M.P.; Vicente, O. Environmentally induced changes in antioxidant phenolic compounds levels in wild plants. Acta Physiol. Plant 2016, 38, 9. [Google Scholar] [CrossRef]
- Hssaini, L.; Charafi, J.; Hanine, H.; Ennahli, S.; Mekaoui, A.; Mamouni, A.; Razouk, R. Comparative analysis and physio-biochemical screening of an ex-situ fig (Ficus carica L.) collection. Hortic. Environ. Biotechnol. 2019, 60, 671–683. [Google Scholar] [CrossRef]
- USDA. U.S. Department of Agriculture, Agricultural Research Service. Database for the Flavonoid Content of Selected Foods, Release 3.0; Nutrient Data Laboratory: Beltsville, MD, USA, 2011.
- Hssaini, L.; Hanine, H.; Razouk, R.; Ennahli, S.; Mekaoui, A.; Guirrou, I.; Charafi, J. Diversity Screening of Fig (Ficus Carica, L.) Germplasm through Integration of Morpho-agronomic and Biochemical Traits. Int. J. Fruit Sci. 2019, 20, 1–20. [Google Scholar] [CrossRef]
- Vinson, J.A.; Hao, Y.; Su, X.; Zubik, L. Phenol antioxidant quantity and quality in foods: Vegetables. J. Agric. Food Chem. 1998, 46, 3630–3634. [Google Scholar] [CrossRef]
- Vallejo, F.; Marín, J.G.; Tomás-Barberán, F.A. Phenolic compound content of fresh and dried figs (Ficus carica L.). Food Chem. 2012, 130, 485–492. [Google Scholar] [CrossRef]
- Hssaini, L.; Charafi, J.; Razouk, R.; Hernández, F.; Fauconnier, M.L.; Ennahli, S.; Hanine, H. Assessment of Morphological Traits and Fruit Metabolites in Eleven Fig Varieties (Ficus Carica L.). Int. J. Fruit Sci. 2020, 1–21, 8–28. [Google Scholar] [CrossRef]
- Stinco, C.M.; Rodríguez-Pulido, F.J.; Escudero-Gilete, M.L.; Gordillo, B.; Vicario, I.M.; Meléndez-Martínez, A.J. Lycopene isomers in fresh and processed tomato products: Correlations with instrumental color measurements by digital image analysis and spectroradiometry. Food Res. Int. 2013, 50, 111–120. [Google Scholar] [CrossRef]
- Kuś, P.M.; Congiu, F.; Teper, D.; Sroka, Z.; Jerković, I.; Tuberoso, C.I.G. Antioxidant activity, color characteristics, total phenol content and general HPLC fingerprints of six Polish unifloral honey types. LWT-Food Sci. Technol. 2014, 55, 124–130. [Google Scholar]
- Silva, B.M.; Andrade, P.B.; Ferreres, F.; Domingues, A.L.; Seabra, R.M.; Ferreira, M.A. Phenolic profile of quince fruit (Cydonia oblonga Miller) (pulp and peel). J. Agric. Food Chem. 2002, 50, 4615–4618. [Google Scholar] [CrossRef]
- Fan, X.; Jiao, W.; Wang, X.; Cao, J.; Jiang, W. Polyphenol composition and antioxidant capacity in pulp and peel of apricot fruits of various varieties and maturity stages at harvest. Int. J. Food Sci. Technol. 2018, 53, 327–336. [Google Scholar] [CrossRef]
- Yang, Y.; Yao, G.; Yue, W.; Zhang, S.; Wu, J. Transcriptome profiling reveals differential gene expression in proanthocyanidin biosynthesis associated with red/green skin color mutant of pear (Pyrus communis L.). Front. Plant Sci. 2015, 6, 795. [Google Scholar] [CrossRef] [PubMed]
- Çalişkan, O.; Polat, A.A. Phytochemical and antioxidant properties of selected fig (Ficus carica L.) accessions from the eastern Mediterranean region of Turkey. Sci. Hortic. 2011, 128, 473–478. [Google Scholar]
- Del Caro, A.; Piga, A. Polyphenol composition of peel and pulp of two Italian fresh fig fruits cultivars (Ficus carica L.). Eur. Food Res. Technol. 2008, 226, 715–719. [Google Scholar] [CrossRef]
- Solomon, A.; Golubowicz, S.; Yablowicz, Z.; Grossman, S.; Bergman, M.; Gottlieb, H.E.; Flaishman, M.A. Antioxidant activities and anthocyanin content of fresh fruits of common fig (Ficus carica L.). J. Agric. Food Chem. 2006, 54, 7717–7723. [Google Scholar] [CrossRef]
- Harzallah, A.; Bhouri, A.M.; Amri, Z.; Soltana, H.; Hammami, M. Phytochemical content and antioxidant activity of different fruit parts juices of three figs (Ficus carica L.) varieties grown in Tunisia. Ind. Crop. Prod. 2016, 83, 255–267. [Google Scholar] [CrossRef]
- Palmeira, L.; Pereira, C.; Dias, M.I.; Abreu, R.M.; Corrêa, R.C.; Pires, T.C.; Ferreira, I.C. Nutritional, chemical and bioactive profiles of different parts of a Portuguese common fig (Ficus carica L.) variety. Food Res. Int. 2019, 126, 108572. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Barberán, F.A.; Gil, M.I.; Cremin, P.; Waterhouse, A.L.; Hess-Pierce, B.; Kader, A.A. HPLC−DAD−ESIMS analysis of phenolic compounds in nectarines, peaches, and plums. J. Agric. Food Chem. 2001, 49, 4748–4760. [Google Scholar] [CrossRef]
- Kamiloglu, S.; Capanoglu, E. Polyphenol content in figs (Ficus carica L.): Effect of sun-drying. Int. J. Food Prop. 2015, 18, 521–535. [Google Scholar] [CrossRef]
- Viuda-Martos, M.; Barber, X.; Pérez-Álvarez, J.A.; Fernández-López, J. Assessment of chemical, physico-chemical, techno-functional and antioxidant properties of fig (Ficus carica L.) powder co-products. Ind. Crop. Prod. 2015, 69, 472–479. [Google Scholar] [CrossRef]
- Pande, G.; Akoh, C.C. Organic acids, antioxidant capacity, phenolic content and lipid characterisation of Georgia-grown underutilized fruit crops. Food Chem. 2010, 120, 1067–1075. [Google Scholar] [CrossRef]
- Ammar, S.; Del Mar Contreras, M.; Belguith-Hadrich, O.; Segura-Carretero, A.; Bouaziz, M. Assessment of the distribution of phenolic compounds and contribution to the antioxidant activity in Tunisian fig leaves, fruits, skins and pulps using mass spectrometry-based analysis. Food Funct. 2015, 6, 3663–3677. [Google Scholar] [CrossRef]
- Konak, R.; Kösoğlu, İ.; Yemenıcıoğlu, A. Effects of different drying methods on phenolic content, antioxidant capacity and general characteristics of selected dark colored Turkish fig cultivars. V Int. Symp. Fig. 2015, 1173, 335–340. [Google Scholar] [CrossRef]
- Chiva-Blanch, G.; Visioli, F. Polyphenols and health: Moving beyond antioxidants. J. Berry Res. 2012, 2, 63–71. [Google Scholar] [CrossRef]
- Martínez-Valverde, I.; Periago, M.J.; Provan, G.; Chesson, A. Phenolic compounds, lycopene and antioxidant activity in commercial varieties of tomato (Lycopersicum esculentum). J. Sci. Food Agric. 2002, 82, 323–330. [Google Scholar] [CrossRef]
- Rothwell, J.A.; Perez-Jimenez, J.; Neveu, V.; Medina-Remon, A.; M’Hiri, N.; García-Lobato, P.; Scalbert, A. Phenol-Explorer 3.0: A major update of the Phenol-Explorer database to incorporate data on the effects of food processing on polyphenol content. Database 2013, 2013, bat070. [Google Scholar] [CrossRef] [PubMed]
- Clark, N.R.; Ma’ayan, A. Introduction to statistical methods to analyze large data sets: Principal components analysis. Sci. Signal 2011, 4, tr3. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Bolling, B.W. Characterisation of stilbenes in California almonds (Prunus dulcis) by UHPLC–MS. Food Chem. 2014, 148, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. In Methods in Enzymology; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 152–178. [Google Scholar]
- Barreira, J.C.; Ferreira, I.C.; Oliveira, M.B.P.; Pereira, J.A. Antioxidant activities of the extracts from chestnut flower, leaf, skins and fruit. Food Chem. 2008, 107, 1106–1113. [Google Scholar] [CrossRef]
- Cheng, G.W.; Breen, P.J. Activity of phenylalanine ammonia-lyase (PAL) and concentrations of anthocyanins and phenolics in developing strawberry fruit. J. Am. Soc. Hortic. Sci. 1991, 116, 865–869. [Google Scholar] [CrossRef]
- Porter, L.J.; Hrstich, L.N.; Chan, B.G. The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochemistry 1985, 25, 223–230. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C.L.W.T. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Freire, A.L.; Ramos, C.L.; Da Costa Souza, P.N.; Cardoso, M.G.B.; Schwan, R.F. Nondairy beverage produced by controlled fermentation with potential probiotic starter cultures of lactic acid bacteria and yeast. Int. J. Food Microbiol. 2017, 248, 39–46. [Google Scholar] [CrossRef]
- Genskowsky, E.; Puente, L.A.; Pérez-Álvarez, J.A.; Fernández-López, J.; Muñoz, L.A.; Viuda-Martos, M. Determination of polyphenolic profile, antioxidant activity and antibacterial properties of maqui [Aristotelia chilensi s (Molina) Stuntz] a Chilean blackberry. J. Sci. Food Agric. 2016, 96, 4235–4242. [Google Scholar] [CrossRef] [PubMed]
- Haarman, B.C.B.; Riemersma-Van der Lek, R.F.; Nolen, W.A.; Mendes, R.; Drexhage, H.A.; Burger, H. Feature-expression heat maps–A new visual method to explore complex associations between two variable sets. J. Biomed. Inform. 2015, 53, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Walkley, A.; Black, I.A. An examination of Degtjareff method for determining soil organic matter and a proposed modification of the chromic acid titration method. Soil Sci. 1934, 37, 29–37. [Google Scholar] [CrossRef]
| Variables | Fruit Part | Mini | Max | Mean | Std. Deviation | ANOVA p-Value |
|---|---|---|---|---|---|---|
| Gallic acid * | Peel | 0 | 11.29 | 0.54 | 2.24 | <0.001 |
| (+)-Catechin * | 0 | 24.06 | 5.89 | 5.95 | <0.001 | |
| (−)-Epicatechin * | 2.61 | 55.44 | 17.31 | 12.89 | <0.001 | |
| Chlorogenic acid * | 0 | 10.67 | 3.03 | 2.94 | <0.001 | |
| Quercetin-3-O-rutinoside * | 5.3 | 147.42 | 58.46 | 38.66 | <0.001 | |
| Quercetin-3-O-glucoside * | 2.52 | 35.58 | 11.48 | 7.76 | <0.001 | |
| Luteolin-7-O-glucoside * | 0 | 18.24 | 6.75 | 4.87 | <0.001 | |
| Quercetin * | 0 | 59.61 | 4.49 | 12.48 | <0.001 | |
| Apigenin * | 0 | 4.91 | 0.41 | 1.04 | <0.001 | |
| Cyanidin-3,5-diglucoside * | 0 | 495.76 | 48.58 | 109.91 | <0.001 | |
| Cyanidin-3-O-rutinoside * | 0 | 478.9 | 46.78 | 105.29 | <0.001 | |
| Pelargonidin-3-O-rutinoside * | 0 | 12.67 | 0.67 | 2.58 | <0.001 | |
| TPC (mg GAE/100 g dw) | 370 | 3162.86 | 1368.67 | 671.01 | <0.001 | |
| TFC (mg CE/100 g dw) | 188.57 | 2013.57 | 690.19 | 371.47 | <0.001 | |
| TPAC (mg Cyan /100 g dw) | 0.2 | 3.09 | 0.83 | 0.83 | <0.001 | |
| TAC (mg cy-3-r /100 g dw) | 4.14 | 192.5 | 37.17 | 41.9 | <0.001 | |
| DPPH (mMol TE/g dw) | 21.23 | 367.26 | 156.76 | 21.53 | <0.001 | |
| ABTS (mMol TE/g dw) | 7.57 | 563.53 | 231.52 | 19.59 | <0.001 | |
| LPIC (mMol TE/g dw) | 139.17 | 353.11 | 226.26 | 10.44 | <0.001 | |
| L* | 19.81 | 73.51 | 49.51 | 15.54 | <0.001 | |
| c* | 0.89 | 62.76 | 37.42 | 16.36 | <0.001 | |
| h* | -3.41 | 360.95 | 78.58 | 57.02 | <0.001 | |
| Gallic acid | Pulp | nd | nd | nd | nd | <0.001 |
| (+)-Catechin | 0 | 6.65 | 1.47 | 1.4 | <0.001 | |
| (−)-Epicatechin | 1.25 | 19.06 | 5.23 | 4.03 | <0.001 | |
| Chlorogenic acid | 0 | 4.84 | 0.77 | 1.09 | <0.001 | |
| Quercetin-3-O-rutinoside | 0 | 26.85 | 1.89 | 5.16 | <0.001 | |
| Quercetin-3-O-glucoside | 0 | 4.05 | 0.44 | 0.95 | <0.001 | |
| Luteolin-7-O-glucoside | 0 | 4.5 | 0.21 | 0.89 | <0.001 | |
| Quercetin | nd | nd | nd | nd | <0.001 | |
| Apigenin | nd | nd | nd | nd | <0.001 | |
| Cyanidin-3,5-diglucoside | 0 | 28.45 | 5.82 | 6.68 | <0.001 | |
| Cyanidin-3-O-rutinoside | 0.94 | 34.43 | 9.01 | 8.67 | <0.001 | |
| Pelargonidin-3-O-rutinoside | nd | nd | nd | nd | <0.001 | |
| TPC | 105.71 | 1255.71 | 426.38 | 234.32 | <0.001 | |
| TFC | 13.57 | 331.43 | 157.57 | 79.96 | <0.001 | |
| TPAC | 0.2 | 1.06 | 0.37 | 0.13 | <0.001 | |
| TAC | 2.27 | 19.44 | 7.71 | 4.49 | <0.001 | |
| DPPH | 13.92 | 151.24 | 73.99 | 7.05 | <0.001 | |
| ABTS | 6.59 | 207.49 | 76.19 | 7.35 | <0.001 | |
| LPIC | 42.89 | 226.88 | 121.25 | 7.7 | <0.001 | |
| L* | 12.17 | 34645 | 493.3 | 6.81 | 0.622 | |
| c* | 12.35 | 59.85 | 28.02 | 13.64 | <0.001 | |
| h* | 4.87 | 74.8 | 32.33 | 16.59 | <0.001 | |
| Effect | Wilks Lambda’s value | F | Hypothesis df | Error df | Sig. | |
| Variety | 0 | 477.23 | 560 | 1376.367 | 0 | |
| Fruit part | 0 | 496,075.72 | 20 | 79 | 0 | |
| Variety * Fruit part | 0 | 464.37 | 440 | 1242.807 | 0 |
| Cultivars | TPC | TFC | TPAC | TAC | DPPH | ABTS | LPIC | IC50 (DPPH) | IC50 (ABTS) | IC50 (LPIC) | L* | c* | h° |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioudie | 1346.19 | 602.86 | 0.90 | 10.82 | 332.96 | 452.52 | 154.84 | 172.38 | 292.70 | 114.83 | 55.46 | 45.97 | 91.43 |
| Breba Blanca | 1093.81 | 590.95 | 0.43 | 14.27 | 100.19 | 192.06 | 278.94 | 125.46 | 185.47 | 83.56 | 59.81 | 47.67 | 85.01 |
| Breval Blanca | 796.19 | 669.52 | 0.51 | 15.78 | 254.75 | 364.80 | 231.87 | 306.04 | 285.62 | 24.45 | 62.62 | 54.83 | 90.86 |
| Chaari | 696.19 | 388.57 | 0.39 | 9.93 | 299.74 | 527.25 | 169.11 | 129.47 | 112.22 | 51.81 | 44.85 | 39.14 | 90.87 |
| Chetoui | 1100.95 | 708.81 | 2.20 | 18.13 | 329.04 | 336.61 | 160.55 | 27.42 | 119.33 | 124.80 | 65.56 | 50.4 | 96.1 |
| Cuello Dama Blanco | 1391.43 | 1177.86 | 0.33 | 34.88 | 333.99 | 491.90 | 155.56 | 80.47 | 163.46 | 86.35 | 63.32 | 51.12 | 92.73 |
| El Quoti Lbied | 1241.43 | 493.33 | 0.50 | 11.65 | 68.83 | 175.95 | 255.40 | 156.99 | 154.30 | 99.83 | 48.8 | 37.29 | 95.72 |
| Fassi | 2020.00 | 935.00 | 0.58 | 47.90 | 332.13 | 493.69 | 232.58 | 0.28 | 97.35 | 216.92 | 31.78 | 10.28 | 215.93 |
| Ghoudan | 927.14 | 602.86 | 0.46 | 19.16 | 133.21 | 210.85 | 289.63 | 76.47 | 225.29 | 81.13 | 28.13 | 28.96 | 42.36 |
| Herida | 389.05 | 214.76 | 0.33 | 21.50 | 56.86 | 77.49 | 240.43 | 234.03 | 230.18 | 125.68 | 51.25 | 44.53 | 90.85 |
| INRA 1302 | 1627.14 | 807.62 | 1.97 | 53.00 | 37.87 | 77.49 | 191.22 | 3.97 | 56.49 | 76.19 | 31.36 | 18.39 | 18.33 |
| INRA 1305 | 912.86 | 417.14 | 1.04 | 22.33 | 40.76 | 157.60 | 216.89 | 92.67 | 114.38 | 136.79 | 36.42 | 19.02 | 36.25 |
| INRA 2105 | 2070.00 | 1282.62 | 1.67 | 51.97 | 79.97 | 137.01 | 251.84 | 3.88 | 73.70 | 101.26 | 34.61 | 13.85 | 19.96 |
| INRA 2201 | 1865.24 | 689.76 | 1.13 | 126.41 | 54.38 | 147.31 | 295.34 | 109.17 | 68.01 | 263.49 | 36.79 | 24.53 | 25.41 |
| INRA 2304 | 2396.19 | 385.00 | 0.76 | 63.75 | 16.62 | 91.81 | 238.29 | 2.12 | 21.84 | 124.04 | 24.52 | 38.88 | 23.35 |
| INRA1301 | 2860.48 | 1944.52 | 2.59 | 192.23 | 152.40 | 190.72 | 198.35 | 19.85 | 40.58 | 124.78 | 25.72 | 22.09 | 20.99 |
| Kadata | 1208.10 | 492.14 | 0.54 | 17.16 | 29.41 | 126.27 | 301.76 | 201.52 | 174.22 | 98.09 | 59.62 | 48.67 | 98.26 |
| Nabout | 810.48 | 699.29 | 1.21 | 82.29 | 121.45 | 459.23 | 239.00 | 16.98 | 177.38 | 132.85 | 52.86 | 55.04 | 89.07 |
| Noukali | 822.38 | 677.86 | 0.50 | 21.23 | 128.26 | 212.64 | 203.34 | 25.04 | 202.71 | 55.03 | 28.7 | 41.67 | 53.68 |
| Ouarraksi | 1384.29 | 763.57 | 0.48 | 4.82 | 218.02 | 293.65 | 278.94 | 161.00 | 297.22 | 88.32 | 59.26 | 38.39 | 103.53 |
| Palmeras | 2855.71 | 1070.71 | 0.33 | 18.06 | 29.21 | 157.15 | 199.06 | 106.34 | 179.49 | 87.90 | 65.15 | 36.95 | 93.99 |
| Sarilop | 1155.71 | 263.57 | 0.38 | 17.16 | 21.78 | 12.82 | 278.22 | 175.77 | 200.03 | 78.28 | 59.21 | 48.26 | 90.94 |
| Snowden | 1700.95 | 598.10 | 0.42 | 15.58 | 69.45 | 440.43 | 248.98 | 123.15 | 190.12 | 80.01 | 72.62 | 54.57 | 94.84 |
| Trojana | 415.24 | 317.14 | 0.56 | 11.10 | 5.27 | 12.01 | 189.79 | 170.40 | 495.99 | 295.07 | 73.16 | 50.95 | 90.38 |
| White Adriatic | 1129.52 | 461.19 | 0.44 | 28.05 | 22.66 | 0.07 | 156.27 | 162.94 | 299.49 | 103.07 | 59.7 | 37.58 | 99.14 |
| Cultivar | TPC | TFC | TPAC | TAC | DPPH | ABTS | LPIC | IC50 (DPPH) | IC50 (ABTS) | IC50 (LPIC) | L* | c* | h° |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bioudie | 339.05 | 95.71 | 0.40 | 3.58 | 22.21 | 24.37 | 40.48 | 275.08 | 418.35 | 152.16 | 20.67 | 44.04 | 7.83 |
| Breba Blanca | 255.71 | 170.71 | 0.36 | 5.86 | 19.43 | 24.32 | 35.86 | 206.83 | 226.64 | 231.46 | 34.91 | 17.05 | 36.51 |
| Breval Blanca | 731.90 | 257.62 | 0.45 | 6.75 | 35.01 | 16.42 | 23.51 | 163.29 | 455.25 | 169.11 | 19.05 | 55.05 | 16.46 |
| Chaari | 284.29 | 250.48 | 0.26 | 3.03 | 25.41 | 23.44 | 24.85 | 202.38 | 220.80 | 241.85 | 34.10 | 29.50 | 32.35 |
| Chetoui | 491.43 | 257.62 | 0.64 | 8.27 | 39.50 | 25.03 | 38.99 | 168.57 | 204.48 | 231.55 | 25.18 | 15.33 | 27.07 |
| Cuello Dama Dlanco | 439.05 | 93.33 | 0.25 | 3.45 | 33.75 | 40.83 | 21.13 | 199.52 | 221.21 | 238.04 | 19.95 | 46.79 | 6.52 |
| El Quoti Lbied | 422.38 | 162.38 | 0.41 | 3.24 | 32.69 | 33.51 | 21.43 | 139.85 | 198.06 | 201.63 | 44.51 | 16.53 | 42.33 |
| Fassi | 234.29 | 93.33 | 0.38 | 11.37 | 27.66 | 22.73 | 31.40 | 245.24 | 201.32 | 130.18 | 18.62 | 44.07 | 36.60 |
| Ghoudan | 1186.67 | 271.90 | 0.29 | 12.48 | 33.60 | 33.95 | 32.59 | 201.06 | 241.50 | 85.47 | 33.24 | 14.72 | 54.88 |
| Herida | 174.76 | 107.62 | 0.29 | 7.72 | 18.70 | 17.67 | 35.86 | 151.47 | 233.68 | 148.96 | 34.18 | 15.14 | 62.97 |
| INRA 1302 | 520.00 | 250.48 | 0.47 | 7.31 | 34.06 | 35.82 | 26.79 | 167.29 | 316.70 | 392.39 | 35.10 | 22.29 | 36.35 |
| INRA 1305 | 262.86 | 131.43 | 0.22 | 16.89 | 15.35 | 14.43 | 43.45 | 269.32 | 665.58 | 138.37 | 40.04 | 17.22 | 55.95 |
| INRA 2105 | 753.33 | 160.00 | 0.56 | 9.58 | 38.78 | 32.85 | 33.33 | 117.46 | 138.88 | 164.76 | 34.31 | 17.98 | 38.18 |
| INRA 2201 | 374.76 | 106.43 | 0.43 | 3.24 | 25.75 | 33.20 | 35.12 | 160.10 | 186.17 | 145.74 | 45.91 | 18.83 | 42.91 |
| INRA 2304 | 465.24 | 229.05 | 0.31 | 5.17 | 30.21 | 23.26 | 24.55 | 273.64 | 299.00 | 138.24 | 41.24 | 27.78 | 40.09 |
| INRA1301 | 398.57 | 186.19 | 0.41 | 14.06 | 31.43 | 22.99 | 35.42 | 124.58 | 309.90 | 149.39 | 36.55 | 19.90 | 39.46 |
| Kadata | 331.90 | 146.90 | 0.43 | 10.68 | 29.18 | 26.01 | 24.55 | 152.18 | 225.52 | 187.61 | 33.31 | 14.83 | 33.58 |
| Nabut | 662.86 | 285.00 | 0.42 | 3.10 | 25.22 | 31.69 | 28.27 | 202.18 | 292.24 | 241.83 | 27.72 | 40.87 | 24.90 |
| Noukali | 329.52 | 21.90 | 0.30 | 5.03 | 30.51 | 28.81 | 23.96 | 290.33 | 242.72 | 212.67 | 20.45 | 46.30 | 23.42 |
| Ouarraksi | 255.71 | 137.38 | 0.55 | 3.58 | 26.48 | 22.50 | 18.01 | 173.75 | 288.42 | 306.69 | 37.63 | 31.32 | 35.36 |
| Palmeras | 450.95 | 199.29 | 0.22 | 11.85 | 32.38 | 29.69 | 30.36 | 187.54 | 138.48 | 185.15 | 19.22 | 49.20 | 13.68 |
| Sarilop | 408.10 | 110.00 | 0.26 | 5.44 | 24.23 | 20.86 | 16.07 | 308.37 | 353.84 | 300.35 | 41.90 | 22.04 | 57.91 |
| Snowden | 274.76 | 20.71 | 0.39 | 10.34 | 27.01 | 35.86 | 26.49 | 152.55 | 359.54 | 138.24 | 36.62 | 15.71 | 44.56 |
| Trojana | 205.71 | 133.81 | 0.31 | 3.86 | 19.54 | 15.85 | 28.57 | 563.91 | 892.67 | 231.46 | 24.06 | 34.28 | 15.21 |
| White Adriatic | 405.71 | 60.00 | 0.35 | 16.82 | 42.63 | 24.01 | 31.40 | 166.52 | 415.02 | 125.67 | 38.22 | 20.90 | 46.45 |
| Cultivars | Gallic Acid | (+)-Catechin | (−)-Epicatechin | Chlorogenic Acid | Quercetin-3-O-rutinoside | Quercetin-3-O-Glucoside | Luteolin-7-O-Glucoside | Quercetin | Apigenin | Cyanidin-3,5-Diglucoside | Cyanidin-3-O-Rutinoside | Pelargonidin-3-O-Rutinoside | Total (µg/g) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Biondie | - | 3.38 | 14.05 | 3.34 | 55.39 | 11.66 | 7.19 | 0.86 | - | - | 95.86 | ||
| Breba Blanca | - | 1.74 | 6.37 | 0.56 | 10.98 | 6.16 | - | - | - | 0.86 | 1.06 | - | 27.74 |
| Breval Blanca | - | 5.14 | 19.01 | 1.66 | 65.05 | 7.29 | 12.43 | - | - | 0.76 | 0.84 | - | 112.18 |
| Chaari | - | 1.30 | 9.84 | 0.59 | 44.18 | 10.07 | 5.83 | - | - | 3.76 | 6.31 | - | 81.88 |
| Chetoui | 11.25 | 2.86 | 10.21 | 2.93 | 125.68 | 18.66 | 8.96 | - | - | 0.86 | 1.08 | - | 182.49 |
| Cuello Dama Blanco | - | 16.54 | 42.35 | 8.77 | 46.49 | 5.32 | 17.91 | 59.52 | 4.84 | - | - | - | 201.73 |
| El Quoti Lbied | - | 2.81 | 16.24 | 0.49 | 28.94 | 6.92 | - | 1.01 | 1.13 | 7.03 | - | - | 64.57 |
| Fassi | - | 7.76 | 19.01 | 4.12 | 147.33 | 21.06 | 15.21 | 1.00 | - | 81.08 | 83.91 | - | 380.49 |
| Ghoudan | - | 2.35 | 16.67 | 1.16 | 85.87 | 21.91 | 11.86 | 1.40 | - | 21.06 | 20.17 | - | 182.46 |
| Herida | - | 0.99 | 5.12 | 0.46 | 17.88 | 3.39 | 2.58 | - | - | 30.42 | |||
| INRA 1302 | - | 4.02 | 12.23 | 4.28 | 53.78 | 10.34 | 5.69 | 4.45 | - | 97.69 | 100.32 | - | 292.81 |
| INRA 1305 | - | 3.82 | 18.96 | 8.71 | 70.98 | 14.67 | 1.17 | 21.14 | 21.61 | - | 161.05 | ||
| INRA 2105 | - | 23.87 | 13.18 | 0.67 | 38.35 | 8.63 | 3.82 | 5.52 | 1.70 | 43.03 | 40.55 | - | 179.31 |
| INRA 2201 | - | 4.67 | 24.83 | 5.49 | 54.77 | 13.46 | 5.82 | 1.20 | 275.20 | 246.86 | 4.16 | 632.31 | |
| INRA 2304 | - | 7.45 | 11.07 | 0.86 | 64.49 | 14.19 | 7.26 | 3.87 | 130.12 | 131.23 | - | 370.54 | |
| INRA1301 | - | 14.36 | 54.66 | 7.81 | 141.08 | 35.48 | 11.50 | 2.17 | 494.08 | 478.66 | 12.56 | 1239.80 | |
| Kadata | - | 0.92 | 6.21 | 1.40 | 20.23 | 2.56 | 3.34 | 0.91 | 1.11 | - | 36.69 | ||
| Nabout | 2.22 | 7.01 | 9.86 | 0.90 | 33.78 | 6.03 | 6.71 | 1.46 | - | 1.04 | 1.15 | - | 70.15 |
| Noukali | - | 6.28 | 31.98 | 8.39 | 112.91 | 21.47 | 1.86 | - | - | 35.89 | 34.88 | - | 253.66 |
| Ouarraksi | - | 3.19 | 12.42 | 3.71 | 92.49 | 11.22 | 8.61 | - | - | 0.83 | 0.94 | - | 133.41 |
| Palmeras | - | 18.25 | 44.22 | 4.48 | 23.15 | 18.31 | 15.03 | - | 1.40 | - | - | - | 151.77 |
| Sarilop | - | 3.60 | 14.91 | 2.30 | 41.88 | 4.64 | 4.30 | - | - | - | - | - | 71.63 |
| Snowden | - | 2.83 | 11.52 | 2.24 | 49.46 | 6.86 | 5.33 | - | - | - | - | - | 78.23 |
| Trojana | - | 2.67 | 16.47 | 3.25 | 2.39 | - | - | - | - | - | 24.78 | ||
| White Adriatic | - | 2.24 | 5.09 | 0.57 | 19.79 | 3.53 | 5.17 | 0.91 | - | - | - | - | 37.30 |
| Cultivars (Pulp) | (+)-Catechin | (−)-Epicatechin | Chlorogenic Acid | Quercetine-3-O-Rutinoside | Quercetine-3-O-Glucoside | Luteoline-7-O-Glucoside | Cyanidine-3.5-Diglucoside | Cyanidine-3-O-Rutinoside | Total (µg/g) |
|---|---|---|---|---|---|---|---|---|---|
| Biondie | - | 2.15 | - | - | - | - | 0.82 | 1.31 | 1.43 |
| Breba Blanca | 1.12 | 3.38 | 0.46 | - | - | - | 3.86 | 1.85 | 2.13 |
| Breval Blanca | 1.71 | 4.79 | 0.33 | 0.92 | - | - | 5.53 | 8.05 | 3.56 |
| Chaari | 1.43 | 4.09 | - | 1.23 | - | - | 2.59 | 3.8 | 2.63 |
| Chetoui | 1 | 7.99 | 1.3 | 26.8 | 4.03 | 0.75 ± 0.35 | 6.87 | 11.87 | 8.55 |
| Cuello Dama Blanca | - | 3.85 | 3.14 | 1.23 | 0.98 | - | 4.74 | 11.05 | 4.17 |
| El Quoti Lbied | - | 1.75 | 0.38 | - | - | - | 3.91 | 7.93 | 3.49 |
| Fassi | 2.63 | 9.08 | 0.68 | 1.23 | - | - | 11.93 | 16.94 | 7.08 |
| Ghoudan | 1.92 | 2.47 | 0.31 | 1.51 | 1.31 | - | 2.41 | 5.42 | 2.19 |
| Herida | 0.67 | 2.03 | - | - | - | - | 0.9 | 1.04 | 1.16 |
| INRA 1302 | 2.96 | 9.91 | 1.22 | 1.06 | - | - | 14.74 | 20.37 | 8.38 |
| INRA 1305 | 2.11 | 7.83 | 1.04 | - | - | - | - | 1.52 | 3.13 |
| INRA 2105 | 6.63 | 19.05 | 4.83 | 2.21 | 1.45 | - | 20.38 | 28.78 | 11.90 |
| INRA 2201 | 1.34 | 1.75 | 0.4 | - | - | - | 4.73 | 10.44 | 3.73 |
| INRA 2304 | - | 1.27 | - | 0.81 | - | - | 2.83 | 5.12 | 2.51 |
| INRA 1301 | 0.73 | 1.3 | - | - | - | - | 0.83 | 1.39 | 1.06 |
| Kadota | 1.2 | 8.37 | 0.44 | 1.02 | - | - | 8.98 | 16.6 | 6.10 |
| Nabout | 1.5 | 2.52 | 0.36 | 1.03 | 1.05 | - | 1.11 | 3.03 | 1.51 |
| Noukali | 1.67 | 4.28 | 0.33 | 1.48 | - | - | 6.15 | 11.1 | 4.17 |
| Ouarraksi | 1.39 | 1.65 | 0.47 | 1.42 | - | - | 4.11 | 9.68 | 3.12 |
| Palmeras | 3.06 | 7.23 | 2.07 | 1.92 | 2.19 | 4.47 ± 0.04 | 6.88 | 8.81 | 4.59 |
| Sarilop | 1.82 | 10.88 | 0.49 | 0.89 | - | - | 0.93 | 2.25 | 2.88 |
| Snowden | - | 5.08 | 0.37 | - | - | - | 0.89 | 1.57 | 1.98 |
| Trojana | - | 4.15 | - | 1.2 | - | - | 0.84 | 0.96 | 1.79 |
| White Adriatic | 1.83 | 3.91 | 0.58 | 1.22 | - | - | 28.43 | 34.42 | 11.73 |
| Cultivars | Geographical Origin | August | September | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| (1–5) | (6–10) | (11–15) | (16–20) | (21–25) | (26–30) | (31–4) | (5–9) | |||
| Local | El Quoti Lbied | Morocco | ||||||||
| Nabout | ||||||||||
| Fassi | ||||||||||
| Noukali | ||||||||||
| Ghoudan | ||||||||||
| Chetoui | ||||||||||
| Bioudie | ||||||||||
| Chaari | ||||||||||
| Ournaksi | ||||||||||
| INRA 1305 | ||||||||||
| INRA 2105 | ||||||||||
| INRA 1302 | ||||||||||
| INRA 2201 | ||||||||||
| INRA 2304 | ||||||||||
| INRA 1301 | ||||||||||
| Introduced | Snowden | USA | ||||||||
| White Adriatic | Italy | |||||||||
| Kadota | Italy | |||||||||
| Triana | Italy | |||||||||
| Cuello Dama Blanca | Spain | |||||||||
| Breval Blanca | Spain | |||||||||
| Palmeras | Spain | |||||||||
| Herida | Spain | |||||||||
| Breba Blanca | Spain | |||||||||
| Total rainfall (mm) | 0 | 0 | 0 | 0 | 0 | 26.4 | 0 | 0 | ||
| Average temperature (°C) | 25.84 | 28.5 | 27.56 | 29.24 | 29.44 | 23.64 | 25.6 | 25.42 | ||
| Average solar radiation (W/m²) | 169.29 | 208.74 | 243.83 | 238.28 | 185.35 | 123.5 | 270.21 | 271.38 | ||
| Soil type | Sandy clay loam with an average organic matter of 1% [0–30 cm soil layer] | |||||||||
| Soil pH | 7.2 | |||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hssaini, L.; Hernandez, F.; Viuda-Martos, M.; Charafi, J.; Razouk, R.; Houmanat, K.; Ouaabou, R.; Ennahli, S.; Elothmani, D.; Hmid, I.; et al. Survey of Phenolic Acids, Flavonoids and In Vitro Antioxidant Potency Between Fig Peels and Pulps: Chemical and Chemometric Approach. Molecules 2021, 26, 2574. https://doi.org/10.3390/molecules26092574
Hssaini L, Hernandez F, Viuda-Martos M, Charafi J, Razouk R, Houmanat K, Ouaabou R, Ennahli S, Elothmani D, Hmid I, et al. Survey of Phenolic Acids, Flavonoids and In Vitro Antioxidant Potency Between Fig Peels and Pulps: Chemical and Chemometric Approach. Molecules. 2021; 26(9):2574. https://doi.org/10.3390/molecules26092574
Chicago/Turabian StyleHssaini, Lahcen, Francisca Hernandez, Manuel Viuda-Martos, Jamal Charafi, Rachid Razouk, Karim Houmanat, Rachida Ouaabou, Said Ennahli, Driss Elothmani, Ilham Hmid, and et al. 2021. "Survey of Phenolic Acids, Flavonoids and In Vitro Antioxidant Potency Between Fig Peels and Pulps: Chemical and Chemometric Approach" Molecules 26, no. 9: 2574. https://doi.org/10.3390/molecules26092574
APA StyleHssaini, L., Hernandez, F., Viuda-Martos, M., Charafi, J., Razouk, R., Houmanat, K., Ouaabou, R., Ennahli, S., Elothmani, D., Hmid, I., Fauconnier, M. L., & Hanine, H. (2021). Survey of Phenolic Acids, Flavonoids and In Vitro Antioxidant Potency Between Fig Peels and Pulps: Chemical and Chemometric Approach. Molecules, 26(9), 2574. https://doi.org/10.3390/molecules26092574











