Graphene Assisted in the Analysis of Coumarins in Angelicae Pubescentis Radix by Dispersive Liquid–Liquid Microextraction Combined with 1H-qNMR
Abstract
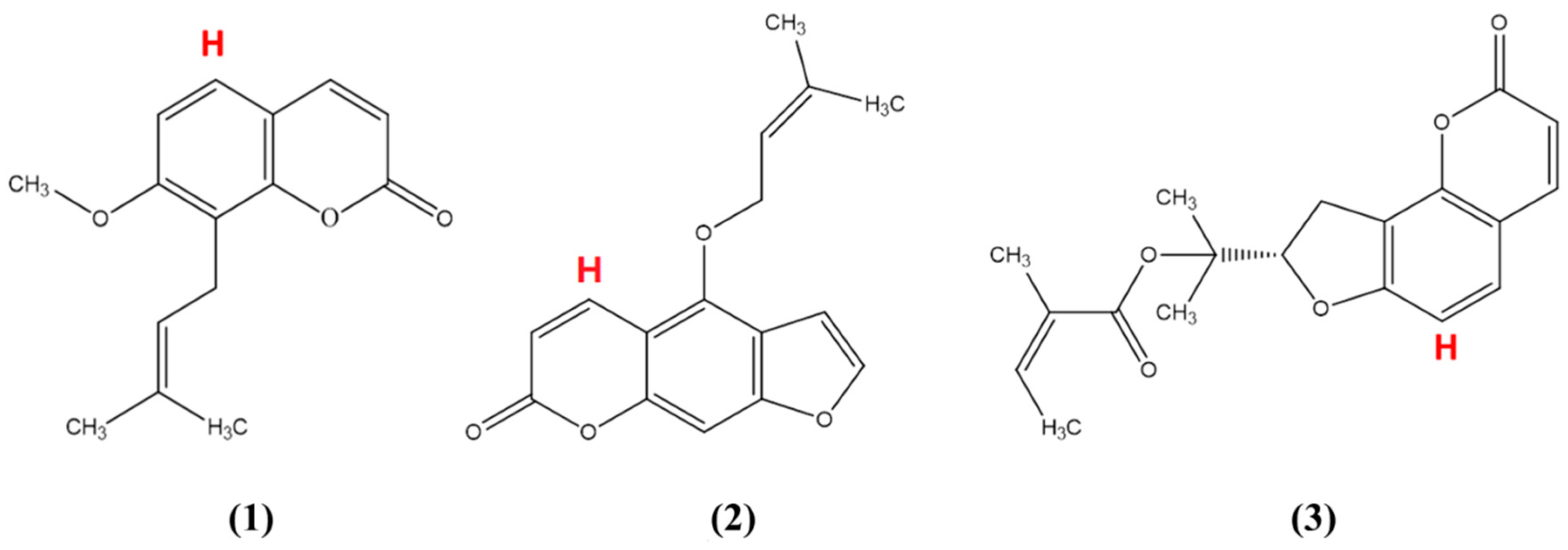
1. Introduction
2. Results and Discussion
2.1. Optimization of NMR Parameters
2.1.1. Choice of D1
2.1.2. Choice of NS
2.2. Optimization of DLLME-SFO Conditions
2.2.1. Choice of Extractant and Dispersant
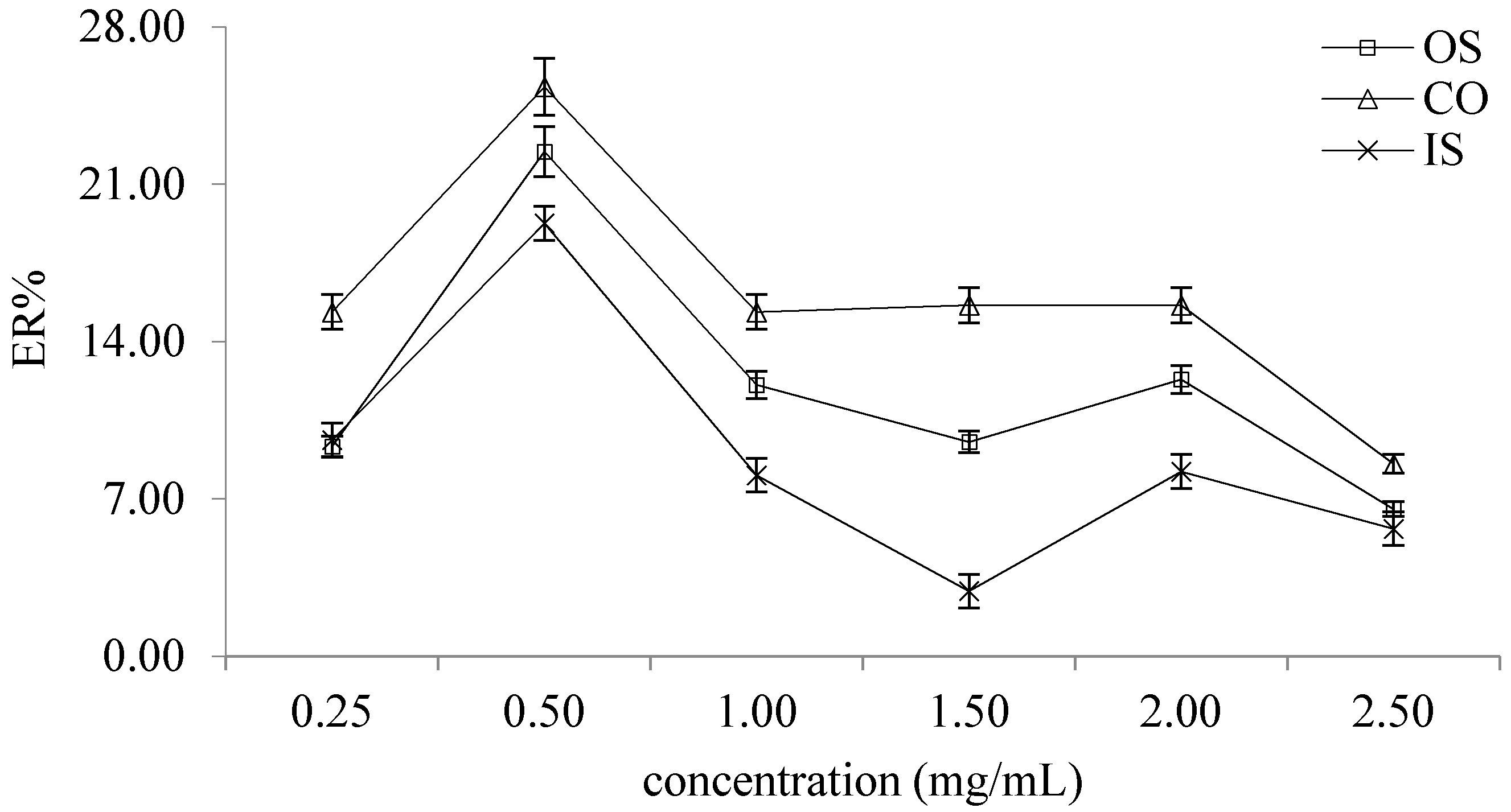
2.2.2. Selection of Volume Ratio of Distilled Water to Test Sample Solution
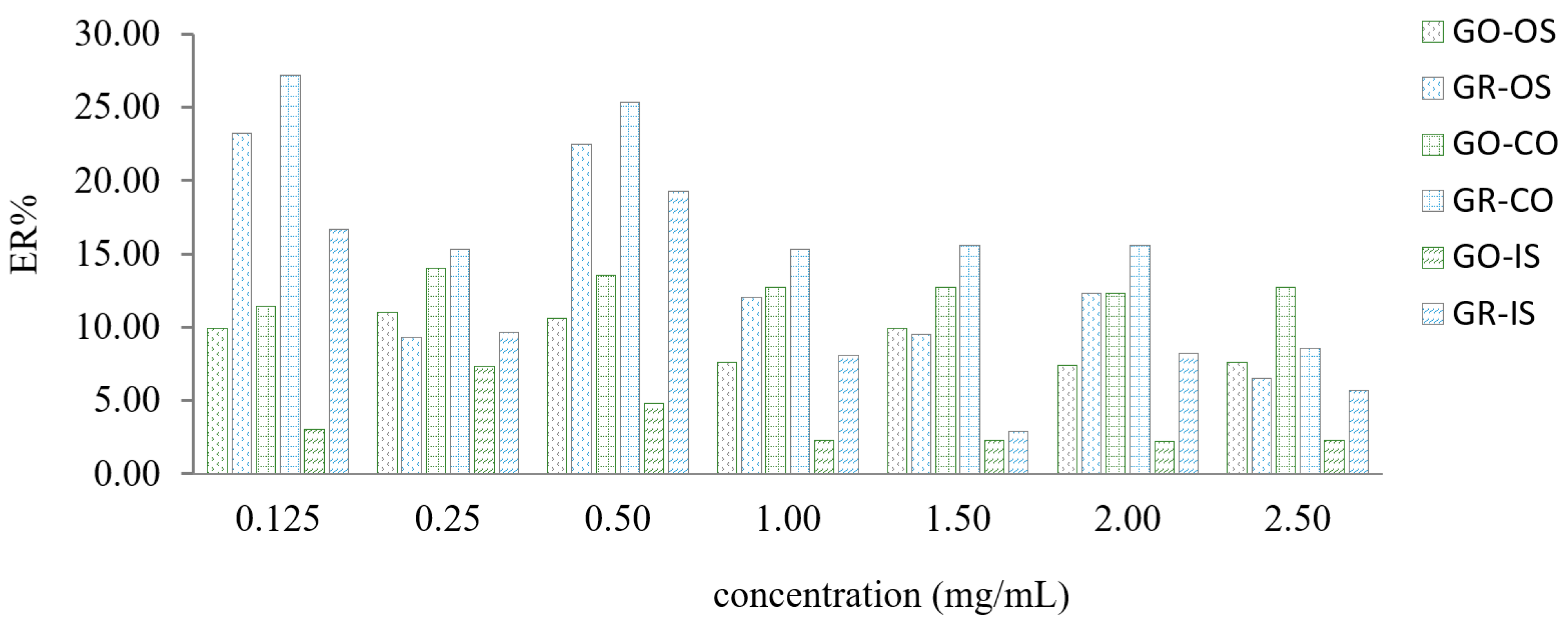
2.2.3. Selection of Graphene Concentration in 1-Dodecyl Alcohol
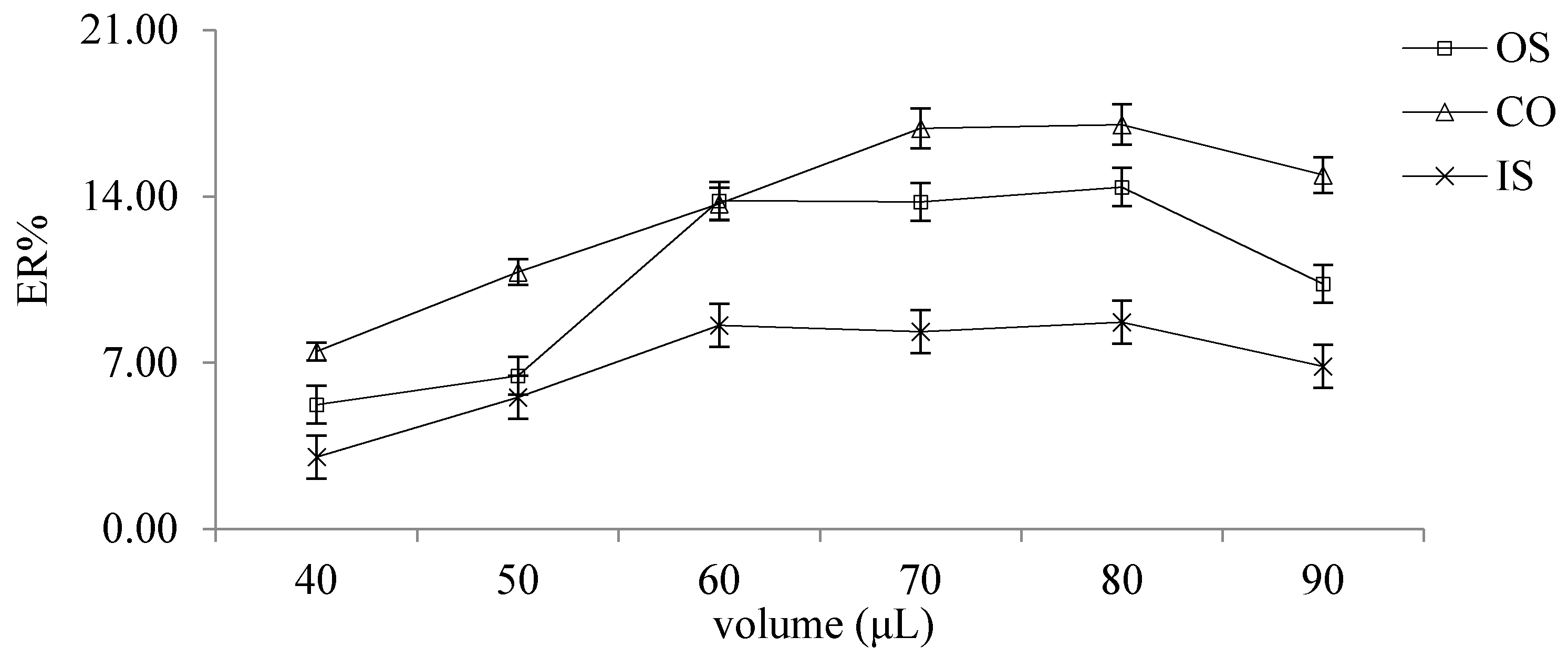
2.2.4. Selection of Extractant Volume
2.2.5. Selection of Extractant Volume
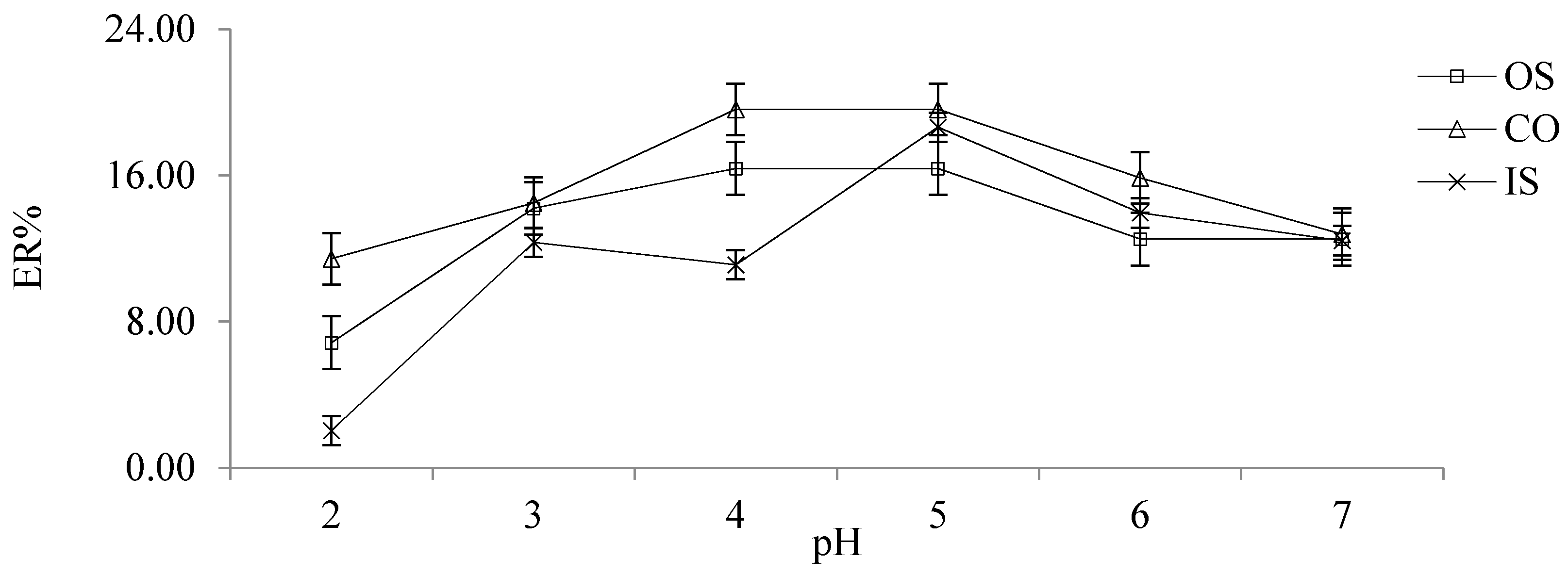
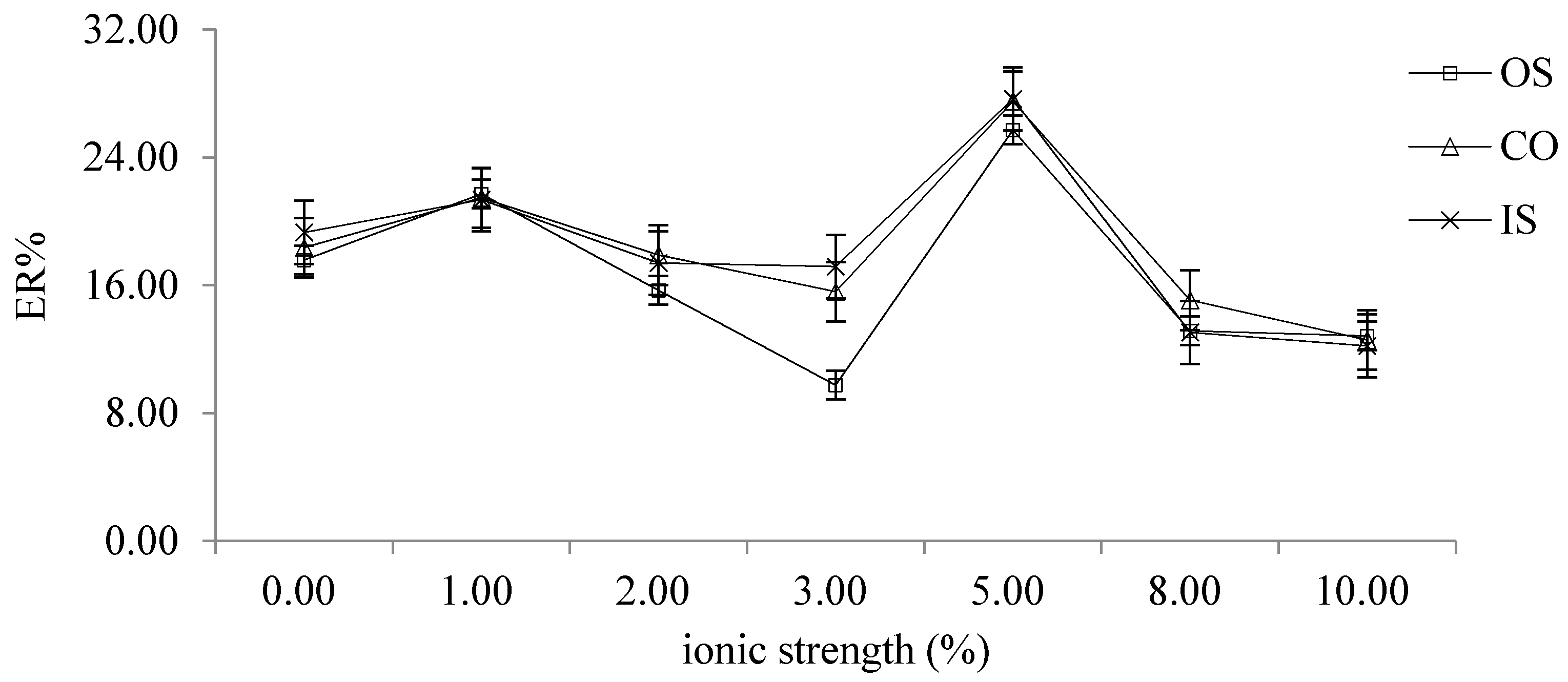
2.2.6. Choice of Ionic Strength
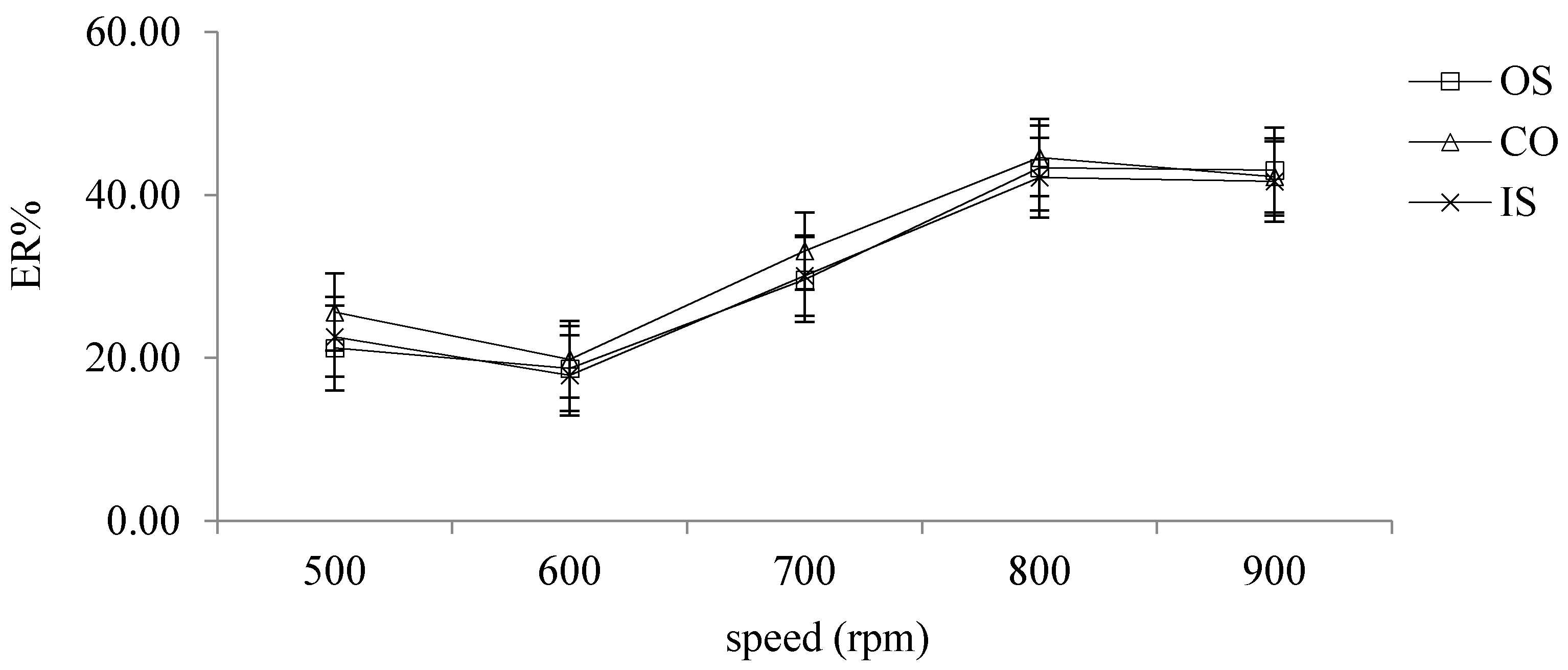
2.2.7. Selection of Different Speed
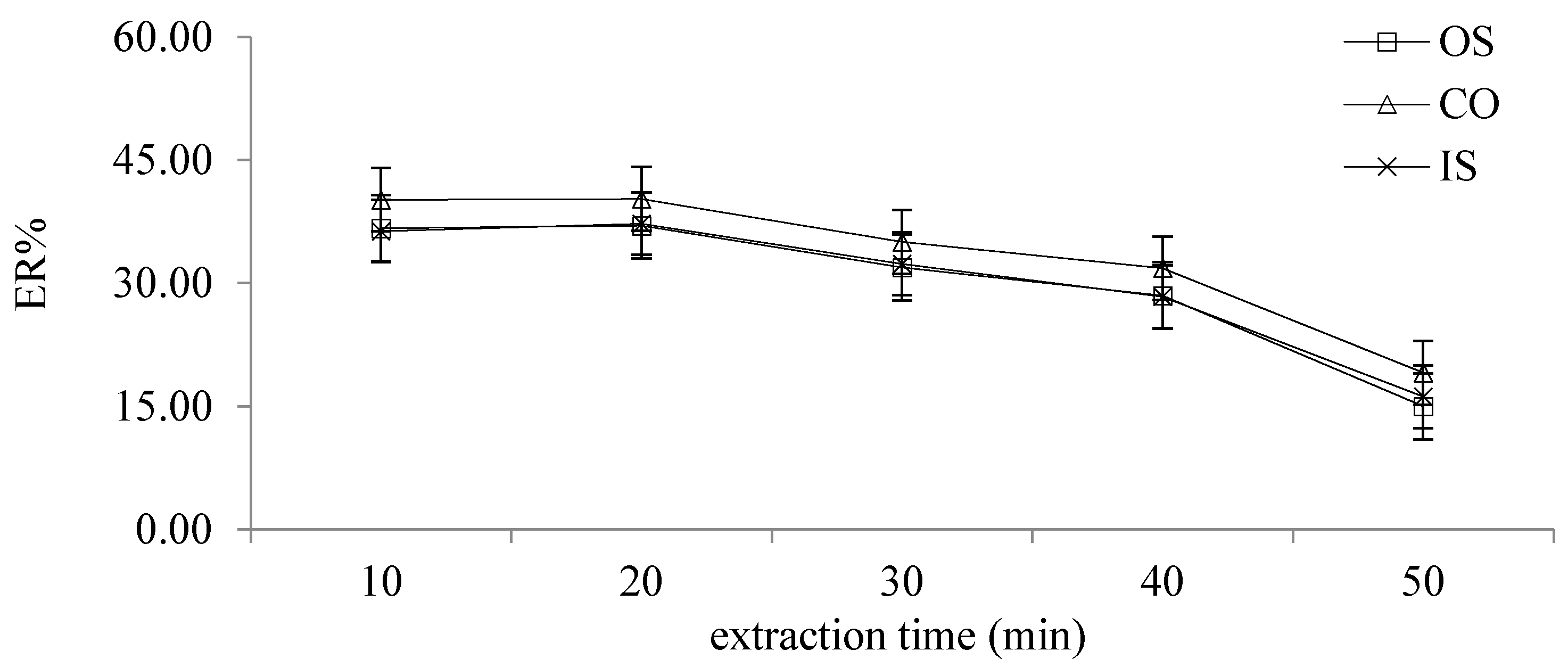
2.2.8. Different Extraction Times
2.3. Methodology Validation
2.3.1. Linearity
2.3.2. Limit of Detection and Limit of Quantification
2.3.3. Precision
2.3.4. Stability and Repeatability
2.3.5. Recovery
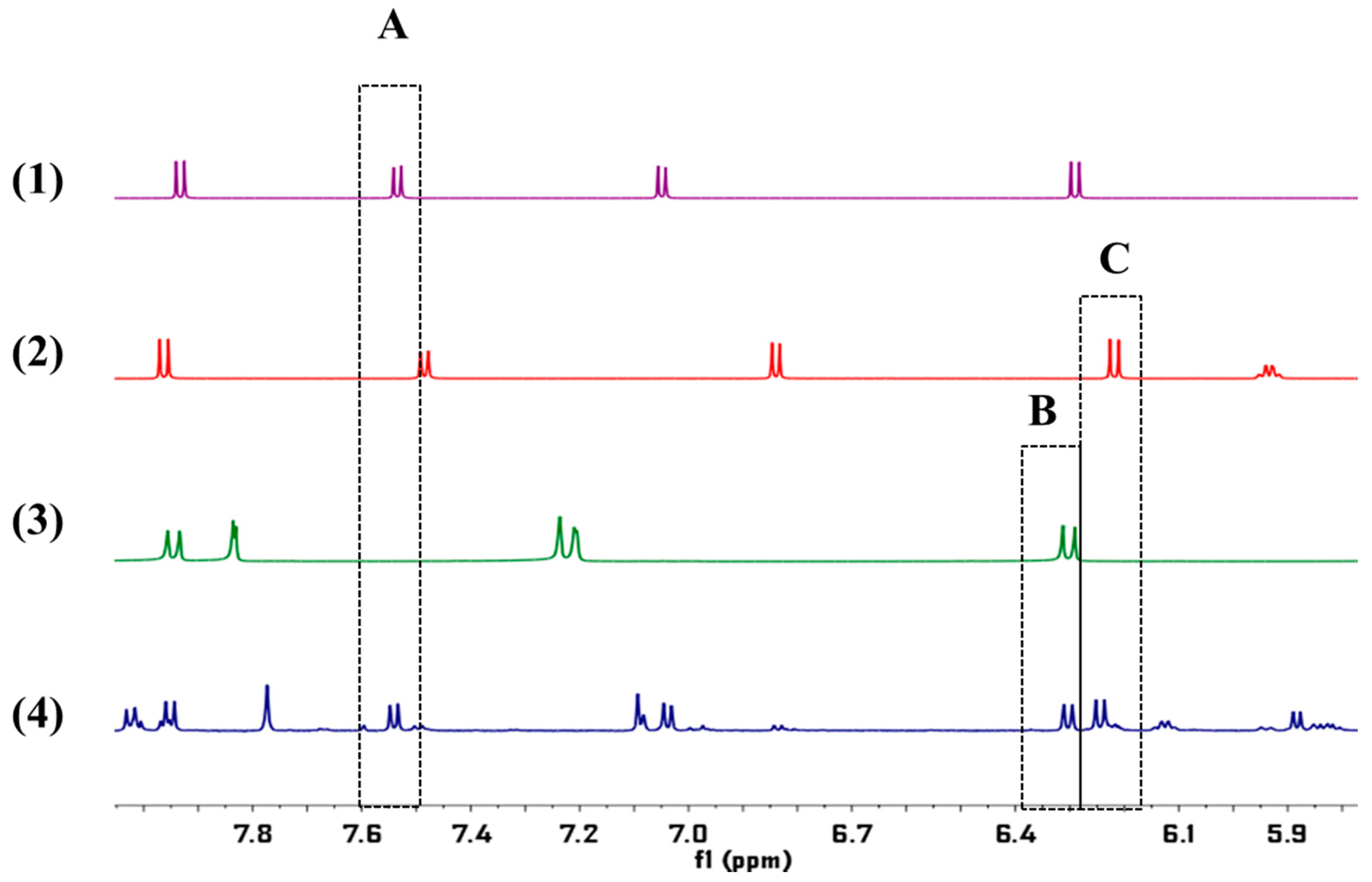
2.4. Determination of the Samples
3. Materials and Methods
3.1. Reagent
3.2. Chemical
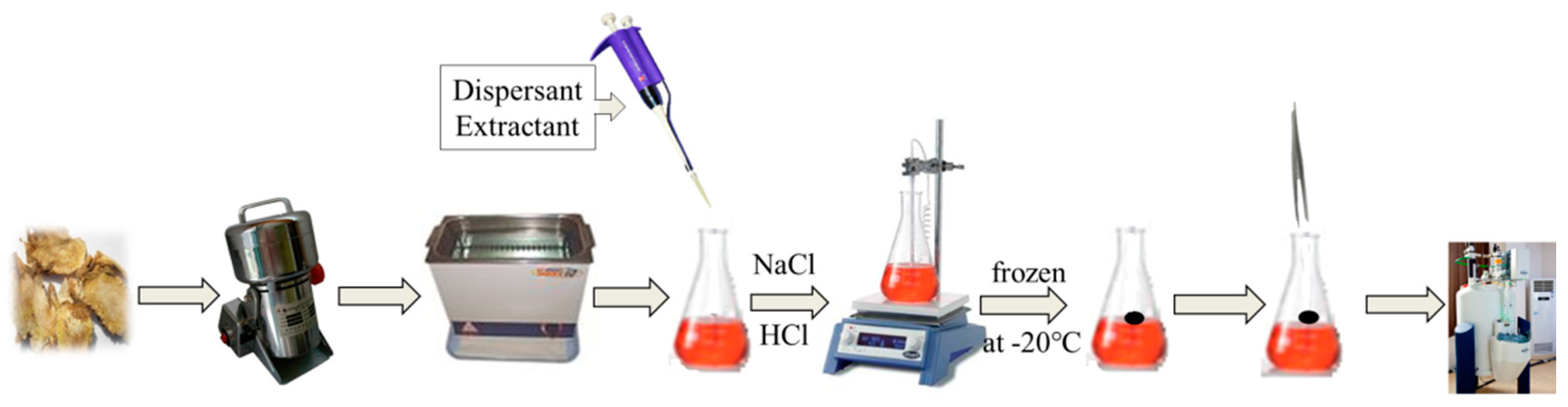
3.3. Method
3.3.1. Preparation of Sample Solution
3.3.2. Extraction Process of DLLME-SFO
3.3.3. NMR Acquisition and Processing Parameters
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Chinese Pharmacopoeia Commission. Pharmacopoeia of People’s Republic of China; China Medical Science and Technology Press: Beijing, China, 2020; Volume 1, pp. 133–134. [Google Scholar]
- Lu, Y.Q.; Wu, H.W.; Yu, X.K.; Zhang, X.; Luo, H.Y.; Tang, L.Y.; Wang, Z.J. Traditional Chinese medicine of Angelicae pubescentis radix: A review of phytochemistry, pharmacology and pharmacokinetics. Front. Pharmacol. 2020, 11, 355. [Google Scholar] [CrossRef]
- Zhao, Y.S.; Hou, X.Y.; Gao, Z.H.; Wang, T. Tracing sources of prescriptions for treating dampness in initial stage of Corona Virus disease 2019. Chin. Arch. Trad. Chin. Med. 2020, 4, 4–7. [Google Scholar]
- Chen, X. Enlightenment of “lung wind” in Nei Jing on the treatment of COVID-19 JIN Jue. Shanghai J. Trad. Chin. Med. 2020, 5, 6–10. [Google Scholar]
- Yang, Y.F.; Zhang, L.; Zhang, Y.B.; Yang, X.W. Simultaneous assessment of absorption characteristics of coumarins from Angelicae Pubescentis Radix: In vitro transport across Caco-2 cell and in vivo pharmacokinetics in rats after oral administration. J. Chromatogr. B 2017, 1060, 308–315. [Google Scholar] [CrossRef]
- Li, X.; Wang, J.; Gao, L. Anti-inflammatory and analgesic activity of r.a.p. (Radix Angelicae Pubescentis) ethanol extracts. Afr. J. Trad. Complement. Altern. Med. 2013, 10, 422–426. [Google Scholar] [CrossRef]
- Li, R.; Zhao, C.; Yao, M.; Song, Y.; Wu, Y.; Wen, A. Analgesic effect of coumarins from Radix angelicae pubescentis is mediated by inflammatory factors and TRPV1 in a spared nerve injury model of neuropathic pain. J. Ethnopharmacol. 2017, 195, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Jiao, X.; Li, J.; Yu, X.; Liu, W.; Tian, J.; He, J.; Chang, Y. The pharmacokinetics, bioavailability and excretion of columbianetin acetate and its metabolite columbianetin were analysed in rat plasma by LC-MS/MS after administration of columbianetin acetate and Angelicae pubescentis radix extract. RSC Adv. 2015, 5, 95882–95893. [Google Scholar] [CrossRef]
- Emami, S.; Dadashpour, S. Current developments of coumarin-based anti-cancer agents in medicinal chemistry. Eur. J. Med. Chem. 2015, 102, 611–630. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Sun, M.; Zhang, Y.; Zhang, J.; He, L. EGFR/cell membrane chromatography-online-high performance liquid chromatography/mass spectrometry method for screening EGFR antagonists from Radix Angelicae Pubescentis. Sci. China Chem. 2010, 53, 2357–2362. [Google Scholar] [CrossRef]
- Horstkotte, B.; Maya, F.; Duarte, C.M.; Cerdà, V. Determination of ppb-level phenol index using in-syringe dispersive liquid-liquid microextraction and liquid waveguide capillary cell spectrophotometry. Microchim. Acta 2012, 179, 91–98. [Google Scholar] [CrossRef]
- Alexovič, M.; Andruch, V.; Balogh, I.S.; Šandrejová, J. A single-valve sequential injection manifold (SV-SIA) for automation of air-assisted liquid-phase microextraction: Stopped flow spectrophotometric determination of chromium(vi). Anal. Meth. UK 2013, 5, 2497. [Google Scholar] [CrossRef]
- Chen, P.S.; Haung, W.Y.; Huang, S.D. Analysis of triazine herbicides using an up-and-down-shaker-assisted dispersive liquid–liquid microextraction coupled with gas chromatography–mass spectrometry. J. Chromatogr. B 2014, 955–956, 116–123. [Google Scholar] [CrossRef]
- Horstkotte, B.; Suárez, R.; Solich, P.; Cerdà, V. In-syringe magnetic stirring assisted dispersive liquid–liquid micro-extraction with solvent washing for fully automated determination of cationic surfactants. Anal. Meth. UK 2014, 6, 9601–9609. [Google Scholar] [CrossRef]
- Alexovič, M.; Wieczorek, M.; Kozak, J.; Kościelniak, P.; Balogh, I.S.; Andruch, V. An automatic, vigorous-injection assisted dispersive liquid–liquid microextraction technique for stopped-flow spectrophotometric detection of boron. Talanta 2015, 133, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Leong, M.I.; Huang, S.D. Dispersive liquid–liquid microextraction method based on solidification of floating organic drop combined with gas chromatography with electron-capture or mass spectrometry detection. J. Chromatogr. A 2008, 1211, 8–12. [Google Scholar] [CrossRef]
- Berijani, S.; Assadi, Y.; Anbia, M.; Milani Hosseini, M.R.; Aghaee, E. Dispersive liquid–liquid microextraction combined with gas chromatography-flame photometric detection. J. Chromatogr. A 2006, 1123, 1–9. [Google Scholar] [CrossRef]
- Rezaee, M.; Assadi, Y.; Milani Hosseini, M.R.; Aghaee, E.; Ahmadi, F.; Berijani, S. Determination of organic compounds in water using dispersive liquid–liquid microextraction. J. Chromatogr. A 2006, 1116, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Farahani, H.; Norouzi, P.; Dinarvand, R.; Ganjali, M.R. Development of dispersive liquid–liquid microextraction combined with gas chromatography–mass spectrometry as a simple, rapid and highly sensitive method for the determination of phthalate esters in water samples. J. Chromatogr. A 2007, 1172, 105–112. [Google Scholar] [CrossRef]
- Zang, X.H.; Wu, Q.H.; Zhang, M.Y.; Xi, G.H.; Wang, Z. Developments of dispersive liquid-liquid microextraction technique. Chin. J. Anal. Chem. 2009, 37, 161–168. [Google Scholar] [CrossRef]
- Khalili Zanjani, M.R.; Yamini, Y.; Shariati, S.; Jönsson, J.Å. A new liquid-phase microextraction method based on solidification of floating organic drop. Anal. Chim. Acta 2007, 585, 286–293. [Google Scholar] [CrossRef]
- Shamsipur, M.; Fattahi, N.; Sadeghi, M.; Pirsaheb, M. Determination of ultra traces of lead in water samples after combined solid-phase extraction–dispersive liquid–liquid microextraction by graphite furnace atomic absorption spectrometry. J. Iran. Chem. Soc. 2013, 11, 249–256. [Google Scholar] [CrossRef]
- Sadeghi, M.; Nematifar, Z.; Irandoust, M.; Fattahi, N.; Hamzei, P.; Barati, A.; Shamsipur, M. Efficient and selective extraction and determination of ultra trace amounts of Hg2+ using solid phase extraction combined with ion pair based surfactant-assisted dispersive liquid–liquid microextraction. RSC Adv. 2015, 5, 100511–100521. [Google Scholar] [CrossRef]
- Sun, X.; Xing, X.; Du, Z. Application of dispersive liquid–liquid microextraction based on solidification of floating organic drop for the determination of extractables from pharmaceutical packaging materials. Talanta 2019, 120540. [Google Scholar] [CrossRef] [PubMed]
- Pirsaheb, M.; Fattahi, N. Trace determination of heavy metals in farmed trout fish using dispersive liquid–liquid microextraction based on solidification of floating organic drop and graphite furnace atomic absorption spectrometry. Anal. Meth. UK 2015, 7, 6266–6273. [Google Scholar] [CrossRef]
- Ahmadi-Jouibari, T.; Fattahi, N. Speciation of inorganic arsenic species and total inorganic arsenic in rice using microwave-assisted dispersive liquid–liquid micro-extraction and electrothermal atomic absorption spectrometry. Food Addit. Contam. A 2015, 32, 1140–1147. [Google Scholar] [CrossRef] [PubMed]
- Rezaee, M.; Khalilian, F.; Mashayekhi, H.A.; Fattahi, N. A novel method for the high preconcentration of trace amounts of the aflatoxins in pistachios by dispersive liquid–liquid microextraction after solid-phase extraction. Anal. Meth. UK 2014, 6, 3456–3461. [Google Scholar] [CrossRef]
- Taheri, S.; Jalali, F.; Fattahi, N.; Bahrami, G. Sensitive determination of atorvastatin in human plasma by dispersive liquid-liquid microextraction and solidification of floating organic drop followed by high-performance liquid chromatography. J. Sep. Sci. 2014, 38, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Hryniewicka, M.; Starczewska, B.; Gołębiewska, A. Determination of budesonide and sulfasalazine in water and wastewater samples using DLLME-SFO-HPLC-UV Method. Water 2019, 11, 1581. [Google Scholar] [CrossRef]
- Ahmadi-Jouibari, T.; Fattahi, N.; Shamsipur, M.; Pirsaheb, M. Dispersive liquid–liquid microextraction followed by high-performance liquid chromatography–ultraviolet detection to determination of opium alkaloids in human plasma. J. Pharm. Biomed. 2013, 85, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi-Jouibari, T.; Fattahi, N.; Shamsipur, M. Rapid extraction and determination of amphetamines in human urine samples using dispersive liquid–liquid microextraction and solidification of floating organic drop followed by high performance liquid chromatography. J. Pharm. Biomed. 2014, 94, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Zheng, X.; Zhang, C.; Ma, X.; Ling, Q.; Zhao, L. Ultrasound-assisted dispersive liquid–liquid microextraction based on the solidification of a floating organic droplet followed by gas chromatography for the determination of eight pyrethroid pesticides in tea samples. J. Chromatogr. B 2014, 969, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Cheng, J.; Matsadiq, G.; Liu, L.; Li, J.-K. Dispersive liquid–liquid microextraction based on the solidification of floating organic droplet for the determination of polychlorinated biphenyls in aqueous samples. Anal. Chim. Acta 2010, 674, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Sanagi, M.M.; Abbas, H.H.; Ibrahim, W.A.W.; Aboul-Enien, H.Y. Dispersive liquid–liquid microextraction method based on solidification of floating organic droplet for the determination of triazine herbicides in water and sugarcane samples. Food Chem. 2012, 133, 557–562. [Google Scholar] [CrossRef] [PubMed]
- El-Shahawi, M.S.; Al-Saidi, H.M. Dispersive liquid-liquid microextraction for chemical speciation and determination of ultra-trace concentrations of metal ions. Trac. Trend Anal. Chem. 2013, 44, 12–24. [Google Scholar] [CrossRef]
- Rezaee, M.; Yamini, Y.; Khanchi, A.; Faraji, M.; Saleh, A. A simple and rapid new dispersive liquid–liquid microextraction based on solidification of floating organic drop combined with inductively coupled plasma-optical emission spectrometry for preconcentration and determination of aluminium in water samples. J. Hazard Mater. 2010, 178, 766–770. [Google Scholar] [CrossRef] [PubMed]
- Mandlate, J.S.; Soares, B.M.; Seeger, T.S.; Vecchia, P.D.; Mello, P.A.; Flores, E.M.M.; Duarte, F.A. Determination of cadmium and lead at sub-ppt level in soft drinks: An efficient combination between dispersive liquid-liquid microextraction and graphite furnace atomic absorption spectrometry. Food Chem. 2017, 221, 907–912. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. Nature 2017, 6, 183–191. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Fal′ko, V.I.; Colombo, L.; Gellert, P.R.; Schwab, M.G.; Kim, K. A roadmap for graphene. Nature 2012, 490, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Geim, A.K. Graphene: Status and Prospects. Science 2009, 324, 1530–1534. [Google Scholar] [CrossRef]
- Ma, X.; Wang, J.; Wu, Q.; Wang, C.; Wang, Z. Extraction of carbamate pesticides in fruit samples by graphene reinforced hollow fibre liquid microextraction followed by high performance liquid chromatographic detection. Food Chem. 2014, 157, 119–124. [Google Scholar] [CrossRef]
- Sun, M.; Tang, R.; Wu, Q.; Wang, C.; Wang, Z. Graphene Reinforced Hollow Fiber Liquid Phase Microextraction for the Enrichment of some Phenylurea Residues in Milk Sample. Food Anal. Meth. 2013, 7, 1097–1102. [Google Scholar] [CrossRef]
- Zhang, W.; Zhou, W.; Chen, Z. Graphene/polydopamine-modified polytetrafluoroethylene microtube for the sensitive determination of three active components in Fructus Psoraleaeby online solid-phase microextraction with high-performance liquid chromatography. J. Sep. Sci. 2014, 37, 3110–3116. [Google Scholar] [CrossRef] [PubMed]
- Yang, X. The Establishment of Novel Dispersive Liquid Phase Microextraction Methods and Their Application for Analysis of Cinnamic Acid Derivatives in Traditional Chinese Medicines; Shanxi Medical University: Taiyuan, China, 2018. [Google Scholar]
- Feng, Y.; Li, Q.; Yang, L.; Zhang, Y.; Qiu, D. The Use of 1H-qNMR Method for Simultaneous Determination of Osthol, Columbianadin, and Isoimperatorin in Angelicae Pubescentis Radix. J. Aoac. Int. 2020, 103, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Owczarek, A.; Klys, A.; Olszewska, M.A. A validated 1H qNMR method for direct and simultaneous quantification of esculin, fraxin and (−)-epicatechin in Hippocastani cortex. Talanta 2019, 192, 263–269. [Google Scholar] [CrossRef] [PubMed]

| Detection Method | Extraction Method | ||||
|---|---|---|---|---|---|
| 1H-qNMR | HPLC | DLLME-SFO | Other extraction methods | ||
| Requirement on sample form | No specific requirements | Liquid | Volume of samples required | 1 mL | ≥10 mL |
| Demand for reference substances | No | Yes | Need to stand layer | No | Yes |
| Detection test | ≤5 min | >10 min | Extraction time | 20 min | >20 min |
| Recycling of samples after measurement | Yes | No | Simultaneous separation and extraction | Yes | No |
| Analyte | Linear Regression Equation | R2 | LOD (mg/mL) | LOQ (mg/mL) |
|---|---|---|---|---|
| OS | y = 0.0798x + 0.0008 | 0.9994 | 0.0660 | 0.2201 |
| CO | y = 0.0822x + 0.0007 | 0.9994 | 0.0720 | 0.2401 |
| IS | y = 0.0784x − 0.0011 | 0.9995 | 0.0620 | 0.2066 |
| Analyte | Original Content (g) | Added Content (g) | Total Observed (g) | Recovery (%) | Average Recovery (%) | RSD (%) |
|---|---|---|---|---|---|---|
| OS | 0.0057 | 0.0057 | 0.0113 | 98.19 | 102.26 | 3.62 |
| 0.0117 | 104.46 | |||||
| 0.0117 | 106.12 | |||||
| 0.0112 | 97.37 | |||||
| 0.0117 | 105.13 | |||||
| CO | 0.0085 | 0.0085 | 0.0170 | 100.07 | 99.89 | 3.86 |
| 0.0168 | 97.66 | |||||
| 0.0166 | 94.29 | |||||
| 0.0172 | 101.64 | |||||
| 0.0175 | 105.79 | |||||
| IS | 0.0058 | 0.0057 | 0.0114 | 97.93 | 103.28 | 4.15 |
| 0.0121 | 109.33 | |||||
| 0.0118 | 103.68 | |||||
| 0.0115 | 99.11 | |||||
| 0.0119 | 106.34 |
| Number | Content (g/g) | ||
|---|---|---|---|
| OS | CO | IS | |
| 1 | 0.0023 | 0.0040 | 0.0017 |
| 2 | 0.0016 | 0.0021 | - |
| 3 | 0.0024 | 0.0040 | - |
| 4 | 0.0003 | 0.0006 | 0.0007 |
| 5 | 0.0002 | 0.0010 | 0.0002 |
| 6 | 0.0026 | 0.0049 | 0.0030 |
| 7 | 0.0043 | 0.0061 | 0.0002 |
| 8 | 0.0042 | 0.0069 | 0.0007 |
| 9 | 0.0042 | 0.0069 | 0.0008 |
| 10 | 0.0020 | 0.0055 | 0.0015 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, Y.; Li, Q.; Qiu, D.; Li, G. Graphene Assisted in the Analysis of Coumarins in Angelicae Pubescentis Radix by Dispersive Liquid–Liquid Microextraction Combined with 1H-qNMR. Molecules 2021, 26, 2416. https://doi.org/10.3390/molecules26092416
Feng Y, Li Q, Qiu D, Li G. Graphene Assisted in the Analysis of Coumarins in Angelicae Pubescentis Radix by Dispersive Liquid–Liquid Microextraction Combined with 1H-qNMR. Molecules. 2021; 26(9):2416. https://doi.org/10.3390/molecules26092416
Chicago/Turabian StyleFeng, Yanmei, Qian Li, Daiyu Qiu, and Guichen Li. 2021. "Graphene Assisted in the Analysis of Coumarins in Angelicae Pubescentis Radix by Dispersive Liquid–Liquid Microextraction Combined with 1H-qNMR" Molecules 26, no. 9: 2416. https://doi.org/10.3390/molecules26092416
APA StyleFeng, Y., Li, Q., Qiu, D., & Li, G. (2021). Graphene Assisted in the Analysis of Coumarins in Angelicae Pubescentis Radix by Dispersive Liquid–Liquid Microextraction Combined with 1H-qNMR. Molecules, 26(9), 2416. https://doi.org/10.3390/molecules26092416






