Production of Pyracantha Polysaccharide-Iron(III) Complex and Its Biologic Activity
Abstract
1. Introduction
2. Results and Discussion
2.1. Establishment of a Standard Curve for Iron
2.2. Response Surface Optimization of PPI Complexsynthesis Conditions
2.3. Characterisation of PPI
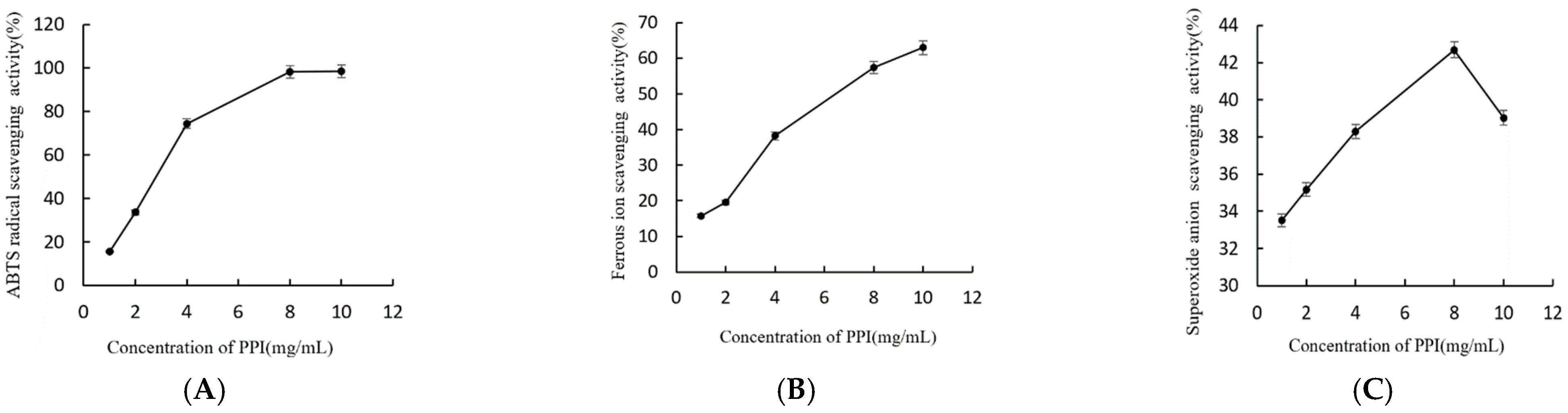
2.4. Antioxidant Activity of PPI
2.5. Effect of PPI on Cell Viability and Cell Morphology
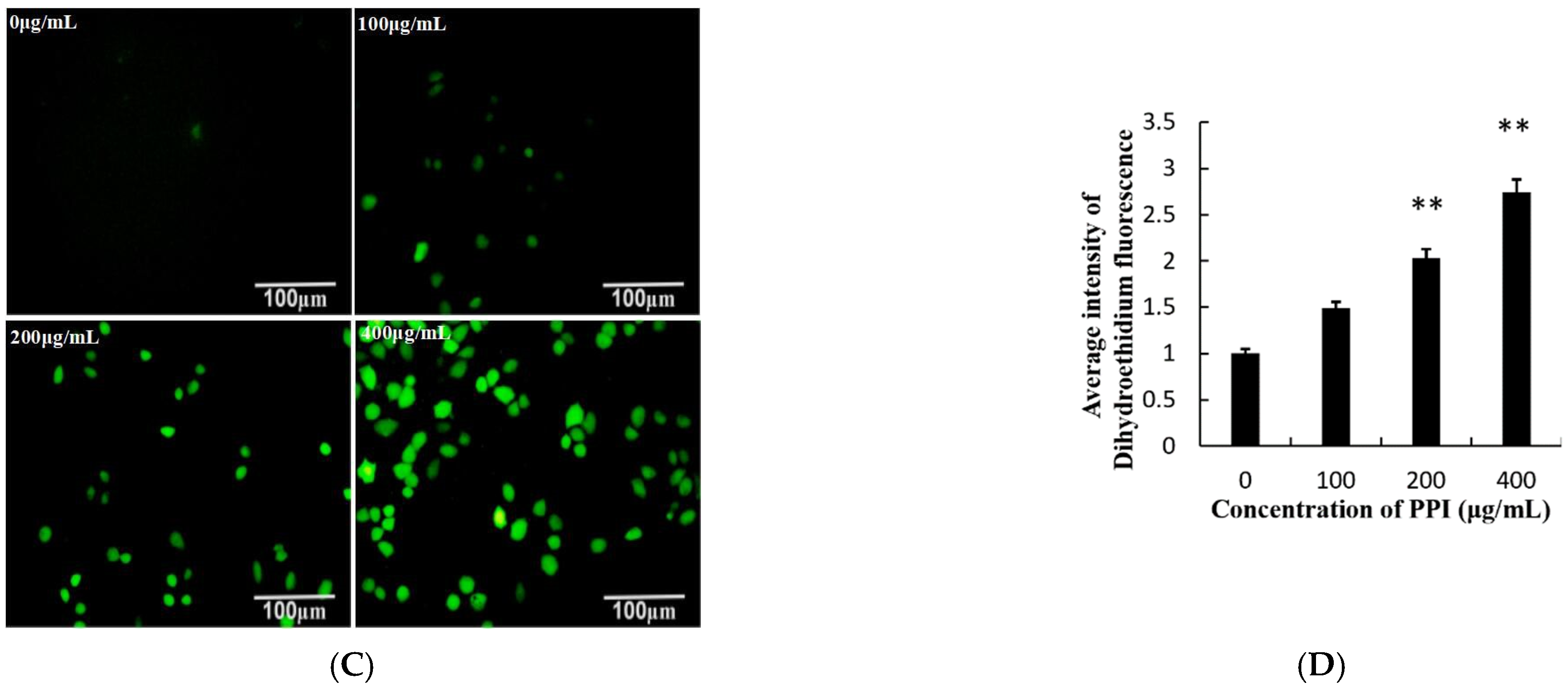
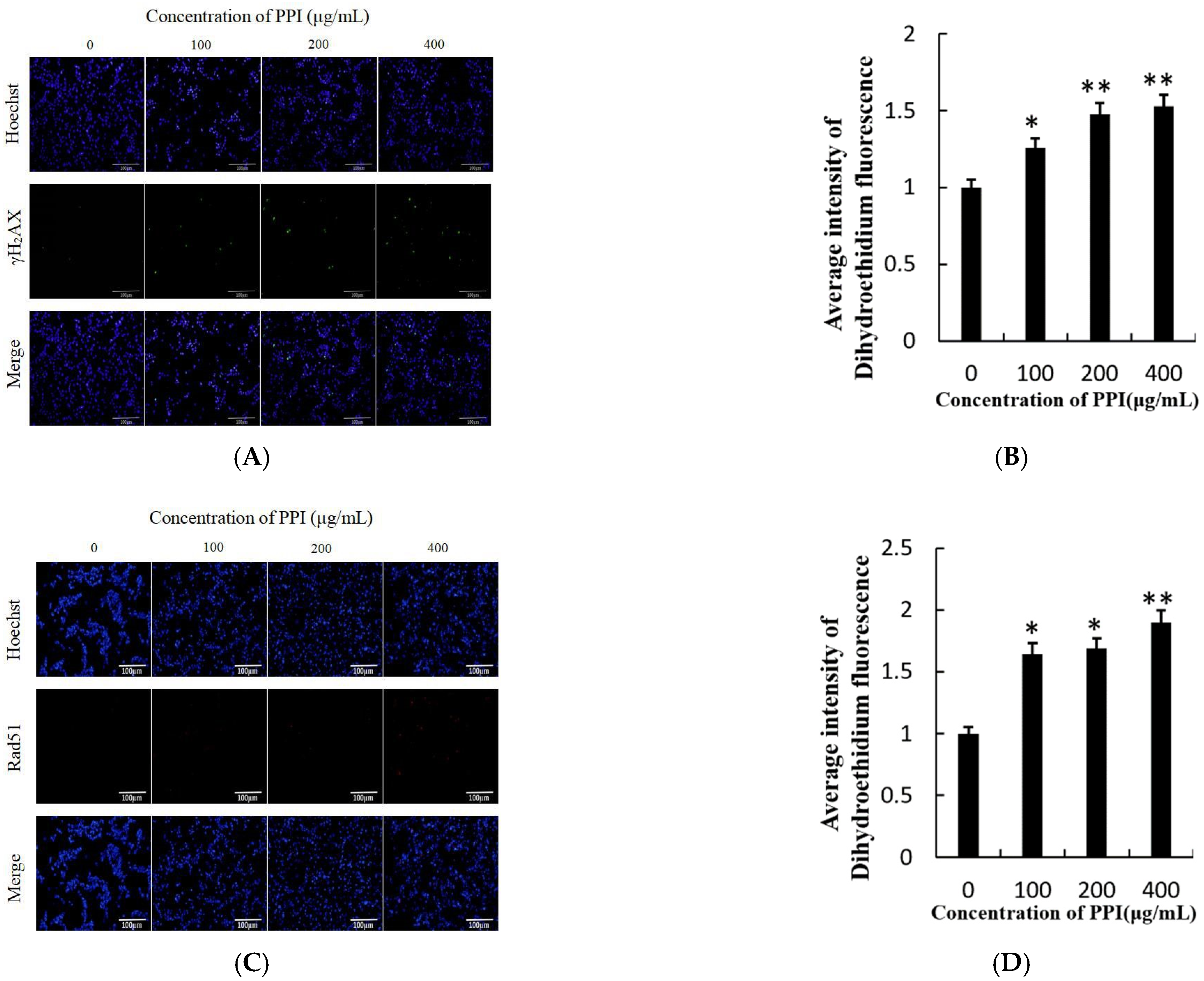
2.6. PPI Increased Levels of Reactive Oxygen Species
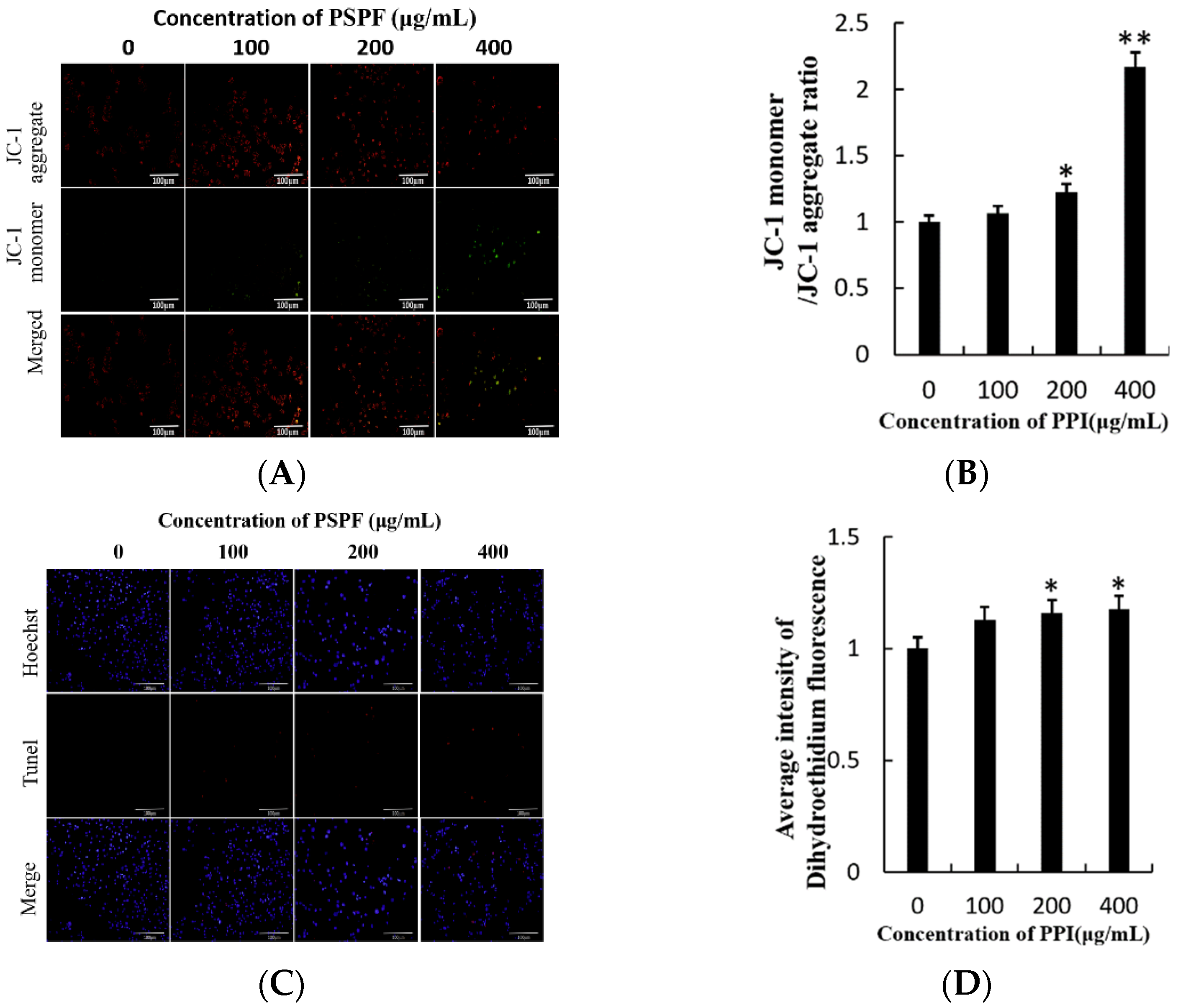
2.7. Loss of Mitochondrial Membrane Potential (∆ψm) and Apoptosis Induction
2.8. PPI Induces Nuclear DNA Breakage
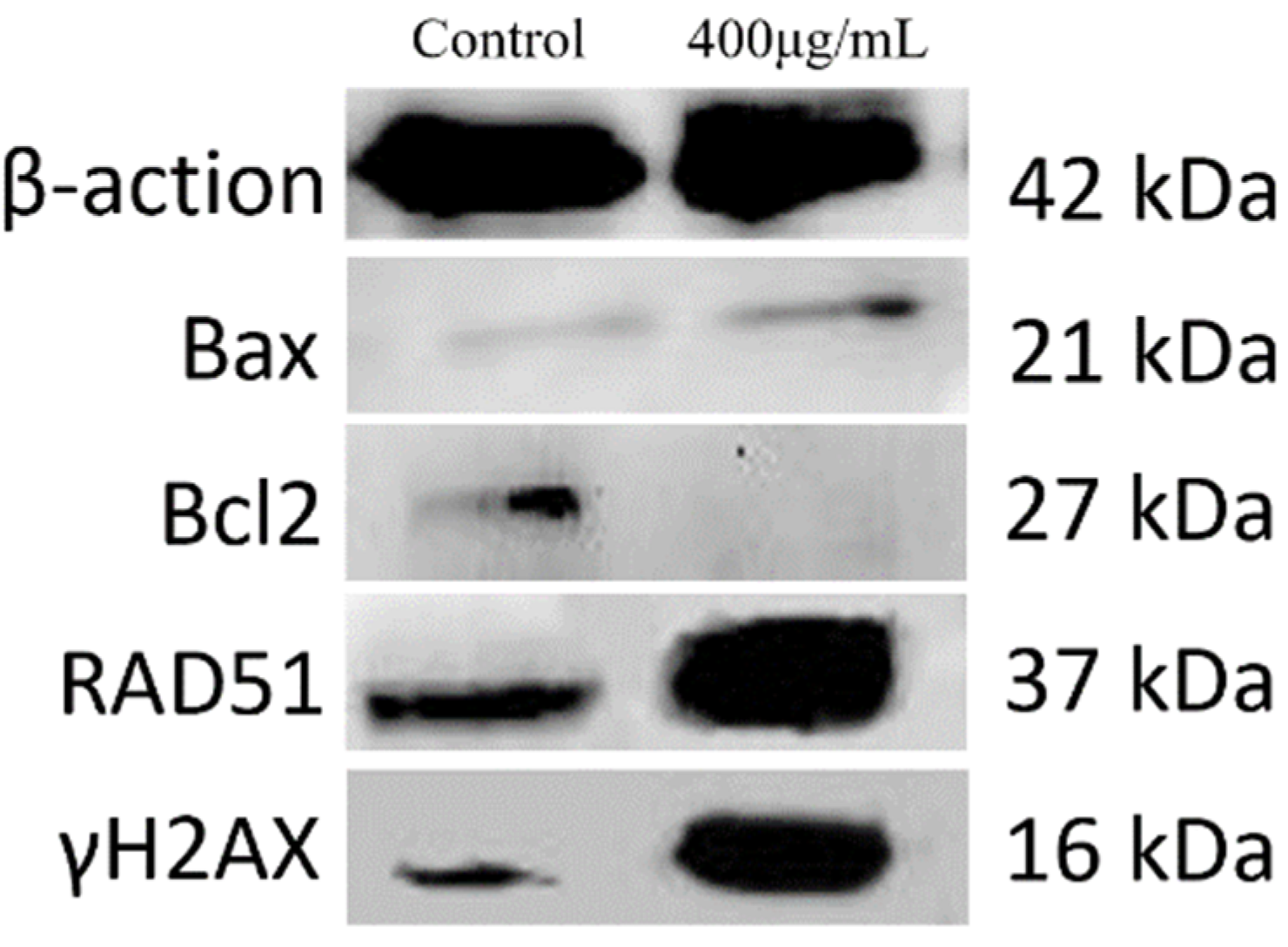
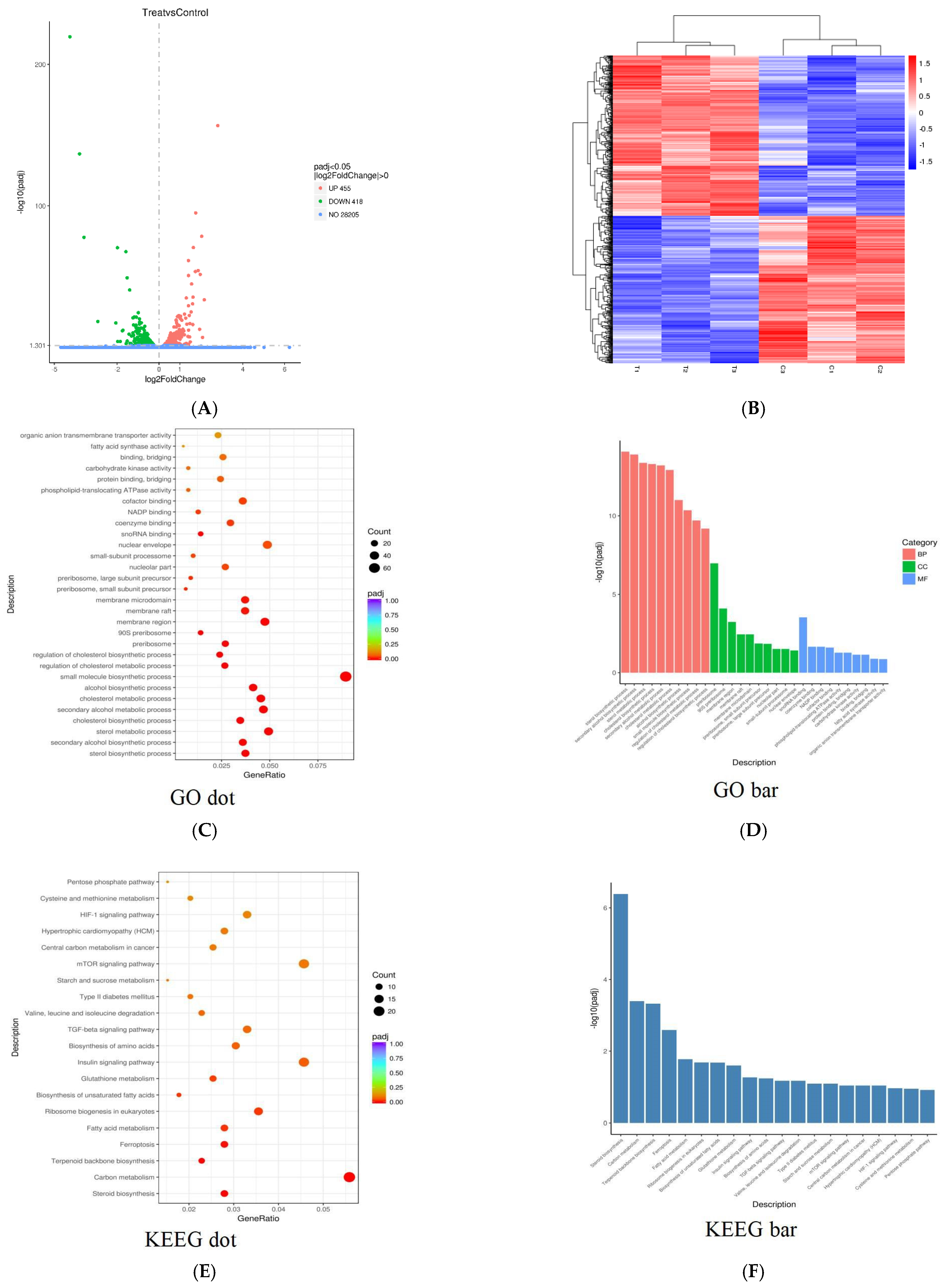
2.9. PPI Affected Gene Expression in Skov3 Cells
3. Conclusions
4. Materials and Methods
4.1. Drugs and Reagents
4.2. Preparation of Polysaccharide
4.3. Single Factor Experimental Data Analysis
4.4. Synthesis of Pyracantha Polysaccharide-Iron(III) Complex (PPI)
4.5. Response Surface Design
4.6. Determination of PPI
4.6.1. Establishment of Standard Curve for Iron
4.6.2. Determination of the Iron Content in PPI
4.6.3. Experimental Design
4.6.4. Antioxidant Activities of PPI
4.7. Characterisation of PPI
4.7.1. UV and FTIR Analysis
4.7.2. SEM Analysis
4.7.3. Circular Dichroism (CD) Analysis
4.7.4. Structural Characteristics of PSPF and PPI
4.7.5. Thermogravimetric Analysis (TGA)
4.7.6. Nuclear Magnetic Resonance (NMR) Analysis
4.7.7. Cytotoxicity of PPI Assay and ROS Assay
4.7.8. Jc-1 Assays, TUNEL Assay and Immunohistochemistry
4.7.9. Western Blot Analysis
4.7.10. Bioinformatics Analysis of the mRNA Expression Profile
4.7.11. Statistical Method
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Wang, X.H.; Bai, H.Q.; Wulangerile. Progress in the Studies of Polysaccharide Metal Complex. J. Inn. Mong. Univ. Natl. 2014, 29, 516–519. [Google Scholar] [CrossRef]
- Danielson, B.G. Structure, chemistry, and pharmacokinetics of intravenous iron agents. J. Am. Soc. Nephrol. 2004, 15, 93–98. [Google Scholar]
- Eichbaum, Q.; Foran, S.; Dzik, S. Is iron gluconate really safer than iron dextran? Blood 2003, 101, 3756. [Google Scholar] [CrossRef]
- Johnson, C.A.; Rosowski, E.; Zimmerman, S.W. A prospective open-label study evaluating the efficacy and adverse reactions of the use of Niferex-150 in ESRD patients receiving EPOGEN. Adv. Perit. Dial. Conf. Perit. Dial. 1992, 8, 444–447. [Google Scholar]
- Floyd, R.A.; Lewis, C.A. Hydroxyl free radical formation from hydrogen peroxide by ferrous iron-nucleotide complexes. Biochemistry 1983, 22, 2645–2649. [Google Scholar] [CrossRef] [PubMed]
- Oates, P.S.; Morgan, E.H. Defective iron uptake by the duodenum of Belgrade rats fed diets of different iron contents. Am. J. Physiol. Liver Physiol. 1996, 270, G826–G832. [Google Scholar] [CrossRef] [PubMed]
- Kudasheva, D.S.; Lai, J.; Ulman, A.; Cowman, M.K. Structure of carbohydrate-bound polynuclear iron oxyhydroxide nanoparticles in parenteral formulations. J. Inorg. Biochem. 2004, 98, 1757–1769. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.W.; Zhang, Q.; Wang, S.H.; Shi, L. Study on the synthesis technique of angelica polysaccharides iron complex by orthogonal test. Med. J. Defending Forces Southwest China 2003, 13, 374–376. [Google Scholar] [CrossRef]
- He, R.X.; Gao, W.H.; Zhu, S.M. Preparation and characters of soluble soybean polysaccharide-iron (Ⅲ) complex. Sci. Technol. Food Ind. 2012, 2, 326–330, doiorg/101155/2019/6416941. [Google Scholar]
- Wu, G.J.; Li, Y.P.; Hu, L.; Chen, Z.Q.; Hong, L.M. Synthesis of Portulace oleracea L. polysaccharide iron complex and its physical-chemical properties. Food Sci. Technol. 2012, 37, 250–252. [Google Scholar] [CrossRef]
- Li, S.J. Study on Chemical and Antioxidation of Fruit of Pyracantha Resources; Huazhong University of Science and Technology: Wuhan, China, 2011. [Google Scholar]
- Yao, Y.-L.; Shu, C.; Feng, G.; Wang, Q.; Yan, Y.-Y.; Yi, Y.; Wang, H.-X.; Zhang, X.-F.; Wang, L.-M. Polysaccharides from Pyracantha fortuneana and its biological activity. Int. J. Biol. Macromol. 2020, 150, 1162–1174. [Google Scholar] [CrossRef]
- Yang, T.; Zhang, S.; Wang, R.; Li, D.; Hu, Y.; Nie, J.; Zhao, X.; Wang, Q.; Chen, Y.; Zheng, Y.; et al. Polysaccharides from Rhizoma Panacis Majoris and its anti-oxidant activity. Int. J. Biol. Macromol. 2016, 86, 756–763. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.-Y.; Zhang, Q.; Shu-Lin, L.; Dong, L.-L.; Liu, S.-L. Optimization of ultrasound extraction of Alisma orientalis polysaccharides by response surface methodology and their antioxidant activities. Carbohydr. Polym. 2015, 119, 101–109. [Google Scholar] [CrossRef]
- Mehmood, T.; Ahmed, A.; Ahmad, A.; Ahmad, M.S.; Sandhu, M.A. Optimization of mixed surfactants-based β-carotene nanoemulsions using response surface methodology: An ultrasonic homogenization approach. Food Chem. 2018, 253, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Celebi, N.; Yildiz, N.; Demir, A.S.; Çalımlı, A.; Calimli, A.; Yıldız, N. Optimization of benzoin synthesis in supercritical carbon dioxide by response surface methodology (RSM). J. Supercrit. Fluids 2008, 47, 227–232. [Google Scholar] [CrossRef]
- Chang, H.Y.; Ho, Y.L.; Sheu, M.J.; Lin, Y.H.; Tseng, M.C.; Wu, S.H.; Huang, G.J.; Chang, Y.S. Antioxidant and free radical scavenging activities of Phellinus merrillii extracts. Bot. Stud. 2007, 48, 407–417. [Google Scholar]
- Pitarresi, G.; Tripodo, G.; Cavallaro, G.; Palumbo, F.S.; Giammona, G. Inulin–iron complexes: A potential treatment of iron deficiency anaemia. Eur. J. Pharm. Biopharm. 2008, 68, 267–276. [Google Scholar] [CrossRef]
- Marshall, P.; Rutherford, D. Physical investigations on colloidal iron-dextran complexes. J. Colloid Interface Sci. 1971, 37, 390–402. [Google Scholar] [CrossRef]
- Park, J.; Chakrabarti, B. Optical characteristics of carboxyl group in relation to the circular dichroic properties and dissociation constants of glycosaminoglycans. Biochim. et Biophys. Acta (BBA)—Gen. Subj. 1978, 544, 667–675. [Google Scholar] [CrossRef]
- Chi, Y.; Li, Y.; Zhang, G.; Gao, Y.; Ye, H.; Gao, J.; Wang, P. Effect of extraction techniques on properties of polysaccharides from Enteromorpha prolifera and their applicability in iron chelation. Carbohydr. Polym. 2018, 181, 616–623. [Google Scholar] [CrossRef]
- Bertini, I.; Luchinat, C.; Parigi, G.; Pierattelli, R. NMR Spectroscopy of Paramagnetic Metalloproteins. ChemBioChem 2005, 6, 1536–1549. [Google Scholar] [CrossRef]
- Kästele, X.; Sturm, C.; Klüfers, P. 13C NMR spectroscopy as a tool for the in situ characterisation of iron-supplementing preparations. Eur. J. Pharm. Biopharm. 2014, 86, 469–477. [Google Scholar] [CrossRef]
- Bai, D.-P.; Zhang, X.-F.; Zhang, G.-L.; Huang, Y.-F.; Gurunathan, S. Zinc oxide nanoparticles induce apoptosis and autophagy in human ovarian cancer cells. Int. J. Nanomed. 2017, 12, 6521–6535. [Google Scholar] [CrossRef]
- Carmody, R.; Cotter, T. Signalling apoptosis: A radical approach. Redox Rep. 2001, 6, 77–90. [Google Scholar] [CrossRef]
- Ishikawa, K.; Takenaga, K.; Akimoto, M.; Koshikawa, N.; Yamaguchi, A.; Imanishi, H.; Nakada, K.; Honma, Y.; Hayashi, J.-I. ROS-Generating Mitochondrial DNA Mutations Can Regulate Tumor Cell Metastasis. Science 2008, 320, 661–664. [Google Scholar] [CrossRef] [PubMed]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Darzynkiewicz, Z.; Galkowski, D.; Zhao, H. Analysis of apoptosis by cytometry using TUNEL assay. Methods 2008, 44, 250–254. [Google Scholar] [CrossRef]
- Redon, C.E.; Dickey, J.S.; Bonner, W.M.; Sedelnikova, O.A. γ-H2AX as a biomarker of DNA damage induced by ionizing radiation in human peripheral blood lymphocytes and artificial skin. Adv. Space Res. 2009, 43, 1171–1178. [Google Scholar] [CrossRef] [PubMed]
- Rothkamm, K.; Horn, S. gamma-H2AX as protein biomarker for radiation exposure. Annali dell’Istituto Superiore di Sanità 2009, 45, 265–271. [Google Scholar]
- He, D.; Yuan, X.; Lin, G.; Chen, W. Determination of iron ion in polysaccharide iron complex pellets. Chin. J. Hosp. Pharm. 2006, 26, 107–108. [Google Scholar] [CrossRef]
- Tang, C.N. Research Progress of Polysaccharide-iron Complex(PIC). Anhui Chem. Ind. 2012, 38, 1–36. [Google Scholar] [CrossRef]
- Saywell, L.G.; Cunningham, B.B. Determination of Iron: Colorimetric o-Phenanthroline Method. Ind. Eng. Chem. Anal. Ed. 1937, 9, 67–69. [Google Scholar] [CrossRef]
- Azargohar, R.; Dalai, A. Production of activated carbon from Luscar char: Experimental and modeling studies. Microporous Mesoporous Mater. 2005, 85, 219–225. [Google Scholar] [CrossRef]
- Adegoroye, A.; Wang, L.; Omotoso, O.; Xu, Z.; Masliyah, J. Characterization of organic-coated solids isolated from different oil sands. Can. J. Chem. Eng. 2010, 88, 462–470. [Google Scholar] [CrossRef]
- Du, X.; Mu, H.; Zhou, S.; Zhang, Y.; Zhu, X. Chemical analysis and antioxidant activity of polysaccharides extracted from Inonotus obliquus sclerotia. Int. J. Biol. Macromol. 2013, 62, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Wang, Y.; Wang, C.; Wang, Z. Antioxidant and immunological activity in vitro of polysaccharides from Gomphidius rutilus mycelium. Carbohydr. Polym. 2013, 92, 2187–2192. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Sheng, Y.; Yu, M.; Li, K.; Ren, G.; Xu, X.; Qu, J. Antioxidant activity of Inonotus obliquus polysaccharide and its amelioration for chronic pancreatitis in mice. Int. J. Biol. Macromol. 2016, 87, 348–356. [Google Scholar] [CrossRef]
- Ben Jeddou, K.; Chaari, F.; Maktouf, S.; Nouri-Ellouz, O.; Helbert, C.B.; Ghorbel, R.E. Structural, functional, and antioxidant properties of water-soluble polysaccharides from potatoes peels. Food Chem. 2016, 205, 97–105. [Google Scholar] [CrossRef]
- Paździoch-Czochra, M.; Wideńska, A. Spectrofluorimetric determination of hydrogen peroxide scavenging activity. Anal. Chim. Acta 2002, 452, 177–184. [Google Scholar] [CrossRef]
- Gurunathan, S.; Zhang, X.-F.; Choi, Y.-J.; Han, J.W.; Kim, E.; Park, J.H.; Kim, J.-H. Differential nanoreprotoxicity of silver nanoparticles in male somatic cells and spermatogonial stem cells. Int. J. Nanomed. 2015, 10, 1335–1357. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liu, J.-C.; Zhao, Y.; Lai, F.-N.; Yang, F.; Ge, W.; Dou, C.-L.; Shen, W.; Zhang, X.-F.; Chen, H. Impact of diethylhexyl phthalate on gene expression and development of mammary glands of pregnant mouse. Histochem. Cell Biol. 2015, 144, 389–402. [Google Scholar] [CrossRef] [PubMed]
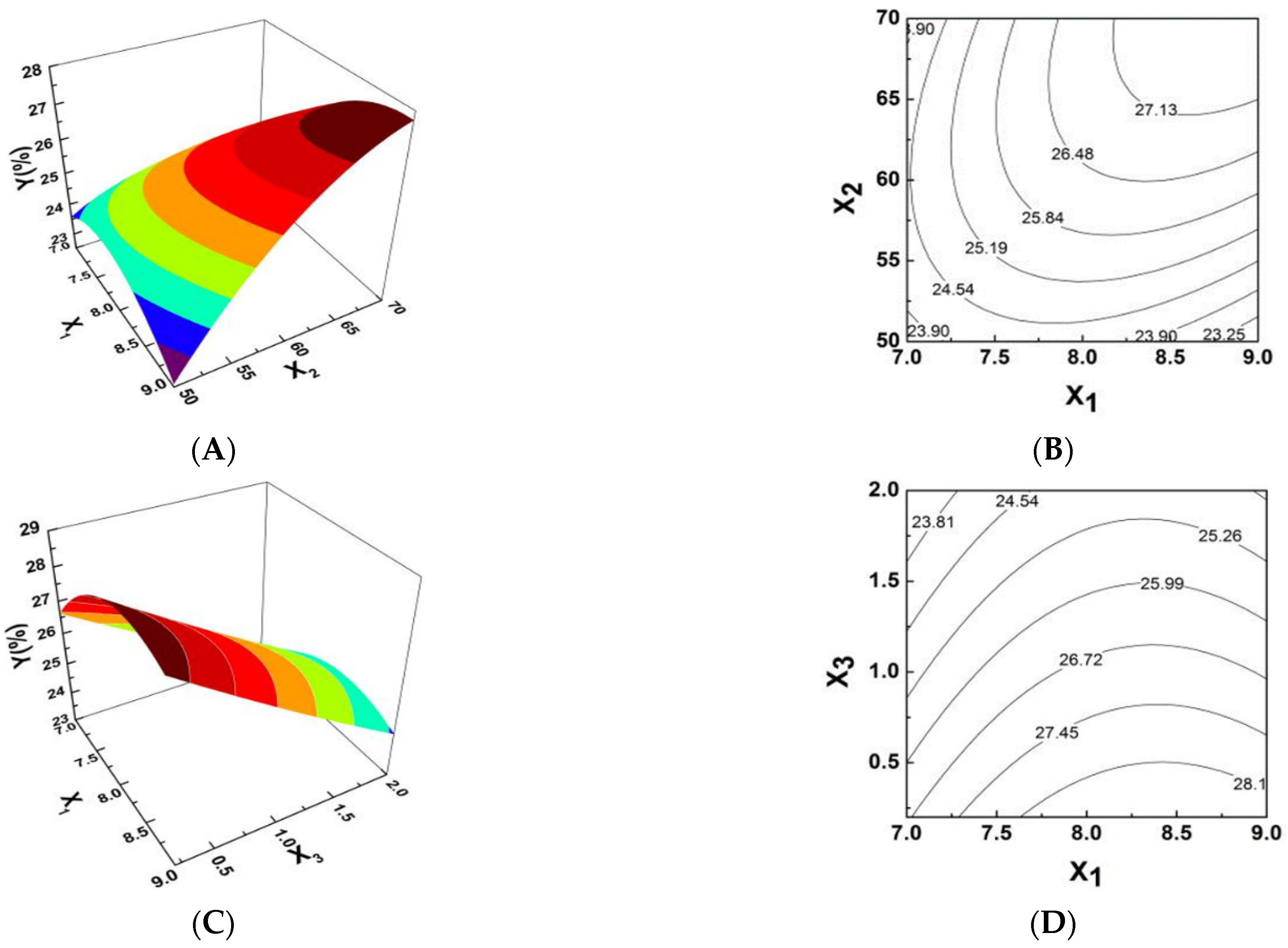
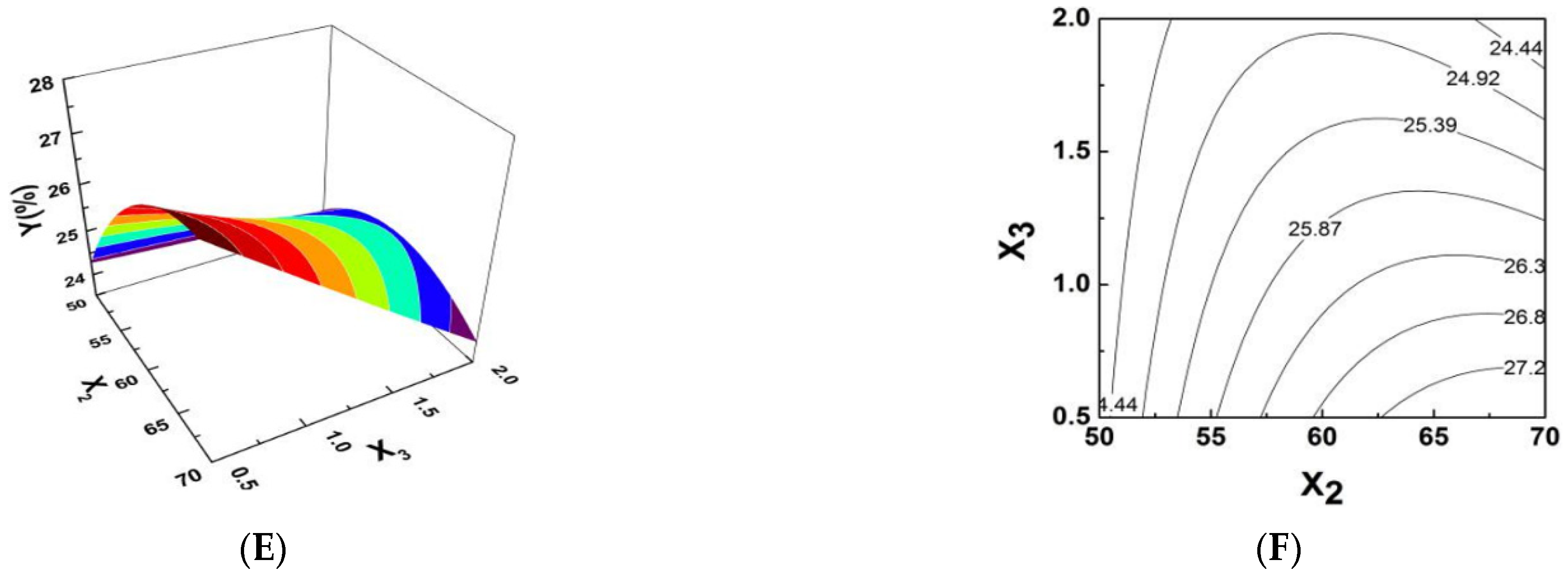



| Source | Sum of Squares | Degree of Freedom | Mean Square | F Value | p Value |
|---|---|---|---|---|---|
| Model | 60.38 | 9 | 6.710 | 89.09 | <0.0001 *** |
| X1 | 4.96 | 1 | 4.961 | 65.88 | <0.0001 *** |
| X2 | 13.65 | 1 | 13.650 | 181.26 | <0.0001 *** |
| X3 | 19.75 | 1 | 19.750 | 262.25 | <0.0001 *** |
| X1 × X2 | 6.55 | 1 | 6.550 | 87.02 | <0.0001 *** |
| X1 × X3 | 0.06 | 1 | 0.058 | 0.76 | 0.4108 |
| X2 × X3 | 6.79 | 1 | 6.790 | 90.11 | <0.0001 *** |
| X12 | 4.98 | 1 | 4.980 | 66.15 | <0.0001 *** |
| X22 | 3.19 | 1 | 3.190 | 42.34 | <0.0003 *** |
| X32 | 0.02 | 1 | 0.015 | 0.20 | 0.6685 |
| Residual | 0.53 | 7 | 0.075 | ||
| Lack of Fit | 0.27 | 3 | 0.091 | 1.43 | 0.3586 |
| Pure Error | 0.25 | 4 | 0.064 | ||
| Cor Total | 60.91 | 16 | |||
| R2 = 0.99 | R2Adj = 0.98 | R2pred = 0.92 | Adeq Precisior = 33.86 | ||
| Run | Level | |||
|---|---|---|---|---|
| X1 | X2 | X3 | Fe (%) | |
| 1 | 0 | 0 | 0 | 26.27 |
| 2 | 1 | 1 | 0 | 27.90 |
| 3 | 0 | 1 | 1 | 23.88 |
| 4 | 0 | 1 | −1 | 29.86 |
| 5 | 1 | 0 | −1 | 27.55 |
| 6 | 1 | 0 | 1 | 24.40 |
| 7 | 0 | 0 | 0 | 26.80 |
| 8 | −1 | 0 | 1 | 23.35 |
| 9 | 0 | 0 | 0 | 26.20 |
| 10 | 0 | −1 | 1 | 23.84 |
| 11 | 1 | −1 | 0 | 22.76 |
| 12 | 0 | −1 | −1 | 24.61 |
| 13 | 0 | 0 | 0 | 26.2 |
| 14 | −1 | 1 | 0 | 23.48 |
| 15 | −1 | −1 | 0 | 23.46 |
| 16 | 0 | 0 | 0 | 26.32 |
| 17 | −1 | 0 | −1 | 26.02 |
| Variable | Symbols | Coded Levels | ||
|---|---|---|---|---|
| Coded | −1 | 0 | 1 | |
| pH | X1 | 7 | 8 | 9 |
| Reaction temperature | X2 | 50 | 60 | 70 |
| Sodium Citrate tribasic/Polysaccharide ratio | X3 | 0.5 | 1.25 | 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, W.-F.; Ma, H.-H.; Yuan, S.; Zhang, X.-F. Production of Pyracantha Polysaccharide-Iron(III) Complex and Its Biologic Activity. Molecules 2021, 26, 1949. https://doi.org/10.3390/molecules26071949
Li W-F, Ma H-H, Yuan S, Zhang X-F. Production of Pyracantha Polysaccharide-Iron(III) Complex and Its Biologic Activity. Molecules. 2021; 26(7):1949. https://doi.org/10.3390/molecules26071949
Chicago/Turabian StyleLi, Wan-Fen, Hao-Hai Ma, Shuai Yuan, and Xi-Feng Zhang. 2021. "Production of Pyracantha Polysaccharide-Iron(III) Complex and Its Biologic Activity" Molecules 26, no. 7: 1949. https://doi.org/10.3390/molecules26071949
APA StyleLi, W.-F., Ma, H.-H., Yuan, S., & Zhang, X.-F. (2021). Production of Pyracantha Polysaccharide-Iron(III) Complex and Its Biologic Activity. Molecules, 26(7), 1949. https://doi.org/10.3390/molecules26071949





