Extraction and Analysis of Six Effective Components in Glycyrrhiza uralensis Fisch by Deep Eutectic Solvents (DES) Combined with Quantitative Analysis of Multi-Components by Single Marker (QAMS) Method
Abstract
1. Introduction
2. Results and Discussion
2.1. The fm/k of Five Components
2.2. Screening of Deep Eutectic Solvents
2.3. Effects of Single Factor of DES
2.4. Influence of Different Extraction Methods on Microstructure
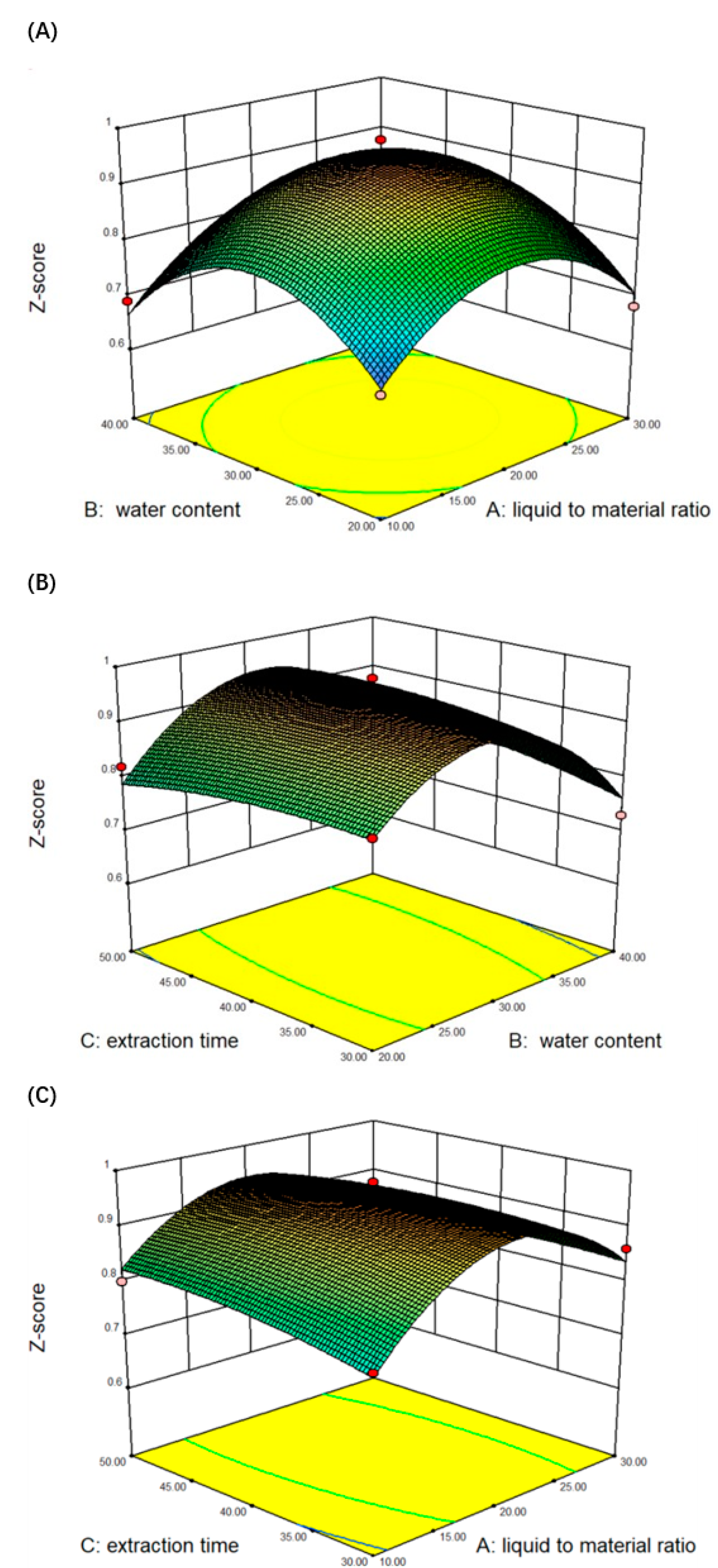
2.5. Optimization of the Extraction Conditions
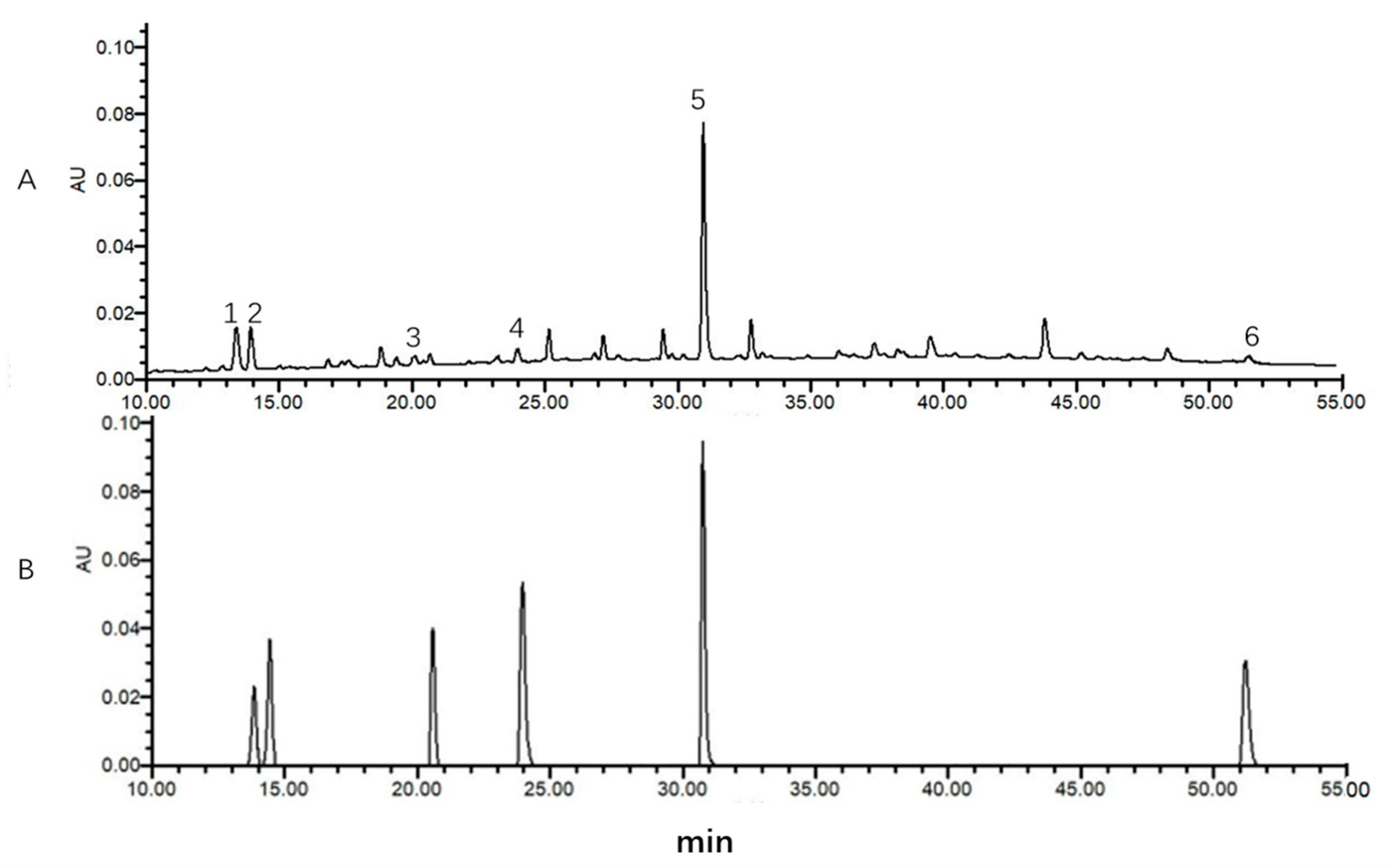
2.6. Method Validation
2.6.1. Linearity, Limit of Detection (LOD), and Limit of Quantification (LOQ)
2.6.2. Precision, Repeatability, Stability, and Recovery
2.7. Verification of Predictive Model
3. Materials and Methods
3.1. Plant Materials
3.2. Chemicals and Reagents
3.3. Preparation of DESs
3.4. Extraction Procedure
3.4.1. Reference Extraction Methods
3.4.2. Preparation of Standard Solution
3.5. Establishment of QAMS Method
3.5.1. Quantitative Analysis of HPLC
3.5.2. Relative Correction Factor (fm/k) Calculation
3.5.3. Comprehensive Evaluation Index
3.6. Experimental Design
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Hayashi, H.; Sudo, H. Economic importance of licorice. Plant Biotechnol. 2009, 26, 101–104. [Google Scholar] [CrossRef]
- Han, C.K.; Wei, B.L.; Wen, F.C. The effect of medicinal plants used in Chinese folk medicine on RANTES secretion by virus-infected human epithelial cells. J. Ethnopharmacol. 2006, 107, 205–210. [Google Scholar]
- Patiguli, M.; Guo, X.F. Research progress in the development and utilization of licorice extracts. Mod. Agric. Sci. Technol. 2019, 3, 211–215. [Google Scholar]
- Wang, Q.; Shen, J.; Yan, Z. Dietary Glycyrrhiza uralensis extracts supplementation elevated growth performance, immune responses and disease resistance against Flavobacterium columnare in yellow catfish (Pelteobagrus fulvidraco). Fish Shellfish Immunol. 2020, 97, 153–164. [Google Scholar] [CrossRef]
- Kim, Y.H.; Shin, E.K.; Kim, D.H. Antiangiogenic effect of licochalcone A. Biochem. Pharmacol. 2010, 80, 1152–1159. [Google Scholar] [CrossRef]
- Sun, Y.; Wumaier, A. Research progress on extraction methods and pharmacological effects of licorice flavonoids. Xinjiang J. Tradit. Chin. Med. 2009, 27, 72–75. [Google Scholar]
- Gao, X.Y.; Wang, W.Q.; Wei, S.L. Review of pharmacological effects of glycyrrhiza radix and its bioactive compounds. China J. Chin. Mater. Med. 2009, 34, 2695–2700. [Google Scholar]
- Wang, A.Z.; Ma, X.C. Research progress in the clinical application of Zhigancao Decoction. World Tradit. Chin. Med. 2020, 15, 1662–1665. [Google Scholar]
- Wei, J.; Yan, Y.L.; Zeng, S.S. Clinical application and analysis of compound glycyrrhizin injection. China Pharm. 2019, 28, 79–81. [Google Scholar]
- Li, F.Z. Examples of clinical application of Gancao Xiexin Decoction in Synopsis of the Golden Chamber. J. Chin. Med. 2019, 34, 2576–2579. [Google Scholar]
- Zhao, Y.S.; Hou, X.Y.; Gao, Z.H. Tracing the source of prescriptions for the treatment of dampness at the initial stage of novel coronavirus pneumonia. Chin. J. Tradit. Chin. Med. 2020, 38, 4–7. [Google Scholar]
- Jin, J.; Chen, X. On the enlightenment of “Lung Wind” in Neijing on the treatment of new coronavirus pneumonia. Shanghai J. Tradit. Chin. Med. 2020, 54, 6–10. [Google Scholar]
- Zhang, F.X.; Song, J.X.; Liu, X.D. Antioxidant and immunological activity of glycyrrhiza flavonoids. China Vet. News 2019, 39, 1180–1183. [Google Scholar]
- Wang, J. Preparation, Anticancer Activity and Mechanism of Licorice Chalcone A and B. Ph.D.Thesis, Hefei University of Technology, Hefei, China, April 2019. [Google Scholar]
- Zhang, M.F.; Shen, Y.Q. Research progress on cardioprotective effects of glycyrrhizic acid and licorice flavonoids. Mod. Med. Clin. 2012, 27, 429. [Google Scholar]
- Bachmetov, L.; Galtanamy, M.; Shapira, A. Suppression of hepaatitis C virus by the flavonoid quercetin is mediated by inhibition of NS3 protease activity. J. Viral. Hepat. 2012, 19, 81. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, F.; Zhao, J. Network pharmacology study of saponins in licorice. J. Ningxia Med. Univ. 2019, 41, 658–665. [Google Scholar]
- Wang, X.; Qin, Y.; Wang, D.J. Application progress of one-test-multiple-evaluation method in quality control of traditional Chinese medicine. Chin. Pat. Med. 2016, 38, 395–402. [Google Scholar]
- Feng, Y.M.; Li, Q.; Yang, L. Simultaneous determination of osthol, columbianadin, and isoimperatorin in Angelicae pubescentis Radix by high-performance liquid chromatography (HPLC) using a quantitative analysis of multi-components by single marker (QAMS) calibration method. Instrum. Sci. Technol. 2020, 48, 550–560. [Google Scholar] [CrossRef]
- Wang, Y.C.; Yu, Y. Quality evaluation of powdered poppy capsule extractive by systematic quantified fingerprint method combined with quantitative analysis of multi-components by single marker method. J. Pharm. Biomed. Anal. 2020, 185, 113247. [Google Scholar] [CrossRef]
- Li, Z.P.; Wang, J.L.; Zhang, Y.L. Simultaneous determination of 9 ingredients in Jiuwei Chenxiang Capsules with one test and multiple evaluations. Chin. Pat. Med. 2020, 42, 1129–1134. [Google Scholar]
- He, C.X. Determination of the content of phenylpropanoid glycosides and nucleosides in Shenqi Fuzheng injection with one test and multiple evaluations. China Pharm. 2020, 29, 52–57. [Google Scholar]
- Wang, Z.Y. Determination of 3 phenolic acids in dried peach medicinal materials by one measurement and multiple evaluation method. Chin. Pharm. Stand. 2020, 21, 371–375. [Google Scholar]
- Abbota, P.; Boothby, D.; Capper, G. Deep eutectic solvents formed between choline chloride and carboxylic acids: Versatile alternatives to ionic liquids. J. Am. Chem. Soc. 2004, 126, 9142–9147. [Google Scholar] [CrossRef]
- Wang, L.; Xu, L.L.; Chen, X.; Jing, H. Response surface method to optimize the eutectic solvent extraction of mulberry leaf DNJ. Food Ind. 2020, 41, 14–17. [Google Scholar]
- Yu, J.O.; Park, S.; Jung, D. Effect of hydrogen bond donor on the choline chloride-based deep eutectic solvent-mediated extraction of lignin from pine wood. Int. J. Biol. Macromol. 2020, 165, 187–197. [Google Scholar]
- Yan, L.; Song, R.P.; Xu, Z. Non-productive celluase binding onto deep eutectic solvent (DES) extracted lignin from willow and corn stover with inhibitory effects on enzymatic hydrolysis of cellulose. Carbohydr. Polym. 2020, 250, 116956. [Google Scholar]
- Li, J.; Wu, Y.F.; Wang, X.S.; Wang, Y. Study on the eutectic solvent extraction of flavonoids from Fructus Aurantii. Appl. Chem. Ind. 2020, 15, 1–7. [Google Scholar]
- Qi, C.; Peng, X.; Yao, X.H. Deep eutectic solvent-based microwave-assisted extraction of genistin, genistein and apigenin from pigeon pea roots. Sep. Purif. Technol. 2015, 150, 63–72. [Google Scholar]
- Hui, W.; Ma, X.D.; Cheng, Q.D. Deep Eutectic Solvent-Based Microwave-Assisted Extraction of Baicalin from Scutellaria baicalensis Georgi. J. Chem. 2018, 10, 1155. [Google Scholar]
- Wei, Z.; Qi, X.; Li, T. Application of natural deep eutectic solvents for extraction and determination of phenolics in Cajanus cajan leaves by ultraperformance liquid chromatography. Sep. Purif. Technol. 2015, 149, 237–244. [Google Scholar] [CrossRef]
- Liu, Z.W.; Que, Z.L.; Ye, Z.W. Chemical constituents from the aerial parts of Abrus mollis. China J. Nat. Med. 2008, 6, 415–417. [Google Scholar] [CrossRef]
- Lai, H.F.; Deng, Q.F. Optimization of extraction technology for total triterpene acids from Abri Herba by semi-bionic method. Chin. J. Exp. Tradit. Med. From. 2013, 19, 46–48. [Google Scholar]
- Dai, Y.T.; Witkamp, G.J.; Verpoorte, R. Tailoring properties of natural deep eutectic solvents with water to facilitate their applications. Food Chem. 2015, 187, 14–19. [Google Scholar] [CrossRef]
- Wang, H. Study on the Application of Eutectic Solvents in the Extraction and Preparation of Effective Components of Scutellaria Baicalensis Georgi. Ph.D. Thesis, Shanxi University, Taiyuan, China, 2019. [Google Scholar]
- Wang, F.; Liu, W.; Chen, F.S. Ultrasound-assisted eutectic solvent extraction of resveratrol from peanut red coat. Food Oils 2020, 33, 90–93. [Google Scholar]
- Chinese Pharmacopoeia Commission. Chinese Pharmacopoeia; China Medical Science Press: Beijing, China, 2015. [Google Scholar]
- Duan, X.J.; Liu, P.; Ye, H.; Wang, K. Multiple indicators, one test, multiple evaluations-response surface method to optimize the best processing technology of honey-dried licorice. Chin. Herb. Med. 2020, 51, 364–371. [Google Scholar]


| Injection Volume/μL | fisoliquiritin apioside/glycyrrhizic acid | fliquiritin/glycyrrhizic acid | fisoliquiritin/glycyrrhizic acid | fliquiritigenin/glycyrrhizic acid | fglycyrrhetinic acid/glycyrrhizic acid |
|---|---|---|---|---|---|
| 4 | 1.57 | 1.34 | 0.88 | 0.73 | 0.78 |
| 8 | 1.60 | 1.33 | 0.87 | 0.71 | 0.80 |
| 10 | 1.59 | 1.35 | 0.86 | 0.70 | 0.80 |
| 16 | 1.61 | 1.34 | 0.87 | 0.71 | 0.77 |
| 20 | 1.58 | 1.33 | 0.87 | 0.71 | 0.79 |
| average | 1.59 | 1.34 | 0.87 | 0.71 | 0.79 |
| RSD | 0.86% | 0.51% | 1.05% | 1.66% | 1.64% |
| Single Factor | Factor Level | Extraction Rate/% | Z-Score | |||||
|---|---|---|---|---|---|---|---|---|
| Isoliquiritin Apioside | Liquiritin | Isoliquiritin | Liquiritigenin | Glycyrrhizic Acid | Glycyrrhetinic Acid | |||
| Water content/% | 10 | 0.59 ± 0.0047 | 0.39 ± 0.0037 | 0.08 ± 0.0041 | 0.05 ± 0.0045 | 1.62 ± 0.0033 | 0.07 ± 0.0045 | 0.84 |
| 20 | 0.59 ± 0.0065 | 0.41 ± 0.0042 | 0.08 ± 0.0044 | 0.04 ± 0.0044 | 1.7 ± 0.0040 | 0.07 ± 0.0042 | 0.83 | |
| 30 | 0.66 ± 0.0057 | 0.44 ± 0.0027 | 0.1 ± 0.0042 | 0.05 ± 0.0034 | 1.83 ± 0.0043 | 0.07 ± 0.0047 | 0.95 | |
| 40 | 0.61 ± 0.0036 | 0.42 ± 0.0045 | 0.12 ± 0.0045 | 0.03 ± 0.0027 | 1.72 ± 0.0034 | 0.05 ± 0.0040 | 0.83 | |
| 50 | 0.66 ± 0.0028 | 0.43 ± 0.0041 | 0.14 ± 0.0040 | 0.03 ± 0.0047 | 1.73 ± 0.0034 | 0.03 ± 0.0037 | 0.82 | |
| Liquid to Material ratio/mL·g−1 | 10 | 0.59 ± 0.0032 | 0.4 ± 0.0033 | 0.08 ± 0.0034 | 0.03 ± 0.0044 | 1.67 ± 0.0050 | 0.12 ± 0.0033 | 0.83 |
| 20 | 0.64 ± 0.0041 | 0.44 ± 0.0047 | 0.1 ± 0.0045 | 0.05 ± 0.0047 | 1.83 ± 0.0034 | 0.07 ± 0.0043 | 0.91 | |
| 30 | 0.59 ± 0.0023 | 0.4 ± 0.0033 | 0.11 ± 0.0029 | 0.03 ± 0.0029 | 1.69 ± 0.0035 | 0.07 ± 0.0040 | 0.8 | |
| 40 | 0.62 ± 0.0034 | 0.42 ± 0.0025 | 0.09 ± 0.0050 | 0.05 ± 0.0047 | 1.71 ± 0.0029 | 0.06 ± 0.0035 | 0.83 | |
| 50 | 0.67 ± 0.0043 | 0.44 ± 0.0050 | 0.1 ± 0.0047 | 0.05 ± 0.0050 | 1.8 ± 0.0040 | 0.07 ± 0.0036 | 0.9 | |
| Extraction time/min | 20 | 0.62 ± 0.0033 | 0.42 ± 0.0045 | 0.09 ± 0.0037 | 0.03 ± 0.0042 | 1.74 ± 0.0047 | 0.06 ± 0.0037 | 0.79 |
| 30 | 0.62 ± 0.0042 | 0.44 ± 0.0034 | 0.1 ± 0.0050 | 0.05 ± 0.0043 | 1.87 ± 0.0045 | 0.07 ± 0.0043 | 0.9 | |
| 40 | 0.7 ± 0.0034 | 0.47 ± 0.0033 | 0.13 ± 0.0039 | 0.03 ± 0.0048 | 1.89 ± 0.0038 | 0.07 ± 0.0042 | 0.93 | |
| 50 | 0.6 ± 0.0035 | 0.42 ± 0.0044 | 0.11 ± 0.0038 | 0.03 ± 0.0036 | 1.74 ± 0.0039 | 0.06 ± 0.0043 | 0.82 | |
| 60 | 0.62 ± 0.0045 | 0.43 ± 0.0023 | 0.11 ± 0.0037 | 0.03 ± 0.0039 | 1.75 ± 0.0034 | 0.07 ± 0.0040 | 0.83 | |
| Extraction temperature/°C | 30 | 0.63 ± 0.0044 | 0.42 ± 0.0045 | 0.11 ± 0.0045 | 0.03 ± 0.0036 | 1.78 ± 0.0039 | 0.04 ± 0.0045 | 0.77 |
| 40 | 0.6 ± 0.0035 | 0.41 ± 0.0047 | 0.06 ± 0.0047 | 0.02 ± 0.0042 | 1.72 ± 0.0043 | 0.04 ± 0.0029 | 0.64 | |
| 50 | 0.67 ± 0.0043 | 0.43 ± 0.0042 | 0.13 ± 0.0049 | 0.03 ± 0.0037 | 1.92 ± 0.0032 | 0.12 ± 0.0034 | 0.93 | |
| 60 | 0.67 ± 0.0032 | 0.44 ± 0.0033 | 0.1 ± 0.0038 | 0.05 ± 0.0043 | 1.83 ± 0.0037 | 0.07 ± 0.0050 | 0.89 | |
| 70 | 0.63 ± 0.0033 | 0.44 ± 0.0043 | 0.1 ± 0.0042 | 0.05 ± 0.0044 | 1.83 ± 0.0040 | 0.07 ± 0.0043 | 0.88 | |
| 80 | 0.63 ± 0.0021 | 0.43 ± 0.0045 | 0.13 ± 0.0040 | 0.02 ± 0.0018 | 1.85 ± 0.0036 | 0.08 ± 0.0047 | 0.84 | |
| Ultrasonic power/W | 240 | 0.63 ± 0.0023 | 0.46 ± 0.0044 | 0.12 ± 0.0039 | 0.02 ± 0.0022 | 1.84 ± 0.0045 | 0.07 ± 0.0040 | 0.87 |
| 300 | 0.63 ± 0.0041 | 0.44 ± 0.0047 | 0.1 ± 0.0040 | 0.05 ± 0.0037 | 1.83 ± 0.0042 | 0.07 ± 0.0042 | 0.94 | |
| 360 | 0.7 ± 0.0042 | 0.47 ± 0.0035 | 0.08 ± 0.0036 | 0.01 ± 0.0046 | 1.93 ± 0.0042 | 0.06 ± 0.0036 | 0.79 | |
| 420 | 0.59 ± 0.0022 | 0.39 ± 0.0039 | 0.12 ± 0.0034 | 0.02 ± 0.0047 | 1.63 ± 0.0034 | 0.06 ± 0.0047 | 0.8 | |
| 480 | 0.62 ± 0.0043 | 0.38 ± 0.0040 | 0.12 ± 0.0037 | 0.02 ± 0.0034 | 1.63 ± 0.0047 | 0.04 ± 0.0041 | 0.76 | |
| Independent Factor | Levels | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| A mL/g (liquid to material ratio) | 10 | 20 | 30 |
| B% (water content) | 20 | 30 | 40 |
| C min (extraction time) | 30 | 40 | 50 |
| Serial Number | A/mL·g−1 | B/% | C/min | Z |
|---|---|---|---|---|
| 1 | 10 | 20 | 40 | 0.68 |
| 2 | 30 | 20 | 40 | 0.68 |
| 3 | 10 | 40 | 40 | 0.69 |
| 4 | 30 | 40 | 40 | 0.71 |
| 5 | 10 | 30 | 30 | 0.78 |
| 6 | 30 | 30 | 30 | 0.86 |
| 7 | 10 | 30 | 50 | 0.80 |
| 8 | 30 | 30 | 50 | 0.80 |
| 9 | 20 | 20 | 30 | 0.83 |
| 10 | 20 | 40 | 30 | 0.73 |
| 11 | 20 | 20 | 50 | 0.82 |
| 12 | 20 | 40 | 50 | 0.82 |
| 13 | 20 | 30 | 40 | 0.95 |
| 14 | 20 | 30 | 40 | 0.98 |
| 15 | 20 | 30 | 40 | 0.94 |
| 16 | 20 | 30 | 40 | 0.94 |
| 17 | 20 | 30 | 40 | 0.95 |
| Source | Sum of | Mean | F | p-Value | ||
|---|---|---|---|---|---|---|
| Squares | df | Square | Value | Prob > F | ||
| Model | 0.16 | 9 | 0.018 | 21.91 | 0.0003 | significant |
| A-Liquid to material ratio | 0.00125 | 1 | 0.00125 | 1.51 | 0.2583 | |
| B-Water content | 0.00045 | 1 | 0.00045 | 0.54 | 0.4844 | |
| C-Extraction time | 0.0002 | 1 | 0.0002 | 0.24 | 0.6377 | |
| AB | 0.0001 | 1 | 0.0001 | 0.12 | 0.7381 | |
| AC | 0.0016 | 1 | 0.0016 | 1.94 | 0.2065 | |
| BC | 0.0025 | 1 | 0.0025 | 3.03 | 0.1254 | |
| A2 | 0.067 | 1 | 0.067 | 80.96 | < 0.0001 | |
| B2 | 0.078 | 1 | 0.078 | 94.32 | < 0.0001 | |
| C2 | 0.001078 | 1 | 0.001078 | 1.31 | 0.2908 | |
| Residual | 0.00578 | 7 | 0.0008257 | |||
| Lack of Fit | 0.0047 | 3 | 0.001567 | 5.8 | 0.0612 | not significant |
| Pure Error | 0.00108 | 4 | 0.00027 | |||
| Cor Total | 0.17 | 16 | ||||
| R2 | 0.9657 | |||||
| Adj R2 | 0.9216 |
| Analyte | Calibration Curve | Linearity Range (μg/μL) | R2 | LOD | LOQ |
|---|---|---|---|---|---|
| isoliquiritin apioside | y = 5000,000x − 1202.6 | 0.0172–0.82 | 0.9996 | 0.00064 | 0.00194 |
| liquiritin | y = 6000,000x − 1945.8 | 0.018–0.09 | 0.9997 | 0.00205 | 0.00621 |
| Isoliquiritin | y = 9000,000x − 1581.9 | 0.0104–0.052 | 1 | 0.00029 | 0.00088 |
| liquiritigenin | y = 10000,000x − 5257.6 | 0.014–0.07 | 1 | 0.00042 | 0.00128 |
| glycyrrhizic acid | y = 80000,00x − 2242.8 | 0.0224–0.112 | 0.9999 | 0.00189 | 0.00573 |
| glycyrrhetinic acid | y = 1000,000x − 3551.6 | 0.0088–0.044 | 0.9996 | 0.00225 | 0.00683 |
| Serial Number | Extraction Rate/% | Z-Score | |||||
|---|---|---|---|---|---|---|---|
| Isoliquiritin Apioside | Liquiritin | Isoliquiritin | Liquiritigenin | Glycyrrhizic Acid | Glycyrrhetinic Acid | ||
| 1 | 0.66 | 0.46 | 0.12 | 0.11 | 1.84 | 0.021 | 0.92 |
| 2 | 0.65 | 0.46 | 0.12 | 0.11 | 1.81 | 0.021 | 0.92 |
| 3 | 0.66 | 0.45 | 0.12 | 0.11 | 1.87 | 0.021 | 0.93 |
| DESs Combination | HBAs | HBDs | Mol Ratio |
|---|---|---|---|
| DESs-1 | ChCl | Glycol | 1:2 |
| DESs-2 | ChCl | Glycol | 1:3 |
| DESs-3 | ChCl | Glycol | 1:4 |
| DESs-4 | ChCl | Glycol | 1:5 |
| DESs-5 | ChCl | Glycol | 1:6 |
| DESs-6 | ChCl | Glycerol | 1:2 |
| DESs-7 | ChCl | Glycerol | 1:3 |
| DESs-8 | ChCl | Glycerol | 1:4 |
| DESs-9 | ChCl | Glycerol | 1:5 |
| DESs-10 | ChCl | Glycerol | 1:6 |
| DESs-11 | ChCl | 1,3-Butanediol | 1:2 |
| DESs-12 | ChCl | 1,3-Butanediol | 1:3 |
| DESs-13 | ChCl | 1,3-Butanediol | 1:4 |
| DESs-14 | ChCl | 1,3-Butanediol | 1:5 |
| DESs-15 | ChCl | 1,3-Butanediol | 1:6 |
| DESs-16 | ChCl | Lactic acid | 1:2 |
| DESs-17 | ChCl | Lactic acid | 1:3 |
| DESs-18 | ChCl | Lactic acid | 1:4 |
| DESs-19 | ChCl | Lactic acid | 1:5 |
| DESs-20 | ChCl | Lactic acid | 1:6 |
| DESs-21 | ChCl | Urea | 1:2 |
| DESs-22 | ChCl | Urea | 1:3 |
| DESs-23 | ChCl | Malic acid | 1:1 |
| DESs-24 | ChCl | Xylitol | 1:1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, P.; Li, Q.; Feng, Y.; Ma, S.; Chen, Y.; Li, G. Extraction and Analysis of Six Effective Components in Glycyrrhiza uralensis Fisch by Deep Eutectic Solvents (DES) Combined with Quantitative Analysis of Multi-Components by Single Marker (QAMS) Method. Molecules 2021, 26, 1310. https://doi.org/10.3390/molecules26051310
Yu P, Li Q, Feng Y, Ma S, Chen Y, Li G. Extraction and Analysis of Six Effective Components in Glycyrrhiza uralensis Fisch by Deep Eutectic Solvents (DES) Combined with Quantitative Analysis of Multi-Components by Single Marker (QAMS) Method. Molecules. 2021; 26(5):1310. https://doi.org/10.3390/molecules26051310
Chicago/Turabian StyleYu, Ping, Qian Li, Yanmei Feng, Sinan Ma, Yuying Chen, and Guichen Li. 2021. "Extraction and Analysis of Six Effective Components in Glycyrrhiza uralensis Fisch by Deep Eutectic Solvents (DES) Combined with Quantitative Analysis of Multi-Components by Single Marker (QAMS) Method" Molecules 26, no. 5: 1310. https://doi.org/10.3390/molecules26051310
APA StyleYu, P., Li, Q., Feng, Y., Ma, S., Chen, Y., & Li, G. (2021). Extraction and Analysis of Six Effective Components in Glycyrrhiza uralensis Fisch by Deep Eutectic Solvents (DES) Combined with Quantitative Analysis of Multi-Components by Single Marker (QAMS) Method. Molecules, 26(5), 1310. https://doi.org/10.3390/molecules26051310






