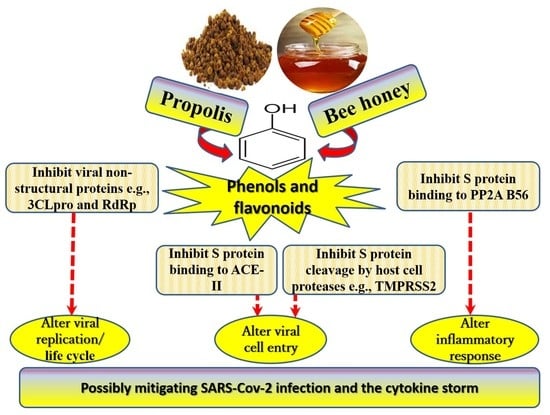
Propolis, Bee Honey, and Their Components Protect against Coronavirus Disease 2019 (COVID-19): A Review of In Silico, In Vitro, and Clinical Studies
Abstract
1. Introduction
2. SARS-CoV-2 and Associated Immune Response
3. Current Gaps in the Treatment of COVID-19
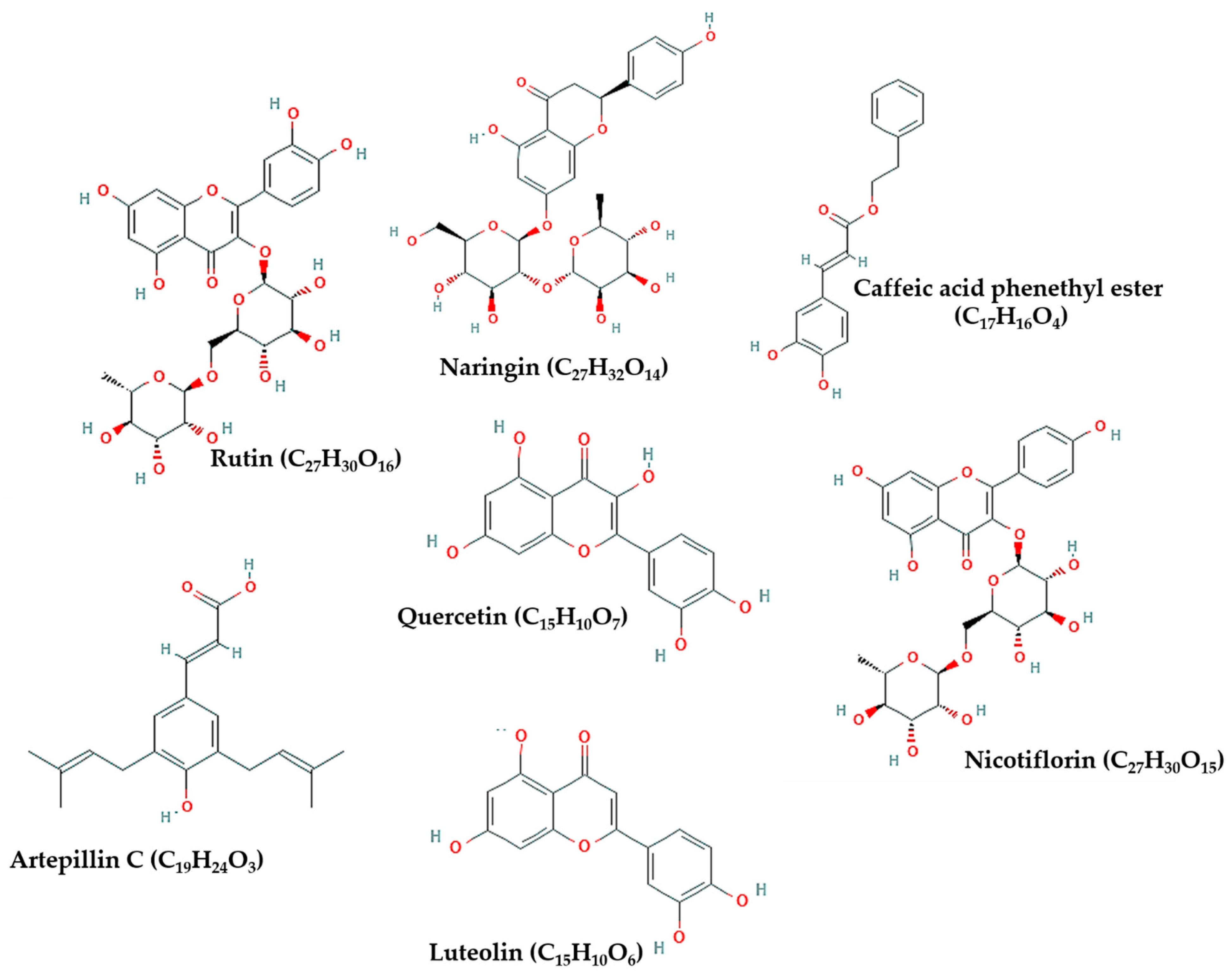
4. Propolis Composition and Biological Activities
5. Bee Honey Composition and Biological Activities
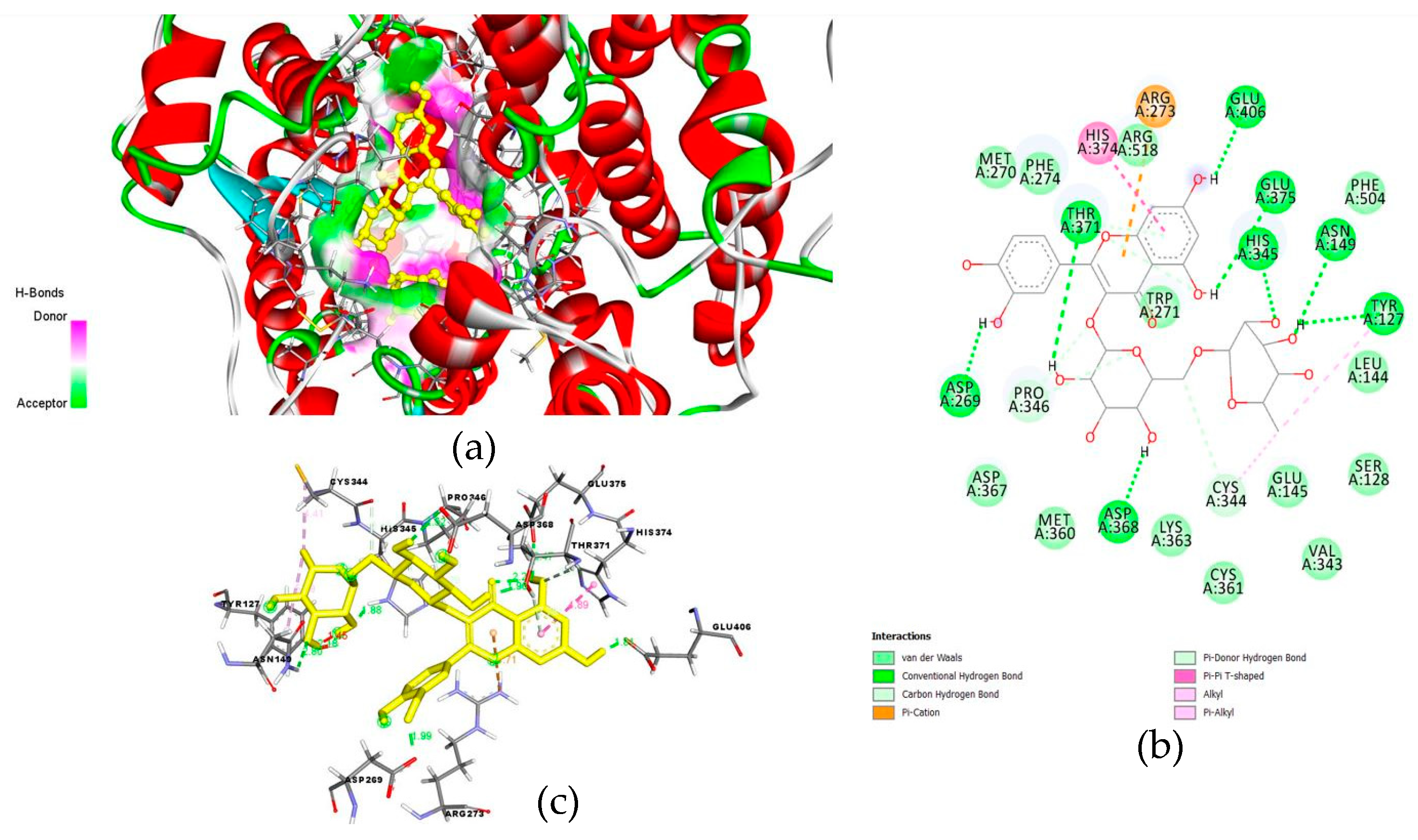
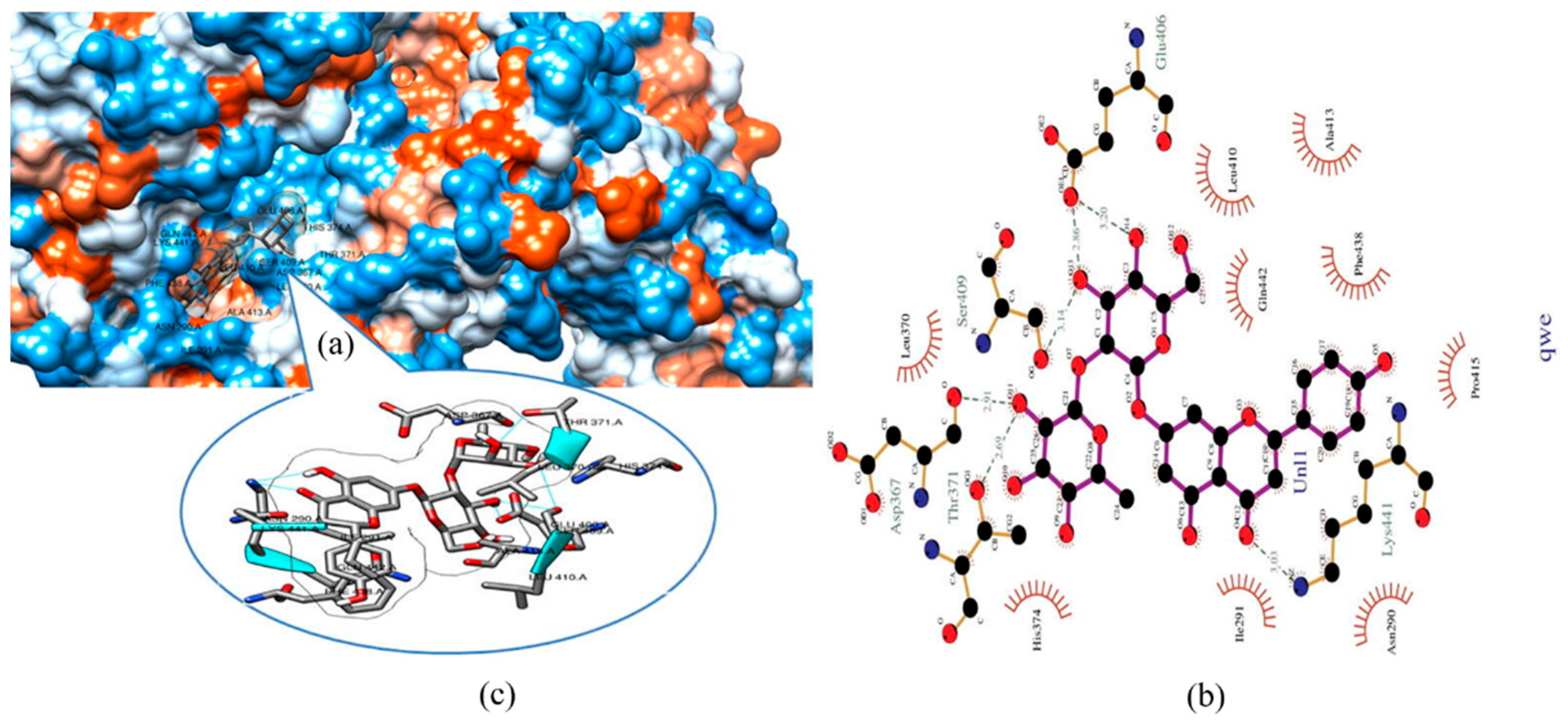
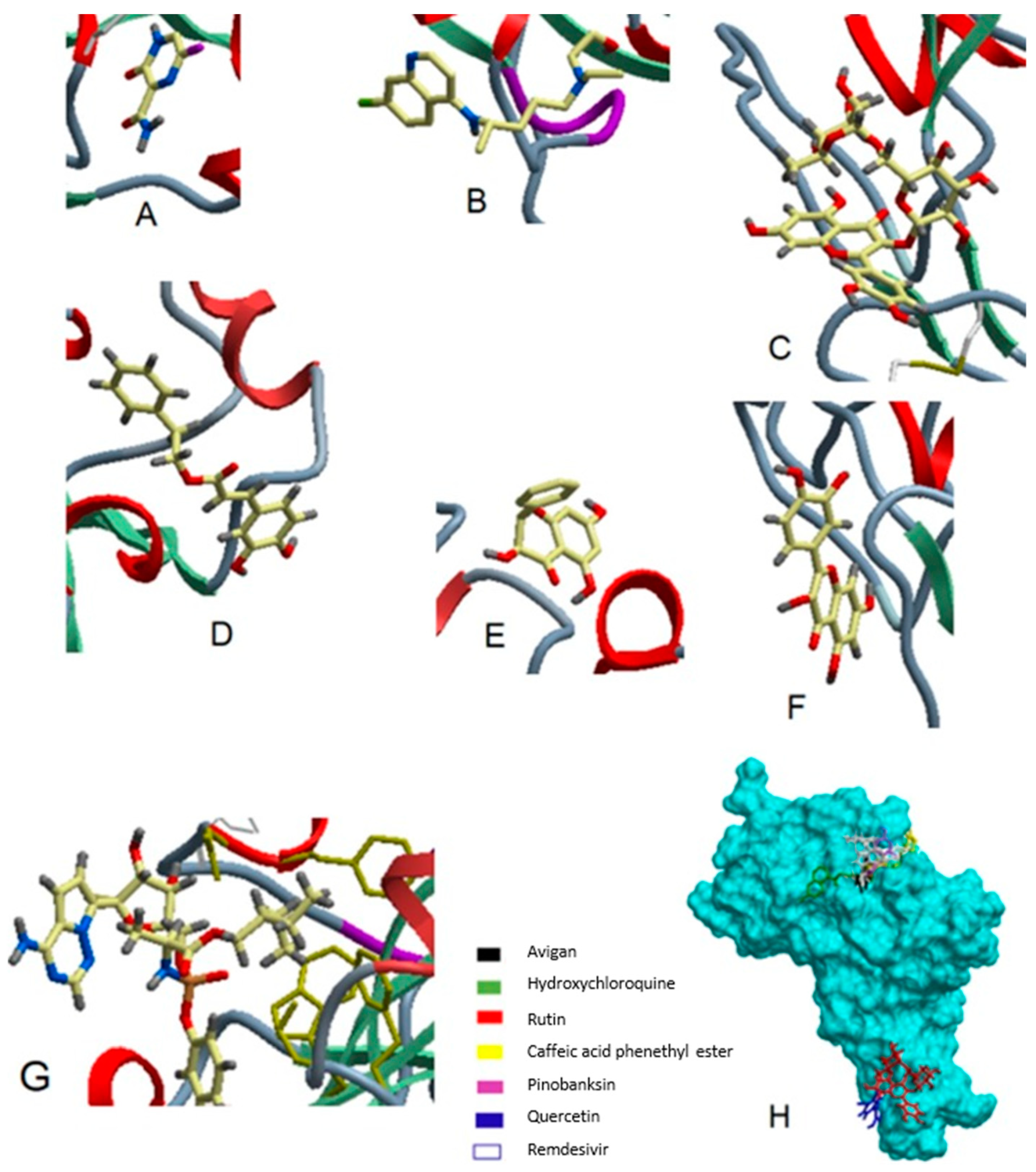
6. Possible Anti-COVID-19 Effects of Flavonoids in Propolis and Bee Honey Reported by Molecular Docking Studies
6.1. Flavonoids in Propolis May Inhibit the Proteolytic Processing of S Protein by Host Proteases
6.2. Flavonoids in Propolis May Inhibit the Binding of SARS-CoV-2 to Host Cell Receptors
6.3. Flavonoids in Propolis May Interrupt Viral-Host Protein Interactions That Induce an Inflammatory Response
6.4. Flavonoids in Propolis and Honey May Interrupt SARS-CoV-2 Life Cycle
7. Evidence from Experimental Studies Evaluating the Effect of Bee Products on SARS-CoV-2
8. Evidence from Human Trials Evaluating the Effect of Bee Products on Patients with COVID-19
9. Discussion
10. Conclusion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| 13b | Tert-butyl(1-((S)-1-(((S)-4-(benzylamino)-3:4-dioxo-1-((S)-2-oxopyrrolidin-3-yl)butan-2-yl)-amino)-3-cyclopropyl-1-oxopropan-2-yl)-2-oxo-1,2-dihydro-pyridin-3-yl) carbamate |
| COVID-19 | Coronavirus Disease 2019 |
| SARS-CoV-2 | Severe acute respiratory syndrome-coronavirus-2 |
| ARDS | Severe acute pneumonia-associated respiratory syndrome |
| M protein | Membrane protein |
| E protein | Envelope protein |
| S protein | Spike protein |
| N protein | Nucleocapsid protein |
| NSPs | Non-structural proteins |
| TM | Transmembrane |
| MW | Molecular weight |
| IL | Interleukin |
| RBD | Receptor-binding domain |
| ACE-II | Angiotensin-converting enzyme-related carboxypeptidase |
| TMPRSS2 | Transmembrane protease serine 2 |
| PLpro | Papain-like protease |
| RdRp | RNA-dependent RNA polymerase |
| 3CLpro | Chymotrypsin-like protease |
| Mpro | Main protease |
| SLiMs | Short linear motifs |
| PP2A | Protein phosphatase 2A |
| Tregs | Regulatory T cells |
| BGP | Brazilian green propolis |
| CAPE | Caffeic acid phenethyl ester |
| RCSBPDB | Research Collaboratory for Structural Bioinformatics Protein Data Bank |
| MLN-4760 | (S,S)-2-[1-Carboxy-2-[3-(3,5-Dichloro-Benzyl)-3h-Imidazole-4-Yl]-Ethylamino]xx-4-Methyl-Pentanoic Acid |
| DUB | Deubiquitinating |
| MOI | Multiplicity of infection |
| TPC2 | Two-pore channel 2 |
| HNS | Honey plus Nigella sativa |
| LOS | Length of hospital stay |
| RCTs | Randomized controlled clinical trials |
| HR | Hazard ratio |
| OR | Odds ratio |
References
- Goyal, B.; Goyal, D. Targeting the Dimerization of the Main Protease of Coronaviruses: A Potential Broad-Spectrum Therapeutic Strategy. ACS Comb. Sci. 2020, 22, 297–305. [Google Scholar] [CrossRef]
- Maaroufi, H. The Spike Protein S1 Subunit of SARS-CoV-2 Contains an LxxIxE-like Motif that is Known to Recruit the Host PP2A-B56 Phosphatase. bioRxiv 2020, 020941. [Google Scholar] [CrossRef]
- Zhou, Y.; Fu, B.; Zheng, X.; Wang, D.; Zhao, C.; Qi, Y.; Sun, R.; Tian, Z.; Xu, X.; Wei, H. Pathogenic T-cells and inflammatory monocytes incite inflammatory storms in severe COVID-19 patients. Natl. Sci. Rev. 2020, 7, 998–1002. [Google Scholar] [CrossRef]
- Ali, A.M.; Kunugi, H. Corona Virus Disease 2019 (COVID-19): A pandemic that threatens physical and mental health by promoting physical inactivity. Sports Med. Health Sci. 2020, 2, 221–223. [Google Scholar] [CrossRef]
- Kumar, V.; Dhanjal, J.K.; Bhargava, P.; Kaul, A.; Wang, J.; Zhang, H.; Kaul, S.C.; Wadhwa, R.; Sundar, D. Withanone and withaferin-A are predicted to interact with transmembrane protease serine 2 (TMPRSS2) and block entry of SARS-CoV-2 into cells. J. Biomol. Struct. Dyn. 2020, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.R.; Cao, Q.D.; Hong, Z.S.; Tan, Y.Y.; Chen, S.D.; Jin, H.J.; Tan, K.S.; Wang, D.Y.; Yan, Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak—An update on the status. Mil. Med. Res. 2020, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- WHO. WHO Coronavirus Disease (COVID-19) Dashboard. Available online: https://covid19.who.int/?gclid=Cj0KCQiA88X_BRDUARIsACVMYD_Ifm2pvYQL4ZMQ0bjHffc9RMSg74GLm2q8WWTV4B_XsfAeuiSsldUaAlfVEALw_wcB (accessed on 24 February 2021).
- Ruan, Q.; Yang, K.; Wang, W.; Jiang, L.; Song, J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020, 46, 846–848. [Google Scholar] [CrossRef] [PubMed]
- Tufan, A.; Güler, A.A.; Matucci-Cerinic, M. COVID-19, immune system response, hyperinflammation and repurposing antirheumatic drugs. Turk. J. Med. Sci 2020, 50, 620–632. [Google Scholar] [CrossRef] [PubMed]
- Zabetakis, I.; Lordan, R.; Norton, C.; Tsoupras, A. COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation. Nutrients 2020, 12, 1466. [Google Scholar] [CrossRef] [PubMed]
- De Biasi, S.; Meschiari, M.; Gibellini, L.; Bellinazzi, C.; Borella, R.; Fidanza, L.; Gozzi, L.; Iannone, A.; Lo Tartaro, D.; Mattioli, M.; et al. Marked T cell activation, senescence, exhaustion and skewing towards TH17 in patients with COVID-19 pneumonia. Nat. Commun. 2020, 11, 1–17. [Google Scholar] [CrossRef]
- Wen, W.; Su, W.; Tang, H.; Le, W.; Zhang, X.; Zheng, Y.; Liu, X.; Xie, L.; Li, J.; Ye, J.; et al. Immune cell profiling of COVID-19 patients in the recovery stage by single-cell sequencing. Cell Discov. 2020, 6, 1–18. [Google Scholar] [CrossRef]
- Li, M.; Zhang, Z.; Cao, W.; Liu, Y.; Du, B.; Chen, C.; Liu, Q.; Uddin, M.N.; Jiang, S.; Chen, C.; et al. Identifying novel factors associated with COVID-19 transmission and fatality using the machine learning approach. Sci. Total Environ. 2021, 764, 142810. [Google Scholar] [CrossRef] [PubMed]
- Louca, P.; Murray, B.; Klaser, K.; Graham, M.S.; Mazidi, M.; Leeming, E.R.; Thompson, E.; Bowyer, R.; Drew, D.A.; Nguyen, L.H.; et al. Dietary supplements during the COVID-19 pandemic: Insights from 1.4M users of the COVID Symptom Study app—A longitudinal app-based community survey. medRxiv 2020, 20239087. [Google Scholar] [CrossRef]
- Gasmi, A.; Tippairote, T.; Mujawdiya, P.K.; Peana, M.; Menzel, A.; Dadar, M.; Benahmed, A.G.; Bjørklund, G. Micronutrients as immunomodulatory tools for COVID-19 management. Clin. Immunol. 2020, 108545. [Google Scholar] [CrossRef]
- Boisard, S.; Shahali, Y.; Aumond, M.-C.; Derbré, S.; Blanchard, P.; Dadar, M.; Le Ray, A.-M.; Richomme, P. Anti-AGE activity of poplar-type propolis: Mechanism of action of main phenolic compounds. Int. J. Food. Sci. 2020, 55, 453–460. [Google Scholar] [CrossRef]
- Egawa, T.; Ohno, Y.; Yokoyama, S.; Yokokawa, T.; Tsuda, S.; Goto, K.; Hayashi, T. The Protective Effect of Brazilian Propolis against Glycation Stress in Mouse Skeletal Muscle. Foods 2019, 8, 439. [Google Scholar] [CrossRef]
- Shaha, A.; Mizuguchi, H.; Kitamura, Y.; Fujino, H.; Yabumoto, M.; Takeda, N.; Fukui, H. Effect of Royal Jelly and Brazilian Green Propolis on the Signaling for Histamine H1 Receptor and Interleukin-9 Gene Expressions Responsible for the Pathogenesis of the Allergic Rhinitis. Biol. Pharm. Bull. 2018, 41, 1440–1447. [Google Scholar] [CrossRef]
- Ali, A.M.; Kunugi, H. Bee honey protects astrocytes against oxidative stress: A preliminary in vitro investigation. Neuropsychopharmacol. Rep. 2019, 39, 312–314. [Google Scholar] [CrossRef]
- Ali, A.M.; Kunugi, H. Apitherapy for age-related skeletal muscle dysfunction (sarcopenia): A review on the effects of royal jelly, propolis, and bee pollen. Foods 2020, 9, 1362. [Google Scholar] [CrossRef]
- Ye, Q.; Wang, B.; Mao, J. The pathogenesis and treatment of the ‘Cytokine Storm’ in COVID-19. J. Infect. 2020, 80, 607–613. [Google Scholar] [CrossRef]
- Zang, R.; Gomez Castro, M.F.; McCune, B.T.; Zeng, Q.; Rothlauf, P.W.; Sonnek, N.M.; Liu, Z.; Brulois, K.F.; Wang, X.; Greenberg, H.B.; et al. TMPRSS2 and TMPRSS4 promote SARS-CoV-2 infection of human small intestinal enterocytes. Sci. Immunol. 2020, 5, eabc3582. [Google Scholar] [CrossRef] [PubMed]
- Rensi, S.; Altman, R.B.; Liu, T.; Lo, Y.C.; McInnes, G.; Derry, A.; Keys, A. Homology Modeling of TMPRSS2 Yields Candidate Drugs That May Inhibit Entry of SARS-CoV-2 into Human Cells. ChemRxiv 2020. [Google Scholar] [CrossRef]
- Jegouic, S.M.; Loureiro, S.; Thom, M.; Paliwal, D.; Jones, I.M. Recombinant SARS-CoV-2 spike proteins for sero-surveillance and epitope mapping. bioRxiv 2020, 109298. [Google Scholar] [CrossRef]
- da Silva, F.M.; da Silva, K.P.; de Oliveira, L.P.; Costa, E.V.; Koolen, H.H.; Pinheiro, M.L.; de Souza, A.Q.; de Souza, A.D. Flavonoid glycosides and their putative human metabolites as potential inhibitors of the SARS-CoV-2 main protease (Mpro) and RNA-dependent RNA polymerase (RdRp). Mem. Inst. Oswaldo Cruz 2020, 115, e200207. [Google Scholar] [CrossRef] [PubMed]
- Tsai, P.H.; Wang, M.L.; Yang, D.M.; Liang, K.H.; Chou, S.J.; Chiou, S.H.; Lin, T.H.; Wang, C.T.; Chang, T.J. Genomic variance of Open Reading Frames (ORFs) and Spike protein in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). J. Chin. Med. Assoc. 2020, 83, 725–732. [Google Scholar] [CrossRef]
- Toyoshima, Y.; Nemoto, K.; Matsumoto, S.; Nakamura, Y.; Kiyotani, K. SARS-CoV-2 genomic variations associated with mortality rate of COVID-19. J. Hum. Genet. 2020, 65, 1075–1082. [Google Scholar] [CrossRef]
- van Dorp, L.; Acman, M.; Richard, D.; Shaw, L.P.; Ford, C.E.; Ormond, L.; Owen, C.J.; Pang, J.; Tan, C.C.S.; Boshier, F.A.T.; et al. Emergence of genomic diversity and recurrent mutations in SARS-CoV-2. Infect. Genet. Evol. 2020, 83, 104351. [Google Scholar] [CrossRef]
- Yin, C. Genotyping coronavirus SARS-CoV-2: Methods and implications. Genomics 2020, 112, 3588–3596. [Google Scholar] [CrossRef]
- Bouhaddou, M.; Memon, D.; Meyer, B.; White, K.M.; Rezelj, V.V.; Marrero, M.C.; Polacco, B.J.; Melnyk, J.E.; Ulferts, S.; Kaake, R.M.; et al. The Global Phosphorylation Landscape of SARS-CoV-2 Infection. Cell 2020, 182, 685–712. [Google Scholar] [CrossRef]
- Kunugi, H.; Ali, A.M. Royal Jelly and Its Components Promote Healthy Aging and Longevity: From Animal Models to Humans. Int. J. Mol. Sci. 2019, 20, 4662. [Google Scholar] [CrossRef]
- Ali, A.M.; Kunugi, H. Apitherapy for Parkinson’s disease: A focus on the effects of propolis and royal jelly. Oxid. Med. Cell. Longev. 2020, 2020, 1727142. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Kunugi, H. Royal jelly as an intelligent anti-aging—A focus on cognitive aging and Alzheimer’s disease: A review. Antioxidants 2020, 9, 937. [Google Scholar] [CrossRef]
- Pitsillou, E.; Liang, J.; Ververis, K.; Hung, A.; Karagiannis, T.C. Interaction of small molecules with the SARS-CoV-2 papain-like protease: In silico studies and in vitro validation of protease activity inhibition using an enzymatic inhibition assay. J. Mol. Graph. Model. 2021, 107851. [Google Scholar] [CrossRef] [PubMed]
- Sharabi, A.; Kasper, I.R.; Tsokos, G.C. The serine/threonine protein phosphatase 2A controls autoimmunity. Clin. Immunol. 2018, 186, 38–42. [Google Scholar] [CrossRef]
- Ali, A.M.; Ahmed, A.H.; Smail, L. Psychological Climacteric Symptoms and Attitudes toward Menopause among Emirati Women. Int. J. Environ. Res. Public Health 2020, 17, 5028. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Kunugi, H. Intermittent fasting, dietary modifications, and exercise for the control of gestational diabetes and maternal mood dysregulation: A review and a case report. Int. J. Environ. Res. Public Health 2020, 17, 9379. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Wang, Y.; Zhang, Z.L.; Liu, Y.X.; Le, K.J.; Cui, M.; Yu, Y.T.; Gu, Z.C.; Gao, Y.; Lin, H.W. Efficacy and safety of current therapeutic options for COVID-19—Lessons to be learnt from SARS and MERS epidemic: A systematic review and meta-analysis. Pharmacol. Res. 2020, 157, 104872. [Google Scholar] [CrossRef]
- Ali, A.M.; Hendawy, A.O. Vitamin K. Involvement in COVID-19 and possible benefits of vitamin K antagonists (VKA). Aging Clin. Exp. Res. 2021. under review. [Google Scholar]
- Ali, A.M.; Kunugi, H. Approaches to nutritional screening in patients with Coronavirus Disease 2019 (COVID-19). Int. J. Environ. Res. Public Health 2021. under review. [Google Scholar]
- Ali, A.M.; Kunugi, H. Hypoproteinemia predicts disease severity and mortality in COVID-19: A call for action. Diagn. Pathol. 2021. under review. [Google Scholar]
- Sahlan, M.; Irdiani, R.; Flamandita, D.; Aditama, R.; Alfarraj, S.; Ansari, M.J.; Khayrani, A.C.; Pratami, D.K.; Lischer, K. Molecular interaction analysis of Sulawesi propolis compounds with SARS-CoV-2 main protease as preliminary study for COVID-19 drug discovery. J. King Saud Univ. Sci. 2021, 33, 101234. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Hendawy, A.O. Royal Jelly Acid, 10-Hydroxy-Trans-2-Decenoic Acid, for Psychiatric and Neurological Disorders: How helpful could it be?! Edelweiss J. Food Sci. Technol. 2019, 1, 1–4. [Google Scholar] [CrossRef]
- Ali, A.M.; Hendawy, A.O. Bee Honey as a Potentially Effective Treatment for Depression: A Review of Clinical and Preclinical Findings. JOJ Nurse Health Care 2018, 9, 555764. [Google Scholar] [CrossRef]
- Anjum, S.I.; Ullah, A.; Khan, K.A.; Attaullah, M.; Khan, H.; Ali, H.; Bashir, M.A.; Tahir, M.; Ansari, M.J.; Ghramh, H.A.; et al. Composition and functional properties of propolis (bee glue): A review. Saudi J. Biol. Sci. 2019, 26, 1695–1703. [Google Scholar] [CrossRef] [PubMed]
- Kocot, J.; Kielczykowska, M.; Luchowska-Kocot, D.; Kurzepa, J.; Musik, I. Antioxidant Potential of Propolis, Bee Pollen, and Royal Jelly: Possible Medical Application. Oxid. Med. Cell. Longev. 2018, 2018, 7074209. [Google Scholar] [CrossRef] [PubMed]
- Hashem, H. IN Silico Approach of Some Selected Honey Constituents as SARS-CoV-2 Main Protease (COVID-19) Inhibitors. EJMO 2020, 4, 96–200. [Google Scholar] [CrossRef]
- Güler, H.I.; Tatar, G.; Yildiz, O.; Belduz, A.O.; Kolayli, S. Investigation of potential inhibitor properties of ethanolic propolis extracts against ACE-II receptors for COVID-19 treatment by Molecular Docking Study. ScienceOpen 2020. [Google Scholar] [CrossRef]
- Wang, Q.; Sui, X.; Sui, D.J.; Yang, P. Flavonoid Extract from Propolis Inhibits Cardiac Fibrosis Triggered by Myocardial Infarction through Upregulation of SIRT1. Evid. Based Complement. Alternat. Med. 2018, 2018, 4957573. [Google Scholar] [CrossRef]
- Falcão, S.I.; Vale, N.; Gomes, P.; Domingues, M.R.M.; Freire, C.; Cardosoe, S.M.; Vilas-Boasa, M. Phenolic Profiling of Portuguese Propolis by LC–MS Spectrometry: Uncommon Propolis Rich in Flavonoid Glycosides. Phytochem. Anal. 2012. [Google Scholar] [CrossRef]
- Refaat, H.; Mady, F.M.; Sarhan, H.A.; Rateb, H.S.; Alaaeldin, E. Optimization and evaluation of propolis liposomes as a promising therapeutic approach for COVID-19. Int. J. Pharm. 2020, 592, 120028. [Google Scholar] [CrossRef]
- Bhargava, P.; Grover, A.; Nigam, N.; Kaul, A.; Doi, M.; Ishida, Y.; Kakuta, H.; Kaul, S.C.; Terao, K.; Wadhwa, R. Anticancer activity of the supercritical extract of Brazilian green propolis and its active component, artepillin C: Bioinformatics and experimental analyses of its mechanisms of action. Int. J. Oncol. 2018, 52, 925–932. [Google Scholar] [CrossRef]
- Veiga, R.S.; De Mendonça, S.; Mendes, P.B.; Paulino, N.; Mimica, M.J.; Lagareiro Netto, A.A.; Lira, I.S.; López, B.G.; Negrão, V.; Marcucci, M.C. Artepillin C and phenolic compounds responsible for antimicrobial and antioxidant activity of green propolis and Baccharis dracunculifolia DC. J. Appl. Microbiol. 2017, 122, 911–920. [Google Scholar] [CrossRef]
- Paulino, N.; Abreu, S.R.; Uto, Y.; Koyama, D.; Nagasawa, H.; Hori, H.; Dirsch, V.M.; Vollmar, A.M.; Scremin, A.; Bretz, W.A. Anti-inflammatory effects of a bioavailable compound, Artepillin C, in Brazilian propolis. Eur. J. Pharmacol. 2008, 587, 296–301. [Google Scholar] [CrossRef]
- Menniti-Ippolito, F.; Mazzanti, G.; Vitalone, A.; Firenzuoli, F.; Santuccio, C. Surveillance of Suspected Adverse Reactions to Natural Health Products The Case of Propolis. Drug Saf. 2008, 31, 419–423. [Google Scholar] [CrossRef]
- Silveira, M.A.D.; Teles, F.; Berretta, A.A.; Sanches, T.R.; Rodrigues, C.E.; Seguro, A.C.; Andrade, L. Effects of Brazilian green propolis on proteinuria and renal function in patients with chronic kidney disease: A randomized, double-blind, placebo-controlled trial. BMC Nephrol. 2019, 20, 1–12. [Google Scholar] [CrossRef]
- Ali, A.M.; Ali, E.M.; Ahmed, M.S.; Hendawy, A.O. Targeting gut microbiome and the recovery of muscle loss associated with cancer (cachexia): An overview of the possible effect of bee products. Med. Legal Update 2021, 21. in press. [Google Scholar]
- Kwon, M.J.; Shin, H.M.; Perumalsamy, H.; Wang, X.; Ahn, Y.-J. Antiviral effects and possible mechanisms of action of constituents from Brazilian propolis and related compounds. J. Apic. Res. 2020, 59, 413–425. [Google Scholar] [CrossRef]
- Maruta, H.; He, H. PAK1-blockers: Potential Therapeutics against COVID-19. Med. Drug Discov. 2020, 6, 100039. [Google Scholar] [CrossRef]
- Maruta, H.; Ahn, M.R. From bench (laboratory) to bed (hospital/home): How to explore effective natural and synthetic PAK1-blockers/longevity-promoters for cancer therapy. Eur. J. Med. Chem. 2017, 142, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Arruda, C.; Pena Ribeiro, V.; Oliveira Almeida, M.; Aldana Mejía, J.A.; Casoti, R.; Kenupp Bastos, J. Effect of light, oxygen and temperature on the stability of artepillin C and p-coumaric acid from Brazilian green propolis. J. Pharm. Biomed. Anal. 2020, 178, 112922. [Google Scholar] [CrossRef] [PubMed]
- Bulgasem, B.Y.; Lani, M.N.; Hassan, Z.; Yusoff, W.M.W.; Fnaish, S.G. Antifungal Activity of Lactic Acid Bacteria Strains Isolated from Natural Honey against Pathogenic Candida Species. Mycobiology 2016, 44, 302–309. [Google Scholar] [CrossRef]
- Hatchett, A.; Berry, C.; Oliva, C.; Wiley, D.; St Hilaire, J.; LaRochelle, A. A Comparison between Chocolate Milk and a Raw Milk Honey Solution’s Influence on Delayed Onset of Muscle Soreness. Sports 2016, 4, 18. [Google Scholar] [CrossRef]
- Rao, P.V.; Krishnan, K.T.; Salleh, N.; Gan, S.H. Biological and therapeutic effects of honey produced by honey bees and stingless bees: A comparative review. Rev. Bras. Farmacogn. 2016, 26, 657–664. [Google Scholar] [CrossRef]
- Eteraf-Oskouei, T.; Najafi, M. Traditional and Modern Uses of Natural Honey in Human Diseases: A Review. Iran. J. Basic Med. Sci. 2013, 16, 731–742. [Google Scholar]
- Semprini, A.; Singer, J.; Braithwaite, I.; Shortt, N.; Thayabaran, D.; McConnell, M.; Weatherall, M.; Beasley, R. Kanuka honey versus aciclovir for the topical treatment of herpes simplex labialis: A randomised controlled trial. BMJ Open 2019, 9, e026201. [Google Scholar] [CrossRef]
- Hashemipour, M.A.; Tavakolineghad, Z.; Arabzadeh, S.A.M.; Iranmanesh, Z.; Nassab, S.A.H.G. Antiviral Activities of Honey, Royal Jelly, and Acyclovir Against HSV-1. Wounds 2014, 26, 47–54. [Google Scholar]
- Tantawy, M.A. Efficacy of Natural Honey Treatment in Patients with Novel Coronavirus. 2020. Available online: https://clinicaltrials.gov/ct2/show/NCT04323345 (accessed on 16 January 2021).
- Tang, J.S.; Compton, B.J.; Marshall, A.; Anderson, R.; Li, Y.; van der Woude, H.; Hermans, I.F.; Painter, G.F.; Gasser, O. Mānuka honey-derived methylglyoxal enhances microbial sensing by mucosal-associated invariant T cells. Food Funct. 2020, 11, 5782–5787. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Hendawy, A.O. Bee honey in combination with exercise—better physical performance and less fatigue for a more active lifestyle during the COVID-19 era. Sports Med. Health Sci. 2021. under review. [Google Scholar]
- Waheed, M.; Hussain, M.B.; Javed, A.; Mushtaq, Z.; Hassan, S.; Shariati, M.A.; Khan, M.U.; Majeed, M.; Nigam, M.; Mishra, A.P.; et al. Honey and cancer: A mechanistic review. Clin. Nutr. 2019, 38, 2499–2503. [Google Scholar] [CrossRef] [PubMed]
- Khan, R.U.; Naz, S.; Abudabos, A.M. Towards a better understanding of the therapeutic applications and corresponding mechanisms of action of honey. Environ. Sci. Pollut. Res. Int. 2017, 24, 27755–27766. [Google Scholar] [CrossRef]
- Ahmad, R.S.; Hussain, M.B.; Saeed, F.; Waheed, M.; Tufail, T. Phytochemistry, metabolism, and ethnomedical scenario of honey: A concurrent review. Int. J. Food Prop. 2017, 20, S254–S269. [Google Scholar] [CrossRef]
- Ali, A.M.; Hendawy, A.O. So, Antidepressant Drugs have Serious Adverse Effects, but what are the Alternatives? Nov. Appro Drug Des. Dev. 2018, 4, 555636. [Google Scholar] [CrossRef]
- Vasquez, A.; Forsgren, E.; Fries, I.; Paxton, R.J.; Flaberg, E.; Szekely, L.; Olofsson, T.C. Symbionts as major modulators of insect health: Lactic acid bacteria and honeybees. PLoS ONE 2012, 7, e33188. [Google Scholar] [CrossRef]
- Brudzynski, K.; Flick, R. Accumulation of soluble menaquinones MK-7 in honey coincides with death of Bacillus spp. present in honey. Food Chem. X 2019, 1, 100008. [Google Scholar] [CrossRef]
- Sundaram, K.S.; Fan, J.H.; Engelke, J.A.; Foley, A.L.; Suttie, J.W.; Lev, M. Vitamin K status influences brain sulfatide metabolism in young mice and rats. J. Nutr. 1996, 126, 2746–2751. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.S.; Sushma, P.; Dharmashekar, C.; Beelagi, M.S.; Prasad, S.K.; Shivamallu, C.; Prasad, A.; Syed, A.; Marraiki, N.; Prasad, K.S. In silico evaluation of flavonoids as effective antiviral agents on the spike glycoprotein of SARS-CoV-2. Saudi J. Biol. Sci. 2021, 28, 1040–1051. [Google Scholar] [CrossRef] [PubMed]
- Vijayakumar, B.G.; Ramesh, D.; Joji, A.; Jayachandra prakasan, J.; Kannan, T. In silico pharmacokinetic and molecular docking studies of natural flavonoids and synthetic indole chalcones against essential proteins of SARS-CoV-2. Eur. J. Pharmacol. 2020, 886, 173448. [Google Scholar] [CrossRef] [PubMed]
- Pitsillou, E.; Liang, J.; Ververis, K.; Lim, K.W.; Hung, A.; Karagiannis, T.C. Identification of Small Molecule Inhibitors of the Deubiquitinating Activity of the SARS-CoV-2 Papain-Like Protease: In silico Molecular Docking Studies and in vitro Enzymatic Activity Assay. Front. Chem. 2020, 8, 1171. [Google Scholar] [CrossRef] [PubMed]
- Arora, S.; Lohiya, G.; Moharir, K.; Shah, S.; Yende, S. Identification of Potential Flavonoid Inhibitors of the SARS-CoV-2 Main Protease 6YNQ: A Molecular Docking Study. DCM 2020, 3, 239–248. [Google Scholar] [CrossRef]
- Walls, A.C.; Park, Y.J.; Tortorici, M.A.; Wall, A.; McGuire, A.T.; Veesler, D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell 2020, 181, 281–292.e286. [Google Scholar] [CrossRef]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Zhang, Y.; Wu, L.; Niu, S.; Song, C.; Zhang, Z.; Lu, G.; Qiao, C.; Hu, Y.; Yuen, K.-Y.; et al. Structural and Functional Basis of SARS-CoV-2 Entry by Using Human ACE2. Cell 2020, 181, 894–904.e899. [Google Scholar] [CrossRef] [PubMed]
- Barros, E.P.; Casalino, L.; Gaieb, Z.; Dommer, A.C.; Wang, Y.; Fallon, L.; Raguette, L.; Belfon, K.; Simmerling, C.; Amaro, R.E. The Flexibility of ACE2 in the Context of SARS-CoV-2 Infection. Biophys. J. 2020. [Google Scholar] [CrossRef]
- Shawan, M.M.A.K.; Halder, S.K.; Hasan, M.A. Luteolin and abyssinone II as potential inhibitors of SARS-CoV-2: An in silico molecular modeling approach in battling the COVID-19 outbreak. Bull. Natl. Res. Cent. 2021, 45, 1–21. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Zhao, W.; Gong, Y.; Gao, X. Molecules inhibit the enzyme activity of 3-chymotrypsin-like cysteine protease of SARS-CoV-2 virus: The experimental and theory studies. bioRxiv 2020, 120642. [Google Scholar] [CrossRef]
- Shivanika, C.; Kumar, D.; Ragunathan, V.; Tiwari, P.; Sumitha, A. Molecular docking, validation, dynamics simulations, and pharmacokinetic prediction of natural compounds against the SARS-CoV-2 main-protease. J. Biomol. Struct. Dyn. 2020, 1–27. [Google Scholar] [CrossRef]
- Clementi, N.; Scagnolari, C.; D’Amore, A.; Palombi, F.; Criscuolo, E.; Frasca, F.; Pierangeli, A.; Mancini, N.; Antonelli, G.; Clementi, M.; et al. Naringenin is a powerful inhibitor of SARS-CoV-2 infection in vitro. Pharmacol. Res. 2021, 163, 105255. [Google Scholar] [CrossRef]
- Sakurai, Y.; Kolokoltsov, A.A.; Chen, C.-C.; Tidwell, M.W.; Bauta, W.E.; Klugbauer, N.; Grimm, C.; Wahl-Schott, C.; Biel, M.; Davey, R.A. Two-pore channels control Ebola virus host cell entry and are drug targets for disease treatment. Science 2015, 347, 995–998. [Google Scholar] [CrossRef]
- Kim, D.E.; Min, J.S.; Jang, M.S.; Lee, J.Y.; Shin, Y.S.; Song, J.H.; Kim, H.R.; Kim, S.; Jin, Y.-H.; Kwon, S. Natural Bis-Benzylisoquinoline Alkaloids-Tetrandrine, Fangchinoline, and Cepharanthine, Inhibit Human Coronavirus OC43 Infection of MRC-5 Human Lung Cells. Biomol. Ther. 2019, 9, 696. [Google Scholar] [CrossRef] [PubMed]
- El Sayed, S.M.; Aboonq, M.S.; El Rashedy, A.G.; Aljehani, Y.T.; Abou El-Magd, R.M.; Okashah, A.M.; El-Anzi, M.E.; Alharbi, M.B.; El-Tahlawi, R.; Nabo, M.M.H.; et al. Promising preventive and therapeutic effects of TaibUVID nutritional supplements for COVID-19 pandemic: Towards better public prophylaxis and treatment (A retrospective study). Am. J. Blood Res. 2020, 10, 266–282. [Google Scholar]
- Ulasli, M.; Gurses, S.A.; Bayraktar, R.; Yumrutas, O.; Oztuzcu, S.; Igci, M.; Igci, Y.Z.; Cakmak, E.A.; Arslan, A. The effects of Nigella sativa (Ns), Anthemis hyalina (Ah) and Citrus sinensis (Cs) extracts on the replication of coronavirus and the expression of TRP genes family. Mol. Biol. Rep. 2014, 41, 1703–1711. [Google Scholar] [CrossRef] [PubMed]
- Fiorini, A.C.; Scorza, C.A.; de Almeida, A.-C.G.; Fonseca, M.C.M.; Finsterer, J.; Fonseca, F.L.A.; Scorza, F.A. Antiviral activity of Brazilian Green Propolis extract against SARS-CoV-2 (Severe Acute Respiratory Syndrome—Coronavirus 2) infection: Case report and review. Clinics 2021, 76, e2357. [Google Scholar] [CrossRef] [PubMed]
- Duarte Silveira, M.A.; De Jong, D.; dos Santos Galvão, E.B.; Ribeiro, J.C.; Silva, T.C.; Berretta, A.A.; Amorim, T.C.; San Martin, R.L.A.; da Rebelo Conceição, L.F.M.; Dantas Gomes, M.M.; et al. Efficacy of propolis as an adjunct treatment for hospitalized COVID-19 patients: A randomized, controlled clinical trial. medRxiv 2021, 20248932. [Google Scholar] [CrossRef]
- Ashraf, S.; Ashraf, S.; Ashraf, M.; Imran, M.A.; Kalsoom, L.; Siddiqui, U.N.; Farooq, I.; Habib, Z.; Ashraf, S.; Ghufran, M.; et al. Honey and Nigella sativa against COVID-19 in Pakistan (HNS-COVID-PK): A multi-center placebo-controlled randomized clinical trial. medRxiv 2020, 20217364. [Google Scholar] [CrossRef]
- Miryan, M.; Soleimani, D.; Dehghani, L.; Sohrabi, K.; Khorvash, F.; Bagherniya, M.; Sayedi, S.M.; Askari, G. The effect of propolis supplementation on clinical symptoms in patients with coronavirus (COVID-19): A structured summary of a study protocol for a randomised controlled trial. Trials 2020, 21, 1–2. [Google Scholar] [CrossRef]
- Perola, E.; Walters, W.P.; Charifson, P.S. A detailed comparison of current docking and scoring methods on systems of pharmaceutical relevance. Proteins Struct. Funct. Bioinf. 2004, 56, 235–249. [Google Scholar] [CrossRef] [PubMed]
- Nader, C.P.; Cidem, A.; Verrills, N.M.; Ammit, A.J. Protein phosphatase 2A (PP2A): A key phosphatase in the progression of chronic obstructive pulmonary disease (COPD) to lung cancer. Respir. Res. 2019, 20, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.R.; Ohlmeyer, M. Protein phosphatase 2A as a therapeutic target in inflammation and neurodegeneration. Pharmacol. Ther. 2019, 201, 181–201. [Google Scholar] [CrossRef]
- Aliboni, A.; D’Andrea, A.; Massanisso, P. Treatment of propolis specimens from Central Italy to yield a product with a lower charge of allergenic species. Sep. Purif. Technol. 2011, 82, 71–75. [Google Scholar] [CrossRef]
- Helbling, A.; Peter, C.; Berchtold, E.; Bogdanov, S.; Müller, U. Allergy to honey: Relation to pollen and honey bee allergy. Allergy 1992, 47, 41–49. [Google Scholar] [CrossRef] [PubMed]
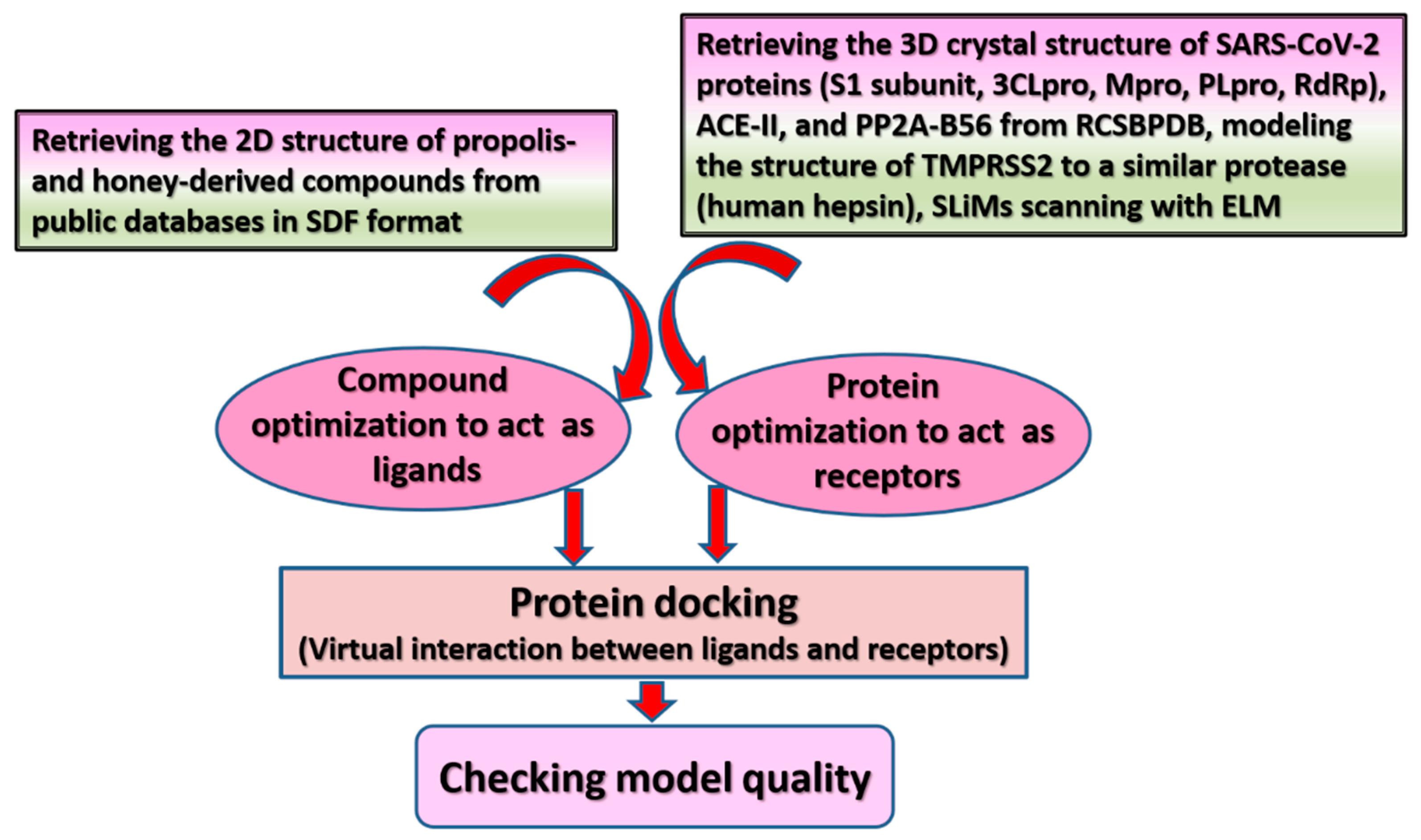

| Bee-Related Compounds | SARS-CoV-2 Proteins | Possible Anti-COVID-19 Effects | References |
|---|---|---|---|
| CAPE | TMPRSS2 | Inhibiting S protein cleavage | [5] |
| Rutin, luteolin, and CAPE | ACE-II | Inhibiting viral binding to host cell receptor | [48,51,79] |
| Naringin, rutin, and quercetin | S protein | Inhibiting viral fusion in host cell membrane | [51,78] |
| Rutin, nicotiflorin, luteolin, and CAPE | 3CLpro/Mpro, PLpro, and RdRp | Inhibiting viral replication and inflammatory reaction | [25,34,47,51,80,81] |
| Artepillin C | PP2A-B56 | Inhibiting viral-host interactions that induce inflammation | [2] |
| Bee Products | Sample and Design | Treatment | Study Outcomes | Results | References |
|---|---|---|---|---|---|
| Natural honey plus Nigella sativa, Anthemis hyaline (TaibUVID). | Confirmed COVID-19 patients (n = 20), contacts of COVID-19 patients (n = 20). Retrospective study. | TaibUVID orally and herbal solution inhalation. | Symptom recovery, blood count profile, and development of SARS-CoV-2 infection. | Improvement of the lymphocyte profile and earlier symptom recovery in regular users of TaibUVID. Lower incidence of SARS-CoV-2 infection in contacts. | [92] |
| BGP (EPP-AF). | Confirmed COVID-19 patient aged 52 years (n = 1). Case report. | EPP-AF® 45 drops/3 times/day/2 weeks. | Early symptom recovery and viral clearance within 12 days of treatment. | Patient’s condition improved considerably. Negative nasopharyngeal swab (PCR). | [94] |
| BGP (EPP-AF). | Hospitalized adult COVID-19 patients (n = 82). An open-label, single center RCT. | EG1: propolis 400 mg (n = 40). EG1: propolis 800 mg (n = 42). EG1: standard care alone (n = 42). | LOS, dependence on oxygen therapy, development of acute kidney injury, ICU admission, use of vasoactive drugs. | Decreased LOS in EG1 and EG2 to 7 and 6 days compared with 12 days in CG. No effect of propolis on oxygen dependency. Decreased renal injury in EGs compared with CG (2 vs. 10 patients). No adverse effects of propolis were depicted. | [95] |
| Natural honey plus Nigella sativa (HNS). | Adults (n = 313) with moderate (n = 210) and severe (n = 103) COVID-19. A multicenter, placebo-controlled RCT. | EG: HNS (n = 107 moderate + 50 severe patients). CG: empty capsules placebo (n = 103 moderate + 53 severe patients). | Symptom recovery, viral clearance, a 30-day mortality, resumption of normal activity, oxygen saturation, and percentage of discharge from the hospital. | A 59% reduction in the time of symptom recovery in EG. Earlier virus clearance in EG. Decreased mortality by 4-folds in EG compared with CG. Higher resumption of normal activity on day 6 in moderate patients in EG than in CG. Achieving a mean oxygen saturation above 90% six days earlier in severe patients in EG. Higher hospital discharge of severe patients in EG. No adverse effects of HNS were depicted. | [96] |
| Iranian green propolis extract | Confirmed COVID-19 patients aged 18–75 years (n = 80). Double-blind, placebo-controlled, RCT | EG: propolis (n = 40). CG: placebo (n = 40). | COVID-19 severity and duration over 2 weeks. | NR | [97] |
| Natural honey | Non-severe COVID-19 patients aged 5–75 years (n = 1000). Single blind multicenter RCT. | EG: honey (n = 500). CG: standard care (n = 500). | Symptom recovery and viral clearance at day 14, lung recovery at day 30, mortality and viral clearance within 30 days. | NR | [68] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ali, A.M.; Kunugi, H. Propolis, Bee Honey, and Their Components Protect against Coronavirus Disease 2019 (COVID-19): A Review of In Silico, In Vitro, and Clinical Studies. Molecules 2021, 26, 1232. https://doi.org/10.3390/molecules26051232
Ali AM, Kunugi H. Propolis, Bee Honey, and Their Components Protect against Coronavirus Disease 2019 (COVID-19): A Review of In Silico, In Vitro, and Clinical Studies. Molecules. 2021; 26(5):1232. https://doi.org/10.3390/molecules26051232
Chicago/Turabian StyleAli, Amira Mohammed, and Hiroshi Kunugi. 2021. "Propolis, Bee Honey, and Their Components Protect against Coronavirus Disease 2019 (COVID-19): A Review of In Silico, In Vitro, and Clinical Studies" Molecules 26, no. 5: 1232. https://doi.org/10.3390/molecules26051232
APA StyleAli, A. M., & Kunugi, H. (2021). Propolis, Bee Honey, and Their Components Protect against Coronavirus Disease 2019 (COVID-19): A Review of In Silico, In Vitro, and Clinical Studies. Molecules, 26(5), 1232. https://doi.org/10.3390/molecules26051232